Abstract
Introduction
The role of smoking in risk of death among patients with COVID-19 remains unclear. We examined the association between in-hospital mortality from COVID-19 and smoking status and other factors in the United States Veterans Health Administration (VHA).
Methods
This is an observational, retrospective cohort study using the VHA COVID-19 shared data resources for February 1 to September 11, 2020. Veterans admitted to the hospital who tested positive for SARS-CoV-2 and hospitalized by VHA were grouped into Never (as reference, NS), Former (FS), and Current smokers (CS). The main outcome was in-hospital mortality. Control factors were the most important variables (among all available) determined through a cascade of machine learning. We reported adjusted odds ratios (aOR) and 95% confidence intervals (95%CI) from logistic regression models, imputing missing smoking status in our primary analysis.
Results
Out of 8 667 996 VHA enrollees, 505 143 were tested for SARS-CoV-2 (NS = 191 143; FS = 240 336; CS = 117 706; Unknown = 45 533). The aOR of in-hospital mortality was 1.16 (95%CI 1.01, 1.32) for FS vs. NS and 0.97 (95%CI 0.78, 1.22; p > .05) for CS vs. NS with imputed smoking status. Among other factors, famotidine and nonsteroidal anti-inflammatory drugs (NSAID) use before hospitalization were associated with lower risk while diabetes with complications, kidney disease, obesity, and advanced age were associated with higher risk of in-hospital mortality.
Conclusions
In patients admitted to the hospital with SARS-CoV-2 infection, our data demonstrate that FS are at higher risk of in-hospital mortality than NS. However, this pattern was not seen among CS highlighting the need for more granular analysis with high-quality smoking status data to further clarify our understanding of smoking risk and COVID-19-related mortality. Presence of comorbidities and advanced age were also associated with increased risk of in-hospital mortality.
Implications
Veterans who were former smokers were at higher risk of in-hospital mortality compared to never smokers. Current smokers and never smokers were at similar risk of in-hospital mortality. The use of famotidine and nonsteroidal anti-inflammatory drugs (NSAIDs) before hospitalization were associated with lower risk while uncontrolled diabetes mellitus, advanced age, kidney disease, and obesity were associated with higher risk of in-hospital mortality.
Background
The impact of cigarette smoking on the risk of death or serious outcomes among patients with COVID-19 remains unclear.1 Current and former smoking status are both associated with more frequent and severe upper and lower respiratory illnesses, and associated complications.2,3 One might expect that smoking would have a similar negative impact on health outcomes in COVID-19 infected patients, but the evidence is unclear and contradictory. For example, smoking is associated with lower BMI,4 while high BMI is a risk factor for more severe illness from COVID-19 infection and is an established major contributor to COVID-19 mortality.5 Furthermore, the effects of smoking on gene expression in current smokers is complex and incompletely understood,6,7 the fact that may also impact the course of COVID-19 infection. For example, the effects of nicotine on upregulation of the angiotensin-converting enzyme (ACE-2) receptors on COVID-19 may influence the course of illness.8,9
A systematic review of the observed effects of smoking on COVID-19-related outcomes from March 2020 identified five studies from China with small sample sizes (N ranged from 41–1099).10 The combined analysis showed no differences between current smokers and other groups while the constituent study with the largest sample size (N = 1099) estimated higher relative risk (RR) of severe symptoms, ICU admission, and mortality among the current smoker group compared to others.10 A subsequent meta-analysis of 12 different studies (11 from China and one from Netherlands) demonstrated that smoking was associated with a 1.54-fold higher risk of a severe outcome among smokers vs. nonsmoking patients infected with COVID-19. A more recent systematic review which included 47 studies (including studies previously reviewed) showed that current smokers had an increased risk of severe COVID‐19, such as respiratory failure necessitating mechanical ventilation compared to nonsmokers.11 In contrast, a letter to the editor after reviewing 11 articles reported that prevalence of hospitalization among smokers is low6 due to possible interaction between SARS-CoV-2 and the nicotinic cholinergic receptor.7,12 A recent study showed a significant association between former smoker status and hospitalization, although the statistical difference faded after adjusting the model with confounding factors.13 Limitations of the above studies include lack of adjustment for confounding factors, imprecise and incomplete documentation of smoking status, and small sample sizes.
There is an urgency to understand better the associations between smoking status and adverse outcomes due to COVID-19 infection to guide public health messaging and inform individual decisions about COVID risk mitigation measures. While the extant literature supports an association between current smoking and increased risk of complications of COVID-19, several questions remain, such as the relationship between former smoking status and COVID-19 outcomes. To examine such an association, we utilized the US Department of Veterans Affairs (VHA) COVID-19 Shared Data Resource. VHA is a national integrated healthcare provider that operates more than 170 medical centers and more than 1000 outpatient clinics across the US It provides medical care and related social services for more than nine million enrolled Veterans with robust racial and ethnic diversity generally representative of the military population14 The Shared Data Resource provides a large, curated data source from VHA national electronic medical records (EMR) for answering important COVID-19 related questions..
The main objective of the study report was to test the association between smoking status and in-hospital mortality. Our approach also allowed us to identify demographic, clinical, pre-existing condition, and prehospitalization medication factors associated with in-hospital mortality of hospitalized patients with COVID-19.
Methods
Study Database, Design, and Population
We performed an observational, retrospective cohort study using the VHA’s Corporate Data Warehouse (CDW), which is a relational database that aggregates patient data from all VHA facilities from 1999 to present.15 We used the COVID-19 Shared Data Resource that contains information related to COVID-19 treated within VHA.16 It encompasses a wide range of information of SARS-CoV-2 tested patients (e.g., timing and nature of test results, pharmacological and nonpharmacological interventions, patient outcomes, and pre-existing conditions and medication).17
For the present study, we included all veterans who were active users of VHA services and excluded patients who did not have at least one outpatient visit or one inpatient stay during the period from January 2018 to December 2019. From this population, we identified all VHA veteran patients who underwent SARS-CoV-2 testing at a VHA facility or outside lab between February 1 and September 11, 2020. A patient was considered a case if he/she had a positive reverse transcriptase polymerase chain reaction SARS-CoV-2 test. We then defined COVID-19 related hospitalization to be the admission that occurred within 7 days after the SARS-CoV-2 test date, or within 15 days prior to the test date, if the test coincided with inpatient status. The index date was defined as the date of the first positive SARS-CoV-2 test or the hospital admission date preceding the positive test. Patients were followed for a minimum of 30 days unless death or hospital discharge occurred (dataset locked on October 11, 2020). The study was approved by the institutional review board of Baylor College of Medicine and the Research and Development Committee of the Michael E. DeBakey VA Medical Center.
Key Study Variables
Our key variable was smoking status (never, former, current, and unknown) gathered from VHA Electronic Medical Records (EMR) Health Factors (HF) dataset. HF table contains data that are derived from clinical reminders within the EMR and include information on a variety of clinically relevant risk behaviors, such as smoking status. Smoking status in the COVID-19 Shared Data Resource included data from the 2 years prior to index date. Health Factor data were mapped to distinct smoking categories (current, former, never) based on the most recent health factor data if the most recent indicated current or former smoker. If the most recent was never, prior data was used to confirm and resolve inconsistencies. For example, a most recent never smoker who had a prior former smoker health factor was defined as former smoker. A most recent never smoker with a past current smoker was defined as former. Patients with no smoking health factors were defined as unknown. Previous studies showed agreement between EMR records and self-reported smoking from a survey with reported kappa statistics ranging from 0.66 to 0.7418–20
Our primary outcome for the statistical modeling was in-hospital mortality. Mortality was assessed from the patient treatment file reflecting death prior to hospital discharge.21
Imputation of Smoking Status: To address the uncertainty related to unknown smoking status, we imputed current, former, or never smoking status from other variables in the dataset using Multiple Imputations by Chained Equations (MICE).22,23 MICE is an empirically and theoretically supported tool to address missing data.24 To increase the generalizability of the imputation model, we included all available variables. We performed five iterations with five levels of imputation in each iteration.
Statistical Analysis
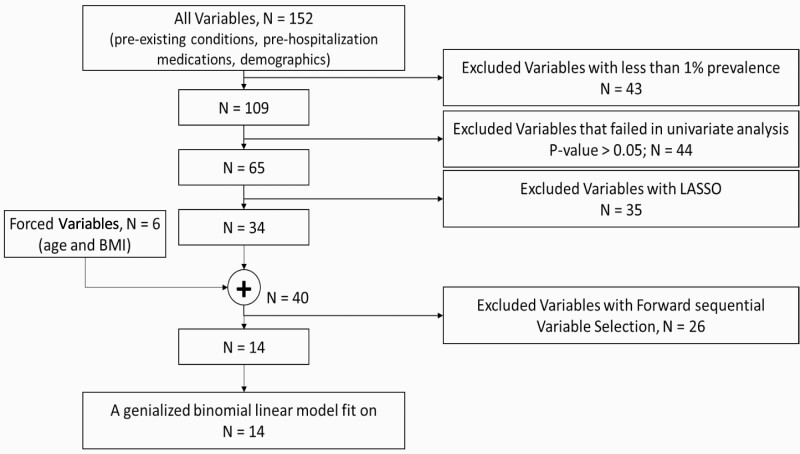
Potential confounders of COVID-19 in-hospital mortality were abstracted from the VA COVID-19 Shared Data Resources.25 These included demographics, pre-existing conditions, and prehospitalization medications. The Elixhauser comorbidity index and Charlson Comorbidity index were determined using International Classification of Diseases (ICD)-10 codes from patients’ encounters (inpatient and outpatient) during the 2 years prior to index date.26,27 The prescribed medications were also reported for the two years prior to index date. To select the most important predictors, we used machine learning feature (or variable) selection techniques (Figure 1) with four steps. The dependent variable for the most important variable selection process was in-hospital mortality. Age and body mass index (BMI) were converted to categorical variables; age was mapped to seven variables (<30, ≥30 <40, ≥40–<50, ≥50 <65, ≥65 <75, ≥75 <85, and ≥85 years) and BMI was mapped to five variables (<18.5, ≥18.5 <25, ≥25 <30, ≥30 <40, and ≥ 40 kg/m2).
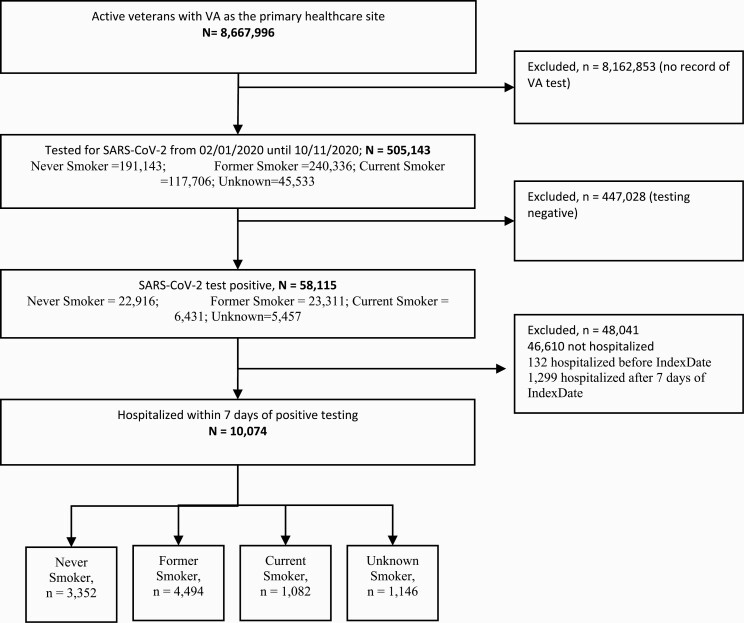
Figure 1.
Strobe diagram.
We utilized a cascade of variable selection approach to identify the most salient features. These steps were performed twice: a) without imputing “unknown smoking” and b) with imputing “unknown smoking” status into three other groups, i.e., “Never smoker,” “Former smoker,” and “Current smoker.” Step 1, prevalence: any variables with prevalence less than 1%, were removed. Step 2, univariate filter: we excluded variables that were not statistically associated with in-hospital mortality at p < .05. Step 3, least absolute shrinkage and selection operator (LASSO): we used LASSO with 10-fold cross-validation to select the most important candidate variables. The LASSO is a regression analysis method that performs both variable selection (filter method) and regularization (wrapper method) to enhance prediction accuracy and interpretability of the statistical model.28 Step 4, sequential forward variable selection: To avoid inter-variable correlations, we used sequential forward variable selection. Before we applied the sequential method, we enriched the list of variables from step 3 by forcing BMI and age variables into the model.
To understand the tendency of the most important variables to raise or lower risk of in-hospital mortality, we applied a binary logistic regression. The positive coefficients from binary logistic regression indicate association with increased risk and negative coefficients indicate association with decreased risk of in-hospital mortality. Odds ratios and confidence intervals were obtained by exponentiating the beta coefficients and confidence intervals around these coefficients.
To characterize the cohort, continuous variables are presented as mean with standard deviation, and categorical variables as number and percentage. We used logistic regression models to test the association between smoking status (current, former, and never) and in-hospital mortality on imputed and unimputed datasets. Odds ratios (OR) with 95% confidence intervals (95% CI) were reported. Each model was adjusted using the most important variables from the variable selection method described above. All statistical analyses were performed using SPSS, version 26 (SPSS Inc, Chicago, IL), the machine learning algorithms were performed using MATLAB (MathWorks Inc., Natick, MA), and MICE imputation was performed using R (MICE-package 3.12.0).
Results
Cohort and Key Variables
Among 8 667 996 active veterans, 505 143 were tested for SARS-CoV-2 from February to September 11, 2020. Smoking status was Never Smoker (n = 191 143; 37.8%), Former Smoker (n = 240 336), Current Smoker (n =117 706; 47.6%), and Unknown (n = 45 533; 23.3%). Among those tested, 58 115 (11.5%) were positive for SARS-CoV-2 (Never Smoker, n = 22,916 [39.4%]; Former Smoker, n = 23,311 [40.1%]; Current Smoker, n = 6431 [11.1%]; Unknown, n = 5457 [9.4%]). Among those who tested positive, 10 074 (17.3%) were hospitalized due to COVID-19 (Never Smoker, n = 3352 [33.3%]; Former Smoker, n = 4494 [44.6%]; Current Smoker, n = 1082 [10.7%]; Unknown, n = 1146 [11.4%]) (Figure 2). The mean (standard deviation (SD)) age of former smokers (70.6 (12.2) years) was higher than never smokers (65.6 (14.8) years) and current smokers (64.3 (12.8) years). The mean (SD) of the Charlson Comorbidity Index in former smokers (3.5 (2.8)) was higher than never smokers (2.8 (2.6)) and current smokers (3.4(2.9)). We observed 1,299 deaths with the lowest proportion observed in current smokers (9.0% (n = 97)) compared to never smokers (11.8% (n = 362)) and former smokers (14.8% (n = 665)) (Table 1, Supplementary Material 4).
Figure 2.
Cascade of machine learning selection of most important variables.
Table 1.
Characteristics of 10 074 Veterans Hospitalized With SARS-CoV-2 Positive Tests
| Variables | Never smoker | Former smoker | Current smoker | Unknown smoker |
|---|---|---|---|---|
| Male sex, N(%)* | 3068(91.5) | 4323(96.2) | 1043(96.4) | 1107(96.6) |
| BMI, M(SD) | 30.8(7.1) | 29.6(7.0) | 28.0(7.0) | 28.2(6.7) |
| BMI < 18.5, N(%)* | 56(1.7) | 140(3.1) | 57(5.3) | 47(4.1) |
| BMI 18.5–25, N(%)* | 610(18.2) | 1042(23.2) | 348(32.2) | 324(28.3) |
| BMI 25–30, N(%)* | 1019(30.4) | 1385(30.8) | 334(30.9) | 360(31.4) |
| BMI 30–40, N(%)* | 1340(40.0) | 1568(34.9) | 278(25.7) | 305(26.6) |
| BMI >40, N(%)* | 320(9.5) | 358(8.0) | 64(5.9) | 61(5.3) |
| Race, N(%) | ||||
| White, N(%)* | 1330(39.7) | 2232(49.7) | 509(47.0) | 552(48.2) |
| Black, N(%)* | 1405(41.9) | 1595(35.5) | 442(40.9) | 315(27.5) |
| Hispanic, N(%)* | 390(11.6) | 403(9.0) | 79(7.3) | 114(9.9) |
| Asian, N(%) | 34(1.0) | 30(0.7) | 6(0.6) | 12(1.0) |
| American Indian, N(%) | 16(0.5) | 35(0.8) | 9(0.8) | 14(1.2) |
| Unknown, N(%)* | 177(5.3) | 199(4.4) | 37(3.4) | 139(12.1) |
| age, M(SD) | 65.6(14.8) | 70.6(12.2) | 64.3(12.8) | 70.9(16.3) |
| age 18–30, N(%)* | 39(1.2) | 15(0.3) | 14(1.3) | 15(1.3) |
| age 30–40, N(%)* | 149(4.4) | 88(2.0) | 64(5.9) | 52(4.5) |
| age 40–50, N(%)* | 290(8.7) | 145(3.2) | 48(4.4) | 67(5.8) |
| age 50–65, N(%)* | 989(29.5) | 933(20.8) | 335(31.0) | 199(17.4) |
| age 65–75, N(%)* | 1025(30.6) | 1762(39.2) | 430(39.7) | 297(25.9) |
| age 75–80, N(%)* | 513(15.3) | 992(22.1) | 152(14.0) | 255(22.3) |
| age > 85, N(%)* | 347(10.4) | 559(12.4) | 39(3.6) | 261(22.8) |
| Pre-existing conditions † | ||||
| Acute kidney failure, N(%)* | 127(3.8) | 261(5.8) | 74(6.8) | 43(3.8) |
| Acute cardiac injury, N(%)* | 126(3.8) | 261(5.8) | 74(6.8) | 43(3.8) |
| Asthma, N(%)* | 266(7.9) | 309(6.9) | 72(6.7) | 36(3.1) |
| Coronary atherosclerosis and other heart disease, N(%)* | 850(25.4) | 1617(36.0) | 340(31.4) | 183(16.0) |
| Congestive heart failure, N(%)* | 855(25.5) | 1395(31.0) | 310(28.7) | 147(12.8) |
| Chronic kidney disease, N(%)* | 228(6.8) | 345(7.7) | 89(8.2) | 36(3.1) |
| Chronic obstructive pulmonary disease, N(%)* | 483(14.4) | 926(20.6) | 208(19.2) | 119(10.4) |
| Cancer, N(%)* | 53(1.6) | 100(2.2) | 23(2.1) | 10(0.9) |
| Cerebrovascular disease, N(%)* | 852(25.4) | 1417(31.5) | 263(24.3) | 176(15.4) |
| Diabetes type 1, N(%)* | 126(3.8) | 165(3.7) | 41(3.8) | 17(1.5) |
| Diabetes type 2, N(%)* | 1688(50.4) | 2401(53.4) | 489(45.2) | 308(26.9) |
| Diabetes with complications, N(%)* | 1302(38.8) | 1934(43.0) | 386(35.7) | 251(21.9) |
| Nonalcohol drug dependence, N(%)* | 135(4.0) | 251(5.6) | 241(22.3) | 24(2.1) |
| Emphysema, N(%)* | 47(1.4) | 168(3.7) | 47(4.3) | 12(1.0) |
| Liver disease, N(%)* | 287(8.6) | 413(9.2) | 130(12.0) | 50(4.4) |
| Lower respiratory infection, N(%)* | 472(14.1) | 629(14.0) | 141(13.0) | 68(5.9) |
† Pre-existing conditions reported for past two years.16
* p-value < .05
After imputation of smoking behavior, the number of cases increased in the Never Smoker category from 3352 to 3789 (+437), Former Smokers from 4494 to 5090 (+596), and Current Smokers from 1082 to 1197 (+115). The number of deaths increased slightly in all groups: Never Smoker mortality increased from 362 to 430 (+68), Former Smoker mortality increased from 665 to 756 (+91), and Current Smoker mortality increased from 97 to 113 (+16). The in-hospital mortality rate, defined as number of deaths per group, increased slightly in Former Smokers from 14.8% to 14.9% and Current Smokers from 9.0% to 9.4% while it slightly decreased slightly in Never Smokers from 11.8% to 11.3%.
Predictors of In-Hospital Mortality
Out of 152 variables (demographics, pre-existing conditions, and prehospitalization medications), 14 variables remained after the variable selection process (Figure 1). The details of selected variables at each step of the selection process are reported in Supplementary Material 2. Among the remaining 14 variables, six variables were demographic or clinical (age ≥50 <65, ≥65 <75, ≥75 <85, and ≥85 years, BMI >40, and former smoker), six variables were pre-existing conditions (acute kidney failure, chronic kidney disease, diabetes with complications, emphysema, lower respiratory infection, and drug dependency), and two variables were prehospitalization medications (famotidine and nonsteroidal anti-inflammatory drugs [NSAIDs]). Utilizing the raw, unimputed data produced the same list of variables after the variable selection process (Supplementary Material 3).
Out of the 14 variables, four were protective factors (famotidine, NSAIDs, nonalcohol drug dependency, and lower respiratory infection,) and the rest (Former Smoker, age 50–65, age 65–75, age 75–80, age ≥ 85, BMI >40, Acute Kidney Failure, Chronic Kidney Disease, Diabetes with Complications, Emphysema) were risk factors for in-hospital mortality (Supplementary Material 2). Among the confounding factors, age was prominently associated with probability of in-hospital death: 50–60 years, OR = 5.15 (95%CI 2.77, 9.57); 65–75 years, OR = 11.07 (95%CI 6.04, 20.30); 75–85 years, OR = 16.11 (95%CI 8.75, 29.68), and age ≥ 85 years, OR = 33.67 (95%CI 18.26, 62.10). Among the categorized BMI (i.e., BMI < 18.5, BMI≥ 18.5 & <25, BMI≥25 & <30, BMI≥30 & <40, BMI≥40), only BMI ≥ 40 kg/m2 was selected as a most important variable. The univariate analysis selected smoking status as a significant factor for predicting mortality, but in the final step of variable selection, only Former Smoker status was kept as a most important factor. The initial variable list included several factors related to diabetes: Diabetes Any, Diabetes Other, Diabetes Type 1, Diabetes Type 2, Diabetes with Complications, and Diabetes Without Complications. In the univariate analysis, all of them except Type 1 Diabetes remained in the model as a risk factor, but by the final step, only Diabetes with Complications was selected as a risk factor for in-hospital mortality with an OR of 1.22 (95%CI: 1.08, 1.39) (Supplementary Material 2). From the other preconditions list of variables, both Acute Kidney Failure (OR = 1.24; 95%CI 1.06, 1.45) and Chronic Kidney Disease (OR = 1.20; 95% CI 1.04, 1.38) were selected as most important risk factors. The univariant analysis selected Emphysema (Beta = 0.45), Chronic Obstructive Pulmonary Disease (COPD, Beta = 0.21) and Lower Respiratory Infection (Beta = –0.31), but in the final list of most important factors, only Emphysema (OR = 1.44; 95% CI 1.05, 1.99) was retained as a risk factor and Lower Respiratory Infection (OR = 0.81; 95%CI 0.67, 0.99) was retained as a protective factor. Among the prehospitalization medication variables, two prehospitalization medications were selected as most important protective factors: Famotidine (OR = 0.73; 95% CI 0.59, 0.92) and NSAID (OR = 0.81; 95% CI 0.71, 0.92). Racial-ethnicity factors were not selected as most important factors for predicting in-hospital mortality (Supplementary Material 2). Although the univariate analysis for variable selection included White (Beta = 0.21), Black (Beta = –0.15), and Hispanic (Beta = –0.24), these variables did not meet criteria for inclusion in later steps of the selection process. The machine learning step (LASSO) excluded hypertension from the final list of the most important variables.
COVID-19 In-hospital Mortality and Smoking Status
The imputed, unadjusted OR of in-hospital mortality was 1.36 (95% CI 1.20, 1.55) for Former Smoker vs. Never Smoker and 0.81 (95% CI 0.65, 1.01) for Current Smoker vs. Never Smoker (Supplementary Material 3). After adjusting for the most important variables with the imputed data, the adjusted OR of in-hospital mortality was 1.16 (95% CI 1.01, 1.32) for Former Smoker vs. Never Smoker and 0.98 (95% CI 0.78, 1.22) for Current Smoker vs. Never Smoker (Tables 2 and 3). Identical analyses of the unimputed data revealed essentially the same strength of associations and confidence intervals for the associations between in-hospital mortality and smoking status (Supplementary Material 3 & 4). Additionally, the association between smoking status and in-hospital mortality remained consistent after adjusting with each group of most important variables (demographics, prehospitalization conditions, and prehospitalization medications) (Supplementary Material 5).
Table 2.
Association Between Smoking Status and COVID-19 In-Hospital Mortality
| Odds ratio (95%Confidence intervals) | ||
|---|---|---|
| variables | Unadjusted | Adjusted† |
| Former smoker vs never smoker | 1.43(1.25, 1.64) | 1.18(1.02, 1.36) |
| Current smoker vs never smoker | 0.81(0.64, 1.03) | 0.96(0.75, 1.22) |
| Unknown smoker vs never smoker | 1.49(1.22, 1.80) | 1.09(0.89, 1.35) |
| Imputed unknown smoker to never, former, and current smoker | ||
| Former smoker vs never smoker | 1.36(1.20, 1.55) | 1.16(1.01, 1.32) |
| Current smoker vs never smoker | 0.81(0.65, 1.01) | 0.97(0.78, 1.22) |
† Adjusted by Age 50–65, Age 65–75, Age 75–85, Age ≥ 85, BMI ≥ 40, Former Smoker, acute kidney failure, chronic kidney disease, diabetes with complications, emphysema, lower respiratory infection, drug dependency, famotidine and NonSteroidal Anti-Inflammatory Drugs (NSAIDs).
Table 3.
Association Between Smoking Status and COVID-19 In-Hospital Mortality
| Unadjusted | Adjusted | |||
|---|---|---|---|---|
| Variables† | p | Or (95%CI) ‡ | p | Or (95%CI) ‡ |
| Never smoker | Reference | Reference | ||
| Former smoker | < .001 | 1.36(1.20, 1.55) | .030 | 1.16(1.01, 1.32) |
| Current smoker | .065 | 0.81(0.65, 1.01) | .826 | 0.97(0.78, 1.22) |
| Age 50–65 | < .001 | 5.15(2.77, 9.57) | ||
| Age 65–75 | < .001 | 11.07(6.04, 20.30) | ||
| Age 75–80 | < .001 | 16.11(8.75, 29.68) | ||
| Age > 85 | < .001 | 33.67(18.26, 62.10) | ||
| Bmi >40 | .008 | 1.39(1.09, 1.78) | ||
| Acute kidney failure | .008 | 1.24(1.06, 1.45) | ||
| Chronic kidney disease | .011 | 1.20(1.04, 1.38) | ||
| Diabetes with complications | .002 | 1.22(1.08, 1.39) | ||
| Nonalcohol drug dependence | .045 | 0.70(0.50, 0.99) | ||
| Emphysema | .026 | 1.44(1.05, 1.99) | ||
| Lower respiratory infection | .039 | 0.81(0.67, 0.99) | ||
| Famotidine | .006 | 0.73(0.59, 0.92) | ||
| Nonsteroidal anti-inflammatory drug | .002 | 0.81(0.71, 0.92) |
† Pre-existing conditions and prehospitalization medications are reported for past two years.
‡ OR (95%CI) = Odds Ratio and 95 percentage confidence intervals.
Discussion
In this analysis of a large, national, retrospective cohort of veterans cared for by the VHA health care system, we found that odds of in-hospital mortality among Former Smoker veterans infected with SARS-CoV-2 is higher than Never Smokers and Current Smokers.
The role of smoking on mortality due to COVID-19 infection has been unclear and controversial. A series of systematic reviews reported an association between smoking status (Former Smoker and Current Smoker) and adverse COVID-19 outcomes, but these reports were limited by small sample sizes, homogeneous populations, incomplete capture of smoking status, and other flaws.10,11,29 Our results perhaps indicate a limited or more nuanced role for smoking in risk of adverse outcomes. In our analysis, the adjusted odds ratio (aOR) of in-hospital mortality among former compared to never smokers was small, albeit statistically significant, indicating an elevated risk of mortality for former smokers. Also, we found a lower, but not statistically significant aOR of mortality among current smokers compared to never smokers. These findings were consistent in unadjusted and unimputed analyses, as well. Our findings add to the literature by highlighting the different risk of in-hospital mortality from COVID-19 among Former, Current, and Never Smokers using a large sample with well-curated data and more complete information on smoking status. As a possible explanation of our findings, we hypothesize that the ongoing exposure to oxidative stress and the resultant increased mucus secretion and neutrophil accumulation in the airways among active smokers may potentially protect them from severe COVID-19 outcomes. The ACE-2 receptor is the main receptor for entry of SARS-CoV-2 in the host cells.30 We and others have previously demonstrated that smokers (Former and Current Smokers) have higher expression of the ACE-2 receptor31 which is particularly upregulated in the goblet cells.31 Active smoking results in an increase the number of goblet cells and decrease ciliated cells resulting in the replacement of normal epithelium and mucous metaplasia.32 It is, therefore, possible that the decreased in ciliary cells and increased mucus secretion by goblet cells in active smokers have a protective effect in current smokers from complications of COVID-19 infection.1 Future studies should explore our hypothesis further.
To our knowledge, this is the first study to demonstrate that former smokers are at higher risk of in-hospital mortality from COVID-19. If corroborated in future studies, public health messaging about the increased risk of smoking with regard to death and complications from COVID-19 may need to be more nuanced. The CDC currently highlights “current or former cigarette smoker” status as a single risk factor.33 Further studies in organoid cultures might help refine the mechanisms by which tobacco exposure influences COVID-19 infection and why former smokers are at higher risk for death from COVID-19 compared to never or current smokers.
Among individual factors that increase risk for in-hospital mortality from COVID, we corroborate some previously reported results, but the sample sizes and quality of data available through the VHA provide clearer and more precise information about the effects than has been available. Of note, after adjusting for confounders such as BMI, we did not find that ethnicity was a significant predictor of in-hospital mortality (Supplement 2) consistent with previous reports.34 Among the most important predictors of in-hospital mortality, older adults were at disproportionate risk of mortality, likely reflecting disease severity, comorbidity burden, and frailty not captured in the structured EMR data. This finding is consistent with Centers for Disease Control and Prevention (CDC) reports.35,36 CDC reported that mortality increased with age and eight out of ten COVID-19 deaths reported were in adults aged ≥ 65 years.37–39 We observed that patients with obesity (BMI ≥ 40 kg/m2) are at higher risk of mortality from COVID-19 infection which is also consistent with previous publications.40,41 Our study also showed that diabetes with complications, typically reflecting more severe, poorly controlled, or long-standing diabetes, are at greater risk of mortality, which is consistent with previous studies.42,43 The immune dysfunction associated with long-standing diabetes may contribute to the elevated risk in this group.43 The fact that this diabetes variable, and not other diabetes variables included in the variable selection process, remained important likely reflects ICD documentation practices more than a clinically relevant distinction among these variables. The association between Emphysema and higher in-hospital mortality is also consistent with previous reports.44,45 Once again, ICD coding practices likely explain why emphysema made the final list of important variables while Chronic Obstructive Pulmonary Disease did not and why Lower Respiratory Infection was marginally protective. The observed association between in-hospital mortality and two kidney-related pre-existing conditions (i.e., Acute Kidney Failure and Chronic Kidney Disease) was also reported previously.46 It is speculated that relative immune dysregulation and exaggerated inflammatory responses in kidney disease contributes to worse COVID outcomes.46 Although hypertension proposed by CDC as a risk factor for severe COIVD-19 illness,47 in our study it was excluded from the most important list of variables in the machine learning (LASSO) step. A systematic review of factors associated with COVID-19 mortality; hypertension also expressed its importance as a risk factor as 46% of patient who died from COVID-19 had such a precondition. In our veteran population, 74% (Supplementary Table 2) who died from COVID-19 had hypertension, hence, the LASSO step excluded this comorbidity due to low variation among hospitalized COVID-19 patients with in-hospital mortality.
Of interest, we did find that prehospital use of famotidine and NSAIDs was significantly protective after allowing for confounders. A recent retrospective study found that continuation of famotidine use in patients hospitalized with COVID-19 was associated with reduced risk of clinical deterioration leading to intubation or death.48 Similar benefit was observed in another study of patients who received famotidine (either oral or intravenous at any dose) within +/−7 days of COVID-19 positive test results and/or hospital admission.49 Thus, famotidine has previously been reported as protective of severe COVID-19 outcomes but detailed analysis of its effects allowing for confounders in a large, national sample have been lacking.48,49 An early, rapid systematic review reported in March 2020 on NSAIDs and viral respiratory infections showed no evidence of increased severe adverse events as a result of the use of NSAIDs.50 An early concerning announcement in LANCET about potential harm from NSAID use in patients with COVID was quickly contradicted in a short report which demonstrated no evidence of an increased risk of death with the use of NSAIDs in COVID-19.51 Our study is the first study to suggest a protective role of NSAID use before hospitalization in hospitalized COVID-19 patients. The protective effects of NSAIDs may reflect their effects on coagulation or the inflammatory response or both. Further investigation is needed and warranted.
Strengths and Weaknesses
The strengths of the study include a large cohort of patients from a healthcare system of national scope with a distribution of race and ethnicity approximating the US population. Furthermore, we used a well-curated dataset with near-complete demographics, prehospitalization medication prescriptions, pre-existing conditions, and up-to-date information of direct clinical and operational relevance to accurately assess testing, results, and outcomes. We applied a standard cross-sectional analytic approach to examine the association between smoking status and in-hospital mortality. Our approach to identifying the most important variables from the dataset was state of the art and our imputation of unknown smoking status enhances the internal validity and clinical relevance of our findings.
Importantly, our analysis is among the first and largest to segregate former from current smokers, revealing a potentially important difference in risk of adverse COVID outcomes between these two groups. The mortality assessment was limited to hospitalized patients due to delays in reporting out-of-hospital deaths. Our data did not capture all possible confounding factors, including the social determinants of health that may confound the differences seen among different smoking status groups. Nicotine replacement therapy, behavioral tobacco cessation, vaping status, and details of smoking history (e.g., duration, years since quitting, total pack years) were not captured.
We focused our current study on examining the effect of smoking status and in-hospital mortality although the relationship between smoking status and the probability testing positive for COVID-19 and need for hospitalization is also very interesting. At this time, VHA data do not capture medical information for veterans who were tested or cared for outside of the VHA system unless the care is ordered and paid for by VHA. The data also captures only limited information about social determinants of health. The risk of incomplete information on the non-VHA outcomes and confounding factors limit the use of the VA COVID-19 Shared Data Resource for some analyses.
Conclusion
Our analyses of this large, national cohort of VHA users showed that former smokers are at increased risk of in-hospital mortality due to COVID-19, but current smokers and never smokers have a similar risk. Older adults, obese patients, kidney disease, and patients with diabetes mellitus with complications are at higher risk of mortality due to COVID-19 while use of famotidine and NSAID medications before hospital admission was associated with lower in-hospital mortality. These findings contribute to our understanding of how smoking affects COVID-19 disease course.
Supplementary Material
A Contributorship Form detailing each author’s specific involvement with this content, as well as any supplementary data, are available online at https://academic.oup.com/ntr.
Funding
The analysis was supported by seed funding from Baylor College of Medicine, Houston, Texas, United States, the Center for Innovations in Quality, Effectiveness and Safety (CIN 13–413), Michael E. DeBakey VA Medical Center, Houston, TX, United states and a national institute of health (NIH), National Heart, Lung, and Blood Institute (BHLBI) K25 funding (#:1K25HL152006-01). We are grateful to the VA Informatics and Computing Infrastructure (VINCI) and VA COVID-19 Shared Data Resource.
Declaration of Interests
The authors do not have any competing interest.
Data Availability Statement
The data are owned by the Veterans Health Administration (VHA) and can be maintained and analyzed only behind the VHA firewall. At this time, the data cannot be made available to non-VHA approved persons. For more details, please contact the corresponding author.
References
- 1. Polverino F. Cigarette smoking and COVID-19: a complex interaction. Am J Respir Crit Care Med. 2020;202(3):471–472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Akl EA, Gaddam S, Gunukula SK, Honeine R, Jaoude PA, Irani J. The effects of waterpipe tobacco smoking on health outcomes: a systematic review. Int J Epidemiol. 2010;39(3):834–857. [DOI] [PubMed] [Google Scholar]
- 3. Ryu JH, Colby TV, Hartman TE, Vassallo R. Smoking-related interstitial lung diseases: a concise review. Eur Respir J. 2001;17(1):122–132. [DOI] [PubMed] [Google Scholar]
- 4. Murphy CM, Rohsenow DJ, Johnson KC, Wing RR. Smoking and weight loss among smokers with overweight and obesity in Look AHEAD. Health Psychol. 2018;37(5):399–406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Tartof SY, Qian L, Hong V, et al. Obesity and mortality among patients diagnosed with COVID-19: results from an integrated health care organization. Ann Intern Med. 2020;173(10):773–781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Farsalinos K, Angelopoulou A, Alexandris N, Poulas K. COVID-19 and the nicotinic cholinergic system. Eur Respir J. 2020;56(1):2001589. doi: 10.1183/13993003.01589-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Oliveira ASF, Ibarra AA, Bermudez I, et al. A potential interaction between the SARS-CoV-2 spike protein and nicotinic acetylcholine receptors. Biophys J. 2021;120(6):983–993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Leung JM, Yang CX, Sin DD. COVID-19 and nicotine as a mediator of ACE-2. Eur Respir J. 2020;55(6):2001261. doi: 10.1183/13993003.01261-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Kickbusch I, Leung G. Response to the emerging novel coronavirus outbreak. BMJ. 2020;368:m406. [DOI] [PubMed] [Google Scholar]
- 10. Vardavas CI, Nikitara K. COVID-19 and smoking: a systematic review of the evidence. Tob Induc Dis. 2020;18:20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Reddy RK, Charles WN, Sklavounos A, Dutt A, Seed PT, Khajuria A. The effect of smoking on COVID-19 severity: a systematic review and meta-analysis. J Med Virol. 2021;93(2):1045–1056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Lagoumintzis G, Chasapis CT, Alexandris N, et al. Nicotinic cholinergic system and COVID-19: In silico identification of interactions between α7 nicotinic acetylcholine receptor and the cryptic epitopes of SARS-Co-V and SARS-CoV-2 Spike glycoproteins. Food Chem Toxicol. 2021;149:112009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Puebla Neira D, Watts A, Seashore J, Polychronopoulou E, Kuo YF, Sharma G. Smoking and risk of COVID-19 hospitalization. Respir Med. 2021;182:106414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Development OoR. Overview of VA research on Health Equity. https://www.research.va.gov/topics/health_equity.cfm. Published 2020. Accessed August 13, 2021.
- 15. Bhargava A, Kim T, Quine DB, Hauser RG. A 20-year evaluation of LOINC in the United States’ largest integrated health system. Arch Pathol Lab Med. 2020;144(4):478–484. [DOI] [PubMed] [Google Scholar]
- 16. Development OoR. ORD COVID-19 Research Update for VHA Partners. https://www.research.va.gov/programs/orppe/education/webinars/orppe-101520.cfm. Published 2020. Accessed August 13, 2021.
- 17. Luo J, Jeyapalina S, Stoddard GJ, Kwok AC, Agarwal JP. Coronavirus disease 2019 in veterans receiving care at veterans health administration facilities. Ann Epidemiol. 2021;55:10–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Calhoun PS, Wilson SM, Hertzberg JS, et al. ; VA Mid-Atlantic MIRECC Workgroup . Validation of veterans affairs electronic medical record smoking data among Iraq- and Afghanistan-era veterans. J Gen Intern Med. 2017;32(11):1228–1234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Golden SE, Hooker ER, Shull S, et al. Validity of Veterans Health Administration structured data to determine accurate smoking status. Health Informatics J. 2020;26(3):1507–1515. [DOI] [PubMed] [Google Scholar]
- 20. McGinnis KA, Brandt CA, Skanderson M, et al. Validating smoking data from the Veteran’s Affairs Health Factors dataset, an electronic data source. Nicotine Tob Res. 2011;13(12):1233–1239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Maynard C. Ascertaining Veterans’ Vital Status: VA data sources for mortality ascertainment and cause of death. VA: Database & Methods Cyberseminar Series Web site. https://www.hsrd.research.va.gov/for_researchers/cyber_seminars/archives/1242-notes.pdf. Published 2017. Accessed August 18, 2021. [Google Scholar]
- 22. Williams DM, Jylhävä J, Pedersen NL, Hägg S. A Frailty index for UK biobank participants. J Gerontol A Biol Sci Med Sci. 2019;74(4):582–587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Azur MJ, Stuart EA, Frangakis C, Leaf PJ. . Multiple imputation by chained equations: what is it and how does it work?. Int J Methods Psychiatr Res. 2011;20(1):40–49. doi: 10.1002/mpr.329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Wulff JN, Ejlskov L. Multiple imputation by chained equations in praxis: guidelines and review. Electron J Bus Res Methods. 2017;15(1). [Google Scholar]
- 25. Scott DuVall JS. Introduction to the VA COVID-19 Shared Data Resource and its Use for Research. VA: Health Services Research & Development Web site. https://www.hsrd.research.va.gov/cyberseminars/catalog-upcoming-session.cfm?UID=3810. Published 2020. Accessed August 13, 2021. [Google Scholar]
- 26. Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47(11):1245–1251. [DOI] [PubMed] [Google Scholar]
- 27. Viernes B, Lynch KE, Robison B, Gatsby E, DuVall SL, Matheny ME. SNOMED CT Disease Hierarchies and the Charlson Comorbidity Index (CCI): An analysis of OHDSI methods for determining CCI. https://www.ohdsi.org/wp-content/uploads/2020/10/Ben-Viernes-Benjamin-Viernes_CCIBySNOMED_2020Symposium.pdf. Accessed August 18, 2021.
- 28. Tang J, Alelyani S, Liu H. Feature selection for classification: A review. In: Data classification: Algorithms and applications. Boca Raton, FL: CRC Press. 2014:37–64. https://www.scopus.com/record/display.uri?eid=2-s2.0-85054068951&origin=inward&txGid=c221c736394899697110a19623d529dd&featureToggles=FEATURE_VIEW_PDF:1. [Google Scholar]
- 29. Del Sole F, Farcomeni A, Loffredo L, et al. Features of severe COVID-19: A systematic review and meta-analysis. Eur J Clin Invest. 2020;50(10):e13378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Hasan A, Paray BA, Hussain A, et al. A review on the cleavage priming of the spike protein on coronavirus by angiotensin-converting enzyme-2 and furin. J Biomol Struct Dyn. 2021;39(8):3025–3033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Cai G, Bossé Y, Xiao F, Kheradmand F, Amos CI. Tobacco smoking increases the lung gene expression of ACE2, the receptor of SARS-CoV-2. Am J Respir Crit Care Med. 2020;201(12):1557–1559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Martinez FJ, Han MK, Allinson JP, et al. At the root: defining and halting progression of early chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2018;197(12):1540–1551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. CDC. People with Certain Medical Conditions. https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html#obesity. Published 2020. Accessed August 13, 2021.
- 34. Razjouyan J, Helmer DA, Li A, et al. Differences in COVID-19-related testing and healthcare utilization by race and ethnicity in the veterans health administration[published online ahead of print March 10, 2021]. J Racial Ethn Health Disparities. 2021;1–8. doi: 10.1007/s40615-021-00982-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Levin AT, Hanage WP, Owusu-Boaitey N, Cochran KB, Walsh SP, Meyerowitz-Katz G. Assessing the age specificity of infection fatality rates for COVID-19: systematic review, meta-analysis, and public policy implications. Eur J Epidemiol. 2020;35(12):1123–1138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. COVID TC, Team R. Severe outcomes among patients with Coronavirus Disease 2019 (COVID-19)-United States, February 12-March 16, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(12):343–346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Sharma A. Estimating older adult mortality from COVID-19. J Gerontol B Psychol Sci Soc Sci. 2021;76(3):e68–e74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Shahid Z, Kalayanamitra R, McClafferty B, et al. COVID-19 and older adults: what we know. J Am Geriatr Soc. 2020;68(5):926–929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. CDC. COVID-19: Older Adults. https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/older-adults.html. Published 2021. August 13, 2021.
- 40. Hussain A, Mahawar K, Xia Z, Yang W, El-Hasani S. Obesity and mortality of COVID-19. Meta-analysis. Obes Res Clin Pract. 2020;14(4):295–300. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 41. Pettit NN, MacKenzie EL, Ridgway JP, et al. Obesity is associated with increased risk for mortality among hospitalized patients with COVID‐19. Obesity. 2020;28(10):1806–1810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Bhandari S, Rankawat G, Singh A, Gupta V, Kakkar S. Impact of glycemic control in diabetes mellitus on management of COVID-19 infection[published online ahead of print September 2, 2020]. Int J Diabetes Dev Ctries. 2020;1–6. doi: 10.1007/s13410-020-00868-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Pal R, Banerjee M. Are people with uncontrolled diabetes mellitus at high risk of reinfections with COVID-19? Prim Care Diabetes. 2021;15(1):18–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Higham A, Mathioudakis A, Vestbo J, Singh D. COVID-19 and COPD: a narrative review of the basic science and clinical outcomes. Eur Respir Rev. 2020;29(158):200199. doi: 10.1183/16000617.0199-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Leung JM, Niikura M, Yang CWT, Sin DD. COVID-19 and COPD. Eur Respir J. 2020;56(2):2002108. doi: 10.1183/13993003.02108-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Robbins-Juarez SY, Qian L, King KL, et al. Outcomes for patients With COVID-19 and acute kidney injury: a systematic review and meta-analysis. Kidney Int Rep. 2020;5(8):1149–1160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. CDC. People with certain medical conditions. https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html. Published 2021. Accessed August 13, 2021.
- 48. Freedberg DE, Conigliaro J, Wang TC, Tracey KJ, Callahan MV, Abrams JA; Famotidine Research Group . Famotidine use is associated with improved clinical outcomes in hospitalized COVID-19 patients: a propensity score matched retrospective cohort study. Gastroenterology. 2020;159(3):1129–1131.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Mather JF, Seip RL, McKay RG. Impact of famotidine use on clinical outcomes of hospitalized patients with COVID-19. Am J Gastroenterol. 2020;115(10):1617–1623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.World Health Organization. The use of non-steroidal anti-inflammatory drugs (NSAIDs) in patients with COVID-19: scientific brief, 19 April 2020. World Health Organization; 2020. https://www.who.int/news-room/commentaries/detail/the-use-of-non-steroidal-anti-inflammatory-drugs-(nsaids)-in-patients-with-covid-19. [Google Scholar]
- 51. Vosu J, Britton P, Howard-Jones A, et al. Is the risk of ibuprofen or other non-steroidal anti-inflammatory drugs increased in COVID-19? J Paediatr Child Health. 2020;56(10):1645–1646. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data are owned by the Veterans Health Administration (VHA) and can be maintained and analyzed only behind the VHA firewall. At this time, the data cannot be made available to non-VHA approved persons. For more details, please contact the corresponding author.