Abstract
Hematopoietic stem cells (HSCs) reside in distinct sites throughout fetal and adult life and give rise to all cells of the hematopoietic system. Because of their multipotency, HSCs are capable of curing a wide variety of blood disorders through hematopoietic stem cell transplantation (HSCT). However, due to HSC heterogeneity, site-specific ontogeny and current limitations in generating and expanding HSCs in vitro, their broad use in clinical practice remains challenging. To assess HSC multipotency, evaluation of their capacity to generate T lymphocytes has been regarded as a valid read-out. Several in vitro models of T cell development have been established which are able to induce T-lineage differentiation from different hematopoietic precursors, although with variable efficiency. Here, we review the potential of human HSCs from various sources to generate T-lineage cells using these different models in order to address the use of both HSCs and T cell precursors in the clinic.
Keywords: HSC, huma, in vitro model, T cell development
Introduction
Human hematopoiesis in vivo
Hematopoiesis is a strictly regulated process in which hematopoietic stem cells (HSCs) gradually lose multipotency in order to generate the full repertoire of blood cells that comprise the hematopoietic system. These multipotent HSCs originate or reside in specific niches that are located at different sites throughout life [1–4]. In the human embryo, hematopoiesis is characterized by a primitive and a definitive wave which are spatiotemporally separated and have different characteristics [5–7]. The primitive program of hematopoiesis occurs after 3 weeks of gestation in the blood islands of the extra-embryonic yolk sac (YS) and generates a transient wave of mainly primitive erythroblasts, megakaryocytes and macrophages [8]. Following primitive hematopoiesis, the definitive program is initiated, at day 32 of gestation, within the dorsal aorta of the aorta-gonad-mesonephros (AGM) region which originates from the embryonic mesoderm [9]. The definitive wave of embryonic hematopoiesis is characterized by the generation of the first self-renewing HSCs that have multilineage hematopoietic potential. These HSCs will subsequently migrate to the fetal liver (FL) where they expand and further support hematopoiesis during fetal life [10,11]. Finally, HSCs will colonize the developing bone marrow (BM) which is the most dominant site of hematopoiesis starting from 20 weeks of gestation [12]. Upon birth, these HSCs will cease to proliferate and acquire a quiescent state in order to maintain lifelong postnatal hematopoiesis [13,14]. Understanding the nature of HSCs has important implications for their in vitro generation and use in the clinic.
Human T cell development in vivo
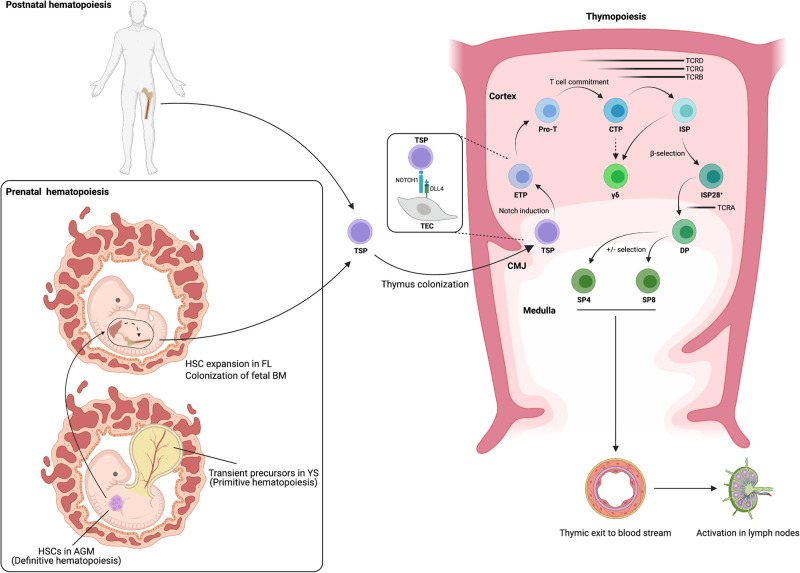
T-lineage potential has been regarded as a hallmark of definitive HSCs [15]. T cell development occurs in the thymus, a small organ located in the thoracic cavity that instructs HSC-derived thymus seeding precursors (TSPs) to differentiate along the T cell lineage in response to the micro-environmental stimuli that it provides (Figure 1) [16–18]. Because of its distinct anatomical site compared with where HSCs reside, immature precursors need to egress from the BM in order to colonize the thymus, starting from week 8 of gestation [19–21]. Upon intra-thymic Notch signaling, predominantly initiated by the NOTCH1–DLL4 axis, the expression of CD7 is highly up-regulated in the TSPs as they differentiate into early T cell progenitors (ETPs). Subsequently, upon induction of IL-7 signaling and continuous Notch stimulation, these ETPs develop into progenitor T cells (pro-T cells) and display a decreased potential to develop into other hematopoietic lineages [22–25]. These pro-T cells will then fully lose the capacity to differentiate into non-T cells by committing to the T cell lineage which is generally characterized by the up-regulation of CD1a [26,27], although more recently the loss of CD44 was found to more accurately define human T cell commitment [28]. During the induction of T cell development, rearrangements of the TCRD, TCRG and TCRB loci are initiated in a process called V(D)J recombination which leads to the generation of functional T cell receptors (TCRs). The outcome of these TCR rearrangement events is the main driver of the developmental bifurcation of the αβ- and γδ-lineages which mainly occurs at the ISP stage, and this allows both lineages to mature into distinct T cell subsets that have unique functions in the periphery [29]. During αβ-lineage development, an in-frame rearranged TCRβ chain associates with the invariant pre-T cell receptor α chain (pTα) in order to generate the pre-TCR that induces β-selection, an event that mainly occurs at the CD4+ immature single positive (ISP) stage [30–33]. Upon pre-TCR signaling, developing thymocytes receive proliferation and survival signals and up-regulate CD8β to differentiate into CD4+CD8β+ double positive (DP) T cells [34]. These DP T cells then enter a quiescent state to permit V(D)J recombination of the TCRA locus [35]. Upon replacement of the pTα by an in-frame rearranged TCRα chain, a functional TCRαβ is expressed on the cell surface together with the CD3 co-receptor [36]. Subsequently, these αβ-lineage T cells undergo positive and negative selection in the thymus in order to establish major histocompatibility complex (MHC)-restricted pathogen recognition and central tolerance, respectively [37,38]. During positive selection, the thymocytes develop into either CD4+ or CD8+ single positive (SP) T cells that will eventually acquire an immunomodulatory or cytotoxic function, respectively. Finally, these mature naïve CD4+ and CD8+ SP T cells exit the human thymus and migrate to the lymph nodes where they can be activated by MHC:antigen complexes on antigen presenting cells (APCs) to enable the elimination of both pathogens and cancer cells (Figure 1).
Figure 1. Human hematopoiesis and thymopoiesis in vivo.
Human embryonal hematopoiesis is characterized by a primitive program capable of generating transient precursors after which true HSCs are generated in a definitive wave. These HSCs further expand in the FL before migrating to the fetal BM which supports hematopoiesis throughout postnatal life. HSCs then egress as TSPs into the bloodstream in order to colonize the thymus where the NOTCH1/DLL4 axis induces the T cell program. T cell development occurs through sequential progression through distinct intermediate stages in which germline rearrangements of TCR loci primarily instructs the bifurcation of the αβ and γδ T cell lineages. HSC: hematopoietic stem cell; AGM: aorta-gonad-mesonephros; YS: yolk sac; FL: fetal liver; BM: bone marrow; CMJ: cortico-medullary junction; TSP: thymus seeding precursor; TEC: thymic epithelial cell; ETP: early T cell progenitor; pro-T: progenitor T cell; CTP: committed T cell precursor; ISP: immature single positive; ISP28+: β-selected ISPs; DP: double positive; SP4/8: CD4/CD8 single positive; γδ: γδ T cell. (Created with BioRender.com).
The clinical application of various sources of HSCs
The access to HSCs is crucial to cure a wide range of blood disorders via hematopoietic stem cell transplantation (HSCT). HSCs are mainly characterized by their self-renewal capacity while preserving multilineage potential. However, our understanding of the biology and immunophenotypic landscape of true HSCs remains rather elusive due to their heterogeneous nature and origin. Substantial efforts have been made to characterize the immunophenotype and multilineage potential of HSCs in more detail using serial transplantation studies in immunocompromised mice, clonal lineage tracing experiments and single cell approaches [39–43]. Several populations within the CD34+CD38-CD45RA- multipotent compartment were found to be enriched for true HSCs, such as CD90+ and CD49f+ fractions [44,45]. Next to their immunophenotypic heterogeneity, HSCs gradually display altered functional properties upon aging since adult HSCs often show a myeloid bias because of changing epigenetic and transcriptional programs [46]. In addition, impaired DNA damage repair and increased production of reactive oxygen species (ROS) in aged HSCs results in lower reconstitution activity upon HSCT [47,48]. Furthermore, the yield of HSCs from different sources can be limited and the search for human leukocyte antigen (HLA)-matching donors in order to avoid graft failure and graft-versus-host disease (GvHD) remains challenging [49]. Therefore, providing larger numbers of HLA-matching HSCs in these transplantations would substantially improve hematopoietic reconstitution and broaden its usage. Only recently, the small molecules StemRegenin 1 (SR1) and UM171 have been identified to promote HSC self-renewal in long-term ex vivo cultures, while maintaining engraftment potential [50–53]. By antagonizing the aryl hydrocarbon receptor (AHR), SR1 prevents HSC differentiation while UM171 promotes HSC self-renewal by indirectly depositing H3K4me2 and H3K27Ac activation marks in the proximity of stem cell genes [54]. Although the retained long-term lymphoid potential of these ex vivo expanded HSCs still remains to be verified in vivo, these small molecules have important therapeutical implications. In addition to the ex vivo expansion of HSCs, pluripotent stem cells (PSCs), such as embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), are actively being studied to exploit their potential for generating HSCs in vitro. As such, iPSCs may provide an inexhaustive source of patient-specific HSCs that can be used for autologous HSCT or to model disease [55,56]. The ex vivo expansion and in vitro generation of HSCs might furthermore improve fundamental studies since larger numbers of HSCs could be generated to perform perturbation studies using gene editing approaches such as CRISPR/Cas9 [57]. Since assessment of the T-lineage potential of HSCs has been regarded as a valid evaluation strategy of their multilineage capacity, modeling of human T cell development can serve as a valuable read-out to address HSC multipotency after in vitro generation or ex vivo expansion, and thus to evaluate their therapeutic use.
In vitro modeling of human T cell development
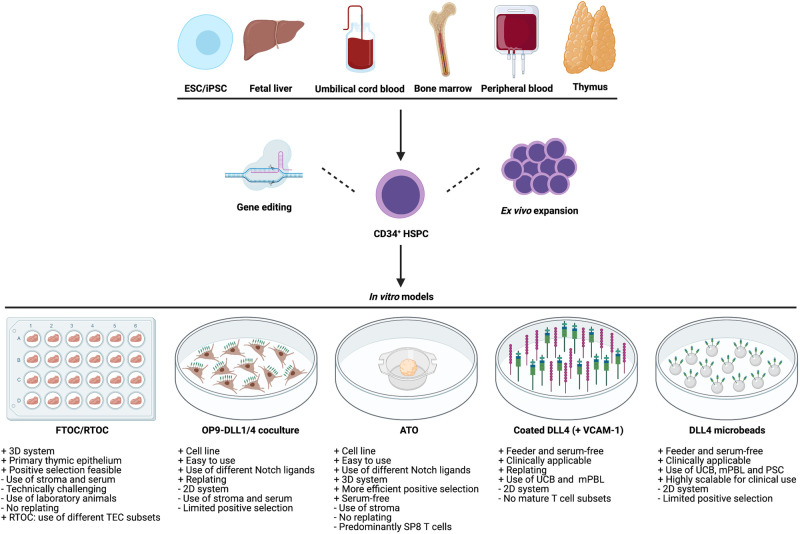
T cell development can be studied by multiple in vitro approaches. While the establishment of fetal thymic organ cultures (FTOCs) provided the first in vitro model to study human T cell development around 30 years ago, the OP9-Delta-like ligand 1/4 (OP9-DLL1/4) coculture system is generally more accessible and technically less challenging. Recently, the artificial thymic organoid (ATO) model has shown to be a reliable 3D format to study the more mature stages of T cell development from different hematopoietic stem and precursor cell (HSPC) sources. In addition, feeder-free approaches have been designed for the generation of clinical-grade T cell precursors (Figure 2).
Figure 2. HSPCs from various sources can be exploited to model human T cell development in vitro.
CD34+ HSPCs can be isolated from either bone marrow, thymus, fetal liver, umbilical cord blood, peripheral blood or in vitro differentiated from ESCs and iPSCs. These HSPCs can be expanded and manipulated ex vivo before subsequent application in in vitro T cell differentiation models. Green and purple sticks represent DLL1/4 Notch ligand and VCAM-1, respectively. The OP9 stromal cells and microbeads are depicted as brown cells and silver spheres, respectively. ESC: embryonic stem cell; iPSC: induced pluripotent stem cell; HSPC: hematopoietic stem and precursor cell; FTOC: fetal thymic organ culture; RTOC: reaggregated thymic organ culture; TEC: thymic epithelial cell; OP9-DLL1/4: OP9-Delta-like ligand 1/4; ATO: artificial thymic organoid; SP8: CD8 single positive; VCAM-1: vascular cell adhesion molecule 1; UCB: umbilical cord blood; mPBL: mobilized peripheral blood; PSC: pluripotent stem cell. (Created with BioRender.com).
Fetal thymic organ cultures
FTOCs were the first approach to study human T cell development in vitro. Here, thymic lobes from E14-E15 immunodeficient mice are isolated and reconstituted with human HSPCs by the hanging drop method [58]. Initially, thymic lobes from wildtype mice were used and these were depleted from endogenous thymocytes using deoxyguanosine to allow more efficient engraftment of human precursors. Since these FTOCs do not permit to study the specific impact of different subsets of thymic epithelial cells (TECs) or other stromal cells, reaggregated FTOCs (RTOCs) were developed in which HSPCs and murine TECs were combined in small organoids [58]. FTOCs provided the first insights into the developmental potential of human HSPCs that were derived from different sources. Precursors from fetal BM, FL and umbilical cord blood (UCB) differentiated more efficiently towards mature T cell subsets compared with those from adult BM, which highlights important differences in T cell potential of fetal versus adult hematopoietic programs [12,59–61]. Particularly aging affects T lymphoid potential since CD34+ precursors from older BM donors displayed decreased T cell potential compared with younger counterparts as shown by the low efficiency of aged HSPCs to generate CD4+CD8β+ DP T cells in FTOCs [62]. In addition, it also has been shown that aging predominantly affects lymphoid progenitors as illustrated by the decreased growth and survival of aged lymphoid precursors, mediated by elevated levels of the cell cycle regulators p16INK4a and p14ARF [63]. To further underscore that HSPCs are highly heterogeneous, the expression of CD38 within bulk CD34+ BM-derived HSPCs was shown to be inversely correlated with T lymphoid potential in FTOCs [64]. Although these 3D in vitro organ cultures provided valuable insights in HSC/HSPC heterogeneity and allowed to study human T cell development, their use might raise ethical concerns and the assembly of FTOCs is technically challenging which can lead to variable results.
OP9-DLL1/4 coculture models
Because of these issues, the OP9-DLL1 system was developed as an alternative in vitro model to improve the study of human T cell development. OP9 cells are derived from the BM stroma of op/op mice, a strain that has defects in myeloid development since these mice do not express macrophage colony-stimulating factor (M-CSF). These OP9 cells were transduced with a DLL1 Notch ligand to support the generation of T cells since Notch signaling is indispensable for T cell development in vivo [65]. In addition, the OP9-DLL4 variant was developed since it has been shown that the NOTCH1/DLL4 axis is the physiological driving force of early T cell development in vivo [66,67]. In these models, HSPCs are seeded onto confluent layers of OP9-DLL1/4 stroma in the presence of SCF, FLT3-L and IL-7. Upon detaching of the exhausted OP9 monolayer, the coculture can easily be replated onto new OP9-DLL1/4 stromal cells which allows to continuously and kinetically track the development of the initial HSPC population, even from single cells [68,69]. This avoids the need for multiple parallel cultures, in contrast with when using FTOCs. Similar to FTOCs, however, HSPCs from various sources display variable T lymphoid potential in OP9-DLL1/4 cocultures as shown by the inefficient generation of mature T cell subsets from BM-derived CD34+CD38−/low HSPCs compared with from their UCB-derived counterparts. This phenomenon, which is cell-autonomous as demonstrated by mixed cocultures, is probably caused by a bias towards the myeloid program in BM-derived HSPCs [69]. Interestingly, pro-T cells that were generated on OP9-DLL1 cocultures from CD34+CD38−/low UCB-derived HSPCs are capable of reconstituting the thymus of immunodeficient mice, illustrating their clinical potential [70,71]. While this model has revolutionized the study of early human T cell development, OP9-DLL1/4 cocultures unfortunately do not very efficiently support the generation of mature T cells.
OP9-DLL1/4 models have also been used to evaluate the T cell potential of HSPCs that are derived from PSCs. In order to generate HSCs, PSCs first need to differentiate into mesoderm which then gives rise to hemogenic endothelium (HE) from which HSCs originate during definitive hematopoiesis [6]. The first protocol described by Vodyanik et al. [72] seeded single ESCs onto a confluent monolayer of OP9 stroma in the presence of serum, without the addition of growth factors. This approach resulted in the generation of CD34+KDR+ HE-derived CD43+ HSPCs with multilineage capacity although T cell development was not addressed [73]. As a modification of this pioneering approach, Timmermans et al. [74] initiated OP9 cocultures by seeding ESC clumps in the presence of both serum and hematopoietic growth factors. After 10–12 days of coculture, hematopoietic zones (HZs) could be identified in which both CD34+ HSPCs and surrounding CD34+ endothelial cells were present. Upon transfer of these HZs onto OP9-DLL1 stromal cells, CD34+ HSPCs gave rise to T-lineage cells after 4–5 weeks. Since these approaches are serum-based and use xenogeneic OP9 stroma, Kennedy et al. [15] designed a protocol to generate embryoid bodies (EBs) in a well-defined medium and low oxygen environment in order to better mimic the in vivo situation [75]. During mesoderm induction, the GSK3-β antagonist CHIR99021 is added to stimulate Wnt signaling which promotes definitive hematopoiesis [76]. After 8 days, CD34+CD43−CD84−CD173− HE cells can be sorted to generate definitive CD34+CD45+ HSPCs in a cytokine-rich medium. Finally, these HSPCs can be isolated and seeded onto OP9-DLL1 stroma to induce T cell development in vitro. This EB approach was further employed by Themeli et al. [77,78] where a less complex defined medium was used to either generate iPSC-derived T cells having anti-tumor activity or to model severe combined immune deficiency (SCID) using patient-specific iPSCs. Overall, the OP9-DLL1/4 coculture model has significantly boosted the study of early human T cell development from a large variety of HSPC sources and this model can easily be applied, also from a limited amount of precursor cells.
Artificial thymic organoids
While the OP9-DLL1/4 coculture model has been instrumental to unravel the molecular mechanisms that drive early human T cell development, this 2D model does not efficiently support the generation of SP4 and SP8 T cells. Therefore, the 3D ATO in vitro model was recently developed in which CD34+ HSPCs and MS5-DLL1/4 stromal cells are combined at defined ratios, similar to RTOCs, and incubated on a transwell insert that is in contact with serum-free T cell induction medium [79]. This system has been shown to support T cell differentiation using CD34+ HSPCs from different sources, including UCB, peripheral blood (PBL), thymus and BM, highlighting the high T-lineage potential of ATOs. However, HSPCs derived from either BM or PBL show slower kinetics during T cell maturation, again underscoring the heterogeneous nature of HSPCs. Although no primary thymic epithelium is used, the spatiotemporal network of ATOs might provide thymic-like niches that allow T cell progenitors to migrate through these niches which may facilitate the generation of more mature stages of T cell development. This might be further enhanced by the altered oxygen levels in these ATOs which are cultured at the air/liquid interface, compared with OP9-DLL1/4 cocultures in which the T cell precursors are submerged in the culture medium. In addition, the prolonged cross-talk between stromal cells and T cell precursors, in combination with the high density of these small organoids that enables reciprocal stimulation and selection of progenitors, may also enhance thymocyte maturation. The development of SP8 T cells is favored in ATOs because of the ubiquitous expression of MHC-I molecules on the developing thymocytes while the low abundance of MHC-II-expressing dendritic cells complicates the maturation of SP4 T cells. Because of the technical simplicity of ATOs and the efficient generation of the more mature stages of T cell development, ATOs may become increasingly important to assess the multilineage potential of HSPCs. Indeed, ATOs have been recently used to model SCID using patient-derived HSPCs [80]. However, as opposed to OP9-DLL1/4 cocultures but similar to FTOCs, well-to-well variation might be observed since ATOs cannot be reaggregated after analysis for kinetic studies within the same organoid. Nevertheless, as with OP9-DLL1/4 cocultures, ATOs can serve as a suitable preclinical model but will need to be further optimized to enable the generation of clinical-grade T cells since murine stromal cells are used.
Importantly, ATOs are also capable of generating human ESC- and iPSC-derived SP T cells. Here, following the initial generation of a CD326+CD56+ embryonic mesodermal progenitors (EMPs) from PSCs, the emergence of HSPCs is induced in embryonic mesodermal organoids (EMOs) by mixing these EMPs together with MS5-DLL4 stromal cells. Subsequently, the medium is switched to the T cell induction medium to initiate T cell development in the same initial organoid [81]. This serum-free PSC-ATO protocol is capable of generating PSC-derived T cells at a higher efficiency compared with the 2D OP9 model. This presumably not only results from the 3D architecture that is provided by the organoids during T cell development, but may also be facilitated by the presence of Notch signaling during the hematopoietic specification step in the EMOs since definitive hematopoiesis is indeed dependent on Notch signaling in vivo [82,83].
Feeder-free cultures
Since the stroma-based in vitro models rely on the use of murine cells, they are not yet suitable for the generation of HSPC-derived T cells or T cell precursors for clinical application. Therefore, a feeder-free model was developed in which coated DLL4 and vascular cell adhesion molecule 1 (VCAM-1) provided a fully defined thymic-like niche that is capable of synergistically inducing early human T cell development from UCB-derived HSPCs [84]. Here, VCAM-1 engages with α4-integrin on HSPCs to enhance Notch signaling for the generation of CD7+CD5+CD45RA+ pro-T cells. Upon injection in immunodeficient mice, these pro-T cells home to the thymus and differentiate into functional SP8 T cells. Also specific cytokines such as TNFα, combined with coated DLL4, have been shown to promote the development of early human T cell precursors from both UCB and mobilized PBL (mPBL) sources [85,86]. More recently, a novel method has been designed for the generation of clinical-grade T cells by incubating HSPCs with DLL4-coated microbeads in suspension which significantly enhances the scalability of T cell precursor generation out of HSPCs from either ESCs, UCB and mobilized peripheral blood (mPBL) and the therapeutic applicability [87]. Importantly, the impact of thymic involution and injury on T cell reconstitution upon infusion of these clinical-grade T cell progenitors remains to be addressed. Nonetheless, these results show that off-the-shelf pro-T cells can easily be generated in vitro in a fully defined serum-free and stroma-free environment. As such, these feeder-free approaches might become increasingly important in the clinic to improve T cell reconstitution after myeloablative conditioning and HSCT.
Concluding remarks
Investigating the multilineage potential of human HSCs that are isolated from different sources, or that are expanded or generated in vitro, is instrumental to evaluate their clinical potential. Studying the T cell developmental capacity of these highly diverse sources of HSCs has been very useful in this context since T lymphoid potential has been regarded as a valid read-out of HSC multilineage potential. In addition, the generation of off-the-shelf T cell precursors has important implications in the clinic to improve and accelerate reconstitution of the T-cell lineage which can take months or even years following classical HSCT. However, their application is still suboptimal and requires further optimization of existing protocols or the development of novel innovative tools in order to bring such patient-tailored clinical applications from bench to bedside. Fortunately, significant advancements have been made over the past years that facilitate the evaluation of T-lineage potential which will significantly accelerate the functional evaluation of these novel approaches.
Perspectives
Importance of the field: The in vitro generation and expansion of clinical-grade HSPCs, T cell precursors and functional mature T cells holds enormous therapeutic potential for treating hematological disorders through HSCT, and for improving and broadening immune therapeutic approaches.
Current state of the research: While in vitro models allow to study human T cell development and can be used to evaluate the multilineage potential of ex vivo isolated or in vitro generated and expanded HSC/HSPCs, they are not fully suitable yet to generate mature functional T cells that can be used in the clinic. Although clinical-grade human T cell precursors can be generated in vitro out of HSPCs, the in vitro expansion and generation of human HSCs requires further research.
Future directions: By optimizing current in vitro models, or by designing novel innovative tools, the generation of off-the-shelf HSPCs and T cells might become a revolutionary curative strategy to improve both HSCT and immune cell therapy.
Abbreviations
- AGM
aorta-gonad-mesonephros
- ATO
artificial thymic organoid
- BM
bone marrow
- DP
double positive
- EMOs
embryonic mesodermal organoids
- EMPs
embryonic mesodermal progenitors
- ESCs
embryonic stem cells
- ETPs
early T cell progenitors
- FL
fetal liver
- FTOCs
fetal thymic organ cultures
- HE
hemogenic endothelium
- HLA
human leukocyte antigen
- HSCT
hematopoietic stem cell transplantation
- HSCs
hematopoietic stem cells
- HSPC
hematopoietic stem and precursor cell
- HZs
hematopoietic zones
- ISP
immature single positive
- MHC
major histocompatibility complex
- PBL
peripheral blood
- PSCs
pluripotent stem cells
- SCID
severe combined immune deficiency
- SP
single positive
- SR1
StemRegenin 1
- TEC
thymic epithelial cell
- TSPs
thymus seeding precursors
- UCB
umbilical cord blood
- VCAM-1
vascular cell adhesion molecule 1
- YS
yolk sac
Competing Interests
The authors declare that there are no competing interests associated with the manuscript.
Funding
Work on this topic in the Taghon lab was supported by the Fund for Scientific Research Flanders (FWO project grants, E-Rare-3 JTC 2017 and fellowship grant to SS), the Foundation Against Cancer (STK) and grants from the Ghent University Research Fund (GOA and Starting Credit).
Author Contributions
S.S. and T.T. wrote the manuscript.
References
- 1.Ceredig, R., Rolink, A.G. and Brown, G. (2009) Models of haematopoiesis: seeing the wood for the trees. Nat. Rev. Immunol. 9, 293–300 10.1038/nri2525 [DOI] [PubMed] [Google Scholar]
- 2.Doulatov, S., Notta, F., Laurenti, E. and Dick, J.E. (2012) Hematopoiesis: a human perspective. Cell Stem Cell 10, 120–136 10.1016/j.stem.2012.01.006 [DOI] [PubMed] [Google Scholar]
- 3.Mahony, C.B. and Bertrand, J.Y. (2019) How HSCs colonize and expand in the fetal niche of the vertebrate embryo: an evolutionary perspective. Front. Cell Dev. Biol. 7, 34 10.3389/fcell.2019.00034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sánchez, M.-J., Holmes, A., Miles, C. and Dzierzak, E. (1996) Characterization of the first definitive hematopoietic stem cells in the agm and liver of the mouse embryo. Immunity 5, 513–525 10.1016/S1074-7613(00)80267-8 [DOI] [PubMed] [Google Scholar]
- 5.Jagannathan-Bogdan, M. and Zon, L.I. (2013) Hematopoiesis. Development 140, 2463–2467 10.1242/dev.083147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ditadi, A., Sturgeon, C.M. and Keller, G. (2017) A view of human haematopoietic development from the petri dish. Nat. Rev. Mol. Cell Biol. 18, 56–67 10.1038/nrm.2016.127 [DOI] [PubMed] [Google Scholar]
- 7.Julien, E., El Omar, R. and Tavian, M. (2016) Origin of the hematopoietic system in the human embryo. FEBS Lett. 590, 3987–4001 10.1002/1873-3468.12389 [DOI] [PubMed] [Google Scholar]
- 8.Palis, J., Robertson, S., Kennedy, M., Wall, C. and Keller, G. (1999) Development of erythroid and myeloid progenitors in the yolk sac and embryo proper of the mouse. Development 126, 5073–5084 10.1242/dev.126.22.5073 [DOI] [PubMed] [Google Scholar]
- 9.Medvinsky, A. and Dzierzak, E. (1996) Definitive hematopoiesis is autonomously initiated by the AGM region. Cell 86, 897–906 10.1016/S0092-8674(00)80165-8 [DOI] [PubMed] [Google Scholar]
- 10.Ema, H. and Nakauchi, H. (2000) Expansion of hematopoietic stem cells in the developing liver of a mouse embryo. Blood 95, 2284–2288 10.1182/blood.V95.7.2284 [DOI] [PubMed] [Google Scholar]
- 11.Kumaravelu, P., Hook, L., Morrison, A.M., Ure, J., Zhao, S., Zuyev, S.et al. (2002) Quantitative developmental anatomy of definitive haematopoietic stem cells/long-term repopulating units (HSC/RUs): role of the aorta-gonad-mesonephros (AGM) region and the yolk sac in colonisation of the mouse embryonic liver. Development 129, 4891–4899 10.1242/dev.129.21.4891 [DOI] [PubMed] [Google Scholar]
- 12.Popescu, D.-M., Botting, R.A., Stephenson, E., Green, K., Webb, S., Jardine, L.et al. (2019) Decoding human fetal liver haematopoiesis. Nature 574, 365–371 10.1038/s41586-019-1652-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Morrison, S.J. and Scadden, D.T. (2014) The bone marrow niche for haematopoietic stem cells. Nature 505, 327–334 10.1038/nature12984 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bowie, M.B., McKnight, K.D., Kent, D.G., McCaffrey, L., Hoodless, P.A. and Eaves, C.J. (2006) Hematopoietic stem cells proliferate until after birth and show a reversible phase-specific engraftment defect. J. Clin. Invest. 116, 2808–2816 10.1172/JCI28310 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kennedy, M., Awong, G., Sturgeon, C.M., Ditadi, A., LaMotte-Mohs, R., Zuniga-Pflucker, J.C.et al. (2012) T lymphocyte potential marks the emergence of definitive hematopoietic progenitors in human pluripotent stem cell differentiation cultures. Cell Rep. 2, 1722–1735 10.1016/j.celrep.2012.11.003 [DOI] [PubMed] [Google Scholar]
- 16.Miller, J.F.A.P. (2020) The function of the thymus and its impact on modern medicine. Science 369, eaba2429 10.1126/science.aba2429 [DOI] [PubMed] [Google Scholar]
- 17.Lavaert, M., Liang, K.L., Vandamme, N., Park, J.E., Roels, J., Kowalczyk, M.S.et al. (2020) Integrated scRNA-seq identifies human postnatal thymus seeding progenitors and regulatory dynamics of differentiating immature thymocytes. Immunity 52, 1088–1104.e6 10.1016/j.immuni.2020.03.019 [DOI] [PubMed] [Google Scholar]
- 18.Roels, J., Kuchmiy, A., De Decker, M., Strubbe, S., Lavaert, M., Liang, K.L.et al. (2020) Distinct and temporary-restricted epigenetic mechanisms regulate human alphabeta and gammadelta T cell development. Nat. Immunol. 21, 1280–1292 10.1038/s41590-020-0747-9 [DOI] [PubMed] [Google Scholar]
- 19.Haddad, R., Guimiot, F., Six, E., Jourquin, F., Setterblad, N., Kahn, E.et al. (2006) Dynamics of thymus-colonizing cells during human development. Immunity 24, 217–230 10.1016/j.immuni.2006.01.008 [DOI] [PubMed] [Google Scholar]
- 20.Farley, A.M., Morris, L.X., Vroegindeweij, E., Depreter, M.L.G., Vaidya, H., Stenhouse, F.H.et al. (2013) Dynamics of thymus organogenesis and colonization in early human development. Development 140, 2015–2026 10.1242/dev.087320 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Six, E.M., Bonhomme, D., Monteiro, M., Beldjord, K., Jurkowska, M., Cordier-Garcia, C.et al. (2007) A human postnatal lymphoid progenitor capable of circulating and seeding the thymus. J. Exp. Med. 204, 3085–3093 10.1084/jem.20071003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jaleco, A.C., Neves, H., Hooijberg, E., Gameiro, P., Clode, N., Haury, M.et al. (2001) Differential effects of Notch ligands delta-1 and jagged-1 in human lymphoid differentiation. J. Exp. Med. 194, 991–1002 10.1084/jem.194.7.991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.De Smedt, M., Reynvoet, K., Kerre, T., Taghon, T., Verhasselt, B., Vandekerckhove, B.et al. (2002) Active form of Notch imposes T cell fate in human progenitor cells. J. Immunol. 169, 3021–3029 10.4049/jimmunol.169.6.3021 [DOI] [PubMed] [Google Scholar]
- 24.Hao, Q.-L., George, A.A., Zhu, J., Barsky, L., Zielinska, E., Wang, X.et al. (2008) Human intrathymic lineage commitment is marked by differential CD7 expression: identification of CD7− lympho-myeloid thymic progenitors. Blood 111, 1318–1326 10.1182/blood-2007-08-106294 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Magri, M., Yatim, A., Benne, C., Balbo, M., Henry, A., Serraf, A.et al. (2009) Notch ligands potentiate IL-7-driven proliferation and survival of human thymocyte precursors. Eur. J. Immunol. 39, 1231–1240 10.1002/eji.200838765 [DOI] [PubMed] [Google Scholar]
- 26.Dik, W.A., Pike-Overzet, K., Weerkamp, F., de Ridder, D., de Haas, E.F., Baert, M.R.et al. (2005) New insights on human T cell development by quantitative T cell receptor gene rearrangement studies and gene expression profiling. J. Exp. Med. 201, 1715–1723 10.1084/jem.20042524 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Weerkamp, F., Baert, M.R.M., Brugman, M.H., Dik, W.A., de Haas, E.F.E., Visser, T.P.et al. (2006) Human thymus contains multipotent progenitors with T/B lymphoid, myeloid, and erythroid lineage potential. Blood 107, 3131–3137 10.1182/blood-2005-08-3412 [DOI] [PubMed] [Google Scholar]
- 28.Canté-Barrett, K., Mendes, R.D., Li, Y., Vroegindeweij, E., Pike-Overzet, K., Wabeke, T.et al. (2017) Loss of CD44dim expression from early progenitor cells marks T-cell lineage commitment in the human thymus. Front. Immunol. 8, 32 10.3389/fimmu.2017.00032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ciofani, M. and Zúñiga-Pflücker, J.C. (2010) Determining γδ versus αβ T cell development. Nat. Rev. Immunol. 10, 657–663 10.1038/nri2820 [DOI] [PubMed] [Google Scholar]
- 30.von Boehmer, H. (2005) Unique features of the pre-T-cell receptor alpha-chain: not just a surrogate. Nat. Rev. Immunol. 5, 571–577 10.1038/nri1636 [DOI] [PubMed] [Google Scholar]
- 31.Yamasaki, S., Ishikawa, E., Sakuma, M., Ogata, K., Sakata-Sogawa, K., Hiroshima, M.et al. (2006) Mechanistic basis of pre-T cell receptor-mediated autonomous signaling critical for thymocyte development. Nat. Immunol. 7, 67–75 10.1038/ni1290 [DOI] [PubMed] [Google Scholar]
- 32.Carpenter, A.C. and Bosselut, R. (2010) Decision checkpoints in the thymus. Nat. Immunol. 11, 666–673 10.1038/ni.1887 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Dutta, A., Zhao, B. and Love, P.E. (2021) New insights into TCR β-selection. Trends Immunol. 42, 735–750 10.1016/j.it.2021.06.005 [DOI] [PubMed] [Google Scholar]
- 34.Taghon, T., Van de Walle, I., De Smet, G., De Smedt, M., Leclercq, G., Vandekerckhove, B.et al. (2009) Notch signaling is required for proliferation but not for differentiation at a well-defined β-selection checkpoint during human T-cell development. Blood 113, 3254–3263 10.1182/blood-2008-07-168906 [DOI] [PubMed] [Google Scholar]
- 35.Yannoutsos, N., Wilson, P., Yu, W., Chen, H.T., Nussenzweig, A., Petrie, H.et al. (2001) The role of recombination activating gene (RAG) reinduction in thymocyte development in vivo. J. Exp. Med. 194, 471–480 10.1084/jem.194.4.471 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Krangel, M.S. (2009) Mechanics of T cell receptor gene rearrangement. Curr. Opin. Immunol. 21, 133–139 10.1016/j.coi.2009.03.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sohn, S.J., Thompson, J. and Winoto, A. (2007) Apoptosis during negative selection of autoreactive thymocytes. Curr. Opin. Immunol. 19, 510–515 10.1016/j.coi.2007.06.001 [DOI] [PubMed] [Google Scholar]
- 38.Takaba, H. and Takayanagi, H. (2017) The mechanisms of T cell selection in the thymus. Trends Immunol. 38, 805–816 10.1016/j.it.2017.07.010 [DOI] [PubMed] [Google Scholar]
- 39.Velten, L., Haas, S.F., Raffel, S., Blaszkiewicz, S., Islam, S., Hennig, B.P.et al. (2017) Human haematopoietic stem cell lineage commitment is a continuous process. Nat. Cell Biol. 19, 271–281 10.1038/ncb3493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Notta, F., Zandi, S., Takayama, N., Dobson, S., Gan, O.I., Wilson, G.et al. (2016) Distinct routes of lineage development reshape the human blood hierarchy across ontogeny. Science 351, aab2116 10.1126/science.aab2116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Laurenti, E. and Göttgens, B. (2018) From haematopoietic stem cells to complex differentiation landscapes. Nature 553, 418–426 10.1038/nature25022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yamamoto, R., Morita, Y., Ooehara, J., Hamanaka, S., Onodera, M., Rudolph, K.L.et al. (2013) Clonal analysis unveils self-renewing lineage-restricted progenitors generated directly from hematopoietic stem cells. Cell 154, 1112–1126 10.1016/j.cell.2013.08.007 [DOI] [PubMed] [Google Scholar]
- 43.Wilson, A., Laurenti, E., Oser, G., van der Wath, R.C., Blanco-Bose, W., Jaworski, M.et al. (2008) Hematopoietic stem cells reversibly switch from dormancy to self-renewal during homeostasis and repair. Cell 135, 1118–1129 10.1016/j.cell.2008.10.048 [DOI] [PubMed] [Google Scholar]
- 44.Majeti, R., Park, C.Y. and Weissman, I.L. (2007) Identification of a hierarchy of multipotent hematopoietic progenitors in human cord blood. Cell Stem Cell 1, 635–645 10.1016/j.stem.2007.10.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Notta, F., Doulatov, S., Laurenti, E., Poeppl, A., Jurisica, I. and Dick, J.E. (2011) Isolation of single human hematopoietic stem cells capable of long-term multilineage engraftment. Science 333, 218–221 10.1126/science.1201219 [DOI] [PubMed] [Google Scholar]
- 46.Dorshkind, K., Höfer, T., Montecino-Rodriguez, E., Pioli, P.D. and Rodewald, H.-R. (2020) Do haematopoietic stem cells age? Nat. Rev. Immunol. 20, 196–202 10.1038/s41577-019-0236-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Geiger, H. and Van Zant, G. (2002) The aging of lympho-hematopoietic stem cells. Nat. Immunol. 3, 329–333 10.1038/ni0402-329 [DOI] [PubMed] [Google Scholar]
- 48.de Haan, G. and Lazare, S.S. (2018) Aging of hematopoietic stem cells. Blood 131, 479–487 10.1182/blood-2017-06-746412 [DOI] [PubMed] [Google Scholar]
- 49.Gragert, L., Eapen, M., Williams, E., Freeman, J., Spellman, S., Baitty, R.et al. (2014) HLA match likelihoods for hematopoietic stem-cell grafts in the U.S. registry. N. Engl. J. Med. 371, 339–348 10.1056/NEJMsa1311707 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Boitano, A.E., Wang, J., Romeo, R., Bouchez, L.C., Parker, A.E., Sutton, S.E.et al. (2010) Aryl hydrocarbon receptor antagonists promote the expansion of human hematopoietic stem cells. Science 329, 1345–1348 10.1126/science.1191536 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fares, I., Chagraoui, J., Gareau, Y., Gingras, S., Rjean, R., Csaszar, E.et al. (2013) UM171 is a novel and potent agonist Of human hematopoietic stem cell renewal. Blood 122, 798. - 10.1182/blood.V122.21.798.798 [DOI] [Google Scholar]
- 52.Fares, I., Chagraoui, J., Gareau, Y., Gingras, S., Ruel, R., Mayotte, N.et al. (2014) Pyrimidoindole derivatives are agonists of human hematopoietic stem cell self-renewal. Science 345, 1509–1512 10.1126/science.1256337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wagner JE, J., Brunstein, C.G., Boitano, A.E., DeFor, T.E., McKenna, D., Sumstad, D.et al. (2016) Phase I/II trial of stemRegenin-1 expanded umbilical cord blood hematopoietic stem cells supports testing as a stand-alone graft. Cell Stem Cell 18, 144–155 10.1016/j.stem.2015.10.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Chagraoui, J., Girard, S., Spinella, J.-F., Simon, L., Bonneil, E., Mayotte, N.et al. (2021) UM171 preserves epigenetic marks that are reduced in Ex vivo culture of human HSCs via potentiation of the CLR3-KBTBD4 complex. Cell Stem Cell 28, 48–62.e6 10.1016/j.stem.2020.12.002 [DOI] [PubMed] [Google Scholar]
- 55.Takahashi, K., Tanabe, K., Ohnuki, M., Narita, M., Ichisaka, T., Tomoda, K.et al. (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 10.1016/j.cell.2007.11.019 [DOI] [PubMed] [Google Scholar]
- 56.Robinton, D.A. and Daley, G.Q. (2012) The promise of induced pluripotent stem cells in research and therapy. Nature 481, 295–305 10.1038/nature10761 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bak, R.O., Dever, D.P. and Porteus, M.H. (2018) CRISPR/cas9 genome editing in human hematopoietic stem cells. Nat. Protoc. 13, 358–376 10.1038/nprot.2017.143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ueno, T., Liu, C., Nitta, T. and Takahama, Y. (2005) Development of T-lymphocytes in mouse fetal thymus organ culture. Methods Mol. Biol. 290, 117–133 10.1385/1-59259-838-2:117 [DOI] [PubMed] [Google Scholar]
- 59.Robin, C., Bennaceur-Griscelli, A., Louache, F., Vainchenker, W. and Coulombel, L. (1999) Identification of human T-lymphoid progenitor cells in CD34+ CD38low and CD34+ CD38+ subsets of human cord blood and bone marrow cells using NOD-SCID fetal thymus organ cultures. Br. J. Haematol. 104, 809–819 10.1046/j.1365-2141.1999.01266.x [DOI] [PubMed] [Google Scholar]
- 60.Plum, J., De Smedt, M., Defresne, M., Leclercq, G. and Vandekerckhove, B. (1994) Human CD34+ fetal liver stem cells differentiate to T cells in a mouse thymic microenvironment. Blood 84, 1587–1593 10.1182/blood.V84.5.1587.1587 [DOI] [PubMed] [Google Scholar]
- 61.Blom, B., Res, P., Noteboom, E., Weijer, K. and Spits, H. (1997) Prethymic CD34+ progenitors capable of developing into T cells are not committed to the T cell lineage. J. Immunol. 158, 3571–3577 PMID: [PubMed] [Google Scholar]
- 62.Offner, F., Kerre, T., De Smedt, M. and Plum, J. (1999) Bone marrow CD34+ cells generate fewer T cells in vitro with increasing age and following chemotherapy. Br. J. Haematol. 104, 801–808 10.1046/j.1365-2141.1999.01265.x [DOI] [PubMed] [Google Scholar]
- 63.Signer, R.A.J., Montecino-Rodriguez, E., Witte, O.N. and Dorshkind, K. (2008) Aging and cancer resistance in lymphoid progenitors are linked processes conferred by p16Ink4a and Arf. Genes Dev. 22, 3115–3120 10.1101/gad.1715808 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Weekx, S.F.A., Snoeck, H.W., Offner, F., De Smedt, M., Van Bockstaele, D.R., Nijs, G.et al. (2000) Generation of T cells from adult human hematopoietic stem cells and progenitors in a fetal thymic organ culture system: stimulation by tumor necrosis factor-α. Blood 95, 2806–2812 10.1182/blood.V95.9.2806.009k01_2806_2812 [DOI] [PubMed] [Google Scholar]
- 65.Schmitt, T.M. and Zuniga-Pflucker, J.C. (2002) Induction of T cell development from hematopoietic progenitor cells by delta-like-1 in vitro. Immunity 17, 749–756 10.1016/S1074-7613(02)00474-0 [DOI] [PubMed] [Google Scholar]
- 66.Koch, U., Fiorini, E., Benedito, R., Besseyrias, V., Schuster-Gossler, K., Pierres, M.et al. (2008) Delta-like 4 is the essential, nonredundant ligand for Notch1 during thymic T cell lineage commitment. J. Exp. Med. 205, 2515–2523 10.1084/jem.20080829 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Besseyrias, V., Fiorini, E., Strobl, L.J., Zimber-Strobl, U., Dumortier, A., Koch, U.et al. (2007) Hierarchy of Notch-Delta interactions promoting T cell lineage commitment and maturation. J. Exp. Med. 204, 331–343 10.1084/jem.20061442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Taghon, T.N., David, E.S., Zúñiga-Pflücker, J.C. and Rothenberg, E.V. (2005) Delayed, asynchronous, and reversible T-lineage specification induced by Notch/Delta signaling. Genes Dev. 19, 965–978 10.1101/gad.1298305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.De Smedt, M., Leclercq, G., Vandekerckhove, B., Kerre, T., Taghon, T. and Plum, J. (2011) T-lymphoid differentiation potential measured in vitro is higher in CD34+CD38-/lo hematopoietic stem cells from umbilical cord blood than from bone marrow and is an intrinsic property of the cells. Haematologica 96, 646–654 10.3324/haematol.2010.036343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Awong, G., Herer, E., Surh, C.D., Dick, J.E., La Motte-Mohs, R.N. and Zúñiga-Pflücker, J.C. (2009) Characterization in vitro and engraftment potential in vivo of human progenitor T cells generated from hematopoietic stem cells. Blood 114, 972–982 10.1182/blood-2008-10-187013 [DOI] [PubMed] [Google Scholar]
- 71.Awong, G., Singh, J., Mohtashami, M., Malm, M., La Motte-Mohs, R.N., Benveniste, P.M.et al. (2013) Human proT-cells generated in vitro facilitate hematopoietic stem cell-derived T-lymphopoiesis in vivo and restore thymic architecture. Blood 122, 4210–4219 10.1182/blood-2012-12-472803 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Vodyanik, M.A., Bork, J.A., Thomson, J.A. and Slukvin, I.I. (2005) Human embryonic stem cell–derived CD34+ cells: efficient production in the coculture with OP9 stromal cells and analysis of lymphohematopoietic potential. Blood 105, 617–626 10.1182/blood-2004-04-1649 [DOI] [PubMed] [Google Scholar]
- 73.Vodyanik, M.A., Thomson, J.A. and Slukvin, I.I. (2006) Leukosialin (CD43) defines hematopoietic progenitors in human embryonic stem cell differentiation cultures. Blood 108, 2095–2105 10.1182/blood-2006-02-003327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Timmermans, F., Velghe, I., Vanwalleghem, L., De Smedt, M., Van Coppernolle, S., Taghon, T.et al. (2009) Generation of T cells from human embryonic stem cell-derived hematopoietic zones. J. Immunol. 182, 6879–6888 10.4049/jimmunol.0803670 [DOI] [PubMed] [Google Scholar]
- 75.Ditadi, A. and Sturgeon, C.M. (2016) Directed differentiation of definitive hemogenic endothelium and hematopoietic progenitors from human pluripotent stem cells. Methods 101, 65–72 10.1016/j.ymeth.2015.10.001 [DOI] [PubMed] [Google Scholar]
- 76.Sturgeon, C.M., Ditadi, A., Awong, G., Kennedy, M. and Keller, G. (2014) Wnt signaling controls the specification of definitive and primitive hematopoiesis from human pluripotent stem cells. Nat. Biotechnol. 32, 554–561 10.1038/nbt.2915 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Themeli, M., Kloss, C.C., Ciriello, G., Fedorov, V.D., Perna, F., Gonen, M.et al. (2013) Generation of tumor-targeted human T lymphocytes from induced pluripotent stem cells for cancer therapy. Nat. Biotechnol. 31, 928–933 10.1038/nbt.2678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Themeli, M., Chhatta, A., Boersma, H., Prins, H.J., Cordes, M., de Wilt, E.et al. (2020) iPSC-based modeling of RAG2 severe combined immunodeficiency reveals multiple T cell developmental arrests. Stem Cell Rep. 14, 300–311 10.1016/j.stemcr.2019.12.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Seet, C.S., He, C., Bethune, M.T., Li, S., Chick, B., Gschweng, E.H.et al. (2017) Generation of mature T cells from human hematopoietic stem and progenitor cells in artificial thymic organoids. Nat. Methods 14, 521–530 10.1038/nmeth.4237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Bosticardo, M., Pala, F., Calzoni, E., Delmonte, O.M., Dobbs, K., Gardner, C.L.et al. (2020) Artificial thymic organoids represent a reliable tool to study T-cell differentiation in patients with severe T-cell lymphopenia. Blood Adv. 4, 2611–2616 10.1182/bloodadvances.2020001730 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Montel-Hagen, A., Seet, C.S., Li, S., Chick, B., Zhu, Y., Chang, P.et al. (2019) Organoid-Induced differentiation of conventional T cells from human pluripotent stem cells. Cell Stem Cell 24, 376–389 e8 10.1016/j.stem.2018.12.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Pajcini, K.V., Speck, N.A. and Pear, W.S. (2011) Notch signaling in mammalian hematopoietic stem cells. Leukemia 25, 1525–1532 10.1038/leu.2011.127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Benveniste, P., Serra, P., Dervovic, D., Herer, E., Knowles, G., Mohtashami, M.et al. (2014) Notch signals are required for in vitro but not in vivo maintenance of human hematopoietic stem cells and delay the appearance of multipotent progenitors. Blood 123, 1167–1177 10.1182/blood-2013-07-505099 [DOI] [PubMed] [Google Scholar]
- 84.Shukla, S., Langley, M.A., Singh, J., Edgar, J.M., Mohtashami, M., Zúñiga-Pflücker, J.C.et al. (2017) Progenitor T-cell differentiation from hematopoietic stem cells using delta-like-4 and VCAM-1. Nat. Methods 14, 531–538 10.1038/nmeth.4258 [DOI] [PubMed] [Google Scholar]
- 85.Smits, K., De Smedt, M., Naessens, E., De Smet, G., Stove, V., Taghon, T.et al. (2007) Tumor necrosis factor promotes T-cell at the expense of B-cell lymphoid development from cultured human CD34+ cord blood cells. Exp. Hematol. 35, 1272–1278 10.1016/j.exphem.2007.04.009 [DOI] [PubMed] [Google Scholar]
- 86.Moirangthem, R.D., Ma, K., Lizot, S., Cordesse, A., Olivré, J., de Chappedelaine, C.et al. (2021) A DL-4- and TNFα-based culture system to generate high numbers of nonmodified or genetically modified immunotherapeutic human T-lymphoid progenitors. Cell. Mol. Immunol. 18, 1662–1676 10.1038/s41423-021-00706-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Trotman-Grant, A.C., Mohtashami, M., De Sousa Casal, J., Martinez, E.C., Lee, D., Teichman, S.et al. (2021) DL4-μbeads induce T cell lineage differentiation from stem cells in a stromal cell-free system. Nat. Commun. 12, 5023 10.1038/s41467-021-25245-8 [DOI] [PMC free article] [PubMed] [Google Scholar]