Abstract
Musculoskeletal pains are sometimes misdiagnosed in some diseases, like rheumatoid and psoriatic arthritis, erosive OA, etc. Secondary hyperparathyroidism was not considered a differential diagnosis for RA, despite the fact that it can cause arthralgia or arthritis. Also, fibromyalgia is a psychosomatic condition marked by widespread pain and tenderness. This study included 400 patients attended certain outpatient clinics of Rheumatology in Egypt and Saudi Arabia, who were not fulfilling criteria for RA diagnosis. Criteria for classification of fibromyalgia syndrome were applied to all patients. We did lab tests and radiological imaging modalities for diagnosis or exclusion of suspected diseases were applied. All patients were fulfilling both old and new criteria of fibromyalgia syndrome, and not fulfilling any RA criteria, and had vitamin D3 deficiency or insufficiency. 75% of patients had abnormally high levels of PTH, without parathyroid gland pathology. Radiology showed subperiosteal and subchondral resorption of mainly thumbs, subchondral osteopenia of proximal and middle phalanges, mild subperiosteal resorption along the radial aspect of the middle phalanx and mild tuft erosions, besides changes in the carpus closely resembling those of rheumatoid arthritis, of ulnar styloid resorption, radiocarpal and scapho-trapezoid joint arthritis. Of special interest, the presence of tuft spur-like excrescences.
Subject terms: Musculoskeletal system, Fibromyalgia, Rheumatoid arthritis, Rheumatology, Metabolic bone disease, Osteoporosis
Introduction
Secondary hyperparathyroidism (SHPT) is a medical disorder in which the parathyroid glands secrete too much parathyroid hormone (PTH) in reaction to hypocalcemia, resulting in gland hyperplasia. The most prevalent cause of SHPT is chronic renal failure, which is caused by the kidneys' inability to convert enough vitamin D2 to its active form D3, and hence to not adequately excrete phosphate. Insoluble calcium phosphate occurs in the body as a result, removing calcium from circulation. Both of these mechanisms result in hypocalcemia and, as a result, SHPT1. Malabsorption (chronic pancreatitis, small bowel disease, malabsorption-dependent bariatric surgery) can also cause SHPT because fat-soluble vitamin D cannot be reabsorbed. Consequently, hypocalcemia develops, and parathyroid hormone secretion rises in an attempt to raise serum calcium levels. Other factors include calcium shortage in the diet, vitamin D deficiency, or steatorrhea2.
Muscle cramps and bone pain are common symptoms of HPT, as are perioral tingling or paresthesia in the fingers or toes, and positive Chvostek and Trousseau signs. Only a few cases of arthritis mimicking rheumatoid arthritis as a presenting characteristic of HPT have been described in the literature3.
Rheumatoid arthritis (RA) is a chronic autoimmune disease that affects the joints principally which become warm, swollen, and painful. Wrists and hands are the most commonly affected joints, with the same joints being impacted bilaterally. It was previously established that RF is not exclusive to RA and can be present in other disorders such as Epstein–Barr virus or Parvovirus infections, as well as in 5 to 10% of the normal population, especially elderly people4,5.
Anti-CCP antibodies are produced locally at areas of inflammation, not just in the synovium of RA patients, but also in non-RA conditions. As a result, anti-CCP positivity may be seen in a subset of patients with diseases other than RA6.
One of the reasons of SHPT, as we know, is vitamin D3 deficiency, which has been discovered to be frequent in the Middle East, despite the region's abundance of sun rays and hence UV exposure. Heavy metal pollution, such as cadmium and lead, is one of the most likely causes of vitamin D insufficiency in the Middle East7,8.
The mitochondria of the kidney's proximal convoluted tubule cells produce circulating 1α,25-dihydroxyvitamin D3 [1,25(OH)2D3], and parathyroid hormone (PTH) evidently increases 1,25(OH)2D3 production while 1,25(OH)2D3 suppresses PTH production9.
Cadmium prefers to accumulate in the kidneys. The accumulation of cadmium-metallothionein (CdMT) in the proximal convoluted tubules causes damage as a consequence to renal cadmium deposition.
The preferential absorption of CdMT into the S1 and S2 segments of the proximal tubules, which is responsible for converting 25(OH)D3 to 1,25(OH)2D3, may contribute to CdMT-induced nephrotoxicity10.
Vitamin D3 deficiency and hyperparathyroidism have been linked to Fibromyalgia syndrome11,12. Meanwhile, gout, pseudogout, juxtaarticular erosions, tenosynovitis, periarthritis, and pericapsular calcification are just a few of the articular symptoms linked to hyperparathyroidism13.
Patients and methods
This study included 400 patients attended outpatient clinics of Al-Azhar University Faculty of Medicine Hospitals, Egypt, and Elaj Specialized Clinics, Saudi Arabia, in the period between Nov 2014 to Dec 2019, who were previously diagnosed as RA, but we found them not fulfilling any criteria for RA diagnosis, and not responding to treatments including NSAIDs, and DMARDs, (Table 1). This study was carried out according to regulations and approval of Ethics Committee of Faculty of Medicine for Girls, Al-Azhar University, Nasr City, Cairo, Egypt, Registered at Central Administration of Research & Development; Egyptian Ministry of Health: Reg No. RHBIRB2018122001.
Table 1.
Demographic data of MEPS study group.
| MEPS | No | % | Age years | Female sex | Duration years | |
|---|---|---|---|---|---|---|
| N | % | |||||
| Seropositive | 72 | 18 | 40 ± 13 | 220 | 55 | 1.27 ± 0.82 |
| Seronegative | 328 | 82 | 39 ± 16 | 180 | 45 | 1.17 ± 0.90 |
| Total | 400 | 100 | 1.22 ± 0.86 | |||
Inclusion and exclusion criteria
We got informed consent from all patients to re-evaluate and re-diagnose them. All methods were carried out in accordance with relevant guidelines and regulations. Patients were investigated to establish existence of RA, using criteria for classification of RA14,15, with exclusion of other mimicking diseases like psoriatic arthritis, erosive OA, viral arthritis, reactive arthritis, IBD arthritis, Lyme’s disease, and palindromic rheumatism. In addition, we applied old and new criteria for classification of fibromyalgia syndrome to all patients16,17 to establish the diagnosis of FMS.
We did lab tests of RF, ACPA, ESR, CRP, LFT, RFT, serum 25 cholcalciferol, PTH, total and ionized calcium, phosphorus, and SUA. In addition, all patients did plain X-ray (Toshiba Digital Radiography System, DIGIX U) for hands, knees, and lumbar regions. When recommended, they were exposed to MRI (Philips1.5 T), and CT (Multi-Slice spiral CT Aston). High-resolution peripheral quantitative computed tomography (HR-pQCT) imaging allowed for highly reliable assessment of erosion in patients suspected to have RA to exclude them.
Consent for publication
we confirm hereby that the manuscript has not been submitted or is not simultaneously being submitted elsewhere, is not at the time of submission under consideration by another journal or other publication, and that no portion of the data has been or will be published elsewhere while the manuscript is under review by the journal, unless rejected by the Journal, or withdrawn by the author.
Results
All patients scheduled were fulfilling both old and new criteria of fibromyalgia syndrome, and not fulfilling any RA criteria, 82% of them were seronegative and 18% were seropositive with low RF titers and negative ACPA. All patients had vitamin D3 deficiency or insufficiency. 75% of patients had abnormally high levels of PTH (96 ± 12 pg/ml), and had no parathyroid gland pathology (Tables 2, 3).
Table 2.
Laboratory chemistry and PTH results of MEPS study group.
| No | ESR (mm) | SUA (mg/dl) | Ca (mg/dl) | Ca++ (mg/dl) | D3 (ng/ml) | PTH (pg/ml) | P (mg/dl) | |
|---|---|---|---|---|---|---|---|---|
| MEPS | 400 | 40 ± 14 | 6.4 ± 1.3 | 8.6 ± 1.4 | 1.11 ± 0.11 | 15.58 ± 6.66 | 96 ± 12 | 4.8 ± 1.2 |
ESR erythrocyte sedimentation rate, SUA serum uric acid, Ca total calcium, Ca++ ionized calcium, D3 vitamin D3, PTH parathyroid hormone, P serum phosphorus.
Table 3.
Different Criteria Domains in MEPS study group.
| NO | RN | Dur | CCP | RF | TTP | SSS | WPI | |
|---|---|---|---|---|---|---|---|---|
| MEPS | 400 | 0% | 1.22 ± 0.86 | 0% | 18% | 15.2 ± 2.3 | 9.62 ± 2.11 | 15.32 ± 3.34 |
RN rheumatoid nodules, Dur duration, CCP anti-CCP, RF rheumatoid factor, TTP tender trigger points, SSS symptom severity score, WPI widespread pain index.
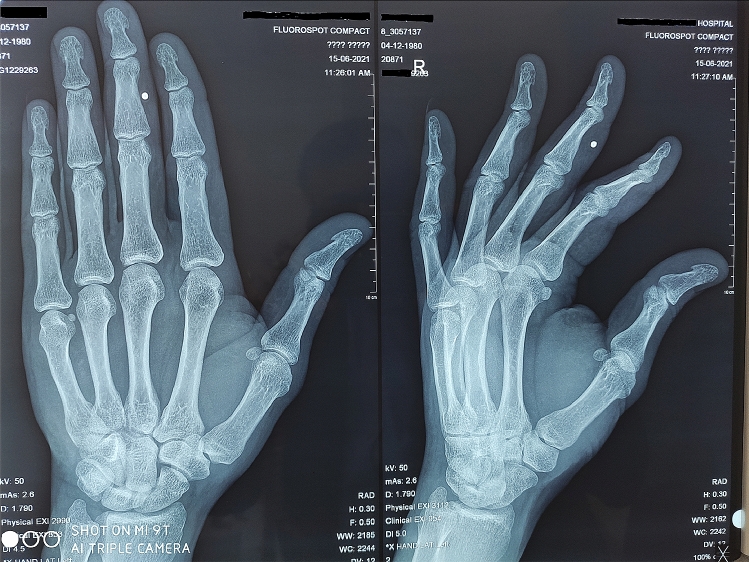
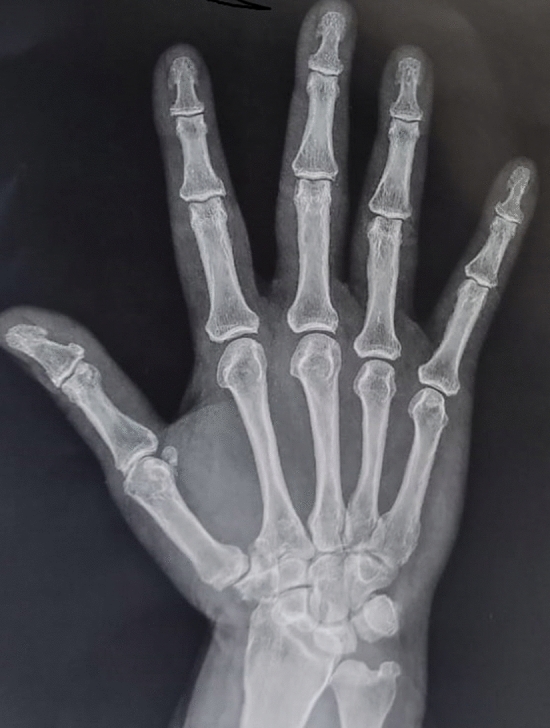
X-rays of our patients hands showed subperiosteal and subchondral resorption of mainly thumbs, subchondral osteopenia of proximal and middle phalanges, mild subperiosteal resorption along the radial aspect of the middle phalanx (88%) and mild tuft erosions (12%), besides changes in the carpus closely resembling those of rheumatoid arthritis, of ulnar styloid resorption, radiocarpal and scapho-trapezoid joint arthritis (35%) (Fig. 1). Of special interest, the presence of tuft spurs-like excrescences mimicking that of (spade phalanx sign) of acromegaly (95%), but without any other criteria of acromegaly (Figs. 2, 3, 4). Plain X ray of knees showed chondrocalcinosis (20%), intracortical resorption, and osteopenia. Lumbar spine shows mild sclerotic bands of the Superior and inferior end plates mimicking rugger jersey sign (30%), as well as osteopenia and brown tumors (30%).we also detected calcaneal spurs in 45% of patients (Table 4).
Figure 1.
Plain X ray hand PA view shows mild subperiosteal bone resorption affecting radial aspects of the proximal and middle phalanges of the 2nd and 3rd fingers, terminal tuft erosions (acro-osteolysis), as well as spur-like excrescences. Figure shows changes in the carpus closely resembling those of rheumatoid arthritis, radiocarpal and scapho-trapezoid joint arthritis.
Figure 2.
Plain X ray hand PA view shows mild subperiosteal bone resorption affecting radial aspects of the proximal and middle phalanges of the 2nd and 3rd fingers, terminal tuft erosions (acro-osteolysis), as well as spur-like excrescences.
Figure 3.
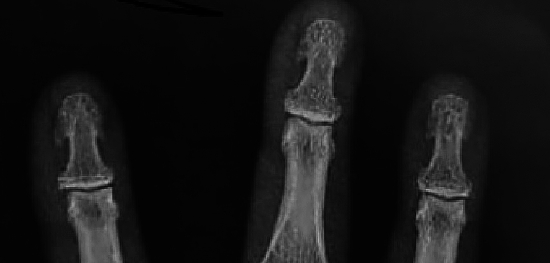
Zoom of previous Plain X ray hands PA view shows tuft spur-like excrescences mimicking that of (spade phalanx sign) of acromegaly.
Figure 4.
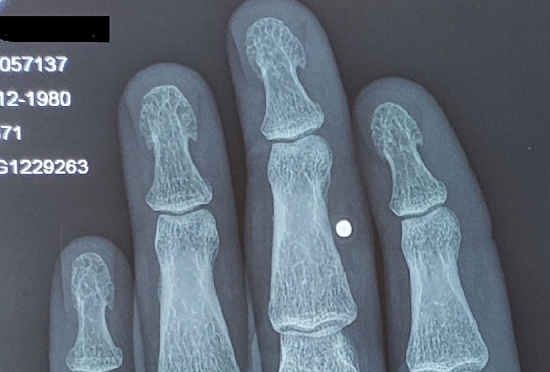
Zoom of previous Plain X ray hands PA view shows tuft spur-like excrescences mimicking that of (spade phalanx sign) of acromegaly.
Table 4.
Different radiological findings in MEPS study group.
| Group | N0 | Sr | Odp | Bt | Rj | Cs | ch | Ts | WA |
|---|---|---|---|---|---|---|---|---|---|
| MEPS | 400 | 88% | 12 | 30% | 30% | 45% | 20% | 95% | 35 |
Sr Subperiosteal resorption, Odp Osteolysis of distal phalanges, Bt Brown tumours, Rj Rugger jersey appearance of vertebral column, Cs Calcaneal spurs, ch chondrocalcinosis, Ts Tuft spur-like excrescences, WA wrist arthritis.
Discussion
We found many patients with bilateral wrist and hand arthritis had a combination of vitamin D3 deficiency, fibromyalgia, and sometimes increased serum parathyroid hormone. They were misdiagnosed as having either seronegative or seropositive rheumatoid arthritis, resulting in failure of treatment and long-term pain. We revaluated them as having a new syndrome, of vitaminD3 deficiency, FMS, and SHPT; we named it Middle East Pain Syndrom (MEPS), since all patients were from Middle East.
A study by Helliwell showed arthritis in 11% of HPT patients, including two cases of erosive arthritis18. Also, HPT presenting as arthritis had been described in other case reports18–22. Meanwhile, HPT itself, can occasionally co-exist with inflammatory arthritis like rheumatoid arthritis23. In addition to clinical and biochemical features, radiographical features were helpful to differentiate both. HPT does not cause only osteoporosis and pathological fractures, it can cause also bone erosions, which are shaggy in appearance and distributed in the radiocarpal, radioulnar, metacarpophalangeal and distal interphalangeal joints, in contrast to that which predominate PIP joints of rheumatoid arthritis24.
HPT is characterized by both bone resorption and formation. Plain radiography results may include resorption and sclerosis of multiple places in the skeletal system as a result25. Bone findings were characterized by the osteoclastic resorption of bone, osteoblastic bone formation, and fibrous replacement of marrow, with radiographic findings of subperiosteal resorption, brown tumors, bone cysts, and sclerosis26.
The disease may progress with further destruction that results in a ground-glass appearance in bone trabeculae26. Bone resorption may be interpreted as subperiosteal, intracortical, trabecular, endosteal, subchondral, subligamentous, or subtendinous. Subperiosteal bone resorption is an early and virtually pathognomonic sign of HPT, which is marked by marginal erosions with adjacent resorption of bone and sclerosis. Although subperiosteal bone resorption can affect many sites, the most common site in HPT is the middle phalanges of the index and middle fingers, primarily on the radial aspect26. HPT osteoporosis attains a characteristic tubal, cigar or pyramidal like appearance, other than that found in RA or other collagen diseases. Of special interest, the presence of tuft spur-like excrescences mimicking that of spade phalanx sign of acromegaly27, but without any other criteria of acromegaly. One literature reported this sign in an unexplained case report28.
Of course, the secondary hyperparathyroidism found in our patients is a sequence of chronic vitamin D3 deficiency or insufficiency, and not associated with renal problems, nor malignancies, or other precipitating factors. We attributed this chronic vitamin D3 deficiency or insufficiency to probable long term exposure to pollutants, as the heavy metals cadmium and lead, present in some mineral waters, fizzy waters, fried snacks, and tobacco smoke7,8,29.
Vitamin D is produced in the skin from 7-dehydrocholesterol by UV irradiation. The liver and other tissues metabolize vitamin D, whether from the skin or oral ingestion, to 25OHD, the principal circulating form of vitamin D. The steroidogenic enzyme cytochrome P450scc (CYP11A1) as well as CYP27B1 (1α-hydroxylase) play an important role in these pathways. They were proved to be produced in placenta, adrenal glands, and epidermal keratinocytes. 25OHD is then further metabolized to 1,25(OH)2D principally in the kidney, by the enzyme CYP27B1. 1,25(OH)2D is the principal hormonal form of vitamin D, responsible for most of its biologic actions. Other novel products include 20-hydroxyvitamin D3 [20(OH)D3], 22(OH)D3, 20,23(OH)2D3, 20,22(OH)2D3, 1,20(OH)2D3, 1,20,23(OH)3D3, and 17,20,23(OH)3D3 were found to be produced too by placenta, adrenal glands, and epidermal keratinocytes30–33.
Cadmium preferentially selects to settle in kidneys. Renal cadmium deposition induces injury resulted from accumulation of cadmium-metallothionein (CdMT) in the proximal convoluted tubules. CdMT-induced nephrotoxicity might be due, at least in part, to its preferential uptake of CdMT into the S1 and S2 segments of the proximal tubules, which is responsible for converting 25(OH)D3 to 1,25(OH)2D334. In addition, cadmium was found to inhibit expression of the p450scc (CYP11A1) gene and consequently its steroidogenic function35.
The mitochondria of proximal convoluted tubule cells of the kidney are the site of production of circulating 1α,25-dihydroxyvitamin D3. Parathyroid hormone (PTH) markedly stimulates 1,25(OH)2D3 production, while 1,25(OH)2D3 suppresses production of PTH itself9.
Vitamin D3 deficiency and HPT are established to be associated with Fibromyalgia syndrome36,37. Hyperparathyroidism is known to be associated with multiple articular manifestations including gout, pseudogout, juxtaarticular erosions, tenosynovitis, periarthritis, and pericapsular calcification12. Hyperparathyroidism is a cause of bone pain and aching joints due to osteoporosis or osteopenia because of the continued removal of calcium from bone as well as chondrocalcinosis and pseudogout. Bone pain may also be caused by hemorrhages within the bone. Joint pains, degenerative arthritis, joint laxity and muscle weakness can result from calcium pyrophosphate deposition38. This chronic suffering from vitamin D3 deficiency or insufficiency induces SHPT, which in turn induces musculoskeletal perturbance in the form of muscle and tendon pain and weakness, bone and joint pains.
This syndrome responds well to treatment of fibromyalgia regimen with SSRI, muscle relaxants, and vitamin D3 supplementation 5000–10,000 iu/day, antioxidants including selenium, vitamins E and C. In cases with arthritis we add sulphasalazine 1 gm/bid or Plaquenil 200 mg/bid to which their inflamed joints respond significantly.
Conclusion
Middl East Pain Syndrome is a new disease, which must be given more attention for diagnosis, and more extensive studies for its pathogenesis, prevention. and cure. Cadmium pollution must be considered as one of the new millennium big challenges that should be treated efficiently, to prevent many economically affecting health hazards.
Author contributions
A.A.E., B.M.E., and H.M.E. collected study subjects, data, and made tables. A.A.E. and A.M.B. wrote the main manuscript text A.A.E. and A.M.B. prepared figures All authors reviewed the manuscript.
Funding
We confirm also that there are not any financial support or other benefits from commercial sources for the work reported on in the manuscript, or any other financial interests that any of the authors may have, which could create a potential conflict of interest or the appearance of a conflict of interest with regard to the work.
Data availability
All data and materials of this manuscript is available when requested by the Editors of Journal.
Competing interests
The authors declare no competing interests.
Footnotes
This article has been retracted. Please see the retraction notice for more detail: 10.1038/s41598-024-66447-6
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
7/8/2024
This article has been retracted. Please see the Retraction Notice for more detail: 10.1038/s41598-024-66447-6
References
- 1.Taylor JG, Bushinsky DA. Calcium and phosphorus homeostasis. Blood Purif. 2009;27:387–394. doi: 10.1159/000209740. [DOI] [PubMed] [Google Scholar]
- 2.Pepe J, Cipriani C, Pilotto R, De Lucia F, Castro C, Lenge L, et al. Sporadic and hereditary primary hyperparathyroidism. J. Endocrinol. Invest. 2011;34:40–44. [PubMed] [Google Scholar]
- 3.Madkhali T, Alhefdhi A, Chen H, Elfenbein D. Primary hyperparathyroidism. Ulus Cerrahi Derg. 2016;32:58–66. doi: 10.5152/UCD.2015.3032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Falkenburg WJJ. IgG subclass specificity discriminates restricted IgM rheumatoid factor responses from more mature anti-citrullinated protein antibody-associated or isotype-switched iga responses. Arthritis Rheumatol. 2015;67:3124–3134. doi: 10.1002/art.39299. [DOI] [PubMed] [Google Scholar]
- 5.Edkins A, Cushley W. The Jekyll and Hyde nature of antibodies. Biol. Sci. Rev. 2012;25:2–4. [Google Scholar]
- 6.Vossenaar ER, Smeets TJ, Kraan MC, Raats JM, van Venrooij WJ, Tak PP. The presence of citrullinated proteins is not specific for rheumatoid synovial tissue. Arthritis Rheum. 2004;50:3485–3494. doi: 10.1002/art.20584. [DOI] [PubMed] [Google Scholar]
- 7.Elbeialy, A. & Hesham, E. AB1261 Cadmium toxicity as a probable cause of osteopenia in adolescents and its relation to bad dietary habits. 2091–2092 (2019).
- 8.Kazantzis G. Cadmium, osteoporosis and calcium metabolism. Biometals. 2004;17:493–498. doi: 10.1023/B:BIOM.0000045727.76054.f3. [DOI] [PubMed] [Google Scholar]
- 9.Wang Y, Zhu J, De Luca HF. The vitamin D receptor in the proximal renal tubule is a key regulator of serum 1α,25-dihydroxyvitamin D3. Am. J. Physiol. Endocrinol. Metab. 2015;308:E201–E205. doi: 10.1152/ajpendo.00422.2014. [DOI] [PubMed] [Google Scholar]
- 10.Kurata Y, Katsuta O, Doi T, et al. Chronic cadmium treatment induces tubular nephropathy and osteomalacic osteopenia in ovariectomized cynomolgus monkeys. Vet. Pathol. 2014;51:919–931. doi: 10.1177/0300985813509384. [DOI] [PubMed] [Google Scholar]
- 11.Costa JM, Ranzolin A, Costa Neto CA, Marques CD, Duarte AL. High frequency of asymptomatic hyperparathyroidism in patients with fibromyalgia: Random association or misdiagnosis? Rev. Bras. Reumatol. 2016;56:391–397. doi: 10.1016/j.rbr.2016.01.003. [DOI] [PubMed] [Google Scholar]
- 12.Borgia AR, Cavallasca JA, Costa CA, Musuruana JL. Hyperparathyroidism, a forgotten cause of musculoskeletal pain. Reumatol. Clin. 2012;8:299–301. doi: 10.1016/j.reuma.2012.02.008. [DOI] [PubMed] [Google Scholar]
- 13.Mackenzie-Feder, J., Sirrs, S., Anderson, D., Sharif, J. & Khan, A. Primary hyperparathyroidism: An overview. Int. J. Endocrinol. (2011). [DOI] [PMC free article] [PubMed]
- 14.Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988;31:315–324. doi: 10.1002/art.1780310302. [DOI] [PubMed] [Google Scholar]
- 15.Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO, et al. Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010;62:2569–2581. doi: 10.1002/art.27584. [DOI] [PubMed] [Google Scholar]
- 16.Wolfe F, Smythe HA, Yunus MB, Bennett RM, Bombardier C, Goldenberg DL, et al. The American college of rheumatology 1990 criteria for the classification of fibromyalgia. Report of the Multicenter Criteria Committee. Arthritis Rheum. 1990;33:160–172. doi: 10.1002/art.1780330203. [DOI] [PubMed] [Google Scholar]
- 17.Wolfe F, Clauw DJ, Fitzcharles MA, Goldenberg DL, Katz RS, Mease P, et al. The American College of Rheumatology preliminary diagnostic criteria for fibromyalgia and measurement of symptom severity. Arthritis Care Res. 2010;62:600–610. doi: 10.1002/acr.20140. [DOI] [PubMed] [Google Scholar]
- 18.Helliwell M. Rheumatic symptoms in primary hyperparathyroidism. Postgrad. Med. J. 1983;59:236–240. doi: 10.1136/pgmj.59.690.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bywaters EGL, Dixon ASJ, Scott JT. Joint lesions of hyperparathyroidism. Ann. Rheumatic Dis. 1963;22:171. doi: 10.1136/ard.22.3.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zvaifler NJ, Reefe WE, Black RL. Articular manifestations in primary hyperparathyroidism. Arthritis Rheum. 1962;5:237–249. doi: 10.1002/art.1780050304. [DOI] [Google Scholar]
- 21.Lipson RL, Williams LE, Zvaifler NJ, Howell DS. The “connective tissue disorder” of hyperparathyroidism. Arthritis Rheum. Off. J. Am. College Rheumatol. 1968;11:198–205. doi: 10.1002/art.1780110211. [DOI] [PubMed] [Google Scholar]
- 22.Duraywish AA. Chronic unclassified polyarthritis: A rare presentation of primary hyperparathyroidism. Med. J. Cairo Univ. 2016;84:265–269. [Google Scholar]
- 23.Salari M, Aboutorabi RB, Rezaieyazdi Z. Hypercalcemia and lytic bone lesions masquerading inflammatory arthritis treated as rheumatoid arthritis. Arch. Iran Med. 2015;18:720–723. [PubMed] [Google Scholar]
- 24.Feng X, McDonald JM. Disorders of bone remodeling. Annu. Rev. Pathol. 2011;6:121–145. doi: 10.1146/annurev-pathol-011110-130203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li, N., Li, X. M., Xu, L., Sun, W. J., Cheng, X. G. & Tian, W. Comparison of QCT and DXA: Osteoporosis detection rates in postmenopausal women. Int. J. Endocrinol. (2013). [DOI] [PMC free article] [PubMed]
- 26.Patel, A. A., Ramanathan, R., Kuban, J. & Willis, M. H. Imaging findings and evaluation of metabolic bone disease. Adv. Radiol. (2015).
- 27.Killinger Z, Payer J, Lazúrová I, Imrich R, Homérová Z, Kužma M, Rovenský J. Arthropathy in acromegaly. Rheum. Dis. Clin. North Am. 2010;36:713–720. doi: 10.1016/j.rdc.2010.09.004. [DOI] [PubMed] [Google Scholar]
- 28.Freyschmidt, J., Brossmann, J., Sternberg, A. et al. Freyschmidt's "koehler/zimmer's borderlands of normal and early pathological findings in skeletal radiography" (2003).
- 29.Elbeialy, A. A. & Eldosouky, H. A. AB0984 Cadmium toxicity as a probable cause of smoking induced bone loss. 1614–1614 (2018).
- 30.Bikle D, et al. Vitamin D: Production, metabolism, and mechanisms of action. In: Feingold KR, Anawalt B, Boyce A, et al., editors. Endotext. MDText.com Inc; 2000. [PubMed] [Google Scholar]
- 31.Slominski AT, Kim TK, Shehabi HZ, et al. In vivo evidence for a novel pathway of vitamin D3 metabolism initiated by P450scc and modified by CYP27B1. FASEB J. 2012;26(9):3901–3915. doi: 10.1096/fj.12-208975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Slominski AT, Kim TK, Shehabi HZ, et al. In vivo production of novel vitamin D2 hydroxy-derivatives by human placentas, epidermal keratinocytes, Caco-2 colon cells and the adrenal gland. Mol. Cell Endocrinol. 2014;383(1–2):181–192. doi: 10.1016/j.mce.2013.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Slominski A, Kim TK, Li W, et al. Detection of novel CYP11A1-derived secosteroids in the human epidermis and serum and pig adrenal gland. Sci. Rep. 2015;5:14875. doi: 10.1038/srep14875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Johri N, Jacquillet G, Unwin R. Heavy metal poisoning: The effects of cadmium on the kidney. Biometals. 2010;23(5):783–792. doi: 10.1007/s10534-010-9328-y. [DOI] [PubMed] [Google Scholar]
- 35.Smida AD, Valderrama XP, Agostini MC, Furlan MA, Chedrese J. Cadmium stimulates transcription of the cytochrome p450 side chain cleavage gene in genetically modified stable porcine granulosa cells. Biol. Reprod. 2004;70(1):25–31. doi: 10.1095/biolreprod.103.019000. [DOI] [PubMed] [Google Scholar]
- 36.Okumus M, Koybası M, Tuncay F, Ceceli E, Ayhan F, Yorgancioglu R, Borman P. Fibromyalgia syndrome: Is it related to vitamin D deficiency in premenopausal female patients? Pain Manag. Nurs. 2013;14(4):e156–e163. doi: 10.1016/j.pmn.2011.09.006. [DOI] [PubMed] [Google Scholar]
- 37.Bellato E, Marini E, Castoldi F, et al. Fibromyalgia syndrome: Etiology, pathogenesis, diagnosis, and treatment [published correction appears in Pain Res Treat. 2013;2013:960270] Pain Res. Treat. 2012;2012:426130. doi: 10.1155/2012/426130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jones JG. Chapter 32: Biochemistry of articular disorders. In: Marshall WJ, Lapsley M, Day AP, Ayling RM, editors. Clinical Biochemistry: Metabolic and Clinical Aspects. 3. Churchill Livingstone; 2014. pp. 636–645. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data and materials of this manuscript is available when requested by the Editors of Journal.