Abstract
Background
Postoperative delirium is a serious complication of surgery associated with prolonged hospitalisation, long-term cognitive decline, and mortality. This study aimed to determine whether targeting bispectral index (BIS) readings of 50 (light anaesthesia) was associated with a lower incidence of POD than targeting BIS readings of 35 (deep anaesthesia).
Methods
This multicentre randomised clinical trial of 655 at-risk patients undergoing major surgery from eight centres in three countries assessed delirium for 5 days postoperatively using the 3 min confusion assessment method (3D-CAM) or CAM-ICU, and cognitive screening using the Mini-Mental State Examination at baseline and discharge and the Abbreviated Mental Test score (AMTS) at 30 days and 1 yr. Patients were assigned to light or deep anaesthesia. The primary outcome was the presence of postoperative delirium on any of the first 5 postoperative days. Secondary outcomes included mortality at 1 yr, cognitive decline at discharge, cognitive impairment at 30 days and 1 yr, unplanned ICU admission, length of stay, and time in electroencephalographic burst suppression.
Results
The incidence of postoperative delirium in the BIS 50 group was 19% and in the BIS 35 group was 28% (odds ratio 0.58 [95% confidence interval: 0.38–0.88]; P=0.010). At 1 yr, those in the BIS 50 group demonstrated significantly better cognitive function than those in the BIS 35 group (9% with AMTS ≤6 vs 20%; P<0.001).
Conclusions
Among patients undergoing major surgery, targeting light anaesthesia reduced the risk of postoperative delirium and cognitive impairment at 1 yr.
Clinical trial registration
Keywords: anaesthesia, bispectral index, cognitive dysfunction, delirium, electroencephalography, postoperative delirium
Editor's key points.
-
•
Consistent with findings in other large studies, the 6644-patient BALANCED Anaesthesia Study provided evidence that deliberately targeting light anaesthesia (bispectral index reading of 50) with a volatile-agent-based general anaesthetic is not associated with significantly superior outcomes, compared with deliberately targeting deep anaesthesia (bispectral index reading of 35).
-
•
The current study was a 515-patient sub-study of the BALANCED Anaesthesia Study, focusing on postoperative delirium, and was conducted mainly in patients in Hong Kong and China.
-
•
This study found that there were significantly fewer patients with syndromal delirium when a bispectral index reading of 50 was targeted, but no reduction was found in sub-syndromal delirium.
-
•
It is unlikely that deliberately targeting light general anaesthesia alone leads to clinically important changes in patient outcomes, apart from increasing the risk of intraoperative awareness, but further research should address whether there are vulnerable populations to deep anaesthesia based on factors, such as age, genetics, or comorbidities.
Perioperative neurocognitive disorders, including new onset postoperative delirium (POD), are common in older patients after anaesthesia and surgery, and are associated with poor short- and long-term outcomes, including worsening cognitive decline, surgical complications, increased risk of institutionalisation, and death after both cardiac and noncardiac surgery.1, 2, 3, 4, 5 POD is more frequent with increasing age, occurring in up to 65% of older patients, and its incidence is increased in patients with even subtle cognitive impairment.6 With increasing numbers of older people undergoing surgery and anaesthesia, POD has become a significant global health challenge that requires urgent attention. Preventive strategies involving multidisciplinary perioperative interventions may be of some benefit overall, but the influence on POD of modifying anaesthesia technique remains inconclusive.7
A number of studies have investigated whether bispectral index (BIS)-guided anaesthesia is associated with a decreased risk of POD, compared with ‘standard of care’ anaesthesia or use of targeted end-tidal volatile agent concentrations, with the inference being that the use of BIS guidance would result in less anaesthetic exposure and therefore ‘lighter’ anaesthesia.8, 9, 10 Small studies have used the frontal EEG-based BIS monitor as a measure of anaesthetic depth, and have shown associations between POD and very low BIS values or burst suppression.9 Recently, the Electroencephalography Guidance of Anesthesia to Alleviate Geriatric Syndromes (ENGAGES) trial investigated minimising anaesthetic administration and minimising EEG suppression during surgical anaesthesia to reduce POD.11 They found no difference between BIS-guided anaesthesia and routine care on POD outcomes (26.0% vs 23.0%; P=0.22). This study had several limitations likely associated with the comparator being ‘routine care’, most importantly, the separation of anaesthetic dose was small (0.69 vs 0.80 minimal alveolar concentration), the median time difference between groups with EEG burst suppression was small (7 vs 13 min), and the mean BIS level in the two groups was not reported.
We designed and conducted a sub-study of the ‘Anaesthetic depth and complications after major surgery (BALANCED) trial’ to determine whether light general anaesthesia (BIS target 50) was associated with a lower incidence of POD than deep general anaesthesia (BIS target 35).12
Methods
The main BALANCED study
The BALANCED Anaesthesia Study investigated 1 yr all-cause mortality in adult patients aged 60 yr or more scheduled to undergo major surgery lasting 2 h or more who were randomised to receive either light (BIS target 50) or deep (BIS target 35) general anaesthesia.12 Blood pressure targeting was implemented to prevent blood pressure from being a confounding factor.12,13 Inclusion criteria were patients aged 60 yr or more, ASA physical status of 3 or 4, expected duration of surgery 2 h or more, and an anticipated hospital stay of at least 2 days. Exclusion criteria included site of surgery interfered with placing BIS electrodes, planned wake-up test, use of nitrous oxide, propofol infusion, ketamine infusion greater than 25 mg h−1, or not expected to be contactable after 1 yr. An a priori MAP target suitable to the patient was chosen by the attending anaesthetist, and patients were then randomised to maintenance of anaesthesia being targeted to BIS 50 or BIS 35. For the delirium sub-study, all BIS and anaesthetic data were captured electronically. Assessors undertaking POD and cognition assessments were blind to group allocation.
The BALANCED delirium sub-study
The POD sub-study protocol was approved by the trial steering committee in 2014. All sites were invited to participate. Institutional ethics approval for the delirium sub-study was obtained at each participating site. Patients were required to provide specific written informed consent for the delirium sub-study (either as part of a modified main study consent form, an addendum to the main study consent form, or an alternative consent form that included the sub-study). An additional exclusion criterion for the POD sub-study was the identification of preoperative delirium at baseline. Baseline assessments for the delirium sub-study were completed before randomisation.
Delirium was assessed at baseline and twice daily (between 05:00–11:00 h and 18:00–22:00 h) using the confusion assessment method (CAM) 3 min diagnostic interview (3D-CAM) or if patients were in the ICU, the CAM-ICU.14,15 Sub-syndromal delirium was classified where patients experienced at least two features of delirium without meeting the criteria for delirium.16 All research staff were fully trained via the online training as per the guidelines for the 3D-CAM and CAM-ICU. Staff training was overseen by the corresponding author, and all staff undertaking assessments were required to have approval before commencing assessments. Delirium training and assessments were overseen by a collaborating neuropsychologist (Dr Paul Maruff of The Royal Melbourne Hospital, Melbourne, VIC, Australia). Once weekly, the research staff would discuss any cases that required adjudication and refer to a third assessor or the corresponding author. These adjudicated outcomes were then compiled and checked for final classification by the corresponding author. Delirium was assessed for 5 days postoperatively or until discharge, including weekends. The CAM-ICU was used for patients admitted to the ICU postoperatively and who were unable to talk on these days. POD assessments were performed when patients could be roused sufficiently in order to be assessed (Richmond Agitation and Sedation Scale score above –4). Patients with a complete baseline assessment and at least one postoperative assessment were included in the analysis.
Cognitive screening
Cognitive function was assessed at baseline, discharge, 30 days, and 1 yr postoperatively. The Mini-Mental State Examination (MMSE) was used at baseline to identify undiagnosed significant cognitive impairment, and at discharge to explore any association with POD. The abbreviated mental test score (AMTS) was administered by telephone at 30 days and 1 yr postoperatively to identify long-term cognitive consequences of anaesthesia and POD. The AMTS is a 10-item test assessing recall, concentration, orientation, and registration.17 Scores 6 or below have been shown to correlate well with dementia.18 Both the MMSE and the AMTS are cognitive screening tools designed to identify at-risk patients.19
Quantitative EEG analysis
The BIS is a quantitative EEG parameter between 0 (isoelectric EEG) and 100 (fully conscious) commonly used during general anaesthesia to titrate drug effect. Duration of anaesthesia was defined as the time from when the BIS value decreased below 60 after induction of anaesthesia until the inspired anaesthetic agent was stopped after completion of surgery. The signal quality ratio (SQR) is a proprietary algorithm assessing the signal quality of the EEG data from which BIS is derived. An SQR <50 was considered poor and was discarded before further analysis. The suppression ratio (SR) was defined as the percentage of time that the EEG was isoelectric during each BIS time series epoch (with SQR ≥50). If the SR was equal to zero, then no EEG burst suppression was recorded. If the SR was greater than zero, burst suppression was defined as being present, and the duration of burst suppression during that epoch was calculated. Durations of suppression for each epoch were summed to derive the total duration of EEG suppression for each patient.
Sample size calculation
Chan and colleagues8 reported that POD incidence was reduced from 24.1% to 15.6% with the use of BIS-guided anaesthesia vs routine care in a healthier older patient group (83% ASA physical status 1 or 2) than the BALANCED trial cohort. Based on this study, we hypothesised that the incidence of POD would be reduced from a conservative incidence of 25% in the BIS 35 group to 15% in the BIS 50 group. A total of 270 patients would be required in each group to achieve a power of 80% at two-tailed alpha value of 0.05. We therefore planned to recruit 600 patients to allow for all-cause loss to follow-up.
Intention-to-treat population
All randomised patients in the sub-study were included in the intention-to-treat (ITT) population. The primary and secondary endpoints were analysed using the full ITT population, which included all randomised patients who did not have delirium on preoperative screening and underwent induction of general anaesthesia for surgery.
Statistics
The primary outcome compared the presence of POD on any of the 10 assessments in the ITT population between randomised groups using a Mantel–Haenszel χ2 test stratified by study-centre groups (Australia, China, and the USA). The pooled estimate of the odds ratio (OR) and 95% confidence interval (CI) was used to describe the statistical comparison. The dichotomous secondary outcomes, thresholds for MMSE and AMTS scores, were compared using the same models as for the primary outcome. The scores and score changes for MMSE and AMTS were compared using a general linear model, which included study-centre groups as a factor. The number of episodes of delirium was compared between randomised groups using a Poisson regression model, which also included study-centre groups as a factor. Intraoperative characteristics were compared between randomised groups using Mann–Whitney U-tests for continuous data, including drug doses and χ2 or Fisher's exact tests as appropriate for dichotomous data. All analyses were undertaken using SPSS version 26 (IBM, Armonk, NY, USA), and a two-tailed P-value <0.05 was taken to indicate statistical significance with no adjustment for multiple comparisons.
A per-protocol analysis of the primary outcome and secondary outcomes was performed after removing all patients with a mean BIS more than 5 BIS units from target, patients who were lost to follow-up, and patients who had major protocol deviations.
Results
This POD sub-study enrolled 547 of the 655 eligible participants from the BALANCED study over eight sites in Australia, China (including Hong Kong Special Administrative Region), and the USA. After excluding four patients because of the presence of delirium on their preoperative assessment and 32 patients without baseline delirium assessment, 515 patients with a baseline delirium assessment and at least one postoperative assessment were included in the final analysis (Fig. 1). Most delirium assessments were conducted with the 3D-CAM. Only two patients were assessed with the CAM-ICU (one assessment each while in the ICU). The patient and surgical characteristics are presented in Table 1.
Fig 1.
Enrolment, randomisation, and outcome assessment for the intention-to-treat and per-protocol groups (Consolidated Standards of Reporting Trials diagram). BIS, bispectral index.
Table 1.
Patient and surgical characteristics by BIS group. Data are mean (standard deviation), n (%), or median (inter-quartile range). BIS, bispectral index; MMSE, Mini-Mental State Examination; SAR, Special Administrative Region; WHODAS, WHO Disability Assessment Schedule.
| BIS 50 (n=253) | BIS 35 (n=262) | |
|---|---|---|
| Age (yr) | 70.8 (6.9) | 71.1 (6.8) |
| Sex | ||
| Female | 91 (36) | 92 (35) |
| Male | 162 (64) | 170 (65) |
| Body weight (kg) | 70 (58–78) | 70 (60–78) |
| BMI (kg m−2) | 26 (23–28) | 26 (22–28) |
| ASA physical status | ||
| 3 | 249 (98) | 256 (98) |
| 4 | 4 (2) | 6 (2) |
| Operation for cancer | 178 (70) | 170 (65) |
| Emergency operation | 3 (1.2) | 2 (1.0) |
| Preoperative WHODAS 2.0 score | 18 (12–21) | 18 (12–21) |
| Preoperative Charlson Comorbidity Index | 7 (5–8) | 7 (5–8) |
| MMSE | 28 (2) | 28 (2) |
| Preoperative haemoglobin (g L−1) | 128 (115–141) | 126 (113–140) |
| Preoperative creatinine (mmol L−1) | 81 (69–99) | 80 (68–101) |
| Preoperative albumin (g L−1) | 39 (35–42) | 39 (36–42) |
| Type of surgery | ||
| Cardiac | 0 (0) | 1 (0.4) |
| Head and neck | 5 (2) | 8 (3) |
| Intra-abdominal | 178 (73) | 184 (70) |
| Orthopaedic | 6 (2) | 12 (5) |
| Spinal | 20 (8) | 15 (6) |
| Thoracic | 16 (6) | 12 (5) |
| Vascular | 13 (5) | 16 (6) |
| Other | 9 (4) | 14 (5) |
| Coexisting medical conditions | ||
| Cardiovascular | 50 (20) | 76 (29) |
| Stroke/neurological | 49 (19) | 39 (15) |
| Respiratory | 36 (14) | 47 (18) |
| Rheumatic/connective tissue | 11 (4) | 5 (2) |
| Gastric/peptic ulcer | 6 (2) | 7 (3) |
| Diabetes mellitus | 77 (30) | 78 (30) |
| Hepatic | 117 (46) | 125 (48) |
| Malignancy | 140 (55) | 138 (53) |
| Ethnicity | ||
| Asian | 199 (79) | 201 (78) |
| Black or African American | 4 (2) | 2 (1) |
| Hispanic/Latino | 2 (1) | 2 (1) |
| White/Caucasian | 41 (16) | 50 (19) |
| Not specified | 7 (3) | 7 (3) |
| Recruitment by country | ||
| Australia | 21 (8) | 22 (8) |
| China, including Hong Kong SAR | 199 (74) | 203 (73) |
| USA | 49 (18) | 53 (19) |
The median (inter-quartile range [IQR]) BIS for each group in the POD study was 51 (48–53) for the BIS 50 group and 38 (36–40) for the BIS 35 group, and the median volatile anaesthetic concentrations were 0.59 (0.51–0.69) and 0.79 (0.66–0.95) minimum alveolar concentration (MAC), respectively (Table 2). Time with SR >0 was 2.0 (0–7.0) min and 5.3 (0–27.7) min, respectively. Mean arterial pressure was 4 mm Hg higher and inotrope or vasopressor use was 20% lower in the BIS 50 group compared with the BIS 35 group.
Table 2.
Intraoperative characteristics. Data are median (inter-quartile range) or n (%). BIS, bispectral index; MAC, minimum alveolar concentration; SR, suppression ratio. ∗One patient with missing intraoperative data.
| BIS 50 (n=253) | BIS 35 (n=261)∗ | P-value | |
|---|---|---|---|
| Duration of surgery (min) | 221 (164–293) | 216 (162–298) | 0.973 |
| Major regional anaesthesia | 23 (9) | 28 (11) | 0.535 |
| BIS | 51 (48–53) | 38 (36–40) | <0.001 |
| MAP (mm Hg) | 85 (77–93) | 81 (74–87) | <0.001 |
| MAC fraction of volatile anaesthetic | 0.59 (0.51–0.69) | 0.79 (0.66–0.95) | <0.001 |
| Midazolam | 92 (36) | 134 (51) | <0.001 |
| mg | 2.0 (2.0–2.0) | 2.0 (2.0–2.0) | 0.535 |
| Propofol | 214 (85) | 223 (85) | 0.786 |
| mg | 100 (80–140) | 100 (80–140) | 0.728 |
| Ketamine | 12 (4.7) | 9 (3.4) | 0.458 |
| mg | 25 (12–25) | 20 (10–25) | 0.754 |
| Fentanyl | 196 (78) | 198 (76) | 0.667 |
| μg | 100 (100–300) | 113 (100–350) | 0.331 |
| Remifentanil | 88 (35) | 97 (37) | 0.574 |
| mcg | 470 (296–804) | 365 (234–757) | 0.503 |
| Morphine | 102 (40) | 96 (37) | 0.410 |
| mg | 4.5 (4.0–6.0) | 4.5 (4.0–6.5) | 0.695 |
| Alpha-2 agonist | 26 (10) | 26 (10) | 0.906 |
| Paracetamol | 26 (10) | 28 (11) | 0.868 |
| Nonsteroidal anti-inflammatory | 21 (8) | 21 (8) | 0.916 |
| Tramadol | 42 (17) | 36 (14) | 0.375 |
| Inotrope or vasopressor | 133 (53) | 189 (72) | <0.001 |
| PACU duration of stay (min) | 80 (59–120) | 84 (64–136) | 0.138 |
| Time with burst suppression (SR >0) (min) | 2.0 (0–7.0) | 5.3 (0–27.7) | 0.001 |
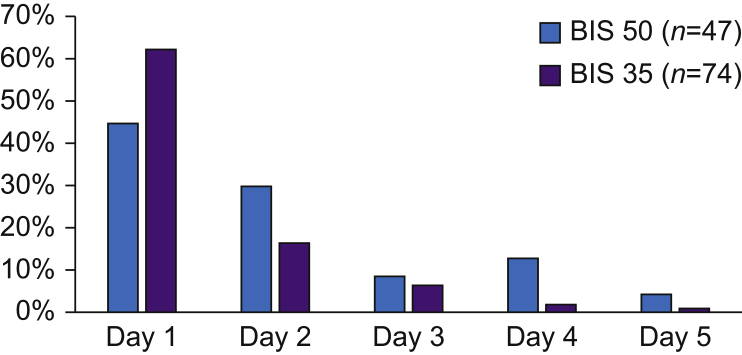
Two patients died postoperatively and before completion of any POD assessments, both in the BIS 35 group. They were excluded from subsequent analyses. Adjudication of POD proceeded as described in the methods, with no final adjudications nor reclassifications by the corresponding author. The incidence of POD was 19% in the BIS 50 group and 28% in the BIS 35 group, OR 0.58 (95% CI: 0.38–0.88), P=0.010 (Table 3) with a number needed to treat (NNT) of 10 (95% CI: 6–43). This corresponded to an absolute risk reduction of 9.7% (95% CI: 2.3–16.8). POD mostly occurred on the first 2 days after surgery (Fig. 2). There was no difference in the MMSE on discharge, but the mean AMTS at 1 yr was statistically significantly lower in the BIS 35 group compared with the BIS 50 group (7.8 [0.2] vs 8.2 [0.2], respectively; P=0.012). The clinical relevance of this difference would require further testing of patients to interpret. At 1 yr, significantly more patients in the BIS 35 group had an AMTS score of ≤6 compared with the BIS 50 group, 20% vs 9%, respectively; P=0.001. The per-protocol analysis also supported the finding of less POD and higher AMTS scores (better cognitive function) at 1 yr in the BIS 50 compared with the BIS 35 group (Table 4).
Table 3.
Primary and secondary outcomes. Data are n (%) or mean (standard error of the mean). AMTS, abbreviated mental test score; CI, confidence interval; MMSE, Mini-Mental State Examination.
| BIS 50 (n=253) | BIS 35 (n=262) | Odds ratio (95% CI) or mean difference | P-value | |
|---|---|---|---|---|
| Primary outcome | ||||
| Incidence of postoperative delirium | 47 (19) | 74 (28) | 0.58 (0.38–0.88) | 0.010 |
| Number of assessments for delirium | 2135 | 2157 | ||
| Secondary outcomes | ||||
| Number of episodes of delirium/patient | 0.45 | 0.77 | –0.32 (–0.45 to –0.15) | <0.001 |
| MMSE at discharge (n=417) | n=207 | n=210 | ||
| Score | 28.0 (2.8) | 27.7 (2.8) | 0.31 (–0.22 to 0.84) | 0.25 |
| Change from preoperative score | 0.36 (0.36) | 0.12 (0.34) | 0.24 (–0.11 to 0.58) | 0.18 |
| Score <26 | 11 (7.0) | 18 (11.3) | 0.58 (0.27–1.28) | 0.18 |
| AMTS at 30 days (n=476) | n=230 | n=246 | ||
| Score | 8.1 (0.1) | 8.1 (0.1) | 0.06 (–0.16 to 0.28) | 0.58 |
| Score ≤6 | 20 (8.7) | 22 (8.9) | 0.96 (0.51–1.81) | 0.90 |
| AMTS at 1 yr (n=413) | n=208 | n=205 | ||
| Score | 8.2 (0.2) | 7.8 (0.2) | 0.34 (0.07–0.60) | 0.012 |
| Score ≤6 | 18 (8.7) | 41 (20.0) | 0.37 (0.20–0.67) | <0.001 |
Fig 2.
Percentage of new delirium diagnoses for each group on each day. BIS, bispectral index.
Table 4.
Per-protocol analysis, primary and secondary. Data are n (%) or mean (standard error of the mean). AMTS, Abbreviated Mental Test score; BIS, bispectral index; CI, confidence interval; MMSE, Mini-Mental State Examination.
| BIS 50 (n=196) | BIS 35 (n=199) | Odds ratio (95% CI) or mean difference | P-value | |
|---|---|---|---|---|
| Primary outcome | ||||
| Incidence of postoperative delirium | 34 (17) | 57 (29) | 0.53 (0.32–0.85) | 0.009 |
| Number of assessments for delirium | 1655 | 1665 | ||
| Secondary outcomes | ||||
| Number of episodes of delirium/patient | 0.44 (0.06) | 0.86 (0.10) | –0.42 (–0.59 to –0.25) | 0.002 |
| MMSE at discharge (n=290) | n=143 | n=147 | ||
| Score | 27.1 (3.0) | 26.7 (2.9) | 0.33 (–0.30 to 0.96) | 0.30 |
| Change from preoperative score | 0.59 (0.6) | 0.23 (0.6) | 0.35 (–0.05 to 0.76) | 0.085 |
| Score <26 | 10 (8.1) | 14 (11.4) | 0.69 (0.29–1.63) | 0.401 |
| AMTS at 30 days (n=370) | n=178 | n=192 | ||
| Score | 8.2 (0.1) | 8.2 (0.1) | 0.04 (–0.21 to 0.29) | 0.77 |
| Score ≤6 | 17 (9.6) | 19 (9.9) | 0.95 (0.48–1.90) | 0.90 |
| AMTS 1 yr (n=326) | n=164 | n=162 | ||
| Score | 8.1 (0.2) | 7.8 (0.2) | 0.27 (–0.04 to 0.58) | 0.09 |
| Score ≤6 | 17 (10.4) | 33 (20.4) | 0.45 (0.24–0.85) | 0.012 |
Postoperative delirium was significantly associated with increased unplanned ICU admission rates, OR 2.99 (95% CI: 1.3–6.6), and duration of hospital stay, which was increased from a median (IQR) of 6 (IQR: 4–10) to 8 (6–13) days. The incidence of myocardial infarction was significantly higher in those who experienced delirium (6% with delirium vs 1% without delirium; P=0.002). Duration of surgery was 11% longer in patients with POD (Table 4). Exploratory analysis to inform future hypotheses showed patients with delirium declined significantly more than those without delirium at discharge on the MMSE (change 1.0 vs 0.1, respectively; P<0.001). They were also significantly more likely to have an AMTS score ≤6 suggestive of clinically important impairment than those without delirium, at both 30 days (19% vs 6%, respectively; P<0.001) and 1 yr (35% vs 8%, respectively; P<0.001).
Burst suppression occurred in both groups with time in burst suppression being median (IQR) 2 (0–7) min in the BIS 50 group vs 6 (0–23) min in the BIS 35 group (Table 2). Time in burst suppression was significantly longer in those patients who went on to experience delirium (Table 5). Mortality at 1 yr was significantly greater in those patients who experienced delirium than those who did not (12% vs 6%, respectively; P=0.024).
Table 5.
Association between postoperative delirium, various outcomes, and use of various drugs during anaesthesia. Data are n (%) or median (inter-quartile range). CI, confidence interval; CVS, cardiovascular system; SR, burst suppression ratio. ∗Intraoperative data missing for one patient in the no delirium group.
| Delirium (N=122) | No delirium (N=393) | Odds ratio (95% CI) or P-value | |
|---|---|---|---|
| Intraoperative use of∗ | |||
| Midazolam | 51 (42) | 175 (45) | 0.9 (0.6–1.4) |
| Propofol | 106 (88) | 331 (84) | 1.3 (0.7–2.4) |
| Ketamine | 7 (6) | 14 (4) | 1.7 (0.7–4.2) |
| Opioids | 121 (100) | 391 (99) | — |
| Vasopressors or inotropes | 85 (70) | 237 (60) | 1.6 (1.0–2.4) |
| Duration of surgery (min) | 237 (180–320) | 211 (157–287) | P=0.005 |
| Duration of hospital stay (days) | 8 (6–13) | 6 (4–10) | P<0.001 |
| Time with SR >0 (min) | 7 (0–28) (n=47) | 2 (0–10) (n=152) | P=0.028 |
| Mortality | 15 (12) | 24 (6) | 2.15 (1.1–4.2) |
| Myocardial infarction | 7 (6) | 4 (1) | 6.0 (1.7–20.8) |
| Pulmonary embolism | 0 (0) | 1 (0.3) | — |
| Stroke | 1 (0.8) | 2 (0.5) | — |
| Sepsis | 11 (9) | 18 (5) | 2.09 (1.0–3.0) |
| Surgical site infection | 7 (6) | 19 (5) | 1.21 (0.5–3.0) |
| Unplanned ICU admission | 12 (10) | 14 (4) | 3.00 (1.3–6.6) |
| Composite of CVS and mortality | 20 (17) | 32 (8) | 2.2 (1.2–4.1) |
Although not part of the original analysis plan, our data also enabled us to calculate sub-syndromal POD. Patients who were classified as positive for delirium were not included in the sub-syndromal data. Sub-syndromal POD was detected in 117 (45%) of the 262 patients in the BIS 35 group and 125 (49%) of 253 patients in the BIS 50 group (OR 1.21 [95% CI: 0.86–1.71]; P=0.278) (Supplementary Table S1; Supplementary Fig. S1). Bispectral index, MAP, MAC, POD, and sub-syndromal POD data for each site for the BIS 35 and BIS 50 groups are also presented in Supplementary Table S1.
Discussion
In this randomised trial of patients undergoing major surgery, BIS-guided lighter anaesthesia decreased the risk of POD compared with BIS-guided deeper general anaesthesia. Targeting BIS 50 resulted in a 34% relative reduction in patients suffering an episode of POD, an absolute reduction of 9% when compared with targeting BIS 35. Patients with an episode of POD had more unplanned ICU admissions, stayed 2 days longer in hospital, had a higher incidence of myocardial infarction, were more likely to have impaired cognitive function on the MMSE at discharge and the AMTS score at 30 days and 1 yr. This result confirms past research on the importance of delirium as an indicator of the potential causes of poor outcomes in a healthcare environment where the number of older at-risk patients having major surgery is steadily increasing.4 Although we did not find an association between POD and other postoperative outcomes, the size of the study meant the number of adverse outcomes was relatively small.
Previous studies addressing depth of anaesthesia and POD have predominantly compared BIS guidance (either targeted or untargeted) vs no BIS (i.e. ‘standard of care’), and have yielded inconclusive results. In 2014, Whitlock and colleagues10 reported on POD in ICU patients after cardiothoracic surgery with BIS-guided anaesthesia compared with end-tidal anaesthetic-concentration-guided anaesthesia (18.8% vs 28.0% [OR 0.60; 95% CI: 0.35–1.02; P=0.058]). This study confined the assessment of POD to the ICU, but included a meta-analysis of other studies, identifying a reduction in POD overall compared with standard of care (OR 0.56; 95% CI: 0.42–0.73). A Cochrane review by Punjasawadwong and colleagues20 in 2018 in noncardiac surgery patients that included three studies found there was moderate quality evidence that processed EEG-guided anaesthesia (such as BIS) reduced the incidence of POD.8,9 The patients included in this meta-analysis underwent a variety of surgeries, from minor day procedures to major noncardiac surgery.
Our results contrast with the results of the ENGAGES trial; however, we achieved a wider separation of anaesthetic dose of 0.21 MAC units as compared with 0.11 MAC units in the ENGAGES trial.11 Duration of burst suppression was longer in the two arms of the ENGAGES trial (7 vs 13 min) compared with our BALANCED delirium study (2.0 vs 5.3 min), making burst suppression, as distinct from BIS, a less likely cause of the difference in our study. The decision to actively target two BIS values (i.e. 35 and 50) rather than use a standard-care group improved BIS separation, and therefore increased anaesthetic dose separation and consequent anaesthetic dose reduction in the BIS 50 group.
Some of the previous literature on this topic has focused on patients with hip fracture, in whom there is a very high incidence of POD.21 Sieber and colleagues22 studied depth of sedation using i.v. propofol during spinal anaesthesia, targeted to a sedation scale, in patients undergoing hip fracture repair. BIS was monitored in both groups (although not targeted), with no difference in POD being found with light sedation (34% POD; mean [standard deviation {sd}] BIS 82.3 [9.4]) vs deep sedation (39% POD; mean [sd] BIS 57.0 [14.8]). Of note, most of these patients were female, and many had high frailty indices and significant comorbidities, making them less generalisable to other patient cohorts. Indeed, these investigators found that patients with the least comorbidity (lowest Charlson Comorbidity Index scores) had a 2.3-fold increase in POD risk (95% CI: 1.1–3.2) with deep sedation compared with light sedation. This suggests that the influence of comorbidities in high-risk patients may mask any benefit from changing the depth of anaesthesia or sedation.
POD has been confirmed by many studies to be associated with poor outcomes, although there are limited long-term data in perioperative patients to date.4,23,24 Our study confirmed an association between POD occurrence and unplanned ICU admission, length of hospital stay, risk of myocardial infarction, and 1 yr mortality; however, it was not designed to determine whether POD triggered these outcomes, and it was not sufficiently powered to identify if the reduction in POD associated with higher BIS targets affected these outcomes.
The association between POD and mortality at 1 yr (12% in those with POD vs 6% in those without) supports previous work, which has demonstrated a higher rate of mortality in patients who experience POD.25
Limitations and strengths
Because of an oversight, the delirium sub-study was retrospectively registered. This is a limitation; however, given the sub-study was undertaken in an entirely prospective design, we believe any effect would be minimal. Because of time limitations, the cognitive assessments undertaken in this study were screening measures designed to identify at-risk patients who may require further investigation. Nonetheless, we have demonstrated that patients randomised to BIS 50 had a significantly lower incidence of poor cognition screening score (AMTS) at 1 yr compared with those randomised to BIS 35, despite there being no difference in cognitive screening score between the groups at baseline, discharge, and 30 days. The reasons for this are unclear. Blood pressure targeting in this study ensured that blood pressure was not a source of bias. It is possible that the higher volatile anaesthetic concentrations or increased use of vasopressors and inotropes may have contributed to this difference, but the study was not designed to explore these factors. There was no evidence in this study that midazolam was associated with a higher incidence of postoperative cognitive consequences. The identification of four patients who met the criteria for delirium at baseline reflects the age and comorbidity of this population, and emphasises the importance of preoperative screening before undertaking surgical procedures and research.26 Although not assessed as part of this study, such patients would be expected to also have a higher risk for POD.
The study design did not control for other factors that may influence POD outcomes, such as benzodiazepine exposure, anticholinergic drugs, or opioid use. POD is a complex clinical entity, and the current best practice for prevention and care involves the implementation of a range of strategies as part of a ‘bundle’ of perioperative care.25 This study maintained tightly controlled MAP, ensuring outcomes were not confounded by major differences in blood pressure or episodes of hypotension. We did not adjust for multiplicity of testing with respect to secondary outcomes. Finally, although most baseline variables appeared well matched, a chance difference in cardiovascular disease (higher incidence in the BIS 35 group) may have confounded the relationship between depth of anaesthesia and POD, although this risk may have been balanced by a higher incidence of stroke in the BIS 50 group. Exploration of these associations was not pre-planned for our study and requires a much larger RCT.
Conclusions
This sub-study of the BALANCED Anaesthesia Study demonstrated a protective effect of targeting a BIS of 50 to reduce POD compared with a BIS of 35. This is the first study to investigate POD with two specific BIS targets. Consequently, there was significant separation in BIS and anaesthetic dose between the groups and a significant reduction in POD when targeting a BIS of 50. We have also shown that patients randomised to light anaesthesia (BIS 50) have a lower incidence of poor cognitive screening scores at 1 yr compared with those randomised to deep anaesthesia (BIS 35). The NNT of 10 means lighter anaesthesia was an efficient therapy to reduce POD in this population. We also observed a reduction in the risk of mortality at 1 yr in patients who did not suffer delirium. Targeting light anaesthesia using EEG prevented 1 in 10 cases of delirium. Reducing the incidence of POD has the potential to improve outcomes for older individuals undergoing surgery worldwide, but this conclusion needs to be confirmed in a larger study.
Authors' contributions
Trial conception: LAE, DC, KL, TGS
Day-to-day running of the trial: LAE, MTVC, RH, MHMC, BPC, DAS, KOP, DIS, RV, MS, AA, PSM
Statistical analysis: CF
Writing of first draft of paper: LAE, DC, KL, TGS
Editing of draft: all authors
Revising of paper: LAE, DC, KL, TGS, CF
Approval of final version of paper: all authors
Acknowledgements
The overarching BALANCED study from which this sub-study was drawn was supported by grants from the Health Research Council of New Zealand (12-308-Short), the Australian National Health and Medical Research Council (APP1042727), the Research Grants Council of Hong Kong (number 61513), and the National Institutes of Health in the USA (P30 CA 008748).
Handling editor: Michael Avidan
Footnotes
This article is accompanied by the following editorials: The quagmire of postoperative delirium: does dose matter? by Gaskell & Sleigh, Br J Anaesth 2021:127:664–666, doi: 10.1016/j.bja.2021.08.008 Anaesthetic depth and delirium: a challenging balancing act by Whitlock et al., Br J Anaesth 2021:127:667–671, doi: 10.1016/j.bja.2021.08.003
Supplementary data to this article can be found online at https://doi.org/10.1016/j.bja.2021.07.021.
Declarations of interest
TGS is a consultant to Becton Dickinson (Melbourne, Victoria, Australia) and has received research funding from Boehringer Ingelheim. All other authors declare no competing interests.
Funding
Health Research Council of New Zealand; National Health and Medical Research Council, Australia; Research Grant Council of Hong Kong; National Institute for Health and Research, UK; National Institutes of Health, USA; Health and Medical Research Fund (13140851); Food and Health Bureau, Hong Kong Special Administrative Region, China.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Evered L., Silbert B., Knopman D.S., et al. Recommendations for the nomenclature of cognitive change associated with anaesthesia and surgery—2018. Br J Anaesth. 2018;121:1005–1012. doi: 10.1016/j.bja.2017.11.087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Silbert B.S., Scott D.A., Evered L.A., et al. A comparison of the effect of high- and low-dose fentanyl on the incidence of postoperative cognitive dysfunction after coronary artery bypass surgery in the elderly. Anesthesiology. 2006;104:1137–1145. doi: 10.1097/00000542-200606000-00007. [DOI] [PubMed] [Google Scholar]
- 3.Evered L.A., Silbert B.S., Scott D.A., Maruff P., Ames D. Prevalence of dementia 7.5 years after coronary artery bypass graft surgery. Anesthesiology. 2016;125:62–71. doi: 10.1097/ALN.0000000000001143. [DOI] [PubMed] [Google Scholar]
- 4.Inouye S.K., Marcantonio E.R., Kosar C.M., et al. The short-term and long-term relationship between delirium and cognitive trajectory in older surgical patients. Alzheimers Dement. 2016;12:766–775. doi: 10.1016/j.jalz.2016.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.O’Regan N.A., Fitzgerald J., Timmons S., O’Connell H., Meagher D. Delirium: a key challenge for perioperative care. Int J Surg. 2013;11:136–144. doi: 10.1016/j.ijsu.2012.12.015. [DOI] [PubMed] [Google Scholar]
- 6.Rudolph J.L., Marcantonio E.R. Review articles: postoperative delirium: acute change with long-term implications. Anesth Analg. 2011;112:1202–1211. doi: 10.1213/ANE.0b013e3182147f6d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chan M.T.V., Hedrick T.L., Egan T.D., et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on the role of neuromonitoring in perioperative outcomes: electroencephalography. Anesth Analg. 2020;130:1278–1291. doi: 10.1213/ANE.0000000000004502. [DOI] [PubMed] [Google Scholar]
- 8.Chan M.T., Cheng B.C., Lee T.M., Gin T. BIS-guided anesthesia decreases postoperative delirium and cognitive decline. J Neurosurg Anesthesiol. 2013;25:33–42. doi: 10.1097/ANA.0b013e3182712fba. [DOI] [PubMed] [Google Scholar]
- 9.Radtke F.M., Franck M., Lendner J., Kruger S., Wernecke K.D., Spies C.D. Monitoring depth of anaesthesia in a randomized trial decreases the rate of postoperative delirium but not postoperative cognitive dysfunction. Br J Anaesth. 2013;110:i98–105. doi: 10.1093/bja/aet055. [DOI] [PubMed] [Google Scholar]
- 10.Whitlock E.L., Torres B.A., Lin N., et al. Postoperative delirium in a substudy of cardiothoracic surgical patients in the BAG-RECALL clinical trial. Anesth Analg. 2014;118:809–817. doi: 10.1213/ANE.0000000000000028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wildes T.S., Mickle A.M., Ben Abdallah A., et al. Effect of electroencephalography-guided anesthetic administration on postoperative delirium among older adults undergoing major surgery: the ENGAGES randomized clinical trial. JAMA. 2019;321:473–483. doi: 10.1001/jama.2018.22005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Short T.G., Campbell D., Frampton C., et al. Anaesthetic depth and complications after major surgery: an international, randomised controlled trial. Lancet. 2019;394:1907–1914. doi: 10.1016/S0140-6736(19)32315-3. [DOI] [PubMed] [Google Scholar]
- 13.Short T.G., Leslie K., Chan M.T., Campbell D., Frampton C., Myles P. Rationale and design of the Balanced Anesthesia Study: a prospective randomized clinical trial of two levels of anesthetic depth on patient outcome after major surgery. Anesth Analg. 2015;121:357–365. doi: 10.1213/ANE.0000000000000797. [DOI] [PubMed] [Google Scholar]
- 14.Ely E.W., Margolin R., Francis J., et al. Evaluation of delirium in critically ill patients: validation of the confusion assessment method for the intensive care unit (CAM-ICU) Crit Care Med. 2001;29:1370–1379. doi: 10.1097/00003246-200107000-00012. [DOI] [PubMed] [Google Scholar]
- 15.Marcantonio E.R., Ngo L.H., O’Connor M., et al. 3D-CAM: derivation and validation of a 3-minute diagnostic interview for CAM-defined delirium: a cross-sectional diagnostic test study. Ann Intern Med. 2014;161:554–561. doi: 10.7326/M14-0865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ely E.W., Inouye S.K., Bernard G.R., et al. Delirium in mechanically ventilated patients: validity and reliability of the confusion assessment method for the intensive care unit (CAM-ICU) JAMA. 2001;286:2703–2710. doi: 10.1001/jama.286.21.2703. [DOI] [PubMed] [Google Scholar]
- 17.Maxwell M.J., Moran C.G., Moppett I.K. Development and validation of a preoperative scoring system to predict 30 day mortality in patients undergoing hip fracture surgery. Br J Anaesth. 2008;101:511–517. doi: 10.1093/bja/aen236. [DOI] [PubMed] [Google Scholar]
- 18.Sheehan B. Assessment scales in dementia. Ther Adv Neurol Disord. 2012;5:349–358. [Google Scholar]
- 19.Jitapunkul S., Pillay I., Ebrahim S. The abbreviated mental test: its use and validity. Age Ageing. 1991;20:332–336. doi: 10.1093/ageing/20.5.332. [DOI] [PubMed] [Google Scholar]
- 20.Punjasawadwong Y., Chau-In W., Laopaiboon M., Punjasawadwong S., Pin-On P. Processed electroencephalogram and evoked potential techniques for amelioration of postoperative delirium and cognitive dysfunction following non-cardiac and non-neurosurgical procedures in adults. Cochrane Database Syst Rev. 2018;5:CD011283. doi: 10.1002/14651858.CD011283.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.O’Regan N.A., Law T.W., Dunne D., et al. Delirium in older postoperative hip fracture patients. Hip Int. 2013;23:424–425. doi: 10.5301/hipint.5000067. [DOI] [PubMed] [Google Scholar]
- 22.Sieber F., Neufeld K.J., Gottschalk A., et al. Depth of sedation as an interventional target to reduce postoperative delirium: mortality and functional outcomes of the Strategy to Reduce the Incidence of Postoperative Delirium in Elderly Patients randomised clinical trial. Br J Anaesth. 2019;122:480–489. doi: 10.1016/j.bja.2018.12.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hshieh T.T., Saczynski J., Gou R.Y., et al. Trajectory of functional recovery after postoperative delirium in elective surgery. Ann Surg. 2017;265:647–653. doi: 10.1097/SLA.0000000000001952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Vasunilashorn S.M., Fong T.G., Albuquerque A., et al. Delirium severity post-surgery and its relationship with long-term cognitive decline in a cohort of patients without dementia. J Alzheimers Dis. 2018;61:347–358. doi: 10.3233/JAD-170288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Oh E.S., Fong T.G., Hshieh T.T., Inouye S.K. Delirium in older persons: advances in diagnosis and treatment. JAMA. 2017;318:1161–1174. doi: 10.1001/jama.2017.12067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Silbert B.S., Scott D.A. Informed consent in patients with frailty syndrome. Anesth Analg. 2020;130:1474–1481. doi: 10.1213/ANE.0000000000004629. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.