Abstract
Purpose of review
Dysphonia has been described as a major symptom of coronavirus disease-2019 (COVID-19). A literature review examining this topic was undertaken and is presented here, combined with insights from our experience in managing patients with laryngeal complications following mechanical ventilation for severe COVID-19 pneumonitis.
Recent findings
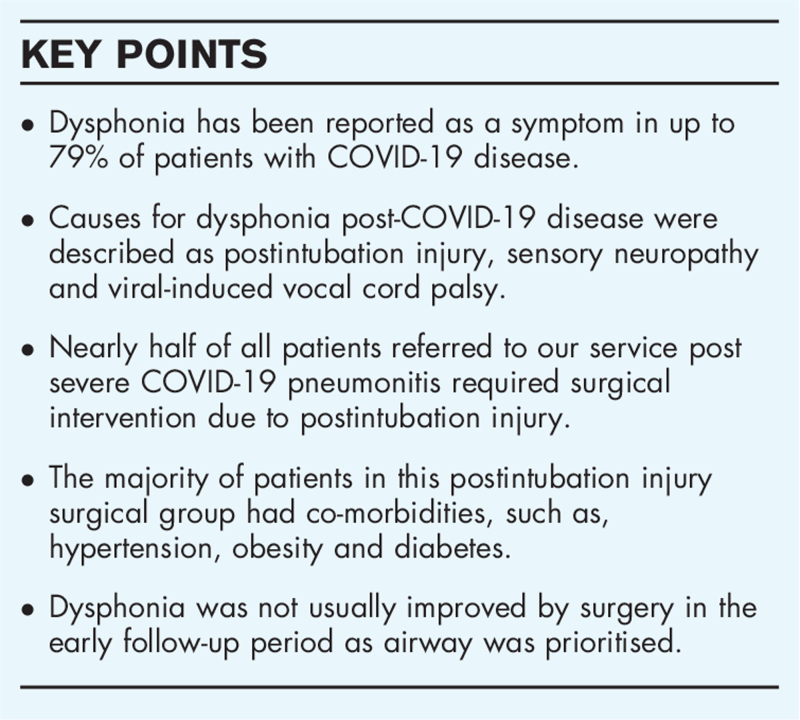
Naunheim et al. reported that patients who are most at risk of needing intubation with COVID-19 disease are those with patient-specific risk factors and these are at an increased risk for subsequent laryngotracheal injury following intubation (1). In our cohort of 105 patients referred with laryngological symptoms postintubation for COVID-19 pneumonitis, 40% presented as urgent reviews, of which almost half had severe postintubation complications requiring surgery. Perceptual voice ratings and patient-reported voice ratings varied widely, but there was no significant change in voice scores postoperatively. The reflux symptom index (RSI) scores did improve significantly (p = 0.0266). The need for surgery was associated with the presence of comorbidities for instance hypertension, diabetes and obesity in our cohort. This is in support of reported association of comorbidity as a risk factor for intubation and subsequent development of postintubation airway complications.
Summary
Dysphonia following COVID-19 infection may have multiple causes. Literature reports demonstrate intubation injury, sensory neuropathy, and postviral neuropathy are associated with voice changes. Our personal experience has confirmed postintubation injury markedly affects glottic function with resultant dysphonia attributable to scar formation, posterior glottic stenosis, granulation and subglottic stenosis. Frequent surgical intervention is required for airway patency and may have short-term further deleterious effects on phonation, although in our cohort this is not statistically significant analysing Grade, Roughness, Breathiness, Asthenia, Strain, Voice Handicap Index-10 or Airway, Voice, Swallow scores. Maximal antireflux medications and advice statistically improved RSI scores postoperatively.
Keywords: coronavirus disease-2019 pandemic, infection, dysphonia, voice, airway
INTRODUCTION
When the coronavirus disease-2019 (COVID-19) pandemic caused by the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection was declared in 2020, the effects and complications of this infection on the larynx were not known. Over the succeeding months, patients who were discharged from the intensive care unit following mechanical ventilation for severe COVID-19 pneumonitis began to present to the laryngology service with laryngeal complications, such as, stridor, dysphonia and dysphagia.
The aim of this article is to share our experience in the management of patients presenting with laryngeal complications following mechanical ventilation used as part of the management of severe COVID-19 pneumonitis and to review the existing literature on the wider effects of COVID-19 on voice.
Box 1.
no caption available
METHODS
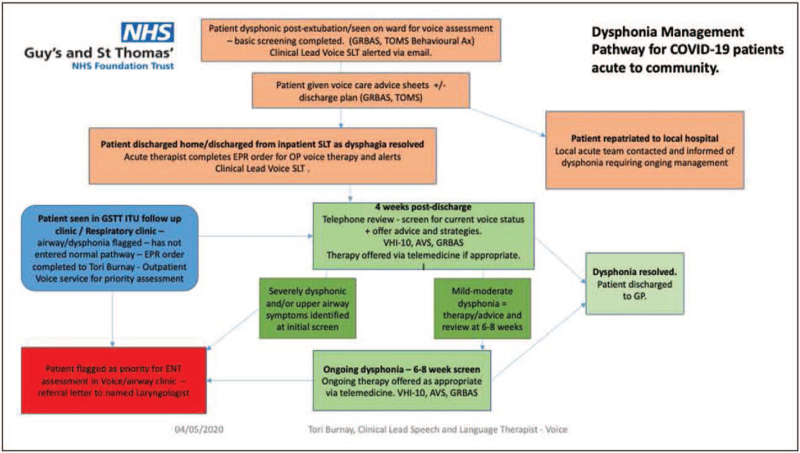
Patients who developed laryngeal complications following mechanical ventilation for severe COVID-19 pneumonitis were referred to our multidisciplinary laryngology service either directly by the critical care team, via the emergency department or via a new outpatient dysphonia management pathway (DMP) set up by the speech and language therapy (SLT) department to allow effective screening and treatment of patients in a timely manner (Fig. 1).
FIGURE 1.
Dysphonia management pathway for COVID-19 patients. COVID-19, coronavirus disease-2019.
Patients diagnosed with significant postintubation laryngeal complications requiring surgery were identified and the voice was assessed subjectively by clinicians and speech pathologists using the Grade, Breathiness, Asthenia and Strain (GRBAS) scale [2] and by patients scoring the Voice Handicap Index-10 (VHI-10) [3]. Patient perception of other laryngological symptoms was assessed using the Reflux Symptom Index (RSI) [4], and Airway–Voice–Swallow (AVS) scale [5] pre and postintervention whenever possible. Posttreatment scores were obtained between 2 and 6 weeks postoperatively. Paired t-tests were calculated using Graphpad.
RESULTS
Between March 2020 and June 2021 inclusive, 105 patients were referred to our unit with laryngeal symptoms (stridor, dysphonia and/or dysphagia) following mechanical ventilation for severe COVID-19 pneumonitis. Common laryngological complaints postintubation for COVID-19 at our 4-week screen were breathing difficulties, weak voice, vocal fatigue, difficulties with respiratory-phonatory coordination and altered voice quality. Other associated symptoms reported included, laryngeal irritability, persistent cough and a sensation of something sticking in the throat as well as a report of ‘noisy breathing’. These symptoms were identified as markers for possible airway risk factors with 40/105 (38%) being urgently referred via the DMP for assessment by the multidisciplinary laryngology clinic.
In total, 18/40 (45%) patients were diagnosed with significant postintubation laryngeal complications and listed for surgery. These patients had a combination of pathologies (14 granulomas, 3 subglottic abscesses, 15 posterior glottic scars and scar bridge between the vocal cords) and all had restriction of movement of the vocal cords with reduced abduction, a narrowed glottis, resulting in stridor and dysphonia. 3/18 had existing tracheostomies following discharge from the intensive care unit. A further 7/40 (17.5%) patients presented with minor posterior glottic scar or a small granuloma requiring only SLT input, and 42/105 (40%) of the total cohort were discharged as their symptoms had either resolved spontaneously or they had achieved optimal function following SLT intervention. Of note, 12/105 patients did not return for review.
To date, we have operated on 14/18 patients who had significant laryngeal injury (see Table 1 for a summary of outcome measures). 4/14 operated patients required a second airway procedure for persisting symptoms. Postsurgery, the impact on the voice was variable; however, looking at the GRBAS, VHI-10, and AVS scores there was not a significant difference between the pre and postoperative scores (using paired t-tests). Of note, the RSI scores postoperatively showed a significant improvement P = 0.0266. The average patient age was 57 years (range 31–72 years of age), and 11/18 patients were female. Patient co-morbidities included hypertension (13/18), diabetes (10/18) and obesity (8/18).
Table 1.
Summary table of 18 patients who required surgery for postintubation complications post-COVID-19 pneumonitis
| Patient | Age | Sex | Laryngeal pathology | GRBAS preop | GRBAS post op | VHI-10 preop | VHI-10 postop | RSI preopa | RSI postopa | AVS Preop | AVS Post op | Co-morbidities |
| 1 | 71 | F | Unilateral vc immobility, posterior glottic scar, granuloma | 20212 | 11101 | 11 | 29 | 40 | 30 | 432 | 321 | T2DM, HTN, obesity |
| 2 | 31 | M | Bilateral vc immobility, posterior glottic scar, granuloma | 22221 | 30331 | 40 | 21 | 38 | 20 | 533 | 335 | OSA |
| 3 | 64 | F | Bilateral vc immobility, posterior glottic scar, granuloma | 0 | 20211 | 6 | 6 | 2 | 0 | 111 | 221 | T2DM, HTN, obesity |
| 4 | 60 | M | Bilateral vc immobility, posterior glottic scar | 22221 | 20212 | 38 | 36 | 44 | 38 | 433 | 222 | T2DM, HTN, oesophagectomy |
| 5 | 65 | M | Bilateral vc immobility, posterior glottic scar | 32223 | 33333 | 37 | 35 | 17 | 17 | 441 | 431 | T2DM, HTN, hypercholesterolaemia |
| 6 | 72 | F | Unilateral vc immobility, posterior glottic scar, granuloma, abscess | No preop PROMs | 32323 | Not done | Not done | Not done | Not done | Not done | 445 | T2DM, HTN, AF, OSA, asthma, obesity, hyperthyroidism |
| 7 | 56 | F | Granuloma | 11001 | 21102 | 29 | 24 | 23 | 26 | 222 | Not done | HTN, obesity |
| 8 | 62 | F | Bilateral vc immobility, posterior glottic scar, granuloma | No pre/postoperative PROMS as the patient was non-English speaking | T2DM, HTN, CVAs | |||||||
| 9 | 60 | F | Unilateral vc immobility, cricoarytenoid ankylosis granuloma, abscess | 10110 | 10110 | 33 | 35 | 31 | 27 | Not done | 532 | HTN, hypercholesterolaemia |
| 10 | 67 | M | Granuloma of glottis, tracheal granulation tissue related to tracheostomy, cricoarytenoid joint fixation and abscess | 0 | 10001 | 19 | 9 | 28 | 15 | 322 | 112 | T2DM, cardiac stents, HTN, RA |
| 11 | 56 | M | Bilateral vc immobility, posterior glottic scar | 10111 | 10100 | 37 | 34 | 35 | 33 | 321 | Not done | asthma, obesity, DOAC for AF, previous alcohol excess, back pain |
| 12 | 56 | F | Posterior glottic scar, granuloma and tracheal granulation tissue related to trachesotomy | 22222 | 21222 | 24 | 27 | 36 | 34 | 231 | Not done | RSV and common coronavirus, HTN, hypothyroidism (previous tracheostomy with covid) |
| Awaiting follow-up postsurgery | ||||||||||||
| 13 | 46 | M | Bilateral vc immobility, posterior glottic scar, granuloma | 10111 | 21 | 23 | 421 | Hepatitis B, HTN, obesity | ||||
| 14 | 58 | F | Bilateral vc immobility, posterior glottic scar, granuloma and tracheal granulation tissue related to trachesotomy | 11111 | 5 | 14 | T2DM, HTN, hypothyroid, hypercholesterolaemia, OA, angina | |||||
| Awaiting surgery | ||||||||||||
| 15 | 51 | F | Bilateral vc immobility, posterior glottic scar | 0 | 31 | 38 | 322 | T2DM, HTN, hypercholesterolaemia, obesity, hay fever | ||||
| 16 | 61 | M | Bilateral vc immobility, posterior glottic scar, granuloma | 10100 | 23 | 24 | 222 | Klippel-Trenaunay syndrome | ||||
| 17 | 44 | F | Bilateral vc immobility, posterior glottic scar, granuloma | 32303 | 25 | 15 | 331 | T2DM, obesity, hiatus hernia, depression, fibromyalgia | ||||
| 18 | 44 | F | Posterior glottic scar, granuloma | 21212 | 31 | 29 | 331 | Borderline DM, obesity, PDA closed age 4 | ||||
Pre and postoperative scores for the voice were assessed subjectively by clinicians and speech pathologists using the GRBAS scale (Grade, Breathiness, Asthenia and Strain) and by patients using the Voice Handicap Index-10 (VHI-10). Patient assessment of other laryngological symptoms was reported using the Reflux Symptom Index (RSI), and Airway-Voice-Swallow (AVS) scale. Case 8 was non-English speaking and therefore no PROMS were collected. Cases 13 and 14 have had surgery but are awaiting follow-up and Cases 15–18 await surgery. AF, atrial fibrillation; CVAs, cerebral vascular accidents; DOAC, direct oral anticoagulant; HTN, hypertension; OSA, obstructive sleep apnoea; PDA, patent ductus arteriosus; T2DM, type 2 diabetes mellitus; vc, vocal cord.
RSI scores were the only parameter to show significance (using a paired t-test P = 0.0266) pre and postoperatively.
Surgical management approach
In our cohort, the surgical technique used in the treatment of stridor secondary to postintubation injuries or posttracheostomy depended on the location of the complication in the larynx. Postintubation glottic injuries, including posterior glottic scars, are notoriously difficult to treat and the long-term impact of these scars and success of surgical and SLT intervention is yet to be evaluated.
Concerning the postintubation injuries, an initial treatment using a Blue Laser Wolf Trublue (Neomed UK) to vaporize the granulomas on vocal cord or trachea were carried out as ‘in office’ procedures, where possible. Ultimately, the severity of the vocal cord immobility and size of the granulomas would dictate whether a general anaesthetic was necessary. In the operating room, our unit uses either a Lumenis CO2 laser fibre (where access to trachea is required) or an acublade scanner mode CO2 laser to divide posterior glottic scars. In addition, laryngeal plasma radiofrequency ablation (coblation), and/or Straightshot M4 Microdebrider (Medtronic) help excise bulky granulomas or where access with laser is difficult. Palpation for a subglottic swelling and incision to confirm or exclude an abscess is crucial (Fig. 2). In three patients, we uncovered subglottic abscesses intraoperatively, which were incised and drained and a pus swab taken and sent for microbiology, culture and sensitivity. Organisms cultured included Pseudomonas aeruginosa, Klebsiella pneumoniae, Escherichia coli, Streptococcus milleri and Candida albicans. In all three cases, the abscesses had not been apparent on the preoperative contrast CT scans of the neck. These were missed by the CT scan because the abscesses were small and in a small subglottic compartment.
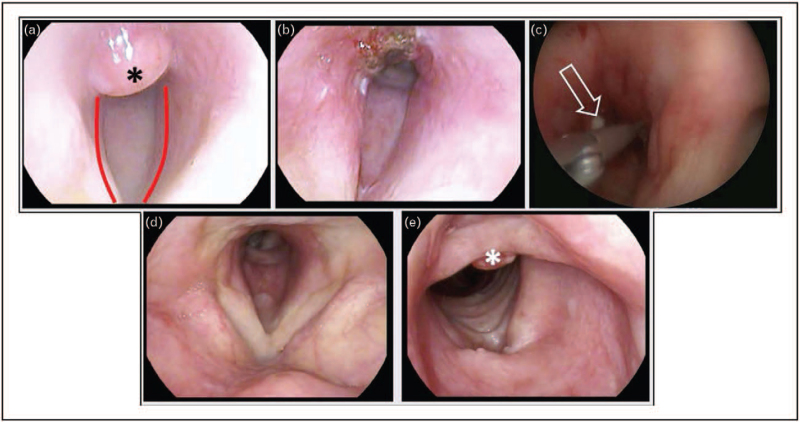
FIGURE 2.
Case 10. (a) Area of subglottic granulation tissue visualised on flexible nasal endoscopy (FNE) (black asterisk). Note (red lines) grade 3 subglottic stenosis. (b) Post operative image immediately after blue laser ablation. (c) Intraoperative image from emergency procedure to restore the diameter of the subglottis; note the presence of purulent discharge (hollow arrow). (d,e) Images taken during clinic review, approximately 4 weeks postoperatively, demonstrating relative normalisation of the anatomy, consistent with symptomatic resolution. Note the presence of residual granulation tissue (white asterisk). FNE, flexible nasendoscopy; post op, post-operative.
In some patients with fixation of the cricoarytenoid joints, which may be due to cricoarytenoid ankyloses or scar, it has been necessary to divide the scar around the cricoarytenoid joint. Depo-medrone, a long-acting methylprednisolone (40–80 mg) is injected into the scar tissue to reduce recurrence of scarring and inflammation around the cricoarytenoid joint. Balloon dilation of the stenosed sections of the airway, inflated for 60 s at 8 atmospheric pressure, is applied using a 15 mm Boston medical balloon. Medialised cords are lateralised at the vocal process using a temporary 3/0 prolene suture delivered passed with a Lichtenberger endo-extralaryngeal needle carrier. This suture is brought through the skin of the neck and tied over a silicone barrier to avoid cheese-wiring and to protect the integrity of the skin. The suture is routinely removed in the office 4 weeks after placement using transnasal laryngoscopy for visualisation of the larynx. If bilateral sutures are placed, we recommend removing these sutures in a staged manner 7 days apart. Postoperative prescriptions include: antibiotics for 2 weeks orally (if sutures are used), a 7-day course of steroid tablets and antireflux medication (twice daily proton pump inhibitor and an alginate three times a day). Inpatient stays ranged between 1 day and 4 weeks (length of stay largely depended on swallowing outcome postoperatively). None of the patients underwent a medial arytenoidectomy thus reducing the risk of further granulation, which may occur with exposed arytenoid cartilage. Nor did we carry out a rib graft procedure to expand the posterior glottic as this area is still actively inflamed with abscesses and granulation in some cases. In our opinion, the rib graft is therefore unlikely to survive and may only exacerbate formation of more inflammation and abscesses if the graft fails. In some cases, a partial posteriorly placed thyro-arytenoid myectomy was performed lateral to the tip of the vocal process to encourage a permanent lateralisation when the sutures are finally removed.
Speech and language therapy intervention for patients undergoing airway surgery
A patient information leaflet was developed to help consent patients and further explain the surgery and postoperative recovery process. A specialist dysphagia SLT performed pre and postoperative fibreoptic endoscopic evaluation of the swallow (FEES). A voice SLT provided pre and postoperative therapy advocating initially 2 weeks voice rest to avoid adduction of the vocal cords when sutures were in situ, and hourly brisk sniffing exercises to encourage vocal cord abduction and to improve the glottic airway space.
Literature review in relation to our experience
Dysphonia as a symptom of coronavirus disease-2019
SARS-CoV-2 infection causes inflammation of the upper and lower respiratory tract commonly causing a loss of sense of smell, high temperature, cough and, with some variants, headache and rhinorrhoea among many other symptoms [6]. Dysphonia as a major symptom of COVID-19 disease has been reported in 25–79% in four studies [7,8,9▪▪,10]. Two groups have reported dysphonia in up to 25% of patients with COVID-19 [7,8]. Cantarella et al. published one of the early reports on the effects of SARS-CoV-2 infection on the voice, airway and swallow reporting that 47% of their patients experienced dysphonia following COVID-19 disease [9▪▪]. The grade of dysphonia was positively related to cough and rhinitis symptoms. Furthermore, Azzam, et al. reported 79% incidence of dysphonia in SARS-CoV-2 positive patients [10].
Consequences of coronavirus disease-2019 causing dysphonia
In the remaining section we relate our experience and those of others in the literature by expanding upon the three categories of causes of dysphonia from SARS-CoV-2 infection initially described by Helding et al.[11].
Dysphonia postintubation from coronavirus disease-2019
Vocal cord immobility is only one presentation of many consequences of postintubation injury. Naunheim et al. describe presumed iatrogenic causes of dysphonia from prolonged intubation and intubation laryngeal injury; posterior glottic (15%) and subglottic (10%) stenoses and granulation tissue (10%) [1▪▪], which reflects our experience of the type of injuries with this cohort of postintubated patients for COVID-19 pneumonitis. Stroboscopic findings included reduction of vocal fold closure and abnormal wave, phase symmetry, periodicity and amplitude.
Dysphonia is a known risk factor in critical care patients who have been intubated and ventilated [12,13]. Archer et al. published their data on patients hospitalized with COVID-19 in relation to swallowing and voice outcomes based on the first wave of the pandemic (N = 164) [14▪]. Half (52.4%) had a tracheostomy, 78.7% had been intubated and 13.4% had new neurologic impairment. On discharge, 17.1% had dysphonia requiring ongoing intervention and monitoring [14▪].
A similar prospective, multisite study in Ireland by Regan et al. reported 20% needed intervention for dysphonia postextubation and 37% of those cases continued to be dysphonic on discharge [15]. Rouhani et al. reported an abnormal voice perception as evaluated by GRBAS scale postintubation for COVID-19 who underwent tracheostomy insertion and subsequent decannulation [16]. Surgery in our cohort largely did not cause complete resolution of dysphonia and, in some cases at this early postoperative stage, worsened vocal outcomes both subjectively and objectively, but not significantly as an overall cohort. It is recognised that surgery primarily performed to improve the airway can be at the expense of voice quality, which may or may not be temporary. RSI improved significantly postoperatively as all patients were given maximal antireflux medication (twice daily proton pump inhibitor and three times daily alginate) and dietary/lifestyle adjustments as advised by clinicians.
Patients that require intubation for COVID-19 disease are typically those with co-morbidities such as, type 2 diabetes mellitus, obesity, and hypertension [1▪▪,17,18]. Our patient cohort aligns with Naunheim et al.'s findings [1▪▪], as patients who are most at risk of needing intubation with COVID-19 disease are those with patient-specific risk factors and these are at an increased risk for subsequent laryngotracheal injury following intubation (Table 1).
Sensory neuropathy
Sensory neuropathy of the larynx from SARS-CoV-2 infection causes chronic cough and swallow dysfunction [11]. SARS-CoV-2 is also thought to lead to a physical effect on the true vocal folds from acute and chronic inflammation, and subsequent chronic vocal fatigue, which relate to associated voice complaints. The laryngeal damage from the sensory neuropathy may be exacerbated by the mucosal inflammation of the upper respiratory tract thought to be mediated from the virus itself, in addition to the inflammation caused by the physical trauma of coughing, related hypersensitivity of the larynx and laryngopharyngeal reflux. Hence, on this theoretical basis in our surgical cohort, all patients were given maximal antireflux medication in order to minimise further inflammatory risk. Chronic cough and hypersensitivity of the airways, including the larynx has also been reported [8]. In their nonintubated group with COVID-19 described by Naunheim et al., 42.9% reported dysphonia [1▪▪]. In the longer term, 26% complained of vocal fatigue 3 months after the onset of infection, which may result in dysphonia [11]. As yet, our service has not seen many patients with LONG-COVID including dysphonia as a presentation in nonintubated patients. It is important to note, however, that sensory neuropathy may have a contributory role in those patients with postintubation laryngeal injury.
Viral-induced vocal cord paralysis
Amin and Koufman introduced the term postviral vagal neuropathy to the group of sensory and motor effects involving dysphonia, vocal fatigue, odynophagia, neuropathic pain, cough, globus, laryngospasm, excessive throat clearing and laryngopharyngeal reflux [19]. These symptoms have been reported multiple times by patients presenting with lower cranial neuropathy post-COVID-19. We have seen a small number of nonintubated patients presenting with these symptoms in our clinic and expect to see many more cases as we manage our GP referrals over the next few months.
The reported studies support the notion that dysphonia is a significant symptom of SARS-CoV-2 infection. They also explain the many different laryngeal diagnoses that may account for the dysphonia post-COVID-19 disease in both intubated and nonintubated patients. Our experience, supported by the literature, highlights the importance of postextubation endoscopy and ongoing multidisciplinary review of patients following discharge from hospital after COVID-19 disease in the laryngology clinic. We would also encourage GP/family doctor referral of community treated patients reporting the above persistent laryngeal symptoms for laryngeal assessment post-COVID-19 disease.
CONCLUSION
SARS-CoV-2 is a systemic infection that impacts the voice significantly as demonstrated in the literature in both patients who were intubated and not-intubated due to the COVID-19 pneumonitis. The pathophysiology of dysphonia is assumed to be acute and chronic inflammation of the laryngeal mucosa, which may or may not be associated with a sensory neuropathy and/or vocal cord palsy. Viral-induced vocal cord palsy is recognised but has not been prevalent at our centre in patients being treated with COVID-19 disease. Those with vocal cord immobility in the majority of our cohort of patients required surgical intervention for other pathologies.
Laryngeal intubation injury is postulated to be primarily due to direct trauma from the endotracheal tube on the adjacent posterior glottis and cricoarytenoid joint with resultant granulation, scarring and cricoarytenoid ankylosis and in some cases subglottic abscesses, as we have found in our reported cohort. Postintubation glottic injuries may cause some dysphonia, but managing the airway compromise is the most important aspect to prioritise and may not alleviate or improve the dysphonia. We recognise that the long-term impact of these laryngeal injuries and success of our surgical and SLT intervention has not yet been fully evaluated.
The literature suggests that up to three-quarters of all patients with COVID-19 disease have dysphonia and so we believe this is a significant symptom to report. If dysphonia persists, there may be significant morbidity for patients and, particularly in those who are postintubation, this may be a sign of potential airway compromise. Identifying dysphonia early and managing this symptom in a multidisciplinary approach, involving a laryngologist and SLT is undoubtedly the gold standard for care of the post-COVID dysphonic patient.
Acknowledgements
None.
Financial support and sponsorship
None.
Conflicts of interest
There are no conflicts of interest.
REFERENCES AND RECOMMENDED READING
Papers of particular interest, published within the annual period of review, have been highlighted as:
▪ of special interest
▪▪ of outstanding interest
REFERENCES
- 1▪▪.Naunheim MR, Zhou AS, Puka E, et al. Laryngeal complications of COVID-19. Laryngoscope Investig Otolaryngol 2020; 5:1117–1124. Dec 1 [cited 2021 Jul 28]. [DOI] [PMC free article] [PubMed] [Google Scholar]; The authors described presumed iatrogenic causes of dysphonia from prolonged intubation and intubation laryngeal injury which reflects our experience of the type of injuries with this cohort of postintubated patients for COVID-19 pneumonitis. Stroboscopic findings included reduction of vocal fold closure and abnormal wave, phase symmetry, periodicity and amplitude.
- 2.Hirano M. Clinical examination of voice. 1981; Wien; New York: Springer-Verlag, 100 p. [Google Scholar]
- 3.Rosen CA, Lee AS, Osborne J, et al. Development and validation of the voice handicap index-10. Laryngoscope 2004; 114:1549–1556. [DOI] [PubMed] [Google Scholar]
- 4.Belafsky PC, Postma GN, Koufman JA. Validity and reliability of the reflux symptom index (RSI). J Voice 2002; 16:274–277. [DOI] [PubMed] [Google Scholar]
- 5.SaSandhu GS, Nouraei SAR. Laryngeal and Tracheobronchial Stenosis. Chapter 5. Assessment of Patients and outcomes in laryngotracheal stenosis. Plural publishing; 2015:53. [Google Scholar]
- 6.Cascella M, Rajnik M, Aleem A, et al. Features, evaluation, and treatment of coronavirus (COVID-19). StatPearls. 2021. [PubMed] [Google Scholar]
- 7.Lechien JR, Chiesa-Estomba CM, Cabaraux P, et al. Features of mild-to-moderate COVID-19 patients with dysphonia. J Voice 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Could non-classic symptoms indicate mild COVID? [Internet]. [cited 28 July 2021]. Available from: https://covid.joinzoe.com/post/symptoms. [Google Scholar]
- 9▪▪.Cantarella G, Aldè M, Consonni D, et al. Prevalence of dysphonia in non hospitalized patients with COVID-19 in Lombardy, the Italian Epicenter of the Pandemic. J Voice 2021; S0892-1997(21)00108-9. doi: 10.1016/j.jvoice.2021.03.009. [Online ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]; The authors highlighted the effects of Covid-19 on voice and relates it to inflammatory effects of Covid-19 on respiratory mucosa causing cough and rhinitis. It may be same pathophysiology that causes dysphonia.
- 10.Azzam AAA, Samy A, Sefein I, ElRouby I. Vocal Disorders in Patients with COVID 19 in Egypt. Indian J Otolaryngol Head Neck Surg 2021; 1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Helding L, Carroll TL, Nix J, et al. COVID-19 after effects: concerns for singers. J Voice 2020; S0892-1997(20)30281-2. doi: 10.1016/j.jvoice.2020.07.032.[Online ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brodsky MB, Levy MJ, Jedlanek E, et al. Laryngeal injury and upper airway symptoms after oral endotracheal intubation with mechanical ventilation during critical care: a systematic review. Crit Care Med 2018; 46:2010–2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hamdan AL, Sibai A, Rameh C, Kanazeh G. Short-term effects of endotracheal intubation on voice. J Voice 2007; 21:762–768. [DOI] [PubMed] [Google Scholar]
- 14▪.Archer SK, Lezzi CM, Gilpin L. Swallowing and voice outcomes in patients hospitalized with COVID-19: an observational cohort study. Arch Phys Med Rehabil 2021; 102:1084–1090. [DOI] [PMC free article] [PubMed] [Google Scholar]; The authors published their data on patients hospitalized with COVID-19 in relation to swallowing and voice outcomes based on the first wave of the pandemic. Half (52.4%) had a tracheostomy, 78.7% had been intubated and 13.4% had new neurologic impairment. On discharge, 17.1% had dysphonia requiring ongoing intervention and monitoring.
- 15.Regan J, Walshe M, Lavan S, et al. Post-extubation dysphagia and dysphonia amongst adults with COVID-19 in the Republic of Ireland: A prospective multi-site observational cohort study. Clin Otolaryngol 2021; 10.1111/coa.13832. doi: 10.1111/coa.13832. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rouhani MJ, Clunie G, Thong G, et al. A Prospective study of voice, swallow, and airway outcomes following tracheostomy for COVID-19. Laryngoscope 2021; 131:E1918–E1925. [DOI] [PubMed] [Google Scholar]
- 17.Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City Area. JAMA 2020; 323:2052–2059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hur K, Price CP, Gray EL, et al. “covid19” Factors Associated With Intubation and Prolonged Intubation in Hospitalized Patients With COVID-19. Otolaryngology–Head and Neck Surgery 2020; 163:170–178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Amin MR, Koufman JA. Vagal neuropathy after upper respiratory infection: a viral etiology? Am J Otolaryngol 2001; 22:251–256. [DOI] [PubMed] [Google Scholar]