Abstract
Objective
Guillain-Barré Syndrome (GBS) is an acute inflammatory polyneuropathy characterized by a rapid progressive symmetric weakness. The GBS is the most common cause of acute flaccid paralysis (AFP) in most parts of the world. This study was carried out to investigate the epidemiological features of GBS in Iranian children.
Materials & Methods
The data were extracted using the AFP surveillance system that is a national screening program to detect all cases of AFP aged 0-15 years around the country. National Population Statistics data and AFP demographic data during 2008-2013 intervals were obtained from the relevant authorities in the Ministry of Health in Iran. The GBS cases were then extracted from the aforementioned database. The Chi-square test and Fisher’s exact test were used for statistical analysis.
Results
A total of 1884 cases of GBS were identified in the study period, and the average annual incidence rate was 1.72 per 100,000 individuals. The highest incidence rate was within the range of 0-5 years. There was no statistically significant relationship between the incidence of GBS and the season in the whole country.
Conclusion
High costs of GBS treatment, morbidity and occasional mortality, and number of new cases, which is estimated to be approximately 300 individuals per year, need the particular attention of the health system.
Key Words: Guillain-Barré Syndrome, Incidence, Children, Epidemiology, Iran
Introduction
Guillain-Barré Syndrome (GBS) is an acute acquired peripheral neuropathy characterized by rapidly developing motor weakness (1). The GBS has an autoimmune etiology and in the two-third of cases is triggered by respiratory or gastrointestinal infections (2, 3). The pathology is an inflammatory disorder affecting the myelin sheath and the axon leading to diffuse demyelination and axonal degeneration (4).
The GBS is the most common cause of subacute and acute flaccid paralysis (AFP) in neonates and children. All age groups can be affected by GBS, and it seems that adults are more susceptible than children. The worldwide incidence of GBS for all ages has been estimated to be within the range of 0.4-2.4 per 100,000 individuals. The annual incidence in children seems to be lower estimated to be within the range of 0.4-1.3 per 100,000 individuals.
The prognosis of GBS in children is generally excellent that is quite different from adults who have higher morbidity and mortality (4). There is no significant difference in GBS incidence among seasons, although a limited number of studies suggested a seasonal variation (1). With this background in mind, the current study aimed to determine the annual incidence of GBS in children, analyze morbidity and mortality rates, and compare these rates according to age, gender, and geographical and seasonal variations.
Materials & Methods
This cross-sectional study was conducted on Iranian children under 15 years of age with GBS within 2008 and 2013. Patients’ information was obtained using the database extracted from the AFP surveillance system, which is designed by the World Health Organization (WHO) to screen children aged 0-15 years with AFP in polio-free countries with a high risk of imported polio. This program is implemented by the Preventable Diseases Department of the Ministry of Health in Iran and is annually evaluated and approved by the WHO.
These AFP cases are classified in each province by regional AFP committees (composed of infection, epidemiologist, and neurologist experts). Then, GBS patients were separated from other AFP cases and analyzed. The GBS children information sheet included patients’ code, name, age, gender, date of birth, date of paralysis, early-onset symptoms of the disease, a remnant of paralysis after 60 days of occurrence, and home address (within 2008 and 2013).
Based on the 2008 census and demographic estimates, the populations of each province and the whole country were obtained from the Statistics National Center for the years 2008-2013. Total Iranian populations were 70,495,882 and 75,149,669 individuals after the 2006 and 2011 censuses, respectively. The numbers of children under 15 years of age were 17,681,629 and 17,561,778 in 2006 and 2011, respectively.
The data were collected from information sheets and entered in SPSS software (version 20) and analyzed. Mean and standard deviation and relative and absolute frequency were used to analyze quantitative and qualitative variables, respectively. Moreover, the Chi-square test and Fisher’s exact test were performed to investigate the association between demographic and epidemiological factors with GBS. A p-value of less than 0.05 was considered statistically significant.
Results
Within 2008 and 2013, 1884 cases of GBS were diagnosed, including 333 cases in 2008, 309 cases in 2009, 322 cases in 2010, 307 cases in 2011, 321 cases in 2012, and 292 cases in 2013 with the mean age values of 4.96±3.54, 5.60±3.96, 5.47±3.77, 5.66±3.97, 5.25±3.54, and 6.05±3.79 years, respectively. Altogether, 1061 (56.3%) and 823 (43.7%) children were male and female, respectively. Furthermore, there was no statistically significant difference between the genders of patients in each year (P=0.587).
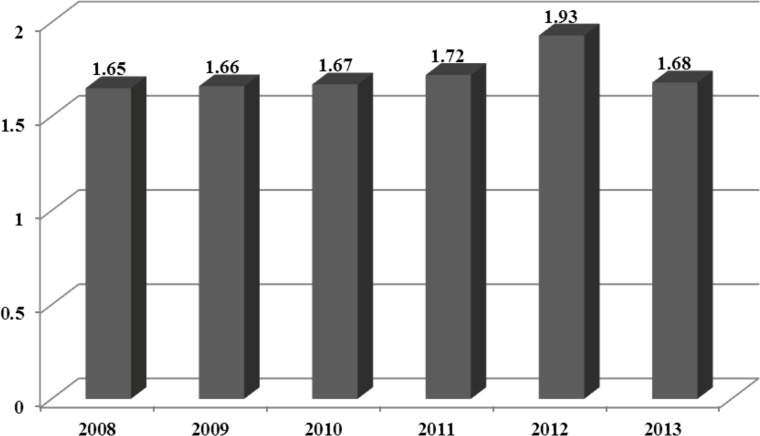
The mean incidence rate of GBS during the study period (2008-2013) was 1.72 per 100,000 individuals in children under 15 years of age. The highest (1.93%) and lowest (1.65%) incidence rates were reported in 2012 and 2008, respectively (Figure 1). Overall, during the study, the investigation of the age distribution of cases showed 4% of cases under 1 year, 50% of cases within 1-5 years, 30% of cases within 5-10 years, and 16% of cases within 10-15 years of age.
Fig 1.
Guillain-Barre syndrome incidence in children under 15 years old during 2008-13
In this study, 579 (30.7%) cases had a fever during the last week before paralysis. Residual motor deficit after 60 days of paralysis onset was in the order of 31.8% in 2008, 34.4% in 2009, 35.1% in 2010, 34.0% in 2011, 27.1% in 2012, and 22.7% in 2013. The rest of the patients, excluding death cases, were recovered before 60 days. Residual paralysis or weakness (at least one muscle group at ≤ grade 6 level) ranged from steppage gait to non-ambulatory state (i.e., bed or chair bound). Grade 6 is the ability to move a muscle against gravity with minimal resistance to force.
Out of 1884 cases, 29 children (1.5%) with GBS died. The results also showed that there was a significant difference between the mortality rate and age group (P=0.024) with maximum mortality in children under 5 years of age; however, there was no significant difference between the mortality rate and the gender of children with GBS (P=0.451; Table 1)
Table 1.
Age Distribution and Gender Influence on Guillain-Barré Syndrome Outcomes in Children Under 15 Years of Age
| Outcome n (%) |
|||||
|---|---|---|---|---|---|
| Alive | Death | Full remission | Residual motor deficit | ||
| Gender | |||||
|
809 (98.3)
1046 (98.6) |
14 (1.7)
15 (1.4) |
576 (67)
729 (68.7) |
248 (33)
332 (31.3) |
|
| Age (year) | |||||
|
72 (96)
922 (97.9) 562 (99.3) 299 (99.3) |
3 (4)
20 (2.1) 4 (0.7) 2 (0.7) |
54 (72)
649 (69) 396 (70) 206 (68.4) |
21 (28)
293 (31) 170 (30) 95 (31.6) |
|
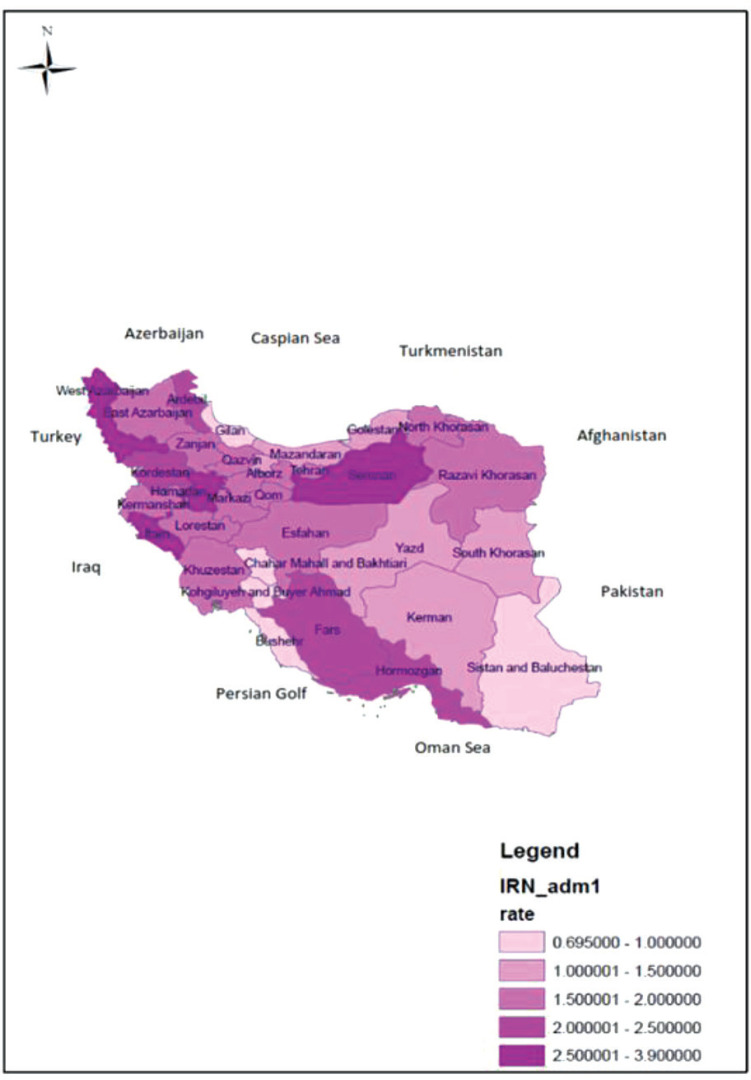
Figure 2 shows the distribution of incidence rates of GBS in different Iranian provinces. According to Figure 2, Semnan (3.7 in 100,000) and Ilam (2.8 in 100,000) have the highest incidence rates of GBS, and Bushehr (0.7 in 100,000) and Gilan (0.8 in 100,000) have the lowest incidence rates of GBS.
Fig 2.
the distribution incidence rate of Guillain Barre Syndrome per 100,000 populations of children less than 15 years old in each province of Iran (2008-2013)
During 6 years of study (2008-2013), the incidence rates of GBS were 1.6 and 1.9 in girls and boys per 100,000 children under 15 years of age, respectively.
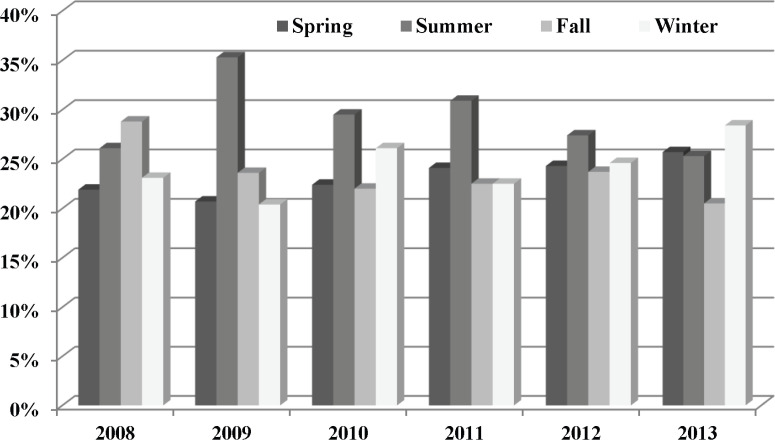
Figure 3 shows seasonal variability of GBS incidence in children illustrating the percentage of patients who were affected during each season based on the study years. The incidence rate of GBS according to the season during 2008-2013 showed no significant difference among the seasons in different years (P=0.157).
Fig 3.
Seasonal variability of GBS incidence in children less than 15 years (2008-2013)
Discussion
This descriptive cross-sectional retrospective study was based on the data from the AFP surveillance system in Iranian children aged 0-15 years with GBS during 2008-2013 intervals. Historically, the overall incidence rate of GBS at all ages was within the range of 0.4/100,000 in Sardinia, Italy, (5) to 4/100,000 in Larimer County, Colorado (6). The incidence of GBS in children (age range: 0-15 years) seems to be lower than that in adults with a range of 0.34-1.34/100,000 per year (1). The highest rate was reported in adult patients above 75 years of age (4.5/100,000) (1).
With 1884 cases of GBS, the present study reported the largest number of patients aged 0-15 years from one country (1), preceded by olive et al. reporting 1678 affected children from Brazil (7). The prospective method of studying GBS incidence is considered to be more reliable, compared to the retrospective one (1). Nevertheless, in the current retrospective study, each case with AFP was reviewed by an expert committee and the diagnosis of GBS was confirmed, minimizing the risk of overestimation of GBS reports. Regarding two national censuses in 2006 and 2011, the total population of the present study is accurate.
The case definition in the current study was based on AFP patients reported from all around the country. The GBS cases were identified after further clinical and electrophysiological studies; this means that the major manifestation was a motor deficit. In addition, theoretically, this study could have missed rare cases of acute sensory neuropathy with only sensory nerves or roots involvement and atypical cases, such as Miller Fisher syndrome that is not presented with flaccid paralysis. However, this fact can have a negligible effect since these atypical cases are rare (less than 5%), and they can only change the incidence of GBS by approximately 0.1 per 100.000 individuals (8, 9).
A recent study from Bangladesh showed an incidence rate of 1.5 to 2.5/100,000 individuals per year in children under 15 years of age (10), indicating a probable increase in childhood GBS incidence. With a mean incidence of 1.72 per 100,000 individuals, the present study supports the aforementioned hypothesis. This presumed rise in incidence could be explained by a better diagnosis of the disease by physicians, a better national registry network, and the emergence of many new environmental factors (i.e., chemical, physical, and microbiological) that can modulate the immune system. However, researchers should wait for performing extensive surveys from other countries to document the possible rise in GBS incidence.
In a 6-year cross-sectional study (2001-2006) in East Azerbaijan, Iran, Barzegar et al. reported an incidence of GBS, in a 0-to-15-year interval, ranging from 0.16 to 4 per 100.000 individuals (11). There was a peak incidence in 2003 (probably after a campylobacter outbreak in the northwest of Iran) with a similar peak in GBS mortality during this year). All studies showed a male predominance in GBS which is unusual in a presumed autoimmune disease. The male: female GBS ratios in all ages were reported as 1.25:1 in the UK (3), 1.7:1 in Western Balkans countries, and 1.1:1 in Spain (12). The male to female ratio was 1.27:1 in the present cohort that is almost similar to those of other countries in all age groups.
Childhood GBS generally has a good prognosis in contrast to adult GBS with higher morbidity and mortality (4). The mortality rate in childhood GBS was 0% in Toronto, Canada (13); nonetheless, there was a mortality rate of 1.5% which could be explained by delay in patients’ referral to tertiary hospitals when sophisticated respiratory and intensive care unit care was available. During an 11-year study period (1989 -1999) in Honduras, the mortality rate in childhood GBS was 7.3%. Most of the mortality cases of the present study occurred in children under 5 years of age. The reason for this age distribution is the non-specificity of symptoms in early childhood, delay in the correct diagnosis, and vulnerability of young children to respiratory failure and autonomic dysfunction (14).
Residual motor deficit after 60 days of paralysis onset annually ranged from 22.7% to 35.1%. In this study, there was no longer follow-up for the patients. Vajsar J et al. reported a persistently decreased muscle power (with at least one muscle group having movement against gravity but with minimal resistance against a force) in 23% of children with GBS during an average 5-year follow-up in Toronto (13).
No significant seasonal variation in GBS incidence was reported in other studies (15-18), although a Brazilian study showed outbreaks in spring and summer (19) and another study in Netherland showed an increase in incidence in winter and June (20). Xiujuan Wu et al. reported a higher incidence of childhood GBS in summer (21). Despite different climates in Iran, there was no significant difference in the seasonal incidence of GBS in the patients of this study.
The age distribution of GBS is bimodal according to some studies, with peaks occurring in young adults and the elderly (2, 22, 23). In the current study, 50% of all GBS cases were within the age range of 1-5 years, for which there was no explanation and no similar results were obtained in other studies.
Acknowledgments
The authors would like to express their gratitude to all colleagues in the Polio Eradication Office in the Health Deputy of the Ministry of Health and Medical Education and all the children with GBS who were cited in this article. In addition, the authors would like to appreciate the consults of Mofid Clinical Research Development Center, Tehran, Iran. IR.SBMU.REC.1394.183
Authors’ Contribution
Tonekaboni Seyed Hassan, Mahmoudi Sussan, Abdollah Gorji Fatemeh, Nejad Biglari Habibe, Taghdiri Mohammad Mahdi, Etemadi Koroush, Mohammad Ghofrani, Abdollah Karimi, and Rezaei Zadeh Rukerd Mohammad contributed to the design and implementation of the study, the analysis of the results, and the writing of the manuscript.
Conflicts of Interest
The authors declare that there is no conflict of interest.
References
- 1.McGrogana A, Madleb GC, Seamanb HE, de Vries CS. The Epidemiology of Guillain-BarréSyndrome Worldwide:a Systematic Literature Review. Neuroepidemiology. 2009;32:150–63. doi: 10.1159/000184748. [DOI] [PubMed] [Google Scholar]
- 2.Govoni V, Granieri E. Epidemiology of the Guillain-Barre syndrome. Curr Opin Neurol. 2001;14:605–613. doi: 10.1097/00019052-200110000-00009. [DOI] [PubMed] [Google Scholar]
- 3.Hughes RAC, Rees JH. Clinical and epidemiologic features of Guillain-Barre syndrome. J Infect Dis. 1997;176 :S92–S98. doi: 10.1086/513793. [DOI] [PubMed] [Google Scholar]
- 4.Rosen AB. Guillain-Barre´ Syndrome. Pediatrics in Review April. 2012;33(4):164–71. doi: 10.1542/pir.33-4-164. [DOI] [PubMed] [Google Scholar]
- 5.Congia S, Melis M, Carboni MA. Epidemiologic and clinical features of the Guillain-Barre´ syndrome in Sardinia in the 1961–1980 periods. Acta Neurol. 1989;11:15–20. [PubMed] [Google Scholar]
- 6.Kaplan JE, Poduska PJ, McIntosh GC, Hopkins RS, Ferguson SW, Schon berger LB. GBS in Lorimer County Colorado: a high incidence area. Neurology. 1985;35:581–4. doi: 10.1212/wnl.35.4.581. [DOI] [PubMed] [Google Scholar]
- 7.Hughes RA, Rees JH. Clinical and epidemiologic features of Guillain-Barre syndrome. JInfect Dis. 1997;176:92–98. doi: 10.1086/513793. [DOI] [PubMed] [Google Scholar]
- 8.Olive JM, Castillo C, Garcia Castro R, de Quadros CA. Epidemiologic study of Guillain-Barre syndrome in children <15 years of age in Latin America. J Infect Dis. 1997;175(suppl):S160–S164. doi: 10.1093/infdis/175.supplement_1.s160. [DOI] [PubMed] [Google Scholar]
- 9.Peric1 S, Milosevic V, Berisavac I, Stojiljkovic O, Bumbasirevic LB, Marjanovic I, et al. Clinical and epidemiological features of Guillain-Barré syndrome in the Western Balkans. Journal of the Peripheral Nervous System. 2014;19:317–321. doi: 10.1111/jns.12096. [DOI] [PubMed] [Google Scholar]
- 10.Islam Zh, Jacobs BC, Islam MB, Mohammad QD, Diorditsa S, Endtz HP. High Incidence of Guillain-Barré Syndrome in Children, Bangladesh. Emerging Infectious Diseases July. 2011;17(7):1317–18. doi: 10.3201/eid1707.101999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Barzegar M, Dastgiri S, Karegarmaher MH, Varshochiani A. Epidemiology of childhood Guillan-Barre syndrome in the north west of Iran. BMC Neurology. 2007;7:1–5. doi: 10.1186/1471-2377-7-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.González-Suárez I, Sanz-Gallego I, Rodríguez de Rivera FJ, Arpa J. Guillain-Barré syndrome: natural history and prognostic factors: a retrospective review of 106 cases. BMC Neurol. 2013;13:95. doi: 10.1186/1471-2377-13-95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Vajsar J, Fehlings D, Stephens D. Long-term outcome in children with Guillain-Barré syndrome. J Pediatr. 2003 Mar;142(3):305–9. doi: 10.1067/mpd.2003.115. [DOI] [PubMed] [Google Scholar]
- 14.Molinero MR, Varon D, Holden KR, Sladky JT, Molina IB, Cleaves F. Epidemiology of Childhood Guillain-Barré Syndrome as a Cause of Acute Flaccid Paralysis in Honduras: 1989–1999. Journal of Child Neurology. 2003 Nov;18(11):741–7. doi: 10.1177/08830738030180110801. [DOI] [PubMed] [Google Scholar]
- 15.Storey E, Cook M, Peppard R, Newton-John H, Byrne E. Guillain-Barre syndrome andrelated conditions in Victorian teaching hospitals 1980–1984. Aust NZ J Med. 1989;19:687–693. doi: 10.1111/j.1445-5994.1989.tb00335.x. [DOI] [PubMed] [Google Scholar]
- 16.Arami MA, Yazdchi M, Khandaghi R. Epidemiology and characteristics of Guillain Barre syndrome in the northwest of Iran. Ann Saudi Med. 2006;26:22–27. doi: 10.5144/0256-4947.2006.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Govoni V, Granieri E, Tola MR, Casetta I, Ruppi P, Vaghi L. The frequency of clinical variants of Guillain-Barre syndrome in Ferrara, Italy. J Neurol. 1999;246:1010–1014. doi: 10.1007/s004150050505. [DOI] [PubMed] [Google Scholar]
- 18.Sedano MJ, Calleja J, Canga E, Berciano J. Guillain-Barre syndrome in Cantabria, Spain: an epidemiological and clinical study. Acta Neurol Scand. 1994;89:287–292. doi: 10.1111/j.1600-0404.1994.tb01682.x. [DOI] [PubMed] [Google Scholar]
- 19.Rocha MS, Brucki SM, Carvalho AA, Lima UW. Epidemiologic features of Guillain Barre Syndrome in Sao Paulo, Brazil. Arq Neuropsiquiatr. 2004;62:33–37. doi: 10.1590/s0004-282x2004000100006. [DOI] [PubMed] [Google Scholar]
- 20.van Koningsveld R, van Doorn PA, Schmitz PI, Ang CW, van der Meche FG. Mild forms of Guillain-Barre syndrome in an epidemiologic survey in The Netherlands. Neurology. 2000;54:620–625. doi: 10.1212/wnl.54.3.620. [DOI] [PubMed] [Google Scholar]
- 21.Wu X, Shen D, Li T, Zhang B, Li Ch, Mao M, et al. Distinct clinical characteristics of pediatric Guillain-Barré Syndrome: A comparative study between children and adults in northeast China. PLoS ONE. 11:3. doi: 10.1371/journal.pone.0151611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rees JH, Thompson RD, Smeeton NC, Hughes RAC. Epidemiological study of Guillain-Barre Syndrome in south east England. J Neurol Neurosurg Psychiatry. 1998;64:74–77. doi: 10.1136/jnnp.64.1.74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Winer JB. Guillain Barre Syndrome. Mol Pathol. 2001;54:381–385. [PMC free article] [PubMed] [Google Scholar]