Abstract
Background:
Neurofilament light chain (NfL) and glial fibrilliary acidic protein (GFAP) have been suggested to be biomarkers of the pathophysiological process of neuromyelitis optica spectrum disorders (NMOSD), but the relationship between the plasma levels of these molecules with disease activity and treatment is incompletely understood.
Objective:
To investigate the treatment effects of disease-modifying drugs on plasma neurofilament light chain (pNfL) and plasma glial fibrillary acidic protein (pGFAP) and explore the predictive value of pNfL and pGFAP in the activity of NMOSD.
Methods:
pNfL and pGFAP levels were measured using single-molecule arrays in 72 patients with NMOSD and 38 healthy controls (HCs). Patients with NMOSD received tocilizumab (n = 29), rituximab (n = 23), oral prednisone (n = 16), and oral azathioprine or mycophenolate mofetil (n = 4).
Results:
NMOSD patients had significantly higher pNfL and pGFAP levels than HCs (pNfL, 18.3 (11.2–39.3) versus 11.5 (7.0–23.3) pg/mL; p = 0.001; pGFAP, 149.7 (88.6–406.5) versus 68.7 (59.4–80.8) pg/mL; p < 0.001). Multivariable regression analyses indicated that baseline pNfL concentration was associated with age (p = 0.017), Expanded Disability Status Scale (EDSS) score (p = 0.002), and recent relapses (p < 0.001). Baseline pGFAP concentration was also associated with EDSS (p < 0.001) and recent relapses (p < 0.001). Compared with prednisone, tocilizumab and rituximab significantly reduced pNfL [tocilizumab, exp(β), 0.65; 95% confidence interval (CI), 0.56–0.75; p < 0.001; rituximab, exp(β), 0.79; 95% CI = 0.68–0.93; p = 0.005] and pGFAP levels [tocilizumab, exp(β), 0.64; 95% CI, 0.51–0.80; p < 0.001; rituximab, exp(β), 0.77; 95% CI, 0.61–0.98; p = 0.041] at the end of the study. The pNfL levels in the tocilizumab and rituximab groups were reduced to those of HCs [tocilizumab, 8.5 (7.06–17.90) pg/mL; p = 0.426; rituximab, 14.0 (9.94–21.80) pg/mL; p = 0.216]. However, the pGFAP levels did not decrease to those of HCs in NMOSD patients at the end of study [tocilizumab, 88.9 (63.4–131.8) pg/mL; p = 0.012; rituximab, 141.7 (90.8–192.7) pg/mL; p < 0.001].
Conclusion:
pNfL and pGFAP may serve as biomarkers for NMOSD disease activity and treatment effects.
Keywords: glial fibrillary acidic protein, neurofilament light chain, NMOSD, treatment response
Introduction
Neuromyelitis optica spectrum disorder (NMOSD), a devastating autoimmune disease of the central nervous system, is characterized by recurrent inflammation of the optic nerve and spinal cord. It is frequently associated with a pathogenic autoantibody against water channel aquaporin-4 (AQP4-IgG) on the perivascular astrocyte endfeet, resulting in astrocyte destruction with secondary neuronal damage.1,2
Glial fibrillary acidic protein (GFAP) is an intermediate filament protein that forms the astrocyte cytoskeleton and could leak into the cerebrospinal fluid (CSF) after astrocyte injury. GFAP was significantly increased during acute NMOSD relapses and may reflect astrocytic injury3,4 Neurofilament light chain (NfL), a structural element of the neuron, could also be released into the CSF and peripheral blood after neuroaxonal damage. 5 Recently, single-molecule array (SIMOA) has provided highly sensitive measurements of GFAP and NfL levels in the blood and has been in use in various neurological diseases.6–8 Serum NfL and GFAP detected by SIMOA were strongly correlated with NfL and GFAP in the CSF of patients with NMOSD. Increased NfL and GFAP levels were found to correlate with NMOSD disease activity and worsening disability. 9
However, the dynamic changes under different disease-modifying treatments in NMOSD have been rarely reported. We therefore assessed plasma NfL (pNfL) and plasma GFAP (pGFAP) as potential biomarkers to monitor treatment response in NMOSD. Our aim was to assess the treatment effects of disease-modifying drugs on pNfL and pGFAP and investigate the predictive roles of pNfL and pGFAP in NMOSD disease activity.
Methods
Participants
In this prospective study, we enrolled 72 patients with NMOSD who had received different disease-modifying drugs in the Department of Neurology, Tianjin Medical University General Hospital. Twenty-nine patients were enrolled from the TANGO clinical trial (ClinicalTrial.gov, NCT 03350633) and received regular tocilizumab infusion at a dose of 8 mg/kg every 4 weeks. Sixteen patients were treated with oral prednisone (10 mg/day) and four patients were treated with conventional immunosuppressants (azathioprine or mycophenolate mofetil). In addition, 23 patients receiving low-dose rituximab treatment were enrolled in our analysis, and the doses of rituximab administered to the patients were according to the peripheral blood CD19+ B-cell counts, which were determined by flow cytometry.10,11 We also collected blood samples from 38 age- and sex-matched healthy controls (HCs).
Blood samples were centrifuged at 3000 rpm for 10 min at room temperature and then stored at −80°C within 4 h after collection. All samples were collected between September 2017 and November 2020. All patients with NMOSD were diagnosed according to the 2015 International Consensus Diagnostic criteria. 12 Clinical records of participants were collected at baseline and follow-ups, including sex, age at sampling, disease duration since the first onset, relapses in the past 60 days (yes/no), prior treatment before sample collection, Expanded Disability Status Scale (EDSS) score.
Measurement of NfL and GFAP
To measure pNfL and pGFAP concentrations in plasma, we used a similar methodology described previously. 13 In brief, a SIMOA HD-1 Analyzer and a SIMOA Neurology 2-Plex assay reagent kit (103520; both from Quanterix, Billerica, MA, USA) were used according to the protocol provided by the manufacturer. The limits of detection were 0.065 pg/mL for pNfL and 0.475 pg/mL for pGFAP. The intra- and inter-assay coefficients of variations for duplicate measurements were both within 10%. The laboratory investigators were blinded to clinical data and treatments of the patients.
Statistical analysies
Categorical variables were described by counts and percentages, and continuous and ordinal variables were described by median and interquartile ranges (IQRs). Demographic features of participants at baseline were compared using the Fisher exact test or the Mann–Whitney U test. Before further analysis, the pNfL and pGFAP levels were log-transformed to better meet the normal assumption. For clarity, figures were visualized with the original biomarker levels. In the HCs, the associations of age and sex with log-pNfL or log-pGFAP were tested with linear regression model, and regression coefficients are denoted with β. The differences in log-NfL and log-GFAP between NMOSD and HCs were determined using a mixed-effect model, which included age and sex as covariables. To clarify the association between baseline plasma biomarkers and clinical variables in NMOSD, a multiple linear regression analysis of log-pNfL or log-pGFAP was conducted, which included eight clinical variables: age, sex, disease duration since the first onset, recent relapses (<60 days) before the collection of samples (yes/no), numbers of relapse event, annualized relapse rates at baseline, EDSS score at baseline, treatment at baseline (no treatment/treated). Back-transformed regression coefficients (exp(β)) are reported with 95% confidence intervals (CIs) and p values.
To clarify the treatment response in NMOSD, we used a mixed-effects model for repeated measurements with log-NfL or log-GFAP as the dependent variable and with or without adjustments for sex, age, recent relapses (<60 days) at baseline, treatment at baseline, EDSS score at baseline, and log (baseline NfL or GFAP). Four patients treated with conventional immunosuppressants at follow-up were excluded due to the small sample size, and five patients who provided only baseline samples were also excluded in subsequent analysis. Thus, 63 patients who were regularly treated with disease-modifying drugs were included in a subcohort. According to our previous study protocol, patients in the tocilizumab and rituximab groups who were previously treated with concomitant corticosteroids could be included without prior washout and were allowed to continue previous treatment in the first 3 months.10,14 Two patients who switched from tocilizumab to rituximab had stopped their treatment 6 months prior to baseline. Most patients provided three blood samples (at baseline, month 6, and month 12). The treatment effects in NMOSD were visualized as line plots using original, untransformed data with geometric means, and the 95% CIs and p values were obtained from mixed-effect models. High pNfL and pGFAP concentrations were defined as concentrations that were approximately twice as high as the geometric means in HCs from our dataset (NfL ⩾ 26 pg/mL, GFAP ⩾ 140 pg/mL), according to a previous report. 15 At the end of the follow-up, we also compared the proportion of patients with high pNfL or pGFAP levels but without clinical attack in different treatment groups.
The pGFAP, pNfL, and their ratio (pGFAP/pNfL) were used to distinguish NMOSD from HCs using novel machine learning algorithms, including logistic regression, random forest, and neural network (R packages: e1071 for SVM, nnet for NNET, random Forest for Random Forest, xgboost for XGBoost, stats for logistic regression). Models included sex, age, and other clinical variables as covariates. Receiver operating characteristic (ROC) curves via different methodologies were generated to estimate the predictive accuracy of biomarkers. The mean decrease in Gini score obtained from random forest was defined as the relative importance score, and a higher mean decrease in Gini score indicates greater predictor variable in predicting outcomes. Violin plots represent ranking distributions generated by rerunning the model 50 times.
All analyses were performed via SPSS version 23.0 (IBM Corp., Armonk, NY, USA) or R version 4.0.3 (The R Project, Vienna, Austria). Figures were generated with GraphPad Prism version 8.4.2 (GraphPad Software Inc, San Diego, CA, USA) and R version 4.0.3 (The R Project). The significance level was set at p < 0.05, two-tailed.
Results
Demographic features and biomarker levels in different groups
The baseline characteristics of the participants are presented in Table 1. The median ages were 49.0 (IQR, 33.3–59.0) years in patients with NMOSD and 41.0 (IQR, 29.8–55.3) years in HCs. Most participants were women (NMOSD, 64/72, 88.9%; HCs, 32/38, 84.2%). For patients with NMOSD, the median EDSS scores were 3.3 (IQR, 2.0–7.0), and the median disease duration since first onset was 2.7 (IQR, 1.8–7.2) years. Most (64/72, 88.9%) patients were seropositive with AQP4-IgG.
Table 1.
Demographic features of all participants at baseline.
| HCs (n = 38) | NMOSD (n = 72) | p value | |
|---|---|---|---|
| Female, n (%) | 32 (84.2) | 64 (88.9) | 0.552 |
| Age at baseline, median (IQR) | 41.0 (29.8–55.3) | 49.0 (33.3–59.0) | 0.407 |
| Age at onset, median (IQR) | — | 43.5 (28.3–56.0) | |
| Disease duration(y), median (IQR) | — | 2.71 (1.81–7.24) | |
| Recent relapse (<2 month, n %) | — | 42 (58.3) | |
| Number of relapse event, median (IQR) | — | 2.0 (1.0–4.0) | |
| ARR at baseline, median (IQR) | — | 0.46 (0.00–0.78) | |
| EDSS score, median (IQR) | — | 3.3 (2.0–7.0) | |
| AQP4-ab positive, n (%) | — | 64 (88.9) | |
| Prior treatment at baseline, n (%) | |||
| a No treatment | 23 (31.9) | ||
| Prednisone | 21 (29.2) | ||
| Azathioprine | 2 (2.8) | ||
| Prednisone + Azathioprine | 1 (1.4) | ||
| Mycophenolate mofetil | 3 (4.3) | ||
| Prednisone + Mycophenolate mofetil | 1 (1.4) | ||
| Rituximab | 8 (11.1) | ||
| Tocilizumab | 13 (18.1) | ||
| NfL values, median (IQR) | 11.5 (7.0–23.3) | 18.3 (11.2–39.3) | 0.001 |
| GFAP values, median (IQR) | 68.7 (59.44–80.79) | 149.7 (88.56–406.46) | <0.001 |
Values indicate median (interquartile range) or number (percent). ARR, Annualized relapse rates; DMD, Disease-Modifying Drug; EDSS, Expanded Disability Status Scale; GFAP, glial fibrillary acidic protein; HC, healthy control; IQR, interquartile range; NMOSD, neuromyelitis optica spectrum disorders.
No treatment was defined as patients who first diagnosed, or at relapse stage did not receive any prior treatment, including high dose intravenous steroids, plasma exchange (PE), intravenous immunoglobulin (IVIG), or any DMD treatment at baseline.
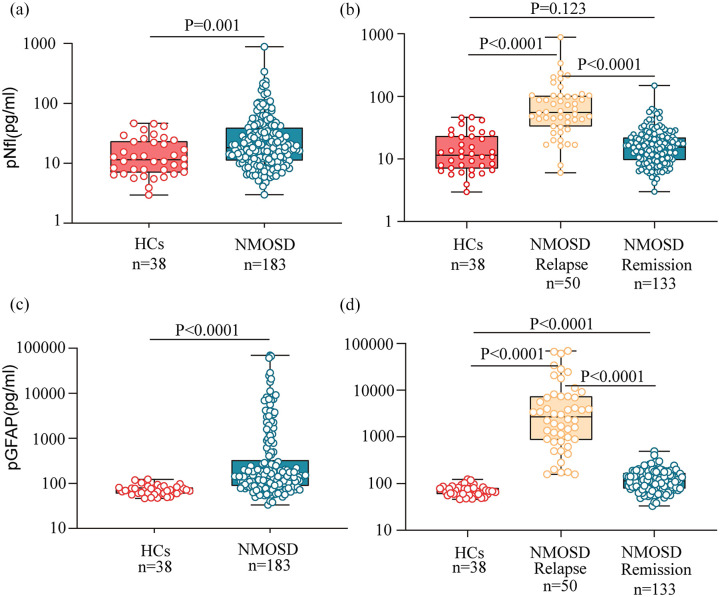
The median pNfL and pGFAP levels were 11.5 (IQR, 7.0–23.3) and 68.7 (IQR, 59.4–80.8) pg/mL in the HCs, and both were positively associated with age (n = 38; log-pNfL, β = 1.001; p < 0.001; log-pGFAP, β = 1.004; p < 0.001) but did not differ between sexes. In patients with NMOSD, the median pNfL levels were 18.3 (IQR, 11.2–39.3) pg/mL, and the median pGFAP levels were 149.7 (IQR, 88.6–406.5) pg/mL. Both the pNfL and pGFAP levels were significantly higher in patients with NMOSD than in the HCs [p = 0.001 and p < 0.001, respectively; Figure 1(a) and (b)]. We then compared the pNfL and pGFAP levels during NMOSD relapse and remission. We found that both pNfL and pGFAP levels during relapses were significantly higher than those in remission [NfL: relapse, 55.4 (IQR, 32.8–101.4) pg/mL; remission, 15.5 (IQR, 9.4–21.9) pg/mL; p < 0.001; GFAP: relapse, 2691.3 (IQR, 867.7–7362.0) pg/mL; remission, 114.0 (IQR, 78.6–168.2) pg/mL; p < 0.001; Figure 1(c) and (d)]. The pNfL levels showed no difference in NMOSD remission compared with HCs (p = 0.123). However, the pGFAP levels during NMOSD remission remained significantly higher than those in HCs (p < 0.001). Both the pNfL and pGFAP levels significantly decreased over time after relapses in patients with NMOSD (both p < 0.001; Supplemental material Figure S1).
Figure 1.
pNfL and pGFAP levels in NMOSD and healthy controls. The box represents the median and the interquartile range with the original data, in panel a and b, plasma NfL levels and plasma GFAP levels were compared between patients with NMOSD and HCs, in panel c and d, NMOSD disease group were subdivided into relapse and remission states, and compared to HCs; p values were obtained from analysis of mixed effect model for log-NfL and log-GFAP, which were adjusted for age and sex; GFAP, glial fibrillary acidic protein; HCs, healthy controls; NfL, neurofilament light chain; NMOSD, neuromyelitis optica spectrum disorders.
Association of the biomarker levels with demographic features of NMOSD at baseline
Univariate analysis showed that the baseline log-pNfL levels were positively associated with age at baseline (p < 0.001), EDSS score (p < 0.001), and recent relapses (p < 0.001) and negatively associated with treatment status (p < 0.001) in patients with NMOSD (Table 2). Multivariable analysis showed that higher baseline log-pNfL levels were independently associated with age (p = 0.017), EDSS score (p = 0.002), and recent relapses (p < 0.001; Table 2). However, the association of treatment intervention with baseline log-pNfL levels did not remain under the multivariate analysis (p = 0.253).
Table 2.
The associations between blood NfL levels and clinical variables in NMOSD patients at baseline.
| Variable | Univariate analysis | Multivariate analysis | ||||
|---|---|---|---|---|---|---|
| exp(β) value | 95% CI | p value | exp(β) value | 95% CI | p value | |
| Age at baseline | 1.01 | 0.74–1.40 | <0.001 | 1.01 | 1.00–1.01 | 0.017 |
| Sex (Ref = female) | 1.26 | 0.91–1.74 | 0.162 | Not included | ||
| EDSS | 1.11 | 1.08–1.15 | <0.001 | 1.06 | 1.02–1.09 | 0.002 |
| ARR | 0.94 | 0.80–1.11 | 0.470 | Not included | ||
| Number of relapse event | 1.00 | 0.96–1.04 | 0.888 | Not included | ||
| Disease duration | 1.00 | 0.98–1.02 | 0.983 | Not included | ||
| Recent relapse (<60d) | ||||||
| No | Ref | |||||
| Yes | 1.73 | 1.47–2.04 | <0.001 | 1.37 | 1.14–1.63 | <0.001 |
| Treatment at baseline | ||||||
| No treatment a | Ref | |||||
| Treated | 0.62 | 0.51–0.75 | <0.001 | 0.90 | 0.75–1.08 | 0.253 |
ARR, Annualized relapse rates; CI, confidence interval; DMD, Disease-Modifying Drug; EDSS, Expanded Disability Status Scale; NfL, neurofilament light chain; NMOSD, neuromyelitis optica spectrum disorders; pNfL, plasma neurofilament light chain; Ref, reference.
No treatment was defined as patients who first diagnosed, or at relapse stage did not receive any prior treatment, including high dose intravenous steroids, plasma exchange (PE), intravenous immunoglobulin (IVIG), or any DMD treatment at baseline; Univariable estimates are given for all tested variables, multivariable estimates are only provided for the variables used in the final multiple lineal model; the estimates β were back transformed (exp(β)) because dependent variable pNfL was log-transformed.
The univariate model also revealed that baseline log-pGFAP levels in patients with NMOSD were significantly associated with age (p = 0.002), EDSS score (p < 0.001), recent relapse (p < 0.001), and treatment intervention (p < 0.001). The multivariable model revealed that log-pGFAP levels were independently associated with the EDSS score (p < 0.001) and recent relapses (p < 0.001). Treatment intervention tended to be an independent factor (p = 0.054) that affected pGFAP levels in our analysis (Table 3).
Table 3.
The associations between blood GFAP levels and clinical variables in NMOSD patients at baseline. .
| Variable | Univariate analysis | Multivariate analysis | ||||
|---|---|---|---|---|---|---|
| exp (β) value | 95% CI | p value | exp (β) value | 95% CI | p value | |
| Age at baseline | 1.02 | 1.01–1.03 | 0.002 | 1.00 | 0.99–1.01 | 0.687 |
| Sex (Ref = female) | 1.18 | 0.62–2.25 | 0.607 | Not included | ||
| EDSS | 1.23 | 1.15–1.32 | <0.001 | 1.12 | 1.06–1.18 | <0.001 |
| ARR | 1.04 | 0.76–1.43 | 0.800 | Not included | ||
| Number of relapse event | 1.02 | 0.94–1.10 | 0.689 | Not included | ||
| Disease duration | 1.00 | 0.96–1.04 | 0.910 | Not included | ||
| Recent relapse (<60d) | ||||||
| No | Ref | |||||
| Yes | 3.84 | 2.95–5.00 | <0.001 | 2.62 | 1.95–3.52 | <0.001 |
| Treatment at baseline | ||||||
| No treatmenta | Ref | |||||
| Treated | 0.33 | 0.23–0.47 | <0.001 | 0.74 | 0.54–1.00 | 0.054 |
ARR, Annualized relapse rates; CI, confidence interval; DMD, Disease-Modifying Drug; EDSS, Expanded Disability Status Scale; GFAP, glial fibrillary acidic protein; NMOSD, neuromyelitis optica spectrum disorders; pGFAP, plasma glial fibrillary acidic protein; Ref, reference.
No treatment was defined as patients who first diagnosed, or at relapse stage did not receive any prior treatment, including high dose intravenous steroids, plasma exchange (PE), intravenous immunoglobulin (IVIG), or any DMD treatment at baseline; Univariable estimates are given for all tested variables, multivariable estimates are only provided for the variables used in the final multiple lineal model; the estimates β were back transformed [exp(β)] because dependent variable pGFAP was log-transformed.
Treatment response of log-pNfL and log-pGFAP levels in NMOSD
We next assessed the change rate of each marker treated with different drugs in NMOSD. The subcohort included 32 samples from 14 (22.2%) patients with oral prednisone, 57 samples from 21 (33.3%) patients with low-dose rituximab, and 81 samples from 28 (44.4%) patients with tocilizumab. The median follow-up in these 63 patients with repeated plasma samples was 371 (IQR, 343–406) days. Detailed baseline demographics and clinical characteristics are shown in Supplemental material Table S1.
Compared with prednisone, both rituximab and tocilizumab had significantly lower pNfL concentrations at the end of study (Table 4 and Figure 2(a)). The reductions in pNfL were 35% [exp(β), 0.65; 95% CI, 0.56–0.75; p < 0.001] in the tocilizumab group and 21% [exp(β), 0.79; 95% CI, 0.68–0.93; p = 0.005] in the rituximab group relative to those in the prednisone group. In the analysis of log-pGFAP (Table 5 and Figure 2(b)), the concentrations of pGFAP decreased by 36% [exp(β): 0.64; 95% CI, 0.51–0.80; p < 0.001] in the tocilizumab group and by 23% [exp(β), 0.77; 95% CI, 0.61–0.98; p = 0.041] in the rituximab group. During the follow-up period, one patient treated with tocilizumab, two patients treated with rituximab, and two patients treated with prednisone experienced relapses. The pNfL and pGFAP concentrations increased rapidly in these patients during the relapse phase. The median pNfL and pGFAP levels were 92.9 (IQR, 38.0–132.3) and 2138.9 (867.7–14,013.5) pg/mL, respectively.
Table 4.
Mean changes in log-neurofilament light in plasma (pNfL) values between baseline and on-treatment measures using unadjusted and adjusted analysis.
| Changes in log-pNfL values without adjustment analysis; exp(β) (95% CI) | p value | Changes in log-pNfL values with adjustment variables; exp(β) (95% CI) | p value | |
|---|---|---|---|---|
| PRE | Ref | Ref | ||
| RTX | 0.74 (0.60–0.91) | 0.008 | 0.79 (0.68–0.93) | 0.005 |
| TCZ | 0.65 (0.53–0.79) | <0.001 | 0.65 (0.56–0.75) | <0.001 |
All estimates were calculated with a mixed effect model for repeated measurements with three different treatment group as the main explanatory variable, adjusted with or without baseline clinical variable: age, EDSS score, recent relapse(yes/no), prior treatment, and baseline pNfL levels; The estimates have been back transformed to the original scale. CI, confidence interval; EDSS, Expanded Disability Status Scale; pNfL, plasma neurofilament light chain; PRE, prednisone; Ref, reference; RTX, Rituximab; TCZ, tocilizumab.
Figure 2.
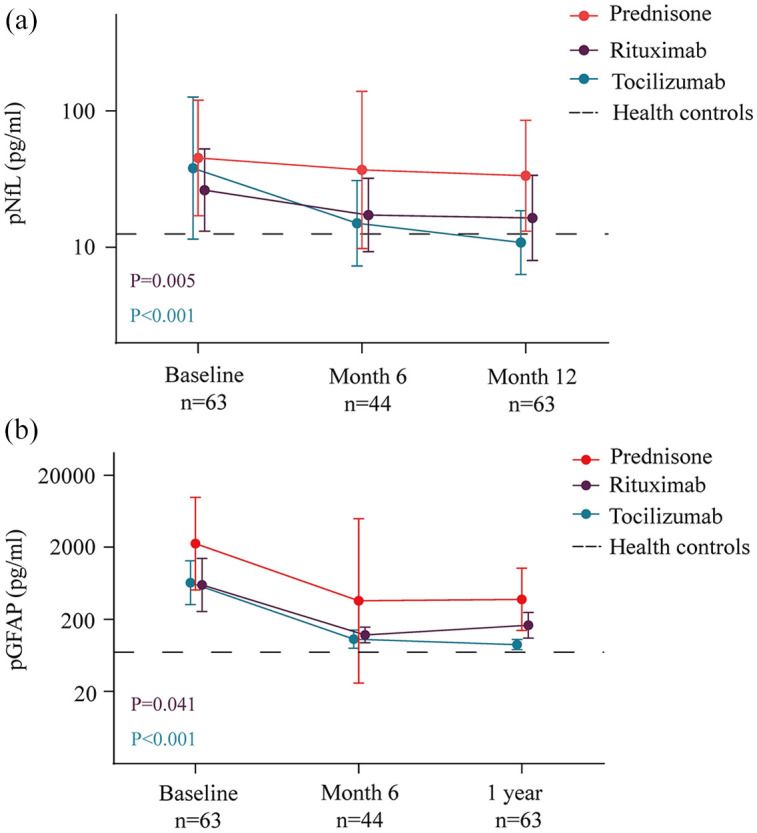
Treatment effects on pNfL and pGFAP levels in follow-up. On treatment effects of tocilizumab and rituximab on pNfL and pGFAP levels were compared to Prednisone, the figures show the original, untransformed data with geometric means of NfL with 95% confidence intervals, p values were from mixed models for repeated measurements of log-NfL and log-GFAP. Dotted line represents plasma NfL (geometric means) concentrations in healthy controls; GFAP, glial fibrillary acidic protein; n, number of evaluable samples; NfL, neurofilament light chain.
Table 5.
Mean changes in log–Glial fibrillary acidic protein (pGFAP) values between baseline and on-treatment measures provided by unadjusted and adjusted analysis.
| Changes in log-pGFAP values without adjustment analysis; exp(β) (95% CI) | p value | Changes in log-pGFAP values with adjustment variables; exp(β) (95% CI) | p value | |
|---|---|---|---|---|
| PRE | Ref | Ref | ||
| RTX | 0.67 (0.52–0.88) | 0.005 | 0.77 (0.61–0.98) | 0.041 |
| TCZ | 0.55 (0.43–0.71) | <0.001 | 0.64 (0.51–0.80) | <0.001 |
All estimates were calculated with a mixed effect model for repeated measurements with 3 different treatment group as the main explanatory variable, adjusted with or without baseline clinical variable: age, EDSS score, recent relapse(yes/no), prior treatment and baseline pGFAP levels; The estimates have been back transformed to the original scale. CI, confidence interval; pGFAP, plasma glial fibrillary acidic protein; Ref, reference; PRE, prednisone; RTX, Rituximab; TCZ, tocilizumab.
At the end of study, the NfL levels in the tocilizumab and rituximab groups were comparable to those of HCs [tocilizumab, 8.5 (IQR, 7.1–17.9) pg/mL; p = 0.426; rituximab, 14.0 (IQR, 9.9–21.8) pg/mL; p = 0.216; prednisone, 25.6 (IQR, 15.4–63.3) pg/mL; p < 0.001]. In patients who did not experience clinical relapses, 4 (28.6%) of 14 patients in the prednisone group had high NfL concentrations, compared with 1 (4.8%) of 21 patients in the rituximab group and 2 (7.1%) of 28 patients in the tocilizumab group. However, the pGFAP concentrations remained even higher than those of HCs in all treatment groups [tocilizumab, 88.9 (IQR, 63.4–131.8) pg/mL; p = 0.012; rituximab, 136.9 (IQR, 86.8–163.6) pg/mL; p < 0.001; prednisone, 193.7 (IQR, 162.4–590.7) pg/mL; p < 0.001]. Using a similar method to define high pGFAP concentrations (GFAP ⩾ 140 pg/mL), 8 (57.1%) of 14 patients in the prednisone group had high pGFAP levels; the corresponding proportions in the rituximab group and tocilizumab group were 33.3% and 21.4%, respectively.
The predictive value of biomarker levels in NMOSD
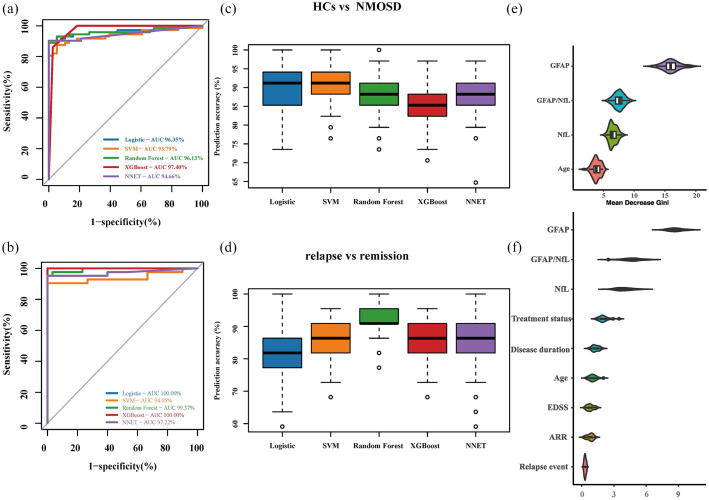
Finally, we used the two biomarkers (pGFAP and pNfL) and their ratio to differentiate NMOSD patients from HCs and to determine the relapse status versus remission in NMOSD. We conducted different statistical models to test the categorizing power of the biomarkers and ROC analyses for plasma biomarkers. Overall, multiple models showed greater areas under the curve in differentiating NMOSD from HCs (range, 93.79%–97.40%) and in distinguishing relapse versus remission in NMOSD (range, 94.05%–100%; Figure 3(a)–(d)). We then used random forest analysis to directly illustrate the relative predictive ability of these biomarkers and found that pGFAP ranked as a stronger predictor to discriminate NMOSD from HCs (Figure 3(e)). Similar results were obtained for the discriminating factor of relapse in NMOSD (Figure 3(f)).
Figure 3.
Discriminatory ability of GFAP and NfL for disease state. ROC curves with AUC indicating the discriminatory ability of blood biomarkers and clinical variables as predictors to distinguish (a) between HCs and NMOSD and (b) between NMOSD relapse and remission stage via different machine learning algorithms. Distributions were generated from rerunning each model 100 times, containing 70% of the samples as the Training set, and 30% samples as the test set (randomly selected). The boxes (c and d) represent with the median and 25% and 75% percentiles, dots are values under and above the adjacent values outliers. In panel E and F, relative importance scores were defined as the mean decrease in Gini score obtained from random forest models. Higher relative importance score indicates a greater relative importance of the variable in predicting value HCs versus NMOSD, and NMOSD relapse versus remission stage. Violin plots represent ranking distributions generated by rerunning the model 50 times. AUC, area under the curve; HCs, healthy controls; ROC, receiver-operating characteristic.
Discussion
In this study, we used SIMOA to measure pNfL and pGFAP concentrations in patients with NMOSD and compared their responsiveness to different disease-modifying drugs. Our results revealed that baseline pNfL and pGFAP levels in patients with NMOSD were independently associated with age, EDSS score, and the presence of relapse stage and that pGFAP levels tended to be affected by treatments. Further analysis showed that both tocilizumab and rituximab significantly reduced the pNfL and pGFAP levels at the end of follow-up compared with corticosteroid. Our research indicated that the pNfL and pGFAP concentrations appear to be feasible biomarkers in monitoring treatment response in NMOSD.
Several studies have revealed that blood NfL may be a good biomarker of treatment response in retrospective cohorts in multiple sclerosis (MS). Serum NfL levels have been shown to predict the course and response to treatment of MS from both relapsing and progressive cohorts. Longitudinal NfL dynamic changes have been reported with disease-modifying drugs for relapsing MS, including dimethyl fumarate, 16 IFN-β1a, 15 fingolimod,15,17 rituximab, 18 and alemtuzumab. 19 Blood NfL levels respond consistently within 3–6 months of the start of these therapies. The reductions in NfL appeared to be associated with clinical and imaging outcomes, and the response to NfL is also in line with the treatment efficacies of these drugs. 20 Recently, a Swedish cohort study included more than 1000 patients with relapsing MS receiving six different treatments, and the largest reductions in pNfL concentration were found in patients treated with alemtuzumab (48%) and the smallest reductions were in patients treated with teriflunomide (7%). 21 In ASCLEPIOS I and II, serum NfL was used to compare efficacy and safety between ofatumumab and teriflunomide for relapsing MS. In ASCLEPIOS I, the serum NfL levels were lower in the ofatumumab group than in the teriflunomide group by 7% at month 3, by 27% at month 12, and by 23% at month 24. Corresponding differences in ASCLEPIOS II were 11%, 26%, and 24%, respectively. 22 In progressive MS, treatment was associated with lower NfL concentrations in phase III trials. 23 In the ORATORIO trial (a phase III randomized trial of ocrelizumab in primary progressive MS), serum NfL was 15.7% lower with ocrelizumab versus 0.2% lower with placebo. In progressive MS trials, targeting disease progression may be confounded by intercurrent disease activity, which restricts the application of measurement and requires the integration of more clinical, biological, and imaging outcomes in the future. 23 We postulated that NfL may also indicate treatment responses in NMOSD. Previous studies have reported no significant change in serum NfL levels between different treatment status in NMOSD patients.9,24 However, these studies included only patients treated with corticosteroid or conventional oral immunosuppressants but not with newly emerging molecular targeted drugs. A recent Korean study reported that serum NfL levels in NMOSD patients treated with rituximab remained stable in the 2-year follow-up period. 25 In our analysis, tocilizumab reduced plasma NfL levels by 35% compared with prednisone by the end of study, and the corresponding reductions were 21% in the rituximab group. Meanwhile, the median pNfL levels during NMOSD remission were comparable to the levels in the HCs in our cohort at the end of follow-up, both in the tocilizumab and rituximab groups. Therefore, our results suggest that the treatment effects of these molecular-targeted drugs on the reduction of blood NfL levels were greater than those of corticosteroid and might better reflect results in the real world. Taken together, blood NfL could be a feasible biofluid marker of treatment response and could be a potential key endpoint that reveals treatment effect in future clinical trial of NMOSD.
Previous studies showed that increased GFAP levels during NMOSD relapse is consistent with primary astrocytopathy, which is mediated by AQP4-IgG.26,27 An earlier study reported that NMO-related optic neuritis can be reflected by increased serum GFAP levels. 28 With the improvement of detection assay sensitivity, increasing evidence has shown that increased blood GFAP levels in patients with NMOSD were strongly associated with disease activity by SIMOA assay.9,29,30 Our results support previous reports that blood GFAP is a sensitive biomarker of disability and disease activity in NMOSD. Of interest, our results indicated that the pGFAP levels also tended to be affected by treatment status. In the N-Momentum clinical trial, a double-masked, placebo-controlled clinical trial of inebilizumab, the serum GFAP levels of patients who did not experience NMOSD attack during follow-up started to decrease with inebilizumab treatment after week 4. In contrast, patients who were administered with placebo have shown greater proportions of increased serum GFAP concentration. 31 The reductions in pGFAP in the tocilizumab and rituximab groups were both greater than that in the prednisone group in our analysis, which was consistent with treatment response. In addition, the blood GFAP concentrations in some patients with NMOSD in the remission stage remained higher than those of HCs. One possible explanation is that patients with NMOSD have continued astrocytic damage even during clinical remission. This was also supported by the observation that patients with high serum GFAP levels tended to experience relapse more frequently in future follow-up.9,31 These results indicate that subclinical disease activity occurs during the disease progression of NMOSD, independent of classical attacks, which were consistent with previous research in the cervical spinal cord atrophy, white matter demyelination, and retinal thinning.32–34 Furthermore, lower GFAP levels in the tocilizumab and rituximab group might reflect the treatment effects of these drugs in other aspects.
Previous studies showed that GFAP and NfL could classify MS patients with different disease courses.35,36 The serum GFAP/NfL quotient during relapse could be a useful biomarker for differentiating NMOSD from MS, 9 and the serum tau levels may discriminate patients with myelin oligodendrocyte glycoprotein antibody–associated disease from AQP4-IgG–seropositive patients with NMOSD. 30 Our analysis explored how the two blood biomarkers, GFAP and NfL, and their ratio could discriminate NMOSD patients from those with different disease courses within our study cohort. With novel machine learning algorithms, the relative importance scores in our diagnostic model indicate that GFAP is a more precise and sensitive event in predicting the current disease course of NMOSD. Our results provide a different perspective on these biomarkers and thus have future diagnostic value.
This study has several limitations. First, the patients were from a single center, and only those with Chinese ethnicity were included. Second, the number of patients in each group was small; thus, the results should be interpreted cautiously. Only a few patients were treated with azathioprine or mycophenolate mofetil; hence, the effects of these drugs on pNfL and pGFAP could not be compared. Third, as the patients were unable to get access to other Food and Drug Administration–approved drugs, including inebilizumab, eculizumab, and satralizumab, we could not compare the efficacy of drugs with different mechanisms on the longitudinal dynamics of pNfL and pGFAP. Larger longitudinal prospective real-world studies are warranted.
Conclusion
We demonstrated in this study that blood GFAP and NfL are markedly increased in NMOSD patients compared with HCs. Simultaneous measurement of pNfL and pGFAP would be potentially useful for the evaluation of markers of disease activity and disease progression and possibly as predictors of different disease courses. It is also feasible to measure the pNfL and pGFAP levels longitudinally as indicators of treatment response in the real world. The dynamics of pNfL and pGFAP may help decide adjustments of disease-modifying drugs in individualized treatments.
Supplemental Material
Supplemental material, sj-docx-1-tan-10.1177_17562864211054952 for Longitudinal treatment responsiveness on plasma neurofilament light chain and glial fibrillary acidic protein levels in neuromyelitis optica spectrum disorder by Tian-Xiang Zhang, Jing-Shan Chen, Chen Du, Pei Zeng, Huiming Zhang, Xuejiao Wang, Ye Liu, Zhenning Huang, Meng Yuan, Yu-Lin Li, Dongmei Jia, Fu-Dong Shi and Chao Zhang in Therapeutic Advances in Neurological Disorders
Acknowledgments
We thank all patients for participating in the study, and all the members of Neuroimmune Team in the Department of Neurology for various supports.
Footnotes
Author contributions: F.-D.S. and C.Z. designed the study. C.Z., J.-S.C., and T.-X.Z. analyzed data. T.-X.Z., C.D., P.Z., and C.Z. interpreted data. T.-X.Z., J.-S.C., and C.Z. drafted the manuscript. T.X.-Z., C.Z., and F.-D.S. revised the manuscript for content. X.J.W., Y.L., Z.N.H., Y.-L.L., H.M.Z., M.Y., D.M.J., and T.-X.Z. were involved in data collection. All authors critically reviewed the manuscript.
Conflict of interest statement: The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by National Science Foundation of China (81601019) and National Science Foundation of Tianjin City (18JCQNJC13200, 20JCJQJC280). Advanced Innovation Center for Human Brain Protection, Capital Medical University, Beijing, China; National Key Research and Development Program of China (2018YFC1312200).
Ethics statement: The study was conducted in accordance with the principles of the Declaration of Helsinki. The Ethical Committee of Tianjin Medical University General Hospital reviewed and approved this study (IRB2017-YX-009). All patients and HCs provided written informed consent.
ORCID iD: Tian-Xiang Zhang  https://orcid.org/0000-0001-9957-9478
https://orcid.org/0000-0001-9957-9478
Data sharing statement: Anonymized individual participant data collected for this study will be made available by request from any qualified investigator.
Supplemental material: Supplemental material for this article is available online.
Contributor Information
Tian-Xiang Zhang, Department of Neurology, Tianjin Medical University General Hospital, Tianjin Neurological Institute, Tianjin, P.R. China.
Jing-Shan Chen, Department of Neurology, Tianjin Medical University General Hospital, Tianjin Neurological Institute, Tianjin, P.R. China.
Chen Du, Department of Neurology, Tianjin Medical University General Hospital, Tianjin Neurological Institute, Tianjin, P.R. China.
Pei Zeng, Department of Neurology, Tianjin Medical University General Hospital, Tianjin Neurological Institute, Tianjin, P.R. China.
Huiming Zhang, Department of Neurology, The Third People’s Hospital of Datong, Datong, China.
Xuejiao Wang, Department of Neurology, The Third People’s Hospital of Datong, Datong, China.
Ye Liu, Department of Neurology, Tianjin Medical University General Hospital, Tianjin Neurological Institute, Tianjin, P.R. China.
Zhenning Huang, Department of Neurology, Tianjin Medical University General Hospital, Tianjin Neurological Institute, Tianjin, P.R. China.
Meng Yuan, Department of Neurology, Tianjin Medical University General Hospital, Tianjin Neurological Institute, Tianjin, P.R. China; China National Clinical Research Center for Neurological Diseases, Beijing Tiantan Hospital, Capital Medical University, Beijing, China.
Yu-Lin Li, Department of Neurology, Tianjin Medical University General Hospital, Tianjin Neurological Institute, Tianjin, P.R. China.
Dongmei Jia, Department of Neurology, Tianjin Medical University General Hospital, Tianjin Neurological Institute, Tianjin, P.R. China.
Fu-Dong Shi, Department of Neurology, Tianjin Medical University General Hospital, Tianjin Neurological Institute, Tianjin, P.R. China; China National Clinical Research Center for Neurological Diseases, Beijing Tiantan Hospital, Capital Medical University, Beijing, China.
Chao Zhang, Department of Neurology, Tianjin Medical University General Hospital, Tianjin Neurological Institute, Tianjin 300052, P.R. China; China National Clinical Research Center for Neurological Diseases, Beijing Tiantan Hospital, Capital Medical University, Beijing, China.
References
- 1. Lennon VA, Wingerchuk DM, Kryzer TJ, et al. A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis. Lancet 2004; 364: 2106–2112. DOI: 10.1016/S0140-6736(04)17551. [DOI] [PubMed] [Google Scholar]
- 2. Wingerchuk DM, Lennon VA, Lucchinetti CF, et al. The spectrum of neuromyelitis optica. Lancet Neurol 2007; 6: 805–815. DOI: 10.1016/S1474-4422(07)70216. [DOI] [PubMed] [Google Scholar]
- 3. Misu T, Takano R, Fujihara K, et al. Marked increase in cerebrospinal fluid glial fibrillar acidic protein in neuromyelitis optica: an astrocytic damage marker. J Neurol Neurosurg Psychiatry 2009; 80: 575–577. DOI: 10.1136/jnnp.2008.150698. [DOI] [PubMed] [Google Scholar]
- 4. Takano R, Misu T, Takahashi T, et al. Astrocytic damage is far more severe than demyelination in NMO: a clinical CSF biomarker study. Neurology 2010; 75: 208–216. DOI: 10.1212/WNL.0b013e3181e2414b. [DOI] [PubMed] [Google Scholar]
- 5. Khalil M, Teunissen CE, Otto M, et al. Neurofilaments as biomarkers in neurological disorders. Nat Rev Neurol 2018; 14: 577–589. DOI: 10.1038/s41582-018-0058-z. [DOI] [PubMed] [Google Scholar]
- 6. Disanto G, Barro C, Benkert P, et al. Serum neurofilament light: a biomarker of neuronal damage in multiple sclerosis. Ann Neurol 2017; 81: 857–870. DOI: 10.1002/ana.24954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Mattsson N, Cullen NC, Andreasson U, et al. Association between longitudinal plasma neurofilament light and neurodegeneration in patients with Alzheimer disease. JAMA Neurol 2019; 76: 791–799. DOI: 10.1001/jamaneurol.2019.0765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Backstrom D, Linder J, Jakobson Mo S, et al. NfL as a biomarker for neurodegeneration and survival in Parkinson disease. Neurology 2020; 95: e827–e838. DOI: 10.1212/WNL.0000000000010084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Watanabe M, Nakamura Y, Michalak Z, et al. Serum GFAP and neurofilament light as biomarkers of disease activity and disability in NMOSD. Neurology 2019; 93: e1299–e1311. DOI: 10.1212/WNL.0000000000008160. [DOI] [PubMed] [Google Scholar]
- 10. Yang CS, Yang L, Li T, et al. Responsiveness to reduced dosage of rituximab in Chinese patients with neuromyelitis optica. Neurology 2013; 81: 710–713. DOI: 10.1212/WNL.0b013e3182a1aac7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Li H, Huang Z, Jia D, et al. Low-dose rituximab treatment for new-onset generalized myasthenia gravis. J Neuroimmunol 2021; 354: 577528. DOI: 10.1016/j.jneuroim.2021.577528. [DOI] [PubMed] [Google Scholar]
- 12. Wingerchuk DM, Banwell B, Bennett JL, et al. International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 2015; 85: 177–189. DOI: 10.1212/WNL.0000000000001729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Chen J, Yang X, Zhang Y, et al. Reference values for plasma neurofilament light chain (NfL) in healthy Chinese. Clin Chem Lab Med. Epub ahead of print 19 October 2020. DOI: 10.1515/cclm-2020-1030. [DOI] [PubMed] [Google Scholar]
- 14. Zhang C, Zhang M, Qiu W, et al. Safety and efficacy of tocilizumab versus azathioprine in highly relapsing neuromyelitis optica spectrum disorder (TANGO): an open-label, multicentre, randomised, phase 2 trial. Lancet Neurol 2020; 19: 391–401. DOI: 10.1016/S1474-4422(20)30070-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Kuhle J, Kropshofer H, Haering DA, et al. Blood neurofilament light chain as a biomarker of MS disease activity and treatment response. Neurology 2019; 92: e1007–e1015. DOI: 10.1212/WNL.0000000000007032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Sejbaek T, Nielsen HH, Penner N, et al. Dimethyl fumarate decreases neurofilament light chain in CSF and blood of treatment naive relapsing MS patients. J Neurol Neurosurg Psychiatry 2019; 90: 1324–1330. DOI: 10.1136/jnnp-2019-321321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Piehl F, Kockum I, Khademi M, et al. Plasma neurofilament light chain levels in patients with MS switching from injectable therapies to fingolimod. Mult Scler 2018; 24: 1046–1054. DOI: 10.1177/1352458517715132. [DOI] [PubMed] [Google Scholar]
- 18. de Flon P, Laurell K, Sundstrom P, et al. Comparison of plasma and cerebrospinal fluid neurofilament light in a multiple sclerosis trial. Acta Neurol Scand 2019; 139: 462–468. DOI: 10.1111/ane.13078. [DOI] [PubMed] [Google Scholar]
- 19. Akgun K, Kretschmann N, Haase R, et al. Profiling individual clinical responses by high-frequency serum neurofilament assessment in MS. Neurol Neuroimmunol Neuroinflamm 2019; 6: e555. DOI: 10.1212/NXI.0000000000000555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Canto E, Barro C, Zhao C, et al. Association between serum neurofilament light chain levels and long-term disease course among patients with multiple sclerosis followed up for 12 years. JAMA Neurol 2019; 76: 1359–1366. DOI: 10.1001/jamaneurol.2019.2137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Delcoigne B, Manouchehrinia A, Barro C, et al. Blood neurofilament light levels segregate treatment effects in multiple sclerosis. Neurology 2020; 94: e1201–e1212. DOI: 10.1212/WNL.0000000000009097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Hauser SL, Bar-Or A, Cohen JA, et al. Ofatumumab versus teriflunomide in multiple sclerosis. N Engl J Med 2020; 383: 546–557. DOI: 10.1056/NEJMoa1917246. [DOI] [PubMed] [Google Scholar]
- 23. Kapoor R, Smith KE, Allegretta M, et al. Serum neurofilament light as a biomarker in progressive multiple sclerosis. Neurology 2020; 95: 436–444. DOI: 10.1212/WNL.0000000000010346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Chang X, Huang W, Wang L, et al. Serum neurofilament light and GFAP are associated with disease severity in inflammatory disorders with aquaporin-4 or myelin oligodendrocyte glycoprotein antibodies. Front Immunol 2021; 12: 647618. DOI: 10.3389/fimmu.2021.647618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Hyun JW, Kim Y, Kim SY, et al. Investigating the presence of interattack astrocyte damage in neuromyelitis optica spectrum disorder: longitudinal analysis of serum glial fibrillary acidic protein. Neurol Neuroimmunol Neuroinflamm 2021; 8: e965. DOI: 10.1212/NXI.0000000000000965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Misu T, Fujihara K, Kakita A, et al. Loss of aquaporin 4 in lesions of neuromyelitis optica: distinction from multiple sclerosis. Brain 2007; 130: 1224–1234. DOI: 10.1093/brain/awm047. [DOI] [PubMed] [Google Scholar]
- 27. Petzold A, Marignier R, Verbeek MM, et al. Glial but not axonal protein biomarkers as a new supportive diagnostic criteria for Devic neuromyelitis optica? Preliminary results on 188 patients with different neurological diseases. J Neurol Neurosurg Psychiatry 2011; 82: 467–469. DOI: 10.1136/jnnp.2009.196550. [DOI] [PubMed] [Google Scholar]
- 28. Storoni M, Petzold A, Plant GT. The use of serum glial fibrillary acidic protein measurements in the diagnosis of neuromyelitis optica spectrum optic neuritis. PLoS ONE 2011; 6: e23489. DOI: 10.1371/journal.pone.0023489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Lee EJ, Lim YM, Kim S, et al. Clinical implication of serum biomarkers and patient age in inflammatory demyelinating diseases. Ann Clin Transl Neurol 2020; 7: 992–1001. DOI: 10.1002/acn3.51070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Kim H, Lee EJ, Kim S, et al. Serum biomarkers in myelin oligodendrocyte glycoprotein antibody-associated disease. Neurol Neuroimmunol Neuroinflamm 2020; 7: e708. DOI: 10.1212/NXI.0000000000000708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Aktas O, Smith MA, Rees WA, et al. Serum glial fibrillary acidic protein: a neuromyelitis optica spectrum disorder biomarker. Ann Neurol 2021; 89: 895–910. DOI: 10.1002/ana.26067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Ventura RE, Kister I, Chung S, et al. Cervical spinal cord atrophy in NMOSD without a history of myelitis or MRI-visible lesions. Neurol Neuroimmunol Neuroinflamm 2016; 3: e224. DOI: 10.1212/NXI.0000000000000224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Jeong IH, Choi JY, Kim SH, et al. Normal-appearing white matter demyelination in neuromyelitis optica spectrum disorder. Eur J Neurol 2017; 24: 652–658. DOI: 10.1111/ene.13266. [DOI] [PubMed] [Google Scholar]
- 34. Ringelstein M, Harmel J, Zimmermann H, et al. Longitudinal optic neuritis-unrelated visual evoked potential changes in NMO spectrum disorders. Neurology 2020; 94: e407–e418. DOI: 10.1212/WNL.0000000000008684. [DOI] [PubMed] [Google Scholar]
- 35. Hogel H, Rissanen E, Barro C, et al. Serum glial fibrillary acidic protein correlates with multiple sclerosis disease severity. Mult Scler 2020; 26: 210–219. DOI: 10.1177/1352458518819380. [DOI] [PubMed] [Google Scholar]
- 36. Huss A, Otto M, Senel M, et al. A score based on NfL and glial markers may differentiate between relapsing-remitting and progressive MS course. Front Neurol 2020; 11: 608. DOI: 10.3389/fneur.2020.00608. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-docx-1-tan-10.1177_17562864211054952 for Longitudinal treatment responsiveness on plasma neurofilament light chain and glial fibrillary acidic protein levels in neuromyelitis optica spectrum disorder by Tian-Xiang Zhang, Jing-Shan Chen, Chen Du, Pei Zeng, Huiming Zhang, Xuejiao Wang, Ye Liu, Zhenning Huang, Meng Yuan, Yu-Lin Li, Dongmei Jia, Fu-Dong Shi and Chao Zhang in Therapeutic Advances in Neurological Disorders