Graphical abstract
Keywords: Gracilaria lemaneiformis, Polysaccharide, Extraction, Purification, Structural characteristics, Physiological activities
Highlights
-
•
Gracilaria lemaneiformis, rich in polysaccharide, is a kind of economic red algae.
-
•
The extraction, purification, structure and bioactivities of GLP were provided.
-
•
The mechanisms of bioactivities of GLP were described.
-
•
The urgent problems to be solved are raised in this paper.
Abstract
Gracilaria lemaneiformis is a kind of edible economic red algae, which is rich in polysaccharide, phycobiliprotein, pigments, minerals and other nutrients and functional components. Polysaccharide is one of the main active components of Gracilaria lemaneiformis, which has been reported to present various physiological bioactivities, including regulation of glycolipid metabolism, immune, anti-tumor, anti-inflammatory and other biological activities. This paper aims to provide a brief summary of extraction, purification, structural characteristics, and physiological activities of Gracilaria lemaneiformis polysaccharide (GLP). This article is able to provide theoretical basis for the future research and exploitation of GLP, and improve its potential development to promote the healthy and sustainable processing and high value utilization industry of Gracilaria lemaneiformis
1. Introduction
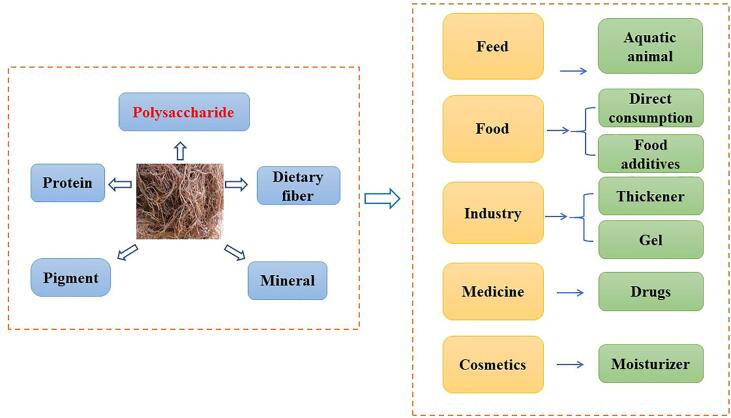
Gracilaria lemaneiformis, also named as asparagus, sea hair vegetable, thread vegetable, and gracilaria, belongs to Rhodophyta Phylum, Gigartinales Order, Gracilariaceae Family, and Gracilaria Genus, which is a kind of economic red algae used as agar, feed, food and drug resources, and has a long edible history (Veeraperumal et al., 2020, Wang et al., 2013). At present, Gracilaria lemaneiformis has been widely cultivated in coastal areas in recent years, including Japan, Korea, Russia and several provinces in China (Shandong, Liaoning, Guangdong, Heilongjiang, Jilin, Fujian, and other coastal cities). The output of dry Gracilaria has already reached 368,967 ton in China, and the cultivated area is 10,459 ha in 2020, as well as it is the second largest cultivated seaweed in China (Fisheries Administration of Ministry of Agriculture and Rural, 2021, Fan et al., 2012, Liu et al., 2016). As shown in Fig. 1, Gracilaria lemaneiformis can not only be eaten directly, but also be processed into feed, food additives, moisturizers, gels, medicines, and so on, which has been widely used in areas of feed, food, cosmetics, medicine, chemical industry and other industries (Cardoso et al., 2016, Li et al., 2008). Especially, it has been reported that Gracilaria lemaneiformis is widely used as feed for fish, shrimp, abalone and other aquatic animals, which can enhance the weight and growth rate of aquatic animals, increase the content of protein, amino acids and other nutrients, improve their antioxidant capacity, and reduce the lipid level (Yu, Chen, Liu, Chen, & Tian, 2016). Moreover, Gracilaria lemaneiformis is also mostly used to produce agar, and 91% of the agar production in 2015 came from Gracilaria algae, such as Gracilaria lemaneiformis, according to incomplete statistics (Porse, & Rudolph, 2017). In fact, Gracilaria lemaneiformis can not only bring economic benefits via processing into various products, but also bring ecological benefits, which has the ability to balance the level of nitrogen and phosphorus, reduce heavy metal pollution, effectively improve the water environment and repair the ecosystem (Wang et al., 2013, Wei et al., 2017).
Fig. 1.
The main components and applications of Gracilaria lemaneiformis.
Gracilaria lemaneiformis contains low fat, high dietary fiber, protein, polysaccharide, phycobiliprotein, pigments and other active ingredients of edible algae (Wen et al., 2006, Yu et al., 2016).
From Table 1, Gracilaria lemaneiformis contains 0.28–21% protein, 0.8–0.87% fat, 14.65–98.7% carbohydrate, 9.2% dietary fiber, 1.7–20.8% ash, 0.0232–0.0334% vitamin C (VC)(dry weight). In addition, the content of chlorophyll a (Chla) is 0.0121–0.045%, carotenoid (Car) is 0.3–0.68%, phycoerythrin (PE) is 0.035–0.0276% and phycocyanin (PC) is 0.006–0.020% (dry weight) (Han, Qi, Huang, & Fu, 2017; Wu, Jiang, & Liu, 2015; Xu and Gao, 2010, Yu and Yang, 2008). Moreover, the levels of essential amino acids (EAAs) are 3.619–6.79%, and the contents of non-essential amino acids (NEAAs) are 6.116–11.74% (dry weight). The content of each amino acid is showed in Table 1, Glu (1.373–2.49%), Asp (0.98–2.31%), Leu (0.835–1.61%), and Taurine (0.801–0.949%) are the major amino acids of Gracilaria lemaneiformis (Chen, Zou, Du, & Ji, 2018). Furthermore, the contents of Ca, Mg, K, Na, Fe and Zn in Gracilaria lemaneiformis are 0.048, 0.249, 1.15, 0.792, 0.218 and 0.0036% (dry weight), respectively. K and Na are the primary minerals. Therefore, Gracilaria lemaneiformis is beneficial to maintain the balance of sodium and potassium in the body and prevent hypertension (Zhou, He, & Ma, 2010) (Table 1).
Table 1.
The nutritional and biological components of Gracilaria lemaneiformis.
| Components | Content (dry weight) | Reference | |
|---|---|---|---|
| Protein (%) | 0.28–21 | (Chen et al., 2020, Chen et al., 2010, Fan et al., 2012, Han et al., 2020, Han et al., 2020, Jin et al., 2017, Kang et al., 2017, Wen et al., 2006, Wu et al., 2017, Zhou et al., 2010) | |
| Fat (%) | 0.8–0.87 | ||
| Carbohydrate (%) | 7.4–98.7 | ||
| Dietary fiber (%) | 9.2 | ||
| Ash (%) | 1.7–20.8 | ||
| Vc (%) | 0.0232–0.0334 | ||
| Pigment (%) | Chlorophyll a (Chla) | 0.0121–0.045 | (Han et al., 2017, Wu et al., 2015; Xu et al., 2010; Yu et al., 2008) |
| Carotenoid (Car) | 0.3–0.68 | ||
| phycoerythrin (PE) | 0.035–0.0276 | ||
| phycocyanin (PC) | 0.006–0.020 | ||
| Amino acid (%) | EAAs | 0.3619–0.679 | (Chen et al., 2018, Zhou et al., 2010) |
| NEAAs | 0.6116–1.174 | ||
| Asp | 0.098–0.231 | ||
| Thr | 0.0571–0.088 | ||
| Ser | 0.0618–0.118 | ||
| Glu | 0.1373–0.249 | ||
| Gly | 0.0623–0.115 | ||
| Ala | 0.0725–0.139 | ||
| Val | 0.067–0.109 | ||
| Cys | 0.0027–0.0097 | ||
| Met | 0.0141–0.027 | ||
| Ile | 0.0565–0.09 | ||
| Leu | 0.0835–0.161 | ||
| Tyr | 0.0285–0.077 | ||
| Phe | 0.0533–0.098 | ||
| His | 0.0175–0.036 | ||
| Lys | 0.056–0.097 | ||
| Arg | 0.0567–0.13 | ||
| Pro | 0.493–0.75 | ||
| Taurine | 0.801–0.949 | ||
| Mineral (%) | Ca | 0.048 | (Zhou et al., 2010) |
| Mg | 0.249 | ||
| K | 1.15 | ||
| Na | 0.792 | ||
| Fe | 0.218 | ||
| Zn | 0.0036 | ||
According to the literatures, polysaccharide is one of the prime functional components from Gracilaria lemaneiformis, which is mainly composed of 3,6-dehydrated l-galactose and d-galactose, as well as belongs to acidic polysaccharide and has linear structure of repeated disaccharide-agarose unit (Chen et al., 2020, Chen et al., 2010, Sun et al., 2018, Xu et al., 2018). Kang, Xie, Sun, Yang, & Zhao (2017) detected the contents of 3,6-dehydrated l-galactose (41.2%) and d-galactose (57.38%) of Gracilaria lemaneiformis polysaccharide. Li, Yu, Jin, Zhang, & Liu (2008), and Liao, Yang, Chen, Yu, Zhang, & Ju (2015) observed that the content of 3,6-dehydrated l-galactose was 26.2–41.4%. The polysaccharide extracted from Gracilaria lemaneiformis is mainly belong to agar polysaccharide, which is a poly-galactan chain composed of 1,3-linked β-d-galactopyranose and 1,4-linked 3,6-anhydro-l-galactose with some ester sulfate, pyruvic acid acetal or methyl groups (Xu et al., 2018). Additionally, Gracilaria lemaneiformis polysaccharide exhibits a variety of biological activities, including regulation of glycolipid metabolism, immune, anti-tumor, anti-inflammatory and other biological activities, which possesses the high nutritional value for health care products and functional foods (Cardoso et al., 2016, Courtois, 2009, Jiang et al., 2014). Han, Wang, Zhao, You, Pedisic, and Kulikouskaya (2020) indicated that GLP (with 200, 400 and 600 mg/kg) improved the body weight and appetite of mice, and inhibited tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6) and interleukin-1β (IL-1β) in colon tissue, as well as repaired colon injury of mice. Moreover, these biological activities may be related to the molecular weight and functional groups (carboxyl group, hydroxyl group, acetyl group, sulfate group, and other groups) of GLP, which may enhance or weaken the physiological activities of GLP (Anand et al., 2018, Chattopadhyay et al., 2008). For example, Zhang et al., 2020, Han et al., 2020 found that sulphated polysaccharide of Gracilaria lemaneiformis had the ability to regulate the abundance and diversity of intestinal microorganisms, and then presented the potential of prebiotics.
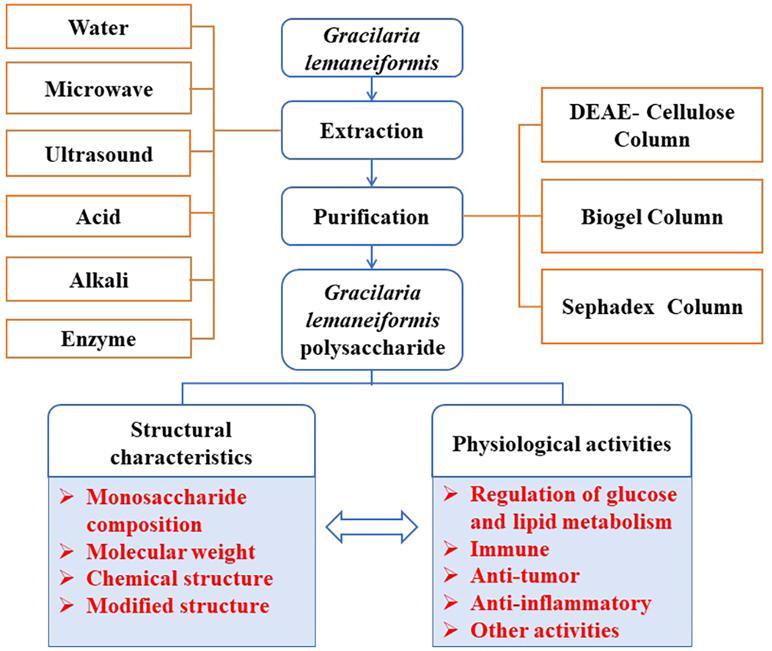
In this review, recent advances in the study of the extraction, purification, structural characteristics, and physiological activities of GLP have been investigated and summarized systematacially. Therefore, it is beneficial to afford theoretical foundation for researchers to open up further functional foods and nutraceuticals of Gracilaria lemaneiformis, so that to promote the sustainable processing and high value utilization industry of Gracilaria lemaneiformis.
2. Extraction and purification of GLP
Gracilaria lemaneiformis is relatively hard, so it must be mechanically crushed into powder to be fully extracted, and the polysaccharide can be extracted in a greater extent. After the Gracilaria lemaneiformis collected from the ocean, the visible impurities, such as sand and insects, are removed by clean water, dried in the oven, and then crushed into powder, which is used as the original extraction after screening.
Polysaccharide is a macromolecular compound with large polarity and low solubility in organic solution, which is easy to precipitate. Generally, in order to obtain high purity polysaccharide and simplify purification steps, acetone, ethanol, dichloromethane, methanol/chloroform and other organic solvents are selected for preliminary treatment of Gracilaria lemaneiformis powder to remove pigment, fat and other non-polar components (Zhang, Wen, Chen, Gu, Zhou, & Duan, 2019). There are numerous ways of extraction and purification of polysaccharide, so we should find the best way to extract and purify polysaccharide by comparing other’s researches, in order to attain more yield of polysaccharide.
2.1. Extraction technology of GLP
The main extraction methods of GLP include water extraction method (Kang et al., 2016), microwave extraction (Sousa, Alves, Morais, Delerue-Matos, & Gonçalves, 2010), ultrasonic extraction (Youssouf et al., 2017), enzyme extraction (Chen, Jia, Zhu, Zou, & Huang, 2019), and acid/alkali extraction (Teng et al., 2020).
Polysaccharide pertains to the polyhydroxyaldehyde polymer with good water solubility. Consequently, the water extraction method is conducive to the full extraction of GLP, which is mild, high safety, low cost and easy operation, as well as will not introduce new impurities. What’s more, water extraction does not destroy the structure of GLP. However, compared with other methods, the solvent extraction method takes longer time. For instance, Khan et al. (2019) prepared the GLP (the yield is 24%) by hot water extraction for 2 h. In Table 2, GLP was extracted by cold/hot water for 1 to 5 h, and the yield of polysaccharide was from 29.7% to 34.84%. The content of sulfurated polysaccharide was on a scale of 0.45% to 22.16%. The difference in yield of polysaccharide may be connected with the extraction temperature, time, solid–liquid ratio or other factors (Chen et al., 2010, Fan et al., 2012, Li et al., 2008, Liao et al., 2015, Liu et al., 2016, Xu et al., 2018, Zhang et al., 2020).
Table 2.
The extraction, purification, monosaccharide, molecular weight and biological activities of Gracilaria lemaneiformis.
| Component | Extraction | Purification | Yield of polysaccharide/total sugar | Yield of sulfurated polysaccharide | Monosaccharide | Molecular weight | Biological activities | Reference |
|---|---|---|---|---|---|---|---|---|
| GLP | cold water | dialyze | – | 6.13% | galactose, rhamnose, arabinose, xylose ,mannose(29.64:0.19:0.21:0.59:1.00) | 1.37 × 10 6 Da | anti-tumor | (Fan et al., 2012) |
| GLP | hot water | DEAE-cellulose 52 | – | 11.26% | galactose | 152481 Da | anti-food allergic | (Liu et al., 2016) |
| GLP | hot water | Sephadex LH-20 | – | 7.65% | galactose (56.6%) | – | antioxidant | (Xu et al., 2018) |
| GL-1, GL-2,GL-3 | hot water | DEAE-cellulose-52, sephadex G-100 | – | 0.45%,12.63%,22.16% | – | 4900 Da, 52000 Da, 67000 Da, | anti-influenza virus | (Chen et al., 2010) |
| GLP,GLP1,GLP2, | hot water | dialyze | 98.2%(total sugar) | 8.24%,8.12%,8.14% | – | 121.8 kDa,57.02 kDa,14.29 kDa | hypoglycemic,antioxidant | (Liao et al., 2015) |
| GLP | hot water | – | 34.84 % | 4.80% | galactose | 2.15 × 105Da | prebiotic | (Zhang et al., 2020) |
| GLP | hot water | DEAE-C | – | – | – | – | antitumor, anticancer | (Kang et al., 2016) |
| GLP2 | ultrasonic/microwave-assisted | DEAE-cellulose 52 | – | – | galactose and3,6-anhydrogalactose (1.0:1.5) | 1.57 × 105 Da | wound healing | (Veeraperumal et al., 2020) |
| GLP | water | – | 29.7% | 2.96% | – | – | – | (Li et al., 2008) |
| NaOH | 25.8% | 0.89% | ||||||
| NaOH + sodium hypochlorite | 25.4% | 0.81% | ||||||
| photobleached | 25.4% | 0.76% | ||||||
| GLP | hydrogenperoxide-assisted enzymatic method(EHA) | – | 16.08 % | 3.56 % | – | – | – | (Chen et al., 2020) |
| Alkali(AA) | 13.11 % | 1.8 % | ||||||
| Enzymatic(EA) | 17.79 % | 4.51 % | ||||||
| GLP | citricacid | – | 59.93% (total sugar) | 29.82% | galactose (71.78%) ,glucose (13.48%) | 21055 Da | attenuatingintestinal inflammation | (Han et al., 2020) |
| GLP | citricacid | dialyze | 59.93% (total sugar) | 29.82% | rhamnose, arabinose, fucose, xylose, mannose, glucose, galactose | 21.85 kDa | digestibility and prebiotic | (Han et al., 2020) |
| GLP2 | citric acid | DEAE-Sepharose | 25.55% | 3.06% | xylose (18.12%), mannose (0.33%), glucose (35.17%), galactose (45.84%) | 18501 Da. | immunomodulation | (Ren et al., 2017) |
| GLP | a-amylase | – | 49.15% (total sugar) | – | rhamnose, arabinose, xylose ,mannose | – | antioxidant | (Wu et al., 2017) |
| algal oligosaccharides (AOS) | fermentation | DEAE-cellulose 52, Bio-gel P2 | 7.4% | – | – | – | antioxidant, attenuated alcohol-inducedhepatopathy | (Jin et al., 2017) |
| GLP,P-1,P-2,P-3 | – | DEAE-A25, SephadexG-100 | 93.57% (total sugar) | 9.24%,8.15%,9.16%,8.14% | rhamnose, fucose, arabinose, xylose, mannose,glucose, and galactose | 123.06 kDa,14.29 kDa,64.78 kDa,57.02 kDa, | antitumor, anticancer | (Kang et al., 2017) |
Ultrasonic and microwave extraction methods can break the cell wall of Gracilaria lemaneiformis, which have the advantage of high safety, environment-friendly and mild conditions. Both of these two methods can promote the molecule to transfer into the extraction solution, and improve the extraction efficiency. They can be used together or combined with other extraction methods as auxiliary means in order to raise the yield of polysaccharide (Chen, Gu, Huang, Li, Wang, & Tang, 2010). Shi, Yan, Cheong, & Liu (2018) used both ultrasonic and microwave to extract GLP (the microwave power was 800 W, the frequency was 2450 MHz, the ultrasonic power and frequency were 50 W and 40KHz respectively, the extraction time was 31.7 min, the temperature was 87° C, and the solid–liquid ratio was 1.0:60.7), finally the polysaccharide content was 34.8%. Veeraperumal et al. (2020) also used ultrasonic/microwave-assisted method to extract GLP. Compared with the traditional hot water extraction method, ultrasonic microwave extraction has the advantages of short extraction time, high extraction efficiency, and less solvent demand (Chen et al., 2010). On the other hand, ultrasound and microwave are able to break down cell walls and may cause the depolymerization of polysaccharide, which may change its molecule and active groups.
Compared with other methods, enzymatic extraction has the advantages of high catalysis, high specificity and mild reaction conditions. The specific enzyme may selectively promote the cell wall decomposition and accelerate the dissolution of intercellular polysaccharide. It acts on specific glyosidic bonds to produce specific polysaccharide. In addition, the yield of polysaccharide is correlated with the time of enzymatic hydrolysis and the type of enzyme. Zhang et al. (2019) used agar hydrolase to prepare polysaccharide from Gracilaria lemaneiformis, and the yield of polysaccharide enhanced with the increase of enzymatic hydrolysis time at the initial stage of enzymatic hydrolysis. However, the yield of polysaccharide remained unchanged and kept at a stable level as the enzymatic hydrolysis time reached 160 min. As given in Table 2, Chen et al. (2020) used catalase to extract agar polysaccharide from Gracilaria lemaneiformis, the yield of polysaccharide was 16.08 %, and the content of sulfate group was 3.56 %, which was 1.76% higher than that obtained by alkali extraction. Wu et al. (2017) extracted GLP by amylase, and its polysaccharide content reached as high as 49.15%. It was observed that the content of GLP was related to the type of enzyme, and the yield of polysaccharide could be as high as 40% by selecting the right enzyme. Nevertheless, enzymatic extraction may not be suitable for industrial production because of its high cost and limited source.
Researches have shown that polysaccharide obtained from Gracilaria lemaneiformis by citric acid is a common extraction method in recent years. Compared with hot water and ultrasonic extraction methods, citric acid can effectively improve the yield of polysaccharide, increase the content of aldehyde acid, reduce the molecular weight and viscosity, and improve the relative homogeneity of polysaccharide (You, Zhang, Wen, Liu, & Fu, 2016). It was proved that the level of total sugar and sulfated polysaccharide could reach 59.93% and 29.82%, separately (Han et al., 2020, Han et al., 2020) (Table 2). In alkaline treatment, sodium hydroxide was generally used for extraction. The content of polysaccharide and the sulfate group of galactose was lower than other methods, and the antioxidant activity and other physiological activities were reduced (Ibrahim et al., 2017). For example, Fan et al. (2012) extracted agar polysaccharide from Gracilaria lemaneiformis by NaOH, and the yield was only 2.06% (Table 2). As shown in Table 2, GLP was extracted by the modified alkaline method, and the yield of polysaccharide was 25.4–25.8%, lower than water extraction (29.7%), but the level of 3,6-Anhydrogalactose was higher than water extraction (39.5–41.4% vs 26.2%) (Li, Yu, Jin, Zhang, & Liu, 2008). Compared with alkaline method, the yield of polysaccharide extracted by citric acid method was higher, and the content of polysaccharide extracted by alkali method was lower. On the other hand, alkali extraction of polysaccharides has a great negative impact on the environment, may also destroy the structure of polysaccharide, and reduce the active groups of polysaccharides. (Courtois, 2009, Jin et al., 2017).
In general, water and acid extraction can attain higher yield of polysaccharide compared with enzymatic, alkaline and fermentation methods, which are also more commonly used extraction methods.
2.2. Isolation and purification of GLP
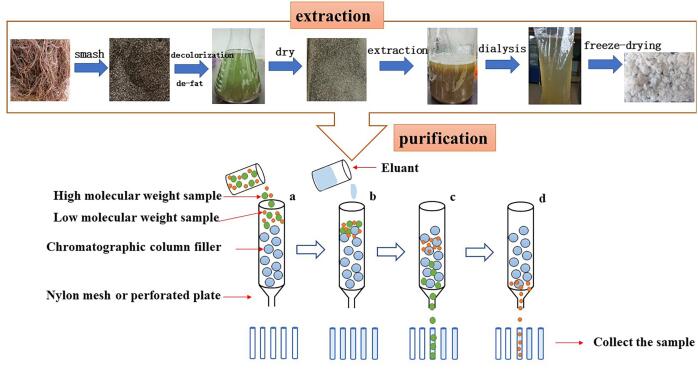
The polysaccharide, obtained by different methods, just is a kind of crude component with pigment, protein and other substance, so it is necessary to purify through alcohol, dialysis and column chromatography. Column chromatography, including ion-exchange chromatography, gel permeation chromatography and affinity chromatography, is a frequently used purification method for polysaccharide. As we know, ion-exchange chromatography mainly includes DEAE-cellulose, EDAE/QAE/CM/SP-sephadex, Q/SP sepharose high performance, and so on, which have exchange agents with different bond strength (Mazumder, Lerouge, Loutelier-Bourhis, Driouich, & Ray, 2005). Gel permeation chromatography primarily contains sephadex, sephacryl, biogel, and sepharose, which are made of molecular sieve with porous network structure, and the elution separation is carried out according to the difference of molecular weight of the separated samples. With the flow of the eluent, sample with relatively large molecular weight flow directly out from the voids of the filler, while small molecules can pass through the filler to prolong the outflow time. Therefore, the larger molecules elute faster than the smaller ones (Fig. 2). Sephadex is a bead shaped gel containing a large number of hydroxyl groups, which is easy to swell in water and electrolyte solution. Moreover, Sephadex is a macroporous compound with porous network structure formed by cross-linking of dextran molecules and crosslinking agent 3- chloro −1, 2- propylene oxide. According to the degree of cross-linking, it can be divided into G-10, G-15, G-25, G-50, G-75, G-100, G-150 and G-200, and the number after G means 10 times of the water value of the gel , which can attain polysaccharide with different molecular (Zhao, Quyang, Chen, Zhao, & Qiu, 2018). Sephacryl is composed of acrylamide and a small amount of crosslinking agent methylene bis acrylamide, which is polymerized under the action of ammonium persulfate and divided into biogel P-2, P-4, P-6 and so on, according to the degree of cross-linking (Loa et al., 2002). Affinity chromatography is a kind of chromatographic method, which uses the binding properties of stationary phase to separate molecules. When the sample flows through the chromatographic column with the mobile phase, the ligand on the affinity adsorbent selectively adsorbs the substance that can be bound with it, while other proteins and impurities are not adsorbed and flow out from the chromatographic column. The purified target product can be obtained by using appropriate buffer to desorb the separated substance and ligand (Gagnon et al., 2021).
Fig. 2.
The extraction and purification in gel permeation chromatography of polysaccharide.
The crude polysaccharide is isolated and purified by column chromatography, mainly including DEAE-cellulose, biogel and sephadex column, so that the GLP with different purity were obtained (Shang, Chu, Zhang, Zheng, & Li, 2021). The purification degree, molecular weight and content of the obtained polysaccharide will be different with various column models. In general, deionized water, salt solution, buffer solution and other eluent are selected to wash out the samples to attain different purified composition. In order to gain polysaccharide components with higher purity, DEAE-cellulose, biogel and sephadex column are usually used for fractional purification of polysaccharide one after another to purify the same specimen. As exhibited in Table 2, the crude polysaccharide was slowly added into DEAE-52 cellulose column and GLP was collected after eluting by NaCl solution, and then a second separation was performed on a biogel P2 column with washing and elution by NH4HCO3 solution. Finally, the content of total sugar of GLP was 98.7%, and reducing sugar of oligosaccharide was 95.2%, detected by phonel-sulfate method and the 3,5–2 nitro salicylic acid method, respectively (Jin et al., 2017). Di, Chen, Sun, Ou, Zeng, & Ye (2017) reported that polysaccharide from Gracilaria lemaneiformis was eluted by DEAE-52 cellulose column and sephadex G-50 successively, and then three purified components (the contents were 6.00%, 12.47% and 26.87%, respectively) were obtained. Shi et al. (2018) obtained three polysaccharide components of Gracilaria lemaneiformis after purification by DEAE-A50 cellulose column, the contents of which were 14.3%, 65.2% and 20.6%, respectively. Kang et al. (2017) purified GLP through DEAE-A25 cellulose column and sephadex column G-100 successively, and got three purified components, with contents of 12.61%, 69.26% and 18.70%, respectively (Table 2).
In addition to the above frequently used methods of purification, membrane separation (natural or synthetic polymer film as medium, and external energy or chemical potential difference as driving force), stepwise precipitation (the solubility of polysaccharide with different molecular weight in different concentrations of organic solvents is variant), and salt fractionation (a large amount of inorganic salts will precipitate macromolecular substances) are also used for the purification of polysaccharide.
3. Structural characteristics of GLP
3.1. Monosaccharide composition
Studies have already demonstrated that the monosaccharide composition is analyzed by gas chromatography-mass spectrometry (GC–MS) or liquid mass chromatography (HPLC-MS) after hydrolysis and derivatization of the polysaccharide (Han et al., 2020). GC–MS is an analytical method that combines gas chromatography with mass spectrometry. Mass spectrometry can be used for effective qualitative analysis, but it cannot analyze more complex organic compounds. However, gas chromatography is an effective separation and analysis method for organic compounds, especially suitable for quantitative analysis of organic compounds, but qualitative analysis is more difficult (Jung et al., 2021, Wang et al., 2021). Therefore, the effective combination of these two methods, namely GC–MS, can be used for the efficient analysis of complex organic compounds such as carbohydrate. GC–MS analysis has the advantages of high selectivity, good resolution, high sensitivity and fast analysis speed. However, carbohydrate is almost nonvolatile, so hydrolysis and derivative reactions of carbohydrate should be carried out before GC–MS analysis, which is converted into volatile and thermal stable derivatives for further analysis (Yun, Cui, Zhang, Zhu, Peng, & Cai, 2021). Moreover, isomers of carbohydrate may be produced in the process of preparing some derivatives. In chromatographic analysis, a kind of carbohydrates may produce several peaks, which usually affect the separation and quantitative analysis of components. Liquid chromatography-mass spectrometry (HPLC-MS) is another detection technique, which uses liquid chromatography as separation system and mass spectrometry as detection system (Gremmel, Fromel, & Knepper, 2017). The sample is separated from the mobile phase in the mass spectrum, and detector obtains the ion fragments are separated by mass analyzer after ionization. High performance liquid chromatography (HPLC) embodies the complementary advantages of chromatography and mass spectrometry, which combines the high separation ability of chromatography for complex samples with the advantages of high selectivity, high sensitivity and the ability to provide relative molecular weight and structure information of mass spectrometry (Strickley, Brandl, Chan, Straub, & Gu, 1990). Naturally, it has been widely used in many fields such as pharmaceutical analysis, food analysis and environmental analysis. The original place (growing condition) and collection season of Gracilaria lemaneiformis may affect the composition and content of monosaccharide and polysaccharide. On the other hand, the purification and degradation of polysaccharide backbone during sulfation may also lead to the change of monosaccharide composition (Soriano and Bourret, 2003). GLP is mainly composed of d-galactose and 3,6-dehydrate-galactose units, with a linear structure of repeated disaccharide-agarose units. The monosaccharide composition of Gracilaria lemaneiformis is mainly galactose, and contains a small amount of rhamnose, arabinose, fucose, glucose, xylose and mannose. As illustrated in the Table 2, Ren, You, Wen, Li, Fu, & Zhou (2017) found that the galactose (45.84%) and glucose (35.17%) were the central monosaccharide components of Gracilaria lemaneiformis. Fan et al. (2012) detected GLP by gas chromatography-mass spectrometry (GC–MS), which was mainly composed of galactose, and also contained a small amount of rhamnose, arabinose, xylose and mannose (their molar ratio was 29.64:0.19:0.21:0.59:1.00). Kang et al. (2017) hydrolyzed and derivatized GLP and then determined its monosaccharide components by GC–MS as rhamnose, fucose, arabinose, xylose, mannose, glucose and galactose, among which galactose was the main component.
In addition to the above methods, there are other methods for detecting monosaccharide composition, such as ion chromatography coupled with pulsed amperometric detection (IC-PAD) and high-performance anion-exchange chromatography (HPAEC), which have prominent advantages in sensitivity, accuracy, ease of operation and no consumption of organic solvents (Zhu, Zhang, Wang, Li, Wang, & Huang, 2019).
3.2. Molecular weight
It has been found that molecular weight is a key factor to regulate physiological activities of polysaccharide, such as antioxidant, immunomodulation, and other activities (Pengzhan, 2003). The physiological effects of polysaccharide are closely related to the molecular weight. According to literatures, polysaccharide molecule is hard to form the active polymer structure when the molecular weight is too small, and hard to cross the cell membrane when the molecular weight is too large (Yuan et al., 2020). Therefore, it is necessary to select the appropriate extraction and purification methods to prepare polysaccharides and control the size of their molecular weight. In a certain molecular weight range, the activities of polysaccharide increased with the decrease of relative molecular weight (Hou et al., 2017). In addition, the variation of molecular weight may be related to species, purification degree and analytical method. The molecular weight of polysaccharide was determined by high-performance size exclusion chromatography (HPSEC), high performance gel chromatography (HPGPC), high performance liquid chromatography (HPLC), polyacrylamide gel electrophoresis (PAGE), and so on. The molecular weight of GLP has measured to be 103 Da ∼ 106 Da. As given in Table 2, Khan et al. (2019) determined that the molecular weight of GLP was 7.185 × 105 Da by HPSEC. Shi et al. (2018) detected the molecular weight of GLP (GLP1 was 5.5 kDa, GLP 2 was 85 kDa and GLP 3 was 82 kDa) with HPLC which illustrated that the molecular weight of polysaccharide components obtained by using different eluents was significantly different. GLP1 had the smallest molecular weight, but it did not show the best activity, and GLP3 exhibited the highest anti-tumor activities. Di et al. (2017) measured the molecular weight of GLP as 1310 (GLP 1), 691 (GLP 2) and 923 kDa (GLP 3), respectively. GLP 3 exhibited the best antioxidant and immune activities. Kang et al. (2017) used PAGE to measure the molecular weight of three purified polysaccharide from GLP as 123.06, 14.29, 64.78 and 57.02 kDa, respectively (Table 2). Fan et al. (2012) determined the molecular weight of GLP by HPGPC was 1.37 × 106 Da. Liao et al. (2015) tested the molecular weight of GLP by gel chromatography was 121.89 kDa, and the molecular weight of these two polysaccharides after purification were 57.02 (GLP1) and 14.29 (GLP2) kDa, respectively. GLP, GLP1, GLP2 had no significance on structure and hypoglycemic activity, which was not consistent with other studies. The possible reason was that the molecular weight of these polysaccharides were so large that there were no significant change with each other. All of these results explained that the range of molecular weight of polysaccharide was large, and it declined after purification or degradation. The biological activities of polysaccharide may be related to its molecular weight, and the polysaccharide with low molecular weight may show the stronger activities than that of high molecular weight in a certain range. Beyond a certain range, the biological activities also can decrease as molecular weight declines. Moreover, some studies also have shown that the activities of polysaccharide are not related to its molecular weight, but most studies have shown that they are relevant.
3.3. Chemical structure
It is well known that the methods used to explore the structure of polysaccharide include High performance liquid chromatography (HPLC), Fourier-transform infrared spectrometry (FTIR), Nuclear magnetic resonance spectrum (NMR), methylation analysis, High performance gel permeation chromatography (HPGPC), and Gas chromatography (GC) (Li et al., 2020, Yang et al., 2021). Among them, IR and NMR are used to detect the chemical structure, sequence and primary structure of polysaccharide.
FTIR is the common method for detecting the chemical structure of polysaccharide, which causes the transition of vibrational energy levels and rotational energy levels in molecules. It can detect the position and intensity of absorption peaks of functional groups of polysaccharide, thus reflecting the characteristics of polysaccharide (Beratto-Ramos et al., 2020, Shi et al., 2020). After detecting the structure of GLP by FTIR, it was found that it had sulfate group, 3,6-anhydrous galactose, C—H, C—O, O—H and glycoside bonds, in which the absorbance bonds of sulfate group mainly occurred at 1260–804 cm−1. As we known that 1200–950 cm−1 is the specific region of polysaccharide and the characteristics of polysaccharide are showed according to the position and intensity of the bands. There is a strong peak at 3449 cm−1, indicating C—H tensile absorbance. The strong peaks at 1031 and 1072 cm−1 are attributed to the tensile absorbance of C—O—H and C—O—C from the glycoside bonds. Generally, the peak at 932 cm−1 is the C—O stretching of 3,6-anhydrous-α-l-galactose. The strong signals at 1259 cm -1 and 804 cm -1 are usually due to the stretching of the S O and C—O—S of sulfate group from polysaccharide (Fan et al., 2012). Shi et al. (2018) detected functional groups of GLP by FTIR that there was hydroxyl stretching at 3400 cm−1, C—O—C stretching of glycoside bond at 1072 cm -1, strong signal of S O bond of sulfate group at 1260 cm -1, and d-galactose absorption peak at 870 cm -1. It confirmed that GLP had the sulfate group, which may affect its antitumor activity. Wu et al. (2017) found O—H and C—H stretching at 2904–3346 cm−1, sulfate group at 1038–892 cm−1 and α (1 → 4) glyosidic bond at 1196–1147 cm−1. This indicated that GLP contained α (1 → 4) glyosidic bond and sulfate group, and showed strong antioxidant activity due to its sulfate group. The chemical structure of GLP was determined using FTIR by Wang et al., 2019, Chen et al., 2010, and the results showed that –OH, C—H and C—O existed at 3289, 2920 and 1070 cm−1, respectively. Moreover, the characteristic peak of 3,6-anhydrous-galactose was detected at 931 cm−1, and the signal of sulfate group appeared at 1260 cm−1 and 850 cm−1. These results showed that GLP had sulfate group, 3,6-anhydrous galactose and α (1 → 4) glyosidic bond. GLP with sulfate group presented the obvious activities, which demonstrated that sulfate group was able to provide electrophilic groups to enhance the activities.
NMR spectroscopy is commonly used to analyze the structure of polysaccharide, including one-dimensional NMR (1D-NMR) and two-dimensional NMR (2D-NMR), which can be used to determine the primary structure, sequence of monosaccharide residues and the configuration of glyosidic bonds (Beri, & Gandhi, 2021). 1D-NMR mainly includes 1H NMR, 13C NMR and DEPT-135. 1H NMR is mainly used to estimate the configuration of glyosidic bond in polysaccharide structure, and the number of heterohead hydrogen signal can reflect the composition of monosaccharide (Doumert, Lecomte, & Tricot, 2020). The chemical shifts from 4.56 to 5.28 ppm in 1H NMR spectrum were corresponded to anomeric protons that were related with glyosidic bonds. It suggested that oligosaccharide of Gracilaria lemaneiformis contained the α-configuration of the pyranose unit (Liu et al., 2020). β-Gal and α-(1 → 4) and β-(1 → 3) linked galactopyranosyl units was presented at 3.81 and 4.67 ppm in 1H NMR spectrum (Li et al., 2020). AnGalp-2-sulphate linked to Galp, galactopyranosyl and 3,6-α-l-AnGal displayed C-1/H-1 has been found at the signal of 5.28, 4.56, 4.96/97.94 ppm, respectively (Liu et al., 2020). In addition, 1H NMR spectrum showed anomeric carbon 3,6-anhydrogalactose at 5.09, galactose-4-sulfate at δ 4.6 and 3,6-anhydrogalactose-2-sulfate at δ 5.2 (Veeraperumal et al., 2020). 13C NMR has the ability to determine the number and relative content of sugar residues, the position of sugar chain connection and sulfate acid substitution, and the configuration of heterocephalic carbon (Roslund et al., 2011). The 13C NMR spectrum presented 3,6-anhydro-l-galactose and C-1 of β-d-galactose and from the enzymatic GLP at the chemical shift of 90.72/98.5 and 102.6 ppm, respectively (Veeraperumal et al., 2020, Xu et al., 2018). The signals at 92.13, 74.95, 77.8, 78.43, 76.88 and 69.17 (H6 δ 4.67) ppm in the 13C spectrum were attributed to the carbon of 2-O-sulfate-3,6-anhydro-α-d-galactose (Li, Huang, Chen, Xu, Ma, & You, et al., 2020), which implied sulfate group and 3,6-anhydro-α-d-galactose existed in the polysaccharides chain, and both of these two were linked by glyosidic bond. The content of sulfate group was 23%, which was consistent with the result of NMR. GLP could regulate the glycolipid metabolism in mice, which may be attributed to the sulfate group. Chen et al. (2020) found that agar of Gracilaria lemaneiformis had the backbones of the type G → LA and G → L6S. d-galactose-6-sulphate and d-galactose-4-sulphate were observed at 67.3 and 69.1 ppm, and C-6 and C-4 sulfate groups were absent in 13C NMR. In this study, the sulfate group was attached to d-galactose. Furthermore, it showed that GLP included nine anomeric residues, including → 4)-α-d-Glcp-(1→,→6)-→-d-Glcp-(1→,→6)-β-d-Galp-(1→,→3,6)-α → -l-Manp-(1→, α-d-Galp-(1→,α-d-Xylp-(1→,→3)-α-d-Galp-(1→,→4)-α-d-Galp-(1→,and → 3,6)-β-d-Galp-(1→, respectively, via the 1H and 13C NMR spectrum (Ren et al., 2017). NMR results showed that d-galactose was the main monosaccharide component of polysaccharide, which was consistent with the results of monosaccharide composition (xylose: mannose: glucose: galactose = 18.12:0.33:35.17:45.84). The main connection way of glyosidic bonds were made of (1→,→3) and (1→,→6). Moreover, GLP formed by this connection had obvious immune activity. On the other hand, 2D-NMR mainly includes COSY, HSQC, HMBC and NOESY, which can provide the type of monosaccharide residues, the C and H chemical shifts of each sugar residue, and determine the connection position and sequence between sugar residues, so that to reflect the complete structural information of polysaccharide (Martineau, Khantache, Pupier, Sepulcri, & Giraudeau, 2015). The HSQC spectrum displays the connection of anomeric carbons with their respective protons at δ 102./4.47 for 3-linked β-d-galacto-pyranose, δ 101.9/4.54 for α-l-galactose-6-sulfate and δ 98.0/5.1 for 4-linked 3,6-anhydro-α-l-galactopyranosyl residue. The H-2 protons assigned using the COSY spectrum that showed a correlation on the HSQC spectrum with the C-2 atom (C-2 of 4-linked 3,6-anhydro-α-l-galacto-pyranose δ 69.2, C-2 of 3-linked β-d-galactopyranose δ 69.7) (Veeraperumal et al., 2020). The order of monosaccharide composition and the position of active groups are decided by the type and connection of glyosidic bonds. As mentioned above, the physiological activities of polysaccharide have obvious correlation with its glycoside bonds, monosaccharide composition, molecular weight and active groups.
3.4. Modified structure
Sulfate group is the main functional and active group, which plays a vital role in the characterization of physiological activities of organism, as well as accelerates algal polysaccharide to express the biological activities of anticoagulant, antithrombotic and other physiological activities (Lee, Chao, Hsu, & Lu, 2020). The glyosidic bonds of sulfate group include (1 → 2), (1 → 3), (1 → 4) and (1 → 6), of which (1 → 3) - glycoside bond is the main one, accounting for 86% (Han et al., 2020). The literatures have shown that sulfate modification has the ability to improve the solution properties of polysaccharide, enhance the negative charge on the surface of polysaccharide, and strengthen its antioxidant, antitumor, immune and antiviral activities (Cardoso, Costa, & Mano, 2020; Di et al., 2017). Chen et al. (2010) extracted GLP with hot water, and purified by DEAE-cellulose column and sephadex column G-100, and three sulfated polysaccharides were obtained successively (Table 2). Their sulfated degree was 0.45%, 12.63% and 22.16%, respectively. All of them presented significant anti-influenza virus activity, which was enhanced with the increase of the content of sulfate group. A study was conducted to investigate the effect of sulfated content of GLP on its antiviral activity, and the result showed that the antiviral activity was also reinforced with the increase of content of sulfate group in a certain range (Chen, Liao, & Chen, 2008). The sulfate group is connected to the main chain or branch chain of polysaccharide. The order of main chain or branch chain are related to the type and mode of glyosidic bonding, and the number and position of monosaccharides, which lead to the difference of content and position of the sulfate group. In addition, the yield of polysaccharide and content of sulfate group are different by diverse methods (water extraction, acid extraction, alkali extraction, etc.).
The position and number of sulfate group on the polysaccharide skeleton have great influence on its activities, which can significantly promote the activities of polysaccharide. Sulfate group is able to enhance the activities of GLP because it can provide electrophilic groups to enhance the activities, fortify the digestive enzyme activities of colon contents and restore the production of short-chain fatty acids (Xu et al., 2012, Huang et al., 2021)
Phosphate and acetyl groups also have an important role in the physiological activities of GLP. The content and position of these active groups may potentially decrease the ability of the polysaccharide to induce the desired effect. On the other hand, hydrolysis with acetic acid or other methods may change the content and position of phosphate and acetyl group, and then change the activities of polysaccharide (Chang et al., 2012). At present, there are numerous studies on the sulfated groups of polysaccharides in Gracilaria lemaneiformis, but the effects of phosphate and acetyl groups on the activities of GLP need to study further.
4. Physiological activities of GLP
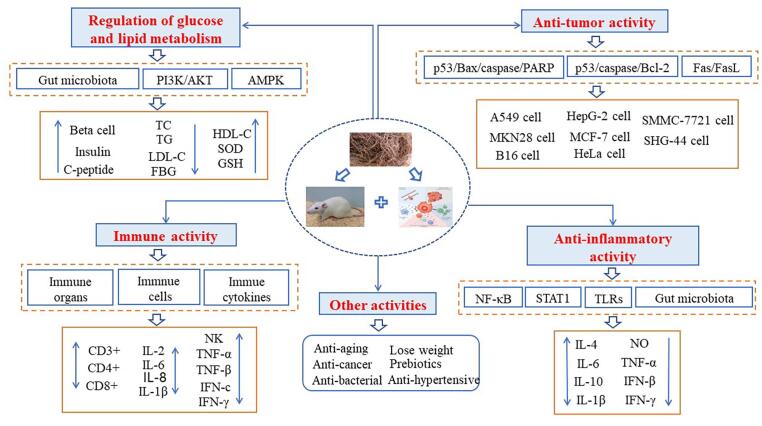
Recent studies have shown that GLP has physiological activities of regulation of glycolipid metabolism, immune, anti-tumor, anti-inflammatory and other biological activities (Fig. 3), which can be used to develop functional food or drugs. Its molecular weight, monosaccharide composition, active groups and glyosidic bond may affect these biological activities. However, the mechanism of physiological activities and the relationship between structure and physiological activities of GLP have not been systematically elucidated.
Fig. 3.
Physical activity of Gracilaria lemaneiformis polysaccharide.
4.1. Regulation of glycolipid metabolism
Long-term consumption of high-fat food will lead to disorder of glucose and lipid metabolism, leading to chronic metabolic diseases, such as obesity, diabetes, hyperlipidemia and so on, thus inducing more syndrome (Chen et al., 2021).
At present, several possible mechanisms of regulation of glycolipid metabolism have been reported, principally including gut microbiota, phosphatidylinositol 3-hydroxykinase/Akt (PI3K/Akt) and adenosine monophosphate-activated protein kinase (AMPK) signaling pathways. As reported, gut microbiota plays an important role in the regulation of lipid metabolism. It is able to utilize the undigested and decomposed carbohydrates to produce short chain fatty acids or other small functional molecules, which participate in the organism’s glycolipid metabolism (Zhao et al., 2020). Furthermore, gut microbiota may have the ability to attenuate the glycolipid metabolism of human body via improving the insulin sensitivity or activities of glycolipid-related enzymes, and ameliorating the status of glycolipid metabolism, including the level of blood glucose and lipid, and other serum indexes (Karlsson et al., 2013). Huang et al. (2019) proved that sulfated polysaccharide from Gracilaria Lemaneiformis regulated the relative abundances of Bacteroides, Ruminococcus_1 and Lactobacillus, which significantly improved the lipid metabolism and ameliorated the symptom induced by high-fat diet. The sulfate group and gut microbiota played an important role in the regulation of glycolipid metabolism by GLP. Liao, Long, Zou, Liu, & Li (2021) discovered that the reduction of Firmicutes/Bacteroidetes ratio improved the bioaccessibility, bioavailability and lipid metabolism, thus reducing the Lee’s index and perfecting the level of lipid (TG, TC, LDL-C, HDL-C). Polysaccharide can not be digested in gastric and intestinal juice, but degraded into functional small molecules by gut microbiota in colon, exerting their activity in regulating glucose and lipid metabolism.
The PI3K/Akt and AMPK signaling pathways also belong to the possible mechanism pathways to play the role of anti-hyperglycemic and anti-hyperlipidemic activities. Normally, PI3K is an intracellular phosphatidylinositol kinase, a key factor to regulate glucose and lipid metabolism, and protein kinase B (Akt) is the main factor of downstream signaling network (Xu et al., 2019). Glucose transport has the closed connection with numerous insulin signaling pathways, such as PI3K and MAPK signaling pathways. Elevated glucose is easy to induce insulin secretion and activate insulin receptor, which lead to the tyrosine phosphorylation of insulin receptor substrate (IRS), thereby activating PI3K and Akt, and regulating glucose metabolism, including up regulation of glycogen synthesis and down regulation of gluconeogenesis (Long et al., 2020). The activation of Akt also inhibited the phosphorylation of GSK-3β, and then activated GS and glucose transport expression, which led to the increase of glycogen synthesis (Guo et al., 2019). Li, Tao, Yang, Fan, Zhang, & Du et al. (2020) verified that polysaccharides, isolated from P. kingianum, accelerated the glucose metabolism by activating the PI3K/AKT signaling pathway, which dramatically enhanced the expression of insulin receptor substrate-1 (IRS-1), PI3K and AKT, and improved insulin secretion level, so that to regulate glucose and lipid metabolism.
AMPK is an important factor in regulating glucose and lipid metabolism to ameliorate insulin resistance via activating the related factors in this signaling pathway (Alhamami, Uddin, Mahmood, & Briski, 2018). Moreover, AMPK stimulates glucose utilization through phosphorylation of the target involved in glucose transporter transport, thus increasing glucose uptake by cells, inhibiting oxidative stress and improving mitochondrial respiratory function (Hou et al., 2020). A research proved that activated AMPK stimulated lipid oxidation as evidenced by the decreased in FFAs, and promoted the augment of glucose transporter 2(Glut2) and glucose transporter 4 (Glut4) levels, which regulated the lipid and glycolipid metabolism (Long et al., 2020). Although there have a lot of signaling pathways, it is basically related to insulin metabolism, thus affecting downstream factors in signaling pathways and playing an important role in glucose transporters to reinforce glucose and lipid metabolism.
The report pointed out that seaweed polysaccharide had the effect to regulate glucose and lipid metabolism, and show the physiological activities of improving the symptoms of diabetes and lipid metabolism (Zhong et al., 2021). Certainly, researchers confirmed the effect of GLP on glucose and lipid metabolism. Both crude and purified polysaccharide of Gracilaria lemaneiformis inhibited the activity of α - glucosidase, and the purified polysaccharide had the better inhibitory effect. The hypoglycemic activity of GLP may be closely related to the connection way of glyosidic bond, monosaccharide composition, molecular weight and active groups. At the same time, it also improved the level of fasting blood glucose, repaired the damage of pancreas and kidney, and then improved the condition of diabetes in mice (Li et al., 2011, Liao et al., 2015). Sun et al. (2018) revealed that GLP was able to alleviate the disorder of glucose and lipid metabolism in mice, reduce the weight of obese mice, and improve its blood lipid level including Low density lipoprotein (LDL-C), High density lipoprotein (HDL-C), Triglyceride (TG), Cholesterol (TC). These effects could be relevant to the modulation of the gut microbiota. Changes in the gut microbiota may affect the expression of lipid-related genes via short chain fatty acids-dependent pathways. In the meanwhile, it improved the diversity of intestinal microflora in mice, and repaired the intestinal damage caused by the disorder of glucose metabolism. In addition, GLP showed strong antioxidant activity in vitro and in vivo, and scavenged ABTS +, DPPH, hydroxyl and superoxide anion (Di et al., 2017, Wen et al., 2017). Moreover, GLP (with dosage of 50, 150 and 250 mg/kg) was found to raise the activities of superoxide dismutase (SOD), glutathione (GSH) and alcohol dehydrogenase (ADH) in diabetic mice, diminish the contents of malondialdehyde (MDA) and free fatty acid (FFA), raise antioxidant activity and then mitigate its liver loss (Jin et al., 2017, Liao et al., 2015) (Table 2).
In recent years, studies have shown that GLP has remarkable physiological effects to lower blood glucose and blood lipid, alleviate the symptoms of diabetic mice, improve the blood glucose level and body damage of diabetic mice, increase the body's antioxidant capacity, and inhibit lipid peroxidation (Cardoso et al., 2016, Courtois, 2009, Jiang et al., 2014). What's more, the purified GLP showed better effect, probably because the purified polysaccharide had less impurity and smaller molecular weight than the crude polysaccharide, which was easy to be absorbed by the body. On the other hand, there may be different in the content and properties of purified polysaccharide, sulfate group or other functional groups. But so far, there are only a few articles have elucidated the activities of regulating glucose and lipid metabolism from GLP, but the mechanism of glycolipid metabolism is still not well comprehensively expounded. Therefore, it is necessary for us to research the glycolipid mechanism of GLP, and provide theoretical basis for the development of hypoglycemic and hypolipidemic products.
4.2. Immune activity
The immune system consists of immune organs (bone marrow, spleen, lymph nodes, tonsils, small intestinal collecting lymph nodes, thymus, etc.), immune cells (lymphocytes, mononuclear phagocytes, neutrophils, basophils, eosinophils, mast cells, etc.), and immunoactive substances (antibodies, lysozyme, complement, immunoglobulin, interferon, interleukin, tumor necrosis factor, and other cytokines) (Xiao, Mo, & Tu, 2020). It has been reported that the immune system includes nonspecific immunity and specific immunity. Nonspecific immunity (innate immunity) can quickly respond to a variety of invasive pathogenic microorganisms, and play an important role in the initiation and effect of specific immunity (Yakubu, Talba, Chong, Ismail, & Shaari, 2020). However, specific immunity is generally formed after the stimulation of antigen substances, and reacts specifically with the antigen. It mainly involves immune cells such as B lymphocytes and T lymphocytes, and its function is activated immediately after the initial antigen stimulation to regulate the levels of immune active substances (Xing, Luo, Xiao, Tang, & Zhan, 2018). According to the literatures, natural plant polysaccharide has immunomodulatory activity, which can promote the proliferation of immune cells, and improve the body immunity (Isaka et al., 2015, Yanagido et al., 2018). Polysaccharide mainly activates immune response or control immune cells, such as macrophages and lymphocytes, directly participate in antigen presentation and killing of tumor cells. These immunoactive polysaccharides can also regulate the levels of natural killer cells (NK), including tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), interleukin-8 (IL-8), interleukin-1β (IL-1β), interferon-c (IFN-c) and interferon-γ (IFN-γ) (Ren, Zhang, & Zhang, 2021). Furthermore, polysaccharide is able to enhance the activities of macrophages acid phosphatase and phagocyte, activate NK cells, increase the index of thymus and spleen, delay the apoptosis of neutrophils, and regulate the expression of iNOS, IL-6 and TNF-α (Liu et al., 2017).
Animal experiments showed that GLP could advance the proliferation of spleen cell and the phagocytic activity of macrophages in mice, promote the proliferation of interleukin-2 (IL-2) and CD8 + cells, and improve the immunity of mice (Fan et al., 2012). Liu et al. (2020) confirmed that the sulfated oligosaccharide of GLP was capable of inhibiting the production of immunoglobulin E, histamine and related cytokines through the animal and cell experiment. Meanwhile, it also controlled the expression of T helper cells and B cells subsets. It significantly increased proliferation of regulatory T cells (Tregs), optimized the proportion of Firmicutes and Bacteroidetes, and provided a good immune activity. In Table 2, Ren et al. (2017) found that GLP observably improved the proliferation and pinocytosis ability of mouse macrophage RAW264.7 cells, and promoted the gene expression of reactive oxygen species (ROS), nitric oxide (NO), interleukin-6 (IL-6) and tumor necrosis factor (TNF-α) in apoptosis. Its strong immune activity is closely related to the connection way of glycoside bond, sulfate group and low molecular weight. It was reported that it reduced neutrophils migration, regulated the production of interleukin-1β, reduced the peripheral mechanisms of traumatic pain, and slowed down the occurrence of inflammation (Costa et al., 2020). These results suggested that GLP could be used as an effective immunoregulation in functional foods or dietary fiber supplementation. GLP has obvious immune activity by regulating related immune factors in immune system, but its mechanism and pathway is not clear. However, it is certain that the glyosidic bond, molecular composition and active groups of GLP affect its activities, and the mechanism of these factors on its activities can be used as a follow-up study.
4.3. Anti-tumor activity
Tumor is a global health problem and the proliferation of tumor cell is so fast that is hard to cure. Most of the compounds found to be toxic to tumor cells are also toxic to normal cells (Zhang et al., 2021). Therefore, it is of great significance to develop new anti-tumor drugs with low toxicity or no toxicity. Numerous studies reported that polysaccharide extracted from alga (such as Gracilaria lemaneiformis and carrageenin) showed excellent anti-tumor activity and no obvious toxicity. (Khotimchenko et al., 2020). It was found that GLP could not only directly inhibit tumor cells, but also affect different stages of tumor development, and could be used in the prevention and adjuvant treatment of tumors (Han et al., 2020, Moghadamtousi et al., 2014). Polysaccharide produced by Pleurotus ostreatus could up-regulate E-cadherin and down-regulate vimentin in A549 cells (Zhang et al., 2021). Furthermore, polysaccharide is able to activate macrophages to secrete nitric oxide (NO) and tumor necrosis factor alpha (TNF-α), which immediately blocks the growth of tumor cell. The lower concentration presented a good effect of macrophage activation, but the higher concentration of polysaccharide really induced macrophage proliferation rate, which showed a dose dependence (Lopez-Legarda et al., 2020). NO, an important molecule in the signaling pathway of anti-tumor can kill invading pathogens and tumor cells, which is produced through iNOS gene activated by macrophages (Lopez-Legarda et al., 2020). Some researches described the anti-tumor regulation of polysaccharide via activation of the p53 signaling pathway, including p53/Bax/caspase-9/caspase-3/PARP (poly (ADP-ribose) polymerase) and p53/caspase-8/Bcl-2 signaling pathway. As was reported, p53 is an important tumor suppressor that can regulate cell growth and death and activate downstream target factors to inhibit the growth of tumor cells (Wan et al., 2020). Caspase-3, caspase-8, and caspase-9, the key proteases of cell apoptosis, play an important role in the cell inhibition mechanism and have the ability to destroy the tumor cells. Caspase-3 is the most important terminal cleavage enzyme in the process of apoptosis. The upstream factors activate mitochondria to release cytochrome and promote the combination of caspase-9 to form apoptotic bodies that promote the activation of other caspases such as caspase-3, and then induce the apoptosis of tumor cells (Meng, Fraser, Feller, & Ziegler, 2000). As we known, PARP is a modifying enzyme translated by multifunctional protein existing in most eukaryotic cells, which is the cleavage substrate of caspase, and the core member of apoptosis. Moreover, it can be cleaved by a variety of caspases in vitro and is the main cleavage target of caspase 3 in vivo. PARP can be activated by recognizing DNA fragments with structural damage, which plays an important role in DNA damage and the apoptosis of tumor cells (Kieseritzky, 2017). Bax and Bcl-2 play the regulating effect in mitochondria-mediated apoptosis pathway. Bax is a pro-apoptotic protein, which can decompose and grind the outer membrane of mitochondria, causing cells to release chromium C and induce cell apoptosis. However, Bcl-2 is an anti-apoptotic protein, which can block the release of cytochrome C and inhibit the apoptosis process of cell. Therefore, the ratio of Bax/Bcl-2 protein reflects the inhibition rate of apoptosis (Kirkin, Joos, & Zornig, 2004). The expression of genes and proteins of p53, Bax, Bcl-2, caspase-3, caspase-8, and caspase-9 were observably up-regulated or down-regulated after treated by polysaccharide, thus showing a distinct effect on apoptosis, metastasis, and invasion of tumor cells (Wan et al., 2020).
On the other hand, the Fas/FasL pathway is one of the main mechanisms of anti-tumor action and has an important effect in the regulation of cell apoptosis. Fas is a transmembrane protein, which belongs to the tumor necrosis factor receptor superfamily. Fas ligand can bind with FasL, which can initiate apoptosis signal transduction, induce apoptosis, and start lethal signal transduction. The activation of Fas ligand promotes the apoptosis of Fas-dependent cell, which eventually causes a series of characteristic changes in cells and lead to cell death, and enhances the immune activity of cells, thus playing an anti-tumor effect (Svandova, Vesela, Lesot, Poliard, & Matalova, 2016). Specifically, its activation includes a series of steps: first, Fas ligand induces the trimerization of receptor, and then forms the apoptosis of cell membrane, which includes FADD, caspase-8, caspase-9, and caspase-3. Researches proved that GLP had the up-regulation effect on the gene and protein levels of Fas and FasL, and then activates apoptosis pathway induced by Fas/FasL. The morphology, growth and nuclear number of tumor cells, including A549 cells (human non-small cell lung cancer cells), MKN28 cells (human gastric cancer high transfer cells), B16 cells (melanoma cells), SHG-44 (human glioma cells), SMMC-7721 (Human hepatoma cell), and MCF-7 cells (human breast cancer cells), were changed significantly. To be more specific, the proliferation of tumor cells were inhibited due to the reduction of cell size and number, the destruction of organelles, and the rupture of part of nuclear membrane and nucleus (Kang et al., 2017). Shi et al. (2018) studied the antitumor effect of GLP on MCF-7 cells, HepG-2 cells (human liver cancer cells) and HeLa cells (cervical cancer cells), and the results showed that GLP had obvious inhibitory effect on the three tumor cells. As revealed in Table 2, Kang et al. (2016) reported that GLP had the good inhibition effect on HeLa, MKN45 (human gastric cancer cells) and A549 tumor cells, with dose-dependent. GLP presented the better inhibition effect as the augment of content, within a certain range. GLP significantly modulated the expression of apoptosis-related and cell cycle-related genes involved in the death receptor-mediated apoptosis pathway (DR4 and Fas) and the p53 pathway (MDM2, DR4, and BNIP3L). At the same time, the experimental results displayed that GLP had no cytotoxicity on BEAS-2b (human lung cells) and MRC-5 (human lung fibroblasts).
4.4. Anti-inflammatory activity
Inflammation is a kind of defense response of the body to stimulation, divided into infectious (caused by viruses, bacteria, etc.) and non-infectious (trauma) inflammation, which is characterized by redness, swelling, heat, pain and dysfunction. In serious cases, it will cause organ lesions and damage the body, which has close relationship with many diseases, such as diabetes, arthritis, cancer, and so on (Isaka et al., 2015). At present, there are two main types of anti-inflammatory drugs on the market: steroidal and non-steroidal (aspirin, acetaminophen, indomethacin, etc) anti-inflammatory drugs, which may cause the side effect to gastrointestinal or renal or other organ. Nevertheless, polysaccharide has anti-inflammatory activity without side effect. In addition, there are numerous possible mechanisms to inhibit inflammation, including NF-κB and STAT1 signaling pathways. Polysaccharide stimulates the phosphorylation of IkBα, a key inhibitor of NF-κB restraining the translocation of NF-κB p65 subunit to the nucleus, can activate NF-κB and STAT1 (Liu, Ye, Hu, & Wang, 2021). Activated NF-κB can enhance the expression of IL-4, IL-6 and IL-10 (anti-inflammatory cytokine), and activate STAT-1 to reduce the expression of iNOS and IL-1β, TNF-α, IFN-β and IFN-γ (proinflammatory cytokine) (Sun, Huo, Zhong, Zhu, Li, & Li, 2021). Both macrophages/TLRs (mainly TLR2 and TLR4)/(MAPK)/NF-кB and TLR4/Reactive oxygen species (ROS)/PI3K/Akt/MAPK/NF-кB signaling pathways had a vital effect on decreasing the expression of IL-1β and TNF-α and promoting the production of macrophages and TNF-α (Yu, Hu, Wang, Zhang, Xu, & Su, 2018). Studies have reported that gut microbiota is closely related to the inflammation, the reduction of the abundance of pathogenic bacteria (E. coli, Staphylococcus, Clostridium, Verrucomicrobia and Akkermansia, etc.) and the promotion of the dominant flora (such as Bifidobacterium, Lactobacillus and Clostridium tenella), which can amplify the anti-inflammatory activity (Shepherd, DeLoache, Pruss, Whitaker, & Sonnenburg, 2018). You et al. (2021) showed that the abundance ratio of Pseudomonas and Bacteroides (F/B) significantly decreased, and the levels of Verrucomicrobia and Akkermansia reduced after treated with polysaccharide. Polysaccharide promoted to change the gut microbiota and short-chain fatty acids, thus enhancing its anti-inflammatory activity. Furthermore, the inhibition of NO production and expression of iNOS are also belong to the mechanisms of anti-inflammation. NO (produced by iNOS in the activated macrophages) is a gaseous free radical involved in inflammatory processes, which is responsible for numerous severe inflammatory reactions (Yanagido et al., 2018)
Marine algae polysaccharide also has been reported to show anti-inflammatory activity without side effects. Gong and Cheung (2021) manifested that GLP treated by UV-H2O2 had the remarkable anti-inflammatory activity, which inhibited the production of nitric oxide, tumor necrosis factor-α and interleukin-6 by 60.49%, 62.81% and 36.29%, respectively in IEC-6 cells. The degraded polysaccharide with sulfate group showed the better anti-inflammatory activity due to its low molecular weight. It was reported that sulfate polysaccharide from Gracilaria lemaneiformis significantly inhibited the release of pro-inflammatory cytokines and suppressed the secretion of immunoglobulin A via reducing the population of B cells (Liu et al., 2019). The anti-inflammatory activity was related to its molecular and sulfate group. A research proved that GLP could restrain the production of tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6) and interleukin-1β (IL-1β) in colon tissue, which presented the ability to strengthen intestinal barrier, protect colonic microstructure and attenuate DSS-induced colonic injuries (Han et al., 2020, Han et al., 2021) (Table 2). These results demonstrated that GLP had the significant anti-inflammatory activity by inhibiting pro-inflammatory cytokines and enhancing the expression of anti-inflammatory cytokines. The anti-inflammatory activity of GLP may be related to its molecular weight and sulfate group.
4.5. Other activities
In addition to the four physiological effects mentioned above, GLP also has the function of anti-aging, anti-microbial, anti-cancer, lowering blood pressure, weight-loss, prebiotics and other physiological activities. For example, Wang et al.(2019) studied the anti-aging effect of GLP on nematodes by observing their life span, reproduction, pharyngeal pump function and stress response, and the results showed that GLP could increase the average life span of nematodes by more than 16.47%, prolong their reproductive time, and improve their heat resistance and antioxidant ability. Chen et al. (2010) investigated the anti-influenza virus activity of sulfated polysaccharide fractions from Gracilaria lemaneiformis in vitro, and the results exhibited that viral adsorption and replication on host cells were inhibited by sulfated polysaccharide. Sulphated polysaccharide from Gracilaria lemaneiformis had anti-diarrhea activity via Escherichia coli (ETEC)-K88, which alleviated mice diarrhea symptoms (Liu et al., 2019). Moreover, GLP (extracted by H2O2) achieved anti-glioblastoma efficacy via MAPK cascade pathway (Jiang et al., 2014). Furthermore, GLP had the obvious effects on obesity and the gut microbiota in high fat diet-fed mice, which improved serum lipids profile (TC, TG, HDL, and LDL), decreased fat accumulation of organs, and regulated the intestinal microbial abundance (Sun, et al., 2018). It was also found that GLP was not degraded by saliva, gastric juice and intestinal juice, which would be fermented by microbiota and have an important effect on prebiotic activities (Han et al., 2020).
5. Conclusions and prospect
GLP is rich in nutritious and active ingredients, which can create high economic value for society. For the moment, Gracilaria lemaneiformis is mostly used in the extraction of agar and feed for fish, shrimp, abalone and other aquatic animals. It also has great potential in the application of functional food, medicine, and cosmetics, which can be used to produce high-value products.
In this review, a good deal of advanced researches on the extraction, purification, structure and physiological activities of GLP were summarized in the passage. It was known from the existing literatures that there are numerous researches on polysaccharide of Gracilaria lemaneiformis. The extraction and purification methods were relatively mature, and the composition and structural characteristics of polysaccharide were thoroughly discussed. It was confirmed that GLP had physiological activities, such as regulation of glucose and lipid metabolism, immune, anti-tumor, anti-inflammatory activities and other physiological activities.
Nevertheless, the mechanism of the physiological activities of GLP needs to be further elaborated, and the relationship between structure and physiological activities still needs to be further studied, so that to fully explore the potential development of polysaccharide from Gracilaria lemaneiformis, and promote the healthy and sustainable development of the deep processing and high-value utilization industry of Gracilaria lemaneiformis.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
This work was supported by the Key-Area Research and Development Program of Guangdong Province (2020B1111030004), the National Key R&D Program of China (2019YFD0901905), the China Agriculture Research System of MOF and MARA, the Special Scientific Research Funds for Central Non-profit Institutes, Chinese Academy of Fishery Sciences (2020TD69), and the Central Public-interest Scientific Institution Basal Research Funds, South China Sea Fisheries Research Institute, CAFS (2021SD06).
Contributor Information
Xiao Hu, Email: hnhuxiao@163.com.
Xianqing Yang, Email: yxqgd@163.com.
References
- Alhamami H.N., Uddin M.M., Mahmood A.S.M.H., Briski K.P. Lateral but not medial hypothalamic AMPK activation occurs at the hypoglycemic nadir in insulin-injected male rats: impact of caudal dorsomedial hindbrain catecholamine signaling. Neuroscience. 2018;379:103–114. doi: 10.1016/j.neuroscience.2018.03.001. [DOI] [PubMed] [Google Scholar]
- Anand J., Sathuvan M., Babu G.V., Sakthivel M., Palani P., Nagaraj S. Bioactive potential and composition analysis of sulfated polysaccharide from Acanthophora spicifera (Vahl) Borgeson[J] International Journal of Biological Macromolecules. 2018;111:1238–1244. doi: 10.1016/j.ijbiomac.2018.01.057. [DOI] [PubMed] [Google Scholar]
- Beratto-Ramos A., Agurto-Muñoz C., Pablo Vargas-Montalba J., Castillo R.D.P. Fourier-transform infrared imaging and multivariate analysis for direct identification of principal polysaccharides in brown seaweeds [J] Carbohydrate Polymers. 2020;230:115561. doi: 10.1016/j.carbpol.2019.115561. [DOI] [PubMed] [Google Scholar]
- Beri S., Gandhi D. Quantification of residual cetyltrimethylammonium bromide (CTAB) and sodium deoxycholate (DOC) in Haemophilus influenzae type b (Hib) polysaccharide using NMR[J] Biologicals. 2021;70:22–27. doi: 10.1016/j.biologicals.2021.02.001. [DOI] [PubMed] [Google Scholar]
- Cardoso M., Costa R., Mano J. Marine origin polysaccharides in drug delivery systems [J] Marine Drugs. 2016;14(2):34. doi: 10.3390/md14020034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang J., Serrano Y., Garrido R., Rodríguez L.M., Pedroso J., Cardoso F., et al. Relevance of O-acetyl and phosphoglycerol groups for the antigenicity of Streptococcus pneumoniae serotype 18C capsular polysaccharide. Vaccine. 2012;30(49):7090–7096. doi: 10.1016/j.vaccine.2012.09.047. [DOI] [PubMed] [Google Scholar]
- Chattopadhyay K., Ghosh T., Pujol C.A., Carlucci M.J., Damonte E.B., Ray B. Polysaccharides from Gracilaria corticata: Sulfation, chemical characterization and anti-HSV activities[J] International Journal Biological Macromolecules. 2008;43(4):346–351. doi: 10.1016/j.ijbiomac.2008.07.009. [DOI] [PubMed] [Google Scholar]
- Chen H., Cheng J., Zhou S., Chen D., Qin W., Li C., et al. Arabinoxylan combined with different glucans improve lipidmetabolism disorder by regulating bile acid and gutmicrobiota in mice fed with high-fat diet[J] International Journal Biological Macromolecules. 2021;168:279–288. doi: 10.1016/j.ijbiomac.2020.12.036. [DOI] [PubMed] [Google Scholar]
- Chen H., Xiao Q., Weng H., Zhang Y., Yang Q., Xiao A. Extraction of sulfated agar from Gracilaria lemaneiformis using hydrogen peroxide-assisted enzymatic method[J] Carbohydrate Polymers. 2020;232:115790. doi: 10.1016/j.carbpol.2019.115790. [DOI] [PubMed] [Google Scholar]
- Chen M.Z., Liao Z.H., Chen H.L. Effect of sulfate content of Gracilaria lemaneiformis polysaccharide on anti-nfluenza virus activity[J] Food science. 2008;2008(08):587–590. [Google Scholar]
- Chen M.-Z., Xie H.-G., Yang L.-W., Liao Z.-H., Yu J. In vitro anti-influenza virus activities of sulfated polysaccharide fractions from Gracilaria lemaneiformis[J] Virol Sin. 2010;25(5):341–351. doi: 10.1007/s12250-010-3137-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen W., Jia Z., Zhu J., Zou Y., Huang G., Hong Y. Optimization of ultrasonic-assisted enzymatic extraction of polysaccharides from thick-shell mussel (Mytilus coruscus) and their antioxidant activities[J] International Journal of Biological Macromolecules. 2019;140:1116–1125. doi: 10.1016/j.ijbiomac.2019.08.136. [DOI] [PubMed] [Google Scholar]
- Chen Y., Gu X., Huang S.-Q., Li J., Wang X., Tang J. Optimization of ultrasonic/microwave assisted extraction (UMAE) of polysaccharides from Inonotus obliquus and evaluation of its anti-tumor activities[J] International Journal of Biological Macromolecules. 2010;46(4):429–435. doi: 10.1016/j.ijbiomac.2010.02.003. [DOI] [PubMed] [Google Scholar]
- Chen B., Zou D., Du H., Ji Z. Carbon and nitrogen accumulation in the economic seaweed Gracilaria lemaneiformis affected by ocean acidification and increasing temperature [J] Aquaculture. 2018;482:176–182. [Google Scholar]
- Costa L.E.C., Brito T.V., Damasceno R.O.S., Sousa W.M., Barros F.C.N., Sombra V.G., et al. Chemical structure, anti-inflammatory and antinociceptive activities of a sulfated polysaccharide from Gracilaria intermedia algae[J] International Journal of Biological Macromolecules. 2020;159:966–975. doi: 10.1016/j.ijbiomac.2020.05.166. [DOI] [PubMed] [Google Scholar]
- Courtois J. Oligosaccharides from land plants and algae: Production and applications in therapeutics and biotechnology[J] Current Opinion in Microbiology. 2009;12(3):261–273. doi: 10.1016/j.mib.2009.04.007. [DOI] [PubMed] [Google Scholar]
- Di T., Chen G., Sun Y.i., Ou S., Zeng X., Ye H. Antioxidant and immunostimulating activities in vitro of sulfated polysaccharides isolated from Gracilaria rubra[J] Journal of Functional Foods. 2017;28:64–75. [Google Scholar]
- Doumert B., Lecomte F., Tricot G. Advanced solid state 1D/2D NMR investigation of the B2O3-Zn(PO3)2 glasses[J] Journal of Non-Crystalline Solids. 2020;548:120325. doi: 10.1016/j.jnoncrysol.2020.120325. [DOI] [Google Scholar]
- Fan Y., Wang W., Song W., Chen H., Teng A., Liu A. Partial characterization and anti-tumor activity of an acidic polysaccharide from Gracilaria lemaneiformis. Carbohydrate Polymers. 2012;88(4):1313–1318. [Google Scholar]
- Fisheries Administration of Ministry of Agriculture and Rural Affairs. (2021). China Fishery Statistical Yearbook [M]. China Agriculture Press.
- Gagnon P., Leskovec M., Prebil S.D., Žigon R., Štokelj M., Raspor A., et al. Removal of empty capsids from adeno-associated virus preparations by multimodal metal affinity chromatography[J] Journal of Chromatography A. 2021;1649 doi: 10.1016/j.chroma.2021.462210. [DOI] [PubMed] [Google Scholar]
- Gong Y., Ma Y., Cheung P.-K., You L., Liao L., Pedisić S., et al. Structural characteristics and anti-inflammatory activity of UV/H2O2-treated algal sulfated polysaccharide from Gracilaria lemaneiformis. Food and Chemical Toxicology. 2021;152:112157. doi: 10.1016/j.fct.2021.112157. [DOI] [PubMed] [Google Scholar]
- Gremmel C., Frömel T., Knepper T.P. HPLC-MS/MS methods for the determination of 52 perfluoroalkyl and polyfluoroalkyl substances in aqueous samples. Analytical and Bioanalytical Chemistry. 2017;409(6):1643–1655. doi: 10.1007/s00216-016-0110-z. [DOI] [PubMed] [Google Scholar]
- Guo Q., Chen Z., Santhanam R.K., Xu L., Gao X., Ma Q., et al. Hypoglycemic effects of polysaccharides from corn silk (Maydis stigma) and their beneficial roles via regulating the PI3K/Akt signaling pathway in L6 skeletal muscle myotubes. International Journal of Biological Macromolecules. 2019;121:981–988. doi: 10.1016/j.ijbiomac.2018.10.100. [DOI] [PubMed] [Google Scholar]
- Han R., Pang D., Wen L., You L., Huang R., Kulikouskaya V. In vitro digestibility and prebiotic activities of a sulfated polysaccharide from Gracilaria Lemaneiformis[J] Journal of Functional Foods. 2020;64:103652. doi: 10.1016/j.jff.2019.103652. [DOI] [Google Scholar]
- Han R., Wang L., Zhao Z., You L., Pedisić S., Kulikouskaya V., et al. Polysaccharide from Gracilaria Lemaneiformis prevents colitis in Balb/c mice via enhancing intestinal barrier function and attenuating intestinal inflammation[J] Food Hydrocolloids. 2020;109:106048. doi: 10.1016/j.foodhyd.2020.106048. [DOI] [Google Scholar]
- Han R., Ma Y., Xiao J., You L., Pedisić S., Liao L. The possible mechanism of the protective effect of a sulfated polysaccharide from Gracilaria Lemaneiformis against colitis induced by dextran sulfate sodium in mice. Food and Chemical Toxicology. 2021;149:112001. doi: 10.1016/j.fct.2021.112001. [DOI] [PubMed] [Google Scholar]
- Han T., Qi Z., Huang H., Fu G. Biochemical and uptake responses of the macroalga Gracilaria lemaneiformis under urea enrichment conditions[J] Aquatic Botany. 2017;136:197–204. [Google Scholar]
- HOU B.-Y., ZHAO Y.-R., MA P., XU C.-Y., HE P., YANG X.-Y., et al. Hypoglycemic activity of puerarin through modulation of oxidative stress and mitochondrial function via AMPK. Chinese Journal of Natural Medicines. 2020;18(11):818–826. doi: 10.1016/S1875-5364(20)60022-X. [DOI] [PubMed] [Google Scholar]
- Hou N., Zhang M., Xu Y., Sun Z., Wang J., Zhang L., et al. Polysaccharides and their depolymerized fragments from Costaria costata: Molecular weight and sulfation-dependent anticoagulant and FGF/FGFR signal activating activities[J] International Journal of Biological Macromolecules. 2017;105:1511–1518. doi: 10.1016/j.ijbiomac.2017.06.042. [DOI] [PubMed] [Google Scholar]
- Huang S., Pang D., Li X., You L., Zhao Z., Cheung P.-K., et al. A sulfated polysaccharide from Gracilaria Lemaneiformis regulates cholesterol and bile acid metabolism in high-fat diet mice. Food & Function. 2019;10(6):3224–3236. doi: 10.1039/c9fo00263d. [DOI] [PubMed] [Google Scholar]
- Huang R., Xie J., Liu X., Shen M. Sulfated modification enhances the modulatory effect of yam polysaccharide on gut microbiota in cyclophosphamide-treated mice. Food Research International. 2021;145:110393. doi: 10.1016/j.foodres.2021.110393. [DOI] [PubMed] [Google Scholar]
- Ibrahim S.M., Althagafi I., Takagi H.D., Hassan R.M. Kinetics and mechanism of oxidation of chondroitin-4-sulfate polysaccharide as a sulfated polysaccharide by hexacyanoferrate(III) in alkaline solutions with synthesis of novel coordination biopolymer chelating agent[J] Journal of Molecular Liquids. 2017;244:353–359. [Google Scholar]
- Isaka S., Cho K., Nakazono S., Abu R., Ueno M., Kim D., et al. Antioxidant and anti-inflammatory activities of porphyran isolated from discolored nori (Porphyra yezoensis)[J] International Journal of Biological Macromolecules. 2015;74:68–75. doi: 10.1016/j.ijbiomac.2014.11.043. [DOI] [PubMed] [Google Scholar]
- Jiang W., Fu Y., Yang F., Yang Y., Liu T., Zheng W., et al. Gracilaria lemaneiformis Polysaccharide as Integrin-Targeting Surface Decorator of Selenium Nanoparticles to Achieve Enhanced Anticancer Efficacy[J] ACS Applied Materials & Interfaces. 2014;6(16):13738–13748. doi: 10.1021/am5031962. [DOI] [PubMed] [Google Scholar]
- Jin M., Liu H., Hou Y., Chan Z., Di W., Li L.i., et al. Preparation, characterization and alcoholic liver injury protective effects of algal oligosaccharides from Gracilaria lemaneiformis[J] Food Research International. 2017;100:186–195. doi: 10.1016/j.foodres.2017.08.032. [DOI] [PubMed] [Google Scholar]
- Jung J.K., Kim I.G., Chung K.S., Baek U.B. Analyses of permeation characteristics of hydrogen in nitrile butadiene rubber using gas chromatography[J] Materials Chemistry and Physics. 2021;267:124653. doi: 10.1016/j.matchemphys.2021.124653. [DOI] [Google Scholar]
- Kang, Y. Li, H., Wu, J., Xu, X. T., Sun, X., Zhao, X. D., et al. (2016). Transcriptome Profiling Reveals the Antitumor Mechanism of Polysaccharide from Marine Algae Gracilariopsis lemaneiformis [J]. PLoS One, 11(6), e0158279. [DOI] [PMC free article] [PubMed]
- Kang Y., Wang Z.-J., Xie D., Sun X., Yang W., Zhao X., et al. Characterization and Potential Antitumor Activity of Polysaccharide from Gracilariopsis lemaneiformis [J] Marine Drugs. 2017;15(4):100. doi: 10.3390/md15040100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlsson F.H., Tremaroli V., Nookaew I., Bergstrom G., Behre C.J., Fagerberg B., et al. Gut metagenome in European women with normal, impaired and diabetic glucose control[J] Nature. 2013;498(7452):99–103. doi: 10.1038/nature12198. [DOI] [PubMed] [Google Scholar]
- Khan B.M., Qiu H.-M., Wang X.-F., Liu Z.-Y., Zhang J.-Y., Guo Y.-J., et al. Physicochemical characterization of Gracilaria chouae sulfated polysaccharides and their antioxidant potential[J] International Journal of Biological Macromolecules. 2019;134:255–261. doi: 10.1016/j.ijbiomac.2019.05.055. [DOI] [PubMed] [Google Scholar]
- Khotimchenko M., Tiasto V., Kalitnik A., Begun M., Khotimchenko R., Leonteva E., et al. Antitumor potential of carrageenans from marine red algae. Carbohydrate Polymers. 2020;246:116568. doi: 10.1016/j.carbpol.2020.116568. [DOI] [PubMed] [Google Scholar]
- Kieseritzky, K. V. (2017). PARP-1/2-Inhibitor beim rezidivierten platinsensitiven Ovarialkarzinom. Im Focus Onkologie, 20(4), 32–32. doi:10.1007/s15015-017-3240-z.
- Kirkin V., Joos S., Zörnig M. The role of Bcl-2 family members in tumorigenesis[J] Biochimica et Biophysica Acta. 2004;1644(2-3):229–249. doi: 10.1016/j.bbamcr.2003.08.009. [DOI] [PubMed] [Google Scholar]
- Lee M.-H., Chao C.-H., Hsu Y.-C., Lu M.-K. Production, characterization, and functions of sulfated polysaccharides from zinc sulfate enriched cultivation of Antrodia cinnamomea [J] International Journal of Biological Macromolecules. 2020;159:1013–1021. doi: 10.1016/j.ijbiomac.2020.05.068. [DOI] [PubMed] [Google Scholar]
- Li F., Zhang Y., Zhong Z. Antihyperglycemic Effect of Ganoderma Lucidum Polysaccharides on Streptozotocin-Induced Diabetic Mice[J] International Journal of Molecular Sciences. 2011;12(9):6135–6145. doi: 10.3390/ijms12096135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H., Yu X., Jin Y., Zhang W., Liu Y. Development of an eco-friendly agar extraction technique from the red seaweed Gracilaria lemaneiformis[J] Bioresource Technology. 2008;99(8):3301–3305. doi: 10.1016/j.biortech.2007.07.002. [DOI] [PubMed] [Google Scholar]
- Li R., Tao A., Yang R., Fan M., Zhang X., Du Z., et al. Structural characterization, hypoglycemic effects and antidiabetic mechanism of a novel polysaccharides from Polygonatum kingianum Coll. et Hemsl. Biomedicine & Pharmacotherapy. 2020;131:110687. doi: 10.1016/j.biopha.2020.110687. [DOI] [PubMed] [Google Scholar]
- Li X., Huang S., Chen X.i., Xu Q., Ma Y., You L., et al. Structural characteristic of a sulfated polysaccharide from Gracilaria Lemaneiformis and its lipid metabolism regulation effect[J] Food & Function. 2020;11(12):10876–10885. doi: 10.1039/d0fo02575e. [DOI] [PubMed] [Google Scholar]
- Li Y.T., Guo X., Tian Y.H., Zhang T.L., Luo Z.W., Liu X.J., et al. Methylation in combination with temperature programming enables rapid identification of polysaccharides by ambient micro-fabrication glow discharge plasma (MFGDP) desorption ionization mass spectrometry [J] Talanta. 2020;218 doi: 10.1016/j.talanta.2020.121156. [DOI] [PubMed] [Google Scholar]
- Liao S., Long X., Zou Y., Liu F., Li Q. Mulberry leaf phenolics and fiber exert anti-obesity through the gut microbiota-host metabolism pathway. Journal of Food Science. 2021;86(4):1432–1447. doi: 10.1111/1750-3841.15679. [DOI] [PubMed] [Google Scholar]
- Liao X., Yang L., Chen M., Yu J., Zhang S., Ju Y. The hypoglycemic effect of a polysaccharide (GLP) from Gracilaria lemaneiformis and its degradation products in diabetic mice[J] Food & Function. 2015;6(8):2542–2549. doi: 10.1039/c4fo01185f. [DOI] [PubMed] [Google Scholar]
- Liu B.o., Liu Q.-M., Li G.-L., Sun L.-C., Gao Y.-Y., Zhang Y.-F., et al. The anti-diarrhea activity of red algae-originated sulphated polysaccharides on ETEC-K88 infected mice [J] RSC Advances. 2019;9(5):2360–2370. doi: 10.1039/c8ra09247h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Q.-M., Xu S.-S., Li L., Pan T.-M., Shi C.-L., Liu H., et al. In vitro and in vivo immunomodulatory activity of sulfated polysaccharide from Porphyra haitanensis[J] Carbohydrate Polymers. 2017;165:189–196. doi: 10.1016/j.carbpol.2017.02.032. [DOI] [PubMed] [Google Scholar]
- Liu Q.-M., Yang Y., Maleki S.J., Alcocer M., Xu S.-S., Shi C.-L., et al. Anti-Food Allergic Activity of Sulfated Polysaccharide from Gracilaria lemaneiformis is Dependent on Immunosuppression and Inhibition of p38 MAPK[J] Journal of Agriculture and Food Chemistry. 2016;64(22):4536–4544. doi: 10.1021/acs.jafc.6b01086. [DOI] [PubMed] [Google Scholar]
- Liu Q., Zhang Y., Shu Z., Liu M., Zeng R., Wang Y., et al. Sulfated oligosaccharide of Gracilaria lemaneiformis protect against food allergic response in mice by up-regulating immunosuppression[J] Carbohydrate Polymers. 2020;230:115567. doi: 10.1016/j.carbpol.2019.115567. [DOI] [PubMed] [Google Scholar]
- Liu, Y., YE, Y., Hu, X., Wang, J. H. (2021). Structural characterization and anti-inflammatory activity of a polysaccharide from the lignified okra[J]. Carbohydr Polym, 265, 118081. [DOI] [PubMed]
- Loa C.C., Lin T.L., Wu C.C., Bryan T.A., Thacker H.L., Hooper T., et al. Purification of turkey coronavirus by Sephacryl size-exclusion chromatography[J] Journal of Virological Methods. 2002;104(2):187–194. doi: 10.1016/S0166-0934(02)00069-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Long X.-S., Liao S.-T., Wen P., Zou Y.-X., Liu F., Shen W.-Z., et al. Superior hypoglycemic activity of mulberry lacking monosaccharides is accompanied by better activation of the PI3K/Akt and AMPK signaling pathways. Food & Function. 2020;11(5):4249–4258. doi: 10.1039/d0fo00427h. [DOI] [PubMed] [Google Scholar]
- Lopez-Legarda X., Arboleda-Echavarría C., Parra-Saldívar R., Rostro-Alanis M., Alzate J.F., Villa-Pulgarín J.A., et al. Biotechnological production, characterization and in vitro antitumor activity of polysaccharides from a native strain of Lentinus crinitus[J] International Journal of Biological Macromolecules. 2020;164:3133–3144. doi: 10.1016/j.ijbiomac.2020.08.191. [DOI] [PubMed] [Google Scholar]
- Martineau E., El Khantache K., Pupier M., Sepulcri P., Akoka S., Giraudeau P. Non-linear effects in quantitative 2D NMR of polysaccharides: Pitfalls and how to avoid them[J] Journal of Pharmaceutical and Biomedical Analysis. 2015;108:78–85. doi: 10.1016/j.jpba.2015.01.056. [DOI] [PubMed] [Google Scholar]
- Mazumder S., Lerouge P., Loutelierbourhis C., Driouich A., Ray B. Structural characterisation of hemicellulosic polysaccharides from using specific enzyme hydrolysis, ion exchange chromatography and MALDI-TOF mass spectroscopy[J] Carbohydrate Polymers. 2005;59(2):231–238. [Google Scholar]
- Meng X.W., Fraser M.J., Feller J.M., Ziegler J.B. Caspase-3-dependent and caspase-3-independent pathways leading to chromatin DNA fragmentation in HL-60 cells[J] Apoptosis An International Journal on Programmed Cell Death. 2000;5(1):61. doi: 10.1023/a:1009689710184. [DOI] [PubMed] [Google Scholar]
- Moghadamtousi S.Z., Karimian H., Khanabdali R., Razavi M., Firoozinia M., Zandi K., et al. Anticancer and Antitumor Potential of Fucoidan and Fucoxanthin, Two Main Metabolites Isolated from Brown Algae[J] The Scientific World Journal. 2014;2014:1–10. doi: 10.1155/2014/768323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pengzhan Y. Antihyperlipidemic effects of different molecular weight sulfated polysaccharides from Ulva pertus (Chlorophyta)[J] Pharmacological Research. 2003;48(6):543–549. doi: 10.1016/s1043-6618(03)00215-9. [DOI] [PubMed] [Google Scholar]
- Porse H., Rudolph B. The seaweed hydrocolloid industry: 2016 updates, requirements, and outlook [J] Journal of Applied Phycology. 2017;29(5):2187–2200. [Google Scholar]
- Ren L., Zhang J., Zhang T. Immunomodulatory activities of polysaccharides from Ganoderma on immune effector cells[J] Food Chemistry. 2021;340:127933. doi: 10.1016/j.foodchem.2020.127933. [DOI] [PubMed] [Google Scholar]
- Ren Y., Zheng G., You L., Wen L., Li C., Fu X., et al. Structural characterization and macrophage immunomodulatory activity of a polysaccharide isolated from Gracilaria lemaneiformis[J] Journal of Functional Foods. 2017;33:286–296. [Google Scholar]
- Roslund M.U., Säwén E., Landström J., Rönnols J., Jonsson K.H.M., Lundborg M., et al. Complete (1)H and (13)C NMR chemical shift assignments of mono-, di-, and trisaccharides as basis for NMR chemical shift predictions of polysaccharides using the computer program casper[J] Carbohydrate Research. 2011;346(11):1311–1319. doi: 10.1016/j.carres.2011.04.033. [DOI] [PubMed] [Google Scholar]
- Shang X.C., Chu D., Zhang J.X., Zheng Y.F., Li Y. Microwave-assisted extraction, partial purification and biological activity in vitro of polysaccharides from bladder-wrack (Fucus vesiculosus) by using deep eutectic solvents [J] Separation and Purification Technology. 2021;259:118169. doi: 10.1016/j.seppur.2020.118169. [DOI] [Google Scholar]
- Shepherd E.S., DeLoache W.C., Pruss K.M., Whitaker W.R., Sonnenburg J.L. An exclusive metabolic niche enables strain engraftment in the gut microbiota[J] Nature. 2018;557(7705):434–438. doi: 10.1038/s41586-018-0092-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi F., Yan X., Cheong K.-L., Liu Y. Extraction, purification, and characterization of polysaccharides from marine algae Gracilaria lemaneiformis with anti-tumor activity[J] Process Biochemistry. 2018;73:197–203. [Google Scholar]
- Shi H., Sun J., Han R., Ding C., Hu F., Yu S. The strategy for correcting interference from water in Fourier transform infrared spectrum based bacterial typing - ScienceDirect[J] Talanta. 2020;208:120347. doi: 10.1016/j.talanta.2019.120347. [DOI] [PubMed] [Google Scholar]
- Soriano E.M., Bourret E. Effects of season on the yield and quality of agar from Gracilaria species (Gracilariaceae, Rhodophyta)[J] Bioresource Technology. 2003;90(3):329–333. doi: 10.1016/s0960-8524(03)00112-3. [DOI] [PubMed] [Google Scholar]
- Sousa A.M.M., Alves V.D., Morais S., Delerue-Matos C., Gonçalves M.P. Agar extraction from integrated multitrophic aquacultured Gracilaria vermiculophylla: Evaluation of a microwave-assisted process using response surface methodology[J] Bioresource Technology. 2010;101(9):3258–3267. doi: 10.1016/j.biortech.2009.12.061. [DOI] [PubMed] [Google Scholar]
- Strickley R.G., Brandl M., Chan K.W., Straub K., Gu L. High-Performance Liquid Chromatographic (HPLC) and HPLC-Mass Spectrometric (MS) Analysis of the Degradation of the Luteinizing Hormone-Releasing Hormone (LH-RH) Antagonist RS-26306 in Aqueous Solution[J] Pharmaceutical Research. 1990;7(5):530–536. doi: 10.1023/a:1015829119270. [DOI] [PubMed] [Google Scholar]
- Sun X., Duan M., Liu Y., Luo T., Ma N., Song S., et al. The beneficial effects of Gracilaria lemaneiformis polysaccharides on obesity and the gut microbiota in high fat diet-fed mice[J] Journal of Functional Foods. 2018;46:48–56. [Google Scholar]
- Sun Y., Huo J., Zhong S., Zhu J., Li Y., Li X. Chemical structure and anti-inflammatory activity of a branched polysaccharide isolated from Phellinus baumii[J] Carbohydrate Polymers. 2021;268:118214. doi: 10.1016/j.carbpol.2021.118214. [DOI] [PubMed] [Google Scholar]
- Svandova E.B., Vesela B., Lesot H., Poliard A., Matalova E. Expression of Fas, FasL, caspase-8 and other factors of the extrinsic apoptotic pathway during the onset of interdigital tissue elimination [J] Histochemistry and Cell Biology. 2016;147(4):497–510. doi: 10.1007/s00418-016-1508-6. [DOI] [PubMed] [Google Scholar]
- Teng C., Qin P., Shi Z., Zhang W., Yang X., Yao Y., et al. Structural characterization and antioxidant activity of alkali-extracted polysaccharides from quinoa [J] Food Hydrocolloids. 2020;113:106392. doi: 10.1016/j.foodhyd.2020.106392. [DOI] [Google Scholar]
- Veeraperumal S., Qiu H.-M., Zeng S.-S., Yao W.-Z., Wang B.-P., Liu Y., et al. Polysaccharides from Gracilaria lemaneiformis promote the HaCaT keratinocytes wound healing by polarised and directional cell migration[J] Carbohydrate Polymers. 2020;241:116310. doi: 10.1016/j.carbpol.2020.116310. [DOI] [PubMed] [Google Scholar]
- Wan X., Jin X., Xie M., Liu J., Gontcharov A.A., Wang H., et al. Characterization of a polysaccharide from Sanghuangporus vaninii and its antitumor regulation via activation of the p53 signaling pathway in breast cancer MCF-7 cells[J] International Journal of Biological Macromolecules. 2020;163:865–877. doi: 10.1016/j.ijbiomac.2020.06.279. [DOI] [PubMed] [Google Scholar]
- Wang W., Xu F., Jin L., Ding C.-F. Rapid identification of illegal drugs and explosives using resonance excitation in miniaturized photoionization ion trap mass spectrometry[J] International Journal of Mass Spectrometry. 2021;467:116625. doi: 10.1016/j.ijms.2021.116625. [DOI] [Google Scholar]
- Wang X., Zhang Z., Zhou H., Sun X., Chen X., Xu N. The anti-aging effects of Gracilaria lemaneiformis polysaccharide in Caenorhabditis elegans[J] International Journal of Biological Macromolecules. 2019;140:600–604. doi: 10.1016/j.ijbiomac.2019.08.186. [DOI] [PubMed] [Google Scholar]
- Wang Z., Wang X., Ke C. Bioaccumulation of trace metals by the live macroalga Gracilaria lemaneiformis[J] Journal of Applied Phycology. 2013;26(4):1889–1897. [Google Scholar]
- Wei Z., You J., Wu H., Yang F., Long L., Liu Q., et al. Bioremediation using Gracilaria lemaneiformis to manage the nitrogen and phosphorous balance in an integrated multi-trophic aquaculture system in Yantian Bay, China [J] Marine Pollution Bulletin. 2017;121(1-2):313–319. doi: 10.1016/j.marpolbul.2017.04.034. [DOI] [PubMed] [Google Scholar]
- Wen X., Peng C., Zhou H., Lin Z., Lin G., Chen S., et al. Nutritional Composition and Assessment of Gracilaria lemaneiformis Bory[J] Journal of Integrative Plant Biology. 2006;48(9):1047–1053. [Google Scholar]
- Wen L., Zhang Y., Sun-Waterhouse D., You L., Fu X. Advantages of the polysaccharides from Gracilaria lemaneiformis over metformin in antidiabetic effects on streptozotocin-induced diabetic mice. RSC Advances. 2017;7(15):9141–9151. [Google Scholar]
- Wu H., Jiang H., Liu C., Deng Y. Growth, pigment composition, chlorophyll fluorescence and antioxidant defenses in the red alga Gracilaria lemaneiformis (Gracilariales, Rhodophyta) under light stress[J] South African Journal of Botany. 2015;100:27–32. [Google Scholar]
- Wu, S. J., Lu, M. S., & Wang, S. J. (2017). Amylase-assisted extraction and antioxidant activity of polysaccharides from Gracilaria lemaneiformis [J]. 3 Biotech, 7(1). [DOI] [PMC free article] [PubMed]
- Xiao X., Mo H., Tu K. CTNNB1 mutation suppresses infiltration of immune cells in hepatocellular carcinoma through miRNA-mediated regulation of chemokine expression [J] International Immunopharmacology. 2020;89:107043. doi: 10.1016/j.intimp.2020.107043. [DOI] [PubMed] [Google Scholar]
- Xing J., Luo K.K., Xiao Y., Tang X.Q., Zhan W.B. Influence of CD4-1+, CD4-2+ and CD8+ T lymphocytes subpopulations on the immune response of B lymphocytes in flounder (Paralichthys olivaceus) immunized with thymus-dependent or thymus-independent antigen[J] Fish & Shellfish Immunology. 2018:979–986. doi: 10.1016/j.fsi.2018.11.004. [DOI] [PubMed] [Google Scholar]
- Xu J., Gao K. UV-A enhanced growth and UV-B induced positive effects in the recovery of photochemical yield in Gracilaria lemaneiformis (Rhodophyta)[J] Journal of Photochemistry and Photobiology B. 2010;100(3):117–122. doi: 10.1016/j.jphotobiol.2010.05.010. [DOI] [PubMed] [Google Scholar]
- Xu L.L., Li W.W., Chen Z.Q., Guo Q.W., Wang C.L., Santhanam R.K., et al. Inhibitory effect of epigallocatechin-3-O-gallate on alpha-glucosidase and its hypoglycemic effect via targeting PI3K/AKT signaling pathway in L6 skeletal muscle cells. International Journal of Biological Macromolecules. 2019;125:605–611. doi: 10.1016/j.ijbiomac.2018.12.064. [DOI] [PubMed] [Google Scholar]
- Xu X.-Q., Su B.-M., Xie J.-S., Li R.-K., Yang J., Lin J., et al. Preparation of bioactive neoagaroligosaccharides through hydrolysis of Gracilaria lemaneiformis agar: A comparative study [J] Food Chemistry. 2018;240:330–337. doi: 10.1016/j.foodchem.2017.07.036. [DOI] [PubMed] [Google Scholar]
- Xu R., Ye H., Sun Y., Tu Y., Zeng X. Preparation, preliminary characterization, antioxidant, hepatoprotective and antitumor activities of polysaccharides from the flower of tea plant (Camellia sinensis) Food and Chemical Toxicology, Toxicology. 2012;50(7):2473–2480. doi: 10.1016/j.fct.2011.10.047. [DOI] [PubMed] [Google Scholar]
- Yakubu Y., Talba A.M., Chong C.M., Ismail I.S., Shaari K. Effect of Terminalia catappa methanol leaf extract on nonspecific innate immune responses and disease resistance of red hybrid tilapia against Streptococcus agalactiae [J] Aquaculture Reports. 2020;18:100555. doi: 10.1016/j.aqrep.2020.100555. [DOI] [Google Scholar]
- Yanagido A., Ueno M., Jiang Z., Cho K., Yamaguchi K., Kim D., et al. Increase in anti-inflammatory activities of radical-degraded porphyrans isolated from discolored nori (Pyropia yezoensis)[J] International Journal of Biological Macromolecules. 2018;117:78–86. doi: 10.1016/j.ijbiomac.2018.05.146. [DOI] [PubMed] [Google Scholar]
- Yang J., Shen M.Y., Luo Y., Wu T., Wen H.L., Xie J.H. Construction and characterization of Mesona chinensis polysaccharide-chitosan hydrogels, role of chitosan deacetylation degree [J] Carbohydrate Polymers. 2021;257 doi: 10.1016/j.carbpol.2020.117608. [DOI] [PubMed] [Google Scholar]
- You L.J., Zhang Y.L., Wen L.R., Liu R.H., Fu X. Effects of different extraction methods on the properties of Gracilaria lemaneiformis polysaccharide[J] Modern Food Science and Technology. 2016;32(6):148–155. [Google Scholar]
- You S., Liu X., Xu G., Ye M., Bai L., Lin R., et al. Identification of bioactive polysaccharide from Pseudostellaria heterophylla with its anti-inflammatory effects[J] Journal of Functional Foods. 2021;78:104353. doi: 10.1016/j.jff.2021.104353. [DOI] [Google Scholar]
- Youssouf L., Lallemand L., Giraud P., Soulé F., Bhaw-Luximon A., Meilhac O., et al. Ultrasound-assisted extraction and structural characterization by NMR of alginates and carrageenans from seaweeds[J] Carbohydrate Polymers. 2017;166:55–63. doi: 10.1016/j.carbpol.2017.01.041. [DOI] [PubMed] [Google Scholar]
- Yu J., Yang Y.-F. Physiological and biochemical response of seaweed Gracilaria lemaneiformis to concentration changes of N and P[J] Journal of Experimental Marine Biology and Ecology. 2008;367(2):142–148. [Google Scholar]
- Yu J., Hu M., Wang Y., Zhang Q., Xu W., Su W. Extraction, partial characterization and bioactivity of polysaccharides from Senecio scandens Buch.-Ham[J] International Journal of Biological Macromolecules. 2018;109:535–543. doi: 10.1016/j.ijbiomac.2017.12.119. [DOI] [PubMed] [Google Scholar]
- Yu Y.-Y., Chen W.-D., Liu Y.-J., Niu J., Chen M., Tian L.-X. Effect of different dietary levels of Gracilaria lemaneiformis dry power on growth performance, hematological parameters and intestinal structure of juvenile Pacific white shrimp (Litopenaeus vannamei)[J] Aquaculture. 2016;450:356–362. [Google Scholar]
- Yuan Q.X., Zhang J., Xiao C.L., Harqin C.L., Ma M.Y., Long T., et al. Structural characterization of a low-molecular-weight polysaccharide from Angelica pubescens Maxim. f. biserrata Shan et Yuan root and evaluation of its antioxidant activity [J] Carbohydrate Polymers. 2020;236 doi: 10.1016/j.carbpol.2020.116047. [DOI] [PubMed] [Google Scholar]
- Yun J., Cui C., Zhang S., Zhu J., Peng C., Cai H., et al. Use of headspace GC/MS combined with chemometric analysis to identify the geographic origins of black tea[J] Food Chemistry. 2021;360:130033. doi: 10.1016/j.foodchem.2021.130033. [DOI] [PubMed] [Google Scholar]
- Zhang J., Wen C., Chen M., Gu J., Zhou J., Duan Y., et al. Antioxidant activities of Sagittaria sagittifolia L. polysaccharides with subcritical water extraction [J] International Journal of Biological Macromolecules. 2019;134:172–179. doi: 10.1016/j.ijbiomac.2019.05.047. [DOI] [PubMed] [Google Scholar]
- Zhang X., Aweya J.J., Huang Z.-X., Kang Z.-Y., Bai Z.-H., Li K.-H., et al. In vitro fermentation of Gracilaria lemaneiformis sulfated polysaccharides and its agaro-oligosaccharides by human fecal inocula and its impact on microbiota[J] Carbohydrate Polymers. 2020;234:115894. doi: 10.1016/j.carbpol.2020.115894. [DOI] [PubMed] [Google Scholar]
- Zhang Y.-H., Song X.-N., Lin Y., Xiao Q., Du X.-P., Chen Y.-H., et al. Antioxidant capacity and prebiotic effects of Gracilaria neoagaro oligosaccharides prepared by agarase hydrolysis [J] International Journal of Biological Macromolecules. 2019;137:177–186. doi: 10.1016/j.ijbiomac.2019.06.207. [DOI] [PubMed] [Google Scholar]
- Zhang Z., Zhang Y., Liu H., Wang J., Wang D., Deng Z., et al. A water-soluble selenium-enriched polysaccharide produced by Pleurotus ostreatus: Purification, characterization, antioxidant and antitumor activities in vitro[J] International Journal of Biological Macromolecules. 2021;168:356–370. doi: 10.1016/j.ijbiomac.2020.12.070. [DOI] [PubMed] [Google Scholar]
- Zhao Y., Ouyang X., Chen J., Zhao L., Qiu X. Separation of aromatic monomers from oxidatively depolymerized products of lignin by combining Sephadex and silica gel column chromatography[J] Separation and Purification Technology. 2018;191:250–256. [Google Scholar]
- Zhao Y., Yan Y., Zhou W., Chen D., Huang K., Yu S., et al. Effects of polysaccharides from bee collected pollen of Chinese wolfberry on immune response and gut microbiota composition in cyclophosphamide-treated mice. Journal of Functional Foods. 2020;72:104057. doi: 10.1016/j.jff.2020.104057. [DOI] [Google Scholar]
- Zhong Q.-W., Zhou T.-S., Qiu W.-H., Wang Y.-K., Xu Q.-L., Ke S.-Z., et al. Characterization and hypoglycemic effects of sulfated polysaccharides derived from brown seaweed Undaria pinnatifida [J] Food Chemistry. 2021;341:128148. doi: 10.1016/j.foodchem.2020.128148. [DOI] [PubMed] [Google Scholar]
- Zhou Z.M., He Q., Ma X.Y. Analysis and evaluation of nutritional components in Gracilaria lemaneiformis[J] food science. 2010;31(9):284–287. [Google Scholar]
- Zhu Z., Zhang Y., Wang J., Li X., Wang W., Huang Z. Characterization of sugar composition in Chinese royal jelly by ion chromatography with pulsed amperometric detection. Journal of Food Composition and Analysis. 2019;78:101–107. doi: 10.1016/j.jfca.2019.01.003. [DOI] [Google Scholar]