Abstract
Introduction
Exposure to antibiotics (ABX) during pregnancy can have a systematic effect on both fetal and maternal health. Although previous biomonitoring studies have indicated the effects on children of extensive exposure to ABX, studies on pregnant women remain scarce. To explore the effect on pregnant women of environmental exposure to ABX through accidental ingestion and identify potential health risks, the present study investigated 122 pregnant women in East China between 2019 and 2020.
Research design and methods
The presence of six categories of ABX (quinolones, sulfonamides, lincosamides, tetracyclines, amide alcohol ABX, and β-lactams) in plasma samples taken from the pregnant women was investigated using an ABX kit and a time-resolved fluorescence immunoassay.
Results
All six ABX were detected in the plasma, with a detection rate of 17.2%. It was discovered that the composition of intestinal flora in pregnant women exposed to ABX was different from that of pregnant women who had not been exposed to ABX. The intestinal flora of pregnant women exposed to ABX also changed at both the phylum and genus levels, and several genera almost disappeared. Furthermore, the metabolic levels of glucose and insulin and the alpha diversity of pregnant women exposed to ABX were higher than those of pregnant women not exposed to ABX.
Conclusion
Pregnant women are potentially at higher risk of adverse microbial effects. Glucose metabolism and insulin levels were generally higher in pregnant women exposed to ABX than in unexposed women. Also, the composition and color of the gut microbiome changed.
Keywords: gastrointestinal tract, gynecology, nutritional sciences, antibodies
Significance of this study.
What is already known about this subject?
Antibiotic (ABX) administration and dietary changes have been associated with changes in the population structure of the gut microbiome. The effects on pregnant women of exposure to subtherapeutic ABX dosages in food have not been previously studied.
What are the new findings?
The concentration of six different ABX in the plasma of pregnant women were detected, with tetracyclines found to be the most common.
It was also found that gut microbiome composition and coloration changed as a result of exposure to antibiotics.
The levels of glucose metabolism and insulin in pregnant women who had been exposed to ABX were generally higher than in those who had not.
How might these results change the focus of research or clinical practice?
This study explored the environmental exposure of pregnant women to ABX through accidental ingestion by examining the approximate concentration of ABX in their plasma. Few previous studies have done this. Plasma ABX can indicate the exposure of pregnant women to ABX in food and drinking water, so the findings of the present study complement those of previous studies that are based on urinary antibiotic concentration, questionnaires, or prescription examination.
Introduction
As a group of pharmaceuticals and personal care products, antibiotics (ABX) are emerging environmental contaminants, and there is concern about the threat they pose to both aquatic life and human health.1 Since the 1950s, growth-promoter ABX administered in low doses have been widely used in the agricultural industry to promote the growth of farm animals and aquatic life, resulting in an increased weight gain in the animals of as much as 15%.2 The effect of this low dose of ABX is broad across vertebrate species, including cattle, pigs, sheep, chickens, and fish. Seafood, including fish and shellfish, is a major source of dietary proteins, especially elements and omega-3 fatty acids,3 which are very important for the health of pregnant women.
The effects observed with many categories of antibacterial agents, such as macrolides, tetracyclines, and sulfonamides, indicate that the side effects are not agent specific and are not the same as those observed with antifungal or antiviral agents. Antimicrobial agents are used in animal husbandry to treat disease, to control infections and prevent them from spreading in herds and flocks, and to promote animal growth.4 Two safety precautions have been implemented for antimicrobial agents: the first relates to their use in animals, which can lead to the development of antimicrobial-resistant bacteria and resistance determinants, which can be passed on to humans via the food chain or zoonotic spread; the second addresses the potential impacts on the human gut microbiome of ingesting animal-derived foods (such as meat, milk, eggs, and edible tissues) containing antimicrobial residue from the metabolism of a parent drug or other compounds used to treat the animal.5 In the USA, the most widespread use of low doses of ABX in animals is in farming for food production, where the ABX increase the animals’ weight gain. The effects of these ABX follow oral administration, either in feed or water, indicating that the microbiota of the gastrointestinal (GI) tract is a major target.6
The vertebrate GI tract contains an exceptionally complex and dense microbial environment, with the gut microbiota influencing the immune response of populations of reactive host cells7 and stimulating a rich matrix of effector mechanisms involved in innate and adaptive immune responses.8 The GI tract also produces hormones, including those involved in energy homeostasis—insulin, glucagon, leptin, and ghrelin, for example—and growth factors, such as glucose-dependent insulinotropic polypeptide and glucagon-like peptide 1.9 Alterations in the populations of the gut microbiota may change the intra-community metabolic interactions.10 However, therapeutic doses of antibiotic treatments alter both the composition of the gut microbiota and the host responses to specific microbial signals.11
Intrapartum antibiotic prophylaxis (IAP) disrupts the colonization of the neonatal gut flora, potentially further altering its development pattern. In addition, significant disruption of intestinal flora, including a decrease in bacterial diversity and a change in taxonomic distribution, has been observed after neonatal antibiotic treatment at birth.12 13 Previous studies have also identified a sustained drop in Bifidobacterium for up to 7 days after antibiotic treatment for group B streptococcus during pregnancy.14 Another study found that IAP can reduce the richness of intestinal microflora, decrease the abundance of Bacteroidetes, and increase the abundance of Firmicutes in 3-month-old newborns.15 Similar effects were observed within 8 weeks of intravenous β-lactam combination therapy in neonates or perinatal ABX in mothers.13 Furthermore, one study found that the use of ABX during delivery or after an emergency cesarean section changes the composition of neonatal intestinal flora, such as increased Clostridiales and decreased Bacteroidaceae, but that breast feeding narrows the differences in this composition.15
The use of ABX during pregnancy can also have effects on children later in life. One study found that children were overweight at the age of 7 if their mother received broad-spectrum ABX, such as ampicillin and amoxicillin, in the second trimester of her pregnancy.16 This finding is supported by three studies that reported larger body size among children exposed to ABX in utero.17–19
Antimicrobial agents of different classes and varying activity are effective across several vertebrate species; however, antibiotic administration and dietary changes have been associated with changes in the population structure of the gut microbiome.20 The effects on pregnant women of exposure to subtherapeutic ABX dosages in food have not been previously studied. Therefore, the present study hypothesized that such subtherapeutic administration alters the composition of the gut microbiota and metabolic capabilities of pregnant women.
Research design and methods
Study population and sample collection
A total of 122 pregnant women who were given a 75 g oral glucose tolerance test between 24 and 28 weeks of pregnancy at Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, between September 2019 and June 2020, were studied. Based on their homogenization of age, pre-pregnancy body mass index, gestational age, blood pressure, race, diet, and living environment, 37 of the patients were included in the study. These patients were separated into two cohorts: those patients without any detectable ABX in circulation (controls, n=25) and those patients who had detectable ABX in circulation (ABX detected, n=12).
Inclusion criteria:1 residents in Songjiang District of Shanghai had a normal diet;2 they did not have diabetes or impaired glucose tolerance before pregnancy.
Exclusion criteria:1 multiple births;2 diabetes, hypertension, thyroid disease, gastrointestinal disease, or cardiovascular disease before pregnancy;3 use of assisted reproductive technology;4 all patients consuming a “prescribed antibiotics”;5 active smoking.
Most maternal characteristics, fasting plasma glucose (FPG) levels, 1-hour plasma glucose (1 h PLG), 2-hour plasma glucose (2 h PLG), fasting insulin levels (FINS), 1-hour plasma insulin (1 h PIN), 2-hour plasma insulin (2 h PIN), glycosylated hemoglobin (HbA1c), homeostasis model assessment (HOMA-IR), triglyceride (TG) levels, total cholesterol (TC), high-density lipoprotein (HDL), and low-density lipoprotein (LDL) levels were obtained from the patients’ medical records. All serum aliquots and fresh feces were collected and immediately stored at –80°C prior to DNA extraction.
DNA extraction
A frozen aliquot (200 mg) of each fecal sample was suspended in 250 µL of guanidine thiocyanate (Sigma-Aldrich, GER), 0.1 M tris (pH 7.5) (Amresco, USA), and 40 µL of 10% N-lauroyl sarcosine (Sigma-Aldrich, GER). Total bacterial genomic DNA samples were extracted from 53 samples using a QIAamp DNA stool mini kit (Qiagen, California, USA). A NanoDrop 2000 spectrophotometer (Thermo Scientific, USA) was used to detect the concentration of the extracted DNA. Any samples that did not meet the detection standards were removed.
16S ribosomal RNA (16S rRNA) amplicon pyrosequencing
The V3 and V4 variable regions of 16S rRNA were specifically amplified by PCR with the forward primer 338F (5′-ACTCCTACGGGAGGCAGCAG-3′) and the reverse primer 806R (5′-GGACTACHVGGGTWTCTAAT-3′), where the barcode was an eight-base sequence unique to each sample. PCR was performed by DNA Extraction Kit (OMEGA-soil DNA Kit; Omega Bio-Tek, USA) that in a triplicate 20 µL mixture containing 4 µL of 5× FastPfu Buffer, 2 µL of 2.5 mM dNTPs, 0.8 µL of each primer (5 µM), 0.4 µL of FastPfu Polymerase (FastPfu Polymerase; TransGen, China), and 10 ng of template DNA, and it was conducted under the following conditions: initial denaturation (95°C, 2 min), 25 cycles at 95°C (30 s), annealing at 55°C (30 s), extension at 72°C (30 s), final extension at 72°C (5 min), 10°C until halted by user. The PCR products were extracted from 2% agarose gels (Biowest agArose; Biowest, Spain) and purified using an AxyPrep DNA gel extraction kit (Axygen Biosciences, Union City, CA, USA) according to the manufacturer’s instructions. They were then quantified using a QuantiFluor-ST fluorimeter (Promega, USA) before being sequenced. A database was established using an Illumina MiSeq instrument (Illumina, San Diego, California, USA) at SHBIO Biotechnology Corporation (Shanghai, China).
Sequence analysis
Raw FASTQ files were demultiplexed and quality filtered using QIIME V.1.9.1 (Knight and Caporaso Labs, USA) with default parameters. Operational taxonomic units (OTUs) were clustered with 97% similarity cut-off using UPARSE (V.7.1, http://drive5.com/uparse/), and chimeric sequences were identified and removed using UCHIME (usearch 8.1.1861, Robert C. Edgar lab, independent scientist, USA). The taxonomy of each 16S rRNA gene sequence was analyzed using the Ribosomal Database Project classifier (http://rdp.cme.msu.edu/) against the SILVA (SSU123) 16S rRNA database, using a confidence threshold of 70%.
Detection of ABX in the serum aliquots
Six major categories of ABX (quinolones, sulfonamides, lincosamides, tetracyclines, amide alcohol antibiotics, and β-lactams) were studied using a time-resolved fluorescence immunoassay (version SS-030, Shanghai Tracing-Bio Technology Co., Ltd., Shanghai, China). The serum aliquots, which had been stored at –80℃, and the detection ABX kits (Antibiotics, Shanghai, China) were returned to room temperature. A total of 150 mL diluent was then fully mixed and dissolved into the lyophilized sample for a tetracycline drug test. Next, 10 µL of serum sample was added to the lyophilized product and mixed well, and a reagent strip was inserted vertically and the time counted. After 6 min, the reagent strip was removed and placed in the time-resolved immunoassay analyzer for the results. The limit of positive judgment for tetracycline concentration was 15 ng/mL. A + indicated that the concentration of tetracycline was between 15 and 60 ng/mL, while ++ indicated that the concentration was greater than 60 ng/mL. The concentration limits of β-lactams, quinolones, sulfonamides, lincosamides, and amide alcohol ABX were 4, 25, 4, 10, and 1 ng/mL, respectively.
Bioinformatics and statistical analysis
The QIIME (V.1.9.1) and R packages were used for gut microbiota sequence analyses. Alpha diversity indices, such as Chao richness estimator, Shannon diversity index, observed species, and coverages, were calculated using the OTUs table in QIIME to investigate gut microbiota community richness. Beta diversity was measured using the Bray-Curtis dissimilarity and unweighted and weighted UniFrac distance.21 Beta diversity analysis, including principal co-ordinates analysis based on UniFrac distance matrix analysis and visualized via non-metric multidimensional scaling, was used to evaluate the structural variation of microbial communities. The total microbial composition difference between the two groups was indicated by permutational multivariate analysis of variance.22 Tax4Fun,21 an R package obtained from the SILVA dataset version 132, was used for functional profiling. Pattern recognition analysis based on forward feature selection and linear discriminant analysis (LDA) was performed using R V.3.5.1.23 Unique OTUs and those shared between samples were illustrated using a Venn diagram that was created in the R package “Venn Diagram”. The relative abundance of taxa at all levels was statistically compared between the two groups using the Kruskal-Wallis test in the R stats package, and the microbiota correlation network was constructed using the SparCC algorithm24 and visualized with Cytoscape V.3.4.0.25
Normal distributed continuous variables were presented as mean±SD and analyzed using Student’s t-test, while non-normal distributed continuous variables were reported as median with IQRs (Q1–Q3) and analyzed using either the Wilcoxon signed-rank test or the Mann-Whitney U test, which were conducted in SPSS V.23.0 (SPSS Inc., Chicago, IL, USA). A p value <0.05 was considered statistically significant.
For dichotomous variables, the McNemar χ2 test, Pearson’s χ2 test, or Fisher’s exact test was applied. LDA effect size26 with a p-value cut-off of 0.05 and an LDA score cut-off of 2 was used to obtain the differential taxa and functions of the two groups. Spearman’s rank correlation was used for correlation analysis between the groups.
Results
General characteristics
The characteristics of the participants are presented in table 1. There were no differences between the two groups in markers of age, pre-pregnancy body mass index, gestational age, systolic blood pressure, diastolic blood pressure, gravidity, or parity. The patients in the ABX detection group had higher FPG, 1 h PLG, HbA1c, FINS, 1 h PIN, and HOMA-IR levels than those in the control group, although there were no statistically significant differences. The two groups had similar 2 h PLG, 2 h PIN, TG, TC, HDL, and LDL levels.
Table 1.
Maternal characteristics and biochemical data
| Maternal characteristics and biochemical variables | Controls (n=25) | ABX detected (n=12) | P value |
| Age (years) | 30.6±4.85 | 31±5.31 | 0.839 |
| Pre-BMI (kg/m2) | 22.5±4.98 | 21.3±2.0 | 0.442 |
| Gestational age (weeks) | 25.7±1.22 | 25.2±1.11 | 0.208 |
| SBP (mmHg) | 107.4±10.0 | 110.7±10.4 | 0.347 |
| DBP (mmHg) | 70.2 (66~74) | 67.8 (65~70.7) | 0.282 |
| Gravidity | 1.8 (1~2.5) | 2.1 (1.3~3) | 0.184 |
| Parity | 0.5 (0~1) | 0.6 (0~1) | 0.657 |
| Fasting glucose (mmol/L) | 4.89 (4.56~4.96) | 4.94 (4.76~4.95) | 0.411 |
| 1 h glucose (mmol/L) | 7.61±2.17 | 7.89±1.15 | 0.746 |
| 2 h glucose (mmol/L) | 6.69±2.11 | 6.09±0.60 | 0.335 |
| HbA1c (mmol/L) | 4.94±0.57 | 4.99±0.44 | 0.773 |
| Fasting insulin (μU/L) | 50.1 (33.1~62.1) | 52.4 (34.3~63.0) | 0.546 |
| 1 h insulin (μU/L) | 319.0±170.7 | 397.7±139.1 | 0.176 |
| 2 h insulin (μU/L) | 279.1±159.2 | 208.2±68.8 | 0.152 |
| HOMA-IR | 1.56 (0.99~2.01) | 1.65 (1.01~2.18) | 0.535 |
| Triglyceride (mmol/L) | 2.71±0.84 | 2.24±0.77 | 0.114 |
| Total cholesterol (mmol/L) | 6.38±1.07 | 5.64±1.11 | 0.064 |
| HDL (mmol/L) | 2.05 (1.72~2.15) | 2.01 (1.68~2.31) | 0.687 |
| LDL (mmol/L) | 3.13±0.89 | 2.69±1.01 | 0.184 |
Clinical characteristics, biochemical and hormonal variables of antibiotic exposure and normal healthy pregnant women at 24~28 weeks’ gestation are presented as mean±SEM when normally distributed or median with 25–75th IQR when non-normal distributed. Statistically significant difference between the GDM and normoglycemic women group are highlighted (*p<0.05, **p<0.01). HOMA-IR, HOMA-IR=FPG (mmol/L) × FINS (μU/mL)/22.5.
ABX, antibiotics; BMI, body mass index; DBP, diastolic blood pressure; FINS, fasting insulin levels; FPG, fasting plasma glucose; HbA1c, glycosylated hemoglobin; HDL, high-density lipoprotein; HOMA-IR, homeostasis model assessment; LDL, low-density lipoprotein; SBP, systolic blood pressure.
Exposure to antibiotics
Of the 122 patients initially studied, 21 tested positive for ABX in venous blood tests (table 2). One patient was exposed to β-lactams, one to quinolones, one to amide alcohol ABX, three to lincosamides, four to sulfonamides, and eighteen to tetracyclines. Six of the patients had two or more ABX in their blood. The antibiotic exposure rate was 17.2%.
Table 2.
Holistic situation of antibiotic exposure in 122 pregnant women at 24~28 weeks’ gestation
| β-Lactams | Sulfonamides | Quinolones | Amide alcohol antibiotics | Tetracyclines | Lincosamides |
| n=1 | n=4 | n=1 | n=1 | n=18 | n=3 |
n represents the number of pregnant women whose ABX examination was positive.
ABX, antibiotics.
The limit of positive judgment for detected tetracycline concentration was 15 ng/mL. The detected concentration limit of β-lactams was 4 ng/mL. The detected concentration limit of quinolones was 25 ng/mL. The limit of positive judgment for sulfonamide detected concentration was 4 ng/mL. The limit of positive judgment for lincosamides detected concentration was 10 ng/mL. The limit of positive judgment for amide alcohol ABX detected concentration was 1 ng/mL.
Altered gut microbiota in pregnant women exposed to ABX
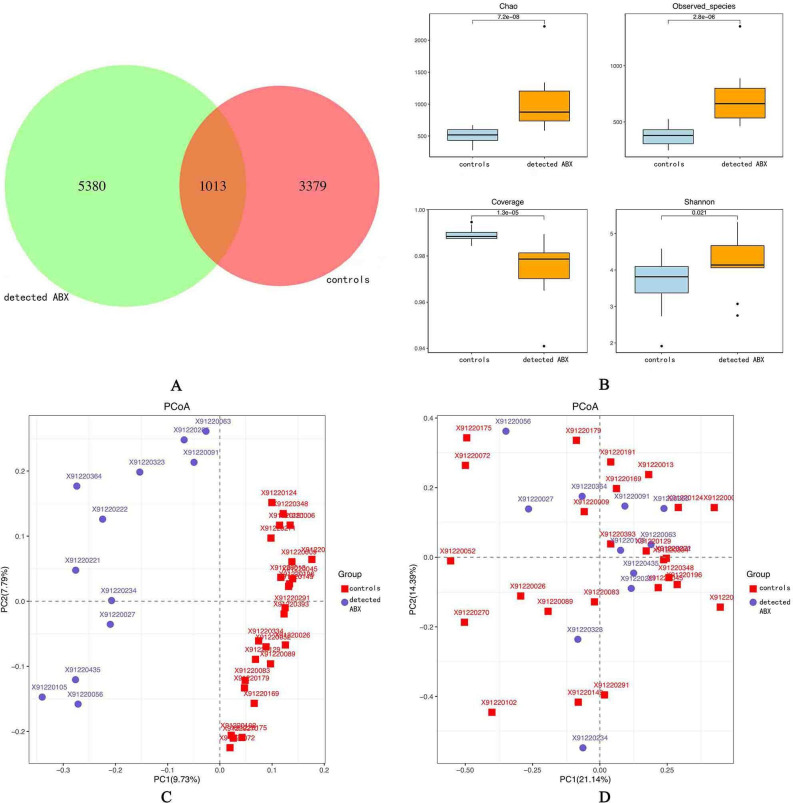
In the control group, the total number of OTUs in common was 4392, while the ABX detection group had 6393 OTUs in common; 1013 OTUs were shared between the two groups (figure 1A). Alpha diversity analysis (figure 1B) indicated that the antibiotic exposure of the ABX detection group presented higher richness (higher Chao estimator, p=7.2e-08; higher observed species, p=2.8e-06) and higher diversity (lower coverage p=1.3e-05; higher Shannon index, p=0.021) than that detected in the control group. Principal coordinate analysis was conducted based on unweighted UniFrac distance to evaluate the overall diversity of microbial composition. Permutational multivariate ANOVA demonstrated significant differences in gut taxonomic composition between the two groups (figure 1C). The differences in microbial community between the two groups were also significant for weighted UniFrac distance (figure 1D).
Figure 1.
Richness and diversity of the gut microbiota in pregnant women who had been exposed to antibiotics (ABX) and those who had not. (A) Venn diagram of exposure to ABX in pregnant women; controls had not been exposed to ABX while detected ABX had. Detected ABX had more operational taxonomic units than controls. (B) Alpha diversity was calculated using QIIME2 V.2018.2 based on a sequence similarity level of 100%, including the Chao richness estimator, observed species, coverage, and the Shannon diversity index. Detected ABX showed higher alpha diversity than controls. Principal coordinates analysis based on the unweighted UniFrac matrix (C) and weighted UniFrac matrix (D) showed that the overall fecal microbiota composition was different between the two groups. Each point represents one sample of antibiotic exposure (red, controls, n=25; blue, detected ABX, n=12). The distances between different samples reflect the comparability of the two groups.
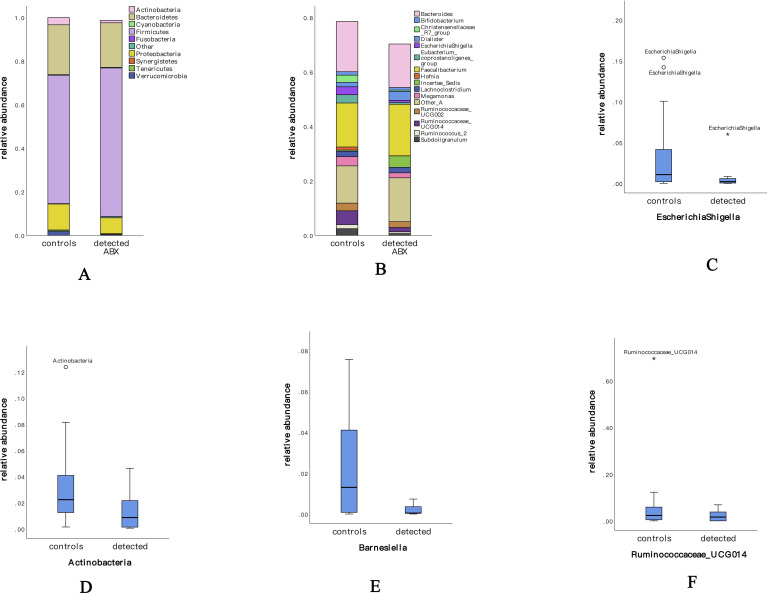
The diversity of the fecal microbiome in the two groups was found to be mainly related to six phyla—Firmicutes, Bacteroidetes, Proteobacteria, Actinobacteria, Verrucomicrobia, and Tenericutes—and those under the category, “Other” (table 3). The 10 most common phyla in the two groups are shown in figure 2A. The predominant genus in both groups was Firmicutes (control group: 58.9%; ABX detection group: 68.2%), and there was no significant difference between the groups. Actinobacteria was lower in the ABX detection group than in the control group, and the difference was statistically significant (control group: 3.3% vs ABX detection group: 1.2%; p=0.004). The other four genera were lower in the ABX detection group than in the control group but with no statistically significant differences (Bacteroidetes, control group: 22.9% vs ABX detection group: 20.4%; Proteobacteria, control group: 11.9% vs ABX detection group: 7.3%; Verrucomicrobia, control group: 1.7% vs ABX detection group: 0.3%; Tenericutes, control group: 0.3% vs ABX detection group: 0.6%).
Table 3.
Microbiome composition in phylum and genera levels of antibiotic exposure and normal healthy pregnant women are different at 24~28 weeks’ gestation
| Controls (n=25) | ABX detected (n=12) | P value | |
| Chao | 681.8±140.2 | 1537.3±564.6 | <0.01 |
| Shannon | 3.82±0.67 | 4.41±0.73 | 0.022 |
| OTUs | 459.7±85.0 | 899.0±284.5 | <0.01 |
| Phylum | |||
| Firmicutes | 0.589±0.192 | 0.682±0.138 | 0.145 |
| Bacteroidetes | 0.229±0.155 | 0.204±0.126 | 0.639 |
| Proteobacteria | 0.119 (0.025~0.160) | 0.073 (0.021~0.080) | 0.119 |
| Actinobacteria | 0.033 (0.013~0.044) | 0.012 (0.015~0.021)** | 0.004 |
| Verrucomicrobia | 0.017 (0~0.015) | 0.003 (0~0002) | 0.135 |
| Tenericutes | 0.006 (0~0.005) | 0.003 (0~0.001) | 0.289 |
| Other | 0.002 (0~0.002) | 0.006 (0.0002~0.008) | <0.01 |
| Genera | |||
| Bacteroides | 0.184 (0.066~0.240) | 0.160 (0.072~0.208) | 0.721 |
| Faecalibacterium | 0.162 (0.072~0.222) | 0.189 (0.188~0.247) | 0.119 |
| Other_A | 0.137 (0.071~0.161) | 0.161 (0.094~0.219) | 0.256 |
| Megamonas | 0.034 (0~0.011) | 0.018 (0~0.004) | 0.088 |
| Lachnoclostridium | 0.020 (0.004~0.024) | 0.020 (0.008~0.031) | 0.721 |
| EscherichiaShigella | 0.029 (0.001~0.041) | 0.008 (0~0.006) | 0.098 |
| Ruminococcaceae_UCG014 | 0.051 (0~0.05) | 0.016 (0~0.037) | 0.459 |
| Incertae_sedis | 0.004 (0~0.004) | 0.042 (0.002~0.025)* | 0.019 |
|
Eubacterium_coprostanoligenes
_group |
0.03 (0.005~0.050) | 0.007 (0~0.007)** | 0.009 |
| Ruminococcaceae_UCG002 | 0.028 (0~0.017) | 0.021 (0~0.023) | 0.407 |
| Christensenellaceae_R7_group | 0.026 (0.005~0.035) | 0.004 (0~0.08)** | 0.001 |
| Dialister | 0.017 (0~0.011) | 0.033 (0~0.048) | 0.060 |
| Bifidobacterium | 0.013 (0.002~0.016) | 0.010 (0.001~0.015) | 0.338 |
| Hafnia | 0.011 (0~0) | 0 (0~0)** | 0.845 |
| Subdoligranulum | 0.024 (0~0.029) | 0.006 (0~0.01)* | 0.024 |
| Ruminococcus_2 | 0.015 (0.001~0.024) | 0.006 (0~0.016)* | 0.031 |
| Prevotella_9 | 0.001 (0~0) | 0.008 (0~0.020) | 0.017 |
| Lachnospiraceae_UCG004 | 0.003 (0~0.003) | 0.006 (0.004~0.009) | 0.004 |
| Rhodococcus | 0.008 (0.007~0.007) | 0.0002 (0~0.0003)** | <0.01 |
Significant differences between two groups show in */**, *p<0.05,**p<0.01, the diversity and microbiome composition are presented as mean±SEM when normally distributed or median with 25–75th IQR when non-normal distributed the decreased genera in GDM group are in boldface.
ABX, antibiotic; OTU, operational taxonomic unit.
Figure 2.
Composition of the gut microbiome at phylum and genera levels and several almost-disappeared genera in the second trimester. (A, B) The composition of the gut biome microflora of the two groups, indicating the dominant microflora. (C) The genera of Escherichia–Shigella, which are sensitive to sulfonamide antibiotics, almost disappeared in the gut microbiomes of pregnant women who had been exposed to antibiotics. (D) The phylum of Actinobacteria and (E) the genus of Barnesiella were sensitive to tetracycline antibiotics and declined or disappeared in pregnant women who had been exposed to antibiotics. (F) Ruminococcaceae UCG-014 was affected by both sulfonamides and tetracyclines and declined in women who had been exposed to antibiotics. ABX, antibiotics.
To explore the altered gut microbiota in pregnant women exposed to antibiotics, the 16 most common genera (figure 2B) were studied. The predominant genus in both groups was Bacteroidetes, although it was present in lower levels in the ABX detection group (control group: 18.4% vs ABX detection group: 16%; p=0.721). At the genus level, the ABX detection group showed a significantly lower abundance of the Eubacterium coprostanoligenes group (p=0.009), the Christensenellaceae R7 group (p=0.001), Subdoligranulum (p=0.024), Ruminococcus 2 (p=0.031), Prevotella 9 (p=0.017), Lachnospiraceae UCG-004 (p=0.004), and Rhodococcus (p=0). The relative abundance of Megamonas, Escherichia–Shigella, Ruminococcaceae UCG-014, and Ruminococcaceae UCG-002 in the ABX detection group was also lower than in the control group, although there were no significant differences (table 3). These findings point to changes in the gut microbiota of pregnant women exposed to antibiotics.
Effects on the gut microbiome of exposure to ABX
Two sulfonamide and tetracycline ABX were found to have an influence on the gut microbiome (table 4). Escherichia–Shigella (p=0.034), Ruminococcus 2 (p=0.016), and Alistipes (p=0.045) were found to be sensitive to sulfonamide antibiotics, and their relative abundance decreased significantly in pregnant women whose blood samples were positive for sulfonamides. The presence of Escherichia-Shigella (figure 2C) could hardly be detected. The abundance of Actinobacteria (p=0.035) (figure 2D), the Eubacterium coprostanoligenes group (p=0.034), the Christensenellaceae R7 group (p=0.004), and Subdoligranulum (p=0.025) in the intestines of pregnant women who were positive for tetracyclines was significantly decreased, while Barnesiella (p=0.04) (figure 2E) almost disappeared. Ruminococcaceae UCG-014 declined in the ABX detection group (figure 2F) and was affected by both sulfonamides and tetracyclines.
Table 4.
Holistic situation of antibiotic exposure in 122 pregnant women at 24~28 weeks’ gestation
| Sulfonamides | Tetracyclines | ||||||
| Controls | Detected ABX | P value | Controls | Detected ABX | P value | ||
| Phylum | |||||||
| – | – | – | – | Actinobacteria | 0.031 (0.012~0.044) | 0.014 (0.001~0.02) | 0.035 |
| – | – | – | – | Other | 0.002 (0~0.002) | 0.006 (0.002~0.008) | 0.001 |
| Genera | |||||||
| EscherichiaShigella | 0.024 (0.001~0.036) | 0 | 0.034 | Eubacterium_coprostanoligenes_group | 0.028 (0.004~0.024) | 0.008 (0.001~0.008) | 0.034 |
| Ruminococcus_2 | 0.027 (0.0015~0.015) | 0.016 (0.027~0) | 0.016 | Christensenellaceae_R7_group | 0.025 (0.004~0.03) | 0.004 (0~0.007) | 0.004 |
| Alistipes | 0.0096 (0.0012~0.012) | 0 | 0.045 | Subdoligranulum | 0.023 (0.004~0.029) | 0.006 (0~0.007) | 0.025 |
| Barnesiella | 0.012 (0~0.016) | 0 | 0.04 | ||||
ABX, antibiotics.
Association of microbial composition with glycemic traits
Spearman correlation analysis was used to identify deeper-level taxa associated with glycemic traits in pregnant women (table 5). It was found that Actinobacteria was not significantly positively or negatively associated with blood glucose and lipid metabolism–related indicators. Escherichia–Shigella was significantly affected by sulfonamides and was only found to be negatively correlated with HDL. Barnesiella was influenced by tetracyclines and had a negative correlation with HbA1c (r = –0.330, p=0.046), FINS (r = –0.421, p=0.009), and HOMA-IR (r = –0.435, p=0.007). Ruminococcaceae UCG-014 was lower in the ABX detection group than in the control group and had a negative association with HbA1c (r = –0.397, p=0.015), 1 h PLG (r = –0.490, p=0.002), FINS (r = –0.396, p=0.015), 1 h PIN (r = –0.362, p=0.027), and HOMA-IR (r = –0.408, p=0.012).
Table 5.
Gut microbiota abundance (phylum and genus) and their correlation with clinical characteristics and biochemical variables in antibiotic exposure and normal health pregnant women
| HbA1c (mmol/L) |
1 h plasma glucose (mmol/L) |
Fasting insulin (μU/L) | 1 h plasma insulin (μU/L) | HOMA-IR | HDL (mmol/L) |
|||||||
| r value | P value | r value | P value | r value | P value | r value | P value | r value | P value | r value | P value | |
| p_Actinobacteria | – | – | – | – | – | – | – | – | – | – | – | – |
| g_EscherichiaShigella | – | – | – | – | – | – | – | – | – | – | −0.345* | 0.037 |
| g_Barnesiella | −0.330* | 0.046 | – | – | −0.421** | 0.009 | – | – | −0.435** | 0.007 | – | – |
| g_Ruminococcaceae_ UCG014 |
−0.397* | 0.015 | −0.490* | 0.002 | −0.396* | 0.015 | −0.362* | 0.027 | −0.408* | 0.012 | – | |
Data are Spearman correlation coefficients Significant correlations showed. *p<0.05,**p<0.01. p_, phylum level; g_, genus level.
HbA1c, glycosylated hemoglobin; HDL, high-density lipoprotein; HOMA-IR, homeostasis model assessment.
Discussion
This is the first study to explore the effect of environmental exposure to ABX through accidental ingestion on the gut microbiome of pregnant women. The blood concentration of ABX is very low—far lower than that of oral or intravenous antibiotics. The study population was strictly screened to exclude those who had used prescribed ABX, so it was concluded that the exposure to ABX was more likely to be from food or drinking water. ABX of accidental ingestion are emerging contaminants, and there is concern about their possible threat to human health and aquatic life.1 ABX consumed in human and animal medicine can directly or indirectly enter the human body, other animals, food, drinking water, and the environment.27 In humans, ABX entering the body through medical use, contaminated food, or the environment can reach the urinary and intestinal tracts through excretion in urine and feces, facilitating the emergence or immigration of resistant genes and bacteria in these sites. This poses a potential risk of infection by resistant bacteria.28 Exposure to ABX may influence childhood-onset asthma, childhood obesity, and colorectal carcinoma.29 Recently, it has been found that the disturbance of the gut microbiome through exposure to ABX has a profound influence on human physiology.30
Compared with children and other adults, pregnant women have different requirements for dietary nutrients and are treated more prudently with regard to drug use; as such, they may have a different ABX exposure profile.31 The present study measured six types of antibiotics: β-lactams (which is the only one routinely given to pregnant women), tetracyclines, sulfonamides, quinolones, amide alcohol ABX, and lincosamides (which are rarely given to pregnant women). The study found that 21 of 122 pregnant women were positive for ABX in venous blood tests. Tetracyclines and sulfonamides were found to have the greatest effect on the gut microbiome; for example, Escherichia–Shigella, Alistipes, and Barnesiella almost disappeared in response to exposure to these antibiotics.
Because of the side effects of enamel hypoplasia and the irreversible staining of deciduous teeth,32 tetracyclines are not given to children under 8 years of age33; instead, they are mainly used as a veterinary ABX.34 In clinical practice, sulfonamides have been largely replaced by the safer β-lactams or macrolides.35 36 Moreover, these two ABX are used in animals for growth promotion, disease prevention, as feed additives, and for the treatment of infection,37 and most of them are frequently detected in surface water or food in China.38 39
Given that ABX are extensively used in animal husbandry and aquaculture to prevent and treat disease or promote growth,40 meat may have substantial antibiotic residues; a previous study found that fish, shrimp, pork, chicken, and milk are the most common sources of exposure to veterinary ABX.31 Based on the present study, it can be suggested that the presence of tetracyclines, quinolones, and sulfonamides in the plasma samples of pregnant women mainly resulted from contaminated environments or food.
A previous study41 analyzed the biological effect of low-dose ABX exposure on pregnant mice and its impact on the body-fat content of newborn mice. It found that (1) maternal low-dose antibiotic exposure in late pregnancy affected the intestinal flora structure of the offspring after birth and had a profound impact on their subsequent development process and health, (2) maternal low-dose antibiotic exposure led to a significant increase in the body weight and body fat of the offspring, and (3) maternal antibiotic exposure during pregnancy led to excessive growth of Firmicutes and Bacteroidetes in the intestinal flora of the offspring. An unbalanced Firmicutes-to-Bacteroidetes ratio causes intestinal flora disorder, indirectly promotes intestinal flora by affecting the short-chain fatty acids and blood-glucose metabolism, and eventually leads to gestational diabetes mellitus. Furthermore, Yoshimoto et al found that administration of low-dose ABX from 1 week before pregnancy to birth affected the composition of the gut microbiota and abdominal fat percentage of the pups. Microbiome changes mainly included the increased abundance of Firmicutes and Clostridium; Clostridium IV and XIVa were also positively associated with increased abdominal fat percentage, while Firmicutes and Bacteroidetes did not have any correlation with body-fat percentage.41 Chao et al reported that the penicillin, vancomycin, and chlortetracycline ABX treatment of pups at subtherapeutic levels for 7 weeks increased the abundance of Firmicutes, body-weight gain, and levels of colonic short-chain fatty acids.6 The present study found that the group of women exposed to ABX had a higher alpha diversity and more OTUs. Among the six most common phyla, the relative abundance of Firmicutes, which has been previously found to be associated with gestational weight gain at a nominal significance threshold,42 increased by 10%. Bacteroides, Megamonas, Escherichia–Shigella, Ruminococcaceae UCG-014, and Rhodococcus, however, all decreased. Bacteroides, which can harbor butyrate-producing members, has been previously found to be negatively associated with blood pressure, body weight, and fat mass.43 The present study also found that the decreased abundance of Escherichia–Shigella, Barnesiella, and Ruminococcaceae UCG-014 had negative associations with HbA1c, 1 h PLG, FINS (μU/L), 1 h PIN, and HOMA-IR. Ruminococcaceae, which can break down indigestible polysaccharides (ie, dietary fiber) into short-chain fatty acids, such as butyrate, have been previously found to have a strong positive correlation with adipokine levels.44
One previous study assessed the effect of amoxicillin on the diversity of intestinal flora and found no effect.45 Rifaximin, however, has been found to decrease the abundance of Streptococcus spp and Veillonella spp and increase the abundance of Eubacterium spp, although it has not been found to reduce the diversity of the intestinal flora.46 47 Other studies have found that minocycline can increase the relative abundance of Alistipes spp, Bacteroides spp, and Marvinbryantia spp. In addition, it has been found that the abundance reduction of Anaerostipes spp, Bifidobacterium spp, Collinsella spp, Coprococcus spp, Dialister spp, Dorea spp, and Faecalibacterium spp eventually reduced the diversity of the bacteria community.45 Some studies have found that amoxicillin, which belongs to β-lactams, can increase the abundance of Alistipes, Bacteroides,45 and Ruminococcus,48 while clindamycin, which belongs to lincosamides, has been found to significantly reduce the abundance of Alistipes,45 which is consistent with the findings of the present study. Alistipes and Ruminococcus can produce short-chain fatty acids, and butyric acid can prevent colon cancer and improve insulin sensitivity.49
In a mouse model of autoimmune encephalomyelitis before delivery and during weaning, antibiotic exposure increased the abundance of Proteobacteria and Bacteroidetes and decreased the abundance of Firmicutes and Actinomycetes; this imbalance of field-intestinal flora coincided with high expression of pro-inflammatory factors, such as IFN-γ and IL-17.50 Antibiotic-induced intestinal microflora imbalance can reduce corneal nerve density by reducing the production of CCR2-corneal macrophages, and it has been suggested that it can be reversed by fecal transplanting or practical probiotics.51 Ruminococcaceae UCG-014 belongs to Ruminococcaceae, and both sulfonamides and tetracyclines can reduce its abundance. Lachnospiraceae and Ruminococcaceae, both belonging to Firmicutes, are dominant bacteria in energy metabolism, which decomposes dietary fiber to produce butyrate;52 they have been found to be negatively correlated with 1-hour blood glucose levels in an oral glucose tolerance test53 and positively correlated with serum leptin levels.44
This study explored the environmental exposure of pregnant women to ABX through accidental ingestion by examining the approximate concentration of ABX in their plasma. Few previous studies have done this. Plasma ABX can indicate the exposure of pregnant women to ABX in food and drinking water, so the findings of the present study complement those of previous studies that are based on urinary antibiotic concentration, questionnaires, or prescription examination. However, there are several limitations that should be considered when interpreting the findings of this study. First, pregnant women who undergo obstetric examination at a local health service organization may have different education and economic levels that influence their dietary habits and attitudes to the use of medicine. Second, due to the fact that most of the ABX examined in this study have a fast metabolic rate, with a half-life of less than 12 hours, it is difficult to capture every instance of exposure to ABX with one spot plasma sample; therefore, the actual level of exposure may be underestimated. Third, ABX were only detected in the patients’ blood, and the intermediate metabolites of ABX were not tested. Lastly, the sample size and sampling location should be expanded in future studies to further validate the experimental conclusion.
Conclusion
The concentration of six different ABX in the plasma of pregnant women were detected, with tetracyclines found to be the most common. It was also found that gut microbiome composition and coloration changed as a result of exposure to antibiotics. The levels of glucose metabolism and insulin in pregnant women who had been exposed to ABX were generally higher than in those who had not. In view of the very low blood concentration of ABX and the rules regarding the use of ABX during pregnancy, it is suggested that the presence of ABX in the plasma of pregnant women may come from food and drinking water. In addition to the findings of this study, the effects on the growth and development of pregnant women and fetuses need to be further studied and given greater attention by both medical staff and pregnant women.
Acknowledgments
We would like to acknowledge the hard and dedicated work of all the staff that implemented the intervention and evaluation components of the study. X-MX is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
Contributors: YS, X-MX, and X-PG conceived the idea and conceptualized the study. YS, H-HQ, and F-FL collected the data. X-MX, D-CC, and D-YZ analyzed the data. YS, X-MX, W-YL, and H-KW drafted the manuscript, then LC, J-FZ, and Y-NC reviewed the manuscript. All authors read and approved the final draft. X-MX is responsible for the overall content as guarantor. The guarantor accepts full responsibility for the finished work and/or the conduct of the study, had access to the data, and controlled the decision to publish.
Funding: Project Category: Three-year Action Plan Project for the promotion of clinical skills and clinical innovation in municipal hospitals. Project Unit: Shanghai Shenkang Hospital Development Center. Project Name: Randomized, double-blind, controlled clinical study on the prevention of Gestational Diabetes Mellitus by supplementation of fermentable dietary fiber in early and middle pregnancy. Project number: SHDC2020CR2060B.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement
All data relevant to the study are included in the article.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
Written informed consent was obtained from the participants prior to enrollment, and approval for the research was obtained from the Ethics Committee of Shanghai General Hospital.
References
- 1.Liu J-L, Wong M-H. Pharmaceuticals and personal care products (PPCPs): a review on environmental contamination in China. Environ Int 2013;59:208–24. 10.1016/j.envint.2013.06.012 [DOI] [PubMed] [Google Scholar]
- 2.Butaye P, Devriese LA, Haesebrouck F. Antimicrobial growth promoters used in animal feed: effects of less well known antibiotics on gram-positive bacteria. Clin Microbiol Rev 2003;16:175–88. 10.1128/CMR.16.2.175-188.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fang GC, Nam DH, Basu N. Mercury and selenium content of Taiwanese seafood. Food Addit Contam Part B Surveill 2011;4:212–7. 10.1080/19393210.2011.605526 [DOI] [PubMed] [Google Scholar]
- 4.Mu C, Zhu W. Antibiotic effects on gut microbiota, metabolism, and beyond. Appl Microbiol Biotechnol 2019;103:9277–85. 10.1007/s00253-019-10165-x [DOI] [PubMed] [Google Scholar]
- 5.Cerniglia CE, Kotarski S. Approaches in the safety evaluations of veterinary antimicrobial agents in food to determine the effects on the human intestinal microflora. J Vet Pharmacol Ther 2005;28:3–20. 10.1111/j.1365-2885.2004.00595.x [DOI] [PubMed] [Google Scholar]
- 6.Cho I, Yamanishi S, Cox L, et al. Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature 2012;488:621–6. 10.1038/nature11400 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Abreu MT, Fukata M, Arditi M. TLR signaling in the gut in health and disease. J Immunol 2005;174:4453–60. 10.4049/jimmunol.174.8.4453 [DOI] [PubMed] [Google Scholar]
- 8.Qin J, Li R, Raes J, et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010;464:59–65. 10.1038/nature08821 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hansotia T, Drucker DJ. Gip and GLP-1 as incretin hormones: lessons from single and double incretin receptor knockout mice. Regul Pept 2005;128:125–34. 10.1016/j.regpep.2004.07.019 [DOI] [PubMed] [Google Scholar]
- 10.Gesta S, Tseng Y-H, Kahn CR. Developmental origin of fat: tracking obesity to its source. Cell 2007;131:242–56. 10.1016/j.cell.2007.10.004 [DOI] [PubMed] [Google Scholar]
- 11.Wlodarska M, Willing B, Keeney KM, et al. Antibiotic treatment alters the colonic mucus layer and predisposes the host to exacerbated Citrobacter rodentium-induced colitis. Infect Immun 2011;79:1536–45. 10.1128/IAI.01104-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Penders J, Thijs C, van den Brandt PA, et al. Gut microbiota composition and development of atopic manifestations in infancy: the KOALA Birth Cohort Study. Gut 2007;56:661–7. 10.1136/gut.2006.100164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fouhy F, Guinane CM, Hussey S, et al. High-throughput sequencing reveals the incomplete, short-term recovery of infant gut microbiota following parenteral antibiotic treatment with ampicillin and gentamicin. Antimicrob Agents Chemother 2012;56:5811–20. 10.1128/AAC.00789-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tanaka S, Kobayashi T, Songjinda P, et al. Influence of antibiotic exposure in the early postnatal period on the development of intestinal microbiota. FEMSImmunol Med Microbiol 2009;56:80–7. 10.1111/j.1574-695X.2009.00553.x [DOI] [PubMed] [Google Scholar]
- 15.Azad MB, Konya T, Persaud RR, et al. Impact of maternal intrapartum antibiotics, method of birth and breastfeeding on gut microbiota during the first year of life: a prospective cohort study. BJOG 2016;123:983–93. 10.1111/1471-0528.13601 [DOI] [PubMed] [Google Scholar]
- 16.Jess T, Morgen CS, Harpsøe MC, et al. Antibiotic use during pregnancy and childhood overweight: a population-based nationwide cohort study. Sci Rep 2019;9:11528. 10.1038/s41598-019-48065-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cassidy-Bushrow AE, Burmeister C, Havstad S, et al. Prenatal antimicrobial use and early-childhood body mass index. Int J Obes 2018;42:1–7. 10.1038/ijo.2017.205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mor A, Antonsen S, Kahlert J, et al. Prenatal exposure to systemic antibacterials and overweight and obesity in Danish schoolchildren: a prevalence study. Int J Obes 2015;39:1450–5. 10.1038/ijo.2015.129 [DOI] [PubMed] [Google Scholar]
- 19.Mueller NT, Whyatt R, Hoepner L, et al. Prenatal exposure to antibiotics, cesarean section and risk of childhood obesity. Int J Obes 2015;39:665–70. 10.1038/ijo.2014.180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schwartz DJ, Langdon AE, Dantas G. Understanding the impact of antibiotic perturbation on the human microbiome. Genome Med 2020;12:82. 10.1186/s13073-020-00782-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lozupone C, Knight R. UniFrac: a new phylogenetic method for comparing microbial communities. Appl Environ Microbiol 2005;71:8228–35. 10.1128/AEM.71.12.8228-8235.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Aßhauer KP, Wemheuer B, Daniel R, et al. Tax4Fun: predicting functional profiles from metagenomic 16S rRNA data. Bioinformatics 2015;31:2882–4. 10.1093/bioinformatics/btv287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.RCTR N, Team R, Null RCT, et al. Team R. R: a language and environment for statistical computing. Computing 2011;1:12–21. [Google Scholar]
- 24.Friedman J, Alm EJ. Inferring correlation networks from genomic survey data. PLoS Comput Biol 2012;8:e1002687. 10.1371/journal.pcbi.1002687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shannon P, Markiel A, Ozier O, et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 2003;13:2498–504. 10.1101/gr.1239303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Segata N, Izard J, Waldron L, et al. Metagenomic biomarker discovery and explanation. Genome Biol 2011;12:R60. 10.1186/gb-2011-12-6-r60 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Davies J, Davies D. Origins and evolution of antibiotic resistance. Microbiol Mol Biol Rev 2010;74:417–33. 10.1128/MMBR.00016-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Marshall BM, Levy SB. Food animals and antimicrobials: impacts on human health. Clin Microbiol Rev 2011;24:718–33. 10.1128/CMR.00002-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Riley LW, Raphael E, Faerstein E. Obesity in the United States – dysbiosis from exposure to low-dose antibiotics? Front Public Health 2013;1:69. 10.3389/fpubh.2013.00069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cho I, Blaser MJ. The human microbiome: at the interface of health and disease. Nat Rev Genet 2012;13:260–70. 10.1038/nrg3182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang H, Wang N, Qian J, et al. Urinary antibiotics of pregnant women in eastern China and cumulative health risk assessment. Environ Sci Technol 2017;51:3518–25. 10.1021/acs.est.6b06474 [DOI] [PubMed] [Google Scholar]
- 32.Ayaslioglu E, Erkek E, Oba AA, et al. Doxycycline-induced staining of permanent adult dentition. Aust Dent J 2005;50:273–5. 10.1111/j.1834-7819.2005.tb00373.x [DOI] [PubMed] [Google Scholar]
- 33.MOH (Ministry of Health, People’s Republic of China) . Principles for clinical use of antibiotics, 2004. Available: http://www.moh.gov.cn/mohyzs/s3573/200804/18544.shtml [Accessed 20 Sep 2014].
- 34.MOA (Ministry of Agriculture of the People’s Republic of China) . Variety catalogue of veterinary drugs, 2013. [Google Scholar]
- 35.MOA (Ministry of Agriculture of the people’s Republic of China) . Application regulations of feed drug additives (No. 168 Decree), 2001. [Google Scholar]
- 36.Zhang W, Shen X, Wang Y, et al. Outpatient antibiotic use and assessment of antibiotic guidelines in Chinese children's hospitals. Eur J Clin Pharmacol 2008;64:821–8. 10.1007/s00228-008-0489-3 [DOI] [PubMed] [Google Scholar]
- 37.Zhao L, Dong YH, Wang H. Residues of veterinary antibiotics in manures from feedlot livestock in eight provinces of China. Sci Total Environ 2010;408:1069–75. 10.1016/j.scitotenv.2009.11.014 [DOI] [PubMed] [Google Scholar]
- 38.Bu Q, Wang B, Huang J, et al. Pharmaceuticals and personal care products in the aquatic environment in China: a review. J Hazard Mater 2013;262:189–211. 10.1016/j.jhazmat.2013.08.040 [DOI] [PubMed] [Google Scholar]
- 39.Cai Z, Zhang Y, Pan H, et al. Simultaneous determination of 24 sulfonamide residues in meat by ultra-performance liquid chromatography tandem mass spectrometry. J Chromatogr A 2008;1200:144–55. 10.1016/j.chroma.2008.05.095 [DOI] [PubMed] [Google Scholar]
- 40.Sarmah AK, Meyer MT, Boxall ABA. A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 2006;65:725–59. 10.1016/j.chemosphere.2006.03.026 [DOI] [PubMed] [Google Scholar]
- 41.Yoshimoto A, Uebanso T, Nakahashi M, et al. Effect of prenatal administration of low dose antibiotics on gut microbiota and body fat composition of newborn mice. J Clin Biochem Nutr 2018;62:155–60. 10.3164/jcbn.17-53 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Crusell MKW, Hansen TH, Nielsen T, et al. Gestational diabetes is associated with change in the gut microbiota composition in third trimester of pregnancy and postpartum. Microbiome 2018;6:89. 10.1186/s40168-018-0472-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Gomez-Arango LF, Barrett HL, McIntyre HD, et al. Increased systolic and diastolic blood pressure is associated with altered gut microbiota composition and butyrate production in early pregnancy. Hypertension 2016;68:974–81. 10.1161/HYPERTENSIONAHA.116.07910 [DOI] [PubMed] [Google Scholar]
- 44.Gomez-Arango LF, Barrett HL, McIntyre HD, et al. Connections between the gut microbiome and metabolic hormones in early pregnancy in overweight and obese women. Diabetes 2016;65:2214–23. 10.2337/db16-0278 [DOI] [PubMed] [Google Scholar]
- 45.Zaura E, Brandt BW, Teixeira de Mattos MJ, et al. Same exposure but two radically different responses to antibiotics: resilience of the salivary microbiome versus long-term microbial shifts in feces. mBio 2015;6:e01693–15. 10.1128/mBio.01693-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kaji K, Takaya H, Saikawa S, et al. Rifaximin ameliorates hepatic encephalopathy and endotoxemia without affecting the gut microbiome diversity. World J Gastroenterol 2017;23:8355–66. 10.3748/wjg.v23.i47.8355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bajaj JS, Heuman DM, Sanyal AJ, et al. Modulation of the metabiome by rifaximin in patients with cirrhosis and minimal hepatic encephalopathy. PLoS One 2013;8:e60042. 10.1371/journal.pone.0060042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.De La Cochetière MF, Durand T, Lepage P, et al. Resilience of the dominant human fecal microbiota upon short-course antibiotic challenge. J Clin Microbiol 2005;43:5588–92. 10.1128/JCM.43.11.5588-5592.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rashid M-U, Weintraub A, Nord CE. Development of antimicrobial resistance in the normal anaerobic microbiota during one year after administration of clindamycin or ciprofloxacin. Anaerobe 2015;31:72–7. 10.1016/j.anaerobe.2014.10.004 [DOI] [PubMed] [Google Scholar]
- 50.Stanisavljević S, Čepić A, Bojić S, et al. Oral neonatal antibiotic treatment perturbs gut microbiota and aggravates central nervous system autoimmunity in Dark Agouti rats. Sci Rep 2019;9:918. 10.1038/s41598-018-37505-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Liu J, Wu M, He J, et al. Antibiotic-induced dysbiosis of gut microbiota impairs corneal nerve regeneration by affecting CCR2-negative macrophage distribution. Am J Pathol 2018;188:2786–99. 10.1016/j.ajpath.2018.08.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tun HM, Bridgman SL, Chari R, et al. Roles of birth mode and infant gut microbiota in intergenerational transmission of overweight and obesity from mother to offspring. JAMA Pediatr 2018;172:368–77. 10.1001/jamapediatrics.2017.5535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Chen T, Zhang Y, Zhang Y, et al. Relationships between gut microbiota, plasma glucose and gestational diabetes mellitus. J Diabetes Investig 2021;12:641–50. 10.1111/jdi.13373 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data relevant to the study are included in the article.