Abstract
Interactions between estrogen and growth factor signaling pathways at the level of gene expression play important roles in the function of reproductive tissues. For example, estrogen regulates transforming growth factor beta (TGFβ) in the uterus during the proliferative phase of the mammalian reproductive cycle. Bone morphogenetic protein 7 (BMP-7), a member of the TGFβ superfamily, is also involved in the development and function of reproductive tissues. However, relatively few studies have addressed the expression of BMP-7 in reproductive tissues, and the role of BMP-7 remains unclear. As part of an ongoing effort to understand how estrogen represses gene expression and to study its interactions with other signaling pathways, chick BMP-7 (cBMP-7) was cloned. cBMP-7 mRNA levels are repressed threefold within 8 h following estrogen treatment in the chick oviduct, an extremely estrogen-responsive reproductive tissue. This regulation occurs at the transcriptional level. Estrogen has a protective role in many tissues, and withdrawal from estrogen often leads to tissue regression; however, the mechanisms mediating regression of the oviduct remain unknown. Terminal transferase-mediated end-labeling and DNA laddering assays demonstrated that regression of the oviduct during estrogen withdrawal involves apoptosis, which is a novel observation. cBMP-7 mRNA levels during estrogen withdrawal increase concurrently with the apoptotic index of the oviduct. Furthermore, addition of purified BMP-7 induces apoptosis in primary oviduct cells. This report demonstrates that the function of BMP-7 in the oviduct involves the induction of apoptosis and that estrogen plays an important role in opposing this function.
Reproductive tissues exist in a delicate balance between the processes of proliferation, differentiation, cell cycle inhibition, and apoptosis (41). These processes are largely mediated by the actions and interactions of growth factors and steroid hormones (26, 67). Estrogen is a potent mitogenic steroid hormone involved in many aspects of the development and maintenance of numerous target tissues (65). The mammalian female reproductive tract, for example, is extremely sensitive to plasma estrogen levels during normal estrus cycles, as proliferation and regression of the endometrium depend largely on the estrogen concentration (6). These modulatory actions of estrogen are mediated by the estrogen receptor (ER), which affects gene expression by binding to estrogen response elements (EREs) in the regulatory regions of target genes (39). The interplay between estrogen and growth factor signaling at the level of gene expression is clearly important, since estrogen induces the genes that encode such growth factors as epidermal growth factor, transforming growth factor alpha (TGF-α), and insulin-like growth factor-1, which are involved in various aspects of reproductive homeostasis (25, 44, 45). In contrast, the transforming growth factor beta (TGF-β) superfamily of growth factors inhibit epithelial cell proliferation in reproductive tissue and often act to antagonize the proliferative estrogenic signals (15, 55). Although both growth hormone and steroid hormone pathways play important roles in reproductive physiology, little is known about the mechanisms of interaction between these pathways at the level of gene expression and the potential consequences of the interaction to reproductive tissues.
The bone morphogenetic proteins (BMPs) are a large subset of the TGF-β superfamily of extracellular signaling proteins that have been implicated in differentiation, development, and tissue remodeling (22). BMPs were initially identified by the ability of bone extracts to induce bone formation at extraskeletal sites (60, 73). BMP-7 (also called osteogenic protein-1 and OP-1) was originally cloned from a human genomic library in a screen designed to isolate genes involved in bone formation (50) and has since been cloned from a mouse (51), Xenopus laevis (76), and partially cloned from a chick (23). BMP-7 is synthesized as an inactive preprohormone and is proteolytically cleaved to produce a mature protein of 139 amino acids (50). The mature form of BMP-7 binds the type II serine/threonine kinase receptor, ActRII or ActRIIB, subsequently recruiting and phosphorylating the type I receptor, ALK-2 (38). The active form of ALK-2 signals through Smad1, -5, and -8, culminating in the regulation of BMP-7 target genes such as the genes for type X collagen (17, 74) and Tlx2 (70). Although some downstream events in the BMP-7 pathway are known, the regulation of the BMP-7 gene remains a mystery since neither the transcriptional start site nor the promoter of the BMP-7 gene has been characterized in any system.
BMP-7 has been implicated in such diverse processes as murine hind brain development (2), tooth development (19), nephrogenesis, eye development, and skeletal patterning (36). A BMP-7 null mutation in mice results in severe developmental defects in the skeleton, eye, and kidney (28). BMP-7-deficient mice also exhibit polydactyly in their hind limbs, indicating a role for BMP-7 in digit formation (28). These data suggest that BMP-7 may also have important roles in tissue remodeling. In addition to the role of BMP-7 in development, a virtually unexplored role in reproductive tissue is emerging. Because estrogen injection into nonpregnant mice decreases BMP-7 mRNA levels in the uterus (49) and because withdrawal of estrogen induces apoptosis of the uterine epithelium (75), we speculated that estrogen withdrawal leads to an increase in BMP-7, which triggers apoptosis in estrogen-responsive organs such as the uterus and oviduct. However, no direct evidence exists to suggest that BMP-7 has a role in induction of apoptosis in the uterine epithelium, and no biochemical data exist as to the exact function of BMP-7 in the uterus.
To further define the relationship between estrogen and BMP-7 and to determine the physiological significance of BMP-7 expression in reproductive organs, BMP-7 mRNA levels were examined in the chick oviduct. The oviduct is the functional equivalent of the human Fallopian tube and is extremely estrogen responsive (6), making it an ideal system for studying estrogen regulation of transcription. Additionally, the development of the oviduct can be precisely controlled in immature chicks, allowing detailed examination of specific developmental processes. This study provides additional characterization of the inverse relationship between estrogen and BMP-7 mRNA expression by demonstrating that estrogen represses chick BMP-7 (cBMP-7) gene expression at the transcriptional level. We also report that estrogen deficiency leads to increased cBMP-7 mRNA production and stimulates apoptosis of the oviduct. Finally, addition of purified BMP-7 to primary oviduct cells directly induces apoptosis, demonstrating that BMP-7 has an important role in tissue remodeling by driving apoptotic events.
MATERIALS AND METHODS
Animals and estrogen treatments.
Two-week-old, sexually immature White Leghorn chicks were implanted with 2 diethylstilbestrol (DES) pellets (Hormone Pellet Press, Leawood, Kans.) for at least 2 weeks to induce development of the oviduct. Withdrawn oviducts were produced by withdrawal of the DES pellets. DES was withdrawn for 5 days, unless otherwise noted. Secondary stimulation of 5-day estrogen-withdrawn chicks was performed for the indicated times by wing vein injection of 17-β-estradiol (25 mg/kg of body weight). After all treatments, the magnum portion of the oviduct was frozen in liquid nitrogen prior to storage at −70°C.
Cloning of cBMP-7.
In order to clone the complete cBMP-7 coding sequence, a 5-day estrogen-withdrawn chick oviduct cDNA library was screened with a PCR-generated probe designed to a chick partial cDNA sequence (23). The probe was constructed using a 5′ primer located at +367 to +382 and a 3′ primer located at +1306 to +1328 relative to the translational start codon (Fig. 1). The previously cloned cBMP-7 sequence included nucleotides +367 to +1364. Our efforts resulted in a more complete sequence ranging from +111 to +1728 (GenBank accession number AF205877). A 3′-RACE (rapid amplification of cDNA ends) reaction using a 5-day estrogen-withdrawn library constructed using the Marathon cDNA amplification kit (Stratagene, La Jolla, Calif.) and the 5′ primer described above generated a clone extending to +1974 that included a polyadenylation signal and a polyadenylated [poly(A)+] tail. Sequence comparison with human and mouse BMP-7 suggested that the extreme 5′ end of the coding sequence was absent. Further screening of the 5-day W/D oviduct cDNA library and the 5-day estrogen-withdrawn oviduct RACE library yielded no additional 5′ sequence. Therefore, a chicken genomic library (courtesy of Kathleen Conklin, University of Minnesota) was screened using a probe located from +121 to +324, which is contained entirely on exon 1 in the mouse and is located on the same exon in the chicken (data not shown). A clone located from −1808 to +374 (GenBank accession number AF223970) relative to the start site of translation was isolated that included an in-frame translational start codon. The translated sequence was aligned with both human (GenBank accession number X51801) and mouse (GenBank accession number X56906) sequences (Fig. 2) using the GCG computer package (Genetics Computer Group, Madison, Wis.).
FIG. 1.
Full-length cBMP-7 cDNA clone obtained as described in Materials and Methods. The complete cBMP-7 sequence (GenBank accession numbers AF205877 and AF223970) was translated, and the consensus ATG translational start codon is in bold. The nucleotides are numbered on the left using ATG as nucleotide +1 because the promoter and 5′-UTR have not been mapped. The predicted form of cBMP-7 following processing at the RSIR cleavage site is underlined. An asterisk denotes the translational stop codon, and the polyadenylation recognition sequence is boxed. The previously cloned cBMP-7 sequence included nucleotides +367 to +1364 (23).
FIG. 2.
cBMP-7 amino acid sequence is highly homologous to human and mouse BMP-7. The cBMP-7 amino acid sequence was aligned with the human (X51801) and mouse (X56906) BMP-7 sequences using the GCG computer package and found to be 86% identical throughout the entire protein. The dashes represent identical amino acid matches, and the dots represent gaps in the sequence. The amino acid sequence is numbered on the left.
Tissue preparation and Northern blot analysis.
Total RNA was isolated using the phenol-guanidinium isothiocyanate-based RNAzol B reagent (Tel-Test, Friendsville, Tex.), and poly(A)+-selected RNA was isolated using the Poly(A)+ Tract isolation kit (Promega, Madison, Wis.). Northern blot analysis was conducted by electrophoresing 2 μg of poly(A)+ RNA on an 0.8% formaldehyde gel. The separated RNA was transferred to nitrocellulose (Schleicher & Schuell, Keene, N.H.) using an electrotransfer apparatus (Bio-Rad, Hercules, Calif.) and cross-linked by UV irradiation (Stratagene). The Northern blots were hybridized overnight at 42°C with the indicated random primer-labeled cDNA probes (random primer labeling kit from Stratagene) in hybridization solution. The blots were washed in a final wash of 0.2× SSC (1× SSC is 0.15 M NaCl plus 0.015 M sodium citrate)–0.1% sodium dodecyl sulfate for 15 min and exposed to Hyperfilm with an intensifying screen at −70°C for 3 days. The housekeeping gene glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as a non-estrogen-regulated control (40) to demonstrate equal loading of the lanes. Densitometric analysis of RNA bands was conducted using a Bio-Rad densitometer and the Molecular Analyst software package (Bio-Rad). Statistics were generated using Scheffe's analysis of variance test for statistical significance. Differences were considered significant at P < 0.05.
Nuclear run-on transcription assays.
Nuclear run-on assays were performed as previously described with a few modifications (47). Briefly, nuclei were obtained from 5-day estrogen-withdrawn and estrogen-stimulated oviducts by Dounce homogenization followed by ultracentrifugation. The nuclei were placed in a transcription reaction that included 125 μCi each of 32P-labeled UTP and CTP (700 Ci/mmol; New England Nuclear) for 40 min and subsequently treated with proteinase K and DNase. The labeled RNA was extracted using RNAzol, and approximately 2 × 107 cpm were hybridized for 36 h at 65°C to 4 μg of cDNA immobilized on a filter. The cBMP-7 cDNA consisted of a fragment spanning the 3′ end of the coding region and the 3′ untranslated region (UTR) (nucleotides 360 to 1328). Full-length hepatocyte nuclear factor 3β (HNF-3β) cDNA (3) was used as a non-estrogen-regulated control. The blots were washed, and unhybridized RNA was removed by treatment with RNase A and RNase T1 at 37°C for 1 h. Filters were exposed to a phosphorimager screen overnight, and quantitation was performed using the Molecular Analyst software (Bio-Rad).
Detection of apoptosis.
The induction of apoptosis was determined by using a terminal transferase-mediated end-labeling procedure (TUNEL) on serial sections of paraffin-embedded oviduct tissue (Surgical Pathology Lab, University of Minnesota), using the ApopTag assay kit (Intergen, Purchase, N.Y.). TUNEL-positive cells were scored by examination of four consecutive fields (×200 magnification) and totaled as a percentage of 4,000 cells each. Negative controls for each tissue consisted of adjacent sections where terminal transferase was omitted. No significant background staining was observed in any of the control sections (data not shown). Detection of apoptosis in primary oviduct cell culture was accomplished by measuring internucleosomal DNA laddering using the TACS apoptotic DNA laddering kit-isotopic (Sigma, St. Louis, Mo.) per the manufacturer's protocol. Briefly, DNA was extracted from primary oviduct cells and Klenow-labeled using 0.5 μCi of dCTP (3,000 Ci/mmol; New England Nuclear). The entire reaction was separated on a 1.5% agarose gel, dried, and exposed to film for 8 h. Quantitation was performed by densitometry of the three smallest oligonucleosomal fragments.
Primary oviduct cell culture.
Preparation of the primary oviduct cells was performed as previously described (61). Briefly, 2-day estrogen-withdrawn chick oviducts were dissociated using a mixture of collagenase, protease, DNase, and trypsin. Dissociated cells were washed three times with culture medium, and cells from 100 mg of tissue were plated into each dish containing serum-free Dulbecco's modified Eagle's medium-F12 medium (1:1) and the indicated concentrations of mouse BMP-7 (Creative BioMolecules, Hopkinton, Mass.). Cells were harvested 20 h later, resuspended in Tris buffer, and frozen until the intranucleosomal DNA laddering assay was performed.
RESULTS
Cloning of the cBMP-7 coding region indicates that it is highly homologous to human and mouse BMP-7.
Characterization of the promoter and enhancer sequences involved in regulation of the BMP-7 gene would facilitate the understanding of BMP-7 function and responsiveness to cellular signals. However, these cis-acting sequences have not been identified in any system. As a first step in studying the regulation of the BMP-7 gene in the chick oviduct, and since a previously cloned chick cDNA lacks the 5′ end of the open reading frame (23), an effort to clone the complete open reading frame of cBMP-7 and the 5′-flanking region was initiated. Cloning of the coding region of cBMP-7 was accomplished by screening an estrogen withdrawn chick oviduct cDNA library, a 3′-RACE estrogen-withdrawn chick oviduct cDNA library, and a chicken genomic library (see Materials and Methods for specifics). The deduced amino acid sequence reveals that the cBMP-7 gene encodes a 435-amino-acid protein (Fig. 1), whereas the human and mouse BMP-7 genes encode proteins of 431 and 430 amino acids, respectively (50, 51). The similarity of the predicted size of cBMP-7 to human and mouse BMP-7 suggests that the entire coding region has been cloned. Alignment of the coding region of cBMP-7 with human and mouse BMP-7 sequences (Fig. 2) reveals 86% identity. A predicted dibasic protease cleavage sequence (RSIR) exists (amino acids 293 to 296), as observed in human and mouse BMP-7 sequences. Cleavage at this site would produce a mature protein of 139 amino acids that is 96% identical to human and mouse BMP-7. This predicted protein contains the conserved 7-cysteine domain observed in many members of the TGF-β superfamily. The 3′-UTR of cBMP-7 contains a polyadenylation signal sequence (11), indicating that the entire 3′ coding sequence is cloned. Cloning of the extreme 5′ open reading frame was accomplished by screening a chicken genomic library and is characterized by an extremely GC-rich sequence. Thus, the cloning of the cBMP-7 coding region including a previously undescribed 3′-UTR has been accomplished. However, S1 analysis of ∼500 bp upstream of the start site of translation (data not shown) did not reveal an obvious promoter or start site for transcription, so the location and nature of the cBMP-7 regulatory sequences remain undetermined.
cBMP-7 mRNAs are repressed by estrogen at the transcriptional level in the oviduct.
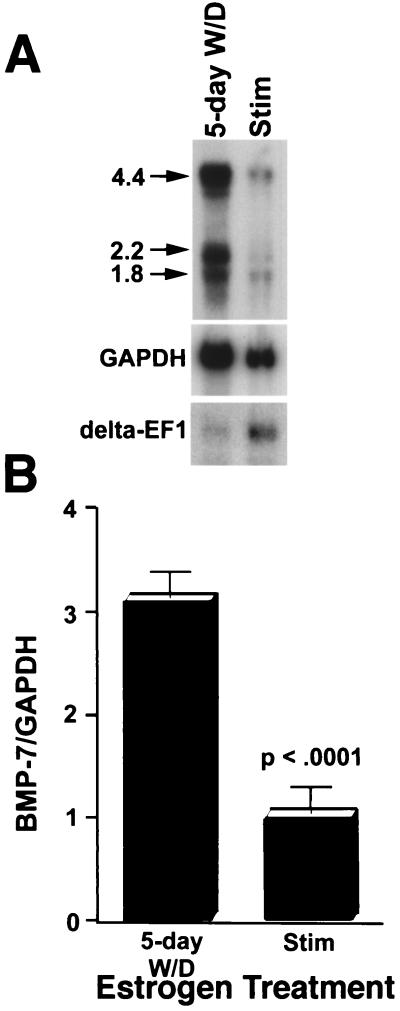
Regulation of reproductive processes often includes the modulatory actions of estrogen on gene expression, including genes involved in growth hormone signaling (15, 25, 44, 45, 55). In order to determine whether this generalization can be extended to BMP-7 signaling in the oviduct, Northern blot analysis of poly(A)+ RNA from estrogen withdrawn and estrogen-stimulated oviducts was performed using a probe specific for cBMP-7 (data not shown). cBMP-7 mRNA levels are decreased threefold in estrogen-stimulated oviducts compared to estrogen-withdrawn oviducts (Fig. 3A). A similar fourfold repression in the amount of cBMP-7 mRNA was observed in untreated laying hens, indicating that repression by estrogen does indeed occur in a physiologically relevant state (data not shown). Three major BMP-7 mRNA species are observed, consistent with a previously published report for mouse BMP-7 (51). Densitometry indicates that all mRNA species are repressed to roughly the same extent. The exact nature of the multiple transcripts is currently unknown in any system. The estrogen-inducible transcription factor delta-EF1 (7) is shown to illustrate that a concomitant inductive response occurs in estrogen-stimulated oviducts, confirming that estrogen does not have a generalized negative effect on transcription in the oviduct. To the contrary, the genes for ovalbumin (62), connexin 43 (20), glycoprotein (27), cystic fibrosis transmembrane regulator (56), leukemia inhibitory factor (54), H-ras (9), and myosin light-chain kinase (57) are induced by estrogen in the oviducts of various species. Additionally, the genes for epidermal growth factor and TGF-α are induced in the oviduct (63), demonstrating that some members of the TGF-β superfamily can respond positively to estrogen in this tissue and that there is some specificity in the repression of the cBMP-7 gene by estrogen.
FIG. 3.
cBMP-7 gene expression is repressed by chronic estrogen treatment. (A) Poly(A)+ RNA (2 μg) was isolated from 5-day estrogen-withdrawn (5-day W/D) and estrogen-stimulated (Stim) oviducts and subjected to Northern blot analysis. The blot was probed with a cBMP-7 cDNA fragment spanning nucleotides 360 to 1328 (Fig. 1). This probe was determined to be specific to cBMP-7, since a probe designed exclusively to the 3′-UTR yielded identical results upon Northern blot analysis (data not shown). Arrows point to the main mRNA species (labeled in kilobases). The blot was probed a second time with GAPDH, a gene not regulated by estrogen (40), to normalize for loading of the lanes. The estrogen-inducible gene delta-EF1 (7) is shown to demonstrate that estrogen does not have a generalized repressive effect on the oviduct. (B) The experiment was repeated five times and the 4.4-kb band was subjected to densitometric analysis. The BMP-7-GAPDH ratio was plotted relative to stimulation. The error bars represent the standard deviation, and the repression was determined to be statistically significant (P < 0.0001).
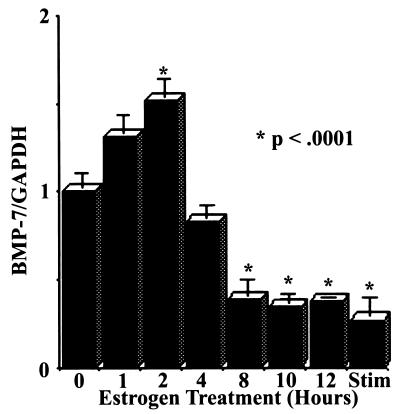
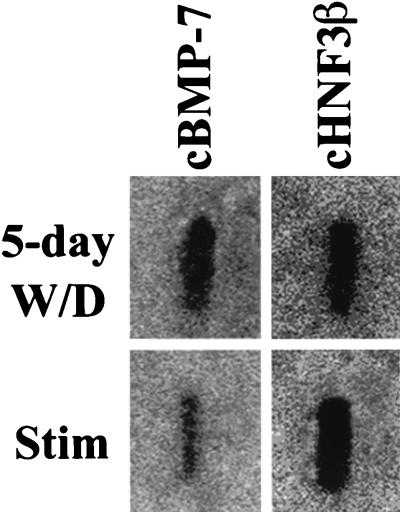
In order to determine the kinetics of repression of cBMP-7 gene expression by estrogen, estrogen-withdrawn chicks were injected with 17β-estradiol for the indicated times, and Northern blot analysis was conducted (Fig. 4). cBMP-7 mRNA levels are decreased to estrogen-stimulated levels within 8 h of treatment. Surprisingly, a 1.5-fold induction of cBMP-7 mRNAs was observed 2 h following estrogen treatment. The reason for this rapid but transient increase in cBMP-7 mRNA remains unknown. In order to determine whether the decrease in cBMP-7 mRNA is due to transcriptional or posttranscriptional events, a nuclear run-on experiment was conducted (Fig. 5). The transcriptional activity of the cBMP-7 gene is repressed 3.5-fold in the estrogen-treated oviduct compared with the estrogen-withdrawn oviduct. The extent of repression agrees well with the Northern blot data (Fig. 3 and 4), indicating that the regulation of cBMP-7 by estrogen occurs at the transcriptional level. These data indicate that estrogen represses cBMP-7 gene expression and that an inverse relationship between estrogen concentration and cBMP-7 mRNA levels exists in the oviduct. This is the first information garnered in any system that indicates how the expression of the BMP-7 gene is regulated.
FIG. 4.
17β-Estradiol represses cBMP-7 mRNAs within 8 h. Poly(A)+ RNA (2 μg) was isolated from oviducts after injection of 5-day estrogen-withdrawn animals with 17β-estradiol, and Northern blot analysis was performed as in Fig. 3. This experiment was performed twice in duplicate. The asterisks signify samples that are statistically different from the 5-day estrogen-withdrawn or zero time point samples (P < 0.0001).
FIG. 5.
Repression of cBMP-7 gene expression by estrogen occurs at the transcriptional level. Nuclei were isolated from 5-day estrogen-withdrawn (W/D) and estrogen-stimulated (Stim) oviducts and placed in a nuclear run-on transcription reaction. RNA was isolated and hybridized to a filter containing either cBMP-7 or HNF-3β cDNAs. HNF-3β, a gene not transcriptionally regulated by estrogen (3), was used as a control.
Estrogen withdrawal induces apoptosis of the oviduct.
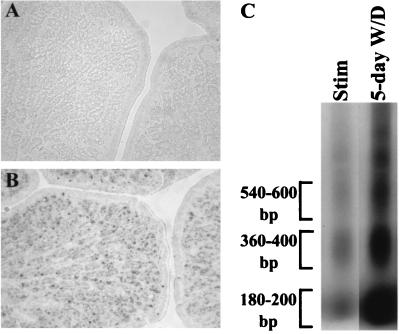
Steroid hormones are essential for maintaining reproductive vitality. However, during the female reproductive cycle and during aging, steroid levels fluctuate or diminish, respectively (6). Few studies have examined the resultant effects on reproductive tissues. In periods of low plasma estrogen levels, the oviduct regresses in size due to unknown mechanisms (8, 64). TUNEL (13) and internucleosomal DNA laddering (1) analyses were performed on estrogen-stimulated and estrogen-withdrawn oviducts in order to determine whether estrogen withdrawal induces apoptosis. TUNEL analysis indicated that in estrogen-stimulated oviduct, 0.44% ± 0.38% (standard deviation) of the total number of cells are apoptotic, whereas in estrogen-withdrawn oviduct, 7.92% ± 2.2% of the cells are apoptotic (Fig. 6A and B, respectively). This demonstrates that estrogen withdrawal triggers an 18-fold increase in apoptosis. To further verify that estrogen withdrawal induces apoptosis of the chick oviduct, an internucleosomal DNA laddering assay was performed (Fig. 6C). DNA fragmentation of chromatin into oligonucleosome-length fragments is a hallmark of the apoptotic response (34, 35). The internucleosomal DNA laddering assay indicated that estrogen withdrawal induces a sixfold increase in DNA laddering compared to the estrogen-stimulated oviduct (Fig. 6C). The discrepancy between the fold induction of apoptosis between the TUNEL and DNA laddering assays can be attributed to the different sensitivities of each individual assay (16). Nonetheless, the data indicate that estrogen withdrawal specifically induces apoptosis. This observation is the first direct evidence that the regression of the oviduct due to estrogen withdrawal is the result of apoptosis.
FIG. 6.
Withdrawal of estrogen induces apoptosis in the oviduct. Oviducts from (A) estrogen-stimulated and (B) 5-day estrogen-withdrawn chicks were sectioned and subjected to TUNEL analysis. The dark spots represent TUNEL-positive cells, which were scored by examination of four consecutive fields and totaled as a percentage of ∼4,000 cells. An 18-fold induction of apoptosis was observed in the 5-day estrogen-withdrawn oviduct. The experiment was repeated twice with similar results. (C) Apoptosis was also determined using an internucleosomal DNA fragmentation laddering assay. DNA (1 μg) was Klenow-labeled and separated by gel electrophoresis. The brackets denote the areas subjected to densitometric analysis, and the numbers indicate the sizes of the oligonucleosomal DNA fragments. The values of the three areas were totaled to quantitate the amount of DNA laddering per lane. A sixfold increase in DNA laddering was observed in the 5-day estrogen-withdrawn oviduct. The experiment was repeated twice with similar results.
Regression of the oviduct correlates with increased cBMP-7 mRNA levels.
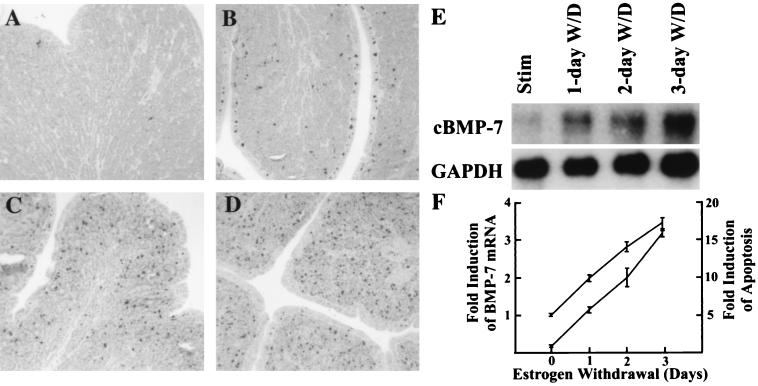
cBMP-7 gene expression is repressed by estrogen (Fig. 3 to 5), suggesting an inverse relationship between estrogen concentration and both the production of cBMP-7 mRNA and the induction of apoptosis (Fig. 6). Since both events occur following estrogen withdrawal, it seems probable that cBMP-7 is involved in the apoptotic response. To determine whether these events follow a similar time course during shorter withdrawal periods, estrogen was withdrawn for 1, 2, or 3 days. TUNEL and Northern blot analyses were done to determine whether apoptosis and an increase in cBMP-7 mRNA levels occur concomitantly. As shown in Fig. 7, little apoptosis is observed in the estrogen-stimulated oviduct (Fig. 7A). However, a sixfold increase in apoptosis is observed in the 1-day estrogen-withdrawn oviduct (Fig. 7B), with a 10- and 16-fold induction of apoptosis in 2-day and 3-day estrogen-withdrawn oviducts, respectively (Fig. 7C and D). Northern blot analysis indicated cBMP-7 mRNA levels follow the same temporal pattern as apoptosis, with a 2-fold increase in the 1-day estrogen-withdrawn oviduct, a 2.8-fold increase in 2-day withdrawn oviduct, and a 3.5-fold increase in 3-day withdrawn oviduct (Fig. 7E). The increase in cBMP-7 mRNA mirrors the onset of apoptosis induced by estrogen withdrawal (Fig. 7F). These data demonstrate that the induction of apoptosis and the increase in cBMP-7 mRNA levels are concomitant.
FIG. 7.
Increasing apoptosis in oviduct withdrawn from estrogen for various times is correlated with an increase in BMP-7 mRNA. Chicks stimulated with estrogen for 21 days were withdrawn from estrogen for 1, 2, or 3 days. Part of the oviduct was paraffin embedded, sectioned, and subjected to TUNEL analysis. The rest of the oviduct was used to isolate poly(A)+ RNA for Northern blot analysis. (A) Results of the TUNEL assay on estrogen-stimulated chicks. The other panels are the results of the TUNEL assay on chicks withdrawn from estrogen for (B) 1, (C) 2, or (D) 3 days, indicating an increase in apoptotic cells throughout the withdrawal time course. (E) Northern blot analysis indicates that the increase in BMP-7 mRNA correlates with the increase in apoptosis. Only the 4.4-kb mRNA of BMP-7 is shown. GAPDH mRNA was measured as the loading control. (F) The induction of cBMP-7 mRNA and an increase in apoptosis over time are graphically represented as the top and bottom line, respectively. This experiment was repeated with comparable results. Data points and error bars represent the mean and the range, respectively.
BMP-7 induces apoptosis in primary oviduct cell culture.
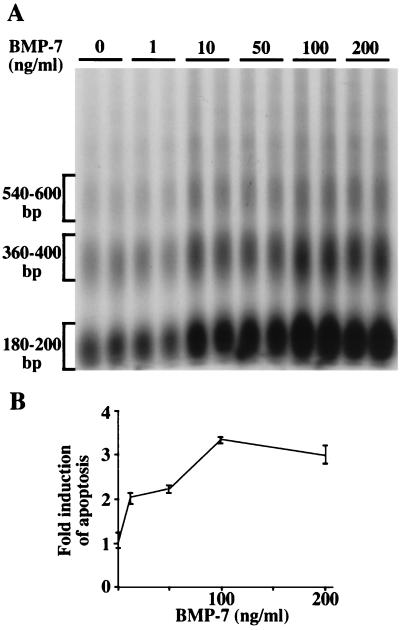
To directly test the possibility that BMP-7 induces apoptosis in the oviduct, primary oviduct cells were cultured with increasing concentrations of mouse BMP-7 and harvested 20 h later. Induction of apoptosis in primary oviduct cells was dose dependent, with a twofold increase at 10 and 50 ng/ml and a threefold increase at 100 and 200 ng/ml (Fig. 8). The primary oviduct cell culture model system that has been developed (61) utilizes 2-day estrogen-withdrawn oviduct as a source of cells. Since apoptosis is already occurring in this tissue (Fig. 7C), the fold induction of apoptosis reported in Fig. 8 represents an increase in the apoptotic index over a 2-day estrogen-withdrawn oviduct. Thus, the level of apoptosis induced in primary oviduct cell cultures by treatment with BMP-7 for 20 h is comparable to that induced by estrogen withdrawal in vivo. These data demonstrate that BMP-7 induces molecular events leading to apoptosis.
FIG. 8.
Addition of mouse BMP-7 to primary oviduct cell culture induces apoptosis. Oviducts withdrawn from estrogen for 2 days were dissociated and placed into tissue culture as previously described (61). Cultures were treated with the indicated BMP-7 concentrations for 20 h and harvested. Internucleosomal DNA laddering was performed as in Fig. 6. Treatment of primary oviduct cells with 10 and 50 ng of BMP-7 per ml resulted in a twofold induction of apoptosis, whereas the highest concentrations induced a threefold response. Each BMP-7 treatment was done in duplicate, and the experiment was conducted twice with similar results. Data points and error bars represent means and ranges, respectively.
DISCUSSION
This study demonstrates that estrogen represses cBMP-7 gene expression in the oviduct (Fig. 3), agreeing well with a previous observation that estrogen treatment leads to decreased BMP-7 mRNAs in the mouse uterus (49). Repression of cBMP-7 mRNA levels was observed within 8 h following 17β-estradiol injection (Fig. 4) and occurs at the transcriptional level (Fig. 5). In an attempt to identify an ER binding site or an estrogen-dependent repressor element to explain the repression of cBMP-7 by estrogen, a 3.8-kb genomic DNA fragment upstream of the translational start codon of cBMP-7 was isolated. Sequence analysis did not reveal any estrogen response elements or any known estrogen-responsive repressor elements (data not shown). The genomic sequence contained no obvious promoter elements (TATA box, CCAAT box, etc.); however, an extensive GC-rich region containing numerous SP-1 binding sites upstream of the coding region is reminiscent of TATA-less promoters (66). Since the transcriptional start site and promoter for the BMP-7 gene have not yet been characterized in any species, the mechanisms of estrogen-mediated repression of cBMP-7 gene expression remain unknown.
Estrogen-specific repression of gene expression has been observed for the interleukin-6 (53), follicle-stimulating hormone-β (43), glycoprotein hormone-α subunit (30), and gonadotropin releasing hormone (29) genes. In all cases, repression of gene expression by estrogen occurs in the absence of a high-affinity DNA-binding site for ER, although mutation or deletion of the DNA-binding domain of ER abolishes repression (43, 53). It is thought that the DNA-binding domain may participate in protein-protein interactions or may be required for the proper function of the ER. Indeed, the repression of the interleukin-6 gene is mediated by a protein interaction between ER and the necessary activator, NF-κB, preventing it from activating transcription (12, 31, 53). Interestingly, there is a potential NF-κB site upstream of the start site of translation of cBMP-7 (data not shown), raising the possibility that ER may repress the BMP-7 gene by squelching the effects of NF-κB. However, functional characterization of the cBMP-7 promoter is needed to determine the mechanism of repression.
Regulation of the BMP-7 gene by estrogen appears to be a complex process, since a statistically significant 1.5-fold induction occurs within 2 h following estrogen treatment. The function of this transient increase in mRNA levels is not clear, although it is interesting to speculate that the initial increase in BMP-7 may play a role in its repression at later times. Similarly, TGF-β1-3 mRNA levels are also induced quickly (<2 h) following estrogen treatment in the uterus but decrease to below control levels by 6 h (68), suggesting that this type of regulation may represent a general theme for estrogen-regulated TGF-β family members. Regulation of BMP-7 mRNA levels also appears to be multihormonal, since androgen treatment of castrated mice results in a fourfold increase in BMP-7 mRNA levels in the prostate within 72 h (71), lending support to the contention that steroid hormones regulate expression of the BMP-7 gene. Regulation of the BMP-7 signaling pathway in the oviduct also occurs at the level of the receptor, as estrogen withdrawal increases the sensitivity of the oviduct to cBMP-7 by upregulating its receptor, ALK-2 (38), approximately 2.5-fold (data not shown). Similarly, estrogen withdrawal also induces an increase in TGF-β type II receptor mRNA in the uterine endometrium, leading to an increased sensitivity of the tissue to TGF-β ligands (75). It appears clear that the convergence of the estrogen and BMP-7 pathways occurs at multiple points, generating a greater response of the oviduct to BMP-7 signaling.
We report the novel observation that estrogen withdrawal initiates cellular events leading to apoptosis in the oviduct (Fig. 6 and 7). Induction of apoptosis following estrogen withdrawal has been observed in other tissues, such as the uterine epithelium (75), vagina (4), osteocytes (72), and MCF-7 xenografts in nude mice (10). These observations are not confined to estrogen withdrawal, since androgen withdrawal also induces apoptosis of the ventral prostate gland (5, 32). This is consistent with the concept that steroid hormones are involved in the maintenance of their target tissues, and withdrawal from hormone results in cellular events that often culminate in apoptosis. However, the molecular mediators directly driving apoptosis in these tissues are unknown. Our observation that BMP-7 drives apoptosis in the oviduct raises the possibility that BMP-7 mediates apoptosis in these other reproductive tissues as well. Additionally, while some members of the BMP family have been implicated in apoptotic processes (14, 37), BMP-7 is the first family member implicated in apoptosis of a reproductive tissue, which may represent a previously unrecognized function for BMP molecules.
Cellular events leading to apoptosis are mediated by modulation of the expression of specific genes or alteration of existing gene products (48). However, the specific intracellular mechanisms involved in the activation of apoptotic pathways by BMP-7 signaling are unknown. We expect that the caspase (42, 48, 52) and Bcl (24, 46) gene families are of importance in regulating apoptotic events in the oviduct due to their nearly universal involvement in known apoptotic pathways (33). Delineation of the molecular target(s) of BMP-7 signaling in the apoptotic pathway may provide a general model of the molecular mechanisms behind tissue remodeling in both normal and pathological reproductive function.
The observation that BMP-7 induces apoptosis in a reproductive tissue implies that BMP-7 may be involved in tissues remodeling during periods of low estrogen in normal reproductive cycles. One report demonstrated that in molting laying hens, which are characterized by total regression of the oviduct, a significant increase in apoptosis is observed (21). However, the estrogen status was undetermined, although an earlier study suggested that estrogen levels are decreased in molting laying hens (69). Although cBMP-7 mRNA levels in the oviduct of a molting hen are unknown, our data suggest that cBMP-7 mRNA levels are induced in the oviduct of a molting hen, triggered by declining estrogen concentrations. Similarly, the onset of menstruation in mammalian reproductive cycles is also characterized by a decrease in plasma estrogen levels followed by extensive endometrial cell death (6). We provide the first direct evidence implicating a specific protein, BMP-7, in inducing apoptotic reproductive tissue remodeling.
Previous studies have demonstrated that purified bovine osteogenic protein exists as a BMP-7/BMP-2A heterodimer (58). However, recombinant human BMP-7 existing as a homodimer induces similar levels of osteogenic activity (59), suggesting that both the heterodimer and homodimer forms are functional in vivo. Another report demonstrated that the heterodimeric form exhibits greater activity than the homodimeric form (18). Thus, the biologically relevant signaling forms remain controversial. However, since the BMP-7 used in this report (Fig. 8) exists as a homodimer, our data suggest that the homodimeric form of BMP-7 is sufficient to induce apoptosis of the oviduct. Whether a heterodimeric form also exists in chickens must await the cloning of other potential partners. For the same reason, these studies also do not address the issue of whether other BMP family members elicit apoptosis in the oviduct.
In conclusion, our data indicate that BMP-7 is a molecular mediator of apoptosis, linking the processes of decreased plasma estrogen levels with the initiation of tissue remodeling. Furthermore, estrogen suppresses these effects by evoking events leading to the transcriptional repression of the BMP-7 gene. Thus, the reproductive health of the oviduct is critically dependent upon the relative levels of estrogen and BMP-7. Identification of the BMP-7 promoter and sequence elements involved in the response to estrogen will provide further information regarding the mechanisms by which estrogen represses BMP-7 gene expression and thus opposes apoptosis.
ACKNOWLEDGMENTS
We thank Donald F. Jin (Creative BioMolecules) for providing the purified mouse BMP-7 protein and Kathleen Conklin (University of Minnesota) for the chick genomic library.
This research was supported by NIH grant R01 DK40082 to M.M.S.
REFERENCES
- 1.Arends M J, Morris R G, Wyllie A H. Apoptosis: the role of the endonuclease. Am J Pathol. 1990;136:593–608. [PMC free article] [PubMed] [Google Scholar]
- 2.Arkell R, Beddington R S P. BMP-7 influences pattern and growth of the developing hindbrain of mouse embryos. Development. 1997;124:1–12. doi: 10.1242/dev.124.1.1. [DOI] [PubMed] [Google Scholar]
- 3.Berger R R, Sanders M M. Estrogen modulates HNF-3β mRNA levels in the developing oviduct. DNA Cell Biol. 2000;19:103–112. doi: 10.1089/104454900314618. [DOI] [PubMed] [Google Scholar]
- 4.Berman J R, McCarthy M M, Kyprianou N. Effect of estrogen withdrawal on nitric oxide synthase expression and apoptosis in the rat vagina. Urology. 1998;51:650–655. doi: 10.1016/s0090-4295(97)00683-3. [DOI] [PubMed] [Google Scholar]
- 5.Brodin G, ten Dijke P, Funa K, Heldin C-H, Landstrom M. Increased Smad expression and activation are associated with apoptosis in normal and malignant prostate after castration. Cancer Res. 1999;59:2731–2738. [PubMed] [Google Scholar]
- 6.Carr B R. Disorders of the ovary and female reproductive tract. 8th ed. Philadelphia, Pa: W. B. Saunders Company; 1992. [Google Scholar]
- 7.Chamberlain E M, Sanders M M. Identification of the novel player δEF1 in estrogen transcriptional cascades. Mol Cell Biol. 1999;19:3600–3606. doi: 10.1128/mcb.19.5.3600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cohrs R, Goswami B B, Sharma O K. Occurrence of 2-5A and RNA degradation in the chick oviduct during rapid estrogen withdrawal. Biochemistry. 1988;27:3246–3252. doi: 10.1021/bi00409a019. [DOI] [PubMed] [Google Scholar]
- 9.Cohrs R J, Goswami B B, Sharma O K. Down regulation of c-myc, c-fos and erb-B during estrogen induced proliferation of the chick oviduct. Biochem Biophys Res Commun. 1988;150:82–88. doi: 10.1016/0006-291x(88)90489-5. [DOI] [PubMed] [Google Scholar]
- 10.Detre S, Salter J, Barnes D M, Riddler S, Hills M, Johnston S R D, Gillett C, A'Hern R, Dowsett M. Time-related effects of estrogen withdrawal on proliferation and cell death-related events in MCF-7 xenografts. Int J Cancer. 1999;81:309–313. doi: 10.1002/(sici)1097-0215(19990412)81:2<309::aid-ijc23>3.0.co;2-s. [DOI] [PubMed] [Google Scholar]
- 11.Fitzgerald M, Schenk T. The sequence 5′-AAUAAA-3′ forms part of the recognition sequence for polyadenylation of late SV40 mRNAs. Cell. 1981;24:251–260. doi: 10.1016/0092-8674(81)90521-3. [DOI] [PubMed] [Google Scholar]
- 12.Galien R, Garcia T. Estrogen receptor impairs interleukin-6 expression by preventing protein binding on the NF-κB site. Nucleic Acids Res. 1997;25:2424–2429. doi: 10.1093/nar/25.12.2424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gavrieli Y, Sherman Y, Ben-Sasson S A. Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol. 1992;119:493–501. doi: 10.1083/jcb.119.3.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Glozak M A, Rogers M B. Specific induction of apoptosis in P19 embryonal carcinoma cells by retinoic acid and BMP2 or BMP4. Dev Biol (Orlando) 1996;179:458–470. doi: 10.1006/dbio.1996.0275. [DOI] [PubMed] [Google Scholar]
- 15.Gold L I, Parekh T V. Loss of growth regulation by transforming growth factor-β (TGF-β) in human cancers: studies on endometrial carcinoma. Semin Reprod Endocrinol. 1999;17:73–92. doi: 10.1055/s-2007-1016214. [DOI] [PubMed] [Google Scholar]
- 16.Gong J, Traganos F, Darzynkiewicz Z. A selective procedure for DNA extraction from apoptotic cells applicable for gel electrophoresis and flow cytometry. Anal Biochem. 1994;218:314–319. doi: 10.1006/abio.1994.1184. [DOI] [PubMed] [Google Scholar]
- 17.Harada S, Sampath T K, Aubin J E, Rodan G A. Osteogenic protein-1 up-regulation of the collagen X promoter activity is mediated by a MEF-2-like sequence and requires an adjacent AP-1 sequence. Mol Endocrinol. 1997;11:1832–1845. doi: 10.1210/mend.11.12.0022. [DOI] [PubMed] [Google Scholar]
- 18.Hazama M, Aono A, Ueno N, Fujisawa Y. Efficient expression of a heterodimer of bone morphogenetic protein subunits using a baculovirus expression system. Biochem Biophys Res Commun. 1995;209:859–866. doi: 10.1006/bbrc.1995.1578. [DOI] [PubMed] [Google Scholar]
- 19.Helder M N, Karg A, Bervoets T J M, Vukicevic S, Burger E H, D'Souza R N, Woltgens J H M, Karsenty G, Bronckers A L J J. Bone morphogenetic protein-7 (osteogenic protein-1, OP-1) and tooth development. J Dent Res. 1998;77:545–554. doi: 10.1177/00220345980770040701. [DOI] [PubMed] [Google Scholar]
- 20.Hermoso M, Saez J C, Villalon M. Identification of gap junctions in the oviduct and regulation of connexins during development and by sexual hormones. Eur J Cell Biol. 1997;74:1–9. [PubMed] [Google Scholar]
- 21.Heryanto B, Yoshimura Y, Tamura T, Okamoto T. Involvement of apoptosis and lysosomal hydrolase activity in the oviducal regression during induced molting in chickens: a cytochemical study for end labeling of fragmented DNA and acid phosphatase. Poultry Sci. 1997;76:67–72. doi: 10.1093/ps/76.1.67. [DOI] [PubMed] [Google Scholar]
- 22.Hogan B L M. Bone morphogenetic proteins: multifunctional regulators of vertebrate development. Genes Dev. 1996;10:1580–1594. doi: 10.1101/gad.10.13.1580. [DOI] [PubMed] [Google Scholar]
- 23.Houston B, Thorpe B H, Burt D W. Molecular cloning and expression of bone morphogenetic protein-7 in the chick epiphyseal growth plate. J Mol Endocrinol. 1994;13:289–301. doi: 10.1677/jme.0.0130289. [DOI] [PubMed] [Google Scholar]
- 24.Huang D C, Cory S, Strasser A. Bcl-2, Bcl-xL and adenovirus protein E1B19kD are functionally equivalent in their ability to inhibit cell death. Oncogene. 1997;14:405–414. doi: 10.1038/sj.onc.1200848. [DOI] [PubMed] [Google Scholar]
- 25.Huet-Hudson Y M, Chakraborty C, De S K, Suzuki Y, Andrews G K, Dey S K. Estrogen regulates the synthesis of epidermal growth factor in mouse uterine epithelial cells. Mol Endocrinol. 1990;4:510–523. doi: 10.1210/mend-4-3-510. [DOI] [PubMed] [Google Scholar]
- 26.Ignar-Trowbridge D M, Pimentel M, Teng C T, Korach K S, McLachlan J A. Cross talk between peptide growth factor and estrogen receptor signaling systems. Environ Health Perspect. 1995;103:35–38. doi: 10.1289/ehp.95103s735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jaffe R C, Arias E B, O'Day-Bowman M B, Donnelly K M, Mavrogianis P A, Verhage H G. Regional distribution and hormone control of estrogen-dependent oviduct-specific glycoprotein messenger ribonucleic acid in the baboon (Papio anubis) Biol Reprod. 1996;55:421–426. doi: 10.1095/biolreprod55.2.421. [DOI] [PubMed] [Google Scholar]
- 28.Jena N, Martin-Seisdedos C, McCue P, Croce C M. BMP7 null mutation in mice: developmental defects in skeleton, kidney, and eye. Exp Cell Res. 1997;230:28–37. doi: 10.1006/excr.1996.3411. [DOI] [PubMed] [Google Scholar]
- 29.Kepa J K, Neely C I, Jacobsen B M, Bruder J M, McDonnell D P, Leslie K K, Wierman M E. Estrogen receptor mediated repression of rat gonadotropin releasing hormone (GnRH) promoter activity in hypothalamic cells. Endocr Rev. 1994;2:1–10. [Google Scholar]
- 30.Keri R A, Andesen B, Kennedy G C, Hamernik D L, Clay C M, Brace A D, Nett T M, Notides A C, Nilson J H. Estradiol inhibits transcription of the human glycoprotein hormone α-subunit despite the absence of a high affinity binding site for estrogen receptor. Mol Endocrinol. 1991;5:725–733. doi: 10.1210/mend-5-5-725. [DOI] [PubMed] [Google Scholar]
- 31.Kurebayashi S, Miyashita Y, Hirose T, Kasayama S, Akira S, Kishimoto T. Characterization of mechanisms of interleukin-6 repression by estrogen receptor. J Steroid Biochem Mol Biol. 1997;60:11–17. doi: 10.1016/s0960-0760(96)00175-6. [DOI] [PubMed] [Google Scholar]
- 32.Kyprianou N, Isaacs J T. Activation of programmed cell death in the rat ventral prostate after castration. Endocrinology. 1988;122:552–562. doi: 10.1210/endo-122-2-552. [DOI] [PubMed] [Google Scholar]
- 33.Lincz L F. Deciphering the apoptotic pathway: all roads lead to death. Immunol Cell Biol. 1998;76:1–19. doi: 10.1046/j.1440-1711.1998.00712.x. [DOI] [PubMed] [Google Scholar]
- 34.Llen R T, Hunter W J, 3rd, Agrawal D K. Morphological and biochemical characterization and analysis of apoptosis. J Pharmacol Toxicol Methods. 1997;37:215–228. doi: 10.1016/s1056-8719(97)00033-6. [DOI] [PubMed] [Google Scholar]
- 35.Loo D T, Rillema J R. Measurement of cell death. Methods Cell Biol. 1998;57:251–264. doi: 10.1016/s0091-679x(08)61583-6. [DOI] [PubMed] [Google Scholar]
- 36.Luo G, Hofmann C, Bronckers A L J J, Sohocki M, Bradley A, Karsenty G. BMP-7 in an inducer of nephrogenesis, and is also required for eye development and skeletal patterning. Genes Dev. 1995;9:2808–2820. doi: 10.1101/gad.9.22.2808. [DOI] [PubMed] [Google Scholar]
- 37.Macias D, Ganan Y, Sampath T K, Piedra M E, Ros M A, Hurle J M. Role of BMP2 and OP-1 (BMP-7) in programmed cell death and skeletogenesis during chick limb development. Development. 1997;124:1109–1117. doi: 10.1242/dev.124.6.1109. [DOI] [PubMed] [Google Scholar]
- 38.Macias-Silva M, Hoodless P A, Tang S J, Buchwald M, Wrana J L. Specific activation of Smad1 signaling pathways by the BMP7 type I receptor, ALK2. J Biol Chem. 1998;273:25628–25636. doi: 10.1074/jbc.273.40.25628. [DOI] [PubMed] [Google Scholar]
- 39.Malayer J R, Gorski J. An integrated model of estrogen receptor action. Domest Anim Endocrinol. 1993;10:159–177. doi: 10.1016/0739-7240(93)90021-3. [DOI] [PubMed] [Google Scholar]
- 40.Malayer J R, Woods V M. Expression of estrogen receptor and maintenance of hormone-responsive phenotype in bovine fetal uterine cells. Domest Anim Endocrinol. 1998;15:141–154. doi: 10.1016/s0739-7240(98)00002-2. [DOI] [PubMed] [Google Scholar]
- 41.McLachlan J A, Nelson K G, Takahashi T, Bossert N L, Newbold R R, Korach K S. Estrogens and growth factors in the development, growth, and function of the female reproductive tract. New York, N.Y: Springer-Verlag; 1991. [Google Scholar]
- 42.Medema J P, Scaffidi C, Kischkel F C, Shevchenko A, Mann M, Krammer P H, Peter M E. FLICE is activated by association with the CD95 death-inducing signaling complex (DISC) EMBO J. 1997;16:2794–2804. doi: 10.1093/emboj/16.10.2794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Miller C D, Miller W L. Transcriptional repression of the ovine follicle-stimulating hormone-β gene by 17β-estradiol. Endocrinology. 1996;137:3437–3446. doi: 10.1210/endo.137.8.8754772. [DOI] [PubMed] [Google Scholar]
- 44.Murphy L J, Murphy L C, Friesen H G. Estrogen induces insulin-like growth factor-I expression in the rat uterus. Mol Endocrinol. 1987;1:445–450. doi: 10.1210/mend-1-7-445. [DOI] [PubMed] [Google Scholar]
- 45.Nelson K G, Takahashi T, Lee D C, Luetteke N C, Bossert N L, Ross K, Eitzman B E, McLachlan J A. Transforming growth factor-α is a potential mediator of estrogen action in the mouse uterus. Endocrinology. 1992;131:1657–1664. doi: 10.1210/endo.131.4.1396310. [DOI] [PubMed] [Google Scholar]
- 46.Newmeyer D D, Farschon D M, Reed J C. Cell-free apoptosis in Xenopus egg extracts: inhibition by Bcl-2 and requirement for an organelle fraction enriched in mitochondria. Cell. 1994;79:353–364. doi: 10.1016/0092-8674(94)90203-8. [DOI] [PubMed] [Google Scholar]
- 47.Nordstrom L A, Dean D M, Sanders M M. A complex array of double-stranded and single-stranded DNA-binding proteins mediates induction of the ovalbumin gene by steroid hormones. J Biol Chem. 1993;268:13193–13202. [PubMed] [Google Scholar]
- 48.Nunez G, Benedict M A, Hu Y, Inohara N. Caspases: the proteases of the apoptotic pathway. Oncogene. 1998;17:3237–3245. doi: 10.1038/sj.onc.1202581. [DOI] [PubMed] [Google Scholar]
- 49.Ozkaynak E, Jin D F, Jelic M, Vukicevic S, Oppermann H. Osteogenic protein-1 mRNA in the uterine endometrium. Biochem Biophys Res Commun. 1997;234:242–246. doi: 10.1006/bbrc.1997.6624. [DOI] [PubMed] [Google Scholar]
- 50.Ozkaynak E, Rueger D C, Drier E A, Corbett C, Ridge R J, Sampath T K, Oppermann H. OP-1 cDNA encodes an osteogenic protein in the TGF-β family. EMBO J. 1990;9:2085–2093. doi: 10.1002/j.1460-2075.1990.tb07376.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ozkaynak E, Schnegelsberg P N J, Oppermann H. Murine osteogenic protein (OP-1): high levels of mRNA in kidney. Biochem Biophys Res Commun. 1991;179:116–123. doi: 10.1016/0006-291x(91)91342-a. [DOI] [PubMed] [Google Scholar]
- 52.Porter A G, Janicke R U. Emerging roles of caspase-3 in apoptosis. Cell Death Differ. 1999;6:99–104. doi: 10.1038/sj.cdd.4400476. [DOI] [PubMed] [Google Scholar]
- 53.Ray P, Ghosh S K, Zhang D-H, Ray A. Repression of interleukin-6 gene expression by 17β-estradiol: inhibition of the DNA-binding activity of the transcription factors NF-IL6 and NF-κB by the estrogen receptor. FEBS Lett. 1997;409:79–85. doi: 10.1016/s0014-5793(97)00487-0. [DOI] [PubMed] [Google Scholar]
- 54.Reinhardt K C, Dubey R K, Mummery C L, van Rooijen M, Keller P J, Marinella R. Synthesis and regulation of leukaemia inhibitory factor in cultured bovine oviduct cells by hormones. Mol Human Reprod. 1998;4:301–308. doi: 10.1093/molehr/4.3.301. [DOI] [PubMed] [Google Scholar]
- 55.Reiss M, Barcellos-Hoff M H. Transforming growth factor-β in breast cancer: a working hypothesis. Breast Cancer Res Treat. 1997;45:81–95. doi: 10.1023/a:1005865812918. [DOI] [PubMed] [Google Scholar]
- 56.Rochwerger L, Buchwald M. Stimulation of the cystic fibrosis transmembrane regulator expression by estrogen in vivo. Endocrinology. 1993;133:921–930. doi: 10.1210/endo.133.2.7688293. [DOI] [PubMed] [Google Scholar]
- 57.Russo M A, Guerriero V, Means A R. Hormonal regulation of a chicken oviduct messenger ribonucleic acid that shares a common domain with gizzard myosin light chain kinase. Mol Endocrinol. 1987;1:60–67. doi: 10.1210/mend-1-1-60. [DOI] [PubMed] [Google Scholar]
- 58.Sampath T K, Coughlin J E, Whetstone R M, Banach D, Corbett C, Ridge R J, Ozkaynak E, Oppermann H, Rueger R C. Bovine osteogenic protein is composed of dimers of OP-1 and BMP-2A, two members of the transforming growth factor-β superfamily. J Biol Chem. 1990;265:13198–13205. [PubMed] [Google Scholar]
- 59.Sampath T K, Maliakal J C, Hauschka P V, Jones W K, Sasak H, Tucker R F, White K H, Coughlin J E, Tucker M M, Pang R H L, Corbett C, Ozkaynak E, Oppermann H, Rueger D C. Recombinant human osteogenic protein-1 (hOP-1) induces new bone formation in vivo with a specific activity comparable with natural bovine osteogenic protein and stimulates osteoblast proliferation and differentiation in vitro. J Biol Chem. 1992;267:20352–20362. [PubMed] [Google Scholar]
- 60.Sampath T K, Reddi A H. Dissociative extraction and reconstitution of extracellular matrix components involved in local bone differentiation. Proc Natl Acad Sci USA. 1981;78:7599–7603. doi: 10.1073/pnas.78.12.7599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Sanders M M, McKnight G S. Chicken egg white genes: multihormonal regulation in a primary cell culture system. Endocrinology. 1985;116:398–405. doi: 10.1210/endo-116-1-398. [DOI] [PubMed] [Google Scholar]
- 62.Sanders M M, McKnight G S. Positive and negative regulatory elements control the steroid-responsive ovalbumin promoter. Biochemistry. 1988;27:6550–6557. doi: 10.1021/bi00417a053. [DOI] [PubMed] [Google Scholar]
- 63.Schell D L, Mavrogianis P A, Fazleabas A T, Verhage H G. Epidermal growth factor, transforming growth factor-alpha, and epidermal growth factor receptor localization in the baboon (Papio anubis) oviduct during steroid treatment and the menstrual cycle. J Soc Gynecol Investig. 1994;1:269–276. doi: 10.1177/107155769400100405. [DOI] [PubMed] [Google Scholar]
- 64.Shepherd J H, Mulvihill E R, Thomas P S, Palmiter R D. Commitment of chick oviduct tubular gland cells to produce ovalbumin mRNA during hormonal withdrawal and restimulation. J Cell Biol. 1980;87:142–151. doi: 10.1083/jcb.87.1.142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Shoham Z, Schachter M. Estrogen biosynthesis--regulation, action, remote effects, and value of monitoring in ovarian stimulation cycles. Fertility Sterility. 1996;65:687–701. doi: 10.1016/s0015-0282(16)58197-7. [DOI] [PubMed] [Google Scholar]
- 66.Smale S T. Transcription initiation from TATA-less promoters within eukaryotic protein-coding genes. Biochim Biophys Acta. 1997;1351:73–88. doi: 10.1016/s0167-4781(96)00206-0. [DOI] [PubMed] [Google Scholar]
- 67.Smith C L. Cross-talk between peptide growth factor and estrogen signaling pathways. Biol Reprod. 1998;58:627–632. doi: 10.1095/biolreprod58.3.627. [DOI] [PubMed] [Google Scholar]
- 68.Takahashi T, Eitzman B, Bossert N L, Walmer D, Sparrow K, Flanders K C, McLachlan J, Nelson K G. Transforming growth factors β1, β2, and β3 messenger RNA and protein expression in mouse uterus and vagina during estrogen-induced growth: a comparison to other estrogen-regulated genes. Cell Growth Differ. 1994;5:919–935. [PubMed] [Google Scholar]
- 69.Tanabe Y, Ogawa T, Nakamura T. The effect of short term starvation on pituitary and plasma LH, plasma estradiol and progesterone, and on pituitary response to LH-RH in the laying hens. Gen Comp Endocrinol. 1981;43:392–398. doi: 10.1016/0016-6480(81)90299-9. [DOI] [PubMed] [Google Scholar]
- 70.Tang S G, Hoodless P A, Lu Z, Breitman M L, McInnes R R, Wrana J L, Buchwald M. The Tlx-2 homeobox gene is a downstream target of BMP signaling and is required for mouse mesoderm development. Development. 1998;125:1877–1887. doi: 10.1242/dev.125.10.1877. [DOI] [PubMed] [Google Scholar]
- 71.Thomas R, Anderson W A, Raman V, Reddi A H. Androgen-dependent gene expression of bone morphogenetic protein 7 in mouse prostate. Prostate. 1998;37:236–245. doi: 10.1002/(sici)1097-0045(19981201)37:4<236::aid-pros5>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- 72.Tomkinson A, Reeve J, Shaw R W, Noble B S. The death of osteocytes via apoptosis accompanies estrogen withdrawal in human bone. J Clin Endocrinol Methods. 1997;82:3128–3135. doi: 10.1210/jcem.82.9.4200. [DOI] [PubMed] [Google Scholar]
- 73.Urist M R. Bone: formation by autoinduction. Science. 1965;150:893–899. doi: 10.1126/science.150.3698.893. [DOI] [PubMed] [Google Scholar]
- 74.Volk S W, Luvalle P, Leask T, LeBoy P S. A BMP responsive transcriptional region in the chicken type X collagen gene. J Bone Miner Res. 1998;13:1521–1529. doi: 10.1359/jbmr.1998.13.10.1521. [DOI] [PubMed] [Google Scholar]
- 75.Wada K, Nomura S, Morii E, Kitamura Y, Nishizawa Y, Miyake A, Terada N. Changes in levels of mRNAs of transforming growth factor (TGF)-β1, -β2, -β3, TGF-β type II receptor and sulfated glycoprotein-2 during apoptosis of mouse uterine epithelium. J Steroid Biochem Mol Biol. 1996;59:367–375. doi: 10.1016/s0960-0760(96)00139-2. [DOI] [PubMed] [Google Scholar]
- 76.Wang S, Krinks M, Kleinwaks L, Moos J M. A novel Xenopus homologue of bone morphogenetic protein-7 (BMP-7) Genes Function. 1997;1:259–271. doi: 10.1046/j.1365-4624.1997.00023.x. [DOI] [PubMed] [Google Scholar]