Abstract
Hematopoietic progenitor cells from Fanconi anemia (FA) group C (FA-C) patients display hypersensitivity to the apoptotic effects of gamma interferon (IFN-γ) and constitutively express a variety of IFN-dependent genes. Paradoxically, however, STAT1 activation is suppressed in IFN-stimulated FA cells, an abnormality corrected by transduction of normal FANCC cDNA. We therefore sought to define the specific role of FANCC protein in signal transduction through receptors that activate STAT1. Expression and phosphorylation of IFN-γ receptor α chain (IFN-γRα) and JAK1 and JAK2 tyrosine kinases were equivalent in both normal and FA-C cells. However, in coimmunoprecipitation experiments STAT1 did not dock at the IFN-γR of FA-C cells, an abnormality corrected by transduction of the FANCC gene. In addition, glutathione S-transferase fusion genes encoding normal FANCC but not a mutant FANCC bearing an inactivating point mutation (L554P) bound to STAT1 in lysates of IFN-γ-stimulated B cells and IFN-, granulocyte-macrophage colony-stimulating factor- and stem cell factor-stimulated MO7e cells. Kinetic studies revealed that the initial binding of FANCC was to nonphosphorylated STAT1 but that subsequently the complex moved to the receptor docking site, at which point STAT1 became phosphorylated. The STAT1 phosphorylation defect in FA-C cells was functionally significant in that IFN induction of IFN response factor 1 was suppressed and STAT1-DNA complexes were not detected in nuclear extracts of FA-C cells. We also determined that the IFN-γ hypersensitivity of FA-C hematopoietic progenitor cells does not derive from STAT1 activation defects because granulocyte-macrophage CFU and erythroid burst-forming units from STAT1−/− mice were resistant to IFN-γ. However, BFU-E responses to SCF and erythropoietin were suppressed in STAT−/− mice. Consequently, because the FANCC protein is involved in the activation of STAT1 through receptors for at least three hematopoietic growth and survival factor molecules, we reason that FA-C hematopoietic cells are excessively apoptotic because of an imbalance between survival cues (owing to a failure of STAT1 activation in FA-C cells) and apoptotic and mitogenic inhibitory cues (constitutively activated in FA-C cells in a STAT1-independent fashion).
Gamma interferon (IFN-γ) induces transcription of a distinct set of genes by activating STAT1, one member of a family of latent cytoplasmic transcription factors that are activated via phosphorylation on tyrosine residues (6). The IFN-γ receptor (IFN-γR) lacks intrinsic tyrosine kinase activity, but on ligand binding, receptor multimerization results in reciprocal activation of JAK1 and JAK2, two Janus tyrosine kinases noncovalently attached to the IFN-γR α and β chains (2, 20, 22). Phosphorylation of the tyrosine residue on IFN-γRα by JAK molecules creates a docking site for the Src homology 2 domain of STAT1, which is then phosphorylated on tyrosine and ultimately forms a homodimeric transcription factor that translocates to the nucleus (6, 21). The factors that govern the traffic of cytoplasmic STAT molecules to the docking site on the IFN-γR are unknown. We have recently found that the activation of STAT1 in response to IFN-γ is suppressed in hematopoietic cells from children with Fanconi anemia of type C (FA-C) and in mice nullizygous at the FA-C locus. However, in the ground state (uninduced by IFN), IFN response factor 1 (IRF-1) is expressed at high levels in mutant FA-C cells (35), suggesting that a non-STAT1 pathway is involved in constitutive activation of IRF-1 in FA cells. In addition, complementation of the defect by retrovirus mediated transfer of normal FANCC cDNA reconstitutes the normal STAT1 response (10, 38).
Linkage of FANCC function with that of STAT1 provided us with an opportunity to test whether the relationship of these two molecules was direct or indirect. We report herein results of experiments in which the assembly of the fully activated IFN-γR complex, including STAT1, JAK1, and JAK2, was examined in isogenic murine and human FA-C cells. We report that in IFN-γ-stimulated FA-C cells, phosphorylation of JAK1, JAK2, and IFN-γRα occurs normally, but STAT1 does not dock at the receptor α chain. In FA-C cells nuclear STAT1 is reduced, and IFN fails to induce STAT1-specific DNA-binding complexes and expression of IRF-1. Expression of the normal FANCC cDNA in mutant cells results in normal STAT1 docking and phosphorylation as well as normal induction of nuclear STAT1-DNA complex and normal induction of IRF-1. We also find that a variety of cytokines and hematopoietic growth factors stimulate the association of STAT1 with glutathione S-transferase (GST) fusion proteins encoding the normal FANCC but not a naturally occurring inactivating mutant FANCC (L554P) and that the association occurs rapidly and prior to STAT1 phosphorylation on Y701. Coimmunoprecipitation experiments confirmed the IFN-inducible association of endogenous FANCC and STAT1 in normal but not mutant cells. We propose that FANCC is a control factor for STAT1 traffic to receptor docking sites, and that mutations that inactivate FANCC function interdict this function. We propose that the hematopoietic defects in FA derive, at least in part, from a loss of balance between mitogenic cues (owing to a reduction of signals transduced through receptors for growth factors that activate STAT1) and mitogenic inhibitory cues (secondary to FANCC-dependent, STAT1-independent constitutive activation of mitotic inhibitory factors), in particular IRF-1, a factor known to effect apoptotic responses in hematopoietic progenitor cells (43).
MATERIALS AND METHODS
Cell cultures and IFN-γ stimulation.
Epstein-Barr virus-transformed lymphoblasts were maintained in RPMI 1640 medium (RPMI; Life Technologies, Grand Island, N.Y.) supplemented with 15% heat-inactivated fetal calf serum and grown in a humidified 5% CO2-containing atmosphere at 37°C. The lymphoblast lines JY and HSC536N (FA-C) have been described elsewhere (38). Unless otherwise indicated, cells were stimulated with recombinant human IFN-γ (1 ng/ml; R & D Systems, Minneapolis, Minn.) for the indicated time periods. For detection of STAT1 phosphorylation, cells were serum starved for 2 h prior to IFN-γ treatment. The leukemia cell line MO7e and its high erythropoietin receptor (EPOR)-expressing subline MO7ER (51) were maintained in RPMI containing 20% fetal calf serum and granulocyte-macrophage colony-stimulating factor (GM-CSF; 100 U/ml; Genetics Institute, Cambridge, Mass.) or EPO (2 U/ml; Amgen Inc., Thousand Oaks, Calif.). Before stimulation, cells were washed and incubated for 18 h at 37°C in serum-free RPMI containing 0.5% bovine serum albumin (Sigma Chemical Co., St. Louis, Mo.). After starvation, cells were washed and then exposed to IFN-γ (10 ng/ml), IFN-α (20,000 U/ml; Schering Corp., Kenilworth, N.J.), GM-CSF (100 U/ml), SCF (50 ng/ml; R & D Systems), and EPO (40 U/ml) for 10 min.
Retroviral infection and transduction of FA cell lines.
FANCC cDNA (47) was subcloned into the retroviral vector pLXSN as described previously (38). Plasmid pLXSN-FANCC (10 μg) was transfected by the calcium phosphate precipitation method into PA12 producer cells expressing the vesicular stomatitis virus G envelope protein (26). Retroviral supernatants were collected and used for transduction of FA-C cell lymphoblasts. HSC536N cells were exposed to the filtered supernatants containing virus in the presence of Polybrene (8 μg/ml; Sigma). Sets of isogenic lines were selected in G418 and then exposed to multiple doses of mitomycin C in cytotoxicity (trypan blue viability) and apoptosis (TUNEL [terminal deoxynucleotidyltransferase-mediated dUTP-biotin nick end labeling] and annexin V binding) assays to ensure that the mutant cells transduced with the normal cDNA were fully complemented. HSC5326N/FANCC cells are HS536N cells transduced by the FANCE vector. HSC536N/neo cells are HS536N cells transduced by the vector alone.
Preparation of whole-cell lysates (WCL).
Following treatment with IFN-γ, cells were washed twice with ice-cold phosphate-buffered saline (PBS) and lysed with either radioimmunoprecipitation assay (RIPA) buffer (150 mM NaCl, 10 mM Tris-HCl [pH 7.6], 1% sodium deoxycholate, 1% Triton X-100, 0.1% sodium dodecyl sulfate [SDS]) or Nonidet P-40 (NP-40) lysis buffer (1% NP-40, 20 mM Tris-HCl [pH 8.0], 137 mM NaCl, 10% glycerol). Both lysis buffers were supplemented with the following freshly made protease inhibitors: 1% aprotinin, 1 μg of leupeptin per ml, 1 mM phenylmethylsulfonyl fluoride (PMSF), and 2 mM sodium orthovanadate. For cell lysates used specifically for assessment of protein-protein interactions, cells were lysed in digitonin lysis buffer (1% digitonin, 50 mM Tris-HCl [pH 8.0], 150 mM NaCl, 50 mM NaF, 5 mM sodium pyrophosphate) containing the protease inhibitors aprotinin, leupeptin, and PMSF. After rocking at 4°C for 20 min (60 min when cells were lysed with digitonin buffer), cell lysates were cleared by centrifugation at 13,200 rpm for 20 min at 4°C, and protein concentrations were determined by the Bradford method (4) using a protein microassay reagent (Bio-Rad, Hercules, Calif.).
Electromobility shift assay (EMSA).
Nuclear extracts were prepared by the method of Dignam et al. (8). A human IRF-1 IFN-γ activation sequence (GAS) oligonucleotide (5′-ACAACAGCCTGATTTCCCCGAA-3′) was synthesized in the Molecular Biology Core Lab (Portland VA Medical Center) and then labeled with [γ-32P]ATP to 2.5 × 104 cpm/ng, using T4 polynucleotide kinase (Boehringer Mannheim). The binding reaction mixture (20 μl) contained 5.0 to 7.5 μg of nuclear extracts, 0.2 ng of labeled oligonucleotide 2 μg of poly(dl-dC), and 10 μg of bovine serum albumin in 10 mM Tris-Cl (pH 7.4)–50 mM NaCl–1 mM dithiothreitol (DTT)–1 mM EDTA–10% glycerol. Reaction mixtures were incubated at room temperature for 30 min and then resolved on a 4% polyacrylamide gel in 25 mM Tris, 190 mM glycine, and 1 mM EDTA. Gels were dried and autoradiographed with intensifying screens at −80°C.
Reverse transcription-PCR.
Total RNA was isolated from cells using Tri Reagent (Molecular Research Center, Inc., Cincinnati, Ohio) in accordance with the manufacturer's instructions. First-strand cDNA was reverse transcribed from the indicated RNA using random hexanucleotide primers (Gibco BRL) and mouse mammary tumor virus RNase H− reverse transcriptase (Gibco BRL) as previously described (46). The cDNA was then amplified by PCR for 35 cycles (denatured at 94°C for 30 s, primer annealed at 53°C for 30 s, and primer extended at 72°C for 30 s). Control reactions in which no cDNA (i.e., water only) was added to the PCR mixture were analyzed with each experiment. For IRF-1 amplification, the primers used were 5′-GAGCTGGGCCATTCACACAG-3′ and 5′-CATGGCGACAGTGCTGGAGT-3′, which produced an amplimer with a predicted length of 390 nucleotides. The amplification products were separated by electrophoresis on 1.5% agarose, stained with ethidium bromide, and visualized by UV transillumination.
Antibodies.
Antibodies used included antiphosphotyrosine (anti-P-Tyr) antibody 4G10, provided by Brian Druker (Oregon Health Sciences University, Portland), anti-JAK1 monoclonal antiserum (Transduction Laboratories, Lexington, Ky.), anti-JAK2 polyclonal antiserum (Upstate Biotechnology, New York, N.Y.), anti-IFN-γRα polyclonal antibody (Antigenix America Antibodies, New York, N.Y.), anti-STAT1α monoclonal antiserum from Santa Cruz Biotechnology (Santa Cruz, Calif.), rabbit polyclonal anti-Stat3, anti-Stat5, and antibodies specific for the tyrosine-phosphorylated forms of STAT1 (P-STAT1), Stat3, and Stat5 (New England Biolabs, Beverly, Mass.), anti-FANCC polyclonal antiserum as described previously (52), and sheep anti-mouse and donkey anti-rabbit horseradish peroxidase-conjugated antibody (Amersham, Arlington Heights, Ill.). All antibodies were used in accordance with the manufacturer's instructions.
Immunoprecipitation.
WCLs (about 1 mg of total proteins) were precleared with 50 μl of 50% protein A-Sepharose suspension (Pharmacia Biotech, Inc.) for 1 h at 4°C. After separation of the protein A-Sepharose from the lysate by centrifugation at 1,000 rpm for 1 min at 4°C, the lysate was incubated with either anti-P-Tyr antibody 4G10 (12 μg/ml), anti-IFN-γRα (2 μg/ml), or anti-STAT1 (1 μg/ml) for 3 to 5 h at 4°C with constant agitation. Immunocomplexes were then bound to protein A-Sepharose beads (50 μl of 50% slurry) during a 1- to 2-h incubation at 4°C. The immunoprecipitates were recovered by centrifuging at 1,000 rpm for 1 min at 4°C. If cells were lysed in NP-40 lysis buffer, the immunocomplexes were washed three times with 0.05% NP-40 wash buffer (0.05% NP-40 in 1× PBS). If cells were lysed in digitonin lysis buffer, the immunocomplexes were then washed with 0.1% digitonin wash buffer (same as digitonin lysis buffer except that the concentration of digitonin was 0.1%).
Immunoblotting.
WCL, nuclear extracts, or immunocomplexes were prepared as above and subjected to immunoblot analyses. Samples were heated at 94°C for 5 min in 2× Laemmli SDS sample buffer, separated by SDS-polyacrylamide gel electrophoresis (PAGE), and transferred onto Bio-Blot nitrocellulose membranes (Costar, Cambridge, Mass.). Nonspecific binding sites on the membranes were blocked with Tris-buffered saline plus 0.05% Tween 20 containing 5% nonfat dry milk for 1 h at room temperature or overnight at 4°C. Immunoblots were subsequently incubated with the indicated primary antibodies at a dilution according to the manufacturer's instruction. After stringent washing, the blots were incubated with appropriate secondary antibodies (linked to horseradish peroxidase) for 30 min at room temperature and developed by using an enhanced chemiluminescence kit (Amersham).
GST-FANCC fusion protein purification and in vitro binding assays.
The GST-FANCC and GST-L554P fusion constructs were created by insertion of the entire open reading frame of FA-C and its mutant version, L554P, into the BamHI and SmaI sites of yeast overexpression vector pGST (31). The plasmids were transformed into yeast strain Sc334 (19) using a Frozen-EZ yeast transformation kit (ZYMO Research, Orange, Calif.). Expression and purification of the yeast GST fusion proteins were performed as described elsewhere (31, 32), with minor modifications. The GST fusion protein was prepared by suspending the yeast cells containing pGST, pGST-FANCC, or pGST-L554P in an equal volume of buffer A (37.5 mM K2PO4 [pH 9.5], 7.5 mM DTT, 1.5 mM EDTA, 1 M NaCl, 2.5% glycerol, 7.5% ethylene glycol, 0.03% silicon antifoam), and the cells were broken by vortexing with an equal volume of glass beads (eight 15-s bursts). The lysate was removed from the beads, and Triton X-100 was added to 1%. After being clarified twice by centrifugation, the supernatant was incubated for 30 min at 4°C with 0.1 ml of glutathione-Sepharose 4B (Pharmacia Biotech, Piscataway, N.J.) 50% slurry equilibrated with buffer B (50 mM KPO4 [pH 7.2], 1 mM DTT, 0.5 mM EDTA, 1 M NaCl, 10% glycerol, 1% Triton X-100). The Sepharose beads were washed three times with buffer B containing 0.5 M NaCl. For binding, about 10 μg of GST, GST-FANCC, or GST-L554P fusion protein bound to glutathione-Sepharose beads was incubated with WCL from untreated or IFN-γ-treated JY cells (1 mg of total proteins) prepared in 1% digitonin buffer supplemented with 1% aprotinin, 1 μg of leupeptin per ml, 1 mM PMSF, and 2 mM sodium orthovanadate. The final concentration of digitonin in the binding mixture was adjusted to 0.5%. After shaking at 4°C for 40 min, the beads were recovered and washed three times with 0.1% digitonin buffer before boiling in Laemmli SDS sample buffer and immunoblotting as described above. For WCL input controls, 60 to 100 μg of total WCL proteins was loaded in each lane.
In vivo phosphate labeling.
Cells were starved for 90 min in phosphate-free RPMI containing 15% dialyzed fetal bovine serum, and [32P]orthophosphate (150 μCi/ml; DuPont NEN) was added to the medium. After labeling for 3 h, cells were incubated with IFN-γ (10 ng/ml) for the indicated time points. WCL prepared as described above were subjected to affinity precipitation with GST-FANCC and analyzed by SDS-PAGE followed by autoradiography. Western blot analysis was performed on these precipitates to determine the quantity of both tyrosine-phosphorylated and total STAT1 bound to GST-FANCC.
Murine clonal BM cell cultures.
Bone marrow (BM) samples were obtained from an equal number of FANCC−/− and FANCC−/+ mice (55) and STAT−/− and STAT+/− mice (9, 28) for each experiment. Femoral marrow samples obtained from mice after cervical dislocation were washed and resuspended in RPMI medium for viable counts (trypan blue dye exclusion). Unfractionated BM cells (105) were cultured in 1 ml of methoCult H4230 (Stem Cell Technologies, Vancouver, British Columbia, Canada), penicillin-streptomycin (Life Technologies), and either murine SCF (from 0.10 to 10 ng/ml; R & D Systems), murine EPO (2 ng/ml), or both SCF and EPO. A combination of three growth factors was used as a positive control: murine Steel factor (10 ng/ml; R & D Systems), murine interleukin-3 (IL-3; 10 ng/ml; R & D Systems), and human EPO (2 U/ml; Amgen). Granulocyte-macrophage CFU (CFU-GM) were cultured in 35-mm-diameter tissue culture dishes at 37°C in 5% CO2 in air and counted after 7 and 14 days, using a dissecting microscope. Colony growth results were expressed as mean (of triplicate plates) colonies per plate ± standard deviation (SD). Between-group comparisons were made using one-way analysis of variance.
RESULTS
Deficient STAT1α activation in FA-C cells.
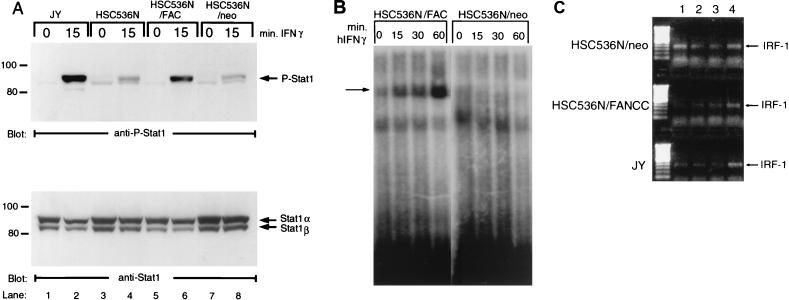
We have recently shown that IFN-γ-induced STAT1 tyrosine phosphorylation is reduced in FA-C cells (38). Equal numbers of cells from each isogenic line were serum starved for 2 h followed by stimulation with IFN-γ at 37°C for the indicated time periods. WCL were prepared in RIPA buffer (see Materials and Methods) and analyzed by immunoblotting with antisera specific to P-STAT1. As shown in Fig. 1, the FA-C mutant cell lines HSC536N and HSC536N/neo contained less P-STAT1 than the normal B-cell line JY following IFN-γ stimulation (Fig. 1A, lane 2 versus lanes 4 and 8). Introduction of the normal FANCC gene into FA-C cells (HSC536N/FANCC) corrected the STAT1 activation defect (Fig. 1A, lane 6), suggesting that STAT1 activation via tyrosine phosphorylation is dependent on expression of a normal FANCC gene product. Reprobing the blot with anti-STAT1 antibody showed that STAT1 protein was expressed at comparable levels in all four cell lines, as indicated in the lower panel.
FIG. 1.
Defective STAT1 tyrosine phosphorylation in IFN γ-stimulated FA-C cells. (A) Cells from the indicated FA-C isogenic lines were serum starved for 2 h, incubated with IFN-γ for the indicated time periods at 37°C, and then solubilized in RIPA lysis buffer as described in Materials and Methods. The WCL were separated by SDS-PAGE and blotted with antibodies to P-STAT1. (B) The blot described above, stripped and reprobed with anti-STAT1. Here and in subsequent figures, the label “FAC” represents FANCC, and the positions of migration of protein standards (in kilodaltons) are indicated on the left. (B) EMSA of STAT1 binding to oligonucleotides corresponding to the STAT1-specific GAS element in the IRF-1 promoter. Nuclear extracts were prepared from HSC536N/FANCC (lanes 1 to 4) and HSC536N (lanes 5 to 8) cells untreated (lanes 1 and 5) or treated with 1 ng of IFN-γ per ml (lanes 2 to 4 and 6 to 8). Arrow denotes STAT1-GAS complexes. (C) IFN-γ-induced expression of IRF-1 transcript. Horizontal rows represent amplification from individual cell lines as indicated. Vertical columns show samples from untreated cells at time zero (lane 1), untreated cells at 3 h (lane 2), cells treated with IFN-γ (0.5 ng/ml) for 3 h (lane 3), and cells treated with IFN-γ (50 ng/ml) for 3 h (lane 4).
To ensure that FA-C cells had reduced STAT1 function, we sought to identify nuclear STAT1 and found none at any time point from 5 to 120 min after IFN-γ stimulation (data not shown). Using an EMSA with a STAT1-specific oligonucleotide as a probe, we observed STAT1-DNA complexes in the nuclear extracts from IFN-stimulated HSC536/FANCC cells (Fig. 1B, lanes 1 to 4). In contrast, no constitutive or inducible STAT1-specific DNA-binding activity was detected in the nuclear extracts from mutant FA-C cells (Fig. 1B, lanes 5 to 8). In addition, IFN-γ induction of IRF-1 was also abolished in mutant cells (Fig. 1C) as we have described elsewhere (38).
Tyrosine phosphorylation of IFN-γRα in IFN-γ-stimulated B lymphocytes.
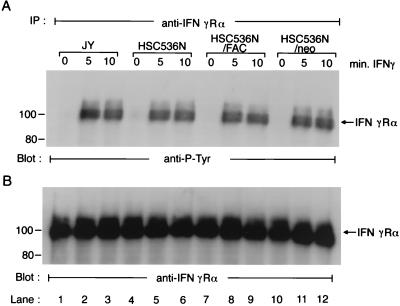
Cellular responsiveness to IFN-γ requires the activation of IFN-γRα through phosphorylation on tyrosine residues (12, 13, 22). We therefore asked whether ligand-induced IFN-γRα tyrosine phosphorylation was different quantitatively and temporally in FA-C cells. Cells were treated with IFN-γ for the indicated time, and lysates were incubated with anti-IFN-γRα antibodies. In the absence of IFN-γ, no tyrosine-phosphorylated IFN-γRα was detected in any of the isogenic cells (Fig. 2A, lanes 1, 4, 7, and 10). Following addition of ligand, equivalent IFN-γRα tyrosine phosphorylation was observed in all four isogenic cell lines (Fig. 2A, lanes 2, 3, 5, 6, 8, 9, 11, and 12). The kinetics of induced IFN-γRα tyrosine phosphorylation, which was maximal at 5 min and remained at maximal levels within 10 min of exposure to IFN-γ, was similar in all four cell lines. The amounts of the receptor proteins loaded in each lane were comparable, as determined by reprobing the membrane with anti-IFN-γRα antibody (Fig. 2B).
FIG. 2.
Activation of IFN-γRα in response to IFN-γ in FA-C cells. Cells were incubated with IFN-γ for the indicated time periods at 37°C and then solubilized in NP-40 lysis buffer as described in Materials and Methods. The WCL were immunoprecipitated (IP) with anti-IFN-γRα antibody. The immunoprecipitates were then analyzed by immunoblotting. (A) Cell lysates prepared from four FA-C isogenic lines were immunoprecipitated with anti-IFN-γRα and probed with anti-P-Tyr; (B) the blot described above was stripped and reprobed with anti-IFN-γRα.
Tyrosine phosphorylation of JAK1 and JAK2 kinases in FA-C cells.
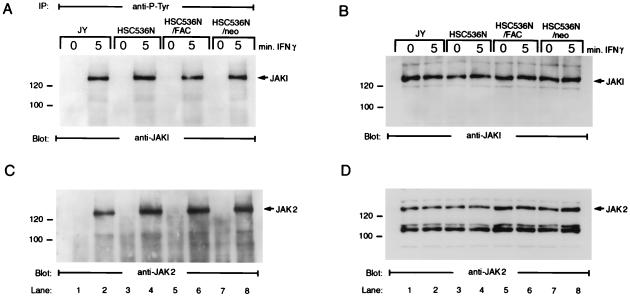
JAK1 and JAK2 are activated by IFN-γ through tyrosine phosphorylation, and these activated kinases play an essential role in transduction of the IFN-γ signal, at least in part, by phosphorylating STAT1 (25, 30, 33, 35, 39, 54). To determine whether IFN-γ-induced tyrosine phosphorylation of the JAK kinases was normal in FA-C cells, WCL prepared from normal and FA-C cells were first immunoprecipitated with anti-P-Tyr antibody 4G10 and then immunoblotted with antibodies against JAK1 and JAK2. As shown in Fig. 3, tyrosine phosphorylation of JAK1 (Fig. 3A) and JAK2 (Fig. 3C) was evident in all four isogenic cell lines after 5 min of IFN-γ treatment. Tyrosine phosphorylation of JAK1 or JAK2 was not detected in unstimulated cells (Fig. 3A and C, lanes 1, 3, 5, and 7). The kinetics of induced JAK phosphorylation were comparable in normal and FA-C cell lines. To ensure that expression of JAK1 and JAK2 proteins was equivalent in both normal and FA-C cells, we performed immunoblotting with anti-JAK1 and -JAK2 antibodies, using WCL from the same cell lines. As expected, both the untreated and IFN-γ-treated samples prepared from each cell line were found to contain similar levels of JAK1 and JAK2 (Fig. 3B and D). These results indicate that after ligation of IFN-γ to its receptor, activation, by tyrosine phosphorylation, of JAK1 and JAK2 was normal in FA-C cells.
FIG. 3.
IFN-γ-induced tyrosine phosphorylation of JAK1 and JAK2 in FA-C cells. Cells were stimulated with IFN-γ for various time periods and lysed in NP-40 lysis buffer. The WCL were either directly analyzed by immunoblotting with anti-JAK1 and -JAK2 antibodies or immunoprecipitated (IP) with anti-P-Tyr antibody, and the immunoprecipitates were then analyzed by immunoblotting. (A) Cell lysates prepared from four FA-C isogenic lines were immunoprecipitated with anti-P-Tyr and probed with anti-JAK1 antibody; (B) cell lysates were directly immunoblotted with anti-JAK1 antibody; (C) cell lysates prepared from FA-C isogenic lines were immunoprecipitated with anti-P-Tyr and probed with anti-JAK2 antibody; (D) cell lysates were directly immunoblotted with anti-JAK2 antibody.
IFN-γ-induced coimmunoprecipitation of IFN-γRα with STAT1 requires the FANCC protein.
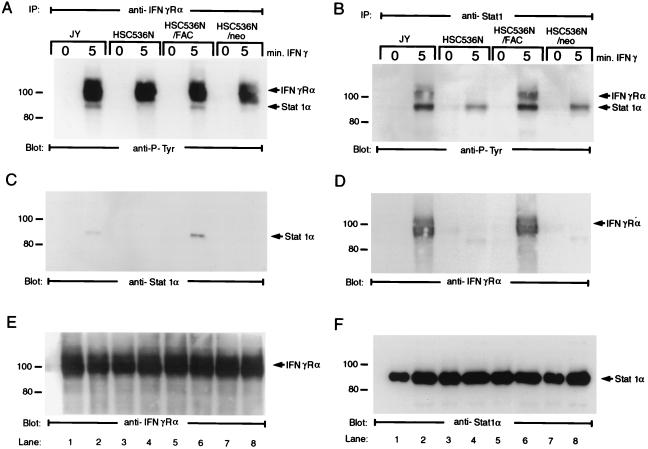
IFN-γ-induced tyrosine phosphorylation of IFN-γRα creates a docking site for latent STAT1, which, after docking, is then activated by the JAK kinases (6, 18, 22, 58). In support of this model, it has been shown that STAT1 associates with tyrosine-phosphorylated IFN-γRα both in vitro and in vivo (12, 13, 42). Having established the normal pattern of IFN-γ-induced tyrosine phosphorylation of IFN-γRα and the two JAK kinases in FA-C cells, we next asked whether STAT1 was recruited to the activated IFN-γR complex. INF-γ-treated isogenic cells were solubilized with 1% digitonin buffer, and protein complexes were immunoprecipitated using antibody to IFN-γRα. The presence of STAT1 in anti-IFN-γRα immunoprecipitates was assessed by immunoblotting with either anti-P-Tyr or anti-STAT1α monoclonal antibody. When material precipitated using lysates from FA-C isogenic cell lines was analyzed with the anti-P-Tyr antibody, a diffusely migrating phosphoprotein of ∼100 kDa was detected in all four cell lines only when cells had been treated with IFN-γ (Fig. 4A, lanes 2, 4, 6, and 8). This 100-kDa phosphoprotein was identified by reprobing the blot with anti-IFN-γRα (Fig. 4E). In addition to the tyrosine-phosphorylated IFN-γRα, a distinct phosphoprotein of ∼90 kDa recognized by anti-P-Tyr immunoblotting was also precipitated by anti-IFN-γRα in lysates prepared from IFN-γ-treated normal (JY) and complemented FA-C (HSC536N/FANCC) cell lines but not in those from stimulated FA-C (HSC536N and HSC536N/neo) cell lines (Fig. 4A, lanes 2 and 6 versus 4 and 8). Reprobing the blot with a monoclonal antibody specific to STAT1α revealed that the 90-kDa band was STAT1α (Fig. 4C, lanes 2 and 6). No signal was detected in the anti-STAT1α immunoblot when anti-IFN-γRα immunoprecipitates had been prepared from IFN-γ-treated mutant cell lines HSC536N and HSC536N/neo (Fig. 4C, lanes 4 and 8) or from cells that had not been incubated with IFN-γ (Fig. 4C, lanes 1, 3, 5, and 7).
FIG. 4.
IFN-γ-induced formation of STAT1α–IFN-γRα complex in B lymphocytes. Cells were stimulated with IFN-γ for up to 10 min and solubilized in digitonin lysis buffer as described in Materials and Methods. The WCL were then incubated with anti-IFN-γRα or anti-STAT1 antibody. The immunoprecipitates were analyzed by SDS-PAGE followed by immunoblotting. (A) Cell lysates prepared from FA-C isogenic lines were immunoprecipitated (IP) with anti-IFN-γRα and probed with anti-P-Tyr antibody; (B) cell lysates prepared from FA-C isogenic lines were immunoprecipitated with anti-STAT1 and probed with anti-P-Tyr antibody; (C) the blot used for panel A was stripped and reprobed with anti-STAT1α monoclonal antibody; (D) the blot used for panel B was stripped and reprobed with anti-STAT1α monoclonal antibody; (E) the blot used for panel A was stripped and reprobed with anti-IFN-γRα; (F) the blot used for panel B was stripped and reprobed with anti-STAT1α.
To confirm the above observations, we conducted coimmunoprecipitation experiments using anti-STAT1 antibody. As shown in Fig. 4B, on the blot probed with anti-P-Tyr, two tyrosine-phosphorylated proteins are evident in both IFN-γ-treated normal (JY) and complemented (HSC536N/FANCC) cell lines but not in stimulated mutant FA-C (HSC536N and HSC536N/neo) cell lines (lanes 2 and 6 versus 4 and 8). The upper band proved to be IFN-γRα, as indicated in Fig. 4D (lanes 2 and 6), when we reprobed the membrane with antibody to the receptor protein. Although a small amount of phosphorylated STAT1 was precipitated by anti-STAT1 antibody in FA-C cell lines HSC536N and HSC536N/neo, no coprecipitating IFN-γRα receptor protein was detected (Fig. 4B and D, lanes 4 and 8), indicating reduced association of STAT1 with the receptor in mutant cells.
As an important technical note, we emphasize that we were unable to detect STAT1 in anti-IFN-γRα or IFN-γRα in anti-STAT1 immunoprecipitates from cell lysates prepared with RIPA buffer or NP-40 lysis buffer (see Materials and Methods). Washing of the immunoprecipitate in 1% Triton X-100, 1% NP-40, or 1% digitonin lysis buffer resulted in the disruption of the STAT1–IFN-γRα complex, suggesting that interaction between STAT1 and the receptor protein may be weak or unstable. Finally, Western blot analysis of the anti-IFN-γRα and -STAT1 immunoprecipitates demonstrated the presence of equivalent IFN-γRα and STAT1 proteins in all four cell lines at 5 min of IFN-γ incubation compared to unstimulated cells (Fig. 4E and F), indicating that both the receptor and STAT1 proteins were constitutively expressed at equivalent levels in all isogenic cell lines. Taken together, these results demonstrate an unambiguous requirement for the FANCC protein in the formation of IFN-γ-induced STAT1α–IFN-γRα complexes.
IFN-γ-induced association of STAT1 with FANCC.
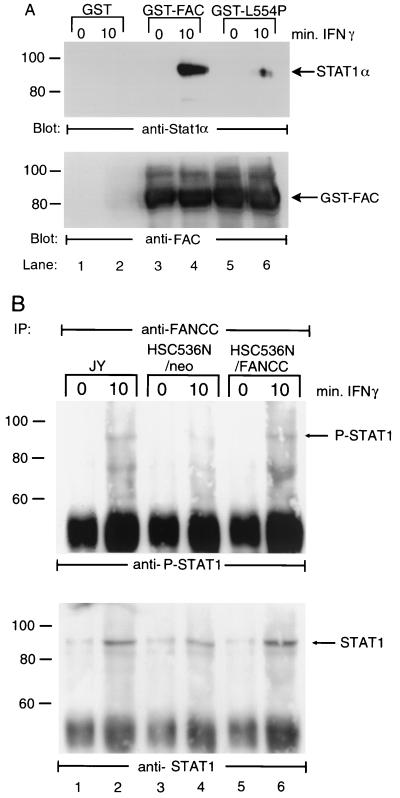
The coimmunoprecipitation result (Fig. 4) demonstrated that FANCC is required for recruitment of STAT1 to the activated IFN-γR complex. Because this suggested that the activation of STAT1 might result from a direct interaction of the FANCC protein with STAT1, we sought to identify a binding interaction between FANCC and STAT1. JY cells were stimulated with IFN-γ or left untreated, and WCL were incubated with glutathione-Sepharose bead-bound GST fusion proteins containing the normal FANCC (GST-FANCC) or a patientderived mutant FANCC (GST-L554P). FANCC-interacting cellular proteins were then isolated from WCL by glutathione-Sepharose affinity precipitation. STAT1 Western blot analysis of the affinity-precipitated proteins (Fig. 5A, top) showed that STAT1 bound strongly to the normal FANCC fused to GST (lane 4) but not to GST alone (lanes 1 and 2). STAT1 bound weakly to the GST fusion protein containing the L554P mutation (lane 6). Reprobing the blot with antibodies to FANCC revealed comparable amounts of GST-fused FANCC and L554P proteins were input in the binding reactions (Fig. 5A, bottom). It is important to note that FANCC-STAT1 interaction was detected only in lysates prepared from IFN-γ-stimulated cells (lanes 3 and 5 versus 4 and 6), suggesting that the association is initiated by early activation elements of the IFN-γ signaling pathway.
FIG. 5.
IFN-γ-induced association of FANCC and STAT1 in B lymphocytes. (A) WCL (1 mg of protein) of JY cells stimulated with IFN-γ for up to 10 min were incubated with yeast-expressed GST fusion proteins (10 μg) bound to glutathione-Sepharose beads. The glutathione-Sepharose affinity precipitates were analyzed by SDS-PAGE followed by immunoblotting with antibodies to STAT1α (top) and reprobed with anti-FANCC to verify the input of the GST fusion proteins. (B) Coimmunoprecipitation (IP) of endogenous FANCC and STAT1. WCL prepared from JY and HSC536N cells (500 μl of each, containing 1 mg of total proteins) were incubated with FANCC-specific antibody in the presence of protein A-agarose. The immunocomplexes were then analyzed by Western blotting with antibody specific for P-STAT1 (top) and for total STAT1 (bottom).
Because STAT1 interacted in vitro with GST-FANCC, we sought to determine whether similar association also occur within cells. To detect complexes formed between endogenous FANCC and STAT1 in IFN-γ-stimulated lymphocytes, we performed immunoprecipitation with an affinity-purified FANCC-specific antiserum. The precipitates were analyzed by Western blotting with antibodies against either P-STAT1 or total STAT1. As shown in Fig. 5B, a detectable P-STAT1 signal was obtained in precipitates from IFN-γ-stimulated JY and HSC536N/FANCC cells, which was absent in samples from the stimulated mutant HSC536N cells (lanes 2 and 6 versus 4). There was no detectable P-STAT1 in precipitates from unstimulated cells (lanes 1, 3, and 5). This result was further substantiated by reprobing the blot with an antibody against total STAT1. The lower panel of Fig. 5B clearly demonstrated that FANCC coimmunoprecipitated with STAT1 in the extracts from the normal JY and corrected HSC536N/FANCC cells treated with IFN-γ (Fig. 5B, bottom, lanes 2 and 6). We also found that there was significantly reduced total STAT1 bound to FANCC in the mutant HSC536N/neo extracts (lane 4), consistent with our observation that STAT1 bound weakly to the GST fusion protein containing the mutant FANCC L554P (Fig. 5A, lane 6). Faint bands were detected in the precipitates from unstimulated cell extracts when the blot was reprobed with antibody against total STAT1 (Fig. 5B, bottom, lanes 1, 3, and 5). This was probably due to a slight reactivity of the antiserum with a protein that was migrating at the same position as the STAT1 protein, as we observed the same faint band in a preimmune precipitation (data not shown). To assess whether the FANCC mutation directly caused a failure of STAT1-FANCC binding, we performed a binding assay using WCL from HSC536N cells. As shown in Fig. 6B, STAT1 from IFN-stimulated HSC536N cells was able to bind to a normal GST-FANCC with association kinetics comparable to that observed in the normal JY cells (Fig. 6A), indicating that the limiting factor for STAT1 complex formation in IFN-stimulated FA-C mutant cells is FANCC itself.
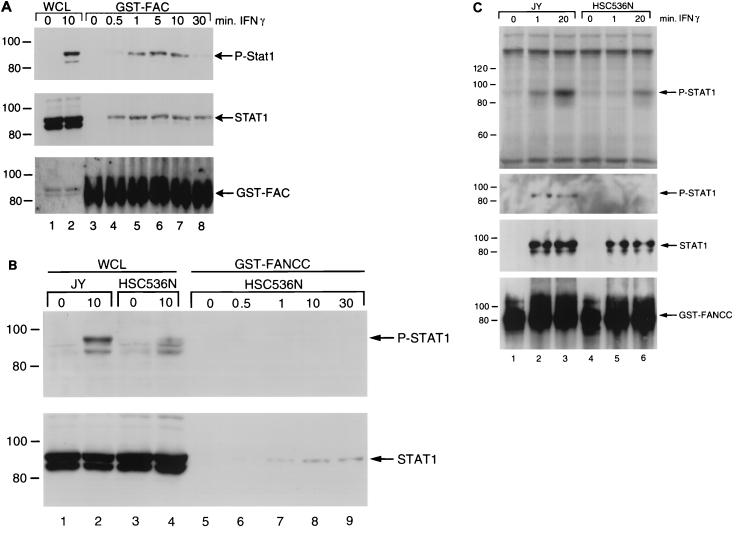
FIG. 6.
Kinetics of FANCC-STAT1 association. WCL (1 mg of total proteins) of JY (A) and HSC536N (B) cells stimulated with IFN-γ (10 ng/ml) for various periods of time were incubated with GST-FANCC (10 μg) bound to glutathione-Sepharose beads, and the glutathione-Sepharose affinity precipitates were analyzed by immunoblotting with antibodies to P-STAT1 (A and B, top), STAT1α/β (A, middle; B, bottom), and FANCC (A, bottom). Lanes 1 and 2 in panel A and lanes 1 to 4 in panel B are the input controls, each of which was loaded with 100 μg of total WCL proteins. The double bands in lanes 1 and 2 of the bottom gel in panel A are nonspecific. (C) In vivo phosphorylation of STAT1 in cells stimulated with IFN-γ. JY and HSC536N cells were metabolically labeled with [32P]orthophosphoric acid for 4 h followed by IFN-γ treatment for the indicated time points. WCL were prepared and affinity precipitated with GST-FANCC and analyzed by SDS-PAGE and autoradiography (top). Western blot analysis, performed on these affinity precipitates to determine the amounts of tyrosine-phosphorylated and total STAT1 proteins, is shown in the lower three panels, which were probed with antibodies specific for P-STAT1, STAT1, and FANCC, respectively. The order of samples is the same in all panels.
IFN-γ initially induces FANCC association with STAT1 form not phosphorylated on Y701.
We further examined kinetically whether the interaction of FANCC-STAT1 in induced cells depended on prior tyrosine phosphorylation of STAT1. We stimulated JY cells with IFN-γ for various periods of time and performed GST fusion protein affinity binding assays. FANCC-bound STAT1 could be detected as early as 30 s after IFN-γ treatment, reached its maximal level at 5 min, and decreased at 30 min after stimulation (Fig. 6A, top). Interestingly, the amounts of total STAT1 proteins associated with FANCC were comparable between 30 s and 1 min of IFN-γ treatment, but the amount of phosphorylated STAT1 bound to FANCC at the 30-s time point was negligible compared to that at the 1-min time point (Fig. 6A, top and middle, compare lanes 4 and 5). While we cannot rule out the possibility that the antibody against total STAT1 may have higher affinity than the one specific for P-STAT1, comparison of P-STAT1 and total STAT1 at the 30-s, 1-min, and 5-min time points clearly demonstrated an increase in the phosphorylated form that occurs only following full binding of the nonphosphorylated form of STAT1. Because the kinetic appearance of STAT1 at the docking site occurs at the time of phosphorylation, our results are most compatible with the notion that IFN-γ initiates the association of FANCC with STAT1 before STAT1 becomes phosphorylated and that the association of FANCC with P-STAT1 results from the delivery by FANCC to the IFN-γR docking site. Because STAT1 is also phosphorylated on serines (e.g., Ser727), additional studies using [32P]orthophosphate-labeled cells were conducted to confirm that the initial association of STAT1 with FANCC is with an unphosphorylated form. Figure 6C (top) shows that significantly more P-STAT1 was bound to GST-FANCC in IFN-stimulated JY cells at 20 min than at the 1-min time point. However, the amounts of FANCC-bound P-STAT1 and total STAT1 were comparable between these two time points (Fig. 6C, middle, compare lanes 2 and 3). This suggests that the more intense signal obtained at 20 min of IFN-γ stimulation is most likely due to serine phosphorylation. In HSC536N cells, there was no P-STAT1 bound to GST-FANCC at 1 min of IFN stimulation, but P-STAT1 was evident at the 20-min time point. Interestingly, however, total bound STAT1 could be detected in equal amount at both 1- and 20-min time points even though none of these bound STAT1 proteins were tyrosine phosphorylated. Taken together, these results suggest that the association of STAT1 with FANCC precedes STAT1 phosphorylation.
FANCC-STAT1 association in MO7e cells and its functional relevance.
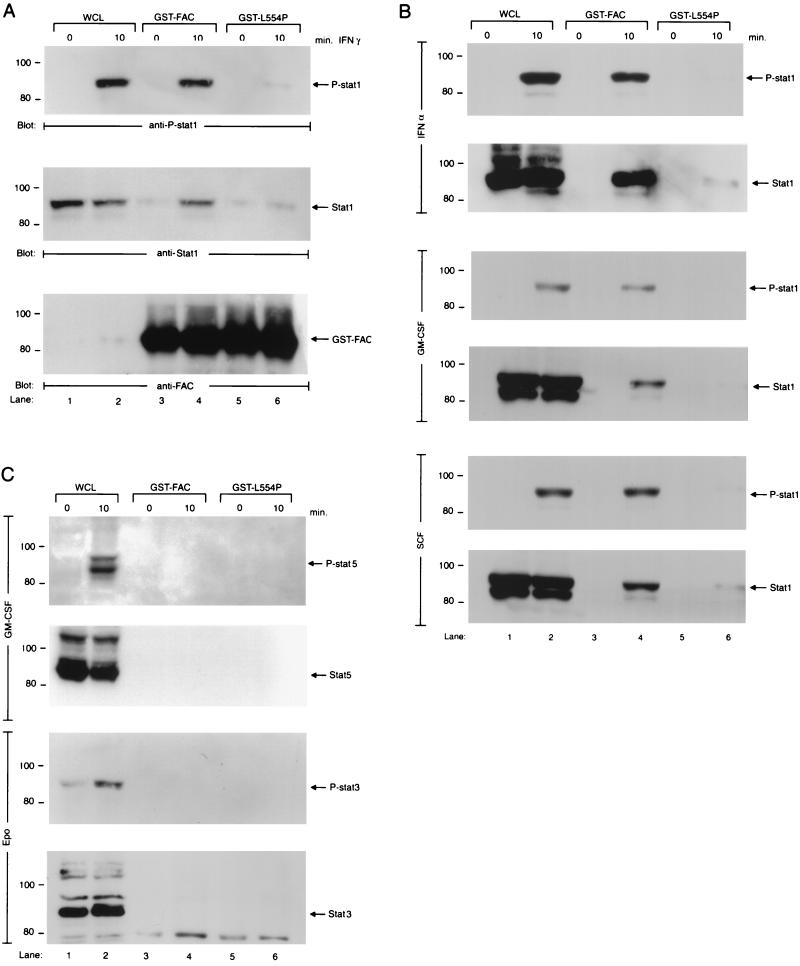
We next sought to determine whether FANCC-STAT1 association occurs in normal myeloid precursor cells. MO7e cells were incubated in the presence of IFN-γ, and analysis of the WCL by Western blotting indicated that IFN-γ induced tyrosine phosphorylation of STAT1 in MO7e cells (Fig. 7A, top, lane 2). A GST fusion protein affinity binding assay showed that IFN-γ induced association of STAT1 with the normal FANCC protein but not with the mutant L554P protein fused to GST (Fig. 7A, top and middle, lanes 4 versus 6). Figure 7A demonstrates that comparable amounts of total STAT1 protein (middle, lanes 1 and 2) and GST fusion proteins (bottom, lanes 3 to 6) were loaded in the binding reactions.
FIG. 7.
FANCC-STAT1 association in stimulated MO7e cells. (A) WCL of MO7e cells stimulated with IFN-γ (10 ng/ml) for 10 min were used for either direct Western blotting (60 μg of total WCL proteins; lanes 1 and 2) or affinity precipitation (1 mg of total WCL proteins) with yeast-expressed GST fusion proteins (10 μg) and then analyzed by immunoblotting (lanes 3 to 6). The blot was probed with anti-P-STAT1 (top), anti-STAT1 (middle), or anti-FAC (bottom). (B) MO7e cells were stimulated with IFN-α (20,000 U/ml), GM-CSF (100 U/ml), or SCF (50 ng/ml) for 10 min, and WCL was subjected to either direct Western analysis (100 μg of total WCL proteins; lanes 1 and 2) or GST affinity binding (1 mg of total WCL proteins; 10 μg of GST fusion proteins; lanes 3 to 6) as described above. All IFN-α, GM-CSF, and SCF blots were first probed with anti-P-STAT1 and then stripped and reprobed with anti-STAT1. (C) MO7e and MO7ER cells were treated with GM-CSF (100 U/ml) and EPO (40 U/ml), respectively, for 10 min. WCL was then subjected to either direct Western blotting (100 μg of total WCL proteins; lanes 1 and 2) or GST affinity binding (1 mg of total WCL proteins; 10 μg of GST fusion proteins; lanes 3 to 6) as described for panel A. The GM-CSF blot was probed first with anti-P-Stat5 and then anti-Stat5; the EPO blot was probed first with anti-P-Stat3 and then anti-Stat3.
Although the STAT1-FANCC association is clear-cut, and while we can state with reasonable certainty that the hypersensitivity does not require STAT1 activation, the mechanism of IFN hypersensitivity in FA-C cells is less clear and seems even counterintuitive. The STAT1 activation defect in FA cells may play a more direct role in signaling for survival or self-replication. Certain growth and survival factors for hematopoietic progenitor cells and stem cells are known to activate STAT1. These include SCF (5, 7), GM-CSF (37), G-CSF (1, 47), and EPO (24, 36). Our hypothesis that the STAT1 defect in FA cells might result in a functional disconnection from survival cues required, first, that FANCC and STAT1 associate in growth factor-responsive hematopoietic cells after stimulation with growth and survival factors other than IFN-γ. To this end, we stimulated MO7e cells with IFN-α, GM-CSF, and SCF, all of which have been shown to induce tyrosine phosphorylation of STAT1 (6, 7, 24) and stimulated MO7ER cells with EPO. As shown in Fig. 7B, IFN-α, GM-CSF, and SCF induced activation of STAT1 by tyrosine phosphorylation (lane 2) and association of STAT1 and FANCC proteins (lane 4). The intensity of the signal owing to STAT1 phosphorylation was greatest for IFN-α and least for GM-CSF (Fig. 7B, lane 2, compare between panels). As has been described by others (24), we also noted low-level STAT1 phosphorylation in MO7ER cells in response to EPO (data not shown). To extend the above observations, we sought to determine whether the FANCC protein also interacts with other STAT molecules. MO7e and MO7ER cells were treated with GM-CSF and EPO, respectively. Both GM-CSF and EPO have been reported to activate STAT3 (7), and the former also induces activation of STAT5 (34) in these cells. Figure 7C demonstrates that GM-CSF and EPO activate Stat5 and Stat3, respectively (lane 2), but that no FANCC-Stat5 or FANCC-Stat3 association was detected in these cells.
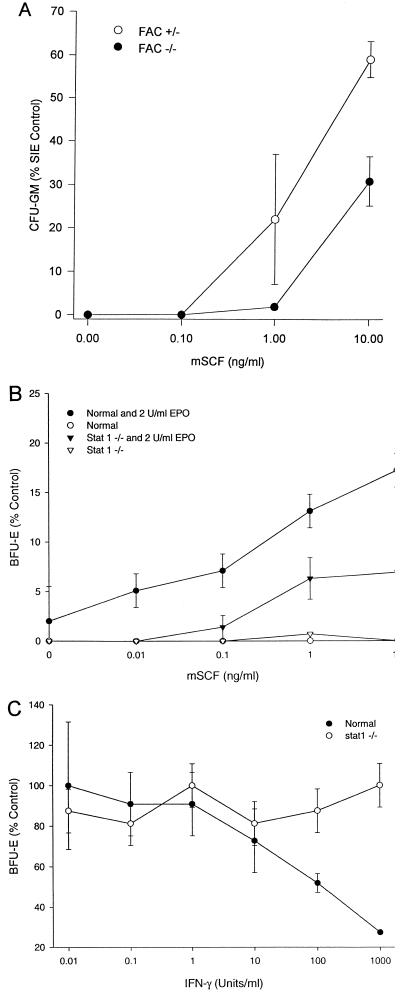
Because Steel factor deficiencies account for both bone marrow and germ cell deficiencies in mice, both of which occur in FANCC knockout mice, we sought to test the hypothesis that FANCC mutations might result in unresponsiveness to Steel factor, a survival factor for hematopoietic cells that induces STAT1 activation (5, 7). We treated BM cells from heterozygotes and FANCC knockout mice with graded doses of Steel factor in the presence of EPO or IL-3 in clonal assays. As shown in Fig. 8A, both normal (heterozygote FANCC−/+) and mutant (FANCC−/−) BM cells proliferated in response to the hematopoietic growth factor SCF, but clonal growth of hematopoietic progenitor cells from FANCC−/− animals was consistently less responsive to SCF stimulation, confirming earlier preliminary reports (3). We found the same to be true in clonal assays from marrows of STAT1−/− mice (Fig. 8B). However, clonal growth of progenitor cells from STAT1−/− mice differed from that from FANCC−/− mice in that they were completely resistant to IFN-mediated suppression (Fig. 8C).
FIG. 8.
SCF induces CFU-GM growth of murine BM cells. (A) BM cells from FANCC−/+ and FANCC−/− mice were exposed to either recombinant murine SCF (0 to 10 ng/ml) or a combination of three growth factors (SIE): murine Steel factor (10 ng/ml), murine IL-3 (10 ng/ml), and human EPO (2 U/ml). CFU-GM and BFU-E were counted after 14 days. Data points represent mean colony growth (±SD) of triplicate cultures. (B) Suboptimal responses of erythroid burst-forming units (BFU-E) from STAT1−/− to EPO and SCF. Results of a single representative experiment (or two independent experiments) are shown. Data points represent mean (±SD) clonal growth derived from colony counts in three replicate plates. Control BFU-E growth was defined in cultures containing IL-3 (10 ng/ml), Steel factor (10 ng/ml), and EPO (2 U/ml). Control BFU-E growth was equivalent in heterozygotes (31 colonies 105 cells) and STAT1−/− mice (48 colonies/105 cells). (C) Suppression of BFU-E growth by IFN-γ is STAT1 dependent. Optimally stimulated (IL-3, SCF, and EPO) clonal cultures of erythroid marrow cells from STAT1−/− mice and control animals were exposed to the indicated doses of IFN-γ throughout the culture period. Results shown are from a single representative experiment. Each point represents mean (±SD) BFU-E growth expressed as percent control. Control colony growth was equivalent (see above), but IFN-γ failed to suppress clonal growth in STAT1−/− mice, even at supraphysiological levels.
DISCUSSION
We have recently demonstrated that hematopoietic progenitor cells from mice nullizygous at the FANCC locus and children with FA-C are hypersensitive to the antiproliferative effect of IFN-γ (38, 55). Because most of the known effects IFN-γ are ignited via STAT1 activation and nuclear translocation of STAT1 homodimers, we initially expected that at IFN-γ doses too low to activate STAT1 in normal cells, FA-C cells would contain high levels of phosphorylated STAT1. We were surprised to discover that in FA-C cells, STAT1 phosphorylation in response to IFN-γ was reduced markedly (10, 38). We ruled out the possibility that reduced STAT1 activation per se was the cause of progenitor cell hypersensitivity to IFN-γ by quantifying growth of clonogenic progenitor cells from the marrow of STAT1−/− mice. Progenitors from these mice were completely resistant to the growth-suppressive activity of IFN-γ (Fig. 8C), indicating that the hypersensitivity derived not from the failure of STAT1 activation but from another biological effect of FANCC mutation. Like FA-C progenitors, however, progenitor cells from STAT1−/− mice were suboptimally responsive to combinations of Steel factor (SCF) and EPO (Fig. 8B), indicating that inactivating FANCC mutations may blunt responses to survival factors for hematopoietic progenitor cells.
We therefore sought to more fully understand the function of the FANCC protein vis-à-vis the JAK/STAT signaling pathway, using the IFN-γ signaling pathway as a paradigm. We also investigated factor-dependent hematopoietic cell lines, seeking to reveal FANCC-JAK-STAT relationships by using other receptor molecules including SCF, EPO, and GM-CSF. We reasoned that the normal FANCC protein, localized primarily in the cytoplasm (16, 56, 57), might functionally interact with key components of the JAK/STAT cytokine signaling cascades induced by survival factors for hematopoietic cells, and that mutations of FANCC might interdict such responses. We have confirmed that notion in this study. We used isogenic cells derived from patients and from nullizygous mice carrying inactivating mutations in the FANCC gene to dissect the ligand-induced assembly of the multimeric IFN-γR–JAK–STAT complex.
We confirmed that STAT1 tyrosine phosphorylation was deficient in FA-C cells (Fig. 1) but determined that the activation kinetics of IFN-γRα (Fig. 2) and JAK1 and JAK2 (Fig. 3) were normal in FA-C cells. In addition, immunoblot analysis of total cell lysates prepared from unstimulated and IFN-γ-treated cells indicated that both normal and FA-C cells constitutively expressed equivalent levels of IFN-γRα, JAK1, and JAK2. These results suggest that the deficiency in STAT1α tyrosine phosphorylation results neither from reduced expression nor activation of the JAK molecules or IFN-γRα. However, STAT1α was detected only in the IFN-γR complexes of cell lysates from IFN-stimulated normal and corrected FA-C cell lines (Fig. 4). Moreover, GST-fused FANCC was shown to bind STAT1 contained in IFN-γ-induced cell lysates, but STAT1 did not bind to a FANCC mutant protein (a point mutant known to inactivate the function of FANCC) fused to GST (Fig. 5). Using activation state-specific antibodies and orthophosphate labeling studies, we have concluded that FANCC-STAT1 association did not require STAT1 phosphorylation, on either tyrosine or serine residues (Fig. 6). We further demonstrated that GST-FANCC-STAT1 association was also induced by IFN-γ, IFN-α, GM-CSF, and SCF in MO7e cells and that mutant GST-FANCC-L554P did not associate with STAT1 in cells stimulated with these factor (Fig. 7). That survival of committed progenitor cells exposed to graded doses of SCF was suppressed in FA-C progenitor cells from FANCC knockout mice confirmed that FANCC augments responses to SCF and EPO in hematopoietic cells (Fig. 8).
Our observation that STAT1α forms a complex with IFN-γRα in normal cells stimulated with IFN-γ is consistent with the previous reports that STAT1α physically associates with IFN-γRα both in vitro and in vivo (12, 13, 42). In these reports, STAT1 was shown to specifically recognize a phosphorylated tyrosine residue in the cytoplasmic domain of the receptor protein and to coimmunoprecipitate with IFN-γRα in stimulated cell lysates. Once recruited into the dimeric IFN-γRα/β complex, STAT1 becomes activated by tyrosine phosphorylation (4, 50, 52). We found that coimmunoprecipitation of STAT1 with IFN-γRα was ligand dependent and additionally depended on the presence of an intact FANCC protein (Fig. 4). The difference in STAT1–IFN-γRα coimmunoprecipitation between normal and FA-C cells (Fig. 4A and B) was not due to the varying levels of the STAT1 proteins, since STAT1 was expressed at comparable levels (Fig. 1B and 4F). Therefore, it appeared that the presence of a functional FANCC protein somehow facilitated the recruitment of STAT1 to the activated IFN-γR complex.
The reason for the complete failure of STAT1 to appear at the docking site of the IFN-γR and subsequently to be activated by tyrosine phosphorylation in FA-C cells is not completely understood. Three possible mechanisms may account for our observation. First, because FANCC and STAT1 associate very rapidly and prior to STAT1 phosphorylation (Fig. 6), FANCC itself might function as a chaperone for STAT1 molecules, moving them from cytoplasm to the receptor docking sites. This shuttling function of FANCC might occur directly or with the help of intermediary molecules. Second, that FANCC interacts with transcription factors including STAT1 in the studies described here and PLZF homologs (17) is compatible with the notion that in FA cells genes whose products govern STAT1 docking and activation might be derepressed. Third, FANCC may suppress the function of proteins that function to inhibit STAT1 activation. There is growing evidence that FANCC interacts with other FA proteins to form a functional nuclear complex (11, 53). It is possible that such nuclear complex functions to modulate gene expression in response to external signals such as IFNs or other cytokines and growth factors. SOCS1, a STAT1-responsive gene whose product interdicts IFN-induced activation of STAT1, is an appealing candidate, particularly since SOCS1 nullizygous mice like FANCC nullizygous mice exhibit IFN-γ hypersensitivity (15, 29). However, this molecule functions by reducing JAK activation (41), and the studies described herein demonstrate that FANCC mutations result in STAT1 inactivation without reduction in JAK1 and JAK2 phosphorylation. Consequently, derepression of SOCS1 expression per se is an unlikely explanation for our results. Nonetheless, while their function is unclear to date, a number of proteins with C-terminal SOCS domains have been identified (15), and it remains quite possible that such proteins or others like them function to inhibit STAT1 activation and are also encoded by genes responsive to transcriptional repressors with which FANCC interacts.
Because the initial association of FANCC and STAT1 did not require tyrosine phosphorylation, we sought to determine whether the initially bound STAT molecule was phosphorylated on other residues. We found that FANCC-STAT1 association required neither tyrosine nor serine phosphorylation (Fig. 6A and C). We also found that STAT1 from induced FA-C cells was capable of binding to normal GST-FANCC (Fig. 6B), demonstrating that early IFN-induced signals influence the receptivity of STAT1 to FANCC, even in mutant cells. Thus, although the nature of the early IFN-induced signaling pathway is unclear at this time, the limiting factor for STAT1 phosphorylation in FA-C cells is clearly FANCC itself, not a FANCC-dependent cofactor. The association of FANCC with STAT1 prior to receptor docking is one of the earliest reported responses to IFN-γ.
While IFN-γ was used as our paradigm, we were also able to demonstrate the association of STAT1 with FANCC in cells exposed to hematopoietic growth and survival factors (Fig. 7A and B). Our results also demonstrate reasonable specificity of FANCC-STAT1 interaction and potential functional consequence of this interaction. While the FANCC-STAT1 binding phenomenon occurred in both myeloid and lymphoid cells (Fig. 7A and B) and occurred in cells stimulated with IFN-γ, IFN-α, GM-CSF, and SCF (Fig. 7A and B), FANCC did not associate with activated STAT3 and STAT5 (Fig. 7C). Therefore, these results suggest that although other STAT molecules are present and activated in appropriately stimulated cells, STAT1 is a specific FANCC partner.
Others have reported that SCF-stimulated BM colony growth is defective in FA patients (3). Since we have shown that SCF induces FANCC-STAT1 interaction in MO7e cells, our results are consistent with a role of the interaction in cell proliferation induced by SCF and certain other hematopoietic growth factors. This notion is further strengthened by our findings that BM progenitor cells from FANCC−/− and STAT1−/− mice exhibited blunted responses to SCF. This suggests a potential role of the FANCC-STAT1 interaction in stem cell and progenitor cell survival mediated by SCF and possibly other growth factors.
That constitutive activation of IFN-responsive genes like IRF-1 and MxA (9, 27) is disconnected with STAT1 activation in FA-C cells not only indicates that the FANCC protein is multifunctional in its capacity to suppress hematopoiesis but also explains why FA patients are not susceptible to opportunistic infections as might be predicted by extrapolation from observations of STAT1−/− mice (9, 28). That is, some of the constitutively expressed proteins in FA mediate the antiviral state (27, 37, 46) and would likely be protective. While the mechanisms by which FANCC mutations result in constitutive activation of ordinarily repressed genes are unclear, that FANCC binds to a PLZF homolog (17) suggests that the failure of FAZF-FANCC interaction in mutant cells may result in selective gene derepression.
From the hematopoietic standpoint, FA-C cells are hypersensitive to a variety of mitogenic inhibitory molecules and apoptotic cues, including cytokines that utilize pathways not known to utilize STAT1 (13). Consequently, we do not argue that the function of FANCC vis-à-vis STAT1 is sufficient to account for every element of the FA phenotype. If this were the case, we would need to propose that STAT1−/− mice should share this phenotype, and they do not. It is clear, however, that FA-C cells, whose receptor complexes are disconnected from STAT1 signaling pathways, are likely deprived of survival signals that utilize this pathway. With STAT1 deficiency alone, this state of disconnection from survival cues may not be sufficient to cause BM failure (a universal finding in FA), but when combined with the constitutive expression of mitogenic inhibitory and apoptotic factors, and hypersensitivity to IFN and tumor necrosis factor (38), the impact of STAT1 disconnection in hematopoietic cells may be substantial. Additional studies are warranted on the structural elements of FANCC required for STAT1 activation and both IRF-1 and MxA repression. We also argue that a comprehensive investigation of molecular hematopoietic control is warranted in STAT1−/− mice, particularly using factors known to influence expansion, survival, and responsiveness of myeloid and erythroid progenitor cells.
ACKNOWLEDGMENTS
We thank Manuel Buchwald for providing the lymphoblast cell line HSC536N from a type C Fanconi anemia patient, A. D. Miller for the retroviral vector pLXSN, David C. Hinkle for the pGST vector, Brian Druker for antiphosphotyrosine antibody 4G10, and Tara Koretsky for valuable technical assistance. We also thank Markus Grompe for the FANCC mutant cell lines, knockout mice, and helpful discussions.
This work was supported by NIH grant HL48546 and a Department of Veterans Affairs merit review grant to G.C.B.
REFERENCES
- 1.Avalos B R, Parker J M, Ware D A, Hunter M G, Sibert K A, Druker B J. Dissociation of the Jak kinase pathway from G-CSF receptor signaling in neutrophils. Exp Hematol. 1997;25:160–168. [PubMed] [Google Scholar]
- 2.Bach E A, Aguet M, Schreiber R D. The IFN gamma receptor: a paradigm for cytokine receptor signaling. Annu Rev Immunol. 1997;15:563–591. doi: 10.1146/annurev.immunol.15.1.563. [DOI] [PubMed] [Google Scholar]
- 3.Bagnara G P, Strippoli P, Bonsi L, Brizzi M F, Avanzi G C, Timeus F, Ramenghi U, Piaggio G, Tong J, Podesta M, et al. Effect of stem cell factor on colony growth from acquired and constitutional (Fanconi) aplastic anemia. Blood. 1992;80:382–387. [PubMed] [Google Scholar]
- 4.Bradford M M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- 5.Brizzi M F, Dentelli P, Rosso A, Yarden Y, Pegoraro L. STAT protein recruitment and activation in c-Kit deletion mutants. J Biol Chem. 1999;274:16965–16972. doi: 10.1074/jbc.274.24.16965. [DOI] [PubMed] [Google Scholar]
- 6.Darnell J E., Jr STATs and gene regulation. Science. 1997;277:1630–1635. doi: 10.1126/science.277.5332.1630. [DOI] [PubMed] [Google Scholar]
- 7.DeBerry C, Mou S, Linnekin D. STAT1 associates with c-kit and is activated in response to stem cell factor. Biochem J. 1997;327:73–80. doi: 10.1042/bj3270073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dignam J D, Lebovitz R M, Roeder R G. Accurate transcription initiation by RNA polymerase II in a soluble extract from isolated mammalian nuclei. Nucleic Acids Res. 1983;11:1475–1489. doi: 10.1093/nar/11.5.1475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Durbin J E, Hackenmiller R, Simon M C, Levy D E. Targeted disruption of the mouse Stat1 gene results in compromised innate immunity to viral disease. Cell. 1996;84:443–450. doi: 10.1016/s0092-8674(00)81289-1. [DOI] [PubMed] [Google Scholar]
- 10.Fagerlie S, Diaz J, Faulkner G, Grompe M, Keeble W, Tower P, Christianson T, Bagby G C. The Fanconi anemia group C gene product is necessary for normal Stat1 phosphorylation. Blood. 1997;90(Suppl. 1):571a. [Google Scholar]
- 11.Garcia-Higuera I, Kuang Y, Naf D, Wasik J, D'Andrea A D. Fanconi anemia proteins FANCA, FANCC, and FANCG/XRCC9 interact in a functional nuclear complex. Mol Cell Biol. 1999;19:4866–4873. doi: 10.1128/mcb.19.7.4866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Greenlund A C, Farrar M A, Viviano B L, Schreiber R D. Ligand-induced IFN gamma receptor tyrosine phosphorylation couples the receptor to its signal transduction system (p91) EMBO J. 1994;13:1591–1600. doi: 10.1002/j.1460-2075.1994.tb06422.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Greenlund A C, Morales M O, Viviano B L, Yan H, Krolewski J, Schreiber R D. Stat recruitment by tyrosine-phosphorylated cytokine receptors: an ordered reversible affinity-driven process. Immunity. 1995;2:677–687. doi: 10.1016/1074-7613(95)90012-8. [DOI] [PubMed] [Google Scholar]
- 14.Haneline L S, Broxmeyer H E, Cooper S, Hangoc G, Carreau M, Buchwald M, Clapp D W. Multiple inhibitory cytokines induce deregulated progenitor growth and apoptosis in hematopoietic cells from Fac−/− mice. Blood. 1998;91:4092–4098. [PubMed] [Google Scholar]
- 15.Hilton D J, Richardson R T, Alexander W S, Viney E M, Willson T A, Sprigg N S, Starr R, Nicholson S E, Metcalf D, Nicola N A. Twenty proteins containing a C-terminal SOCS box form five structural classes. Proc Natl Acad Sci USA. 1998;95:114–119. doi: 10.1073/pnas.95.1.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hoatlin M E, Christianson T A, Keeble W W, Hammond A T, Zhi Y, Heinrich M C, Tower P A, Bagby G C., Jr The Fanconi anemia group C gene product is located in both the nucleus and cytoplasm of human cells. Blood. 1998;91:1418–1425. [PubMed] [Google Scholar]
- 17.Hoatlin M E, Zhi Y, Ball H, Silvey K, Melnick A, Stone S, Arai S, Hawe N, Owen G, Zelent A, Licht J D. A novel BTB/POZ transcriptional repressor protein interacts with the Fanconi anemia group C protein and PLZF. Blood. 1999;94:3737–3747. [PubMed] [Google Scholar]
- 18.Horvath C M, Darnell J E. The state of the STATs: recent developments in the study of signal transduction to the nucleus. Curr Opin Cell Biol. 1997;9:233–239. doi: 10.1016/s0955-0674(97)80067-1. [DOI] [PubMed] [Google Scholar]
- 19.Hovland P, Flick J, Johnston M, Sclafani R A. Galactose as a gratuitous inducer of GAL gene expression in yeasts growing on glucose. Gene. 1989;83:57–64. doi: 10.1016/0378-1119(89)90403-4. [DOI] [PubMed] [Google Scholar]
- 20.Ihle J N. Cytokine receptor signalling. Nature. 1995;377:591–594. doi: 10.1038/377591a0. [DOI] [PubMed] [Google Scholar]
- 21.Ihle J N. STATs: signal transducers and activators of transcription. Cell. 1996;84:331–334. doi: 10.1016/s0092-8674(00)81277-5. [DOI] [PubMed] [Google Scholar]
- 22.Ihle J N, Kerr I M. Jaks and Stats in signaling by the cytokine receptor superfamily. Trends Genet. 1995;11:69–74. doi: 10.1016/s0168-9525(00)89000-9. [DOI] [PubMed] [Google Scholar]
- 23.Ihle J N, Nosaka T, Thierfelder W, Quelle F W, Shimoda K. Jaks and Stats in cytokine signaling. Stem Cells. 1997;15(Suppl 1):105–111. doi: 10.1002/stem.5530150814. [DOI] [PubMed] [Google Scholar]
- 24.Kirito K, Uchida M, Takatoku M, Nakajima K, Hirano T, Miura Y, Komatsu N. A novel function of STAT1 and Stat3 proteins in erythropoietin-induced erythroid differentiation of a human leukemia cell line. Blood. 1998;92:462–471. [PubMed] [Google Scholar]
- 25.Kohlhuber F, Rogers N C, Watling D, Feng J, Guschin D, Briscoe J, Witthuhn B A, Kotenko S V, Pestka S, Stark G R, Ihle J N, Kerr I M. A JAK1/JAK2 chimera can sustain alpha and gamma interferon responses. Mol Cell Biol. 1997;17:695–706. doi: 10.1128/mcb.17.2.695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kozak S L, Kabat D. Ping-pong amplification of a retroviral vector achieves high-level gene expression: human growth hormone production. J Virol. 1990;64:3500–3508. doi: 10.1128/jvi.64.7.3500-3508.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li Y, Youssoufian H. MxA overexpression reveals a common genetic link in four Fanconi anemia complementation groups. J Clin Investig. 1997;100:2873–2880. doi: 10.1172/JCI119836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Meraz M A, White J M, Sheehan K C, Bach E A, Rodig S J, Dighe A S, Kaplan D H, Riley J K, Greenlund A C, Campbell D, Carver-Moore K, DuBois R N, Clark R, Aguet M, Schreiber R D. Targeted disruption of the Stat1 gene in mice reveals unexpected physiologic specificity in the JAK-STAT signaling pathway. Cell. 1996;84:431–442. doi: 10.1016/s0092-8674(00)81288-x. [DOI] [PubMed] [Google Scholar]
- 29.Metcalf D, Alexander W S, Elefanty A G, Nicola N A, Hilton D J, Starr R, Mifsud S, DiRago L. Aberrant hematopoiesis in mice with inactivation of the gene encoding SOCS-1. Leukemia. 1999;13:926–934. doi: 10.1038/sj.leu.2401440. [DOI] [PubMed] [Google Scholar]
- 30.Muller M, Briscoe J, Laxton C, Guschin D, Ziemiecki A, Silvennoinen O, Harpur A G, Barbieri G, Witthuhn B A, Schindler C. The protein tyrosine kinase JAK1 complements defects in interferon-alpha/beta and -gamma signal transduction. Nature. 1993;366:129–135. doi: 10.1038/366129a0. [DOI] [PubMed] [Google Scholar]
- 31.Nelson J R, Lawrence C W, Hinkle D C. Thymine-thymine dimer bypass by yeast DNA polymerase zeta. Science. 1996;272:1646–1649. doi: 10.1126/science.272.5268.1646. [DOI] [PubMed] [Google Scholar]
- 32.Nelson J R, Lawrence C W, Hinkle D C. Deoxycytidyl transferase activity of yeast REV1 protein. Nature. 1996;382:729–731. doi: 10.1038/382729a0. [DOI] [PubMed] [Google Scholar]
- 33.Neubauer H, Cumano A, Muller M, Wu H, Huffstadt U, Pfeffer K. Jak2 deficiency defines an essential developmental checkpoint in definitive hematopoiesis. Cell. 1998;93:397–409. doi: 10.1016/s0092-8674(00)81168-x. [DOI] [PubMed] [Google Scholar]
- 34.Pallard C, Gouilleux F, Benit L, Cocault L, Souyri M, Levy D, Groner B, Gisselbrecht S, Dusanter-Fourt I. Thrombopoietin activates a STAT5-like factor in hematopoietic cells. EMBO J. 1995;14:2847–2856. doi: 10.1002/j.1460-2075.1995.tb07284.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Parganas E, Wang D, Stravopodis D, Topham D J, Marine J-C, Teglund S, Vanin E F, Bodner S, Colamonici O R, van Deusen J M, Grosveld G, Ihle J N. Jak2 is essential for signaling through a variety of cytokine receptors. Cell. 1998;93:385–395. doi: 10.1016/s0092-8674(00)81167-8. [DOI] [PubMed] [Google Scholar]
- 36.Penta K, Sawyer S T. Erythropoietin induces the tyrosine phosphorylation, nuclear translocation, and DNA binding of STAT1 and STAT5 in erythroid cells. J Biol Chem. 1995;270:31282–31287. doi: 10.1074/jbc.270.52.31282. [DOI] [PubMed] [Google Scholar]
- 37.Rajotte D, Sadowski H B, Haman A, Gopalbhai K, Meloche S, Liu L, Krystal G, Hoang T. Contribution of both STAT and SRF/TCF to c-fos promoter activation by granulocyte-macrophage colony-stimulating factor. Blood. 1996;88:2906–2916. [PubMed] [Google Scholar]
- 38.Rathbun R K, Faulkner G R, Ostroski M H, Christianson T A, Hughes G, Jones G, Cahn R, Maziarz R, Royle G, Keeble W, Heinrich M C, Grompe M, Tower P A, Bagby G C. Inactivation of the Fanconi anemia group C gene augments interferon-gamma-induced apoptotic responses in hematopoietic cells. Blood. 1997;90:974–985. [PubMed] [Google Scholar]
- 39.Rodig S J, Meraz M A, White J M, Lampe P A, Riley J K, Arthur C D, King K L, Sheehan K C F, Yin L, Pennica D, Johnson E M, Schreiber R D. Disruption of the Jak1 gene demonstrates obligatory and nonredundant roles of the Jaks in cytokine-induced biologic responses. Cell. 1998;93:373–383. doi: 10.1016/s0092-8674(00)81166-6. [DOI] [PubMed] [Google Scholar]
- 40.Ronni T, Sareneva T, Pirhonen J, Julkunen I. Activation of IFN-alpha, IFN-gamma, MxA, and IFN regulatory factor 1 genes in influenza A virus-infected human peripheral blood mononuclear cells. J Immunol. 1995;154:2764–2774. [PubMed] [Google Scholar]
- 41.Sakamoto H, Yasukawa H, Masuhara M, Tanimura S, Sasaki A, Yuge K, Ohtsubo M, Ohtsuka A, Fujita T, Ohta T, Furukawa Y, Iwase S, Yamada H, Yoshimura A. A Janus kinase inhibitor, JAB, is an interferon-gamma-inducible gene and confers resistance to interferons. Blood. 1998;92:1668–1676. [PubMed] [Google Scholar]
- 42.Sakatsume M, Igarashi K, Winestock K D, Garotta G, Larner A C, Finbloom D S. The Jak kinases differentially associate with the alpha and beta (accessory factor) chains of the interferon gamma receptor to form a functional receptor unit capable of activating STAT transcription factors. J Biol Chem. 1995;270:17528–17534. doi: 10.1074/jbc.270.29.17528. [DOI] [PubMed] [Google Scholar]
- 43.Sato T, Selleri C, Young N S, Maciejewski J P. Inhibition of interferon regulatory factor-1 expression results in predominance of cell growth stimulatory effects of interferon-gamma due to phosphorylation of Stat1 and Stat3. Blood. 1997;90:4749–4758. [PubMed] [Google Scholar]
- 44.Schindler C, Darnell J E., Jr Transcriptional responses to polypeptide ligands: the JAK-STAT pathway. Annu Rev Biochem. 1995;64:621–651. doi: 10.1146/annurev.bi.64.070195.003201. [DOI] [PubMed] [Google Scholar]
- 45.Schindler C, Shuai K, Prezioso V R, Darnell J E., Jr Interferon-dependent tyrosine phosphorylation of a latent cytoplasmic transcription factor. Science. 1992;257:809–813. doi: 10.1126/science.1496401. [DOI] [PubMed] [Google Scholar]
- 46.Segal G M, Smith T D, Heinrich M C, Ey F S, Bagby G C., Jr Specific repression of granulocyte-macrophage and granulocyte colony-stimulating factor gene expression in interleukin-1-stimulated endothelial cells with antisense oligodeoxynucleotides. Blood. 1992;80:609–616. [PubMed] [Google Scholar]
- 47.Shimoda K, Feng J, Murakami H, Nagata S, Watling D, Rogers N C, Stark G R, Kerr I M, Ihle J N. Jak1 plays an essential role for receptor phosphorylation and Stat activation in response to granulocyte colony-stimulating factor. Blood. 1997;90:597–604. [PubMed] [Google Scholar]
- 48.Shuai K, Stark G R, Kerr I M, Darnell J E., Jr A single phosphotyrosine residue of Stat91 required for gene activation by interferon-gamma. Science. 1993;261:1744–1746. doi: 10.1126/science.7690989. [DOI] [PubMed] [Google Scholar]
- 49.Staeheli P, Sutcliffe J G. Identification of a second interferon-regulated murine Mx gene. Mol Cell Biol. 1988;8:4524–4528. doi: 10.1128/mcb.8.10.4524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Strathdee C A, Gavish H, Shannon W R, Buchwald M. Cloning of cDNAs for Fanconi's anaemia by functional complementation. Nature. 1992;356:763–767. doi: 10.1038/356763a0. [DOI] [PubMed] [Google Scholar]
- 51.Tauchi T, Feng G S, Shen R, Hoatlin M, Bagby G C, Jr, Kabat D, Lu L, Broxmeyer H E. Involvement of SH2-containing phosphotyrosine phosphatase Syp in erythropoietin receptor signal transduction pathways. J Biol Chem. 1995;270:5631–5635. doi: 10.1074/jbc.270.10.5631. [DOI] [PubMed] [Google Scholar]
- 52.Tower P A, Christianson T A, Peters S T, Ostroski M L, Hoatlin M E, Zigler A J, Heinrich M C, Rathbun R K, Keeble W, Faulkner G R, Bagby G C., Jr Expression of the Fanconi anemia group C gene in hematopoietic cells is not influenced by oxidative stress, cross-linking agents, radiation, heat, or mitotic inhibitory factors. Exp Hematol. 1998;26:19–26. [PubMed] [Google Scholar]
- 53.Waisfisz Q, de Winter J P, Kruyt F A, de Groot J, van der Weel L, Dijkmans L M, Zhi Y, Arwert F, Scheper R J, Youssoufian H, Hoatlin M E, Joenje H. A physical complex of the Fanconi anemia proteins FANCG/XRCC9 and FANCA. Proc Natl Acad Sci USA. 1999;96:10320–10325. doi: 10.1073/pnas.96.18.10320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Watling D, Guschin D, Muller M, Silvennoinen O, Witthuhn B A, Quelle F W, Rogers N C, Schindler C, Stark G R, Ihle J N, et al. Complementation by the protein tyrosine kinase JAK2 of a mutant cell line defective in the interferon-gamma signal transduction pathway. Nature. 1993;366:166–170. doi: 10.1038/366166a0. [DOI] [PubMed] [Google Scholar]
- 55.Whitney M A, Royle G, Low M J, Kelly M A, Axthelm M K, Reifsteck C, Olson S, Braun R E, Heinrich M C, Rathbun R K, Bagby G C, Grompe M. Germ cell defects and hematopoietic hypersensitivity to gamma-interferon in mice with a targeted disruption of the Fanconi anemia C gene. Blood. 1996;88:49–58. [PubMed] [Google Scholar]
- 56.Yamashita T, Barber D L, Zhu Y, Wu N, D'Andrea A D. The Fanconi anemia polypeptide FACC is localized to the cytoplasm. Proc Natl Acad Sci USA. 1994;91:6712–6716. doi: 10.1073/pnas.91.14.6712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Youssoufian H. Localization of Fanconi anemia C protein to the cytoplasm of mammalian cells. Proc Natl Acad Sci USA. 1994;91:7975–7979. doi: 10.1073/pnas.91.17.7975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zhu X, Wen Z, Xu L Z, Darnell J E., Jr STAT1 serine phosphorylation occurs independently of tyrosine phosphorylation and requires an activated Jak2 kinase. Mol Cell Biol. 1997;17:6618–6623. doi: 10.1128/mcb.17.11.6618. [DOI] [PMC free article] [PubMed] [Google Scholar]