Abstract
COVID-19 is a pandemic requiring immediate solution for treatment because of its complex pathophysiology. Exploration of novel targets and thus treatment will be life savers which is the need of the hour. 2 host factors- TMPRSS2 and ACE2 are responsible for the way the virus will enter and replicate in the host. Also NRF2 is an important protein responsible for its anti-inflammatory role by multiple mechanisms of action like inhibition of NF-kB, suppression of pro-inflammatory genes, etc. NRF2 is deacetylated by Sirtuins and therefore both have a direct association. Absence of SIRT indicates inhibition of NRF2 expression and thus no anti-oxidative and anti-inflammatory protection for the cell. Therefore, we propose that NRF2 activators and/or SIRT activators can be evaluated to check their efficacy in ameliorating the symptoms of COVID-19.
Keywords: COVID-19, NRF2, SIRT, ACE2, TMPRSS2, SIRT activators
1. Introduction
Corona Virus disease 19 (COVID-19) has become one of the most terrifying pandemic in the last couple of years, prevalence of which varies from country to country, of which around 195 countries have already been affected by it. Risk of death due to COVID-19 varies from country to country, as 1% in Germany to 11% in Italy. Studies have also shown that the mortality is high for subjects aged 60 and above [1].
According to many evidences it is suggested that a transmembrane protease which is encoded by a gene named TMPRSS2 plays a major role in the entry for Severe Acute Respiratory Syndrome Coronavirus, this is the same protease responsible for entry of Middle East Respiratory Syndrome Coronavirus and influenza Virus [2]. This leads to an idea that targeting this TMPRSS2 can be a novel target for Coronavirus [3]. Hoffman and coworkers found out that the virus responsible for SARS-CoV-2, i.e. Covid-19 depends upon two major host cell factors mainly: ACE2 and TMPRSS2. ACE2 is a carboxypeptidase that converts Angiotensin 1 to angiotensin [[1], [2], [3], [4], [5], [6], [7], [8], [9]] and Angiotensin 2 to angiotensin [[1], [2], [3], [4], [5], [6], [7]]. ACE2 has been majorly found in lung tissues. Coronavirus has Spike (S) Proteins on its cellular membrane, which helps viral entry into target host cells. This entry depends on attachment of S Proteins on the cellular receptor, ACE2, which then helps viral attachment to the surface of target host cells. Recent studies have shown that the novel coronavirus targets the ACE2 via Spike Proteins(S) in the receptor binding domain (RBD) through Van der Waals forces and study shows that novel coronavirus-2019 RBD binds strongly than SARS-CoV because novel coronavirus has glutamine residue at 479 to bind with lysine 31 on ACE2 Receptor on host cellular surface while SARS-CoV lack the glutamate residue. After this, for the entry of virus into host target cells, S protein must undergo “Priming” by the cellular protease TMPRSS2, which performs the S protein cleavage and thus leads to fusion of viral and cellular membranes. This facilitates entry of virus into host target cells. It is seen that the viral entry is increased 2.6 fold in the presence of TMPRSS2 and decreased by five-fold in absence of TMPRSS2. From various sources it is proved that ACE2 expression in the lung tissues of White and African-American(Black) is much lower than that of Asian people [4]. As soon as the virus enters into the cells of the host, it initiates the host defense mechanisms. One of the mechanism involved is Retinoic acid inducible Gene 1 (RIG-1) like Receptors (RLRs) which recognizes the virus and then binds to it, upon binding it undergoes ubiquitination by the Ubiquitin E3 ligases, leading to activation of NF-kB via phosphorylation. This leads to production of IFN-β, having anti-viral activity [5]. The novel coronavirus-2019 inhibits the RLRs ubiquitination by E3 ligases and thus inhibition of IFN-β [6,7]. ACE2 protein was downregulated in novel coronavirus-2019 and SARS-CoV which leads to lung injury, because ACE2 has a protective role in lung injury [8]. ACE2 downregulation also causes an increase in NF-kB phosphorylation and thus its activation. Due to activation of NF-kB, many other pro-inflammatory cytokines are secreted in large quantities, mainly IL-6 and IL-8 which in turn stimulate migration of many other cytokines and immune cells at the site of infection [9]. Angiotensin 2 has pro-inflammatory properties via phosphorylation and activation of NF-kB, while ACE2 has anti-inflammatory effects by degrading Angiotensin 2 to Angiotensin [[1], [2], [3], [4], [5], [6], [7],10]. Major mechanism leading to inflammation and tissue injury upon virus entry is due to generation of free radicals [11]. Upon entry of virus into the host cells, oxidative stress is induced to facilitate its replication [12]. It is also known that, Angiotensin 2 when binds to AT1R Receptors located on the cell surface, it leads to induction of NADPH Oxidase, which is responsible for excessive generation of ROS and thus causing oxidative stress [13], which in case of coronavirus exposure is increased due to depletion of ACE2 and increase in Angiotensin 2 levels. Studies have shown that increase in ACE2 levels may also increase the competition of coronavirus for binding and thus decreasing its entry into the host cell [14]. It was shown in a study that ACE2 on injection in a soluble form slows down the entering of virus into host cells [15]. Also, Zhang R et al. proved that ACE2 protects the lung from injury, as ACE2 levels are highest in lungs [16].
2. Inflammation involved in COVID-19 pathology
Clinical observations of various coronavirus outbreaks such as Severe acute respiratory syndrome coronavirus(SARS-CoV), Middle east respiratory syndrome (MERS-CoV), and novel SARS-CoV-2019 shows that not only viral propagation inside the host but also, the host inflammatory response is responsible for disease outcome [17]. Similarly, it can be seen with influenza virus for which the lethality of the disease is not due to viral propagation but due to inflammatory response by the host immune system upon viral entry [18]. Major mechanism involved in inflammatory and tissue injury due to virus is by generation of free radicals. Oxidative stress is necessary for viral replication and for inflammatory response by immune cells of the host. During severe condition of a disease, a cytokine storm(excessive production of cytokines) along with T-cell depletion, pulmonary inflammation and lung damage is seen [19]. Not only these, but also patients having Acute Respiratory Distress Syndrome(ARDS) and also other types of pneumonia induced by virus shows Macrophage Activation Syndrome (MAS)(inflammatory stress) [19,20]. Clinical observations also shows that there are two more effects of viral entry into host body: leukopenia [21](decrease in T-cell count due to pyroptosis, which is a cell death mainly affecting immune cells) [19,22], and second is granulocytosis, which is responsible for generation of Reactive Oxygen Species(ROS) [23] by a strong bust of superoxide [19], and thus increasing production of pro- inflammatory cytokines [24].
So far, it is clear that one of the most deleterious effects of the novel coronavirus-2019 is hyperactivation of the host immune system, leading to inflammation in the host body. Thus, anti inflammatory therapy should be given to complement general anti-viral therapy, to manage the symptoms of Covid-19 efficiently [19]. In this article we will discuss role of cytoprotective transcription factor NRF2 and ways of boosting endogenous cellular defenses by increasing the cytoprotective transcription factor NRF2(Gene NFE2L2), which will help to resolve Covid-19 associated inflammation and also restore redox homeostasis along with tissue repair [19].
3. NRF2 activators in viral infection
3.1. NRF2 and its anti-inflammatory roles
NRF2 is a cap ‘n’ collar (CNC) transcription factor, it dimerizes and forms complexes with small musculoaponeurotic fibrosarcoma (sMAF) proteins, which are mainly K, G and F [25], or they may bind to other transcription factors like C-JUN and JUND [26], and then binds to anti-oxidant response elements(ARE) and regulate transcription of various target genes, involving in cellular redox homeostasis, detoxification, tissue damage and metabolic homeostasis [27]. Under normal conditions, NRF2 is complexed with E3 ligase substrate adapter Kelch-like ECH-associated Protein 1(KEAP-1) and thus leading to ubiquitination and proteosomal degradation of the transcription factor [[27], [28], [29]]. ROS generation within a cell leads to modification of specific cysteine residues of KEAP1 leading to its inactivation [30], and thus due to inactivation of KEAP1, it leads to accumulation of NRF2 and resulting in enhanced target gene transcription.
NRF2 is disregulated in various disease conditions like diabetes, and various inflammatory diseases [31] and it also declines on aging [32]. Due to this, people with disease conditions like diabetes and old age are at high risk for factors associated with SARS-CoV induced ARDS [33]. Importantly, activation of NRF2 has been shown to preserve lung architecture in response to inflammation and activation of NRF2 in various animal models has shown to be beneficial in several lung disorders and respiratory infections including ARDS [34]. Single nucleotide polymorphism(SNPs) which is present in the promoter region of NFE2L2 gene(which encodes for NRF2)is seen to be implicated with lung diseases susceptibility in humans, showing NRF2 as a therapeutic target for pulmonary diseases [35].
NRF2 plays anti-inflammatory role by suppressing pro-inflammatory genes such as IL6 and IL1B [36]. NRF2 also induces expression of several macrophage-specific genes that help in tissue repair. One of the gene induced is of Macrophage Receptor with collagenous structure (MARCO), which is responsible for bacterial phagocytosis. It also induces gene for cluster of differentiation-36(CD36), which is a scavenger for oxidized low density lipoproteins(LDL) [37], and IL-17D [38], to give protection against viral infections [39]. Similarly, NRF2 activation leads to restoration of redox homeostasis, this is done by upregulation of various anti-oxidant genes, like Glutathione(GSH), NADPH, thioredoxin, thioredoxin reductase and peroxiredoxin, which protect the cells from ROS and also NRF2 activation provides an alternative pathway for wound healing apart from the one involving inflammatory cascade [19,40].
3.2. NRF2 activation and alveolar protection
Alveolar epithelial type 2 cells that are present in lungs are the most abundant cells that express ACE2 which is about 83% [4]. ACE2 protein is mostly expressed in apical airway epithelia, where they are linked to epithelia differentiation [41]. These differentiated cells are more likely to be infected by viral infection like SARS-CoV [42]. NRF2 activation prevents lung infection by increasing ACE2 and AT2R levels. NRF2 also decreases serum Angiotensin-2 and AT1R levels, thus suppressing oxidative stress, mediated inflammation and fibrosis caused by Angiotensin-2/AT1R axis [43]. NRF2 also plays an important role in increasing interferon gene expression leading to anti-viral activity. In a study, Kesic et al. showed that NRF2 activators increases the expression of RIG-1 and IFN-β, thus enhancing anti-viral activity [44].
3.3. NRF2 activation and inhibition of NF-kB and inflammatory modulators
NRF2 inhibits IKK activation and thus inhibiting proteosomal degradation of IKB. Thus, inhibiting activation of NF-kB. Secondly, NRF2 and NF-kB binds to the same ARE region of the genome. Thus, activation of NRF2 leads to increased levels of NRF2 and thus more NRF2 binds to ARE Region of the genome and NF-kB is inactivated [42]. Along with this, activation of NRF2 also suppresses generation of inflammatory factors such as TNF-α, IL-6, MCP-1, MIP2 [45]. In addition to this, NRF2 activation also downregulates expression of various adhesion molecules such as Selectins and VCAM-1 [46]. TLR expression is upregulated in NRF2 Knockout mice, when compared to wild type, showing the role of NRF2 in TLR expression [47].
3.4. NRF2 activation and anti-apoptotic effect
NRF2 downregulates and decreases the expression of Bcl-2/Bax in lungs, which is upregulated in viral infections. In viral infections, activity of caspases 3 and 9 are also upregulated, which is downregulated by NRF2, thus decreasing overall apoptosis [48].
4. Biology of SARS-CoV and its crosstalk with NRF2
SARS-CoV genome consists of both structural and non-structural proteins. Structural proteins consists of Spike(S), Envelope(E), Membrane(M) and Nucleocapsid(N) and other accessory proteins like ORF3, 6, 7a, 8 and 9b, that interacts with the host cells and are responsible for replication of the genome [49]. The RBD is located on the S-protein of the SARS-CoV(structural protein) which interacts with the ACE2 of the host cells, allowing entry of the virus into the host body [50]. ACE2 converts Angiotensin-2 to Angiotensin- [[1], [2], [3], [4], [5], [6], [7]]. Use of ACE inhibitors like Captopril and angiotension receptor blockers like losartan, which are generally prescribed to cardiovascular patients [51] across the globe, has been considered for COVID-19, because Angiotension-2, which is a target for ACE inhibitors, has vasoconstrictive, proinflammatory, pro-oxidative and prothrombic effects [52]. But, use of these inhibitors alters the ACE/ACE2 ratios and increases the ACE2 levels, thus potentially increasing the docking sites for entry of the virus [53]. NRF2 deficiency has been shown to increase ACE2 levels, whereas NRF2 activators have been shown to reduce ACE2 levels, thus suggesting that ACE2 levels may decrease upon NRF2 activation, thus decreasing the overall docking sites for viral entry [54].
Also upon activation of NRF2, secretory leukocyte protease inhibitor (SLPI) production is increased, which is an anti-protease which inhibits serine protease activity. It also protects the target cells from viral infection by decreasing TMPRSS2 expression by binding to its promoter region [55]. Hence, these activators can prevent the cell against any viral infection and replication [42]. In a study by Lizuka et al. (2005), it is found that SLPI is not expressed in NRF2 knockout mice and thus it disturbs the protease/anti-protease balance, and thus exposes the cells to inflammation. They also found out that SLPI gene expression was elevated by activation of NRF2 and by thus maintaining protease/anti protease balance [56]. In a similar study conducted by Ling JX et al. (2012), it was proved that an oral administration of NRF2 activator(EGCG), decreased the viral infection and viral replication in lungs, thus increasing the survival [42].
Like other coronavirus, novel SARS-CoV-2019 modulates the host translational mechanisms to generation its own proteins [57]. To counteract this, the host inactivates the eukaryotic initiation factor(EIF2)by inactivating two cellular kinases, protein kinase R(PKR) and PKR-like endoplasmic reticulum kinase(PERK), both of which is activated in SARS-CoV infection [58]. PKR has been known to upregulate the autography cargo protein p62, that competes for KEAP1 binding with NRF2 [59], thus promoting autographic degradation of KEAP1 [60] and releasing free NRF2, thus increasing its transcriptional activity. SARS-CoV infection triggers the host Unfolded Protein Response (UPR), as host tries to block the translation of coronavirus proteins. UPR leads to activation of PERK [61]. PERK phosphorylates and activates NRF2 [62]. Thus, this is the first point that shows that NRF2 activation may slow down corona viral infection [19].
Coronavirus is a RNA Virus and it is shown that cells that are infected with RNA virus recognize the viral molecular patterns of nucleic acids by the help of cytoplasmic and endosomal receptors. Some of the RNA sensor receptors that are present are retinoic acid-inducible gene 1 (RIG-1) and Melanoma differentiation-associated protein 5 (MDA-5) [63], and DNA sensor receptors that are present are cyclic GMP-AMP Synthase(CGAS). These sensor receptors mediate immune response via Adaptor Protein stimulator of interferon genes(STING) [64]. Upon activation of STING it leads to increase in transcription of IFN 1 and IFN 3 genes via activation of interferon regulatory factor 3(IRF-3) [64]. SARS-CoV has been shown to antagonize and decrease STING-Mediated immune response [65,66]. IFN 1 is necessary for restricting viral replication and thus spread, mainly by activating autocrine and paracrine type 1 IFN receptor, but excessive release of IFN 1 by infected pulmonary alveolar cells and macrophages may lead to more infiltration of monocyte derived macrophages, thus may potentiate inflammatory damage [67]. NRF2 prevents this exacerbation of inflammatory response by IFN-1, by partly downregulating STING expression [68]. Therefore, NRF2 reduces inflammation by decreasing excessive production of IFNs(IFN-1 in particular) [69].
Apart from this, upregulation of NRF2 also activates heme oxygenase 1(OH-1), which has been known to be effective against many viruses including HIV, Hepatitis and Ebola viruses [70]. OH-1 performs anti-viral actions mainly by forming a heterodimer complex with IRF3 [71]. Due to the formation of this heterodimer complex, it leads to phosphorylation of IRF3 and thus translocation of IRF3 into nucleus, thus inducing the expression of type-1 IFNs [19]. Therefore, we have seen that NRF2 activation leads to partial decrease in IFN1 levels by antagonizing STING expression, and also partly increasing IFN1 levels by increasing OH-1 transcription and thus increasing phosphorylation and translocation of IRF3 into the nucleus (Refer Fig. 1 ).
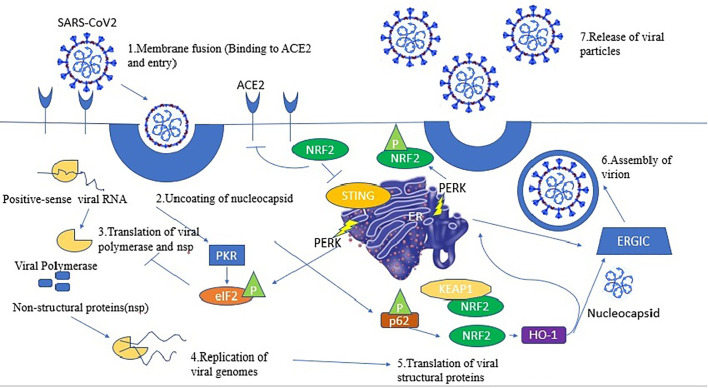
Fig. 1.
This reflects the various steps of viral cycle (1) The viral spike (S) protein binds to to ACE2 causing virion entry (2) Uncoating of the viral nucleocapsid in the cytoplasm of the host cell. (3) Specific viral proteins are formed as a result of translation of the viral positive-sense single-stranded RNA (+ssRNA) and final cleavage of the translation product. The DNA/RNA sensor cGAS is activated by the viral RNA inside the host cell, which signals through the adaptor STING, and leads to induction of type I and type III interferons (IFNs). In turn IFN production is repressed by NRF2 by downregulation of STING expression [56]. (4) NRF2 brings about induction of HO-1 expression, producing Fe2+ that could bind to the divalent metal-binding pocket of the RNA-dependent RNA polymerase (RdRp) of SARS-CoV2 and inhibit its catalytic activity bringing about replication of viral genome (5) Double-stranded RNA-activated protein kinase R (PKR) conducts the host defense, which also phosphorylates eIF2 and inhibits protein translation. p62 is phosphorylated by PKR, thus bringing about NRF2 activation upon removal of its repressor KEAP1 by autophagy. Inhibition of protein translation in turn activates the unfolded protein response (UPR). PERK, a crucial Ser/Thr protein kinase in UPR signaling, brings about NRF2 phosphorylation leading to its stabilization and increased transcriptional activity (6) Virion assembly (7) Release of viral particles. Abbreviations: ACE2, angiotensin-converting enzyme 2; eIF2, eukaryotic initiation factor 2; ER, endoplasmic reticulum; ERGIC, ER–Golgi intermediate compartment; HO-1, heme oxygenase 1; IFN, interferon; KEAP1, Kelch-like ECH-associated protein 1; NRF2, nuclear factor erythroid 2 p45-related factor 2; PERK, PKR-like endoplasmic reticulum kinase; P, phosphorylation; PKR, protein kinase R; STING, stimulator of interferon genes[141]
Apart from the above mechanism, many other mechanisms has been known for the anti-viral activity by OH-1, that is extrapolated for SARS-CoV-2019 [19]. One of the mechanism by which(OH), can help in SARS-CoV infection is by helping in degradation of heme to form three products, biliverdin, Fe+2 and CO, with each of them having anti-SARS CoV activity [19]. Coronaviruses require two major viral proteases viz.3C-like protease(3CL-pro) and papain like protease(PLpro), for the process of viral polyproteins and essential for viral replication [72]. Both of these proteases share a high degree of homology and known to be inhibited by biliverdin [73,74]. Biliverdin is thus expected to inhibit both of these proteases in SARS-CoV and thus its production of viral polyproteins and thus viral replication. Free Fe+2 released by degradation of heme binds to metal binding domain of RdRP of various viruses resulting in inhibition of its enzymatic activity [75,76]. SARS-CoV is also having such metal binding domain and thus a similar mechanism is expected in SARS-CoV [77], as well for Novel Coronavirus. CO inhibits an increase in ROS levels, by activating soluble guanyl cyclase (sGC), this leads to increase in levels of c-GMP locally, and thus activates protein kinase G(PKG) [19], PKG further inhibits NADPH oxidase(NOX), which is responsible for ROS production, thus decreasing overall ROS load [78]. When these mechanisms are viewed in context with SARS-CoV, it is believed to hold promising result against SARS-CoV.
5. NRF2 activators versus other anti-inflammatory therapies used in Covid-19
The worsened inflammation seen in COVID-19 patients could potentially be treated with anti-inflammatory drugs such as corticosteroids and non-steroidal anti-inflammatory drugs (NSAIDs), and by other therapies like monoclonal antibodies [79]. While results with NSAIDs are inconclusive in people with COVID-19 [80], ibuprofen, an NSAID, has been shown to impair the function of neutrophils, their recruitment to the inflammatory site, and the resolution of inflammatory processes in patients with pneumonia [81]. However, ibuprofen is associated with higher rates of nephrotoxicity [82], cardiovascular disease and stroke [83] and appears to increase the risk of these outcomes in ARDS [84]. A significant difference between NSAIDs and NRF2 activators is that NRF2 elicits a much more integrated regulation of the inflammatory response as it is necessary for both execution and resolution. In addition, by regulating the endogenous cytoprotective systems, NRF2, may be a more physiological role in achieving a balance between the beneficial and adverse effects of inflammation [19].
Corticosteroids are used in COVID-19 for the treatment of cytokine storm. Indeed, the RECOVERY trial (randomized evaluation of COVID-19 therapy), a multi-center randomized clinical trial in COVID-19 patients from National Health Service (NHS) hospitals in the UK, found that low-dose dexamethasone, a corticosteroid, reduced mortality in ventilated-driven patients and in patients receiving oxygen alone, although it had no effect in patients receiving no respiratory support. However, use of corticosteroids have some limitations like, use of corticosteroids for the treatment of COVID19 infection showed restrictions primarily related to the suppression of specific immunity and an increased risk of secondary bacterial pneumonia [85]. Another, alternative to these conventional therapies that are used in COVID-19 and very much effective are the drugs that target cytokines involved in the cytokine storm of COVID-19 includes use of monoclonal antibodies. Currently monoclonal antibodies that are used in COVID-19 for the treatment of cytokine storm includes tocilizumab (anti IL6), anakinra(anti IL1), secukinumab (anti IL 17A), canakinumab (anti IL1β), ruksolitinib phosphate (JAK kinase inhibitor), etc. But, these monoclonal antibodies can only target one of the many inflammatory cytokines, and thus have a limited action. Thus, it becomes very important to find other therapies to prevent cytokine storm in COVID-19 [86]. Use of NRF2 activators represents an excellent alternative or parallel to this monoclonal antibodies as well for corticosteroids because it is known that NRF2 inhibits IL-6, IL-1β, IL-17 gene expression [36].
6. Sirtuins: an introduction
Histone deacetylases (HDACs) are categorized under a group of enzymes that are responsible for the removal of acetyl group from ε-N –acetyl lysine amino acid residues from histone as well as non-histone proteins [87]. Till today there are around 18 HDACs that have been discovered in humans.
HDACs have been classified into two families (‘classical’ and ‘sirtuins’) and into four classes (Class1, Class2, Class3 and Class4) [88]. Sirtuins belong to Class 3 Histone deacetylases with highly conserved NAD+ domain, having high sequence similarity with yeast Saccharomyces cerevisiae Protein Sir2(silent information regulator 2) and having no sequence similarity with traditional HDACs [89]. So, sirtuins are also enzymes that catalyze post translational modification of histone and non-histone proteins. There are seven types of sirtuins in mammalian families, differing in localization, enzymatic activities and targets have been identified [90]. SIRT1 and SIRT6 are found predominantly in nucleus, SIRT2 is found in cytoplasm and SIRT3, SIRT4 and SIRT5 are found in mitochondria [91]. In one of the studies, published in 1999, it was seen that overexpression of Sir2 increases lifespan in yeast [92]. In 2000, a research study showed and identified Sir2 as an NAD+ dependent HDAC that performs deacetylation of lysine 9 and 14 of Histone (H3) and lysine 16 Histone (H4) showing that sirtuins and its deacetylation activity may play a role in longetivity in yeast [93]. Firstly, they were only known to perform deacetylation, but recently they have been found to catalyze a large number of other post translational modifications as well, such as desuccinylation, demalonylation and deglutarylation [94,95]. SIRTs have been classified as class-3 Histone deacetylases(HDACs), as they use Nicotinamide Adenine Dinucleotide(NAD) as a cofactor, differing from class-1 and class-2 which uses zinc as a cofactor instead. They are involved in large number of cellular functions and are operated by various stimuli, including metabolic changes, inflammatory signals and oxidative stress [[96], [97], [98], [99]]. Majorly disruption of redox cellular homeostasis affects SIRTs, by either inducing or repressing its expression and finally leading to post translational modifications like cysteine oxidation or nitrosylation, leading to its inactivation [100].
7. Sirtuins and COVID-19
7.1. SIRT1 and its role in combating COVID-19
Of utmost interest is SIRT1, which downregulates ADAM 17 (A Disintegrin and Metalloproteinase Domain 17), also called TNF-α converting enzyme (TACE), by increasing expression of TIMP3, the gene that encodes for tissue metalloproteinase inhibitor 3 [101]. As a consequence, the levels of TNF-α, IL-1β and IL-6 decreases. An increase in TNF-α causes SIRT1 to down-regulate ADAM 17, thereby controlling TNF-α formation in a negative feedback loop that secondarily influences IL-1 β and IL-6 production, which are dependent on TNF-α [101]. If ADAM17 expression is not downregulated by SIRT1, TNF-α and IL-6 are released, resulting in an uncontrolled hyperinflammatory response as may occur with COVID-19 [101,102]. SIRT 1, by inhibition of ADAM17 and thereby TNF-α and IL-6, performs an anti-inflammatory function. If oxidative stress is severe, increased ADAM17 attempts to ameliorate tissue injury by converting active iron (Fe2+) to its inert form (Fe3+) which is stored in hepatocytes and macrophages and as ferritin by means of the Fenton reaction (Fe2+ + H2O2 → Fe3+ + HO% + OH−), (Fe3+ + H2 O2 → Fe2+ + HO2% + H+). This also potentially transforms haemoglobin to methaemoglobin, reducing its capacity to bind to oxygen [103,104].
SIRT1 not only controls and modifies the inflammatory response, but along with the Sirtuin family (SIRT1–7) is also a primary defense against DNA and RNA viral pathogens [105]. In some respiratory infections and cardiovascular conditions, SIRT1 promotes autophagy (the destruction of damaged or redundant cellular components occurring in vacuoles within the cell), and in so doing inhibits apoptosis and provides protection against hypoxic stress. Upregulation of SIRT1 directly decreases viral replication and inhibits the activation of ADAM17, thereby decreasing TNF-α, IL-1β and IL-6. Conversely depletion of SIRT1 allows for increased viral replication with little or no inhibition of ADAM17 activity, causing uncontrolled increase in TNF-α, IL-6 and IL-1β levels. Whereas an increase in TNF-α would usually increase SIRT1 activity to downregulate ADAM17, in the presence of a deficiency of NAD+ or Zn++, this would not occur due to insufficient activation of SIRT1, causing an unchecked increase in TNF-α. Given the above, it is possible that activation of SIRT1 may be a crucial factor in the prevention of the hyperinflammatory response and may be necessary for a successful defense against viral attack [103].
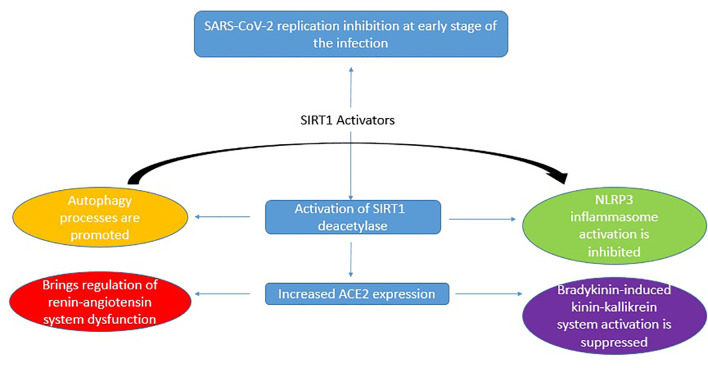
Resveratrol, a SIRT1 activator, directly inhibits viral replication, but apart from this it also performs inhibitory functions on the pathogenetic mechanism involved in COVID-19 severity. These include dysregulated NLRP3 inflammasome activation, renin-angiotensin system dysfunction, and kinin−kallikrein system stimulation. These inhibitory functions are mediated by the induction of Sirt1 protein and of Sirt1-induced upregulation of ACE2 protein expression. This proves that resveratrol functions as a SIRT1 activator in treatment of COVID-19 [106](Refer Fig. 2 ).
Fig. 2.
Antiviral activity of SIRT1 activators in curbing the pathogenetic mechanism involved in COVID-19 severity [142].
7.2. Role of nutrition and diet as Sirtuin activators for prophylaxis in COVID-19
7.2.1. Natural polyphenols
It is shown that supplementation of polyphenols in Covid-19 may help in preventing SARS-CoV2 viral infection by two ways: 1)Prevention of viral entry by binding to the ACE2 receptor and 2)Regulation of consequences of SARS-COV2 on lungs by regulating the expression of ACE2 [107]. According to 2013 study published in the Journal of Nutrition, eating a diet rich in polyphenols, results in longevity and decreases mortality in adults [108]. Dietary foods that are rich in polyphenols includes berries, olive oil, coffee, green tea, dark chocolate, turmeric, ginger, etc. Consuming these foods can help the body have balanced and optimized Sirtuin activation.
7.2.2. Zinc rich foods
Zinc is one of the most important nutrient to keep your immune system strong. Supplementation of zinc in case of Covid-19 has not been studied extensively. But the data available from literature suggests great benefits of zinc supplementation. It improves the mucociliary clearance, strengthens the integrity of the epithelium, decreases viral replication, preserves antiviral immunity, attenuates the risk of hyper-inflammation, supports anti-oxidative effects and thus reduces lung damage and minimized secondary infections [109]. However, in randomized clinical trials on ambulatory patients diagnosed with SARS-COV2 infection, when treated with high dose of zinc gluconate, ascorbic acid or a combination of both, did not significantly decrease the duration of symptoms when compared to standard care [110]. In a study published in 2016 by PLoS One, zinc may increase both mean and maximum lifespan by various longevity pathways [111]. One of those pathway is the activation of Sirtuin pathway. Regular diet that are rich in zinc includes fish, red meat, dairy products, eggs, nuts, spinach, etc. Consuming these foods can help the body have balanced and optimized Sirtuin activation.
7.2.3. Omega-3 fatty acids rich foods
Omega-3 Fatty Acids are one of the most important healthy fats that help to reduce chronic inflammation. In case of Covid-19, it presents various other benefits like prevention of the viral entry by changing the composition of the fats in the lipid membrane of the cells. Other benefits include prevention of activation of NF-KB, IL-6, and other similar pro-inflammatory mediators by various mechanisms, and thus decreasing the inflammation associated with SARS-CoV2. It also plays a role in increasing the phagocytic clearance capacity of the macrophages, thus helps to clear viral load. Apart from this, it also helps to modulate both innate and acquired immunity [112]. According to a study in 2013, supplements of omega-3 fish oil helps to lower inflammation and oxidative stress, and thus increasing lifespan and decreasing mortality [113]. In a study conducted in 2018, it was observed that Omega-3 Fatty acid attenuates the inflammatory response through SIRT-1 [114]. Foods that are rich in Omega-3 Fatty acids includes Fish and other seafoods, algae, flax seeds, chia seeds, walnut, etc. Consuming these foods can help the body have balanced and optimized Sirtuin activation.
7.2.4. Vitamin(B3) rich foods
Vitamin B3 or Niacin is very important for brain and mental activity, along with many other cellular and metabolic functions. Vit-B3 serves as an important precursor for NAD and NADP biosynthesis, both of them vital during systemic chronic inflammation [115]. Recent, evidences showed that increase in NAD+ levels, helps to decrease pro-inflammatory cytokines like IL-1, IL-6 and TNF-α, of which IL-6 is the major cytokines involved in Covid-19 Cytokine Storm [116]. It also reduces neutrophil infiltration and thus inflammation in acute lung injury [117]. Apart from these, it helps to prevents viral replication and strengthens the body's defense mechanisms. Taking this account this can be used as an adjunct therapy for Covid-19 [118]. A study conducted in 2013 shows that niacin supports energy metabolism and lifespan. Activation of Sirtuins by the study conducted in 2019, by niacin by metabolism to NAD+ [119]. Foods that are high in niacin include liver, other organ meats, grass-fed red meat, pasture-raised poultry, wild-caught fish, nutritional yeast, grass-fed dairy, algae, seaweed, and mushrooms. Consuming these foods can help the body have balanced and optimized Sirtuin activation.
7.3. Effect of lifestyle (activities promoting SIRT1 activation) on prophylaxis to COVID-19
7.3.1. Calorie restrictions (fasting)
Fasting, an intentional abstinence from food for a period of time, is practiced as a religious ritual known to have a myriad of health benefits, including strengthening immunity, resistance to stress, slowing the aging process, and prolonging the health of the body longevity without noticeable side effects. Along with these it also activates autophagy [120], which helps to boost the immunity [121]. Since, one of the symptoms of Covid-19 is low immunity and inhibition of autophagy, fasting can be a measure to increase the immunity by either directly activating the immune response or by inducing autophagy, both which are compromised in Covid-19 [122]. A paper published in 2014 [123], tried to show a molecular link between Sirtuin activation and Calorie Restrictions(Fasting) [123]. It was observed that Sirtuins sense low calories and mediate the beneficial effects of Calorie Restrictions via its activation. Thus, fasting or calorie restrictions can help the body to activate Sirtuins.
7.3.2. Exercise
Emotionally and physically, the benefits of exercise are enormous. It has two roles in Covid-19, one is via enhancing the mood, sleep quality and decreasing the stress and anxiety that may be aroused due to this pandemic [124]. On the other hand, exercise with an appropriate intensity is associated with better immune system responses against viral respiratory infections [125]. However, the connection between the immune system and the physical activity is a J shaped one. The physical activity must be kept moderate enough, otherwise uncontrolled and prolonged high-intensity physical activity may have other opposite consequences like immunosuppression [125]. These depends on the duration and intensity of exercise. High intensity exercises results in leukocytosis that is mediated by an increased number of Neutrophils, T and B lymphocytes and NK cells in the systemic circulation, making the immune response against antigens a little blunt [126]. This is explained by activation of the hypothalamic-pituitary-adrenal axis [126]. As the intensity of exercise increases, more the sympathetic system is stimulated and more is the release epinephrine and nor-epinephrine. This has to correlated with increased release of catecholamines and corticosteroids upon sympathetic stimulation [127]. Catecholamines when circulating in the blood then recruit lymphocytes [127], and thus decreasing the immune response against virus. In contrast to this, moderate intensity exercises serves as “immuno-enhancing” [126]. Low-intensity exercise results in an increased NK-Cells cytotoxic activity, increase in IL-2 production and increase in neutrophil phagocytic activity [126]. Hence, this shows that low to moderate exercise may be helpful in case of Covid-19 by enhancing immune response against antigen(viral in this case) [128]. According to a study conducted in 2009, exercise offers anti-inflammatory and anti-oxidative benefits and may increase Sirtuin's activity [129]. Sirtuins can be activated by Cyclic-AMP(c-AMP), which is activated by exercises and physical activity [130]. Thus, exercise can be used as a mean of activating Sirtuins in the body.
8. Crosstalk between Sirtuins and Nrf2
We have already discussed the role of NRF2 in combating oxidative stress by mainly two mechanisms: 1)By upregulation of mitochondrial anti-oxidant enzymes 2)By inhibition of NF-k B, which is a key modulator of inflammatory cascade leading to more oxidative stress [131].
The fact that sirtuins are involved in Antioxidant and Redox Signaling(ARS), comes from the fact that many of the molecules of the Antioxidant Response Element(ARE), mediates signaling processes via transcriptional regulation of gene expression in cells exposed to oxidative stress [91]. The ARE uniquely sense alterations in redox homeostasis due to biological and structural features and thus triggers transcriptional responses mediated mainly by Nuclear Factor E2-related factor 2(NRF2).
Sirtuins are NAD+ dependent class-3 Histone deacetylases(HDACs), which deacetylases various proteins and genes involved in oxidative stress and anti-oxidant pathways. One of the proteins that is involved in anti-oxidant pathway and also is deacetylated by Sirtuins is NRF2. Under normal conditions, NRF2 is bound to CUL3 and KEAP1, and labelled for proteosomal degradation via polyubiquitination [132]. But in case of oxidative stress, this CUL3/KEAP1 mediated proteosomal degradation is hindered and thus it makes NRF2 free. This free NRF2 then translocates to the nucleus and there it binds to small maf proteins forming a heterodimer [133,134]. This heterodimer then binds to promoter region of the DNA which is responsible for transcription of anti-oxidant enzymes, known as anti-oxidant response element(ARE) or Electrophilic response element(ERE), increasing transcription of anti-oxidant enzymes and thus anti-oxidant redox signaling [135].
In a very recent study it was shown that SIRT2 deacetylates NRF2, resulting in decrease in its total and nuclear levels [136]. Also SIRT2 has been shown to modulate nuclear NRF2 levels by AKT phosphorylation, leading to modulation in levels of Glutathione(GSH) and glutamate cysteine ligase(GCL), indicating important role of SIRT2 in ARE [137]. SIRT1 has also been shown to be involved, as knockdown of SIRT1 has been shown to inhibit the expression of NRF2, Heme Oxygenase(OH-1) and SOD, eliminating the neuroprotective action of SIRT1 induced by hyperbaric oxygen preconditioning against transient focal cerebral ischemia [138]. SIRT1 activator, resveratrol have been found to modulate NRF2-mediated antioxidant protein expression to promote neuroprotection against cerebral ischemic injuries [139]. BML-278, EX-527, Piceatannol, Salermide, SirtAct, Sirtinol, Splitomicin, Triacetyl resveratrol are other SIRT1 activators which could be possibly tested for their role in alleviating the pathophysiology of COVID-19. SIRT6 has been found to activate NRF2 in order to protect Human Mesenchymal cells(hMSCs) from oxidative stress [140].
NRF2 interacts with sirtuins for increasing anti-oxidant redox signaling and to maintain redox homeostasis. Sirtuins deacetylates NRF2 and thus increasing its transcription by epigenetic modifications [91].
9. Conclusion
We therefore conclude that the two major host cell factors TMPRSS2 and ACE2 are responsible for the virus action in host body and could be the novel targets for further drug discovery. Not only in viral propagation inside the host cells but also the inflammatory response of the host is crucial in determining the disease spell on the target. Inflammatory response of the host is inflicted by increased number of free radicals, oxidative stress, cytokine storm, leukopenia and granulocytosis. NRF2 activators can thus be a torch of hope because they have multiple facets to their mode of action. Suppression of pro-inflammatory genes, IL-6 and IL-1β depict the anti-inflammatory role of NRF2. It also induces macrophage-specific genes responsible for tissue repair. Also, SLPI production is increased on activation of NRF2 which inhibits serine protease activity. NRF2 decreases TMPRSS2 expression by binding to its promoter region and protects the viral cells from replication. NRF2 also elevates ACE2 and AT2R levels and thus helps in preventing lung infection. It suppresses OS mediated fibrosis as well as inflammation by decreasing serum Angiotensin 2 and AT1R levels. NRF2 also inhibits NF-kβ as NRF2 and NF-kβ both compete to bind to the same ARE region of the genome and when NRF2 is activated more levels of NRF2 will ultimately lead to NF-kβ inhibition. NRF2's anti-inflammatory mechanism is also linked to its ability of suppressing generation of other inflammatory factors like TNF-α, IL-6, MCF-1, etc. Another mechanism important for its action is that it downregulates expression of apoptotic factors like Bax-BCl2 in lungs alone with upregulatory activity of Caspases 3 and 9 that is downregulated by NRF2 and finally decreases apoptosis. Sirtuins are also linked to NRF2. Sirtuins are involved in Anti-oxidant and Redox Signaling (ARS) and NRF2 is one of the proteins deacetylated by Sirtuins and thus increases its transcription by epigentic modifications. Both SIRT1 and SIRT2 are linked with NRF2, most prominent being SIRT1, as absence of SIRTs by knocking them down will inhibit the expression of NRF2. Therefore SIRT activators are also crucial and can play an important role in alleviating COVID-19 symptoms by various mechanisms and SIRT1 activators in the form of food and lifestyle too can help in combating COVID-19 as described in detail above. We therefore propose that NRF2 activators /and SIRT activators can be potential therapeutic agents in the treatment and alleviation of COVID-19.
Declaration
The authors have no conflict of interest to declare.
References
- 1.Naser N., Masic I., Zildzic M. Public health aspects of COVID19 infection with focus on cardiovascular diseases. Mater. Socio Med. 2020;32(1):71. doi: 10.5455/msm.2020.32.71-76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Iwata-Yoshikawa N., Okamura T., Shimizu Y., Hasegawa H., Takeda M., Nagata N. TMPRSS2 contributes to virus spread and immunopathology in the airways of murine models after coronavirus infection. J. Virol. 2019;93(6):1–15. doi: 10.1128/JVI.01815-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wen L., Juan H., Ling Y., Tanaka Y., Zhang W. 2020. Since January 2020 Elsevier has Created a COVID-19 Resource Centre with Free Information in English and Mandarin on the Novel Coronavirus COVID- 19. The COVID-19 Resource Centre is Hosted on Elsevier Connect, the Company’s Public News and Information. (January) [Google Scholar]
- 4.Zhao Y., Zhao Z., Wang Y., Zhou Y., Ma Y., Zuo W. Single-cell RNA expression profiling of ACE2, the receptor of SARS-CoV-2. Am. J. Respir. Crit. Care Med. 2020;202(5):756–759. doi: 10.1164/rccm.202001-0179LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Malterer M.B., Glass S.J., Newman J.P. Interferon-stimulated genes: a complex web of host defenses. Annu. Rev. Immunol. 2014;44(3):735–745. doi: 10.1146/annurev-immunol-032713-120231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Frieman M., Heise M., Baric R. SARS coronavirus and innate immunity. Virus Res. 2008;133(1):101–112. doi: 10.1016/j.virusres.2007.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hu Yong, Li Wei, Gao Ting, Cui Yan, Jin Yanwen, Li Ping, Ma Qingjun, Liu Xuan, Cao C. The severe acute respiratory syndrome coronavirus nucleocapsid inhibits type I interferon production by interfering with TRIM25-Mediated RIG-I ubiquitination. J. Virol. 2017;91(8):1–15. doi: 10.1128/JVI.02143-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kuba K., Imai Y., Rao S., Gao H., Guo F., Guan B., et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat. Med. 2005;11(8):875–879. doi: 10.1038/nm1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dosch S.F., Mahajan S.D., Collins A.R. SARS coronavirus spike protein-induced innate immune response occurs via activation of the NF-κB pathway in human monocyte macrophages in vitro. Virus Res. 2009;142(1–2):19–27. doi: 10.1016/j.virusres.2009.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Boehm M., Nabel E.G. Angiotensin-converting enzyme 2–a new cardiac regulator. N. Engl. J. Med. 2002 Nov;347(22):1795–1797. doi: 10.1056/NEJMcibr022472. [DOI] [PubMed] [Google Scholar]
- 11.Paracha U.Z., Fatima K., Alqahtani M., Chaudhary A., Abuzenadah A., Damanhouri G., et al. Oxidative stress and hepatitis C virus. Virol. J. 2013;10(1):1. doi: 10.1186/1743-422X-10-251. [Internet]. Available from: Virology Journal. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Salihefendic N., Zildzic M., Ahmetagic S. Acute respiratory distress syndrome (ARDS) from endemic influenza A/H1N1: prehospital management. Med. Arch. (Sarajevo, Bosnia Herzegovina) 2015;69(1):62–63. doi: 10.5455/medarh.2015.69.62-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.de Queiroz T.M., Monteiro M.M.O., Braga V.A. Angiotensin-II-derived reactive oxygen species on baroreflex sensitivity during hypertension: new perspectives. Front. Physiol. 2013;4(May):1–6. doi: 10.3389/fphys.2013.00105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Imai Y., Kuba K., Rao S., Huan Y., Guo F., Guan B., et al. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436(7047):112–116. doi: 10.1038/nature03712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Li F., Li W., Farzan M., Harrison S.C. Structural biology: structure of SARS coronavirus spike receptor-binding domain complexed with receptor. Science (80-). 2005;309(5742):1864–1868. doi: 10.1126/science.1116480. [DOI] [PubMed] [Google Scholar]
- 16.Zhang R., Pan Y., Fanelli V., Wu S., Luo A.A., Islam D., et al. Mechanical stress and the induction of lung fibrosis via the midkine signaling pathway. Am. J. Respir. Crit. Care Med. 2015 Aug;192(3):315–323. doi: 10.1164/rccm.201412-2326OC. [DOI] [PubMed] [Google Scholar]
- 17.Guan W., Ni Z., Hu Y., Liang W., Ou C., He J., et al. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020;382(18):1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Galani I.E., Andreakos E. Neutrophils in viral infections: current concepts and caveats. J. Leukoc. Biol. 2015 Oct;98(4):557–564. doi: 10.1189/jlb.4VMR1114-555R. [DOI] [PubMed] [Google Scholar]
- 19.Cuadrado A., Pajares M., Benito C., Jiménez-Villegas J., Escoll M., Fernández-Ginés R., et al. Can activation of NRF2 be a strategy against COVID-19? Trends Pharmacol. Sci. 2020;41(9):598–610. doi: 10.1016/j.tips.2020.07.003. [Internet]. Available from: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Qin C., Zhou L., Hu Z., Zhang S., Yang S., Tao Y., Xie C., Ma K., Shang K., Wang W., Tian D.S. Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan, China. Clin Infect Dis. 2020;71(15):762–768. doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fung S.Y., Yuen K.S., Ye Z.W., Chan C.P., Jin D.Y. A tug-of-war between severe acute respiratory syndrome coronavirus 2 and host antiviral defence: lessons from other pathogenic viruses. Emerg. Microbes Infect. 2020;9(1):558–570. doi: 10.1080/22221751.2020.1736644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sies H., Jones D.P. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat. Rev. Mol. Cell Biol. 2020 Jul;21(7):363–383. doi: 10.1038/s41580-020-0230-3. [DOI] [PubMed] [Google Scholar]
- 24.DeDiego M.L., Nieto-Torres J.L., Regla-Nava J.A., Jimenez-Guardeno J.M., Fernandez-Delgado R., Fett C., et al. Inhibition of NF- B-mediated inflammation in severe acute respiratory syndrome coronavirus-infected mice increases survival. J. Virol. 2014;88(2):913–924. doi: 10.1128/JVI.02576-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Otsuki A., Yamamoto M. Cis-element architecture of Nrf2-sMaf heterodimer binding sites and its relation to diseases. Arch. Pharm. Res. 2020 Mar;43(3):275–285. doi: 10.1007/s12272-019-01193-2. [DOI] [PubMed] [Google Scholar]
- 26.Venugopal R., Jaiswal A.K. Nrf2 and Nrf1 in association with Jun proteins regulate antioxidant response element-mediated expression and coordinated induction of genes encoding detoxifying enzymes. Oncogene. 1998 Dec;17(24):3145–3156. doi: 10.1038/sj.onc.1202237. [DOI] [PubMed] [Google Scholar]
- 27.Cuadrado A., Rojo A.I., Wells G., Hayes J.D., Cousin S.P., Rumsey W.L., et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat. Rev. Drug Discov. 2019 Apr;18(4):295–317. doi: 10.1038/s41573-018-0008-x. [DOI] [PubMed] [Google Scholar]
- 28.Cuadrado A., Manda G., Hassan A., Alcaraz M.J., Barbas C., Daiber A., et al. Transcription factor NRF2 as a therapeutic target for chronic diseases: a systems medicine approach. Pharmacol. Rev. 2018;70(2):348–383. doi: 10.1124/pr.117.014753. [DOI] [PubMed] [Google Scholar]
- 29.Hayes J.D., Dinkova-Kostova A.T. The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem. Sci. 2014 Apr;39(4):199–218. doi: 10.1016/j.tibs.2014.02.002. [DOI] [PubMed] [Google Scholar]
- 30.Dinkova-Kostova A.T., Holtzclaw W.D., Cole R.N., Itoh K., Wakabayashi N., Katoh Y., et al. Direct evidence that sulfhydryl groups of Keap1 are the sensors regulating induction of phase 2 enzymes that protect against carcinogens and oxidants. Proc. Natl. Acad. Sci. U. S. A. 2002;99(18):11908–11913. doi: 10.1073/pnas.172398899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rabbani P.S., Soares M.A., Hameedi S.G., Kadle R.L., Mubasher A., Kowzun M., et al. Dysregulation of Nrf2/Keap1 redox pathway in diabetes affects multipotency of stromal cells. Diabetes. 2019;68(1):141–155. doi: 10.2337/db18-0232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cho 乳鼠心肌提取 HHS public access. Physiol. Behav. 2016;176(1):100–106. [Google Scholar]
- 33.Wu C., Chen X., Cai Y., Xia J., Zhou X., Xu S., et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern. Med. 2020 Jul;180(7):934–943. doi: 10.1001/jamainternmed.2020.0994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liu Q., Gao Y., Ci X. Role of Nrf2 and its activators in respiratory diseases. Oxidative Med. Cell. Longev. 2019;2019 doi: 10.1155/2019/7090534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Acosta-Herrera M., Pino-Yanes M., Blanco J., Ballesteros J.C., Ambrós A., Corrales A., et al. Common variants of NFE2L2 gene predisposes to acute respiratory distress syndrome in patients with severe sepsis. Crit. Care. 2015 Jun;19(1):256. doi: 10.1186/s13054-015-0981-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kobayashi E.H., Suzuki T., Funayama R., Nagashima T., Hayashi M., Sekine H., et al. Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription. Nat. Commun. 2016;7(May):1–14. doi: 10.1038/ncomms11624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mizumura K., Maruoka S., Shimizu T., Gon Y. Role of Nrf2 in the pathogenesis of respiratory diseases. Respir. 2020 Jan;58(1):28–35. doi: 10.1016/j.resinv.2019.10.003. [DOI] [PubMed] [Google Scholar]
- 38.Liu Q., Gao Y., Ci X. Role of Nrf2 and its activators in respiratory diseases. Oxid. Med. Cell. Longev. 2019 Jan doi: 10.1155/2019/7090534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Roemer Emily J., West Kesley L., Northrup Jessica B., Iverson Jana M. 乳鼠心肌提取 HHS public access. Physiol. Behav. 2016;176(12):139–148. [Google Scholar]
- 40.Brüne B., Dehne N., Grossmann N., Jung M., Namgaladze D., Schmid T., et al. Redox control of inflammation in macrophages. Antioxid. Redox Signal. 2013;19(6):595–637. doi: 10.1089/ars.2012.4785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jia H.P., Look D.C., Shi L., Hickey M., Pewe L., Netland J., et al. ACE2 receptor expression and severe acute respiratory syndrome coronavirus infection depend on differentiation of human airway epithelia. J. Virol. 2005;79(23):14614–14621. doi: 10.1128/JVI.79.23.14614-14621.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hassan S.M., Jawad M.J., Ahjel S.W., Singh R.B., Singh J., Awad S.M., et al. The Nrf2 activator (DMF) and Covid-19: is there a possible role? Med. Arch. (Sarajevo, Bosnia Herzegovina). 2020;74(2):134–138. doi: 10.5455/medarh.2020.74.134-138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Jang I.A., Kim E.N., Lim J.H., Kim M.Y., Ban T.H., Yoon H.E., et al. Effects of resveratrol on the renin-angiotensin system in the aging kidney. Nutrients. 2018;10(11):1–15. doi: 10.3390/nu10111741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kesic M.J., Simmons S.O., Bauer R., Jaspers I. Nrf2 expression modifies influenza a entry and replication in nasal epithelial cells. Free Radic. Biol. Med. 2011;51(2):444–453. doi: 10.1016/j.freeradbiomed.2011.04.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Thimmulappa R.K., Scollick C., Traore K., Yates M., Michael A., Liby K.T., Sporn M.B., Yamamoto M., Kensler T.W., Biswal S. Nrf2-dependent protection from LPS induced inflammatory response and mortality by CDDO-Imidazolide. Biochem. Biophys.Res. Commun. 2006 Dec;351(4):883–889. doi: 10.1016/j.bbrc.2006.10.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Soares M.P., Seldon M.P., Gregoire I.P., Vassilevskaia T., Berberat P.O., Yu J., et al. Heme oxygenase-1 modulates the expression of adhesion molecules associated with endothelial cell activation. J. Immunol. 2004;172(6):3553–3563. doi: 10.4049/jimmunol.172.6.3553. [DOI] [PubMed] [Google Scholar]
- 47.Wang Y., Li L., Deng S., Liu F., He Z. Ursolic acid ameliorates inflammation in cerebral ischemia and reperfusion injury possibly via high mobility group box 1/toll-like receptor 4/NFκB pathway. Front. Neurol. 2018;9(MAY) doi: 10.3389/fneur.2018.00253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Pan H., Wang H., Zhu L., Wang X., Cong Z., Sun K., et al. The involvement of Nrf2-ARE pathway in regulation of apoptosis in human glioblastoma cell U251. Neurol. Res. 2013 Jan;35(1):71–78. doi: 10.1179/1743132812Y.0000000094. [DOI] [PubMed] [Google Scholar]
- 49.Zhu N., Zhang D., Wang W., Li X., Yang B., Song J., et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020;382(8):727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Chen Y., Guo Y., Pan Y., Zhao Z.J. Structure analysis of the receptor binding of 2019-nCoV. Biochem. Biophys. Res. Commun. 2020;525(1):135–140. doi: 10.1016/j.bbrc.2020.02.071. [Internet]. Available from: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Since January 2020 Elsevier has Created a COVID-19 Resource Centre with Free Information in English and Mandarin on the Novel Coronavirus COVID- 19. The COVID-19 Resource Centre is Hosted on Elsevier Connect, the Company’s Public News and Information. 2020. (January) [Google Scholar]
- 52.Mehta P.K., Griendling K.K. Angiotensin II cell signaling: physiological and pathological effects in the cardiovascular system. Am. J. Phys. Cell Physiol. 2007;292(1):1–75. doi: 10.1152/ajpcell.00287.2006. [DOI] [PubMed] [Google Scholar]
- 53.Magrone T., Magrone M., Jirillo E. Focus on receptors for coronaviruses with special reference to angiotensin- converting enzyme 2 as a potential drug target - a perspective. Endocr. Metab. Immune Disord. Drug Targets. 2020;20(6):807–811. doi: 10.2174/1871530320666200427112902. [DOI] [PubMed] [Google Scholar]
- 54.Zhao S., Ghosh A., Lo C.-S., Chenier I., Scholey J.W., Filep J.G., et al. Nrf2 deficiency upregulates intrarenal angiotensin-converting enzyme-2 and angiotensin 1-7 receptor expression and attenuates hypertension and nephropathy in diabetic mice. Endocrinology. 2018 Feb;159(2):836–852. doi: 10.1210/en.2017-00752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Schultz M.A., Hagan S.S., Datta A., Zhang Y., Freeman M.L., Sikka S.C., et al. Nrf1 and Nrf2 transcription factors regulate androgen receptor transactivation in prostate cancer cells. PLoS One. 2014;9(1):11–13. doi: 10.1371/journal.pone.0087204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Iizuka T., Ishii Y., Itoh K., Kiwamoto T., Kimura T., Matsuno Y., et al. Nrf2-deficient mice are highly susceptible to cigarette smoke-induced emphysema. Genes Cells. 2005 Dec;10(12):1113–1125. doi: 10.1111/j.1365-2443.2005.00905.x. [DOI] [PubMed] [Google Scholar]
- 57.Nakagawa K., Lokugamage K.G., Makino S. Free information in English and mandarin on the novel coronavirus COVID- viral and cellular mRNA translation in coronavirus- infected cells. Adv. Virus Res. 2020;96(January):165–192. doi: 10.1016/bs.aivir.2016.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Krähling V., Stein D.A., Spiegel M., Weber F., Mühlberger E. Severe acute respiratory syndrome coronavirus triggers apoptosis via protein kinase R but is resistant to its antiviral activity. J. Virol. 2009;83(5):2298–2309. doi: 10.1128/JVI.01245-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Komatsu M., Kurokawa H., Waguri S., Taguchi K., Kobayashi A., Ichimura Y., et al. The selective autophagy substrate p62 activates the stress responsive transcription factor Nrf2 through inactivation of Keap1. Nat. Cell Biol. 2010 Mar;12(3):213–223. doi: 10.1038/ncb2021. [DOI] [PubMed] [Google Scholar]
- 60.Taguchi K., Fujikawa N., Komatsu M., Ishii T., Unno M., Akaike T., et al. Keap1 degradation by autophagy for the maintenance of redox homeostasis. Proc. Natl. Acad. Sci. U. S. A. 2012;109(34):13561–13566. doi: 10.1073/pnas.1121572109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Chan C.-P., Siu K.-L., Chin K.-T., Yuen K.-Y., Zheng B., Jin D.-Y. Modulation of the unfolded protein response by the severe acute respiratory syndrome coronavirus spike protein. J. Virol. 2006;80(18):9279–9287. doi: 10.1128/JVI.00659-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Cullinan S.B., Zhang D., Hannink M., Arvisais E., Kaufman R.J., Diehl J.A. Nrf2 is a direct PERK substrate and effector of PERK-dependent cell survival. Mol. Cell. Biol. 2003;23(20):7198–7209. doi: 10.1128/MCB.23.20.7198-7209.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Loo Y.-M., Fornek J., Crochet N., Bajwa G., Perwitasari O., Martinez-Sobrido L., et al. Distinct RIG-I and MDA5 signaling by RNA viruses in innate immunity. J. Virol. 2008;82(1):335–345. doi: 10.1128/JVI.01080-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Burdette D.L., Monroe K.M., Sotelo-Troha K., Iwig J.S., Eckert B., Hyodo M., et al. STING is a direct innate immune sensor of cyclic di-GMP. Nature. 2011;478(7370):515–518. doi: 10.1038/nature10429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Sun L., Xing Y., Chen X., Zheng Y., Yang Y., Nichols D.B., et al. Coronavirus papain-like proteases negatively regulate antiviral innate immune response through disruption of STING-mediated signaling. PLoS One. 2012;7(2) doi: 10.1371/journal.pone.0030802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chen X., Yang X., Zheng Y., Yang Y., Xing Y., Chen Z. SARS coronavirus papain-like protease inhibits the type I interferon signaling pathway through interaction with the STING-TRAF3-TBK1 complex. Protein Cell. 2014;5(5):369–381. doi: 10.1007/s13238-014-0026-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Acharya D., Liu G.Q., Gack M.U. Dysregulation of type I interferon responses in COVID-19. Nat. Rev. Immunol. 2020;20(7):397–398. doi: 10.1038/s41577-020-0346-x. [Internet]. Available from: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Olagnier D., Brandtoft A.M., Gunderstofte C., Villadsen N.L., Krapp C., Thielke A.L., et al. Nrf2 negatively regulates STING indicating a link between antiviral sensing and metabolic reprogramming. Nat. Commun. 2018;9(1) doi: 10.1038/s41467-018-05861-7. [Internet]. Available from: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Olagnier D., Lababidi R.R., Hadj S.B., Sze A., Liu Y., Naidu S.D., et al. Activation of Nrf2 signaling augments vesicular stomatitis virus oncolysis via autophagy-driven suppression of antiviral immunity. Mol. Ther. 2017;25(8):1900–1916. doi: 10.1016/j.ymthe.2017.04.022. [Internet]. Available from: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Espinoza J.A., González P.A., Kalergis A.M. Modulation of antiviral immunity by heme oxygenase-1. Am. J. Pathol. 2017 Mar;187(3):487–493. doi: 10.1016/j.ajpath.2016.11.011. [DOI] [PubMed] [Google Scholar]
- 71.Koliaraki V., Kollias G. A new role for myeloid HO-1 in the innate to adaptive crosstalk and immune homeostasis. Adv. Exp. Med. Biol. 2011;780:101–111. doi: 10.1007/978-1-4419-5632-3_9. [DOI] [PubMed] [Google Scholar]
- 72.Bafna K., Krug R.M., Montelione G.T. Structural similarity of SARS-CoV2 Mpro and HCV NS3/4A proteases suggests new approaches for identifying existing drugs useful as COVID-19 therapeutics. ChemRxiv. 2020 Apr the preprint server for chemistry. [Google Scholar]
- 73.Zhu Z., Wilson A.T., Luxon B.A., Brown K.E., Mathahs M.M., Bandyopadhyay S., McCaffrey A.P., Schmidt W.N. Biliverdin inhibits hepatitis C virus nonstructural 3/4A protease activity: mechanism for the antiviral effects of heme oxygenase? Hepatology. 2010 Dec;52(6):1897–1905. doi: 10.1002/hep.23921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Báez-santos Y.M., John S.E.S., Mesecar A.D. The SARS-coronavirus papain-like protease: structure, functionand inhibition by designed antiviral compounds COVID-19 resource centre is hosted on Elsevier Connect, the company’ s public news and information website. Elsevier hereby grants permission t. Antivir. Res. 2015;115(January):21–38. doi: 10.1016/j.antiviral.2014.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Fillebeen C., Rivas-Estilla A.M., Bisaillon M., Ponka P., Muckenthaler M., Hentze M.W., et al. Iron inactivates the RNA polymerase NS5B and suppresses subgenomic replication of hepatitis C virus. J. Biol. Chem. 2005 Mar;280(10):9049–9057. doi: 10.1074/jbc.M412687200. [DOI] [PubMed] [Google Scholar]
- 76.Fillebeen C., Pantopoulos K. Iron inhibits replication of infectious hepatitis C virus in permissive Huh7.5.1 cells. J. Hepatol. 2010 Dec;53(6):995–999. doi: 10.1016/j.jhep.2010.04.044. [DOI] [PubMed] [Google Scholar]
- 77.Gao Y., Yan L., Huang Y., Liu F., Zhao Y., Cao L., et al. Structure of the RNA-dependent RNA polymerase from COVID-19 virus. Science (80-) 2020;368(6492):779–782. doi: 10.1126/science.abb7498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Kalyanaraman H., Schwaerzer G., Ramdani G., Castillo F., Scott B.T., Dillmann W., et al. Protein kinase G activation reverses oxidative stress and restores osteoblast function and bone formation in male mice with type 1 diabetes. Diabetes. 2018;67(4):607–623. doi: 10.2337/db17-0965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Mahase E. Covid-19: demand for dexamethasone surges as RECOVERY trial publishes preprint. BMJ. 2020;369(June) doi: 10.1136/bmj.m2512. [DOI] [PubMed] [Google Scholar]
- 80.Russell B., Moss C., Rigg A., Van Hemelrijck M. COVID-19 and treatment with NSAIDs and corticosteroids: should we be limiting their use in the clinical setting? Ecancermedicalscience. 2020;14:1–3. doi: 10.3332/ecancer.2020.1023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Voiriot G., Philippot Q., Elabbadi A., Elbim C., Chalumeau M., Fartoukh M. Risks related to the use of non-steroidal anti-inflammatory drugs in community-acquired pneumonia in adult and pediatric patients. J. Clin. Med. 2019;8(6):786. doi: 10.3390/jcm8060786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Clavé S., Rousset-Rouvière C., Daniel L., Tsimaratos M. The invisible threat of non-steroidal anti-inflammatory drugs for kidneys. Front. Pediatr. 2019;7(December):1–6. doi: 10.3389/fped.2019.00520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Baigent C., Bhala N., Emberson J., Merhi A., Abramson S., Arber N., et al. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: meta-analyses of individual participant data from randomised trials. Lancet. 2013;382(9894):769–779. doi: 10.1016/S0140-6736(13)60900-9. [Internet]. Available from: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Wen Y.-C., Hsiao F.-Y., Lin Z.-F., Fang C.-C., Shen L.-J. Risk of stroke associated with use of nonsteroidal anti-inflammatory drugs during acute respiratory infection episode. Pharmacoepidemiol. Drug Saf. 2018 Jun;27(6):645–651. doi: 10.1002/pds.4428. [DOI] [PubMed] [Google Scholar]
- 85.Kolilekas L., Loverdos K., Giannakaki S., Vlassi L., Levounets A., Zervas E., et al. Can steroids reverse the severe COVID-19 induced “cytokine storm”? J. Med. Virol. 2020;92(11):2866–2869. doi: 10.1002/jmv.26165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Zhao M. 2020. Since January 2020 Elsevier has Created a COVID-19 Resource Centre with Free Information in English and Mandarin on the Novel Coronavirus COVID-19. The COVID-19 resource centre is hosted on Elsevier Connect, the company’s public news and information website. Elsevier hereby grants permission to make all its COVID-19-related research that is available on the COVID-19 resource centre - including this research content - immediately available in PubMed Central and other publicly funded repositories, such as the WHO COVID database with rights for unrestricted research re-use and analyses in any form or by any means with acknowledgement of the original source. These permissions are granted for free by Elsevier for as long as the COVID-19 resource centre remains active. Cytokine storm and immunomodulatory therapy in COVID-19: Role of chloroquine and anti-IL-6 monoclonal antibodies. (January) [Google Scholar]
- 87.Singh V., Ubaid S. Role of silent information regulator 1 (SIRT1) in regulating oxidative stress and inflammation. Inflammation. 2020;43(5):1589–1598. doi: 10.1007/s10753-020-01242-9. [DOI] [PubMed] [Google Scholar]
- 88.Gregoretti I.V., Lee Y.-M., Goodson H.V. Molecular evolution of the histone deacetylase family: functional implications of phylogenetic analysis. J. Mol. Biol. 2004 Apr;338(1):17–31. doi: 10.1016/j.jmb.2004.02.006. [DOI] [PubMed] [Google Scholar]
- 89.Wager MGT and JFS 基因的改变NIH public access. Bone. 2011;23(1):1–7. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3624763/pdf/nihms412728.pdf [Internet]. Available from: [Google Scholar]
- 90.Deng G.-F., Xu X.-R., Zhang Y., Li D., Gan R.-Y., Li H.-B. Phenolic compounds and bioactivities of pigmented rice. Crit. Rev. Food Sci. Nutr. 2013;53(3):296–306. doi: 10.1080/10408398.2010.529624. [DOI] [PubMed] [Google Scholar]
- 91.Singh C.K., Chhabra G., Ndiaye M.A., Garcia-Peterson L.M., MacK N.J., Ahmad N. The role of Sirtuins in antioxidant and redox signaling. Antioxid. Redox Signal. 2018;28(8):643–661. doi: 10.1089/ars.2017.7290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Gomes P., Fleming Outeiro T., Cavadas C. Emerging role of Sirtuin 2 in the regulation of mammalian metabolism. Trends Pharmacol. Sci. 2015 Nov;36(11):756–768. doi: 10.1016/j.tips.2015.08.001. [DOI] [PubMed] [Google Scholar]
- 93.Imai S., Armstrong C.M., Kaeberlein M., Guarente L. Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature. 2000 Feb;403(6771):795–800. doi: 10.1038/35001622. [DOI] [PubMed] [Google Scholar]
- 94.Du J., Jiang H., Lin H. Investigating the ADP-ribosyltransferase activity of sirtuins with NAD analogues and 32P-NAD. Biochemistry. 2009 Apr;48(13):2878–2890. doi: 10.1021/bi802093g. [DOI] [PubMed] [Google Scholar]
- 95.Peng C., Lu Z., Xie Z., Cheng Z., Chen Y., Tan M., et al. 2011. The First Identification of Lysine Malonylation Substrates and its Regulatory Enzyme *; pp. 1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Blander G., Guarente L. The Sir2 family of protein deacetylases. Annu. Rev. Biochem. 2004;73:417–435. doi: 10.1146/annurev.biochem.73.011303.073651. [DOI] [PubMed] [Google Scholar]
- 97.Burnett C., Valentini S., Cabreiro F., Goss M., Somogyvári M., Piper M.D., Hoddinott M., Sutphin G.L., Leko V., McElwee J.J., Vazquez-Manrique R.P. Absence of effects of Sir2 overexpression on lifespan in C. elegans and Drosophila. nature. 2011 Sep;477(7365):482–485. doi: 10.1038/nature10296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Haigis M.C., Sinclair D.A. Mammalian sirtuins: biological insights and disease relevance. Annu.Rev. Pathol. 2010 Feb;28(5):253–295. doi: 10.1146/annurev.pathol.4.110807.092250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Houtkooper R.H., Pirinen E., Auwerx J. Sirtuins as regulators of metabolism and healthspan. Nat. Rev. Mol. Cell Biol. 2012 Apr;13(4):225–238. doi: 10.1038/nrm3293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Panieri E., Santoro M.M. ROS homeostasis and metabolism: a dangerous liason in cancer cells. Cell Death Dis. 2016;7(6) doi: 10.1038/cddis.2016.105. e2253-12. Available from: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Fontani F. MMPs, ADAMs and their natural inhibitors in inflammatory bowel disease: involvement of oxidative stress. J. Clin. Gastroenterol. Treat. 2017;3(1):1–12. [Google Scholar]
- 102.Yoo C.H., Yeom J.H., Heo J.J., Song E.K., Lee S. Il, Han M.K. Interferon β protects against lethal endotoxic and septic shock through SIRT1 upregulation. Sci. Rep. 2015;4:1–8. doi: 10.1038/srep04220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Miller R., Wentzel A.R., Richards G. COVID-19: NAD+ deficiency may predispose the aged.; obese and type2 diabetics to mortality through its effect on SIRT1 activity. Medical hypotheses. 2020 Nov;144(11004) doi: 10.1016/j.mehy.2020.110044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Maras J.S., Das S., Sharma S., Sukriti S., Kumar J., Vyas A.K., et al. Iron-overload triggers ADAM-17 mediated inflammation in severe alcoholic hepatitis. Sci. Rep. 2018;8(1):1–14. doi: 10.1038/s41598-018-28483-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Budayeva H.G., Rowland E.A., Cristea I.M. Intricate roles of mammalian Sirtuins in defense against viral pathogens. J. Virol. 2016;90(1):5–8. doi: 10.1128/JVI.03220-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Rossi G.A., Sacco O., Capizzi A., Mastromarino P. Can resveratrol-inhaled formulations be considered potential adjunct treatments for COVID-19? Front. Immunol. 2021;12(May):1–11. doi: 10.3389/fimmu.2021.670955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Paraiso I.L., Revel J.S., Stevens J.F. 2020. Since January 2020 Elsevier has Created a COVID-19 Resource Centre with Free Information in English and Mandarin on the Novel Coronavirus COVID- 19. The COVID-19 resource centre is hosted on Elsevier Connect, the company’s public news and information. (January) [Google Scholar]
- 108.Zamora-Ros R., Rabassa M., Cherubini A., Urpí-Sardà M., Bandinelli S., Ferrucci L., et al. High concentrations of a urinary biomarker of polyphenol intake are associated with decreased mortality in older adults. J. Nutr. 2013;143(9):1445–1450. doi: 10.3945/jn.113.177121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Wessels I., Rolles B., Rink L. The potential impact of zinc supplementation on COVID-19 pathogenesis. Front. Immunol. 2020;11(July):1–11. doi: 10.3389/fimmu.2020.01712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Thomas S., Patel D., Bittel B., Wolski K., Wang Q., Kumar A., et al. Effect of high-dose zinc and ascorbic acid supplementation vs usual care on symptom length and reduction among ambulatory patients with SARS-CoV-2 infection: the COVID a to Z randomized clinical trial. JAMA Netw. Open. 2021;4(2):1–10. doi: 10.1001/jamanetworkopen.2021.0369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Kumar J., Barhydt T., Awasthi A., Lithgow G.J., Killilea D.W., Kapahi P. Zinc levels modulate lifespan through multiple longevity pathways in Caenorhabditis elegans. PLoS One. 2016;11(4):1–21. doi: 10.1371/journal.pone.0153513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Hathaway D., Pandav K., Patel M., Riva-Moscoso A., Singh B.M., Patel A., et al. Omega 3 fatty acids and COVID-19: a comprehensive review. Infect Chemother. 2020;52(4):478–495. doi: 10.3947/ic.2020.52.4.478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.de Magalhães J.P., Müller M., Rainger J.E., Steegenga W. Fish oil supplements, longevity and aging. Aging (Albany NY) 2016;8(8):1578–1582. doi: 10.18632/aging.101021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Chen X., Chen C., Fan S., Wu S., Yang F., Fang Z., et al. Omega-3 polyunsaturated fatty acid attenuates the inflammatory response by modulating microglia polarization through SIRT1-mediated deacetylation of the HMGB1/NF-ΚB pathway following experimental traumatic brain injury. J. Neuroinflammation. 2018;15(1):1–15. doi: 10.1186/s12974-018-1151-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Boergeling Y., Ludwig S. Targeting a metabolic pathway to fight the flu. FEBS J. 2017 Jan;284(2):218–221. doi: 10.1111/febs.13997. [DOI] [PubMed] [Google Scholar]
- 116.Shakoor H., Feehan J., Mikkelsen K., Al Dhaheri A.S., Ali H.I., Platat C., Ismail L.C., Stojanovska L., Apostolopoulos V. Be well: A potential role for vitamin B in COVID-19. Maturitas. 2021 Feb;144:108–111. doi: 10.1016/j.maturitas.2020.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Liu B., Li M., Zhou Z., Guan X., Xiang Y. 2020. Since January 2020 Elsevier has Created a COVID-19 Resource Centre with Free Information in English and Mandarin on the Novel Coronavirus COVID-19. The COVID-19 resource centre is hosted on Elsevier Connect, the company’s public news and information. (January) [Google Scholar]
- 118.Mehmel M., Jovanovi N. 2020. Research and Therapeutic Uses; pp. 1–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Yang N.C., Cho Y.H., Lee I. The lifespan extension ability of nicotinic acid depends on whether the intracellular NAD+ level is lower than the sirtuin-saturating concentrations. Int. J. Mol. Sci. 2020;21(1):1–22. doi: 10.3390/ijms21010142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Bagherniya M., Butler A.E., Barreto G.E., Sahebkar A. The effect of fasting or calorie restriction on autophagy induction: a review of the literature. Ageing Res. Rev. 2018;47:183–197. doi: 10.1016/j.arr.2018.08.004. https://www.sciencedirect.com/science/article/pii/S1568163718301478 [Internet]. Available from: [DOI] [PubMed] [Google Scholar]
- 121.Brandhorst S., Choi I.Y., Wei M., Cheng C.W., Sedrakyan S., Navarrete G., et al. A periodic diet that mimics fasting promotes multi-system regeneration, enhanced cognitive performance, and healthspan. Cell Metab. 2015;22(1):86–99. doi: 10.1016/j.cmet.2015.05.012. [Internet] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Hannan A., Rahman A., Rahman S. 2020. Since January 2020 Elsevier has Created a COVID-19 Resource Centre with Free Information in English and Mandarin on the Novel Coronavirus COVID-19. The COVID-19 resource centre is hosted on Elsevier Connect, the company’s public news and information. (January) [Google Scholar]
- 123.Wang Y. Molecular links between caloric restriction and Sir2/SIRT1 activation. Diabetes Metab. J. 2014;38(5):321–329. doi: 10.4093/dmj.2014.38.5.321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Powell K.E., King A.C., Buchner D.M., Campbell W.W., DiPietro L., Erickson K.I., et al. The scientific foundation for the physical activity guidelines for Americans, 2nd edition. J. Phys. Act. Health. 2018 Dec:1–11. doi: 10.1123/jpah.2018-0618. [DOI] [PubMed] [Google Scholar]
- 125.Martin S.A., Pence B.D., Woods J.A. Exercise and respiratory tract viral infections. Exerc. Sport Sci. Rev. 2009;37(4):157–164. doi: 10.1097/JES.0b013e3181b7b57b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Simpson R.J., Kunz H., Agha N., Graff R. Exercise and the regulation of immune functions. Prog. Mol. Biol. Transl. Sci. 2015;135:355–380. doi: 10.1016/bs.pmbts.2015.08.001. [DOI] [PubMed] [Google Scholar]
- 127.Neves P.R.D.S., Tenório T.R.D.S., Lins T.A., Muniz M.T.C., Pithon-Curi T.C., Botero J.P., et al. Acute effects of high- and low-intensity exercise bouts on leukocyte counts. J. Exerc. Sci. Fit. 2015 Jun;13(1):24–28. doi: 10.1016/j.jesf.2014.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Hammami N., Jdidi H., Frih B. COVID-19 pandemic: physical activity as prevention mean. Open Sports Sci. J. 2020;13(1):120–122. [Google Scholar]
- 129.Grabowska W., Sikora E., Bielak-Zmijewska A. Sirtuins, a promising target in slowing down the ageing process. Biogerontology. 2017;18(4):447–476. doi: 10.1007/s10522-017-9685-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Gerhart-Hines Z., Dominy J.E., Blättler S.M., Jedrychowski M.P., Banks A.S., Lim J.-H., et al. The cAMP/PKA pathway rapidly activates SIRT1 to promote fatty acid oxidation independently of changes in NAD+ Mol. Cell. 2011;44(6):851–863. doi: 10.1016/j.molcel.2011.12.005. https://www.sciencedirect.com/science/article/pii/S1097276511009440 [Internet]. Available from: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Ganesh V., Negi G., Sharma S.S., Kumar A. Potential therapeutic effects of the simultaneous targeting of the Nrf2 and NF- κ B pathways in diabetic neuropathy Redox Biology Potential therapeutic effects of the simultaneous targeting of the Nrf2 and NF- κ B pathways in diabetic neuropathy $ Redox Biol. 2014;1(1):394–397. doi: 10.1016/j.redox.2013.07.005. [Internet]. Available from: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Kobayashi A., Kang M., Okawa H., Ohtsuji M., Zenke Y., Chiba T., et al. Oxidative stress sensor Keap1 functions as an adaptor for Cul3-based E3 ligase to regulate proteasomal degradation of Nrf2. Mol. Cell. Biol. 2004;24(16):7130–7139. doi: 10.1128/MCB.24.16.7130-7139.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Nguyen T., Sherratt P.J., Pickett C.B. Regulatory mechanisms controlling gene expression mediated by the antioxidant response element. Annu. Rev. Pharmacol. Toxicol. 2003;43:233–260. doi: 10.1146/annurev.pharmtox.43.100901.140229. [DOI] [PubMed] [Google Scholar]
- 134.Itoh K., Chiba T., Takahashi S., Ishii T., Igarashi K., Katoh Y., et al. An Nrf2/small Maf heterodimer mediates the induction of phase II detoxifying enzyme genes through antioxidant response elements. Biochem. Biophys. Res. Commun. 1997 Jul;236(2):313–322. doi: 10.1006/bbrc.1997.6943. [DOI] [PubMed] [Google Scholar]
- 135.Yan F., Polk D.B. Probiotics and immune health. Curr. Opin. Gastroenterol. 2011 Oct;27(6):496–501. doi: 10.1097/MOG.0b013e32834baa4d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Yang X., Park S.H., Chang H.C., Shapiro J.S., Vassilopoulos A., Sawicki K.T., Chen C., Shang M., Burridge P.W., Epting C.L., Wilsbacher L.D. Sirtuin 2 regulates cellular iron homeostasis via deacetylation of transcription factor NRF2. J. Clin. Investig. 2017 Apr;127(5):1505–1516. doi: 10.1172/JCI88574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Cao W., Hong Y., Chen H., Wu F., Wei X., Ying W. SIRT2 mediates NADH-induced increases in Nrf2, GCL, and glutathione by modulating Akt phosphorylation in PC12 cells. FEBS Letters. England. 2016;590:2241–2255. doi: 10.1002/1873-3468.12236. [DOI] [PubMed] [Google Scholar]
- 138.Xue F., Huang J.-W., Ding P.-Y., Zang H.-G., Kou Z.-J., Li T., et al. Nrf2/antioxidant defense pathway is involved in the neuroprotective effects of Sirt1 against focal cerebral ischemia in rats after hyperbaric oxygen preconditioning. Behav. Brain Res. 2016 Aug;309:1–8. doi: 10.1016/j.bbr.2016.04.045. [Internet]. Available from: [DOI] [PubMed] [Google Scholar]
- 139.Narayanan S.V., Dave K.R., Saul I., Perez-Pinzon M.A. Resveratrol preconditioning protects against cerebral ischemic injury via nuclear erythroid 2–related factor 2. Stroke. 2015 Jun;46(6):1626–1632. doi: 10.1161/STROKEAHA.115.008921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Carcel C., Sato S., Zheng D., Heeley E., Arima H., Yang J., et al. Prognostic significance of hyponatremia in acute intracerebral hemorrhage: pooled analysis of the intensive blood pressure reduction in acute cerebral hemorrhage trial studies. Crit. Care Med. 2016 Jul;44(7):1388–1394. doi: 10.1097/CCM.0000000000001628. [DOI] [PubMed] [Google Scholar]
- 141.Bousquet J., Cristol J.P., Czarlewski W., Anto J.M., Martineau A., Haahtela T., Fonseca S.C., Iaccarino G., Blain H., Fiocchi A., Canonica G.W. Nrf2‐interacting nutrients and COVID‐19: time for research to develop adaptation strategies. Clin. Transl. Allergy. 2020;10(1) doi: 10.1186/s13601-020-00362-7. 58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Rossi G.A., Sacco O., Capizzi A., Mastromarino P. Can resveratrol-inhaled formulations be considered potential adjunct treatments for COVID-19? Front. Immunol. 2021 May 19;(12):1591. doi: 10.3389/fimmu.2021.670955. [DOI] [PMC free article] [PubMed] [Google Scholar]