Abstract
Gaucher disease is an inborn error of metabolism resulting from the deficiency of the enzyme glucocerebrosidase and consequent accumulation of glucocerebroside within the lysosomes of macrophages. The clinical presentation is very diverse, depending on the age of onset and the severity of the disease, and results from the progressive infiltration of lipid-laden cells in various organs. Common manifestations of Gaucher disease include enlarged liver and/or spleen (hepatosplenomegaly), bone marrow disease (pancytopenia) and bone abnormalities, which are extremely variable and can affect multiple skeletal sites. While bone involvement of long bones and vertebrae is a well-recognised feature of Gaucher disease, jawbone involvement is less commonly noted. Here, we describe a case of a 63-year-old patient with type 1 Gaucher disease with a history of long-term use of bisphosphonates and who had presented with dental pain, with subsequent investigations confirming the radiological features of jaw involvement in Gaucher disease, including periodontal disease.
Keywords: lipid disorders, dentistry and oral medicine, haematology (incl blood transfusion)
Background
Gaucher disease is an ultra-rare disorder caused by biallelic mutations in GBA1, resulting in a marked decrease in glucocerebrosidase activity and consequent intralysosomal accumulation of glucosylceramide in cells of the monocytes/macrophage lineage, inducing their transformation into Gaucher cells.1 Lipid-laden cells infiltrate various tissues, including the bone marrow, spleen and liver, leading to a complex multisystem disease characterised by haematological, visceral and skeletal abnormalities and, in certain form, by neurological impairment.1 Gaucher disease is typically divided into three main phenotypes: non-neuronopathic (type I Gaucher disease, GD1) variant, which typically lacks primary involvement of the nervous system, and acute (type II, GD2) and subacute or chronic (type III, GD3) neuronopathic variants, where neurodegenerative manifestations occur. However, it is increasingly recognised that neurological symptoms and signs can also occur in patients with GD1 and that the three types of Gaucher disease represent a phenotypic continuum.2
Although the visceral changes can be significant in patients with Gaucher disease, the most debilitating and potentially irreversible complications result from a complex pattern of bone involvement, which can range from asymptomatic bone disease, such as Erlenmeyer flask deformity of the distal femur, to painful bone crisis, chronic bone pain and fragility fracture that markedly impact the patient’s quality of life and can be poorly responsive to enzyme replacement treatment.3
Femur and vertebrae are the most commonly involved bones in Gaucher disease; however, other sites can be affected such as the jawbones.1 4–9 Oral manifestations in patients with Gaucher disease are usually asymptomatic and frequently discovered incidentally on panoramic radiographs performed as part of routine clinical care. It is believed to be caused by two processes: bone marrow hyperplasia and abnormal haematopoiesis (similar to those observed in patients with thalassaemia and sickle cell disease) and infiltration of Gaucher cells.5 6
Generalised osteopenia with rarefaction of the trabecular structure, effacement of lamina dura, displacement of mandibular canal, pseudocystic radiolucent lesions in the molars and premolars with ‘worm eaten appearance’, apical root resorption, delayed eruption and cortical thinning have all been described in case series and observational studies.4–9 These features can occur early in life and require detailed dental assessment to enable appropriate treatment planning and dental management.9
Here, we report the case of a 63-year-old man with GD1 with symptomatic mandibular-maxillofacial involvement, in addition to haematological, splenic, hepatic and bone manifestations. We present this case to emphasise the importance of oral and periodontal examination, including radiographic investigations to be part of routine clinical care.
Case presentation
A 63-year-old man with a history of GD1 was referred for dental assessment in May 2018 due to a history of toothache and dentinal hypersensitivity to thermal stimuli involving the upper left and right posterior teeth of 4 months’ duration. The GD1 diagnosis had been preceded by a lifelong history of easy bruising and abdominal distension, with subsequent development of hepatosplenomegaly and pancytopenia. Bone marrow aspirate was suggestive of Gaucher disease and confirmed by enzymatic and molecular investigation: reduced β-glucocerebrosidase activity in leucocytes (0.8 nmol/hour/mg; normal 5.4–16.8 nmol/hour/mg) and GBA1 variant N370S/L444P, respectively. Treatment included fortnightly intravenous enzyme replacement therapy since October 2012 (60 units/kg body weight), calcium/vitamin D supplements and intravenous bisphosphonate (pamidronate every 6–12 months over a period of 5 years up to 2017). Additional diagnoses included cholecystectomy due to symptomatic gallstone disease at the age of 55 years, non-cirrhotic portal hypertension, Barrett’s oesophagus and Gaucher-related bone manifestations, including bone marrow infiltration by Gaucher cells in both femora and tibiae, prior femoral osteonecrosis and osteoporosis (figure 1). The patient had no history of diabetes, hypertension, autoimmune disease or smoking. Alcohol intake was 8 units/week.
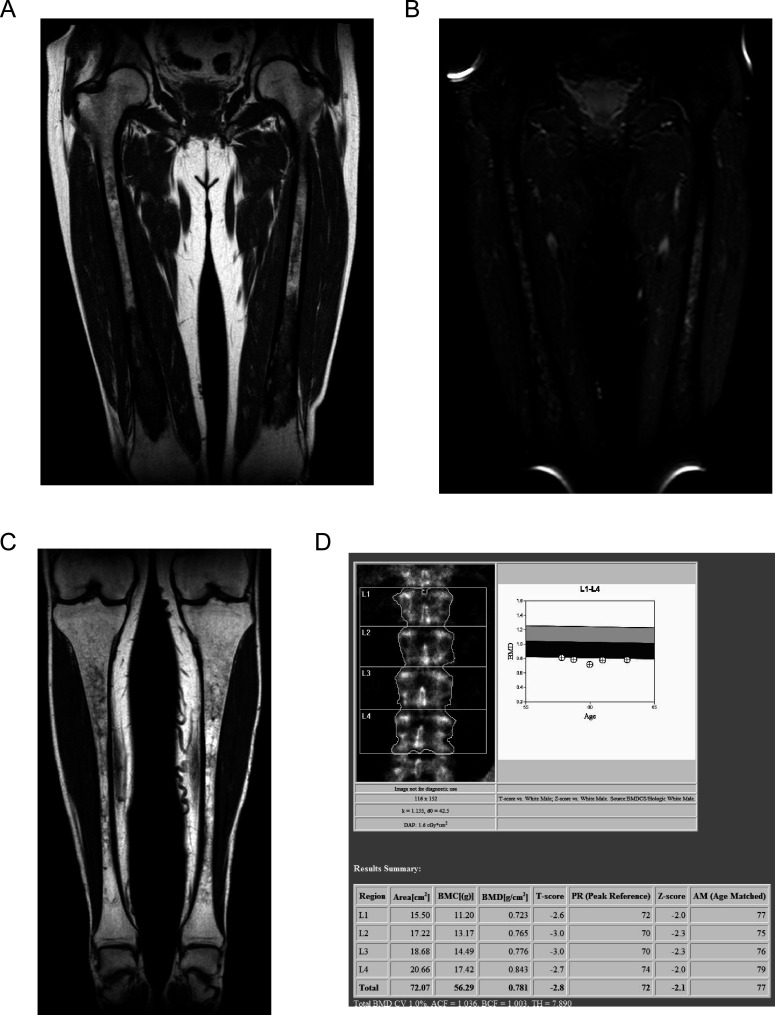
Figure 1.
(A) T1-weighted MR image shows low abnormal signal within both femora, indicating marrow infiltration by Gaucher cells. (B) T2-weighted MR image shows areas of low signal within the distal diaphysis of both femora in keeping with established bony infarction. (C) T1-weighted MR image shows diffuse low signal within both tibiae, indicating marrow involvement by Gaucher cells. (D) Bone mineral density by dual-energy X-ray absorptiometry shows osteoporotic values (T-score −2.8) in the lumbar spine.
The patient reported that he had received numerous dental restorations over the years, including two root canal treatments over the last 12 months. There was no history of spontaneous gingival bleeding or epistaxis.
Clinical examination revealed significant masticatory muscle pain, check biting bilaterally and tongue biting on the left side, tooth surface loss affecting the buccal sides of the teeth, ledges on the restorations of three teeth (lower left second molar, lower right second molar and upper left third molar), missing teeth (lower right premolar and upper wisdom teeth bilaterally) and staining due to tea and mild periodontal disease (1–2 mm clinical attachment loss; no increased teeth mobility). Gingival bleeding was not noted. The filled surface indices were not calculated. The patient had good oral hygiene status, which was assessed using the Simplified Oral Hygiene Index through the visual observation of debris and calculus.10 Gingival tissue was non-inflamed and there were no mucosal ulcers or areas of exposed or non-healing bones in the maxilla or mandible. The floor mouth, larynx and glottis were normal. Vital signs, body temperature, and submaxillary and cervical lymph nodes were within normal range. Cranial nerve examination was unremarkable.
Investigations
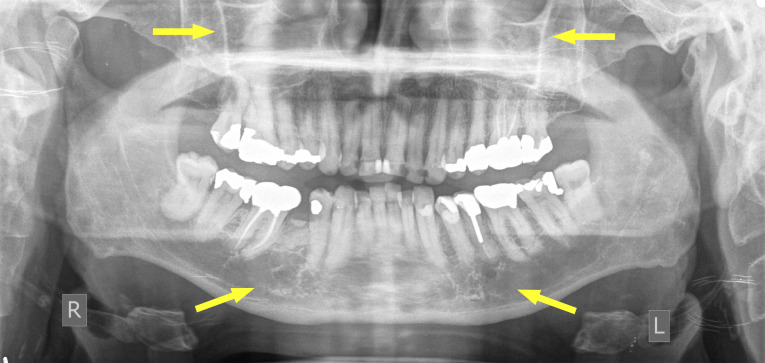
Abnormal laboratory studies included elevated levels of alanine transaminase (61 U/L; normal <41 U/L), gamma-glutamyl transferase (472 U/L; normal 8–61 U/L), alkaline phosphatase (185 U/L; normal <129 U/L) and low platelet count (84×109/L; normal 140–400×109/L). Haemoglobin (148.0 g/L), white cell count (4.98×109/L), glucose (5.1 mmol/L), calcium (2.35 mmol/L), corrected calcium (2.36 mmol/L), phosphate (0.97 mmol/L), vitamin D (108 nmol/L) and C reactive protein (4 mg/L) were within normal range. Dried blood spot glucosylsphingosine (Lyso-Gb1) 101 ng/ml (reference range less than 6.8ng/ml). Coagulation tests were normal. A full panoramic radiograph showed extensive restorations on the dentition, retained root of the lower right second premolar, periodontal bone loss affecting lower molars bilaterally and upper left molar, a sparse trabecular pattern bilaterally in the body of the mandible and opacification of the maxillary sinuses bilaterally (figure 2). These changes were considered to be likely secondary to proliferation of haematopoietic tissue and medullary expansion associated with Gaucher disease.
Figure 2.
Panoramic radiograph showing extensive restorations on the dentition, sparse trabecular pattern of the bone in the mandible inferior to the molars bilaterally and opacification of the maxillary sinuses bilaterally secondary to proliferation of haematopoietic tissue and medullary expansion associated with Gaucher disease.
Differential diagnosis
The differential diagnosis included simple odontogenic conditions, periodontal and periapical infections, osteonecrosis of the jaw due to the long-term use of bisphosphonates and other systemic disorders that can result in loss of periodontal tissue. The patient did not present signs of pulp disease or dental caries extending into the dentin or pulp. The odontogenic infections were ruled out as the oral examination and the panoramic radiograph did not show signs of local or systemic inflammation, nor evidence of dental abscess, sinus tracts or deep pockets.11 The medication-related osteonecrosis of the jaw (MRONJ) was excluded by both clinical evaluation and the absence of exposed bone, and radiological investigation, which did not demonstrate typical radiographic features of MRONJ, such as bone sclerosis, widening of the periodontal ligament space and osteolytic changes.12 13 Other systemic disorders affecting the periodontal disease were excluded as there were no clinical features suggestive of neoplasms, hormonal changes or autoimmune disease.14
Treatment
The patient was diagnosed with periodontitis and temporomandibular disorder exacerbated by the underlying Gaucher disease.6 7 14 15 The dental symptoms were due to tooth surface loss exacerbated by toothbrushing trauma and excessive consumption of dietary acids (citric fruits), for which dietary and oral hygiene instruction were given. Treatment also included removal of the ledges on the restorations and temporomandibular joint exercise. Bisphosphonate treatment was put on hold as a precautionary measure and an information sheet was given to the patient highlighting clinical symptoms related to MRONJ. Gaucher treatment with enzyme replacement therapy was continued.
Outcome and follow-up
The patient practised good oral care at home, limited consumption of dietary acids and performed Temporomandibular Joint (TMJ) exercises. He was discharged back to his local dentist for removing the ledges and following dental check-ups. At 6-month follow-up appointment in the Gaucher clinic, the patient reported resolution of the dental symptoms. A bisphosphonate holiday of 3 years was advised based on the low fracture risk at reassessment (age <75 years; no history of fragility fractures; no fracture during therapy; stable bone mineral density). He continues to be reviewed every 6 months in the Gaucher clinic.
Discussion
Gaucher-related bone manifestations range from asymptomatic radiological findings to pathological growth pattern, disabling bone deformities, osteonecrosis and osteoporosis with fragility fractures.3 Patients with Gaucher disease with skeletal involvement often have bone pain in various parts of their skeleton, but very rarely in the jawbones.5 6 Few cases have been described where patients were asymptomatic or presented with non-specific clinical presentations such as pain, submaxillary lymphadenopathy, alveolar abscess, recurrent gingival haemorrhage, loosening of teeth as signs of mandible involvement, or persistent fever and bifrontal headaches as signs of maxillary sinus involvement.4 7 It has been suggested that involvement of jaws in Gaucher disease is more common than previously thought, affecting 90% of patients, with nearly all cases involving the jaw and one-quarter involving the maxilla.5 6 9
In our case, a patient with a long history of Gaucher disease with visceral and bone complications—specifically chronic bone pain (femurs and spine), osteonecrosis of both femurs and osteoporosis in treatment with intravenous bisphosphonate had presented to our attention with toothache and dentinal hypersensitivity to thermal stimuli. The history, clinical and radiological investigations allowed us to diagnose a periodontitis with temporomandibular joint disease exacerbated by the underlying Gaucher disease. Gaucher disease has been recognised to have a moderate impact on the periodontal apparatus because it can cause loss of the alveolar bone trabecular architecture, which in turn has been suggested to favour the development of periodontal disease.15 16 This association has been confirmed in isolated case reports and narrative review.6 15 In the present case, the radiological investigation also showed sparse trabecular pattern and opacification of the maxillary sinuses: these findings are consistent with previous studies and reflect, respectively, a condition of generalised osteopenia and Gaucher cell infiltration.7
In view of the extensive restoration and the overall greater risk of infection observed in some patients with severe Gaucher disease due to an abnormal neutrophil chemotaxis, we ruled out the presence of odontogenic infections, which are very common dental conditions and can vary from well-localised infections to potentially life-threatening deep facial space infections.11 Early recognition and appropriate therapy are therefore essential to avoid progression and additional complications. In the present case, the clinical examination excluded the presence of pulpitis, deep periodontal pocket, periodontal abscess, as well as signs of systemic inflammation such as fever, swelling or lymphadenopathy.11 In addition, the panoramic radiograph did not show some typical features of the odontogenic infections such as periapical cysts or fractures.
Given the history of long-term use of bisphosphonates in this patient, the diagnostic investigation also focused on ruling out MRONJ.12 13 17 The risk of MRONJ varies depending on the individual bisphosphonate potency and the total dose, and its incidence in patients receiving bisphosphonate for non-cancer indications is very low and is most common following a dental extraction, although rarely could occur spontaneously.12 13 17 However, it is unknown whether patients with Gaucher disease may be at higher risk of developing MRONJ due to the underlying bony changes and the often needed long-term use of bisphosphonate in these individuals.3 18
In conclusion, we emphasise the importance of considering the possibility of Gaucher disease-related jaw involvement when encountering a patient with dental or oral symptoms as this can mimic other odontogenic conditions and pose a diagnostic challenge. Familiarity with the maxillofacial radiographic findings in this lysosomal storage disorder can facilitate the definitive diagnosis. We recommend regular dental examinations in the management of Gaucher disease.
Learning points.
Jawbone involvement in Gaucher disease is usually asymptomatic and underestimated.
Oral manifestations in Gaucher disease can pose a diagnostic challenge that could be mistaken for other conditions such as osteonecrosis of the jaw during bisphosphonate treatment.
The diagnosis of Gaucher involvement of the jaw is by a thorough medical history and clinical examination to rule out odontogenic lesions and radiographic evaluation for definitive diagnosis.
Acknowledgments
The authors thank the patient for providing consent for this manuscript publication.
Footnotes
Twitter: @damore_simona
Contributors: SD was involved in literature search and drafting of the manuscript; NK was involved in patient care and drafting of manuscript; UR was involved in patient care, conceived and supervised manuscript content, and final drafting of the manuscript. All authors read and approved the final manuscript.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Case reports provide a valuable learning resource for the scientific community and can indicate areas of interest for future research. They should not be used in isolation to guide treatment choices or public health policy.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Consent obtained directly from patient(s).
References
- 1.Beutler E, Grabowski GA. Gaucher disease. In: Scriver CR, Beaudet AL, Sly WS, eds. The metabolic and molecular bases of inherited disease. New York: McGraw-Hill, 2001: 3635–68. [Google Scholar]
- 2.Grabowski GA, Zimran A, Ida H. Gaucher disease types 1 and 3: phenotypic characterization of large populations from the ICGG Gaucher registry. Am J Hematol 2015;90 Suppl 1:S12–18. 10.1002/ajh.24063 [DOI] [PubMed] [Google Scholar]
- 3.Hughes D, Mikosch P, Belmatoug N, et al. Gaucher disease in bone: from pathophysiology to practice. J Bone Miner Res 2019;34:996–1013. 10.1002/jbmr.3734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Schwartz MR, Weycer JS, McGavran MH. Gaucher's disease involving the maxillary sinuses. Arch Otolaryngol Head Neck Surg 1988;114:203–6. 10.1001/archotol.1988.01860140101032 [DOI] [PubMed] [Google Scholar]
- 5.Carter LC, Fischman SL, Mann J, et al. The nature and extent of jaw involvement in Gaucher disease: observations in a series of 28 patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1998;85:233–9. 10.1016/s1079-2104(98)90432-2 [DOI] [PubMed] [Google Scholar]
- 6.Saranjam HR, Sidransky E, Levine WZ, et al. Mandibular and dental manifestations of Gaucher disease. Oral Dis 2012;18:421–9. 10.1111/j.1601-0825.2011.01898.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nobre RM, Ribeiro ALR, Alves-Junior SM, et al. Dentomaxillofacial manifestations of Gaucher's disease: preliminary clinical and radiographic findings. Dentomaxillofac Radiol 2012;41:541–7. 10.1259/dmfr/43023353 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zeevi I, Anavi Y, Kaplan I, et al. Jaws features in type 1 Gaucher disease. J Oral Maxillofac Surg 2013;71:694–701. 10.1016/j.joms.2012.09.020 [DOI] [PubMed] [Google Scholar]
- 9.Mohamed YSA, Zayet MK, Omar OM, et al. Jaw bones' involvement and dental features of type I and type III Gaucher disease: a radiographic study of 42 paediatric patients. Eur Arch Paediatr Dent 2020;21:241–7. 10.1007/s40368-019-00471-3 [DOI] [PubMed] [Google Scholar]
- 10.Greene JC, Vermillion JR. The simplified oral hygiene index. J Am Dent Assoc 1964;68:7–13. 10.14219/jada.archive.1964.0034 [DOI] [PubMed] [Google Scholar]
- 11.Jevon P, Abdelrahman A, Pigadas N. Management of odontogenic infections and sepsis: an update. Br Dent J 2020;229:363–70. 10.1038/s41415-020-2114-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ruggiero SL, Dodson TB, Fantasia J, et al. American Association of Oral and Maxillofacial Surgeons position paper on medication-related osteonecrosis of the jaw--2014 update. J Oral Maxillofac Surg 2014;72:1938–56. 10.1016/j.joms.2014.04.031 [DOI] [PubMed] [Google Scholar]
- 13.Khan AA, Morrison A, Hanley DA, et al. Diagnosis and management of osteonecrosis of the jaw: a systematic review and international consensus. J Bone Miner Res 2015;30:3–23. 10.1002/jbmr.2405 [DOI] [PubMed] [Google Scholar]
- 14.Horwitz J, Hirsh I, Machtei EE. Oral aspects of Gaucher's disease: a literature review and case report. J Periodontol 2007;78:783–8. 10.1902/jop.2007.060341 [DOI] [PubMed] [Google Scholar]
- 15.Albandar JM, Susin C, Hughes FJ. Manifestations of systemic diseases and conditions that affect the periodontal attachment apparatus: case definitions and diagnostic considerations. J Periodontol 2018;89 Suppl 1:S183–203. 10.1002/JPER.16-0480 [DOI] [PubMed] [Google Scholar]
- 16.Renvert S, Berglund J, Persson RE, et al. Osteoporosis and periodontitis in older subjects participating in the Swedish national survey on aging and care (SNAC-Blekinge). Acta Odontol Scand 2011;69:201–7. 10.3109/00016357.2010.549501 [DOI] [PubMed] [Google Scholar]
- 17.UK Chemotherapy Board . Medication-Related osteonecrosis of the jaw. guidance for the oncology multidisciplinary team. Report of a working Party on behalf of the UK chemotherapy board. UKCB, 2019. Available: https://www.rcplondon.ac.uk/guidelines-policy/medication-related-osteonecrosis-jaw-guidance-oncology-multidisciplinary-team [Accessed Jul 2021].
- 18.Khan A, Hangartner T, Weinreb NJ, et al. Risk factors for fractures and avascular osteonecrosis in type 1 Gaucher disease: a study from the International collaborative Gaucher group (ICGG) Gaucher registry. J Bone Miner Res 2012;27:1839–48. 10.1002/jbmr.1680 [DOI] [PubMed] [Google Scholar]