Abstract
Objectives
To report our early experience using endografts with inner branches for the treatment of complex abdominal aortic aneurysms and thoracoabdominal aortic aneurysms (TAAAs).
Methods
A retrospective analysis of all patients treated in our institution for complex abdominal aortic aneurysms and TAAAs with custom-made stent grafts consisting of one or more inner branches. Data collected included patients demographics, aortic aneurysm morphology, stent grafts features, perioperative morbidity and mortality and short-term reintervention and mortality rates.
Results
Twenty-seven patients (18 males, mean age 70 ± 7.1) were included. Indications for surgery included TAAAs (12, 41%) juxtarenal abdominal aortic aneurysms (10, 37%), type 1A endoleaks (4, 15%) and paraanastamotic aneurysms (1, 4%). A total of 90 inner branches were used. Twenty-one (78%) of the stent grafts consisted only of inner branches and six (22%) had a combination of inner branches with either fenestrations or outer branches. Technical success was achieved in 26/27 (96%) of the patients. There was one perioperative mortality. Six patients suffered from major perioperative adverse events. Mean follow-up was seven months (range 1–23). During the follow-up period, four patients (15%) required reinterventions. Branch-related reinterventions were performed in two (7%) patients. No occlusions of inner branches occurred during the follow-up.
Conclusions
Inner branches in branched endovascular aneurysm repairs offer a feasible option for the treatment of complex abdominal aortic aneurysms and TAAAs. The procedures can be completed with high technical success and with acceptable short-term branch-related reintervention rates. Further follow-up is required to determine the long-term durability of this technology.
Keywords: Inner branches, BEVAR, FEVAR, aneurysm, complex aneurysm, thoracoabdominal aortic aneurysm
Introduction
During the last decade, there has been increasing use of endovascular techniques for the treatment of complex abdominal aortic aneurysms (AAA) and thoracoabdominal aortic aneurysms (TAAA).1–4 Fenestrated endovascular aneurysm repair (FEVAR) and branched endovascular aneurysm repair (BEVAR) with custom-made devices are both considered acceptable configurations in these situations. Fenestrated stent grafts are typically used when the visceral vessels originate from a non-aneurysmal segment of the aorta, whereas outer branches (OBs) are commonly used when the visceral arteries originate from aneurysmal segments. Recently, a third option of inner branches (IBs) has been introduced. The IB configuration has been previously described in stent grafts designed to treat aortic arch pathologies.5,6 They have rarely been described for the endovascular treatment of complex AAAs and TAAAs. 7 Advantages of IBs include increased anatomical suitability in aortas too narrow for OBs, while providing an overlap between the aortic module and the side branch. The aim of this study is to present our early, single-centre experience with the use of IBs for the treatment of complex AAAs and TAAAs. In this study, we assessed the feasibility, technical success and short-term follow-up of patients treated with endograft-containing IBs.
Methods
This study was approved by the local institutional review board.
We performed a retrospective review of a prospectively maintained data base of all patients who underwent elective FEVAR and BEVAR at our institution between the years 2008 and 2020. Patients with endografts that had one or more IBs were included in this study. Data collected included patients demographics, aortic aneurysm morphology, stent grafts characteristics, perioperative morbidity and mortality and short-term reintervention and mortality rates.
Outcome endpoints
Technical success, as advised in the SVS reporting standards for EVAR, 8 was defined as the successful deployment of the endoluminal graft with secure proximal and distal fixation, successful catheterization and bridging of all IBs and their respective target vessels with a patent stent graft, and the absence of either type I or III endoleaks at the completion of the procedure.
Secondary endpoints included perioperative morbidity and mortality and reintervention rates.
Preoperative evaluation
All patients underwent a preoperative computerized tomography angiogram (CTA) with 1.5 mm cuts of the chest, abdomen and pelvis. All patients were considered unfit to undergo open surgical aortic repair due to their advanced age or severe comorbidities (American Society of Anesthesiologists (ASA) III/IV).
Candidates suitable for treatment with BEVAR or FEVAR were patients who required elective repair of complex AAAs or TAAAs. All patients were treated for degenerative aortic aneurysms. Patients excluded were those with symptomatic aneurysms or those who required emergent repair. Others excluded were patients with post-dissection aneurysms.
Stent grafts
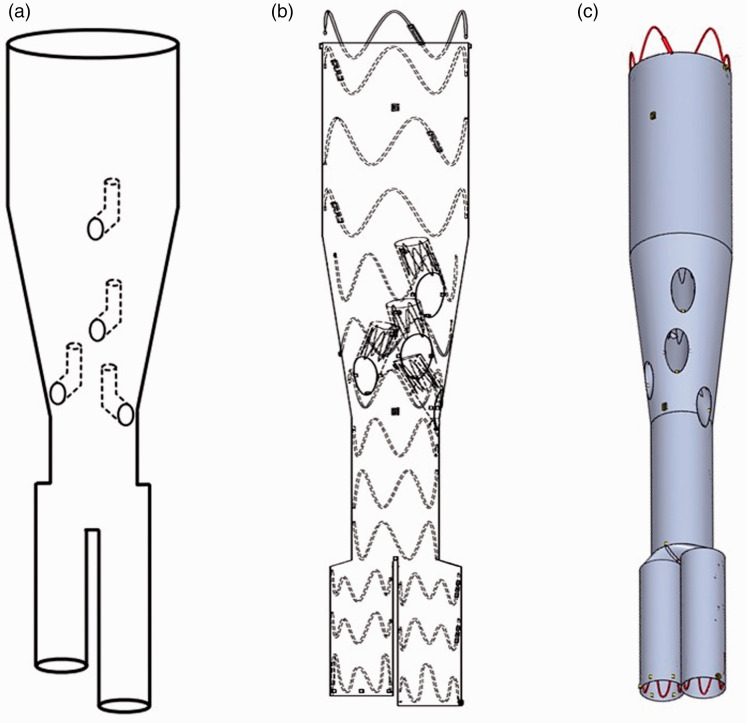
Endografts used were all E-xtra Design Engineering stent-grafts (Jotec GmbH, Hechingen, Germany). This device is a custom-made endograft loaded on a 24 French (F) delivery system. The endografts are tapered in the mid-portion to provide a small gap between the exit site of the IB and the origin of the visceral artery (Figure 1). This gap between the aortic module and the aorta allows for imaging of the visceral vessels and for manipulation of the catheter outside the stent graft.
Figure 1.
Illustration (a), schematic drawing (b), and isometric view (c) of a typical stent graft with four inner branches used in this study. Permission to reproduce illustrations was obtained from manufacturer.
Anatomical inclusion criteria for the E-xtra Design are:
Access vessels >8 mm (due to the 24F delivery system)
Aortic diameter >18 mm
Visceral vessels diameter >4 mm. There are no limitations regarding the angulation of the visceral vessels.
Decision on fenestrations versus IBs or OBs
OBs were chosen when the aortic diameter at the level of the visceral segment was wider than 28 mm. Fenestrations or IBs were used for smaller diameter aortas (≥18 and <28 mm). Fenestrations were typically used in smaller diameter aortas, where the device was anticipated to have circumferential contact with the aortic wall at the level of the fenestrations. IBs were chosen for slightly larger aortas, in which the tapered segment of the endograft would create a gap between the aortic wall and the tapered segment of the stent graft, thus providing a gap that aided in the cannulation of the visceral vessel.
Branch structure and configuration
IBs were preferentially designed in an antegrade configuration (‘upward pointing’) to allow access from the left arm. This configuration is suitable for the caudal direction typically observed in the celiac artery (CA), the superior mesenteric artery (SMA) and renal arteries. In the uncommon anatomical scenario of an upward-pointing visceral artery, retrograde-pointing IBs were designed. Retrograde IBs were also designed in situations where there was limited free-lumen space available for the stent to accommodate multiple IBs at the same level or in patients with severely tortuous aortas where manipulation of long catheters and sheaths and cannulation of target arteries from the arm would be difficult.
IBs were typically 17–19 mm in length. Diameters were 8–9 mm for the CA and SMA and 6 mm for the renal arteries. An attempt was made by the manufacturer not to design a device that had more than two IBs at the same level, as this would increase the profile of the delivery system. No pre-loaded catheters were used.
Perfusion branches
An additional perfusion branch was planned in selected cases of type 1 and 2 TAAAs, in which a spinal drain was contraindicated or a staged procedure was not possible.
Spinal cord protection
A spinal drain was placed preoperatively in nine patients. All were considered high risk for developing spinal cord ischemia (TAAA type 1 or 2, coverage of over 20 cm of the thoracic aorta and previous aortic surgery). Those who were at high risk for spinal cord ischemia were maintained post-operatively with a high mean arterial pressure of 90–100 mmHg. Spinal pressure was maintained post-operatively at 10 cmH2O, and spinal drains were removed after 48hours.
Procedure
All procedures were performed under general anaesthesia in a dedicated hybrid operating room. Bilateral common femoral arteries (CFAs) were exposed with surgical cut downs. The left brachial artery was exposed in the proximal upper arm through a short longitudinal incision. After admission of intravenous heparin, a 12F Gore dry seal sheath (W.L. Gore and Associates, Flagstaff, AZ) was placed through a CFA into the aorta, and through it, a marker guide wire was placed into one of the renal arteries. In cases of TAAAs, thoracic stent grafts were initially deployed in the thoracic aorta, extending to approximately 10–20 mm cephalad to the CA. The custom-made aortic module was then delivered through the contralateral CFA and positioned with the distal opening of a renal branch placed approximately 10 mm cephalad to the origin of the marked renal artery. The aortic module was then fully deployed. The delivery system was removed, and the entry site of the CFA was closed around the stiff wire using purse string sutures, thus restoring flow to the leg. A 10F, 45-cm long Cook sheath (Bloomington, IN) was placed through the left brachial artery and advanced into the descending aorta and into the main aortic module. An 8F, 70 cm long Cook straight sheath was advanced coaxially through the 10 F sheath. A 5F, 100 cm long Bern catheter (Merit Medical, South Jordan, UT) was used to sequentially cannulate each IB and corresponding artery. A balloon expandable stent graft (Advanta V12 (Maquet-Atrium Medical Inc., Hudson, NH) or VBX stent graft (W.L. Gore & Associates, Flagstaff, AZ)) was then positioned with maximum overlap within the IB and at least 2 cm within the visceral vessel and deployed. The same sequence was performed in all the other IBs. Following completion of the IBs, additional modules (bifurcated devices and limbs) were placed, followed by a completion angiogram. The femoral and brachial arteries were closed primarily.
Post-operative care
Patients were monitored in an intensive care unit for 48 hours, after which they were transferred to a regular hospital bed. All patients were placed on clopidogrel 75 mg and aspirin 100 mg once daily. In patients with spinal drains, clopidogrel was initiated only after the removal of the drain. In those without CSF drains, clopidogrel was started on post-operative day 1.
Lower extremity Doppler exams were performed routinely preoperatively and on post-operative day 3. These included ankle/brachial indexes (ABIs) and segmental pressures.
Follow-up
All patients were seen in our outpatient clinic, and all underwent a CTA of their chest and abdomen at 1, 6 and 12 months post-operatively and then every 12 months thereafter (Figures 2 and 3). In the rare occasion of a patient with a juxtarenal AAA (JRAAA) who suffered from chronic renal failure, we occasionally performed an abdominal duplex of the sac and branches together with a non-contrast CT scan to evaluate the sac size.
Figure 2.
Volume rendering image from postoperative CT scan demonstrating endograft with four inner branches.
Figure 3.
Sagittal maximum intensity projection image from postoperative CT scan. Arrow points at inner branch of the celiac artery stent.
Reinterventions
Patients underwent reinterventions if, on follow-up CTA, they were found to have a type I or III endoleak or a type II endoleak associated with sac enlargement compared to the previous exam.
Results
During the study period, we performed a total of 75 BEVARs and FEVARs. Of these, 27 (36%) were endografts containing one or more IBs, all in the last three years. Mean age was 70 ± 7.1 (range 59–84) and 18 (70%) were males. Patient’s demographics and morphologic details of the aneurysms are presented in Table 1. Aneurysms treated included TAAAs (12, 41%), JRAAAs (10, 37%), type 1A endoleaks (4, 15%) and a paraanastamotic aneurysm (1, 4%) (Table 2).
Table 1.
Patients’ baseline characteristics.
| N = 27 (%) | |
|---|---|
| Age (years) | 70 ± 7.1 |
| Male | 18 (70) |
| Coronary artery disease | 16 (59) |
| Congestive heart failure | 6 (22) |
| Hypertension | 19 (70) |
| Diabetes mellitus | 5 (19) |
| Hyperlipidemia | 20 (74) |
| COPD | 6 (22) |
| Smoker (past or present) | 23 (85) |
| Chronic kidney disease | 6 (22) |
| Previous aortic surgery | 6 (22) |
| ASA score | |
| ASA3 | 21 (78) |
| ASA4 | 6 (22) |
COPD: chronic obstructive pulmonary disease; ASA: American Society of Anesthesiology.
Table 2.
Aortic morphology.
| TAAA | 12 (41%) |
| Type 1 | 3 |
| Type 2 | 1 |
| Type 3 | 2 |
| Type 4 | 5 |
| Type 5 | 1 |
| JRAAA | 10 (37%) |
| Type 1A endoleak | 4 (15%) |
| Paraanastamotic aneurysm | 1 (4%) |
| Diameter, mean, mm (range) | 61 (45–88) |
TAAA: thoracoabdominal aortic aneurysm; JRAAA: juxtarenal abdominal aortic aneurysm.
Six patients had prior aortic surgery. Four had previous EVARs and developed type 1A endoleaks, one had a previous open AAA repair and developed a para-anastamotic aneurysm and one had previous Chimney EVAR and had a large gutter endoleak.
Details of the endografts and side branches used are reported in Table 3. Endografts with IBs only were used in 21 patients (78%). Five endografts had a combination of OBs and IBs. One patient required a combination of two IBs with two fenestrations. A total of 90 IBs branches were used, 86 (95%) of them with an antegrade configuration. Four patients required an additional inner perfusion branch intended to perfuse the spinal cord that was occluded one month after the index procedure.
Table 3.
Device data.
| Aortic stent graft configuration | N = 27 (%) |
| Inner branches only | 21 (78) |
| Inner + outer branches | 5 (18) |
| Inner branches + fenestrations | 1 (4) |
| Inner branch configuration | N = 90 (%) |
| Antegrade | 86 (95) |
| Retrograde | 4 (5) |
| Bridging stent grafts | |
| Advanta V12 | 62 (69) |
| VBX | 28 (31) |
Operative data is detailed in Table 4. Technical success was achieved in 26 (96%) patients. In one patient, the aortic module was successfully deployed, but due to the angulation of the aortic arch, we were unsuccessful in advancing a catheter and sheath from the left subclavian artery into the descending aorta, and the procedure was aborted. Catheterization and bridging of the IBs and their respective target vessel with patent stent grafts were achieved in all procedures (100%). No immediate type I endoleak was seen. An expected filling of the aneurysm sac was seen in all four patients who had a perfusion branch. One patient had a significant type II endoleak.
Table 4.
Operative data.
| Technical success | 26 (96%) |
| Procedure time (incision to complete closure) (minutes) | 244 (range 156–382) |
| Contrast, mean (ml) | 310 (range 220–380) |
| Length of stay (days) | 4 (range 3–10 days) |
| Target vessels revascularized | N = 90 (%) |
| Celiac artery | 20 (22) |
| Superior mesenteric artery | 24 (27) |
| Right renal artery | 22 (24) |
| Left renal artery | 24 (27) |
| Diameters of target vessels (mm) | |
| Celiac artery (mean, range) | 8.17 (6.5–10) |
| Superior mesenteric artery (mean, range) | 7.62 (5.5–10) |
| Renal arteries (mean, range) | 5.6 (4–7.5) |
Procedural-related complications and perioperative morbidity and mortality are reported in Table 5. Intraoperative complications included one perforation of the CA that was treated with an additional covered stent. Two patients required iliac conduits after we were unable to pass the device from the femoral artery.
Table 5.
Procedural and perioperative complications.
| Intraoperative complications | |
| Perforation of visceral vessel | 1 |
| Inability to pass delivery system | 2 |
| Postoperative complications | |
| Mortality | 1 |
| Kidney injury (bleeding, artery occlusion) | 2 |
| Acute cholecystitis | 1 |
| Acute leg ischemia | 1 |
| Stenosis of femoral artery | 1 |
| Spinal cord ischemia | 1 |
Despite the small profile of the brachial artery, it accommodated the 10 F sheath, and the artery underwent primary repair in all cases without the need of an interposition graft.
There was one perioperative mortality. This patient was a 61-year-old male with severe chronic obstructive pulmonary disease and chronic kidney disease who underwent an uneventful BEVAR with four IBs. During the post-operative period, his respiratory status gradually deteriorated to the point where he required reintubation, followed by worsening of his hemodynamic status and kidney function. He expired on post-operative day 5 from multi-organ failure. Five perioperative adverse events occurred. One developed bleeding from the kidney parenchyma, most probably due to a wire injury and was treated successfully with coiling. One suffered from occlusion of a renal artery that was diagnosed on post-operative day 3 and resulted in loss of a kidney. There was one case of spinal cord ischemia that partially improved following administration of IV pressors and a spinal drain.
Post-operative Doppler examination
Preoperative femoral and ankle brachial indexes were normal in all patients. In 26/27 (96%) patients, these indexes remained unchanged on the post-operative Doppler study. In one patient, there was a unilateral decrease in ABI on the side from which the aortic module was delivered.
Follow-up
Follow-up was available for all patients. Mean follow-up was seven months (range 1–23 months). Sac size decreased or remained stable in all patients. During this period, four patients (15%) required reinterventions. One developed a type IA endoleak that was treated with an aortic extension. One patient underwent embolization of a type II endoleak originating from a lumbar artery that was initially thought to be a type III endoleak, and one developed an endoleak from the distal SMA stent that was treated with an extension. One patient underwent a suboptimal occlusion of a perfusion branch and required a secondary procedure to completely occlude the branch. All reinterventions were performed more than six months after the index procedure.
During the follow-up, no patient developed occlusion of a side branch. No patient required reintervention due to stenosis or detachment of the bridging stent from the IB. All patients were alive at the time of the study. No patient required explanation of the endograft.
Discussion
FEVAR and BEVAR have evolved over the past decade into an acceptable treatment option for complex AAAs and TAAAs.9–12 Contemporary techniques for bridging between the main aortic module to the target vessels consist of side branches placed through fenestrations or directional OBs. Fenestrations can usually be accessed through the femoral arteries thus avoiding the need for axillary or brachial artery access. They can be placed in non-aneurysmal segments of the aorta, making them particularly attractive for the management of JRAAAs. They are required, however, to be positioned directly across the origin of the target vessel, and a failure to do so can result in the inability to cannulate the target vessel. In addition, the seal between the side branch and the aortic module is based on creation of an hourglass configuration. This attachment can be prone to failure due to stent fracture or migration, resulting in endoleaks or stenosis and occlusion of the branch. 13 Directional OBs offer the advantage of providing an overlap between the aortic and the bridging module, thus allowing for a certain degree of freedom in movement without creating an endoleak due to separation of the components. They are typically manufactured pointing caudally, which is useful when the target vessel is downward pointing, anatomy frequently encountered in the CA and the SMA. Their main disadvantage is that they can only be used in larger aortic diameters such as TAAAs. Due to this restriction, their applicability in narrower aortas such as JRAAAs and suprarenal AAAs is limited.
The IBs are cylindrical components with a wide external opening sewn onto the inner wall of a custom-made stent graft. They have several advantages. As the side branches are fixed to the aortic module by overlapping with the IB, there is no need for flaring. The overlap between the IB and side branch provides a secure seal, similar to that of OBs. In addition, they can be used in relatively narrow aortas, making them applicable for the treatment of JRAAAs, type IA endoleaks and post-dissection TAAA, where the narrow true lumen often precludes the use of directional side branches.
The configuration of IBs is typically ‘upward pointing’. As such, once the IBs are cannulated, the introduction of a wire and catheter into the visceral vessels can be achieved with relative ease, as these vessels are commonly in a caudal orientation.
The data for IBs in JRAAAs and TAAAs is scarce in the literature. The largest series to date reporting on IBs for complex aortic aneurysm comes from Katsargaris and Verhoevens paper. 7 Stent grafts with IBs in combination with fenestrations seemed to be a better configuration than stent grafts with IBs alone. In their paper, IBs were selected for target vessels regarded unsuitable for either fenestrations or directional side branches. They claimed that catheterization of IBs without an indwelling wire proved tedious and time-consuming. The reason for this is the inherent difficulty in visualizing and orientating the IB perpendicularly to facilitate catheterization, in contrast to fenestrations or standard branches. Based on our experience, we found that with familiarity of the marker locations on the IB, together with positioning the image intensifier beam fully perpendicular to the branch and its markers, cannulation can be achieved with relative ease, even without pre-loaded catheters. They also preferred fenestrations as it allows keeping the main graft diameter wide resulting in closer contact with the aortic wall at the level of the fenestrations. This is an obvious advantage of fenestrations; however, we have learned that having IBs in a tapered segment does not compromise the proximal seal. Finally, cannulating a target vessel with a steep take off can be extremely challenging through a downward-pointing IB. This however can be resolved with designing an aortic graft with a upward-pointing IB. Based on the favourable data presented in this manuscript, it is our impression that IBs can be used not only when fenestrations and external branches are not possible but as a primary choice of device.
The mean procedure time and contrast load in our series were 244 min and 310 cc, respectively. The mean operative time of the first five cases was prolonged (mean 290 min), mostly due to our unfamiliarity with the device. With the increasing experience that we obtained with these devices, we significantly shortened the length of the procedures which currently stands at a mean of 180 min. In a similar fashion, we have decreased the contrast load to 260 cc per case. These numbers are similar to non-IB cases performed by our group. We anticipate that both figures will continue to decrease over time with our growing use of fusion technology and experience with these devices.
Reinterventions for FEVAR and BEVAR are not uncommon, and high reintervention rates remain a concern. 3 , 12 , 14 , 15 It is difficult to determine differences in patency and reintervention rates of fenestrations compared to OBs, as results of these two configurations are frequently reported together without a distinction made between the two.15–17 In one recent report, directional branches were more prone to primary endoleaks in comparison to fenestrations; however, branch endoleaks resolved more often spontaneously compared with fenestration endoleaks. 18 In a multi-centre study with an off-the-shelf fenestrated endograft used for pararenal AAAs, 34% underwent secondary interventions at 25 months’ follow-up. 2 Martin-Gonzalez et al. 19 recently published a report comparing the outcome of renal branches used in branched and fenestrated devices. Renal re-intervention rates were similar in both groups (4.7% vs. 5.2%); however, the renal occlusion rate was significantly higher when branches were used compared to fenestrations (9.6% vs. 2.3%; p < 0.01). This was not our experience in the present study. In this study, we report our experience with 90 IBs, of which 46 (51%) were for renal arteries. Despite the use of IBs, we did not encounter occlusions of renal branches during the follow-up.
Experience with IBs in EVAR is derived mostly from the treatment of aortic arch pathologies. 5 , 6 In a recent study by Tsilimparis et al., 6 the authors report their experience treating 54 patients with aortic arch diseases with a custom-made inner-branched endograft with two IBs (Cook Medical, Bloomington, IN). Technical success was achieved in 53 cases (98%), and only three early stent graft-related reinterventions were necessary. In one of the largest studies to date reporting experience with IBs in complex AAAs and TAAAs, 32 patients were treated. Technical success was achieved in all patients. During the follow-up, four renal IBs occluded in three patients. 7
In our study, four patients (15%) required reinterventions. Although follow-up is short, it is important to emphasize that only two of the reinterventions were branch-related. These were required due to an endoleak that originated from the distal landing zone of a branch placed in the SMA and a persistent endoleak from a perfusion branch after an unsuccessful attempt to occlude it. No endoleak originated from the overlap site of the IB with the bridging stent graft, suggesting an additional benefit of IBs.
All patients in this cohort were treated for degenerative aortic aneurysms; however, there may be a role for IBs in the management of post-dissection aneurysms, as the narrow profile of the tapered segment and IBs may prove to be suitable for this pathology.
Conclusions
These results show that IBs in BEVARs offer a feasible option for treatment of complex aortic aneurysms and TAAAs. The procedures can be completed with high technical success and with low short-term branch-related reintervention rates. The current design of these stents offers adequate sealing of the aneurysms; however, longer follow-up is required to assess the durability of this repair.
Supplemental Material
Supplemental material, sj-pdf-1-vas-10.1177_1708538120977279 for Short-term outcomes of inner branches for endovascular repair of complex abdominal and thoracoabdominal aortic aneurysms by Daniel Silverberg, Avner Bar-Dayan, Haitam Hater, Boris Khaitovich and Moshe Halak in Vascular
Footnotes
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD
Haitam Hater https://orcid.org/0000-0002-6631-1173
References
- 1.Farber MA, Oderich GS, Timaran C, et al. ; Zenith p-Branch Feasibility Study Investigators. Results from a prospective multicenter feasibility study of Zenith p-Branch stent graft. J Vasc Surg 2019; 70: 1409–1418. [DOI] [PubMed] [Google Scholar]
- 2.Farber MA, Eagleton MJ, Mastracci TM, et al. Results from multiple prospective single-center clinical trials of the off-the-shelf p-Branch fenestrated stent graft. J Vasc Surg 2017; 66: 982–990. [DOI] [PubMed] [Google Scholar]
- 3.Verhoeven EL, Katsargyris A, Bekkema F, et al. Ten-year experience with endovascular repair of thoracoabdominal aortic aneurysms: results from 166 consecutive patients. Eur J Vasc Endovasc Surg 2015; 49: 524–531. [DOI] [PubMed] [Google Scholar]
- 4.Gargiulo M, Gallitto E, Pini R, et al. Fenestrated endografting is the preferred option for juxta-renal aortic aneurysm reconstruction. J Cardiovasc Surg (Torino) 2020; 61: 2–9. [DOI] [PubMed] [Google Scholar]
- 5.Spear R, Haulon S, Ohki T, et al. Subsequent results for arch aneurysm repair with inner branched endografts. Eur J Vasc Endovasc Surg 2016; 51: 380–385. [DOI] [PubMed] [Google Scholar]
- 6.Tsilimparis N, Detter C, Law Y, et al. Single-center experience with an inner branched arch endograft. J Vasc Surg 2019; 69: 977–985. [DOI] [PubMed] [Google Scholar]
- 7.Katsargyris A, Marques de Marino P, Mufty H, et al. Early experience with the use of inner branches in endovascular repair of complex abdominal and thoraco-abdominal aortic aneurysms. Eur J Vasc Endovasc Surg 2018; 55: 640–646. [DOI] [PubMed] [Google Scholar]
- 8.Chaikof EL, Blankensteijn JD, Harris PL, et al. Ad hoc committee for standardized reporting practices in vascular surgery of The Society for Vascular Surgery/American Association for Vascular Surgery. Reporting standards for endovascular aortic aneurysm repair. J Vasc Surg 2002; 35: 1048–1060. [DOI] [PubMed] [Google Scholar]
- 9.Verhoeven EL, Zeebregts CJ, Kapma MR, et al. Fenestrated and branched endovascular techniques for thoracoabdominal aneurysm repair. J Cardiovasc Surg (Torino ) 2005; 46: 131–140. [PubMed] [Google Scholar]
- 10.Zenith Fenestrated Study Investigators. Results of the United States multicenter prospective study evaluating the Zenith fenestrated endovascular graft for treatment of juxtarenal abdominal aortic aneurysms. J Vasc Surg 2014; 60: 1420–1428. [DOI] [PubMed] [Google Scholar]
- 11.Schanzer A, Simons JP, Flahive J, et al. Outcomes of fenestrated and branched endovascular repair of complex abdominal and thoracoabdominal aortic aneurysms. J Vasc Surg 2017; 66: 687–694. [DOI] [PubMed] [Google Scholar]
- 12.Oderich GS, Ribeiro M, Hofer J, et al. Prospective, nonrandomized study to evaluate endovascular repair of pararenal and thoracoabdominal aortic aneurysms using fenestrated-branched endografts based on supraceliac sealing zones. J Vasc Surg ▪▪▪▪; 65: 1249–1259. [DOI] [PubMed] [Google Scholar]
- 13.Silverberg D, Aburamileh A, Rimon U, et al. Secondary interventions after fenestrated and branched endovascular repair of complex aortic aneurysms. J Vasc Surg 2020; 72: 866–872. Sep [DOI] [PubMed] [Google Scholar]
- 14.Timaran DE, Knowles M, Ali T, et al. Fenestrated endovascular aneurysm repair among octogenarians at high and standard risk for open repair. J Vasc Surg 2017; 66: 354–359. [DOI] [PubMed] [Google Scholar]
- 15.Lindström D, Kettunen H, Engström J, et al. Outcome after fenestrated and branched repair of aortic aneurysms-device failures predict reintervention rates. Ann Vasc Surg 2020; 66: 142–151. [DOI] [PubMed] [Google Scholar]
- 16.Eagleton MJ, Follansbee M, Wolski K, et al. Fenestrated and branched endovascular aneurysm repair outcomes for type II and III thoracoabdominal aortic aneurysms. J Vasc Surg 2016; 63: 930–942. [DOI] [PubMed] [Google Scholar]
- 17.Simons JP, Podder S, Dossabhoy SS, et al. Fenestrated/branched endovascular aortic aneurysm repair using a supraceliac aortic proximal seal zone versus an infraceliac aortic proximal seal zone. Ann Vasc Surg 2020; 62: 213–222. [DOI] [PubMed] [Google Scholar]
- 18.Kärkkäinen JM, Tenorio ER, Jain A, et al. Outcomes of target vessel endoleaks after fenestrated-branched endovascular aortic repair. J Vasc Surg 2020; 72: 445-455. [DOI] [PubMed] [Google Scholar]
- 19.Martin-Gonzalez T, Mastracci T, Carrell T, et al. Mid-term outcomes of renal branches versus renal fenestrations for thoraco-abdominal aneurysm repair. Eur J Vasc Endovasc Surg 2016; 52: 141–148. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-pdf-1-vas-10.1177_1708538120977279 for Short-term outcomes of inner branches for endovascular repair of complex abdominal and thoracoabdominal aortic aneurysms by Daniel Silverberg, Avner Bar-Dayan, Haitam Hater, Boris Khaitovich and Moshe Halak in Vascular