Keywords: Cockle health, coinfection, confounding factors, Haplosporidia, pathogen interactions, Vibrio
Despite coinfections being recognized as the rule in animal populations, most studies focus on single pathogen systems. Pathogen interaction networks and the drivers of such associations are lacking in disease ecology studies. Common cockle Cerastoderma edule populations are exposed to a great diversity of pathogens, thus making them a good model system to investigate. This study examined the diversity and prevalence of pathogens from different taxonomic levels in wild and fished C. edule on the Irish coast. Potential interactions were tested focussing on abiotic (seawater temperature and salinity) and biotic (cockle size and age, and epiflora on shells) factors. No Microsporidia nor OsHV-1μVar were detected. Single infections with Haplosporidia (37.7%) or Vibrio (25.3%) were more common than two-pathogen coinfected individuals (9.5%), which may more easily succumb to infection. Fished C. edule populations with high cockle densities were more exposed to infections. Higher temperature and presence of epiflora on cockle shells promoted coinfection in warmer months. Low seawater salinity, host condition and proximity to other infected host species influenced coinfection distribution. A positive association between two Minchinia spp. was observed, most likely due to their different pathogenic effect. Findings highlight the major influence that ecological factors have on pathogen interactions and host–pathogen interplay.
Introduction
Animal hosts are exposed to complex pathogen assemblages that ultimately form a dynamic community within them (Johnson and Buller, 2011). However, research into host–pathogen interactions remains dominated by the study of one host–one pathogen systems despite the fact that hosts can simultaneously carry several infectious agents, with consequences for the dynamics of each agent and host health. Coinfections have been previously reported in shellfish hosts: including Vibrio tapetis, Perkinsus sp. and digenetic trematodes in cockles (Lassalle et al., 2007); ostreid herpesvirus-1 (OsHV-1) and Vibrio spp. in oysters (Petton et al., 2015; Alfaro et al., 2019); and protozoans and bacteria in clams (Arzul et al., 2012; Carella et al., 2020); and in some of the cases with a dramatic effect on disease susceptibility (Cox, 2001; Lassalle et al., 2007; Arzul et al., 2012; Alfaro et al., 2019). Processes at different scales of ecological organization from the within-host level (e.g. interactions of coinfecting parasites) to the ecosystem level (e.g. the influence of environmental variables) across space and over time have a key influence in complex natural systems (Hellard et al., 2015). Therefore, moving from one host–one pathogen systems towards an ecosystem view of host–pathogen interactions and their ecology is essential.
Detection of interspecific parasite/pathogen interactions in natural populations is not easy, due to complex networks of indirect effects making it difficult to infer underlying processes (Hellard et al., 2012). Interactions among coinfecting infectious agents can, in fact, alter host pathology, parasite transmission and virulence evolution, also influencing the spread of infections at a population level resulting in disease outbreaks, as described previously in bivalves (Lassalle et al., 2007; Arzul et al., 2012; Alfaro et al., 2019). These effects can be the opposite, i.e. coinfections within the host may reduce infection success yet still enhance pathology or, in contrast, co-occurring parasites/pathogens may weaken host pathology increasing the infection success (Johnson and Hoverman, 2012). For example, mortality induced by one parasite can also eliminate the availability of hosts for other parasites (Jolles et al., 2008); likewise, morbidity induced by one parasite can increase exposure to a second, even if within-host interactions are antagonistic (Karvonen et al., 2009).
Simultaneous infections may therefore trigger a whole spectrum of outcomes within the hosts, both synergistic and antagonistic, i.e. the weight of one or both infectious agents may be suppressed (Fukami et al., 2010), one or both agents may be amplified resulting in coexistence (Thomas et al., 1998), or one may be amplified and the other suppressed (Johnson and Hoverman, 2012). Parasite/pathogen interactions can be not only direct, such as competition for attachment sites, competition for host resources and predation upon one another (intra-guild predation) (Hatcher et al., 2006, 2012; Johnson et al., 2010), but interactions can also be indirect or host-mediated often involving changes in immunity such as cross-immunity (apparent competition) and immune suppression (apparent facilitation) (Jolles et al., 2008). For instance, Johnson and Hoverman (2012) suggested cross-reactive immunity as a cause of a decrease in parasite persistence in their experiments with a group of related larval trematodes in Pacific chorus frogs (Pseudacris regilla). Johansen and Sommer (2001) suggested that the immune suppression triggered by a primary infection could be the cause of multiple bacterial infections in Atlantic salmon (Salmo salar).
The co-occurrence of infectious agents may also result simply because the same risk factors promote their presence and not because the different pathogen groups are interacting synergistically (Vaumourin et al., 2015). Risk factors shared by two or more infectious agents can occur at the level of the host individual (e.g. host condition and age), population (e.g. host density) or landscape (e.g. climatic factors), increasing the probability of infection by both/all agents (Hellard et al., 2015). Such confounding factors may influence host exposure and/or host susceptibility promoting simultaneous infections (Vaumourin et al., 2015). Environmental parameters such as seawater temperature and salinity, host density or host physiological conditions and behaviour are common confounding factors that may influence the co-occurrence of different pathogen groups and the epidemiological and geographic patterns of infection and disease (Hellard et al., 2012; Vaumourin et al., 2015). Confounding factors must be considered in field studies since those factors can create statistical associations between infectious agents even if there is no true biological interaction between them (Kuris and Lafferty, 1994).
Generalist host species, such as the common cockle Cerastoderma edule, with wide spatial distributions, are exposed to a large range of environmental conditions, and a greater diversity of parasites and pathogens, and consequently are more likely to be coinfected (Vaumourin et al., 2015; Mahony et al., 2020; de Montaudouin et al., 2021), directly affecting individual cockle health and cockle population dynamics (Longshaw and Malham, 2013). Cockles may be host to multiple macro-parasite infections, particularly trematodes (de Montaudouin et al., 2010, 2021), but may also be infected by a range of pathogens (Rowley et al., 2014; de Montaudouin et al., 2021), providing a microcosm for the parasite/pathogen community. Within this community, there are haplosporidian protists such as the genus Minchinia (Engelsma et al., 2011; Longshaw and Malham, 2013; Ramilo et al., 2018; Lynch et al., 2020b; Albuixech-Martí et al., 2020; de Montaudouin et al., 2021), numerous members of the bacterial family Vibrio (Carballal et al., 2001; Lassalle et al., 2007; de Montaudouin et al., 2021), viruses such as ostreid herpesvirus-1 microVar (OsHV-1 μVar) (Carballal et al., 2003; Evans et al., 2017; Bookelaar et al., 2020; de Montaudouin et al., 2021) and microsporidian species reported in digeneans and paramyxids infecting cockle populations (Goater, 1993; Fermer et al., 2009, 2011; Villalba et al., 2014; de Montaudouin et al., 2021). Cockles are a keystone species responsible for different ecosystem services, i.e. carbon storage, energy cycling, food source for seabirds, etc., being also good sentinel and bioindicator species (Malham et al., 2012; Carss et al., 2020). Moreover, the common cockle is one of the main non-cultured bivalve species harvested in western European waters, with a high economic value (Rowley et al., 2014; Carss et al., 2020). Consequently, cockles have been well-studied and are useful model species. Given the complex parasite/pathogen assemblages within the cockles and their intricate interactions and effects, a comprehensive and integrated approach that considers not only the synergistic impact of coinfections but also the role of ecological factors driving the infection across space and over time is needed.
Future climate scenarios predict a warming marine environment and increased precipitation events resulting in pulse events of reduced/fluctuating salinity in nearshore ecosystems (Allam and Raftos, 2015; Coen and Bishop, 2015). Consequently, climate change may be expected to have a significant effect on disease epidemiology (Coen and Bishop, 2015), and may even have significant consequences on zoonoses emergence (Hoarau et al., 2020). In this context, the need for the understanding of ecological processes involved in the transmission and dynamics of infectious agents in host reservoirs is critical (Hoarau et al., 2020). This study, therefore, aims to assess the interactions between infectious agents, their hosts and the environment.
For that purpose, this study examined the diversity and prevalence of significant pathogen groups, Haplosporidia, Vibrio spp., OsHV-1 μVar and Microsporidia, associated with bivalve mortalities globally as well as coinfections and associations of the different infectious agents at an individual and population level in wild and fished C. edule along the Irish and Celtic Seas. The role of abiotic (seawater temperature and salinity) and biotic (cockle size and age, and epiflora on their shells) factors in the risk of coinfection throughout the study was also assessed. Developing knowledge on pathogen diversity and co-occurrence in an economically and ecologically important species as is C. edule and integrating the role of the environment in the host–pathogen interplay is essential for a better understanding of the interaction networks that can affect disease outcomes and transmissibility in the current changing environment. Findings from this study highlight the complexity of pathogen interactions within a host and the effect of key biotic and environmental drivers shaping disease dynamics, which can also be applied to other coinfected animal host species.
Materials and methods
Sampling
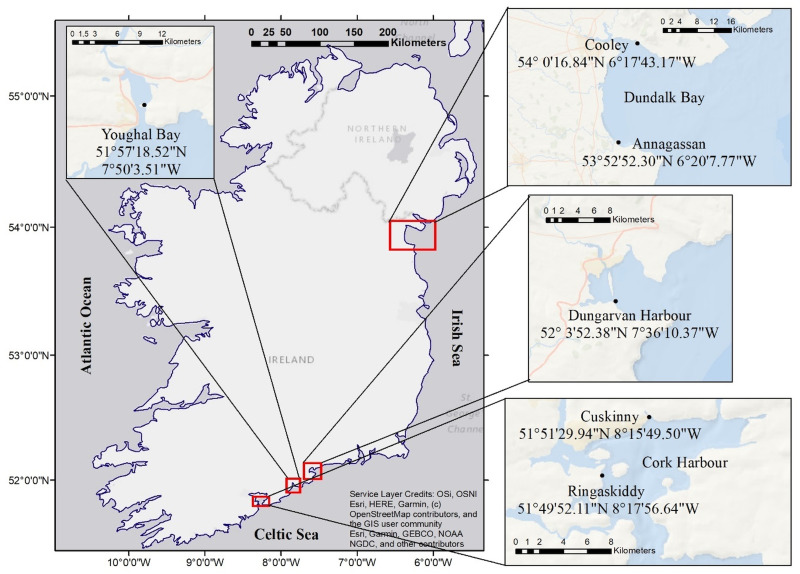
Cockles were sampled from spring 2018 to spring 2019 at Cork Harbour (Ringaskiddy and Cuskinny, n = 257) on the south coast (Celtic Sea) of Ireland and from summer 2018 to spring 2019 at Youghal Bay (n = 69) and Dungarvan Harbour (n = 169), with an extensive farming of Pacific oysters (Crassostrea gigas), on the south coast (Celtic Sea) of Ireland, where there is no commercial fishing activity (Fig. 1). Additionally, 240 cockles were collected from summer 2018 to spring 2019 at the commercial fishery at Dundalk Bay (Annagassan and Cooley) on the northeast coast (Irish Sea) of Ireland (Fig. 1). Dundalk Bay is a classified Bivalve Mollusc Production Area and it has supported a commercial dredge cockle (C. edule) fishery since 2001 (Hervas et al., 2008).
Fig. 1.
Map of Ireland highlighting the cockle sample sites with coordinates.
The four locations are bays with intertidal sand and mudflats, where a variety of bivalves inhabit. Cerastoderma edule densities in the selected sites vary from higher densities, 9.33 ± 3.5 ind m−2, in Dundalk Bay (Shellfish Stocks and Fisheries Review, Marine Institute and BIM, 2017) to lower densities in the no commercial fishing sites: 1.6 ± 2.1 ind m−2 in Youghal Bay; 0.8 ± 1.7 ind m−2 in Cork Harbour; and 0.4 ± 1.3 ind m−2 in Dungarvan Harbour (Fermer et al., 2011). Cockle mortalities associated with disseminated neoplasia have been previously reported in Cork Harbour (Morgan et al., 2012). While a massive cockle mortality event associated with dinoflagellates occurred in Youghal Bay in the past (Ottway et al., 1979). Dungarvan Harbour has also a history of OsHV-1-associated mortality events (Lynch et al., 2012; Prado-Alvarez et al., 2016). However, to the best of our knowledge, Dundalk Bay has not previously recorded mortality events.
Even though Cork Harbour is an important industrial area and is a busy ferry/shipping terminal, its trophic status (based on the nutrient levels such as phosphorus and nitrogen, growth of algae and dissolved oxygen concentration) was qualified as unpolluted by the EPA in 2017 along with Dungarvan Harbour, an extremely sheltered bay, while Youghal Bay, close to an urban hub, was classified as intermediate polluted area. In turn, Dundalk Bay showed a localized effect on the trophic status, with Annagassan classified as an unpolluted area and Cooley as an intermediate polluted area, probably due to the influence of water discharge from the local river.
Cockles were collected by hand raking from the intertidal area of each site at spring tide (0.5–1 m) and seasonal intervals. The sample size, outlined in Table 1, was dependant on the availability of cockles at each site and season. Cockles were kept at ambient temperature during the fieldwork and were processed either on the same day or were held out of water overnight at 4 °C and were processed the next day.
Table 1.
Sample sites, seasons and number of cockles collected
| Sampling dates | Cork Harbour | Youghal Bay | Dungarvan Harbour | Dundalk Bay | Totals |
|---|---|---|---|---|---|
| Spring 2018 | 60 | – | – | – | 60 |
| Summer 2018 | 39 | 11 | 51 | 60 | 161 |
| Autumn 2018 | 60 | 16 | 58 | 60 | 194 |
| Winter 2018/19 | 38 | 30 | 30 | 60 | 158 |
| Spring 2019 | 60 | 12 | 30 | 60 | 162 |
| Totalsa | 257 (34.9%) | 69 (9.4%) | 169 (23.0%) | 240 (32.7%) | 735 |
(–) No samples collected.
In brackets the % representation of all cockles sampled.
Sample processing and diagnostic methods
Morphometrics
Measurements included (A) cockle body size [height (mm)] of each individual that was recorded using Vernier callipers. The (B) cockle's age was established by counting the annual winter growth rings. For verification and validation purposes, the number of growth rings was confirmed by a second researcher, as it is recognized that variations in growth during the same year lead to the possible formation of extra stria (de Montaudouin, 1996). Ultimately, the presence or absence of epiflora on the shell of each individual was also recorded. Epiflora was mainly composed of epiphytic algae, although no specific species were identified.
Histology
A transverse section of each cockle was excised, including the digestive gland, gonads, gills and mantle tissues; as well as a section of the hinge ligament and a section of the tip of the foot were taken for histological examination. The three sections were placed in a labelled histocassette and were preserved in Davidson's fixative for 48 h (Shaw and Battle, 1957) prior to processing. Paraffin-embedded tissue blocks were sectioned at 5 μm and stained using haematoxylin and eosin (Sigma Aldrich, USA) (Howard and Smith, 1983). Tissue slides were screened at a magnification of 40x and under oil at 100x for visualization of the pathogens and tissue pathology.
Molecular techniques for cockle species identification and pathological screening
A small piece of gill tissue (2–5 mm2 of tissue) from each cockle was taken and stored at −20 °C for molecular assays, to determine the European cockle species [C. edule (Linnaeus, 1758) or Cerastoderma glaucum (Poiret, 1789)] and for subsequent pathological screening. Genomic DNA from the cockle gill tissue was extracted using the chelex-100 extraction method (Walsh et al., 1991; Lynch et al., 2008).
Polymerase chain reaction (PCR) was carried out to amplify the nuclear DNA markers ITS-for/ITS Ce-R/ITS Cg-R to differentiate between the presence of C. edule, C. glaucum species or hybrids (Freire et al., 2011; Table 2). Amplification was conducted following the reaction mixture and thermocycling conditions, as well as the visualization of the product, described in Albuixech-Martí et al. (2020).
Table 2.
Description of PCR primer pairs showing sequences for each forward and reverse primer and expected product size
| Primer sequence (5′–3′) | ||||
|---|---|---|---|---|
| Primer pair | Forward | Reverse | Product size (bp) | Primer specificity |
| ITS-for ITS Ce-R |
GTT TCC GTA GGT GAA CCT G | AAG CAG CGA GAA GCC GTT C | 190 | Cerastoderma edule |
| ITS-for ITS Cg-R |
GTT TCC GTA GGT GAA CCT G | AAT TCG CCA TCG TCG G | 470 | Cerastoderma glaucum |
| HAP-F1 HAP-R3 |
GTT CTT TCW TGA TTC TAT GMA | AKR HRT TCC TWG TTC AAG AYG A | 350 | Haplosporidia spp. |
| TAP-For TAP-Rev |
ATC TAA CTA GCT GTC GCT AAC TCG T | CTT TCA AGA TTA CCC GGC TCT GC | 165 | Minchinia tapetis |
| MER-For MER-Rev |
ATC TAA CTA GCT GTC ACT ATG GAA AA | ACG CAC ATT AAA GAT TGC CCA GCT CTT T | 170 | Minchinia mercenariae-like |
| Vib-F3 Vib-R3 |
CAA CAG AAG AAG CAC CGG CT | CAC GCT TTC GCA TCT GAG TG | 286 | Vibrio spp. |
| OHVA OHVB |
TGC TGG CTG ATG TGA TGG CTT TGG | GGA TAT GGA GCT GCG GCG CT | 385 | Ostreid herpesvirus-1 and variants |
| MicIF1 MicIR1 |
GTG GAC GCT AGT CTC ACA GGT T | TTG CAC CAG AAG GTT TAC GAC ACA T | 180 | Microsporidia sp.1 |
| MicIF2 MicIR2 |
ATG CAT GCG TAA GCG AAG CAG TTA T | TCT CTT GCA CCA GAA GGT TTA CGA C | 180 | Microsporidia sp. 2 |
Standard PCR screening for haplosporidian detection was carried out in cockle gill tissue samples (n = 735) using generic haplosporidian HAP-F1 and HAP-R3 primers that amplify small regions of the SSU rDNA of most haplosporidians (Renault et al., 2000; Molloy et al., 2012; Table 2). Amplification was conducted following the reaction mixture and thermocycling conditions, as well as the visualization of the product, described in Albuixech-Martí et al. (2020).
Standard PCR screening for Minchinia spp. detection was carried out in the cockle samples that were positive for Haplosporidia by conventional PCR, using specific primers for Minchinia tapetis (TAP-For/Rev) and Minchinia mercenariae-like (MER-For/Rev) (Albuixech-Martí et al., 2020; Table 2) in the appropriate pairings and separate PCR reactions. Amplification was conducted following the reaction mixture and thermocycling conditions, as well as the visualization of the product, described in Albuixech-Martí et al. (2020).
Standard PCR screening for Vibrio spp. in cockle gill tissues was conducted using generic primers Vib-F3/R3, developed in our lab for the detection of Vibrio genus [Kett et al., unpublished as per Notaro et al. (2021), data confirmed to be specific for Vibrio spp. by Sanger sequencing; Table 2]. The screening was conducted in all the collected cockles (n = 735). A total of 2 μL of DNA per individual was used in a total volume of 27.5 μL of the reaction mixture containing: 5 μL of 5x green buffer, 5 μL of dNTPs (1.25 mm), 0.5 μL of MgCl2, 0.25 μL of forward and reverse primers, 0.1 μL of GoTaq polymerases, 1.5 μL of DMSO and 12.9 μL of ddH2O. Cycling conditions consisted of an initial denaturation of the sample at 95 °C for 1 min followed by 35 cycles of 94 °C for 20 s, 56 °C for 30 s, 72 °C for 30 s and a final elongation at 72 °C for 7 min. Electrophoresis of the amplification products was conducted in a 2% agarose gel.
Standard PCR screening for OsHV-1 and variants, including OsHV-1 μVar, was carried out in cockle gill tissue samples (n = 735) using specific primers OHVA/OHVB that amplify small products of the ORF4 gene in the OsHV-1 virus and its variants (Lynch et al., 2013; Table 2). Amplification was conducted following the reaction mixture and thermocycling conditions described in Lynch et al. (2013). Electrophoresis of the amplification products was conducted in a 2% agarose gel.
Standard PCR using specific primers MicIF1/MicIR1 and MicIF2/MicIR2 (Lynch et al., unpublished data confirmed to be specific to Microsporidia spp. by Sanger sequencing; Table 2) to detect two unidentified microsporidian species was carried out in cockle gill tissue samples (n = 735). Both reaction mixture and thermocycling conditions were the same as used with the PCR for OsHV-1 and variants, as well as the visualization of the product.
Real-time quantitative PCR (qPCR) for detection and quantification of Vibrio aestuarianus in the cockle samples that were positive for Vibrio spp. was performed using specific primers (Table 3) for detection of the molecular chaperone dnaJ gene, following the protocol of McCleary and Henshilwood (2015). All samples were performed in triplicates using 5 μL of DNA. Ct values were used to determine real-time PCR quantification and detection limits. A tested sample was considered positive if its mean Ct value was below 37.
Table 3.
Description of qPCR primer pairs and probe
| Primers | Sequence | Specificity |
|---|---|---|
|
dnaJ f420 dnaJ r456 dnaJ p441 (probe) |
GTGAAGGGACGGGTGCTAAG CCATGACAAGTGCCACAAGTCT FAM-AGGGCACGTCGGC-MGB |
Vibrio aestuarianus |
|
HVDP-F HVDP-R |
ATTGATGATGTGGATAATCTGTG GGTAAATACCATTGGTCTTGTTCC |
Ostreid herpesvirus-1 and variants |
Viral load for OsHV-1 and variants was investigated by qPCR with a small number of individuals (n = 30, whose standard PCR results displayed some smear in the visualization of the product) to verify that there was no viral load by carrying out a more specific screening and increasing the amount of DNA processed. The protocol of Pepin et al. (2008) was followed, using the specific primers HVDP-F/HVDP-R (Table 3). qPCR was conducted in duplicate using 5 μL DNA by a Thermo Hybaid PCR express thermal cycler.
Direct sequencing was carried out on representative PCR products (n = 10) amplified using generic Vibrio Vib-F3/R3 primers (Kett et al., unpublished as per Notaro et al., 2021) to identify the Vibrio species present in the samples. Likewise, direct sequencing was carried out on representative PCR products (n = 5) amplified using generic haplosporidian HAP-F1 and HAP-R3 primers (Renault et al., 2000; Molloy et al., 2012) to identify the species present in the samples beyond the Minchinia species screened. Genomic DNA from selected individuals was isolated and purified using the QIAquick Gel Extraction Kit (QIAgen) prior to direct sequencing. Both the forward and reverse strands of DNA samples were sequenced commercially (Source Bioscience). Each sequence was matched against the National Center for Biotechnology Information (NCBI) nucleotide database with BLASTn (Basic Local Alignment Search Tool), which finds regions of local similarity between sequences to identify and confirm the DNA being detected in the PCRs.
Environmental data
This study was conducted using E.U. Copernicus Marine Service Information (marine.copernicus.eu/services-portfolio/access-to-products). Two environmental parameters critical in shaping host–pathogen interactions in estuarine and marine environments (Coen and Bishop, 2015) were selected for this study as potential confounding factors: seawater temperature and salinity. Recorded data in seawater temperature (°C) and salinity (PSU), taken monthly at a depth of 0.5 m below sea level, were downloaded by ArcGIS Desktop 10.5.1, Redlands, CA, USA (Environmental Systems Research Institute, 2017) throughout the duration of the study (from April 2018 to May 2019) from the different sample sites (coordinate-based) along the Irish coast.
Statistical analysis
Statistical analysis was performed in R version 3.2.3. statistical software. The pathogen occurrence was modelled as pathogen presence/absence in each cockle for each pathogen group and their combinations, and it was used in the subsequent analysis to examine the associations between the different pathogen groups present through the samples.
Association screening approach (SCN), described in detail by Vaumourin et al. (2014), was conducted running 5000 simulations (NS) with the presence/absence of Haplosporidia and Vibrio spp. to determine a potential association between these two pathogen groups, under the null hypothesis of random pathogen associations [P(SCN) < 0.05]. Subsequently, a second SCN (NS = 5000) was performed with the presence/absence of the different pathogen species detected and quantified in the samples, i.e. M. tapetis, M. mercenariae-like and V. aestuarianus.
Corrected Pearson's χ2 test, described in detail by Hellard et al. (2012), was applied to the Haplosporidia and Vibrio spp. presence/absence data to determine if the coinfection was due to an underlying biological interaction or to confounding factors considered (seawater temperature, salinity, cockle size and age, and epiflora on their shells). Likewise, the test was applied to the M. tapetis and M. mercenariae-like presence/absence data. Biological interactions between two pathogens, therefore, will be suspected when the probability of coinfection is not random once confounding factors have been considered [P(Corr χ2) < 0.05]. In the model, running 1000 bootstraps, the effects of the confounding factors are summarized as F1 + F2 + F3 + F4 + F5.
Moreover, Pearson's χ2 tests were used to determine whether the prevalence observed of single infections and two-pathogen coinfections was significantly different [P(Chisq) < 0.05] among the sample sites and throughout the year. Fisher's exact test was conducted when the frequencies of the pathogen occurrence in a site or season were <4, to gain accuracy.
Results
In total, 735 cockles were screened to determine the prevalence of infection and coinfections of the target pathogens (Table 4). The cockle speciation PCR confirmed that all the individuals from all sample sites were C. edule.
Table 4.
Prevalence (%, positive individuals/screened individuals) of Haplosporidia spp., Minchinia tapetis, M. mercenariae-like, Vibrio spp. and Vibrio aestuarianus screening in Cerastoderma edule at each sample site by season
| Sample site | Pathogen identification | Spring 2018 | Summer 2018 | Autumn 2018 | Winter 2018/19 | Spring 2019 | Totals |
|---|---|---|---|---|---|---|---|
| Cork Harbour – Ringaskiddy and Cuskinny | Haplosporidia spp. | 45.0% (27/60)a | 15.4% (6/39) | 41.7% (25/60) | 21.1% (8/38) | 28.3% (17/60) | 32.3% (83/257) |
| Minchinia tapetis | 15.0% (9/60) | 7.7% (3/39) | 18.3% (11/60)a | 2.6% (1/38) | 1.7% (1/60) | 9.7% (25/257) | |
| Minchinia mercenariae-like | 23.3% (14/60)a | 0% (0/39) | 8.3% (5/60) | 0% (0/38) | 10.0% (6/60) | 9.7% (25/257) | |
| Vibrio spp.b | 13.3% (8/60) | 12.8% (5/39) | 15.0% (9/60)a | 5.3% (2/38) | 11.7% (7/60) | 12.1% (31/257) | |
| V. aestuarianus | 1.7% (1/60)a | 0% (0/39) | 0% (0/60) | 0% (0/38) | 1.7% (1/60)a | 0.8% (2/257) | |
| Youghal Bay | Haplosporidia spp. | – | 9.1% (1/11) | 12.5% (2/16) | 56.7% (17/30)a | 8.33% (1/12) | 30.4% (21/69) |
| Minchinia tapetis | – | 0% (0/11) | 0% (0/16) | 0% (0/30) | 0% (0/12) | 0% (0/69) | |
| Minchinia mercenariae-like | – | 0% (0/11) | 12.5% (2/16)a | 3.3% (1/30) | 0% (0/12) | 4.3% (3/69) | |
| Vibrio spp.b | – | 27.3% (3/11) | 43.8% (7/16)a | 16.7% (5/30) | 33.3% (4/12) | 27.5% (19/69) | |
| V. aestuarianus | – | 27.3% (3/11)a | 6.3% (1/16) | 0% (0/30) | 0% (0/12) | 5.8% (4/69) | |
| Dungarvan Harbour | Haplosporidia spp. | – | 39.2% (20/51)a | 10.3% (6/58) | 30% (9/30) | 40% (12/30) | 27.8% (47/169) |
| Minchinia tapetis | – | 2.0% (1/51)a | 0% (0/58) | 0% (0/30) | 0% (0/30) | 0.6% (1/169) | |
| Minchinia mercenariae-like | – | 0% (0/51) | 0% (0/58) | 0% (0/30) | 10.0% (3/30)a | 1.8% (3/169) | |
| Vibrio spp.b | – | 56.9% (29/51)a | 56.9% (33/58)a | 6.7% (2/30) | 36.7% (11/30) | 44.4% (75/169) | |
| V. aestuarianus | – | 43.1% (22/51)a | 0% (0/58) | 0% (0/30) | 3.3% (1/30) | 13.6% (23/169) | |
| Dundalk Bay – Annagassan and Cooley | Haplosporidia spp. | – | 98.3% (59/60)a | 28.3% (17/60) | 45.0% (27/60) | 38.3% (23/60) | 52.5% (126/240) |
| Minchinia tapetis | – | 70.0% (42/60)a | 23.3% (14/60) | 0% (0/60) | 0% (0/60) | 23.3% (56/240) | |
| Minchinia mercenariae-like | – | 11.7% (7/60)a | 0% (0/60) | 1.7% (1/60) | 3.3% (2/60) | 4.2% (10/240) | |
| Vibrio spp.b | – | 38.3% (23/60)a | 33.3% (20/60) | 3.3% (2/60) | 26.7% (16/60) | 25.4% (61/240) | |
| V. aestuarianus | – | 3.3% (2/60) | 10% (6/60)a | 0% (0/60) | 0% (0/60) | 3.3% (8/240) | |
| Totals by season | Haplosporidia spp. | 45.0% (27/60) | 53.4% (86/161)a | 25.8% (50/194) | 38.6% (61/158) | 32.7% (53/162) | 37.7% (277/735) |
| Minchinia tapetis | 15.0% (9/60) | 28.6% (46/161)a | 12.9% (25/194) | 0.6% (1/158) | 0.6% (1/162) | 11.2% (82/735) | |
| Minchinia mercenariae-like | 23.3% (14/60)a | 4.3% (7/161) | 3.6% (7/194) | 1.3% (2/158) | 6.8% (11/162) | 5.6% (41/735) | |
| Vibrio spp.b | 13.3% (8/60) | 37.3% (60/161)a | 35.6% (69/194) | 7.0% (11/158) | 23.5% (38/162) | 25.3% (186/735) | |
| V. aestuarianus | 1.7% (1/60) | 16.8% (27/161)a | 3.6% (7/194) | 0.0% (0/158) | 1.2% (2/162) | 5.0% (37/735) |
(–) No samples.
The highest prevalence (%) for each pathogen group at each site/season is highlighted in bold.
Potential Vibrio species were defined in Table 5.
Diversity and prevalence of single and multiple infections and the associations between them
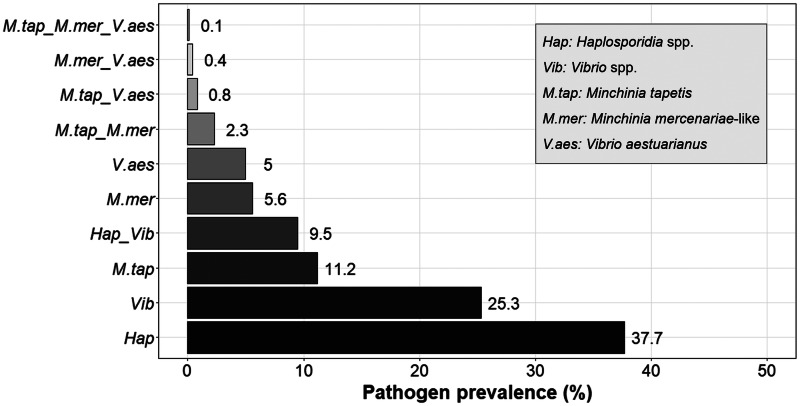
No OsHV-1 μVar nor microsporidian species were detected in any of the cockles (n = 735) screened by PCR. The only pathogen groups detected during the study were Haplosporidia and Vibrio. Overall, Haplosporidia was the most common pathogen found through the samples, with a 37.7% (n = 277) occurrence (Fig. 2, detailed data in Table 4). Among these 277 infected cockles, two haplosporidian species were identified in the study: M. tapetis which was present in 29.6% (n = 82/277) of the tested individuals, and M. mercenariae-like which was present in 14.8% (n = 41/277) (Fig. 2, detailed data in Table 4). Nevertheless, 171 (61.7%) of the tested samples that were positive in the generic haplosporidian PCR did not amplify using the more specific PCR for Minchinia spp., which could indicate the presence of other haplosporidian species occurring in the samples. Thus, five of those samples amplified using generic HAP-F1/R3 primers were commercially sequenced; however, low-complexity sequences were obtained, and no significant similarity was found in GenBank. Therefore, the 277 positive PCR cases were defined as Haplosporidia spp. for statistical and analysis purposes.
Fig. 2.
Overall prevalence of infections and coinfections (%) with the different pathogen groups (Hap: Haplosporidia spp.; Vib: Vibrio spp.; M.tap: Minchinia tapetis; M.mer: Minchinia mercenariae-like; V.aes: Vibrio aestuarianus).
Vibrio was present in 25.3% (n = 186) of the total number of cockles screened (Fig. 2, detailed data in Table 4). Of the 186 samples that were positive for Vibrio, 19.9% (n = 37/186) were confirmed by qPCR to be V. aestuarianus (Fig. 2, detailed data in Table 4). Vibrio aestuarianus-positive controls (diluted purified DNA) have a Ct = 30 (60 copies of V. aestuarianus DNA per μL). Based on that value, 13.5% (n = 5/37) of the individuals were classified as heavily infected individuals (Ct ⩽ 30), while 86.5% (n = 32/37) were classified as low-intensity infections (37 > Ct > 30).
Direct sequencing was carried out on representative PCR products (n = 10) amplified using generic Vibrio Vib-F3/R3 primers (Kett et al., unpublished as per Notaro et al., 2021) to identify other Vibrio species present in the samples apart from V. aestuarianus. Using BLASTn, three of the sequenced samples had a query length of 127–155 bp with an 83–91% query coverage and 92.6–97.9% identity to Vibrio splendidus deposited by Gao (2020) (MT445179.1; MT445177.1) and by Landreau et al. (2020) (MT345091.1) (Table 5). Three other PCR product samples that generated sequences had a query length of 82–179 bp, a query coverage of 78–83% and 92.6–98.6% identity to Vibrio kanaloae deposited by Zheng (2020) (MT505700.1) and by Barcia and Romalde (2020) (CP065151.1; CP065150.1) (Table 5). Another sample, with a query length of 158 bp, had 94% query coverage and 98% identity to V. aestuarianus deposited by Garcia (2018) (MK307696.1) and Vibrio mediterranei deposited by Gao et al. (2020) (MT269602.1) (Table 5). Two other samples were identified as Vibrio spp., as the blasted sequence was quite short (52–69 bp) to differentiate robustly between species (Table 5). The 186 positive PCR cases were defined as Vibrio spp. for statistical and analysis purposes.
Table 5.
Description of the BLASTn results obtained from the sequenced DNA of cockle samples using generic Vibrio primers Vib-F3/R3
| Sample site | Season | Species identification | Per cent identity | Query cover | Query length (bp) |
|---|---|---|---|---|---|
| Youghal Dungarvan Cooley |
Summer 2018 | Vibrio spp. among them: Vibrio splendidus and V. kanaloae | 92.59–98.58% | 78–83% | 82–179 |
| Annagassan Annagassan |
Summer 2018 | Vibrio spp. most likely V. splendidus | 97.84–97.92% | 88–91% | 154–155 |
| Annagassan | Summer 2018 | Vibrio spp. most likely V. aestuarianus or V. mediterranei | 98.01% | 94% | 158 |
| Cuskinny Dungarvan |
Autumn 2018 | Vibrio spp. | 94.23–94.29% | 67–73% | 52–69 |
Overall, 9.5% (n = 70/735) of the individuals displayed coinfection with the two pathogen groups present in the samples, Haplosporidia and Vibrio (Fig. 2). However, no significant association between them was displayed when testing by SCN (P value > 0.05), under the null hypothesis of random pathogen combinations (Table 6), i.e. the pathogen combination was considered not to occur differently than expected by chance.
Table 6.
Association screening analysis (SCN) results for all the pathogen combinations between Haplosporidia (HAP) and Vibrio (VIB) pathogen groups
| Coinfection status | Pathogen groups | Obs | LL | UL | SCN P value | |
|---|---|---|---|---|---|---|
| 2-pathogen coinfection | HAP | VIB | 70 | 51 | 90 | 0.5174 |
| 1-pathogen infection | HAP | 207 | 177 | 236 | 0.9832 | |
| VIB | 116 | 92 | 141 | 0.5142 | ||
| Not infected | 342 | 309 | 376 | 0.5222 | ||
The observed (Obs) frequency of each coinfection status is given along with the lower (LL) and upper (UL) limits of the 95% confidence envelope.
In order to identify the mechanisms driving the pathogen coinfection with Haplosporidia and Vibrio pathogen groups, corrected Pearson's χ2 test was also performed including seawater temperature, salinity, cockle height and age, as well as the presence/absence of epiflora on the shells (Tables 7 and 8), as confounding factors which may promote pathogen presence. Under the null hypothesis of independence between the two pathogens, the test showed that the observed frequencies for each pathogen combination were not significantly different (Corr χ2 = 2.51; ĉ = 0.84; P value 1 = 0.08; P value 2 = 0.08) to the expected frequencies considering the confounding factors as drivers of the infection (Supplementary material). Therefore, the proportion of double infected individuals can be explained because the same factors promote the pathogen presence and not because the different pathogen groups are interacting synergistically. The differences in the considered environmental and biotic factors between sample sites and throughout the time, thus, might have been driving the risk of infection in C. edule populations.
Table 7.
Mean ± s.d. of abiotic (seawater temperature and salinity) and biotic factors (cockle height and age) and the overall percentage of the presence of epiflora on the cockle shells at each sample site
| Mean ± s.d. | Cork Harbour | Youghal Bay | Dungarvan Harbour | Dundalk Bay |
|---|---|---|---|---|
| Temperature (°C) | 11.8 ± 2.6 | 10.7 ± 2.9 | 12.2 ± 3.0 | 10.8 ± 4.2 |
| Salinity (PSU) | 33.6 ± 0.8 | 33.9 ± 0.03 | 33.9 ± 0.2 | 30.4 ± 0.5 |
| Height (mm) | 29.7 ± 7.8 | 27.3 ± 4.8 | 22.0 ± 4.5 | 29.6 ± 5.3 |
| Age (growth rings) | 3.6 ± 1.9 | 2.9 ± 1.7 | 1.7 ± 1.0 | 3.0 ± 1.6 |
| Epiflora (presence/absence) | 10.9% | 7.2% | 4.7% | 1.7% |
The largest value for each factor is highlighted in bold.
Table 8.
Mean ± s.d. of abiotic (seawater temperature and salinity) and biotic factors (cockle height and age) and the overall percentage of the presence of epiflora on the cockle shells by season
| Mean ± s.d. | Spring 2018 | Summer 2018 | Autumn 2018 | Winter 2018/19 | Spring 2019 |
|---|---|---|---|---|---|
| Temperature (°C) | 12.2 ± 3.9 | 12.0 ± 3.3 | 11.9 ± 3.3 | 11.5 ± 3.4 | 11.4 ± 3.4 |
| Salinity (PSU) | 32.2 ± 1.6 | 33.1 ± 1.4 | 32.9 ± 1.6 | 32.7 ± 1.7 | 32.6 ± 1.7 |
| Height (mm) | 28.0 ± 7.4 | 27.2 ± 7.1 | 27.5 ± 6.9 | 27.8 ± 6.7 | 27.9 ± 6.6 |
| Age (growth rings) | 2.7 ± 1.7 | 2.8 ± 1.7 | 2.8 ± 1.8 | 2.9 ± 1.8 | 2.9 ± 1.7 |
| Epiflora (presence/absence) | 26.7% | 8.7% | 2.1% | 1.9% | 4.9% |
Regarding the specific species quantified in the samples, only 0.8% (n = 6/735) of the individuals were coinfected with V. aestuarianus and M. tapetis, while 0.4% (n = 3/735) were coinfected with V. aestuarianus and M. mercenariae-like (Fig. 2). Likewise, 13.8% of the Minchinia-infected cockles showed a coinfection with both Minchinia species (n = 17/123); however, the percentage fell to 2.3% when all the screened cockles were considered (n = 17/735) (Fig. 2). While coinfection with both Minchinia species and V. aestuarianus was only observed in one of the samples (0.1%, n = 1/735) (Fig. 2).
Associations between M. tapetis, M. mercenariae-like and V. aestuarianus and all the possible combinations were tested by SCN, revealing a significant association between the two species of Minchinia (Table 9). The observed number of coinfected individuals with both Minchinia species were significantly overrepresented compared to expected by random chance (P value < 0.001; Table 9), indicating a significant positive two-way association between these two species. While M. mercenariae-like was found to occur alone significantly less often than expected by chance (P value < 0.05; Table 9).
Table 9.
Association screening analysis (SCN) results for all the pathogen combinations between M. tapetis (MT), M. mercenariae-like (MM) and V. aestuarianus (VA)
| Coinfection status | Pathogen species | Obs | LL | UL | SCN P value | ||
|---|---|---|---|---|---|---|---|
| 3-pathogen coinfection | MT | MM | VA | 1 | 0 | 2 | 0.4292 |
| 2-pathogen coinfection | MT | MM | 16 | 0 | 11 | 0.0004 | |
| MT | VA | 5 | 0 | 10 | 0.7036 | ||
| MM | VA | 2 | 0 | 6 | 0.5568 | ||
| 1-pathogen infection | MT | 60 | 53 | 95 | 0.0996 | ||
| MM | 22 | 21 | 50 | 0.0296 | |||
| VA | 29 | 18 | 46 | 0.8016 | |||
| Not infected | 600 | 558 | 612 | 0.1836 | |||
The observed (Obs) frequency of each coinfection status is given along with the lower (LL) and upper (UL) limits of the 95% confidence envelope. Significant associations (P value < 0.05) are highlighted in bold.
The association between Minchinia species was further examined by corrected Pearson's χ2 test in order to assess if the biotic and abiotic factors considered in the study (Tables 7 and 8) influenced this coinfection. The test showed that the observed frequencies for each pathogen combination were significantly different from the expected frequencies considering the confounding factors as drivers of the infection (Supplementary material). The significant outcome (Corr χ2 = 3.04; ĉ = 0.59; P value 1 = 0.02; P value 2 = 0.02) showed that both pathogens are not independent, and the proportion of double infected individuals cannot be due only to shared confounding factors.
Site influence on single infections and coinfections
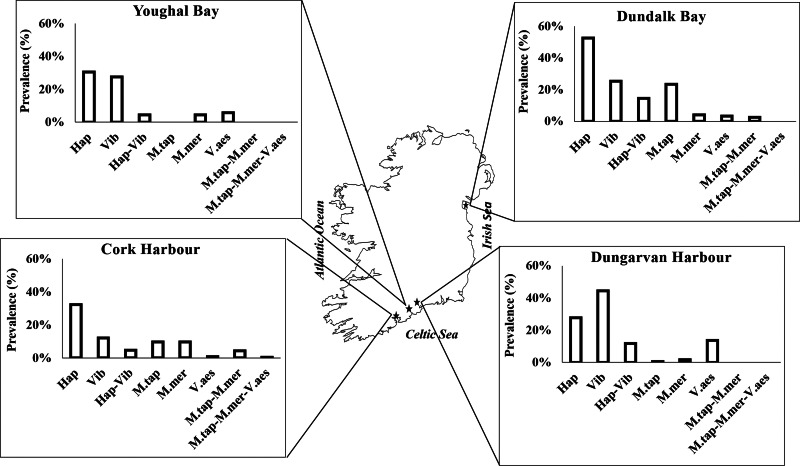
Spatial variability was assessed and significant differences [P(Chisq) < 0.05] were found between the sample sites when the prevalence of single infections and two-pathogen coinfections was considered (Fig. 3). Dundalk Bay was the site with the highest presence of Haploporidia spp. (52.5%, n = 126/240), showing a substantially higher prevalence of M. tapetis (23.3%, n = 56/240) than the other sites, while no presence of M. tapetis was observed in Youghal Bay. Dundalk Bay showed lower seawater salinity and a lower presence of epiflora on the cockle shells than the other sample sites (Table 7). Dungarvan Harbour, with the smallest and youngest cockles collected (Table 7), showed the highest presence of Vibrio spp. (44.4%, n = 75/169) and a peak of V. aestuarianus (13.6%, n = 23/169). Moreover, three out of the five individuals with the highest intensity of infection (Ct ⩽ 30) with V. aestuarianus were observed at Dungarvan Harbour. Accordingly, the highest prevalence of coinfection with Haplosporidia and Vibrio spp. occurred in Dundalk Bay (14.6%, n = 35/240) and Dungarvan Harbour (11.8%, n = 20/169). However, coinfection with M. tapetis and M. mercenariae-like was absent in Dungarvan Harbour and Youghal Bay, with Cork Harbour being the site with the highest prevalence (4.3%, n = 11/257). The other two-pathogen coinfections with Minchinia species and V. aestuarianus showed no significant differences between the sites [P(Chisq) >0.05] since the total number of positive cases detected was too low [M. tapetis and V. aestuarianus (n = 6); M. mercenariae and V. aestuarianus (n = 3)]. Likewise, Cork Harbour was the only site that showed three-pathogen coinfection with both Minchinia species and V. aestuarianus (0.4%, n = 1/257). Cork Harbour, the site with the oldest cockles, had the highest presence of epiflora on the cockle shells compared to the other sample sites (Table 7).
Fig. 3.
Prevalence (%) of Haplosporidia spp. (Hap), Vibrio spp. (Vib), Minchinia tapetis (M.tap), M. mercenariae-like (M.mer) and V. aestuarianus (V.aes) single infections and coinfections (Hap_Vib; M.tap_M.mer; M.tap_M.mer_V.aes) at each sample site.
Seasonal impact on single infections and coinfections
Significant seasonal variability [P(Chisq) <0.001] was exhibited when the prevalence of single infections and two-pathogen coinfections were compared between seasons (Fig. 4). The absence of V. aestuarianus and, in general, a low prevalence of infections were observed in winter 2018/19 (Fig. 4), except for Haplosporidia spp. (38.6%, n = 61/158). Summer 2018 had the highest prevalence for all pathogens (Fig. 4), except for M. mercenariae-like that exhibited the highest prevalence in spring 2018 (23.3%, n = 14/60). Furthermore, the five individuals with the highest intensity of infection (Ct ⩽ 30) with V. aestuarianus were observed during summer 2018. The highest prevalence of coinfection with Haplosporidia and Vibrio spp. occurred in summer 2018 (21.7%, n = 35/161), while the highest coinfection prevalence with M. tapetis and M. mercenariae-like occurred in spring 2018 (11.7%, n = 7/60), with no occurrence in winter 2018/19. The higher pathogen incidence during spring and summer corresponded with higher seawater temperature and a peak in the presence of epiflora on the cockle shells (Table 8). The other two-pathogen coinfections with Minchinia species and V. aestuarianus showed no significant differences throughout the year [P(Chisq) > 0.05] since the total number of positive cases detected was too low [M. tapetis and V. aestuarianus (n = 6); M. mercenariae and V. aestuarianus (n = 3)]. There was only one case of three-pathogen coinfection with M. tapetis, M. mercenariae-like and V. aestuarianus in spring 2019 (0.6%, n = 1/162).
Fig. 4.
(A) Prevalence (%) of Haplosporidia (Hap) and Vibrio (Vib) single infections and coinfection (Hap-Vib) by season. (B) Prevalence (%) of Minchinia tapetis (M.tap), M. mercenariae-like (M.mer) and V. aestuarianus (V.aes) single infections and coinfections (M.tap_M.mer and M.tap_M.mer_V.aes) by season.
Histopathology
The histological screening was used as a validation method of the PCR and qPCR results. Haplosporidian infection was observed in histological sections of 67 cockles out of a subsample of 70 samples (95.7% visual confirmation) that were positive for generic haplosporidian PCR. Mostly haplosporidians were observed as multinucleate plasmodia, multiple haplosporidia-like sporonts and developing spores similar to those described for Minchinia spp. (Ramilo et al., 2018; Lynch et al., 2020b; Fig. 5), and as uninucleate cells stage in lower numbers. Among the sample sites, the haplosporidia-like sporonts were predominantly visualized in the infected individuals from Cork Harbour in spring 2018, autumn 2018 and spring 2019; while the developing spores were observed mainly in the infected individuals from Dundalk Bay in summer 2018 and winter 2018/19. A higher prevalence of haplosporidians was recorded by PCR at both sites. The multinucleate plasmodia were visualized in cockles throughout the year at the different sample sites. The multinucleate plasmodia and haplosporidia-like sporonts were mainly observed throughout the connective tissue of the gills and digestive gland, while the developing spores were principally found in the mantle of infected animals.
Fig. 5.
(A) Multiple haplosporidia-like sporonts (black arrow) along with haemocytes (red arrow) in the connective tissue of Cerastoderma edule; (B) a large number of haplosporidian spores (black arrow) in the mantle tissue of C. edule; and (C) red arrow shows a haemocyte accumulation in the connective tissue of C. edule.
Host pathology associated with Vibrio species included haemocyte accumulation in the connective tissues, as McCleary and Henshilwood (2015) described in C. gigas infected with V. aestuarianus. In this study, some of the sections examined (n = 28) showed haemocyte accumulation in the connective tissues of C. edule (Fig. 5). However, the presence of haemocyte infiltrations throughout the organs and tissues has also been previously associated with Minchinia infection (Longshaw and Malham, 2013). In this study, only in a minor number of screened samples (n = 4), where Vibrio and Haplosporidia were detected by PCR, haemocyte infiltrations were observed, all of them at Cork Harbour.
Although OsHV-1 μVar is not visible using histology, cytopathic effects can be observed in infected hosts, such as hypertrophied cells with a low nucleus-cytoplasm ratio and nuclear abnormalities (Prado-Alvarez et al., 2016). Abnormal cell pathology associated with a viral infection was not observed in the histology (n = 70). Similarly, microsporidial spores may be visualized by microscopy in infected hosts (Garcia, 2002); however, it was not observed in the histological sections screened (n = 70).
Ten histological sections of samples that were negative by PCR were screened and no evident signal of infection was found in those, and tissues, organs and sinuses presented good integrity and a clear structure.
Discussion
The present study describes for the first time, to the best of our knowledge, simultaneous infections with haplosporidians and Vibrio spp. in wild and fished C. edule populations along the Irish coast. Coinfections with two and, especially, three pathogens, were rare in this study compared to single infected individuals. This finding may indicate that coinfected individuals succumb more easily to simultaneous infections and die. The lower number of coinfected individuals screened might indicate that the sampling was carried out on a much smaller cohort of survivors within the population. This survival bias is supported by previous studies on mortalities associated with coinfections of ostreid herpesvirus (OsHV-1 and variants) and Vibrio spp. in C. gigas in France (Petton et al., 2015; Azéma et al., 2016; Petton et al., 2019), as well as with the co-occurrence of different protozoa and bacteria in the noble pen shell Pinna nobilis in the Mediterranean Sea (Carella et al., 2020). Only three individuals presented a three-pathogen coinfection with Minchinia spp. and Vibrio spp., all located at Cork Harbour. Cork Harbour was the site with the oldest cockles, and it is well known that older hosts have more time than younger hosts to accumulate disease-causing pathogens (Lafferty and Kuris, 2009; Breitburg et al., 2015). Nevertheless, older bivalves have also more time available to develop immunotolerance and defence mechanisms compared to their younger cohorts (Renault and Novoa, 2004; Coen and Bishop, 2015). It may be the reason why cockles at Cork Harbour have not succumbed to the multiple infections.
Even though Haplosporidia and Vibrio were found simultaneously in double infected individuals, no significant biological interaction was statistically identified between those pathogen groups. Unlike other studies that have reported that multiple pathogens can interact synergistically, with the presence of one pathogen enhancing the abundance and/or virulence of the other (De Lorgeril et al., 2018; Carella et al., 2020). Concurrent infections by multiple pathogens species are more likely to occur in immune depressed individuals, which are more vulnerable to multiple opportunistic infections (De Lorgeril et al., 2018; Carella et al., 2020). These findings highlight that there can be variation amongst pathogen groups and the primary pathogen–opportunistic pathogen interplay.
It is well known that environmental and ecological factors potentially influence the mechanisms of disease transmission in marine systems, i.e. hydrodynamics, the biomass of infected animals, etc. (Petton et al., 2015). Similarities in the host environment, behaviour or susceptibility can cause correlations in the risk of infection between two parasites (Vaumourin et al., 2014). In this study, the proportion of double infected individuals with Haplosporidia and Vibrio was explained by risk factors (increased seawater temperature, reduced salinity and host condition) shared by the two pathogen groups, increasing the probability of infection by both infectious agents. Such confounding factors may influence host exposure and/or host susceptibility promoting simultaneous infections, even though involved pathogens do not interact biologically (Vaumourin et al., 2015). These factors may be naturally occurring or be outcomes of coastal development (e.g. pollution) or climate change (e.g. warming, increased precipitation events and subsequent freshwater loadings into bays and estuaries), and may impact the expression of diseases directly or indirectly (Coen and Bishop, 2015). The predicted warming, increased precipitation and reduced salinity of coastal waters in temperate regions will provide new areas for the natural occurrence of pathogenic strains and may also impact the resilience of bivalve hosts (Rowley et al., 2014; Lynch et al., 2020a).
Consequently, spatial variability in the pathogen infection was detected driven by environmental and biological variables. The highest prevalence of coinfection with Haplosporidia and Vibrio spp. occurred in Dundalk Bay, which had a lower mean salinity throughout the year compared to the other sites. Gorrasi et al. (2020) observed that the abundance of Vibrio genus in water decreased along the salinity gradient within a marine saltern hypersaline environment. In general, marine vibrios are considered halotolerant, with no extreme halophiles reported within this genus (Gorrasi et al., 2020). Previous studies have highlighted the intolerance of haplosporidians to low salinity in C. edule (Albuixech-Martí et al., 2020), eastern oyster Crassostrea virginica (Ford and Haskin, 1982; Burreson and Ford, 2004) and European flat oyster Ostrea edulis (Flannery et al., 2014). The high prevalence of haplosporidians at Dundalk Bay may be due to its commercial fishery activity. It is recognized that fishing and dredging may unbalance the interplay between infectious disease agents and their hosts in marine ecosystems (Rowley et al., 2014; Coen and Bishop, 2015). Cranfield et al. (1999, 2005) hypothesized that extensive dredging in Foveaux Strait of New Zealand contributed to increased densities of Chilean oysters (Ostrea chilensis) and decreased densities of other filter-feeding organisms, that may have previously reduced the dispersal stages of the haplosporidian Bonamia exitiosa, enhancing disease transmission. The high cockle density found at Dundalk Bay may have facilitated the infection among the cockle population, due to the proximity of infected to non-infected cockles. The cultivated and harvested molluscs tend to have a much greater incidence of disease than wild molluscs, which may be because they are placed at high densities which enhance disease transmission and cause stress (competition for resources, space, etc.) among individuals (Ford, 2002).
Dungarvan Harbour also displayed a high prevalence of coinfection with Haplosporidia and Vibrio spp. compared to the other sites. Cockles from Dungarvan Harbour were the smallest and the youngest ones. Although it is accepted that older hosts have more time to accumulate disease-causing pathogens (Lafferty and Kuris, 2009; Breitburg et al., 2015), the susceptibility of small bivalves to viral and bacterial infections is generally greater than for larger adults (Renault and Novoa, 2004; Coen and Bishop, 2015). Moreover, the proximity of an extensive C. gigas culture site at Dungarvan Harbour, where Vibrio spp. are endemic (pers. comm. with Marine Institute Ireland), can have promoted the presence of Vibrio in the environment. Particularly, V. aestuarianus and V. splendidus, which have been identified in this study, have been described as increasingly harmful pathogens of C. gigas aquaculture (Le Roux et al., 2002; Garnier et al., 2007; McCleary and Henshilwood, 2015). Caballes et al. (2012) demonstrated interspecific transmission of Vibrio rotiferianus between co-occurring echinoderm species.
Even though the natural host for OsHV-1 are C. gigas, this virus can behave opportunistically and infect other cohabiting bivalve species (Bookelaar et al., 2020). It has been previously reported in C. edule, in Ireland (Bookelaar et al., 2020) and in the Sydney cockle, Anadara trapezia, in Australia (Evans et al., 2017). Nevertheless, no presence of OsHV-1 μVar was detected in the study, despite the extensive farming of C. gigas at Dungarvan Harbour, which has a history of OsHV-1-associated mortality events (Lynch et al., 2012; Prado-Alvarez et al., 2016). During summer 2018, the prevalence of OsHV-1 μVar in C. gigas at Dungarvan Harbour was confirmed by Kett et al. (unpublished data) to be 48.4% (n = 92/190), with low mortality rates (max. of 6% at the high shore in July). Moreover, Bookelaar et al. (2020) observed OsHV-1μVar-infected C. edule (14.4%; n = 36/250) and infected C. gigas (range of 0–27% per month; n = 270) at Dungarvan Harbour from April 2015 to August 2015, with no recording of mortalities. Thus, the lack of OsHV-1 μVar detection in C. edule at Dungarvan Harbour in the current study may be because the virus remained within the oysters and was not shed from dying or dead oysters into the environment, as the high prevalence and low mortality recorded by Kett et al. (unpublished data) suggested. The smaller sample size (n = 81), compared to the above studies, taken during spring and summer 2018, when the OsHV-1 μVar can have a major emergence due to the higher temperatures (Renault et al., 2014), may have also influenced the results.
Seasonal variability was also observed in this study, with the highest prevalence of coinfection with Haplosporidia and Vibrio spp. occurring in summer 2018, which coincided with the highest intensity of infection with V. aestuarianus. Additionally, lower occurrences of the haplosporidian and Vibrio species quantified were detected in the cooler spring 2019 compared to spring 2018 in this study. Burreson and Ford (2004) tested that low-temperature conditions caused a dramatic reduction in the prevalence of Haplosporidium nelsoni in C. virginica. Conversely, a higher prevalence of Bonamia ostreae in European flat oyster O. edulis was associated with cooler spring temperatures over a 30-year study period in Ireland (Engelsma et al., 2010; Lynch et al., 2014). These findings would suggest that a thermal tolerance range exists for haplosporidian species. It is also well-known that bacteria belonging to the genus Vibrio are strongly thermo-dependent and their occurrence is expected to rise with increasing temperature conditions (Garnier et al., 2007; Cotter et al., 2010; Vezzulli et al., 2010). Most of the reported Vibrio-associated mass mortalities in marine invertebrates have occurred during the summer when seawater temperatures were higher than 18°C (Le Roux et al., 2002; Paillard et al., 2004; Garnier et al., 2007; Vezzulli et al., 2010). Therefore, high temperatures in summer might have triggered a major pathogen propagation and, consequently, to a higher prevalence of two-pathogen coinfection. While lower temperatures in winter, with the lowest prevalence of two-pathogen coinfection, would have kept the pathogens at a sub-lethal level in the hosts reducing the transmission between cockles. It has been observed that Vibrio spp. may enter into a dormant state when the environmental conditions become unfavourable (i.e. suboptimal or reduced temperature, elevated salinity, low nutrient concentration and extreme pH) (Lin and Schwarz, 2003; Nowakowska and Oliver, 2013; Neogi et al., 2018). The low number of coinfected individuals in winter might also suggest that most C. edule had succumbed to infection in autumn after the peak in summer.
It was also observed that with the higher temperatures in summer and spring, the presence of epiflora on the cockle shells was more frequent, which may have consequences on cockle susceptibility to infectious agents. As seen in shellfish aquaculture, the presence of biofouling on the stock can have a direct impact, causing physical damage, biological competition, mechanical interference and environmental modification, while infrastructure is also impacted (Fitridge et al., 2012). Microalgae have been pointed as a repetitive and substantial source of bacterial inoculation into the bivalve larval culture systems (Salvesen et al., 2000). Microalgae may promote and/or inhibit bacterial growth by the production of organic exudates and toxic metabolites (Salvesen et al., 2000). For instance, Vibrio species such as V. splendidus or V. parahaemolyticus have been found to be associated with microalgae (Kumazawa et al., 1991; Rehnstam-Holm et al., 2010; Notaro et al., 2021). Therefore, the presence of epiflora on the shells might have also exposed the cockles to pathogens, in particular Vibrio species.
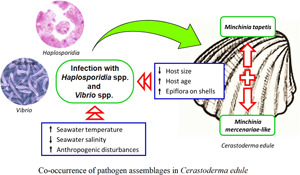
Both species of Minchinia, M. tapetis and M. mercenariae-like, were found to occur together significantly more often than expected by random chance, indicating a positive two-way association between both species. However, previous studies examining pairwise coinfections indicate that antagonistic interactions between similar types of parasites may be common (Dobson, 1985; Johnson and Hoverman, 2012). A possible explanation of the established synergetic interaction is that, despite both Minchinia species being closely related, they exhibit differences in their pathogenic effect and use of resources within the host. Carballal et al. (2020) observed that the distribution of infection with M. tapetis and M. mercenariae-like haplosporidian in cockles C. edule in Galicia was different throughout the cockle tissues. M. mercenariae-like haplosporidian was observed in the connective tissue of the digestive gland, gills and gonad, while M. tapetis was only observed in the digestive gland (Carballal et al., 2020). Further research is needed to reveal the underlying mechanism of this synergetic association in C. edule and the role that host exposure and host habitat play in it.
In brief, coinfected individuals may more easily succumb to pathogen load. The proportion of double infected individuals with haplosporidian and Vibrio was driven by environmental and biological factors, triggering a spatial and seasonal variability in the distribution of coinfection. The higher temperature in warmer months and the presence of epiflora, which can be associated with pathogens, on the cockle shells may have promoted the risk of coinfection, along with low salinity and the host condition in some of the sample sites. The close proximity to other infected host species may have also influenced the coinfection distribution in C. edule. Similarly, fished C. edule populations with high cockle densities might have been more exposed to the infection. Therefore, those factors may conform the ideal scenario for simultaneous infections, although the pathogen groups showed no biological interaction between them. Nevertheless, a positive two-way association between both Minchinia species was observed, which is suggested to be due to their different pathogenic effect and use of resources within the host. Our findings shed light on the complex interactions between multiple aetiological agents associated with host diseases in the frame of a natural system. This study highlights the consequences of the coinfections are not simply the sum of the effects caused by each infectious agent, but the result of a complex combination of direct and indirect pathogens effects, the host ecology and the environmental context.
These results are framed into the current scenario of climate change, which involves oceans warming on average by 0.6°C in the next 100 years (Wiltshire et al., 2008), with European seas especially affected (Philippart et al., 2011). The sea surface temperature around the UK and Ireland has increased at six times the rate of the global average, particularly jeopardizing species that have small thermal tolerance windows (Frost et al., 2012). As a result, previously appropriate habitats for cockles may no longer be suitable due to changes in thermal regime, oxygen levels or altered rainfall patterns (Morgan et al., 2013; Coen and Bishop, 2015). Climate change may be also expected to have a large effect on disease occurrence, especially in host species showing strong relationships between temperature/salinity and diseases, as seen for the cockle populations studied, and for many other molluscan host–parasite systems (Allam and Raftos, 2015; Coen and Bishop, 2015; Lynch et al., 2020a). The physiological stress may also cause the host to become immune-compromised and unable to suppress an infection (Suttle, 2007). Therefore, climate change could be the driver of the spreading and range expansions of infectious agents, such as bacteria, haplosporidians and viruses, in the marine ecosystem and may lead to more frequent pathogen outbreaks (Burreson and Ford, 2004; Suttle, 2007; Rowley et al., 2014; Coen and Bishop, 2015). As global climate change and anthropogenic impacts continue to modify the distribution and abundance of hosts and parasites and the ways in which they interact, integral studies like ours are important since concepts gained might be applied to other associations and to a changing environment.
A final important point to emphasize is that the relative potential of coinfections to cause the emergence of zoonoses in wild animals remains to be fully assessed (Hoarau et al., 2020). Given that two-thirds of emerging infectious diseases are zoonoses, with nearly 70% originating from wildlife (Hoarau et al., 2020), better knowledge of the interactions of infectious agents in wild reservoirs, such as cockles, will provide key insight for the understanding and management of spillover processes. Although challenging, findings from this study highlight the necessity to integrate data pertaining to host ecology, pathogen diversity, biogeography and seasonality of the disease and the environmental influence. This approach should be consistently applied to provide a more detailed picture of coinfection dynamics in individual hosts and communities over time.
Acknowledgements
We are grateful to Dr Eileen Dillane of the University College Cork (Ireland) (Research Support Officer and Genetic Laboratory Manager) for the provision of professional advice during our lab work. We also thank our colleagues Dr Kate Mahony, Dr Katie Costello and Gary Kett of University College Cork (Ireland), who assisted us with the transport, and fieldwork. For the permission to carry out the fieldwork in Dungarvan Harbour, we thank Dungarvan Shellfish Ltd (Ireland); we also thank Martin Hooey from Dundalk Fishery (Ireland) for showing us the best sampling areas in Dundalk Bay and to provide us samples.
Author contributions
Conceptualization and methodology: S.A.M., S.C.C., S.A.L.; formal analysis: S.A.M.; writing – original draft: S.A.M.; writing – review and editing: S.C.C., S.A.L.; supervision: S.C.C., S.A.L. All authors approved the final version of the manuscript before submission.
Financial support
This work was supported by the Bluefish (Grant Agreement No. 80991) Project, part-funded by the European Regional Development Fund (ERDF) through the Ireland Wales Co-operation Programme. A cross-border programme investing in the overall economic, environmental and social well-being of Ireland and Wales.
Ethical standards
Not applicable.
Supplementary material
For supplementary material accompanying this paper visit https://doi.org/10.1017/S0031182021001396.
click here to view supplementary material
Conflict of interest
None.
References
- Albuixech-Martí S, Lynch SA and Culloty SC (2020) Biotic and abiotic factors influencing haplosporidian species distribution in the cockle Cerastoderma edule in Ireland. Journal of Invertebrate Pathology 174, 107425. [DOI] [PubMed] [Google Scholar]
- Alfaro AC, Nguyen TV and Merien F (2019) The complex interactions of ostreid herpesvirus 1, Vibrio bacteria, environment and host factors in mass mortality outbreaks of Crassostrea gigas. Reviews in Aquaculture 11, 1148–1168. [Google Scholar]
- Allam B and Raftos D (2015) Immune responses to infectious diseases in bivalves. Journal of Invertebrate Pathology 131, 121–136. [DOI] [PubMed] [Google Scholar]
- Arzul I, Chollet B, Michel J, Robert M, Garcia C, Joly J-, Francois C and Miossec L (2012) One Perkinsus species may hide another: characterization of Perkinsus species present in clam production areas of France. Parasitology 139, 1757–1771. [DOI] [PubMed] [Google Scholar]
- Azéma P, Travers M, Benabdelmouna A and Dégremont L (2016) Single or dual experimental infections with Vibrio aestuarianus and OsHV-1 in diploid and triploid Crassostrea gigas at the spat, juvenile and adult stages. Journal of Invertebrate Pathology 139, 92–101. [DOI] [PubMed] [Google Scholar]
- Bookelaar B, Lynch SA and Culloty SC (2020) Host plasticity supports spread of an aquaculture introduced virus to an ecosystem engineer. Parasites & Vectors 13, 498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breitburg DL, Hondorp D, Audemard C, Carnegie RB, Burrell RB, Trice M and Clark V (2015) Landscape-level variation in disease susceptibility related to shallow-water hypoxia. PLoS ONE 10, e0116223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burreson E and Ford S (2004) A review of recent information on the Haplosporidia, with special reference to Haplosporidium nelsoni (MSX disease). Aquatic Living Resources 17, 499–517. [Google Scholar]
- Caballes CF, Schupp PJ, Pratchett MS and Rivera-Posada JA (2012) Interspecific transmission and recovery of TCBS-induced disease between Acanthaster planci and Linckia guildingi. Diseases of Aquatic Organisms 100, 263–267. [DOI] [PubMed] [Google Scholar]
- Carballal M, Iglesias D, Santamarina J, Ferro-Soto B and Villalba A (2001) Parasites and pathologic conditions of the cockle Cerastoderma edule populations of the coast of Galicia (NW Spain). Journal of Invertebrate Pathology 78, 87–97. [DOI] [PubMed] [Google Scholar]
- Carballal M, Villalba A, Iglesias D and Hine P (2003) Virus-like particles associated with large foci of heavy hemocytic infiltration in cockles Cerastoderma edule from Galicia (NW Spain). Journal of Invertebrate Pathology 84, 234–237. [DOI] [PubMed] [Google Scholar]
- Carballal MJ, Cao A, Iglesias D, Gonzalez AI and Villalba A (2020) Temporal dynamics of infection of cockles Cerastoderma edule with the protistan parasite Minchinia tapetis (Rhizaria: Haplosporida) in Galicia (NW Spain). Journal of Invertebrate Pathology 172, 107349. [DOI] [PubMed] [Google Scholar]
- Carella F, Elisabetta A, Simone F, Fulvio S, Daniela M, Prado P, Rossella P, Marino F, Eleonora F, Tobia P and De Vico G (2020) In the wake of the ongoing mass mortality events: co-occurrence of mycobacterium, haplosporidium and other pathogens in Pinna nobilis collected in Italy and Spain (Mediterranean Sea). Frontiers in Marine Science 7, 48. [Google Scholar]
- Carss DN, Brito AC, Chainho P, Ciutat A, de Montaudouin X, Fernandez Otero RM, Incera Filgueira M, Garbutt A, Goedknegt MA, Lynch SA, Mahony KE, Maire O, Malham SK, Orvain F, Olivier AVDS and Jones L (2020) Ecosystem services provided by a non-cultured shellfish species: the common cockle Cerastoderma edule. Marine Environmental Research 158, 104931. [DOI] [PubMed] [Google Scholar]
- Coen LD and Bishop MJ (2015) The ecology, evolution, impacts and management of host-parasite interactions of marine molluscs. Journal of Invertebrate Pathology 131, 177–211. [DOI] [PubMed] [Google Scholar]
- Cotter E, Malham SK, O'Keeffe S, Lynch SA, Latchford JW, King JW, Beaumont AR and Culloty SC (2010) Summer mortality of the Pacific oyster, Crassostrea gigas, in the Irish Sea: the influence of growth, biochemistry and gametogenesis. Aquaculture 303, 8–21. [Google Scholar]
- Cox F (2001) Concomitant infections, parasites and immune responses. Parasitology 122, S23–S38. [DOI] [PubMed] [Google Scholar]
- Cranfield H, Michael K and Doonan I (1999) Changes in the distribution of epifaunal reefs and oysters during 130 years of dredging for oysters in Foveaux Strait, southern New Zealand. Aquatic Conservation-Marine and Freshwater Ecosystems 9, 461–483. [Google Scholar]
- Cranfield H, Dunn A, Doonan I and Michael K (2005) Bonamia exitiosa epizootic in Ostrea chilensis from Foveaux Strait, southern New Zealand between 1986 and 1992. ICES Journal of Marine Science 62, 3–13. [Google Scholar]
- De Lorgeril J, Lucasson A, Petton B, Toulza E, Montagnani C, Clerissi C, Vidal-Dupiol J, Chaparro C, Galinier R, Escoubas J, Haffner P, Degremont L, Charriere GM, Lafont M, Delort A, Vergnes A, Chiarello M, Faury N, Rubio T, Leroy MA, Perignon A, Regler D, Morga B, Alunno-Bruscia M, Boudry P, Le Roux F, Destoumieux-Garzon D, Gueguen Y and Mitta G (2018) Immune-suppression by OsHV-1 viral infection causes fatal bacteraemia in Pacific oysters. Nature Communications 9, 4215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Montaudouin X (1996) Factors involved in growth plasticity of cockles Cerastoderma edule (L), identified by field survey and transplant experiments. Journal of Sea Research 36, 251–265. [Google Scholar]
- De Montaudouin X, Paul-Pont I, Lambert C, Gonzalez P, Raymond N, Jude F, Legeay A, Baudrimont M, Dang C, Le Grand F, Le Goic N, Bourasseau L and Paillard C (2010) Bivalve population health: multistress to identify hot spots. Marine Pollution Bulletin 60, 1307–1318. [DOI] [PubMed] [Google Scholar]
- De Montaudouin X, Arzul I, Cao A, Carballal MJ, Bruno C, Correia S, Cuesta J, Culloty S, Daffe G, Darriba S, Díaz S, Freitas R, Garcia C, Goedknegt A, Grade A, Groves E, Iglesias D, Jensen K and Villalba A (2021) Catalogue of parasites and diseases of the common cockle Cerastoderma edule, 1st Edn. Aveiro, Portugal: UA Editora – Universidade de Aveiro. doi: 10.34624/9a9c-9j21 [DOI]
- Dobson A (1985) The population-dynamics of competition between parasites. Parasitology 91, 317–347. [DOI] [PubMed] [Google Scholar]
- Engelsma MY, Kerkhoff S, Roozenburg I, Haenen OLM, van Gool A, Sistermans W, Wijnhoven S and Hummel H (2010) Epidemiology of Bonamia ostreae infecting European flat oysters Ostrea edulis from Lake Grevelingen, The Netherlands. Marine Ecology Progress Series 409, 131–142. [Google Scholar]
- Engelsma MY, Roozenburg I, Voorbergen-Laarman M, Van den Brink A, Troost K, Ysebaert T, Bateman K and Longshaw M (2011) Digenean trematodes and haplosporidian protozoans associated with summer mortality of cockles Cerastoderma edule in the Oosterschelde, The Netherlands. In European Association of Fish Pathologists Conference, Split, Croatia, pp. 14–18.
- Evans O, Paul-Pont I and Whittington RJ (2017) Detection of ostreid herpesvirus 1 microvariant DNA in aquatic invertebrate species, sediment and other samples collected from the Georges River estuary, New South Wales, Australia. Diseases of Aquatic Organisms 122, 247–255. [DOI] [PubMed] [Google Scholar]
- Fermer J, Culloty SC, Kelly TC and O'Riordan RM (2009) Intrapopulational distribution of Meiogymnophallus minutus (Digenea, Gymnophallidae) infections in its first and second intermediate host. Parasitology Research 105, 1231–1238. [DOI] [PubMed] [Google Scholar]
- Fermer J, Culloty SC, Kelly TC and O'riordan RM (2011) Parasitological survey of the edible cockle Cerastoderma edule (Bivalvia) on the south coast of Ireland. Journal of the Marine Biological Association of the United Kingdom 91, 923–928. [Google Scholar]
- Fitridge I, Dempster T, Guenther J and de Nys R (2012) The impact and control of biofouling in marine aquaculture: a review. Biofouling 28, 649–669. [DOI] [PubMed] [Google Scholar]
- Flannery G, Lynch SA, Longshaw M, Stone D, Martin P, Ramilo A, Villalba A and Culloty SC (2014) Interlaboratory variability in screening for Bonamia ostreae, a protistan parasite of the European flat oyster Ostrea edulis. Diseases of Aquatic Organisms 110, 93–99. [DOI] [PubMed] [Google Scholar]
- Ford SE (2002) Development of high disease resistance in a wild oyster population. Journal of Shellfish Research 21, 387. [Google Scholar]
- Ford S and Haskin H (1982) History and epizootiology of Haplosporidium nelsoni (Msx), an oyster pathogen in Delaware Bay, 1957–1980. Journal of Invertebrate Pathology 40, 118–141. [Google Scholar]
- Freire R, Arias A, Mendez J and Insua A (2011) Identification of European commercial cockles (Cerastoderma edule and C. glaucum) by species-specific PCR amplification of the ribosomal DNA ITS region. European Food Research and Technology 232, 83–86. [Google Scholar]
- Frost M, Baxter JM, Buckley PJ, Cox M, Dye SR and Harvey NW (2012) Impacts of climate change on fish, fisheries and aquaculture. Aquatic Conservation-Marine and Freshwater Ecosystems 22, 331–336. [Google Scholar]
- Fukami T, Dickie IA, Wilkie JP, Paulus BC, Park D, Roberts A, Buchanan PK and Allen RB (2010) Assembly history dictates ecosystem functioning: evidence from wood decomposer communities. Ecology Letters 13, 675–684. [DOI] [PubMed] [Google Scholar]
- Garcia L (2002) Laboratory identification of the microsporidia. Journal of Clinical Microbiology 40, 1892–1901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garnier M, Labreuche Y, Garcia C, Robert A and Nicolas J (2007) Evidence for the involvement of pathogenic bacteria in summer mortalities of the Pacific oyster Crassostrea gigas. Microbial Ecology 53, 187–196. [DOI] [PubMed] [Google Scholar]
- Goater C (1993) Population biology of Meiogymnophallus minutus (Trematoda, Gymnophallidae) in cockles from the Exe Estuary. Journal of the Marine Biological Association of the United Kingdom 73, 163–177. [Google Scholar]
- Gorrasi S, Pasqualetti M, Franzetti A, Pittino F and Fenice M (2020) Vibrio communities along a salinity gradient within a marine saltern hypersaline environment (Saline di Tarquinia, Italy). Environmental Microbiology 22, 4356–4366. [DOI] [PubMed] [Google Scholar]
- Hatcher MJ, Dick JTA and Dunn AM (2006) How parasites affect interactions between competitors and predators. Ecology Letters 9, 1253–1271. [DOI] [PubMed] [Google Scholar]
- Hatcher MJ, Dick JTA and Dunn AM (2012) Diverse effects of parasites in ecosystems: linking interdependent processes. Frontiers in Ecology and the Environment 10, 186–194. [Google Scholar]
- Hellard E, Pontier D, Sauvage F, Poulet H and Fouchet D (2012) True versus false parasite interactions: a robust method to take risk factors into account and its application to feline viruses. PLoS ONE 7, e29618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hellard E, Fouchet D, Vavre F and Pontier D (2015) Parasite-parasite interactions in the wild: how to detect them? Trends in Parasitology 31, 640–652. [DOI] [PubMed] [Google Scholar]
- Hervas A, Tully O, Hickey J, O'Keefe E and Kelly K (2008) Assessment, monitoring and management of the Dundalk Bay and Waterford cockle (Cerastoderma edule) Fisheries in 2007. BIM Fisheries Resource Series No. 7 (2008).
- Hoarau AOG, Mavingui P and Lebarbenchon C (2020) Coinfections in wildlife: focus on a neglected aspect of infectious disease epidemiology. PLoS Pathogens 16, e1008790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard DW and Smith CS (1983) Histologic techniques for marine bivalve mollusks. US Dept Commerce, NOAA Tech Memo NMFS-F/NEC-25.
- Johansen L and Sommer A (2001) Infectious pancreatic necrosis virus infection in Atlantic salmon Salmo salar post-smolts affects the outcome of secondary infections with infectious salmon anaemia virus or Vibrio salmonicida. Diseases of Aquatic Organisms 47, 109–117. [DOI] [PubMed] [Google Scholar]
- Johnson PTJ and Buller ID (2011) Parasite competition hidden by correlated coinfection: using surveys and experiments to understand parasite interactions. Ecology 92, 535–541. [DOI] [PubMed] [Google Scholar]
- Johnson PTJ and Hoverman JT (2012) Parasite diversity and coinfection determine pathogen infection success and host fitness. Proceedings of the National Academy of Sciences of the USA 109, 9006–9011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson PTJ, Dobson A, Lafferty KD, Marcogliese DJ, Memmott J, Orlofske SA, Poulin R and Thieltges DW (2010) When parasites become prey: ecological and epidemiological significance of eating parasites. Trends in Ecology & Evolution 25, 362–371. [DOI] [PubMed] [Google Scholar]
- Jolles AE, Ezenwa VO, Etienne RS, Turner WC and Olff H (2008) Interactions between macroparasites and microparasites drive infection patterns in free-ranging African buffalo. Ecology 89, 2239–2250. [DOI] [PubMed] [Google Scholar]
- Karvonen A, Seppala O and Valtonen ET (2009) Host immunization shapes interspecific associations in trematode parasites. Journal of Animal Ecology 78, 945–952. [DOI] [PubMed] [Google Scholar]
- Kumazawa N, Nakagaki E, Yonekawa Y, Ikura K and Morimoto N (1991) Ecological cycle of thermostable direct hemolysin-producing strains of Vibrio parahaemolyticus in a brackish-water area with special reference to mollusks and attached microalgae. Journal of Veterinary Medical Science 53, 263–267. [DOI] [PubMed] [Google Scholar]
- Kuris A and Lafferty K (1994) Community structure – larval trematodes in snail hosts. Annual Review of Ecology and Systematics 25, 189–217. [Google Scholar]
- Lafferty KD and Kuris AM (2009) Parasitic castration: the evolution and ecology of body snatchers. Trends in Parasitology 25, 564–572. [DOI] [PubMed] [Google Scholar]
- Lassalle G, de Montaudouin X, Soudant P and Paillard C (2007) Parasite co-infection of two sympatric bivalves, the Manila clam (Ruditapes philippinarum) and the cockle (Cerastoderma edule) along a latitudinal gradient. Aquatic Living Resources 20, 33–42. [Google Scholar]
- Le Roux F, Gay M, Lambert C, Waechter M, Poubalanne S, Chollet B, Nicolas J and Berthe F (2002) Comparative analysis of Vibrio splendidus-related strains isolated during Crassostrea gigas mortality events. Aquatic Living Resources 15, 251–258. [Google Scholar]
- Lin M and Schwarz J (2003) Seasonal shifts in population structure of Vibrio vulnificus in an estuarine environment as revealed by partial 16S ribosomal DNA sequencing. FEMS Microbiology Ecology 45, 23–27. [DOI] [PubMed] [Google Scholar]
- Longshaw M and Malham SK (2013) A review of the infectious agents, parasites, pathogens and commensals of European cockles (Cerastoderma edule and C. glaucum). Journal of the Marine Biological Association of the United Kingdom 93, 227–247. [Google Scholar]
- Lynch SA, Mulcahy MF and Culloty SC (2008) Efficiency of diagnostic techniques for the parasite, Bonamia ostreae, in the flat oyster, Ostrea edulis. Aquaculture 281, 17–21. [Google Scholar]
- Lynch SA, Carlsson J, Reilly AO, Cotter E and Culloty SC (2012) A previously undescribed ostreid herpes virus 1 (OsHV-1) genotype detected in the pacific oyster, Crassostrea gigas, in Ireland. Parasitology 139, 1526–1532. [DOI] [PubMed] [Google Scholar]
- Lynch SA, Dillane E, Carlsson J and Culloty SC (2013) Development and assessment of a sensitive and cost-effective polymerase chain reaction to detect ostreid herpesvirus 1 and variants. Journal of Shellfish Research 32, 657–664. [Google Scholar]
- Lynch SA, Morgan E, Carlsson J, Mackenzie C, Wooton EC, Rowley AF, Malham S and Culloty SC (2014) The health status of mussels, Mytilus spp., in Ireland and Wales with the molecular identification of a previously undescribed haplosporidian. Journal of Invertebrate Pathology 118, 59–65. [DOI] [PubMed] [Google Scholar]
- Lynch SA, Coghlan A, O'Leary B, Morgan E and Culloty SC (2020a) Northward establishment of the Mediterranean mussel Mytilus galloprovincialis limited by changing climate. Biological Invasions 22, 2725–2736. [Google Scholar]
- Lynch SA, Lepee-Rivero S, Kelly R, Quinn E, Coghlan A, Bookelaar B, Morgan E, Finarelli JA, Carlsson J and Culloty SC (2020b) Detection of haplosporidian protistan parasites supports an increase to their known diversity, geographic range and bivalve host specificity. Parasitology 147, 584–592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahony KE, Lynch SA, Egerton S, Cabral S, de Montaudouin X, Fitch A, Magalhaes L, Rocroy M and Culloty SC (2020) Mobilisation of data to stakeholder communities. Bridging the research-practice gap using a commercial shellfish species model. PLoS ONE 15, e0238446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malham SK, Hutchinson TH and Longshaw M (2012) A review of the biology of European cockles (Cerastoderma spp.). Journal of the Marine Biological Association of the United Kingdom 92, 1563–1577. [Google Scholar]
- McCleary S and Henshilwood K (2015) Novel quantitative TaqMan (R) MGB real-time PCR for sensitive detection of Vibrio aestuarianus in Crassostrea gigas. Diseases of Aquatic Organisms 114, 239–248. [DOI] [PubMed] [Google Scholar]
- Molloy DP, Giamberini L, Stokes NA, Burreson EM and Ovcharenko MA (2012) Haplosporidium raabei n. sp (Haplosporidia): a parasite of zebra mussels, Dreissena polymorpha (Pallas, 1771). Parasitology 139, 463–477. [DOI] [PubMed] [Google Scholar]
- Morgan E, O'Riordan RM, Kelly TC and Culloty SC (2012) Influence of disseminated neoplasia, trematode infections and gametogenesis on surfacing and mortality in the cockle Cerastoderma edule. Diseases of Aquatic Organisms 98, 73–84. [DOI] [PubMed] [Google Scholar]
- Morgan E, O'Riordan RM and Culloty SC (2013) Climate change impacts on potential recruitment in an ecosystem engineer. Ecology and Evolution 3, 581–594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neogi SB, Lara R, Alam M, Harder J, Yamasaki S and Colwell RR (2018) Environmental and hydroclimatic factors influencing Vibrio populations in the estuarine zone of the Bengal delta. Environmental Monitoring and Assessment 190, 565. [DOI] [PubMed] [Google Scholar]
- Notaro DA, Culloty SC and Lynch SA (2021) A pilot study investigating the potential of antimicrobial photodynamic therapy (aPDT) to control Vibrio spp. development in microalgae and seawater. Aquaculture International 29, 355–372. [Google Scholar]
- Nowakowska J and Oliver JD (2013) Resistance to environmental stresses by Vibrio vulnificus in the viable but nonculturable state. FEMS Microbiology Ecology 84, 213–222. [DOI] [PubMed] [Google Scholar]
- Ottway B, Parker M, McGrath D and Crowley M (1979) Observations on a bloom of Gyrodinium aureolum (Hulbert) on the south coast of Ireland, summer 1976, associated with mortalities of littoral and sublittoral organisms. Irish Fisheries Investigations Series B 18, 3–10. [Google Scholar]
- Paillard C, Allam B and Oubella R (2004) Effect of temperature on defense parameters in Manila clam Ruditapes philippinarum challenged with Vibrio tapetis. Diseases of Aquatic Organisms 59, 249–262. [DOI] [PubMed] [Google Scholar]
- Pepin JF, Riou A and Renault T (2008) Rapid and sensitive detection of ostreid herpesvirus 1 in oyster samples by real-time PCR. Journal of Virological Methods 149, 269–276. [DOI] [PubMed] [Google Scholar]
- Petton B, Bruto M, James A, Labreuche Y, Alunno-Bruscia M and Le Roux F (2015) Crassostrea gigas mortality in France: the usual suspect, a herpes virus, may not be the killer in this polymicrobial opportunistic disease. Frontiers in Microbiology 6, 686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petton B, de Lorgeril J, Mitta G, Daigle G, Pernet F and Alunno-Bruscia M (2019) Fine-scale temporal dynamics of herpes virus and vibrios in seawater during a polymicrobial infection in the Pacific oyster Crassostrea gigas. Diseases of Aquatic Organisms 135, 97–106. [DOI] [PubMed] [Google Scholar]
- Philippart CJM, Anadon R, Danovaro R, Dippner JW, Drinkwater KF, Hawkins SJ, Oguz T, O'Sullivan G and Reid PC (2011) Impacts of climate change on European marine ecosystems: observations, expectations and indicators. Journal of Experimental Marine Biology and Ecology 400, 52–69. [Google Scholar]
- Prado-Alvarez M, Darmody G, Hutton S, O'Reilly A, Lynch SA and Culloty SC (2016) Occurrence of OsHV-1 in Crassostrea gigas cultured in Ireland during an exceptionally warm summer. Selection of less susceptible oysters. Frontiers in Physiology 7, 492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramilo A, Abollo E, Villalba A and Carballal MJ (2018) A Minchinia mercenariae-like parasite infects cockles Cerastoderma edule in Galicia (NW Spain). Journal of Fish Diseases 41, 41–48. [DOI] [PubMed] [Google Scholar]
- Rehnstam-Holm A, Godhe A, Harnstrom K, Raghunath P, Saravanan V, Collin B, Karunasagar I and Karunasagar I (2010) Association between phytoplankton and Vibrio spp. Along the southwest coast of India: a mesocosm experiment. Aquatic Microbial Ecology 58, 127–139. [Google Scholar]
- Renault T and Novoa B (2004) Viruses infecting bivalve molluscs. Aquatic Living Resources 17, 397–409. [Google Scholar]
- Renault T, Stokes N, Chollet B, Cochennec N, Berthe F, Gerard A and Burreson E (2000) Haplosporidiosis in the pacific oyster Crassostrea gigas from the French Atlantic coast. Diseases of Aquatic Organisms 42, 207–214. [DOI] [PubMed] [Google Scholar]
- Renault T, Bouquet AL, Maurice J, Lupo C and Blachier P (2014) Ostreid herpesvirus 1 infection among Pacific oyster (Crassostrea gigas) spat: relevance of water temperature to virus replication and circulation prior to the onset of mortality. Applied and Environmental Microbiology 80, 5419–5426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rowley AF, Cross ME, Culloty SC, Lynch SA, Mackenzie CL, Morgan E, O'Riordan RM, Robins PE, Smith AL, Thrupp TJ, Vogan CL, Wootton EC and Malham SK (2014) The potential impact of climate change on the infectious diseases of commercially important shellfish populations in the Irish Sea-a review. ICES Journal of Marine Science 71, 741–759. [Google Scholar]
- Salvesen I, Reitan K, Skjermo J and Oie G (2000) Microbial environments in marine larviculture: impacts of algal growth rates on the bacterial load in six microalgae. Aquaculture International 8, 275–287. [Google Scholar]
- Shaw BL and Battle HI (1957) The gross microscopic anatomy of the digestive tract of Crassostrea virginica (Gmelin). Canadian Journal of Zoology 35, 325–346. [Google Scholar]
- Suttle CA (2007) Marine viruses – major players in the global ecosystem. Nature Reviews Microbiology 5, 801–812. [DOI] [PubMed] [Google Scholar]
- Thomas F, Renaud F and Poulin R (1998) Exploitation of manipulators: ‘hitch-hiking’ as a parasite transmission strategy. Animal Behaviour 56, 199–206. [DOI] [PubMed] [Google Scholar]
- Vaumourin E, Vourc'h G, Telfer S, Lambin X, Salih D, Seitzer U, Morand S, Charbonnel N, Vayssier-Taussat M and Gasqui P (2014) To be or not to be associated: power study of four statistical modeling approaches to identify parasite associations in cross-sectional studies. Frontiers in Cellular and Infection Microbiology 4, 62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaumourin E, Vourc'h G, Gasqui P and Vayssier-Taussat M (2015) The importance of multiparasitism: examining the consequences of co-infections for human and animal health. Parasites & Vectors 8, 545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vezzulli L, Previati M, Pruzzo C, Marchese A, Bourne DG, Cerrano C and VibrioSea Consortium (2010) Vibrio infections triggering mass mortality events in a warming Mediterranean Sea. Environmental Microbiology 12, 2007–2019. [DOI] [PubMed] [Google Scholar]
- Villalba A, Iglesias D, Ramilo A, Darriba S, Parada JM, No E, Abollo E, Molares J and Carballal MJ (2014) Cockle Cerastoderma edule fishery collapse in the Ria de Arousa (Galicia, NW Spain) associated with the protistan parasite Marteilia cochillia. Diseases of Aquatic Organisms 109, 55–80. [DOI] [PubMed] [Google Scholar]
- Walsh P, Metzger D and Higuchi R (1991) Chelex-100 as a medium for simple extraction of DNA for PCR-based typing from forensic material. BioTechniques 10, 506–513. [PubMed] [Google Scholar]
- Wiltshire KH, Malzahn AM, Wirtz K, Greve W, Janisch S, Mangelsdorf P, Manly BFJ and Boersma M (2008) Resilience of North Sea phytoplankton spring bloom dynamics: an analysis of long-term data at Helgoland Roads. Limnology and Oceanography 53, 1294–1302. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
For supplementary material accompanying this paper visit https://doi.org/10.1017/S0031182021001396.
click here to view supplementary material