Abstract
Metabolic syndromes are frequently associated with dementia, suggesting that the dysregulation of energy metabolism can increase the risk of neurodegeneration and cognitive impairment. In addition, growing evidence suggests the link between infections and brain disorders, including Alzheimer’s disease. The immune system and energy metabolism are in an intricate relationship. Infection triggers immune responses, which are accompanied by imbalance in cellular and organismal energy metabolism, while metabolic disorders can lead to immune dysregulation and higher infection susceptibility. In the brain, the activities of brain-resident immune cells, including microglia, are associated with their metabolic signatures, which may be affected by central nervous system (CNS) infection. Conversely, metabolic dysregulation can compromise innate immunity in the brain, leading to enhanced CNS infection susceptibility. Thus, infection and metabolic imbalance can be intertwined to each other in the etiology of brain disorders, including dementia. Insulin and leptin play pivotal roles in the regulation of immunometabolism in the CNS and periphery, and dysfunction of these signaling pathways are associated with cognitive impairment. Meanwhile, infectious complications are often comorbid with diabetes and obesity, which are characterized by insulin resistance and leptin signaling deficiency. Examples include human immunodeficiency virus (HIV) infection and periodontal disease caused by an oral pathogen Porphyromonas gingivalis. This review explores potential interactions between infectious agents and insulin and leptin signaling pathways, and discuss possible mechanisms underlying the relationship between infection, metabolic dysregulation, and brain disorders, particularly focusing on the roles of insulin and leptin.
Keywords: infection, immunometabolism, glia, neuroinflammation, dementia, leptin, insulin
Introduction
Dementia is a general term for debilitating conditions, in which progressive and long-lasting loss of mental ability impairs cognition and simple daily activities. Alzheimer’s disease (AD), the most common form of dementia, is a neurodegenerative disorder characterized by cognitive decline associated with the accumulation of β-amyloid (Aβ) plaques and neurofibrillary tangles in the brain (Ferri et al., 2005). Amyloid cascade hypothesis is a model postulating a linear pathway initiated by Aβ deposition, eventually leading to neuroinflammation and neuronal loss. Although amyloid cascade hypothesis has provided the theoretical framework for the research direction and advanced the knowledge and understanding of AD pathology at molecular levels in the last decades, it remains controversial whether Aβ is the cause of the pathogenesis (De Strooper and Karran, 2016).
While the exact role for Aβ in dementia etiology remains unclear, several elements have been suggested to increase the risk of cognitive decline. Among those suggested are impaired glycemic control [e.g., metabolic syndromes (MetS) including diabetes] and infection (Ott et al., 1999; Peila et al., 2002; Arvanitakis et al., 2004; Biessels et al., 2006; Irie et al., 2008; Biessels and Despa, 2018). Largely due to modern lifestyle and diet (i.e., the lack of exercise and food with high sugar and carbohydrate), overweight, obesity, and associated metabolic disorders are widespread epidemics. Obesity is a common risk factor for many chronic disorders, such as type 2 diabetes (T2DM) and cardiovascular diseases (Haslam and James, 2005). In addition, evidence suggests that metabolic disturbance can cause neurodegenerative disorders, including AD (Whitmer et al., 2005; Kivipelto et al., 2006; Mejido et al., 2020), possibly via blood–brain barrier (BBB) disruption and neuroinflammation (Pugazhenthi et al., 2017). The association may also be compounded and influenced by other factors, such as age (Bos et al., 2017; Deckers et al., 2017; Whitmer et al., 2018), indicating the complexity of mechanisms underlying the link between metabolic disturbances and cognitive decline. Meanwhile, the relationship between infections and dementia, especially late-onset AD, has been repeatedly suggested over the past three decades (Ashraf et al., 2019). The aim of this review is to overview and discuss how these two contributory causes may be intertwined in the etiology of neurodegenerative disorders, particularly focusing on the interaction between infectious agents and two metabolic hormones, namely insulin and leptin, in the brain and periphery.
Metabolic Disturbance and Cognitive Impairment
Insulin and Leptin – Linking Metabolic Syndromes, Infection, and Cognitive Impairment
Energy homeostasis is regulated by intricate interactions between the peripheral organs and central regulatory system in the brain, where insulin and leptin play crucial roles (Boucsein et al., 2021) (Figure 1). Insulin and leptin resistance, as well as dysregulation of related pathways, are associated with obesity and metabolic disorders (Francisco et al., 2019; Gruzdeva et al., 2019) and central nervous system (CNS) dysfunctions (Cereda et al., 2007; Arnold et al., 2018; Kellar and Craft, 2020). Growing evidence suggests that insulin and leptin play significant physiological roles in cognition (Paz-Filho et al., 2008; Morrison, 2009; Gray et al., 2014; Arnold et al., 2018), and these signaling pathways may be promising therapeutic targets to alleviate cognitive impairment accompanied by obesity and MetS (Mejido et al., 2020). On the other hand, obesity, visceral adiposity in particular, is frequently associated with immune dysregulation and infection susceptibility (Hamdy et al., 2006; Conde et al., 2010; Kumari et al., 2019; Obradovic et al., 2021), while infection increases the risk of neurodegeneration and dementia (Heneka et al., 2020; Shinjyo et al., 2021), suggesting the link between MetS, infection, and cognitive impairment.
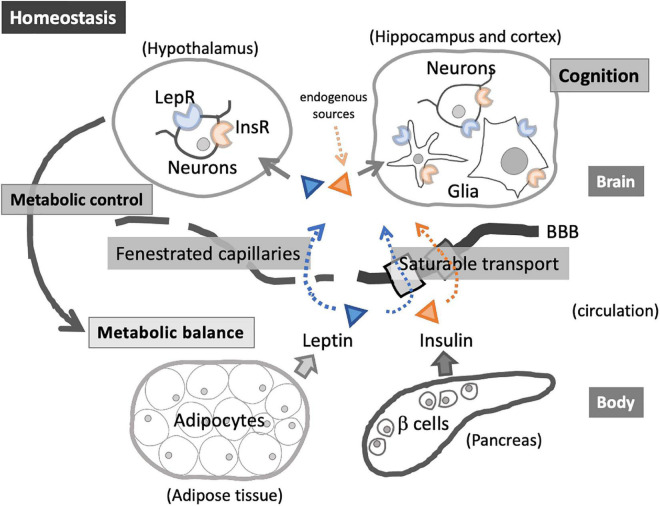
FIGURE 1.
Insulin and leptin in homeostatic interactions between the peripheral organs and central nervous system (CNS). Insulin and leptin enter the brain via saturable transport through blood–brain barrier (BBB) and via fenestrated capillaries in the specific brain regions (e.g., hypothalamus). Acting on InsR and LepR, these hormones exert diverse roles, including energy balance regulation through hypothalamic-pituitary-adrenal axis as well as the modulation of glial immunological activities. InsR and LepR on neuronal cells are involved in cognition.
Insulin
Insulin and Dementia
Insulin is a peptide hormone composed of 51 amino acids, generated from the precursor proinsulin through a series of processing (Rahman et al., 2021). Primarily produced by β cells of the pancreatic islets, insulin is the main anabolic hormone that regulates the energy metabolism throughout the body, i.e., promoting glucose uptake into the liver, fat, and muscle cells (White, 2003). In addition, acting through common receptors (insulin receptor [InsR] and IGF-1 receptor [IGF-1R]), insulin and insulin-like growth factors (IGFs) regulate proliferation and survival of various cell types throughout the body during development, in adulthood, and in aging processes (Nakae et al., 2001; Richardson et al., 2004). InsR and IGF-1R are tyrosine kinase receptors that can be present as homodimers (InsR/InsR, IGF-1R/IGF-1R) or heterodimer (InsR/IGF-1R), sharing the majority of downstream pathways, namely phosphoinositide 3-kinase (PI3K), serine threonine kinase Akt, glycogen synthase kinase 3β (GSK3β), and mammalian target of rapamycin (mTOR) (O’Neill et al., 2012). InsR and IGF-1R are highly expressed in the CNS, including the hippocampus and hypothalamus, and central insulin regulates peripheral energy metabolism (O’Neill et al., 2012; Tiedemann et al., 2017; Barrios et al., 2021; Scherer et al., 2021). Furthermore, it is now evident that insulin and IGF-1 play important roles in higher-order brain functions, such as memory and cognitive processing, and in neuroprotection in response to brain injury (Stewart and Rotwein, 1996; Gerozissis, 2003; Fernandez and Torres-Aleman, 2012), indicating pleiotropic roles for insulin/IGF signaling in the CNS.
Insulin crosses the BBB using a saturable transporter, which is regulated by insulin itself and altered by a number of factors including hyperglycemia and diabetes (Banks et al., 2012). In addition, the choroid plexus, a highly vascularized tissue responsible for the production of cerebrospinal fluid (CSF) at the interface of the CNS and periphery, releases insulin and IGFs (Salehi et al., 2009; Ziegler et al., 2012; Mazucanti et al., 2019; Dani et al., 2021). In the parenchyma, microglia generate IGF-1 during development (Wlodarczyk et al., 2017) as well as in adulthood (Myhre et al., 2019), while astrocytes can produce insulin, which is negatively regulated by Aβ and bacterial lipopolysaccharide (LPS) (Takano et al., 2018). InsR and IGF-1R are expressed on neurons and non-neuronal cells (Shaughness et al., 2020), and are involved in the regulation of synaptic plasticity (Dyer et al., 2016), astroglial energy metabolism, microglial inflammatory phenotypes (Haas et al., 2020), as well as the self-renewal and maintenance of neural stem cells (Ziegler et al., 2015), indicating crucial roles of insulin through multiple targets and mechanisms in the brain parenchyma (Gabbouj et al., 2019). Although the hypothalamus is the most well-studied target region regulating systemic energy metabolism (Mitchell and Begg, 2021), specific inactivation of InsR and IGF-1R in the hippocampus and amygdala led to increased anxiety and cognitive impairment in animal models (Soto et al., 2019), indicating the significance of extrahypothalamic and non-metabolic roles for insulin. Moreover, hippocampal microglia responded to insulin treatment in young rats but not in the aged group (Haas et al., 2020), suggesting that microglia develop insulin resistance during aging. Indeed, experimental evidence suggests the link between impaired insulin signaling in the CNS and cognitive impairment. Intracerebral streptozotocin (STZ) induces AD-like brain pathology in mice, which has been widely used as a model for sporadic AD (Lester-Coll et al., 2006). In mixed AD and diabetic mice using APPswe/PS1dE9 (APP/PS1) transgenic AD model or triple-transgenic model of AD (3xTg-AD) with either STZ treatment or InsR deficiency (db/db), genetic background and dysfunctional insulin signaling cooperatively exacerbated CNS inflammation and AD pathology (Hierro-Bujalance et al., 2020; Imamura et al., 2020; Sankar et al., 2020). In addition, intranasal insulin treatment can alleviate AD pathogenesis and cognitive impairment via reduced neuroinflammation and enhanced neural plasticity (Chen Y. et al., 2014; Guo et al., 2017), possibly via the actions through receptors on microglia and astrocytes (Spielman et al., 2015). Although what roles insulin/IGF signaling play in the CNS could be context-dependent, i.e., either beneficial (Carro et al., 2003; Sukhanov et al., 2007; Tien et al., 2017) or damaging (Labandeira-Garcia et al., 2017), fine-tuning of these signaling pathways is crucial to maintaining brain functions. Considering the anti-inflammatory effects of insulin and IGF-1 on microglia (Labandeira-Garcia et al., 2017; Shaughness et al., 2020) and observations that age-related changes of microglia toward pro-inflammatory phenotypes are implicated in brain aging and neurodegenerative disorders (Gemma et al., 2010; Chowen and Garcia-Segura, 2020), microglial insulin/IGF resistance may underlie chronic inflammation in the brain, which is associated with dementia (Lutshumba et al., 2021). Consequently, dysregulated insulin/IGF signaling and insulin resistance in the CNS have been linked to an increased risk of dementia, including AD, which is sometimes referred to as type 3 diabetes (Biessels et al., 2006; Whitmer et al., 2008; Zemva and Schubert, 2011; Banks et al., 2012; Smolina et al., 2015; Biessels and Despa, 2018; Kuo et al., 2018; Shinjyo et al., 2020).
Insulin as an Immunomodulator – A Possible Link Between Metabolic Syndromes and Infection Susceptibility
Acute and chronic infections induce insulin resistance (Yki-Järvinen et al., 1989; Fernández-Real et al., 2006), whereas metabolic imbalance (e.g., higher body fat mass and diabetes) is associated with higher infection susceptibility (Rayfield et al., 1982; Fernández-Real et al., 2007), indicating bidirectional interactions between the immune system and energy metabolism in human body. Chronic low-grade inflammation and immune dysregulation likely mediate the mutual and possibly synergetic relationship. The strong interaction between the immune and metabolic pathways is apparently rooted in their common evolutionary origin, as represented by the fat body in Drosophila, which senses both infectious and metabolic stresses and perform the functions of the liver, adipose tissue, and immune system (Hotamisligil, 2017). Insulin plays a key role in such evolutionarily conserved immunometabolism, partly through the interactions with tumor necrosis factor (TNF) receptor (Uysal et al., 1997) and Toll-like receptor (TLR) pathways (DiAngelo et al., 2009; Hotamisligil, 2017), as well as through modulating metabolic pathways in immune cells (van Niekerk et al., 2020). Through metabolic regulation, insulin alleviates the harmful effects of hyperglycemia (Sun et al., 2014). For example, due to its glucose-lowering effect, insulin exert anti-inflammatory effects through modulating the release of inflammatory mediators. As glucose is pro-inflammatory, insulin deficiency activates inflammatory reactions in the body, leading to the release of inflammatory mediators including reactive oxygen species and pro-inflammatory cytokines from leukocytes (Mohanty et al., 2000; Esposito et al., 2002). In addition, insulin directly activates phagocytic and bactericidal activity of immune cells and diabetes-induced infection susceptibility is partly mediated by impaired immune responses due to the lack of insulin signaling, as shown in rodent studies (Yano et al., 2012).
Leptin
Leptin as a Metabolic Hormone
Leptin is a 16 kDa polypeptide that regulates metabolic balance and fat storage. Mainly produced by the white adipose tissue (WAT), leptin acts via leptin receptor (LepR) in the brain and plays a pivotal role in the control of appetite and energy expenditure. There are six LepRs (LepRa to LepRf), with identical extracellular N-terminal domain and distinct intracellular C-terminal regions generated by alternative splicing of db (Lee et al., 1996). LepRb, the long isoform with high ligand affinity, is the major isoform expressed in the brain and activates intracellular pathways, including JAK/STAT, ERK/MAPK, and IRS/PI3K (Allison and Myers, 2014). The hypothalamic nuclei, the regulatory center of energy homeostasis, are highly enriched with LepRb (Elmquist et al., 1998; Balthasar et al., 2004; Leshan et al., 2009), and leptin exerts its effects through the action in the hypothalamus to regulate food intake and energy metabolism (Friedman, 2019). Leptin enters the hypothalamus through fenestrated capillary and acts on LepR expressed on neurons in arcuate nucleus (ARC), dorsomedial hypothalamus (DMH), and ventromedial hypothalamus (VMH), enabling a feedback mechanism to maintain energy balance, thereby preventing obesity and metabolic disorders (Pandit et al., 2017). In addition, leptin can cross BBB using a saturable transport system (Banks and Farrell, 2003), as well as the blood-CSF barrier (choroid plexus epithelia) (Merino et al., 2006; Dietrich et al., 2008). LepRb in extrahypothalamic brain regions also plays significant roles in the regulation of energy metabolism (Scott et al., 2009). For example, LepRb in the ventral tegmental area (VTA) regulates energy balance via mesolimbic dopaminergic system (Fulton et al., 2006; Hommel et al., 2006), suggesting that leptin targets multiple brain regions and cellular components. Consequently, dysregulation of leptin signaling results in obesity, diabetes, and associated comorbidities (Myers et al., 2008; Wasim et al., 2016; Fischer et al., 2020). Leptin resistance is the condition where diminished leptin sensitivity occurs, resulting in a defect in satiety detection despite high leptin levels, which has been linked to obesity (Izquierdo et al., 2019). Leptin deficient (ob/ob) and LepR deficient (db/db) mice, carrying mutations in leptin (ob) and LepR (db) genes, respectively, exhibit excessive eating, develop obesity and diabetes, and are widely used as animal models of T2DM (Chen et al., 1996; Ninomiya et al., 2002; Gautron and Elmquist, 2011). Growing evidence suggests the contribution of leptin resistance to neurodegeneration in AD (Bonda et al., 2014). Serum leptin levels showed negative correlation with cognitive decline in the elderly (Holden et al., 2009). In the brain autopsy, CSF leptin levels were significantly higher in AD compared to control and mild cognitive impairment cases, and CSF leptin concentration was correlated with pathological neurofibrillary tangle burden (Bonda et al., 2014), suggesting that leptin resistance develops during AD progression.
Leptin as an Adipokine
Adipokines are the adipose tissue-derived factors that affect whole body homeostasis in autocrine and paracrine manners, targeting a number of biological processes such as glucose metabolism, lipid metabolism, insulin sensitivity, as well as immune response (Fasshauer and Blüher, 2015). Adipokines include leptin, adiponectin, vaspin, fibroblast growth factor 21 (FGF21), and many more, each exerting specific biological effects, and mediate diverse actions throughout the body (Fasshauer and Blüher, 2015). Originally identified as an adipocyte-derived hormone that regulates neuroendocrine axis, leptin is one of the most studied adipokines linking the immune system and energy metabolism (Abella et al., 2017; Jiménez-Cortegana et al., 2021).
Indeed, leptin belongs to the family of long-chain helical cytokines and has similarity to IL-6, IL-12, and granulocyte colony-stimulating factor (G-CSF). LepR is a type I cytokine receptor (La Cava and Matarese, 2004). Although neurons are the most well-established cellular targets of leptin, LepR is also expressed by non-neuronal cells throughout the body. Importantly, most immune cells, including hematopoietic bone-marrow precursors, monocytes/macrophages, and lymphocytes, express LepR, suggesting that leptin directly modulate immune responses and inflammation (La Cava and Matarese, 2004; Procaccini et al., 2012, 2017). Adipose tissue and lymphoid organs are often anatomically associated, and the contiguity between adipocytes and lymphoid cells supports the functional interactions (Matarese et al., 2002). For example, leptin affects thymic function and growth/survival of bone-marrow CD34+ precursors and CD4+ T cells (Lord et al., 1998; Howard et al., 1999; Martín-Romero and Sánchez-Margalet, 2001; Papathanassoglou et al., 2006; Cohen et al., 2017), and modulates both innate and adaptive immunity through diverse mechanisms (Maurya et al., 2018), such as enhancement of neutrophil oxidative burst (Mancuso et al., 2002; Caldefie-Chezet et al., 2003), phagocytosis by monocytes/macrophages (Faggioni et al., 1999; Fantuzzi and Faggioni, 2000; Sánchez-Margalet et al., 2003), and cytotoxic activity of natural killer (NK) cells (Tian et al., 2002), as well as mobilization of macrophages, lymphocytes (Abella et al., 2017), and neutrophils (Souza-Almeida et al., 2018).
Leptin deficiency and resistance are associated with increased susceptibility to infectious diseases, including bacterial (e.g., Mycobacterium tuberculosis and Streptococcus pneumonia), viral [e.g., coronaviruses, influenza A virus, and human immunodeficiency virus (HIV)], and parasitic (e.g., Trypanosoma brucei, Trypanosoma cruzi, and Entamoeba histolytica) infections (Amole et al., 1985; Sánchez-Pozo et al., 2003; Wieland et al., 2005; Hsu et al., 2007; Nagajyothi et al., 2010; Tschöp et al., 2010; Vedantama and Viswanathan, 2012; Zhang et al., 2013; Radigan et al., 2014; Alti et al., 2018; Guglielmi et al., 2021), while infection with certain pathogens, such as Plasmodium spp. and Toxoplasma gondii, can cause dysregulated leptin secretion, independently of adiposity (Pulido-Mendez et al., 2002; Baltaci and Mogulkoc, 2012). The significant association between obesity-induced inflammation (meta-inflammation) and the severity of infectious disease has been highlighted by COVID-19 pandemic (Huizinga et al., 2020; Rebello et al., 2020). It is notable that Streptococcus pneumoniae is a major cause of meningitis, which potentially leads to persistent cognitive disability (Yau et al., 2018), and M. tuberculosis can disseminate into the brain and induce CNS tuberculosis, a cascade of inflammatory responses that can potentially cause brain damage (Leonard, 2017; Davis et al., 2019). HIV-positive individuals frequently suffer neurocognitive disorders (HIV-associated neurocognitive disorders, HAND), and influenza A virus (H1N1) have been associated with neurological manifestation in both young and adult patients (Cárdenas et al., 2014; Wilking et al., 2014), with some developing permanent sequelae (Cárdenas et al., 2014), suggesting that defective leptin signaling could affect brain functions through increased infection susceptibility. While the mechanistic link between leptin resistance and infection susceptibility is complex and multifactorial (Maurya et al., 2018; Rebello et al., 2020), leptin’s actions through immune cells in the periphery and CNS, and dysregulation thereof, likely play a significant role.
The Roles for Leptin and Insulin in Cellular Immunometabolism
Cellular energy metabolism is mainly driven by glycolysis, tricarboxylic acid (TCA) cycle, fatty acid oxidation, and oxidative phosphorylation (OXPHOS). Glutaminolysis, the conversion of glutamine to glutamate, is activated to fuel TCA cycle when glucose availability is limited. Glutaminolysis also plays a crucial role in the brain, where glutamate functions as a major neurotransmitter (Wang et al., 2017). In addition, in the absence of glucose, such as insulin-induced hypoglycemia, microglia utilize glutamine as an alternative fuel to support their immunological functions (Bernier et al., 2020). Upon stimulation by pathogen-derived molecules and endogenous ligands, immune cells undergo metabolic reprogramming into alternative modes of energy metabolism (O’Neill et al., 2016; Gaber et al., 2017; Hotamisligil, 2017; Próchnicki and Latz, 2017), which can be largely classified into the pro-inflammatory phenotypes dominated by glycolysis (similar to the Warburg effect in cancer cells), and anti-inflammatory phenotypes characterized by TCA cycle, fatty acid oxidation, and OXPHOS (O’Neill et al., 2016). Such bioenergetic shifts determine the properties of various immune cell populations, including macrophages (Van den Bossche et al., 2017), neutrophils (Curi et al., 2020), and T cells (O’Neill et al., 2016; Spadaro et al., 2017; Balyan et al., 2020), where insulin/IGFs and leptin come into play. For example, insulin regulates T cell’s metabolic reprogramming, thereby shaping adaptive immunity (Tsai et al., 2018). InsR-deficient T cells showed compromised responses to antigens in vitro, and T cell-specific InsR knockout in mice led to reduced antigen-specific immunity to influenza virus infection in vivo (Tsai et al., 2018), suggesting that InsR signaling reinforces metabolic reprogramming required for T cell activation. Such immunometabolic changes of T cells are likely mediated by mTOR, a key regulator of cellular homeostasis including protein synthesis and autophagy (Rao et al., 2010; Chi, 2012; Martin et al., 2021). mTOR also mediates insulin-induced alteration of metabolic rates and immune responses in myeloid cells (Ratter et al., 2021), and mTOR mediates age-associated microglial priming and neurodegeneration (Keane et al., 2021), suggesting that insulin-induced activation of mTOR pathway plays a role in immunometabolic imbalance in the CNS. IGF-1R signaling is essential for the anti-inflammatory polarization of macrophages upon metabolic stress as well as helminth infection (Labandeira-Garcia et al., 2017; Spadaro et al., 2017), and IGF-2 instructs macrophage precursor cells to become anti-inflammatory through metabolic pre-programming toward OXPHOS (Du et al., 2019). Leptin also induces immunometabolic changes in immune cells, including macrophages and T cells (Cohen et al., 2017; Boutens et al., 2018; Monteiro et al., 2019). Activation of leptin signaling pathways (JAK/STAT and IRS/PI3K) leads to intracellular metabolic changes, such as increased glucose uptake and glycolytic activity and reduced OXPHOS, associated with pro-inflammatory phenotype of macrophages and T cells (Cohen et al., 2017; Boutens et al., 2018; Monteiro et al., 2019). It is conceivable that immunometabolic imbalance due to dysregulated insulin/IGF and leptin signaling pathways underlie immunodeficiency and infection susceptibility in nutritional imbalance, including malnutrition and obesity.
Brain-Resident Immune Cells and Immunometabolism
Microglia and Astrocytes
The brain is the most energy-demanding organ, consuming glucose at a disproportionately high rate compared to the rest of the body. Glucose metabolism in the brain is founded on intricate interactions between the cellular components, i.e., neurons and glia. Neurons are highly aerobic and heavily dependent on OXPHOS, while astrocytes and oligodendrocytes, the major neuroglial populations, are predominantly glycolytic. These cell-type specific metabolic profiles support the overall power system in the CNS: lactate generation via glycolysis in neuroglia fueling OXPHOS in neurons (Fünfschilling et al., 2012; Jha and Morrison, 2018; Rosko et al., 2019) (Figure 2). To protect such elaborate systems, the brain harbors resident innate immune cells, namely resident macrophage populations: microglia, perivascular macrophages, meningeal macrophages, and choroid plexus macrophages. The former three, including microglia, have embryonic origins and are maintained through local self-renewal while choroid plexus macrophages are replenished via constant supply of bone marrow-derived monocytes (Prinz et al., 2017). These brain-resident macrophage populations play significant roles in the brain physiology, as well as in defense against infection (Ransohoff and Brown, 2012; Heneka et al., 2015; Herz et al., 2017; Kierdorf and Prinz, 2017).
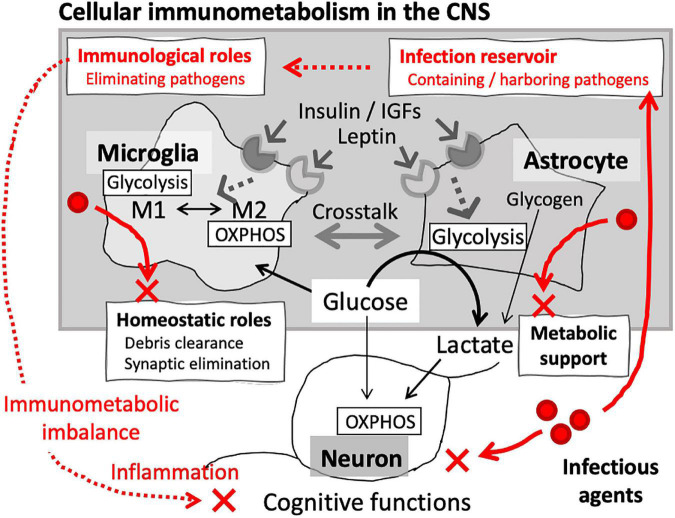
FIGURE 2.
Cellular immunometabolism in the brain. Glia are the main cellular components of immunometabolism in the CNS. Microglial immunological phenotypes are closely associated with their metabolic signature: glycolysis-dominant proinflammatory (M1) and OXPHOS-dominant anti-inflammatory (M2) phenotypes. In addition to the role as resident innate immune cells, microglia also play homeostatic roles, such as eliminating unwanted synapses and debris. Astrocytes are the major metabolic component of CNS, providing bioenergetic support for the neuronal networks. Microglia and astrocytes express InsR and LepR, through which their metabolic signatures and immunological phenotypes are regulated. Crosstalk between microglia and astrocytes constitutes cellular immunometabolism in the CNS. Infection can disrupt the immunometabolic homeostasis, which may lead to neuronal damages and cognitive impairment.
While aberrant activation of resident macrophages contributes to CNS pathology via inflammation (De Strooper and Karran, 2016; Herz et al., 2017), evidence is growing that brain-resident macrophages play homeostatic roles in the brain. Residing in the brain parenchyma, microglia play pivotal roles in the development and maintenance of neuronal networks. The most notable roles of microglia include complement-dependent synaptic pruning during CNS development (Schafer et al., 2012) and synaptic reorganization throughout life (Herz et al., 2017). In addition, microglia-derived neurotropic factors, such as brain-derived neurotrophic factor (BDNF), promote learning-dependent synaptic formation (Parkhurst et al., 2013) and protect neurons from brain injuries (Madinier et al., 2009). Furthermore, CNS macrophages are the major component of glymphatic system, which plays a crucial role in the clearance of waste products and toxic materials, including Aβ (Gordleeva et al., 2020). In the context of AD pathogenesis, microglial activation is a double-edged sword; it can facilitate the clearance of Aβ and tau, while potentially inducing neuroinflammation leading to neuronal damages associated with AD (Lue et al., 2010; Leyns and Holtzman, 2017; Shippy and Ulland, 2020).
Meanwhile, growing evidence suggests that astrocytes are part of the innate immunity in the brain (Sofroniew, 2020). Responding to the changing microenvironment and diverse stimuli (e.g., microbial infections, exposure to environmental toxins, tumor formation, and neurodegenerative diseases), astrocytes undergo morphological and functional changes and influence the outcome of a number of brain disorders (Drögemüller et al., 2008; Soung and Klein, 2018; Katsouri et al., 2020; Sofroniew, 2020). In addition, crosstalk between astrocytes and microglia in the hippocampus is responsible for sensitivity to insults (Lana et al., 2020), pointing to the importance of communication between astroglia and brain-resident macrophages in cognitive dysfunctions (Liddelow et al., 2017; Linnerbauer et al., 2020). Furthermore, astrocytes and microglia participate in Aβ clearance and protection of synaptic connectivity in AD models, suggesting highly context-dependent immunological roles of these glial cell populations against AD development (Davis et al., 2021).
Glial Cells and Central Nervous System Immunometabolism
Microglia and astrocytes, also called macroglia, are major resident non-neuronal cells in the brain. As described above, evidence suggests that the interplay between neurons and these glial population plays pivotal roles in the brain, including the hippocampus, in physiological and pathological conditions (Lana et al., 2021). While neuronal cells express InsR/IGF-1R at high levels and many studies have focused on the roles of neuronal InsR/IGF-1R (Frölich et al., 1998), microglia and astrocytes also express InsR and IGF-1R (Shaughness et al., 2020). Insulin and IGFs support brain functions through regulating astroglial glucose metabolism (Sonnewald et al., 1996; Fernandez et al., 2018). InsR signaling modulates astroglial glucose uptake and bioenergetics (Heni et al., 2011), and specific ablation of astroglial InsR led to altered mood and cognition in mice (González-García et al., 2021). In addition, insulin can modulate inflammatory responses of astrocytes (Spielman et al., 2015), suggesting the immunometabolic regulation of astrocytes by insulin. IGF-1R can also modulate astroglial metabolic and immunological signatures (Hernandez-Garzón et al., 2016), and astroglial IGF-1R signaling mediates synaptic plasticity of cortical inhibitory neurons (Noriega-Prieto et al., 2021). These data suggest that impairment in InsR/IGF-1R signaling pathways may disrupt the metabolic network between glia and neurons, as well as immunological roles of astrocytes, eventually leading CNS dysfunctions. Microglial InsR/IGF-1R also mediate the effects of insulin. Low-dose insulin exerted pro-inflammatory effects on microglial cells in vitro (Spielman et al., 2015), while it was anti-inflammatory at a higher concentration (Brabazon et al., 2018), suggesting the variable roles of microglial InsR/IGF-1R signaling. In addition, microglia are the major source of IGF-1 in the brain, and microglial IGF-1 was found to be increased in AD model mice compared to wild-type (Myhre et al., 2019). These data suggest that insulin and IGFs play crucial roles in immunometabolism via the phenotypic regulation of glial cells, potentially mediating the link between bioenergetics and immunity in the brain.
Microglia (Tang et al., 2007; André et al., 2017; Fujita and Yamashita, 2019), and astrocytes (Naranjo et al., 2020; Pratap and Holsinger, 2020) express LepR. Leptin can enhance microglial pro-inflammatory responses, including IL-6 production through a mechanism involving insulin receptor substrate-1 (IRS-1), PI3K, and Akt (Tang et al., 2007; André et al., 2017; Fujita and Yamashita, 2019), IL-1β release via a caspase 1-independent mechanism (Pinteaux et al., 2007), and lipopolysaccharide (LPS)-induced pro-inflammatory responses (Lafrance et al., 2010). On the other hand, in a spinal cord injury model, leptin reduced microglial inflammatory responses, while inducing neuroprotective phenotypes, Fernández-Martos et al. (2012). In myeloid cell-specific LepR deficient mice, hypothalamic microglia exhibited less ramified morphology and impaired phagocytic capacity (Gao et al., 2018), suggesting that leptin directly regulates homeostatic microglial phenotypes (Davis et al., 2017). Considering multifaceted physiological roles of microglia and potential harm (i.e., neuroinflammation) caused by their aberrant activation (Colonna and Butovsky, 2017; Li and Barres, 2018), it is conceivable that impaired leptin signaling in microglia could lead to significant homeostatic imbalance in the CNS. On the other hand, specific LepR depletion in astrocytes (GFAP-LepR–/–) resulted in impaired neurotransmission in the hippocampus, suggesting that leptin regulates hippocampal plasticity via astroglial LepR, possibly by regulating the glucose and glutamate up-take capacity (Naranjo et al., 2020).
Other Central Nervous System-Resident Immune Cells
In addition to resident macrophages, the CNS harbors multiple leukocyte populations, including dendritic cells (DCs) and T lymphocytes. As a professional antigen-presenting cells, DCs play an essential role in the regulation of adaptive immunity. Although initially believed to be absent in the healthy brain parenchyma, only appearing in response to aging, injury, and infections (Fischer and Reichmann, 2001), studies have shown the presence of DCs within the healthy steady-state brain (Bulloch et al., 2008; Prodinger et al., 2011). Upon activation, brain DCs can migrate out of the brain and induce T-cell homing into the CNS where antigen-specific immune responses may take place (Karman et al., 2004). Correlation between aging and the accumulation of DCs in the CNS (Kaunzner et al., 2012) suggests potential involvement of brain DCs in immunological changes and inflammation associated with aging. In response to immunological stimuli, such as viral and parasitic infections, T cells infiltrate into the brain, where these cells play beneficial roles by eliminating pathogens. In addition, studies have shown that long-lived memory T cells are established after CNS infections (Wakim et al., 2012; Landrith et al., 2017; Mockus et al., 2018), and these tissue-resident memory CD8 cells can provide frontline defense against re-infection (Netherby-Winslow et al., 2021). However, persistence of these cells can also cause neuronal damages (Ai and Klein, 2020; Ghazanfari et al., 2021).
Although it is unknown how these CNS resident immune cells respond to and regulated by leptin and insulin, InsR is expressed on the surface of activated T cells (Helderman et al., 1978). Acting through InsR, insulin enhances and maintains T cell functions after immunological challenges (Helderman, 1984) and potentially induces anti-inflammatory polarization in the periphery (Viardot et al., 2007). These data suggest that insulin may play an immunomodulatory role via actions on T cell populations in the brain.
Glia as the Target and Reservoir of Infectious Agents
Growing evidence supports the infectious etiology of dementia, and a number of causative agents have been proposed, including viruses (e.g., Herpes simplex virus type 1 (HSV-1), cytomegalovirus (CMV)] and bacteria (e.g., Chlamydophila pneumoniae, spirochetes, Helicobacter pylori, Porphyromonas gingivalis) (Harris and Harris, 2015; Sochocka et al., 2017; Nazareth et al., 2021). Suggested mechanisms include persistent inflammation caused by chronic infection, leading to a vicious cycle of neuroinflammation and neurodegeneration, as well as BBB disruption, which may further enhance the entry of infectious agents and proinflammatory mediators from the periphery (Sochocka et al., 2017) (Figure 3).
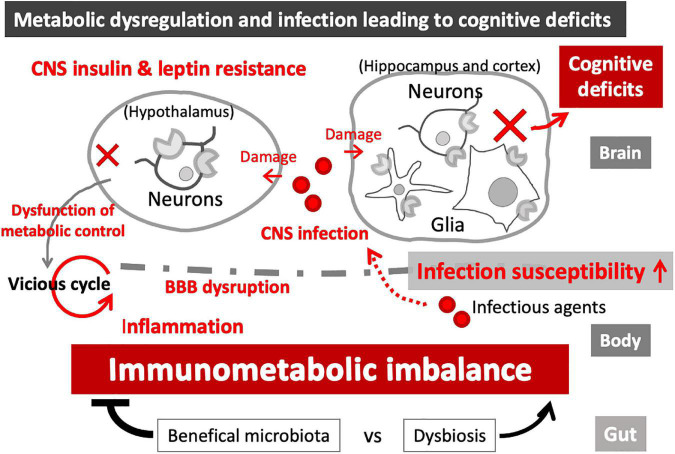
FIGURE 3.
Immunometabolic imbalance in the CNS. Leptin and insulin resistance, associated with metabolic disease, can cause immune dysfunctions and increased infection susceptibility. Metabolic imbalance also causes BBB disruption, leading to neuroinflammation and loss of CNS homeostasis, eventually resulting in functional damages to the affected brain regions. While hypothalamic damages can magnify immunometabolic imbalance in the periphery (vicious cycle), hippocampal and cortical damages can cause cognitive impairment. CNS infection-induced brain damages can further enhance the immunometabolic imbalance and cognitive deficits, as part of the vicious cycle.
Some infectious agents can enter and persistently inhabit the brain, directly causing chronic activation of resident immune cells (Sochocka et al., 2017). For example, Chlamydia pneumoniae, obligate intracellular bacteria, are frequently found in microglia and astrocytes in the brain of AD patients (Balin et al., 1998; Nazareth et al., 2021). Microglia and astrocytes are also the target of some viruses, such as CMV (Cheeran et al., 2001) and HIV (Avalos et al., 2017; Valdebenito et al., 2021). In addition, T. gondii, a neurotropic protozoan parasite causing a lifelong CNS infection, has been implicated in various neurological disorders, including dementia (Nayeri Chegeni et al., 2019). T. gondii is an obligate intracellular parasite, potentially infecting all nucleated cell types. Chronic Toxoplasma infection is linked to microglial activation and persistent neuroinflammation (Li et al., 2019), and CNS energy metabolism is significantly skewed toward glycolysis upon infection (Hargrave et al., 2019), suggesting that T. gondii interferes with host immunometabolism in the brain. Indeed, it has been shown that Toxoplasma infection may lead to reduced IGF-1 signaling in the brain (El Saftawy et al., 2020). On the other hand, In vitro, high glucose and up-regulation of glycolysis in the host cell had a significant impact on the life-stage conversion of T. gondii (i.e., from fast-growing tachyzoite stage into dormant bradyzoite stage) (Weilhammer et al., 2012), indicating that host cell metabolism could modulate the virulence of intracellular pathogens. Intriguingly, T. gondii infection can enhance Aβ clearance by increasing phagocytic activity of recruited monocytes (Möhle et al., 2016), suggesting that the ultimate outcome may depend on the extent and the aggressiveness of the infection. Of note, SARS-CoV-2 is frequently found in astroglia and microglia, rather than neurons, in the autopsy brain, indicating potential involvement of glial activation in the neurological manifestation of COVID-19 (Solomon, 2021). These data suggest that infectious agents could produce immunometabolic imbalance in the CNS via affecting microglial and astroglial phenotypes (Figure 2), which may lead to chronic inflammation and an increased risk of neurodegenerative disorders (Stephenson et al., 2018).
Insulin and Leptin Mediating the Link Between Infection and Neurodegenerative Disorders
Infectious Etiology of Dementia
Recently, it has been shown that Aβ, a hallmark of AD, is an antimicrobial peptide, part of innate immunity to protect the host from various infectious agents (Soscia et al., 2010; Kumar et al., 2016; Gosztyla et al., 2018), suggesting the necessity to revisit the role of amyloid plaque formation in AD pathogenesis. Above mentioned neurotropic viruses, such as HSV-1, HSV-2 and CMV, certain bacterial species, including spirochetes and P. gingivalis, fungi (e.g., Candida albicans), and neurotropic parasite (e.g., T. gondii), may cause disruption of brain functions via chronic inflammation and immune dysregulation in the brain (Sochocka et al., 2017; Shinjyo et al., 2021). The antimicrobial hypothesis for AD proposes that Aβ generation and amyloid plaque formation are not the major culprit in AD pathogenesis, but rather a defense against such infectious agents (Moir et al., 2018; Iqbal et al., 2020; Fulop et al., 2021).
While metabolic disorders and infection may independently increase the risk of dementia, obesity and metabolic disorders may indirectly increase the risk of infection-induced neurological disorders, by enhanced susceptibility to infection and infection-induced complications via immune dysregulation (Shah and Hux, 2003; Muller et al., 2005; Knapp, 2013; Tamara and Tahapary, 2020). Conversely, obesity can occur as a result of infection (Pasarica and Dhurandhar, 2007; Dhurandhar, 2011), and infection-induced chronic low-grade inflammation may lead to insulin resistance (Fernández-Real et al., 2006), suggesting bi-directional interactions between infection and metabolic disorders. Furthermore, several pathogens, such as HSV, CMV, H. pylori, and P. gingivalis, are implicated in both metabolic disorders and AD (Jeon et al., 2012; Harris and Harris, 2015; Fleck-Derderian et al., 2017; Beydoun et al., 2018; Lövheim et al., 2018; Mei et al., 2020; Baradaran et al., 2021; Costa et al., 2021; Salem et al., 2021), supporting the potential mechanistic link between infection, MetS, and dementia etiology.
Metabolic Imbalance Causing Central Nervous System Infection Susceptibility
Studies showing possible interactions between metabolic imbalance and infection in dementia etiology, particularly focusing on data highlighting the roles for insulin and leptin, are listed in Table 1.
TABLE 1.
The roles of insulin and leptin in central nervous system (CNS) infection.
| Pathogen/host | Study design and outcome measures | Findings | Proposed direction of causality and/or mechanism | References |
| Borna disease virus (BDV) | ||||
| BDV/mouse | Animal model: virus-induced obesity model using intracerebral injection of BDV-1 in rats. Outcome measures: neuropathology including inflammatory infiltrates and neurodegeneration in the hypothalamus, hippocampal shrinkage, and leptin resistance | BDV infection-induced obesity was associated with: 1. hypothalamic inflammation. 2. Hippocampal involution and microglial activation in the neocortex. 3. Preferential infection of glutamatergic sites, while sparing GABAergic areas, causing (anabolic appetite-stimulating) GABAergic predominance and fat accumulation 4. Leptin resistance in the brain. | CNS infection → Hypothalamic inflammation, neurotransmitter imbalance (GABA predominance), obesity, and leptin resistance. | Gosztonyi et al., 2020 |
| BDV/rat | Animal model: BDV infection models using two different strains: BDV-ob (obesity-inducing) and BDV-bi (no obesity-inducing effect). Outcome measures: Mononuclear infiltrates into the brain, astrogliosis, and neuronal death. | 1. BDV-ob infection and mononuclear infiltrates were restricted to certain brain areas including hypothalamus, hippocampus, and amygdala. Particularly severe infiltration in the median eminence of hypothalamus. (BDV-ob infection was observed evenly throughout the brain.) 2. Mononuclear infiltrates, astrogliosis, and neuronal death in the hippocampus of BDV-ob infected brain. | CNS infection →Hypothalamic inflammation and disruption of neuroendocrine system →Obesity | Herden et al., 2000 |
| Canine distemper virus (CDV) | ||||
| CDV/mouse | Metabolic disturbance: obesity induced by intracerebral CDV infection. Outcome measures: Plasma insulin and lipid composition. | Hyperinsulinemia and triglyceride accumulation in CDV-induced obesity mice | CDV infection → Hyperinsulinemia and obesity | Bernard et al., 1988 |
| CDV/mouse | Animal model: obesity induced by intracerebral infection with CDV. Outcome measures: Leptin and LepR expression. | Functional LepR was specifically downregulated in the hypothalamus of obese mice. | CNS infection →Leptin resistance in the hypothalamus. → Obesity. | Bernard et al., 1999 |
| Human immunodeficiency virus (HIV) | ||||
| HIV/human | Subjects: patients with HIV-1 infection (n203, Cohort study). Outcome measures: association between HIV-associated dementia (HAND) and diabetes. | HAND was significantly associated with diabetes (odds ratio 5.43, 1.66–17.70), which was not fully explained by age or coexisting vascular risk factors. | Diabetes ↔ HAND | Valcour et al., 2005 |
| HIV/mouse | Animal model: HIV model in mice using EcoHIV, with vs. without intranasal insulin treatment (daily for 9 days). Outcome measures: cognitive functions, hippocampal neuronal integrity, and the expression of genes associated with brain functions. | 1. Infected mice exhibited cognitive impairment. 2. Intranasal insulin restored cognitive functions, hippocampal dendritic integrity, and gene expressions. 3. The beneficial effect of intranasal insulin was independent of HIV burden in the brain. | Infection →Cognitive impairment. Central insulin treatment → Restoration of brain functions | Kim et al., 2019 |
| HIV/human FIV/cat | 1. Human ex vivo model: (1) brain autopsy of patients with HIV/AIDS. (2) HIV-1 infection in primary human neurons and microglia, treated with insulin. Outcome measures: neuroinflammation and neuronal death. 2. Animal in vivo HAND model: feline retrovirus (FIV) intracranial infection in cats. Insulin intranasal treatment for 6 weeks compared to PBS treatment. Outcome measures: morphological changes in the brain, neuroinflammation, neuronal survival, neurobehavioral performance. | 1-1. Increased neuroinflammatory gene expression in the brain of HIV/AIDS. 1-2. Insulin treatment suppressed HIV-1 growth and reduced infection-induced CSCL10 and IL-6 expression in HIV-infected microglia. 1-3. Insulin treatment prevented HIV-1 infection-induced neuronal death. 2-1. Insulin treatment enhanced the preservation of cortical neurons, and improved neurobehavioral performance in FIV-infected cats. | Infection → Cognitive impairment. Central insulin treatment → Restoration of brain functions. | Mamik et al., 2016 |
| Porphyromonas gingivalis | ||||
| P. gingivalis/mouse | Animal model: db/db mouse infected with P. gingivalis (W83, p.o.). Outcome measures: neuroinflammation in the hippocampus; mRNA levels for genes associated with insulin signaling. | 1. Infection induced reactive microglia and astrocytes. 2. Infection enhanced the expression of insulin signaling pathway genes (e.g., InsR, Igf1, Irs, and Gsk3β). 3. Pro-inflammatory genes were also up-regulated. | Infection → Disruption of insulin signaling pathway and inflammation in the brain. | Bahar et al., 2021 |
| Toxoplasma gondii | ||||
| T. gondii/rat | Animal model: T. gondii infection model in rats. Outcome measures: plasma leptin levels | Plasma leptin levels increased in chronic T. gondii infection. | CNS infection → Increase in plasma leptin (metabolic imbalance) | Baltaci and Mogulkoc, 2012 |
| West Nile virus (WNV) | ||||
| WNV/mouse | Animal model: db/db mouse infected with WNV. Outcome measures: leukocyte infiltration and neuroinflammation/neuronal damage. | Infection-induced leukocyte infiltration into the brain was reduced, while neuroinflammation/neuronal death was enhanced, in db/db mice. | LepR dysfunction → Increased CNS infection susceptibility | Kumar et al., 2014 |
Relevant keywords, such as insulin and leptin, are highlighted in bold.
West Nile Virus
West Nile virus (WNV) is a single-stranded RNA virus, genetically related to the Japanese encephalitis virus (JEV). Transmitted by mosquitoes, WNV potentially causes life-threatening encephalitis or meningitis, especially in the elderly (Alli et al., 2021). In addition, it can cause persistent cognitive impairments (Murray et al., 2014; Vittor et al., 2020). While CNS infiltration of lymphocytes, including CD8+ T cells, is essential in eliminating viruses, persistent inflammation can cause synaptic loss and neuronal death, impairing cognitive ability (Garber et al., 2019; Vittor et al., 2020). Of note, diabetes is a frequent comorbidity of severe WNV diseases and considered a risk factor for developing WNV encephalitis (Badawi et al., 2018). In db/db obesity model, WNV infection-induced leukocyte infiltration into the brain was significantly lower, suggesting that obesity compromises protection against viral infection in the brain (Kumar et al., 2014). In particular, infiltration of CD8+ T cells was significantly reduced in obese mice, which was associated with higher viral load and enhanced inflammatory responses in the brain (Kumar et al., 2014), suggesting a role of leptin signaling in protecting the brain via leukocyte recruitment (Rummel et al., 2010).
Porphyromonas gingivalis
Porphyromonas gingivalis is a periodontal disease-causing Gram-negative bacteria found in the oral cavity. Evidence suggests a strong association between P. gingivalis infection and sporadic AD (Kanagasingam et al., 2020), and its presence has been identified in the brain of AD patients (Dominy et al., 2019). P. gingivalis infection can trigger inflammation both in the periphery and CNS in affected individuals, leading to cognitive decline (Dominy et al., 2019; Kanagasingam et al., 2020). In mice, oral P. gingivalis infection led to brain colonization and AD-like pathogenesis, including complement activation and Aβ1–42 formation (Poole et al., 2015; Dominy et al., 2019), suggesting potential mechanistic links between periodontal disease and AD. A systematic review of pre-clinical studies also found that P. gingivalis infection induced inflammatory responses and tissue degeneration in the brain, which were associated with cognitive impairment (Costa et al., 2021).
Periodontitis is also associated with obesity (Suvan et al., 2011; Keller et al., 2015; Nascimento et al., 2015; Khan et al., 2018). High fat diet (HFD) significantly enhanced systemic inflammation induced by periodontal pathogens in rodents (Virto et al., 2018). P. gingivalis infection up-regulated the expression of genes associated with insulin/IGF-1 signaling and induced inflammatory responses in the brain of db/db mice (Virto et al., 2018), suggesting that metabolic imbalance due to dysregulated insulin and leptin signaling pathways may exacerbate the outcome of periodontitis and associated inflammation in the brain.
Human Immunodeficiency Virus
Despite the effectiveness of antiretroviral therapy in saving the lives of many from acquired immunodeficiency syndrome (AIDS), HIV-positive individuals frequently suffer neurocognitive disorders (HAND) (Antinori et al., 2007; Foley et al., 2011). While inflammation and brain atrophy due to persistent viral presence in the CNS may partly provide explanations (Pemberton and Brew, 2001; Woods et al., 2010; Ances et al., 2012; Brew and Barnes, 2019), exact mechanisms underlying HAND remain unclear.
Metabolic syndromes, including diabetes, is prevalent in HIV-infected individuals (Calza et al., 2011; Paik and Kotler, 2011), and insulin resistance was associated with lower cognitive scores in a HIV-1 cohort (Valcour et al., 2005, 2006), suggesting that HIV infection may increase the risk of cognitive impairment via metabolic dysregulation. In fact, in a murine model of HAND, intranasal insulin administration restored hippocampal dendritic integrity and cognitive functions, independently of HIV burden in the brain (Kim et al., 2019). In primary human neurons and microglia in vitro, insulin suppressed infection-induced inflammatory responses and HIV-1 growth in microglia, and prevented infection-induced neuronal death (Mamik et al., 2016). In addition, in a feline HIV model in vivo, intranasal insulin enhanced the preservation of cortical neurons and improved cognitive performance (Mamik et al., 2016), suggesting that impaired insulin signaling in the CNS may underlie cognitive impairment in HIV positive individuals.
Central Nervous System Infections Causing Metabolic Imbalance – Borna Disease Virus and Canine Distemper Virus
Borna disease virus (BDV) is a neurotropic RNA virus infecting a broad host spectrum including humans. Borna disease was originally observed as infectious diseases in domestic animals in the nineteenth century. The confirmation of human infection has begun since the 1980s, mainly in neuropsychiatric patients (Hatalski et al., 1997; Schwemmle, 2001; Taieb et al., 2001). BDV causes persistent infection in the brain and neurobehavioral deficits associated with neuroinflammation. In an experimental infection model in rodents, it takes a biphasic course characterized by hyperactivity associated with inflammatory lesions in the brain during the first acute stage, followed by the development of varying symptoms, including obesity, depending on viral strains and affected brain regions (Narayan et al., 1983; Herden et al., 2000). Intracerebral infection of rats with a BDV variant induced obesity without neurological signs, which is correlated with severe mononuclear cell infiltration into the hypothalamus, suggesting that infection-induced neuroendocrine dysregulations caused the development of obesity (Herden et al., 2000). It was also suggested that BDV infection-induced neuroinflammation and neurotransmitter imbalance underlie the dysfunction of hypothalamus and leptin resistance, leading to obesity (Gosztonyi et al., 2020).
Canine distemper virus (CDV) is an RNA virus closely related to measles virus, infecting a wide range of host species (Martinez-Gutierrez and Ruiz-Saenz, 2016). CDV causes canine distemper, a severe systemic disease in dogs, presenting a variety of symptoms including neurologic disorders (Martella et al., 2008). In a virus CDV infection-induced obesity model in mice using intracerebral infection, hyperinsulinemia and alteration in leptin signaling were observed (Bernard et al., 1988, 1999). In this model, infection caused hyperinsulinemia and obesity, while CDV showed tropism for the hypothalamus. Obesity developed in up to 30% of the surviving mice (Bernard et al., 1988). In addition, functional LepR was specifically down-regulated in the hypothalamus of infected obese mice (Bernard et al., 1999), suggesting that CDV infection in the brain induced leptin resistance in the hypothalamus, which led to obesity, thereby increasing the risk of MetS-associated cognitive impairment (Figure 3).
Peripheral Infection and Cognitive Impairment – Potential Involvement of Insulin and Glycemic Control
Peripheral infection-induced metabolic disturbance can lead to CNS dysfunction (Table 2). Sepsis-associated encephalopathy (SAE) is a brain disease secondary to peripheral infection without overt CNS infection, occurring up to 50–70% of sepsis cases (Chen Q. et al., 2014). After initial response to eliminate pathogens, systemic inflammation and increased BBB permeability occur, causing severe encephalopathy. While SAE is partly reversible, it can lead to persistent neurocognitive deficits, increasing the risk of dementia later in life (Widmann and Heneka, 2014; Seidel et al., 2020). Disrupted glycemic control is frequently encountered in sepsis patients (Hirasawa et al., 2009), and it has been suggested that higher infection susceptibility in patients with hyperglycemia is associated with disease severity (Koh et al., 2012). Experimental sepsis in rodents induced cognitive deficits accompanied by hyperglycemia (Huang et al., 2020). BBB disruption, microglial activation, oxidative damage and inflammation in the hippocampus, cortex and cerebrum occurred in those animal models (Michels et al., 2015; Sonneville et al., 2015; Huang et al., 2020), suggesting glial activation and neuroinflammation underlie SAE-induced cognitive impairment. In addition, the experimental sepsis induced more severe brain damages, including microglial activation and neuronal death, in hyperglycemic mice compared to insulin-treated mice (Sonneville et al., 2015), suggesting that poor glycemic control renders CNS more vulnerable to neuroinflammation, and insulin may protect the brain from sepsis-induced neuroinflammation and neuronal damages (Hache et al., 2015).
TABLE 2.
The roles of insulin in peripheral infection-induced CNS damage.
| Pathogen or disease/host | Study design and outcome measures | Findings | Proposed direction of causality and/or mechanism | References |
| Sepsis/rat | Animal model: surgically induced polymicrobial sepsis model in rats. Glucose treatment, insulin treatment compared to control. Outcome measures: Blood glucose, behavioral deficits, brain activity (EEG), BBB permeability, glial activation and inflammation in the cerebrum. | 1. Sepsis induced hyperglycemia. 2. Glucose treatment led to a decline in survival rate, reduced brain activity, increased BBB permeability, and enhanced microglial and astroglial activation and inflammatory responses in the cerebrum. 3. Glycemic control (insulin treatment) inhibited inflammatory responses and restored BBB integrity and brain activity to near normal. | Peripheral infection →Hyperglycemia. →Glial activation and neuroinflammation. → Cognitive dysfunction. Note: Insulin help restore brain functions by preventing BBB disruption and neuroinflammation. | Huang et al., 2020 |
| Sepsis/mouse | Animal model: surgically induced polymicrobial sepsis model in mice. Mice with moderate hyperglycemia were compared to control (normoglycemia). Outcome measures: neuronal damages, glial activation, and cell death in the hippocampus and frontal cortex. | (A) In hyperglycemic mice (compared to normoglycemia mice), sepsis induced: 1. Higher neuronal damage in frontal cortex. 2. Microglial activation in frontal cortex and hippocampus. 3. More apoptotic cells in frontal cortex. (B) Insulin prevented the above damages | Hyperglycemia →Enhanced infection (sepsis)-induced brain damage. Note: Insulin prevents hyperglycemia-induced susceptibility to sepsis-induced brain damages | Sonneville et al., 2015 |
| Sepsis/mouse | Animal model: surgically induced polymicrobial sepsis model in mice. Outcome measures: cognitive functions, hippocampal synaptic plasticity, and hippocampal insulin signaling in post-septic mice compared to control. | 1. Post-septic mice exhibited cognitive impairment, which was accompanied by reduced synaptic plasticity and disrupted insulin signaling in the hippocampus. 2. Treatment with a GLP-1 receptor agonist (insulinotropic) or GSK3β inhibitor (insulin signaling downstream) rescued cognition. | Peripheral infection →Disruption of hippocampal insulin signaling. → Impaired synaptic plasticity and cognitive deficits. | Neves et al., 2018 |
| Sepsis/rat | Animal model: sepsis model induced by LPS (i.p.) in rats. Insulin treatment: continuous intravenous infusion for 6h after LPS stimulation. Compared to control (saline). Outcome measures: Inflammatory cytokines and oxidative stress in the cortex, hippocampus, and hypothalamus. | In the brain regions (cortex, hippocampus, and hypothalamus): 1.Insulin alleviated sepsis-induced inflammatory response (IL-1β, IL-6, and TNF-α). 2. Insulin suppressed oxidative damage while restoring antioxidants (SOD and GSH). | Peripheral infection →Inflammation and oxidative stress in the brain. Note: Insulin treatment lowered sepsis-induced inflammation in the brain | Chen Q. et al., 2014 |
| Systemic inflammation/mouse | Animal model: LPS (i.p.) challenge in chronic neurodegeneration model (ME7 prion infection) in mice. Treated with glucose and insulin. Outcome measures: blood glucose, cognitive performance | LPS challenge induced hypoglycemia and acute cognitive impairment in mice with brain disease (ME7 prion infection), which was mitigated by glucose and mimicked by insulin. | Peripheral infection →Metabolic imbalance in the CNS. →Cognitive impairment. Note: Brain disease (prion disease) makes the CNS more vulnerable to peripheral inflammation | Kealy et al., 2020 |
Relevant keywords, such as insulin and leptin, are highlighted in bold.
In addition, acute inflammation in the periphery (LPS challenge) induced metabolic changes in the CNS in rodents, and the metabolic imbalance in the brain was associated impaired cognition (Kealy et al., 2020). In addition, LPS-induced acute peripheral inflammation produced hypoglycemia in blood and CSF, and caused severe cognitive impairment selectively in those mice with brain disease (chronic neurodegeneration model induced by prion infection), which was mimicked by insulin and alleviated by glucose administration (Kealy et al., 2020). These data suggest that acute peripheral inflammation can cause neuroinflammation and cognitive deficits via disruption of glycemic control, whereas chronic neuroinflammation, including infection-induced neurodegenerative disorders, renders the brain more vulnerable to metabolic imbalance (Figure 3).
Other Pathogens Affecting Metabolic Balance – Indirect Link to Cognition
‘Infectobesity’ is a concept that proposes the infectious etiology of obesity (Pasarica and Dhurandhar, 2007; van Ginneken et al., 2009). Considering the link between MetS and cognitive decline (Mejido et al., 2020), infectobesity can be an indirect cause of dementia. In addition to BDV and CDV, there are several viruses potentially causing obesity (Dhurandhar, 2001; Atkinson, 2007). Rous-associated virus 7 (RAV-7), a retrovirus causing avian leukosis, can induce stunting and obesity in chickens, which is associated with hyperlipidemia and increased insulin levels, as well as immune dysregulation (Carter and Smith, 1984). Adenovirus 36 (Ad-36) is a human adenovirus associated with obesity (Esposito et al., 2012). Ad-36 infection in adipocytes reduced leptin production while inducing proliferation, differentiation, and lipid accumulation in adipocytes (Vangipuram et al., 2007; van Ginneken et al., 2009), suggesting viral infection of fat cells can directly cause adipogenesis and obesity. Reduced leptin release caused by infection may also compromise CNS-mediated control of energy homeostasis, further enhancing the risk of MetS-associated cognitive impairment (Whitmer et al., 2005; Kivipelto et al., 2006; Mejido et al., 2020).
Gut Microbiome
While the concept of microbial infection and pathogenicity has dominated the mainstream of microbiology, only a small fraction of microorganism are inherently pathogenic. In particular, the gut is inhabited by microbiota, a collection of microorganisms including bacteria, archaea, viruses, and fungi, consisting of at least 1,000 distinct species. Commensal bacterial and fungal species are involved in the regulation/dysregulation of energy homeostasis and immune responses, through extracting and metabolizing nutrients, regulating peripheral and central insulin sensitivity (Schertzer and Lam, 2021), and contributing to intestinal immune control via bidirectional communication with immune cells (Khan et al., 2021). Disruption of the symbiotic relationship between the host and microbiota leads to chronic inflammation and insulin resistance (Patterson et al., 2016). Obesity is associated with altered gut microbial composition in mice and humans, and the trait is transmissible as colonization of germ-free mice with microbiota from obese mice led to a significant increase in body fat compared to colonization with microbiota from lean mice (Turnbaugh et al., 2006), suggesting that gut microbiome is part of the host metabolic system actively regulating energy balance (Bäckhed et al., 2004). It has also been suggested that microbiome plays a crucial role in the communication between the gut and brain (microbiota-gut-brain axis), which is essential for the regulation of energy homeostasis (Romaní-Pérez et al., 2021; van Son et al., 2021) as well as the development and functions of the nervous system (Chen et al., 2021; Gwak and Chang, 2021). Consequently, altered gut microbiota (dysbiosis) has been implicated in a number of chronic inflammatory diseases, such as diabetes (Zawada et al., 2020; Rodriguez and Delzenne, 2021) and neurodegenerative disorders, including AD (Jiang et al., 2017; Chen et al., 2021; Leblhuber et al., 2021; Romanenko et al., 2021). Furthermore, these indigenous microbiota play a critical role in host defense against infection, through stimulating mucosal immune defenses (e.g., of antimicrobial peptides and IgA release) and limiting resource availability/niche opportunity for invading microbes (Libertucci and Young, 2019). Consequently, dysbiosis has been associated with infection susceptibility (Lazar et al., 2018; Libertucci and Young, 2019), which may further accelerate immunometabolic imbalance.
Probiotics are living microorganisms that provide health benefits by improving or restoring the composition of gut microbiota. It has been shown that probiotics can reduce leptin secretion and improve hypothalamic leptin and insulin resistance in high fat diet-induced obesity models in rodents (Al-muzafar and Amin, 2017; Bagarolli et al., 2017; Ji et al., 2018; Cheng and Liu, 2020). Of note, probiotics may prevent AD pathogenesis by improving glucose metabolism. Triple transgenic AD model mice (3xTg-AD) exhibited significant metabolic impairment [increased glycated hemoglobin [HbA1c] in the serum, accumulation of advanced glycation end products (AGE), and impaired glucose uptake due to decreased glucose transporter levels in the brain], and all these parameters were normalized by oral treatment with probiotics (lactic acid bacteria and bifidobacteria) (Bonfili et al., 2020). These data suggest that beneficial gut microbiome can reduce AD pathogenesis via the restoration of metabolic balance (Figure 3).
Discussion and Conclusion
Metabolic diseases and infection are important risk factors for dementia (Whitmer et al., 2005; Kivipelto et al., 2006; Ashraf et al., 2019; Mejido et al., 2020). Metabolic dysregulation enhances infection susceptibility via immune dysfunction, whereas infection, both in the CNS and periphery, can disrupt metabolic balance (Figure 3). In the CNS, infection-induced damage in the regions associated with metabolic control, such as hypothalamus, can disrupt whole-body energy metabolism. In the periphery, infection-induced immune reactions may persist and disarray immunometabolism, resulting in chronic inflammation and increased BBB permeability (Gustafson et al., 2007; Kanoski et al., 2010; Montagne et al., 2015), which can disrupt the CNS integrity, cognitive ability, and central energy control (Montagne et al., 2015). In addition, infection-induced dysregulation of glucose metabolism can lead to glial activation and neuroinflammation (Huang et al., 2020; Bahar et al., 2021). The vicious cycle may further advance the systemic imbalance, while beneficial gut microbiome can restore the balance of immunometabolism. Furthermore, although evidence is still lacking, infectious agents can potentially disrupt the immunometabolism in the brain by infecting microglia and astrocytes. Thus, multidirectional interactions between metabolic imbalance and infection at cellular and systemic levels likely occur during the development of dementia and neurodegenerative disorders. Combined and multifactorial impacts due to these interactions can further exacerbate brain dysfunction. As the key regulators of energy metabolism and immune responses, insulin and leptin play significant roles in those intricate interactions. It should also be noted that other metabolic hormones and adipokines, such as glucagon and adiponectin, similarly take part in immunometabolism, and dysregulation of these molecules are also implicated in neurodegenerative disorders, including AD (Talbot and Wang, 2014; Grieco et al., 2019; Kim et al., 2020). In addition, growing evidence suggests the central actions of thyroid hormones (THs) (Capelli et al., 2021); THs exert immunomodulatory roles in the brain by inducing microglial phenotypic changes (Mallat et al., 2002) and affecting the expression of chemokines (Davis et al., 2016). Thyroid dysfunctions are possibly associated with AD pathogenesis (Figueroa et al., 2021). Future research may unveil the interactions between infection and these metabolic hormones.
While the potential therapeutic benefit of insulin in treating dementia has been increasingly recognized (Frölich et al., 1998; Kim et al., 2019; Kellar and Craft, 2020), it is relatively unexplored how the modulation of metabolic regulators, including insulin and leptin, can impact infection-induced neurological disorders. Considering the diverse roles for these molecules play in the immunometabolic network, including the CNS immunometabolism (Larabee et al., 2020), it is plausible that infection-induced damages to the brain can be alleviated by modulating these signaling pathways. On the other hand, it is largely unresolved how glia - pathogen interactions can impact immunometabolism in the CNS and what roles leptin and insulin may play in the interactions. How these relationships may relate to the etiology of dementia is also an unanswered question. Further research is warranted to understand the mechanisms underlying the interrelationship between infection, metabolic disorders, and dementia.
Author Contributions
NS wrote the first draft of the manuscript. KK critically assessed the content. NS and KK jointly finalized the manuscript. Both authors contributed to the article and approved the submitted version.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Funding
This study was supported by the Nagasaki University “Doctoral Program for World-leading Innovative and Smart Education” for Global Health, “Global Health Elite Programme for Building a Healthier World”.
References
- Abella V., Scotece M., Conde J., Pino J., Gonzalez-Gay M., Gómez-Reino J., et al. (2017). Leptin in the interplay of inflammation, metabolism and immune system disorders. Nat. Rev. Rheumatol. 13 100–109. 10.1038/nrrheum.2016.209 [DOI] [PubMed] [Google Scholar]
- Ai S., Klein R. (2020). Update on T cells in the virally infected brain: friends and foes. Curr. Opin. Neurol. 33 405–412. 10.1097/WCO.0000000000000825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alli A., Ortiz J., Atoot A., Atoot A., Millhouse P. (2021). Management of West Nile encephalitis: an uncommon complication of West Nile virus. Cureus 13:e13183. 10.7759/cureus.13183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allison M., Myers M. J. (2014). 20 years of leptin: connecting leptin signaling to biological function. J. Endocrinol. 223 T25–T35. 10.1530/JOE-14-0404 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-muzafar H., Amin K. (2017). Probiotic mixture improves fatty liver disease by virtue of its action on lipid profiles, leptin, and inflammatory biomarkers. BMC Complement. Altern. Med. 17:43. 10.1186/s12906-016-1540-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alti D., Sambamurthy C., Kalangi S. (2018). Emergence of leptin in infection and immunity: scope and challenges in vaccines formulation. Front. Cell Infect. Microbiol. 8:147. 10.3389/fcimb.2018.00147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amole B., Wittner M., Hewlett D., Tanowitz H. (1985). Trypanosoma brucei: infection in murine diabetes. Exp. Parasitol. 60 342–347. 10.1016/0014-4894(85)90040-90042 [DOI] [PubMed] [Google Scholar]
- Ances B., Ortega M., Vaida F., Heaps J., Paul R. (2012). Independent effects of HIV, aging, and HAART on brain volumetric measures. J. Acquir. Immune Defic. Syndr. 59 469–477. 10.1097/QAI.0b013e318249db17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- André C., Guzman-Quevedo O., Rey C., Rémus-Borel J., Clark S., Castellanos-Jankiewicz A., et al. (2017). Inhibiting microglia expansion prevents diet-induced hypothalamic and peripheral inflammation. Diabetes Metab. Res. Rev. 66 908–919. 10.2337/db16-0586 [DOI] [PubMed] [Google Scholar]
- Antinori A., Arendt G., Becker J., Brew B., Byrd D., Cherner M., et al. (2007). Updated research nosology for HIV-associated neurocognitive disorders. Neurology 69 1789–1799. 10.1212/01.WNL.0000287431.88658.8b [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arnold S., Arvanitakis Z., Macauley-Rambach S., Koenig A., Wang H., Ahima R., et al. (2018). Brain insulin resistance in type 2 diabetes and Alzheimer disease: concepts and conundrums. Nat. Rev. Neurol. 14 168–181. 10.1038/nrneurol.2017.185 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arvanitakis Z., Wilson R., Bienias J., Evans D., Bennett D. (2004). Diabetes mellitus and risk of Alzheimer disease and decline in cognitive function. Arch. Neurol. 61 661–666. 10.1001/archneur.61.5.661 [DOI] [PubMed] [Google Scholar]
- Ashraf G., Tarasov V., Makhmutovà A., Chubarev V., Avila-Rodriguez M., Bachurin S., et al. (2019). The possibility of an infectious etiology of Alzheimer disease. Mol. Neurobiol. 56 4479–4491. 10.1007/s12035-018-1388-y [DOI] [PubMed] [Google Scholar]
- Atkinson R. (2007). Viruses as an etiology of obesity. Mayo Clin. Proc. 82 1192–1198. 10.4065/82.10.1192 [DOI] [PubMed] [Google Scholar]
- Avalos C., Abreu C., Queen S., Li M., Price S., Shirk E., et al. (2017). Brain macrophages in simian immunodeficiency virus-infected, antiretroviral-suppressed macaques. mBio 8:e01186-17. 10.1128/mBio.01186-1117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bäckhed F., Ding H., Wang T., Hooper L., Koh G., Nagy A., et al. (2004). The gut microbiota as an environmental factor that regulates fat storage. Proc. Natl. Acad. Sci. U S A. 101 15718–15723. 10.1073/pnas.0407076101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Badawi A., Velummailum R., Ryoo S., Senthinathan A., Yaghoubi S., Vasileva D., et al. (2018). Prevalence of chronic comorbidities in dengue fever and West Nile virus: a systematic review and meta-analysis. PLoS One 13:e0200200. 10.1371/journal.pone.0200200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bagarolli R., Tobar N., Oliveira A., Araújo T., Carvalho B., Rocha G., et al. (2017). Probiotics modulate gut microbiota and improve insulin sensitivity in DIO mice. J. Nutr. Biochem. 50 16–25. 10.1016/j.jnutbio.2017.08.00 [DOI] [PubMed] [Google Scholar]
- Bahar B., Kanagasingam S., Tambuwala M., Aljabali A., Dillon S., Doaei S., et al. (2021). Porphyromonas gingivalis (W83) infection induces Alzheimer’s disease-like pathophysiology in obese and diabetic mice. J. Alzheimers Dis. 82 1259–1275. 10.3233/JAD-210465 [DOI] [PubMed] [Google Scholar]
- Balin B., Gérard H., Arking E., Appelt D., Branigan P., Abrams J., et al. (1998). Identification and localization of Chlamydia pneumoniae in the Alzheimer’s brain. Med. Microbiol. Immunol. 187 23–42. 10.1007/s004300050071 [DOI] [PubMed] [Google Scholar]
- Baltaci A., Mogulkoc R. (2012). Plasma leptin levels in rats with induced Toxoplasma gondii infection. Bratisl Lek List. 113 67–69. 10.4149/bll_2012_016 [DOI] [PubMed] [Google Scholar]
- Balthasar N., Coppari R., McMinn J., Liu S., Lee C., Tang V., et al. (2004). Leptin receptor signaling in POMC neurons is required for normal body weight homeostasis. Neuron 42 983–991. 10.1016/j.neuron.2004.06.004 [DOI] [PubMed] [Google Scholar]
- Balyan R., Gautam N., Gascoigne N. (2020). The Ups and downs of metabolism during the lifespan of a T Cell. Int. J. Mol. Sci. 21:7972. 10.3390/ijms21217972 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banks W., Farrell C. (2003). Impaired transport of leptin across the blood-brain barrier in obesity is acquired and reversible. Am. J. Physiol. Endocrinol. Metab. 285 E10–E15. 10.1152/ajpendo.00468.2002 [DOI] [PubMed] [Google Scholar]
- Banks W., Owen J., Erickson M. (2012). Insulin in the brain: there and back again. Pharmacol. Ther. 136 82–93. 10.1016/j.pharmthera.2012.07.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baradaran A., Dehghanbanadaki H., Naderpour S., Pirkashani L., Rajabi A., Rashti R., et al. (2021). The association between Helicobacter pylori and obesity: a systematic review and meta-analysis of case-control studies. Clin. Diabetes Endocrinol. 7:15. 10.1186/s40842-021-00131-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrios V., López-Villar E., Frago L., Canelles S., Díaz-González F., Burgos-Ramos E., et al. (2021). Cerebral insulin bolus revokes the changes in hepatic lipid metabolism induced by chronic central leptin infusion. Cells 10:581. 10.3390/cells10030581 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernard A., Cohen R., Khuth S., Vedrine B., Verlaeten O., Akaoka H., et al. (1999). Alteration of the leptin network in late morbid obesity induced in mice by brain infection with canine distemper virus. J. Virol. 73 7317–7327. 10.1128/JVI.73.9.7317-7327.1999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernard A., Zwingelstein G., Meister R., Wild T. (1988). Hyperinsulinemia induced by canine distemper virus infection of mice and its correlation with the appearance of obesity. Comp. Biochem. Physiol. B 91 691–696. 10.1016/0305-0491(88)90193-90199 [DOI] [PubMed] [Google Scholar]
- Bernier L., York E., Kamyabi A., Choi H., Weilinger N., MacVicar B. (2020). Microglial metabolic flexibility supports immune surveillance of the brain parenchyma. Nat. Commun. 11:1559. 10.1038/s41467-020-15267-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beydoun M., Beydoun H., Elbejjani M., Dore G., Zonderman A. (2018). Helicobacter pylori seropositivity and its association with incident all-cause and Alzheimer’s disease dementia in large national surveys. Alzheimers Dement 14 1148–1158. 10.1016/j.jalz.2018.04.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biessels G., Despa F. (2018). Cognitive decline and dementia in diabetes mellitus: mechanisms and clinical implications. Nat. Rev. Endocrinol. 14 591–604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biessels G., Staekenborg S., Brunner E., Brayne C., Scheltens P. (2006). Risk of dementia in diabetes mellitus: a systematic review. Lancet Neurol. 5 64–74. 10.1016/S1474-4422(05)70284-70282 [DOI] [PubMed] [Google Scholar]
- Bonda D., Stone J., Torres S., Siedlak S., Perry G., Kryscio R., et al. (2014). Dysregulation of leptin signaling in Alzheimer disease: evidence for neuronal leptin resistance. J. Neurochem. 128 162–172. 10.1111/jnc.12380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonfili L., Cecarini V., Gogoi O., Berardi S., Scarpona S., Angeletti M., et al. (2020). Gut microbiota manipulation through probiotics oral administration restores glucose homeostasis in a mouse model of Alzheimer’s disease. Neurobiol. Aging 87 35–43. 10.1016/j.neurobiolaging.2019.11.004 [DOI] [PubMed] [Google Scholar]
- Bos I., Vos S., Frölich L., Kornhuber J., Wiltfang J., Maier W., et al. (2017). The frequency and influence of dementia risk factors in prodromal Alzheimer’s disease. Neurobiol. Aging 56 33–40. 10.1016/j.neurobiolaging.2017.03.034 [DOI] [PubMed] [Google Scholar]
- Boucsein A., Kamstra K., Tups A. (2021). Central signalling cross-talk between insulin and leptin in glucose and energy homeostasis. J. Neuroendocr. 33:e12944. 10.1111/jne.12944 [DOI] [PubMed] [Google Scholar]
- Boutens L., Hooiveld G., Dhingra S., Cramer R., Netea M., Stienstra R. (2018). Unique metabolic activation of adipose tissue macrophages in obesity promotes inflammatory responses. Diabetologia 61 942–953. 10.1007/s00125-017-4526-4526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brabazon F., Bermudez S., Shaughness M., Khayrullina G., Byrnes K. (2018). The effects of insulin on the inflammatory activity of BV2 microglia. PLoS One 13:e0201878. 10.1371/journal.pone.0201878 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brew B., Barnes S. (2019). The impact of HIV central nervous system persistence on pathogenesis. AIDS 33 S113–S121. 10.1097/QAD.0000000000002251 [DOI] [PubMed] [Google Scholar]
- Bulloch K., Miller M., Gal-Toth J., Milner T., Gottfried-Blackmore A., Waters E., et al. (2008). CD11c/EYFP transgene illuminates a discrete network of dendritic cells within the embryonic, neonatal, adult, and injured mouse brain. J. Comp. Neurol. 508 687–710. 10.1002/cne.21668 [DOI] [PubMed] [Google Scholar]
- Caldefie-Chezet F., Poulin A., Vasson M. (2003). Leptin regulates functional capacities of polymorphonuclear neutrophils. Free Radic. Res. 37 809–814. 10.1080/1071576031000097526 [DOI] [PubMed] [Google Scholar]
- Calza L., Masetti G., Piergentili B., Trapani F., Cascavilla A., Manfredi R., et al. (2011). Prevalence of diabetes mellitus, hyperinsulinaemia and metabolic syndrome among 755 adult patients with HIV-1 infection. Int. J. STD AIDS 22 43–45. 10.1258/ijsa.2010.010256 [DOI] [PubMed] [Google Scholar]
- Capelli V., Diéguez C., Mittag J., López M. (2021). Thyroid wars: the rise of central actions. Trends Endocrinol. Metab. 32 659–671. 10.1016/j.tem.2021.05.006 [DOI] [PubMed] [Google Scholar]
- Cárdenas G., Soto-Hernández J., Díaz-Alba A., Ugalde Y., Mérida-Puga J., Rosetti M., et al. (2014). Neurological events related to influenza A (H1N1) pdm09. Influ. Other Respir. Viruses 8 339–346. 10.1111/irv.12241 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carro E., Trejo J., Núñez A., Torres-Aleman I. (2003). Brain repair and neuroprotection by serum insulin-like growth factor. Mol. Neurobiol. 27 153–162. [DOI] [PubMed] [Google Scholar]
- Carter J., Smith R. (1984). Specificity of avian leukosis virus-induced hyperlipidemia. J. Virol. 50 301–308. 10.1128/JVI.50.2.301-308.1984 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cereda E., Sansone V., Meola G., Malavazos A. (2007). Increased visceral adipose tissue rather than BMI as a risk factor for dementia. Age Ageing 36 488–491. 10.1093/ageing/afm096 [DOI] [PubMed] [Google Scholar]
- Cheeran M., Hu S., Yager S., Gekker G., Peterson P., Lokensgard J. (2001). Cytomegalovirus induces cytokine and chemokine production differentially in microglia and astrocytes. J. Neurovirol. 7 135–147. 10.1080/13550280152058799 [DOI] [PubMed] [Google Scholar]
- Chen H., Charlat O., Tartaglia L., Woolf E., Weng X., Ellis S., et al. (1996). Evidence that the diabetes gene encodes the leptin receptor: identification of a mutation in the leptin receptor gene in db/db mice. Cell 84 491–495. 10.1016/s0092-8674(00)81294-81295 [DOI] [PubMed] [Google Scholar]
- Chen Q., Yu W., Shi J., Shen J., Gao T., Zhang J., et al. (2014). Insulin alleviates the inflammatory response and oxidative stress injury in cerebral tissues in septic rats. J. Inflamm. 11:18. 10.1186/1476-9255-11-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y., Zhao Y., Dai C., Liang Z., Run X., Iqbal K., et al. (2014). Intranasal insulin restores insulin signaling, increases synaptic proteins, and reduces Abeta level and microglia activation in the brains of 3xTg-AD mice. Exp. Neurol. 261 610–619. 10.1016/j.expneurol.2014.06.004 [DOI] [PubMed] [Google Scholar]
- Chen Y., Xu J., Chen Y. (2021). Regulation of neurotransmitters by the gut microbiota and effects on cognition in neurological disorders. Nutrients 13:2099. 10.3390/nu13062099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Y., Liu J. (2020). Effect of Lactobacillus rhamnosus GG on energy metabolism, leptin resistance, and gut microbiota in mice with diet-induced obesity. Nutrients 12:2557. 10.3390/nu12092557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chi H. (2012). Regulation and function of mTOR signalling in T cell fate decisions. Nat. Rev. Immunol. 12 325–338. 10.1038/nri3198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chowen J., Garcia-Segura L. (2020). Microglia, neurodegeneration and loss of neuroendocrine control. Prog. Neurobiol. 184:101720. 10.1016/j.pneurobio.2019.101720 [DOI] [PubMed] [Google Scholar]
- Cohen S., Danzaki K., MacIver N. (2017). Nutritional effects on T-cell immunometabolism. Eur. J. Immunol. 47 225–235. 10.1002/eji.201646423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colonna M., Butovsky O. (2017). Microglia function in the central nervous system during health and neurodegeneration. Annu. Rev. Immunol. 35 441–468. 10.1146/annurev-immunol-051116-052358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conde J., Scotece M., Gómez R., Gómez-Reino J., Lago F., Gualillo O. (2010). At the crossroad between immunity and metabolism: focus on leptin. Expert Rev. Clin. Immunol. 6 801–808. 10.1586/eci.10.48 [DOI] [PubMed] [Google Scholar]
- Costa M., de Araújo I., da Rocha, Alves L., da Silva R., Dos Santos, et al. (2021). Relationship of Porphyromonas gingivalis and Alzheimer’s disease: a systematic review of pre-clinical studies. Clin. Oral Investig. 25 797–806. 10.1007/s00784-020-03764-w [DOI] [PubMed] [Google Scholar]
- Curi R., Levada-Pires A., Silva E., Poma S., Zambonatto R., Domenech P., et al. (2020). The critical role of cell metabolism for essential neutrophil functions. Cell Physiol. Biochem. 54 629–647. 10.33594/000000245 [DOI] [PubMed] [Google Scholar]
- Dani N., Herbst R., McCabe C., Green G., Kaiser K., Head J., et al. (2021). A cellular and spatial map of the choroid plexus across brain ventricles and ages. Cell 184 3056–3074. 10.1016/j.cell.2021.04.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis A., Rohlwink U., Proust A., Figaji A., Wilkinson R. (2019). The pathogenesis of tuberculous meningitis. J. Leukoc. Biol. 105 267–280. 10.1002/JLB.MR0318-102R [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis B., Salinas-Navarro M., Cordeiro M., Moons L., De Groef L. (2017). Characterizing microglia activation: a spatial statistics approach to maximize information extraction. Sci. Rep. 7:1576. 10.1038/s41598-017-01747-1748 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis N., Mota B., Stead L., Palmer E., Lombardero L., Rodríguez-Puertas R., et al. (2021). Pharmacological ablation of astrocytes reduces Aβ degradation and synaptic connectivity in an ex vivo model of Alzheimer’s disease. J. Neuroinflamm. 18:73. 10.1186/s12974-021-02117-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis P., Glinsky G., Lin H., Mousa S. (2016). Actions of thyroid hormone analogues on chemokines. J. Immunol. Res. 2016:3147671. 10.1155/2016/3147671 [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Strooper B., Karran E. (2016). The cellular phase of Alzheimer’s disease. Cell 164 603–615. 10.1016/j.cell.2015.12.056 [DOI] [PubMed] [Google Scholar]
- Deckers K., Van Boxtel M., Verhey F., Köhler S. (2017). Obesity and cognitive decline in adults: effect of methodological choices and confounding by age in a longitudinal study. J. Nutr. Heal. Aging 21 546–553. 10.1007/s12603-016-0757-753 [DOI] [PubMed] [Google Scholar]
- Dhurandhar N. (2001). Infectobesity: obesity of infectious origin. J. Nutr. 131 2794S–2797S. 10.1093/jn/131.10.2794S [DOI] [PubMed] [Google Scholar]
- Dhurandhar N. (2011). A framework for identification of infections that contribute to human obesity. Lancet Infect. Dis. 11 963–969. 10.1016/S1473-3099(11)70274-70272 [DOI] [PubMed] [Google Scholar]
- DiAngelo J., Bland M., Bambina S., Cherry S., Birnbaum M. (2009). The immune response attenuates growth and nutrient storage in Drosophila by reducing insulin signaling. Proc. Natl. Acad. Sci. U S A. 106 20853–20858. 10.1073/pnas.0906749106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dietrich M., Spuch C., Antequera D., Rodal I., de Yébenes J., Molina J., et al. (2008). Megalin mediates the transport of leptin across the blood-CSF barrier. Neurobiol. Aging 29 902–912. 10.1016/j.neurobiolaging.2007.01.008 [DOI] [PubMed] [Google Scholar]
- Dominy S., Lynch C., Ermini F., Benedyk M., Marczyk A., Konradi A., et al. (2019). Porphyromonas gingivalis in Alzheimer’s disease brains: evidence for disease causation and treatment with small-molecule inhibitors. Sci. Adv. 5:eaau3333. 10.1126/sciadv.aau3333 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drögemüller K., Helmuth U., Brunn A., Sakowicz-Burkiewicz M., Gutmann D., Mueller W., et al. (2008). Astrocyte gp130 expression is critical for the control of Toxoplasma encephalitis. J. Immunol. 181 2683–2693. 10.4049/jimmunol.181.4.2683 [DOI] [PubMed] [Google Scholar]
- Du L., Lin L., Li Q., Liu K., Huang Y., Wang X., et al. (2019). IGF-2 preprograms maturing macrophages to acquire oxidative phosphorylation-dependent anti-inflammatory properties. Cell Metab. 29 1363–1375. 10.1016/j.cmet.2019.01.006 [DOI] [PubMed] [Google Scholar]
- Dyer A., Vahdatpour C., Sanfeliu A., Tropea D. (2016). The role of Insulin-Like Growth Factor 1 (IGF-1) in brain development, maturation and neuroplasticity. Neuroscience 325 89–99. 10.1016/j.neuroscience.2016.03.056 [DOI] [PubMed] [Google Scholar]
- El Saftawy E., Amin N., Sabry R., El-Anwar N., Shash R., Elsebaie E., et al. (2020). Can Toxoplasma gondii pave the road for dementia? J. Parasitol. Res. 2020:8859857. 10.1155/2020/8859857 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elmquist J., Bjørbaek C., Ahima R., Flier J., Saper C. (1998). Distributions of leptin receptor mRNA isoforms in the rat brain. J. Comp. Neurol. 395 535–547. [PubMed] [Google Scholar]
- Esposito K., Nappo F., Marfella R., Giugliano G., Giugliano F., Ciotola M., et al. (2002). Inflammatory cytokine concentrations are acutely increased by hyperglycemia in humans: role of oxidative stress. Circulation 106 2067–2072. 10.1161/01.cir.0000034509.14906.ae [DOI] [PubMed] [Google Scholar]
- Esposito S., Preti V., Consolo S., Nazzari E., Principi N. (2012). Adenovirus 36 infection and obesity. J. Clin. Virol. 55 95–100. 10.1016/j.jcv.2012.06.009 [DOI] [PubMed] [Google Scholar]
- Faggioni R., Fantuzzi G., Gabay C., Moser A., Dinarello C., Feingold K., et al. (1999). Leptin deficiency enhances sensitivity to endotoxin-induced lethality. Am. J. Physiol. 276 R136–R142. 10.1152/ajpregu.1999.276.1.R136 [DOI] [PubMed] [Google Scholar]
- Fantuzzi G., Faggioni R. (2000). Leptin in the regulation of immunity, inflammation, and hematopoiesis. J. Leukoc. Biol. 68 437–446. [PubMed] [Google Scholar]
- Fasshauer M., Blüher M. (2015). Adipokines in health and disease. Trends Pharmacol. Sci. 36 461–470. 10.1016/j.tips.2015.04.014 [DOI] [PubMed] [Google Scholar]
- Fernandez A., Hernandez E., Guerrero-Gomez D., Miranda-Vizuete A., Torres Aleman I. (2018). A network of insulin peptides regulate glucose uptake by astrocytes: potential new druggable targets for brain hypometabolism. Neuropharmacology 136 216–222. 10.1016/j.neuropharm.2017.08.034 [DOI] [PubMed] [Google Scholar]
- Fernandez A., Torres-Aleman I. (2012). The many faces of insulin-like peptide signalling in the brain. Nat. Rev. Neurosci. 13 225–239. [DOI] [PubMed] [Google Scholar]
- Fernández-Martos C., González P., Rodriguez F. (2012). Acute leptin treatment enhances functional recovery after spinal cord injury. PLoS One 7:e35594. 10.1371/journal.pone.0035594 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernández-Real J., Ferri M., Vendrell J., Ricart W. (2007). Burden of infection and fat mass in healthy middle-aged men. Obes (Silver Spring) 15 245–252. 10.1038/oby.2007.541 [DOI] [PubMed] [Google Scholar]
- Fernández-Real J., López-Bermejo A., Vendrell J., Ferri M., Recasens M., Ricart W. (2006). Burden of infection and insulin resistance in healthy middle-aged men. Diabetes Care 29 1058–1064. 10.2337/diacare.2951058 [DOI] [PubMed] [Google Scholar]
- Ferri C., Prince M., Brayne C., Brodaty H., Fratiglioni L., Ganguli M., et al. (2005). Global prevalence of dementia: a Delphi consensus study. Lancet 366 2112–2117. 10.1016/S0140-6736(05)67889-67880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Figueroa P., Ferreira A., Britto L., Doussoulin A., Torrão A. (2021). Association between thyroid function and Alzheimer’s disease: a systematic review. Metab. Brain Dis. 36 1523–1543. 10.1007/s11011-021-00760-761 [DOI] [PubMed] [Google Scholar]
- Fischer A., Cannon B., Nedergaard J. (2020). Leptin: is it thermogenic? Endocr Rev. 41 232–260. 10.1210/endrev/bnz016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer H., Reichmann G. (2001). Brain dendritic cells and macrophages/microglia in central nervous system inflammation. J. Immunol. 166 2717–2726. 10.4049/jimmunol.166.4.2717 [DOI] [PubMed] [Google Scholar]
- Fleck-Derderian S., McClellan W., Wojcicki J. (2017). The association between cytomegalovirus infection, obesity, and metabolic syndrome in U.S. adult females. Obes (Silver Spring) 25 626–633. 10.1002/oby.21764 [DOI] [PubMed] [Google Scholar]
- Foley J., Wright M., Gooding A., Ettenhofer M., Kim M., Choi M., et al. (2011). Operationalization of the updated diagnostic algorithm for classifying HIV-related cognitive impairment and dementia. Int. Psychogeriatr. 23 835–843. 10.1017/S1041610210002085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Francisco V., Ruiz-Fernández C., Pino J., Mera A., González-Gay M., Gómez R., et al. (2019). Adipokines: linking metabolic syndrome, the immune system, and arthritic diseases. Biochem. Pharmacol. 165 196–206. 10.1016/j.bcp.2019.03.030 [DOI] [PubMed] [Google Scholar]
- Friedman J. (2019). Leptin and the endocrine control of energy balance. Nat. Metab. 1 754–764. 10.1038/s42255-019-0095-y [DOI] [PubMed] [Google Scholar]
- Frölich L., Blum-Degen D., Bernstein H., Engelsberger S., Humrich J., Laufer S., et al. (1998). Brain insulin and insulin receptors in aging and sporadic Alzheimer’s disease. J. Neural Transm. (Vienna). 105 423–438. 10.1007/s007020050068 [DOI] [PubMed] [Google Scholar]
- Fujita Y., Yamashita T. (2019). The effects of leptin on glial cells in neurological diseases. Front. Neurosci. 13:828. 10.3389/fnins.2019.00828 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fulop T., Tripathi S., Rodrigues S., Desroches M., Bunt T., Eiser A., et al. (2021). Targeting impaired antimicrobial immunity in the brain for the treatment of Alzheimer’s disease. Neuropsychiatr Dis. Treat 17 1311–1339. 10.2147/NDT.S264910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fulton S., Pissios P., Manchon R., Stiles L., Frank L., Pothos E. N., et al. (2006). Leptin regulation of the mesoaccumbens dopamine pathway. Neuron 51 811–822. 10.1016/j.neuron.2006.09.006 [DOI] [PubMed] [Google Scholar]
- Fünfschilling U., Supplie L., Mahad D., Boretius S., Saab A., Edgar J., et al. (2012). Glycolytic oligodendrocytes maintain myelin and long-term axonal integrity. Nature 485 517–521. 10.1038/nature11007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gabbouj S., Ryhänen S., Marttinen M., Wittrahm R., Takalo M., Kemppainen S., et al. (2019). Altered insulin signaling in Alzheimer’s disease brain - special emphasis on PI3K-Akt pathway. Front. Neurosci. 13:629. 10.3389/fnins.2019.00629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaber T., Strehl C., Buttgereit F. (2017). Metabolic regulation of inflammation. Nat. Rev. Rheumatol. 13 267–279. 10.1038/nrrheum.2017.37 [DOI] [PubMed] [Google Scholar]
- Gao Y., Vidal-Itriago A., Milanova I., Korpel N., Kalsbeek M., Tom R., et al. (2018). Deficiency of leptin receptor in myeloid cells disrupts hypothalamic metabolic circuits and causes body weight increase. Mol. Metab. 7 155–160. 10.1016/j.molmet.2017.11.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garber C., Soung A., Vollmer L., Kanmogne M., Last A., Brown J., et al. (2019). T cells promote microglia-mediated synaptic elimination and cognitive dysfunction during recovery from neuropathogenic flaviviruses. Nat. Neurosci. 22 1276–1288. 10.1038/s41593-019-0427-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gautron L., Elmquist J. (2011). Sixteen years and counting: an update on leptin in energy balance. J. Clin. Invest. 121 2087–2093. 10.1172/JCI45888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gemma C., Bachstetter A., Bickford P. (2010). Neuron-Microglia dialogue and hippocampal neurogenesis in the aged brain. Aging Dis. 1 232–244. [PMC free article] [PubMed] [Google Scholar]
- Gerozissis K. (2003). Brain insulin: regulation, mechanisms of action and functions. Cell Mol. Neurobiol. 23 873–874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghazanfari N., Gregory J., Devi S., Fernandez-Ruiz D., Beattie L., Mueller S., et al. (2021). CD8(+) and CD4(+) T cells infiltrate into the brain during Plasmodium berghei ANKA infection and form long-term resident memory. J. Immunol. 207 1578–1590. 10.4049/jimmunol.2000773 [DOI] [PubMed] [Google Scholar]
- González-García I., Gruber T., García-Cáceres C. (2021). Insulin action on astrocytes: from energy homeostasis to behaviour. J. Neuroendocr. 33:e12953. 10.1111/jne.12953 [DOI] [PubMed] [Google Scholar]
- Gordleeva S., Kanakov O., Ivanchenko M., Zaikin A., Franceschi C. (2020). Brain aging and garbage cleaning: modelling the role of sleep, glymphatic system, and microglia senescence in the propagation of inflammaging. Semin. Immunopathol. 42 647–665. 10.1007/s00281-020-00816-x [DOI] [PubMed] [Google Scholar]
- Gosztonyi G., Ludwig H., Bode L., Kao M., Sell M., Petrusz P., et al. (2020). Obesity induced by Borna disease virus in rats: key roles of hypothalamic fast-acting neurotransmitters and inflammatory infiltrates. Brain Struct. Funct. 225 1459–1482. 10.1007/s00429-020-02063-2060 [DOI] [PubMed] [Google Scholar]
- Gosztyla M., Brothers H., Robinson S. (2018). Alzheimer’s amyloid-beta is an antimicrobial peptide: a review of the evidence. J. Alzheimers Dis. 62 1495–1506. 10.3233/JAD-171133 [DOI] [PubMed] [Google Scholar]
- Gray S., Meijer R., Barrett E. (2014). Insulin regulates brain function, but how does it get there? Diabetes 63 3992–3997. 10.2337/db14-0340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grieco M., Giorgi A., Gentile M., D’Erme M., Morano S., Maras B., et al. (2019). Glucagon-Like Peptide-1: a focus on neurodegenerative diseases. Front. Neurosci. 13:1112. 10.3389/fnins.2019.01112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gruzdeva O., Borodkina D., Uchasova E., Dyleva Y., Barbarash O. (2019). Leptin resistance: underlying mechanisms and diagnosis. Diab. Metab. Syndr. Obes 12 191–198. 10.2147/DMSO.S182406 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guglielmi V., Colangeli L., D’Adamo M., Sbraccia P. (2021). Susceptibility and severity of viral infections in obesity: lessons from influenza to COVID-19. does leptin play a role? Int. J. Mol. Sci. 22:3183. 10.3390/ijms22063183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo Z., Chen Y., Mao Y., Zheng T., Jiang Y., Yan Y., et al. (2017). Long-term treatment with intranasal insulin ameliorates cognitive impairment, tau hyperphosphorylation, and microglial activation in a streptozotocin-induced Alzheimer’s rat model. Sci. Rep. 7:45971. 10.1038/srep45971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gustafson D., Karlsson C., Skoog I., Rosengren L., Lissner L., Blennow K. (2007). Mid-life adiposity factors relate to blood-brain barrier integrity in late life. J. Int. Med. 262 643–650. 10.1111/j.1365-2796.2007.01869.x [DOI] [PubMed] [Google Scholar]
- Gwak M., Chang S. (2021). Gut-Brain connection: microbiome, gut barrier, and environmental sensors. Immune Netw. 21:e20. 10.4110/in.2021.21.e20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haas C., de Carvalho A., Muller A., Eggen B., Portela L. (2020). Insulin activates microglia and increases COX-2/IL-1beta expression in young but not in aged hippocampus. Brain Res. 1741:146884. 10.1016/j.brainres.2020.146884 [DOI] [PubMed] [Google Scholar]
- Hache G., Osuchowski M., Thiemermann C. (2015). Does insulin protect the brain in mice and man with sepsis? Shock 44:287. 10.1097/SHK.0000000000000423 [DOI] [PubMed] [Google Scholar]
- Hamdy O., Porramatikul S., Al-Ozairi E. (2006). Metabolic obesity: the paradox between visceral and subcutaneous fat. Curr. Diab. Rev. 2 367–373. 10.2174/1573399810602040367 [DOI] [PubMed] [Google Scholar]
- Hargrave K., Woods S., Millington O., Chalmers S., Westrop G., Roberts C. (2019). Multi-Omics studies demonstrate Toxoplasma gondii-induced metabolic reprogramming of murine dendritic cells. Front. Cell Infect. Microbiol. 9:309. 10.3389/fcimb.2019.00309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris S., Harris E. (2015). Herpes simplex virus type 1 and other pathogens are key causative factors in sporadic Alzheimer’s disease. J. Alzheimers. Dis. 48 319–353. 10.3233/JAD-142853 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haslam D., James W. (2005). Obesity. Lancet 366 1197–1209. 10.1016/S0140-6736(05)67483-67481 [DOI] [PubMed] [Google Scholar]
- Hatalski C., Lewis A., Lipkin W. (1997). Borna disease. Emerg. Infect. Dis. 3 129–135. 10.3201/eid0302.970205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helderman J. (1984). Acute regulation of human lymphocyte insulin receptors. analysis by the glucose clamp. J. Clin. Invest. 74 1428–1435. 10.1172/JCI111554 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helderman J., Reynolds T., Strom T. (1978). The insulin receptor as a universal marker of activated lymphocytes. Eur. J. Immunol. 8 589–595. 10.1002/eji.1830080810 [DOI] [PubMed] [Google Scholar]
- Heneka M., Carson M., El Khoury J., Landreth G., Brosseron F., Feinstein D., et al. (2015). Neuroinflammation in Alzheimer’s disease. Lancet Neurol. 14 388–405. 10.1016/S1474-4422(15)70016-70015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heneka M., Golenbock D., Latz E., Morgan D., Brown R. (2020). Immediate and long-term consequences of COVID-19 infections for the development of neurological disease. Alzheimers Res. Ther. 12:69. 10.1186/s13195-020-00640-643 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heni M., Hennige A., Peter A., Siegel-Axel D., Ordelheide A., Krebs N., et al. (2011). Insulin promotes glycogen storage and cell proliferation in primary human astrocytes. PLoS One 6:e21594. 10.1371/journal.pone.0021594 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herden C., Herzog S., Richt J., Nesseler A., Christ M., Failing K., et al. (2000). Distribution of borna disease virus in the brain of rats infected with an obesity-inducing virus strain. Brain Pathol. 10 39–48. 10.1111/j.1750-3639.2000.tb00241.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hernandez-Garzón E., Fernandez A., Perez-Alvarez A., Genis L., Bascuñana P., Fernandez de la Rosa R., et al. (2016). The insulin-like growth factor I receptor regulates glucose transport by astrocytes. Glia 64 1962–1971. 10.1002/glia.23035 [DOI] [PubMed] [Google Scholar]
- Herz J., Filiano A., Smith A., Yogev N., Kipnis J. (2017). Myeloid cells in the central nervous system. Immunity 46 943–956. 10.1016/j.immuni.2017.06.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hierro-Bujalance C., Del Marco A., José Ramos-Rodríguez J., Infante-Garcia C., Bella Gomez-Santos S., Herrera M., et al. (2020). Cell proliferation and neurogenesis alterations in Alzheimer’s disease and diabetes mellitus mixed murine models. J. Neurochem. 154 673–692. 10.1111/jnc.14987 [DOI] [PubMed] [Google Scholar]
- Hirasawa H., Oda S., Nakamura M. (2009). Blood glucose control in patients with severe sepsis and septic shock. World J. Gastroenterol. 15 4132–4136. 10.3748/wjg.15.4132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holden K., Lindquist K., Tylavsky F., Rosano C., Harris T., Yaffe K. (2009). Serum leptin level and cognition in the elderly: findings from the Health ABC Study. Neurobiol. Aging 30 1483–1489. 10.1016/j.neurobiolaging.2007.11.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hommel J., Trinko R., Sears R., Georgescu D., Liu Z., Gao X., et al. (2006). Leptin receptor signaling in midbrain dopamine neurons regulates feeding. Neuron 51 801–810. 10.1016/j.neuron.2006.08.023 [DOI] [PubMed] [Google Scholar]
- Hotamisligil G. (2017). Inflammation, metaflammation and immunometabolic disorders. Nature 542 177–185. 10.1038/nature21363 [DOI] [PubMed] [Google Scholar]
- Howard J., Lord G., Matarese G., Vendetti S., Ghatei M., Ritter M., et al. (1999). Leptin protects mice from starvation-induced lymphoid atrophy and increases thymic cellularity in ob/ob mice. J. Clin. Invest. 104 1051–1059. 10.1172/JCI6762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu A., Aronoff D., Phipps J., Goel D., Mancuso P. (2007). Leptin improves pulmonary bacterial clearance and survival in ob/ob mice during Pneumococcal pneumonia. Clin. Exp. Immunol. 150 332–339. 10.1111/j.1365-2249.2007.03491.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C., Lue J., Cheng T., Tsai Y. (2020). Glycemic control with insulin attenuates sepsis-associated encephalopathy by inhibiting glial activation via the suppression of the nuclear factor kappa B and mitogen-activated protein kinase signaling pathways in septic rats. Brain Res. 1738:146822. 10.1016/j.brainres.2020.146822 [DOI] [PubMed] [Google Scholar]
- Huizinga G., Singer B., Singer K. (2020). The Collision of Meta-Inflammation and SARS-CoV-2 Pandemic Infection. Endocrinology 161:bqaa154. 10.1210/endocr/bqaa154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imamura T., Yanagihara Y., Ohyagi Y., Nakamura N., Iinuma K., Yamasaki R., et al. (2020). Insulin deficiency promotes formation of toxic amyloid-beta42 conformer co-aggregating with hyper-phosphorylated tau oligomer in an Alzheimer’s disease model. Neurobiol. Dis. 137:104739. 10.1016/j.nbd.2020.104739 [DOI] [PubMed] [Google Scholar]
- Iqbal U., Zeng E., Pasinetti G. (2020). The use of antimicrobial and antiviral drugs in Alzheimer’s disease. Int. J. Mol. Sci. 21:4920. 10.3390/ijms21144920 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irie F., Fitzpatrick A., Lopez O., Kuller L., Peila R., Newman A., et al. (2008). Enhanced risk for Alzheimer disease in persons with type 2 diabetes and APOE epsilon4: the cardiovascular health study cognition study. Arch. Neurol. 65 89–93. 10.1001/archneurol.2007.29 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Izquierdo A., Crujeiras A., Casanueva F., Carreira M. (2019). Leptin, obesity, and leptin resistance: where are we 25 years later? Nutrients 11:2704. 10.3390/nu11112704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeon C., Haan M., Cheng C., Clayton E., Mayeda E., Miller J., et al. (2012). Helicobacter pylori infection is associated with an increased rate of diabetes. Diab. Care 35 520–525. 10.2337/dc11-1043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jha M., Morrison B. (2018). Glia-neuron energy metabolism in health and diseases: new insights into the role of nervous system metabolic transporters. Exp. Neurol. 309 23–31. 10.1016/j.expneurol.2018.07.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji Y., Park S., Park H., Hwang E., Shin H., Pot B., et al. (2018). Modulation of active gut microbiota by Lactobacillus rhamnosus GG in a diet induced obesity murine model. Front. Microbiol. 9:710. 10.3389/fmicb.2018.00710 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang C., Li G., Huang P., Liu Z., Zhao B. (2017). The gut microbiota and Alzheimer’s disease. J. Alzheimers Dis. 58 1–15. 10.3233/JAD-161141 [DOI] [PubMed] [Google Scholar]
- Jiménez-Cortegana C., López-Saavedra A., Sánchez-Jiménez F., Pérez-Pérez A., Castiñeiras J., Virizuela-Echaburu J., et al. (2021). Leptin, both bad and good actor in Cancer. Biomolecules 11:913. 10.3390/biom11060913 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanagasingam S., Chukkapalli S., Welbury R., Singhrao S. (2020). Porphyromonas gingivalis is a strong risk factor for Alzheimer’s disease. J. Alzheimers Dis. Rep. 4 501–511. 10.3233/ADR-200250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanoski S., Zhang Y., Zheng W., Davidson T. (2010). The effects of a high-energy diet on hippocampal function and blood-brain barrier integrity in the rat. J. Alzheimers Dis. 21 207–219. 10.3233/JAD-2010-091414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karman J., Ling C., Sandor M., Fabry Z. (2004). Initiation of immune responses in brain is promoted by local dendritic cells. J. Immunol. 173 2353–2361. 10.4049/jimmunol.173.4.2353 [DOI] [PubMed] [Google Scholar]
- Katsouri L., Birch A., Renziehausen A., Zach C., Aman Y., Steeds H., et al. (2020). Ablation of reactive astrocytes exacerbates disease pathology in a model of Alzheimer’s disease. Glia 68 1017–1030. 10.1002/glia.23759 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaunzner U., Miller M., Gottfried-Blackmore A., Gal-Toth J., Felger J., McEwen B., et al. (2012). Accumulation of resident and peripheral dendritic cells in the aging CNS. Neurobiol. Aging 33 681–693.e1. 10.1016/j.neurobiolaging.2010.06.007 [DOI] [PubMed] [Google Scholar]
- Kealy J., Murray C., Griffin E., Lopez-Rodriguez A., Healy D., Tortorelli L., et al. (2020). Acute inflammation alters brain energy metabolism in mice and humans: role in suppressed spontaneous activity, impaired cognition, and delirium. J. Neurosci. 40 5681–5696. 10.1523/JNEUROSCI.2876-19.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keane L., Antignano I., Riechers S., Zollinger R., Dumas A., Offermann N., et al. (2021). mTOR-dependent translation amplifies microglia priming in aging mice. J. Clin. Invest. 131:e132727. 10.1172/JCI132727 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kellar D., Craft S. (2020). Brain insulin resistance in Alzheimer’s disease and related disorders: mechanisms and therapeutic approaches. Lancet Neurol. 19 758–766. 10.1016/S1474-4422(20)30231-30233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keller A., Rohde J., Raymond K., Heitmann B. (2015). Association between periodontal disease and overweight and obesity: a systematic review. J. Periodontol. 86 766–776. 10.1902/jop.2015.140589 [DOI] [PubMed] [Google Scholar]
- Khan S., Barrington G., Bettiol S., Barnett T., Crocombe L. (2018). Is overweight/obesity a risk factor for periodontitis in young adults and adolescents: a systematic review. Obes. Rev. 19 852–883. 10.1111/obr.12668 [DOI] [PubMed] [Google Scholar]
- Khan S., Luck H., Winer S., Winer D. (2021). Emerging concepts in intestinal immune control of obesity-related metabolic disease. Nat. Commun. 12:2598. 10.1038/s41467-021-22727-22727 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kierdorf K., Prinz M. (2017). Microglia in steady state. J. Clin. Invest. 127 3201–3209. 10.1172/JCI90602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim B., Kelschenbach J., Borjabad A., Hadas E., He H., Potash M., et al. (2019). Intranasal insulin therapy reverses hippocampal dendritic injury and cognitive impairment in a model of HIV-associated neurocognitive disorders in EcoHIV-infected mice. AIDS 33 973–984. 10.1097/QAD.0000000000002150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J., Barua S., Jeong Y., Lee J. (2020). Adiponectin: the potential regulator and therapeutic target of obesity and Alzheimer’s disease. Int. J. Mol. Sci. 21:6419. 10.3390/ijms21176419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kivipelto M., Ngandu T., Laatikainen T., Winblad B., Soininen H., Tuomilehto J. (2006). Risk score for the prediction of dementia risk in 20 years among middle aged people: a longitudinal, population-based study. Lancet Neurol. 5 735–741. 10.1016/S1474-4422(06)70537-70533 [DOI] [PubMed] [Google Scholar]
- Knapp S. (2013). Diabetes and infection: is there a link?-A mini-review. Gerontology 59 99–104. 10.1159/000345107 [DOI] [PubMed] [Google Scholar]
- Koh G., Peacock S., van der Poll T., Wiersinga W. (2012). The impact of diabetes on the pathogenesis of sepsis. Eur. J. Clin. Microbiol. Infect. Dis. 31 379–388. 10.1007/s10096-011-1337-1334 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar D., Choi S., Washicosky K., Eimer W., Tucker S., Ghofrani J., et al. (2016). Amyloid-β peptide protects against microbial infection in mouse and worm models of Alzheimer’s disease. Sci. Transl. Med. 8:340ra72. 10.1126/scitranslmed.aaf1059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar M., Roe K., Nerurkar P., Orillo B., Thompson K., Verma S., et al. (2014). Reduced immune cell infiltration and increased pro-inflammatory mediators in the brain of Type 2 diabetic mouse model infected with West Nile virus. J. Neuroinflamm. 11:80. 10.1186/1742-2094-11-80 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumari R., Kumar S., Kant R. (2019). An update on metabolic syndrome: metabolic risk markers and adipokines in the development of metabolic syndrome. Diabetes Metab. Syndr. 13 2409–2417. 10.1016/j.dsx.2019.06.005 [DOI] [PubMed] [Google Scholar]
- Kuo C., Lu C., Chang Y., Li C. (2018). Population-Based cohort study on dementia risk in patients with type 1 diabetes mellitus. Neuroepidemiology 50 57–62. 10.1159/000486719 [DOI] [PubMed] [Google Scholar]
- La Cava A., Matarese G. (2004). The weight of leptin in immunity. Nat. Rev. Immunol. 4 371–379. 10.1038/nri1350 [DOI] [PubMed] [Google Scholar]
- Labandeira-Garcia J., Costa-Besada M., Labandeira C., Villar-Cheda B., Rodríguez-Perez A. (2017). Insulin-Like growth factor-1 and neuroinflammation. Front. Aging Neurosci. 9:365. 10.3389/fnagi.2017.00365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lafrance V., Inoue W., Kan B., Luheshi G. (2010). Leptin modulates cell morphology and cytokine release in microglia. Brain Behav. Immun. 24 358–365. 10.1016/j.bbi.2009.11.003 [DOI] [PubMed] [Google Scholar]
- Lana D., Ugolini F., Giovannini M. (2020). Space-Dependent glia-neuron interplay in the hippocampus of transgenic models of β-amyloid deposition. Int. J. Mol. Sci. 21:9441. 10.3390/ijms21249441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lana D., Ugolini F., Nosi D., Wenk G., Giovannini M. (2021). The emerging role of the interplay among astrocytes, microglia, and neurons in the hippocampus in health and disease. Front. Aging Neurosci. 13:651973. 10.3389/fnagi.2021.651973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landrith T., Sureshchandra S., Rivera A., Jang J., Rais M., Nair M., et al. (2017). CD103+ CD8 T cells in the Toxoplasma-infected brain exhibit a tissue-resident memory transcriptional profile. Front. Immunol. 8:335. 10.3389/fimmu.2017.00335 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larabee C., Neely O., Domingos A. (2020). Obesity: a neuroimmunometabolic perspective. Nat. Rev. Endocrinol. 16 30–43. 10.1038/s41574-019-0283-286 [DOI] [PubMed] [Google Scholar]
- Lazar V., Ditu L., Pircalabioru G., Gheorghe I., Curutiu C., Holban A., et al. (2018). Aspects of gut microbiota and immune system interactions in infectious diseases, immunopathology, and cancer. Front. Immunol. 9:1830. 10.3389/fimmu.2018.01830 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leblhuber F., Ehrlich D., Steiner K., Geisler S., Fuchs D., Lanser L., et al. (2021). The immunopathogenesis of Alzheimer’s disease is related to the composition of gut microbiota. Nutrients 13:361. 10.3390/nu13020361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee G., Proenca R., Montez J., Carroll K., Darvishzadeh J., Lee J., et al. (1996). Abnormal splicing of the leptin receptor in diabetic mice. Nature 378 632–635. 10.1038/379632a0 [DOI] [PubMed] [Google Scholar]
- Leonard J. (2017). Central nervous system tuberculosis. Microbiol. Spectr. 5:11. 10.1128/microbiolspec.TNMI7-0044-2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leshan R., Louis G., Jo Y., Rhodes C., Münzberg H., Myers M. J. (2009). Direct innervation of GnRH neurons by metabolic- and sexual odorant-sensing leptin receptor neurons in the hypothalamic ventral premammillary nucleus. J. Neurosci. 29 3138–3147. 10.1523/JNEUROSCI.0155-09.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lester-Coll N., Rivera E., Soscia S., Doiron K., Wands J., de la Monte S. (2006). Intracerebral streptozotocin model of type 3 diabetes: relevance to sporadic Alzheimer’s disease. J. Alzheimers Dis. 9 13–33. 10.3233/jad-2006-9102 [DOI] [PubMed] [Google Scholar]
- Leyns C., Holtzman D. (2017). Glial contributions to neurodegeneration in tauopathies. Mol. Neurodegener. 12:50. 10.1186/s13024-017-0192-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Q., Barres B. (2018). Microglia and macrophages in brain homeostasis and disease. Nat. Rev. Immunol. 18 225–242. 10.1038/nri.2017.125 [DOI] [PubMed] [Google Scholar]
- Li Y., Severance E., Viscidi R., Yolken R., Xiao J. (2019). Persistent toxoplasma infection of the brain induced neurodegeneration associated with activation of complement and microglia. Infect. Immun. 87:e00139-19. 10.1128/IAI.00139-119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Libertucci J., Young V. (2019). The role of the microbiota in infectious diseases. Nat. Microbiol. 4 35–45. 10.1038/s41564-018-0278-274 [DOI] [PubMed] [Google Scholar]
- Liddelow S., Guttenplan K., Clarke L., Bennett F., Bohlen C., Schirmer L., et al. (2017). Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541 481–487. 10.1038/nature21029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linnerbauer M., Wheeler M., Quintana F. (2020). Astrocyte crosstalk in CNS inflammation. Neuron 108 608–622. 10.1016/j.neuron.2020.08.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lord G., Matarese G., Howard J., Baker R., Bloom S., Lechler R. (1998). Leptin modulates the T-cell immune response and reverses starvation-induced immunosuppression. Nature 394 897–901. 10.1038/29795 [DOI] [PubMed] [Google Scholar]
- Lövheim H., Olsson J., Weidung B., Johansson A., Eriksson S., Hallmans G., et al. (2018). Interaction between cytomegalovirus and herpes simplex virus Type 1 associated with the risk of Alzheimer’s disease development. J. Alzheimers Dis. 61 939–945. 10.3233/JAD-161305 [DOI] [PubMed] [Google Scholar]
- Lue L., Kuo Y., Beach T., Walker D. (2010). Microglia activation and anti-inflammatory regulation in Alzheimer’s disease. Mol. Neurobiol. 41 115–128. 10.1007/s12035-010-8106-8108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lutshumba J., Nikolajczyk B., Bachstetter A. (2021). Dysregulation of systemic immunity in aging and dementia. Front. Cell Neurosci. 15:652111. 10.3389/fncel.2021.652111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madinier A., Bertrand N., Mossiat C., Prigent-Tessier A., Beley A., Marie C., et al. (2009). Microglial involvement in neuroplastic changes following focal brain ischemia in rats. PLoS One 4:e8101. 10.1371/journal.pone.0008101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mallat M., Lima F., Gervais A., Colin C., Moura Neto V. (2002). New insights into the role of thyroid hormone in the CNS: the microglial track. Mol. Psychiatry 7 7–8. 10.1038/sj.mp.4000988 [DOI] [PubMed] [Google Scholar]
- Mamik M., Asahchop E., Chan W., Zhu Y., Branton W., McKenzie B., et al. (2016). Insulin treatment prevents neuroinflammation and neuronal injury with restored neurobehavioral function in models of HIV/AIDS neurodegeneration. J. Neurosci. 36 10683–10695. 10.1523/JNEUROSCI.1287-16.2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mancuso P., Gottschalk A., Phare S., Peters-Golden M., Lukacs N., Huffnagle G. (2002). Leptin-deficient mice exhibit impaired host defense in Gram-negative pneumonia. J. Immunol. 168 4018–4024. 10.4049/jimmunol.168.8.4018 [DOI] [PubMed] [Google Scholar]
- Martella V., Elia G., Buonavoglia C. (2008). Canine distemper virus. Vet. Clin. North Am. Small Anim. Pr. 38 787–797. 10.1016/j.cvsm.2008.02.007 [DOI] [PubMed] [Google Scholar]
- Martin D., Torrance B., Haynes L., Bartley J. (2021). Targeting aging: lessons learned from immunometabolism and cellular senescence. Front. Immunol. 12:714742. 10.3389/fimmu.2021.714742 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez-Gutierrez M., Ruiz-Saenz J. (2016). Diversity of susceptible hosts in canine distemper virus infection: a systematic review and data synthesis. BMC Vet. Res. 12:78. 10.1186/s12917-016-0702-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martín-Romero C., Sánchez-Margalet V. (2001). Human leptin activates PI3K and MAPK pathways in human peripheral blood mononuclear cells: possible role of Sam68. Cell Immunol. 212 83–91. 10.1006/cimm.2001.1851 [DOI] [PubMed] [Google Scholar]
- Matarese G., La Cava A., Sanna V., Lord G., Lechler R., Fontana S., et al. (2002). Balancing susceptibility to infection and autoimmunity: a role for leptin? Trends Immunol. 23 182–187. 10.1016/s1471-4906(02)02188-2189 [DOI] [PubMed] [Google Scholar]
- Maurya R., Bhattacharya P., Dey R., Nakhasi H. (2018). Leptin functions in infectious diseases. Front. Immunol. 9:2741. 10.3389/fimmu.2018.02741 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazucanti C., Liu Q., Lang D., Huang N., O’Connell J., Camandola S., et al. (2019). Release of insulin produced by the choroid plexus is regulated by serotonergic signaling. JCI Insight 4:e131682. 10.1172/jci.insight.131682 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mei F., Xie M., Huang X., Long Y., Lu X., Wang X., et al. (2020). Porphyromonas gingivalis and its systemic impact: current status. Pathogens 9:944. 10.3390/pathogens9110944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mejido D., Peny J., Vieira M., Ferreira S., De Felice F. (2020). Insulin and leptin as potential cognitive enhancers in metabolic disorders and Alzheimer’s disease. Neuropharmacology 171:108115. 10.1016/j.neuropharm.2020.108115 [DOI] [PubMed] [Google Scholar]
- Merino B., Díez-Fernández C., Ruiz-Gayo M., Somoza B. (2006). Choroid plexus epithelial cells co-express the long and short form of the leptin receptor. Neurosci. Lett. 393 269–272. 10.1016/j.neulet.2005.10.003 [DOI] [PubMed] [Google Scholar]
- Michels M., Vieira A., Vuolo F., Zapelini H., Mendonça B., Mina F., et al. (2015). The role of microglia activation in the development of sepsis-induced long-term cognitive impairment. Brain Behav. Immun. 43 54–59. 10.1016/j.bbi.2014.07.002 [DOI] [PubMed] [Google Scholar]
- Mitchell C., Begg D. (2021). The regulation of food intake by insulin in the central nervous system. J. Neuroendocr. 33:e12952. 10.1111/jne.12952 [DOI] [PubMed] [Google Scholar]
- Mockus T., Shwetank, Lauver M., Ren H., Netherby C., Salameh T., et al. (2018). CD4 T cells control development and maintenance of brain-resident CD8 T cells during polyomavirus infection. PLoS Pathog. 14:e1007365. 10.1371/journal.ppat.1007365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohanty P., Hamouda W., Garg R., Aljada A., Ghanim H., Dandona P. (2000). Glucose challenge stimulates reactive oxygen species (ROS) generation by leucocytes. J. Clin. Endocrinol. Metab. 85 2970–2973. 10.1210/jcem.85.8.6854 [DOI] [PubMed] [Google Scholar]
- Möhle L., Israel N., Paarmann K., Krohn M., Pietkiewicz S., Müller A., et al. (2016). Chronic Toxoplasma gondii infection enhances beta-amyloid phagocytosis and clearance by recruited monocytes. Acta Neuropathol. Commun. 4:25. 10.1186/s40478-016-0293-298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moir R., Lathe R., Tanzi R. (2018). The antimicrobial protection hypothesis of Alzheimer’s disease. Alzheimers Dement. 14 1602–1614. 10.1016/j.jalz.2018.06.3040 [DOI] [PubMed] [Google Scholar]
- Montagne A., Barnes S., Sweeney M., Halliday M., Sagare A., Zhao Z., et al. (2015). Blood-brain barrier breakdown in the aging human hippocampus. Neuron 85 296–302. 10.1016/j.neuron.2014.12.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monteiro L., Pereira J., Palhinha L., Moraes-Vieira P. (2019). Leptin in the regulation of the immunometabolism of adipose tissue-macrophages. J. Leukoc. Biol. 106 703–716. 10.1002/JLB.MR1218-478R [DOI] [PubMed] [Google Scholar]
- Morrison C. (2009). Leptin signaling in brain: a link between nutrition and cognition? Biochim Biophys. Acta 1792 401–408. 10.1016/j.bbadis.2008.12.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller L., Gorter K., Hak E., Goudzwaard W., Schellevis F., Hoepelman A., et al. (2005). Increased risk of common infections in patients with type 1 and type 2 diabetes mellitus. Clin. Infect. Dis. 41 281–288. 10.1086/431587 [DOI] [PubMed] [Google Scholar]
- Murray K., Garcia M., Rahbar M., Martinez D., Khuwaja S., Arafat R., et al. (2014). Survival analysis, long-term outcomes, and percentage of recovery up to 8 years post-infection among the Houston West Nile virus cohort. PLoS One 9:e102953. 10.1371/journal.pone.0102953 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Myers M., Cowley M., Münzberg H. (2008). Mechanisms of leptin action and leptin resistance. Annu. Rev. Physiol. 70 537–556. 10.1146/annurev.physiol.70.113006.100707 [DOI] [PubMed] [Google Scholar]
- Myhre C., Thygesen C., Villadsen B., Vollerup J., Ilkjær L., Krohn K., et al. (2019). Microglia express insulin-like growth factor-1 in the hippocampus of aged APP swe/PS1 ΔE9 transgenic mice. Front. Cell Neurosci. 13:308. 10.3389/fncel.2019.00308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagajyothi F., Zhao D., Machado F., Weiss L., Schwartz G., Desruisseaux M., et al. (2010). Crucial role of the central leptin receptor in murine Trypanosoma cruzi infection. J. Infect. Dis. 202 1104–1113. 10.1086/656189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakae J., Kido Y., Accili D. (2001). Distinct and overlapping functions of insulin and IGF-I receptors. Endocr. Rev. 22 818–835. 10.1210/edrv.22.6.0452 [DOI] [PubMed] [Google Scholar]
- Naranjo V., Contreras A., Merino B., Plaza A., Lorenzo M., García-Cáceres C., et al. (2020). Specific deletion of the astrocyte leptin receptor induces changes in hippocampus glutamate metabolism, synaptic transmission and plasticity. Neuroscience 447 182–190. 10.1016/j.neuroscience.2019.10.005 [DOI] [PubMed] [Google Scholar]
- Narayan O., Herzog S., Frese K., Scheefers H., Rott R. (1983). Behavioral disease in rats caused by immunopathological responses to persistent borna virus in the brain. Science 220 1401–1403. 10.1126/science.6602380 [DOI] [PubMed] [Google Scholar]
- Nascimento G., Leite F., Do L., Peres K., Correa M., Demarco F., et al. (2015). Is weight gain associated with the incidence of periodontitis? a systematic review and meta-analysis. J. Clin. Periodontol. 42 495–505. 10.1111/jcpe.12417 [DOI] [PubMed] [Google Scholar]
- Nayeri Chegeni T., Sarvi S., Moosazadeh M., Sharif M., Aghayan S., Amouei A., et al. (2019). Is Toxoplasma gondii a potential risk factor for Alzheimer’s disease? a systematic review and meta-analysis. Microb. Pathog. 137:103751. 10.1016/j.micpath.2019.103751 [DOI] [PubMed] [Google Scholar]
- Nazareth L., Walkden H., Chacko A., Delbaz A., Shelper T., Armitage C., et al. (2021). Chlamydia muridarum can invade the central nervous system via the olfactory and trigeminal nerves and infect peripheral nerve glial cells. Front. Cell Infect. Microbiol. 10:607779. 10.3389/fcimb.2020.607779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Netherby-Winslow C., Ayers K., Lukacher A. (2021). Balancing inflammation and central nervous system homeostasis: T cell receptor signaling in antiviral brain T(RM) formation and function. Front. Immunol. 11:624144. 10.3389/fimmu.2020.624144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neves F., Marques P., Barros-Aragão F., Nunes J., Venancio A., Cozachenco D., et al. (2018). Brain-Defective insulin signaling is associated to late cognitive impairment in post-septic mice. Mol. Neurobiol. 55 435–444. 10.1007/s12035-016-0307-303 [DOI] [PubMed] [Google Scholar]
- Ninomiya Y., Shigemura N., Yasumatsu K., Ohta R., Sugimoto K., Nakashima K., et al. (2002). Leptin and sweet taste. Vitam Horm. 64 221–248. 10.1016/s0083-6729(02)64007-64005 [DOI] [PubMed] [Google Scholar]
- Noriega-Prieto J., Maglio L., Zegarra-Valdivia J., Pignatelli J., Fernandez A., Martinez-Rachadell L., et al. (2021). Astrocytic IGF-IRs induce adenosine-mediated inhibitory downregulation and improve sensory discrimination. J. Neurosci. 41 4768–4781. 10.1523/JNEUROSCI.0005-21.2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Obradovic M., Sudar-Milovanovic E., Soskic S., Essack M., Arya S., Stewart A., et al. (2021). Leptin and obesity: role and clinical implication. Front. Endocrinol. 12:585887. 10.3389/fendo.2021.585887 [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Neill C., Kiely A., Coakley M., Manning S., Long-Smith C. (2012). Insulin and IGF-1 signalling: longevity, protein homoeostasis and Alzheimer’s disease. Biochem. Soc. Trans. 40 721–727. 10.1042/BST20120080 [DOI] [PubMed] [Google Scholar]
- O’Neill L., Kishton R., Rathmell J. (2016). A guide to immunometabolism for immunologists. Nat. Rev. Immunol. 16 553–565. 10.1038/nri.2016.70 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ott A., Stolk R., van Harskamp F., Pols H., Hofman A., Breteler M. (1999). Diabetes mellitus and the risk of dementia: the rotterdam study. Neurology 53 1937–1942. 10.1212/wnl.53.9.1937 [DOI] [PubMed] [Google Scholar]
- Paik I., Kotler D. (2011). The prevalence and pathogenesis of diabetes mellitus in treated HIV-infection. Best Pr. Res. Clin. Endocrinol. Metab. 25 469–478. 10.1016/j.beem.2011.04.003 [DOI] [PubMed] [Google Scholar]
- Pandit R., Beerens S., Adan R. (2017). Role of leptin in energy expenditure: the hypothalamic perspective. Am. J. Physiol. Regul. Integr. Comp. Physiol. 312 R938–R947. 10.1152/ajpregu.00045.2016 [DOI] [PubMed] [Google Scholar]
- Papathanassoglou E., El-Haschimi K., Li X., Matarese G., Strom T., Mantzoros C. (2006). Leptin receptor expression and signaling in lymphocytes: kinetics during lymphocyte activation, role in lymphocyte survival, and response to high fat diet in mice. J. Immunol. 176 7745–7752. 10.4049/jimmunol.176.12.7745 [DOI] [PubMed] [Google Scholar]
- Parkhurst C., Yang G., Ninan I., Savas J., Yates J., III, Lafaille J., et al. (2013). Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell 155 1596–1609. 10.1016/j.cell.2013.11.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasarica M., Dhurandhar N. (2007). Infectobesity: obesity of infectious origin. Adv. Food Nutr. Res. 52 61–102. 10.1016/S1043-4526(06)52002-52009 [DOI] [PubMed] [Google Scholar]
- Patterson E., Ryan P., Cryan J., Dinan T., Ross R., Fitzgerald G., et al. (2016). Gut microbiota, obesity and diabetes. Postgr. Med J. 92 286–300. 10.1136/postgradmedj-2015-133285 [DOI] [PubMed] [Google Scholar]
- Paz-Filho G., Babikian T., Asarnow R., Delibasi T., Esposito K., Erol H., et al. (2008). Leptin replacement improves cognitive development. PLoS One 3:e3098. 10.1371/journal.pone.0003098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peila R., Rodriguez B., Launer L. Honolulu-Asia Aging, and Study. (2002). Type 2 diabetes, APOE gene, and the risk for dementia and related pathologies: the honolulu-asia aging study. Diabetes Metab. Res. Rev. 51 1256–1262. 10.2337/diabetes.51.4.1256 [DOI] [PubMed] [Google Scholar]
- Pemberton L., Brew B. (2001). Cerebrospinal fluid S-100beta and its relationship with AIDS dementia complex. J. Clin. Virol. 22 249–253. 10.1016/s1386-6532(01)00196-192 [DOI] [PubMed] [Google Scholar]
- Pinteaux E., Inoue W., Schmidt L., Molina-Holgado F., Rothwell N., Luheshi G. (2007). Leptin induces interleukin-1beta release from rat microglial cells through a caspase 1 independent mechanism. J. Neurochem. 102 826–833. 10.1111/j.1471-4159.2007.04559.x [DOI] [PubMed] [Google Scholar]
- Poole S., Singhrao S., Chukkapalli S., Rivera M., Velsko I., Kesavalu L., et al. (2015). Active invasion of Porphyromonas gingivalis and infection-induced complement activation in ApoE-/- mice brains. J. Alzheimers Dis. 43 67–80. 10.3233/JAD-140315 [DOI] [PubMed] [Google Scholar]
- Pratap A., Holsinger R. (2020). Altered brain leptin and leptin receptor expression in the 5XFAD mouse model of Alzheimer’s disease. Pharm 13:401. 10.3390/ph13110401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prinz M., Erny D., Hagemeyer N. (2017). Ontogeny and homeostasis of CNS myeloid cells. Nat. Immunol. 18 385–392. 10.1038/ni.3703 [DOI] [PubMed] [Google Scholar]
- Procaccini C., Jirillo E., Matarese G. (2012). Leptin as an immunomodulator. Mol. Asp. Med. 33 35–45. 10.1016/j.mam.2011.10.012 [DOI] [PubMed] [Google Scholar]
- Procaccini C., La Rocca C., Carbone F., De Rosa V., Galgani M., Matarese G. (2017). Leptin as immune mediator: interaction between neuroendocrine and immune system. Dev. Comp. Immunol. 66 120–129. 10.1016/j.dci.2016.06.006 [DOI] [PubMed] [Google Scholar]
- Próchnicki T., Latz E. (2017). Inflammasomes on the crossroads of innate immune recognition and metabolic control. Cell Metab. 26 71–93. 10.1016/j.cmet.2017.06.018 [DOI] [PubMed] [Google Scholar]
- Prodinger C., Bunse J., Krüger M., Schiefenhövel F., Brandt C., Laman J., et al. (2011). CD11c-expressing cells reside in the juxtavascular parenchyma and extend processes into the glia limitans of the mouse nervous system. Acta Neuropathol. 121 445–458. 10.1007/s00401-010-0774-y [DOI] [PubMed] [Google Scholar]
- Pugazhenthi S., Qin L., Reddy P. H. (2017). Common neurodegenerative pathways in obesity, diabetes, and Alzheimer’s disease. Biochim Biophys. Acta Mol. Basis Dis. 1863 1037–1045. 10.1016/j.bbadis.2016.04.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pulido-Mendez M., De Sanctis J., Rodriguez-Acosta A. (2002). Leptin and leptin receptors during malaria infection in mice. Folia Parasitol. 49 249–251. 10.14411/fp.2002.046 [DOI] [PubMed] [Google Scholar]
- Radigan K., Morales-Nebreda L., Soberanes S., Nicholson T., Nigdelioglu R., Cho T., et al. (2014). Impaired clearance of influenza a virus in obese, leptin receptor deficient mice is independent of leptin signaling in the lung epithelium and macrophages. PLoS One 9:e108138. 10.1371/journal.pone.0108138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahman M., Hossain K., Das S., Kundu S., Adegoke E., Rahman M., et al. (2021). Role of insulin in health and disease: an update. Int. J. Mol. Sci. 22:6403. 10.3390/ijms22126403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ransohoff R., Brown M. (2012). Innate immunity in the central nervous system. J. Clin. Invest. 122 1164–1171. 10.1172/JCI58644 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao R., Li Q., Odunsi K., Shrikant P. (2010). The mTOR kinase determines effector versus memory CD8+ T cell fate by regulating the expression of transcription factors T-bet and Eomesodermin. Immunity 32 67–78. 10.1016/j.immuni.2009.10.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratter J., van Heck J., Rooijackers H., Jansen H., van Poppel P., Tack C., et al. (2021). Insulin acutely activates metabolism of primary human monocytes and promotes a proinflammatory phenotype. J. Leukoc Biol. 10.1002/JLB.3AB0120-019RR Online ahead of print. [DOI] [PubMed] [Google Scholar]
- Rayfield E., Ault M., Keusch G., Brothers M., Nechemias C., Smith H. (1982). Infection and diabetes: the case for glucose control. Am. J. Med. 72 439–450. 10.1016/0002-9343(82)90511-3 [DOI] [PubMed] [Google Scholar]
- Rebello C., Kirwan J., Greenway F. (2020). Obesity, the most common comorbidity in SARS-CoV-2: is leptin the link? Int. J. Obes (Lond) 44 1810–1817. 10.1038/s41366-020-0640-645 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson A., Liu F., Adamo M., Van Remmen H., Nelson J. (2004). The role of insulin and insulin-like growth factor-I in mammalian ageing. Best Pr. Res. Clin. Endocrinol. Metab. 18 393–406. 10.1016/j.beem.2004.02.002 [DOI] [PubMed] [Google Scholar]
- Rodriguez J., Delzenne N. (2021). Modulation of the gut microbiota-adipose tissue-muscle interactions by prebiotics. J. Endocrinol. 249 R1–R23. 10.1530/JOE-20-0499 [DOI] [PubMed] [Google Scholar]
- Romanenko M., Kholin V., Koliada A., Vaiserman A. (2021). Nutrition, gut microbiota, and Alzheimer’s disease. Front. Psychiatry 12:712673. 10.3389/fpsyt.2021.712673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romaní-Pérez M., Bullich-Vilarrubias C., López-Almela I., Liébana-García R., Olivares M., Sanz Y. (2021). The microbiota and the gut-brain axis in controlling food intake and energy homeostasis. Int. J. Mol. Sci. 22:5830. 10.3390/ijms22115830 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosko L., Smith V., Yamazaki R., Huang J. (2019). Oligodendrocyte bioenergetics in health and disease. Neuroscientist 25 334–343. 10.1177/1073858418793077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rummel C., Inoue W., Poole S., Luheshi G. (2010). Leptin regulates leukocyte recruitment into the brain following systemic LPS-induced inflammation. Mol. Psychiatry 15 523–534. 10.1038/mp.2009.98 [DOI] [PubMed] [Google Scholar]
- Salehi Z., Mashayekhi F., Naji M., Pandamooz S. (2009). Insulin-like growth factor-1 and insulin-like growth factor binding proteins in cerebrospinal fluid during the development of mouse embryos. J. Clin. Neurosci. 16 950–953. 10.1016/j.jocn.2008.09.018 [DOI] [PubMed] [Google Scholar]
- Salem D., Salem N., Hendawy S. (2021). Association between Toxoplasma gondii infection and metabolic syndrome in obese adolescents: a possible immune-metabolic link. Parasitol. Int. 83:102343. 10.1016/j.parint.2021.102343 [DOI] [PubMed] [Google Scholar]
- Sánchez-Margalet V., Martín-Romero C., Santos-Alvarez J., Goberna R., Najib S., Gonzalez-Yanes C. (2003). Role of leptin as an immunomodulator of blood mononuclear cells: mechanisms of action. Clin. Exp. Immunol. 133 11–19. 10.1046/j.1365-2249.2003.02190.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sánchez-Pozo C., Rodriguez-baño J., Domínguez-Castellano A., Muniain M., Goberna R., Sánchez-Margalet V. (2003). Leptin stimulates the oxidative burst in control monocytes but attenuates the oxidative burst in monocytes from HIV-infected patients. Clin. Exp. Immunol. 134 464–469. 10.1111/j.1365-2249.2003.02321.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sankar S., Infante-Garcia C., Weinstock L., Ramos-Rodriguez J., Hierro-Bujalance C., Fernandez-Ponce C., et al. (2020). Amyloid beta and diabetic pathology cooperatively stimulate cytokine expression in an Alzheimer’s mouse model. J. Neuroinflamm. 17:38. 10.1186/s12974-020-1707-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schafer D., Lehrman E., Kautzman A., Koyama R., Mardinly A., Yamasaki R., et al. (2012). Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron 74 691–705. 10.1016/j.neuron.2012.03.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scherer T., Sakamoto K., Buettner C. (2021). Brain insulin signalling in metabolic homeostasis and disease. Nat. Rev. Endocrinol. 17 468–483. 10.1038/s41574-021-00498-x [DOI] [PubMed] [Google Scholar]
- Schertzer J., Lam T. (2021). Peripheral and central regulation of insulin by the intestine and microbiome. Am. J. Physiol. Endocrinol. Metab. 320 E234–E239. 10.1152/ajpendo.00547.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwemmle M. (2001). Borna disease virus infection in psychiatric patients: are we on the right track? Lancet Infect. Dis. 1 46–52. 10.1016/S1473-3099(01)00021-24 [DOI] [PubMed] [Google Scholar]
- Scott M., Lachey J., Sternson S., Lee C., Elias C., Friedman J., et al. (2009). Leptin targets in the mouse brain. J. Comp. Neurol. 514 518–532. 10.1002/cne.22025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seidel G., Gaser C., Götz T., Günther A., Hamzei F. (2020). Accelerated brain ageing in sepsis survivors with cognitive long-term impairment. Eur. J. Neurosci. 52 4395–4402. 10.1111/ejn.14850 [DOI] [PubMed] [Google Scholar]
- Shah B., Hux J. (2003). Quantifying the risk of infectious diseases for people with diabetes. Diabetes Care 26 510–513. 10.2337/diacare.26.2.510 [DOI] [PubMed] [Google Scholar]
- Shaughness M., Acs D., Brabazon F., Hockenbury N., Byrnes K. (2020). Role of insulin in neurotrauma and neurodegeneration: a review. Front. Neurosci. 14:547175. 10.3389/fnins.2020.547175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinjyo N., Kagaya W., Pekna M. (2021). Interaction between the complement system and infectious agents - a potential mechanistic link to neurodegeneration and dementia. Front. Cell Neurosci. 15:710390. 10.3389/fncel.2021.710390 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinjyo N., Parkinson J., Bell J., Katsuno T., Bligh A. (2020). Berberine for prevention of dementia associated with diabetes and its comorbidities: a systematic review. J. Int. Med. 18 125–151. 10.1016/j.joim.2020.01.004 [DOI] [PubMed] [Google Scholar]
- Shippy D., Ulland T. (2020). Microglial immunometabolism in Alzheimer’s disease. Front. Cell Neurosci. 14:56446. 10.3389/fncel.2020.563446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smolina K., Wotton C., Goldacre M. (2015). Risk of dementia in patients hospitalised with type 1 and type 2 diabetes in England, 1998-2011: a retrospective national record linkage cohort study. Diabetologia 58 942–950. 10.1007/s00125-015-3515-x [DOI] [PubMed] [Google Scholar]
- Sochocka M., Zwolińska K., Leszek J. (2017). The infectious etiology of Alzheimer’s disease. Curr. Neuropharmacol. 15 996–1009. 10.2174/1570159X15666170313122937 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sofroniew M. (2020). Astrocyte reactivity: subtypes, states, and functions in CNS innate immunity. Trends Immunol. 41 758–770. 10.1016/j.it.2020.07.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solomon T. (2021). Neurological infection with SARS-CoV-2 - the story so far. Nat. Rev. Neurol. 17 65–66. 10.1038/s41582-020-00453-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sonneville R., Derese I., Marques M., Langouche L., Derde S., Chatre L., et al. (2015). Neuropathological correlates of hyperglycemia during prolonged polymicrobial sepsis in mice. Shock 44 245–251. 10.1097/SHK.0000000000000403 [DOI] [PubMed] [Google Scholar]
- Sonnewald U., Wang A., Schousboe A., Erikson R., Skottner A. (1996). New aspects of lactate metabolism: IGF-I and insulin regulate mitochondrial function in cultured brain cells during normoxia and hypoxia. Dev. Neurosci. 18 443–448. 10.1159/000111439 [DOI] [PubMed] [Google Scholar]
- Soscia S., Kirby J., Washicosky K., Tucker S., Ingelsson M., Hyman B., et al. (2010). The Alzheimer’s disease-associated amyloid beta-protein is an antimicrobial peptide. PLoS One 5:e9505. 10.1371/journal.pone.0009505 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soto M., Cai W., Konishi M., Kahn C. (2019). Insulin signaling in the hippocampus and amygdala regulates metabolism and neurobehavior. Proc. Natl. Acad. Sci. U S A. 116 6379–6384. 10.1073/pnas.1817391116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soung A., Klein R. (2018). Viral encephalitis and neurologic diseases: focus on astrocytes. Trends Mol. Med. 24 950–962. 10.1016/j.molmed.2018.09.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Souza-Almeida G., D’Avila H., Almeida P., Luna-Gomes T., Liechocki S., Walzog B., et al. (2018). Leptin mediates in vivo neutrophil migration: involvement of tumor necrosis factor-alpha and CXCL1. Front. Immunol. 9:111. 10.3389/fimmu.2018.00111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spadaro O., Camell C., Bosurgi L., Nguyen K., Youm Y., Rothlin C., et al. (2017). IGF1 shapes macrophage activation in response to immunometabolic challenge. Cell Rep. 19 225–234. 10.1016/j.celrep.2017.03.046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spielman L., Bahniwal M., Little J., Walker D., Klegeris A. (2015). Insulin modulates in vitro secretion of cytokines and cytotoxins by human glial cells. Curr. Alzheimer Res. 12 684–693. 10.2174/1567205012666150710104428 [DOI] [PubMed] [Google Scholar]
- Stephenson J., Nutma E., van der Valk P., Amor S. (2018). Inflammation in CNS neurodegenerative diseases. Immunology 154 204–219. 10.1111/imm.12922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart C., Rotwein P. (1996). Growth, differentiation, and survival: multiple physiological functions for insulin-like growth factors. Physiol. Rev. 76 1005–1026. 10.1152/physrev.1996.76.4.1005 [DOI] [PubMed] [Google Scholar]
- Sukhanov S., Higashi Y., Shai S., Vaughn C., Mohler J., Li Y., et al. (2007). IGF-1 reduces inflammatory responses, suppresses oxidative stress, and decreases atherosclerosis progression in ApoE-deficient mice. Arter. Thromb Vasc. Biol. 27 2684–2690. 10.1161/ATVBAHA.107.156257 [DOI] [PubMed] [Google Scholar]
- Sun Q., Li J., Gao F. (2014). New insights into insulin: the anti-inflammatory effect and its clinical relevance. World J. Diabetes 5 89–96. 10.4239/wjd.v5.i2.89 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suvan J., D’Aiuto F., Moles D., Petrie A., Donos N. (2011). Association between overweight/obesity and periodontitis in adults. a systematic review. Obes Rev. 12 e381–e404. 10.1111/j.1467-789X.2010.00808.x [DOI] [PubMed] [Google Scholar]
- Taieb O., Baleyte J., Mazet P., Fillet A. (2001). Borna disease virus and psychiatry. Eur. Psychiatry 16 3–10. 10.1016/s0924-9338(00)00529-520 [DOI] [PubMed] [Google Scholar]
- Takano K., Koarashi K., Kawabe K., Itakura M., Nakajima H., Moriyama M., et al. (2018). Insulin expression in cultured astrocytes and the decrease by amyloid beta. Neurochem. Int. 119 171–177. 10.1016/j.neuint.2017.10.017 [DOI] [PubMed] [Google Scholar]
- Talbot K., Wang H. (2014). The nature, significance, and glucagon-like peptide-1 analog treatment of brain insulin resistance in Alzheimer’s disease. Alzheimers Dement. 10 S12–S25. 10.1016/j.jalz.2013.12.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamara A., Tahapary D. (2020). Obesity as a predictor for a poor prognosis of COVID-19: a systematic review. Diab. Metab. Syndr. 14 655–659. 10.1016/j.dsx.2020.05.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang C., Lu D., Yang R., Tsai H., Kao M., Fu W., et al. (2007). Leptin-induced IL-6 production is mediated by leptin receptor, insulin receptor substrate-1, phosphatidylinositol 3-kinase, Akt, NF-kappaB, and p300 pathway in microglia. J. Immunol. 179 1292–1302. 10.4049/jimmunol.179.2.1292 [DOI] [PubMed] [Google Scholar]
- Tian Z., Sun R., Wei H., Gao B. (2002). Impaired natural killer (NK) cell activity in leptin receptor deficient mice: leptin as a critical regulator in NK cell development and activation. Biochem. Biophys. Res. Commun. 298 297–302. 10.1016/s0006-291x(02)02462-2462 [DOI] [PubMed] [Google Scholar]
- Tiedemann L., Schmid S., Hettel J., Giesen K., Francke P., Büchel C., et al. (2017). Central insulin modulates food valuation via mesolimbic pathways. Nat. Commun. 8:16052. 10.1038/ncomms16052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tien L., Lee Y., Pang Y., Lu S., Lee J., Tseng C., et al. (2017). Neuroprotective effects of intranasal IGF-1 against neonatal lipopolysaccharide-induced neurobehavioral deficits and neuronal inflammation in the substantia nigra and locus coeruleus of juvenile rats. Dev. Neurosci. 39 443–459. 10.1159/000477898 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai S., Clemente-Casares X., Zhou A., Lei H., Ahn J., Chan Y., et al. (2018). Insulin receptor-mediated stimulation boosts T cell immunity during inflammation and infection. Cell Metab. 28 922–934. 10.1016/j.cmet.2018.08.003 [DOI] [PubMed] [Google Scholar]
- Tschöp J., Nogueiras R., Haas-Lockie S., Kasten K., Castañeda T., Huber N. (2010). CNS leptin action modulates immune response and survival in sepsis. J. Neurosci. 30 6036–6047. 10.1523/JNEUROSCI.4875-09.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turnbaugh P., Ley R., Mahowald M., Magrini V., Mardis E., Gordon J. (2006). An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444 1027–1031. 10.1038/nature05414 [DOI] [PubMed] [Google Scholar]
- Uysal K., Wiesbrock S., Marino M., Hotamisligil G. (1997). Protection from obesity-induced insulin resistance in mice lacking TNF-alpha function. Nature 389 610–614. 10.1038/39335 [DOI] [PubMed] [Google Scholar]
- Valcour V., Sacktor N., Paul R., Watters M., Selnes O., Shiramizu B., et al. (2006). Insulin resistance is associated with cognition among HIV-1-infected patients: the Hawaii Aging With HIV cohort. J. Acquir. Immune Defic. Syndr. 43 405–410. 10.1097/01.qai.0000243119.67529.f5 [DOI] [PubMed] [Google Scholar]
- Valcour V., Shikuma C., Shiramizu B., Williams A., Watters M., Poff P., et al. (2005). Diabetes, insulin resistance, and dementia among HIV-1-infected patients. J. Acquir. Immune Defic. Syndr. 38 31–36. 10.1097/00126334-200501010-200501016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valdebenito S., Castellano P., Ajasin D., Eugenin E. (2021). Astrocytes are HIV reservoirs in the brain: a cell type with poor HIV infectivity and replication but efficient cell-to-cell viral transfer. J. Neurochem. 158 429–443. 10.1111/jnc.15336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van den Bossche J., O’Neill L., Menon D. (2017). Macrophage immunometabolism: where are we (going)? Trends Immunol. 38 395–406. 10.1016/j.it.2017.03.001 [DOI] [PubMed] [Google Scholar]
- van Ginneken V., Sitnyakowsky L., Jeffery J. (2009). ”Infectobesity: viral infections (especially with human adenovirus-36: Ad-36) may be a cause of obesity. Med. Hypotheses 72 383–388. 10.1016/j.mehy.2008.11.034 [DOI] [PubMed] [Google Scholar]
- van Niekerk G., Christowitz C., Conradie D., Engelbrecht A. (2020). Insulin as an immunomodulatory hormone. Cytokine Growth. Factor. Rev. 52 34–44. 10.1016/j.cytogfr.2019.11.006 [DOI] [PubMed] [Google Scholar]
- van Son J., Koekkoek L., La Fleur S., Serlie M., Nieuwdorp M. (2021). The role of the gut microbiota in the gut-brain axis in obesity: mechanisms and future implications. Int. J. Mol. Sci. 22:2993. 10.3390/ijms2206299 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vangipuram S., Yu M., Tian J., Stanhope K., Pasarica M., Havel P., et al. (2007). Adipogenic human adenovirus-36 reduces leptin expression and secretion and increases glucose uptake by fat cells. Int. J. Obes (Lond) 31 87–96. 10.1038/sj.ijo.0803366 [DOI] [PubMed] [Google Scholar]
- Vedantama G., Viswanathan V. (2012). Leptin signaling protects the gut from Entamoeba histolytica infection. Gut Microbes 3 2–3. 10.4161/gmic.19424 [DOI] [PubMed] [Google Scholar]
- Viardot A., Grey S., Mackay F., Chisholm D. (2007). Potential antiinflammatory role of insulin via the preferential polarization of effector T cells toward a T helper 2 phenotype. Endocrinology 148 346–353. 10.1210/en.2006-2686 [DOI] [PubMed] [Google Scholar]
- Virto L., Cano P., Jiménez-Ortega V., Fernández-Mateos P., González J., Esquifino A., et al. (2018). Obesity and periodontitis: an experimental study to evaluate periodontal and systemic effects of comorbidity. J. Periodontol. 89 176–185. 10.1902/jop.2017.170355 [DOI] [PubMed] [Google Scholar]
- Vittor A., Long M., Chakrabarty P., Aycock L., Kollu V., DeKosky S. (2020). West nile virus-induced neurologic sequelae—relationship to neurodegenerative cascades and dementias. Curr. Trop. Med. Rep. 7 25–36. 10.1007/s40475-020-00200-207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wakim L., Woodward-Davis A., Liu R., Hu Y., Villadangos J., Smyth G., et al. (2012). The molecular signature of tissue resident memory CD8 T cells isolated from the brain. J. Immunol. 189 3462–3471. 10.4049/jimmunol.1201305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y., Li Y., Zhao R., Wu B., Lanoha B., Tong Z., et al. (2017). Glutaminase C overexpression in the brain induces learning deficits, synaptic dysfunctions, and neuroinflammation in mice. Brain Behav. Immun. 66 135–145. 10.1016/j.bbi.2017.06.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wasim M., Awan F., Najam S., Khan A., Khan H. (2016). Role of leptin deficiency, inefficiency, and leptin receptors in obesity. Biochem. Genet. 54 565–572. 10.1007/s10528-016-9751-z [DOI] [PubMed] [Google Scholar]
- Weilhammer D., Iavarone A., Villegas E., Brooks G., Sinai A., Sha W. (2012). Host metabolism regulates growth and differentiation of Toxoplasma gondii. Int. J. Parasitol. 42 947–959. 10.1016/j.ijpara.2012.07.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- White M. (2003). Insulin signaling in health and disease. Science 302 1710–1711. 10.1126/science.1092952 [DOI] [PubMed] [Google Scholar]
- Whitmer R., Gunderson E., Barrett-Connor E., Quesenberry C. J., Yaffe K. (2005). Obesity in middle age and future risk of dementia: a 27 year longitudinal population based study. BMJ 330:1360. 10.1136/bmj.38446.466238.E0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitmer R., Gunderson E., Barrett-Connor E., Quesenberry C. J., Yaffe K. (2018). Obesity trajectories and risk of dementia: 28 years of follow-up in the Whitehall II Study. Alzheimers Dement. 14 178–186. 10.1016/j.jalz.2017.06.2637 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitmer R., Gustafson D., Barrett-Connor E., Haan M., Gunderson E., Yaffe K. (2008). Central obesity and increased risk of dementia more than three decades later. Neurology 71 1057–1064. 10.1212/01.wnl.0000306313.89165.ef [DOI] [PubMed] [Google Scholar]
- Widmann C., Heneka M. (2014). Long-term cerebral consequences of sepsis. Lancet Neurol. 13 630–636. 10.1016/S1474-4422(14)70017-70011 [DOI] [PubMed] [Google Scholar]
- Wieland C., Florquin S., Chan E., Leemans J., Weijer S., Verbon A., et al. (2005). Pulmonary Mycobacterium tuberculosis infection in leptin-deficient ob/ob mice. Int. Immunol. 17 1399–1408. 10.1093/intimm/dxh317 [DOI] [PubMed] [Google Scholar]
- Wilking A., Elliott E., Garcia M., Murray K., Munoz F. (2014). Central nervous system manifestations in pediatric patients with influenza A H1N1 infection during the 2009 pandemic. Pediatr. Neurol. 51 370–376. 10.1016/j.pediatrneurol.2014.04.026 [DOI] [PubMed] [Google Scholar]
- Wlodarczyk A., Holtman I., Krueger M., Yogev N., Bruttger J., Khorooshi R., et al. (2017). A novel microglial subset plays a key role in myelinogenesis in developing brain. EMBO J. 36 3292–3308. 10.15252/embj.201696056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woods S., Iudicello J., Dawson M., Weber E., Grant I., Letendre S. (2010). HIV-associated deficits in action (verb) generation may reflect astrocytosis. J. Clin. Exp. Neuropsychol. 32 522–527. 10.1080/13803390903264130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yano H., Kinoshita M., Fujino K., Nakashima M., Yamamoto Y., Miyazaki H., et al. (2012). Insulin treatment directly restores neutrophil phagocytosis and bactericidal activity in diabetic mice and thereby improves surgical site Staphylococcus aureus infection. Infect. Immun. 80 4409–4416. 10.1128/IAI.00787-712 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yau B., Hunt N., Mitchell A., Too L. (2018). Blood-Brain barrier pathology and CNS outcomes in Streptococcus pneumoniae meningitis. Int. J. Mol. Sci. 19:3555. 10.3390/ijms19113555 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yki-Järvinen H., Sammalkorpi K., Koivisto V., Nikkilä E. (1989). Severity, duration, and mechanisms of insulin resistance during acute infections. J. Clin. Endocrinol. Metab. 69 317–323. 10.1210/jcem-69-2-317 [DOI] [PubMed] [Google Scholar]
- Zawada A., Rychter A., Ratajczak A., Lisiecka-Masian A., Dobrowolska A., Krela-Kaźmierczak I. (2020). Does gut-microbiome interaction protect against obesity and obesity-associated metabolic disorders? Microorganisms 9:18. 10.3390/microorganisms9010018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zemva J., Schubert M. (2011). Central insulin and insulin-like growth factor-1 signaling: implications for diabetes associated dementia. Curr. Diab. Rev. 7 356–366. 10.2174/157339911797415594 [DOI] [PubMed] [Google Scholar]
- Zhang A., To K., Li C., Lau C., Poon V., Chan C., et al. (2013). Leptin mediates the pathogenesis of severe 2009 pandemic influenza a (h1n1) infection associated with cytokine dysregulation in mice with diet-induced obesity. J. Infect. Dis. 207 1270–1280. 10.1093/infdis/jit031 [DOI] [PubMed] [Google Scholar]
- Ziegler A., Levison S., Wood T. (2015). Insulin and IGF receptor signalling in neural-stem-cell homeostasis. Nat. Rev. Endocrinol. 11 161–170. 10.1038/nrendo.2014.208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziegler A., Schneider J., Qin M., Tyler W., Pintar J., Fraidenraich D., et al. (2012). IGF-II promotes stemness of neural restricted precursors. Stem Cells 30 1265–1276. 10.1002/stem.1095 [DOI] [PMC free article] [PubMed] [Google Scholar]