Abstract
In children and adolescents, chronic low-grade inflammation has been implicated in the pathogenesis of co- and multi-morbid conditions to mental health disorders. Diet quality is a potential mechanism of action that can exacerbate or ameliorate low-grade inflammation; however, the exact way dietary intake can regulate the immune response in children and adolescents is still to be fully understood. In this review, I discuss the current observational and interventional evidence that supports a potential therapeutic role for dietary intake in the amelioration of low-grade inflammation and highlight the need to develop a better understanding of the biological mechanisms underlying and attenuating the associations between dietary intake and low-grade inflammation in children and adolescents.
Keywords: Dietary intake, Dietary pattern, Macronutrients, Biomarkers, Inflammation, CRP, Cytokine, Interleukin, Children, Adolescent
1. Introduction
Inflammation is a physiological response to cellular and tissue damage. It is designed to protect the host from bacteria, viruses and infections by eliminating pathogens, promoting cellular repair and restoring homeostatic conditions (Calder et al., 2011). However, a prolonged inflammatory state through chronic low-grade inflammation has deleterious effects, including irreparable damage to tissues and organs, and increased risk of disease status (Minihane et al., 2015).
Low-grade inflammation, reflected in the overproduction of acute-phase proteins such as C-reactive protein (CRP), pro-inflammatory cytokines such as interleukin-6 (IL-6) and tumour necrosis factor-alpha (TFN-α) has been established as a risk factor for several neuropsychiatric disorders (Giacobbe et al., 2020; Mitchell and Goldstein, 2014), including major depressive disorder (Colasanto, 2020), depression (Kiecolt-Glaser et al., 2015; Zunszain et al., 2012; Sawyer et al., 2019; Cattaneo et al., 2020; Pitharouli et al., 2021) and schizophrenia (Osimo et al., 2018). Moreover, low-grade inflammation in children and adolescents has been associated with the development of co- and multi-morbid conditions to mental health pathologies (Barbaresko et al., 2013; Furman et al., 2019; Stein et al., 2019; Bennett et al., 2018), including cardiovascular disease (Singh et al., 2015; Amaral et al., 2020), metabolic syndrome (Al-Hamad and Raman, 2017), type-II diabetes (Reinehr, 2019) and obesity (Stroescu et al., 2019), therefore making inflammation an important therapeutic target to study, especially for individuals suffering from those conditions.
The potential factors that promote low-grade chronic inflammation are diverse, and increasingly, attention has been given to diet quality as a potential mechanism of action that can exacerbate or ameliorate low-grade inflammation and subsequently influence mental health (Marx et al., 2020; Berk et al., 2013). Certainly, healthy dietary patterns of high quality, such as adherence to a Mediterranean Diet (Grosso et al., 2014), or eating foods such as vegetables and fruit (Calder et al., 2011), or macro/micronutrients, such as omega-3 polyunsaturated fatty acids (PUFAs) (Silveira et al., 2018) or vitamins C and E (Sun et al., 2011), respectively, have been shown to reduce systemic inflammation (Esposito et al., 2004; Giugliano et al., 2006). In contrast, the prevailing Western dietary pattern, which is high in refined grains, red meat, refined sugar and saturated fat, illicit elicits a pro-inflammatory response and increasing levels of circulating inflammatory biomarkers (Silveira et al., 2018).
Moreover, it is well established that a healthy diet in childhood and adolescence is crucial for optimal growth and development and for disease prevention (van der Velde et al., 2019). For example, higher vegetable intake in childhood has been associated with a lower risk of developing mental health pathologies later in life (Lassale et al., 2019), such as depression. In addition, a healthy diet can contribute to the prevention of cardio-metabolic multi-morbidities, often seen in adult patients with neuropsychiatric conditions. However, the efficacy of nutritional strategies for children and adolescents with mental health pathologies has been largely under-explored. While in observational and interventional studies, a higher quality, nutrient-rich diet, has been associated with a reduced risk of adverse mental health in both children (Kohlboeck et al., 2012) and adolescents (Jacka et al., 2010, 2011), to the best of the author's knowledge, there are presently no rigorous intervention studies explicitly designed to take a dietary approach to treatment in children and adolescent populations with existing adverse mental health. Further research studies are urgently needed.
Former literature reviews in children and adolescents have, for the most part, focused on various aspects of diet and various biomarkers that are not specifically related to the immune system function and response (Hilger-Kolb et al., 2017; Suhett et al., 2020; Rocha et al., 2017). In my recent review (Bujtor et al., 2021) I have presented several studies that bring together the current evidence base from observational and interventional studies investigating associations between dietary intake, through dietary patterns, food groups, macronutrients or micronutrients, and biological markers of low-grade inflammation, including CRP, IL-6 and TNF-α among others, in both children and adolescents. Taken together, these studies indicate a good quality diet, high in vegetable and fruit intake, whole grains, fibre and healthy fats ameliorates low-grade inflammation and therefore represents a promising therapeutic approach, as well as an important element for disease prevention in both children and adolescents. However, the mechanisms by which dietary patterns affect the inflammatory process are largely underexplored. In this review, I discuss the current evidence that supports a potential therapeutic role for dietary intake in the amelioration of low-grade inflammation and highlight the need to develop a better understanding of the biological mechanisms underlying and attenuating the associations between dietary intake and low-grade inflammation in children and adolescents.
2. Dietary intake and inflammation
2.1. Dietary patterns and indices
It is important to examine diet–disease relationships through dietary patterns, which are based on a countries dietary guidelines (Barbaresko et al., 2013), as foods are typically eaten in combination, and nutrients have both synergistic and antagonistic biochemical interactions (Tapsell et al., 2016). Dietary patterns capture the whole diet, including the combination of foods and nutrients (Moeller et al., 2007). The Mediterranean dietary pattern, examined in ten studies in this review (Del Mar Bibiloni et al., 2013; Liese et al., 2018; Agostinis-Sobrinho et al., 2019; Arouca et al., 2018; Carvalho et al., 2018; Douros et al., 2019; Karampola et al., 2019; Lazarou et al., 2010; Sureda et al., 2018; Çağiran Yilmaz et al., 2019) is characterized by a high intake of vegetables, fruit, whole grains, legumes, nuts, fish and low-fat dairy, alongside moderate to low consumption of meat and healthy fats (Dai et al., 2008). Results showed adequate adherence was associated with lower levels of CRP (Agostinis-Sobrinho et al., 2019; Sureda et al., 2018), IL-6, and TNF-α (Carvalho et al., 2018), and soluble vascular cell adhesion molecule-1 (sVCAM-1) (Arouca et al., 2018) in healthy males and females, and IL-17 (Douros et al., 2019) in both males and females with asthma. Whereas, the majority of studies conducted in cohorts with underlying pathologies, including type-1 diabetes (Liese et al., 2018), and obesity (Karampola et al., 2019; Lazarou et al., 2010; Çağiran Yilmaz et al., 2019), and one study in a healthy cohort (Del Mar Bibiloni et al., 2013) found no associations with inflammatory biomarkers. In interventional studies examining adherence to a low glycaemic index diet pattern (Rouhani et al., 2016) or to a hypocaloric high glycaemic index diet pattern (Rouhani et al., 2016; Iannuzzi et al., 2009; Parillo et al., 2012) adequate adherence was associated with lower levels of CRP, both in males and females with obesity, while another study found no association (Damsgaard et al., 2013). As well as in one intervention study in females with metabolic syndrome which examined the DASH diet and found adherence associated with lower levels of CRP, in female patients (Saneei et al., 2014), while a second observational study found no association (Liese et al., 2018) in neither males or females with type-1 diabetes. Taken together these results indicate that adequate adherence to healthful dietary patterns is associated with decreased levels of biomarkers, including CRP, IL-6 and TNF-α.
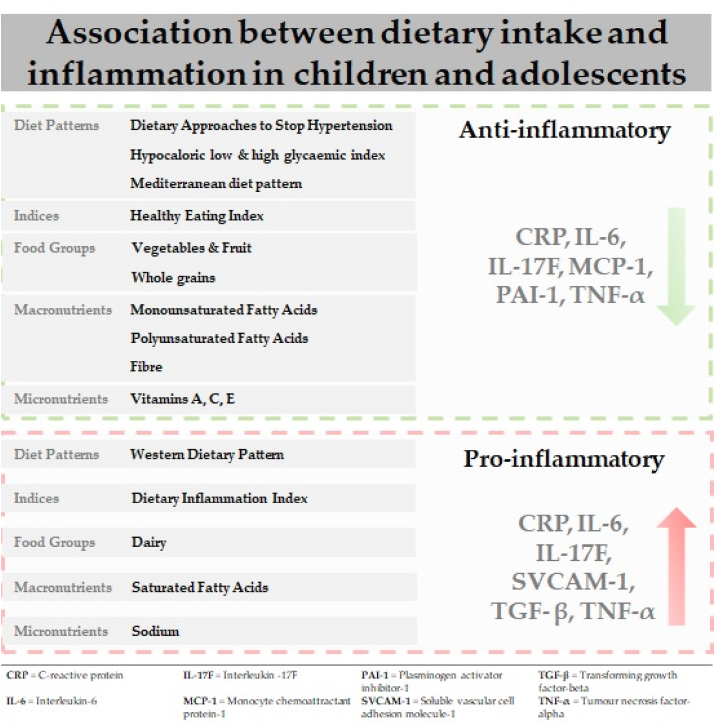
Dietary indices are nutritionally derived indices based on levels of consumption of nutrients or food groups (Bowyer et al., 2018). Of the five observational studies that examined healthful dietary indices, three observational studies focused on the Healthy Eating Index (HEI), a tool that assesses adherence to the Dietary Guidelines of Americans in any given year (Liese et al., 2018; Navarro et al., 2017; Sanjeevi et al., 2018). The first study found that a higher score in the HEI (healthy diet) was associated with lower levels of CRP in females, but not males from a healthy cohort (Navarro et al., 2017). In contrast, two studies found that moderate HEI scores (moderately healthy diet) were not associated with CRP or IL-6 in males or females in cohorts of patients with type-1 diabetes (Liese et al., 2018; Sanjeevi et al., 2018). The remaining two observational studies (Chan She Ping-Delfos et al., 2015; Vyncke et al., 2013) examined the Diet Quality Index (DQI), a composite individual-level diet quality indicator that enables cross-cultural diet quality comparisons. However, neither of the studies found an association between the DQI and CRP (Chan She Ping-Delfos et al., 2015; Vyncke et al., 2013) or IL-1, IL-6, IFN-y and TNF-α (Vyncke et al., 2013). This suggests a lack of sensitivity associated with the DQI as a measure of inflammatory status (Fig. 1).
Fig. 1.
Association between dietary intake and biomarkers of inflammation in children and adolescents (Bujtor et al., 2021).
In contrast to healthful dietary patterns, three studies examined the Western dietary pattern (Del Mar Bibiloni et al., 2013; Oddy et al., 2018; Khayyatzadeh et al., 2018). This pattern is characterised by high amounts of refined grains, red meat, high-fat dairy, ultra-processed food intake and trans fatty acids while being low in omega-3 PUFAs (Li et al., 2020; Okręglicka, 2015). Results showed positive associations with pro-inflammatory markers, specifically CRP and IL-6 in healthy males and females. The first study was longitudinal and found an association between WDP adherence at 14-years and higher levels of hs-CRP at 17-years both in healthy males and females (Oddy et al., 2018). The remaining two observational studies found an association between WDP adherence, and higher levels of CRP and IL-6 in this case in female cohorts of healthy participants (Del Mar Bibiloni et al., 2013; Khayyatzadeh et al., 2018). Similarly, across the six observational studies (Lazarou et al., 2010; Sen et al., 2018; Almeida-de-Souza et al., 2018; Coheley et al., 2019; Seremet Kurklu et al., 2020; Shivappa et al., 2017) that examined the Dietary Inflammatory Index (DII), a tool that assesses the inflammatory potential of a diet (Suhett et al., 2020), diets with high inflammatory potential (a higher score in the DII), inducing a higher inflammatory response were positively associated with pro-inflammatory biomarkers in males and females. Specifically, higher levels of hs-CRP, IL-6 and TNF-α in healthy males and females. Four of the studies found a positive association between adherence to a pro-inflammatory diet (indicated by a higher score in the DII) and IL-6 (Almeida-de-Souza et al., 2018; Seremet Kurklu et al., 2020), IL-1, IL-2, interferon gammon and sVCAM-1 in healthy males and females (Shivappa et al., 2017), but also a positive association between DII and CRP in males and females with obesity (Lazarou et al., 2010). However, two studies did not find any associations between the DII and CRP (Sen et al., 2018), or IL-6, MCP-1 or TNF-α (Coheley et al., 2019) in neither healthy males or females. This was likely due to study design and methodology. For example in one of the studies (Sen et al., 2018) exposure assessment (dietary intake) occurred approximately four years before outcome assessment (CRP) and given dietary patterns change rapidly in childhood (Demory-Luce et al., 2004) timing may have affected any potential associations. Furthermore, in this study, CRP data was only available in a small subset of children from the overall study population.
Overall, the results regarding dietary patterns and indices show adequate adherence to healthful dietary patterns, such as the DASH diet, low glycaemic index diets and the Mediterranean diet are associated with decreased levels of biomarkers, including CRP, IL-6 and TNF-α. While a Western dietary pattern and diets with high inflammatory potential as measured by the DII elicited a pro-inflammatory response increasing levels of the same biomarkers. Associations across the studies were attenuated by gender, as well as the presence of underlying pathologies, independent of dietary intake. However, the mechanisms by which dietary patterns affect the inflammatory process are largely underexplored (Marx et al., 2020). It has been previously hypothesised that the protective effect of healthful dietary patterns like the Mediterranean diet, and the pro-inflammatory response elicited by a Western dietary pattern may be derived from the properties of their constituents (Casas et al., 2014).
2.2. Food groups, macro- and micronutrients
The associations between the individual constituents of dietary patterns (food groups, macro- and micronutrients) and inflammatory markers are attenuated by gender, as well as the presence of underlying pathologies, independent of dietary intake. Concerning food groups, of the nine observational studies, examining vegetable and/or fruit intake (Arouca et al., 2018; Navarro et al., 2017; Sanjeevi et al., 2018; González-Gil et al., 2016; Qureshi et al., 2009; Hagin et al., 2017; Holt et al., 2009; Han et al., 2015; Cabral et al., 2018), two were previously discussed (Navarro et al., 2017; Sanjeevi et al., 2018). Notably, across the studies high dietary intake of vegetables and/or fruits was associated with lower levels of CRP (González-Gil et al., 2016; Qureshi et al., 2009; Hagin et al., 2017) and IL-6 (Holt et al., 2009; Cabral et al., 2018) (both in healthy males and females), TNF-α (Arouca et al., 2018) (only in healthy females), and IL-17F (Han et al., 2015) (both male and female patients with asthma). In contrast, one study found an association between intake and lower levels of hs-CRP in females, but not in males (Navarro et al., 2017), while one study did not find any associations (Sanjeevi et al., 2018). Whole and refined grain intake were examined in one intervention (Hajihashemi et al., 2014) and six observational (Arouca et al., 2018; González-Gil et al., 2016; Qureshi et al., 2009; Han et al., 2015; Cabral et al., 2018; Hur and Reicks, 2012) studies, five of which were previously discussed (Arouca et al., 2018; González-Gil et al., 2016; Qureshi et al., 2009; Han et al., 2015; Cabral et al., 2018). Overall, these studies found that wholegrain dietary intake was associated with lower levels of CRP in a female cohort with obesity (Hajihashemi et al., 2014) and in healthy males and females (Hur and Reicks, 2012) as well as IL-17F in males and females from cohorts with and without underlying pathologies (Han et al., 2015). In contrast, results on the association between refined grain intake and inflammatory markers remain inconclusive (Arouca et al., 2018; González-Gil et al., 2016; Qureshi et al., 2009; Cabral et al., 2018). Similarly, dairy was examined in seven observational studies (Arouca et al., 2018; Sanjeevi et al., 2018; González-Gil et al., 2016; Qureshi et al., 2009; Han et al., 2015; Cabral et al., 2018; Aslam et al., 2020), six of which were previously discussed (Arouca et al., 2018; Sanjeevi et al., 2018; González-Gil et al., 2016; Qureshi et al., 2009; Han et al., 2015; Cabral et al., 2018). While one study found an association between dairy intake and higher levels of IL-17F in males and females, from a cohort of patients with asthma (Han et al., 2015) and a second study found an association between dairy intake and higher levels of IL-6, Il-10 and TGFβ-1 in females, but not in males, and IL-1 in both males and females, and IL-5 in males but not in females in a healthy cohort (Arouca et al., 2018) the majority of studies did not find any associations with inflammatory biomarkers (Arouca et al., 2018; Sanjeevi et al., 2018; González-Gil et al., 2016; Qureshi et al., 2009; Han et al., 2015; Cabral et al., 2018).
Even more interesting is the fact that the majority of studies examining pro-inflammatory foods (Tabung et al., 2016; Kanauchi et al., 2019) including meat, seafood and eggs (Arouca et al., 2018; Sanjeevi et al., 2018; González-Gil et al., 2016; Qureshi et al., 2009; Han et al., 2015; Cabral et al., 2018; Aeberli et al., 2006), and added sugars (snacks—candy, jams, spreads, sugar-sweetened beverages, fruit juice) (Karampola et al., 2019; Sanjeevi et al., 2018; González-Gil et al., 2016; Hagin et al., 2017; Holt et al., 2009; Han et al., 2015; Cabral et al., 2018; Kosova et al., 2013; Jin et al., 2014) did not find an association between dietary intake and CRP, IL-6 and TNF-α in males and females from healthy cohorts. This is particularly interesting in the case of meat, which has previously demonstrated one of the most consistent epidemiological associations between diet and human disease risk (Alisson-Silva et al., 2016) and has been associated with increased levels of CRP in other studies (Montonen et al., 2013). The lack of associations between these constituents and inflammatory markers in the studies in this review may be due to the observational study design and the methodology employed. It is widely accepted that nutrition epidemiology studies are affected by reporting bias. Imprecision in the measurement of dietary intake is often observed, particularly in children and adolescents, which can cause the over- or under-estimation of the impact of exposure (Livingstone and Robson, 2000; Foster and Adamson, 2014; Magarey et al., 2011). Whereas biological, rather than self-reported, dietary measures are a more accurate and reliable way of investigating dietary intake as well as absorption (Potischman, 2003)and should be more often used in future research studies (Dragsted et al., 2018).
In terms of macronutrients, fat was examined in 13 observational studies (Del Mar Bibiloni et al., 2013; Arouca et al., 2018; Karampola et al., 2019; Navarro et al., 2017; Sanjeevi et al., 2018; Qureshi et al., 2009; Aeberli et al., 2006; Prihaningtyas et al., 2019; Thomas et al., 2008; Harris et al., 2019; Arya et al., 2006; Oldewage-Theron and Kruger, 2017; Au et al., 2012), 7 of which were previously discussed (Del Mar Bibiloni et al., 2013; Arouca et al., 2018; Karampola et al., 2019; Navarro et al., 2017; Sanjeevi et al., 2018; Qureshi et al., 2009; Aeberli et al., 2006). Overall, the studies found that saturated fatty acids intake was associated with higher levels of CRP in healthy females but not males (Navarro et al., 2017; Arya et al., 2006) as well as obese males and females (Aeberli et al., 2006) and healthy males but not females (Harris et al., 2019). One study found an inverse association between monounsaturated fatty acid (MUFA) and IL-6 as well as between omega-3 polyunsaturated fatty acid and plasminogen activator inhibitor-1 in healthy females (Del Mar Bibiloni et al., 2013). Another study found a positive association between MUFA : SFA ratio and IL-6 in males and females, TGFβ-1 in females but not in males, and sVCAM-1 in males but not in females, from a cohort of healthy participants (Arouca et al., 2018). The same study also found an inverse association between SFA and sVCAM-1 in females but not in males (Arouca et al., 2018). Lastly, seven studies found no associations between SFA and CRP (Sanjeevi et al., 2018; Thomas et al., 2008) and total fat and CRP (Karampola et al., 2019; Qureshi et al., 2009; Prihaningtyas et al., 2019; Oldewage-Theron and Kruger, 2017; Au et al., 2012) or IL-6 (Au et al., 2012). Fibre was examined in one intervention (Machado et al., 2015) and nine observational (Karampola et al., 2019; Navarro et al., 2017; Qureshi et al., 2009; Arya et al., 2006; Jaacks et al., 2014; Lin et al., 2015; Swann et al., 2020; Parikh et al., 2012; Miller et al., 2016) studies, three of which were discussed previously (Karampola et al., 2019; Navarro et al., 2017; Qureshi et al., 2009). Results show that seven studies did not find an association between fibre intake and CRP in males or females from healthy cohorts (Qureshi et al., 2009; Arya et al., 2006; Machado et al., 2015; Jaacks et al., 2014; Lin et al., 2015; Swann et al., 2020) and an obese cohort (Karampola et al., 2019).Whereas three studies found an association between fibre intake and lower levels of CRP (Navarro et al., 2017; Parikh et al., 2012), from cohorts of healthy participants and lower levels of plasminogen activator inhibitor-1, and monocyte chemoattractant protein-1 in overweight males and females (Miller et al., 2016). Lastly, a total of seven observational studies examined various micronutrients (Del Mar Bibiloni et al., 2013; Navarro et al., 2017; Holt et al., 2009; Oldewage-Theron and Kruger, 2017; Zhu et al., 2014; King et al., 2007; de Sousa et al., 2019), three of which were discussed previously (Navarro et al., 2017; Holt et al., 2009; Oldewage-Theron and Kruger, 2017). These studies found an association between dietary intakes of vitamins, specifically A, C and E, and lower levels of CRP and IL-6, while sodium was associated with higher levels of TNF-α, in males and females from a cohort of healthy participants (Zhu et al., 2014). Taken together these results indicate that high intakes of vegetables, fruit and whole grains resulted in lower levels of inflammatory biomarkers, such as CRP, IL-6 and TNF-α. The same was for healthy fats such as MUFA and PUFA and various micronutrients such and vitamins A, C and E, all of which are considered to have anti-inflammatory properties (Schultz et al., 2019). While higher intakes of saturated fatty acids and sodium elicited an inflammatory response.
Indeed, the Mediterranean diet is characterized by high intakes of anti-inflammatory food groups, macro- and micronutrients (Dai et al., 2008). The diet is rich in vegetables, fruits and whole grains and therefore also antioxidants, folate, and flavonoids which are considered anti-inflammatory. While the high dietary fibre content supports gut health and the growth of microbial species, potentially regulating the inhibition or production of pro-inflammatory chemokines and cytokines (Davis et al., 2020) and omega-3 PUFAs found in the diet, have been shown to regulate the immune response by inhibiting the activation of pro-inflammatory pathways and reducing cytokine expression (Giacobbe et al., 2020; Giacobbe and Mind, 2021). High-dose eicosapentaenoic acid has been shown to improve cognitive symptoms in Attention deficit hyperactivity disorder (ADHD) youth with low baseline levels (Chang et al., 2018, 2019), while research in animal models has demonstrated inflammation-induced reductions in neurogenesis can be prevented through omega-3 PUFAs intake (Borsini et al., 2017). In contrast, the Western dietary pattern, comprised of high intakes of ultra-processed foods, sodium, trans- and saturated fatty acids while being low in omega-3 PUFAs (Li et al., 2020; Okręglicka, 2015) demonstrates positive associations with pro-inflammatory biomarkers.
3. Clinical implications
As discussed extensively in this review, the examined studies demonstrate healthful dietary patterns such as the Mediterranean diet pattern as well as its individual constituents including vegetables and fruit, macronutrients such as fibre and healthy fats such as Omega-3, and micronutrients such as Vitamins A, C and E, have inverse associations with pro-inflammatory biomarkers. As such, modifying dietary intake to encompass these healthful elements as early as during childhood and adolescence represents a promising therapeutic strategy, potentially regulating the immune response and subsequently reducing the risk of adverse mental health disorders and associated co- and multi-morbid conditions later in life.
However, it is important to highlight that there is currently no consensus regarding the inflammatory biomarkers best used to represent chronic low-grade inflammation in children and adolescents, while biomarker measurement error such as sampling, storage and laboratory errors also cannot be excluded (Tworoger and Hankinson, 2006). Moreover, the majority of the studies in this review used a single static measurement of inflammation, however, inflammatory markers owing to their role in homeostasis and immune response are by nature not static and when measured in the fasting state are recognised as being insensitive and producing highly variable results. Several studies examined cohorts with underlying pathologies, with overweight/obesity (Karampola et al., 2019; Lazarou et al., 2010; Çağiran Yilmaz et al., 2019; Rouhani et al., 2016; Iannuzzi et al., 2009; Parillo et al., 2012; Damsgaard et al., 2013; Hajihashemi et al., 2014; Aeberli et al., 2006; Prihaningtyas et al., 2019; Machado et al., 2015; Miller et al., 2016), and type-1 diabetes (Liese et al., 2018; Sanjeevi et al., 2018; Jaacks et al., 2014) being the most studied. In overweight and obese populations excess adipose tissue has been linked to an increase in sub-chronic levels of key pro-inflammatory cytokines, mainly CRP, IL-6 and TNF-α (Reilly and Saltiel, 2017), and this may prevent or attenuate any potential therapeutic effect exerted by a healthful diet (Monteiro and Azevedo, 2010). The evident associations were also attenuated by several different factors (Calder et al., 2011; Navarro et al., 2016; Cunningham-Rundles et al., 2005). For example, gender differences were evident across the studies however, they were not specific to one particular diet, food group, macro- or micronutrient. Nor were they specific to any inflammatory biomarker. This may be in part attributable to the influence of hormones. Sex hormones affect immune function, whereby oestrogens stimulate auto-immunity and androgens exhibit protective properties (Zolin et al., 2015; da Silva et al., 2017; Whitacre, 2001; Ortona et al., 2016). These implications all need to be addressed in future research studies. Moreover, there is limited evidence examining the associations between dietary intake and low-grade inflammation in the context of neuropsychiatric disorders. Only one study included in our review examined the mechanistic pathways between dietary patterns, inflammatory markers and outcomes of depressive symptoms (Oddy et al., 2014), reporting a healthful diet was protective in these pathways. Ultimately, further interventional research is needed to establish the strength of associations between dietary intake and inflammatory biomarkers, as well as studies that examine the biological mechanisms underlying and attenuating such associations. Moreover, research studies should extend to include the examination of these associations specifically in the context of neuropsychiatric conditions and associated co- and multi-morbid conditions in children and adolescents.
4. Conclusion
In this review, I have discussed the current evidence that supports a potential therapeutic role for dietary intake in the amelioration of low-grade inflammation in children and adolescents. These data suggest adherence to healthful patterns and their constituents are associated with decreased levels of inflammatory biomarkers, including CRP, IL-6 and TNF- α, in children and adolescents. However, it is important to highlight the need for further research studies, that address methodological implications and factors that may confound associations, to develop a better understanding of the biological mechanisms underlying and attenuating the associations between dietary intake and low-grade inflammation in children and adolescents, specifically in the context of mental health.
Funding
Melissa Bujtor is funded by the UK Medical Research Council (grants MR/N029488/1). This project has received funding from the European Union's Horizon 2020 research and innovation programme under Grant Agreement No 848158.
Declaration of competing interest
The author declares no conflict of interest.
Acknowledgements
The corresponding author confirms that all authors who contributed significantly to the work have been listed and all authors listed contributed significantly to the work and have read and agreed to the current version of the manuscript. No other acknowledgements applicable.
Biography

Melissa Bujtor is a researcher and PhD candidate working in the Stress, Psychiatry and Immunology (SPI) Lab at King's College London's, Institute of Psychiatry, Psychology and Neuroscience and Deakin University Australia. Her current PhD research examines the associations between modifiable health behaviours, such as diet, and their associations with mental health in children and adolescents. She is particularly interested in advances in the field of nutritional psychiatry and understanding the mechanisms, at the level of the body and the brain, underlying the effect of nutrition on mental health especially in the crucial early years.
References
- Aeberli I., et al. Dietary intakes of fat and antioxidant vitamins are predictors of subclinical inflammation in overweight Swiss children. AJCN (Am. J. Clin. Nutr.) 2006;84(4):748–755. doi: 10.1093/ajcn/84.4.748. [DOI] [PubMed] [Google Scholar]
- Agostinis-Sobrinho C., et al. The combined association of adherence to Mediterranean diet, muscular and cardiorespiratory fitness on low-grade inflammation in adolescents: a pooled analysis. Eur. J. Nutr. 2019;58(7):2649–2656. doi: 10.1007/s00394-018-1812-0. [DOI] [PubMed] [Google Scholar]
- Al-Hamad D., Raman V. Metabolic syndrome in children and adolescents. Transl. Pediatr. 2017;6(4):397. doi: 10.21037/tp.2017.10.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alisson-Silva F., et al. Human risk of diseases associated with red meat intake: analysis of current theories and proposed role for metabolic incorporation of a non-human sialic acid. Mol. Aspect. Med. 2016;51:16–30. doi: 10.1016/j.mam.2016.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Almeida-de-Souza J., et al. Dietary inflammatory index and inflammatory biomarkers in adolescents from LabMed physical activity study. Eur. J. Clin. Nutr. 2018;72(5):710–719. doi: 10.1038/s41430-017-0013-x. [DOI] [PubMed] [Google Scholar]
- Amaral G.A., et al. Interleukin 1-beta is linked to chronic low-grade inflammation and cardiovascular risk factors in overweight Adolescents. Endocrine, metabolic & immune disorders-drug targets (formerly current drug targets-immune. Endocrine & Metabolic Disorders) 2020;20(6):887–894. doi: 10.2174/1871530319666191116141159. [DOI] [PubMed] [Google Scholar]
- Arouca A., et al. Associations between a Mediterranean diet pattern and inflammatory biomarkers in European adolescents. Eur. J. Nutr. 2018;57(5):1747–1760. doi: 10.1007/s00394-017-1457-4. [DOI] [PubMed] [Google Scholar]
- Arya S., et al. C-reactive protein and dietary nutrients in urban Asian Indian adolescents and young adults. Nutrition. 2006;22(9):865–871. doi: 10.1016/j.nut.2006.05.002. [DOI] [PubMed] [Google Scholar]
- Aslam H., et al. The associations between dairy product consumption and biomarkers of inflammation, adipocytokines, and oxidative stress in children: a cross-sectional study. Nutrients. 2020;12(10) doi: 10.3390/nu12103055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Au L.E., et al. Dietary intake and cardiometabolic risk in ethnically diverse urban schoolchildren. J. Acad. Nutr. Diet. 2012;112(11):1815–1821. doi: 10.1016/j.jand.2012.07.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbaresko J., et al. Dietary pattern analysis and biomarkers of low-grade inflammation: a systematic literature review. Nutr. Rev. 2013;71(8):511–527. doi: 10.1111/nure.12035. [DOI] [PubMed] [Google Scholar]
- Bennett J.M., et al. Inflammation–nature's way to efficiently respond to all types of challenges: implications for understanding and managing “the epidemic” of chronic diseases. Front. Med. 2018;5:316. doi: 10.3389/fmed.2018.00316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berk M., et al. So depression is an inflammatory disease, but where does the inflammation come from? BMC Med. 2013;11(1):1–16. doi: 10.1186/1741-7015-11-200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borsini A., et al. Rescue of IL-1β-induced reduction of human neurogenesis by omega-3 fatty acids and antidepressants. Brain Behav. Immun. 2017;65:230–238. doi: 10.1016/j.bbi.2017.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowyer R.C., et al. Use of dietary indices to control for diet in human gut microbiota studies. Microbiome. 2018;6(1):1–11. doi: 10.1186/s40168-018-0455-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bujtor M., et al. Associations of dietary intake on biological markers of inflammation in children and adolescents: a systematic review. Nutrients. 2021;13(2):356. doi: 10.3390/nu13020356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cabral M., et al. Food intake and high-sensitivity C-reactive protein levels in adolescents. Nutr. Metabol. Cardiovasc. Dis. 2018;28(10):1067–1074. doi: 10.1016/j.numecd.2018.06.003. [DOI] [PubMed] [Google Scholar]
- Çağiran Yilmaz F., et al. Adolescent obesity and its association with diet quality and cardiovascular risk factors. Ecol. Food Nutr. 2019;58(3):207–218. doi: 10.1080/03670244.2019.1580581. [DOI] [PubMed] [Google Scholar]
- Calder P.C., et al. Dietary factors and low-grade inflammation in relation to overweight and obesity. Br. J. Nutr. 2011;106(S3):S1–S78. doi: 10.1017/S0007114511005460. [DOI] [PubMed] [Google Scholar]
- Carvalho K.M.B., et al. Does the mediterranean diet protect against stress-induced inflammatory activation in European adolescents? The HELENA study. Nutrients. 2018;10(11) doi: 10.3390/nu10111770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casas R., et al. The immune protective effect of the mediterranean diet against chronic low-grade inflammatory diseases. Endocrine, metabolic & immune disorders-drug targets (formerly current drug targets-immune. Endocrine & Metabolic Disorders) 2014;14(4):245–254. doi: 10.2174/1871530314666140922153350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cattaneo A., et al. Whole-blood expression of inflammasome-and glucocorticoid-related mRNAs correctly separates treatment-resistant depressed patients from drug-free and responsive patients in the BIODEP study. Transl. Psychiatry. 2020;10(1):1–14. doi: 10.1038/s41398-020-00874-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan She Ping-Delfos W.L., et al. Use of the Dietary Guideline Index to assess cardiometabolic risk in adolescents. Br. J. Nutr. 2015;113(11):1741–1752. doi: 10.1017/S0007114515001026. [DOI] [PubMed] [Google Scholar]
- Chang J.P.-C., et al. Omega-3 polyunsaturated fatty acids in youths with attention deficit hyperactivity disorder: a systematic review and meta-analysis of clinical trials and biological studies. Neuropsychopharmacology. 2018;43(3):534–545. doi: 10.1038/npp.2017.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang J.P.-C., et al. High-dose eicosapentaenoic acid (EPA) improves attention and vigilance in children and adolescents with attention deficit hyperactivity disorder (ADHD) and low endogenous EPA levels. Transl. Psychiatry. 2019;9(1):1–9. doi: 10.1038/s41398-019-0633-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coheley L.M., et al. Dietary inflammatory index® and cortical bone outcomes in healthy adolescent children. Osteoporos. Int. : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA. 2019;30(8):1645–1654. doi: 10.1007/s00198-019-04946-3. [DOI] [PubMed] [Google Scholar]
- Colasanto M., et al. Depression and inflammation among children and adolescents: a meta-analysis. J. Affect. Disord. 2020;277:940–948. doi: 10.1016/j.jad.2020.09.025. [DOI] [PubMed] [Google Scholar]
- Cunningham-Rundles S., et al. Mechanisms of nutrient modulation of the immune response. J. Allergy Clin. Immunol. 2005;115(6):1119–1128. doi: 10.1016/j.jaci.2005.04.036. [DOI] [PubMed] [Google Scholar]
- Dai J., et al. Adherence to the Mediterranean diet is inversely associated with circulating interleukin-6 among middle-aged men: a twin study. Circulation. 2008;117(2):169. doi: 10.1161/CIRCULATIONAHA.107.710699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Damsgaard C.T., et al. Higher protein diets consumed ad libitum improve cardiovascular risk markers in children of overweight parents from eight European countries. J. Nutr. 2013;143(6):810–817. doi: 10.3945/jn.112.173427. [DOI] [PubMed] [Google Scholar]
- Davis R., et al. Habitual dietary fibre and prebiotic intake is inadequate in patients with inflammatory bowel disease: findings from a multicentre cross-sectional study. J. Hum. Nutr. Diet. 2020 doi: 10.1111/jhn.12812. [DOI] [PubMed] [Google Scholar]
- Demory-Luce D., et al. Changes in food group consumption patterns from childhood to young adulthood: the Bogalusa Heart Study. J. Am. Diet Assoc. 2004;104(11):1684–1691. doi: 10.1016/j.jada.2004.07.026. [DOI] [PubMed] [Google Scholar]
- Douros K., et al. Adherence to the Mediterranean diet and inflammatory markers in children with asthma. Allergol. Immunopathol. 2019;47(3):209–213. doi: 10.1016/j.aller.2018.04.007. [DOI] [PubMed] [Google Scholar]
- Dragsted L.O., et al. Validation of biomarkers of food intake—critical assessment of candidate biomarkers. Genes Nutr. 2018;13(1):1–14. doi: 10.1186/s12263-018-0603-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esposito K., et al. Effect of a Mediterranean-style diet on endothelial dysfunction and markers of vascular inflammation in the metabolic syndrome: a randomized trial. J. Am. Med. Assoc. 2004;292(12):1440–1446. doi: 10.1001/jama.292.12.1440. [DOI] [PubMed] [Google Scholar]
- Foster E., Adamson A. Challenges involved in measuring intake in early life: focus on methods. Proc. Nutr. Soc. 2014;73(2):201–209. doi: 10.1017/S0029665114000020. [DOI] [PubMed] [Google Scholar]
- Furman D., et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019;25(12):1822–1832. doi: 10.1038/s41591-019-0675-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giacobbe J. Mind IT, editor. Inspire The Mind: Inspire The Mind. [cited 19/05/2021 19/05/2021]. Available from: https://library-guides.ucl.ac.uk/referencing-plagiarism/vancouver#s-lg-box-wrapper-18252573.
- Giacobbe J., et al. The anti-inflammatory role of omega-3 polyunsaturated fatty acids metabolites in pre-clinical models of psychiatric, neurodegenerative and neurological disorders. Front. Psychiatr. 2020;11:122. doi: 10.3389/fpsyt.2020.00122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giugliano D., et al. The effects of diet on inflammation: emphasis on the metabolic syndrome. J. Am. Coll. Cardiol. 2006;48(4):677–685. doi: 10.1016/j.jacc.2006.03.052. [DOI] [PubMed] [Google Scholar]
- González-Gil E., et al. Food intake and inflammation in European children: the IDEFICS study. Eur. J. Nutr. 2016;55(8):2459–2468. doi: 10.1007/s00394-015-1054-3. [DOI] [PubMed] [Google Scholar]
- Grosso G., et al. Mediterranean diet and cardiovascular risk factors: a systematic review. Crit. Rev. Food Sci. Nutr. 2014;54(5):593–610. doi: 10.1080/10408398.2011.596955. [DOI] [PubMed] [Google Scholar]
- Hagin S., et al. Dietary behaviors in newly diagnosed youth with inflammatory bowel disease. Child Health Care. 2017;46(4):408–420. [Google Scholar]
- Hajihashemi P., et al. Whole-grain intake favorably affects markers of systemic inflammation in obese children: a randomized controlled crossover clinical trial. Mol. Nutr. Food Res. 2014;58(6):1301–1308. doi: 10.1002/mnfr.201300582. [DOI] [PubMed] [Google Scholar]
- Han Y.-Y., et al. Diet, interleukin-17, and childhood asthma in Puerto Ricans. Ann. Allergy Asthma Immunol. : Official Public. Am. College Allergy, Asthma, & Immunol. 2015;115(4):288. doi: 10.1016/j.anai.2015.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris C.P., et al. Dietary saturated fat and low-grade inflammation modified by accelerometer-measured physical activity in adolescence: results from the GINIplus and LISA birth cohorts. BMC Publ. Health. 2019;19(1):818. doi: 10.1186/s12889-019-7113-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilger-Kolb J., et al. Associations between dietary factors and obesity-related biomarkers in healthy children and adolescents - a systematic review. Nutr. J. 2017;16:1–12. doi: 10.1186/s12937-017-0300-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holt E.M., et al. Fruit and vegetable consumption and its relation to markers of inflammation and oxidative stress in adolescents. J. Am. Diet Assoc. 2009;109(3):414–421. doi: 10.1016/j.jada.2008.11.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hur I.Y., Reicks M. Relationship between whole-grain intake, chronic disease risk indicators, and weight status among adolescents in the national health and nutrition examination survey, 1999-2004. J. Acad. Nutr. Diet. 2012;112(1):46–55. doi: 10.1016/j.jada.2011.08.028. [DOI] [PubMed] [Google Scholar]
- Iannuzzi A., et al. Comparison of two diets of varying glycemic index on carotid subclinical atherosclerosis in obese children. Heart Ves. 2009;24(6):419–424. doi: 10.1007/s00380-008-1138-6. [DOI] [PubMed] [Google Scholar]
- Jaacks L.M., et al. No association of dietary fiber intake with inflammation or arterial stiffness in youth with type 1 diabetes. J. Diabet. Complicat. 2014;28(3):305–310. doi: 10.1016/j.jdiacomp.2014.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacka F.N., et al. Associations between diet quality and depressed mood in adolescents: results from the Australian Healthy Neighbourhoods Study. Aust. N. Z. J. Psychiatr. 2010;44(5):435–442. doi: 10.3109/00048670903571598. [DOI] [PubMed] [Google Scholar]
- Jacka F.N., et al. A prospective study of diet quality and mental health in adolescents. PLoS One. 2011;6(9) doi: 10.1371/journal.pone.0024805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin R., et al. Dietary fructose reduction improves markers of cardiovascular disease risk in Hispanic-American adolescents with NAFLD. Nutrients. 2014;6(8):3187–3201. doi: 10.3390/nu6083187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanauchi M., et al. A novel dietary inflammatory index reflecting for inflammatory ageing. Annals. Med. Surg. 2019;47:44–46. doi: 10.1016/j.amsu.2019.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karampola M., et al. Study on dietary constituents, hs-CRP serum levels and investigation of correlation between them in excess weight adolescents. Hippokratia. 2019;23(1):3. [PMC free article] [PubMed] [Google Scholar]
- Khayyatzadeh S.S., et al. A Western dietary pattern is associated with elevated level of high sensitive C-reactive protein among adolescent girls. Eur. J. Clin. Invest. 2018;48(4) doi: 10.1111/eci.12897. [DOI] [PubMed] [Google Scholar]
- Kiecolt-Glaser J.K., et al. Inflammation: depression fans the flames and feasts on the heat. Am. J. Psychiatr. 2015;172(11):1075–1091. doi: 10.1176/appi.ajp.2015.15020152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King D.E., et al. Magnesium intake and serum C-reactive protein levels in children. Magnes. Res. 2007;20(1):32–36. [PubMed] [Google Scholar]
- Kohlboeck G., et al. Food intake, diet quality and behavioral problems in children: results from the GINI-plus/LISA-plus studies. Ann. Nutr. Metab. 2012;60(4):247–256. doi: 10.1159/000337552. [DOI] [PubMed] [Google Scholar]
- Kosova E.C., et al. The relationships between sugar-sweetened beverage intake and cardiometabolic markers in young children. J. Acad. Nutr. Diet. 2013;113(2):219–227. doi: 10.1016/j.jand.2012.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lassale C., et al. Healthy dietary indices and risk of depressive outcomes: a systematic review and meta-analysis of observational studies. Mol. Psychiatr. 2019;24(7):965–986. doi: 10.1038/s41380-018-0237-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazarou C., et al. C-reactive protein levels are associated with adiposity and a high inflammatory foods index in mountainous Cypriot children. Clin. Nutr.(Edinburgh, Scotland) 2010;29(6):779–783. doi: 10.1016/j.clnu.2010.05.001. [DOI] [PubMed] [Google Scholar]
- Li T., et al. Systematic review and meta-analysis: the association of a pre-illness Western dietary pattern with the risk of developing inflammatory bowel disease. J Dig Dis. 2020 doi: 10.1111/1751-2980.12910. [DOI] [PubMed] [Google Scholar]
- Liese A.D., et al. Dietary quality and markers of inflammation: No association in youth with type 1 diabetes. J. Diabet. Complicat. 2018;32(2):179–184. doi: 10.1016/j.jdiacomp.2017.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin Y., et al. Dietary fiber intake and its association with indicators of adiposity and serum biomarkers in European adolescents: the HELENA study. Eur. J. Nutr. 2015;54(5):771–782. doi: 10.1007/s00394-014-0756-2. [DOI] [PubMed] [Google Scholar]
- Livingstone M., Robson P. Measurement of dietary intake in children. Proc. Nutr. Soc. 2000;59(2):279–293. doi: 10.1017/s0029665100000318. [DOI] [PubMed] [Google Scholar]
- Machado A.M., et al. Effects of brown and golden flaxseed on the lipid profile, glycemia, inflammatory biomarkers, blood pressure and body composition in overweight adolescents. Nutrition. 2015;31(1):90–96. doi: 10.1016/j.nut.2014.05.002. [DOI] [PubMed] [Google Scholar]
- Magarey A., et al. Assessing dietary intake in children and adolescents: considerations and recommendations for obesity research. Int. J. Pediatr. Obes. 2011;6(1):2–11. doi: 10.3109/17477161003728469. [DOI] [PubMed] [Google Scholar]
- Del Mar Bibiloni M., et al. Dietary factors associated with subclinical inflammation among girls. Eur. J. Clin. Nutr. 2013;67(12):1264–1270. doi: 10.1038/ejcn.2013.196. [DOI] [PubMed] [Google Scholar]
- Marx W., et al. Diet and depression: exploring the biological mechanisms of action. Mol. Psychiatr. 2020:1–17. doi: 10.1038/s41380-020-00925-x. [DOI] [PubMed] [Google Scholar]
- Miller S.J., et al. Dietary fibre linked to decreased inflammation in overweight minority youth. Pediatric obesity. 2016;11(1):33–39. doi: 10.1111/ijpo.12017. [DOI] [PubMed] [Google Scholar]
- Minihane A.M., et al. Low-grade inflammation, diet composition and health: current research evidence and its translation. Br. J. Nutr. 2015;114(7):999–1012. doi: 10.1017/S0007114515002093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell R.H.B., Goldstein B.I. Inflammation in children and adolescents with neuropsychiatric disorders: a systematic review. J. Am. Acad. Child Adolesc. Psychiatr. 2014;53(3):274–296. doi: 10.1016/j.jaac.2013.11.013. [DOI] [PubMed] [Google Scholar]
- Moeller S.M., et al. Dietary patterns: challenges and opportunities in dietary patterns research: an Experimental Biology workshop, April 1, 2006. J. Am. Diet Assoc. 2007;107(7):1233–1239. doi: 10.1016/j.jada.2007.03.014. [DOI] [PubMed] [Google Scholar]
- Monteiro R., Azevedo I. Chronic inflammation in obesity and the metabolic syndrome. Mediat. Inflamm. 2010;2010 doi: 10.1155/2010/289645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montonen J., et al. Consumption of red meat and whole-grain bread in relation to biomarkers of obesity, inflammation, glucose metabolism and oxidative stress. Eur. J. Nutr. 2013;52(1):337–345. doi: 10.1007/s00394-012-0340-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Navarro S.L., et al. Factors associated with multiple biomarkers of systemic inflammation. Cancer Epidemiol. Prevention Biomark. 2016;25(3):521–531. doi: 10.1158/1055-9965.EPI-15-0956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Navarro P., et al. Vegetable and fruit intakes are associated with hs-CRP levels in pre-pubertal girls. Nutrients. 2017;9(3):224. doi: 10.3390/nu9030224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oddy W., et al. A 'Western' dietary pattern, adiposity and inflammation: pathways to mental health problems and depression in adolescents. Obes. Rev. 2014;15:120–121. [Google Scholar]
- Oddy W.H., et al. Dietary patterns, body mass index and inflammation: pathways to depression and mental health problems in adolescents. Brain Behav. Immun. 2018;69:428–439. doi: 10.1016/j.bbi.2018.01.002. [DOI] [PubMed] [Google Scholar]
- Okręglicka K. Health effects of changes in the structure of dietary macronutrients intake in western societies. Rocz. Panstw. Zakl. Hig. 2015;66(2):97–105. [PubMed] [Google Scholar]
- Oldewage-Theron W., Kruger R. The association between diet quality and subclinical inflammation among children aged 6-18 years in the Eastern Cape, South Africa. Publ. Health Nutr. 2017;20(1):102–111. doi: 10.1017/S1368980016001956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ortona E., et al. Sex-based differences in autoimmune diseases. Ann. Ist. Super Sanita. 2016;52(2):205–212. doi: 10.4415/ANN_16_02_12. [DOI] [PubMed] [Google Scholar]
- Osimo E.F., et al. Prevalence and correlates of low-grade systemic inflammation in adult psychiatric inpatients: an electronic health record-based study. Psychoneuroendocrinology. 2018;91:226–234. doi: 10.1016/j.psyneuen.2018.02.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parikh S., et al. Adolescent fiber consumption is associated with visceral fat and inflammatory markers. J. Clin. Endocrinol. Metabol. 2012;97(8):E1451–E1457. doi: 10.1210/jc.2012-1784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parillo M., et al. Metabolic changes after a hypocaloric, low-glycemic-index diet in obese children. J. Endocrinol. Invest. 2012;35(7):629–633. doi: 10.3275/7909. [DOI] [PubMed] [Google Scholar]
- Pitharouli M.C., et al. Elevated C-reactive protein in patients with depression, independent of genetic, health, and psychosocial factors: results from the UK biobank. Am. J. Psychiatr. 2021 doi: 10.1176/appi.ajp.2020.20060947. appi. ajp. 2020. [DOI] [PubMed] [Google Scholar]
- Potischman N. Biologic and methodologic issues for nutritional biomarkers. J. Nutr. 2003;133(3) doi: 10.1093/jn/133.3.875S. 875S-80S. [DOI] [PubMed] [Google Scholar]
- Prihaningtyas R., et al. 2019. Dietary Intakes and High Sensitivity CRP (hsCRP) in Adolescents with Obesity. [Google Scholar]
- Qureshi M.M., et al. A cross-sectional study of food group intake and C-reactive protein among children. Nutr. Metab. 2009;6:40. doi: 10.1186/1743-7075-6-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reilly S.M., Saltiel A.R. Adapting to obesity with adipose tissue inflammation. Nat. Rev. Endocrinol. 2017;13(11):633–643. doi: 10.1038/nrendo.2017.90. [DOI] [PubMed] [Google Scholar]
- Reinehr T. Inflammatory markers in children and adolescents with type 2 diabetes mellitus. Clin. Chim. Acta. 2019;496:100–107. doi: 10.1016/j.cca.2019.07.006. [DOI] [PubMed] [Google Scholar]
- Rocha N.P., et al. Association between dietary pattern and cardiometabolic risk in children and adolescents: a systematic review. J. Pediatr. 2017;93(3):214–222. doi: 10.1016/j.jped.2017.01.002. [DOI] [PubMed] [Google Scholar]
- Rouhani M.H., et al. The impact of a low glycemic index diet on inflammatory markers and serum adiponectin concentration in adolescent overweight and obese girls: a randomized clinical trial. Horm. Metab. Res. 2016;48(4):251–256. doi: 10.1055/s-0042-100467. [DOI] [PubMed] [Google Scholar]
- Saneei P., et al. The Dietary Approaches to Stop Hypertension (DASH) diet affects inflammation in childhood metabolic syndrome: a randomized cross-over clinical trial. Ann. Nutr. Metab. 2014;64(1):20–27. doi: 10.1159/000358341. [DOI] [PubMed] [Google Scholar]
- Sanjeevi N., et al. Cardiovascular biomarkers in association with dietary intake in a longitudinal study of youth with type 1 diabetes. Nutrients. 2018;10(10) doi: 10.3390/nu10101552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawyer K.M., et al. Intergenerational transmission of depression: clinical observations and molecular mechanisms. Mol. Psychiatr. 2019;24(8):1157–1177. doi: 10.1038/s41380-018-0265-4. [DOI] [PubMed] [Google Scholar]
- Schultz H., et al. Rising plasma beta-carotene is associated with diminishing C-reactive protein in patients consuming a dark green leafy vegetable–rich, low inflammatory foods everyday (LIFE) diet. Am. J. Lifestyle Med. 2019 doi: 10.1177/1559827619894954. 1559827619894954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sen S., et al. Associations of prenatal and early life dietary inflammatory potential with childhood adiposity and cardiometabolic risk in Project Viva. Pediatr Obes. 2018;13(5):292–300. doi: 10.1111/ijpo.12221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seremet Kurklu N., et al. Is there a relationship between the dietary inflammatory index and metabolic syndrome among adolescents? JPEM (J. Pediatr. Endocrinol. Metab.) 2020;33(4):495–502. doi: 10.1515/jpem-2019-0409. [DOI] [PubMed] [Google Scholar]
- Shivappa N., et al. Association between dietary inflammatory index and inflammatory markers in the HELENA study. Mol. Nutr. Food Res. 2017;61(6) doi: 10.1002/mnfr.201600707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- da Silva C.T.B., et al. Inflammation and internalizing disorders in adolescents. Prog. Neuro-Psychopharmacol. Biol. Psychiatry. 2017;77:133–137. doi: 10.1016/j.pnpbp.2017.03.023. [DOI] [PubMed] [Google Scholar]
- Silveira B.K.S., et al. Dietary pattern and macronutrients profile on the variation of inflammatory biomarkers: scientific update. Cardiol. Res. Pract. 2018:1–18. doi: 10.1155/2018/4762575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh J., et al. Vitamin D, low-grade inflammation and cardiovascular risk in young children: a pilot study. Pediatr. Cardiol. 2015;36(7):1338–1343. doi: 10.1007/s00246-015-1162-0. [DOI] [PubMed] [Google Scholar]
- de Sousa A.F., et al. No relation between zinc status and inflammatory biomarkers in adolescent judokas. Int. J. Vitam. Nutr. Res. 2019 doi: 10.1024/0300-9831/a000504. [DOI] [PubMed] [Google Scholar]
- Stein D.J., et al. Integrating mental health with other non-communicable diseases. Br. Med. J (Clinical research ed) 2019;364:l295. doi: 10.1136/bmj.l295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stroescu R.F., et al. Adiponectin, leptin and high sensitivity C-reactive protein values in obese children–important markers for metabolic syndrome? J. Pediatr. Endocrinol. Metab. 2019;32(1):27–31. doi: 10.1515/jpem-2018-0378. [DOI] [PubMed] [Google Scholar]
- Suhett L.G., et al. Dietary inflammatory potential, cardiometabolic risk and inflammation in children and adolescents: a systematic review. Crit. Rev. Food Sci. Nutr. 2020:1–10. doi: 10.1080/10408398.2020.1734911. [DOI] [PubMed] [Google Scholar]
- Sun C.-H., et al. The effect of vitamin–mineral supplementation on CRP and IL-6: a systemic review and meta-analysis of randomised controlled trials. Nutr. Metabol. Cardiovasc. Dis. 2011;21(8):576–583. doi: 10.1016/j.numecd.2009.12.014. [DOI] [PubMed] [Google Scholar]
- Sureda A., et al. Adherence to the mediterranean diet and inflammatory markers. Nutrients. 2018;10(1):62. doi: 10.3390/nu10010062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swann O.G., et al. Dietary fibre intake and its association with inflammatory markers in adolescents. Br. J. Nutr. 2020:1–21. doi: 10.1017/S0007114520001609. [DOI] [PubMed] [Google Scholar]
- Tabung F.K., et al. Development and validation of an empirical dietary inflammatory index. J. Nutr. 2016;146(8):1560–1570. doi: 10.3945/jn.115.228718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tapsell L.C., et al. Foods, nutrients, and dietary patterns: interconnections and implications for dietary guidelines. Adv Nutr. 2016;7(3):445–454. doi: 10.3945/an.115.011718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas N.E., et al. C-reactive protein in schoolchildren and its relation to adiposity, physical activity, aerobic fitness and habitual diet. Br. J. Sports Med. 2008;42(5):357–360. doi: 10.1136/bjsm.2007.043604. [DOI] [PubMed] [Google Scholar]
- Tworoger S.S., Hankinson S.E. Use of biomarkers in epidemiologic studies: minimizing the influence of measurement error in the study design and analysis. Cancer Causes Control. 2006;17(7):889–899. doi: 10.1007/s10552-006-0035-5. [DOI] [PubMed] [Google Scholar]
- van der Velde L.A., et al. Diet quality in childhood: the generation R study. Eur. J. Nutr. 2019;58(3):1259–1269. doi: 10.1007/s00394-018-1651-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vyncke K.E., et al. Intake and serum profile of fatty acids are weakly correlated with global dietary quality in European adolescents. Nutrition. 2013;29(2):411–419. doi: 10.1016/j.nut.2012.07.007. [DOI] [PubMed] [Google Scholar]
- Whitacre C.C. Sex differences in autoimmune disease. Nat. Immunol. 2001;2(9):777–780. doi: 10.1038/ni0901-777. [DOI] [PubMed] [Google Scholar]
- Zhu H.D., et al. Dietary sodium, adiposity, and inflammation in healthy adolescents. Pediatrics. 2014;133(3):E635–E642. doi: 10.1542/peds.2013-1794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zolin S.J., et al. The early evolving sex hormone environment is associatd with significant outcome and the inflammatory response differences post-injury. J. Trauma Acute Care Surgery. 2015;78(3):451. doi: 10.1097/TA.0000000000000550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zunszain P.A., et al. Springer; 2012. Inflammation and Depression. Behavioral Neurobiology of Depression and its Treatment; pp. 135–151. [Google Scholar]