Introduction
Pregnancy is a mysterious biological process that presents great challenges to the maternal immune system. In the early 1950s, the “fetal allograft” concept was described for the first time by Peter Medawar, and the unique immunology of the maternal-fetal interface was recognized [1]. Correct and precise interaction between mother and fetus plays an important role during pregnancy process, such as the apposition, adhesion, implantation, and growth of embryo in uterus [2]. In 1991, Colbern and Main proposed that the maternal immune cells directly interact with placenta but not the fetus [3]. Therefore, information concerning the cross-talk between maternal immune cells and placenta during normal pregnancy will provide clues to explore the underlying mechanism of pathological pregnancy. Immune cells, such as natural killer (NK), macrophage, T, and dendritic cells, have been demonstrated to play important roles during normal pregnancy [4]. With the development of single-cell RNA sequencing (scRNA-seq) technologies, researchers are devoted to providing a whole picture about the immune cellular composition and inter-cellular communication events during normal pregnancy [5], [6]. These foundational studies reveal that immune cell subsets, which are classified based on different markers at high resolution, exert specific function during pregnancy establishment. However, the panoramic analysis of immune subsets at high resolution in pathological pregnancy remains lacking.
To reveal the etiology of recurrent miscarriage (RM), also known as recurrent pregnancy loss (RPL), a complex pathological pregnancy affecting about 5% of women of child-bearing age, recently two independent studies have profiled the decidual and peripheral blood immune cells in RM/RPL patients using scRNA-seq and published their data in this journal and Cell Discovery, separately [7], [8]. Both of these two studies find that immune cells in RM/RPL patients preferentially exhibit pro-inflammatory status in decidua or peripheral blood.
As decidual NK (dNK) cells constitute the largest population of decidual leukocytes during first-trimester pregnancy, alternations in the proportion and function of dNK cells in RM/RPL have been explored in depth in both studies. Consistent with previous study [6], they have identified three main dNK subsets (dNK1, dNK2, and dNK3) in normal pregnancy. dNK1 cells may contribute to the secretion of growth-promoting factors (GPFs) through expressing high levels of genes encoding inhibitory receptors, such as KIR2DL1, KIR2DL3, and LILRB1. Both dNK2 and dNK3 cells are prone to exhibiting cytotoxic or cytokine-secreting properties. More importantly, they firstly report that the proportion of dNK1 cells is significantly decreased, and conversely, that of dNK3 cells is significantly increased in RM/RPL patients. In addition to the alternation in proportion, the expression of LILRB1 in dNK1 cells is decreased, whereas the production of pro-inflammatory cytokines in dNK3 cells is enhanced. Collectively, the alternations in proportion and function of dNK1 and dNK3 cells indicate their diminished immune-protective capability, which provides important clue to explore the underlying mechanism of RM/RPL.
Macrophages are the second largest class of leukocytes at the maternal-fetal interface. Both these two studies reveal that the proportion of decidual macrophage (dM) cells is significantly decreased. In contrast, the expression of genes encoding pro-inflammatory factors including CXCL8, TNF, and IFIT2 is increased in RM/RPL patients. The only difference is that Guo et al. find that an increase in the proportion of inflammatory macrophage subset dM1 cells is accompanied by a substantial decrease in the number of regulatory macrophage subset dM2 cells [8] while Wang et al. do not find an alternation in the proportion of dM1 and dM2 cells in RM/RPL patients [7]. This is probably due to the relatively small amount of captured dM cells for sequencing and analysis. Therefore, studies with larger sample size should be further developed.
Cell–cell interactions, including between immune cells and immune cells, immune cells and stromal cells, as well as immune cells and extravillous trophoblast (EVT) cells, have been further evaluated in these two studies. They find that ligand–receptor interactions between the decidual leukocyte subsets are preferentially immune activation in RM/RPL patients. Through integrating their scRNA-seq data from decidual tissue of normal and RM/RPL pregnancies with the scRNA-seq data from healthy EVTs and stromal cells reported previously [6], Guo et al. found that the interactions of immune cell subsets with EVTs and stromal cells are impaired in RM/RPL patients [8]. Taken together, these data indicate that cell–cell interactions at the maternal-fetal interface are in a pro-inflammatory state in RM/RPL patients.
In addition to decidual immune cells, Wang et al. performed a panoramic analysis on peripheral immune cells as well [7]. Consequently, they have identified five peripheral T cell subsets, i.e., CD4+ naïve T, CD8+ naïve T, CD4+ memory T, CD8+ effector T, and mucosal-associated invariant T (MAIT) cells. Moreover, they have observed decreases in the proportion of CD4+ naïve T, CD8+ naïve T, and CD4+ memory T cells, but increases in the proportions of CD8+ effector T and MAIT cells in RM/RPL patients. Importantly, this is first time to report that MAIT activation may be associated with the maternal inflammation condition. Meanwhile, sequencing data analysis further shows an increase in the expression of genes encoding inflammatory cytokines in CD8+ effector T and MAIT cells, indicating their highly immune-activated property in RM/RPL patients. The markedly increased proportion of peripheral CD56dimCD16+ NK cells is also noticed in RM/RPL patients. Furthermore, the expression of pro-inflammatory genes is increased, whereas expression of immunosuppressive genes is decreased in RM/RPL patients. Taken together, these alterations strongly indicate a systematic pro-inflammatory property in RM/RPL patients. Compared to decidua, peripheral blood could be easily collected at multiple time points, such as pre-conception and different gestational weeks. Therefore, the high-resolution data of peripheral immune cells may pave the way for the pre-symptomatic diagnosis of abnormal pregnancy. It is worth noting that immune changes in any organ may affect the immune status of peripheral blood. Therefore, whether and to what extent the immune status of peripheral blood can reflect the immune status of the maternal-fetal interface needs to be carefully verified and interpreted.
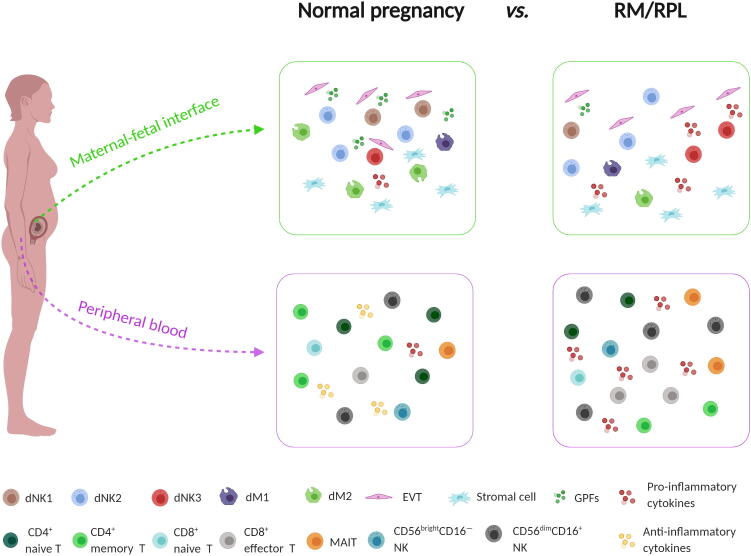
In conclusion, the overactivation of immune status at the maternal-fetal interface caused by the increase in pro-inflammatory immune cell subsets and the decrease in anti-inflammatory immune cell subsets may have deleterious effects on fetal survival (Figure 1). The findings in these two studies provide a panoramic atlas of immune status and generate a data-driven hypothesis about the underlying mechanisms for RM/RPL. The detailed information about immune cell subsets may help explain some contradictive observations based on current low-resolution data. However, some questions remain to be answered in further studies. First, functional studies using appropriate in vitro or in vivo models are warranted to further uncover the immune mechanisms underlying RM/RPL. Second, prognostic value of the alterations in immune landscape in predicting subsequent pregnancy outcomes in RM/RPL patients also needs to be investigated.
Figure 1.
Overactivation of immune status at maternal-fetal interface in RM/RPL patients
The proportion of pro-inflammatory immune cell subsets (e.g., dNK3, dM1, peripheral CD8+ effector T, MAIT, and CD56dimCD16+ NK cells) is increased, whereas the proportion of anti-inflammatory immune cell subsets (e.g., dNK1, dNK2, dM2, peripheral CD4+ naïve T, CD8+ naïve T, CD4+ memory T, and CD56brightCD16− NK cells) is decreased in decidua and peripheral blood, together leading to overactivation of immune status at the maternal-fetal interface in RM/RPL patients. dM, decidual macrophage; dNK, decidual NK; EVT, extravillous trophoblast; GPF, growth-promoting factor; MAIT, mucosal-associated invariant T; RM, recurrent miscarriage; RPL, recurrent pregnancy loss.
CRediT author statement
Chunyu Huang: Investigation, Writing - original draft, Visualization. Yong Zeng: Conceptualization. Wenwei Tu: Supervision, Conceptualization, Writing - review & editing. All authors read and approved the final manuscript.
Competing interests
The authors have declared no competing interests.
Acknowledgments
The present study was supported in part by the Shenzhen Healthcare Research Project (Grant No. SZXJ2018004), Clinical Research Fund of Chinese Medical Association (Grant No. 18010110740), National Key R&D Program of China (Grant Nos. 2018YFC1003900 and 2018YFC1003904), Sanming Project of Medicine in Shenzhen (Grant No. SZSM201502035), and General Research Fund, Research Grants Council of Hong Kong (Grant Nos. 17122519, 17126317, 17115015, and 17121214), China. The figure was created with BioRender.com.
Handled by Fuchou Tang
Footnotes
Peer review under responsibility of Beijing Institute of Genomics, Chinese Academy of Sciences / China National Center for Bioinformation and Genetics Society of China.
References
- 1.Billington W.D. The immunological problem of pregnancy: 50 years with the hope of progress. A tribute to Peter Medawar. J Reprod Immunol. 2003;60:1–11. doi: 10.1016/s0165-0378(03)00083-4. [DOI] [PubMed] [Google Scholar]
- 2.PrabhuDas M., Bonney E., Caron K., Dey S., Erlebacher A., Fazleabas A., et al. Immune mechanisms at the maternal-fetal interface: perspectives and challenges. Nat Immunol. 2015;16:328–334. doi: 10.1038/ni.3131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Colbern G.T., Main E.K. Immunology of the maternal-placental interface in normal pregnancy. Semin Perinatol. 1991;15:196–205. [PubMed] [Google Scholar]
- 4.Orefice R. Immunology and the immunological response to pregnancy. Best Pract Res Clin Obstet Gynaecol. 2021;76:3–12. doi: 10.1016/j.bpobgyn.2020.07.013. [DOI] [PubMed] [Google Scholar]
- 5.Suryawanshi H., Morozov P., Straus A., Sahasrabudhe N., Max K.E.A., Garzia A., et al. A single-cell survey of the human first-trimester placenta and decidua. Sci Adv. 2018;4:eaau4788. doi: 10.1126/sciadv.aau4788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Vento-Tormo R., Efremova M., Botting R.A., Turco M.Y., Vento-Tormo M., Meyer K.B., et al. Single-cell reconstruction of the early maternal-fetal interface in humans. Nature. 2018;563:347–353. doi: 10.1038/s41586-018-0698-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang F., Jia W., Fan M., Shao X., Li Z., Liu Y., et al. Single-cell immune landscape of human recurrent miscarriage. Genomics Proteomics Bioinformatics. 2021;19:208–222. doi: 10.1016/j.gpb.2020.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Guo C., Cai P., Jin L., Sha Q., Yu Q., Zhang W., et al. Single-cell profiling of the human decidual immune microenvironment in patients with recurrent pregnancy loss. Cell Discov. 2021;7:1. doi: 10.1038/s41421-020-00236-z. [DOI] [PMC free article] [PubMed] [Google Scholar]