Abstract
Background:
In March 2020, the Indian Council of Medical Research (ICMR) issued guidelines that all patients presenting with severe acute respiratory infections (SARI) should be investigated for coronavirus disease 2019 (COVID-19). Following the same protocol, in our institute, all patients with SARI were transferred to the COVID-19 suspect intensive care unit (ICU) and investigated for COVID-19.
Methods:
This study was planned to examine the demographical, clinical features, and outcomes of the first 500 suspected patients of COVID-19 with SARI admitted in the COVID-19 suspect ICU at a tertiary care center. Between March 7 and July 20, 2020, 500 patients were admitted to the COVID-19 suspect ICU. We analyzed the demographical, clinical features, and outcomes between COVID-19 positive and negative SARI cases. The records of all the patients were reviewed until July 31, 2020.
Results:
Of the 500 suspected patients admitted to the hospital, 88 patients showed positive results for COVID-19 by reverse transcription-polymerase chain reaction (RT-PCR) of the nasopharyngeal swabs. The mean age in the positive group was higher (55.31 ± 16.16 years) than in the negative group (40.46 ± 17.49 years) (P < 0.001). Forty-seven (53.4%) of these patients in the COVID-19 positive group and 217 (52.7%) from the negative group suffered from previously known comorbidities. The common symptoms included fever, cough, sore throat, and dyspnea. Eighty-five (20.6%) patients died in the COVID-19 negative group, and 30 (34.1%) died in the COVID-19 positive group (P = 0.006). Deaths among the COVID-19 positive group had a significantly higher age than deaths in the COVID-19 negative group (P < 0.001). Among the patients who died with positive COVID-19 status had substantially higher neutrophilia and lymphopenia (P < 0.001). X-ray chest abnormalities were almost three times more likely in COVID-19 deaths (P < 0.001).
Conclusion:
In the present article, 17.6% of SARI were due to COVID-19 infection with significantly higher mortality (34.1%) in COVID-19 positive patients with SARI. Although all patients presenting as SARI have considerable mortality rates, the COVID-19-associated SARI cases thus had an almost one-third risk of mortality.
Keywords: Coronavirus Disease 2019, SARS-CoV2, severe acute respiratory infection
Introduction
Coronavirus Disease 2019 (COVID-19) is a contagious illness primarily affecting the respiratory tract. This disease was not known before the Wuhan outbreak in December 2019 China. This is the third coronavirus attack, the previous two being the 2003 severe acute respiratory syndrome-related coronavirus (SARS-CoV) infection and the 2012 Middle East respiratory coronavirus (MERS-CoV) epidemic.[1] Given its worldwide distribution, the World Health Organization (WHO) declared this disease a pandemic in March 2020.
Symptomatic COVID-19 infection is characterized primarily by fever, cough, dyspnea, and infiltrations in the chest.[2,3] Although many of the observed infections are not dangerous, about 15–20 percent of confirmed COVID-19 patients may have a severe illness, including respiratory failure, shock, and multiple organ dysfunction that require admission to the critical care unit.[4,5]
As per the WHO, severe acute respiratory infection (SARI) is described as an acute respiratory infection with fever or temperature ≥38°C and cough, onset since the last 10 days, and necessitating admission to the hospital.[6] In the era of the current pandemic, all SARI patients need to be viewed with a high index of suspicion of COVID-19. Taking all COVID-19 patients thus provides a sound denominator to study the clinical pattern in all COVID-19 and non-COVID-19 patients.
In March 2020, the Indian Council of Medical Research (ICMR) issued guidelines to investigate all the patients presenting with SARI for COVID-19.[7] In compliance with ICMR protocols at our institute, any patient presenting with SARI from March 7, 2020, was admitted first in the COVID-19 suspect intensive care unit (ICU) and tested for COVID-19. After confirmation of his COVID-19 status, the patient is shifted to a designated area accordingly. If the patient remains stable (not require high oxygen (more than 15 L/min of oxygen to maintain SpO2 > 94%) with a positive COVID-19 report, he is moved to COVID-19 ward; in case, the patient deteriorates (requirement of high oxygen or ventilator support) with a positive COVID-19 report, he is shifted to COVID-19 critical care unit; and with a negative report to the adult intensive care unit [Figure 1].
Figure 1.
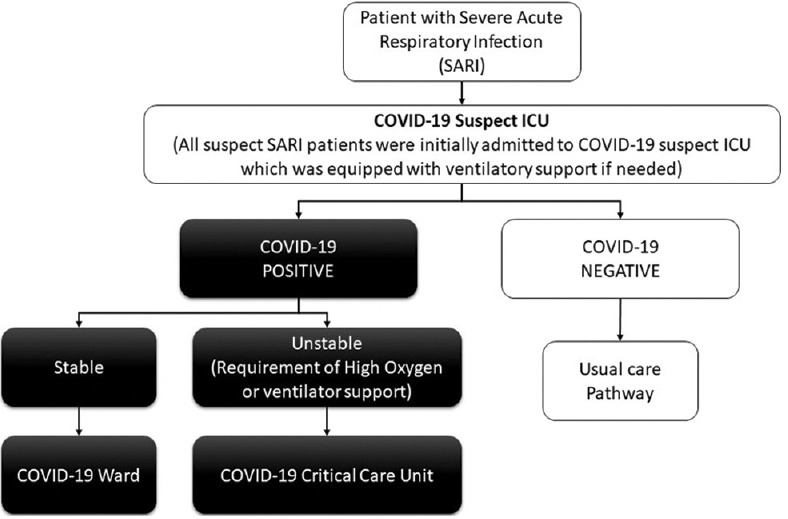
Pathway of care of SARI patients during the COVID-19 Pandemic
Studies have mentioned the clinical features of pneumonia incurred by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).[8,9,10,11,12] This is the first report from western Rajasthan, a dry-arid region, describing the clinicodemographic and management profile of SARI patients in the initial days of COVID-19. Furthermore, this article also compared the mortality between COVID-19 SARI versus non-COVID-19 SARI cases. The plethora of clinical features and laboratory investigations described here give an important insight into the early indicators of disease severity. These should be on the radar of not only the intensivists but also primary care physicians to timely diagnose a potential case of SARI and channelize towards appropriate management.
The objective of the present study was to examine the clinical and laboratory as well as comorbidities and outcomes of the SARI patients admitted in the COVID-19 suspect ICU.
Materials and Methods
This was a hospital-based cross-sectional study done after approval from the institutional ethical committee (AIIMS/IEC/2020-21/2053). Five hundred suspected patients of COVID-19 with SARI were admitted in the COVID-19 suspect ICU from March 7 to July 20, 2020, at a tertiary care center in India. The records of all the patients were reviewed until July 31, 2020.
The data of the first 500 patients with SARI admitted in COVID-19 suspect ICU during this pandemic were analyzed. The information included demographic data, known comorbidities, clinical presentation (signs and symptoms), laboratory investigations, X-ray-chest, mechanical ventilator requirements, and outcomes.
All the suspected COVID-19 patients with SARI enrolled in this study were tested with reverse transcription-polymerase chain reaction (RT-PCR) of the nasopharyngeal swabs. The specimen was obtained for the SARS-CoV-2 RT-PCR examination on the day of admission. The nasopharyngeal swabs were placed into a vial that contained a viral transport medium. The specimens were stored and transported at 4°C to the laboratory. In the present study, the RT-PCR test was done only on admission.
The laboratory investigations collected were hemoglobin, total leukocyte counts, including neutrophil and lymphocyte percentage, platelet counts, serum urea, serum creatinine, serum procalcitonin, and serum D-dimer. Chest radiography was also performed for all inpatients.
Statistical analysis
The data were inserted using Microsoft Excel, and all statistical analyses were done utilizing Windows IBM SPSS statistics software version 21.0.[13] Descriptive analyses were performed and presented as numbers and percentages for the study participants. Comparisons between nominal data were made using the Chi-square test, and between continuous data were done using an independent sample t-test. Ordinal data were described using the median and interquartile range and analyzed using the Mann-Whitney U test. A P value smaller than 0.05 was the cutoff for statistical significance.
Results
In the present study, 500 suspected patients of COVID-19 with SARI admitted in the COVID-19 suspect ICU were enrolled in the study. Among them, 318 (63.6%) were male, and 182 (36.4%) were female. The RT-PCR test for COVID-19 revealed 88 (17.6%) patients positive, and 412 were negative. The mean age in the positive group was 55.31 ± 16.16 years, and in the negative group was 40.46 ± 17.49 years (P < 0.001). Out of the 500 patients with SARI, 264 (52.8%) had comorbidities. Hypertension [n = 127 (25.4%)] and diabetes [n = 79 (15.8%)] were the top two comorbidities in these patients. Others were respiratory (n = 45), neurological (n = 39), renal (n = 32), cardiac (n = 20), and various types of cancers (n = 21). However, there was no statistically significant association of these comorbidities with COVID-19 positive patients [Table 1].
Table 1.
Demographical and clinical characteristics of SARI patients
| Variables | COVID-19 Status | P | |
|---|---|---|---|
|
| |||
| Positive (n=88) | Negative (n=412) | ||
| Age (Years in mean±SD) | 55.31±16.16 | 40.46±17.49 | <0.001^* |
| Males | 64 (72.7%) | 254 (61.7%) | 0.849 |
| Fever | 69 (78.4%) | 327 (79.4%) | 0.840 |
| Cough/Sore Throat | 27 (30.7%) | 135 (32.8%) | 0.704 |
| Dyspnea | 71 (80.7%) | 331 (80.3%) | 0.942 |
| Chest pain | 2 (2.3%) | 6 (1.5%) | 0.580 |
| Comorbidities | 47 (53.4%) | 217 (52.7%) | 0.9 |
| Diabetes | 11 (12.5%) | 68 (16.5%) | 0.35 |
| Hypertension | 28 (31.8%) | 99 (24.0%) | 0.128 |
| Cardiac Disorders (CAD//RHD/CHF/Pul HTN) | 3 (3.4%) | 17 (4.1%) | 0.755 |
| Respiratory Disorders (COPD/TB/ILD/BA) | 4 (4.5%) | 41 (10.0%) | 0.108 |
| Neurological Disorder (Stroke/Seizure/Parkinsonism SDH/Spina Bifida/Spine fracture) | 4 (4.5%) | 35 (8.5%) | 0.210 |
| Renal Disease | 5 (5.7%) | 27 (6.6%) | 0.762 |
| Cancer (Lymphomas/MM/ALL) | 1 (1.1%) | 20 (4.9%) | 0.114 |
| Need for mechanical ventilation | 20 (22.7%) | 66 (16.0%) | 0.156 |
| Death | 30 (34.1%) | 85 (20.6%) | 0.006* |
Figures in brackets represent column-wise percentages. [SARI=Severe Acute Respiratory Infection, COVID-19=Coronavirus disease 2019, SD=standard deviation, CAD=coronary artery disease, RHD- rheumatic heart disease, CHF=congestive heart failure, Pul HTN-pulmonary hypertension, COPD- chronic obstructive pulmonary disease, TB- tuberculosis, ILD- interstitial lung disease, bronchial asthma, MM=multiple myeloma, ALL=acute lymphocytic leukemia]. All P by Chi-Square test (except Age variable, which is derived by t-test)
The common symptoms included dyspnea [71 (80.7%) in positive group; 331 (80.3%) in negative group], fever [69 (78.4%) in positive group; 327 (79.4%) in negative group], cough/sore throat [27 (30.7%) in positive group; 135 (32.8%) in negative group)] at the time of admission [Table 1]. Less common symptoms were chest pain, headache, vomiting, loose stools, abdominal pain, nasal discharge, anosmia, and altered sensorium.
The mean total leukocyte count (TLC) in the positive group was 11.6 ± 4.5 × 103 /μL, and in the negative group was 13.3 ± 3.0 × 103 /μL. Most patients had neutrophilia and lymphopenia in both groups. The percentage of neutrophils in the positive group was (mean ± SD) 79.12 ± 11.99, and in the negative group was 75.64 ± 14.81 (P = 0.056). The percentage of lymphocytes was 13.81 ± 8.60 in the positive group and 15.62 ± 12.52 in the negative group (P = 0.231). In positive group, 66 (94.3%) and in negative group, 180 (94.8%) patients had raised D-dimer ≥0.5 (μg/mL) (P = 0.505) [Table 2]. The median procalcitonin (normal value < 0.02 ng/mL) at the time of admission in the positive group was 0.23 [0.08–1.23] ng/mL, and in the negative group was 0.67 [0.10–4.85] ng/mL (P = 0.035).
Table 2.
Laboratory parameters of SARI patients
| Variables | COVID-19 Status | P | |
|---|---|---|---|
|
| |||
| Positive (n=88) Mean±SD | Negative (n=412) Mean±SD | ||
| Hemoglobin (gm/dL) | 11.50±2.50 | 11.12±6.99 | 0.636 |
| Total leukocyte counts (×103/µL) Normal 4.0-11.0 ×103/µL) | 11.67±6.21 | 13.49±12.24 | 0.208 |
| Neutrophils percentage (Normal 40%-60%) | 79.12±11.99 | 75.64±14.81 | 0.056 |
| Lymphocytes percentage (Normal 25%-35%) | 13.81±8.60 | 15.62±12.52 | 0.231 |
| Platelet counts (× 109/L) (Normal 150-400 ×109/L) | 269.87±146.0 | 266.42±158.71 | 0.865 |
| d dimer ≥0.5 (μg/mL) (Normal Range <0.5 μg/mL) | 66 (94.3%) | 180 (94.8%) | 0.505^ |
| Urea (mg/dL) | 44.97±34.10 | 57.88±61.88 | 0.077 |
| Creatinine (mg/dL) | 1.56±0.94 | 1.84±2.50 | 0.089 |
SARI=Severe Acute Respiratory Infection, COVID-19=Coronavirus disease 2019, SD=Standard deviation, ^P calculated by Chi-Square test, all other P values by independent sample t-test
Sixty-six (75.9%) patients in the positive group showed bilateral chest infiltrates, and 45 (11%) had abnormal X-ray findings in the negative group (P < 0.001). The various patterns on chest X-rays of positive COVID-19 patients were bilateral patchy nodular or interstitial shadows, bilateral peripheral and basal nodular interstitial infiltration, bilateral peripheral interstitial infiltration, and bilateral basal nodular-interstitial infiltration [Figure 2].
Figure 2.
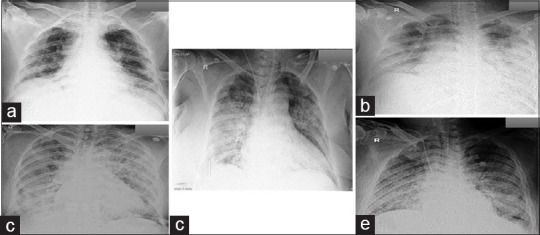
Various patterns of chest X-rays found in COVID-19 positive patients (a), (b) bilateral basal nodular-interstitial infiltration (c) bilateral patchy nodular-interstitial shadows (d) bilateral peripheral interstitial infiltration (e) bilateral peripheral and basal nodular interstitial infiltration
Twenty (22.7%) patients in a positive group and 66 (16.0%) in the negative group required invasive mechanical ventilator support in COVID-19 suspect ICU (P = 0.318). Three (3.4%) patients in the positive group and 15 (3.6%) in the negative group were on high flow nasal oxygen (HFNO) support at the time of admission. The rest of the patients required no or minimal oxygen support in both groups at admission. There was notably greater mortality in COVID-19 positive patients (34.1%) relative to negative patients (20.6%) (P = 0.006). On adjusting the age differences, death continued to be associated with COVID-19 positive status.[aOR = 1.93, 95% CI = 1.14-3.29] [Figure 3]. The cause of mortality in these patients ranged from acute respiratory distress syndrome (ARDS), multiple organ dysfunction syndrome (MODS), and refractory shock.
Figure 3.
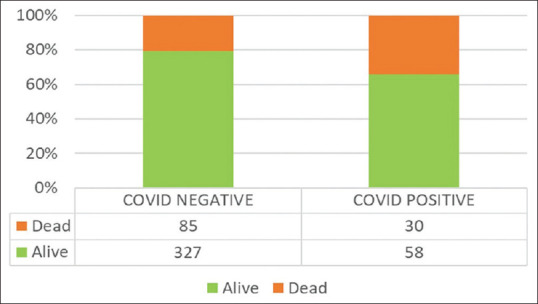
Mortality in COVID-19 negative and COVID-19 positive patients presented with SARI
Analysis of all deaths in SARI patients (n = 115)
Of the 500 SARI patients, 115 died. Of these, the COVID-19 positive patients (n = 30) had a significantly higher age as compared to the COVID-19 negative (n = 85) patients (P < 0.001). There were no differences with respect to gender, presenting symptoms, or any comorbidities. X-ray chest abnormalities were almost three times more likely in COVID-19 deaths (P < 0.001). Among the laboratory parameters, COVID-19 positive patients had a significantly higher median neutrophils percentage {88.2 [85.5–90.5] in COVID-19 positive patients who died; 78.5 [68.5–87.6], P = 0.003} in COVID-19 negative patients who died}, lower median lymphocyte percentage {6.7 [4.8–10.2] in COVID-19 positive patients who died; 11.4 [6.2–19.9], P = 0.017} in COVID-19 negative patients who died} and a lower median procalcitonin value {0.5 [0.1–1.3] ng/mL in COVID-19 positive patients who died; 2.0 [0.4–19.7] ng/mL, P = 0.017} in COVID-19 negative patients who died }.
Analysis of the COVID-19 positive patients (n = 88)
Of the 500 SARI patients, 88 were positive COVID-19 patients, of whom 30 died. There were no significant differences in age (P = 0.204) or gender (P = 0.359) or the presenting symptoms between the positive COVID-19 patients who had died compared to those who were alive. Similarly, none of the comorbidities evaluated had any significance with the mortality of COVID-19 patients. Among the laboratory parameters, dead COVID-19 patients were found to have a higher median total leucocyte count {16.6 [11.8–18.0] ×103 /μL in patients who died; 9.1 [6.2–13.0](×103 /μL, P < 0.001 in alive patients} with significantly higher median neutrophils percentage {88.2 [85.5–90.5] in patients who died; 77.0 [67.7–85.6], P < 0.001 in alive patients} and lower median lymphocytes percentage {15.5 [9.6–21.5] in patients who died; 6.7 [4.8–10.2], P < 0.001 in alive patients} than alive COVID-19 patients.
Analysis of SARI patients requiring ventilator support or HFNO (n = 104)
Of the 500 SARI patients, 104 required ventilator support or HFNO at admission [Figure 4]. There were no significant differences in age or the presenting symptoms between the patients who required ventilator support compared to those who did not need it during admission. Patients who required ventilator support had more comorbidities (65.7%) in comparison to patients who did not (49.2%) require ventilator support at admission (P = 0.002). There were more deaths in patients who required ventilator support (34.3%) in comparison to those who did not need ventilator support (19.9%) at admission (P = 0.002).
Figure 4.
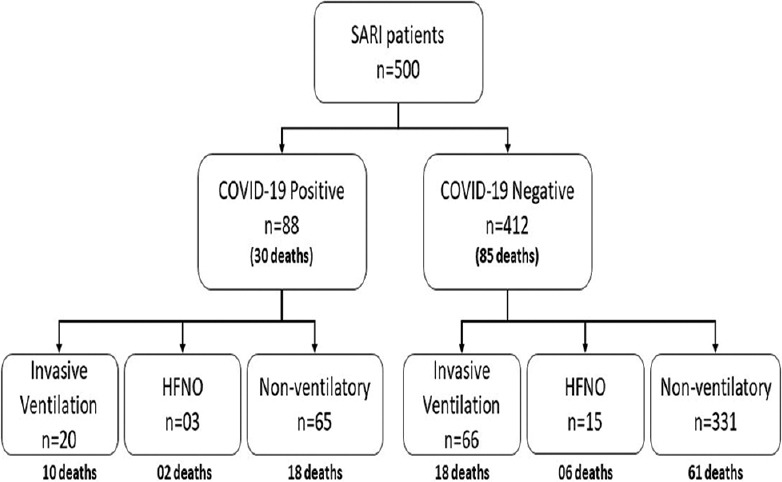
Requirement of ventilator support and high flow nasal oxygen (HFNO) support in patients presented with SARI at the time of admission
Discussion
In the present study, the RT-PCR test revealed 88 SARI patients positive for COVID-19. The previous survey, conducted by the ICMR in India of SARI patients in March 2020, showed COVID-19 positivity of 1.8%.[14] Our study has shown that this number has dramatically increased to 17.6 percent. It may be due to the progression of the transmission of the disease. In a retrospective case series of 82 patients with SARI, Aggarwal, et al.[15] found 39% positivity for COVID-19. The higher rate of positivity in their case series may be due to their sampling for COVID-19 on the day of admission, and again on the day 5 of admission in case, the first sample was negative. In the present study, the RT-PCR test was done only on admission.
The patients diagnosed as COVID-19 positive were older than COVID-19 negative patients (P < 0.001), which was similar to earlier studies.[15,16] Of the total dead SARI patients, the COVID-19 positive patients had a significantly higher age than the COVID-19 negative patients (P < 0.001).
The current study found that patients with various comorbidities had SARI and required hospitalizations. Hypertension (25.4%) and diabetes (15.8%) were the top two comorbidities in patients presenting with SARI. This was similar to previous studies.[15,16,17,18] There was no significant difference between COVID-19 positive and negative groups for these comorbidities. However, patients who required ventilator support had more comorbidities (65.7%) in comparison to patients who did not (49.2%) require ventilator support at admission (P = 0.002). It can be explained by higher chances of progression to multiple organ dysfunction syndrome because of these comorbidities, which necessitated ventilator requirements in these patients.[19,20,21,22] Yang et al.,[16] in their study on 200 patients with COVID-19, found a mortality rate in the ICU as 51.7%. Agarwal et al.[9] observed COVID-positive subject's death rate of 28.4% during the study of 281 SARI patients. Suresh et al.,[21] in their study on 116 COVID-19 patients, concluded that lower SpO2 and Glasgow Coma Scale (GCS) were the main parameters that showed a significant association with mortality and the requirement for mechanical ventilation. Similarly, we also observed 34.1% of mortality in COVID-19 positive patients with SARI. A total of 104 out of 500 patients required ventilator or HFNO support. This pattern of utilization can be used for resource planning and allocation.
Like other studies,[8,14,15] most of our patients had cough, fever, shortness of breath, and bilateral chest infiltrates in chest X-rays. Most of our patients had lymphopenia, which was similar to the finding in the studies done by Bhandari et al.[23] and Zaboli et al.[24] It has been suggested that lymphocytes may be the principal target of SARS-CoV-2 and can be linked to disease progression and mortality. Neutrophilia and lymphopenia were significantly higher in COVID-19 positive patients who died. This result can be explained by the fact that neutrophils are triggered by inflammatory factors associated with viruses, such as interleukin-6, interleukin-8, and tumor necrosis factor.[25] Human immunological response from a viral infection, however, relies primarily on lymphocytes. Systematic inflammation depresses cellular immunity by reducing CD4 + T lymphocytes. Thus, virus-triggered inflammation causes neutrophilia and lymphopenia.[26]
In a low–middle-income country like India, resources for the management of COVID-19 patients are limited. These barriers may occur at the infrastructure level (limited number of isolation beds), testing level (a smaller number of certified test laboratories, particularly in suburban and regional hospitals.), treatment level (inadequate ICU ventilators, oxygen supply, medications, personal protective equipment (PPE), and staff), information level (inconsistency and ambiguity about testing, triage, and management), and transport-level (insufficient transport choices for sufferers). These shortcomings may be overcome by using existing resources optimally. Some examples are utilizing exhaust fans to transform normal pressure rooms to negative pressure rooms to improve isolation, open field hospitals in large public spaces, focused testing on symptomatic patients, enrolling paramedical personnel, and re-use some forms of PPE after disinfection, etc.[27]
There are a few limitations to this study. First, this was a single-center study. Multicenter studies with a bigger sample size will help better understand the clinical and outcome profiles of COVID-19 patients. Secondly, the RT-PCR test was used to diagnose COVID-19 in nasopharyngeal swabs. This test, however, has a false negative rate of 38%, which could have missed the diagnosis of COVID-19 in some patients.[28] We did not use computer tomography (CT) of the chest for these patients. Patients with PCR negative could be considered to be COVID-19 based on the CT.[29,30]
Conclusions
In conclusion, of the 500 suspected patients of coronavirus disease 2019 with severe acute respiratory infection admitted at our hospital, the mortality of COVID-19 positive patients was considerably high.
Key points
This study examined the first 500 suspected patients of COVID-19 with SARI admitted at our center. Out of them, 104 required ventilator support or HFNO at admission. Of the 500 SARI patients, 88 patients showed positive results for COVID-19 by reverse transcription-polymerase chain reaction (RT-PCR). Eighty-five (20.6%) patients died in the COVID-19 negative group, and 30 (34.1%) died in the COVID-19 positive group (P = 0.006). Although all patients presenting as SARI have considerable mortality rates, the COVID-19 associated SARI cases thus had almost one-third risk of mortality.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- 1.Sahu KK, Mishra AK, Lal A. Comprehensive update on the current outbreak of novel coronavirus infection (2019-nCoV) Ann Transl Med. 2020;8:393. doi: 10.21037/atm.2020.02.92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gao Y, Shi C, Chen Y, Shi P, Liu J, Xiao Y, et al. A cluster of the Corona Virus Disease 2019 caused by incubation period transmission in Wuxi, China. J Infect. 2020;80:666–70. doi: 10.1016/j.jinf.2020.03.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sahu KK, Mishra AK, Lal A. COVID-2019: Update on epidemiology, disease spread, and management. Monaldi Arch Chest Dis. 2020:90. doi: 10.4081/monaldi.2020.1292. doi: 10.4081/monaldi. 2020.1292. [DOI] [PubMed] [Google Scholar]
- 4.Sahu KK, Mishra AK, Lal A. Trajectory of the COVID-19 pandemic: Chasing a moving target. Ann Transl Med. 2020;8:694. doi: 10.21037/atm-20-2793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chen T, Wu D, Chen H, Yan W, Yang D, Chen G, et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: A retrospective study. BMJ. 2020;368:m1091. doi: 10.1136/bmj.m1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Government of India Ministry of Health and Family Welfare Directorate General of Health Services (EMR Division) Guidelines on Clinical Management of COVID-19. Available from: https://www.mohfw.gov.in/pdf/GuidelinesonClinicalManagementofCOVID1912020.pdf .
- 7.Indian Council of Medical Research. Revised Strategy of COVID19 testing in India (Version 3, dated 20/03/2020) https://www.icmr.gov.in/pdf/covid/strategy/2020-03-20_covid19_test_v3.pdf. [Google Scholar]
- 8.Gupta N, Agrawal S, Ish P, Mishra S, Gaind R, Usha G, et al. Clinical and epidemiologic profile of the initial COVID-19 patients at a tertiary care center in India. Monaldi Arch Chest Dis. 2020:90. doi: 10.4081/monaldi.2020.1294. doi: 10.4081/monaldi. 2020.1294. [DOI] [PubMed] [Google Scholar]
- 9.Agarwal N, Biswas B, Lohani P. Epidemiological determinants of COVID-19 infection and mortality: A study among patients presenting with severe acute respiratory illness during the pandemic in Bihar, India. Niger Postgrad Med J. 2020;27:293–301. doi: 10.4103/npmj.npmj_301_20. [DOI] [PubMed] [Google Scholar]
- 10.Murthy S, Archambault PM, Atique A, Carrier FM, Cheng MP, Codan C, et al. Characteristics and outcomes of patients with COVID-19 admitted to hospital and intensive care in the first phase of the pandemic in Canada: A national cohort study. CMAJ. 2021;9:E181–8. doi: 10.9778/cmajo.20200250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jalili M, Payandemehr P, Saghaei A, Sari HN, Safikhani H, Kolivand P. Characteristics and mortality of hospitalized patients with COVID-19 in Iran: A national retrospective cohort study. Ann Intern Med. 2021;174:125–7. doi: 10.7326/M20-2911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sonkar SK, Kumar S, Gupta KK, Chaudhary SC, Kumar V, Sawlani KK, et al. A learning experience of pandemic COVID-19 management at our medical institute. J Family Med Prim Care. 2020;25(9):4270–6. doi: 10.4103/jfmpc.jfmpc_836_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.IBM Corp. Released 2012. IB M SPSS Statistics for Windows, Version 21.0. Armonk, NY: IBM Corp; [Google Scholar]
- 14.Gupta N, Praharaj I, Bhatnagar T, Vivian Thangaraj JW, Giri S, Chauhan H, et al. Severe acute respiratory illness surveillance for coronavirus disease 2019, India, 2020. Indian J Med Res. 2020;151:236–40. doi: 10.4103/ijmr.IJMR_1035_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Aggarwal A, Shrivastava A, Kumar A, Ali A. Clinical and epidemiological features of SARS-CoV-2 patients in SARI ward of a tertiary care centre in New Delhi. J Assoc Physicians India. 2020;68:19–26. [PubMed] [Google Scholar]
- 16.Yang L, Liu J, Zhang R, Li M, Li Z, Zhou X, et al. Epidemiological and clinical features of 200 hospitalized patients with coronavirus disease 2019 outside Wuhan, China: A descriptive study. J Clin Virol. 2020;129:104475. doi: 10.1016/j.jcv.2020.104475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang JJ, Cao YY, Tan G, Dong X, Wang BC, Lin J, et al. Clinical, radiological, and laboratory characteristics and risk factors for severity and mortality of 289 hospitalized COVID-19 patients. Allergy. 2021;76:533–50. doi: 10.1111/all.14496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li J, He X, Yuan Yuan, Zhang W, Li X, Zhang Y, et al. Meta-analysis investigating the relationship between clinical features, outcomes, and severity of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pneumonia. Am J Infect Control. 2021;49:82–9. doi: 10.1016/j.ajic.2020.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mishra AK, Sahu KK, George AA, Sargent J, Lal A. Cerebrovascular events in COVID-19 patients. Monaldi Arch Chest Dis. 2020;90:1341. doi: 10.4081/monaldi.2020.1341. [DOI] [PubMed] [Google Scholar]
- 20.Mishra AK, Sahu KK, George AA, Lal A. A review of cardiac manifestations and predictors of outcome in patients with COVID-19. Heart Lung. 2020;49:848–52. doi: 10.1016/j.hrtlng.2020.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Suresh S, Tiwari A, Mathew R, Bhaskararayuni J, Sahu AK, Aggarwal P, et al. Predictors of mortality and the need of mechanical ventilation in confirmed COVID-19 patients presenting to the emergency department in North India. J Family Med Prim Care. 2021;10:542–9. doi: 10.4103/jfmpc.jfmpc_1775_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Douville NJ, Douville CB, Mentz G, Mathis MR, Pancaro C, Tremper KK, et al. Clinically applicable approach for predicting mechanical ventilation in patients with COVID-19. Br J Anaesth. 2021;126:578–89. doi: 10.1016/j.bja.2020.11.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bhandari S, Bhargava A, Sharma S, Keshwani P, Sharma R, Banerjee S. Clinical profile of Covid-19 infected patients admitted in a tertiary care hospital in North India. J Assoc Physicians India. 2020;18:13–7. [PubMed] [Google Scholar]
- 24.Zaboli E, Majidi H, Alizadeh-Navaei R, Hedayatizadeh-Omran A, Asgarian-Omran H, Vahedi Larijani L, et al. Lymphopenia and lung complications in patients with coronavirus disease-2019 (COVID-19): A retrospective study based on clinical data. J Med Virol. 2021 doi: 10.1002/jmv.27060. doi: 10.1002/jmv. 27060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Malik P, Patel U, Mehta D, Patel N, Kelkar R, Akrmah M, et al. Biomarkers and outcomes of COVID-19 hospitalizations: Systematic review and meta-analysis. BMJ Evid Based Med. 2021;26:107–8. doi: 10.1136/bmjebm-2020-111536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yang AP, Liu JP, Tao WQ, Li HM. The diagnostic and predictive role of NLR, d-NLR, and PLR in COVID-19 patients. Int Immunopharmacol. 2020;84:106504. doi: 10.1016/j.intimp.2020.106504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Siow WT, Liew MF, Shrestha BR, Muchtar F, See KC. Managing COVID-19 in resource-limited settings: Critical care considerations. Crit Care. 2020;24:167. doi: 10.1186/s13054-020-02890-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kucirka LM, Lauer SA, Laeyendecker O, Boon D, Lessler J. Variation in false-negative rate of reverse transcriptase polymerase chain reaction-based SARS-CoV-2 Tests by time since exposure. Ann Intern Med. 2020;18(173):262–7. doi: 10.7326/M20-1495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Xie X, Zhong Z, Zhao W, Zheng C, Wang F, Liu J. Chest CT for typical coronavirus disease 2019 (COVID-19) pneumonia: Relationship to negative RT-PCR testing. Radiology. 2020;296:E41–5. doi: 10.1148/radiol.2020200343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lal A, Mishra AK, Sahu KK. CT chest findings in coronavirus disease-19 (COVID-19) J Formos Med Assoc. 2020;119:1000–1. doi: 10.1016/j.jfma.2020.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]


