Introduction
Spatially fractionated radiation therapy is hypothesized to allow for safe, hypofractionated, dose- escalated radiation therapy for large tumors.1, 2, 3 Spatially fractionated radiation therapy can be delivered in a grid-like pattern (ie, GRID) using 3-dimensional conformal planning techniques. It can also be delivered using intensity modulated radiation therapy (ie, lattice radiation therapy). Lattice delivers a low dose to a gross tumor volume (GTV) with high dose spherical vertices spaced within the tumor, which has previously been described in small case series of patients with gynecologic and other malignancies.4, 5, 6, 7, 8 We recently developed a spatially fractionated lattice stereotactic body radiation therapy (SBRT) technique that simultaneously delivers 20 Gy in 5 fractions to a planning target volume (PTV) with a simultaneous integrated boost (SIB) to 66.77 Gy to a geometric lattice of spherical subvolumes confined to the tumor.9 A recently completed phase I trial demonstrated that lattice SBRT is associated with low rates of acute toxicity and suggests that it is a highly efficacious treatment for very large tumors.10 The safety and efficacy of lattice SBRT is now further being examined in an ongoing phase II clinical trial (NCT04553471). A graphical summary of the lattice SBRT radiation therapy field and the most recent version of the protocol are available in the Appendix E1.
Tumor lysis syndrome (TLS) is comprised of a combination of metabolic abnormalities and acute kidney injury that usually occurs in patients with hematologic malignancies undergoing cytotoxic treatment.11, 12, 13, 14 TLS is rare in patients with solid tumors, especially those treated with radiation therapy in the absence of chemotherapy, although a few prior cases have been reported.12,13 Here we describe a case of TLS in a patient with metastatic endometrial clear cell carcinoma treated with lattice SBRT alone.
Case Report
An 85-year-old woman with hypertension and chronic kidney disease (CKD) stage III (baseline creatinine approximately 1.70-1.80 mg/dL) presented to a hospital emergency department with 2 months of vaginal bleeding soaking 3 to 4 pads per day and dysuria. Hemoglobin was 9.4 g/dL and creatinine was 2.07 mg/dL on admission. Initial diagnostic computerized tomography (CT) of the abdomen and pelvis demonstrated marked endometrial thickening as well as bilateral mixed cystic and solid nodular masses inseparable from the uterus. In conglomerate the masses measured 13 × 9 × 9 cm (Fig 1A-C). The patient had peritoneal metastases as well as extensive periportal and retroperitoneal lymphadenopathy.
Figure 1.
The pelvic mass (red arrows) demonstrated on initial computed tomography (CT) abdomen/pelvis in the axial (A), coronal (B), and sagittal (C) planes. Panels (D-F) demonstrate the lattice stereotactic body radiation therapy (SBRT) plan, with the characteristic high dose spherical vertices spaced uniformly within the gross tumor volume (GTV)_2000 (red).
Physical examination revealed pale clear tumor masses circumferentially filling the vaginal vault and approaching within 3 cm of the introitus; the tumor had parametrial extension but no rectal involvement. Vaginal biopsy was obtained and demonstrated high-grade carcinoma. Immunohistochemistry was positive for PAX-8, napsin A, pancytokeratin, and CK7. Stains were negative for WT-1 and CK20 and supported a diagnosis of clear cell carcinoma. CT chest was obtained to complete staging and was negative for pulmonary metastases. Work-up yielded a diagnosis of International Federation of Gynecology and Obstetrics stage IVB clear cell carcinoma of the endometrium. The patient was deemed not to be a surgical candidate and so was referred to radiation oncology for palliative radiation therapy.
Given her pain and bleeding, the patient was offered palliative radiation to the pelvis 20 Gy in 5 fractions or further dose escalation with lattice SBRT on a phase II clinical trial (NCT04553471). She consented to lattice SBRT and was simulated supine without intravenous contrast due to CKD. Iodinated KY jelly was placed intravaginally to help delineate the lower extent of the target volume. Planning target volume (PTV) 20 Gy (ie, PTV_2000) consisted of GTV_2000 with a 1-cm isotropic expansion, which was prescribed to 20 Gy in 5 fractions. PTV_6670 consisted of a geometric arrangement of spherical vertices, each with a diameter of 1.5 cm and a minimum of 4 cm of spacing from center-to-center, placed to achieve the desired rapid dose fall-off within GTV_2000. Each spherical vertex was prescribed to a dose of 66.7 Gy in 5 fractions as an SIB within GTV_2000. Specific care was taken to refrain from placing any high dose spheres within a vagina avoidance structure, which was designed by the treating radiation oncologist and located in the inferior third of the PTV_2000. A total of 7 high-dose spheres were placed within the superior two-thirds of GTV_2000, with appropriate separation from the surrounding organs-at-risk to ensure all clinical dosimetric planning guidelines were achieved. The volumetric-modulated arc therapy plan was delivered every other day. The patient tolerated treatment well with only grade 1 nausea, diarrhea, and fatigue, which were present before initiating lattice SBRT.
She was seen in a scheduled follow-up visit 4 days after the completion of therapy. Her gynecologic oncologist remarked on his examination that the vaginal tumor was “markedly diminished in size after radiation therapy but not gone.” He recommended proceeding with platinum-based chemotherapy, but the patient decided against initiation of chemotherapy at that time. Thirteen days after completion of radiation therapy, the patient was found down and bradycardic at home by emergency medical services after the reporting of seizure-like activity by her family. Family members also reported progressive weakness and diminished urine output for several days before this event. In the emergency room, the patient was noted to be minimally responsive and bradycardic with heart rate in the 20s and mean arterial pressure in the 30s, which improved with transcutaneous pacing, norepinephrine, and epinephrine drips. Admission labs are shown in Table 1.
Table 1.
The patient's 24 days prior and 13 days postradiation therapy labs are listed and compared with the criteria for TLS
| Variable | Reference range | 24 days before radiation | 13 days after radiation | Laboratory TLS criteria |
|---|---|---|---|---|
| Potassium | 3.3-4.9 mmol/L | 5.3 mmol/L | 6.4 mmol/L (↑) | ≥6.0 mmol/L or 25% increase from baseline |
| Calcium | 8.5-10.3 mg/dL | 9.2 mg/dL | 9.0 mg/dL (↔) | ≤7.0 mg/dL or 25% increase from baseline |
| Phosphorus | 2.3-4.5 mg/dL | - | 3.8 mg/dL (↔) | ≥4.5 mg/dL or 25% increase from baseline |
| Uric acid | 2.5-7.0 mg/dL | - | 17.6 mg/dL (↑) | ≥8.0 mg/dL or 25% increase from baseline |
| Creatinine | 0.6-1.1 mg/dL | 1.76 mg/dL | 2.82 mg/dL (↑) | Not applicable, but AKI = clinical TLS |
| Lactate dehydrogenase | 100-250 Units/L | - | 370 units/L (↑) | Not applicable |
Abbreviations: AKI = acute kidney injury; TLS = tumor lysis syndrome.
Labs in bold meet TLS criteria.
Cairo-Bishop definition of TLS: (A) laboratory TLS = 2 or more of the metabolic abnormalities present within a 24-hour period, 3 days before or 7 days after the start of therapy; (B) clinical criteria for TLS = laboratory TLS plus any 1 of the following: (1) AKI, (2) cardiac arrhythmia/sudden death, or (3) seizures.40
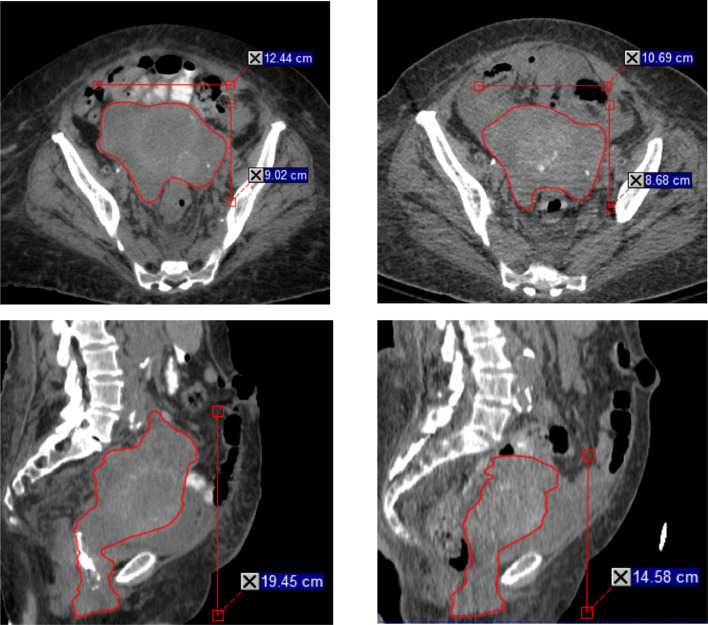
Chest radiograph demonstrated dilatated pulmonary vasculature and bilateral lung opacities due to pulmonary edema. CT head was negative for acute intracranial pathology. CT abdomen and pelvis showed a smaller uterine tumor as well as extensive retroperitoneal, mesenteric, and pericardiophrenic lymphadenopathy and peritoneal metastases (Fig 2). There was no evidence of obstructive nephropathy. Notably, the diagnostic CT was uploaded to the treatment planning system and the GTV_2000 was recontoured yielding a volume of 450 cm3, down from 625 cm3 at time of simulation (28% volume reduction).
Figure 2.
Gross tumor volume (GTV)_2000 (red) in both the axial (top row) and sagittal (bottom row) planes are demonstrated pre- (left column) and 13 days postlattice stereotactic body radiation therapy (SBRT) (right column). Maximum dimensions anterior to posterior, medial to lateral, and superior to inferior are annotated. GTV_2000 contours yielded a volume of 630 cm3 prelattice SBRT and 450 cm3 postlattice SBRT.
The patient was treated with calcium gluconate, insulin-dextrose, albuterol, and sodium zirconium cyclosilicate for hyperkalemia and rasburicase for hyperuricemia. High-rate fluids titrated to a urine output of 100 cc/hr were administered to help with blood pressure, augment renal clearance, and prevent intrarenal crystallization during TLS. Renal dysfunction and metabolic derangements were progressive despite medical management, and the patient declined escalation of care with dialysis. The patient was discharged to home hospice 2 days after admission. She passed away at home.
Discussion
TLS is a severe complication of cytotoxic therapy, which can lead to seizure, cardiac dysrhythmia, and death. TLS is rare in patients with solid tumors.15,16 However, cases of TLS in patients with lung cancers, breast cancers, and gynecologic cancers have previously been reported after chemotherapy.17, 18, 19 Radiation-induced TLS in the absence of chemotherapy is exceedingly rare, and case reports primarily describe cases of patients with myelo- and lymphoproliferative disorders.20, 21, 22, 23
Six cases of radiation-induced TLS in solid tumor malignancies have previously been described.24, 25, 26, 27, 28, 29 The details of these cases are summarized in Table 2. All patients received radiation alone. Notably, in all cases, the patients presented during or up to 7 days after receiving radiation therapy. The timing of radiation-induced TLS in this case is atypical compared with the more common chemotherapy-induced TLS, which generally occurs within 7 days of treatment.12 The exact time at which the patient in our case developed presymptomatic laboratory changes of TLS is unclear in the absence of surveillance labs. Regardless, her presentation 13 days after completion of lattice SBRT, with 28% tumor shrinkage, hyperkalemia, hyperuricemia, acute kidney injury on CKD, arrhythmia, and possible seizure is certainly suggestive of TLS. The highest total dose of radiation received in the prior studies was 30 Gy in 10 fractions, and we are not aware of any studies demonstrating TLS after SBRT. Nevertheless, a slower, radiation-induced “smoldering TLS” would be consistent with the typical slower radiographic responses seen with ablative radiation.30, 31, 32
Table 2.
A summary of the 6 cases of radiation-induced TLS in patients with solid-tumor malignancies available in the literature
| Patient age and sex | Disease | Indication for radiation | Radiation prescription | Timing of TLS presentation | Presenting symptoms | Outcome | Authors (year) |
|---|---|---|---|---|---|---|---|
| 52-year-old-male | Squamous cell carcinoma of the lung | Superior vena cava syndrome | 30 Gy in 10 fractions | After the second fraction of radiation (6 Gy/30 Gy) | Decreased urine output | Death | Noh et al23 (2018) |
| 65-year-old-male | Metastatic melanoma | Pain | 5 fractions (dose N/R) | 7 days after completion of radiation | Renal insufficiency | Death | Dar et al24 (2014) |
| 60-year-old-male | Metastatic prostate cancer | Pain | 30 Gy in 10 fractions | On the sixth day of radiation (18 Gy/30 Gy) | Decreased urine output | Death | Kaplan et al25 (2012) |
| Male (age N/R) | Metastatic bronchogenic carcinoma | N/R | N/R | 3 days after completion of radiation | Seizure | Death | Stuart & Auten26 (2017) |
| 73-year-old-male | Metastatic breast cancer | Pain | 8.5 Gy in 1 fraction (hemi-body irradiation) | 2 days after completion of radiation | Decreased urine output | Death | Rostom et al27 (2000) |
| 34-year-old-female | Metastatic medulloblastoma | Rapidly enlarging abdominal mass | 1 Gy per day (total prescription N/R) | On the third day of radiation (3 Gy) | Decreased urine output | Recovered after medical management | Tomlinson & Solberg28 (1984) |
Abbreviations: N/R = not reported in the literature; TLS = tumor lysis syndrome.
Patients due to receive therapy for malignancy should be risk stratified for TLS. Predictive factors include type and burden of malignancy, anticipated response to cytoreductive therapy, CKD, and pretreatment uric acid and lactate dehydrogenase.33 Pre-emptive management may include the monitoring of laboratory values as well as prophylaxis with intravenous fluids and dose-adjusted allopurinol.12,16,34
Retrospective analysis of clinically available imaging from patients enrolled in the phase I trial of lattice SBRT demonstrated clinically significant shrinkage (median 24.4% by volume) by their first posttreatment imaging a median of 81 days after lattice SBRT.11 More than a quarter of patients had greater than 80% reduction of volume by their second imaging time point 3 to 6 months after treatment. This is consistent with prior retrospective series that also demonstrate partial and complete radiographic responses in a large majority of patients.1,35,36 Of note, it is also possible that the patient's tumor histology is more sensitive to radiation therapy than other tumor types. Clear cell carcinomas lack ARID1A gene expression in roughly 50% of patients, leading to deficient DNA repair after radiation therapy.37, 38, 39, 40
Although the trial is still ongoing, we felt it was important to report this event to reduce the risk that it occurs in the future. Spatially fractionated radiation therapy has a long history, but there is renewed interest in this technique, and it continues to be used outside of prospective studies and without independent safety monitoring. Similar to this event, the literature suggests that TLS could occur greater than 1 week after completion of radiation therapy. Radiation oncologists would not typically consider patients receiving radiation therapy to be at risk for TLS, so it may not be observed with routine clinical follow-up.
Conclusions
Ultimately, the phase II trial protocol was amended to require monitoring of complete blood counts and comprehensive metabolic panels at baseline as well as days 0, 2 to 3, and 7 after completion of treatment for all patients. The details of this protocol amendment are presented in Appendix E2. A similar approach should be used by radiation oncologists treating with spatially fractionated radiation therapy.
Footnotes
Sources of support: This work had no specific funding.
Disclosures: M.S.: Emerson Foundation/Goldman Sachs (grants/contracts), Varian Medical Systems (honoraria). C.R.: Varian Medical Systems (grants/contracts, consulting fees), Merck (grants/contracts), AstraZeneca (consulting fees), EMD Serono (consulting fees), Siemens Healthineers (travel), U.S. provisional application No. 62/598,162, entitled “System and Method for Determining Segments for Ablation” (patent), “Noninvasive Imaging and Treatment System for Cardiac Arrhythmias” WO 2017078757 A1 (patent), Radiologic (leadership, stock). J.K.: Varian Medical Systems (honoraria).
All the data generated and analyzed in this case report are included in this published article and referenced articles.
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.adro.2021.100797.
Appendix. Supplementary materials
References
- 1.Mohiuddin M, Fujita M, Regine WF. High-dose spatially-fractionated radiation (GRID): A new paradigm in the management of advanced cancers. Int J Radiat Oncol Biol Phys. 1999;45:721–727. doi: 10.1016/s0360-3016(99)00170-4. [DOI] [PubMed] [Google Scholar]
- 2.Mohiuddin M, Miller T, Ronjon P. Spatially fractionated grid radiation (SFGRT): A novel approach in the management of recurrent and unresectable soft tissue sarcoma. Int J Radiat Oncol. 2009;75(3, Supplement):S526. [Google Scholar]
- 3.Mohiuddin M, Memon M, Nobah A. Locally advanced high-grade extremity soft tissue sarcoma: Response with novel approach to neoadjuvant chemoradiation using induction spatially fractionated GRID radiotherapy (SFGRT) J Clin Oncol. 2014;32(15_suppl):10575. [Google Scholar]
- 4.Amendola BE, Perez NC, Mayr NA, Wu X, Amendola M. Spatially fractionated radiation therapy using lattice radiation in far-advanced bulky cervical cancer: A clinical and molecular imaging and outcome study. Radiat Res. 2020;194:724–736. doi: 10.1667/RADE-20-00038.1. [DOI] [PubMed] [Google Scholar]
- 5.Amendola B, Perez N, Amendola MA. Lattice radiotherapy with RapidArc for treatment of gynecological tumors: Dosimetric and early clinical evaluations. Cureus. 2010;2 [Google Scholar]
- 6.Amendola BE, Perez NC, Wu X, Amendola MA, Qureshi IZ. Safety and efficacy of lattice radiotherapy in voluminous non-small cell lung cancer. Cureus. 2019;11:e4263. doi: 10.7759/cureus.4263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Griffin RJ, Ahmed MM, Amendola B. Understanding high-dose, ultra-high dose rate, and spatially fractionated radiation therapy. Int J Radiat Oncol Biol Phys. 2020;107:766–778. doi: 10.1016/j.ijrobp.2020.03.028. [DOI] [PubMed] [Google Scholar]
- 8.Wu X, Ahmed MM, Wright J. On modern technical approaches of three-dimensional high-dose lattice radiotherapy (LRT) Cureus. 2010;2 [Google Scholar]
- 9.Duriseti S, Kavanaugh J, Goddu S. Spatially fractionated stereotactic body radiotherapy (Lattice SBRT) for large tumors. Adv Radiat Oncol. 2021;6:100639. doi: 10.1016/j.adro.2020.100639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Duriseti S, Kavanaugh J, Szymanski J, et al. LITE SABR M1: A phase I trail of Lattice SBRT for large tumors [e-pub ahead of print]. medRxiv. 10.1101/2021.06.05.21257825. Accessed October 19, 2021. [DOI]
- 11.Howard SC, Jones DP, Pui C-H. The tumor lysis syndrome. N Engl J Med. 2011;364:1844–1854. doi: 10.1056/NEJMra0904569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wilson FP, Berns JS. Tumor lysis syndrome: New challenges and recent advances. Adv Chronic Kidney Dis. 2014;21:18–26. doi: 10.1053/j.ackd.2013.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Coiffier B, Altman A, Pui C-H. Guidelines for the management of pediatric and adult tumor lysis syndrome: An evidence-based review. J Clin Oncol. 2008;26:2767–2778. doi: 10.1200/JCO.2007.15.0177. [DOI] [PubMed] [Google Scholar]
- 14.Findakly D, Luther RD, Wang J. Tumor lysis syndrome in solid tumors: A comprehensive literature review, new insights, and novel strategies to improve outcomes. Cureus. 2020;12:e8355. doi: 10.7759/cureus.8355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mirrakhimov AE, Ali AM, Khan M. Tumor lysis syndrome in solid tumors: An up to date review of the literature. Rare Tumors. 2014;6 doi: 10.4081/rt.2014.5389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Persons DA, Garst J, Vollmer R. Tumor lysis syndrome and acute renal failure after treatment of non-small-cell lung carcinoma with combination irinotecan and cisplatin. Am J Clin Oncol. 1998;21:426–429. doi: 10.1097/00000421-199808000-00024. [DOI] [PubMed] [Google Scholar]
- 17.Stark ME, Dyer MC, Coonley CJ. Fatal acute tumor lysis syndrome with metastatic breast carcinoma. Cancer. 1987;60:762–764. doi: 10.1002/1097-0142(19870815)60:4<762::aid-cncr2820600409>3.0.co;2-p. [DOI] [PubMed] [Google Scholar]
- 18.Yahata T, Nishikawa N, Aoki Y. Tumor lysis syndrome associated with weekly paclitaxel treatment in a case with ovarian cancer. Gynecol Oncol. 2006;103:752–754. doi: 10.1016/j.ygyno.2006.06.003. [DOI] [PubMed] [Google Scholar]
- 19.Alkan A, Kütük T, Karcı E. Radiation-induced tumor lysis syndrome in chronic lymphocytic leukemia. Turk J Hematol. 2016;33:248–250. doi: 10.4274/tjh.2015.0259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schifter T, Cohen A, Lewinski UH. Severe tumor lysis syndrome following splenic irradiation. Am J Hematol. 1999;60:75–76. doi: 10.1002/(sici)1096-8652(199901)60:1<75::aid-ajh14>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 21.Linck D, Basara N, Tran V. Peracute onset of severe tumor lysis syndrome immediately after 4 Gy fractionated TBI as part of reduced intensity preparative regimen in a patient with T-ALL with high tumor burden. Bone Marrow Transplant. 2003;31:935–937. doi: 10.1038/sj.bmt.1704025. [DOI] [PubMed] [Google Scholar]
- 22.Fleming DR, Henslee-Downey PJ, Coffey CW. Radiation induced acute tumor lysis syndrome in the bone marrow transplant setting. Bone Marrow Transplant. 1991;8:235–236. [PubMed] [Google Scholar]
- 23.Noh GY, Choe DH, Kim CH. Fatal tumor lysis syndrome during radiotherapy for non–small-cell lung cancer. J Clin Oncol. 2008;26:6005–6006. doi: 10.1200/JCO.2008.19.4308. [DOI] [PubMed] [Google Scholar]
- 24.Dar L, Gendelman O, Amital H. Tumor lysis syndrome presenting in a patient with metastatic melanoma treated with radiation therapy. Isr Med Assoc J IMAJ. 2014;16:456–457. [PubMed] [Google Scholar]
- 25.Kaplan MA, Kucukoner M, Alpagat G. Tumor lysis syndrome during radiotherapy for prostate cancer with bone and bone marrow metastases without visceral metastasis. Ann Saudi Med. 2012;32:306–308. doi: 10.5144/0256-4947.2012.306-308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Stuart S, Auten J. A rare seizure: Tumor lysis syndrome after radiation therapy of a solid tumor. Am J Emerg Med. 2017;35:941.e3–941.e4. doi: 10.1016/j.ajem.2017.01.023. [DOI] [PubMed] [Google Scholar]
- 27.Rostom AY, El-Hussainy G, Kandil A. Tumor lysis syndrome following hemi-body irradiation for metastatic breast cancer. Ann Oncol Off J Eur Soc Med Oncol. 2000;11:1349–1351. doi: 10.1023/a:1008347226743. [DOI] [PubMed] [Google Scholar]
- 28.Tomlinson GC, Solberg LA. Acute tumor lysis syndrome with metastatic medulloblastoma. A case report. Cancer. 1984;53:1783–1785. doi: 10.1002/1097-0142(19840415)53:8<1783::aid-cncr2820530829>3.0.co;2-f. [DOI] [PubMed] [Google Scholar]
- 29.Yip C, Hennedige TP, Cook GJR. Imaging assessment after SBRT for hepatocellular carcinoma. Hepatoma Res. 2020;6 [Google Scholar]
- 30.Mastrocostas K, Jang H-J, Fischer S. Imaging post-stereotactic body radiation therapy responses for hepatocellular carcinoma: Typical imaging patterns and pitfalls. Abdom Radiol N Y. 2019;44:1795–1807. doi: 10.1007/s00261-019-01901-y. [DOI] [PubMed] [Google Scholar]
- 31.Alcantara P, Martínez BC, García-Esquinas MG. Evaluation of tumor response after stereotactic body radiation therapy for lung cancer: Role of 18F-fluorodeoxyglucose positron emission tomography/computed tomography. J Clin Transl Res. 2020;6:155–167. [PMC free article] [PubMed] [Google Scholar]
- 32.Wilson FP, Berns JS. Onco-nephrology: Tumor lysis syndrome. Clin J Am Soc Nephrol CJASN. 2012;7:1730–1739. doi: 10.2215/CJN.03150312. [DOI] [PubMed] [Google Scholar]
- 33.Montesinos P, Lorenzo I, Martín G. Tumor lysis syndrome in patients with acute myeloid leukemia: Identification of risk factors and development of a predictive model. Haematologica. 2008;93:67–74. doi: 10.3324/haematol.11575. [DOI] [PubMed] [Google Scholar]
- 34.Sathishkumar S, Dey S, Meigooni AS. The impact of TNF-alpha induction on therapeutic efficacy following high dose spatially fractionated (GRID) radiation. Technol Cancer Res Treat. 2002;1:141–147. doi: 10.1177/153303460200100207. [DOI] [PubMed] [Google Scholar]
- 35.Kaiser A, Mohiuddin MM, Jackson GL. Dramatic response from neoadjuvant, spatially fractionated GRID radiotherapy (SFGRT) for large, high-grade extremity sarcoma. J Radiat Oncol. 2013;2:103–106. [Google Scholar]
- 36.Wiegand KC, Shah SP, Al-Agha OM. ARID1A mutations in endometriosis-associated ovarian carcinomas. N Engl J Med. 2010;363:1532–1543. doi: 10.1056/NEJMoa1008433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jones S, Wang T-L, Shih I-M. Frequent mutations of chromatin remodeling gene ARID1A in ovarian clear cell carcinoma. Science. 2010;330:228–231. doi: 10.1126/science.1196333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Caumanns JJ, Wisman GBA, Berns K. ARID1A mutant ovarian clear cell carcinoma: A clear target for synthetic lethal strategies. Biochim Biophys Acta Rev Cancer. 2018;1870:176–184. doi: 10.1016/j.bbcan.2018.07.005. [DOI] [PubMed] [Google Scholar]
- 39.Park Y, Chui MH, Suryo Rahmanto Y. Loss of ARID1A in tumor cells renders selective vulnerability to combined ionizing radiation and PARP inhibitor therapy. Clin Cancer Res. 2019;25:5584–5594. doi: 10.1158/1078-0432.CCR-18-4222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cairo MS, Bishop M. Tumour lysis syndrome: New therapeutic strategies and classification. Br J Haematol. 2004;127:3–11. doi: 10.1111/j.1365-2141.2004.05094.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.