Abstract
Liver diseases have been a common challenge for people all over the world, which threatens the quality of life and safety of hundreds of millions of patients. China is a major country with liver diseases. Metabolic associated fatty liver disease, hepatitis B virus and alcoholic liver disease are the three most common liver diseases in our country, and the number of patients with liver cancer is increasing. Therefore, finding effective drugs to treat liver disease has become an urgent task. Chinese medicine (CM) has the advantages of low cost, high safety, and various biological activities, which is an important factor for the prevention and treatment of liver diseases. This review systematically summarizes the potential of CM in the treatment of liver diseases, showing that CM can alleviate liver diseases by regulating lipid metabolism, bile acid metabolism, immune function, and gut microbiota, as well as exerting anti-liver injury, anti-oxidation, and anti-hepatitis virus effects. Among them, Keap1/Nrf2, TGF-β/SMADS, p38 MAPK, NF-κB/IκBα, NF-κB-NLRP3, PI3K/Akt, TLR4-MyD88-NF-κB and IL-6/STAT3 signaling pathways are mainly involved. In conclusion, CM is very likely to be a potential candidate for liver disease treatment based on modern phytochemistry, pharmacology, and genomeproteomics, which needs more clinical trials to further clarify its importance in the treatment of liver diseases.
Keywords: liver diseases, natural agents, toxicity, clinical trials, potential application, Chinese medicine
Introduction
Chinese medicine (CM) is an effective drug treatment system with a history of thousands of years. It is used for disease prevention, treatment and diagnosis. CM is characterized by individualized adjustment of multiple components and multiple targets, which makes the body change from an abnormal state to a normal state (Wang et al., 2018). It has made an indelible contribution to human health and is considered a potential natural source of therapeutic drugs (Hesketh and Zhu, 1997; Chan and Ng, 2020). For example, Tu won the 2015 Nobel Prize for discovering and developing artemisinin in Artemisia annua Linn. It is a clear example to prove the therapeutic potential of CM and is of great significance to the continued development of the field (Tu, 2016). Besides, this field has huge and undeveloped resources. Screening and providing effective monomer chemicals are important means of CM to promote the development of medicine in the world (Wang et al., 2018).
Liver diseases are serious diseases threatening the whole human health, mainly including metabolic associated fatty liver disease (MAFLD), alcoholic liver disease (ALD), chronic viral hepatitis (e.g., hepatitis B virus (HBV) and hepatitis C virus (HCV) infections), autoimmune hepatitis, hepatic schistosomiasis, drug-induced liver injury, liver cirrhosis (LC), hepatocellular carcinoma (HCC), and so on (Li, Q. et al., 2018; Wang et al., 2014). China has the highest incidence of liver diseases in the world, and about 300,000–400,000 people die from various liver diseases each year. According to the data, MAFLD, HBV and ALD are the three most common liver diseases in China, with the incidence of 49.3, 22.9 and 14.8% respectively (Wang et al., 2014).
At present, CM has shown significant efficacy in the treatment of liver diseases, such as Rheum palmatum L. (Jin et al., 2005; Yang et al., 2012; Neyrinck et al., 2017), Silybum marianum (L.) Gaertn. (Alaca et al., 2017; Jindal et al., 2019), and Sophora flavescens Ait. (Yang et al., 2018; Yim et al., 2019). Furthermore, liver diseases are various, and the course of each disease is also different. Fortunately, CM can effectively treat a variety of liver diseases, and it has played an important role in the prevention and treatment of liver diseases. For example, Zingiber officinale and Glycyrrhiza uralensis Fischer can effectively treat ALD and MAFLD (Jung et al., 2016; Kandeil et al., 2019), and Rhizoma Coptidis can be used in the treatment of hepatitis virus (Hung et al., 2018). For more serious liver diseases, such as liver cirrhosis and liver cancer, Salvia offificinalis L. and Portulaca oleracea L. have shown good effects (Guoyin et al., 2017; Jiang, Y. et al., 2017). Besides, according to relevant records, the variety of CM commonly used in the treatment of liver diseases is up to 90 kinds (Wu, 2001). It can be seen that the resources of CM for the treatment of liver diseases are rich and valuable, which is worthy of further research and development.
In this review, we collected relevant literature in recent 6 years (2015–2020) through CNKI, PubMed, ScienceDirect and Google academic, and analyzed the application, toxicology and clinical data of CM and their related compounds, aiming to dig out more CM with potential biological activities for liver diseases, and promote their application value in the treatment of liver diseases, further providing relevant reference for the clinical application CM.
Characteristics of Several Important Liver Diseases
The Three Most Common Liver Diseases in China
MAFLD
MAFLD is a clinical syndrome characterized by hepatocyte steatosis and increased lipid deposition with the exception of alcohol and other clear liver-damaging factors (Mantovani et al., 2019). It is associated with obesity, insulin resistance, type 2 diabetes mellitus, hypertension, hyperlipidemia, and metabolic syndrome (Younossi, 2019). MAFLD is a broad umbrella term for a range of liver disorders, from non-alcoholic fatty liver (NAFL) to non-alcoholic steatohepatitis (NASH). It is called NAFL if it is only steatosis (fatty liver) and NASH if there is severe inflammation and liver cell damage (steatohepatitis). The course of MAFLD is complex and variable, which can lead to cirrhosis and liver cancer in severe cases (Friedman et al., 2018).
The pathogenesis of MAFLD mainly includes abnormal lipid metabolism, oxidative stress, inflammasome activation, insulin resistance, mitochondrial dysfunction, and genetic determinants (Buzzetti et al., 2016). Abnormal lipid metabolism in hepatocytes is the initial factor for MAFLD. When the number of fatty acids entering the liver is greater than their oxidation and secretion, the lipid accumulates in the liver, resulting in hepatic lipid deposition (Onyekwere et al., 2015), which leads directly to MAFLD. Furthermore, excessive lipid deposition further aggravates tissue damage by promoting the production of reactive oxygen species (ROS) and a series of pathological changes, such as the peroxidation of cells themselves, the release of pro-inflammatory factors and the infiltration of inflammatory cells, damaged hepatocytes activate the nuclear factor kappa-B (NF-κB) pathway, thus inducing the production of proinflammatory cytokine tumor necrosis factor-α (TNF-α) and interleukin-1β/-6 (IL-1β, IL-6) (Buzzetti et al., 2016; Xiao et al., 2020). These inflammatory factors can not only induce the activation of astrocytes and the remodeling of cell matrix, but also accelerate the progression of the disease by promoting insulin resistance. In addition, MAFLD is strongly associated with gut microbes, some of which carry genes that ferment dietary sugars into ethanol. When released into the bloodstream, they will increase oxidative stress and inflammation in the liver. In the liver, alcohol dehydrogenase metabolizes ethanol into toxic acetaldehyde, which forms adducts with proteins and other molecules in the cell because of its electrophilic properties, resulting in the loss of hepatocyte structure and function (Kolodziejczyk et al., 2019).
HBV Infection
HBV, a part of the Hepadnaviridae family, consists of nucleocapsid, envelope, and three complete membrane proteins (Seitz et al., 2007), which is a partially double-stranded and non-cytopathic DNA virus. The virus replicates the DNA by reverse transcription of the pre-RNA genome and has many serological markers such as HBsAg and anti-HBs, HBeAg and anti-HBe, and anti-HBc IgM and IgG (Trépo et al., 2014; Hu and Liu, 2017). HBV is the most common chronic virus in the world. Infected cells produce covalently closed circular DNA intermediates and integrated sequences that act as transcription templates for viral proteins (Fanning et al., 2019). HBV is transmitted through a number of routes, but mainly in the form of blood and body fluids, including perinatal and mother-to-child transmission, as well as sexual and extraintestinal patterns (Yuen et al., 2018).
At present, vaccination is still the most effective tool to prevent HBV infection, but there are also other therapeutic approaches, such as antiviral drugs that directly act on virus replication (interferon) and immune modulators (including reverse-transcriptase inhibitors, primarily a nucleoside or nucleotide analogue) (Yuen et al., 2018). These treatments can effectively inhibit HBV replication, but the disadvantages are the long-term medication and side effects. In addition, HBV infection can lead to chronic hepatitis and a series of complications, and studies have shown that HBV may persist in the body even after the infected person has fully recovered (Rehermann et al., 1996; Shi and Zheng, 2020). If immunosuppression-mediated host immune control is weakened, or several therapies and drugs have a direct effect on HBV replication, HBV may be reactivated (Shi and Zheng, 2020). Therefore, it is urgent to find a more effective HBV therapy to ensure the health of all human beings.
ALD
ALD refers to hepatocyte necrosis and destruction of normal liver function under the action of ethanol for a long time, which is a series of liver diseases including fatty liver, alcoholic hepatitis, cirrhosis, and its complications (such as ascites, portal hypertension-related bleeding, hepatic encephalopathy, and HCC) (Singal et al., 2018). The disease initially presents as alcoholic fatty liver disease, then gradually develops into alcoholic cirrhosis, even extensive hepatocyte necrosis, eventually inducing liver failure (Penny, 2013; Hu et al., 2019).
Sustained large quantity of alcohol stimulation is the primary factor of ALD. The pathogenesis is complicated and varied, mainly related to genetics, oxidative stress, hepatic steatosis, hepatic inflammation, and so on (2018). There is some evidence that aldehyde dehydrogenase2*2 and alcohol dehydrogenase 1B*3 alleles are closely related to alcoholic liver disease, and they can have some kind of chemical reaction with alcohol to achieve rapid metabolism (Agrawal and Bierut, 2012; Dodge et al., 2014); transmembrane 6 superfamily member 2 gene mutation can lead to the accumulation of liver fat, so that the disease will develop into a bad situation (2018); patatinlike phospholipase domain-containing protein 3, which mediates triglyceride hydrolysis in adipocytes, is closely related to lipid metabolism in the liver, but the mechanism of how it affects ALD is unclear (Salameh et al., 2015; BasuRay et al., 2017). At the same time, membrane-bound O-acetyltransferase domain-containing protein 7 is also an important genetic material related to ALD, but its mechanism is not clear (2018).
Oxidative stress plays a crucial role in the pathogenesis of ALD. In biological systems, free radicals include oxygen free radicals and nitrogen free radicals, among which oxygen free radicals and non-free radicals such as hypochlorite and ozone are called ROS. Under normal circumstances, the body contains antioxidants (such as superoxide dismutase (SOD), catalase, glutathione (GSH), glutathione peroxidase, glutathione transferase, heme oxygenase bilirubin etc.) and ROS in a state of balance, which are not harmful to the human body (Li et al., 2015). But in the case of long-term alcohol abuse, the reduction in the level or activity of antioxidants in the body causes oxidative stress. Alcohol may also increase the level of ROS. For example, ROS and nicotinamide adenine dinucleotide (NADH) are produced when ethanol is oxidized to acetaldehyde by alcohol dehydrogenase in the liver. Acetaldehyde is oxidized to acetic acid in mitochondria, which stimulates the body to produce large amounts of ROS (Li et al., 2014). NADH also interferes with the mitochondrial electron transport system and promotes ROS production (Ceni et al., 2014). Alcohol can also activate the NAD (P) H oxidase in hepatocytes, leading to an increase in the production of superoxide (Kalyanaraman, 2013). There is also evidence that another important pathophysiological mechanism of ALD is the interaction between endotoxin and Kupffer cells (KCs). Long-term high alcohol intake can induce low levels of intestinal endotoxemia, and increase intestinal permeability, causing Gram-negative bacteria to enter the hepatic portal circulation to suppress immune function (Mello et al., 2008; Gao and Liu, 2016). KCs recognize and clear gut-derived endotoxins, and promote oxidative stress and inflammatory response through their interaction (Yang and Wei, 2017).
Other Liver Diseases
HCV Infection
Hepatitis C is an infectious disease caused by HCV. HCV is an RNA virus, 45–65 nm in diameter, encapsulated in a lipid bilayer, belonging to the Flaviviridae family (Manns et al., 2017). HCV enters its target cells by a variety of host factors, including CD81, low-density lipoprotein receptor, dendritic cell-specific ICAM-grabbing non-integrin, claudin-1, and occludin. Among the different types of liver diseases, HCV is unique in requiring liver specific microRNA-122 replication (Luna et al., 2015). In addition, the genotypes of HCV are very rich. By the culture, analysis and identification of HCV strains isolated from all parts of the world, seven major HCV genotypes were found, namely 1–7 (Manns et al., 2017). Genotype 1 is the most prevalent in the world, including 83.4 million cases (46.2% of all HCV cases), about a third of which are in East Asia. Genotype 3 ranks second in the world (54.3 million, 30.1%), genotype 2, 4 and 6 account for 22.8% of all cases, and genotype 5 accounts for less than 1% of the remaining cases (Messina et al., 2015).
HCV transmission is most commonly associated with direct percutaneous exposure to blood via blood transfusions, health-care-related injections, and injecting drug use (Spearman et al., 2019). Alcohol is also a common cofactor for HCV infection, and alcohol use is more strongly associated with the progression of liver fibrosis (Poynard et al., 1997). Secondly, HCV infection can induce the abnormal expression of two host microRNAs (miR-208b and miR-499a-5p) encoded by myosin genes in hepatocytes. MiR-208b and miR-499a-5p inhibit type I IFN signal transduction in infected hepatocytes by directly down-regulating type I IFN receptor expression (Jarret et al., 2016). In addition, chronic HCV infection can also lead to liver fibrosis, cirrhosis, hepatocellular carcinoma and other serious complications.
LC
LC is a pathological stage characterized by diffuse fibrosis, pseudolobules formation, and intrahepatic and extrahepatic vascular proliferation (He and Liu, 2021). It is one of the main causes of death in patients with liver diseases all over the world, and also the final result of the development of a variety of acute and chronic liver diseases. LC shows symptoms such as portal hypertension and liver dysfunction. At present, the diagnosis of LC mainly depends on the imaging of irregular nodular liver by ultrasound, CT or MRI and the evaluation of liver synthesis function. In clinical practices, LC is considered as an end-stage manifestation of liver pathology with a high mortality without liver transplantation treatment (Tsochatzis et al., 2014; Zhou et al., 2014). But liver transplantation requires a lot of ligands and money, which is not an easy thing to solve, so CM has become a more effective approach.
The pathological pathway of LC is very complicated, but the research has shown that it is closely related to the expression of some cells on the wall of hepatic sinus. Hepatic sinus walls are composed of three kinds of non-parenchymal cells (liver sinusoidal endothelial cells (LSECs), KCs and hepatic stellate cells (HSCs)), which are involved in the development of LC (Zhou et al., 2014). In non-diseased liver, HSCs are located in the subendothelial space of Disse and are primarily involved in the storage of retinoic acid, but HSC is activated in the area of liver injury (Friedman, 1993; Hernandez-Gea and Friedman, 2011). In this activated phenotype, HSC is the main source of collagen and non-collagen matrix proteins in fibrosis. Related studies have shown that LSECs can secrete the cytokine IL-33 to activate HSCs and promote fibrosis (Marvie et al., 2010). Secondly, the exfoliation and capillarization of LSECs were proved to be the main contributing factors of liver dysfunction in cirrhosis (Yokomori et al., 2012). Finally, KCs can mediate liver inflammation to aggravate liver damage and fibrosis (López-Navarrete et al., 2011). Cytokines such as platelet-derived growth factor, transforming growth factor-β (TGF-β), TNF-α, and Interferon also play a crucial role in the pathogenesis of liver fibrosis and cirrhosis (Zhou et al., 2014). It is worth mentioning that if a patient has been diagnosed with ALD, concomitant chronic hepatitis B or C infection will directly aggravate the liver injury, leading to more frequent and rapid occurrence of cirrhosis (Poynard et al., 1997).
HCC
HCC is the most common form of liver cancer, accounting for 90% of the total cases of liver cancer. Among the various chronic liver diseases, HCC is the final stage of the disease in some patients with LC. About 80% of HCC patients have the pathological basis of LC, and the rate of HCC in patients with cirrhosis disease base in the short-term can be 5–30% (El-Serag, 2012). HBV and HCV are major risk factors for the development of HCC (Llovet et al., 2021). Others include exposure to aflatoxin, excessive drinking, smoking, diabetes, and knowledge of other risk factors such as MAFLD has been gradually recognized (Forner et al., 2012; Forner et al., 2018). The high incidence of HCC is concentrated in developing countries such as China, mainly due to chronic HBV infection (Jemal et al., 2011). Until now, there has been no nationwide cancer screening in China. Once a patient develops HCC, not only does the patient face tremendous pain from radiation therapy, but the improvement in survival rates is very limited, if more potential anti-cancer drugs can be tapped from the CM system, it will be beneficial to HCC patients.
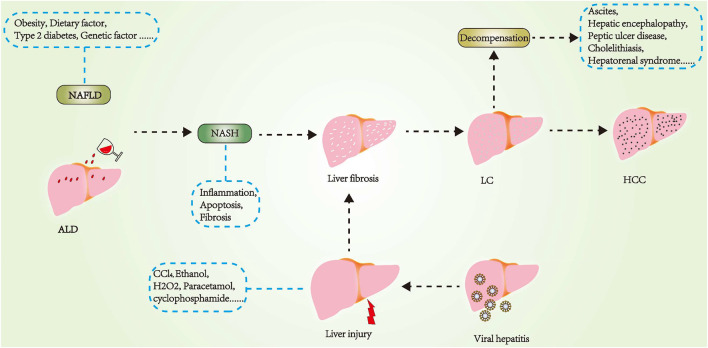
Figure 1 is a map of the major pathogenesis of some important liver diseases.
FIGURE 1.
Main pathogenesis of important liver diseases.
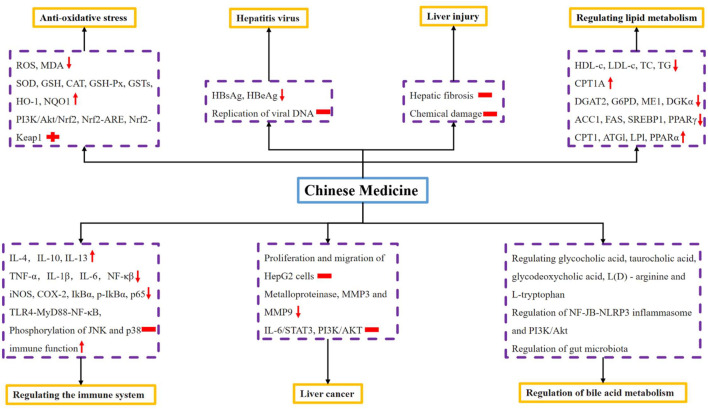
Pharmacological Effects of CM for Management of Liver Disease
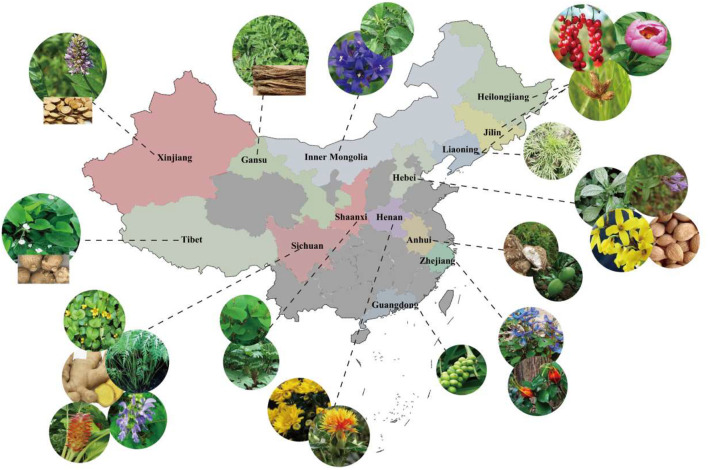
There are abundant varieties of natural CM resources in China, which is worthy of further development and utilization. For example, Figure 2 only shows the distribution of some CM for liver disease in the main producing area (also named “Daodi” producing area). Among them, many of the common CM have shown anti-liver disease activity, see Table 1. In addition, the pharmacological effects of CM on liver disease are summarized in Figure 3.
FIGURE 2.
Distribution of some Chinese medicine for liver diseases in main producing areas (also named “Daodi” producing area). Shaanxi: Rheum palmatum L., Polygonum cuspidatum Sieb.et Zucc.; Sichuan: Salvia miltiorrhiza Bunge., Zingiber officinale Rosc., Ligusticum chuanxiong Hort., Curcuma wenyujin Y. H. Chen et C. Ling, Lysimachia christinae Hance; Gansu: Angelica sinensis (Oliv.) Diels; Tibet: Alisma orientalis (Sam.) Juzep.; Hebei: Prunus persica (L.) Batsch, Forsythia suspensa (Thunb.) Vahl, Isatis indigotica Fort., Scutellaria baicalensis Georgi; Henan: Carthamus tinctorius L., Chrysanthemum morifolium Ramat.; Zhejiang: Gardenia jasminoides Ellis, Corydalis yanhusuo W. T. Wang; Liaoning: Artemisia scoparia Waldst. et Kit.; Guangdong: Alpinia oxyphylla Miq.; Anhui: Chaenomeles speciosa (Sweet) Nakai, Poria cocos (Schw.) Wolf; Xinjiang: Glycyrrhiza uralensis Fisch.; Inner Mongolia: Gentiana scabra Bunge, Isatis indigotica Fort.; Dongbei (Heilongjiang, Jilin, Liaoning): Schisandra chinensis (Turcz.) Baill., Paeonia lactiflora Pall., Sparganium stoloniferum Buch-Ham.
TABLE 1.
Some of the Chinese medicine used for the treatment of liver diseases are described in the standard and their biological activities.
| No | Latin name | English name | Family | Used part | Types of liver diseases that can be treated recorded in the standard | Reported biological activities associated with liver diseases |
|---|---|---|---|---|---|---|
| 1 | Rheum palmatum L | Rhei Radix et Rhizoma | Polygonaceae | Root and rhizome | Damp-heat jaundice a ; Acute infectious hepatitis b | Regulating gut microbiota Neyrinck et al. (2017), protective effect on high fat diet-induced hepatosteatosis, α-naphthylisothiocyanate induced liver injury and diethylnitrosamine (DENA)-induced hepatocellular carcinoma El-Saied et al. (2018); Yang et al. (2012); Yang et al. (2016a), anti-hepatic fibrosis Jin et al. (2005) |
| Rheum offcinale Baill | ||||||
| Rheum tanguticum Maxim.ex Balf | ||||||
| 2 | Angelica sinensis (Oliv.) Diels | Radix Angelicae Sinensis | Apiaceae | Root | Blood deficiency and chlorosis a ; Syndrome of blood deficiency c | Anti-inflammatory, anti-oxidative stress Mo et al. (2018) |
| 3 | Silybum marianum (L.)Gaertn | Herba Silybi | Asteraceae | Whole grass and achene | Fruit and extract for liver disease and jaundice d ; Fatty liver, chronic hepatitis, cirrhosis c ; Acute or chronic hepatitis, liver cirrhosis, fatty liver, metabolic toxic liver injury b | Protective effect on liver injury caused by cholestasis Alaca et al. (2017), protective effect against hepatotoxicity caused by deltamethrin Jindal et al. (2019), anti-oxidative stress Egresi et al. (2020); Zhu.et al. (2018a), regulating lipid metabolism Feng et al. (2019) |
| 4 | Artemisia scoparia Waldst. et Kit | Herba Artemisiae Scopariae | Asteraceae | Aboveground part | Infectious icteric hepatitis a , d | Anti-hepatocellular carcinoma Jang et al. (2017); Jung et al. (2018); Kim et al. (2018); Yan et al. (2018) |
| Artemisia capillaris Thunb | ||||||
| 5 | Gentiana scabra Bunge | Gentianae Radix et Rhizoma | Gentianaceae | Root and rhizome | Liver channel is hot and jaundice d ; Damp-heat jaundice, head distension and headache caused by liver and gallbladder excess fire c | Anti-hepatic fibrosis Qu et al. (2015), protective effect on liver injury caused by B19-NS1 Sheu et al. (2017) |
| 6 | Bupleurum chinense DC. | Radix Bupleuri | Apiaceae | Root | Chest pain, irregular menstruation a , c , d | Protective effect on liver injury caused by acetaminophen and D-galactosamine/lipopolysaccharide Wang et al. (2019a); Zou et al. (2018), anti-oxidative, anti-inflammatory Jia et al. (2019), enhancing immune function Zou et al. (2019) |
| 7 | Polygonum cuspidatum Sieb. et Zucc | Rhizoma Polygoni Cuspidati | Polygonaceae | Root and rhizome | Damp-heat jaundice, amenorrhea in women a , c , d | regulating lipid metabolism, anti-oxidative stress, alleviating insulin resistance Kim. et al. (2020a); Zhao. et al. (2019a) |
| 8 | Atractylodes macrocephala Koidz | Rhizoma | Asteraceae | Rhizome | Jaundice d | Anti-acute liver injury Han et al. (2016) |
| Atractylodis | ||||||
| Macrocephalae | ||||||
| 9 | Scutellaria baicalensis Georgi | Radix | Labiatae | Root | Jaundice a ; Headache due to liver fire, swelling and pain due to red eyes, damp-heat jaundice c | Relieving endoplasmic reticulum stress Dong et al. (2016), anti-hepatocellular carcinoma Wang. et al. (2020a), anti-oxidative stress, anti-inflammatory Park et al. (2017), anti-hepatic fibrosis Pan et al. (2015a) |
| Scutellariae | ||||||
| 10 | Curcuma longa L | Rhizoma Curcumae Longae | Zingiberaceae | Rhizome | Amenorrhea of women a ; Women with blood stasis and amenorrhea d ; Women have dysmenorrhea and amenorrhea c | Relieving endoplasmic reticulum stress Kim et al. (2017a), anti-oxidative stress, anti-inflammatory, protective effect on liver injury caused by CCl4, ethanol and methotrexate Lee. et al. (2017a); Moghadam et al. (2015); Uchio et al. (2017) |
| 11 | Ligusticum chuanxiong Hort | Rhizoma Chuanxiong | Apiaceae | Rhizome | Irregular menstruation, dysmenorrhea, chest pain b , c , d | Anti-hepatocellular carcinoma (Hu et al. 2015), protective effect against D-galactose-induced liver and kidney injury (Mo et al. 2017) |
| 12 | Glycyrrhiza uralensis Fisch | Radix | Leguminosae | Root | Hepatitis b | Hepatoprotective activities against CCl4/alcohol -induced liver injury Jung et al. (2016); Lin et al. (2017), anti-oxidative stress Cao et al. (2017) |
| Glycyrrhiza inflata Bat | Glycyrrhizae | |||||
| Glycyrrhiza glabra L | ||||||
| 13 | Prunus persica (L.) Batsch | Semen Persicae | Rosaceae | Mature seed | Amenorrhea, dysmenorrhea a , c , d | Anti-hepatocellular carcinoma Shen et al. (2017), protective effect on liver injury caused by CCl4 Rehman et al. (2021), anti-oxidative stress, anti-inflammatory Kim. et al. (2017b); Lee et al. (2008) |
| Prunus davidiana (Carr.) Franch | ||||||
| 14 | Sophora flavescens Ait | Radix | Leguminosae | Root | Jaundice a , c , d | Anti-hepatitis B virus Yang et al. (2018) |
| Sophorae Flavescentis | ||||||
| 15 | Sophora tonkinensis Gapnep | Radix Sophorae Tonkinensis | Leguminosae | Root and rhizome | Jaundice b , d | regulating lipid metabolism, anti-oxidative stress, anti-inflammatory Zhao et al. (2020) |
| 16 | Salvia miltiorrhiza Bunge | Radix | Labiatae | Root and rhizome | Irregular menstruation, amenorrhea and dysmenorrhea b , c , d ; Hepatosplenomegaly b | Protective effect on liver injury caused by paracetamol and lipopolysaccharide Gao et al. (2015); Zhou et al. (2015), anti-hepatocellular carcinoma Jiang et al. (2017b), anti-hepatic fibrosis Peng et al. (2018) |
| Salviae Miltiorrhizae | ||||||
| 17 | Aloe barbadensis Miller | Aloe | Liliaceae | The liquid concentrate of plant leaves | Liver heat a ; Liver fire, headache, red eyes, convulsion c ; Liver meridian excess heat, dizziness, headache, tinnitus, irritability, constipation b | Hepatoprotective effect against cartap- and malathion induced toxicityGupta et al. (2019), anti-inflammatory and anti-oxidant Klaikeaw et al. (2020) |
| Aloe ferox Miller | ||||||
| 18 | Coptis chinensis Franch | Rhizoma Coptidis | Ranunculaceae | Rhizome | Liver fire, red eyes, jaundice, disharmony between liver and stomach a ; Liver fire, red eyes, swelling and pain c | Anti-hepatocellular carcinoma Auyeung and Ko. (2009); Lin et al. (2004); Ma et al. (2018a), anti-hepatitis C virus Hung et al. (2018), protective effect on liver injury caused by CCl4 Ma. et al. (2018b) |
| Coptis deltoidea C. Y. Cheng et Hsiao | ||||||
| Coptis teeta Wall | ||||||
| 19 | Paeonia lactiflora Pall | Radix | Ranunculaceae | Root | Hypochondriac pain, blood deficiency and chlorosis, Irregular menstruation a ; Chest and abdomen rib pain, irregular menstruation b , d | Improving liver function, anti-inflammatory and anti-oxidant Wang. et al. (2020b) |
| Paeoniae Alba | ||||||
| 20 | Paeonia lactiflora Pall | Radix Paeoniae Rubra | Ranunculaceae | Root | Eye red swelling and pain, liver depression, hypochondriac pain, amenorrhea and dysmenorrhea a , c | Protective effect on liver injury caused by cholestasis Ma et al. (2018a); Ma et al. (2015) |
| Paeonia veitchii Lynch | ||||||
| 21 | Isatis indigotica Fort | Folium Isatidis | Brassicaceae | leaf | Jaundice a , c ; Jaundice, acute infectious hepatitis d ; Acute hepatitis b | Enhancing the endogenous antioxidant system Ding and Zhu (2020) |
| 22 | Isatis indigotica Fort | Radix Isatidis | Brassicaceae | Root | Acute and chronic hepatitis d ; Hepatitis c | Alleviating insulin resistance Li et al. (2019b) |
| 23 | Lycium barbarum L | Fructus Lycii | Solanaceae | fruit | The eyes are not clear a ; Yin deficiency of liver and kidney, dizziness c , d | Protective effect against paracetamol-induced acute hepatotoxicity Gündüz et al. (2015), anti-hepatocellular carcinoma Ceccarini et al. (2016), Regulating the immune system Tan et al. (2019) |
Cited from “Chinese Pharmacopoeia.”
Cited from “Zhong Yao Da Ci Dian”.
Cited from “Zhong Hua Ben Cao”.
Cited from “Quan Guo Zhong Cao Yao Hui Bian”.
(Note: doctor of traditional Chinese medicine holds that the liver stores blood and the liver is a sea of blood).
FIGURE 3.
Pharmacological effects of Chinese medicine on liver diseases.
Regulating Lipid Metabolism
Lipid uptake, esterification, oxidation, and fatty acid secretion all occur in hepatocytes. These processes are regulated by hormones, nuclear receptors, and transcription factors to maintain liver lipid homeostasis (Nguyen et al., 2008). If the balance of liver lipid metabolism is destroyed, the lipid will accumulate abnormally in the liver. Excessive lipid accumulation will lead to liver steatosis, insulin resistance and the development of fatty liver disease, and even induce oxidative stress, causing inflammation, cytotoxicity and aggravating liver injury. Therefore, maintaining normal lipid metabolism is an important function of the liver (Ding, H.-R. et al., 2018; Li, X. et al., 2019).
Many CMs have shown good effects in regulating lipid metabolism, such as Radix Bupleuri, Pericarpium Citri Reticulatae, Rhubarb, Polygonum Multiflorum, Coptis Chinensis, Artemisia Annua, Flos Lonicera and Radix Sophorae Tonkinensis. The results showed that the serum high-density lipoprotein cholesterol (HDL-C), TC and low-density lipoprotein cholesterol (LDL-C) levels of c57BL/6 mice were reduced by Citrus reticulata Blanco peel extract. The author further revealed that 0.2 and 0.5% of the extract could effectively prevent the micro fatty degeneration and excessive accumulation of lipid droplets in the liver (Ke et al., 2020). Rheum Palmatum L. can continuously reduce the accumulation of excess fat and the expression of lipogenic genes in the liver of male Sprague-Dawley rats induced by a high-fat diet. Concomitantly, increased phosphorylation of adenine monophosphate activated protein kinase (AMPK) and acetyl-CoA carboxylaze was observed (Yang, M. et al., 2016). In addition, Sophorae Tonkinensis water extract and Polygonum Multiflorum Thunb. extract alleviate nonalcoholic liver disease by enhancing hepatic carnitine palmitoyltransferase 1A activity to promote fatty acids β-oxidation, and regulating the protein response to lipid metabolism and expression in the liver to reduce lipid accumulation (Jung et al., 2020; Zhao et al., 2020).
It is worth mentioning that relevant studies of hepatic lipid metabolism were also conducted in fish. Addition of 200–400 mg/kg Radix Bupleuri extract to the daily diet of hybrid grouper fish can reduce the expression of lipogenesis-related genes, such as diacylgycerol acyltransferase 2, glucose-6-phosphate dehydrogenase, malic enzyme 1 and diacylglycerol kinase alpha (Zou et al., 2019). Lonicera japonica extract can effectively reduce the levels of LDL-C, triglyceride (TG) and total cholesterol (TC) in the serum of grass carp as well as the expression of lipogenic genes acc1, fas, SREBP1 and PPARγ, and increase the expression of liposoluble genes CPT1, ATGL, LPL and PPARα (Meng et al., 2019).
Liver Injury
Liver Fibrosis
Liver fibrosis belongs to chronic liver injury, mainly manifested as the accumulation of extracellular matrix (Tsuchida and Friedman, 2017), which is a dynamic process. Hepatocytes, activated hepatic stellate cells, endothelial cells, immune cells, and macrophages all participate in its establishment and regression (Campana and Iredale, 2017). Liver fibrosis is a pathological insult mainly caused by chronic liver disease (viral infection, alcoholic liver disease, NASH, etc). If not treated in time, it will continue to deteriorate and eventually progress to cirrhosis and even liver cancer.
The TGF-β/Smads pathway plays an important role in the regulation of liver fibrosis. In the background of liver fibrosis, Smad3 and Smad4 are pro-fibrosis, while Smad2 and Smad7 are anti-fibrosis (Xu et al., 2016). Meanwhile, TGF-β is also activated by the deposits in the fibrous extracellular matrix, and expressed and released from a variety of cells (Dewidar et al., 2019). The evidence has shown that Forsythiae Fructuse water extract (FSE), Curcuma Wenyujin, and Zingiber Officinale can effectively inhibit the development of liver fibrosis through the TGF-β/Smads signaling pathway (Hasan et al., 2016; Hu et al., 2020a; Xie et al., 2020).
Radix Salvia Miltiorrhiza (RSM) is the dry root and rhizome of Labiatae plant Salvia Miltiorrhiza Bunge, whose main functions include removing blood stasis, relieving pain, activating blood circulation, clearing the heart, and removing trouble (Commission, 2015). It is widely used in the treatment of liver fibrosis in clinic, but the specific mechanisms are not clear. The recent study of Yuan et al. showed that RSM improved liver fibrosis by increasing the activity of natural killer (NK) cells as well as the effects of NKG2D and NKp46 on NK cells, and inhibiting the activation of HSCs in vivo and in vitro (Peng et al., 2018). Another study showed that the mixture of RSM extract and Astragalus Membranaceus extract at a ratio of 1:1 could regulate the expression of TGF-β1 and Cyclin D1 to improve liver fibrosis and the liver functions, especially having a good effect on reducing the cyclin D1 expression (Cao et al., 2020). In addition, many CM have anti-fibrosis activities. For example, Gentiana Scabra bage inhibits fibrosis by reducing the expression of hepatic type I and type III collagen proteins in rats (Qu et al., 2015). Ginkgo biloba is also a common CM mainly used in coronary heart disease, angina pectoris, and hyperlipidemia (Commission, 2015). Wang et al. found that Ginkgo biloba extract could improve liver fibrosis by inhibiting inflammation, HSC activation, and hepatocyte apoptosis, which may be related to the p38MAPK, NF-κB/IκBα, and Bcl-2/Bax signaling pathway (Wang et al., 2015).
Chemical Liver Injury
Chemical liver injury is mainly caused by alcohol, toxic chemicals, and drugs. As we all know, the liver has dual blood supply of hepatic artery and hepatic vein, which is the main detoxification organ of human body. The liver plays a core role in biotransformation and excretion of foreign compounds, so it is the main target of the adverse reactions of drugs and other heterologous organisms (Holt and Ju, 2010). Secondly, the liver is the initial contact site of alcohol, chemical toxic substances, and the oral drugs absorbed through the intestine, so it is vulnerable to chemical damage. At the same time, electrophilic compounds and free radicals are the intermediate products of many chemical substances after liver metabolism. These substances may change the structure and function of cell macromolecules, and even lead to the occurrence of liver cancer (Gu and Manautou, 2012).
At present, a variety of CM are widely used for chemical injuries. Both Schisandra Sphenanthera extract and Polygonatum Sibiricum water extract can regulate alcoholic liver injury in mice through the nuclear factor-erythroid 2-related factor 2 (Nrf2)-antioxidant responsive element (ARE) signaling pathway (Wang, G. et al., 2021; Zeng et al., 2017). The liver damage caused by CCl4 can be alleviated by Curcuma longa L. extract and Prunus persica Seeds Extract, which is mainly related to inhibiting liver oxidative stress, and increasing the Nrf2 and NQO-1 levels, as well as reducing type Ⅲ collagen mRNA expression (Lee, G.-H. et al., 2017; Rehman et al., 2021). In addition, Hedyotis Diffusa water extract, Ligusticum Chuanxiong Hort, and Panax ginseng can also be used to respectively relieve the chemical damage caused by hydrogen peroxide and D-galactose (Gao et al., 2016; Mo et al., 2017). It is worth mentioning that a large number of CM can also alleviate drug-induced liver injuries. Paracetamol (acetaminophen) is a commonly used drug in clinic, which is mainly used for cold-induced fever, headache, joint pain, neuralgia, migraine, dysmenorrhea, and so on. Lycium Barbarum extract can significantly improve paracetamol-induced apoptosis to protect the liver from chemical damage (Gündüz et al., 2015), and Isatidis Folium can enhance the endogenous antioxidant system and reduce paracetamol-induced liver damage in mice (Ding and Zhu, 2020). Ahmed et al. also found that Panax ginseng could be used as a hepatoprotective agent, which prevented cyclophosphamide (with immunosuppressive and anti-cancer potential)-induced liver injury by reducing the expression of TNF-α, IL-1β and Caspase3 genes, as well as increasing the BCL-2 gene expression, and its liver-protective effect is better than vitamin E (Abdelfattah-Hassan et al., 2019).
Anti-oxidative Stress
Oxidative stress is the main influencing factor of the pathogenesis of ALD and MAFLD. It has been briefly discussed in the previous content. When the level or activity of antioxidants in the human body is reduced, oxidative stress will occur. Due to the stimulation of external factors (such as alcohol), the body will produce a large amount of active oxygen, which is the key to the development of fatty liver into steatohepatitis. GSH is an endogenous antioxidant, which is widely present in animals. Excessive oxidative stress can cause GSH consumption and lead to the accumulation of ROS (Li, X. et al., 2019). In addition, cytochrome P4502E1 (CYP2E1) plays a key role in the generation of ROS, which is also induced by alcohol (Leung and Nieto, 2013). Calculus bovis is a commonly used CM for fever, faintness, stroke and phlegm. The evidence showed that calculus bovis could inhibit oxidative stress in hepatocytes by reducing ROS and increasing SOD content, thereby achieving the liver-protective effect on mice with nonalcoholic fatty liver. And curcuma longa hot water extract and zingiber officinale hydroalcoholic extract can reduce the level of GSH to protect the liver.
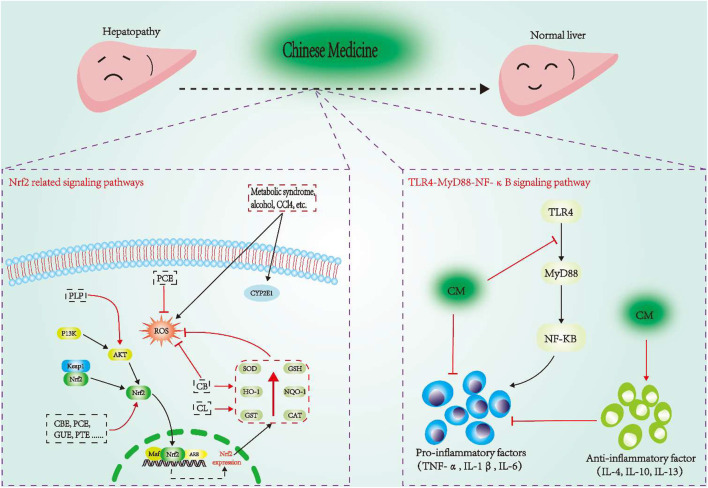
Nrf2 is an important redox-sensitive transcription factor, and controls the basic and induced expression of a series of antioxidant response element-dependent genes, which is beneficial to improve the body’s oxidative stress state, thus regulating the physiological and pathological consequences under oxidant exposure (Ma, 2013). Under normal physiological conditions, Nrf2 is locked in the cytoplasm by Keap1. But when the cells are attacked by ROS or electrophiles, Nrf2 will dissociate from Keap1 and quickly translocate into the nucleus, first forming a heterodimer with the small Maf protein, and then combining with the ARE, which finally transcribes and activates the expression of the antioxidant enzyme genes regulated by Nrf2 (Ho et al., 2012; Heiss et al., 2013). In addition, the signal pathways related to Nrf2 (such as Nrf2-Keap1 and Nrf2-ARE) in the oxidative stress system have been widely recognized, especially the Nrf2-Keap1 pathway, which is an anti-stress mechanism inherited from our ancestors, as well as a defense system to maintain the homeostasis of the cells (Buendia et al., 2016; Bellezza et al., 2018). As reported, Polygonum Cuspidatum extract could reduce oxidative stress by targeting the Keap1/Nrf2 pathway, and down-regulate the levels of sterol regulatory element bending protein 1, fatty acid synthase, and stearoyl coenzyme alpha desaturase-1 to prevent hepatic lipid accumulation in fructose-fed rats (Zhao, X.-J. et al., 2019). Paeonia Lactiflora Pall. (PLP) can increase the expression of AKt, Nrf2, HO-1, NQO1 and GCLC, and activate the PI3K/Akt/Nrf2 pathway to enhance the antioxidant system, thereby reducing ANIT-induced liver tissue damage (Ma et al., 2015). In addition, Citrus Reticulata Blanco peel extract, Glycyrrhiza Uralensis ethanol extract, and Polygonum Multiflorum Thunb. ethanolic extract can directly activate the Nrf2 to regulate the redox state of liver injury (Cao et al., 2017; Ke et al., 2020; Lin, E.-Y. et al., 2018). The details are showed in Figure 4.
FIGURE 4.
Some CM treat liver disease through Nrf2 and TLR4-MyD88-NF-κB signaling pathway. CBE, Citrus reticulata Blanco peel extract; GUE, Glycyrrhiza uralensis ethanol extract; PTE, Polygonum multiflorum Thunb. ethanolic extract; PLP, Paeonia lactiflflora Pall.; CL, Curcuma longa; CB, Calculus bovis; PCE, Polygonum cuspidatum extract.
Regulation of Bile Acid Metabolism
Bile acids (BAs) are important components of bile, which have the functions of regulating metabolism, endocrine and immune (Chávez-Talavera et al., 2017). The liver is the site of bile acid synthesis. The primary bile acids, such as cholic acid and chenodeoxycholic acid, combine with glycine or taurine to form bound BAs, which are secreted into bile canaliculus through the transport proteins such as bile salt export pump and multidrug resistance associated protein 2, and are temporarily stored in the gallbladder and released through the bile duct. When BAs and other components of bile are discharged into the intestine together, they can promote the emulsification and absorption of dietary fat, cholesterol, and fat-soluble vitamins. About 90–95% of BAs are reabsorbed in the ileum through apical sodium-dependent bile acid transporter and ileal bile acid transporter (IBAT), and the remaining 5–10% of BAs are excreted in feces (Li and Chiang, 2014; Tripathi et al., 2018). BAs are the important physiological basis involved in the regulation of liver function and disease states. According to the data, the metabolism and inflammation related to obesity, type 2 diabetes, dyslipidemia, and MAFLD are all regulated by BAs (Chávez-Talavera et al., 2017). Therefore, BAs’ normal synthesis, transportation and excretion are vital factors for the homeostasis.
Cholestasis means that the bile cannot flow from the liver to the duodenum, and its flow is decreased, which is characterized by the excessive accumulation of bile acids and other toxic compounds (Crocenzi et al., 2012). Excessive accumulation of bile acids in the liver may cause liver damage, liver fibrosis, and eventually liver failure and biliary cirrhosis (Padda et al., 2011). The study has shown that PLP can regulate glycocholic acid, taurocholic acid, glycodeoxycholic acid, L (D)-arginine, and L-tryptophan, and these metabolites are related to bile acid secretion and amino acid metabolism, which is concluded that bile acid metabolism may be involved in the therapeutic effects of PLP on cholestasis (Ma et al., 2016). Ma et al. further demonstrated that PLP could alleviate cholestasis by regulating the NF-κB-NLRP3 inflammasome and the PI3K/Akt-dependent pathways (Ma, X. et al., 2018; Ma et al., 2015). Another study showed that the ethanol extract of Schisandra Chinensis could significantly protect the mice from intrahepatic cholestasis induced by cholic acid (Zeng et al., 2016). In addition, Schisandra Chinensis extract can also enhance the excretion of bile acids from the serum and liver to the intestine and feces, and adjust the intestinal microorganisms disturbed by the external factors to achieve the protective effects on liver injury caused by cholestasis (Li, D.-S. et al., 2020).
Regulating the Immune System
Inhibition of Inflammatory Response
Inflammation is the basis of a variety of physiological and pathological processes, mainly induced by infection and tissue damage (Medzhitov, 2008). When natural antioxidants are out of balance, the free radicals produced by different organisms and environments can further lead to various inflammation-related diseases (Arulselvan et al., 2016). As we all know, there are many kinds of cytokines involved in the inflammatory response. For example, TNF-α, IL-1β, and IL-6 play a pro-inflammatory role, by contrary, TGF-β, IL-4, IL-10, and IL-13 can inhibit the occurrence and progress of inflammation. There is evidence that the inflammatory mechanisms of the liver are essential for maintaining the homeostasis of the tissues and organs. When the inflammatory mechanisms are out of balance, the hepatic pathological process will be drived, such as chronic infection, autoimmunity, and malignant tumor (Robinson et al., 2016). FSE, Gentianae Macrophyllae extract, and Aloe vera can reduce inflammatory liver injury by reducing the serum concentration of TNF-α, IL-1β, IL-6, NF-κB, and other cytokines (Zhao et al., 2017; Cui et al., 2019; Hu et al., 2020a; Klaikeaw et al., 2020). Moreover, Radix Bupleuri extract and Schisandra Sphenanthera extract can directly inhibit the mRNA expression of TNF-α, IL-1β, and IL-6 to protect the liver (Chen et al., 2019; Jia et al., 2019). In addition, Angelica Sinensis Supercritical Fluid CO2 Extract can significantly inhibit D-galactose-mediated expression of inflammatory cytokines, such as iNOS, COX-2, IKBα, p-IκBα, and p65, protecting the liver and kidney tissues (Mo et al., 2018).
Toll-like receptor4 (TLR4)-myeloid differentiation factor 88 (MyD88)-NF-κB signaling pathway is a key pathway in the physiologic and biochemical reactions of diseases. It widely exists in various tissues and cells, which is one of the important signaling pathways that mediate the expression of inflammatory factors (Wu et al., 2017). As one of the important pathways associated with inflammatory response and hepatic fibrosis, its activation can lead to the release of downstream inflammatory factors and induce the production of TNF-α, IL-1β, and IL-6. Hu et al. found that FSE could improve the inflammatory state of liver fibrosis through the TLR4-MyD88-NF-κB pathway (Hu et al., 2020a). Jia et al. found that RBE could inhibit TLR4-MyD88-NF-κB signaling pathway to reduce H2O2-induced liver inflammation in tilapia (Jia et al., 2019). Another study showed that GME could also attenuate ALD by inhibiting the phosphorylation of JNK and p38 to inhibit the initiation of inflammation (Cui et al., 2019).
The molecular mechanisms of the CM alleviating liver diseases through inflammatory pathways are shown in Figure 4.
Enhancing Immune Function
Zou et al. found that adding 200–800 mg/kg RBE to the diet of hybrid grouper could effectively reduce the serum ALP, ALT, AST, and LDH contents. In addition, it could down-regulate the expression of apoptosis-related genes (caspase-9), and up-regulate the antioxidant genes (CAT) and immune-related genes (MHC2, IKKα, and TGF-β1) (Zou et al., 2019). Tan et al. reported that dietary supplementation of Lycium barbarum extract (0.50–2.00 g/kg) could effectively increase IL-10 and TGF-β1 mRNA levels in the liver of HFD-fed hybrid grouper (Tan et al., 2019). In addition, Ginkgo biloba extract not only improves the hepatic antioxidant status of HFD-fed hybrid grouper, and maintains normal liver histology and preserves liver function, but also up-regulates the expression of immune-related genes (MHC2 and TLR3) (Tan et al., 2018).
Hepatitis Virus
Some CM have inhibitory effects on hepatitis virus and can assist the treatment of patients with viral hepatitis. Some studies have shown that most of the terpenoids isolated from Flos Lonicerae can inhibit the secretion of HBsAg and HBeAg, as well as the DNA replication of HBV (Ge et al., 2019). In addition, Yang et al. found that the methanolic extract of Rhizoma Coptidis could block the attachment of HCV and the entry/fusion with host cells, which effectively inhibited the infection of pseudoparticles of HCV in Huh-7.5 cells, and hindered the infection of several HCV genotypes (Hung et al., 2018).
Liver Cancer
Currently, Western medicine and therapies are the main treatment strategies for liver cancer, but the overall prognosis of liver cancer patients is still very poor. Under such circumstances, it is extremely urgent to find a better method for the treatment of liver cancer. CM contains abundant treatment resources and has been used for the prevention of liver cancer for thousands of years. In modern China, CM has also been proven to be an effective method for the treatment of liver cancer. However, the theory of CM prevention and treatment of liver cancer is more widely accepted in China than abroad (Liao et al., 2020). According to relevant data, most CM can show anti-liver cancer effects. Ethanol extract of root of Prunus Persica can significantly inhibit the migration of liver cancer HepG2 cells and the expression of extracellular matrix metalloproteinases, MMP3 and MMP9. It is worth mentioning that it can also inhibit tumor growth in nude mice in vivo (Shen et al., 2017). Artemisia capillaris extract can inhibit the growth, migration and invasion of Huh7 and HepG2 liver cancer cells. This inhibitory effect is closely related to blocking the PI3K/AKT signaling pathway (Yan, Honghua et al., 2018). Jiang et al. further found that the anti-liver cancer effect of Artemisia capillaris extract is also related to the inhibition of the IL-6/STAT3 signal axis (Jang et al., 2017). Futhermore, Zheng et al. found that oral administration of portulaca oleracea extract to male AKR mice for seven consecutive days could contribute to the treatment of liver cancer. The results showed that the serum levels of IL-6, IL-1β, TNF-α and MDA in mice decreased after 7 days of treatment, while the activity of SOD increased. The pathological changes of the liver were significantly alleviated. Meanwhile, portulaca oleracea extract could effectively inhibit PI3K, Akt, mTOR, NF-κB and IκBα, and up regulate the expression of Nrf2 and HO-1. These effects are attributed to the protective effect of Portulaca oleracea extract on liver cancer by regulating PI3K/Akt/mTOR and Nrf2/HO-1/NF-κB pathway (Guoyin et al., 2017).
In addition, some CM can also achieve protection against liver cancer through various other effects. For examples, Astragalus membranaceus and Curcuma wenyujin promote the normalization of blood vessels in liver tumor endothelial cells by increasing the expression of CD34 and reducing the expression of HIF1a (Zang et al., 2019). Artemisia capillaris leaves can achieve pro-apoptotic effects on liver cancer cells by reducing the expression of XIAP and the release of cytochrome C through mitochondrial membrane potential (Kim et al., 2018). Besides, Ligustrum lucidum Ait. fruit extract can induce apoptosis and cell senescence of human liver cancer cell Bel-7402 by up-regulating p21. All in all, there are abundant resources of CM against liver cancer, which are worthy of our further development and utilization.
Other Anti-liver Disease Mechanisms
A large number of studies have shown that the occurrence of liver diseases is also closely related to endoplasmic reticulum stress and insulin resistance. Scutellaria baicalensis Georgi extract can regulate the endoplasmic reticulum stress and protect the liver by reducing the expression of glucose-related protein 78 (Dong et al., 2016). HFD increased the expression of adipose-derived carbohydrate response element binding protein and endoplasmic reticulum stress genes CHOP, x-box binding protein 1, and glucose regulated protein 78 in male wistar rats, and Ginger extract could restore these changes to normal state (Kandeil et al., 2019). Jung et al. reported that Polygonum multiflfluorum thunb. reduced nonalcoholic steatosis and insulin resistance by regulating the expression of the proteins on lipid metabolism and glucose transport in the liver (Jung et al., 2020).
Recently, the evidence has shown that gut microbiota play an important role in metabolism, immune system, and so on. The changes of gut microbiota and their function can promote the development of acute and chronic liver diseases. In addition, the destruction of intestinal barrier can make microorganisms transfer to the blood, and continuously cause inflammatory reaction, thus promoting liver injury, hepatic fibrosis, cirrhosis, and carcinogenic transformation (Shen et al., 2018; Chopyk and Grakoui, 2020). Rhubarb extract can promote some intestinal bacteria (such as Akkermansia muciniphila and Parabacteroides goldsteinii.) to participate in the intestinal barrier function, and alleviate liver inflammation caused by acute alcohol intake (Neyrinck et al., 2017). In addition, Schisandra chinensis bee pollen could inhibit the expression of LXR-α, SREBP-1c, and FAS genes, and regulate the structure of intestinal microflora in obese mice, so as to achieve the protective effect on MAFLD (Cheng et al., 2019).
Natural Agents From CM for Liver Disease Treatment
Polysaccharides and Glycosides
Polysaccharide is one of the active components of CM. The polysaccharides in CM have a wide range of biological activities in enhancing immunity, antiviral, anti-inflammation, anti-oxidation, and anti-tumor (Chen et al., 2016). Ginkgo biloba leaf polysaccharides and Astragalus polysaccharides can effectively inhibit liver steatosis (Yan et al., 2015; Huang et al., 2017). The polysaccharides from roots of Sophora flavescens can significantly inhibit the HBsAg and HBeAg secretion of HepG2.2.15 cells, and have good anti-HBV activity (Yang et al., 2018). In addition, the polysaccharides extracted from many CM have obvious protective effects on acute liver injury, such as Rhizoma Atractylodis Macrocephalae polysaccharides (Han et al., 2016), Angelica sinensis polysaccharides (Wang, K. et al., 2020), Poria Cocos polysaccharides (Wu, K. et al., 2018), Lycium barbarum polysaccharides (Wei et al., 2020), and Schizandra chinensis acidic polysaccharides (Yuan et al., 2018). Wang et al. reported that Paeoniae Radix Alba polysaccharides inhibited the NF-κB signaling pathway (including the liver infiltration of inflammatory CD4+ and CD8+ cells, and the overexpression of inflammatory cytokines IL-2, IL-6, and IL-10) to inhibit the immune inflammatory response in experimental autoimmune hepatitis mice (Wang, S. et al., 2020). Finally, it is also important that APS is the main active component extracted from Astragalus, which has been proved to have a significant inhibitory effect on many types of human solid tumors. A recent study showed that APS could reduce the activity of hepatoma cells and induce the apoptosis of HCC cells in a concentration-dependent manner. The study further showed that the results might be related to inhibiting the expression of Notch 1 in HCC cells (Huang et al., 2016).
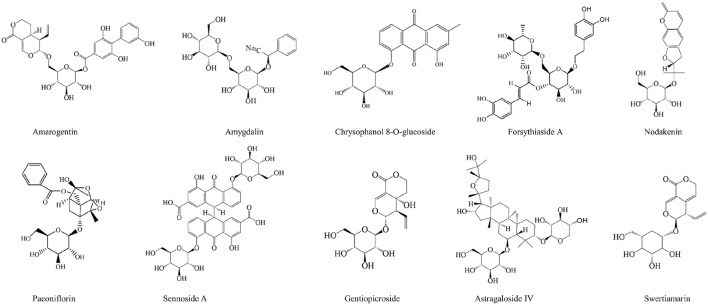
Glycosides are a class of compounds formed by linking the sugar or sugar derivative with another non-sugar substance through the terminal carbon atom of the sugar. The studies have shown that most glycosides have good hepatoprotective effects on liver, such as amygdalin, amarogentin, and forsythiaside A (Pan, C.-W. et al., 2015; Tang et al., 2019; Zhang et al., 2017). Chrysophanol 8-o-glucoside, extracted from Rheum palmatum, can significantly inhibit the gene expression of α-SMA and collagen I, and inhibit the phosphorylation of STAT3 by inhibiting the nuclear translocation of p-STAT3, thus alleviating fibrosis and achieving liver protection (Park et al., 2020). What’s more, Gentiopicroside not only protects alcoholic liver disease by improving lipid metabolism imbalance and mitochondrial dysfunction caused by alcohol (Yang, H.-X. et al., 2020; Zhang et al., 2021), but also treats alcoholic liver cancer by regulating the activation of P2x7R-NLRP3 inflammasome (Li, Xia et al., 2018). It is worth mentioning that astragaloside IV can inhibit hepatoma cells by inhibiting multidrug resistance-associated protein 2, and long noncoding RNA ATB (Li, Y. et al., 2018; Qu et al., 2020).
The specific information of polysaccharides and glycosides is shown in Table 2. In addition, the chemical structures of the glycosides with therapeutic effects on liver diseases are shown in Figure 5.
TABLE 2.
Summary of polysaccharides and glycosides with significant anti-liver disease activity.
| Compounds | Source | The species investigated | Dose | Mechanisms | References |
|---|---|---|---|---|---|
| Polysaccharides | |||||
| PRAM2 | Rhizoma Atractylodis Macrocephalae | Male ICR mice | 50, 100, 200 mg/kg | Inhibition of NOS activity and NO level and its reduction of the production of free radicals | Han et al. (2016) |
| Radix isatidis polysaccharide | Radix isatidis | 3T3-L1 preadipocytes | 25, 50, 100 μg/ml | Improvement of the glucose metabolism, lipid metabolism and oxidative stress | Li, et al. (2019c) |
| Male Wistar rats | 25, 50, 100 mg/kg | ||||
| Salvia miltiorrhiza polysaccharide | Salvia miltiorrhiza | Chickens | 0.5, 1, 2 g/L | Down-regulation of the contents of ALT, AST, and MDA, and up-regulation of the contents of GSH and CYP450 | Han et al. (2019) |
| Chicken hepatocytes | 100, 200, 500 μg/ml | ||||
| Angelica sinensis polysaccharide | Angelica sinensis | L02 cells | 200, 400, 800 μg/ml | Through regulating lipid metabolism, anti-inflammation, anti-oxidation and inhibiting HSC activation | Ma et al. (2020); Wang et al. (2016); Wang. et al. (2020c) |
| ICR male mice | 100, 300, 500 mg/kg | ||||
| Male Balb/c mice | 1.5, 6 mg/kg | ||||
| Murine splenocytes | 5, 25,125 μg/ml | ||||
| Male C57BL/6J mice | 200 mg/kg | ||||
| Primary splenocytes | 50, 100, 200 μg/ml | ||||
| Codonopsis pilosula polysaccharide | Codonopsis pilosula | Female ICR mice | 100, 150, 200 mg/kg | Through antioxidant effect | Liu et al. (2015) |
| Poria cocos polysaccharide | Poria cocos | Male Kunming mice | 200, 400 mg/kg | By suppressing cell death, reducing hepatocellular inflammatory stress and apoptosis, and Hsp90 bioactivity | Wu et al. (2018c); Wu. et al. (2019b) |
| AML12 cells | 20, 40 g/L | ||||
| Lycium barbarum polysaccharide | Lycium barbarum | L02 cells | 24 μg/ml | By reversing oxidative injury, inflammatory response and TLRs/NF-κB signaling pathway expression | Gan. et al. (2018b); Wei et al. (2020) |
| Male wistar rats | 400, 800, 1600 mg/kg | ||||
| Astragalus membranaceus-Polysaccharide | Astragalus membranaceus | HFSTZ Mice | 500 mg/kg | Through improving peripheral metabolic stress, activating hepatic insulin signaling | Huang et al. (2016); Huang et al. (2017); Sun et al. (2019) |
| C57BL/6 mice | 800 mg/kg | ||||
| HCC cells | 0.1, 0.5, 1 mg/ml | ||||
| SFP-100 | Sophora flavescens | Female Balb/c mice | 500 mg/kg | By decreasing hepatocytes apoptosis, inhibit the infiltration of neutrophils and macrophages into liver | Yang et al., (2018) |
| L02 cells | 10, 50, 250 μg/ml | ||||
| HepG2.2.15 cells | 50, 100, 250, 500 μg/ml | ||||
| Codonopsis lanceolata polysaccharide | Codonopsis lanceolata | Male C57BL/6 mice | 100 mg/kg | Through activating anti-oxidative signaling pathway | Zhang, et al. (2020a) |
| STRP | Sophora tonkinensis | Male ICR mice | 50, 100, 200 mg/kg | By inhibiting MDA, ROS generation and increasing liver GSH, GPx, T-SOD, CAT levels | Cai et al. (2018); Shan et al. (2019) |
| Schisandra chinensis Polysaccharide | Schisandra chinensis | Mice | 200, 400, 800 mg/kg | Regulation of Nrf2/antioxidant response element and TLR4/NF-κB signaling pathways | Shan et al. (2019) |
| Schisandra chinensis acidic polysaccharide | Schisandra chinensis | Male ICR mice | 5, 10, 20 mg/kg | By inhibiting the expression of CYP2E1 protein and then alleviating oxidative stress injury | Yuan et al. (2018) |
| HepG2 cells | 3.12, 6.25, 12.5 μg/ml | ||||
| GBLP | Ginkgo biloba | Male Wistar rats | 100, 200, 400 mg/kg | By attenuating IR, preserving liver function, enhancing antioxidant defense system, and reducing lipid peroxidation | Yan et al. (2015) |
| Paeoniae radix alba polysaccharides | Paeoniae radix alba | Male Kunming mice | 0.2, 0.4, 0.8 g/kg | Inhibition of the NF-κB signaling pathway | Wang et al. (2020b) |
| Glycosides | |||||
| Chrysophanol 8-O-glucoside | Rheum palmatum | LX-2 cells | 1, 5, 20 μg/ml | Regulation of the STAT3 signaling pathway | Park et al. (2020) |
| Sennoside A | Rheum officinale Baill | HepG2 cells | 25, 50, 100 μM | Down-regulation of KRT7 and KRT81, and inhibition of the AKT and ERK pathways | Le et al. (2020); Zhu et al. (2020) |
| SMMC-7721 cells | 25, 50, 100 μM | ||||
| Male C57BL/6J mice | 15, 30, 60 mg/kg | ||||
| HSC-T6 cells | 10 μM | ||||
| Astragaloside IV | Astragalus membranaceus | SMMC-7721 cells | 80 μg/ml | Inhibition of lncRNA-ATB, MRP2, PTP1B and anti-apoptotic signaling, and improvement insulin resistance | Li et al. (2018g); Qu et al. (2020); Su et al. (2020); Zhou et al. (2021) |
| Huh-7 cells | 80 μg/ml | ||||
| HepG2 cells | 0.4, 4, 40 μM | ||||
| H22 cells | 0.4, 4, 40 μM | ||||
| Male BALB/c mice | 50 mg/kg | ||||
| HepG2 cells | 6.4, 12.8, 25.6, 51.2, 102.4 μM | ||||
| SK-Hep1 cells | 200, 400 μM | ||||
| Hep3B cells | 200, 400 μM | ||||
| Amarogentin | Swertia and Gentiana roots | HSCs | 0.01, 0.1, 1 mg/ml | By anti-oxidative properties and suppressing the mitogen-activated protein kinase signaling pathway | Zhang et al. (2017) |
| Male C57BL/6 mice | 25, 50, 100 mg/kg | ||||
| Amygdalin | Armeniaca semen | Female BALB/c mice | 4, 8 mg/kg | regulation of the NLRP3, NF-κB, Nrf2/NQO1, PI3K/AKT and JAK2/STAT3 signaling pathways | Tang et al. (2019); Wang et al. (2021a); Yang et al. (2019a) |
| HepG2 cells | 80 μM | ||||
| Male Sprague–Dawley rats | 0.5, 1, 1.5, 3 mg/kg | ||||
| LX-2 cells | 1.25, 2.5, 5 mg/ml | ||||
| Forsythiaside A | Forsythia suspensa | Male BALB/c mice | 15, 30, 60 mg/kg | Through modulating the remolding of extracellular matrix, PI3K/AKT and Nrf2 signaling pathway, and inhibition of NF-κB activation | Gong et al. (2021); Pan, et al. (2015a) |
| Transgenic zebrafish | 25, 50, 100 μM | ||||
| Gentiopicroside | Gentiana manshurica Kitagawa | Male Sprague–Dawley rats | 20 mg/kg | Improvement of mitochondrial dysfunction and activation of LKB1/AMPK signaling | Li, et al. (2018e); Yang et al. (2020a); Zhang et al. (2021) |
| Male C57BL/6 mice | 40, 80 mg/kg | ||||
| HepG2 cells | 100 μM | ||||
| RAW 264.7 macrophages | 25, 50, 100 μM | ||||
| Paeoniflorin | Paeonia lactiflora | Male Sprague-Dawley rats | 10, 20, 40, 80, 200 mg/kg | By activating LKB1/AMPK and AKT pathways, and inhibiting HMGB1-TLR4 signaling pathway and HIF-1α expression | Li, et al. (2018d); Xie et al. (2018); Zhao et al. (2014) |
| Male C57BL/6 mice | 100 mg/kg | ||||
| Swertiamarin | Gentiana manshurica Kitag | HSCs cells | 2.4, 6, 15 μM | By suppressing angiotensin II–angiotensin type 1 receptor–extracellular signal-regulated kinase signaling | Li et al., (2016) |
| MaleWistar rats | 15, 20 mg/kg | ||||
| Nodakenin | Angelica biserrata | Male ICR mice | 10, 30 mg/kg | By regulating apoptosis-related mitochondrial proteins | Lim et al. (2021) |
| Geniposide | Gardenia jasminoides frui | HepG2 cells | 65, 130, 260 μmol/L | Regulation of Nrf2/AMPK/mTOR signaling pathways | Shen et al. (2020) |
| Male wild-type mice | 50, 75, 100 mg/kg |
FIGURE 5.
The chemical structures of glycosides showing anti-hepatopathy activity.
Phenols and Flavonoids
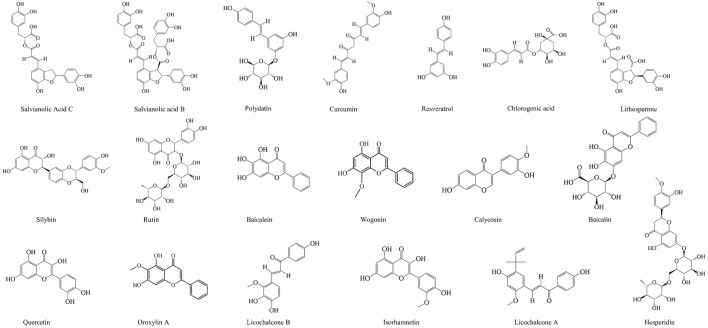
Phenolic compounds are composed of the aromatic rings with one or more hydroxyl groups. They play an important role on oxidative stress in the human by maintaining the balance between oxidants and antioxidants, which are divided into phenolic acids, flavonoids, coumarins, and tannins (Van Hung, 2016). A large number of phenolic compounds in CM have obvious antioxidant capacity, which can reduce the oxidative damage of the liver, such as Lithospermic acid, Chlorogenic acid, Curcumin, Polydatin, and Salvianolic acid C (Chan and Ho, 2015; Koneru et al., 2017; Shi et al., 2016; Wu, C.-T. et al., 2019; Zhong et al., 2016). Yang et al. further found that Chlorogenic acid could reduce the expression of α-SMA, collagen I in the liver tissue and serum TGF-β1 by increasing the mRNA and protein expression of Smad7 and MMP-9, thus alleviating liver fibrosis (Wu, C. et al., 2019). The studies have shown that Curcumin and Polydatin can inhibit lipid accumulation by regulating endoplasmic reticulum stress and the Keap1/Nrf2 pathway (Lee, H.-Y. et al., 2017; Zhao, X.-J. et al., 2018). In addition, Yan et al. demonstrated that Chlorogenic acid could improve liver injury and insulin resistance by inactivating the JNK pathway and inhibiting the autophagy in MAFLD rats (Yan, Hua et al., 2018).
Flavonoids, a part of phenolic compounds, also have significant hepatoprotective effects. For example, Isorhamnetin suppresses the TGF-β/Smad pathway and reduces oxidative stress to alleviate hepatic fibrosis (Yang, J.H. et al., 2016), and Wogonin reduces hepatic fibrosis by regulating the activation and apoptosis of HSCs (Du et al., 2019). Quercetin can effectively alleviate MAFLD, which depends on its regulation of intestinal microbiota imbalance and related gut-liver axis activation (Porras et al., 2017). Hesperidin and Oxylin A have significant anti-hepatoma activity (Mo'men et al., 2019; Wei et al., 2017). In addition, Licochalcone A can increase the expression of antioxidant enzymes by reducing the apoptosis, mitochondrial dysfunction, and reactive oxygen production stimulated by tert butyl peroxide and Acetaminophen, thus protecting APAP-induced hepatotoxicity, which is largely dependent on the antioxidant Nrf2 pathway (Lv et al., 2018). What’s more, rutin has a good protective effect on various acute liver injury induced by carbon tetrachloride, lipopolysaccharide, and mercury chloride (Caglayan et al., 2019; Elsawy et al., 2019; Rakshit et al., 2021).
Bacalin, a kind of flavonoid extracted from Scutellaria baicalensis, has significant biological activity, which is widely used in the treatment of liver diseases. The study has shown that bacalin suppresses the production of IL-1β, IL-6, and TNF-α, as well as regulates the TLR4 expression and inhibits the NF-κB activation, protecting the inflammation of chicken’s liver induced by LPS through the negative regulation of inflammatory medium (Cheng et al., 2017). Another study showed that the inhibition of the proliferation, apoptosis, invasion, migration, and activation of HSCs induced by platelet derived growth factor-BB through mir-3595/acsl4 axis is one of the mechanisms of bacalin in anti-hepatic fibrosis (Wu, X. et al., 2018).
The specific information of the phenols and flavonoids is shown in Table 3, and the chemical structures of the phenols and flavonoids are shown in Figure 6.
TABLE 3.
Summary of phenols and flavonoids with significant anti-liver disease activity.
| Compounds | Source | The species investigated | Dose | Mechanisms | References |
|---|---|---|---|---|---|
| Phenols | |||||
| Resveratrol | Polygonum cuspidatum | Male C57BL/6J mice | 60 mg/kg | Through improving insulin sensitivity and glucose levels | Hajighasem et al. (2018); Zhao et al. (2019a) |
| HepG2 cells | 20, 50, 100 μM | ||||
| Male Wistar rats | 25 mg/kg | ||||
| Salvianolic acid B | Salvia miltiorrhiza | Male Kunming mice | 15, 30 mg/kg | Inhibition of MAPK-mediated P-Smad2/3L signaling | Wu et al. (2019b) |
| HSC-T6 cells | 25, 50, 100 μM | ||||
| LX-2 cells | 25, 50, 100 μM | ||||
| Salvianolic Acid C | Salvia miltiorrhiza | Male ICR mice | 5, 10, 20 mg/kg | By attenuating inflammation, oxidative stress, and apoptosis through inhibition of the Keap1/Nrf2/HO-1 signaling | Wu, et al. (2019c) |
| Polydatin | Polygonum cuspidatum | Male Sprague-Dawley rats | 7.5, 15, 30 mg/kg | Through increasing miR-200a to regulate Keap1/Nrf2 pathway, and restoring the antioxidant balance as well as the MMP/TIMP balance | Koneru et al. (2017); Zhao, et al. (2018a) |
| BRL-3A cells | 10, 20, 40 μM | ||||
| HepG2 cells | 10, 20, 40 μM | ||||
| Male C57BL/6 mice | 50, 100 mg/kg | ||||
| Curcumin | Curcumin longa | Pregnant NMRI mice | 10 mg/kg | By suppression of oxidative stress-related inflammation via PI3K/AKT and NF-kB related signaling | Barandeh et al. (2019); Lee et al. (2017b); Zhong et al. (2016) |
| Male Sprague-Dawley rats | 200 mg/kg | ||||
| Male C57BL/6 mice | 20, 40, 80 mg/kg | ||||
| HSCs | 0.5, 1, 2 μM | ||||
| Chlorogenic acid | Oriental Wormwood | Female Sprague-Dawley rats | 50 mg/kg | Inhibition of oxidative stress, JNK pathway and miR-21-Regulated TGF-β1/Smad7 signaling pathway | Shi et al. (2016); Yang et al. (2017) |
| Male Sprague-Dawley rats | 15, 30, 60 mg/kg | ||||
| HSCs | 12.5, 25, 50 mg/ml | ||||
| LX2 cells | 20, 40, 80 μg/ml | ||||
| Lithospermic acid | Salvia miltiorrhiza | Huh-7 cells | 5, 10, 20, 40 μg/ml | Reduction of free radicals, restoration of liver functions and inhibition of caspase activity associated with apoptosis | Chan and Ho (2015) |
| Male BALB/c mice | 50, 100 mg/kg | ||||
| Flavonoids | |||||
| Hesperidin | Citrus | Male Wistar rats | 200 mg/kg | Inhibition of free radicals, NF-κB activation and PI3K/Akt pathway, and activation of the Akt pathway | Li et al. (2020b); Mo'men et al. (2019); Pérez-Vargas et al. (2014) |
| Male C57BL/6J mice | 100, 200, 400 mg/kg | ||||
| Hepatocytes | 10, 20 ng/ml | ||||
| Licochalcone A | Licorice Glycyrrhiza | Nrf2−/− C57BL/6 mice | 50, 100 mg/kg | Up-regulation of the Nrf2 antioxidant and sirt-1/AMPK pathway | Liou et al. (2019); Lv et al. (2018) |
| HepG2 cells | 1.5, 3, 3.7, 6, 12 μM | ||||
| Male C57BL/6 mice | 5, 10 mg/kg | ||||
| Licochalcone B | Licorice Glycyrrhiza | HepG2 cells | 40, 80, 120 μM | Inhibition of Caspase 8 and Caspase 9 proteins | Wang et al. (2019b) |
| Wogonin | Scutellaria radix | Male C57BL/6 mice | 10, 20, 40 mg/kg | Regulation of hepatic stellate cell activation and apoptosis | Du et al. (2019) |
| HSC-T6 cells | 1.25 μg/ml | ||||
| LX-2 cells | 20 μg/ml | ||||
| Quercetin | Radix Bupleuri | Male C57BL/6J mice | 0.05% (wt/wt) | By ameliorating inflammation, oxidative stress, and lipid metabolism, and modulating intestinal microbiota imbalance and related gut-liver axis activation | Li et al. (2018b); Porras et al. (2017); Yang et al. (2019a); Zhu et al. (2018a) |
| Male BALB/c mice | 50 mg/kg | ||||
| Raw 264.7 cells | 50 μM | ||||
| Male db/db mice | 100 mg/kg | ||||
| Male Sprague-Dawley rats | 100 mg/kg | ||||
| HepG2 cells | 100 μM | ||||
| Baicalin | Scutellariae radix | Male C57BL/6 mice | 15, 30, 60 mg/kg | By regulating the ERK signaling pathway, TLR4-Mediated NF-κB pathway and miR-3595/ACSL4 axis | Cheng et al. (2017); Liao et al. (2017); Wu et al. (2018a) |
| HSC-T6 cells | 50, 100, 150 μM | ||||
| Young chicken | 50, 100, 200 mg/kg | ||||
| Baicalein | Scutellariae radix | BEL-7402 cells | 5, 10 μg/ml | By activating apoptosis and ameliorating P-glycoprotein activity | Li. et al. (2018a) |
| BEL-7402/5-FU cells | 5, 10 μg/ml | ||||
| Rutin | Forsythia suspensa | Male db/db mice | 60, 120 mg/kg | By interfering with oxidative stress, inflammation and apoptosis, and facilitating signal transduction and activated state of insulin IRS-2/PI3K/Akt/GSK-3β signal pathway | D'Atanasio et al. (2018); Elsawy et al. (2019); Liang et al. (2018); Liu et al. (2017) |
| HepG2 cells | 8, 16, 32, 64 μg/ml | ||||
| Male albino rats | 70 mg/kg | ||||
| Male Sprague Dawley rats | 50, 100 mg/kg | ||||
| Male C57BL/6 mice | 200 mg/kg | ||||
| Calycosin | Radix astragali | Male C57BL/6 mice | 12.5, 25, 50 mg/kg | By activating farnesoid X receptor | Duan et al. (2017) |
| Silybin | Silybum marianum | Male C57BL/6 mice | 105 mg/kg | By reducing oxidative damage to mitochondria, proteins, lipids, and involvement with the NF-κB pathway | Goh et al. (2020); Ou et al. (2018) |
| LO2 cells | 25, 50 μM | ||||
| Isorhamnetin | / | Male C57BL/6J mice | 50 mg/kg | By inhibiting de novo lipogenic pathway, by inhibiting TGF-β/Smad signaling and relieving oxidative stress, inhibiting Extracellular Matrix Formation via the TGF-β1/Smad3 and TGF-β1/p38 MAPK Pathways (via inhibition of TGF-β1-mediated Smad3 and p38 MAPK signaling pathways.) | Ganbold et al. (2019); Liu et al. (2019a); Yang et al. (2016b) |
| LX-2 cells | 25, 50, 100 μM | ||||
| HepG2 cells | 25, 50, 100 μM | ||||
| Male ICR mice | 10, 30 mg/kg | ||||
| Oroxylin A | Scutellaria baicalensis | Male ICR mice | 30 mg/kg | Inhibition of hypoxia inducible factor 1alpha, and activation PKM1/HNF4 alpha | Jin et al. (2018); Wei et al. (2017) |
| LO2 cells | 10, 20, 40 μM | ||||
| HepG2 cells | 6, 8, 10 μM | ||||
| SMMC-7721 cells | 15, 20, 25 μM | ||||
| C57BL/6J mice | 75 mg/kg |
FIGURE 6.
The chemical structures of phenols and flavonoids showing anti-hepatopathy activity.
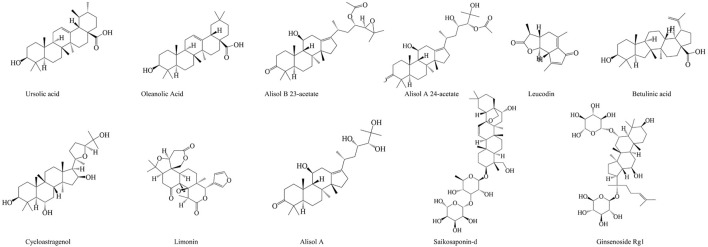
Terpenoids
Terpenoids (isoprenoids) are the most abundant chemical compounds in plants (Tholl, 2015), which has a wide range of biological activities, such as anti-inflammation (Kim, T. et al., 2020), anti-depressant (Agatonovic-Kustrin et al., 2020), anti-cancer (Ateba et al., 2018), and so on. Many studies have shown that terpenoids are also widely used in the treatment of liver diseases. Leucodin is a sesquiterpene lactone isolated from Artemisia capillaris, which can inhibit the inflammatory response of macrophages, and P2x7R-NLRP3-mediated lipid accumulation in hepatocytes (Shang et al., 2018). Saikosaponin-d is an active component isolated from Radix Bupleuri, which can inhibit the COX2 expression through the p-STAT3/C/EBPβ signaling pathway in HCC (Ren et al., 2019). Oleanolic acid (OA) is a kind of triterpenoid widely existing in fruits, vegetables, and herbs. It is liver-specific and can selectively inhibit adipogenesis (Lin, Y.-N. et al., 2018). In addition, OA can regulate antioxidant status, and induce mitochondria-mediated apoptosis and regulate inflammation, which effectively inhibits 7,12-Dimethylbenz[a]anthracene-induced liver cancer (Hosny et al., 2021).
Rhizoma Alismatis is a kind of common CM, which is often used in clinic for adverse urination, edema, diarrhea, and so on. Modern studies have shown that many compounds extracted from Rhizoma Alismatis have hepatoprotective effects. For example, Alisol A 24-acetate, a natural triterpene extracted from Rhizoma Alismatis, can improve NASH by inhibiting oxidative stress, and stimulating autophagy through the AMPK/mTOR signaling pathway (Wu, C. et al., 2018). Meng et al. found that Alisol A 23-acetate could also improve NASH in the mice, which was achieved by the activation of X-like receptor (Meng et al., 2017). Futhermore, Meng et al. found that Alisol A 23-acetate activated FXR to induced the phosphorylation of STAT3 and the expression of its target genes, Bcl-xl and SOCS3. And it reduced the expression of the liver uptake transporter NTCP, and bile acid synthases CYP7A1 and Cyp8b1, as well as increased the expression of the outflow transporters BSEP and MRP2, reducing the hepatic bile acid deposition, which achieved the protective effect on CCl4-induced hepatotoxicity in the mice (Meng et al., 2015).
The specific information of the terpenoids in the treatment of liver diseases is shown in Table 4, and the chemical structures of the terpenoids with therapeutic effects on liver diseases are shown in Figure 7.
TABLE 4.
Summary of terpenoids with significant anti-liver disease activity.
| Compounds | Source | The species investigated | Dose | Mechanisms | References |
|---|---|---|---|---|---|
| Betulinic acid | Betula pubescens | Male C57BL/6J mice | 15, 30, 60, 150 mg/kg | Through the YY1/FAS, MAPK/ERK and PI3K/AKT/mTOR signaling pathway | Liu et al. (2019b); Liu et al. (2019c); Mu et al. (2020) |
| SMMC-7721 cells | 2.5, 5, 10, 20, 40 μM | ||||
| HepG2 cells | 2.5, 5, 10, 20, 40 μM | ||||
| Saikosaponin-d | Radix Bupleuri | SMMC-7721 cells | 2.5, 5, 10 μg/L | Through SENP5- Dependent Inhibition of Gli1 SUMOylation Under Hypoxia, and p-STAT3/C/EBPβ signaling | Ren et al. (2019); Zhang et al. (2019) |
| HepG2 cells | 2.5, 5, 10 μg/L | ||||
| Cycloastragenol | Astragali Radix | HepG2 cells | 12, 25, 50 μM | By activating farnesoid X receptor signaling | Gu et al. (2017) |
| Female C57BL/6 mice | 100 mg/100 g diet | ||||
| Limonin | Citrus fruit and plants | Male Wistar rats | 100 mg/kg | By activating Nrf2 antioxidative pathway and inhibiting NF-κB inflammatory response and TLR-signaling pathway | Mahmoud et al. (2014); Yang et al. (2020b) |
| L-02 cells | 10, 25, 50 μM | ||||
| Male C57BL/6 mice | 40, 80 mg/kg | ||||
| Oleanolic acid | Forsythia suspensa | Male Swiss albino mice | 75 mg/kg | Through induction of mitochondrial-mediated apoptosis and autophagy, and inhibition of Liver X Receptor Alpha and Pregnane X Receptor | Hosny et al. (2021) |
| EAC cells | 9.32 μM | ||||
| HepG2 cells | 10, 20, 32.58, 27.56 μM | ||||
| SMMC-7721 cells | 10, 30, 60 μmol/L | ||||
| Ginsenoside Rg1 | Panax ginseng | Male C57BL/6 mice | 15, 30, 60 mg/kg | By activating Nrf2 signaling pathway | Ning et al. (2018) |
| Ursolic acid | Forsythia suspensa | C57BL/6 mice | 40 mg/kg | Through RhoA-related signaling pathways, and inhibition of interactive NOX4/ROS, RhoA/R and CASP3 | Gan et al. (2018a); Ma et al. (2021); Wan et al. (2019); Wan et al. (2020) |
| HepG2 cells | 10 μM | ||||
| Male Kunming mice | 20, 40, 80 mg/kg | ||||
| Sprague–Dawley rats | 40 mg/kg | ||||
| Alisol A | Rhizoma Alismatis | C57BL/6 mice | 100 mg/kg | Through the AMPK/ACC/SREBP-1c pathway | Ho et al. (2019) |
| Alisol B 23-acetate | Rhizoma Alismatis | Male C57BL/6 mice | 10, 15, 20, 30, 40, 60 mg/kg | Regulation of the FXR and STAT3 signaling pathway | Meng et al. (2015); Meng et al. (2017) |
| Leucodin | Artemisia capillaris | HepG2 cells | 1, 5 μM | Through the P2x7 receptor pathway | Shang et al. (2018) |
FIGURE 7.
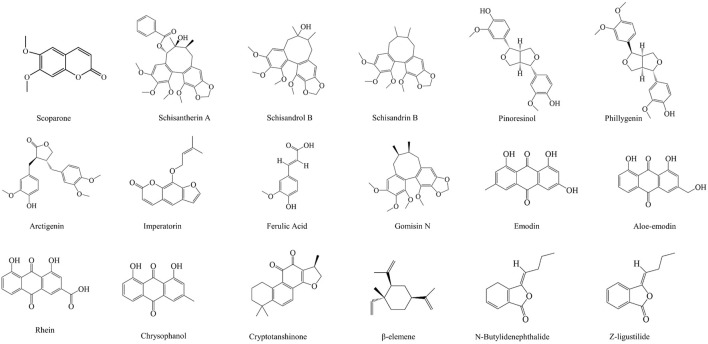
The chemical structures of terpenoids showing anti-hepatopathy activity.
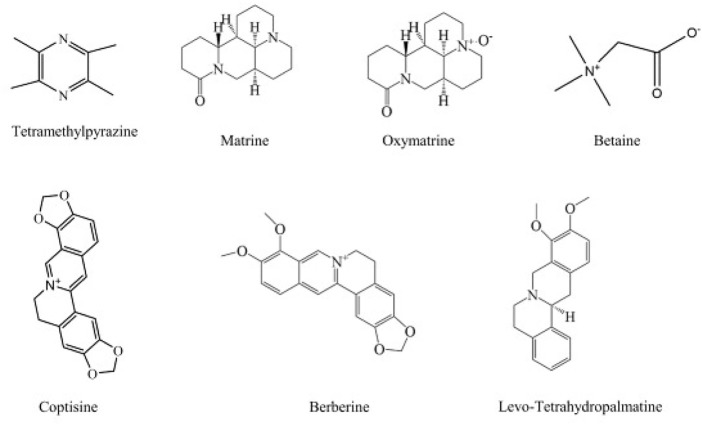
Alkaloids
Alkaloids are an important class of natural products, which have a wide range of biological activities, and have been used in folk medicine for many years (Stöckigt et al., 2011). We are surprised to find that alkaloids play an important role in the treatment of liver diseases. Matrine and Oxymatrine are the main active substances extracted from the roots of Sophora flavescens, and are widely used (Yuan et al., 2010). They have significant biological activities against MAFLD, liver injury, and liver cancer (Gao et al., 2018; Shi et al., 2020; Wei et al., 2018; Xu et al., 2018; Zhang, H. et al., 2020). Ligustrazine is an alkaloid extracted from Ligusticum chuanxiong. It not only activates Nrf2 to inhibit hepatic steatosis, but also induces the apoptosis and autophagy of hepatoma cells to exert an anti-hepatoma effect (Cao et al., 2015; Lu et al., 2017). And coptisine exerts an anti-hepatoma effect by activating the 67 kDa laminin receptor/cGMP signal to induce the apoptosis of human hepatoma cells, and the proliferation and migration of HCC cells (Chai et al., 2018a; Zhou et al., 2018).
The specific information of various alkaloids in the treatment of liver diseases is shown in Table 5. In addition, the chemical structure formulas are shown in Figure 8.
TABLE 5.
Summary of alkaloids with significant anti-liver disease activity.
| Compounds | Source | The species investigated | Dose | Mechanisms | References |
|---|---|---|---|---|---|
| Tetramethylpyrazine | Ligusticum chuanxiong Hort | Male Sprague-Dawley rats | 50, 100, 200 mg/kg | Through PDGF-bR/NLRP3/caspase1 pathway to reduce liver inflammation, and exerts antitumor effects by inducing apoptosis and autophagy in hepatocellular carcinoma, and inhibition of hepatic steatosis by activating the Nrf2 signaling pathway | Cao et al. (2015); Lu et al. (2017); Wu et al. (2015) |
| Human HCC HepG2 cells | 50, 100, 200 μM | ||||
| Male BALB/c nude mice | 50, 100, 150 mg/kg | ||||
| Male ICR mice | 100 mg/kg | ||||
| Human LO2 hepatocytes | 20 μM | ||||
| Coptisine | Rhizoma Coptidis | Kunming mice | 37.5, 150 mg/kg | Through up-regulating expression of miR-122, and activating 67-kDa laminin receptor/cGMP signaling | Chai et al. (2018a); Chai et al. (2018b); Zhou et al. (2018) |
| HepG2 cells | 12.5, 25, 50, 100 μg/ml | ||||
| L02 cells | 25 μg/ml | ||||
| SMMC7721 cells | 12.5, 25, 50, 100 μM | ||||
| Male BALB/c nude mice | 150 mg/kg | ||||
| HepG2 cells | 25 μg/ml | ||||
| Huh7 cells | 25 μg/ml | ||||
| Matrine | Sophora flavescens, Sophora subprostrata | Male C57BL/6J mice | 0.5, 2.5, 10 mg/kg | Regulation of SERCA pathway, and inhibition of mitophagy, PINK1/Parkin pathways and Notch signaling pathway | Gao et al. (2018); Wei et al. (2018) |
| L02 cells | 200, 400, 800 μM | ||||
| HepG2 cells | 1, 5 nM | ||||
| Huh7 cells | 1, 5 nM | ||||
| Betaine | Lycium chinensis | Male Sprague-Dawley rats | 20 g/kg | Regulation of oxidative stress, inflammation, apoptosis, autophagy and Akt/mTOR signaling | Abu Ahmad et al. (2019); Veskovic et al. (2019) |
| Male C57BL/6 mice | 1.5% (w/v) | ||||
| Berberine | Rhizoma Coptidis | Male C57BL/6 mice | 2, 5 mg/kg | Inhibition of oxidative stress, hepatocyte necrosis, inflammatory response, and AKT-mTOR-S6K signaling pathway | Li. et al. (2018a); Zhao et al. (2018b) |
| MIHA cells | 10, 20, 100 μM | ||||
| HepG2 cells | 10, 20 μM | ||||
| Oxymatrine | Sophora alopecuroides | Male Sprague-Dawley rats | 30, 60, 120 mg/kg | Activation of Nrf2/HO-1, regulation of miR-182, and modulation of TLR4-dependent inflammatory and TGF-β1 signaling pathways | Xu et al. (2018); Zhang et al. (2020b); Zhao et al. (2016) |
| BMDMs | 1.0 mg/ml | ||||
| HSC-T6 cells | 250, 500, 1000 μg/ml | ||||
| Male C57BL/6 mice | 120 mg/kg | ||||
| Wistar male rats | 80 mg/kg | ||||
| Levo-tetrahydropalmatine | Corydalis yanhusuo | Male C57 mice | 20, 40 mg/kg | Modulation of PPARγ/NF-κB, TGF-β1/Smad and TRAF6/JNK signaling pathway | Yu et al. (2021); Yu et al. (2018) |
| LX-2 cells | 34.01 μmol/L | ||||
| Male Balb/c mice | 20, 40 mg/kg |
FIGURE 8.
The chemical structures of alkaloids showing anti-hepatopathy activity.
Other Bioactive Ingredients
In addition to the above compounds, many compounds have the activities of anti-liver diseases, including phenylpropanoids (such as simple phenylpropanoids, coumarins, and lignans), anthraquinones, and volatile oils. Some lignans extracted from CM have been proved to have the effects on improving liver diseases. For example, Gomisin N extracted from Schisandra chinensis not only has protective effects on endoplasmic reticulum stress-induced hepatic steatosis, but also alleviates the liver injury caused by ethanol by improving lipid metabolism and oxidative stress (Jang et al., 2016; Nagappan et al., 2018). Futhermore, Arctigenin can inhibit the proliferation of HepG2 cells and block the autophagy cells that lead to the accumulation of sequestosome 1/p62, so as to achieve the therapeutic effects on liver cancer. It will become a new drug for the autophagy research and cancer chemoprevention. It is worth noting that many anthraquinones in Rhubarb have good activities of anti-liver diseases, including chrysophanol, emodin, rhein, and aloe emodin (Bai et al., 2020; Dong et al., 2017; Kuo et al., 2020; Li, Y. et al., 2019). Cryptotanshinone, the main anthraquinone extracted from Salvia miltiorrhiza Bunge, can protect liver by activating the AMPK/SIRT1 and Nrf2, and inhibiting CYP2E1 to inhibit adipogenesis, oxidative stress, and inflammation (Nagappan et al., 2019). Other bioactive components against liver diseases are shown in Table 6. In addition, the related chemical structures are also shown in Figure 9.
TABLE 6.
Summary of other bioactive ingredients with significant anti-liver disease activity.
| Compounds | Source | The species investigated | Dose | Mechanisms | References |
|---|---|---|---|---|---|
| Phenylpropanoids | |||||
| Ferulic Acid | Angelica sinensis | Male Swiss albino mice | 50, 100 mg/kg | Upregulation of Nrf2/HO-1 signaling, and inhibition of TGF-β/smad signaling pathway, and modulation of the gut microbiota composition | Ma et al. (2019); Mahmoud et al. (2020); Mu et al. (2018); Roghani et al. (2020) |
| Male ApoE−/− mice | 30 mg/kg | ||||
| Male Wistar rats | 10, 25, 50 mg/kg | ||||
| LX-2 cells | 5, 15, 30 μM | ||||
| Phillygenin | Forsythia suspensa | LX2 cells | 12.5, 25, 50 μM | Through TLR4/MyD88/NF-κB signaling pathway | Hu et al. (2020b) |
| Arctigenin | Arctium lappa | HepG2 cells | 10 μM | Through autophagy inhibition in hepatocellular carcinoma cells | Okubo et al. (2020) |
| MCF-7 cells | 10 μM | ||||
| Imperatorin | Angelica dahurica | Male C57BL/6 mice | 50, 100 mg/kg | By stimulating the SIRT1-FXR pathway | Gao et al. (2020) |
| Hepatocytes | 5, 10 μM | ||||
| Pinoresinol | Forsythiae Fructus | Male ICR mice | 25, 50, 100, 200 mg/kg | Through inhibition of NF-κB and AP-1 | Kim et al. (2010) |
| Schisandrol B | Schisandra sphenanthera | Male C57BL/6 mice | 12.5, 50, 200 mg/kg | Inhibition of CYP-mediated bioactivation and regulation of liver regeneration | Jiang et al. (2015) |
| Schisantherin A | Schisandra sphenanthera | Male C57BL/6 mice | 25, 50, 100, 200, 400, 800 mg/kg | Inhibition of mitogen-activated protein kinase pathway | Zheng et al. (2017) |
| Schisandrin B | Schisandra sphenanthera | Male Wistar rats | 25, 50 mg/kg | Regulation of Nrf2-ARE and TGF-β/smad signaling pathways | Chen et al. (2017) |
| HSC-T6 cells | 5, 10, 30 μM | ||||
| Gomisin N | Schisandra sphenanthera | Male C57BL/6N mice | 5, 20 mg/kg | Through ameliorating lipid metabolism, oxidative Stress and ER stress | Nagappan et al. (2018); Yun et al. (2017) |
| HepG2 cells | 10, 50, 100 μM | ||||
| C57BL/6 mice | 1, 30 mg/kg | ||||
| Scoparone | Artemisia capillaris | AML12 cells | 200 mM | By regulating the ROS/P38/Nrf2 axis, PI3K/AKT/mTOR pathway, and TLR4/NF-κB signaling pathway | Liu et al. (2020b) |
| RAW264.7 cells | 25, 50, 100, 200 mM | ||||
| Male C57BL/6 J mice | 20, 40, 80 mg/kg | ||||
| Anthraquinones | |||||
| Chrysophanol | Rheum palmatum | HSC-T6 cells | 30 mM | By regulating endoplasmic reticulum stress and ferroptosis | Kuo et al. (2020) |
| Emodin | Rheum palmatum | Male BALB/c nude mice | 15, 25, 30, 50, 60 mg/kg | By regulating VEGFR2, miR-34a, AMPK with Hippo/Yap signalling pathway, MAPK, PI3K/AKT signaling pathways, and inhibiting the TLR4 signaling pathway and epithelial-mesenchymal transition and transforming growth factor-β1 | Bai et al. (2020); Ding et al. (2018a); Lee et al. (2020); Lin et al. (2016); Liu et al. (2018) |
| HepG2 cells | 3, 10, 30, 100 μM | ||||
| SK-Hep-1 cells | 30 μM | ||||
| Male C57B/6 mice | 10, 30 mg/kg | ||||
| Male Sprague-Dawley rats | 10, 20, 40 mg/kg | ||||
| RAW264.7 cells | 15, 30, 60 μg/ml | ||||
| Male Balb/c mice | 20, 40, 80 mg/kg | ||||
| SMMC-7721 cells | 25, 50, 100 μM | ||||
| Rhein | Polygonum multiflorum | Male Sprague-Dawley rats | 10, 30, 1000 mg/kg | Through regulating the Fas death pathway and the mitochondrial pathway, and promoting bile acid transport and reduce bile acid accumulation | Li et al. (2019c); Xian et al. (2020) |
| L02 cells | 25, 50, 100 μM | ||||
| Aloe-emodin | Rheum palmatum | HepG2 cells | 1, 15, 30 μM | Regulation of the Fas death pathway and the mitochondrial pathway, and inhibition of multidrug resistance protein 2 | Dong et al. (2017); Liu et al. (2020b) |
| Male and female Kunming mouse | 0.8, 1.6 g/kg | ||||
| HL-7702 cells | 5, 10, 20, 40 μM | ||||
| Cryptotanshinone | Salvia miltiorrhiza | Male C57BL/6 mice | 20, 40 mg/kg | Inhibition of MAPKs phosphorylation regulated by TAK1, and activation of AMPK/SIRT1 and Nrf2 signaling pathways | Jin et al. (2014); Nagappan et al. (2019) |
| HepG2 cells | 2.5, 5 μM | ||||
| AML-12 cells | 2.5, 5 μM | ||||
| Volatile oil | |||||
| Z-ligustilide and n-Butylidenephthalide | Angelica tenuissima | MaleC57BL/6mice | 10, 50 mg/kg | Inhibition of fatty acid uptake and esterification | Lee et al. (2019) |
| HepG2 cells | 10, 50, 100 μg/ml | ||||
| Butylidenephthalide | Angelica sinensis | HSC-T6 cells | 15, 25, 35 μg/ml | Reduction of EMT, decreasing inflammatory reaction, and liver cell proliferation | Chuang et al. (2016) |
| Male Wistar rats | 15, 80 mg/kg | ||||
| Ligustilide | Angelica sinensis | Male Sprague-Dawley | 10, 20, 40 mg/kg | Promotion of phosphorylation of Nrf2 and AMPKa1 | Guo et al. (2021) |
FIGURE 9.
The related chemical structures of other anti-hepatopathy bioactive components.
Toxicity
After the above discussion, it is not difficult to find the important position of CM in the treatment of liver diseases. As we all know, CM is a relatively safe class of drugs, but we can’t ignore its toxic and side effects on the liver when we use CM to treat liver diseases. The studies have found that some CM show certain hepatotoxicity. For example, Rhubarb extract had a certain protective effect on the rats with chronic renal failure, but the incidence of mild hepatotoxicity was also observed in normal rats (Wang et al., 2009). Ginkgo biloba extract induced DNA damage by inhibiting the topoisomerase II activity in human hepatocytes (Zhang et al., 2015). Interestingly, the hepatotoxicity of some CM comes from their own hydrolysates. For example, after the intragastric administration of Sophora flavescens extract to the rats, kurarinone glucosides was hydrolyzed into Kurarinone in liver cells, which eventually led to the lipid accumulation and liver injury through a series of actions (Jiang, P. et al., 2017). In addition, the use of herbal products is also a crucial cause of acute liver injury. It has been reported that a 68-year-old woman suffered from acute liver injury caused by aloe, after stopping taking aloe, her liver functions returned to normal levels (Parlati et al., 2017). It is worth noting that the first case of autoimmune hepatitis caused by turmeric supplements has been reported (Lukefahr et al., 2018).
The dosage of CM is often closely related to hepatotoxicity. In order to study the hepatotoxicity of Cortex Dictamni, fan et al. used its water extract and alcohol extract to carry out the toxicity experiments in vivo and in vitro. The results showed that high dose of water extract and alcohol extract significantly increased the levels of ALT and AST, absolute and relative liver weight, and the liver-to-brain ratio, and the histological examination of the liver showed the cell enlargement and nuclear contraction. In vitro cell experiment also showed that water extract and alcohol extract reduced the cell viability in a dose-dependent manner (Fan et al., 2018). A single oral dose of 60 g/kg Cortex Dictamni ethanol extract for 24 h resulted in severe hepatocyte necrosis in mice, and the induced liver injury showed a dose and time-dependent manner (Huang et al., 2020). Saikosaponins, a major bioactive component extracted from Radix Bupleuri, enhances the CYP2E1 expression in a dose and time-dependent manner, and induces oxidative stress in vivo and in vitro, leading to liver injury in mice (Li et al., 2017). In another study, the rats were fed with 300, 1250 and 2500 mg kg−1·D−1 Radix Scutellariae Baicalensis ethanol extract for 26 weeks. It was found that the liver tissues of the rats in the high-dose group showed some inflammatory changes mainly characterized by leukocyte infiltration. In addition, there were also some changes in the levels of glucose, electrolyte, and lipid (Yi et al., 2018). It can be seen that the hepatotoxicity of many CM are closely related to the dosage.
In addition, the abuse of CM without the guidance of doctors is also the source of toxic reactions. Because traditional Chinese medical science thinks that “toxicity” refers to the biases of drugs, the toxic components of CM are often the effective components for treating diseases. The key to judging whether CM is toxic or non-toxic is to see whether it is used according to the syndrome. As long as the treatment is besed on the syndrome, toxic drugs are also safe. If the treatment is not for the syndrome, non-toxic drugs may be harmful. It is worth noting that there are also some CM products considered non-toxic or low toxic, which have obvious toxicological effects on different organs in animal and human models (Liu, R. et al., 2020). So it is a great problem to control the toxic and non-toxic boundaries reasonably, and every traditional medical scholar should make efforts to do so.
Clinical Trials
Most drugs for anti-liver diseases used in clinic are CM compounds, and less clinical research and application involve only one CM or one compound. Table 7 shows some CM (excluding CM compounds) used in the clinical treatment of liver diseases. The purpose is to improve the richness of clinical medication, so that more CM with potential and significant therapeutic effects can be noticed.
TABLE 7.
Some Chinese medicine are used to treat liver diseases in clinic.
| CM or its compounds | disease | Subject | Study design | Treatment groups | Length | Clinical outcome | References |
|---|---|---|---|---|---|---|---|
| Turmeric supplementation (Combined use of chicory seeds) | MAFLD | 92 patients (aged 20–60 years) | Double-blind, randomized, controlled clinical trial | Control group: placebo | 12 weeks | Significantly decreased serum alkaline phosphatase and increased serum HDL-C | Ghaffari et al. (2019) |
| Experimental group: turmeric supplementation (3 g/d, TUR); Chicory seed supplementation (9 g/d, CHI); Turmeric and chicory seed supplementation (3 g/d, TUR + CHI) | |||||||
| Curcuminoids supplementation | MAFLD | 55 patients | Double-blind, randomized, placebo-controlled trial | Control group: placebo capsules | 8 weeks | Improved the severity of MAFLD; serum concentrations of TNF-α, MCP-1 and EGF were improved | Saberi-Karimian et al. (2020) |
| Experimental group: 500 mg curcuminoids (plus 5 mg piperine to increase intestinal absorption) | |||||||
| Curcumin (amorphous dispersion formulation) | MAFLD | 80 cases | Randomized double-blind placebo-controlled trial | Control group: placebo | 8 weeks | The liver fat content, biochemical parameters and anthropometry were significantly improved in patients with MAFLD | Rahmani et al. (2016) |
| Experimental group: 500 mg/day equivalent to 70 mg curcumin | |||||||
| Curcumin supplementation | LC | 70 patients (aged 20–70 years) | Randomized, double-blind, placebo-controlled trial | Control group: placebo | 3 months | MELD(i), MELD, MELD-Na and Child-Pugh scores decreased significantly | Nouri-Vaskeh et al. (2020b) |
| Experimental group: 1000 mg/day curcumin | |||||||
| Curcumin | LC | 70 cases (aged 20–70 years) | Randomized double-masked placebo-controlled trial | Control group: placebo | 12 weeks | The total score and most of the CLDQ, physical and mental health scores and most of the SF-36 areas were significantly improved, and the LDSI2.0 domain was significantly decreased | Nouri-Vaskeh et al. (2020a) |
| Experimental group: 1000 mg/day curcumin | |||||||
| Resveratrol | MAFLD | 60 subjects | Double-blind, randomized, placebo-controlled trial | Control group: placebo | 3 months | Significantly reduced aspartate aminotransferase (AST), glucose, LDL-C, total cholesterol; reduced the levels of tumour necrosis factor-alpha, cytokeratin 18 and fifibroblast growth factor 21 | Chen et al. (2015) |
| Experimental group: 150 mg resveratrol twice daily | |||||||
| Portulaca oleracea L. hydroalcohoic extract | MAFLD | 74 patients | Randomized, double-blind clinical trial | Control group: placebo capsules | 12 weeks | The levels of alanine aminotransferase (ALT), aspartate transaminase, γ-glutamyltransferase, fasting blood glucose, insulin resistance, triglyceride and LDL-C were significantly reduced | Darvish Damavandi et al. (2021) |
| Experimental group: 300 mg purslane extract | |||||||
| Portulaca oleracea L. seeds | MAFLD | Sixty eligible individuals (12 men and 48 women) | Randomized controlled clinical trial | Control group: low-calorie diet | 8 weeks | Reduced fasting blood glucose, total cholesterol, and LDL-C | Gheflati et al. (2019) |
| Experimental group: 10 g/day of purslane seeds and low-calorie diet | |||||||
| Hesperidin (Combined use of flaxseed) | MAFLD | One hundred eligible patients | Randomized, controlled, clinical trial | Control group: lifestyle modification program | 12 weeks | The levels of ALT, insulin resistance, insulin sensitivity index, fasting blood glucose and fatty liver index decreased significantly | Yari et al. (2021) |
| Experimental group: lifestyle modification program with 30 g whole flaxseed powder; lifestyle modification program with 1 g hesperidin supplementation; lifestyle modification program with combination of 30 g flaxseed and 1 g hesperidin | |||||||
| Artemisia annua L. Extract | Nonalcoholic liver dysfunction | 79 subjects | Randomized, double-Blind, placebo-controlled | Control group: placebo | 4 weeks | Levels of AST and ALT were significantly reduced, and scores on the multidimensional fatigue scale were reduced, significantly enhancing liver function and health | Han et al. (2020) |
| Experimental group: powdered-water extract of Artemisia annua | |||||||
| Silymarin | NASH | 78 patients | Randomized, double-blind, placebo controlled trial | Control group: placebo | 12 months | After 48 weeks of treatment, the MAFLD activity score (NAS) decreased by at least two points, fibrosis stage improved, baseline changes, serum ALT and AST decreased | Navarro et al. (2019) |
| Experimental group: proprietary standardized silymarin preparation 420 mg or 700 mg | |||||||
| Silymarin | NASH | 99 patients | Randomized, double-blind, placebo-controlled trial | Control group: placebo | 48 weeks | The fibrosis was reduced and the ratio of AST to platelet index was also significantly decreased | Wah Kheong et al. (2017) |
| Experimental group: Silymarin (three times daily) |
Single extract or chemical component of CM showed good activity of anti-liver diseases in clinical research. Artemisia annua L. extract can improve the liver function in the patients with mild to moderate nonalcoholic liver dysfunction, and no obvious adverse reactions were observed in all subjects (Han et al., 2020). Futhermore, Portulaca oleracea extract can improve liver enzyme, blood lipid, and blood glucose in the patients with MAFLD (Darvish Damavandi et al., 2021). It is worth noting that Carcuma longa has a wide range of clinical applications, with a large number of clinical data, suggesting its position in the clinical treatment of liver diseases. To assess the effect of Carcuma longa on MAFLD, 92 MAFLD patients aged 20–60 years were enrolled in a 12-week study. The results showed that Carcuma longa supplement was very useful in controlling MAFLD-related risk factors (Darvish Damavandi et al., 2021). Curcumin, the main active component of Curcuma longa, can increase the serum inflammatory cytokine levels in the patients with MAFLD, which may be partly dependent on the anti-steatosis effect (Saberi-Karimian et al., 2020). In addition, curcumin can improve the quality of life of the patients with liver cirrhosis (Nouri-Vaskeh et al., 2020a). Although the clinical application of Curcuma longa has surpassed other CM against liver diseases, it still fails to solve the problem of its optimal dosage, and the molecular mechanisms on treating liver diseases is unclear. More importantly, in view of the widespread use of Curcuma longa, we need larger, more impartial and high-quality controlled randomized trials to conduct a deeper evaluation.
In the future, more clinical experiments should be studied, which makes more CM into clinical application, and even go to the international stage. There are still many deficiencies in the current clinical research. First, the dosage is single and the sample size is small, which is not good for screening the best treatment dose. Secondly, the existing clinical experiments mainly focus on the study of MAFLD, but there are many kinds of liver diseases. In the future, the research can be expanded to make more patients with liver diseases benefit from CM. Finally, the mechanisms of many CM (especially CM compounds) used in the treatment of liver diseases are not clear. We should further explore the mechanism of action of CM, making its fuzzy mechanism clearer and letting more people accept it.
Conclusion and Perspectives
In conclusion, CM can prevent and treat liver diseases through many ways, including regulating lipid metabolism, anti-liver injury (such as CCl4, H2O2, alcohol, and drug damage), anti-oxidant stress (including reducing ROS, increasing SOD, GSH and CAT content, and regulating Nrf2 and other related pathways), regulating bile acid metabolism (including regulating the excreted and ingested receptors), regulating the immune system, anti-hepatitis virus, and anti-liver cancer. In terms of the current situation, a large number of studies have proved the potential of CM in the treatment of liver diseases. However, the resources of CM are huge, and it is probably known that the effective CM for liver diseases are only one corner of the iceberg. More tasks need the joint efforts of all traditional medicine scholars. In addition, a large part of the current research has not only been focused on the study of efficacy, but also the expression level of genes and proteins. But it is not enough, and more new methods should be explored, such as using multi-group analysis (metabolomics, proteomics), so as to promote the progress of CM in the treatment of liver diseases.
It is worth noting that there is also relevant evidence that the new technology of CM combined with other preparations can greatly enhance the therapeutic effects on liver diseases. For example, due to the characteristics of unstable chemical structure, low bioavailability, easy oxidation, and UV degradation, the toxic effect of curcumin on hepatoma cells is limited. Therefore, Kong et al. used curcumin loaded mesoporous silica nanoparticles, and found that the complex had better antioxidant activity than curcumin alone, as well as significantly enhanced the cytotoxic effect on hepatoma cells (Kong et al., 2019). Another study showed that curcumin liposome had a greater inhibitory effect on the growth and apoptosis of cancer cells (Feng et al., 2017). But these studies are still very few, which should be increased later.
This paper lists and elaborates the active ingredients of some CM against liver diseases, such as polysaccharides, glycosides, phenols, flavonoids, terpenoids, alkaloids, etc. We found the research on the mechanism of action of each ingredient was relatively single, and CM showed the joint action of multi-component and multi-target in the treatment of liver diseases. Therefore, screening more effective components and studying their molecular mechanisms should be greatly strengthened. For example, recent studies have shown that iron is essential for life, but excessive iron may be cytotoxic, which may lead to cell death and some diseases (Bogdan et al., 2016; Nakamura et al., 2019). In addition, in the previous discussion, we also know that the gut microbiota plays an important role in the treatment of liver diseases. Therefore, it is suggested that we can refer to these relevant mechanisms in the future research of CM on treating liver diseases.
CM, including Tibetan medicine, has shown good effects of anti-liver diseases (Li, Qi et al., 2018; Fu et al., 2020), which is indispensable in the treatment of liver diseases. This paper is a comprehensive review of CM and the related compounds, toxicology, and clinical research, which is aimed to provide scientific and effective references for the treatment of liver diseases, and to better use and develop the treasure of CM.
Author Contributions
KF and YL designed this article and established the structure. CW, CM, and HZ assisted in data collection and form establishment. YL helped to revise the manuscript.
Funding
The study was supported by National Natural Science Foundation of China (No: 81891012, 81630101, and U19A2010), Sichuan Science and Technology Program (2021JDRC0041).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
ALD, alcoholic liver disease; AMPK, adenine monophosphate activated protein kinase; ARE, antioxidant responsive element; BAs, bile acids; CM, Chinese medicine; CYP, cytochrome P450; FL, fatty liver; FSE, Forsythiae Fructuse water extract; GSH, glutathione; HBV, hepatitis B virus; HCC, hepatocellular carcinoma; HCV, hepatitis C virus; HSCs, hepatic stellate cells; IL-1β, interleukin-1β; IL-6, interleukin-6; KCs, kupffer cells; LC, liver cirrhosis; LSECs, liver sinusoidal endothelial cells; MAFLD, metabolic associated fatty liver disease; NAFL, non-alcoholic fatty liver; NASH, non-alcoholic steatohepatitis; NF-κB, nuclear factor kappa-B; NK, natural killer; Nrf2, nuclear factor-erythroid 2-related factor 2; ROS, reactive oxygen species; RSM, Radix Salvia Miltiorrhiza; SOD, superoxide dismutase; TC, total cholesterol; TG, triglyceride; TGF-β, transforming growth factor-β; TNF-α, tumor necrosis factor-α.
References
- Abdelfattah-Hassan A., Shalaby S. I., Khater S. I., El-Shetry E. S., Abd El Fadil H., Elsayed S. A. (2019). Panax Ginseng Is superior to Vitamin E as a Hepatoprotector Against Cyclophosphamide-Induced Liver Damage. Complement. Ther. Med. 46, 95–102. 10.1016/j.ctim.2019.08.005 [DOI] [PubMed] [Google Scholar]
- Abu Ahmad N., Raizman M., Weizmann N., Wasek B., Arning E., Bottiglieri T., et al. (2019). Betaine Attenuates Pathology by Stimulating Lipid Oxidation in Liver and Regulating Phospholipid Metabolism in Brain of Methionine-Choline-Deficient Rats. FASEB J. 33 (8), 9334–9349. 10.1096/fj.201802683R [DOI] [PubMed] [Google Scholar]
- Agatonovic-Kustrin S., Kustrin E., Gegechkori V., Morton D. W. (2020). Anxiolytic Terpenoids and Aromatherapy for Anxiety and Depression. Adv. Exp. Med. Biol. 1260, 283–296. 10.1007/978-3-030-42667-5_11 [DOI] [PubMed] [Google Scholar]
- Agrawal A., Bierut L. J. (2012). Identifying Genetic Variation for Alcohol Dependence. Alcohol. Res. 34 (3), 274–281. [PMC free article] [PubMed] [Google Scholar]
- Alaca N., Özbeyli D., Uslu S., Şahin H. H., Yiğittürk G., Kurtel H., et al. (2017). Treatment with Milk Thistle Extract (Silybum marianum), Ursodeoxycholic Acid, or Their Combination Attenuates Cholestatic Liver Injury in Rats: Role of the Hepatic Stem Cells. Turk J. Gastroenterol. 28 (6), 476–484. 10.5152/tjg.2017.16742 [DOI] [PubMed] [Google Scholar]
- Arulselvan P., Fard M. T., Tan W. S., Gothai S., Fakurazi S., Norhaizan M. E., et al. (2016). Role of Antioxidants and Natural Products in Inflammation. Oxid Med. Cel Longev 2016, 5276130. 10.1155/2016/5276130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ateba S. B., Mvondo M. A., Ngeu S. T., Tchoumtchoua J., Awounfack C. F., Njamen D., et al. (2018). Natural Terpenoids Against Female Breast Cancer: A 5-year Recent Research. Curr. Med. Chem. 25 (27), 3162–3213. 10.2174/0929867325666180214110932 [DOI] [PubMed] [Google Scholar]
- Auyeung K. K., Ko J. K. (2009). Coptis Chinensis Inhibits Hepatocellular Carcinoma Cell Growth Through Nonsteroidal Anti-inflammatory Drug-Activated Gene Activation. Int. J. Mol. Med. 24 (4), 571–577. 10.3892/ijmm_00000267 [DOI] [PubMed] [Google Scholar]
- Bai J., Wu J., Tang R., Sun C., Ji J., Yin Z., et al. (2020). Emodin, A Natural Anthraquinone, Suppresses Liver Cancer In Vitro and In Vivo by Regulating VEGFR2 and miR-34a. Invest. New Drugs 38 (2), 229–245. 10.1007/s10637-019-00777-5 [DOI] [PubMed] [Google Scholar]
- Barandeh B., Amini Mahabadi J., Azadbakht M., Gheibi Hayat S. M., Amini A. (2019). The Protective Effects of Curcumin on Cytotoxic and Teratogenic Activity of Retinoic Acid in Mouse Embryonic Liver. J. Cel Biochem 120 (12), 19371–19376. 10.1002/jcb.28934 [DOI] [PubMed] [Google Scholar]
- BasuRay S., Smagris E., Cohen J. C., Hobbs H. H. (2017). The PNPLA3 Variant Associated with Fatty Liver Disease (I148M) Accumulates on Lipid Droplets by Evading Ubiquitylation. Hepatology 66 (4), 1111–1124. 10.1002/hep.29273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bellezza I., Giambanco I., Minelli A., Donato R. (2018). Nrf2-Keap1 Signaling in Oxidative and Reductive Stress. Biochim. Biophys. Acta Mol. Cel Res 1865 (5), 721–733. 10.1016/j.bbamcr.2018.02.010 [DOI] [PubMed] [Google Scholar]
- Bogdan A. R., Miyazawa M., Hashimoto K., Tsuji Y. (2016). Regulators of Iron Homeostasis: New Players in Metabolism, Cell Death, and Disease. Trends Biochem. Sci. 41 (3), 274–286. 10.1016/j.tibs.2015.11.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buendia I., Michalska P., Navarro E., Gameiro I., Egea J., León R. (2016). Nrf2-ARE Pathway: An Emerging Target Against Oxidative Stress and Neuroinflammation in Neurodegenerative Diseases. Pharmacol. Ther. 157, 84–104. 10.1016/j.pharmthera.2015.11.003 [DOI] [PubMed] [Google Scholar]
- Buzzetti E., Pinzani M., Tsochatzis E. A. (2016). The Multiple-Hit Pathogenesis of Non-alcoholic Fatty Liver Disease (NAFLD). Metabolism 65 (8), 1038–1048. 10.1016/j.metabol.2015.12.012 [DOI] [PubMed] [Google Scholar]
- Caglayan C., Kandemir F. M., Darendelioğlu E., Yıldırım S., Kucukler S., Dortbudak M. B. (2019). Rutin Ameliorates Mercuric Chloride-Induced Hepatotoxicity in Rats via Interfering with Oxidative Stress, Inflammation and Apoptosis. J. Trace Elem. Med. Biol. 56, 60–68. 10.1016/j.jtemb.2019.07.011 [DOI] [PubMed] [Google Scholar]
- Cai L., Zou S., Liang D., Luan L. (2018). Structural Characterization, Antioxidant and Hepatoprotective Activities of Polysaccharides from Sophorae Tonkinensis Radix. Carbohydr. Polym. 184, 354–365. 10.1016/j.carbpol.2017.12.083 [DOI] [PubMed] [Google Scholar]
- Campana L., Iredale J. P. (2017). Regression of Liver Fibrosis. Semin. Liver Dis. 37 (1), 1–10. 10.1055/s-0036-1597816 [DOI] [PubMed] [Google Scholar]
- Cao J., Miao Q., Miao S., Bi L., Zhang S., Yang Q., et al. (2015). Tetramethylpyrazine (TMP) Exerts Antitumor Effects by Inducing Apoptosis and Autophagy in Hepatocellular Carcinoma. Int. Immunopharmacol 26 (1), 212–220. 10.1016/j.intimp.2015.03.028 [DOI] [PubMed] [Google Scholar]
- Cao L. J., Hou Z. Y., Li H. D., Zhang B. K., Fang P. F., Xiang D. X., et al. (2017). The Ethanol Extract of Licorice (Glycyrrhiza Uralensis) Protects Against Triptolide-Induced Oxidative Stress Through Activation of Nrf2. Evid. Based Complement. Alternat Med. 2017, 2752389. 10.1155/2017/2752389 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao T., Lu Y., Zhu M., Cheng J., Ye B., Fang N., et al. (2020). Effects of Salvia Miltiorrhiza and Radix Astragali on the TGF-β/Smad/Wnt Pathway and the Pathological Process of Liver Fibrosis in Rats. Cel Mol Biol (Noisy-le-grand) 66 (6), 46–51. 10.14715/cmb/2020.66.6.9 [DOI] [PubMed] [Google Scholar]
- Ceccarini M. R., Vannini S., Cataldi S., Moretti M., Villarini M., Fioretti B., et al. (2016). In Vitro Protective Effects of Lycium Barbarum Berries Cultivated in Umbria (Italy) on Human Hepatocellular Carcinoma Cells. Biomed. Res. Int. 2016, 7529521. 10.1155/2016/7529521 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ceni E., Mello T., Galli A. (2014). Pathogenesis of Alcoholic Liver Disease: Role of Oxidative Metabolism. World J. Gastroenterol. 20 (47), 17756–17772. 10.3748/wjg.v20.i47.17756 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chai F. N., Ma W. Y., Zhang J., Xu H. S., Li Y. F., Zhou Q. D., et al. (2018a). Coptisine from Rhizoma Coptidis Exerts an Anti-cancer Effect on Hepatocellular Carcinoma by Up-Regulating miR-122. Biomed. Pharmacother. 103, 1002–1011. 10.1016/j.biopha.2018.04.052 [DOI] [PubMed] [Google Scholar]
- Chai F. N., Zhang J., Xiang H. M., Xu H. S., Li Y. F., Ma W. Y., et al. (2018b). Protective Effect of Coptisine from Rhizoma Coptidis on LPS/D-GalN-induced Acute Liver Failure in Mice Through Up-Regulating Expression of miR-122. Biomed. Pharmacother. 98, 180–190. 10.1016/j.biopha.2017.11.133 [DOI] [PubMed] [Google Scholar]
- Chan H. H. L., Ng T. (2020). Traditional Chinese Medicine (TCM) and Allergic Diseases. Curr. Allergy Asthma Rep. 20 (11), 67. 10.1007/s11882-020-00959-9 [DOI] [PubMed] [Google Scholar]
- Chan K. W., Ho W. S. (2015). Anti-oxidative and Hepatoprotective Effects of Lithospermic Acid Against Carbon Tetrachloride-Induced Liver Oxidative Damage In Vitro and In Vivo. Oncol. Rep. 34 (2), 673–680. 10.3892/or.2015.4068 [DOI] [PubMed] [Google Scholar]
- Chávez-Talavera O., Tailleux A., Lefebvre P., Staels B. (2017). Bile Acid Control of Metabolism and Inflammation in Obesity, Type 2 Diabetes, Dyslipidemia, and Nonalcoholic Fatty Liver Disease. Gastroenterology 152 (7), 1679–1694.e3. 10.1053/j.gastro.2017.01.055 [DOI] [PubMed] [Google Scholar]
- Chen Q., Zhang H., Cao Y., Li Y., Sun S., Zhang J., et al. (2017). Schisandrin B Attenuates CCl4-Induced Liver Fibrosis in Rats by Regulation of Nrf2-ARE and TGF-β/Smad Signaling Pathways. Drug Des. Devel Ther. 11, 2179–2191. 10.2147/DDDT.S137507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen S., Zhao X., Ran L., Wan J., Wang X., Qin Y., et al. (2015). Resveratrol Improves Insulin Resistance, Glucose and Lipid Metabolism in Patients with Non-alcoholic Fatty Liver Disease: a Randomized Controlled Trial. Dig. Liver Dis. 47 (3), 226–232. 10.1016/j.dld.2014.11.015 [DOI] [PubMed] [Google Scholar]
- Chen Y., Yao F., Ming K., Wang D., Hu Y., Liu J. (2016). Polysaccharides from Traditional Chinese Medicines: Extraction, Purification, Modification, and Biological Activity. Molecules 21 (12), 1705. 10.3390/molecules21121705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Z., Liu F., Zheng N., Guo M., Bao L., Zhan Y., et al. (2019). Wuzhi Capsule (Schisandra Sphenanthera Extract) Attenuates Liver Steatosis and Inflammation During Non-alcoholic Fatty Liver Disease Development. Biomed. Pharmacother. 110, 285–293. 10.1016/j.biopha.2018.11.069 [DOI] [PubMed] [Google Scholar]
- Cheng N., Chen S., Liu X., Zhao H., Cao W. (2019). Impact of SchisandraChinensis Bee Pollen on Nonalcoholic Fatty Liver Disease and Gut Microbiota in HighFat Diet Induced Obese Mice. Nutrients 11 (2), 346. 10.3390/nu11020346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng P., Wang T., Li W., Muhammad I., Wang H., Sun X., et al. (2017). Baicalin Alleviates Lipopolysaccharide-Induced Liver Inflammation in Chicken by Suppressing TLR4-Mediated NF-Κb Pathway. Front. Pharmacol. 8, 547. 10.3389/fphar.2017.00547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chopyk D. M., Grakoui A. (2020). Contribution of the Intestinal Microbiome and Gut Barrier to Hepatic Disorders. Gastroenterology 159 (3), 849–863. 10.1053/j.gastro.2020.04.077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chuang H. M., Su H. L., Li C., Lin S. Z., Yen S. Y., Huang M. H., et al. (2016). The Role of Butylidenephthalide in Targeting the Microenvironment Which Contributes to Liver Fibrosis Amelioration. Front. Pharmacol. 7, 112. 10.3389/fphar.2016.00112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Commission, C.P. (2015). Pharmacopoeia of the People’s republic of China. Beijing: China medical science and technology press. [Google Scholar]
- Crocenzi F. A., Zucchetti A. E., Boaglio A. C., Barosso I. R., Sanchez Pozzi E. J., Mottino A. D., et al. (2012). Localization Status of Hepatocellular Transporters in Cholestasis. Front. Biosci. (Landmark Ed. 17, 1201–1218. 10.2741/3981 [DOI] [PubMed] [Google Scholar]
- Cui Y., Jiang L., Shao Y., Mei L., Tao Y. (2019). Anti-alcohol Liver Disease Effect of Gentianae Macrophyllae Extract Through MAPK/JNK/p38 Pathway. J. Pharm. Pharmacol. 71 (2), 240–250. 10.1111/jphp.13027 [DOI] [PubMed] [Google Scholar]
- D'Atanasio E., Trombetta B., Bonito M., Finocchio A., Di Vito G., Seghizzi M., et al. (2018). The Peopling of the Last Green Sahara Revealed by High-Coverage Resequencing of Trans-saharan Patrilineages. Genome Biol. 19 (1), 20. 10.1186/s13059-018-1393-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Darvish Damavandi R., Shidfar F., Najafi M., Janani L., Masoodi M., Akbari-Fakhrabadi M., et al. (2021). Effect of Portulaca Oleracea (Purslane) Extract on Liver Enzymes, Lipid Profile, and Glycemic Status in Nonalcoholic Fatty Liver Disease: A Randomized, Double-Blind Clinical Trial. Phytother Res. 35 (6), 3145–3156. 10.1002/ptr.6972 [DOI] [PubMed] [Google Scholar]
- Dewidar B., Meyer C., Dooley S., Meindl-Beinker A. N. (2019). TGF-β in Hepatic Stellate Cell Activation and Liver Fibrogenesis-Updated 2019. Cells 8 (11), 1419. 10.3390/cells8111419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding C. H., Zhu H. (2020). Isatidis Folium Alleviates Acetaminophen-Induced Liver Injury in Mice by Enhancing the Endogenous Antioxidant System. Environ. Toxicol. 35 (11), 1251–1259. 10.1002/tox.22990 [DOI] [PubMed] [Google Scholar]
- Ding H. R., Wang J. L., Ren H. Z., Shi X. L. (2018a). Lipometabolism and Glycometabolism in Liver Diseases. Biomed. Res. Int. 2018, 1287127. 10.1155/2018/1287127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding Y., Liu P., Chen Z. L., Zhang S. J., Wang Y. Q., Cai X., et al. (2018b). Emodin Attenuates Lipopolysaccharide-Induced Acute Liver Injury via Inhibiting the TLR4 Signaling Pathway In Vitro and In Vivo. Front. Pharmacol. 9, 962. 10.3389/fphar.2018.00962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dodge N. C., Jacobson J. L., Jacobson S. W. (2014). Protective Effects of the Alcohol Dehydrogenase-Adh1b*3 Allele on Attention and Behavior Problems in Adolescents Exposed to Alcohol During Pregnancy. Neurotoxicol Teratol 41, 43–50. 10.1016/j.ntt.2013.11.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong Q., Chu F., Wu C., Huo Q., Gan H., Li X., et al. (2016). Scutellaria Baicalensis Georgi Extract Protects Against Alcohol Induced Acute Liver Injury in Mice and Affects the Mechanism of ER Stress. Mol. Med. Rep. 13 (4), 3052–3062. 10.3892/mmr.2016.4941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong X., Fu J., Yin X., Yang C., Ni J. (2017). Aloe-emodin Induces Apoptosis in Human Liver HL-7702 Cells Through Fas Death Pathway and the Mitochondrial Pathway by Generating Reactive Oxygen Species. Phytother Res. 31 (6), 927–936. 10.1002/ptr.5820 [DOI] [PubMed] [Google Scholar]
- Du X. S., Li H. D., Yang X. J., Li J. J., Xu J. J., Chen Y., et al. (2019). Wogonin Attenuates Liver Fibrosis via Regulating Hepatic Stellate Cell Activation and Apoptosis. Int. Immunopharmacol 75, 105671. 10.1016/j.intimp.2019.05.056 [DOI] [PubMed] [Google Scholar]
- Duan X., Meng Q., Wang C., Liu Z., Liu Q., Sun H., et al. (2017). Calycosin Attenuates Triglyceride Accumulation and Hepatic Fibrosis in Murine Model of Non-alcoholic Steatohepatitis via Activating Farnesoid X Receptor. Phytomedicine 25, 83–92. 10.1016/j.phymed.2016.12.006 [DOI] [PubMed] [Google Scholar]
- Egresi A., Süle K., Szentmihályi K., Blázovics A., Fehér E., Hagymási K., et al. (2020). Impact of Milk Thistle (Silybum marianum) on the Mycotoxin Caused Redox-Homeostasis Imbalance of Ducks Liver. Toxicon 187, 181–187. 10.1016/j.toxicon.2020.09.002 [DOI] [PubMed] [Google Scholar]
- El-Saied M. A., Sobeh M., Abdo W., Badr O. M., Youssif L. T., Elsayed I. H., et al. (2018). Rheum Palmatum Root Extract Inhibits Hepatocellular Carcinoma in Rats Treated with Diethylnitrosamine. J. Pharm. Pharmacol. 70 (6), 821–829. 10.1111/jphp.12899 [DOI] [PubMed] [Google Scholar]
- El-Serag H. B. (2012). Epidemiology of Viral Hepatitis and Hepatocellular Carcinoma. Gastroenterology 142 (6), 1264–e1. 10.1053/j.gastro.2011.12.061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elsawy H., Badr G. M., Sedky A., Abdallah B. M., Alzahrani A. M., Abdel-Moneim A. M. (2019). Rutin Ameliorates Carbon Tetrachloride (CCl4)-Induced Hepatorenal Toxicity and Hypogonadism in Male Rats. PeerJ 7, e7011. 10.7717/peerj.7011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan Q., Zhao B., Wang C., Zhang J., Wu J., Wang T., et al. (2018). Subchronic Toxicity Studies of Cortex Dictamni Extracts in Mice and its Potential Hepatotoxicity Mechanisms In Vitro. Molecules 23 (10), 2486. 10.3390/molecules23102486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fanning G. C., Zoulim F., Hou J., Bertoletti A. (2019). Therapeutic Strategies for Hepatitis B Virus Infection: Towards a Cure. Nat. Rev. Drug Discov. 18 (11), 827–844. 10.1038/s41573-019-0037-0 [DOI] [PubMed] [Google Scholar]
- Feng R., Chen J. H., Liu C. H., Xia F. B., Xiao Z., Zhang X., et al. (2019). A Combination of Pueraria Lobata and Silybum marianum Protects Against Alcoholic Liver Disease in Mice. Phytomedicine 58, 152824. 10.1016/j.phymed.2019.152824 [DOI] [PubMed] [Google Scholar]
- Feng T., Wei Y., Lee R. J., Zhao L. (2017). Liposomal Curcumin and its Application in Cancer. Int. J. Nanomedicine 12, 6027–6044. 10.2147/IJN.S132434 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forner A., Llovet J. M., Bruix J. (2012). Hepatocellular Carcinoma. Lancet 379 (9822), 1245–1255. 10.1016/S0140-6736(11)61347-0 [DOI] [PubMed] [Google Scholar]
- Forner A., Reig M., Bruix J. (2018). Hepatocellular Carcinoma. Lancet 391 (10127), 1301–1314. 10.1016/S0140-6736(18)30010-2 [DOI] [PubMed] [Google Scholar]
- Friedman S. L., Neuschwander-Tetri B. A., Rinella M., Sanyal A. J. (2018). Mechanisms of NAFLD Development and Therapeutic Strategies. Nat. Med. 24 (7), 908–922. 10.1038/s41591-018-0104-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedman S. L. (1993). Seminars in Medicine of the Beth Israel Hospital, Boston. The Cellular Basis of Hepatic Fibrosis. Mechanisms and Treatment Strategies. N. Engl. J. Med. 328 (25), 1828–1835. 10.1056/NEJM199306243282508 [DOI] [PubMed] [Google Scholar]
- Fu K., Xu M., Zhou Y., Li X., Wang Z., Liu X., et al. (2020). The Status Quo and Way Forwards on the Development of Tibetan Medicine and the Pharmacological Research of Tibetan Materia Medica. Pharmacol. Res. 155, 104688. 10.1016/j.phrs.2020.104688 [DOI] [PubMed] [Google Scholar]
- Gan D., Zhang W., Huang C., Chen J., He W., Wang A., et al. (2018a). Ursolic Acid Ameliorates CCl4-Induced Liver Fibrosis Through the NOXs/ROS Pathway. J. Cel Physiol 233 (10), 6799–6813. 10.1002/jcp.26541 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gan F., Liu Q., Liu Y., Huang D., Pan C., Song S., et al. (2018b). Lycium Barbarum Polysaccharides Improve CCl4-Induced Liver Fibrosis, Inflammatory Response and TLRs/NF-kB Signaling Pathway Expression in Wistar Rats. Life Sci. 192, 205–212. 10.1016/j.lfs.2017.11.047 [DOI] [PubMed] [Google Scholar]
- Ganbold M., Owada Y., Ozawa Y., Shimamoto Y., Ferdousi F., Tominaga K., et al. (2019). Isorhamnetin Alleviates Steatosis and Fibrosis in Mice with Nonalcoholic Steatohepatitis. Sci. Rep. 9 (1), 16210. 10.1038/s41598-019-52736-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao L. N., Yan K., Cui Y. L., Fan G. W., Wang Y. F. (2015). Protective Effect of Salvia Miltiorrhiza and Carthamus tinctorius Extract Against Lipopolysaccharide-Induced Liver Injury. World J. Gastroenterol. 21 (30), 9079–9092. 10.3748/wjg.v21.i30.9079 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao X., Guo S., Zhang S., Liu A., Shi L., Zhang Y. (2018). Matrine Attenuates Endoplasmic Reticulum Stress and Mitochondrion Dysfunction in Nonalcoholic Fatty Liver Disease by Regulating SERCA Pathway. J. Transl Med. 16 (1), 319. 10.1186/s12967-018-1685-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao X., Li C., Tang Y. L., Zhang H., Chan S. W. (2016). Effect of Hedyotis Diffusa Water Extract on Protecting Human Hepatocyte Cells (LO2) from H2O2-Induced Cytotoxicity. Pharm. Biol. 54 (7), 1148–1155. 10.3109/13880209.2015.1056310 [DOI] [PubMed] [Google Scholar]
- Gao X., Liu L. (2016). Research Progress on Epidemiology and Pathogenesis of Alcoholic Liver Disease. Chin. J. Gastroenterol. Imaging 6 (02), 62–65. [Google Scholar]
- Gao Z., Zhang J., Wei L., Yang X., Zhang Y., Cheng B., et al. (2020). The Protective Effects of Imperatorin on Acetaminophen Overdose-Induced Acute Liver Injury. Oxid Med. Cel Longev 2020, 8026838. 10.1155/2020/8026838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ge L., Xiao L., Wan H., Li J., Lv K., Peng S., et al. (2019). Chemical Constituents from Lonicera japonica Flower Buds and Their Anti-hepatoma and Anti-HBV Activities. Bioorg. Chem. 92, 103198. 10.1016/j.bioorg.2019.103198 [DOI] [PubMed] [Google Scholar]
- Ghaffari A., Rafraf M., Navekar R., Sepehri B., Asghari-Jafarabadi M., Ghavami S. M. (2019). Turmeric and Chicory Seed Have Beneficial Effects on Obesity Markers and Lipid Profile in Non-alcoholic Fatty Liver Disease (NAFLD). Int. J. Vitam Nutr. Res. 89 (5-6), 293–302. 10.1024/0300-9831/a000568 [DOI] [PubMed] [Google Scholar]
- Gheflati A., Adelnia E., Nadjarzadeh A. (2019). The Clinical Effects of Purslane (Portulaca Oleracea) Seeds on Metabolic Profiles in Patients with Nonalcoholic Fatty Liver Disease: A Randomized Controlled Clinical Trial. Phytother Res. 33 (5), 1501–1509. 10.1002/ptr.6342 [DOI] [PubMed] [Google Scholar]
- Goh Z. H., Tee J. K., Ho H. K. (2020). An Evaluation of the In Vitro Roles and Mechanisms of Silibinin in Reducing Pyrazinamide- and Isoniazid-Induced Hepatocellular Damage. Int. J. Mol. Sci. 21 (10), 3714. 10.3390/ijms21103714 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gong L., Zhou H., Wang C., He L., Guo C., Peng C., et al. (2021). Hepatoprotective Effect of Forsythiaside a Against Acetaminophen-Induced Liver Injury in Zebrafish: Coupling Network Pharmacology with Biochemical Pharmacology. J. Ethnopharmacol 271, 113890. 10.1016/j.jep.2021.113890 [DOI] [PubMed] [Google Scholar]
- Gu M., Zhang S., Zhao Y., Huang J., Wang Y., Li Y., et al. (2017). Cycloastragenol Improves Hepatic Steatosis by Activating Farnesoid X Receptor Signalling. Pharmacol. Res. 121, 22–32. 10.1016/j.phrs.2017.04.021 [DOI] [PubMed] [Google Scholar]
- Gu X., Manautou J. E. (2012). Molecular Mechanisms Underlying Chemical Liver Injury. Expert Rev. Mol. Med. 14, e4. 10.1017/S1462399411002110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gündüz E., Dursun R., Zengin Y., İçer M., Durgun H. M., Kanıcı A., et al. (2015). Lycium Barbarum Extract Provides Effective protection Against Paracetamol-Induced Acute Hepatotoxicity in Rats. Int. J. Clin. Exp. Med. 8 (5), 7898–7905. [PMC free article] [PubMed] [Google Scholar]
- Guo S., Wang G., Yang Z. (2021). Ligustilide Alleviates the Insulin Resistance, Lipid Accumulation, and Pathological Injury with Elevated Phosphorylated AMPK Level in Rats with Diabetes Mellitus. J. Recept Signal. Transduct Res. 41 (1), 85–92. 10.1080/10799893.2020.1789877 [DOI] [PubMed] [Google Scholar]
- Guoyin Z., Hao P., Min L., Wei G., Zhe C., Changquan L. (20172017). Antihepatocarcinoma Effect of Portulaca Oleracea L. In Mice by PI3K/Akt/mTOR and Nrf2/HO-1/nf-Κb Pathway. Evid. Based Complement. Alternat Med. 2017, 8231358. 10.1155/2017/8231358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta V. K., Siddiqi N. J., Ojha A. K., Sharma B. (2019). Hepatoprotective Effect of Aloe Vera Against Cartap- and Malathion-Induced Toxicity in Wistar Rats. J. Cel Physiol 234 (10), 18329–18343. 10.1002/jcp.28466 [DOI] [PubMed] [Google Scholar]
- Hajighasem A., Farzanegi P., Mazaheri Z., Naghizadeh M., Salehi G. (2018). Effects of Resveratrol, Exercises and Their Combination on Farnesoid X Receptor, Liver X Receptor and Sirtuin 1 Gene Expression and Apoptosis in the Liver of Elderly Rats with Nonalcoholic Fatty Liver. PeerJ 6, e5522. 10.7717/peerj.5522 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han B., Gao Y., Wang Y., Wang L., Shang Z., Wang S., et al. (2016). Protective Effect of a Polysaccharide from Rhizoma Atractylodis Macrocephalae on Acute Liver Injury in Mice. Int. J. Biol. Macromol 87, 85–91. 10.1016/j.ijbiomac.2016.01.086 [DOI] [PubMed] [Google Scholar]
- Han B., Kim S. M., Nam G. E., Kim S. H., Park S. J., Park Y. K., et al. (2020). A Randomized, Double-Blind, Placebo-Controlled, Multi-Centered Clinical Study to Evaluate the Efficacy and Safety of Artemisia Annua L. Extract for Improvement of Liver Function. Clin. Nutr. Res. 9 (4), 258–270. 10.7762/cnr.2020.9.4.258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han C., Wei Y., Wang X., Ba C., Shi W. (2019). Protective Effect of Salvia Miltiorrhiza Polysaccharides on Liver Injury in Chickens. Poult. Sci. 98 (9), 3496–3503. 10.3382/ps/pez153 [DOI] [PubMed] [Google Scholar]
- Hasan I. H., El-Desouky M. A., Hozayen W. G., Abd el Aziz G. M. (2016). Protective Effect of Zingiber Officinale Against CCl4-Induced Liver Fibrosis Is Mediated Through Downregulating the TGF-β1/Smad3 and NF-ĸb/iĸb Pathways. Pharmacology 97 (1-2), 1–9. 10.1159/000441229 [DOI] [PubMed] [Google Scholar]
- He Y., Liu Y. (2021). Research Progress of Intestinal Bacterial Overgrowth in Patients with Liver Cirrhosis. J. integrated Chin. West. Med. Liver Dis. 31 (03), 286–288. [Google Scholar]
- Heiss E. H., Schachner D., Zimmermann K., Dirsch V. M. (2013). Glucose Availability Is a Decisive Factor for Nrf2-Mediated Gene Expression. Redox Biol. 1, 359–365. 10.1016/j.redox.2013.06.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hernandez-Gea V., Friedman S. L. (2011). Pathogenesis of Liver Fibrosis. Annu. Rev. Pathol. 6, 425–456. 10.1146/annurev-pathol-011110-130246 [DOI] [PubMed] [Google Scholar]
- Hesketh T., Zhu W. X. (1997). Health in China. Traditional Chinese Medicine: One Country, Two Systems. BMJ 315 (7100), 115–117. 10.1136/bmj.315.7100.115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho C., Gao Y., Zheng D., Liu Y., Shan S., Fang B., et al. (2019). Alisol A Attenuates High-Fat-Diet-Induced Obesity and Metabolic Disorders via the AMPK/ACC/SREBP-1c Pathway. J. Cel Mol Med 23 (8), 5108–5118. 10.1111/jcmm.14380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho C. Y., Cheng Y. T., Chau C. F., Yen G. C. (2012). Effect of Diallyl Sulfide on In Vitro and In Vivo Nrf2-Mediated Pulmonic Antioxidant Enzyme Expression via Activation ERK/p38 Signaling Pathway. J. Agric. Food Chem. 60 (1), 100–107. 10.1021/jf203800d [DOI] [PubMed] [Google Scholar]
- Holt M., Ju C. (2010). “Drug-induced Liver Injury,” in Handb Exp Pharmacol. Editor Barrett J. E. (New York, NY: Springer; ), 196. [DOI] [PubMed] [Google Scholar]
- Hosny S., Sahyon H., Youssef M., Negm A. (2021). Oleanolic Acid Suppressed DMBA-Induced Liver Carcinogenesis Through Induction of Mitochondrial-Mediated Apoptosis and Autophagy. Nutr. Cancer 73 (6), 968–982. 10.1080/01635581.2020.1776887 [DOI] [PubMed] [Google Scholar]
- Hu B., Gongji G., Ta N., Wuli J. (2019). Research Progress of Alcoholic Liver Disease. J. inner mongolia Univ. nationalities 34 (06), 535–538. [Google Scholar]
- Hu J., Liu K. (2017). Complete and Incomplete Hepatitis B Virus Particles: Formation, Function, and Application. Viruses 9 (3), 56. 10.3390/v9030056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu N., Guo C., Dai X., Wang C., Gong L., Yu L., et al. (2020a). Forsythiae Fructuse Water Extract Attenuates Liver Fibrosis via TLR4/MyD88/NF-Κb and TGF-Β/smads Signaling Pathways. J. Ethnopharmacol 262, 113275. 10.1016/j.jep.2020.113275 [DOI] [PubMed] [Google Scholar]
- Hu N., Wang C., Dai X., Zhou M., Gong L., Yu L., et al. (2020b). Phillygenin Inhibits LPS-Induced Activation and Inflammation of LX2 Cells by TLR4/MyD88/NF-Κb Signaling Pathway. J. Ethnopharmacol 248, 112361. 10.1016/j.jep.2019.112361 [DOI] [PubMed] [Google Scholar]
- Hu Y., Bi X., Zhao P., Zheng H., Huang X. (2015). Cytotoxic Activities, SAR and Anti-invasion Effects of Butylphthalide Derivatives on Human Hepatocellular Carcinoma SMMC7721 Cells. Molecules 20 (11), 20312–20319. 10.3390/molecules201119699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang L., Li Y., Pan H., Lu Y., Zhou X., Shi F. (2020). Cortex Dictamni-Induced Liver Injury in Mice: The Role of P450-Mediated Metabolic Activation of Furanoids. Toxicol. Lett. 330, 41–52. 10.1016/j.toxlet.2020.05.004 [DOI] [PubMed] [Google Scholar]
- Huang W. H., Liao W. R., Sun R. X. (2016). Astragalus Polysaccharide Induces the Apoptosis of Human Hepatocellular Carcinoma Cells by Decreasing the Expression of Notch1. Int. J. Mol. Med. 38 (2), 551–557. 10.3892/ijmm.2016.2632 [DOI] [PubMed] [Google Scholar]
- Huang Y. C., Tsay H. J., Lu M. K., Lin C. H., Yeh C. W., Liu H. K., et al. (2017). Astragalus Membranaceus-Polysaccharides Ameliorates Obesity, Hepatic Steatosis, Neuroinflammation and Cognition Impairment without Affecting Amyloid Deposition in Metabolically Stressed APPswe/PS1dE9 Mice. Int. J. Mol. Sci. 18 (12), 2746. 10.3390/ijms18122746 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hung T. C., Jassey A., Lin C. J., Liu C. H., Lin C. C., Yen M. H., et al. (2018). Methanolic Extract of Rhizoma Coptidis Inhibits the Early Viral Entry Steps of Hepatitis C Virus Infection. Viruses 10 (12), 669. 10.3390/v10120669 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jang E., Kim S. Y., Lee N. R., Yi C. M., Hong D. R., Lee W. S., et al. (2017). Evaluation of Antitumor Activity of Artemisia Capillaris Extract Against Hepatocellular Carcinoma Through the Inhibition of IL-6/STAT3 Signaling Axis. Oncol. Rep. 37 (1), 526–532. 10.3892/or.2016.5283 [DOI] [PubMed] [Google Scholar]
- Jang M. K., Yun Y. R., Kim S. H., Kim J. H., Jung M. H. (2016). Protective Effect of Gomisin N Against Endoplasmic Reticulum Stress-Induced Hepatic Steatosis. Biol. Pharm. Bull. 39 (5), 832–838. 10.1248/bpb.b15-01020 [DOI] [PubMed] [Google Scholar]
- Jarret A., McFarland A. P., Horner S. M., Kell A., Schwerk J., Hong M., et al. (2016). Hepatitis-C-virus-induced microRNAs Dampen Interferon-Mediated Antiviral Signaling. Nat. Med. 22 (12), 1475–1481. 10.1038/nm.4211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jemal A., Bray F., Center M. M., Ferlay J., Ward E., Forman D. (2011). Global Cancer Statistics. CA Cancer J. Clin. 61 (2), 69–90. 10.3322/caac.20107 [DOI] [PubMed] [Google Scholar]
- Jia R., Gu Z., He Q., Du J., Cao L., Jeney G., et al. (2019). Anti-oxidative, Anti-inflammatory and Hepatoprotective Effects of Radix Bupleuri Extract Against Oxidative Damage in Tilapia (Oreochromis niloticus) via Nrf2 and TLRs Signaling Pathway. Fish. Shellfish Immunol. 93, 395–405. 10.1016/j.fsi.2019.07.080 [DOI] [PubMed] [Google Scholar]
- Jiang P., Zhang X., Huang Y., Cheng N., Ma Y. (2017a). Hepatotoxicity Induced by Sophora Flavescens and Hepatic Accumulation of Kurarinone, a Major Hepatotoxic Constituent of Sophora Flavescens in Rats. Molecules 22 (11), 1809. 10.3390/molecules22111809 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang Y., Fan X., Wang Y., Chen P., Zeng H., Tan H., et al. (2015). Schisandrol B Protects Against Acetaminophen-Induced Hepatotoxicity by Inhibition of CYP-Mediated Bioactivation and Regulation of Liver Regeneration. Toxicol. Sci. 143 (1), 107–115. 10.1093/toxsci/kfu216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang Y., Zhang L., Rupasinghe H. P. (2017b). Antiproliferative Effects of Extracts from Salvia Officinalis L. And Saliva Miltiorrhiza Bunge on Hepatocellular Carcinoma Cells. Biomed. Pharmacother. 85, 57–67. 10.1016/j.biopha.2016.11.113 [DOI] [PubMed] [Google Scholar]
- Jin H., Lian N., Bian M., Zhang C., Chen X., Shao J., et al. (2018). Oroxylin A Prevents Alcohol-Induced Hepatic Steatosis Through Inhibition of Hypoxia Inducible Factor 1alpha. Chem. Biol. Interact 285, 14–20. 10.1016/j.cbi.2018.02.025 [DOI] [PubMed] [Google Scholar]
- Jin H., Sakaida I., Tsuchiya M., Okita K. (2005). Herbal Medicine Rhei Rhizome Prevents Liver Fibrosis in Rat Liver Cirrhosis Induced by a Choline-Deficient L-Amino Acid-Defined Diet. Life Sci. 76 (24), 2805–2816. 10.1016/j.lfs.2004.09.041 [DOI] [PubMed] [Google Scholar]
- Jin Q., Jiang S., Wu Y. L., Bai T., Yang Y., Jin X., et al. (2014). Hepatoprotective Effect of Cryptotanshinone from Salvia Miltiorrhiza in D-Galactosamine/lipopolysaccharide-Induced Fulminant Hepatic Failure. Phytomedicine 21 (2), 141–147. 10.1016/j.phymed.2013.07.016 [DOI] [PubMed] [Google Scholar]
- Jindal R., Sinha R., Brar P. (2019). Evaluating the Protective Efficacy of Silybum marianum Against Deltamethrin Induced Hepatotoxicity in Piscine Model. Environ. Toxicol. Pharmacol. 66, 62–68. 10.1016/j.etap.2018.12.014 [DOI] [PubMed] [Google Scholar]
- Jung J. C., Lee Y. H., Kim S. H., Kim K. J., Kim K. M., Oh S., et al. (2016). Hepatoprotective Effect of Licorice, the Root of Glycyrrhiza Uralensis Fischer, in Alcohol-Induced Fatty Liver Disease. BMC Complement. Altern. Med. 16, 19. 10.1186/s12906-016-0997-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung K. H., Rumman M., Yan H., Cheon M. J., Choi J. G., Jin X., et al. (2018). An Ethyl Acetate Fraction of Artemisia Capillaris (ACE-63) Induced Apoptosis and Anti-angiogenesis via Inhibition of PI3K/AKT Signaling in Hepatocellular Carcinoma. Phytother Res. 32 (10), 2034–2046. 10.1002/ptr.6135 [DOI] [PubMed] [Google Scholar]
- Jung S., Son H., Hwang C. E., Cho K. M., Park S. W., Kim H., et al. (2020). The Root of Polygonum Multiflorum Thunb. Alleviates Non-alcoholic Steatosis and Insulin Resistance in High Fat Diet-Fed Mice. Nutrients 12 (8), 2353. 10.3390/nu12082353 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalyanaraman B. (2013). Teaching the Basics of Redox Biology to Medical and Graduate Students: Oxidants, Antioxidants and Disease Mechanisms. Redox Biol. 1, 244–257. 10.1016/j.redox.2013.01.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kandeil M. A., Hashem R. M., Mahmoud M. O., Hetta M. H., Tohamy M. A. (2019). Zingiber Officinale Extract and omega-3 Fatty Acids Ameliorate Endoplasmic Reticulum Stress in a Nonalcoholic Fatty Liver Rat Model. J. Food Biochem. 43 (12), e13076. 10.1111/jfbc.13076 [DOI] [PubMed] [Google Scholar]
- Ke Z., Zhao Y., Tan S., Chen H., Li Y., Zhou Z., et al. (2020). Citrus Reticulata Blanco Peel Extract Ameliorates Hepatic Steatosis, Oxidative Stress and Inflammation in HF and MCD Diet-Induced NASH C57BL/6 J Mice. J. Nutr. Biochem. 83, 108426. 10.1016/j.jnutbio.2020.108426 [DOI] [PubMed] [Google Scholar]
- Kim H. G., Lee S. B., Lee J. S., Kim W. Y., Choi S. H., Son C. G. (2017a). Artemisia Iwayomogi Plus Curcuma Longa Synergistically Ameliorates Nonalcoholic Steatohepatitis in HepG2 Cells. Evid. Based Complement. Alternat Med. 2017, 4390636. 10.1155/2017/4390636 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim H. J., Park K. K., Chung W. Y., Lee S. K., Kim K. R. (2017b). Protective Effect of White-fleshed Peach (Prunus Persica (L.) Batsch) on Chronic Nicotine-Induced Toxicity. J. Cancer Prev. 22 (1), 22–32. 10.15430/JCP.2017.22.1.22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim H. Y., Kim J. K., Choi J. H., Jung J. Y., Oh W. Y., Kim D. C., et al. (2010). Hepatoprotective Effect of Pinoresinol on Carbon Tetrachloride-Induced Hepatic Damage in Mice. J. Pharmacol. Sci. 112 (1), 105–112. 10.1254/jphs.09234fp [DOI] [PubMed] [Google Scholar]
- Kim J., Jung K. H., Yan H. H., Cheon M. J., Kang S., Jin X., et al. (2018). Artemisia Capillaris Leaves Inhibit Cell Proliferation and Induce Apoptosis in Hepatocellular Carcinoma. BMC Complement. Altern. Med. 18 (1), 147. 10.1186/s12906-018-2217-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J., Kim C. S., Jo K., Lee I. S., Kim J. H., Kim J. S. (2020a). POCU1b, the n-Butanol Soluble Fraction of Polygoni Cuspidati Rhizoma et Radix, Attenuates Obesity, Non-Alcoholic Fatty Liver, and Insulin Resistance via Inhibitions of Pancreatic Lipase, cAMP-Dependent PDE Activity, AMPK Activation, and SOCS-3 Suppression. Nutrients 12 (12), 3612. 10.3390/nu12123612 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim T., Song B., Cho K. S., Lee I. S. (2020b). Therapeutic Potential of Volatile Terpenes and Terpenoids from Forests for Inflammatory Diseases. Int. J. Mol. Sci. 21 (6), 2187. 10.3390/ijms21062187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klaikeaw N., Wongphoom J., Werawatganon D., Chayanupatkul M., Siriviriyakul P. (2020). Anti-inflammatory and Anti-oxidant Effects of Aloe Vera in Rats with Non-alcoholic Steatohepatitis. World J. Hepatol. 12 (7), 363–377. 10.4254/wjh.v12.i7.363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kolodziejczyk A. A., Zheng D., Shibolet O., Elinav E. (2019). The Role of the Microbiome in NAFLD and NASH. EMBO Mol. Med. 11 (2), e9302. 10.15252/emmm.201809302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koneru M., Sahu B. D., Gudem S., Kuncha M., Ravuri H. G., Kumar J. M., et al. (2017). Polydatin Alleviates Alcohol-Induced Acute Liver Injury in Mice: Relevance of Matrix Metalloproteinases (MMPs) and Hepatic Antioxidants. Phytomedicine 27, 23–32. 10.1016/j.phymed.2017.01.013 [DOI] [PubMed] [Google Scholar]
- Kong Z. L., Kuo H. P., Johnson A., Wu L. C., Chang K. L. B. (2019). Curcumin-Loaded Mesoporous Silica Nanoparticles Markedly Enhanced Cytotoxicity in Hepatocellular Carcinoma Cells. Int. J. Mol. Sci. 20 (12), 2918. 10.3390/ijms20122918 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo C. Y., Chiu V., Hsieh P. C., Huang C. Y., Huang S. J., Tzeng I. S., et al. (2020). Chrysophanol Attenuates Hepatitis B Virus X Protein-Induced Hepatic Stellate Cell Fibrosis by Regulating Endoplasmic Reticulum Stress and Ferroptosis. J. Pharmacol. Sci. 144 (3), 172–182. 10.1016/j.jphs.2020.07.014 [DOI] [PubMed] [Google Scholar]
- Le J., Fu Y., Han Q., Ma Y., Ji H., Wei X., et al. (2020). Transcriptome Analysis of the Inhibitory Effect of Sennoside A on the Metastasis of Hepatocellular Carcinoma Cells. Front. Pharmacol. 11, 566099. 10.3389/fphar.2020.566099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee C. K., Park K. K., Hwang J. K., Lee S. K., Chung W. Y. (2008). The Pericarp Extract of Prunus Persica Attenuates Chemotherapy-Induced Acute Nephrotoxicity and Hepatotoxicity in Mice. J. Med. Food 11 (2), 302–306. 10.1089/jmf.2007.545 [DOI] [PubMed] [Google Scholar]
- Lee E. H., Baek S. Y., Park J. Y., Kim Y. W. (2020). Emodin in Rheum Undulatum Inhibits Oxidative Stress in the Liver via AMPK with Hippo/Yap Signalling Pathway. Pharm. Biol. 58 (1), 333–341. 10.1080/13880209.2020.1750658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee G. H., Lee H. Y., Choi M. K., Chung H. W., Kim S. W., Chae H. J. (2017a). Protective Effect of Curcuma Longa L. Extract on CCl4-Induced Acute Hepatic Stress. BMC Res. Notes 10 (1), 77. 10.1186/s13104-017-2409-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee H. Y., Kim S. W., Lee G. H., Choi M. K., Chung H. W., Lee Y. C., et al. (2017b). Curcumin and Curcuma Longa L. Extract Ameliorate Lipid Accumulation Through the Regulation of the Endoplasmic Reticulum Redox and ER Stress. Sci. Rep. 7 (1), 6513. 10.1038/s41598-017-06872-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee W., Koo H. R., Choi Y. J., Choi J. G., Oh M. S., Jin X., et al. (2019). Z-ligustilide and N-Butylidenephthalide Isolated from the Aerial Parts of Angelica Tenuissima Inhibit Lipid Accumulation In Vitro and In Vivo. Planta Med. 85 (9-10), 719–728. 10.1055/a-0901-1307 [DOI] [PubMed] [Google Scholar]
- Leung T. M., Nieto N. (2013). CYP2E1 and Oxidant Stress in Alcoholic and Non-alcoholic Fatty Liver Disease. J. Hepatol. 58 (2), 395–398. 10.1016/j.jhep.2012.08.018 [DOI] [PubMed] [Google Scholar]
- Li C. H., Tang S. C., Wong C. H., Wang Y., Jiang J. D., Chen Y. (2018a). Berberine Induces miR-373 Expression in Hepatocytes to Inactivate Hepatic Steatosis Associated AKT-S6 Kinase Pathway. Eur. J. Pharmacol. 825, 107–118. 10.1016/j.ejphar.2018.02.035 [DOI] [PubMed] [Google Scholar]
- Li D. S., Huang Q. F., Guan L. H., Zhang H. Z., Li X., Fu K. L., et al. (2020a). Targeted Bile Acids and Gut Microbiome Profiles Reveal the Hepato-Protective Effect of WZ Tablet (Schisandra Sphenanthera Extract) Against LCA-Induced Cholestasis. Chin. J. Nat. Med. 18 (3), 211–218. 10.1016/S1875-5364(20)30023-6 [DOI] [PubMed] [Google Scholar]
- Li J., Duan B., Guo Y., Zhou R., Sun J., Bie B., et al. (2018b). Baicalein Sensitizes Hepatocellular Carcinoma Cells to 5-FU and Epirubicin by Activating Apoptosis and Ameliorating P-Glycoprotein Activity. Biomed. Pharmacother. 98, 806–812. 10.1016/j.biopha.2018.01.002 [DOI] [PubMed] [Google Scholar]
- Li J. P., Yuan Y., Zhang W. Y., Jiang Z., Hu T. J., Feng Y. T., et al. (2019a). Effect of Radix Isatidis Polysaccharide on Alleviating Insulin Resistance in Type 2 Diabetes Mellitus Cells and Rats. J. Pharm. Pharmacol. 71 (2), 220–229. 10.1111/jphp.13023 [DOI] [PubMed] [Google Scholar]
- Li J., Zhang L., Gao H., Song X., Wu X. (2014). Progress in Pathogenesis and Treatment of Alcoholic Cirrhosis. Med. innovation China 11 (35), 147–149. [Google Scholar]
- Li Q., Li H. J., Xu T., Du H., Huan Gang C. L., Fan G., et al. (2018c). Natural Medicines Used in the Traditional Tibetan Medical System for the Treatment of Liver Diseases. Front. Pharmacol. 9, 29. 10.3389/fphar.2018.00029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li S., Qin Q., Luo D., Pan W., Wei Y., Xu Y., et al. (2020b). Hesperidin Ameliorates Liver Ischemia/reperfusion Injury via Activation of the Akt Pathway. Mol. Med. Rep. 22 (6), 4519–4530. 10.3892/mmr.2020.11561 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li S., Tan H. Y., Wang N., Zhang Z. J., Lao L., Wong C. W., et al. (2015). The Role of Oxidative Stress and Antioxidants in Liver Diseases. Int. J. Mol. Sci. 16 (11), 26087–26124. 10.3390/ijms161125942 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li S., Wang Q., Tao Y., Liu C. (2016). Swertiamarin Attenuates Experimental Rat Hepatic Fibrosis by Suppressing Angiotensin II-Angiotensin Type 1 Receptor-Extracellular Signal-Regulated Kinase Signaling. J. Pharmacol. Exp. Ther. 359 (2), 247–255. 10.1124/jpet.116.234179 [DOI] [PubMed] [Google Scholar]
- Li T., Chiang J. Y. (2014). Bile Acid Signaling in Metabolic Disease and Drug Therapy. Pharmacol. Rev. 66 (4), 948–983. 10.1124/pr.113.008201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X., Jin Q., Yao Q., Xu B., Li L., Zhang S., et al. (2018d). The Flavonoid Quercetin Ameliorates Liver Inflammation and Fibrosis by Regulating Hepatic Macrophages Activation and Polarization in Mice. Front. Pharmacol. 9, 72. 10.3389/fphar.2018.00072 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X., Li X., Lu J., Huang Y., Lv L., Luan Y., et al. (2017). Saikosaponins Induced Hepatotoxicity in Mice via Lipid Metabolism Dysregulation and Oxidative Stress: A Proteomic Study. BMC Complement. Altern. Med. 17 (1), 219. 10.1186/s12906-017-1733-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X., Sun R., Liu R. (2019b). Natural Products in Licorice for the Therapy of Liver Diseases: Progress and Future Opportunities. Pharmacol. Res. 144, 210–226. 10.1016/j.phrs.2019.04.025 [DOI] [PubMed] [Google Scholar]
- Li X., Zhang Y., Jin Q., Xia K. L., Jiang M., Cui B. W., et al. (2018e). Liver Kinase B1/AMP-Activated Protein Kinase-Mediated Regulation by Gentiopicroside Ameliorates P2X7 Receptor-dependent Alcoholic Hepatosteatosis. Br. J. Pharmacol. 175 (9), 1451–1470. 10.1111/bph.14145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y., Shen F., Bao Y., Chen D., Lu H. (2019c). Apoptotic Effects of Rhein Through the Mitochondrial Pathways, Two Death Receptor Pathways, and Reducing Autophagy in Human Liver L02 Cells. Environ. Toxicol. 34 (12), 1292–1302. 10.1002/tox.22830 [DOI] [PubMed] [Google Scholar]
- Li Y., Ye Y., Chen H. (2018g). Astragaloside IV Inhibits Cell Migration and Viability of Hepatocellular Carcinoma Cells via Suppressing Long Noncoding RNA ATB. Biomed. Pharmacother. 99, 134–141. 10.1016/j.biopha.2017.12.108 [DOI] [PubMed] [Google Scholar]
- Li Y. C., Qiao J. Y., Wang B. Y., Bai M., Shen J. D., Cheng Y. X. (2018f). Paeoniflorin Ameliorates Fructose-Induced Insulin Resistance and Hepatic Steatosis by Activating LKB1/AMPK and AKT Pathways. Nutrients 10 (8), 1024. 10.3390/nu10081024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang W., Zhang D., Kang J., Meng X., Yang J., Yang L., et al. (2018). Protective Effects of Rutin on Liver Injury in Type 2 Diabetic Db/db Mice. Biomed. Pharmacother. 107, 721–728. 10.1016/j.biopha.2018.08.046 [DOI] [PubMed] [Google Scholar]
- Liao C. C., Day Y. J., Lee H. C., Liou J. T., Chou A. H., Liu F. C. (2017). ERK Signaling Pathway Plays a Key Role in Baicalin Protection Against Acetaminophen-Induced Liver Injury. Am. J. Chin. Med. 45 (1), 105–121. 10.1142/S0192415X17500082 [DOI] [PubMed] [Google Scholar]
- Liao X., Bu Y., Jia Q. (2020). Traditional Chinese Medicine as Supportive Care for the Management of Liver Cancer: Past, Present, and Future. Genes Dis. 7 (3), 370–379. 10.1016/j.gendis.2019.10.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim J. Y., Lee J. H., Yun D. H., Lee Y. M., Kim D. K. (2021). Inhibitory Effects of Nodakenin on Inflammation and Cell Death in Lipopolysaccharide-Induced Liver Injury Mice. Phytomedicine 81, 153411. 10.1016/j.phymed.2020.153411 [DOI] [PubMed] [Google Scholar]
- Lin C. C., Ng L. T., Hsu F. F., Shieh D. E., Chiang L. C. (2004). Cytotoxic Effects of Coptis Chinensis and Epimedium Sagittatum Extracts and Their Major Constituents (Berberine, Coptisine and Icariin) on Hepatoma and Leukaemia Cell Growth. Clin. Exp. Pharmacol. Physiol. 31 (1-2), 65–69. 10.1111/j.1440-1681.2004.03951.x [DOI] [PubMed] [Google Scholar]
- Lin E. Y., Chagnaadorj A., Huang S. J., Wang C. C., Chiang Y. H., Cheng C. W. (2018a2018). Hepatoprotective Activity of the Ethanolic Extract of Polygonum Multiflorum Thunb. Against Oxidative Stress-Induced Liver Injury. Evid. Based Complement. Alternat Med. 2018, 4130307. 10.1155/2018/4130307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin W., Zhong M., Yin H., Chen Y., Cao Q., Wang C., et al. (2016). Emodin Induces Hepatocellular Carcinoma Cell Apoptosis Through MAPK and PI3K/AKT Signaling Pathways In Vitro and In Vivo. Oncol. Rep. 36 (2), 961–967. 10.3892/or.2016.4861 [DOI] [PubMed] [Google Scholar]
- Lin Y., Kuang Y., Li K., Wang S., Ji S., Chen K., et al. (2017). Nrf2 Activators from Glycyrrhiza Inflata and Their Hepatoprotective Activities Against CCl4-Induced Liver Injury in Mice. Bioorg. Med. Chem. 25 (20), 5522–5530. 10.1016/j.bmc.2017.08.018 [DOI] [PubMed] [Google Scholar]
- Lin Y. N., Chang H. Y., Wang C. C. N., Chu F. Y., Shen H. Y., Chen C. J., et al. (2018b). Oleanolic Acid Inhibits Liver X Receptor Alpha and Pregnane X Receptor to Attenuate Ligand-Induced Lipogenesis. J. Agric. Food Chem. 66 (42), 10964–10976. 10.1021/acs.jafc.8b03372 [DOI] [PubMed] [Google Scholar]
- Liou C. J., Lee Y. K., Ting N. C., Chen Y. L., Shen S. C., Wu S. J., et al. (2019). Protective Effects of Licochalcone A Ameliorates Obesity and Non-alcoholic Fatty Liver Disease via Promotion of the Sirt-1/AMPK Pathway in Mice Fed a High-Fat Diet. Cells 8 (5), 447. 10.3390/cells8050447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu B., Deng X., Jiang Q., Li G., Zhang J., Zhang N., et al. (2020a). Scoparone Improves Hepatic Inflammation and Autophagy in Mice with Nonalcoholic Steatohepatitis by Regulating the ROS/P38/Nrf2 axis and PI3K/AKT/mTOR Pathway in Macrophages. Biomed. Pharmacother. 125, 109895. 10.1016/j.biopha.2020.109895 [DOI] [PubMed] [Google Scholar]
- Liu C., Chen J., Li E., Fan Q., Wang D., Li P., et al. (2015). The Comparison of Antioxidative and Hepatoprotective Activities of Codonopsis Pilosula Polysaccharide (CP) and Sulfated CP. Int. Immunopharmacol 24 (2), 299–305. 10.1016/j.intimp.2014.12.023 [DOI] [PubMed] [Google Scholar]
- Liu D. M., Yang D., Zhou C. Y., Wu J. S., Zhang G. L., Wang P., et al. (2020b). Aloe-emodin Induces Hepatotoxicity by the Inhibition of Multidrug Resistance Protein 2. Phytomedicine 68, 153148. 10.1016/j.phymed.2019.153148 [DOI] [PubMed] [Google Scholar]
- Liu F., Zhang J., Qian J., Wu G., Ma Z. (2018). Emodin Alleviates CCl4-induced L-iver F-ibrosis by S-uppressing E-pithelial-mesenchymal T-ransition and T-ransforming G-rowth F-actor-β1 in R-ats. Mol. Med. Rep. 18 (3), 3262–3270. 10.3892/mmr.2018.9324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu N., Feng J., Lu X., Yao Z., Liu Q., Lv Y., et al. (2019a). Isorhamnetin Inhibits Liver Fibrosis by Reducing Autophagy and Inhibiting Extracellular Matrix Formation via the TGF-β1/Smad3 and TGF-Β1/p38 MAPK Pathways. Mediators Inflamm. 2019, 6175091. 10.1155/2019/6175091 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Q., Pan R., Ding L., Zhang F., Hu L., Ding B., et al. (2017). Rutin Exhibits Hepatoprotective Effects in a Mouse Model of Non-alcoholic Fatty Liver Disease by Reducing Hepatic Lipid Levels and Mitigating Lipid-Induced Oxidative Injuries. Int. Immunopharmacol 49, 132–141. 10.1016/j.intimp.2017.05.026 [DOI] [PubMed] [Google Scholar]
- Liu R., Li X., Huang N., Fan M., Sun R. (2020c). Toxicity of Traditional Chinese Medicine Herbal and Mineral Products. Adv. Pharmacol. 87, 301–346. 10.1016/bs.apha.2019.08.001 [DOI] [PubMed] [Google Scholar]
- Liu W., Li S., Qu Z., Luo Y., Chen R., Wei S., et al. (2019b). Betulinic Acid Induces Autophagy-Mediated Apoptosis Through Suppression of the PI3K/AKT/mTOR Signaling Pathway and Inhibits Hepatocellular Carcinoma. Am. J. Transl Res. 11 (11), 6952–6964. [PMC free article] [PubMed] [Google Scholar]
- Liu Y., Bi Y., Mo C., Zeng T., Huang S., Gao L., et al. (2019c). Betulinic Acid Attenuates Liver Fibrosis by Inducing Autophagy via the Mitogen-Activated Protein Kinase/extracellular Signal-Regulated Kinase Pathway. J. Nat. Med. 73 (1), 179–189. 10.1007/s11418-018-1262-2 [DOI] [PubMed] [Google Scholar]
- Llovet J. M., Kelley R. K., Villanueva A., Singal A. G., Pikarsky E., Roayaie S., et al. (2021). Hepatocellular Carcinoma. Nat. Rev. Dis. Primers 7 (1), 6. 10.1038/s41572-020-00240-3 [DOI] [PubMed] [Google Scholar]
- López-Navarrete G., Ramos-Martínez E., Suárez-Álvarez K., Aguirre-García J., Ledezma-Soto Y., León-Cabrera S., et al. (2011). Th2-associated Alternative Kupffer Cell Activation Promotes Liver Fibrosis Without Inducing Local Inflammation. Int. J. Biol. Sci. 7 (9), 1273–1286. 10.7150/ijbs.7.1273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu C., Xu W., Shao J., Zhang F., Chen A., Zheng S. (2017). Nrf2 Activation Is Required for Ligustrazine to Inhibit Hepatic Steatosis in Alcohol-Preferring Mice and Hepatocytes. Toxicol. Sci. 155 (2), 432–443. 10.1093/toxsci/kfw228 [DOI] [PubMed] [Google Scholar]
- Lukefahr A. L., McEvoy S., Alfafara C., Funk J. L. (2018). Drug-induced Autoimmune Hepatitis Associated with Turmeric Dietary Supplement Use. BMJ Case Rep. 2018, bcr2018224611. 10.1136/bcr-2018-224611 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luna J. M., Scheel T. K., Danino T., Shaw K. S., Mele A., Fak J. J., et al. (2015). Hepatitis C Virus RNA Functionally Sequesters miR-122. Cell 160 (6), 1099–1110. 10.1016/j.cell.2015.02.025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lv H., Xiao Q., Zhou J., Feng H., Liu G., Ci X. (2018). Licochalcone A Upregulates Nrf2 Antioxidant Pathway and Thereby Alleviates Acetaminophen-Induced Hepatotoxicity. Front. Pharmacol. 9, 147. 10.3389/fphar.2018.00147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma B. X., Meng X. S., Tong J., Ge L. L., Zhou G., Wang Y. W. (2018a). Protective Effects of Coptis Chinensis Inflorescence Extract and Linarin Against Carbon Tetrachloride-Induced Damage in HepG2 Cells Through the MAPK/Keap1-Nrf2 Pathway. Food Funct. 9 (4), 2353–2361. 10.1039/c8fo00078f [DOI] [PubMed] [Google Scholar]
- Ma P., Sun C., Li W., Deng W., Adu-Frimpong M., Yu J., et al. (2020). Extraction and Structural Analysis of Angelica Sinensis Polysaccharide with Low Molecular Weight and its Lipid-Lowering Effect on Nonalcoholic Fatty Liver Disease. Food Sci. Nutr. 8 (7), 3212–3224. 10.1002/fsn3.1581 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma Q. (2013). Role of Nrf2 in Oxidative Stress and Toxicity. Annu. Rev. Pharmacol. Toxicol. 53, 401–426. 10.1146/annurev-pharmtox-011112-140320 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma X., Chi Y. H., Niu M., Zhu Y., Zhao Y. L., Chen Z., et al. (2016). Metabolomics Coupled with Multivariate Data and Pathway Analysis on Potential Biomarkers in Cholestasis and Intervention Effect of Paeonia Lactiflora Pall. Front. Pharmacol. 7, 14. 10.3389/fphar.2016.00014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma X., Wen J. X., Gao S. J., He X., Li P. Y., Yang Y. X., et al. (2018b). Paeonia Lactiflora Pall. Regulates the NF-Κb-NLRP3 Inflammasome Pathway to Alleviate Cholestasis in Rats. J. Pharm. Pharmacol. 70 (12), 1675–1687. 10.1111/jphp.13008 [DOI] [PubMed] [Google Scholar]
- Ma X., Zhao Y. L., Zhu Y., Chen Z., Wang J. B., Li R. Y., et al. (2015). Paeonia Lactiflora Pall. Protects Against ANIT-Induced Cholestasis by Activating Nrf2 via PI3K/Akt Signaling Pathway. Drug Des. Devel Ther. 9, 5061–5074. 10.2147/DDDT.S90030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma X. Y., Zhang M., Fang G., Cheng C. J., Wang M. K., Han Y. M., et al. (2021). Ursolic Acid Reduces Hepatocellular Apoptosis and Alleviates Alcohol-Induced Liver Injury via Irreversible Inhibition of CASP3 In Vivo. Acta Pharmacol. Sin 42 (7), 1101–1110. 10.1038/s41401-020-00534-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma Y., Chen K., Lv L., Wu S., Guo Z. (2019). Ferulic Acid Ameliorates Nonalcoholic Fatty Liver Disease and Modulates the Gut Microbiota Composition in High-Fat Diet Fed ApoE-/- Mice. Biomed. Pharmacother. 113, 108753. 10.1016/j.biopha.2019.108753 [DOI] [PubMed] [Google Scholar]
- Mahmoud A. M., Hussein O. E., Hozayen W. G., Bin-Jumah M., Abd El-Twab S. M. (2020). Ferulic Acid Prevents Oxidative Stress, Inflammation, and Liver Injury via Upregulation of Nrf2/HO-1 Signaling in Methotrexate-Induced Rats. Environ. Sci. Pollut. Res. Int. 27 (8), 7910–7921. 10.1007/s11356-019-07532-6 [DOI] [PubMed] [Google Scholar]
- Mahmoud M. F., Gamal S., El-Fayoumi H. M. (2014). Limonin Attenuates Hepatocellular Injury Following Liver Ischemia and Reperfusion in Rats via Toll-like Receptor Dependent Pathway. Eur. J. Pharmacol. 740, 676–682. 10.1016/j.ejphar.2014.06.010 [DOI] [PubMed] [Google Scholar]
- Manns M. P., Buti M., Gane E., Pawlotsky J. M., Razavi H., Terrault N., et al. (2017). Hepatitis C Virus Infection. Nat. Rev. Dis. Primers 3, 17006. 10.1038/nrdp.2017.6 [DOI] [PubMed] [Google Scholar]
- Mantovani A., Gatti D., Zoppini G., Lippi G., Bonora E., Byrne C. D., et al. (2019). Association Between Nonalcoholic Fatty Liver Disease and Reduced Bone Mineral Density in Children: A Meta-Analysis. Hepatology 70 (3), 812–823. 10.1002/hep.30538 [DOI] [PubMed] [Google Scholar]
- Marvie P., Lisbonne M., L'Helgoualc'h A., Rauch M., Turlin B., Preisser L., et al. (2010). Interleukin-33 Overexpression Is Associated with Liver Fibrosis in Mice and Humans. J. Cel Mol Med 14 (6B), 1726–1739. 10.1111/j.1582-4934.2009.00801.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medzhitov R. (2008). Origin and Physiological Roles of Inflammation. Nature 454 (7203), 428–435. 10.1038/nature07201 [DOI] [PubMed] [Google Scholar]
- Mello T., Ceni E., Surrenti C., Galli A. (2008). Alcohol Induced Hepatic Fibrosis: Role of Acetaldehyde. Mol. Aspects Med. 29 (1-2), 17–21. 10.1016/j.mam.2007.10.001 [DOI] [PubMed] [Google Scholar]
- Meng Q., Chen X., Wang C., Liu Q., Sun H., Sun P., et al. (2015). Protective Effects of Alisol B 23-acetate from Edible Botanical Rhizoma Alismatis Against Carbon Tetrachloride-Induced Hepatotoxicity in Mice. Food Funct. 6 (4), 1241–1250. 10.1039/c5fo00082c [DOI] [PubMed] [Google Scholar]
- Meng Q., Duan X. P., Wang C. Y., Liu Z. H., Sun P. Y., Huo X. K., et al. (2017). Alisol B 23-acetate Protects Against Non-alcoholic Steatohepatitis in Mice via Farnesoid X Receptor Activation. Acta Pharmacol. Sin 38 (1), 69–79. 10.1038/aps.2016.119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meng X. L., Zhu Z. X., Lu R. H., Li S., Hu W. P., Qin C. B., et al. (2019). Regulation of Growth Performance and Lipid Metabolism in Juvenile Grass Carp (Ctenopharyngodon Idella) with Honeysuckle (Lonicera japonica) Extract. Fish. Physiol. Biochem. 45 (5), 1563–1573. 10.1007/s10695-019-00644-3 [DOI] [PubMed] [Google Scholar]
- Messina J. P., Humphreys I., Flaxman A., Brown A., Cooke G. S., Pybus O. G., et al. (2015). Global Distribution and Prevalence of Hepatitis C Virus Genotypes. Hepatology 61 (1), 77–87. 10.1002/hep.27259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mo Z. Z., Lin Z. X., Su Z. R., Zheng L., Li H. L., Xie J. H., et al. (2018). Angelica Sinensis Supercritical Fluid CO2 Extract Attenuates D-Galactose-Induced Liver and Kidney Impairment in Mice by Suppressing Oxidative Stress and Inflammation. J. Med. Food 21 (9), 887–898. 10.1089/jmf.2017.4061 [DOI] [PubMed] [Google Scholar]
- Mo Z. Z., Liu Y. H., Li C. L., Xu L. Q., Wen L. L., Xian Y. F., et al. (2017). Protective Effect of SFE-CO2 of Ligusticum Chuanxiong Hort Against D-Galactose-Induced Injury in the Mouse Liver and Kidney. Rejuvenation Res. 20 (3), 231–243. 10.1089/rej.2016.1870 [DOI] [PubMed] [Google Scholar]
- Mo'men Y. S., Hussein R. M., Kandeil M. A. (2019). Involvement of PI3K/Akt Pathway in the Protective Effect of Hesperidin Against a Chemically Induced Liver Cancer in Rats. J. Biochem. Mol. Toxicol. 33 (6), e22305. 10.1002/jbt.22305 [DOI] [PubMed] [Google Scholar]
- Moghadam A. R., Tutunchi S., Namvaran-Abbas-Abad A., Yazdi M., Bonyadi F., Mohajeri D., et al. (2015). Pre-administration of Turmeric Prevents Methotrexate-Induced Liver Toxicity and Oxidative Stress. BMC Complement. Altern. Med. 15, 246. 10.1186/s12906-015-0773-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mu M., Zuo S., Wu R. M., Deng K. S., Lu S., Zhu J. J., et al. (2018). Ferulic Acid Attenuates Liver Fibrosis and Hepatic Stellate Cell Activation via Inhibition of TGF-β/Smad Signaling Pathway. Drug Des. Devel Ther. 12, 4107–4115. 10.2147/DDDT.S186726 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mu Q., Wang H., Tong L., Fang Q., Xiang M., Han L., et al. (2020). Betulinic Acid Improves Nonalcoholic Fatty Liver Disease Through YY1/FAS Signaling Pathway. FASEB J. 34 (9), 13033–13048. 10.1096/fj.202000546R [DOI] [PubMed] [Google Scholar]
- Nagappan A., Jung D. Y., Kim J. H., Lee H., Jung M. H. (2018). Gomisin N Alleviates Ethanol-Induced Liver Injury Through Ameliorating Lipid Metabolism and Oxidative Stress. Int. J. Mol. Sci. 19 (9), 2601. 10.3390/ijms19092601 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagappan A., Kim J. H., Jung D. Y., Jung M. H. (2019). Cryptotanshinone from the Salvia Miltiorrhiza Bunge Attenuates Ethanol-Induced Liver Injury by Activation of AMPK/SIRT1 and Nrf2 Signaling Pathways. Int. J. Mol. Sci. 21 (1), 265. 10.3390/ijms21010265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura T., Naguro I., Ichijo H. (2019). Iron Homeostasis and Iron-Regulated ROS in Cell Death, Senescence and Human Diseases. Biochim. Biophys. Acta Gen. Subj 1863 (9), 1398–1409. 10.1016/j.bbagen.2019.06.010 [DOI] [PubMed] [Google Scholar]
- Navarro V. J., Belle S. H., D'Amato M., Adfhal N., Brunt E. M., Fried M. W., et al. (2019). Silymarin in Non-cirrhotics with Non-alcoholic Steatohepatitis: A Randomized, Double-Blind, Placebo Controlled Trial. PLoS One 14 (9), e0221683. 10.1371/journal.pone.0221683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neyrinck A. M., Etxeberria U., Taminiau B., Daube G., Van Hul M., Everard A., et al. (2017). Rhubarb Extract Prevents Hepatic Inflammation Induced by Acute Alcohol Intake, an Effect Related to the Modulation of the Gut Microbiota. Mol. Nutr. Food Res. 61 (1), 1–12. 10.1002/mnfr.201500899 [DOI] [PubMed] [Google Scholar]
- Nguyen P., Leray V., Diez M., Serisier S., Le Bloc'h J., Siliart B., et al. (2008). Liver Lipid Metabolism. J. Anim. Physiol. Anim. Nutr. (Berl) 92 (3), 272–283. 10.1111/j.1439-0396.2007.00752.x [DOI] [PubMed] [Google Scholar]
- Ning C., Gao X., Wang C., Huo X., Liu Z., Sun H., et al. (2018). Hepatoprotective Effect of Ginsenoside Rg1 from Panax Ginseng on Carbon Tetrachloride-Induced Acute Liver Injury by Activating Nrf2 Signaling Pathway in Mice. Environ. Toxicol. 33 (10), 1050–1060. 10.1002/tox.22616 [DOI] [PubMed] [Google Scholar]
- Nouri-Vaskeh M., Afshan H., Malek Mahdavi A., Alizadeh L., Fan X., Zarei M. (2020a). Curcumin Ameliorates Health-Related Quality of Life in Patients with Liver Cirrhosis: A Randomized, Double-Blind Placebo-Controlled Trial. Complement. Ther. Med. 49, 102351. 10.1016/j.ctim.2020.102351 [DOI] [PubMed] [Google Scholar]
- Nouri-Vaskeh M., Malek Mahdavi A., Afshan H., Alizadeh L., Zarei M. (2020b). Effect of Curcumin Supplementation on Disease Severity in Patients with Liver Cirrhosis: A Randomized Controlled Trial. Phytother Res. 34 (6), 1446–1454. 10.1002/ptr.6620 [DOI] [PubMed] [Google Scholar]
- Okubo S., Ohta T., Shoyama Y., Uto T. (2020). Arctigenin Suppresses Cell Proliferation via Autophagy Inhibition in Hepatocellular Carcinoma Cells. J. Nat. Med. 74 (3), 525–532. 10.1007/s11418-020-01396-8 [DOI] [PubMed] [Google Scholar]
- Onyekwere C. A., Ogbera A. O., Samaila A. A., Balogun B. O., Abdulkareem F. B. (2015). Nonalcoholic Fatty Liver Disease: Synopsis of Current Developments. Niger. J. Clin. Pract. 18 (6), 703–712. 10.4103/1119-3077.163288 [DOI] [PubMed] [Google Scholar]
- Ou Q., Weng Y., Wang S., Zhao Y., Zhang F., Zhou J., et al. (2018). Silybin Alleviates Hepatic Steatosis and Fibrosis in NASH Mice by Inhibiting Oxidative Stress and Involvement with the Nf-Κb Pathway. Dig. Dis. Sci. 63 (12), 3398–3408. 10.1007/s10620-018-5268-0 [DOI] [PubMed] [Google Scholar]
- Padda M. S., Sanchez M., Akhtar A. J., Boyer J. L. (2011). Drug-induced Cholestasis. Hepatology 53 (4), 1377–1387. 10.1002/hep.24229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan C. W., Zhou G. Y., Chen W. L., Zhuge L., Jin L. X., Zheng Y., et al. (2015a). Protective Effect of Forsythiaside A on Lipopolysaccharide/d-Galactosamine-Induced Liver Injury. Int. Immunopharmacol 26 (1), 80–85. 10.1016/j.intimp.2015.03.009 [DOI] [PubMed] [Google Scholar]
- Pan T. L., Wang P. W., Huang C. H., Leu Y. L., Wu T. H., Wu Y. R., et al. (2015b). Herbal Formula, Scutellariae Radix and Rhei Rhizoma Attenuate Dimethylnitrosamine-Induced Liver Fibrosis in a Rat Model. Sci. Rep. 5, 11734. 10.1038/srep11734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park C. H., Shin M. R., An B. K., Joh H. W., Lee J. C., Roh S. S., et al. (2017). Heat-Processed Scutellariae Radix Protects Hepatic Inflammation through the Amelioration of Oxidative Stress in Lipopolysaccharide-Induced Mice. Am. J. Chin. Med. 45 (6), 1233–1252. 10.1142/S0192415X17500689 [DOI] [PubMed] [Google Scholar]
- Park Y. J., Lee K. H., Jeon M. S., Lee Y. H., Ko Y. J., Pang C., et al. (2020). Hepatoprotective Potency of Chrysophanol 8-O-Glucoside from Rheum Palmatum L. Against Hepatic Fibrosis via Regulation of the STAT3 Signaling Pathway. Int. J. Mol. Sci. 21 (23), 9044. 10.3390/ijms21239044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parlati L., Voican C. S., Perlemuter K., Perlemuter G. (2017). Aloe Vera-Induced Acute Liver Injury: A Case Report and Literature Review. Clin. Res. Hepatol. Gastroenterol. 41 (4), e39–e42. 10.1016/j.clinre.2016.10.002 [DOI] [PubMed] [Google Scholar]
- Peng Y., Yang T., Huang K., Shen L., Tao Y., Liu C. (2018). Salvia Miltiorrhiza Ameliorates Liver Fibrosis by Activating Hepatic Natural Killer Cells In Vivo and In Vitro. Front. Pharmacol. 9, 762. 10.3389/fphar.2018.00762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penny S. M. (2013). Alcoholic Liver Disease. Radiol. Technol. 84 (6), 577–585. [PubMed] [Google Scholar]
- Pérez-Vargas J. E., Zarco N., Shibayama M., Segovia J., Tsutsumi V., Muriel P. (2014). Hesperidin Prevents Liver Fibrosis in Rats by Decreasing the Expression of Nuclear Factor-Κb, Transforming Growth Factor-β and Connective Tissue Growth Factor. Pharmacology 94 (1-2), 80–89. 10.1159/000366206 [DOI] [PubMed] [Google Scholar]
- Porras D., Nistal E., Martínez-Flórez S., Pisonero-Vaquero S., Olcoz J. L., Jover R., et al. (2017). Protective Effect of Quercetin on High-Fat Diet-Induced Non-alcoholic Fatty Liver Disease in Mice Is Mediated by Modulating Intestinal Microbiota Imbalance and Related Gut-Liver Axis Activation. Free Radic. Biol. Med. 102, 188–202. 10.1016/j.freeradbiomed.2016.11.037 [DOI] [PubMed] [Google Scholar]
- Poynard T., Bedossa P., Opolon P. (1997). Natural History of Liver Fibrosis Progression in Patients with Chronic Hepatitis C. The OBSVIRC, METAVIR, CLINIVIR, and DOSVIRC Groups. Lancet 349 (9055), 825–832. 10.1016/s0140-6736(96)07642-8 [DOI] [PubMed] [Google Scholar]
- Qu X., Gao H., Zhai J., Sun J., Tao L., Zhang Y., et al. (2020). Astragaloside IV Enhances Cisplatin Chemosensitivity in Hepatocellular Carcinoma by Suppressing MRP2. Eur. J. Pharm. Sci. 148, 105325. 10.1016/j.ejps.2020.105325 [DOI] [PubMed] [Google Scholar]
- Qu Z. X., Li F., Ma C. D., Liu J., Li S. D., Wang W. L. (2015). Effects of gentiana Scabra Bage on Expression of Hepatic Type I, III Collagen Proteins in Paragonimus Skrjabini Rats with Liver Fibrosis. Asian Pac. J. Trop. Med. 8 (1), 60–63. 10.1016/S1995-7645(14)60188-7 [DOI] [PubMed] [Google Scholar]
- Rahmani S., Asgary S., Askari G., Keshvari M., Hatamipour M., Feizi A., et al. (2016). Treatment of Non-alcoholic Fatty Liver Disease with Curcumin: A Randomized Placebo-Controlled Trial. Phytother Res. 30 (9), 1540–1548. 10.1002/ptr.5659 [DOI] [PubMed] [Google Scholar]
- Rakshit S., Shukla P., Verma A., Kumar Nirala S., Bhadauria M. (2021). Protective Role of Rutin Against Combined Exposure to Lipopolysaccharide and D-Galactosamine-Induced Dysfunctions in Liver, Kidney, and Brain: Hematological, Biochemical, and Histological Evidences. J. Food Biochem. 45 (2), e13605. 10.1111/jfbc.13605 [DOI] [PubMed] [Google Scholar]
- Rehermann B., Ferrari C., Pasquinelli C., Chisari F. V. (1996). The Hepatitis B Virus Persists for Decades after Patients' Recovery from Acute Viral Hepatitis Despite Active Maintenance of a Cytotoxic T-Lymphocyte Response. Nat. Med. 2 (10), 1104–1108. 10.1038/nm1096-1104 [DOI] [PubMed] [Google Scholar]
- Rehman S., Nazar R., Butt A. M., Ijaz B., Tasawar N., Sheikh A. K., et al. (2021). Phytochemical Screening and Protective Effects of Prunus Persica Seeds Extract on Carbon Tetrachloride-Induced Hepatic Injury in Rats. Curr. Pharm. Biotechnol. 10.2174/1389201022666210203142138 [DOI] [PubMed] [Google Scholar]
- Ren M., McGowan E., Li Y., Zhu X., Lu X., Zhu Z., et al. (2019). Saikosaponin-d Suppresses COX2 Through p-STAT3/C/EBPβ Signaling Pathway in Liver Cancer: A Novel Mechanism of Action. Front. Pharmacol. 10, 623. 10.3389/fphar.2019.00623 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson M. W., Harmon C., O'Farrelly C. (2016). Liver Immunology and its Role in Inflammation and Homeostasis. Cell Mol Immunol 13 (3), 267–276. 10.1038/cmi.2016.3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roghani M., Kalantari H., Khodayar M. J., Khorsandi L., Kalantar M., Goudarzi M., et al. (2020). Alleviation of Liver Dysfunction, Oxidative Stress and Inflammation Underlies the Protective Effect of Ferulic Acid in Methotrexate-Induced Hepatotoxicity. Drug Des. Devel Ther. 14, 1933–1941. 10.2147/DDDT.S237107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saberi-Karimian M., Keshvari M., Ghayour-Mobarhan M., Salehizadeh L., Rahmani S., Behnam B., et al. (2020). Effects of Curcuminoids on Inflammatory Status in Patients with Non-alcoholic Fatty Liver Disease: A Randomized Controlled Trial. Complement. Ther. Med. 49, 102322. 10.1016/j.ctim.2020.102322 [DOI] [PubMed] [Google Scholar]
- Salameh H., Raff E., Erwin A., Seth D., Nischalke H. D., Falleti E., et al. (2015). PNPLA3 Gene Polymorphism Is Associated with Predisposition to and Severity of Alcoholic Liver Disease. Am. J. Gastroenterol. 110 (6), 846–856. 10.1038/ajg.2015.137 [DOI] [PubMed] [Google Scholar]
- Seitz S., Urban S., Antoni C., Böttcher B. (2007). Cryo-electron Microscopy of Hepatitis B Virions Reveals Variability in Envelope Capsid Interactions. EMBO J. 26 (18), 4160–4167. 10.1038/sj.emboj.7601841 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shan Y., Jiang B., Yu J., Wang J., Wang X., Li H., et al. (2019). Protective Effect of Schisandra Chinensis Polysaccharides Against the Immunological Liver Injury in Mice Based on Nrf2/ARE and TLR4/NF-Κb Signaling Pathway. J. Med. Food 22 (9), 885–895. 10.1089/jmf.2018.4377 [DOI] [PubMed] [Google Scholar]
- Shang Y., Li X. F., Jin M. J., Li Y., Wu Y. L., Jin Q., et al. (2018). Leucodin Attenuates Inflammatory Response in Macrophages and Lipid Accumulation in Steatotic Hepatocytes via P2x7 Receptor Pathway: A Potential Role in Alcoholic Liver Disease. Biomed. Pharmacother. 107, 374–381. 10.1016/j.biopha.2018.08.009 [DOI] [PubMed] [Google Scholar]
- Shen B., Feng H., Cheng J., Li Z., Jin M., Zhao L., et al. (2020). Geniposide Alleviates Non-alcohol Fatty Liver Disease via Regulating Nrf2/AMPK/mTOR Signalling Pathways. J. Cel Mol Med 24 (9), 5097–5108. 10.1111/jcmm.15139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen H., Wang H., Wang L., Wang L., Zhu M., Ming Y., et al. (2017). Ethanol Extract of Root of Prunus Persica Inhibited the Growth of Liver Cancer Cell HepG2 by Inducing Cell Cycle Arrest and Migration Suppression. Evid. Based Complement. Alternat Med. 2017, 8231936. 10.1155/2017/8231936 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen T. D., Pyrsopoulos N., Rustgi V. K. (2018). Microbiota and the Liver. Liver Transpl. 24 (4), 539–550. 10.1002/lt.25008 [DOI] [PubMed] [Google Scholar]
- Sheu M. J., Chiu C. C., Yang D. J., Hsu T. C., Tzang B. S. (2017). The Root Extract of Gentiana Macrophylla Pall. Alleviates B19-NS1-Exacerbated Liver Injuries in NZB/W F1 Mice. J. Med. Food 20 (1), 56–64. 10.1089/jmf.2016.3817 [DOI] [PubMed] [Google Scholar]
- Shi H., Shi A., Dong L., Lu X., Wang Y., Zhao J., et al. (2016). Chlorogenic Acid Protects Against Liver Fibrosis In Vivo and In Vitro Through Inhibition of Oxidative Stress. Clin. Nutr. 35 (6), 1366–1373. 10.1016/j.clnu.2016.03.002 [DOI] [PubMed] [Google Scholar]
- Shi J., Han G., Wang J., Han X., Zhao M., Duan X., et al. (2020). Matrine Promotes Hepatic Oval Cells Differentiation into Hepatocytes and Alleviates Liver Injury by Suppression of Notch Signalling Pathway. Life Sci. 261, 118354. 10.1016/j.lfs.2020.118354 [DOI] [PubMed] [Google Scholar]
- Shi Y., Zheng M. (2020). Hepatitis B Virus Persistence and Reactivation. BMJ 370, m2200. 10.1136/bmj.m2200 [DOI] [PubMed] [Google Scholar]
- Singal A. K., Bataller R., Ahn J., Kamath P. S., Shah V. H. (2018). ACG Clinical Guideline: Alcoholic Liver Disease. Am. J. Gastroenterol. 113 (2), 175–194. 10.1038/ajg.2017.469 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spearman C. W., Dusheiko G. M., Hellard M., Sonderup M. (2019). Hepatitis C. Lancet 394 (10207), 1451–1466. 10.1016/S0140-6736(19)32320-7 [DOI] [PubMed] [Google Scholar]
- Stöckigt J., Antonchick A. P., Wu F., Waldmann H. (2011). The Pictet-Spengler Reaction in Nature and in Organic Chemistry. Angew. Chem. Int. Ed. Engl. 50 (37), 8538–8564. 10.1002/anie.201008071 [DOI] [PubMed] [Google Scholar]
- Su C. M., Wang H. C., Hsu F. T., Lu C. H., Lai C. K., Chung J. G., et al. (2020). Astragaloside IV Induces Apoptosis, G1-phase Arrest and Inhibits Anti-apoptotic Signaling in Hepatocellular Carcinoma. In Vivo 34 (2), 631–638. 10.21873/invivo.11817 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun J., Liu Y., Yu J., Wu J., Gao W., Ran L., et al. (2019). APS Could Potentially Activate Hepatic Insulin Signaling in HFD-Induced IR Mice. J. Mol. Endocrinol. 63 (1), 77–91. 10.1530/JME-19-0035 [DOI] [PubMed] [Google Scholar]
- Tan X., Sun Z., Liu Q., Ye H., Zou C., Ye C., et al. (2018). Effects of Dietary Ginkgo Biloba Leaf Extract on Growth Performance, Plasma Biochemical Parameters, Fish Composition, Immune Responses, Liver Histology, and Immune and Apoptosis-Related Genes Expression of Hybrid Grouper (Epinephelus Lanceolatus♂ × Epinephelus Fuscoguttatus♀) Fed High Lipid Diets. Fish. Shellfish Immunol. 72, 399–409. 10.1016/j.fsi.2017.10.022 [DOI] [PubMed] [Google Scholar]
- Tan X., Sun Z., Ye C., Lin H. (2019). The Effects of Dietary Lycium Barbarum Extract on Growth Performance, Liver Health and Immune Related Genes Expression in Hybrid Grouper (Epinephelus Lanceolatus♂ × E. Fuscoguttatus♀) Fed High Lipid Diets. Fish. Shellfish Immunol. 87, 847–852. 10.1016/j.fsi.2019.02.016 [DOI] [PubMed] [Google Scholar]
- Tang F., Fan K., Wang K., Bian C. (2019). Amygdalin Attenuates Acute Liver Injury Induced by D-Galactosamine and Lipopolysaccharide by Regulating the NLRP3, NF-Κb and Nrf2/NQO1 Signalling Pathways. Biomed. Pharmacother. 111, 527–536. 10.1016/j.biopha.2018.12.096 [DOI] [PubMed] [Google Scholar]
- Tholl D. (2015). Biosynthesis and Biological Functions of Terpenoids in Plants. Adv. Biochem. Eng. Biotechnol. 148, 63–106. 10.1007/10_2014_295 [DOI] [PubMed] [Google Scholar]
- Trépo C., Chan H. L., Lok A. (2014). Hepatitis B Virus Infection. Lancet 384 (9959), 2053–2063. 10.1016/S0140-6736(14)60220-8 [DOI] [PubMed] [Google Scholar]
- Tripathi A., Debelius J., Brenner D. A., Karin M., Loomba R., Schnabl B., et al. (2018). The Gut-Liver Axis and the Intersection with the Microbiome. Nat. Rev. Gastroenterol. Hepatol. 15 (7), 397–411. 10.1038/s41575-018-0011-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsochatzis E. A., Bosch J., Burroughs A. K. (2014). Liver Cirrhosis. Lancet 383 (9930), 1749–1761. 10.1016/S0140-6736(14)60121-5 [DOI] [PubMed] [Google Scholar]
- Tsuchida T., Friedman S. L. (2017). Mechanisms of Hepatic Stellate Cell Activation. Nat. Rev. Gastroenterol. Hepatol. 14 (7), 397–411. 10.1038/nrgastro.2017.38 [DOI] [PubMed] [Google Scholar]
- Tu Y. (2016). Artemisinin-A Gift from Traditional Chinese Medicine to the World (Nobel Lecture). Angew. Chem. Int. Ed. Engl. 55 (35), 10210–10226. 10.1002/anie.201601967 [DOI] [PubMed] [Google Scholar]
- Uchio R., Higashi Y., Kohama Y., Kawasaki K., Hirao T., Muroyama K., et al. (2017). A Hot Water Extract of Turmeric (Curcuma Longa) Suppresses Acute Ethanol-Induced Liver Injury in Mice by Inhibiting Hepatic Oxidative Stress and Inflammatory Cytokine Production. J. Nutr. Sci. 6, e3. 10.1017/jns.2016.43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Hung P. (2016). Phenolic Compounds of Cereals and Their Antioxidant Capacity. Crit. Rev. Food Sci. Nutr. 56 (1), 25–35. 10.1080/10408398.2012.708909 [DOI] [PubMed] [Google Scholar]
- Veskovic M., Mladenovic D., Milenkovic M., Tosic J., Borozan S., Gopcevic K., et al. (2019). Betaine Modulates Oxidative Stress, Inflammation, Apoptosis, Autophagy, and Akt/mTOR Signaling in Methionine-Choline Deficiency-Induced Fatty Liver Disease. Eur. J. Pharmacol. 848, 39–48. 10.1016/j.ejphar.2019.01.043 [DOI] [PubMed] [Google Scholar]
- Wah Kheong C., Nik Mustapha N. R., Mahadeva S. (2017). A Randomized Trial of Silymarin for the Treatment of Nonalcoholic Steatohepatitis. Clin. Gastroenterol. Hepatol. 15 (12), 1940–1949.e8. 10.1016/j.cgh.2017.04.016 [DOI] [PubMed] [Google Scholar]
- Wan S., Luo F., Huang C., Liu C., Luo Q., Zhu X. (2020). Ursolic Acid Reverses Liver Fibrosis by Inhibiting Interactive NOX4/ROS and RhoA/ROCK1 Signalling Pathways. Aging (Albany NY) 12 (11), 10614–10632. 10.18632/aging.103282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan S. Z., Liu C., Huang C. K., Luo F. Y., Zhu X. (2019). Ursolic Acid Improves Intestinal Damage and Bacterial Dysbiosis in Liver Fibrosis Mice. Front. Pharmacol. 10, 1321. 10.3389/fphar.2019.01321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang F. S., Fan J. G., Zhang Z., Gao B., Wang H. Y. (2014). The Global Burden of Liver Disease: The Major Impact of China. Hepatology 60 (6), 2099–2108. 10.1002/hep.27406 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang G., Fu Y., Li J., Li Y., Zhao Q., Hu A., et al. (2021a). Aqueous Extract of Polygonatum Sibiricum Ameliorates Ethanol-Induced Mice Liver Injury via Regulation of the Nrf2/ARE Pathway. J. Food Biochem. 45 (1), e13537. 10.1111/jfbc.13537 [DOI] [PubMed] [Google Scholar]
- Wang J., Liao A. M., Thakur K., Zhang J. G., Huang J. H., Wei Z. J. (2019a). Licochalcone B Extracted from Glycyrrhiza Uralensis Fisch Induces Apoptotic Effects in Human Hepatoma Cell HepG2. J. Agric. Food Chem. 67 (12), 3341–3353. 10.1021/acs.jafc.9b00324 [DOI] [PubMed] [Google Scholar]
- Wang J., Wong Y. K., Liao F. (2018). What Has Traditional Chinese Medicine Delivered for Modern Medicine? Expert Rev. Mol. Med. 20, e4. 10.1017/erm.2018.3 [DOI] [PubMed] [Google Scholar]
- Wang J., Zhao Y., Xiao X., Li H., Zhao H., Zhang P., et al. (2009). Assessment of the Renal protection and Hepatotoxicity of Rhubarb Extract in Rats. J. Ethnopharmacol 124 (1), 18–25. 10.1016/j.jep.2009.04.018 [DOI] [PubMed] [Google Scholar]
- Wang K., Song Z., Wang H., Li Q., Cui Z., Zhang Y. (2016). Angelica Sinensis Polysaccharide Attenuates Concanavalin A-Induced Liver Injury in Mice. Int. Immunopharmacol 31, 140–148. 10.1016/j.intimp.2015.12.021 [DOI] [PubMed] [Google Scholar]
- Wang K., Wang J., Song M., Wang H., Xia N., Zhang Y. (2020a). Angelica Sinensis Polysaccharide Attenuates CCl4-Induced Liver Fibrosis via the IL-22/STAT3 Pathway. Int. J. Biol. Macromol 162, 273–283. 10.1016/j.ijbiomac.2020.06.166 [DOI] [PubMed] [Google Scholar]
- Wang Q., Liang Y., Peng C., Jiang P. (2020b). Network Pharmacology-Based Study on the Mechanism of Scutellariae Radix for Hepatocellular Carcinoma Treatment. Evid. Based Complement. Alternat Med. 2020, 8897918. 10.1155/2020/8897918 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang R., Zhang D., Tang D., Sun K., Peng J., Zhu W., et al. (2021b). Amygdalin Inhibits TGFβ1-Induced Activation of Hepatic Stellate Cells (HSCs) In Vitro and CCl4-Induced Hepatic Fibrosis in Rats In Vivo. Int. Immunopharmacol 90, 107151. 10.1016/j.intimp.2020.107151 [DOI] [PubMed] [Google Scholar]
- Wang S., Xu J., Wang C., Li J., Wang Q., Kuang H., et al. (2020c). Paeoniae Radix alba Polysaccharides Obtained via Optimized Extraction Treat Experimental Autoimmune Hepatitis Effectively. Int. J. Biol. Macromol 164, 1554–1564. 10.1016/j.ijbiomac.2020.07.214 [DOI] [PubMed] [Google Scholar]
- Wang Y., Wang R., Wang Y., Peng R., Wu Y., Yuan Y. (2015). Ginkgo Biloba Extract Mitigates Liver Fibrosis and Apoptosis by Regulating P38 MAPK, NF-Κb/iκbα, and Bcl-2/Bax Signaling. Drug Des. Devel Ther. 9, 6303–6317. 10.2147/DDDT.S93732 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y. X., Du Y., Liu X. F., Yang F. X., Wu X., Tan L., et al. (2019b). A Hepatoprotection Study of Radix Bupleuri on Acetaminophen-Induced Liver Injury Based on CYP450 Inhibition. Chin. J. Nat. Med. 17 (7), 517–524. 10.1016/S1875-5364(19)30073-1 [DOI] [PubMed] [Google Scholar]
- Wei J., Zhang L., Liu J., Pei D., Wang N., Wang H., et al. (2020). Protective Effect of Lycium Barbarum Polysaccharide on Ethanol-Induced Injury in Human Hepatocyte and its Mechanism. J. Food Biochem., e13412. 10.1111/jfbc.13412 [DOI] [PubMed] [Google Scholar]
- Wei L., Dai Y., Zhou Y., He Z., Yao J., Zhao L., et al. (2017). Oroxylin A Activates PKM1/HNF4 Alpha to Induce Hepatoma Differentiation and Block Cancer Progression. Cell Death Dis 8 (7), e2944. 10.1038/cddis.2017.335 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Wei R., Cao J., Yao S. (2018). Matrine Promotes Liver Cancer Cell Apoptosis by Inhibiting Mitophagy and PINK1/Parkin Pathways. Cell Stress Chaperones 23 (6), 1295–1309. 10.1007/s12192-018-0937-7 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Wu C., Chen W., Ding H., Li D., Wen G., Zhang C., et al. (2019b). Salvianolic Acid B Exerts Anti-liver Fibrosis Effects via Inhibition of MAPK-Mediated Phospho-Smad2/3 at Linker Regions In Vivo and In Vitro. Life Sci. 239, 116881. 10.1016/j.lfs.2019.116881 [DOI] [PubMed] [Google Scholar]
- Wu C., Jing M., Yang L., Jin L., Ding Y., Lu J., et al. (2018a). Alisol A 24-acetate Ameliorates Nonalcoholic Steatohepatitis by Inhibiting Oxidative Stress and Stimulating Autophagy through the AMPK/mTOR Pathway. Chem. Biol. Interact 291, 111–119. 10.1016/j.cbi.2018.06.005 [DOI] [PubMed] [Google Scholar]
- Wu C. T., Deng J. S., Huang W. C., Shieh P. C., Chung M. I., Huang G. J. (2019a). Salvianolic Acid C Against Acetaminophen-Induced Acute Liver Injury by Attenuating Inflammation, Oxidative Stress, and Apoptosis through Inhibition of the Keap1/Nrf2/HO-1 Signaling. Oxid Med. Cel Longev 2019, 9056845. 10.1155/2019/9056845 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu K., Fan J., Huang X., Wu X., Guo C. (2018b). Hepatoprotective Effects Exerted by Poria Cocos Polysaccharides Against Acetaminophen-Induced Liver Injury in Mice. Int. J. Biol. Macromol 114, 137–142. 10.1016/j.ijbiomac.2018.03.107 [DOI] [PubMed] [Google Scholar]
- Wu K., Guo C., Yang B., Wu X., Wang W. (2019c). Antihepatotoxic Benefits of Poria Cocos Polysaccharides on Acetaminophen-Lesioned Livers In Vivo and In Vitro. J. Cel Biochem 120 (5), 7482–7488. 10.1002/jcb.28022 [DOI] [PubMed] [Google Scholar]
- Wu Q. (2001). Traditional Chinese Medicine for Liver Disease. Beijing: China medical science and technology press. [Google Scholar]
- Wu X., Zhang F., Xiong X., Lu C., Lian N., Lu Y., et al. (2015). Tetramethylpyrazine Reduces Inflammation in Liver Fibrosis and Inhibits Inflammatory Cytokine Expression in Hepatic Stellate Cells by Modulating NLRP3 Inflammasome Pathway. IUBMB Life 67 (4), 312–321. 10.1002/iub.1348 [DOI] [PubMed] [Google Scholar]
- Wu X., Zhi F., Lun W., Deng Q., Zhang W. (2018c). Baicalin Inhibits PDGF-BB-Induced Hepatic Stellate Cell Proliferation, Apoptosis, Invasion, Migration and Activation via the miR-3595/ACSL4 axis. Int. J. Mol. Med. 41 (4), 1992–2002. 10.3892/ijmm.2018.3427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Z., Meng X., Hu J., Ding Y., Peng Y. (2017). Research Progress on the Relationship between TLR4-MyD88-NF-kB Signaling Pathway and Hepatitis Liver Fibrosis Liver Cancer axis. Int. J. Pharm. Res. 44 (05), 396–401. [Google Scholar]
- Xian Z., Tian J., Wang L., Zhang Y., Han J., Deng N., et al. (2020). Effects of Rhein on Bile Acid Homeostasis in Rats. Biomed. Res. Int. 2020, 8827955. 10.1155/2020/8827955 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao Y., Kim M., Lazar M. A. (2020). Nuclear Receptors and Transcriptional Regulation in Non-alcoholic Fatty Liver Disease. Mol. Metab. 50, 101119. 10.1016/j.molmet.2020.101119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie H., Su D., Zhang J., Ji D., Mao J., Hao M., et al. (2020). Raw and Vinegar Processed Curcuma Wenyujin Regulates Hepatic Fibrosis via Bloking TGF-β/Smad Signaling Pathways and Up-Regulation of MMP-2/TIMP-1 Ratio. J. Ethnopharmacol 246, 111768. 10.1016/j.jep.2019.01.045 [DOI] [PubMed] [Google Scholar]
- Xie T., Li K., Gong X., Jiang R., Huang W., Chen X., et al. (2018). Paeoniflorin Protects Against Liver Ischemia/reperfusion Injury in Mice via Inhibiting HMGB1-TLR4 Signaling Pathway. Phytother Res. 32 (11), 2247–2255. 10.1002/ptr.6161 [DOI] [PubMed] [Google Scholar]
- Xu F., Liu C., Zhou D., Zhang L. (2016). TGF-β/SMAD Pathway and its Regulation in Hepatic Fibrosis. J. Histochem. Cytochem. 64 (3), 157–167. 10.1369/0022155415627681 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J., Li C., Li Z., Yang C., Lei L., Ren W., et al. (2018). Protective Effects of Oxymatrine Against lipopolysaccharide/D-galactosamine-induced A-cute L-iver F-ailure through O-xidative D-amage, via A-ctivation of Nrf2/HO-1 and M-odulation of I-nflammatory TLR4-signaling P-athways. Mol. Med. Rep. 17 (1), 1907–1912. 10.3892/mmr.2017.8060 [DOI] [PubMed] [Google Scholar]
- Yan H., Gao Y. Q., Zhang Y., Wang H., Liu G. S., Lei J. Y. (2018). Chlorogenic Acid Alleviates Autophagy and Insulin Resistance by Suppressing JNK Pathway in a Rat Model of Nonalcoholic Fatty Liver Disease. J. Biosci. 43 (2), 287–294. 10.1007/s12038-018-9746-5 [DOI] [PubMed] [Google Scholar]
- Yan H., Jung K. H., Kim J., Rumman M., Oh M. S., Hong S. S. (2018). Artemisia Capillaris Extract AC68 Induces Apoptosis of Hepatocellular Carcinoma by Blocking the PI3K/AKT Pathway. Biomed. Pharmacother. 98, 134–141. 10.1016/j.biopha.2017.12.043 [DOI] [PubMed] [Google Scholar]
- Yan Z., Fan R., Yin S., Zhao X., Liu J., Li L., et al. (2015). Protective Effects of Ginkgo Biloba Leaf Polysaccharide on Nonalcoholic Fatty Liver Disease and its Mechanisms. Int. J. Biol. Macromol 80, 573–580. 10.1016/j.ijbiomac.2015.05.054 [DOI] [PubMed] [Google Scholar]
- Yang F., Luo L., Zhu Z. D., Zhou X., Wang Y., Xue J., et al. (2017). Chlorogenic Acid Inhibits Liver Fibrosis by Blocking the miR-21-Regulated TGF-β1/Smad7 Signaling Pathway In Vitro and In Vivo. Front. Pharmacol. 8, 929. 10.3389/fphar.2017.00929 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang F., Xu Y., Xiong A., He Y., Yang L., Wan Y. J., et al. (2012). Evaluation of the Protective Effect of Rhei Radix et Rhizoma Against α-naphthylisothiocyanate Induced Liver Injury Based on Metabolic Profile of Bile Acids. J. Ethnopharmacol 144 (3), 599–604. 10.1016/j.jep.2012.09.049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang G., Wei W. (2017). Research Progress on Immune Mechanism of Alcoholic Liver Disease and Prevention and Treatment of Traditional Chinese Medicine. J. southwest Med. Univ. 40 (03), 319–321. [Google Scholar]
- Yang H., Yang T., Heng C., Zhou Y., Jiang Z., Qian X., et al. (2019a). Quercetin Improves Nonalcoholic Fatty Liver by Ameliorating Inflammation, Oxidative Stress, and Lipid Metabolism in Db/db Mice. Phytother Res. 33 (12), 3140–3152. 10.1002/ptr.6486 [DOI] [PubMed] [Google Scholar]
- Yang H., Zhou Z., He L., Ma H., Qu W., Yin J., et al. (2018). Hepatoprotective and Inhibiting HBV Effects of Polysaccharides from Roots of Sophora Flavescens. Int. J. Biol. Macromol 108, 744–752. 10.1016/j.ijbiomac.2017.10.171 [DOI] [PubMed] [Google Scholar]
- Yang H. X., Shang Y., Jin Q., Wu Y. L., Liu J., Qiao C. Y., et al. (2020a). Gentiopicroside Ameliorates the Progression from Hepatic Steatosis to Fibrosis Induced by Chronic Alcohol Intake. Biomol. Ther. (Seoul) 28 (4), 320–327. 10.4062/biomolther.2020.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J. H., Kim S. C., Kim K. M., Jang C. H., Cho S. S., Kim S. J., et al. (2016a). Isorhamnetin Attenuates Liver Fibrosis by Inhibiting TGF-β/Smad Signaling and Relieving Oxidative Stress. Eur. J. Pharmacol. 783, 92–102. 10.1016/j.ejphar.2016.04.042 [DOI] [PubMed] [Google Scholar]
- Yang M., Li X., Zeng X., Ou Z., Xue M., Gao D., et al. (2016b). Rheum Palmatum L. Attenuates High Fat Diet-Induced Hepatosteatosis by Activating AMP-Activated Protein Kinase. Am. J. Chin. Med. 44 (3), 551–564. 10.1142/S0192415X16500300 [DOI] [PubMed] [Google Scholar]
- Yang R., Song C., Chen J., Zhou L., Jiang X., Cao X., et al. (2020b). Limonin Ameliorates Acetaminophen-Induced Hepatotoxicity by Activating Nrf2 Antioxidative Pathway and Inhibiting NF-Κb Inflammatory Response via Upregulating Sirt1. Phytomedicine 69, 153211. 10.1016/j.phymed.2020.153211 [DOI] [PubMed] [Google Scholar]
- Yang Y., Zhao J., Song X., Li L., Li F., Shang J., et al. (2019b). Amygdalin Reduces Lipopolysaccharide-Induced Chronic Liver Injury in Rats by Down-Regulating PI3K/AKT, JAK2/STAT3 and NF-Κb Signalling Pathways. Artif. Cell Nanomed Biotechnol 47 (1), 2688–2697. 10.1080/21691401.2019.1634084 [DOI] [PubMed] [Google Scholar]
- Yari Z., Cheraghpour M., Alavian S. M., Hedayati M., Eini-Zinab H., Hekmatdoost A. (2021). The Efficacy of Flaxseed and Hesperidin on Non-alcoholic Fatty Liver Disease: An Open-Labeled Randomized Controlled Trial. Eur. J. Clin. Nutr. 75 (1), 99–111. 10.1038/s41430-020-0679-3 [DOI] [PubMed] [Google Scholar]
- Yi Y., Zhao Y., Li C., Zhang Y., Bin Y., Yuan Y., et al. (2018). Potential Chronic Liver Toxicity in Rats Orally Administered an Ethanol Extract of Huangqin (Radix Scutellariae Baicalensis). J. Tradit Chin. Med. 38 (2), 242–256. [PubMed] [Google Scholar]
- Yim D., Kim M. J., Shin Y., Lee S. J., Shin J. G., Kim D. H. (2019). Inhibition of Cytochrome P450 Activities by Sophora Flavescens Extract and its Prenylated Flavonoids in Human Liver Microsomes. Evid. Based Complement. Alternat Med. 2019, 2673769. 10.1155/2019/2673769 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yokomori H., Oda M., Yoshimura K., Hibi T. (2012). Recent Advances in Liver Sinusoidal Endothelial Ultrastructure and Fine Structure Immunocytochemistry. Micron 43 (2-3), 129–134. 10.1016/j.micron.2011.08.002 [DOI] [PubMed] [Google Scholar]
- Younossi Z. M. (2019). Non-alcoholic Fatty Liver Disease - A Global Public Health Perspective. J. Hepatol. 70 (3), 531–544. 10.1016/j.jhep.2018.10.033 [DOI] [PubMed] [Google Scholar]
- Yu Q., Liu T., Li S., Feng J., Wu L., Wang W., et al. (20182018). The Protective Effects of Levo-Tetrahydropalmatine on ConA-Induced Liver Injury Are via TRAF6/JNK Signaling. Mediators Inflamm. 2018, 4032484. 10.1155/2018/4032484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu Q., Cheng P., Wu J., Guo C. (2021). Pparγ/NF-Κb and TGF-β1/Smad Pathway Are Involved in the Anti-fibrotic Effects of Levo-Tetrahydropalmatine on Liver Fibrosis. J. Cel Mol Med 25 (3), 1645–1660. 10.1111/jcmm.16267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan F., Chen J., Wu W. J., Chen S. Z., Wang X. D., Su Z., et al. (2010). Effects of Matrine and Oxymatrine on Catalytic Activity of Cytochrome P450s in Rats. Basic Clin. Pharmacol. Toxicol. 107 (5), 906–913. 10.1111/j.1742-7843.2010.00596.x [DOI] [PubMed] [Google Scholar]
- Yuan R., Tao X., Liang S., Pan Y., He L., Sun J., et al. (2018). Protective Effect of Acidic Polysaccharide from Schisandra Chinensis on Acute Ethanol-Induced Liver Injury through Reducing CYP2E1-dependent Oxidative Stress. Biomed. Pharmacother. 99, 537–542. 10.1016/j.biopha.2018.01.079 [DOI] [PubMed] [Google Scholar]
- Yuen M. F., Chen D. S., Dusheiko G. M., Janssen H. L. A., Lau D. T. Y., Locarnini S. A., et al. (2018). Hepatitis B Virus Infection. Nat. Rev. Dis. Primers 4, 18035. 10.1038/nrdp.2018.35 [DOI] [PubMed] [Google Scholar]
- Yun Y. R., Kim J. H., Kim J. H., Jung M. H. (2017). Protective Effects of Gomisin N Against Hepatic Steatosis Through AMPK Activation. Biochem. Biophys. Res. Commun. 482 (4), 1095–1101. 10.1016/j.bbrc.2016.11.164 [DOI] [PubMed] [Google Scholar]
- Zang W., Bian H., Huang X., Yin G., Zhang C., Han L. I., et al. (2019). Traditional Chinese Medicine (TCM) Astragalus Membranaceus and Curcuma Wenyujin Promote Vascular Normalization in Tumor-Derived Endothelial Cells of Human Hepatocellular Carcinoma. Anticancer Res. 39 (6), 2739–2747. 10.21873/anticanres.13400 [DOI] [PubMed] [Google Scholar]
- Zeng H., Li D., Qin X., Chen P., Tan H., Zeng X., et al. (2016). Hepatoprotective Effects of Schisandra Sphenanthera Extract Against Lithocholic Acid-Induced Cholestasis in Male Mice Are Associated with Activation of the Pregnane X Receptor Pathway and Promotion of Liver Regeneration. Drug Metab. Dispos 44 (3), 337–342. 10.1124/dmd.115.066969 [DOI] [PubMed] [Google Scholar]
- Zeng X., Li X., Xu C., Jiang F., Mo Y., Fan X., et al. (2017). Schisandra Sphenanthera Extract (Wuzhi Tablet) Protects Against Chronic-Binge and Acute Alcohol-Induced Liver Injury by Regulating the NRF2-ARE Pathway in Mice. Acta Pharm. Sin B 7 (5), 583–592. 10.1016/j.apsb.2017.04.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang C. Y., Jiang Z. M., Ma X. F., Li Y., Liu X. Z., Li L. L., et al. (2019). Saikosaponin-d Inhibits the Hepatoma Cells and Enhances Chemosensitivity through SENP5-dependent Inhibition of Gli1 SUMOylation Under Hypoxia. Front. Pharmacol. 10, 1039. 10.3389/fphar.2019.01039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H., Yang L., Wang Y., Huang W., Li Y., Chen S., et al. (2020a). Oxymatrine Alleviated Hepatic Lipid Metabolism via Regulating miR-182 in Non-alcoholic Fatty Liver Disease. Life Sci. 257, 118090. 10.1016/j.lfs.2020.118090 [DOI] [PubMed] [Google Scholar]
- Zhang Y., Wang H., Zhang L., Yuan Y., Yu D. (2020b). Codonopsis Lanceolata Polysaccharide CLPS Alleviates High Fat/high Sucrose Diet-Induced Insulin Resistance via Anti-oxidative Stress. Int. J. Biol. Macromol 145, 944–949. 10.1016/j.ijbiomac.2019.09.185 [DOI] [PubMed] [Google Scholar]
- Zhang Y., Yang X., Wang S., Song S., Yang X. (2021). Gentiopicroside Prevents Alcoholic Liver Damage by Improving Mitochondrial Dysfunction in the Rat Model. Phytother Res. 35 (4), 2230–2251. 10.1002/ptr.6981 [DOI] [PubMed] [Google Scholar]
- Zhang Y., Zhao H., Li H., Cao W., Wang F., Zhang T., et al. (2017). Protective Effects of Amarogentin Against Carbon Tetrachloride-Induced Liver Fibrosis in Mice. Molecules 22 (5). 10.3390/molecules22050754 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Z., Chen S., Mei H., Xuan J., Guo X., Couch L., et al. (2015). Ginkgo Biloba Leaf Extract Induces DNA Damage by Inhibiting Topoisomerase II Activity in Human Hepatic Cells. Sci. Rep. 5, 14633. 10.1038/srep14633 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao H., Zhang Y., Shu L., Song G., Ma H. (2019a). Resveratrol Reduces Liver Endoplasmic Reticulum Stress and Improves Insulin Sensitivity In Vivo and In Vitro. Drug Des. Devel Ther. 13, 1473–1485. 10.2147/DDDT.S203833 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao H. W., Zhang Z. F., Chai X., Li G. Q., Cui H. R., Wang H. B., et al. (2016). Oxymatrine Attenuates CCl4-Induced Hepatic Fibrosis via Modulation of TLR4-dependent Inflammatory and TGF-Β1 Signaling Pathways. Int. Immunopharmacol 36, 249–255. 10.1016/j.intimp.2016.04.040 [DOI] [PubMed] [Google Scholar]
- Zhao P., Piao X., Pan L., Zeng Z., Li Q., Xu X., et al. (2017). Forsythia Suspensa Extract Attenuates Lipopolysaccharide-Induced Inflammatory Liver Injury in Rats via Promoting Antioxidant Defense Mechanisms. Anim. Sci. J. 88 (6), 873–881. 10.1111/asj.12717 [DOI] [PubMed] [Google Scholar]
- Zhao Q., Wei M., Zhang S., Huang Z., Lu B., Ji L. (2020). The Water Extract of Sophorae tonkinensis Radix et Rhizoma Alleviates Non-alcoholic Fatty Liver Disease and Its Mechanism. Phytomedicine 77, 153270. 10.1016/j.phymed.2020.153270 [DOI] [PubMed] [Google Scholar]
- Zhao X. J., Chen L., Zhao Y., Pan Y., Yang Y. Z., Sun Y., et al. (2019b). Polygonum Cuspidatum Extract Attenuates Fructose-Induced Liver Lipid Accumulation through Inhibiting Keap1 and Activating Nrf2 Antioxidant Pathway. Phytomedicine 63, 152986. 10.1016/j.phymed.2019.152986 [DOI] [PubMed] [Google Scholar]
- Zhao X. J., Yu H. W., Yang Y. Z., Wu W. Y., Chen T. Y., Jia K. K., et al. (2018a). Polydatin Prevents Fructose-Induced Liver Inflammation and Lipid Deposition through Increasing miR-200a to Regulate Keap1/Nrf2 Pathway. Redox Biol. 18, 124–137. 10.1016/j.redox.2018.07.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Y., Ma X., Wang J., Zhu Y., Li R., Wang J., et al. (2014). Paeoniflorin Alleviates Liver Fibrosis by Inhibiting HIF-1α Through mTOR-dependent Pathway. Fitoterapia 99, 318–327. 10.1016/j.fitote.2014.10.009 [DOI] [PubMed] [Google Scholar]
- Zhao Z., Wei Q., Hua W., Liu Y., Liu X., Zhu Y. (2018b). Hepatoprotective Effects of Berberine on Acetaminophen-Induced Hepatotoxicity in Mice. Biomed. Pharmacother. 103, 1319–1326. 10.1016/j.biopha.2018.04.175 [DOI] [PubMed] [Google Scholar]
- Zheng N., Liu F., Lu H., Zhan Y., Zhang M., Guo W., et al. (2017). Schisantherin A Protects Against Liver Ischemia-Reperfusion Injury via Inhibition of Mitogen-Activated Protein Kinase Pathway. Int. Immunopharmacol 47, 28–37. 10.1016/j.intimp.2017.03.019 [DOI] [PubMed] [Google Scholar]
- Zhong W., Qian K., Xiong J., Ma K., Wang A., Zou Y. (2016). Curcumin Alleviates Lipopolysaccharide Induced Sepsis and Liver Failure by Suppression of Oxidative Stress-Related Inflammation via PI3K/AKT and NF-Κb Related Signaling. Biomed. Pharmacother. 83, 302–313. 10.1016/j.biopha.2016.06.036 [DOI] [PubMed] [Google Scholar]
- Zhou L., Yang F., Li G., Huang J., Liu Y., Zhang Q., et al. (2018). Coptisine Induces Apoptosis in Human Hepatoma Cells Through Activating 67-kDa Laminin Receptor/cGMP Signaling. Front. Pharmacol. 9, 517. 10.3389/fphar.2018.00517 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou W. C., Zhang Q. B., Qiao L. (2014). Pathogenesis of Liver Cirrhosis. World J. Gastroenterol. 20 (23), 7312–7324. 10.3748/wjg.v20.i23.7312 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou X., Cheung C. M., Yang J. M., Or P. M., Lee W. Y., Yeung J. H. (2015). Danshen (Salvia Miltiorrhiza) Water Extract Inhibits Paracetamol-Induced Toxicity in Primary Rat Hepatocytes via Reducing CYP2E1 Activity and Oxidative Stress. J. Pharm. Pharmacol. 67 (7), 980–989. 10.1111/jphp.12381 [DOI] [PubMed] [Google Scholar]
- Zhou X., Wang L. L., Tang W. J., Tang B. (2021). Astragaloside IV Inhibits Protein Tyrosine Phosphatase 1B and Improves Insulin Resistance in Insulin-Resistant HepG2 Cells and Triglyceride Accumulation in Oleic Acid (OA)-treated HepG2 Cells. J. Ethnopharmacol 268, 113556. 10.1016/j.jep.2020.113556 [DOI] [PubMed] [Google Scholar]
- Zhu H., He C., Zhao H., Jiang W., Xu S., Li J., et al. (2020). Sennoside A Prevents Liver Fibrosis by Binding DNMT1 and Suppressing DNMT1-Mediated PTEN Hypermethylation in HSC Activation and Proliferation. FASEB J. 34 (11), 14558–14571. 10.1096/fj.202000494RR [DOI] [PubMed] [Google Scholar]
- Zhu S. Y., Jiang N., Yang J., Tu J., Zhou Y., Xiao X., et al. (2018a). Silybum marianum Oil Attenuates Hepatic Steatosis and Oxidative Stress in High Fat Diet-Fed Mice. Biomed. Pharmacother. 100, 191–197. 10.1016/j.biopha.2018.01.144 [DOI] [PubMed] [Google Scholar]
- Zhu X., Xiong T., Liu P., Guo X., Xiao L., Zhou F., et al. (2018b). Quercetin Ameliorates HFD-Induced NAFLD by Promoting Hepatic VLDL Assembly and Lipophagy via the IRE1a/XBP1s Pathway. Food Chem. Toxicol. 114, 52–60. 10.1016/j.fct.2018.02.019 [DOI] [PubMed] [Google Scholar]
- Zou C., Su N., Wu J., Xu M., Sun Z., Liu Q., et al. (2019). Dietary Radix Bupleuri Extracts Improves Hepatic Lipid Accumulation and Immune Response of Hybrid Grouper (Epinephelus Lanceolatus♂ × Epinephelus Fuscoguttatus♀). Fish. Shellfish Immunol. 88, 496–507. 10.1016/j.fsi.2019.02.052 [DOI] [PubMed] [Google Scholar]
- Zou C., Tan X., Ye H., Sun Z., Chen S., Liu Q., et al. (2018). The Hepatoprotective Effects of Radix Bupleuri Extracts Against D-Galactosamine/lipopolysaccharide Induced Liver Injury in Hybrid Grouper (Epinephelus Lanceolatus♂ × Epinephelus Fuscoguttatus♀). Fish. Shellfish Immunol. 83, 8–17. 10.1016/j.fsi.2018.08.047 [DOI] [PubMed] [Google Scholar]