Abstract
Transforming growth factor (TGF)-β1 and mesenchymal stromal cells (MSCs) are two effective immunosuppressive agents for organ transplantation technology. This study aims to explore the molecular mechanism of TGF-β1-overexpressed MSCs on T cell immunosuppression. To achieve that, BM-MSCs were isolated from canine bone marrow, and their osteogenic differentiation and surface markers were detected. The TGF-β1 gene was transferred into lentivirus and modified MSCs (TGF-β1/MSCs) by lentivirus transfection. Furthermore, TGF-β1/MSCs were co-cultured with T cells to investigate their effect on differentiation and immune regulation. Results showed that TGF-β1/MSCs significantly downregulated the proportion of CD4+ CD8+ T cells in lymphocytes and significantly upregulated the proportion of CD4+ CD25+ T cells. Moreover, TGF-β1/MSCs significantly upregulated the expression of IL-10 in CD4+ T cells and downregulated the expression of IL-17A, IL-21, and IL-22. Meanwhile, interferon-γ (IFN-γ) neutralizing antibody blocked the effects of TGF-β1/MSCs on the differentiation inhibition of Th17. Overall, our results confirm the strong immunosuppressive effect of TGF-β1/MSCs in vitro and demonstrate that IFN-γ mediates the immunosuppressive effect of TGF-β1/MSC.
Keywords: TGF-β1, MSC, Th17, Treg, immunosuppression
1. Introduction
With the continuous development and progress of modern medicine, the transplantation of allogeneic tissues and organs has become necessary for various diseases. However, the acute or chronic rejection that often occurs after allotransplantation seriously restricts the further development and application of transplantation technology. At present, immunosuppressants are mainly used to inhibit rejection after transplantation to prolong the survival time of grafts or transplant recipients; however, recipients need to take immunosuppressant drugs for life. Moreover, it may produce various side effects and increase the risk of opportunistic infection, causing a significant burden to patients, which seriously limits the scope of its clinical application. Therefore, it is a crucial problem to be solved urgently in transplantation technology to find a method that can induce allograft immune tolerance, prolong graft survival time, have low side effects, and high safety factors.
Bone marrow mesenchymal stem cells (MSCs) belong to nonterminally differentiated cells, which have the characteristics of mesenchymal cells and the potential of stem cell self-renewal and multidirectional differentiation. Animal experiments and preliminary clinical transplantation studies have found that MSCs play an essential role in supporting stem cell implantation and regulating transplantation immunity. After the infusion of MSCs to patients receiving allogeneic skin transplantation, it was found that MSCs could significantly prolong the survival time of allogeneic skin transplantation [1]. A study on the use of mesenchymal stem cells in cartilage regeneration in osteoarthritis has shown that MSCs have special low immunogenicity and immunomodulatory ability [2]. Another study of 47 patients with intractable graft-versus-host disease showed that bone marrow MSCs isolated from third parties effectively treated intractable graft-versus-host disease [3]. The unique immunological characteristics of MSCs enable them to survive for a long time in an allogeneic or even xenogeneic body. After transplantation, MSCs still maintain the potential of multidirectional differentiation but its antigenicity is not increased, which plays a vital role in studying allogeneic or mismatched transplantation. Preliminary studies have shown that autologous or allogeneic bone marrow-derived MSC transplantation does not cause significant adverse reactions. The low immunogenicity of MSCs enables them to evade immune recognition in immune response and gain the ability to survive in the receptor. Besides, MSCs can inhibit T cell proliferation through direct cell-to-cell contact and the indirect effect of cytokines secreted by MSC; moreover, the inhibitory effect of MSCs is dose-dependent, that is, the greater the number of MSCs, the stronger the inhibitory effect [4].
Transforming growth factor β (TGF-β) is a widely used immunosuppressive agent regulating proliferation, differentiation, and biological function of many kinds of immunoreactive cells [5]. Its role in immunity has attracted wide attention, such as inducing B cells to secrete IgA [6]. Particularly, it can stimulate immature CD4+ T cells to differentiate into Foxp3 Treg and inhibit the activation and proliferation of effector T cells [7]. TGF-β leads to cell inactivation and plays a negative regulatory role, thus maintaining immune tolerance [8,9]. Johnston et al. proved that TGF-β upregulated regulatory T cells (Treg), induce immune tolerance, and prolong grafts’ survival time [10]. The activity of TGF-β1 and MSCs can regulate rejection after transplantation.
Based on the above, this study explored the molecular mechanism of TGF-β1 modification of MSCs on T cell immunosuppression. Compared with the blank control group, TGF-β1/MSCs significantly downregulated the proportion of CD4+ CD8+ T cells in lymphocytes and significantly upregulated the proportion of CD4+ CD25+ T cells in lymphocytes. In addition, we also found that TGF-β1/MSCs significantly upregulated the expression of IL-10 in CD4+ T cells and downregulated the expression of IL-17A, IL-21, and IL-22, which explained the immunosuppressive effect of TGF-β1/MSC. Meanwhile, IFN-γ neutralizing antibody blocked the effects of TGF-β1/MSCs on the differentiation inhibition of Th17. Overall, our results confirmed the strong immunosuppressive effect of TGF-β1/MSCs in vitro and found that IFN-γ mediated the immunosuppressive effect of TGF-β1/MSC.
2. Material and methods
2.1. Preparation of canine bone marrow mesenchymal stem cells and gene transduction
Under an aseptic operation, the experimental dogs were anesthetized and punctured at the left or right skeletal crest with a bone marrow puncture needle. First, 3 mL of the bone marrow fluid was extracted into a preprepared 15 mL centrifuge tube containing anticoagulants, and then 3 mL of PBS buffer containing double antibodies was diluted to make 6 mL bone marrow dilution. Clumps were removed by filtering through a 70 μm cell strainer (Thermo-Fisher Scientific). Cells were then centrifuged at 400×g for 5 min and then resuspended in the complete culture medium of stem cells. The cell suspension was inoculated in a 60 mm Petri dish and cultured at 37°C in a 5% CO2 incubator for 24 h. Lentiviral vectors for canine TGF-β1 gene overexpression were constructed by and purchased from Genscript (Nanjing, China). The cells were transduced with the recombinant virus for 12 h and then cultured in the viral vector-free medium for another 48 h. Subsequently, the MSCs transduced with TGF-β1 were exposed to 5 mg/mL puromycin for 3 days to obtain stable transduction TGF-β1/MSCs. To neutralize the IFN-γ, neutralizing antibodies (NA) for IFN-γ (10 μg/mL) were added to the co-culture; NA to IFN-γ were obtained from R&D Systems (Minneapolis, MN).
Ethical approval: The research related to animal use has been complied with all the relevant national regulations and institutional policies for the care and use of animals and was approved by the Animal Ethics Committee of the Second Affiliated Hospital of Kunming Medical University.
2.2. Identification of osteogenic differentiation of MSCs
When the cell density reaches 80%, 1/3rd of bone marrow MSCs were digested with trypsin. The cell concentration was adjusted to 3 × 104 cells/mL and seeded in 6-well plates. About 2 mL of growth medium (stem cell complete medium) was added to each well and cultured at 37°C in 5% CO2 incubator until the cells were covered with culture plates. After 24 h of induction and differentiation, the growth medium was carefully discarded, and 2 mL of the osteogenic induction medium was added to the experimental group. After alizarin red staining, the experimental group cells were induced to form calcium nodules as far as possible. The induced differentiation solute ion was discarded entirely, washed with PBS, and fixed with 4% formaldehyde solution (2 mL for 30 min); after fixation, it was washed twice with PBS and washed 2–3 times with 1 mL of alizarin red staining solution for 3 min. The staining results were observed under an inverted microscope and photographed.
2.3. Flow cytometry cell sorting and analysis
For cell sorting, splenocytes from 7- to 8-week-old BALB/C mice were used to purify CD4+, CD8+ or CD25+ T cells according to the manufacturer’s instructions (BD FACSAria Ⅲ, BD Biosciences, Germany). The purity of T cells was confirmed by flow cytometry. For cell surface staining, the cells were resuspended in PBS (1 × 105 cells/mL). Fluorescence-labeled monoclonal antibodies anti-CD4, anti-CD25, anti-CD34, anti-CD105, anti-CD44, anti-CD29, and anti-CD90 were, respectively, added and incubated in the refrigerator at 4°C for 30 min. All antibodies for flow cytometry were purchased from BD Biosciences. Flow cytometry analysis was performed with a FACS Aria II Cell Sorter (BD Biosciences) and data analysis was carried out using FlowJo software (TreeStar, Ashland, USA).
2.4. Western blotting
Proteins were extracted and subjected to Western blot analysis. Equal amounts of proteins (20 μg) were conducted by SDS-PAGE (12%) and transferred to the PVDF membrane (Epizyme, Shanghai, China) by semidry blotting. After blocking with 5% (w/v) non-fat milk powder, membranes were probed with the primary antibody against β-actin (1:5,000 dilution), IL-17 (1:1,000 dilution), Foxp3 (1:1,000 dilution), and RORγt (1:1,000 dilution). All antibodies were purchased from CST (Shanghai, China). The secondary antibodies were HRP conjugated goat anti-rabbit IgG (1:5,000 dilutions, Proteintech, Wuhan, China). Signals were detected by the chemiluminescence procedure (Pierce, Rockford, Illinois, USA) with BioMax films (Kodak).
2.5. Real-time quantitative PCR
RNA extraction and quantitative real-time PCR were performed for gene expression analysis. In short, according to the manufacturer’s protocols, the total RNA was extracted from the cell sample with the TRIzol reagent (Invitrogen, Carlsbad, CA, USA). The RNA concentration was measured using a spectrophotometer, and all samples were balanced by reverse transcription with a cDNA synthesis kit (Fermentas, St. Leon-Rot, Germany). Using cDNA as templates, SYBR Green I dye-labeled fluorescence quantitative detection was performed at 95°C for 10 min, followed by 40 cycles of 95°C, 20 s and 60°C, 30 s. Assays were performed in duplicate or triplicate as 20 µL reactions in 96 well plates using the 7,500 Fast Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). Using the 2−ΔΔCt method, GAPDH was used as an internal reference to quantify the expression level of target genes in each experimental group. The primers used were synthesized as follows: GAPDH, 5′-AGA CAG CCG CAT CTT CTT GT-3′ (forward), GAPDH, 5′-TGA TGG CAA CAA TGT CCA CT-3′ (reverse); IL-17, 5′-CTC CAG AAG GCC CTC AGA CTAC-3′ (forward), IL-17, 5′-AGC TTT CCC TCC GCA TTG ACA CAG-3′ (reverse); T-bet, 5′-TCA CTA AGC AAG GAC GGA GAA TG-3′ (forward), T-bet, 5′-ATA AGC GGT TCC CTG GCA TAC-3′ (reverse); Foxp3, 5′-CAA GTT CCA CAA CAT GCG AC-3′ (forward), Foxp3, 5′-ATT GAG TGT CCG CTG CTT CT-3′ (reverse); IL-17F, 5′-ACC AAG GCT GCT CTG TTT CT-3′ (forward), IL-17F, 5′-GGT AAG GAG TGG CAT TTC TA-3′ (reverse); RORγt, 5′-TGG AAG TGG TGC TGG TTA GGA TG-3′ (forward), RORγt, 5′-GGA GTG GGA GAA GTC AAA GAT GGA-3′ (reverse); RORα, 5′-CTA CAT TGA CGG GCA CACC-3′ (forward), RORα, 5′-ACA CAG TTG GGG AAG TCT CG-3′ (reverse).
2.6. Quantification of cytokines
After the cells were treated according to the experimental conditions, the culture medium was collected. Then, the culture medium was centrifuged at 4°C for 10 min. The contents of IFN-γ, IL-4, IL-10, IL-21, IL-22, and IL-17A in the supernatant were detected using the ELISA kit (Dakewe, Beijing, China).
2.7. Inhibitory effects of TGF-β1/MSCs on T cells
CD4+ T and CD8+ T cells were isolated from the bone marrow MSCs. All 2.5 × 105 CD4+ T cells per well were co-cultured with MSCs (3 × 105) or TGF-β1/MSCs. The proliferation of CD4+ T cells was measured after 3, 5, or 7 days using the MTT assay [11]. The percentage of suppressive capacity was calculated by defining the absorption of the control as 100%. All CD8+ T cells were co-cultivated with IL-2 (40.0 U/mL) and co-cultured with MSCs or TGF-β1/MSCs. After 3, 5, or 7 days, an LDH assay (Beyotime Biotechnology, Nantong, China) was performed to analyze the cytotoxic lymphocyte (CTL) activity. Then, MDCK cells were added to the effector cells. All cells were cultured under 5% CO2 at 37°C for 24 h, and the cell-free supernatant was then retrieved after centrifugation at 500×g for 5 min to analyze LDH. Then, the CTL killing activity was calculated as previously described [12].
2.8. Statistical analysis
Origin 9.0 for Windows was employed in the data analysis. GraphPad Prism 5 was also adopted for preliminary data analysis. All data were expressed as mean ± SD of individual values. The statistical analysis was determined using Student’s t-test. P < 0.05 was considered statistically significant.
3. Results
3.1. Isolation and identification of BM-MSCs
The cell suspensions isolated from the bone marrow of the femur and tibia of mice were cultured. The next day, the nonadherent cells and impurities were removed by changing the culture medium. With the growth and proliferation of the cells, the adherent cells gradually became fusiform and showed colony-like growth (Figure 1a). To confirm the stemness of MSCs, the osteogenic and adipogenic differentiation potential were determined. After osteogenic induction and differentiation culture, granular substances could be observed in the cells after about 5–7 days, and their levels increased gradually. After about 3 weeks, a large number of granular substances could be observed in the cells, evenly suspended in the culture medium, and alizarin red staining showed positive results, which proved the osteogenic differentiation ability of the extracted cells. Similarly, oil red staining results also showed many lipid droplets, indicating that the isolated MSCs were functional (Figure 1b). The flow cytometry results showed that CD44, CD90, and CD29 were positively expressed, while CD105 and CD34 were negatively expressed (Figure 1c). These results were consistent with the characteristics of MSCs.
Figure 1.
Characterization of BM-MSCs. (a) The basic morphology of BM-MSCs. (b) Identification of osteogenic differentiation ability of BM-MSCs. (c) Phenotype identification of BM-MSCs was conducted by flow cytometry.
3.2. Detection of transfection efficiency
After 96 h of TGF-β1-lentivirus transfection, massive green fluorescence (GFP expression) was observed under a fluorescence microscope in the TGF-β1/MSCs group (Figure 2a), which was also confirmed by flow cytometry (Figure 2b). We further detected the expression of TGF-β1. The results showed that the mRNA level and protein expression of TGF-β1 were significantly increased in TGF- β1/MSCs compared to MSCs.
Figure 2.
Characterization of TGF-β1/MSCs. (a) MSCs were transduced with lentivirus-GFP and lentivirus-TGF-β1. (b) The transduction efficiency was analyzed using flow cytometry. (c) The mRNA level of TGF-β1 in MSCs and TGF-β1/MSCs. (d) The concentration of TGF-β1 in the supernatants of MSCs and TGF-β1/MSCs. Data are expressed as the mean ± SD. Three independent experiments were analyzed using unpaired Student’s t-tests. *P < 0.05; **P < 0.01.
3.3. TGF-β1/MSCs inhibited T lymphocyte proliferation and the regulation of T lymphocyte immune status
T lymphocytes were isolated and purified, and the inhibition of T lymphocyte proliferation and T lymphocyte immune status regulation was observed in MSCs and TGF-β1/MSCs. The proportion of CD4+ CD25+ T cells in mixed lymphocytes was measured by flow cytometry. As shown in Figure 3a, the proportion of CD4+ CD25+ T cells increased under the stimulation of MSCs and TGF-β1/MSCs, and the TGF-β1/MSCs group increased more than the MSCs group. To evaluate the effect of TGF-β1/MSCs on allogeneic stimulation, the mixed lymphocyte reaction of CD4+ T cells and the CTL activity of CD8+ T cells were evaluated. Figure 4b shows that CD4+ T cells from the TGF-β1/MSCs group had markedly decreased proliferative functions than those from the MSCs group and control group. Further, the control group showed a significant increase in the allospecific CTL activity (Figure 4c).
Figure 3.
The effect of TGF-β1/MSCs on T cells. (a) T cells were co-cultured with MSCs or TGF-β1/MSCs, and the proportion of CD4+ CD25+ T cells in lymphocytes was detected by flow cytometry. (b) CD4+ T cells were co-cultured with MSCs or TGF-β1/MSCs for 3, 5, or 7 days, and proliferative responses were measured. (c) CD8+ T cells were co-cultured with MSCs or TGF-β1/MSCs in the presence of IL-2 after 3, 5, or 7 days. Live cells were collected, and the cytotoxic activity against MDCK cells was assessed. Data are expressed as the mean ± SD. Three independent experiments were analyzed using unpaired Student’s t-tests. *P < 0.05; **P < 0.01; MSCs group vs control group. # P < 0.05; ## P < 0.01; TGF-β1/MSCs group vs MSCs group.
Figure 4.
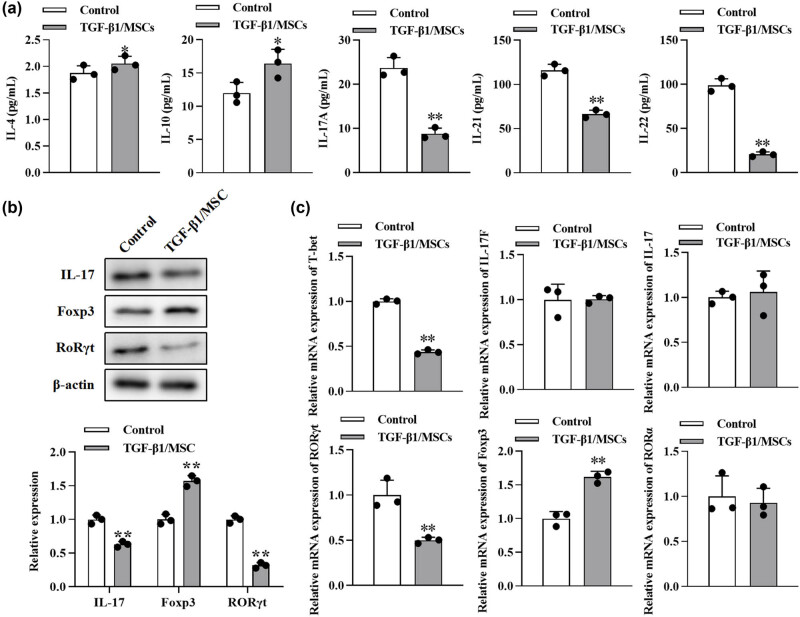
The effect of TGF-β1/MSCs on Th17 differentiation of T cells. (a) After CD4+ CD25− T cells were cultured with TGF-β1/MSCs for 3 days in the presence of IL-2, cytokines (IL-4, IL-10, IL-17A, IL-21, and IL-22) were measured by ELISA from the culture supernatant. (b) The expression of IL17, Foxp3, and RORγt in T cells was measured by Western blotting. (c) The mRNA levels of T-bet, IL-17, IL-17F, RORγt, Foxp3, and RORα were detected using RT-PCR. Data are expressed as the mean ± SD. Three independent experiments were analyzed using unpaired Student’s t-tests. *P < 0.05; **P < 0.01; TGF-β1/MSCs group vs control group.
3.4. TGF-β1/MSCs promoted the generation of Treg cells and suppressed the differentiation to Th17
We further tested the effects of TGF-β1/MSCs on the differentiation of T cells. In order to exclude the effect of TGF-β1 transfection on the proliferation of MSCs, we conducted the CCK-8 assay. The proliferation of MSCs after overexpression of TGF- β1 was not significantly different from that of the control group at 1–5 days but was inhibited at 5–7 days (Figure A1). Next, CD4+ CD25− T cells were co-cultured with TGF-β1/MSCs, and the results showed that the level of IL-10 in the culture supernatant was higher than that in the control group (Figure 4a). However, the levels of IL-17A, IL-21, and IL-22 were lower than those in the control group, and the level of IL-4 was similar to that in the control group. TGF-β1/MSCs could increase the expression of Foxp3 cells but suppressed the expression of IL17 and RORγt in CD4+ CD25− T cells (Figure 4b). Furthermore, CD4+ CD25− T cells in these co-cultures expressed a higher mRNA level of Foxp3 but lower mRNA level of T-bet and RORγt (Figure 4c). These results revealed that TGF-β1/MSCs co-cultured with CD4+ CD25− T cells promoted the generation of Treg cells and suppressed the differentiation to Th17 cells.
3.5. TGF-β1/MSCs inhibited Th17 cell differentiation via IFN-γ
Next, we further explored the intrinsic mechanisms underlying the inhibition of Th17 differentiation. A high level of IFN-γ was observed in the co-culture (Figure 5a), so we conjectured that IFN-γ might be related to the differentiation of Th17. Hence, to neutralize IFN-γ, NA for IFN-γ (10 μg/mL) was added to the co-culture. In the supernatant in the IFN-γ + group, higher levels of IL-17A and IL-22but lower levels of IL-10 were found (Figure 5b). Besides, more IL-17 and RORγt but fewer Foxp3 were detected using Western blot (Figure 5c). These results indicated that IFN-γ mediated the differentiation inhibition of Th17 cells.
Figure 5.
TGF-β1/MSCs regulate Th17 cell differentiation through IFN-γ. (a) CD4+ T cells were co-cultured with TGF-β1/MSCs, and the concentration of IFN-γ in the supernatants was measured. *P < 0.05; **P < 0.01; TGF-β1/MSCs group vs control group. (b) Cytokines were measured by ELISA using the culture supernatant in which CD4+ T cells were co-cultured with TGF-β1/MSCs in the presence of IFN-γ NA. *P < 0.05; **P < 0.01; IFN-γ + group vs IFN-γ- group. (c) The expression of IL17, Foxp3, and RORγt in T cells was measured by Western blotting in the presence of IFN-γ NA. *P < 0.05; **P < 0.01; TGF-β1/MSCs group vs control group. Data are expressed as the mean ± SD. Three independent experiments were analyzed using unpaired Student’s t-tests.
4. Discussion
Organ transplantation is the most effective method for the treatment of patients with end-stage organ failure. However, the therapeutic effect is affected by the possible rejection after the operation and the possible complications of lifelong use of immunosuppressants. Because of their immunosuppressive effect, MSCs have excellent potential in inducing graft immune tolerance to inhibit rejection and reduce the dosage of immunosuppressants. In the study of the ischemia-reperfusion renal transplantation model in rats, it was found that MSCs could reduce the chemotaxis of antigen-presenting cells (APCs), thus effectively reducing the inflammatory reaction in the process of transplantation [13]. Thus, the role of MSCs in renal transplantation has been confirmed. Studies by Miceli et al. [14] have shown that MSCs can reduce the inflammatory response caused by ischemia during renal transplantation, which is beneficial to graft survival. Mohr et al. [15] also showed that infusion of MSCs could significantly improve the decrease of superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) activity caused by ischemia-reperfusion, which is beneficial to the improvement of acute renal failure caused by ischemia-reperfusion.
Treg was first found to inhibit immune response and induce immune tolerance in the 1970s [16]. Natural regulatory T cell (Treg) accounts for about 5–10% of CD4+ T cells, expressing both CD4 and CD25 [17], and specifically expressing Foxp3 [17]. Treg can inhibit the activation and proliferation of CD4+ and CD8+ T lymphocytes, which is related to the direct contact between cells and paracrine cytokines IL-10 and TGF-β [18]. Interleukin-2 (IL-2) and other immunoregulatory molecules have been shown to control the generation of Tregs [19]. It can also indirectly inhibit the immune response by inhibiting the activity of APC while maintaining the microenvironment of immune tolerance. The most significant advantage of Treg in transplantation immunity is that it can actively transplant tolerance, that is, the Treg of the recipient who had reached immune tolerance to organ transplantation can be transferred to other recipients, and the recipient can also achieve the effect of immune tolerance. Sayitoglu et al. [20] confirmed this in an experiment on a mouse skin graft model and found that rejection would occur again after the removal of Treg. Studies of Park [19] and Tahvildari [21] also confirmed that transplantation of Treg could induce immune tolerance to MHC-mismatched heart or skin grafts. Thomann et al. [22] used donor-derived Treg in the islet transplantation model, which also significantly prolonged the survival time of the graft.
TGF-β is a crucial regulator of the immune response, regulating the occurrence and termination of immune response by regulating lymphocyte differentiation and apoptosis. A large number of studies have revealed the mechanism of this regulation. TGF-β can inhibit T lymphocyte proliferation mediated by IL-2 through the Smad3 pathway and directly inhibit T lymphocyte proliferation by downregulating cyclin D2, cyclin E, and c-myc [23,24]; upregulate the expression of FoxP3 and induce the differentiation of Treg. Given the immunosuppressive effect of TGF-β, its application in transplantation immunity has been widely studied. The study of mouse liver transplantation also confirmed a close relationship between TGF-β and liver regeneration [25]. In renal transplantation, TGF-β can significantly inhibit the inflammatory response in the early stage of acute inhibition of rejection, thus achieving the inhibition of chronic rejection [26]. Also, increasing the expression of islet B cell TGF-β in the type 1 diabetic mouse model is helpful to improve the condition of diabetes, which may be related to the increase of T lymphocyte apoptosis [27].
As a well-known immune enhancement factor, IFN-γ is the first discovered cytokine, a necessary condition for stimulating Th1 differentiation [28,29]. At the same time, it can also upregulate the expression of cellular MHC-I molecules, thus improving the ability of immune surveillance in vivo [30]. It can also mediate the activation of macrophages, enhance the lethality of macrophages and NK cells, promote the proliferation of B lymphocytes and the production of antibodies, and upregulate chemokines to promote the chemotaxis of inflammatory cells. According to the report, a large amount of IFN-γ can be detected in the supernatant of the co-culture system of CD4+ T cells and MSCs. Liu et al. believe that this phenomenon is caused by the high expression of TGF-β1 in MSCs [31]. In this study, the IFN-γ-neutralizing antibody significantly blocked the effects of MSCs on T cells, indicating that IFN-γ mediated the differentiation inhibition of Th17.
5. Conclusion
The present study demonstrated that TGF-β1/MSCs inhibited T lymphocyte proliferation and T lymphocyte immune status regulation. Moreover, TGF-β1/MSCs promoted the generation of Treg cells and suppressed the differentiation of Th17 cells. Besides, IFN-γ mediated the differentiation inhibition of Th17.
Appendix
Figure A1.

The effects of overexpressing TGFβ1 on the proliferation of MSCs. The growth curves of the cells after transduction for 7 days were evaluated by the CCK-8 assay. *P < 0.05, **P < 0.01; TGF-β1/MSCs group vs MSCs group.
Footnotes
Funding information: This work was supported by the National Natural Science Foundation of China (81460132) and Yunnan Provincial Science and Technology Department – Kunming Medical University Joint Project (2018FE001).
Author contributions: Ruixue Li and Hong Zhu drafted the manuscript, conceived and designed the study, and revised the manuscript for important intellectual content. Renyong Wang, Shijie Zhong, and Farhan Asghar performed the acquisition of data and conducted the statistical analysis. Tiehan Li and Lei Zhu performed the analysis and interpretation of data. Ruixue Li, Renyong Wang, and Hong Zhu obtained funding. All authors read and approved the final manuscript.
Conflict of interest: The authors state no conflict of interest.
Data availability statement: The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
- [1].Kim YJ, Shin S, Han DJ, Kim YH, Lee JY, Yoon YH, et al. Long-term effects of pancreas transplantation on diabetic retinopathy and incidence and predictive risk factors for early worsening. Transplantation. 2018;102(1):e30–8. [DOI] [PubMed]; Kim YJ, Shin S, Han DJ, Kim YH, Lee JY, Yoon YH. et al. Long-term effects of pancreas transplantation on diabetic retinopathy and incidence and predictive risk factors for early worsening. Transplantation. 2018;102(1):e30–8. doi: 10.1097/TP.0000000000001958. [DOI] [PubMed] [Google Scholar]
- [2].Kristjánsson B, Honsawek S. Mesenchymal stem cells for cartilage regeneration in osteoarthritis. World J Orthopedics. 2017;8(9):674–80. [DOI] [PMC free article] [PubMed]; Kristjánsson B, Honsawek S. Mesenchymal stem cells for cartilage regeneration in osteoarthritis. World J Orthopedics. 2017;8(9):674–80. doi: 10.5312/wjo.v8.i9.674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Zhao K, Lou R, Huang F, Peng Y, Jiang Z, Huang K, et al. Immunomodulation effects of mesenchymal stromal cells on acute graft-versus-host disease after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2015;21(1):97–104. [DOI] [PubMed]; Zhao K, Lou R, Huang F, Peng Y, Jiang Z, Huang K. et al. Immunomodulation effects of mesenchymal stromal cells on acute graft-versus-host disease after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2015;21(1):97–104. doi: 10.1016/j.bbmt.2014.09.030. [DOI] [PubMed] [Google Scholar]
- [4].Fan XL, Zeng QX, Li X, Li CL, Xu ZB, Deng XQ, et al. Induced pluripotent stem cell-derived mesenchymal stem cells activate quiescent T cells and elevate regulatory T cell response via NF-κB in allergic rhinitis patients. Stem Cell Res Ther. 2018;9(1):1–15. [DOI] [PMC free article] [PubMed]; Fan XL, Zeng QX, Li X, Li CL, Xu ZB, Deng XQ. et al. Induced pluripotent stem cell-derived mesenchymal stem cells activate quiescent T cells and elevate regulatory T cell response via NF-κB in allergic rhinitis patients. Stem Cell Res Ther. 2018;9(1):1–15. doi: 10.1186/s13287-018-0896-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Istomine R, Alvarez F, Almadani Y, Philip A, Piccirillo CA. The deubiquitinating enzyme ubiquitin-specific peptidase 11 potentiates TGF-β signaling in CD4+ T cells to facilitate Foxp3 + regulatory T and TH17 cell differentiation. J Immunology. 2019;203(9):2388–400. [DOI] [PubMed]; Istomine R, Alvarez F, Almadani Y, Philip A, Piccirillo CA. The deubiquitinating enzyme ubiquitin-specific peptidase 11 potentiates TGF-β signaling in CD4+ T cells to facilitate Foxp3 + regulatory T and TH17 cell differentiation. J Immunology. 2019;203(9):2388–400. doi: 10.4049/jimmunol.1801689. [DOI] [PubMed] [Google Scholar]
- [6].Weissler KA, Frischmeyer-Guerrerio PA. Genetic evidence for the role of transforming growth factor-β in atopic phenotypes. Curr Opt Immunol. 2019;60:54–62. [DOI] [PMC free article] [PubMed]; Weissler KA, Frischmeyer-Guerrerio PA. Genetic evidence for the role of transforming growth factor-β in atopic phenotypes. Curr Opt Immunol. 2019;60:54–62. doi: 10.1016/j.coi.2019.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Zhang XX, Qiao YC, Li W, Zou X, Chen YL, Shen J, et al. Human amylin induces CD4+ Foxp3 + regulatory T cells in the protection from autoimmune diabetes. Immunologic Res. 2018;66(1):179–86. [DOI] [PubMed]; Zhang XX, Qiao YC, Li W, Zou X, Chen YL, Shen J. et al. Human amylin induces CD4+ Foxp3 + regulatory T cells in the protection from autoimmune diabetes. Immunologic Res. 2018;66(1):179–86. doi: 10.1007/s12026-017-8956-5. [DOI] [PubMed] [Google Scholar]
- [8].Huai G, Markmann JF, Deng S, Rickert CG. TGF‐β‐secreting regulatory B cells: unsung players in immune regulation. Clin Transl Immunol. 2021;10(4):e1270. [DOI] [PMC free article] [PubMed]; Huai G, Markmann JF, Deng S, Rickert CG. TGF‐β‐secreting regulatory B cells: unsung players in immune regulation. Clin Transl Immunol. 2021;10(4):e1270. doi: 10.1002/cti2.1270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Guzman-Genuino RM, Diener KR. Regulatory B cells in pregnancy: lessons from autoimmunity, graft tolerance, and cancer. Front Immunol. 2017;8:172. [DOI] [PMC free article] [PubMed]; Guzman-Genuino RM, Diener KR. Regulatory B cells in pregnancy: lessons from autoimmunity, graft tolerance, and cancer. Front Immunol. 2017;8:172. doi: 10.3389/fimmu.2017.00172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Johnston CJ, Smyth DJ, Dresser DW, Maizels RM. TGF-β in tolerance, development and regulation of immunity. Cell Immunol. 2016;299:14–22. [DOI] [PMC free article] [PubMed]; Johnston CJ, Smyth DJ, Dresser DW, Maizels RM. TGF-β in tolerance, development and regulation of immunity. Cell Immunol. 2016;299:14–22. doi: 10.1016/j.cellimm.2015.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Kuca-Warnawin E, Janicka I, Szczęsny P, Olesińska M, Bonek K, Głuszko P, et al. Modulation of T-Cell activation markers expression by the adipose tissue–derived mesenchymal stem cells of patients with rheumatic diseases. Cell Transplant. 2020;29:0963689720945682. [DOI] [PMC free article] [PubMed]; Kuca-Warnawin E, Janicka I, Szczęsny P, Olesińska M, Bonek K, Głuszko P. et al. Modulation of T-Cell activation markers expression by the adipose tissue–derived mesenchymal stem cells of patients with rheumatic diseases. Cell Transplant. 2020;29:0963689720945682. doi: 10.1177/0963689720945682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Niu J, Wang Y, Liu B, Yao Y. Mesenchymal stem cells prolong the survival of orthotopic liver transplants by regulating the expression of TGF-β1. Turkish J Gastroenterol. 2018;29(5):601–9. [DOI] [PMC free article] [PubMed]; Niu J, Wang Y, Liu B, Yao Y. Mesenchymal stem cells prolong the survival of orthotopic liver transplants by regulating the expression of TGF-β1. Turkish J Gastroenterol. 2018;29(5):601–9. doi: 10.5152/tjg.2018.17395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Bai L, Shao H, Wang H, Zhang Z, Su C, Dong L, et al. Effects of mesenchymal stem cell-derived exosomes on experimental autoimmune uveitis. Sci Rep. 2017;7(1):1–11. [DOI] [PMC free article] [PubMed]; Bai L, Shao H, Wang H, Zhang Z, Su C, Dong L. et al. Effects of mesenchymal stem cell-derived exosomes on experimental autoimmune uveitis. Sci Rep. 2017;7(1):1–11. doi: 10.1038/s41598-017-04559-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Miceli V, Bertani A, Chinnici CM, Bulati M, Pampalone M, Amico G, et al. Conditioned medium from human amnion-derived mesenchymal stromal/stem cells attenuating the effects of cold ischemia-reperfusion injury in an in vitro model using human alveolar epithelial cells. Int J Mol Sci. 2021;22(2):510. [DOI] [PMC free article] [PubMed]; Miceli V, Bertani A, Chinnici CM, Bulati M, Pampalone M, Amico G. et al. Conditioned medium from human amnion-derived mesenchymal stromal/stem cells attenuating the effects of cold ischemia-reperfusion injury in an in vitro model using human alveolar epithelial cells. Int J Mol Sci. 2021;22(2):510. doi: 10.3390/ijms22020510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Mohr A, Zwacka R. The future of mesenchymal stem cell-based therapeutic approaches for cancer–from cells to ghosts. Cancer Lett. 2018;414:239–49. [DOI] [PubMed]; Mohr A, Zwacka R. The future of mesenchymal stem cell-based therapeutic approaches for cancer–from cells to ghosts. Cancer Lett. 2018;414:239–49. doi: 10.1016/j.canlet.2017.11.025. [DOI] [PubMed] [Google Scholar]
- [16].Lin S, Wu H, Wang C, Xiao Z, Xu F. Regulatory T cells and acute lung injury: cytokines, uncontrolled inflammation, and therapeutic implications. Front Immunol. 2018;9:1545. [DOI] [PMC free article] [PubMed]; Lin S, Wu H, Wang C, Xiao Z, Xu F. Regulatory T cells and acute lung injury: cytokines, uncontrolled inflammation, and therapeutic implications. Front Immunol. 2018;9:1545. doi: 10.3389/fimmu.2018.01545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Ferrandino F, Grazioli P, Bellavia D, Campese AF, Screpanti I, Felli MP. Notch and NF-κB: coach and players of regulatory T-cell response in cancer. Front Immunol. 2018;9:2165. [DOI] [PMC free article] [PubMed]; Ferrandino F, Grazioli P, Bellavia D, Campese AF, Screpanti I, Felli MP. Notch and NF-κB: coach and players of regulatory T-cell response in cancer. Front Immunol. 2018;9:2165. doi: 10.3389/fimmu.2018.02165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Apert C, Romagnoli P, van Meerwijk JPM. IL-2 and IL-15 dependent thymic development of Foxp3-expressing regulatory T lymphocytes. Protein Cell. 2018;9(4):322–32. [DOI] [PMC free article] [PubMed]; Apert C, Romagnoli P, van Meerwijk JPM. IL-2 and IL-15 dependent thymic development of Foxp3-expressing regulatory T lymphocytes. Protein Cell. 2018;9(4):322–32. doi: 10.1007/s13238-017-0425-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Park MJ, Baek JA, Kim SY, Jung KA, Choi JW, Park SH, et al. Myeloid-derived suppressor cells therapy enhance immunoregulatory properties in acute graft versus host disease with combination of regulatory T cells. J Transl Med. 2020;18(1):1–14. [DOI] [PMC free article] [PubMed]; Park MJ, Baek JA, Kim SY, Jung KA, Choi JW, Park SH. et al. Myeloid-derived suppressor cells therapy enhance immunoregulatory properties in acute graft versus host disease with combination of regulatory T cells. J Transl Med. 2020;18(1):1–14. doi: 10.1186/s12967-020-02657-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Sayitoglu EC, Freeborn RA, Roncarolo MG. The Yin and Yang of type 1 regulatory T cells: from discovery to clinical application. Front Immunol. 2021;12:2199. [DOI] [PMC free article] [PubMed]; Sayitoglu EC, Freeborn RA, Roncarolo MG. The Yin and Yang of type 1 regulatory T cells: from discovery to clinical application. Front Immunol. 2021;12:2199. doi: 10.3389/fimmu.2021.693105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Tahvildari M, Inomata T, Amouzegar A, Dana R. Regulatory T cell modulation of cytokine and cellular networks in corneal graft rejection. Curr Ophthalmol Rep. 2018;6(4):266–74. [PMC free article] [PubMed]; Tahvildari M, Inomata T, Amouzegar A, Dana R. Regulatory T cell modulation of cytokine and cellular networks in corneal graft rejection. Curr Ophthalmol Rep. 2018;6(4):266–74. [PMC free article] [PubMed] [Google Scholar]
- [22].Thomann AS, Schneider T, Cyran L, Eckert IN, Kerstan A, Lutz MB. Conversion of anergic T cells into Foxp3-IL-10 + regulatory T cells by a second antigen stimulus in vivo. Front Immunol. 2021;12:2598. [DOI] [PMC free article] [PubMed]; Thomann AS, Schneider T, Cyran L, Eckert IN, Kerstan A, Lutz MB. Conversion of anergic T cells into Foxp3-IL-10 + regulatory T cells by a second antigen stimulus in vivo. Front Immunol. 2021;12:2598. doi: 10.3389/fimmu.2021.704578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Geisinger S. Transforming growth factor beta suppression of CD8 + T Cell proliferation. Loyola University Chicago: ProQuest Dissertations Publishing; 2020.; Geisinger S. Transforming growth factor beta suppression of CD8 + T Cell proliferation. Loyola University Chicago: ProQuest Dissertations Publishing; 2020. [Google Scholar]
- [24].Takeuchi H, Konnai S, Maekawa N, Takagi S, Ohta H, Sasaki N, et al. Canine transforming growth factor-β receptor 2-Ig: a potential candidate biologic for melanoma treatment that reverses transforming growth factor-β1 immunosuppression. Front Veterinary Sci. 2021;8:656715. [DOI] [PMC free article] [PubMed]; Takeuchi H, Konnai S, Maekawa N, Takagi S, Ohta H, Sasaki N. et al. Canine transforming growth factor-β receptor 2-Ig: a potential candidate biologic for melanoma treatment that reverses transforming growth factor-β1 immunosuppression. Front Veterinary Sci. 2021;8:656715. doi: 10.3389/fvets.2021.656715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Yin L, Guo X, Zhang C, Cai Z, Xu C. In silico analysis of expression data during the early priming stage of liver regeneration after partial hepatectomy in rat. Oncotarget. 2018;9(14):11794–804. [DOI] [PMC free article] [PubMed]; Yin L, Guo X, Zhang C, Cai Z, Xu C. In silico analysis of expression data during the early priming stage of liver regeneration after partial hepatectomy in rat. Oncotarget. 2018;9(14):11794–804. doi: 10.18632/oncotarget.24370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Park MS, Kim SK, Park HJ, Seok H, Kang SW, Lee SH, et al. Association studies of bone morphogenetic protein 2 gene polymorphisms with acute rejection in kidney transplantation recipients. Transplant Proc. 2017;49(5):1012–7. [DOI] [PubMed]; Park MS, Kim SK, Park HJ, Seok H, Kang SW, Lee SH. et al. Association studies of bone morphogenetic protein 2 gene polymorphisms with acute rejection in kidney transplantation recipients. Transplant Proc. 2017;49(5):1012–7. doi: 10.1016/j.transproceed.2017.03.019. [DOI] [PubMed] [Google Scholar]
- [27].Miani M, Le Naour J, Waeckel-Enée E, Verma SC, Straube M, Emond P, et al. Gut microbiota-stimulated innate lymphoid cells support β-defensin 14 expression in pancreatic endocrine cells, preventing autoimmune diabetes. Cell Metab. 2018;28(4):557–72.e6. [DOI] [PubMed]; Miani M, Le Naour J, Waeckel-Enée E, Verma SC, Straube M, Emond P. et al. Gut microbiota-stimulated innate lymphoid cells support β-defensin 14 expression in pancreatic endocrine cells, preventing autoimmune diabetes. Cell Metab. 2018;28(4):557–72.e6. doi: 10.1016/j.cmet.2018.06.012. [DOI] [PubMed] [Google Scholar]
- [28].Chatterjee S, Chatterjee A, Jana S, Dey S, Roy H, Das MK, et al. Transforming growth factor beta orchestrates PD-L1 enrichment in tumor-derived exosomes and mediates CD8 T-cell dysfunction regulating early phosphorylation of TCR signalome in breast cancer. Carcinogenesis. 2021;42(1):38–47. [DOI] [PubMed]; Chatterjee S, Chatterjee A, Jana S, Dey S, Roy H, Das MK. et al. Transforming growth factor beta orchestrates PD-L1 enrichment in tumor-derived exosomes and mediates CD8 T-cell dysfunction regulating early phosphorylation of TCR signalome in breast cancer. Carcinogenesis. 2021;42(1):38–47. doi: 10.1093/carcin/bgaa092. [DOI] [PubMed] [Google Scholar]
- [29].Okada K, Sadahiro S, Chan LF, Ogimi T, Miyakita H, Saito G, et al. The number of natural killer cells in the largest diameter lymph nodes is associated with the number of retrieved lymph nodes and lymph node size, and is an independent prognostic factor in patients with stage II colon cancer. Oncology. 2018;95(5):288–96. [DOI] [PMC free article] [PubMed]; Okada K, Sadahiro S, Chan LF, Ogimi T, Miyakita H, Saito G. et al. The number of natural killer cells in the largest diameter lymph nodes is associated with the number of retrieved lymph nodes and lymph node size, and is an independent prognostic factor in patients with stage II colon cancer. Oncology. 2018;95(5):288–96. doi: 10.1159/000491019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Qin XY, Zhang YL, Chi YF, Yan B, Zeng XJ, Li HH, et al. Angiotensin II regulates Th1 T cell differentiation through angiotensin II type 1 receptor-PKA-mediated activation of proteasome. Cell Physiol Biochem. 2018;45(4):1366–76. [DOI] [PubMed]; Qin XY, Zhang YL, Chi YF, Yan B, Zeng XJ, Li HH. et al. Angiotensin II regulates Th1 T cell differentiation through angiotensin II type 1 receptor-PKA-mediated activation of proteasome. Cell Physiol Biochem. 2018;45(4):1366–76. doi: 10.1159/000487562. [DOI] [PubMed] [Google Scholar]
- [31].Liu X, Ren S, Qu X, Ge C, Cheng K, Zhao RC. Mesenchymal stem cells inhibit Th17 cells differentiation via IFN-γ-mediated SOCS3 activation. Immunologic Res. 2015;61(3):219–29. [DOI] [PubMed]; Liu X, Ren S, Qu X, Ge C, Cheng K, Zhao RC. Mesenchymal stem cells inhibit Th17 cells differentiation via IFN-γ-mediated SOCS3 activation. Immunologic Res. 2015;61(3):219–29. doi: 10.1007/s12026-014-8612-2. [DOI] [PubMed] [Google Scholar]