Since the OXA-48-type carbapenem-hydrolyzing class D β-lactamase was reported in a Klebsiella pneumoniae isolate from Istanbul (Turkey) in 2001 [1], this carbapenemase has been widely distributed among Enterobacterales, with significant geographical differences [2, 3]. Albeit outbreak of nosocomial infections caused by OXA-48-producing K. pneumoniae has been frequently reported in the Mediterranean area and European countries [4–6]. The situation is less common in China, until the nosocomial outbreak of OXA-48-producing K. pneumoniae ST147 and ST383 was reported in a Chinese Hospital [7]. Subsequently, clonal dissemination of KPC-2- and OXA-48-coproducing K. pneumoniae sequence type 11 and the nosocomial outbreak of OXA-48-producing K. pneumoniae caused by clonal expansion of ST11-KL64 sublineages were found in Taiwan [8, 9].
In this study, we provided data on the genomic characterization of an imipenem intermediate K. pneumoniae strain isolated from the blood culture of a 56-year-old male patient who suffered from bacteremia and septic shock in May 2017. Antimicrobial susceptibility testing toward imipenem, meropenem, piperacillin, ticarcillin, cefoperazone, cefuroxime, cefazolin, cefoxitin, ampicillin, cefepime, ceftriaxone ceftazidime, amikacin, gentamicin, chloramphenicol, levofloxacin, ciprofloxacin, and trimethoprim/sulfamethoxazole was performed by broth microdilution and that of fosfomycin was determined by the agar dilution method using Mueller Hinton medium supplemented with 25 µg/mL glucose-6-phosphate, and the results were interpreted according to the CLSI 2020 (M100-30ED) guideline [10]. To characterize this strain in detail, whole genome sequencing was further performed using an Illumina MiniSeq plus Pacbio Sequencing. Based on the whole genome sequence, subsequent multi-locus sequence typing (MLST) was determined by uploading the genome to the webtool MLST v2.0 (https://cge.cbs.dtu.dk/services/MLST/), and antimicrobial resistance genes and plasmid replicons were identified by ResFinder 3.2 and PlasmidFinder 2.1 (https://cge.cbs.dtu.dk/services/). The plasmid carrying blaOXA-48 was detailly characterized by circular plasmid map and comparison of plasmid structures. Additionally, the phylogenomic tree of blaOXA-48 from global K. pneumoniae isolates was further constructed to observe the evolutionary relationship of blaOXA-48. Briefly, the nucleotide sequences of blaOXA-48 gene of 576 strains were compared by muscle, and then single nucleotide polymorphism (SNP) sites were used to extract SNPs from multiple alignment species. Finally, the maximum likelihood tree was constructed using RAxML software [11].
Antimicrobial susceptibility testing showed that this strain was intermediate to imipenem (2 μg/mL). However, resistance to meropenem (8 μg/mL) was observed. The isolate was resistant to most tested fluoroquinolones and ß-lactam antimicrobials except cefepime (< 2 μg/mL), ceftriaxone (1 μg/mL), and ceftazidime (4 μg/mL), and susceptible to amikacin (< 4 μg/mL) and gentamicin (< 1 μg/mL) (Table 1). Identification of antimicrobial resistance genes displayed the presence of blaOXA-48, fosA5, and blaSHV-145. To the best of our knowledge, this is the first time that we reported K. pneumoniae ST16 carrying OXA-48 in China, since an NDM-5-producing K. pneumoniae isolate also belonging to ST16 has been previously reported [12], thus, we made an in-depth characterization of this strain.
Table 1.
Antimicrobial susceptibility testing results for the OXA-48-producing Klebsiella pneumoniae
| Antimicrobials | MIC (μg/mL)a | Interpretationb |
|---|---|---|
| Fosfomycin | > 256 | R |
| Piperacillin | > 128 | R |
| Ticarcillin | > 128 | R |
| Cefoperazone | > 64 | R |
| Cefuroxime | > 32 | R |
| Cefoxitin | > 32 | R |
| Ampicillin | > 32 | R |
| Meropenem | > 16 | R |
| Chloramphenicol | 16 | R |
| Levofloxacin | 8 | R |
| Cefazolin | > 8 | R |
| Ciprofloxacin | 4 | R |
| Ceftazidime | 4 | S |
| Imipenem | 2 | I |
| Amikacin | < 4 | S |
| Ceftriaxone | 1 | S |
| Gentamicin | < 1 | S |
| Cefepime | < 2 | S |
| Trimethoprim/sulfamethoxazole | < 0.5 | S |
a An antimicrobial susceptibility testing was performed with standard broth microdilution method and interpreted based on the criteria from the Clinical and Laboratory Standards Institute guidelines (M100-30ED-2020)
b R, resistant; S, susceptible; I, intermediate
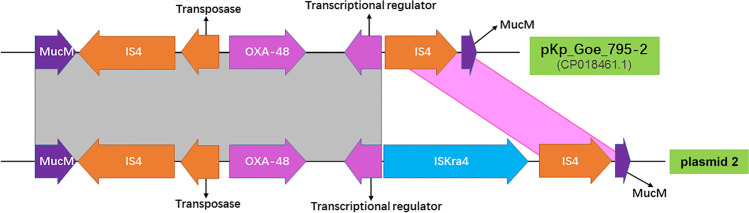
Whole-genome sequencing data by Illumina MiSeq plus Pacbio found that the K. pneumoniae isolate contained a 5.68-Mb genome, including a 5.31-Mb chromosome and seven different plasmids (Table 2). Resistant determinants including fosA5, oqxAB, and blaSHV-145 were found in chromosome, whereas, blaOXA-48 was found in a conjugative IncL/M plasmid (Fig. 1). The size of this plasmid is 66,076 bp, with GC content being 55.23% (Fig. 2). The circular structure of plasmid showed that this plasmid is likely to be a conjugative mobile plasmid because of the frequent binding mobile element tra-associated operons region within this plasmid [13] (Fig. 1). As known, IncL/M plasmid has been reported to be the vector for blaOXA-48 [14]. Analysis of flanking elements of blaOXA-48 gene displayed mobile elements including insertion sequence 4 (IS4), transposase, and ISkra4. We found that IS4 was distributed among both of the downstream and upstream of blaOXA-48 gene (Fig. 2). So far, IS4 family element has been reported to be involved in mobilization and expression of ß-lactam resistance genes including blaVEB-1 and blaOXA-48 [15]. Transposase has been found to mediate chromosomal integration of exogenous genes in Acidithiobacillus ferrooxidans [16]. Altogether, our study suggested that the blaOXA-48 gene might be rapidly spread by a broad host-range conjugative plasmid.
Table 2.
Genomic features of the OXA-48-producing Klebsiella pneumoniae
| Structure | Length (bp) | GC (%) | Antimicrobial resistance genes | Replicon type | Accession no |
|---|---|---|---|---|---|
| Chromosome | 5,314,991 | 57.49 | oqxAB, fosA5, blaSHV-145 | CP058581 | |
| Plasmid 1 | 110,716 | 48.84 | None | Unknown | CP058582 |
| Plasmid 2 | 66,076 | 51.23 | OXA-48 | IncL/M | CP058583 |
| Plasmid 3 | 5,589 | 51.22 | None | Unknown | CP058584 |
| Plasmid 4 | 5,251 | 49.24 | None | IncCoI440II | CP058585 |
| Plasmid 5 | 5,167 | 47.57 | None | Unknown | CP058586 |
| Plasmid 6 | 4,693 | 43.91 | None | IncCoI440I | CP058587 |
| Plasmid 7 | 193,269 | 52.50 | Mph(A) |
IncFIB(K) IncFII(K) |
CP058588 |
Fig. 1.
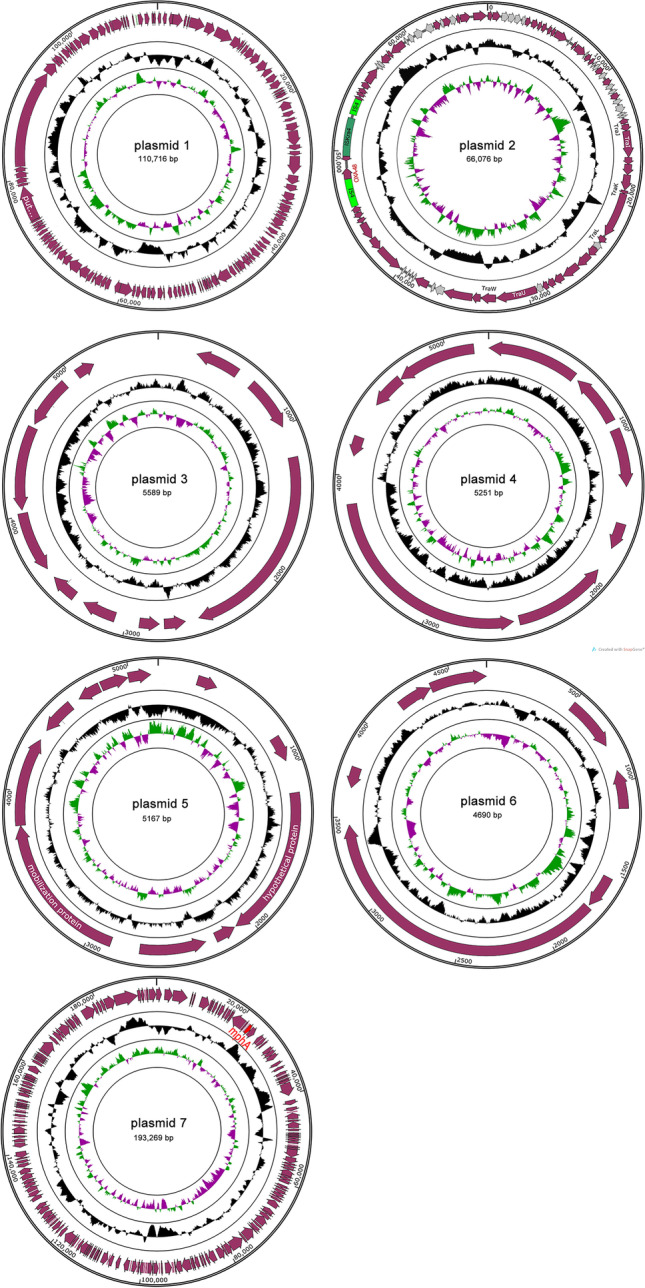
Circular structure of the seven plasmids in the OXA-48-producing Klebsiella pneumoniae. The innermost rings represent the G + C skew of the plasmids, the middle rings represent the G + C content, and the outermost rings represent predicted ORFs. Black, content of G + C; claret, open reading frames; light green, insertion sequence
Fig. 2.
The flanking element analysis of OXA-48 gene between plasmid 2 and its most similar homologue. Colored arrows indicate open reading frames. Orange, blue, and purple arrows represent insertion sequence IS4, ISKra4, and mucM genes, respectively. Gray shading indicates homologous regions among different elements
Noteworthily, this is the first time that we identified SHV-145 in a clinical K. pneumoniae isolate in China. SHV-145 is an extended-spectrum beta-lactamase that has been previously found in a K. pneumonia clinical isolate recovered from a hospitalized patient in Portugal (unpublished data: accession number AFN88952.1). To date, SHV-145 has been predicted by the protein homolog model (AMR detection model) as a beta-lactamase which could lead to the inactivation of penam, cephalosporin and carbapenem (https://card.mcmaster.ca/ontology/37565#resistomes-table). Furthermore, SHV-ESBLs are usually encoded by self-transmissible plasmids. However, in our study, blaSHV-145 was identified in Chromosome, which may be mobilized by mobile elements.
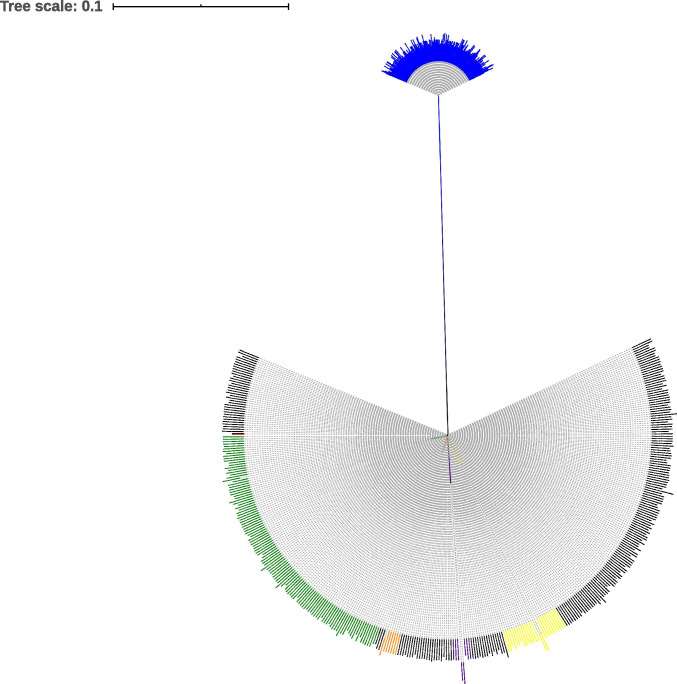
The phylogenetic tree based on the blaOXA-48 gene from global 576 K. pneumoniae isolates displayed 2 clades (Fig. 3). The simple evolutionary relationship suggests that this gene is relatively conservative. Concurrently, this may also indicate that blaOXA-48 has a strong transmission ability among K. pneumoniae isolates.
Fig. 3.
The blaOXA-48 phylogenetic tree based on the global 576 OXA-48-producing Klebsiella pneumoniae isolates. The nucleotide sequences of blaOXA-48 gene of 576 strains were compared by muscle, and then single nucleotide polymorphism (SNP) sites were used to extract SNPs from multiple alignment species. The maximum likelihood tree was constructed using RAxML. Red, the OXA-48-producing K. pneumoniae isolate in our study
To the best of our knowledge, this is the first report on the co-occurrence of fosA5, blaSHV-145 and blaOXA-48 among the K. pneumoniae ST16 in China and for the first time, we prescribed the blaOXA-48 evolutionary phylogenomic of global-producing K. pneumoniae.
Acknowledgements
This research was funded by National Natural Science Foundation of China grants (31770152 and 31400094).
Declarations
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yanmei Sun and Wei Chen equally contributed to this work
Contributor Information
Shiwei Wang, Email: wangsw@nwu.edu.cn.
Xiaoli Cao, Email: cao-xiao-li@163.com.
References
- 1.Poirel L, Heritier C, Tolun V, Nordmann P. Emergence of oxacillinase-mediated resistance to imipenem in Klebsiella pneumoniae. Antimicrob Agents Chemother. 2004;48(1):15–22. doi: 10.1128/AAC.48.1.15-22.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mairi A, Pantel A, Sotto A, Lavigne JP, Touati A. OXA-48-like carbapenemases producing Enterobacteriaceae in different niches. Eur J Clin Microbiol Infect Dis: Official Publication of the European Society of Clinical Microbiology. 2018;37(4):587–604. doi: 10.1007/s10096-017-3112-7. [DOI] [PubMed] [Google Scholar]
- 3.Han R, Shi Q, Wu S, Yin D, Hu F. Dissemination of carbapenemases (kpc, ndm, oxa-48, imp, and vim) among carbapenem-resistant enterobacteriaceae isolated from adult and children patients in China. Front Cell Infect Microbiol. 2020;10:314. doi: 10.3389/fcimb.2020.00314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Potron A, Kalpoe J, Poirel L, Nordmann P. European dissemination of a single OXA-48-producing Klebsiella pneumoniae clone. Clin Microbiol Infect: the official publication of the European Society of Clinical Microbiology and Infectious Diseases. 2011;17(12):E24–E26. doi: 10.1111/j.1469-0691.2011.03669.x. [DOI] [PubMed] [Google Scholar]
- 5.Pitart C, Sole M, Roca I, Fabrega A, Vila J, Marco F. First outbreak of a plasmid-mediated carbapenem-hydrolyzing OXA-48 beta-lactamase in Klebsiella pneumoniae in Spain. Antimicrob Agents Chemother. 2011;55(9):4398–4401. doi: 10.1128/AAC.00329-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cuzon G, Ouanich J, Gondret R, Naas T, Nordmann P. Outbreak of OXA-48-positive carbapenem-resistant Klebsiella pneumoniae isolates in France. Antimicrob Agents Chemother. 2011;55(5):2420–2423. doi: 10.1128/AAC.01452-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Guo L, An J, Ma Y, Ye L, Luo Y, Tao C, et al. Nosocomial outbreak of OXA-48-producing Klebsiella pneumoniae in a Chinese Hospital: clonal transmission of ST147 and ST383. PloS one. 2016;11(8):e0160754. doi: 10.1371/journal.pone.0160754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lu MC, Tang HL, Chiou CS, Wang YC, Chiang MK, Lai YC. Clonal dissemination of carbapenemase-producing Klebsiella pneumoniae: two distinct sub-lineages of Sequence Type 11 carrying blaKPC-2 and blaOXA-48. Int J Antimicrob Agents. 2018;52(5):658–662. doi: 10.1016/j.ijantimicag.2018.04.023. [DOI] [PubMed] [Google Scholar]
- 9.Lu MC, Chen YT, Tang HL, Liu YY, Chen BH, Wang YW, et al. Transmission and evolution of OXA-48-producing Klebsiella pneumoniae ST11 in a single hospital in Taiwan. J Antimicrob Chemother. 2020;75(2):318–326. doi: 10.1093/jac/dkz431. [DOI] [PubMed] [Google Scholar]
- 10.Wayne PA (2020) Performance standards for antimicrobial susceptibility testing, 30th edition, M100Ed30E. Clinical and Laboratory Standards Institute
- 11.Alexandros S. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics. 2014;9:1312–1313. doi: 10.1093/bioinformatics/btu033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Feng Y, Wei L, Zhu S, Qiao F, Zong Z. Handwashing sinks as the source of transmission of ST16 carbapenem-resistant Klebsiella pneumoniae, an international high-risk clone, in icu. J Hosp Infect. 2020;104(4):492–496. doi: 10.1016/j.jhin.2019.10.006. [DOI] [PubMed] [Google Scholar]
- 13.Burbank LP, Van Horn CR. Conjugative plasmid transfer in Xylella fastidiosa is dependent on tra and trb operon functions. J Bacteriol. 2017;199(21):e00388–17. doi: 10.1128/JB.00388-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Solgi H, Giske CG, Badmasti F, Aghamohammad S, Havaei SA, Sabeti S, et al (2017) Emergence of carbapenem resistant Escherichia coli isolates producing blaNDM and blaOXA-48-like carried on IncA/C and IncL/M plasmids at two Iranian university hospitals. Infect Genet Evol 55:318–23 [DOI] [PubMed]
- 15.Aubert D, Naas T, Heritier C, Poirel L, Nordmann P. Functional characterization of IS1999, an IS4 family element involved in mobilization and expression of beta-lactam resistance genes. J Bacteriol. 2006;188(18):6506–6514. doi: 10.1128/JB.00375-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Inaba Y, Banerjee I, Kernan T, Banta S. Transposase-mediated chromosomal integration of exogenous genes in Acidithiobacillus ferrooxidans. Appl Environ Microbiol. 2018;84(21):e01381–18. doi: 10.1128/AEM.01381-18. [DOI] [PMC free article] [PubMed] [Google Scholar]