Abstract
The role of inflammation in cardiovascular pathophysiology has gained a lot of research interest in recent years. Cardiovascular Magnetic Resonance has been a powerful tool in the non-invasive assessment of inflammation in several conditions. More recently, Ultrasmall superparamagnetic particles of iron oxide have been successfully used to evaluate macrophage activity and subsequently inflammation on a cellular level. Current evidence from research studies provides encouraging data and confirms that this evolving method can potentially have a huge impact on clinical practice as it can be used in the diagnosis and management of very common conditions such as coronary artery disease, ischaemic and non-ischaemic cardiomyopathy, myocarditis and atherosclerosis. Another important emerging concept is that of myocardial energetics. With the use of phosphorus magnetic resonance spectroscopy, myocardial energetic compromise has been proved to be an important feature in the pathophysiological process of several conditions including diabetic cardiomyopathy, inherited cardiomyopathies, valvular heart disease and cardiac transplant rejection. This unique tool is therefore being utilized to assess metabolic alterations in a wide range of cardiovascular diseases. This review systematically examines these state-of-the-art methods in detail and provides an insight into the mechanisms of action and the clinical implications of their use.
Keywords: Cardiovascular magnetic resonance (CMR), Ultrasmall superparamagnetic particles of iron oxide (USPIO), Magnetic resonance spectroscopy (MRS)
Background
With more research focus being shifted in the complex pathophysiology of cardiovascular disease it is of paramount importance that we have the appropriate diagnostic techniques to identify the complicated disease processes that lie behind the symptoms. Cardiac energy metabolism in cardiovascular disease represents the foundation of the pathophysiological mechanisms that lie behind the disease processes and phenotypes and can guide the treatment strategy. Better understanding of cardiac metabolism and energetics can aid the diagnosis, prevention and management of each specific disease process. Cardiovascular Magnetic Resonance (CMR) has proved to be a powerful tool for this exact purpose with novel methods allowing for early accurate diagnosis and establishing appropriate therapeutic targets in a timely manner. In this review, we illustrate the critical role of state-of-the-art CMR methods in the assessment of inflammation and myocardial energetics in a variety of cardiovascular diseases.
Ultrasmall superparamagnetic particles of iron oxide CMR for myocardial inflammation
Inflammation is an expanding risk factor for cardiovascular disease [1] and involved in a plethora of conditions including atherosclerosis [2], myocardial infarction [3] cardiomyopathies [4, 5], myocarditis [6] and valve disease [7]. With increasing advancements, CMR has enabled non-invasive assessment of inflammation at a myocardial level using short τ inversion recovery (STIR) sequences, late gadolinium enhancement (LGE), T1 and T2 mapping, negating the need for invasive myocardial biopsy in many patients. However, such sequences do not allow us to detect cellular inflammation. At this cellular level, macrophages play a major part in both the initiation and maintenance, as well as resolution of inflammation [8]. Identifying therefore macrophage activity is crucial as not only can it allow accurate diagnosis and monitoring of disease progression, but importantly it can enable targeted therapeutic interventions, both on macrophages and their by-products. This can allow control of the inflammation and thus slowing down, or even halting completely, specific cardiovascular disease. More recently, Ultrasmall superparamagnetic particles of iron oxide (USPIO) have been used successfully to assess cellular inflammation safely [9]. USPIO consist of nanoparticles with a diameter of < 50 nm which have the ability to be taken up by tissue-resident macrophages and neutrophils [3, 10]. These particles shorten the T2* relaxation time of tissues in which they acscumulate and therefore their impact can be assessed in T2*-weighted imaging and quantified by measuring the changes in the T2* and R2* values (R2* = 1/T2*) [10, 11]. Recently, the USPIO methodology has been validated with histological confirmation that USPIO accumulates in active cardiac macrophages and identifies areas of active cardiac inflammation [11]. The pathophysiology of USPIO and how USPIO CMR can be undertaken has been extensively reviewed elsewhere [3] and in this section we will focus on the potential clinical applications.
Multiple studies have assessed the role of USPIO CMR in STEMI or NSTEMI [11–15] and even following coronary artery bypass graft (CABG) [16]. These studies showed an increase in USPIO R2* in the infarcted and peri-infarct myocardium and this could be seen up to two weeks from the acute event (see Fig. 1). Some also showed that even the remote myocardium had a smaller increase in USPIO R2* lending therefore support that macrophage activation in the remote myocardium can effectively lead to myocardial damage. Interestingly, the study on CABG patients did not reveal significant differences in USPIO R2* indicating that perhaps the myocardial injury following CABG is less likely to be mainly driven by inflammation [16].
Fig. 1.
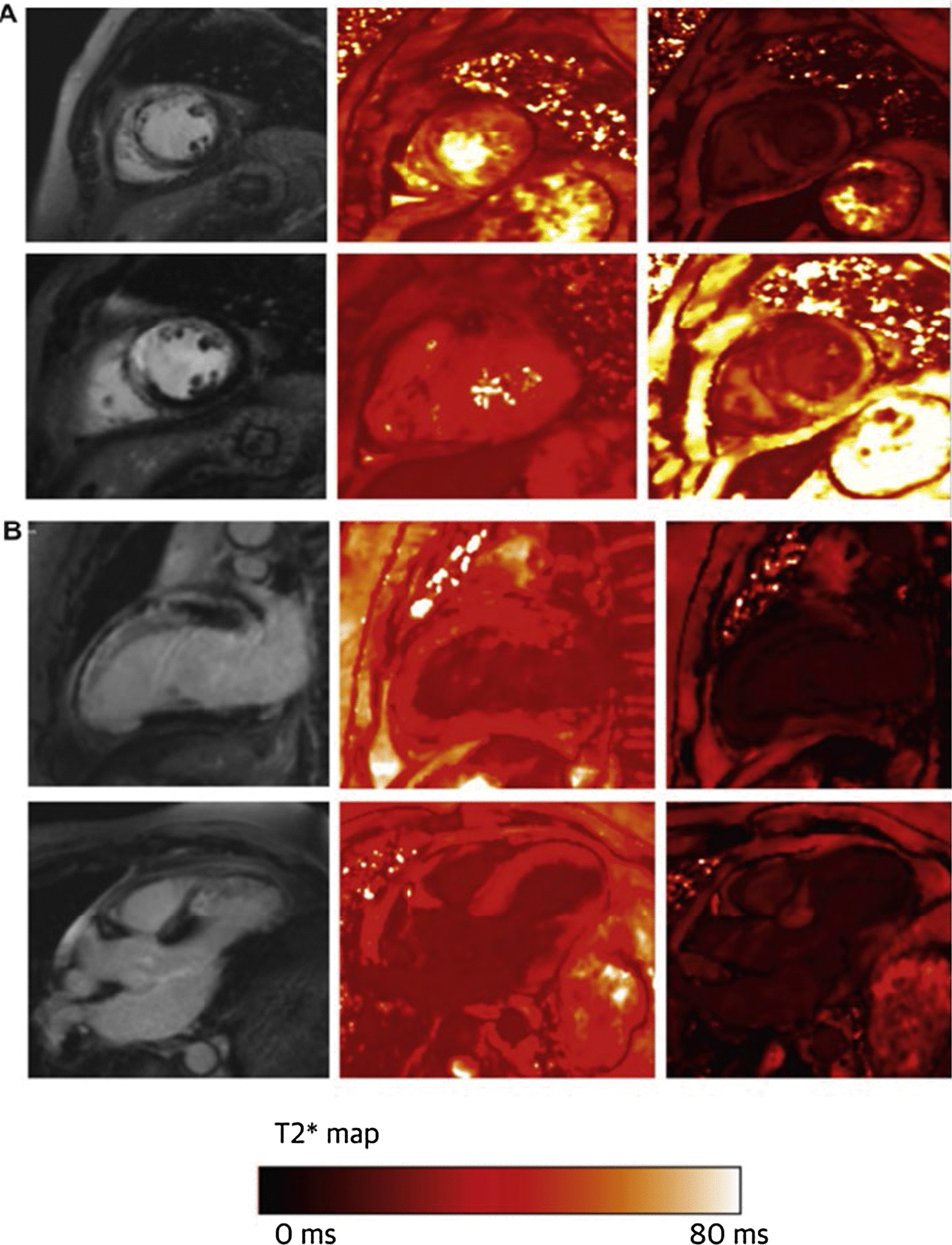
a Patient 1 week post left anterior descending artery infarction with extensive, anterior wall, transmural, mid-ventricle late gadolinium enhancement (LGE) on T1-weighted images (left column), homogeneous myocardial T2 ∗ values before erumoxytol (middle column), but intense dark ferumoxytol uptake in the region of the infarction 24 h post ferumoxytol infusion (right column). b Same patient with anteroapical, transmural LGE, again homogeneous T2 ∗ myocardial values at base, but clear ferumoxytol uptake on T2 ∗ scanning in the region of the LGE 24 h after ferumoxytol infusion. Image reproduced with permission from Merinopoulos et al. [10]
Whilst the role of oedema in Takotsubo cardiomyopathy had been recognized [17], one clinical study utilizing USPIO CMR enabled us to confirm for the first time that macrophage activity is the driving force. Fifty-five patients with acute Takotsubo cardiomyopathy were included as well as controls, and their USPIO CMR acutely showed greater differences between pre and post USPIO T2* in both the ballooning as well as the non-ballooning myocardial segments. At five months, this difference was no longer seen, indicating complete recovery of cellular inflammation. Interestingly, the patients also underwent 31P-CMR spectroscopy, which showed markedly reduced energetic state acutely, which however did not return to normal even at five months. The combination of the two methods allowed us to identify the cause of the acute decompensation (cellular inflammation) but also appreciate why some patients with Takotsubo cardiomyopathy continue to have long-lasting symptomatic and functional impairment (decreased energetics) [18] thus enabling to focus further research either in managing inflammation during the acute phase, or the decreased energetics in the longer term.
The role of inflammation in myocarditis is well acknowledged, and indeed reflected in the recent Expert Recommendation with the Revised Lake Louise Criteria [19] whereby the oedema sequences, LGE and multiparametric parametric mapping play a crucial role in the diagnosis. However, cellular inflammation in myocarditis has been less well established or studied. The largest USPIO CMR study of acute myocarditis included only nine patients [20] with imaging results as shown in Fig. 2, but it failed to reveal any differences in USPIO R2* between the patients and controls, even when only the LGE positive myocardial areas were considered. Similarly, a much smaller study, including only five patients with acute myocarditis [21] likewise failed to show any significant differences compared to controls in USPIO R2*/R1. This is perhaps unexpected and would indicate that macrophages are not the primary medium of inflammation in myocarditis, unlike reported previously by histological studies which confirmed predominantly macrophage-rich inflammation associated with myocyte damage in areas of LGE [22]. It should be considered however, that even between them, the two studies only included 14 patients. Coupled with the known blooming artefacts which are more prominent in the inferolateral myocardium, which is the most common site of myocarditis, it might still be a possibility that USPIO simply failed to detect macrophage activity and further work is warranted.
Fig. 2.
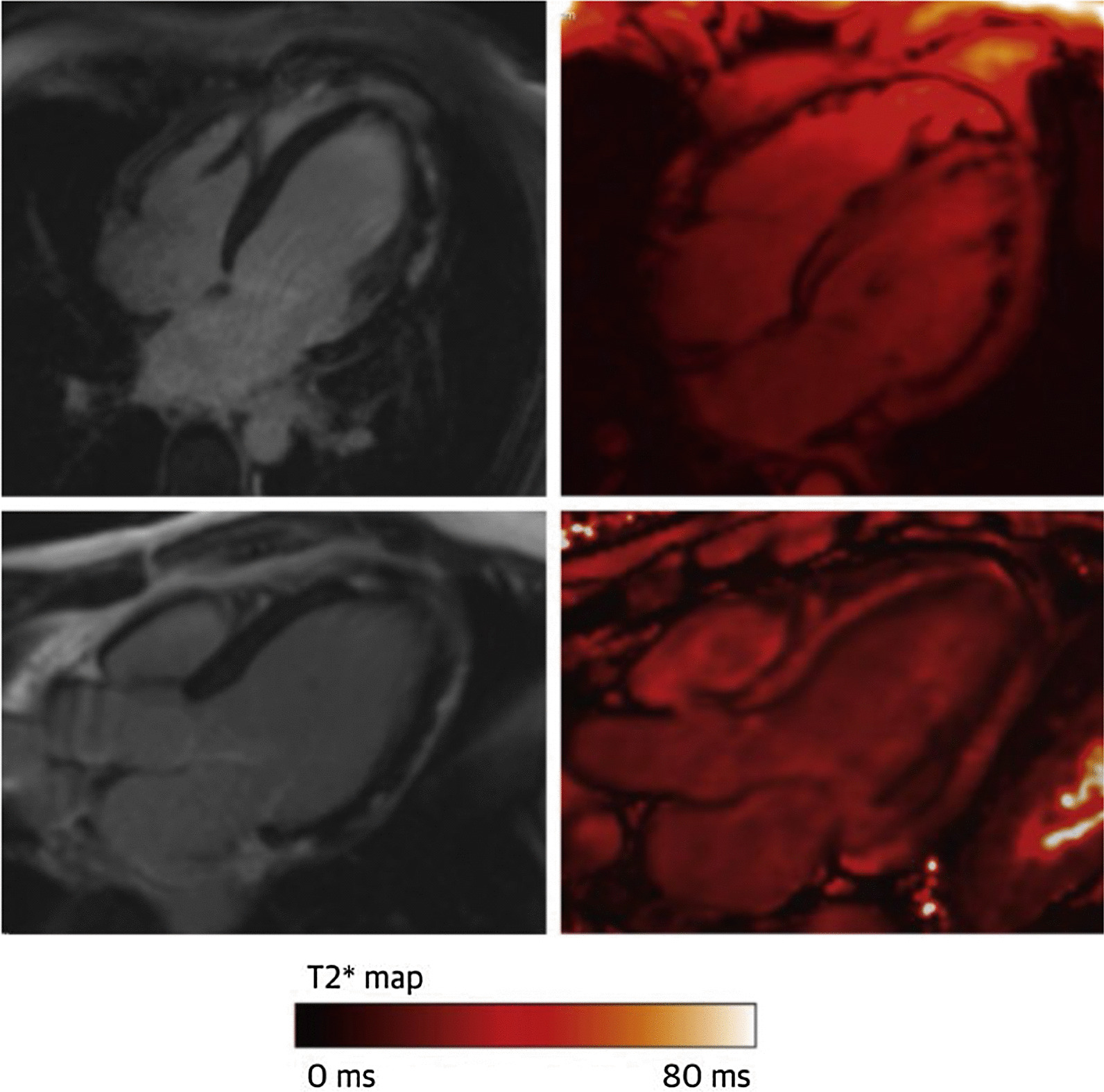
An example of a patient with acute myocarditis showing sub-epicardial LGE, inferiorly and inferolaterally on 4-chamber and 3-chamber views (left) but no evidence of ferumoxytol uptake within the regions displaying LGE 24 h following infusion (right). CMR = cardiac magnetic resonance; LGE = late gadolinium enhancement. Image reproduced with permission from Merinopoulos et al. [10]
The potential role of inflammation in triggering an ischaemic cardiomyopathy has long been considered [23]. However, many studies targeting inflammation therapeutically in this setting have been negative. Could inflammation however still play a role in non-ischaemic cardiomyopathy? A recent study involving only seven patients, studied with USPIO CMR on average 1.4 years following their acute myocardial infarction showed that both infarcted and non-infarcted myocardium had higher R2*. This would lend support to the notion that inflammation could possibly explain why only some patients progress to severe cardiomyopathy and others do not, even with similar index coronary events.
Multiple studies utilising USPIO MRI have investigated the role of inflammation in carotid atherosclerosis, aortic aneurysms and even monitor the effect of statins on atherosclerosis regression [24, 25]. These studies confirmed macrophage activity in carotid arteries which appeared independent from the degree of stenosis, suggesting therefore that both are independent risk factors. In addition, high dose statin therapy also showed a reduction in USPIO-defined inflammation in the carotids, indicating that USPIO can also potentially act as the investigation of choice for early endpoints. Whether USPIO and 18F-FDG correlate well, or whether they might indicate different parts of inflammation remains unclear as to date the studies are conflicting [26, 27]. USPIO MRI can be helpful in other vascular structures such as the abdominal aorta (see Fig. 3), where it can potentially be used to identify patients at higher risk of aneurysmal expansion (than based on diameter alone) and in turn inform the need and timing for surgery [28].
Fig. 3.
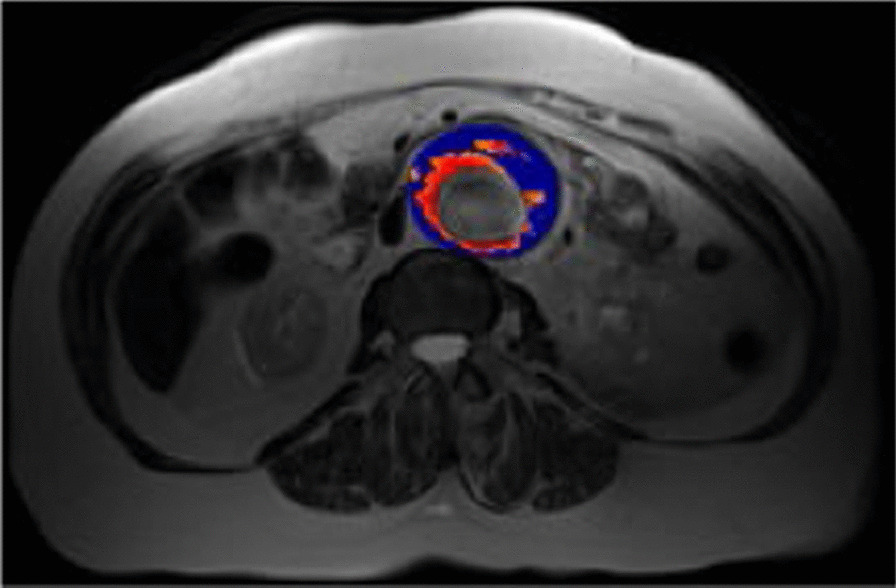
Classification of aneurysms based on uptake of ultrasmall superparamagnetic particles of iron oxide (USPIO). MRI is performed at baseline then 24 h following intravenous administration of USPIO. USPIO causes a reduction in T2* and can be quantified by comparing co-registered T2* images pre- and post- USPIO administration, presented as change in T2* represented as colour maps (as above). 'Positive uptake' of USPIO is denoted by the red colour (thresholded at change in T2* of at least 71% between pre-and post-USPIO administration), whereas blue denotes areas of no positive uptake. Of note, ‘significant’ uptake (i.e. USPIO positive) is defined as at least one focal area of USPIO uptake corresponding to 10 or more contiguous voxels of positive signal change at the aneurysm wall; USPIO uptake at the periluminal area is not thought to be clinically significant. Image courtesy of Dr Rachel Forsythe, University of Edinburgh
The clinical application of USPIO is further promoted by their safety profile. It has been demonstrated that it is a safe alternative to gadolinium-based contrast agents in patients with chronic kidney disease at risk for nephrogenic systemic fibrosis, and therefore can safely be used in this patient cohort [29]. Perhaps one of the main safety concerns is the risk of acute hypersensitivity allergic reaction, a rare but potentially life-threatening complication for which the U.S. Food and Drug Administration issued a boxed warning about the use of ferumoxytol a few years ago [30]. Since then, however, the safety profile of these agents has further improved, and large studies have successfully demonstrated and support their safe use in clinical practice [9, 31].
In summary therefore, USPIO MRI is an emerging safe method, to assess cellular inflammation. Whilst at the moment its role is mainly limited to research, future applications could include diagnostic as well as serial imaging applications, monitoring the response to treatment, or even guiding the timing of intervention.
Phosphorus magnetic resonance spectroscopy and myocardial energetic impairment
Magnetic resonance spectroscopy (MRS) is an optimal non-invasive tool used commonly to analyse cardiac energy metabolism both in the clinical and pre-clinical setting. In the heart, ATP delivery can occur through the creatine kinase (CK) system, which catalyzes the following reversible reaction: Phosphocreatine + ADP + H + ↔ Creatine + ATP. The relative concentration of phosphocreatine to ATP (PCr/ATP) is a marker of the myocardium’s ability to convert substrate into ATP for active processes, and a representantive index of the myocardial energetic state. Phosphorus magnetic resonance spectroscopy (31P-MRS) allows accurate—yet non-invasive—evaluation of the myocardial PCr/ATP ratio [32] as well as absolute levels of high-energy phosphates [33]. Using this technique, studies have shown myocardial energetic compromise to be a feature of diabetic cardiomyopathy [34–36] inherited cardiomyopathies [37, 38], valvular heart disease [39], cardiac transplant rejection [40]. In practice, at clinical field strengths (1.5 and 3 Tesla), a cardiac 31P spectrum is obtained from a 3D-localised myocardial voxel at specific acquisition times predefined by the user [41]. Acquisition times may vary with longer acquisition times required if small voxels and / or high signal to noise ratio is wanted [41]. 31P-MRS has inherently low signal to noise ratio and has low sensitivity to metabolites at low concentrations [41, 42]. This represents the main limitation of this technique as long acquisition time is required to achieve adequate signal to noise ratio [41, 42]. However, developments over the recent years are tackling this issue with the development of fast techniques mapping the phosphate metabolites [41].
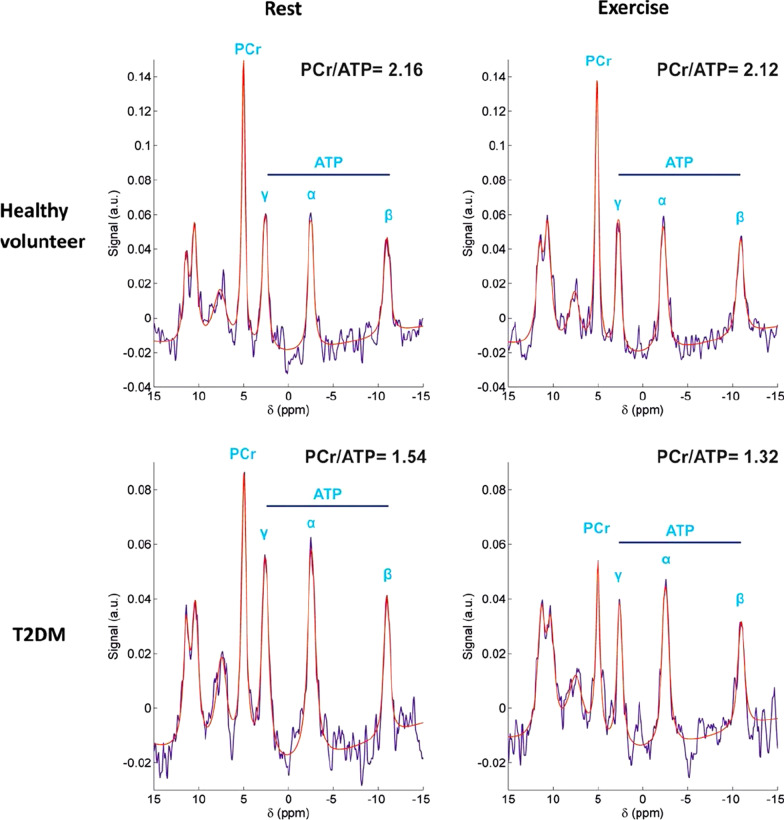
Over the last decade there has been notable progress in the field with the attempt to analyse the cardiac metabolic phenotype in detail non-invasively, and to delineate the relationship between the metabolic remodelling of the myocardium and the structural and functional changes. Due to the constantly varying cardiac workloads, efficient matching of energy supply to demand is essential for maintaining normal cardiac function [43] and myocardial metabolism is profoundly affected by changes in cardiac workload. The onset of exercise triggers a rapid increase in demand for substrate, and oxygen [44]. Rapid changes in the energy demand can be dealt by the fast responsive mechanisms of the healthy myocardium, [45], some of which include increased rates of phosphotransferase reactions [36, 46]. Assessing cardiac energetic response to exercise by 31P-MRS, aggravation of the pre-existing energetic insufficiency in patients with type 2 diabetes (T2D) was shown during increased workload (Fig. 4) [36]. Further, despite having no significant obstructive CAD, mean myocardial perfusion reserve index (MPRI) was significantly reduced in these patients [47, 48]. Pointing to the importance of an appropriate hyperaemic response during exercise to maintain cellular energy metabolism, significant correlations between MPRI with exercise energetics were demonstrated in patients with T2D [47]. While similar reductions in myocardial PCr/ATP during exercise was detected in patients with hypertrophic cardiomyopathy (HCM), in HCM this exacerbation of the energetic impairment was independent of perfusion reserve, as well as the degree of myocardial fibrosis or hypertrophy [37]. No significant changes in myocardial energetics with exercise activity was detected in comparison to the rest values in patients with dilated cardiomyopathy [49].
Fig. 4.
Rest and exercise myocardial 31P-MR spectra in a healthy volunteer (top row) and a T2D patient, suggesting a pre-existing energy deficit in the diabetic heart. Image reproduced with permission from Levelt et al. [47].
The CK system can also act as a buffer to diminish the differences in ATP and ADP levels, a feature which is crucial in maintaining the free energy of ATP hydrolysis in the cytosol. More recently, turnover rates of high-energy phosphates (CK Flux) in humans have also been measured with the saturation transfer method [50]. These dynamic turnover rates seem to be even stronger predictors of outcome in heart failure than steady-state concentrations [51]. Utilising this method, in a cohort of obese individuals and no other cardiac comorbidities, the myocardial CK reaction rate at rest was shown to be enhanced, preserving ATP delivery even with reduced PCr/ATP [52]. However, during augmented workload, ATP delivery through CK was not increased in the obese cohort in contrast with what is detected in non-obese controls suggesting that lower ATP delivery occurs during stress in obesity. This finding was shown to be associated with reduced systolic augmentation and exercise tolerance in patients with obesity. Highlighting the importance of myocardial energy supply through CK as a potential treatment focus to improve symptoms in obesity-related heart disease, weight loss has reversed these energetic changes. Similarly, the total CK flux capacity was recently shown to be reduced in patients with aortic stenosis (AS), with a reduced resting CK flux evident already in patients with moderate AS even with preserved LV systolic function [53]. These findings imply that moderate AS is associated with considerably impaired energetic state of the myocardium which is already established and a decline in the CK flux itself is not essential for the shift towards the systolic failure.
Finally, cellular metabolism can now by measured in vivo and in real-time thanks to the recent advancement of hyperpolarized 13C MRS.. Rider et al. successfully utilised hyperpolarized [1-13C] pyruvate MRS to assess downstream metabolism of [1-13C] pyruvate via PDH (pyruvate dehydrogenase, [13C] bicarbonate), lactate dehydrogenase ([1-13C] lactate), and alanine transaminase ([1-13C] alanine), in 5 patients with T2D and 5 controls at baseline, and repeated these measurements in 5 of these participants (3 T2D, 2 controls) 45 min after a 75 g oral glucose challenge [54]. They showed metabolic flux through cardiac pyruvate dehydrogenase (PDH) was noticeably declined in the individuals with T2D compared to controls. In addition, they have also detected a major increase in metabolic flux through PDH 45 min after the oral administration of 75 g of glucose in patients with T2D and in controls. This was the first study to show that physiological and pathological variations in PDH flux in the human heart can be evaluated with the use of hyperpolarized pyruvate, highlighting in this way that this method has the potential to assess metabolic shifts in a variety of cardiovascular disorders.
Conclusion
The unique CMR methods discussed in this review are without doubt a pillar that can contribute substantially to future research and clinical practice.
USPIO CMR represents a robust novel method that assesses myocardial inflammation on a cellular level by identifying macrophage activity. It can be applied in a variety of diseases, including ischaemic and non-ischaemic cardiomyopathies, enabling in this way not only precise diagnosis and monitoring, but also targeted therapeutic interventions early in the disease process. MRS is another state-of-the-art tool ideal for the non-invasive assessment of cardiac energy metabolism, with its potential expanding in the evaluation of metabolic alterations in various different cardiovascular pathological processes. Further research and clinical studies are needed in order to explore how these novel CMR techniques compare with other imaging methods, such as positron emission tomography, and what clinical information they may add in the pathophysiological processes of a wide spectrum of cardiovascular diseases [55].
Considering that tissue inflammation and myocardial energetics play a key role in the pathophysiology of a wide range of cardiovascular diseases, these novel CMR methods and techniques are proving to meet the research and clinical expectations in assessing the disease processes on a cellular level. This, in turn, can provide clear insights not only in the diagnosis but also in the risk stratification and management strategies adapted in the future.
Acknowledgements
Not applicable.
Abbreviations
- ADP
Adenosine diphosphate
- AS
Aortic stenosis
- ATP
Adenosine triphosphate
- CABG
Coronary artery bypass graft
- CAD
Coronary artery disease
- CK
Creatine kinase
- CMR
Cardiovascular magnetic resonance
- HCM
Hypertrophic cardiomyopathy
- LGE
Late gadolinium enhancement
- LV
Left ventricular
- MRS
Magnetic resonance spectroscopy
- NSTEMI
Non ST segment elevation myocardial infarction
- STIR
Short τ inversion recovery
- STEMI
ST segment elevation myocardial infarction
- T2D
Type 2 diabetes
- 31P-MRS
Phosphorus magnetic resonance spectroscopy
- PDH
Pyruvate dehydrogenase
- USPIO
Ultrasmall superparamagnetic particles of iron oxide
Authors' contributions
All authors contributed to the paper. PG conceived, planned the review and wrote parts of first draft, VT amended significantly the manuscript. EL wrote parts of first draft, AJS, HA, AC, PS, ES, AD, JBC, JRDV, SN, RN, AR, CS and CBD provided critical content review and further helped drafting the manuscript. VV wrote parts of the first draft and significantly amended the manuscript. All authors read and approved the final manuscript.
Funding
This work was funded in part by the Wellcome Trust [215799/Z/19/Z], [220703/Z/20/Z] and [205188/Z/16/Z]. For the purpose of Open Access, the author has applied a CC BY public copyright licence to any Author Accepted Manuscript version arising from this submission. The funding body played no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Availability of data and materials
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
PG is advisor for Pie Medical Imaging and Medis Medical Imaging. The remaining authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Golia E, Limongelli G, Natale F, Fimiani F, Maddaloni V, Pariggiano I, et al. Inflammation and cardiovascular disease: from pathogenesis to therapeutic target, vol 16, Current Atherosclerosis Reports. Current Medicine Group LLC 1; 2014. [DOI] [PubMed]
- 2.Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377(12):1119–1131. doi: 10.1056/NEJMoa1707914. [DOI] [PubMed] [Google Scholar]
- 3.Alam SR, Stirrat C, Richards J, Mirsadraee S, Semple SIK, Tse G, et al. Vascular and plaque imaging with ultrasmall superparamagnetic particles of iron oxide. J Cardiovasc Magn Reson. 2015;17(1):83. doi: 10.1186/s12968-015-0183-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Becker RC, Owens AP, Sadayappan S. Tissue-level inflammation and ventricular remodeling in hypertrophic cardiomyopathy. J Thromb Thrombolysis. 2020;49(2):177–183. doi: 10.1007/s11239-019-02026-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Imanaka-Yoshida K. Inflammation in myocardial disease: From myocarditis to dilated cardiomyopathy. Pathol Int. 2020;70(1):1–11. doi: 10.1111/pin.12868. [DOI] [PubMed] [Google Scholar]
- 6.Arnold JR, McCann GP. Cardiovascular magnetic resonance: Applications and practical considerations for the general cardiologist. Heart. London: BMJ Publishing Group; 2019. [DOI] [PubMed] [Google Scholar]
- 7.Manno G, Bentivegna R, Morreale P, Nobile D, Santangelo A, Novo S, et al. Chronic inflammation: a key role in degeneration of bicuspid aortic valve. J Mol Cell Cardiol. 2019;130:59–64. doi: 10.1016/j.yjmcc.2019.03.013. [DOI] [PubMed] [Google Scholar]
- 8.Fujiwara N, Kobayashi K. Macrophages in inflammation. Current drug targets: inflammation and allergy. Curr Drug Targets Inflamm Allergy. 2005;4:281–286. doi: 10.2174/1568010054022024. [DOI] [PubMed] [Google Scholar]
- 9.Nguyen KL, Yoshida T, Kathuria-Prakash N, Zaki IH, Varallyay CG, Semple SI, et al. Multicenter safety and practice for off-label diagnostic use of ferumoxytol in MRI. Radiology. 2019;293(3):554–564. doi: 10.1148/radiol.2019190477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Merinopoulos I, Gunawardena T, Stirrat C, Cameron D, Eccleshall SC, Dweck MR, et al. Diagnostic applications of ultrasmall superparamagnetic particles of iron oxide for imaging myocardial and vascular inflammation. JACC: Cardiovascular Imaging. 2020. https://www.sciencedirect.com/science/article/pii/S1936878X20306331 [DOI] [PubMed]
- 11.Lagan J, Naish JH, Simpson K, Zi M, Cartwright EJ, Foden P, et al. Substrate for the myocardial inflammation-heart failure hypothesis identified using novel USPIO methodology. JACC Cardiovasc Imaging. 2021;14(2):365–376. doi: 10.1016/j.jcmg.2020.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Alam SR, Shah ASV, Richards J, Lang NN, Barnes G, Joshi N, et al. Ultrasmall superparamagnetic particles of iron oxide in patients with acute myocardial infarction: early clinical experience. Circ Cardiovasc Imaging. 2012;5(5):559–565. doi: 10.1161/CIRCIMAGING.112.974907. [DOI] [PubMed] [Google Scholar]
- 13.Yilmaz A, Rösch S, Klingel K, Kandolf R, Helluy X, Hiller K-H, et al. Magnetic resonance imaging (MRI) of inflamed myocardium using iron oxide nanoparticles in patients with acute myocardial infarction—preliminary results. Int J Cardiol. 2013;163(2):175–182. doi: 10.1016/j.ijcard.2011.06.004. [DOI] [PubMed] [Google Scholar]
- 14.Yilmaz A, Dengler MA, van der Kuip H, Yildiz H, Rösch S, Klumpp S, et al. Imaging of myocardial infarction using ultrasmall superparamagnetic iron oxide nanoparticles: a human study using a multi-parametric cardiovascular magnetic resonance imaging approach. Eur Heart J. 2013;34(6):462–475. doi: 10.1093/eurheartj/ehs366. [DOI] [PubMed] [Google Scholar]
- 15.Stirrat CG, Alam SR, MacGillivray TJ, Gray CD, Dweck MR, Raftis J, et al. Ferumoxytol-enhanced magnetic resonance imaging assessing inflammation after myocardial infarction. Heart. 2017;103(19):1528–1535. doi: 10.1136/heartjnl-2016-311018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Alam SR, Stirrat C, Spath N, Zamvar V, Pessotto R, Dweck MR, et al. Myocardial inflammation, injury and infarction during on-pump coronary artery bypass graft surgery. J Cardiothorac Surg. 2017;12(1):1–10. doi: 10.1186/s13019-017-0681-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Abdel-Aty H, Cocker M, Friedrich MG. Myocardial edema is a feature of Tako-Tsubo cardiomyopathy and is related to the severity of systolic dysfunction: Insights from T2-weighted cardiovascular magnetic resonance. Int J Cardiol. 2009;132(2):291–293. doi: 10.1016/j.ijcard.2007.08.102. [DOI] [PubMed] [Google Scholar]
- 18.Scally C, Rudd A, Mezincescu A, Wilson H, Srivanasan J, Horgan G, et al. Persistent long-term structural, functional, and metabolic changes after stress-induced (takotsubo) cardiomyopathy. Circulation. 2018;137(10):1039–1048. doi: 10.1161/CIRCULATIONAHA.117.031841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ferreira VM, Schulz-Menger J, Holmvang G, Kramer CM, Carbone I, Sechtem U, et al. Cardiovascular magnetic resonance in nonischemic myocardial inflammation: expert recommendations. J Am Coll Cardiol. 2018;72(24):3158–3176. doi: 10.1016/j.jacc.2018.09.072. [DOI] [PubMed] [Google Scholar]
- 20.Stirrat CG, Alam SR, MacGillivray TJ, Gray CD, Dweck MR, Dibb K, et al. Ferumoxytol-enhanced magnetic resonance imaging in acute myocarditis. Heart (British Cardiac Society) 2018;104(4):300–305. doi: 10.1136/heartjnl-2017-311688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lagan J, Naish JH, Simpson K, Zi M, Cartwright EJ, Foden P, et al. Substrate for the myocardial inflammation–heart failure hypothesis identified using novel USPIO methodology. JACC: Cardiovascular Imaging. 2020. [DOI] [PMC free article] [PubMed]
- 22.Mahrholdt H, Goedecke C, Wagner A, Meinhardt G, Athanasiadis A, Vogelsberg H, et al. Cardiovascular magnetic resonance assessment of human myocarditis. Circulation. 2004;109(10):1250–1258. doi: 10.1161/01.CIR.0000118493.13323.81. [DOI] [PubMed] [Google Scholar]
- 23.Dick SA, Epelman S. Chronic heart failure and inflammation. Circulation research. Philadelphia: Lippincott Williams and Wilkins; 2016. pp. 159–176. [DOI] [PubMed] [Google Scholar]
- 24.Tang TY, Howarth SPS, Miller SR, Graves MJ, Patterson AJ, U-King-Im J-M, et al. The ATHEROMA (atorvastatin therapy: effects on reduction of macrophage activity) study: evaluation using ultrasmall superparamagnetic iron oxide-enhanced magnetic resonance imaging in carotid disease. J Am Coll Cardiol. 2009;53(22):2039–2050. doi: 10.1016/j.jacc.2009.03.018. [DOI] [PubMed] [Google Scholar]
- 25.Tang T, Howarth SPS, Miller SR, Trivedi R, Graves MJ, King-Im JU, et al. Assessment of inflammatory burden contralateral to the symptomatic carotid stenosis using high-resolution ultrasmall, superparamagnetic iron oxide-enhanced MRI. Stroke. 2006;37(9):2266–2270. doi: 10.1161/01.STR.0000236063.47539.99. [DOI] [PubMed] [Google Scholar]
- 26.Tang TY, Moustafa RR, Howarth SP, Walsh SR, Boyle JR, Li ZY, et al. Combined PET-FDG and USPIO-enhanced MR imaging in patients with symptomatic moderate carotid artery stenosis. Eur J Vasc Endovasc Surg. 2008;36(1):53–55. doi: 10.1016/j.ejvs.2008.02.006. [DOI] [PubMed] [Google Scholar]
- 27.Smits LP, Tiessens F, Zheng KH, Stroes ES, Nederveen AJ, Coolen BF. Evaluation of ultrasmall superparamagnetic iron-oxide (USPIO) enhanced MRI with ferumoxytol to quantify arterial wall inflammation. Atherosclerosis. 2017;1(263):211–218. doi: 10.1016/j.atherosclerosis.2017.06.020. [DOI] [PubMed] [Google Scholar]
- 28.MA3RS Study Investigators Aortic wall inflammation predicts abdominal aortic aneurysm expansion, rupture, and need for surgical repair. Circulation. 2017;136(9):787–797. doi: 10.1161/CIRCULATIONAHA.117.028433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Neuwelt EA, Hamilton BE, Varallyay CG, Rooney WR, Edelman RD, Jacobs PM, et al. Ultrasmall superparamagnetic iron oxides (USPIOs): a future alternative magnetic resonance (MR) contrast agent for patients at risk for nephrogenic systemic fibrosis (NSF)? Kidney Int. 2009;75(5):465–474. doi: 10.1038/ki.2008.496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Research C for DE and. FDA Drug Safety Communication: FDA strengthens warnings and changes prescribing instructions to decrease the risk of serious allergic reactions with anemia drug Feraheme (ferumoxytol). FDA. 2019 [cited 2021 Aug 7]; https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-strengthens-warnings-and-changes-prescribing-instructions-decrease.
- 31.Adkinson NF, Strauss WE, Macdougall IC, Bernard KE, Auerbach M, Kaper RF, et al. Comparative safety of intravenous ferumoxytol versus ferric carboxymaltose in iron deficiency anemia: a randomized trial. Am J Hematol. 2018;93(5):683–690. doi: 10.1002/ajh.25060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Neubauer S. The failing heart—an engine out of fuel. N Engl J Med. 2007;356(11):1140–1151. doi: 10.1056/NEJMra063052. [DOI] [PubMed] [Google Scholar]
- 33.Bottomley PA, Hardy CJ, Roemer PB. Phosphate metabolite imaging and concentration measurements in human heart by nuclear magnetic resonance. Magn Reson Med. 1990;14(3):425–434. doi: 10.1002/mrm.1910140302. [DOI] [PubMed] [Google Scholar]
- 34.Scheuermann-Freestone M, Madsen PL, Manners D, Blamire AM, Buckingham RE, Styles P, et al. Abnormal cardiac and skeletal muscle energy metabolism in patients with type 2 diabetes. Circulation. 2003;107(24):3040–3046. doi: 10.1161/01.CIR.0000072789.89096.10. [DOI] [PubMed] [Google Scholar]
- 35.Shivu GN, Phan TT, Abozguia K, Ahmed I, Wagenmakers A, Henning A, et al. Relationship between coronary microvascular dysfunction and cardiac energetics impairment in type 1 diabetes mellitus. Circulation. 2010;121(10):1209–1215. doi: 10.1161/CIRCULATIONAHA.109.873273. [DOI] [PubMed] [Google Scholar]
- 36.Levelt E, Rodgers CT, Clarke WT, Mahmod M, Ariga R, Francis JM, et al. Cardiac energetics, oxygenation, and perfusion during increased workload in patients with type 2 diabetes mellitus. Eur Heart J. 2016;37(46):3461–3469. doi: 10.1093/eurheartj/ehv442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dass S, Cochlin LE, Suttie JJ, Holloway CJ, Rider OJ, Carden L, et al. Exacerbation of cardiac energetic impairment during exercise in hypertrophic cardiomyopathy: a potential mechanism for diastolic dysfunction. Eur Heart J. 2015;36(24):1547–1554. doi: 10.1093/eurheartj/ehv120. [DOI] [PubMed] [Google Scholar]
- 38.Dass S, Holloway C, Suttie J, Mahmod M, Sever E, Watkins H, et al. (2013) Patients with Dilated Cardiomyopathy (DCM) have appropriate myocardial oxygenation response to vasodilator stress. J Cardiovasc Magn Reson. 2013;15(1):O68. doi: 10.1186/1532-429X-15-S1-O68. [DOI] [Google Scholar]
- 39.Mahmod M, Francis JM, Pal N, Lewis A, Dass S, De Silva R, et al. Myocardial perfusion and oxygenation are impaired during stress in severe aortic stenosis and correlate with impaired energetics and subclinical left ventricular dysfunction. J Cardiovasc Magn Reson. 2014;29(16):29. doi: 10.1186/1532-429X-16-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bottomley PA, Weiss RG, Hardy CJ, Baumgartner WA. Myocardial high-energy phosphate metabolism and allograft rejection in patients with heart transplants. Radiology. 1991;181(1):67–75. doi: 10.1148/radiology.181.1.1887057. [DOI] [PubMed] [Google Scholar]
- 41.Peterzan MA, Lewis AJM, Neubauer S, Rider OJ. Non-invasive investigation of myocardial energetics in cardiac disease using 31P magnetic resonance spectroscopy. Cardiovasc Diagn Ther. 2020;10(3):625–635. doi: 10.21037/cdt-20-275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Liu Y, Gu Y, Yu X. Assessing tissue metabolism by phosphorous-31 magnetic resonance spectroscopy and imaging: a methodology review. Quant Imaging Med Surg. 2017;7(6):707–726. doi: 10.21037/qims.2017.11.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Taegtmeyer H, McNulty P, Young ME. Adaptation and maladaptation of the heart in diabetes: Part I: general concepts. Circulation. 2002;105(14):1727–1733. doi: 10.1161/01.cir.0000012466.50373.e8. [DOI] [PubMed] [Google Scholar]
- 44.Rolfe DF, Brown GC. Cellular energy utilization and molecular origin of standard metabolic rate in mammals. Physiol Rev. 1997;77(3):731–758. doi: 10.1152/physrev.1997.77.3.731. [DOI] [PubMed] [Google Scholar]
- 45.Carley AN, Taegtmeyer H, Lewandowski ED. Matrix revisited: mechanisms linking energy substrate metabolism to the function of the heart. Circ Res. 2014;114(4):717–729. doi: 10.1161/CIRCRESAHA.114.301863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bittl JA, Ingwall JS. Reaction rates of creatine kinase and ATP synthesis in the isolated rat heart. A 31P NMR magnetization transfer study. J Biol Chem. 1985;260(6):3512–3517. [PubMed] [Google Scholar]
- 47.Levelt E, Rodgers CT, Clarke WT, Mahmod M, Ariga R, Francis JM, et al. Cardiac energetics, oxygenation, and perfusion during increased workload in patients with type 2 diabetes mellitus. Eur Heart J. 2016. [DOI] [PMC free article] [PubMed]
- 48.Levelt E, Piechnik SK, Liu A, Wijesurendra RS, Mahmod M, Ariga R, et al. Adenosine stress CMR T1-mapping detects early microvascular dysfunction in patients with type 2 diabetes mellitus without obstructive coronary artery disease. J Cardiovasc Magn Reson. 2017;19(1):81. doi: 10.1186/s12968-017-0397-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Dass S, Holloway CJ, Cochlin LE, Rider OJ, Mahmod M, Robson M, et al. No evidence of myocardial oxygen deprivation in nonischemic heart failure. Circ Heart Fail. 2015;8(6):1088–1093. doi: 10.1161/CIRCHEARTFAILURE.114.002169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bottomley PA, Ouwerkerk R, Lee RF, Weiss RG. Four-angle saturation transfer (FAST) method for measuring creatine kinase reaction rates in vivo. Magn Reson Med. 2002;47(5):850–863. doi: 10.1002/mrm.10130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Bottomley PA, Panjrath GS, Lai S, Hirsch GA, Wu K, Najjar SS, et al. Metabolic rates of ATP transfer through creatine kinase (CK Flux) predict clinical heart failure events and death. Sci Transl Med. 2013;5(215):215re3. doi: 10.1126/scitranslmed.3007328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Rayner JJ, Peterzan MA, Watson WD, Clarke WT, Neubauer S, Rodgers CT, et al. Myocardial energetics in obesity. Circulation. 2020;141(14):1152–1163. doi: 10.1161/CIRCULATIONAHA.119.042770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Peterzan MA, Clarke WT, Lygate CA, Lake HA, Lau Justin YC, Miller JJ, et al. Cardiac energetics in patients with aortic stenosis and preserved versus reduced ejection fraction. Circulation. 2020;141(24):1971–1985. doi: 10.1161/CIRCULATIONAHA.119.043450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Rider OJ, Apps A, Miller JJJJ, Lau JYC, Lewis AJM, Peterzan MA, et al. Noninvasive in vivo assessment of cardiac metabolism in the healthy and diabetic human heart using hyperpolarized 13C MRI. Circ Res. 2020;126(6):725–736. doi: 10.1161/CIRCRESAHA.119.316260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Fayad ZA, Calcagno C. USPIO-enhanced CMR of myocardial inflammation. JACC Cardiovasc Imaging. 2021;14(2):377–378. doi: 10.1016/j.jcmg.2020.12.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.