Abstract
Background/Aim: Wild yam extract [Dioscorea villosa, (WYE)] is consistently lethal at low IC50s across diverse cancer-lines in vitro. Unlike traditional anti-cancer botanicals, WYE contains detergent saponins which reduce oil-water interfacial tensions causing disintegration of lipid membranes and causing cell lysis, creating an interfering variable. Here, we evaluate WYE at sub-lethal concentrations in MDA-MB-231 triple-negative breast cancer (TNBC) cells. Materials and Methods: Quantification of saponins, membrane potential, lytic death and sub-lethal WYE changes in whole transcriptomic (WT) mRNA, miRNAs and biological parameters were evaluated. Results: WYE caused 346 differentially expressed genes (DEGs) out of 48,226 transcripts tested; where up-regulated DEGS reflect immune stimulation, TNF signaling, COX2, cytokine release and cholesterol/steroid biosynthesis. Down-regulated DEGs reflect losses in cell division cycle (CDC), cyclins (CCN), cyclin-dependent kinases (CDKs), centromere proteins (CENP), kinesin family members (KIFs) and polo-like kinases (PLKs), which were in alignment with biological studies. Conclusion: Sub-lethal concentrations of WYE appear to evoke pro-inflammatory, steroid biosynthetic and cytostatic effects in TNBC cells.
Keywords: Immune stimulation, wild yam, Dioscorea, breast cancer, cell cycle
Dioscorea villosa is a North American native plant within the genus Dioscorea, and the roots and rhizomes of this species are known as wild yam (1). This plant has been widely used as a botanical dietary supplement to treat menopause-related hot flashes, muscular cramps, arthritis, upset stomach, coughs, problems related to childbirth, and in cosmetic topical ointments (1). Research on the medicinal value of wild yam root extract (WYE) has shown evidence suggesting anticancer properties in particular for breast cancer and in both hormone receptor-positive and triple-negative breast cancers (TNBC), where it alters epigenetic 5-hydroxymethylcytosine DNA patterns, induces toxicity, halts cell cycle, inhibits fatty acid synthase and modifies the activity of estrogen and progesterone hormone receptors (1-5). Because TNBC is characterized by the lack of estrogen, progesterone, and human epidermal growth factor receptor 2 receptors, treatment options are limited, leading to highly aggressive metastatic cancers, with poor clinical outcomes in terms of treatment relapse and life expectancy. For this reason, a good deal of research has been focused on finding effective alternative treatments for TNBC, such as the case for WYE, which contains hundreds of constituent saponins such as deltonins, dioscoreavillosides, diarylheptanoids (6), diosgenin, and dioscin, the latter two alone can slow breast tumor growth, migration, deter stem cell phenotype and cause cell death in various models (7-11).
There is a unique element of saponin-rich plants, which vastly differs from most naturally derived plant-based chemotherapies like taxol (Taxus brevifolia), having inherent emulsification properties and a capacity to destroy fats on contact, including those housed within biological membranes (12,13). The "on-contact" cell lytic nature of saponins was first observed in red blood cells (RBCs) in the 1920s, likened to taurocholic acid (12), which disrupts cholesterol or phosphatidylcholine rich triglycerides causing pore formation, micellular structures, lytic permeability, and cell death (13). Steroidal saponins in WYE, such as dioscin and diosgenin target phosphatidylcholine-rich membranes while triterpene saponins tend to destroy cholesterol-rich membranes (14-18); the former can induce membrane lytic destruction within minutes (19-21).
Given that WYE and those of the Dioscorea species, to our knowledge, are of the most consistently cytotoxic herbs in vitro across diverse cancer cell lines by saponification/lytic membrane-mediated lysis (22,23), the question remains as to effects that are occurring at concentrations (sub-lethal) that precede saponin induced lytic membrane destruction. In this work, we evaluate whole transcriptomic patterns induced by WYE at sub-lethal concentrations in MDA-MB-231 triple-negative breast cancer (TNBC) cells, where both immune stimulation and cell-cycle ablation are confirmed.
Materials and Methods
Wild yam extract (WYE) preparation. Wild Yam powder was purchased from Mountain Rose Herbs (Eugene, OR, USA). A crude WYE was prepared by dissolving the powder in absolute ethanol at 50 mg/ml, followed by vortexing and storage in the dark at –20˚C. Serial dilutions of WYE were prepared in sterile HBSS.
Cell culture. MDA-MB-231 HTB-26™cells were purchased from ATCC. The cells were cultured in 75 cm2 flasks with high glucose [4,500 g/l] DMEM supplemented with 7% FBS and 100 U/ml penicillin G sodium/100 μg/ml streptomycin sulfate. Cells were grown at 37˚C in 95% atmosphere 5% CO2 and sub-cultured every three to five days. Experimental studies involving monolayers were conducted in growth media (as described above) in 96 well plates or 75 cm2 flasks. Experimental cultures involving 3D spheroids were seeded in culture media, using low-adhesion spheroid forming 96 well plates, and pelleted by centrifugation at 1,800 × g for 3 min, daily for the first three days. The spheroids were grown at 37˚C in 95% atmosphere 5% CO2 for 7 days prior to experimental treatment. Changes in morphology and live-cell imaging with FDA were captured using an inverted fluorescence microscope.
Human cytokine antibody array. Human cytokine antibody arrays (Cat# AAM-CYT-1000) (Raybiotech Inc, Peachtree Corners, GA, USA) were used to profile supernatant cytokine content. Briefly, antibody-coated array membranes were first incubated for 30 min with 1 ml of blocking buffer. After 30 min, the blocking buffer was decanted and replaced with 1 ml supernatant. Supernatants were pre-diluted in sample buffer to 20% to detect signals in highly expressed control proteins. Membranes were allowed to incubate for 5 h with shaking. Membranes were then washed with the prepared washing buffer and then incubated with 1 ml biotin-conjugated antibodies. After incubation, the mixture of biotin-conjugated antibodies was removed, and membranes were incubated with HRP-conjugated streptavidin (2 h). Detection of chemiluminescent spots was captured on Quantity One software installed on a Bio-Rad Versadoc (Bio-rad, Hercules, CA, USA), followed by densitometry analysis using NIH Image J software, microarray analysis plug-in (24). Data was imported into Raybio software data analysis sheets for final analysis.
Membrane potential and live-cell imaging. Fluorescent live cell imaging was carried out using fluorescein diacetate (FDA), which detects both plasma membrane integrity and the presence of viable cells, which retain the esterase cleaved fluorescein product (25). Tetramethyl rhodamine, ethyl ester (TMRE), was used as a permeable fluorescent cation potentiometric dye that can determine mitochondrial membrane potential in live cells (26). The loss of membrane potential and cell viability with elevated levels of saponins were captured by fluorescent imaging using an inverted fluorescent microscope.
Cell viability and proliferation. For basic toxicity experiments, cells were equally plated in 96 well plates at 0.5×106 cells/ml. Resazurin (Alamar Blue) indicator dye was used to measure cell viability as an indicator of basic metabolic rate (27). Briefly, a working solution of resazurin (0.5 mg/ml) was prepared in sterile PBS; filter sterilized through a 0.2-micron filter, added to the samples [15% (v/v) equivalent], and returned to the incubator for 2-6 h. Reduction of the dye by viable cells reduced the oxidized resazurin, yielding a bright red fluorescent intermediate resorufin quantified using a Synergy HTX multi-mode reader (Bio-Tek, Winooski, VT, USA) using the following settings: 530 nm (excitation)/590 nm (emission) filters. Cell proliferation assays were conducted over 6 days, where cells were equally plated in 96 well plates at 0.05×106 cells/ml in culture media in the absence or presence of a varying concentration of WYE.
Saponin foam test. In this study, the stable foaming properties of the saponins in WYE were tested and correlated to the cytotoxic effects of the yam extract. In brief, 500 mg of wild yam root (powdered) was added to 10 ml of sterile water (50 mg/ml) and boiled for 5 min, followed by filtration through a 0.4-micron mesh filter. Serial dilutions of the filtrate were prepared in sterile water (room temperature) in separate glass test tubes, which were stopped and shaken vigorously for about 30 seconds. Samples were allowed to stand for one-half h. Honeycomb froth indicated saponins, where images were captured, and the foam area quantified using Image J analysis software (20).
Microarray WT 2.1 human datasets. All cells were washed three times in ice-cold HBSS, rapidly frozen, and stored at –80˚C. Total RNA was isolated and purified using the Trizol/chloroform method. The quality was assessed, and concentration was equalized to 82 ng/μl in nuclease-free water. Whole transcriptome analysis was conducted according to the GeneChip TM WT PLUS Reagent Manual for Whole Transcript (WT).
Expression arrays. Briefly, RNA was synthesized to the first-strand cDNA, then second-strand cDNA, followed by a subsequent transcription to cRNA. cRNA was purified and assessed for yield prior to 2nd cycle single-stranded cDNA synthesis, hydrolysis of RNA, and purification of 2nd cycle single-stranded cDNA. cDNA was then quantified for yield and equalized to 176 ng/ml. Subsequently, cDNA was fragmented, labeled, and hybridized onto the arrays prior to being subjected to fluidics and imaging using the Gene Atlas (Affymetrix, ThermoFisher Scientific). The array data quality control and initial processing from CEL to CHP files were conducted using an expression console, prior to data evaluation using the Affymetrix transcriptome analysis console, String Database (String Consortium 2020) and DAVID functional annotation microarray tools (28-30), n=3. The dataset has been deposited to NIH Gene Expression Omnibus located at: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE180621.
ELISA for IL-8. Supernatants from MDA-MB-231 cells (Control vs. WYE Lo 15 μg/ml) were collected and centrifuged at 1,000 × g for 5 min at 4˚C. Specific ELISA was performed using IL-8 ELISA kit following manufacturer’s instructions (Human IL-8/CXCL8 ELISA) (Millipore-Sigma, Saint Louis, MO, USA). The sample was diluted in buffer 20% supernatant/buffer, and 100 μl of prepared supernatant from samples was added to 96 well plates pre-coated with the capture antibody. After incubation, 100 μl of prepared biotinylated antibody mixture was added to each well. After 1 h, the mixture was decanted, and 100 μl streptavidin solution was placed in each well and incubated. Substrate reagent (100 μl) was then added to each well for 30 min followed by a 50 μl stop solution. Plates were read at 450 nm using a Biotek H.T.X. Synergy-multi-mode microplate reader.
Statistical analysis. Statistical analysis was performed for the basic studies using GraphPad Prism (version 3.0; Graph Pad Software Inc., San Diego, CA, USA). The significance of the difference between the groups was assessed using either a student’s t-test or a one-way ANOVA followed by Tukey post hoc analysis.
Results
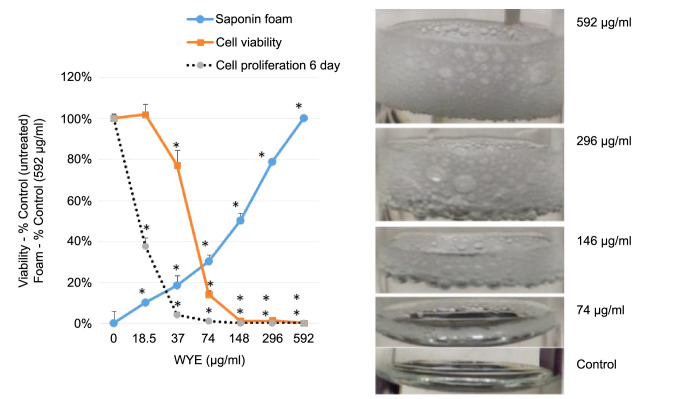
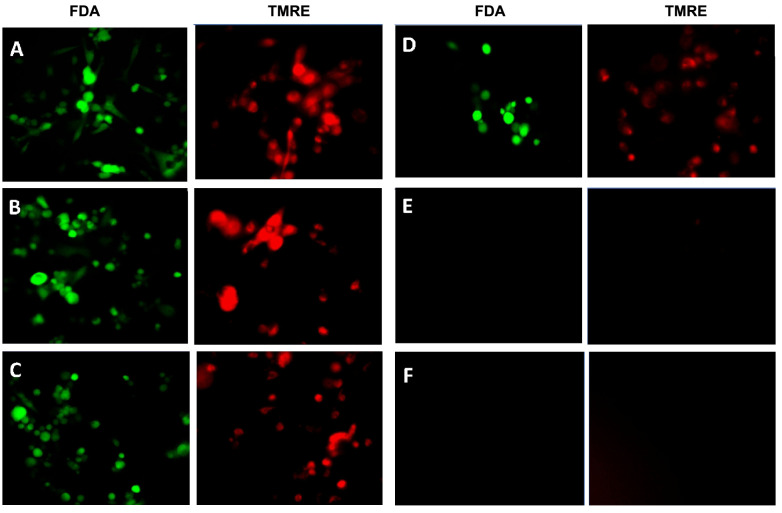
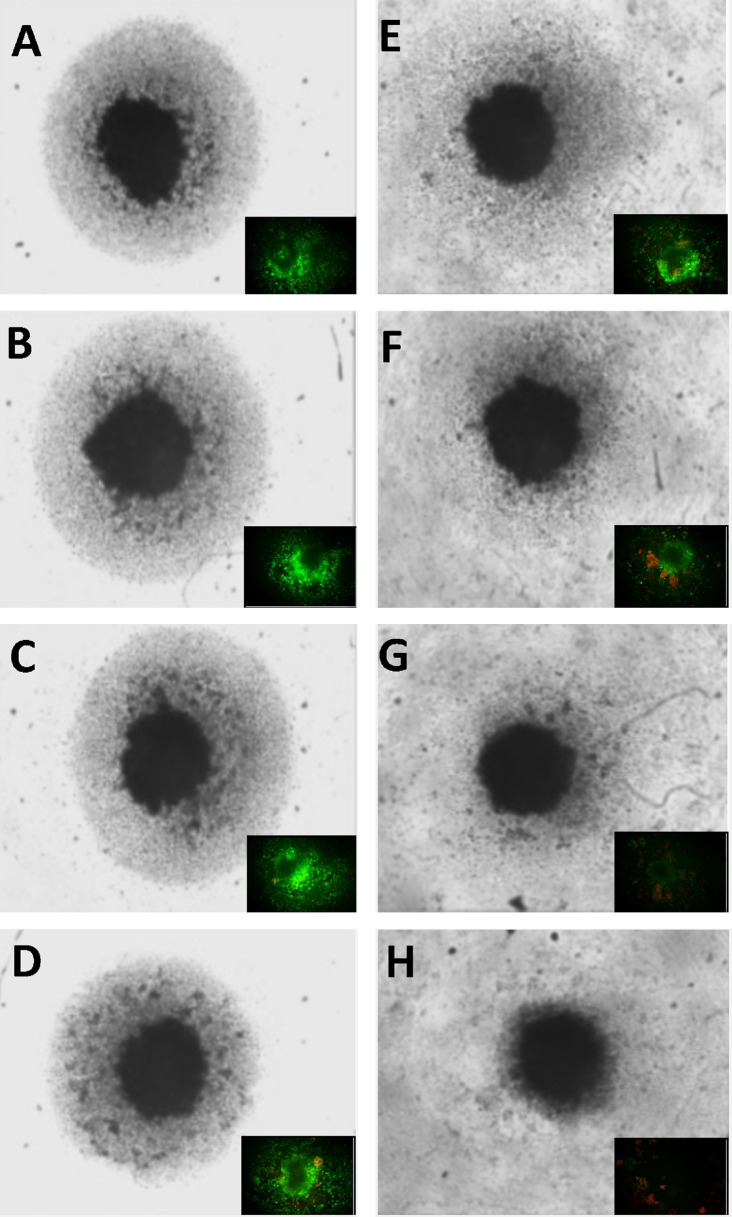
Both cytostatic and cytotoxic curves were generated over a dose-response of WYE, where saponins were quantified by a simple foam test (Figure 1). The data show a close correlation between foam and cell death, with the (anti-proliferative) cytostatic effects being in close proximity to/but slightly preceding sub-lethal concentrations of WYE. At this point, it is uncertain whether the loss of the cell cycle is related to basic cytotoxicity given the proximity of these curves, which will be further elucidated in this work. Next, the data show that greater saponin content in higher concentrations of WYE are associated with cell death, which coincides closely to damage to lipid membrane bio-layers. In Figure 2, we show basic fluorescent cell imaging of plasma membrane integrity/viability (left panel) and mitochondrial membrane (right) using fluorescent probes FDA and TMRE, respectively. The data show complete loss of both with greater saponin foam content in WYE. To determine if WYE saponins would also destroy a small 3D tumor spheroid, we evaluated cell survival (FDA) and morphological changes with increasing concentration of WYE (Figure 3). The data show spheroid tumors to be slightly more resistant to WYE, however, at concentrations exceeding 148 μg/ml there was near complete death of the entire spheroid.
Figure 1. Cytotoxic and cytostatic effects related to saponin content of WYE in MDA-MB-231 cells. The data represent the mean±S.E.M, n=4 for cell viability (at 24 h), cell proliferation (at 6 days) plotted as % (untreated) controls and WYE saponin foam (where 592 μg/ml was set at 100% control), n=3. Significant differences between the controls and treatment groups were determined by a one way ANOVA, followed by a Tukey post hoc test ,*p<0.05. The gene chip icons, represent the 2 concentrations at which microarray analysis work was conducted representing WYE sublethal Low (15 μg/ml) and WYE sub-lethal High (30 μg/ml).
Figure 2. Effect of WYE on cell membrane potentials (plasma/viability) obtained with FDA (left panel) and mitochondrial TMRE (right panel). (A) Control, WYE Treatment (B) 18.5 μg/ml, (C)37 μg/ml, (D) 74 μg/ml, (E) 148 μg/ml, (F) 296 μg/ml).
Figure 3. Effect of WYE on 3D tumor spheroids. Cell viability and morphological changes in MDA-MB-231 tumor spheroids with increasing concentration of WYE are shown. The data represent basic changes in spheroid structure (main image; black and white), with fluorescent FDA viable cell staining (green) in the lower left section of each main image. (A) Control, WYE Treatment (B) 4.62 μg/ml, (C) 9.25 μg/ml, (D) 18.5 μg/ml, (E) 37 μg/ml, (F) 74 μg/ml, (G) 148 μg/ml, (H) 296 μg/ml.
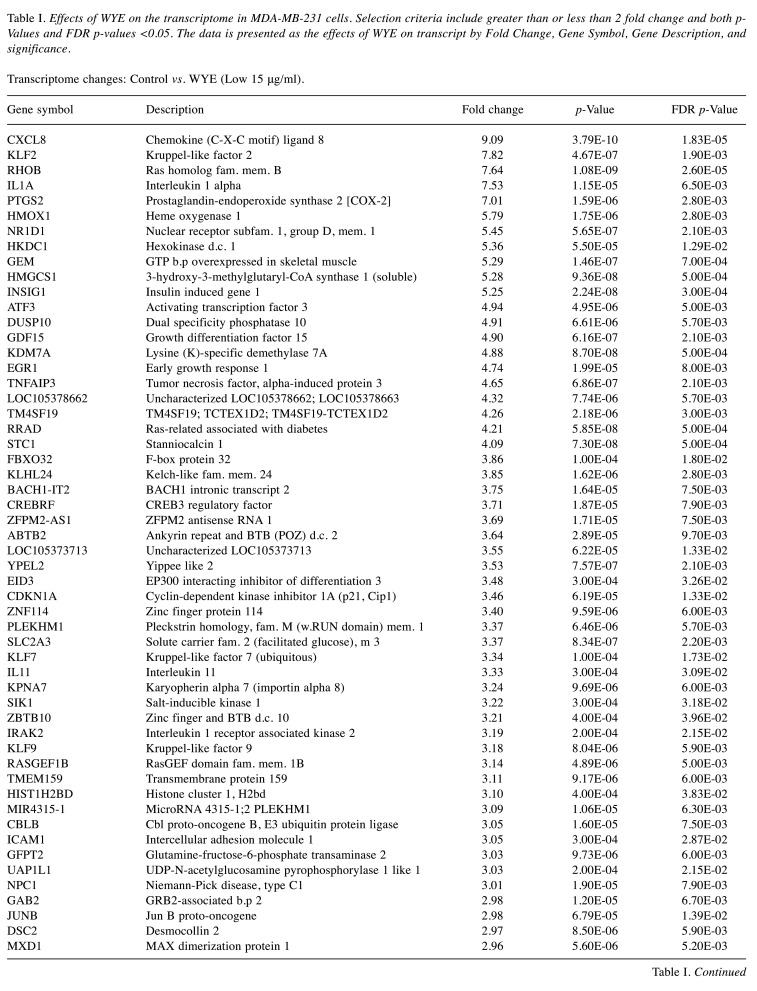
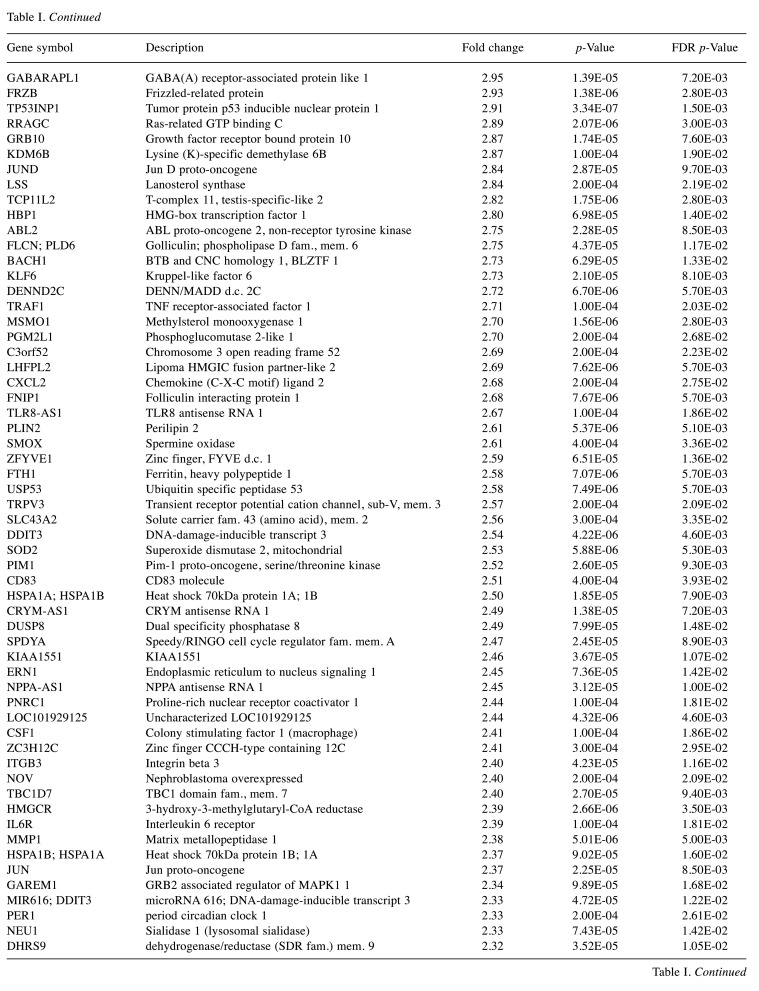
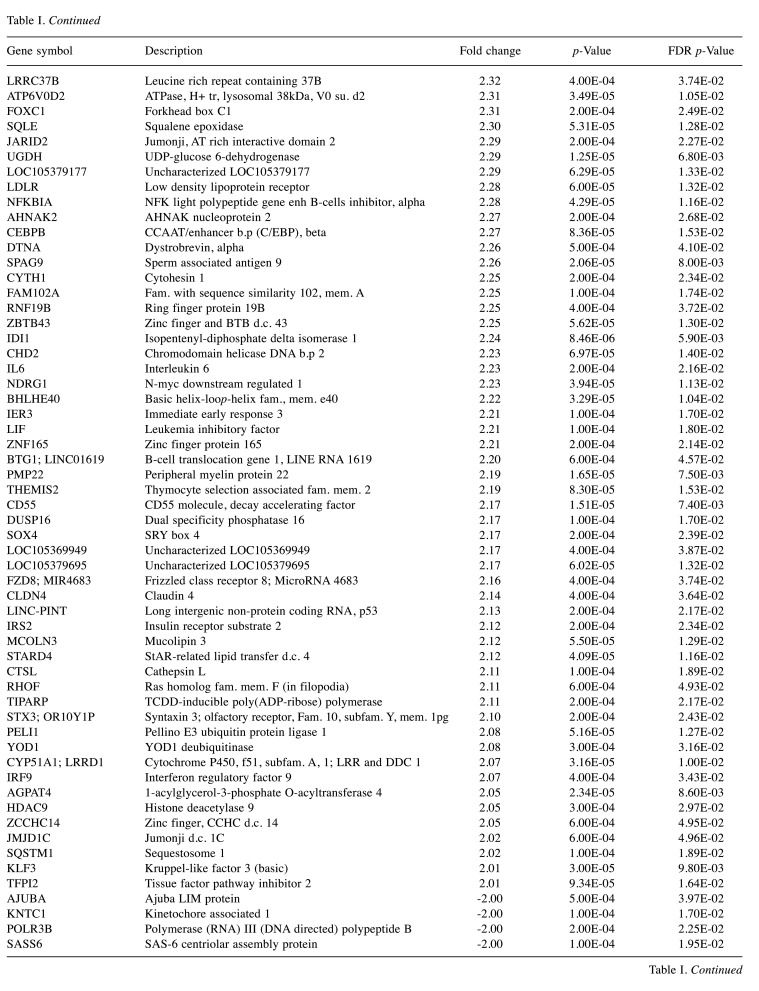
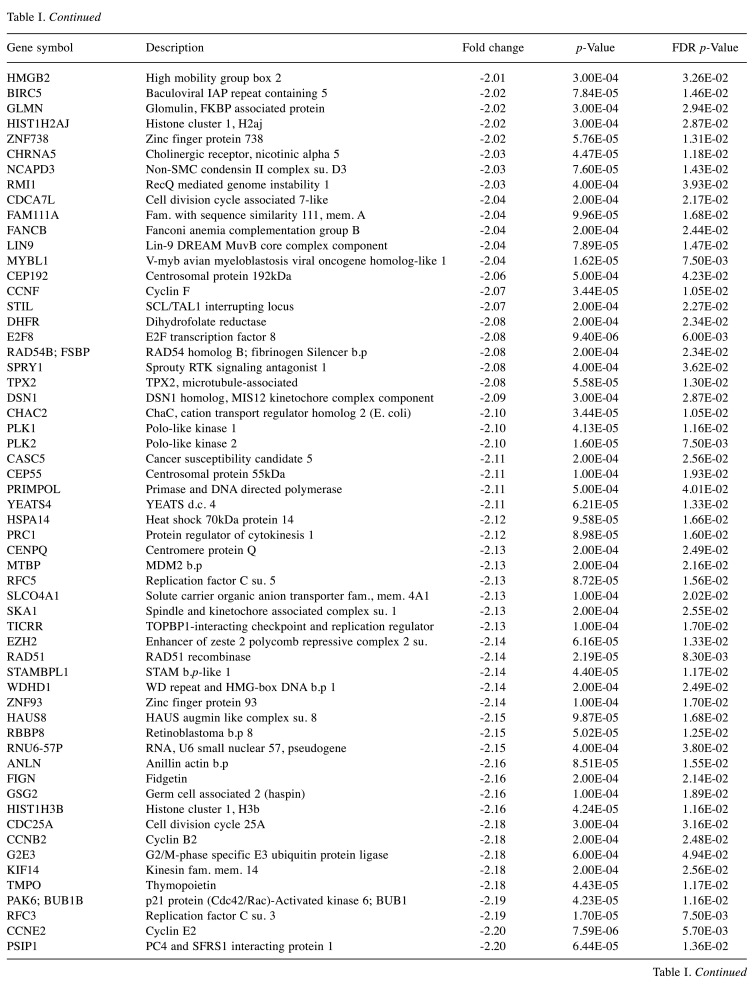
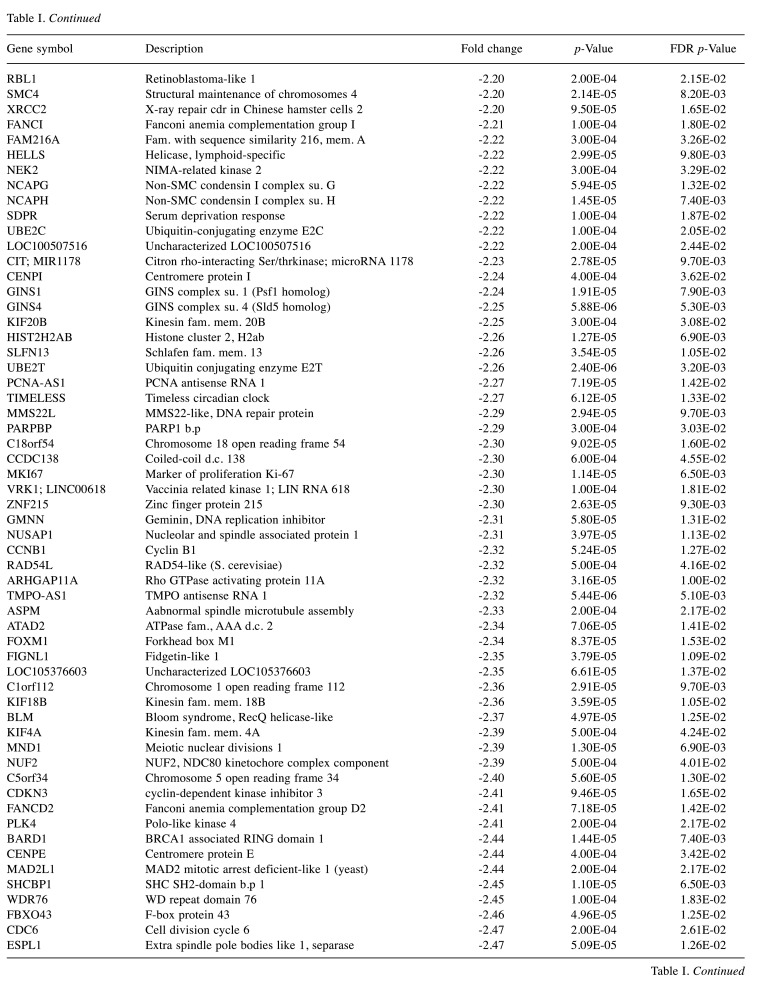
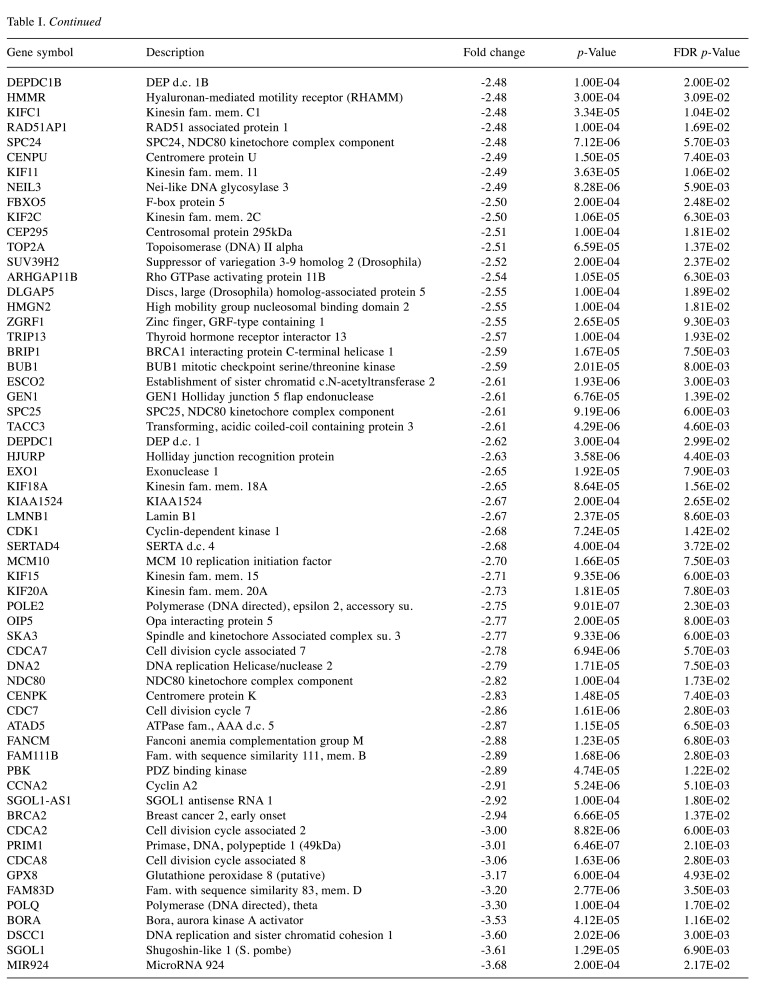
For microarray studies, we chose to conduct experimentation at 2 sub-lethal concentrations denoted as Low (15 μg/ml); sub-lethal and High (30 μg/ml); sub-lethal/cusp of death (Figure 4). Cells for microarray studies were prepared in 75 cm2 flasks and monitored to ensure no morphological structure or attachment changes occurred over 24 h of treatment prior to cell pellet collection. Both cell pellets and supernatant were collected from the same samples and stored at –80˚C. While we provide whole transcriptomic data on both sets, we focus the analysis within this article on only the WYE (low vs. control) data set. In both sets, less than 0.7% of the whole transcriptome showed differentially expressed genes (DEGs), with criteria set at -2<x>+2-fold change, p-Value, and FDR p-Values <0.05. The data for DEGs in the WYE sub-lethal (low) vs. control groups are presented in Table I, with both sets, including sub-lethal WYE (high) vs. control provided at https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE180621.
Figure 4. Microarray gene summary report. Overview of DEGs expressed when comparing controls (untreated) n=3 vs. WYE (Low, sub-lethal at 15 ug/ml) n=3 or WYE (High, sub-lethal cusp of cell death at 30 ug/ml). The data selection criteria were set at -2<x>+2 fold change, p-Value <0.05 and FDR p-Value <0.05.
Table I. Effects of WYE on the transcriptome in MDA-MB-231 cells. Selection criteria include greater than or less than 2 fold change and both p-Values and FDR p-values <0.05. The data is presented as the effects of WYE on transcript by Fold Change, Gene Symbol, Gene Description, and significance. Transcriptome changes: Control vs. WYE (Low 15 μg/ml).
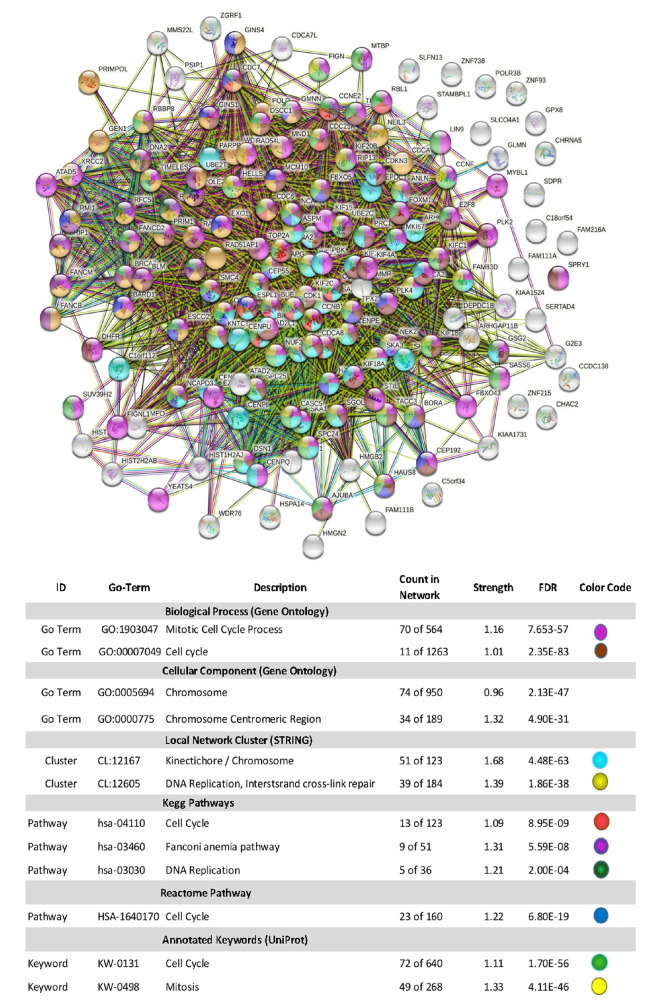
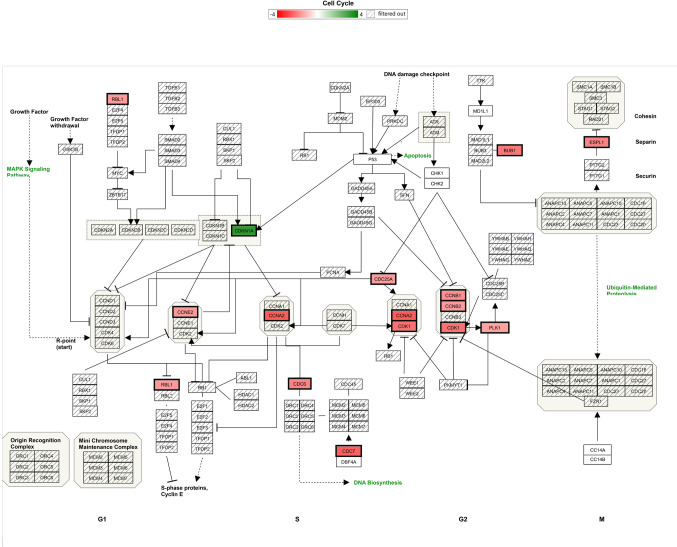
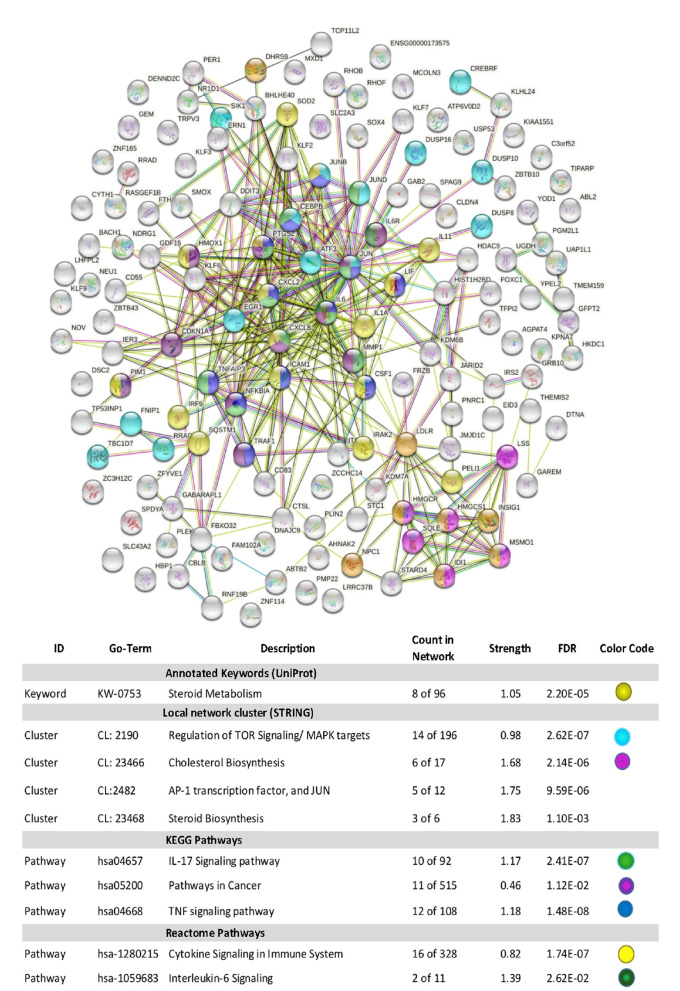
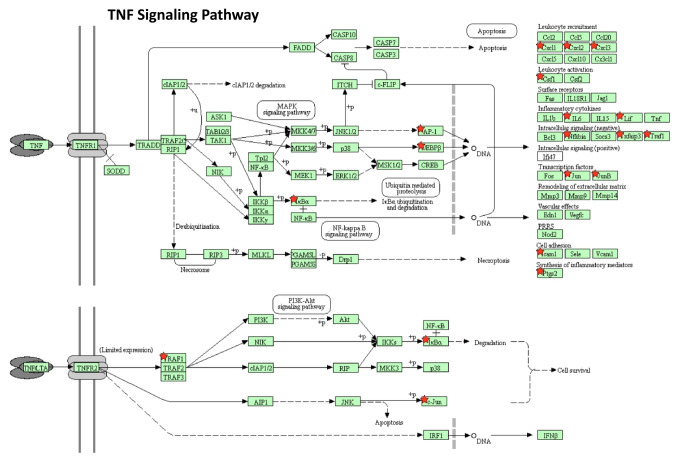
Using Stringdb, all down-regulated genes meeting criteria for WYE (low) were entered, and relational networks were identified (Figure 5). The data show a statistical loss in gene elements that transcribe for cell cycle and mitosis, showing a network FDR value for biological processes analysis (gene ontology) p-Value <0.235e-83. High significance for changes in the chromosome, cell division, DNA replication, and cross strand repair using local network cluster (STRING) also show FDR p-values up to p<4.48e-63. All database platforms pickup up this differential as significant, including the Reactome and Kyoto Encyclopedia of Genes and Genomes (Kegg) pathways, where the latter was also found in the WIKIpathway report of Affymetrix/applied Biosystems transcriptome analysis console report (including up and down DEGs) (Figure 6). Similarly, stringdb analysis was performed on up-regulated DEGs for WYE (Low) (Figure 7), where most significant changes centered on steroid synthesis and cytokine signaling, specifically affecting the TNF-alpha pathway, changes also reflected in the component Kegg overlap map (Figure 8).
Figure 5. Stringdb relational network analysis of down-regulated DEGS caused by WYE (Low) 15 μg/ml vs. untreated controls after meeting selection criteria; -2<x>+2 fold change, p-Value <0.05 and FDR p-Value <0.05. The data in the corresponding table: represent database used, ID #s within a database, description of system altered, count in the network for system, strength of relationship and the false discovery rate (FDR) p-Value. Color codes indicate genes (by symbol) in the string diagram corresponding to the table class description.
Figure 6. Cell cycle Wikipathway analysis report for DEGS caused by WYE (Low) 15 μg/ml vs. untreated controls after meeting selection criteria; -2<x>+2, p-Value <0.05 and FDR p-Value <0.05. DEGS are color coded (red=down), (green=up) with color intensity darker with fold higher fold change. Gene symbol denoted by cross bars /// were not altered.
Figure 7. Stringdb relational network analysis of up-regulated DEGS caused by WYE (Low) 15 μg/mL vs. untreated controls after meeting selection criteria; -2<x>+2, p-Value <0.05 and FDR p-Value <0.05. The data represent database used, ID #s within a database, description of system altered, count in the network for system, strength of relationship and the false discovery rate (FDR) p-Value. Color codes indicate genes (by symbol) in the string diagram corresponding to the table class description.
Figure 8. TNF signaling Kegg Pathway report for up-regulated DEGS caused by WYE (Low) 15 μg/ml vs. untreated controls after meeting selection criteria; -2<x>+2 fold change, p-Value <0.05 and FDR p-Value <0.05. Up-regulated DEGS are demarcated by a red star.
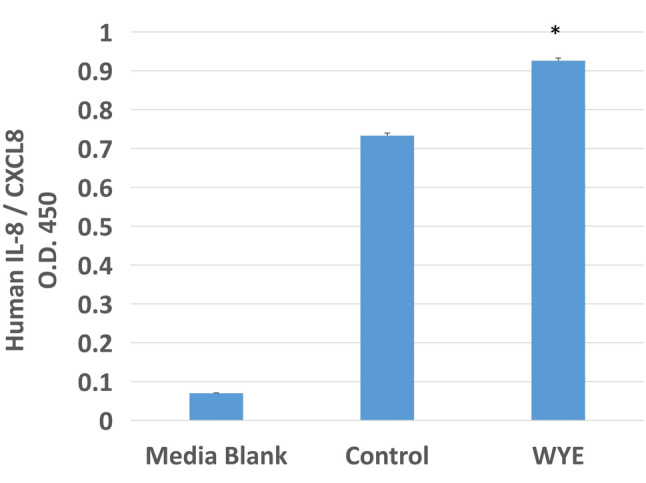
The supernatant of the samples matching the microarray was tested for the presence of cytokines using antibody arrays where the largest up-regulated differential DEG by WYE was for CXCL8/ IL-8, being observed in densitometry values within the anti-body array itself (Figure 9A,B), the Affymetrix microarray (Figure 10) and confirmed by Elisa (Figure 11). Figure 9A,B shows the relative comparison of cytokines released in the supernatant and the mRNA in cells, where the criteria for gene analysis was lowered to less than 2-fold, with a significant p-Value <0.05 and no filter on FDR p-Values. There was a high degree of matching values between proteins released in the supernatant and the mRNA transcription for those proteins in the same pellet sample. In summary, this provides an overall snapshot of the effects of WYE in MDA-MB-231 cells at sub-lethal concentrations.
Figure 9. Supernatant cytokine detection vs. mRNA transcript. Cytokine released in untreated controls vs. (WYE-Low, 15 μg/ml) in supernatants obtained from the same pellets used for microarray analysis and analyzed by a human cytokine array. The data represents matched changes occurring in both the protein antibody with *p-Values <0.05 in both sets, where array values were set at selection criteria; any fold change, p-Value <0.05 and no filter on FDR p-Values (B). There was a high degree of matching values between proteins released in the supernatant and the mRNA produced for those proteins.
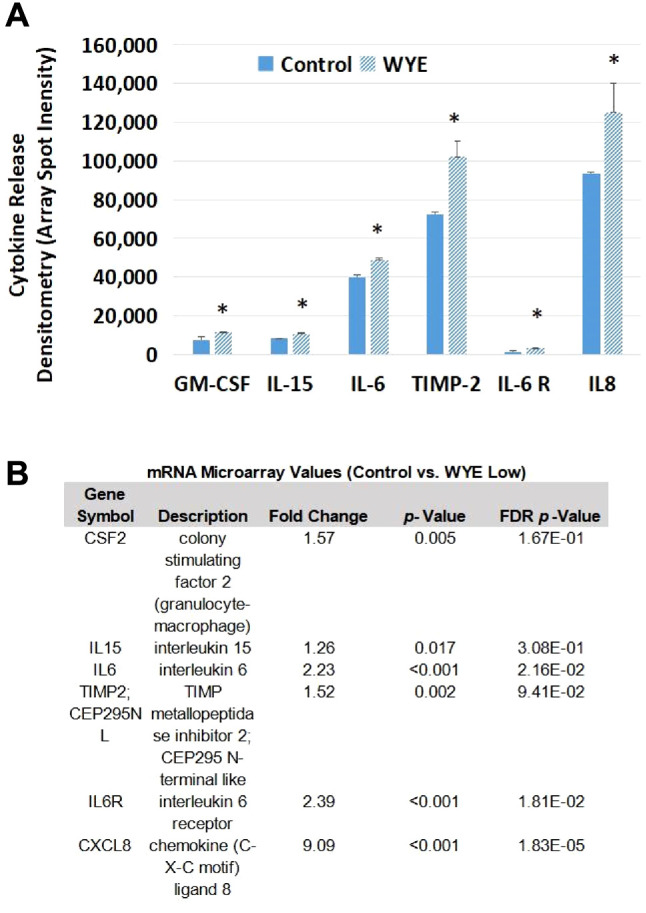
Figure 10. Supernatant cytokine detection. Supernatants collected from pellets used for microarray analysis (WYE-Low, 15ug/ml) show semiquantitative changes in IL-8 by anti-body array (densitometry spot duplicates), demarcated alongside transcriptomic microarray chip spot probe arrays for IL-8 in MDA-MB-231 cells.
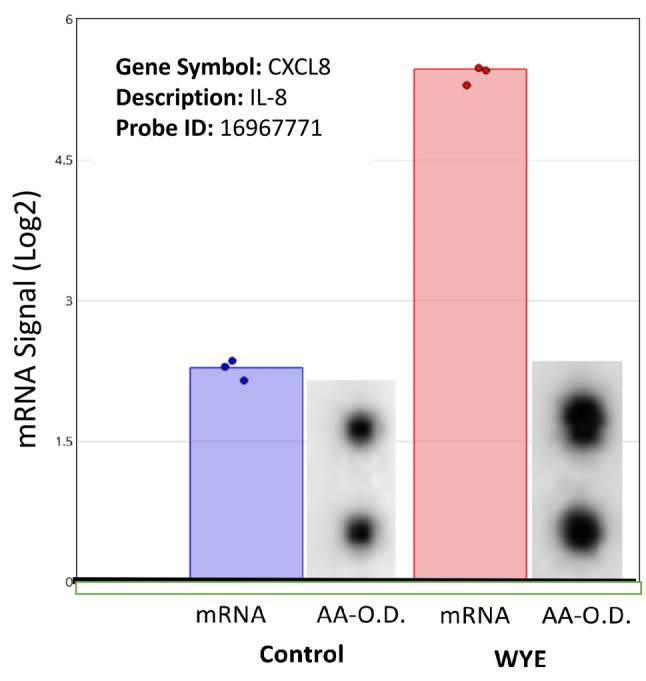
Figure 11. Supernatants collected from pellets used for microarray analysis (WYE-Low, 15 μg/ml) show quantitative changes in IL-8 as determined by ELISA. The data represent the mean±S.E.M for optical densitometry values, n=3 and significant differences between the control and treatment was determined by a students t-test; *p<0.05.
Discussion
In this study, we evaluate the biological and transcriptomic effects of WYE on TNBC cells at sub-lethal concentrations, well below the point involving saponin-mediated cell lysis. WYE shows antiproliferative effects at sub-lethal concentration tantamount to severe downregulation of gene transcripts involved in mitosis and cell division, including transcripts of the following classes: cell division cycle (CDC), cysteine-rich protein 61, connective tissue growth factor, and nephroblastoma overexpressed (CCN), cyclin-dependent kinases (CDK), centromere proteins (CENPs), kinesin superfamily transcripts (KIFS), and polo-like kinases (PLK). These effects were concurrent with single gene up-regulation of the p21 gene CDKN1A, all of which are likely responsible for the observed cytostatic effects of WYE in a 6-day proliferation study. Our findings in this aspect support much of the existing literature for WYE and saponins from the Dioscorea species having the capacity to induce cell-cycle arrest (G2/M) across diverse cancer cell lines with observed downregulation of a similar list of cyclins and cell cycle regulatory transcripts (Cdc25C, Cdk1) (31-34).
While WYE was able to halt cell division effectively at low concentrations (15 μg/ml), the data in this work show an apparent rise in a series of genes that demarcate immune stimulation. As to the compounds responsible for these effects, they could be one or more of the known hundreds of compounds present in the root, such as diterpenes, phenolics, cyanidins, quinones, methyl parvifloside, trigofoenoside A-1, protodeltonin, deltonin, glucosidodeltonin, zingiberensis I, methylprotodioscin, zingiberensis, dioscin, prosapogenin, dioscoreavillosides A and B, diarylheptanoids or possibly lipidated steroid saponins (6).
To our knowledge, this is the first scientific report describing the pro-inflammatory immune-stimulating properties of WYE in a cancer cell line, amongst what appears to be an absence in the literature of similar type studies. Meanwhile, for over 60 years, the research literature has been well established for describing the effects of low-dose saponins as immune-stimulating vaccine adjuvants, which carry out the sole purpose of stimulating a strong, long-lasting learned immune response to administered vaccine antigens. Saponin adjuvants bear a similar molecular similarity to known saponins in WYE, some of which include Quil A, QS-21 from Quillaja, and saponins in the Momordica (35), Glycyrrhiza (36), and the Dioscorea botanical species itself (37). Botanically derived saponin adjuvants boost the immune system by increasing the Th1/Th2 cell-mediated response, antibody production, targeted cytotoxic T-CD4+ cell response with concurrent stimulation of a variety of cytokines [IL-2, IL-10, IL-12 (p70) and IFN-γ] (20,38-44). Yet, interestingly, most of all of the studies on saponin adjuvants have been carried out only in the presence of the antigenic substance containing the vaccine. To add to this gap in the literature, only a few studies have explored the immune-stimulating properties of saponin-rich herbs, which describe an effect on the innate immune response by crude extracts or compounds within Dioscorea species. These changes include capacity to stimulate phagocytosis in macrophages, enhance natural killer (NK) cell activity, activate Toll-like receptor 4 (TLR4) and activate downstream signaling pathways (ERK/JNK, and p38) involving cytokine release (e.g., IL-2, IFN-γ, TNF-α, IL-1b, and IL-6) (45-47). Similarly, Dioscorea glycoproteins alone can trigger infiltration and recruitment of macrophages, lymphocytes, neutrophils, and monocytes while at the same time augmenting NK cytotoxic cell response (48,49). There is some remote similarity in the manner in which biological responses to the FDA approved saponin adjuvant used for the zoster vaccine (50) AS01 (the QS-21 saponin) works to activate the immune system, such as acting on TLR4 signaling, however, reports central around activation of the adaptive immune response (38,51-53). In turn, there are also studies showing that other types of saponin-rich plants can stimulate both the innate and adaptive immune response by activation of TLR4, NK cells, humoral and cell-mediated immunity, macrophage phagocytosis, cytokine and immunoglobulin production, and T CD8+ cell-mediated anticancer immune response, such as the case for Codonopsis pilosula (54,55) and Astragalus (56-58).
While data in this work show WYE to evoke some immune response in the cancer cells itself, the impact of this remains speculative. We cannot exclude the possibility that it could also worsen existing cancers with an inflammatory component. In brief, the immune system is a double-edged sword as it plays a role in both the destruction of/and pathological advancement of cancer. Initiation of cancer can occur from many injurious environmental elements, including; chronic persistent inflammation, exposure to pathogens, irritants, toxins with greater vulnerability occurring in immunocompromised individuals. There is a delicate balance between immunosuppression (under-active) and inflammation (over-active) which controls the initial susceptibility to cancer and various infections.
A healthy immune system will recognize and destroy malignant cells on demand. In established cancers, the host immune system no longer recognizes malignant cells (immune escape) but instead supports the survival, proliferation, and metastasis of cancer. This is carried out by the formation of a protective inflammatory barrier within the tumor microenvironment (TME) (59,60) which fosters cancer growth while at the same time suppresses the body’s immune system to recognize and target cancer (61). The elevation of cytokines released by the tumor tissue itself could worsen pre-existing cancers, driving inward trafficking of leukocyte subpopulations such as monocytes toward the tumor (62), which along with acidity, can polarize and mature into M2- tumor-associated macrophages (TAMS) (63,64). The M2 tumor-promoting phenotype is largely immunosuppressive, aligning with the increased presence of cytokines (IL-6, IL-8, IL-10), COX2, cathepsins, IL-1A, and MMPS within the TME, creating a highly inflammatory aggressive breast cancer with poor clinical outcome (65-72). In the current work, we showed that WYE at very low concentrations, in theory, could perpetuate the M2 phenotype by establishing tumor cell production of COX2, and the release of cathepsins, MMP1, IL-8, IL-6, IL-1A, and CCL2, the consequences of which would be counter-indicated and harmful for cancer patients.
While on the one hand, the WYE used in this study provokes a rise in aggravating tumor cytokines, paradoxically compounds in the Dioscorea species are known to augment the body’s natural capacity to target and destroy tumor cells, as described above. The ability to redirect the body to destroy its own tumor cells is the aim of current-day research in developing immune-modulating anticancer drugs. These therapies aim to restore the body’s natural immune killing capacity by a) penetrating through the TME and b) activating tumor recognition and destruction by the body’s tumor CD8+ T cytotoxic and NK. Cells, both being used in adoptive immune therapies (AIT) (73,74). While there are most certainly natural compounds unidentified to date that can do this, at the current time, AITs involves using the patient’s blood to derive peripheral blood mononuclear cells (PBMCs), which are genetically engineered or modified then reinfused back into the same patient to target cancer (59,75). However, it would suffice to say that any agent, drug, or process can mimic the effect of AITs to boost the body’s own NK cells (76) cytotoxic T (CD8+)/ Th (CD4+), antigen-presenting dendritic cells (DCs)/cytokine-induced killer (CIK) cells (DC-CIK cells therapies) (77-79) would, in theory, establish long term immunity and positive patient outcome including for individuals with stage IV breast and TNBC cancers (80-83).
In conclusion, this work provides an overall transcriptomic analysis of WYE at sub-lethal concentrations, corroborating existing evidence by demonstrating its cytostatic effects and establishing a unique immune-stimulatory response. This work emphasizes the need for future research to investigate the immune stimulatory effects of saponin-rich herbs as it relates to cancer prevention and/ or treatment.
Availability of Data and Material
The dataset has been deposited to NIH. Gene Expression Omnibus located at: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc= GSE180621.
Conflicts of Interest
The Authors declare that they have no conflicts of interest.
Authors’ Contributions
EM and AA conducted studies on cell viability, saponins, antibody arrays, ELISA, and microarray. SDR and KS were involved with troubleshooting, literature review, manuscript preparation, and KS oversaw and guided this project.
Acknowledgements
This research was supported by the National Institute of Minority Health and Health Disparities of the National Institutes of Health through Grant Number U54 MD 007582 and Grant Number P20 MD 006738
References
- 1.Park MK, Kwon HY, Ahn WS, Bae S, Rhyu MR, Lee Y. Estrogen activities and the cellular effects of natural progesterone from wild yam extract in mcf-7 human breast cancer cells. Am J Chin Med. 2009;37(1):159–167. doi: 10.1142/S0192415X09006746. [DOI] [PubMed] [Google Scholar]
- 2.Aumsuwan P, Khan SI, Khan IA, Avula B, Walker LA, Helferich WG, Katzenellenbogen BS, Dasmahapatra AK. Evaluation of wild yam (Dioscorea villosa) root extract as a potential epigenetic agent in breast cancer cells. In Vitro Cell Dev Biol Anim. 2015;51(1):59–71. doi: 10.1007/s11626-014-9807-5. [DOI] [PubMed] [Google Scholar]
- 3.Depypere HT, Comhaire FH. Herbal preparations for the menopause: beyond isoflavones and black cohosh. Maturitas. 2014;77(2):191–194. doi: 10.1016/j.maturitas.2013.11.001. [DOI] [PubMed] [Google Scholar]
- 4.Mazzio E, Badisa R, Mack N, Deiab S, Soliman KF. High throughput screening of natural products for anti-mitotic effects in MDA-MB-231 human breast carcinoma cells. Phytother Res. 2014;28(6):856–867. doi: 10.1002/ptr.5065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chiang CT, Way TD, Tsai SJ, Lin JK. Diosgenin, a naturally occurring steroid, suppresses fatty acid synthase expression in HER2-overexpressing breast cancer cells through modulating Akt, mTOR and JNK phosphorylation. FEBS Lett. 2007;581(30):5735–5742. doi: 10.1016/j.febslet.2007.11.021. [DOI] [PubMed] [Google Scholar]
- 6.Salehi B, Sener B, Kilic M, Sharifi-Rad J, Naz R, Yousaf Z, Mudau FN, Fokou PVT, Ezzat SM, El Bishbishy MH, Taheri Y, Lucariello G, Durazzo A, Lucarini M, Suleria HAR, Santini A. Dioscorea plants: A genus rich in vital nutra-pharmaceuticals-a review. Iran J Pharm Res. 2019;18(Suppl1):68–89. doi: 10.22037/ijpr.2019.112501.13795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.He Z, Chen H, Li G, Zhu H, Gao Y, Zhang L, Sun J. Diosgenin inhibits the migration of human breast cancer MDA-MB-231 cells by suppressing Vav2 activity. Phytomedicine. 2014;21(6):871–876. doi: 10.1016/j.phymed.2014.02.002. [DOI] [PubMed] [Google Scholar]
- 8.Bhuvanalakshmi G, Basappa , Rangappa KS, Dharmarajan A, Sethi G, Kumar AP, Warrier S. Breast cancer stem-like cells are inhibited by diosgenin, a steroidal saponin, by the attenuation of the Wnt β-catenin signaling via the Wnt antagonist secreted frizzled related protein-4. Front Pharmacol. 2017;8:124. doi: 10.3389/fphar.2017.00124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhao J, Xu Y, Wang C, Ding Y, Chen M, Wang Y, Peng J, Li L, Lv L. Soluplus/TPGS mixed micelles for dioscin delivery in cancer therapy. Drug Dev Ind Pharm. 2017;43(7):1197–1204. doi: 10.1080/03639045.2017.1304956. [DOI] [PubMed] [Google Scholar]
- 10.Liao WL, Lin JY, Shieh JC, Yeh HF, Hsieh YH, Cheng YC, Lee HJ, Shen CY, Cheng CW. Induction of G2/M phase arrest by diosgenin via activation of Chk1 kinase and Cdc25C regulatory pathways to promote apoptosis in human breast cancer cells. Int J Mol Sci. 2019;21(1):172. doi: 10.3390/ijms21010172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Masood-Ur-Rahman , Mohammad Y, Fazili KM, Bhat KA, Ara T. Synthesis and biological evaluation of novel 3-O-tethered triazoles of diosgenin as potent antiproliferative agents. Steroids. 2017;118:1–8. doi: 10.1016/j.steroids.2016.11.003. [DOI] [PubMed] [Google Scholar]
- 12.Yeager JF. Hemolysis by saponin and sodium taurocholate, with special reference to the series of Ryvosh. J Gen Physiol. 1928;11(6):779–787. doi: 10.1085/jgp.11.6.779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Efimova SS, Ostroumova OS. Is the membrane lipid matrix a key target for action of pharmacologically active plant saponins. Int J Mol Sci. 2021;22(6):3167. doi: 10.3390/ijms22063167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Orczyk M, Wojciechowski K, Brezesinski G. The influence of steroidal and triterpenoid saponins on monolayer models of the outer leaflets of human erythrocytes, E. coli and S. cerevisiae cell membranes. J Colloid Interface Sci. 2020;563:207–217. doi: 10.1016/j.jcis.2019.12.014. [DOI] [PubMed] [Google Scholar]
- 15.Malabed R, Hanashima S, Murata M, Sakurai K. Interactions of OSW-1 with lipid bilayers in comparison with digitonin and soyasaponin. Langmuir. 2020;36(13):3600–3610. doi: 10.1021/acs.langmuir.9b03957. [DOI] [PubMed] [Google Scholar]
- 16.Verstraeten SL, Albert M, Paquot A, Muccioli GG, Tyteca D, Mingeot-Leclercq MP. Membrane cholesterol delays cellular apoptosis induced by ginsenoside Rh2, a steroid saponin. Toxicol Appl Pharmacol. 2018;352:59–67. doi: 10.1016/j.taap.2018.05.014. [DOI] [PubMed] [Google Scholar]
- 17.Chen M, Balhara V, Jaimes Castillo AM, Balsevich J, Johnston LJ. Interaction of saponin 1688 with phase separated lipid bilayers. Biochim Biophys Acta Biomembr. 2017;1859(7):1263–1272. doi: 10.1016/j.bbamem.2017.03.024. [DOI] [PubMed] [Google Scholar]
- 18.Xu XH, Li T, Fong CM, Chen X, Chen XJ, Wang YT, Huang MQ, Lu JJ. Saponins from Chinese medicines as anticancer agents. Molecules. 2016;21(10):1326. doi: 10.3390/molecules21101326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sudji IR, Subburaj Y, Frenkel N, García-Sáez AJ, Wink M. Membrane disintegration caused by the steroid saponin digitonin is related to the presence of cholesterol. Molecules. 2015;20(11):20146–20160. doi: 10.3390/molecules201119682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lacaille-Dubois MA, Wagner H. A review of the biological and pharmacological activities of saponins. Phytomedicine. 1996;2(4):363–386. doi: 10.1016/S0944-7113(96)80081-X. [DOI] [PubMed] [Google Scholar]
- 21.Koczurkiewicz P, Klaś K, Grabowska K, Piska K, Rogowska K, Wójcik-Pszczoła K, Podolak I, Galanty A, Michalik M, Pękala E. Saponins as chemosensitizing substances that improve effectiveness and selectivity of anticancer drug-Minireview of in vitro studies. Phytother Res. 2019;33(9):2141–2151. doi: 10.1002/ptr.6371. [DOI] [PubMed] [Google Scholar]
- 22.Tong QY, He Y, Zhao QB, Qing Y, Huang W, Wu XH. Cytotoxicity and apoptosis-inducing effect of steroidal saponins from Dioscorea zingiberensis Wright against cancer cells. Steroids. 2012;77(12):1219–1227. doi: 10.1016/j.steroids.2012.04.019. [DOI] [PubMed] [Google Scholar]
- 23.Jaiaree N, Itharat A, Kumapava K. Cytotoxic saponin against lung cancer cells from Dioscorea birmanica Prain & Burkill. J Med Assoc Thai. 2010;93(Suppl 7):S192–S197. [PubMed] [Google Scholar]
- 24.Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012;9(7):671–675. doi: 10.1038/nmeth.2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jones KH, Senft JA. An improved method to determine cell viability by simultaneous staining with fluorescein diacetate-propidium iodide. J Histochem Cytochem. 1985;33(1):77–79. doi: 10.1177/33.1.2578146. [DOI] [PubMed] [Google Scholar]
- 26.Crowley LC, Christensen ME, Waterhouse NJ. Measuring mitochondrial transmembrane potential by TMRE staining. Cold Spring Harb Protoc. 2016;2016(12) doi: 10.1101/pdb.prot087361. [DOI] [PubMed] [Google Scholar]
- 27.McMillian MK, Li L, Parker JB, Patel L, Zhong Z, Gunnett JW, Powers WJ, Johnson MD. An improved resazurin-based cytotoxicity assay for hepatic cells. Cell Biol Toxicol. 2002;18(3):157–173. doi: 10.1023/a:1015559603643. [DOI] [PubMed] [Google Scholar]
- 28.Szklarczyk D, Franceschini A, Kuhn M, Simonovic M, Roth A, Minguez P, Doerks T, Stark M, Muller J, Bork P, Jensen LJ, von Mering C. The STRING database in 2011: functional interaction networks of proteins, globally integrated and scored. Nucleic Acids Res. 2011;39(Database issue):D561–D568. doi: 10.1093/nar/gkq973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Szklarczyk D, Gable AL, Nastou KC, Lyon D, Kirsch R, Pyysalo S, Doncheva NT, Legeay M, Fang T, Bork P, Jensen LJ, von Mering C. The STRING database in 2021: customizable protein-protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res. 2021;49(D1):D605–D612. doi: 10.1093/nar/gkaa1074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Huang DW, Sherman BT, Tan Q, Collins JR, Alvord WG, Roayaei J, Stephens R, Baseler MW, Lane HC, Lempicki RA. The DAVID Gene Functional Classification Tool: a novel biological module-centric algorithm to functionally analyze large gene lists. Genome Biol. 2007;8(9):R183. doi: 10.1186/gb-2007-8-9-r183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Duangprompo W, Aree K, Itharat A, Hansakul P. Effects of 5,6-dihydroxy-2,4-dimethoxy-9,10-dihydrophenanthrene on G2/M cell cycle arrest and apoptosis in human lung carcinoma cells. Am J Chin Med. 2016;44(7):1473–1490. doi: 10.1142/S0192415X16500828. [DOI] [PubMed] [Google Scholar]
- 32.Xie YL, Fan M, Jiang RM, Wang ZL, Li Y. Deltonin induced both apoptosis and autophagy in head and neck squamous carcinoma FaDu cell. Neoplasma. 2015;62(3):419–431. doi: 10.4149/neo_2015_050. [DOI] [PubMed] [Google Scholar]
- 33.Wang G, Chen H, Huang M, Wang N, Zhang J, Zhang Y, Bai G, Fong WF, Yang M, Yao X. Methyl protodioscin induces G2/M cell cycle arrest and apoptosis in HepG2 liver cancer cells. Cancer Lett. 2006;241(1):102–109. doi: 10.1016/j.canlet.2005.10.050. [DOI] [PubMed] [Google Scholar]
- 34.Li X, Qu Z, Jing S, Li X, Zhao C, Man S, Wang Y, Gao W. Dioscin-6’-O-acetate inhibits lung cancer cell proliferation via inducing cell cycle arrest and caspase-dependent apoptosis. Phytomedicine. 2019;53:124–133. doi: 10.1016/j.phymed.2018.09.033. [DOI] [PubMed] [Google Scholar]
- 35.Wang P, Ding X, Kim H, Michalek SM, Zhang P. Structural effect on adjuvanticity of saponins. J Med Chem. 2020;63(6):3290–3297. doi: 10.1021/acs.jmedchem.9b02063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Alexyuk PG, Bogoyavlenskiy AP, Alexyuk MS, Turmagambetova AS, Zaitseva IA, Omirtaeva ES, Berezin VE. Adjuvant activity of multimolecular complexes based on Glycyrrhiza glabra saponins, lipids, and influenza virus glycoproteins. Arch Virol. 2019;164(7):1793–1803. doi: 10.1007/s00705-019-04273-2. [DOI] [PubMed] [Google Scholar]
- 37.Wei WC, Wang JH, Aravindaram K, Wang SJ, Hsu CC, Li CJ, Wen CC, Sheu JH, Yang NS. Polysaccharides from dioscorea (shān yào) and other phytochemicals enhance antitumor effects induced by DNA vaccine against melanoma. J Tradit Complement Med. 2014;4(1):42–48. doi: 10.4103/2225-4110.124342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wang P. Natural and synthetic saponins as vaccine adjuvants. Vaccines (Basel) 2021;9(3):222. doi: 10.3390/vaccines9030222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tian JH, Patel N, Haupt R, Zhou H, Weston S, Hammond H, Logue J, Portnoff AD, Norton J, Guebre-Xabier M, Zhou B, Jacobson K, Maciejewski S, Khatoon R, Wisniewska M, Moffitt W, Kluepfel-Stahl S, Ekechukwu B, Papin J, Boddapati S, Jason Wong C, Piedra PA, Frieman MB, Massare MJ, Fries L, Bengtsson KL, Stertman L, Ellingsworth L, Glenn G, Smith G. SARS-CoV-2 spike glycoprotein vaccine candidate NVX-CoV2373 immunogenicity in baboons and protection in mice. Nat Commun. 2021;12(1):372. doi: 10.1038/s41467-020-20653-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Richou R, Jensen R, Belin C. [Research on saponin, an adjuvant substance which stimulates immunity. I] Rev Immunol Ther Antimicrob. 1964;28:49–62. [PubMed] [Google Scholar]
- 41.Kensil CR. Saponins as vaccine adjuvants. Crit Rev Ther Drug Carrier Syst. 1996;13(1-2):1–55. [PubMed] [Google Scholar]
- 42.Wang P, Škalamera Đ, Sui X, Zhang P, Michalek SM. Synthesis and evaluation of QS-7-based vaccine adjuvants. ACS Infect Dis. 2019;5(6):974–981. doi: 10.1021/acsinfecdis.9b00039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Aguado-Martínez A, Basto AP, Müller J, Balmer V, Manser V, Leitão A, Hemphill A. N-terminal fusion of a toll-like receptor 2-ligand to a Neospora caninum chimeric antigen efficiently modifies the properties of the specific immune response. Parasitology. 2016;143(5):606–616. doi: 10.1017/S0031182016000056. [DOI] [PubMed] [Google Scholar]
- 44.Morein B, Hu KF, Abusugra I. Current status and potential application of ISCOMs in veterinary medicine. Adv Drug Deliv Rev. 2004;56(10):1367–1382. doi: 10.1016/j.addr.2004.02.004. [DOI] [PubMed] [Google Scholar]
- 45.Hao Hao LX, Zhao XH. Report - In vitro immune potentials of a water-soluble polysaccharide extract from Dioscorea opposita planted in Henan Province, China. Pak J Pharm Sci. 2017;30(4):1383–1388. [PubMed] [Google Scholar]
- 46.Lee BH, Hsu WH, Liao TH, Pan TM. Inhibition of leukemia proliferation by a novel polysaccharide identified from Monascus-fermented dioscorea via inducing differentiation. Food Funct. 2012;3(7):758–764. doi: 10.1039/c2fo30026e. [DOI] [PubMed] [Google Scholar]
- 47.Panthong S, Ruangnoo S, Thongdeeying P, Sriwanthana B, Itharat A. Immunomodulatory activity of Dioscorea membranacea Pierre rhizomes and of its main active constituent Dioscorealide B. BMC Complement Altern Med. 2014;14:403. doi: 10.1186/1472-6882-14-403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Huong PT, Jeon YJ. Macrophage activation by glycoprotein isolated from Dioscorea batatas. Toxicol Res. 2011;27(3):167–172. doi: 10.5487/TR.2011.27.3.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Huong PT, Lee CH, Li MH, Lee MY, Kim JK, Lee SM, Seon JH, Lee DC, Jeon YJ. Characterization and immunopotentiating effects of the glycoprotein isolated from dioscorea batatas. Korean J Physiol Pharmacol. 2011;15(2):101–106. doi: 10.4196/kjpp.2011.15.2.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Giordano G, Segal L, Prinsen M, Wijnands MV, Garçon N, Destexhe E. Non-clinical safety assessment of single and repeated administration of gE/AS01 zoster vaccine in rabbits. J Appl Toxicol. 2017;37(2):132–141. doi: 10.1002/jat.3329. [DOI] [PubMed] [Google Scholar]
- 51.Shi S, Zhu H, Xia X, Liang Z, Ma X, Sun B. Vaccine adjuvants: Understanding the structure and mechanism of adjuvanticity. Vaccine. 2019;37(24):3167–3178. doi: 10.1016/j.vaccine.2019.04.055. [DOI] [PubMed] [Google Scholar]
- 52.Fochesato M, Dendouga N, Boxus M. Comparative preclinical evaluation of AS01 versus other Adjuvant Systems in a candidate herpes zoster glycoprotein E subunit vaccine. Hum Vaccin Immunother. 2016;12(8):2092–2095. doi: 10.1080/21645515.2016.1154247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sharma R, Palanisamy A, Dhama K, Mal G, Singh B, Singh KP. Exploring the possible use of saponin adjuvants in COVID-19 vaccine. Hum Vaccin Immunother. 2020;16(12):2944–2953. doi: 10.1080/21645515.2020.1833579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.He LX, Zhang ZF, Sun B, Chen QH, Liu R, Ren JW, Wang JB, Li Y. Sea cucumber (Codonopsis pilosula) oligopeptides: immunomodulatory effects based on stimulating Th cells, cytokine secretion and antibody production. Food Funct. 2016;7(2):1208–1216. doi: 10.1039/c5fo01480h. [DOI] [PubMed] [Google Scholar]
- 55.Peng Y, Song Y, Wang Q, Hu Y, He Y, Ren D, Wu L, Liu S, Cong H, Zhou H. In vitro and in vivo immunomodulatory effects of fucoidan compound agents. Int J Biol Macromol. 2019;127:48–56. doi: 10.1016/j.ijbiomac.2018.12.197. [DOI] [PubMed] [Google Scholar]
- 56.Wang J, Tong X, Li P, Liu M, Peng W, Cao H, Su W. Bioactive components on immuno-enhancement effects in the traditional Chinese medicine Shenqi Fuzheng Injection based on relevance analysis between chemical HPLC fingerprints and in vivo biological effects. J Ethnopharmacol. 2014;155(1):405–415. doi: 10.1016/j.jep.2014.05.038. [DOI] [PubMed] [Google Scholar]
- 57.Bamodu OA, Kuo KT, Wang CH, Huang WC, Wu ATH, Tsai JT, Lee KY, Yeh CT, Wang LS. Astragalus polysaccharides (PG2) enhances the M1 polarization of macrophages, functional maturation of dendritic cells, and T cell-mediated anticancer immune responses in patients with lung cancer. Nutrients. 2019;11(10):2264. doi: 10.3390/nu11102264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Tian Y, Li X, Li H, Lu Q, Sun G, Chen H. Astragalus mongholicus regulate the Toll-like-receptor 4 meditated signal transduction of dendritic cells to restrain stomach cancer cells. Afr J Tradit Complement Altern Med. 2014;11(3):92–96. doi: 10.4314/ajtcam.v11i3.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Salemme V, Centonze G, Cavallo F, Defilippi P, Conti L. The crosstalk between tumor cells and the immune microenvironment in breast cancer: Implications for immunotherapy. Front Oncol. 2021;11:610303. doi: 10.3389/fonc.2021.610303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Steven A, Seliger B. The role of immune escape and immune cell infiltration in breast cancer. Breast Care (Basel) 2018;13(1):16–21. doi: 10.1159/000486585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Gross S, Walden P. Immunosuppressive mechanisms in human tumors: why we still cannot cure cancer. Immunol Lett. 2008;116(1):7–14. doi: 10.1016/j.imlet.2007.11.012. [DOI] [PubMed] [Google Scholar]
- 62.Li X, Yao W, Yuan Y, Chen P, Li B, Li J, Chu R, Song H, Xie D, Jiang X, Wang H. Targeting of tumour-infiltrating macrophages via CCL2/CCR2 signalling as a therapeutic strategy against hepatocellular carcinoma. Gut. 2017;66(1):157–167. doi: 10.1136/gutjnl-2015-310514. [DOI] [PubMed] [Google Scholar]
- 63.Heiskala M, Leidenius M, Joensuu K, Heikkilä P. High expression of CCL2 in tumor cells and abundant infiltration with CD14 positive macrophages predict early relapse in breast cancer. Virchows Arch. 2019;474(1):3–12. doi: 10.1007/s00428-018-2461-7. [DOI] [PubMed] [Google Scholar]
- 64.Ohashi T, Inoue N, Aoki M. The Warburg Effect and M2 Macrophage Polarization in Head and Neck Cancer. Gan To Kagaku Ryoho. 2020;47(1):6–10. [PubMed] [Google Scholar]
- 65.Vendramini-Costa DB, Carvalho JE. Molecular link mechanisms between inflammation and cancer. Curr Pharm Des. 2012;18(26):3831–3852. doi: 10.2174/138161212802083707. [DOI] [PubMed] [Google Scholar]
- 66.Mohamed MM, El-Ghonaimy EA, Nouh MA, Schneider RJ, Sloane BF, El-Shinawi M. Cytokines secreted by macrophages isolated from tumor microenvironment of inflammatory breast cancer patients possess chemotactic properties. Int J Biochem Cell Biol. 2014;46:138–147. doi: 10.1016/j.biocel.2013.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Sousa S, Brion R, Lintunen M, Kronqvist P, Sandholm J, Mönkkönen J, Kellokumpu-Lehtinen PL, Lauttia S, Tynninen O, Joensuu H, Heymann D, Määttä JA. Human breast cancer cells educate macrophages toward the M2 activation status. Breast Cancer Res. 2015;17:101. doi: 10.1186/s13058-015-0621-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Myers KV, Pienta KJ, Amend SR. Cancer cells and M2 macrophages: Cooperative invasive ecosystem engineers. Cancer Control. 2020;27(1):1073274820911058. doi: 10.1177/1073274820911058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Yang M, Liu J, Shao J, Qin Y, Ji Q, Zhang X, Du J. Cathepsin S-mediated autophagic flux in tumor-associated macrophages accelerate tumor development by promoting M2 polarization. Mol Cancer. 2014;13:43. doi: 10.1186/1476-4598-13-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Xu J, Yu Y, He X, Niu N, Li X, Zhang R, Hu J, Ma J, Yu X, Sun Y, Ni H, Wang F. Tumor-associated macrophages induce invasion and poor prognosis in human gastric cancer in a cyclooxygenase-2/MMP9-dependent manner. Am J Transl Res. 2019;11(9):6040–6054. [PMC free article] [PubMed] [Google Scholar]
- 71.Vinnakota K, Zhang Y, Selvanesan BC, Topi G, Salim T, Sand-Dejmek J, Jönsson G, Sjölander A. M2-like macrophages induce colon cancer cell invasion via matrix metalloproteinases. J Cell Physiol. 2017;232(12):3468–3480. doi: 10.1002/jcp.25808. [DOI] [PubMed] [Google Scholar]
- 72.You D, Jeong Y, Yoon SY, Kim SA, Lo E, Kim SW, Lee JE, Nam SJ, Kim S. Entelon® (Vitis vinifera seed extract) prevents cancer metastasis via the downregulation of Interleukin-1 alpha in triple-negative breast cancer cells. Molecules. 2021;26(12):3644. doi: 10.3390/molecules26123644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Seliger B, Massa C. Immune therapy resistance and immune escape of tumors. Cancers (Basel) 2021;13(3):551. doi: 10.3390/cancers13030551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Sanchez CE, Dowlati EP, Geiger AE, Chaudhry K, Tovar MA, Bollard CM, Cruz CRY. NK cell adoptive immunotherapy of cancer: Evaluating recognition strategies and overcoming limitations. Transplant Cell Ther. 2021;27(1):21–35. doi: 10.1016/j.bbmt.2020.09.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Tonn T, Becker S, Esser R, Schwabe D, Seifried E. Cellular immunotherapy of malignancies using the clonal natural killer cell line NK-92. J Hematother Stem Cell Res. 2001;10(4):535–544. doi: 10.1089/15258160152509145. [DOI] [PubMed] [Google Scholar]
- 76.Biswas BK, Guru SA, Sumi MP, Jamatia E, Gupta RK, Lali P, Konar BC, Saxena A, Mir R. Natural killer cells expanded and preactivated exhibit enhanced antitumor activity against different tumor cells in vitro. Asian Pac J Cancer Prev. 2020;21(6):1595–1605. doi: 10.31557/APJCP.2020.21.6.1595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Abbott RC, Cross RS, Jenkins MR. Finding the keys to the CAR: Identifying novel target antigens for T cell redirection immunotherapies. Int J Mol Sci. 2020;21(2):515. doi: 10.3390/ijms21020515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Sommaggio R, Cappuzzello E, Dalla Pietà A, Tosi A, Palmerini P, Carpanese D, Nicolè L, Rosato A. Adoptive cell therapy of triple negative breast cancer with redirected cytokine-induced killer cells. Oncoimmunology. 2020;9(1):1777046. doi: 10.1080/2162402X.2020.1777046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lee H, Kim YA, Kim Y, Park HS, Seo JH, Lee H, Gong G, Lee HJ. Clinicopathological factors associated with tumor-infiltrating lymphocyte reactivity in breast cancer. Cancer Immunol Immunother. 2020;69(11):2381–2391. doi: 10.1007/s00262-020-02633-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Wang ZX, Cao JX, Wang M, Li D, Cui YX, Zhang XY, Liu JL, Li JL. Adoptive cellular immunotherapy for the treatment of patients with breast cancer: a meta-analysis. Cytotherapy. 2014;16(7):934–945. doi: 10.1016/j.jcyt.2014.02.011. [DOI] [PubMed] [Google Scholar]
- 81.Yang L, Lou M, Liu W, Feng D, Cheng T, Ma J. Amplification of immune cells derived from human peripheral blood and cytotoxicity on MDA-MB-231 breast cancer cells in vitro. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2019;35(8):682–688. [PubMed] [Google Scholar]
- 82.Shi YJ, Ren HY, Cen XN, Dong YJ, Ma MX, Zhao YL, Zhu Y, Yu JR. [Dendritic cells elicit cellular immune response by targeting to capture breast cancer cells] Zhonghua Zhong Liu Za Zhi. 2008;30(2):107–111. [PubMed] [Google Scholar]
- 83.Pan K, Guan XX, Li YQ, Zhao JJ, Li JJ, Qiu HJ, Weng DS, Wang QJ, Liu Q, Huang LX, He J, Chen SP, Ke ML, Zeng YX, Xia JC. Clinical activity of adjuvant cytokine-induced killer cell immunotherapy in patients with post-mastectomy triple-negative breast cancer. Clin Cancer Res. 2014;20(11):3003–3011. doi: 10.1158/1078-0432.CCR-14-0082. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The dataset has been deposited to NIH. Gene Expression Omnibus located at: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc= GSE180621.