Abstract
Bacteriophages, bacteria-infecting viruses, are considered by many researchers a promising solution for antimicrobial resistance. On the other hand, some phages have shown contribution to bacterial resistance phenomenon by transducing antimicrobial resistance genes to their bacterial hosts. Contradictory consequences of infections are correlated to different phage lifecycles. Out of four known lifecycles, lysogenic and lytic pathways have been riddles since the uncontrolled conversion between them could negatively affect the intended use of phages. However, phages still can be engineered for applications against bacterial and viral infections to ensure high efficiency. This review highlights two main aspects: (1) the different lifecycles as well as the different factors that affect lytic-lysogenic switch are discussed, including the intracellular and molecular factors control this decision. In addition, different models which describe the effect of phages on the ecosystem are compared, besides the approaches to study the switch. (2) An overview on the contribution of the phage in the evolution of the bacteria, instead of eating them, as a consequence of different mode of actions. As well, how phage display has helped in restricting phage cheating and how it could open new gates for immunization and pandemics control will be tacked.
Keywords: Quorum sensing, Biofilm, lytic-lysogenic switch, Pathogenic bacteria
1. Introduction
Bacteriophages (phages for short) is a word means bacteria eaters since the presence of these viruses introduces a clear plaque when they're applied to bacterial lawn Chanishvili (2016). However, It is not always the case, as it was found that “eating bacteria” is one track phages can pursue out of four possible pathways: lytic, lysogenic, pseudo-lysogenic, and chronic mode of actions (M et al., 2017).
Virulent phages which undergo the lytic lifecycle have been investigated deeply, especially, after the antimicrobial resistance phenomenon. Studies of these phages have provided target specificity, co-evolution with their bacterial host, unlike the chemical antimicrobial agents (Hanifin et al., 2008). To date, the Food and Drug Administration (FDA) has permitted the use of bacteriophages on an individual basis, under the guidelines of investigational new drug application (IND). In addition, virulent phages are also used as biocontrol agents in food industry and as veterinary antimicrobials El-Shibiny and Dawoud (2020). However, the highlighted other phage pathways do not necessitate bacterial host disintegration, which makes phage isolation and purification steps more challenging. This paradox raises the concern that some phages can be beneficial to bacteria instead of killing them. For instance, the non-lytic phages can transfer virulent and antibiotic-resistant genes from pathogenic to non-pathogenic bacterial cells (Penadés et al., 2015). Consequently, the adopted virulent genes may lead to boosting the antibiotic-resistant bacteria rather than controlling their activity (Brown-Jaque et al., 2015, Gómez-Gómez et al., 2019). This process is referred to as “lysogenic conversion” or “phage conversion”.
Although phages are considered a promising alternative to antimicrobial agents, they have a dark, not- well explored side. Stemming from this, it is crucial to dig deep in understanding and predicting their behavior in order to control and use them in several applications. This review aims to highlight the ability of phages to take a decision in moving between lytic and lysogenic cycles. Consequently, the four phage cycles and molecular mechanisms of the decision are discussed, along with how they can adversely affect the micro-ecosystems and human body through the phage-bacterial infection. In addition, illustrating when the phage could become as an enemy for human and how the human can take the maximum use of “non-eaters” phages in pandemic diseases.
2. The four phages life cycle
2.1. Lytic and lysogenic life cycles
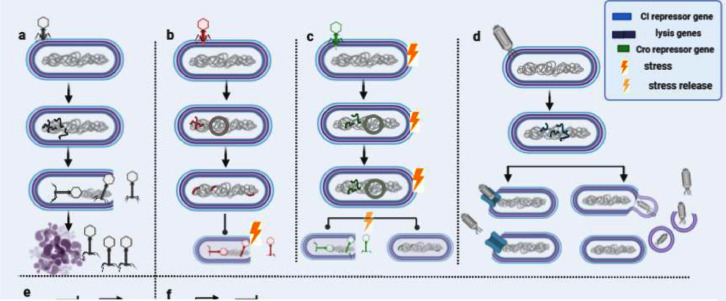
Once the phage infects a bacterial host cell, it releases its genetic material and accordingly takes one of two main routes of replication: lytic or lysogenic. Other replication modes exist such as chronic cycle in filamentous phage (Rakonjac et al., 2017) and pseudo-lysogeny (Filipiak et al., 2020, Łoś and Pseudolysogeny, 2012) but lytic and lysogenic modes are the best studied (Fig. 1) (Z and ST, 2016).
Fig. 1.
The four phage modes of action. a) Lytic life cycle; b) lysogenic life cycle; c) pseudo-lysogenic life cycle; d) chronic life cycle and the release through protein complex (Rakonjac et al., 2011), or budding (Sime-Ngando et al., 2019). e& f are symbolic representations of the antagonistic relation between CI and Coi repressor genes during lytic and lysogenic.
For virulent phages, they enter lytic mode, while temperate phages are characterized by the lysogenic activity. The lytic pathway has distinctive genes such as genes encoding for holins and lysins. Upon these genes expression, the phage disassembles the bacteria's cell membrane and disrupts the peptidoglycan layer. Then, the host cell is transformed into a factory for phages synthesis that in turn lysis, infect and kill other bacteria Young (2014). Lysis genes are governed by lysis repressors, which are controlled by the lysogenic master regulator genes. As the lytic genes become active, lytic phages exclusively express the virulent genes and disrupt their hosts to start the spreading over. Hence lytic decision is called “go” decision. From the phenotype perspective, lytic phages appear in the form of clear plaques in relatively high concentrations on petri dishes.
On the other hand, the lysogenic pathway is mostly abundant in stable bacterial hosts. Temperate phages integrate their genetic materials (prophage) and continue to vertically pass on along with the host divisions. Hence, lysogenic decision is called “stay” decision, as prophages stay as a part of the bacterial genome after the integration, or as a plasmid or linear genes independent from bacterial genome Dolgin (2019). Lysogens can integrate their genetic material at random or at specific sites, called “attachment sites”, via transposition or site recombination respectively. Others may not integrate their genes but save them externally as episome or linear genes that are expressed along with bacterial genes. The variable states of the lysogenic phage can be summarized in three main steps: establishment, maintenance and induction (Howard-Varona et al., 2017). Lysogenicity is dominating when the genes of the lytic proteins are blocked by lysis repressors (e.g CI, CII, CIII repressors). From the phenotype perspective, lysogenic phages usually appear in the form of turbid plaques of incomplete lysis on petri dishes.
2.2. Pseudo-lysogenic and chronic modes of action
Other than the best understood cycles, little is known about filamentous phages and pseudo-lysogenic. Pseudo-lysogeny is one of the phages –bacteria interaction modes that starts in unflavored growth conditions such as low nutrition levels (Cenens et al., 2013). Under stress, pseudo-lysogenic phages neither replicate by lysing the cells and spreading their progeny (lytic cycle), nor integrate and pass their genome during bacterial duplication (lysogenic cycle). Instead, the replication process is hindered due to the lack of cellular energy. After stalling in pseudo-lysogeny, phages can go back to one of the more common replication cycles once a more favorable environment is available Łoś and Pseudolysogeny (2012). In general, phages that follow pseudo-lysogenic cycle, like temperate phages, may offer mutualistic benefit packages between the phage and its bacterial host. Mutual factors will be discussed later.
Unlike all other replication modes, filamentous phages are parasites that do not disrupt the bacterial host. Lytic, lysogenic and pseudo-lysogenic phages become virulent at some point of the replication cycle, but it is not the case with chronic phages. For example, virulent phages are lytic once they enter the cell, while the lysogenic integrates and passes their genome with bacteria till it is activated (switch to lytic cycle). Even the pseudo-lysogenic phages can go back to lytic or lysogenic modes and become lytic under some conditions. However, chronic phages bud out of the bacterial membrane without causing cell lysis. Filamentous phages can build a peer to peer relationship with the bacterial host. In this relation, filamentous phages support the bacteria as they help in composing biofilms, and transferring toxins and other virulent factors to the bacterial host (Hay and Lithgow, 2019).
3. Lytic-lysogenic switch
Although lysogenic phages do not disrupt the bacterial cells, they are metaphorically considered a “time bomb” that will explode the bacterium in harsh or unsustainable conditions. Once the bacterial cells are induced, the phages activate the lysogenic to lytic switch program (Filipiak et al., 2020). Usually the word (phage “switch”) is referred to the transition from temperate phage mode into lytic mode. Another switch exists from lytic to lysogenic, but it is less studied (Williamson et al., 2002, Zhen et al., 2019, Paul, 2008). In this section, we will focus on the switch from lysogenic to lytic mode of action.
3.1. The cellular mechanism behinds the decision
Under physical or chemical cell stress, lysogenicity can be switched into lytic mode thorough a process called “induction”. Induction is an internal or external stress that causes DNA damage, which leads to changes in repressors of the lytic-lysogenic master regulator genes (Filipiak et al., 2020). For instance, during damaging stress, cell takes the SOS response in attempt to repair its DNA. In SOS repair response, RecA protein is activated and suppresses a number of repressors including the lytic repressors, which in turn leads to lysogenic to lytic switch activation (Campoy et al., 2006). While SOS dependent induction activates the bacterial SOS response, the induction can also take other SOS-independent routes. Both involve DNA damage yet each employs different downstream cascades (Modell et al., 2014).
Lambda (λ) temperate phage, as an example, can undergo lysogenic-to-lytic cycle switch in case of cell stress (e.g. temperature, pH change, bacterial growth levels, bacterial cell density, phage density, nutrients availability, chemical treatment, UV radiation) (Casjens and Hendrix, 2015, Choi et al., 2010). In another ecosystem, human microbiota can be affected by numerous external and internal body conditions such as the food type, species of bacterial infection and the antibiotic usage. Moreover, phages and phages communications are expected to be of a high manipulating effect on the mouse gut microbiota diversity (Cornuault et al., 2020), which suggests similar effect in human. Different induction methods have been analyzed to trigger the switch into lytic mode, as discussed in Table 1.
Table 1.
Summary of the inducing factors that triggers lysogenic-to-lytic switch.
| Types of induction | Inducing agents | The effect on lytic-lysogenic switch | References |
|---|---|---|---|
| Universal lysogenic inducer | Mitomycin-C | A positive control of the induction process which turns temperate phages into lytic after incubation for 5h to 7h. | (Cochran et al., 1998) |
| Physical stress | pH change | Change in the optimum pH lead to the lytic mode activation. The study highlighted pH of 5 to have higher induction compared to pH of 8, yet both pH's showed signs of inductions relative to the negative control (pH of 7). | (Choi et al., 2010) |
| Temperature change | In an appropriate range of temperature increase, the temperate phage favors the lytic mode. | (Choi et al., 2010, Schuster et al., 1973) | |
| UV- treatment | Activates Rec-A protease and leads to the lytic switch. | (Osterhout et al., 2007) | |
| Nutrient level | It is a controversial area, yet a study reported syndetic induction upon the additional nutrition and supplementary levels of phosphate with Mitomycin-C in cyanobacteria. | (McDaniel and Paul, 2005) | |
| Seasonal time | At seasons of low-productivity environmental conditions, lysogenic switch is prevalent. On the other hand, in other seasons with boosted environmental conditions, lysis is dominating. | (Williamson et al., 2002, Paul, 2008) | |
| Chemical stress | Organic carbon | When the carbon is consumed, the pH is lowered and phages are induced to go lytic switch. | (Choi et al., 2010) |
| Heavy metals | Relative concentration (<0.1mM) of Chlorine Cr (VI) activates SOS response and leads to activation of the lytic mode. | (Choi et al., 2010) | |
| Toxicity | Relative high concentration of Potassium Cyanate (0.15uM) leads to activation of the lytic mode. | (Choi et al., 2010) | |
| Chemical Oxygen Demand (COD) | The study showed no significant effect of COD variations on the number of phage particles (or VLPs). | (Choi et al., 2010) | |
| Bacterial stress | Cell density | It is a controversial factor (discussed in section 2.4) that leads to lysogenic activation. | (Zhen et al., 2019) |
| Biofilms | Phage induction increases the biofilm formation. | (Tan et al., 2020) | |
| Quorum sensing (QS) | In case of Vibrio cholerae (V. cholerae) and Enterococcus faecalis, QS induces phages to go lytic switch. | (Liang and Radosevich, 2019, Maxwell, 2019) | |
| Antibiotics | Quinolone and other antibiotics lead to lytic switch of phages. | (Zhang et al., 2000, Torres-Barceló, 2018) | |
| Nutrition style | Lysogenic mode is preferred in oligotrophic bacteria. | (Paul, 2008) | |
| Microbiota | Bile salts | Bile salts reduce the efficiency of lytic phages. | (Scanlan et al., 2019, Hernández et al., 2012) |
| Intestinal inflammation | Unlike normal conditions, during inflammation, oxidative species and other secretions encourage the induction of phages into lytic mode. | (Diard et al., 2017) | |
| Mouse gut | The study showed phage goes ultra-virulent inside mouse gut. Further experiments need to be done on human samples. | (Cornuault et al., 2020) |
3.2. The molecular mechanism of the induction
Lytic-lysogenic decision-making depends on the gene expression process that it is sometimes referred to as a “genetic switch” Maxwell (2019). For instance, the expression of some genes “lysis genes” activates the lysis mode; while their absence activates the lysogenic mode Young (2014). These bi-modes are activated or repressed by gene regulation of mainly two antagonistic proteins: CI lysis-repressor and Coi anti-repressor proteins (Chang et al., 2015, Savageau, 2013). CI represses the lysis process, while Coi inactivates CI and activates lysis mode accordingly (Fig. 1.e & 1. f). Throughout this section, we will talk about the bi-modes of lytic and lysogenic switch or what is called “stay or go decision”.
Fig. 2 XXXXX
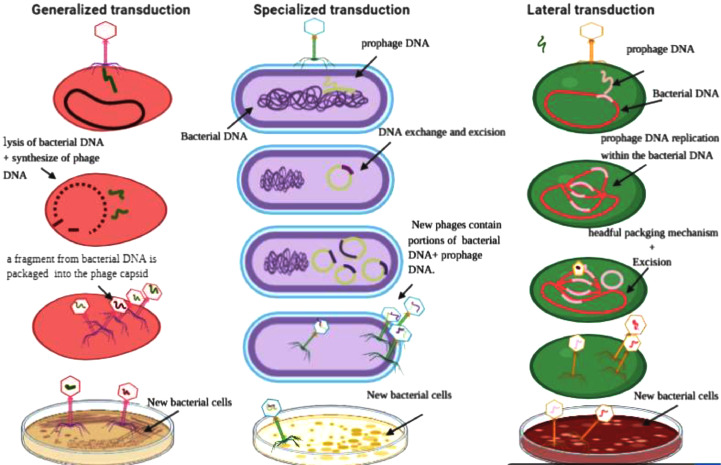
Fig. 2.
The three types of Transduction: Generalized transduction, Specialized transduction, and Lateral transduction adopted from (Griffiths et al., 2000).
Phage genome analysis has shown that lysogenicity phenotype appears due to the presence of a lysogen decision gene cluster. The cluster contains a number of known anti-lytic genes (e.g. CI repressor, anti-repressor, Cro-like repressor, integrase, and excisionase) (Chang et al., 2015). On a molecular level in cases of stress, the CI repressor gene is blocked by Coi anti-repressor due to gene regulation and results in activating the lysis program and prophage excision Savageau (2013). A molecular study has suggested that the regulation of both genes CI and Coi is dependent on environmental factors and the status of the bacterial host (Heinzel et al., 1992). Later, an experiment showed that phage p1 transduction in stationary phase- Escherichia coli (E. coli) bacteria at 42 °C might lead to induction of the P1 phage. It also detected alterations in the genes responsible for the maintenance of lysogenicity Ormaasen (2021).
3.3. The effect of lytic-lysogenic switch on the bacterial ecosystem
In bacteria–phage interactions, competition usually takes place. Ecologists are interested in proposing statistical models to describe and predict the bacteria-phage dynamics in the various ecosystems. For a long time, lytic phages were highly revisited and valued for their ability to shape the bacterial microecosystem through the lysis of the dominating bacteria to control their abundance. Later, other piece of puzzle was added to the picture by questioning the effect of lysogenic phages on the bacteria microecosystem. Temperate phages do not kill the bacteria but pass their genes along with bacterial genome. Moreover, they might benefit the bacteria by developing immunity or horizontally integrate a resistance genes of virulent factors into bacterial genome (Obeng et al., 2016). As the concept of lytic and lysogenic switch becomes clearer, increasing efforts are directed towards studying the effect of phage replication mode on the ecosystems with the aid of the mathematical models. Two hypnotized models are dominating the study of bacteria-phage dynamics: the oldest, Kill-the-Winner (KtW) model; and the relatively new paradigm, Piggyback-the-Winner (PtW) model (Chen et al., 2019).
For a long while, evolutionists believed in KtW model. The old dogma suggested that as the bacteria boost in certain ecosystem, phages viciously reproduce and lyse the bacterial hosts. In KtW theoretical model, the virus is the winner, hence it is the most active not just the abundant opponent that kills the dominating prey, the host bacteria Thingstad (2000). This was backed up by the fact that the phages are much abundant, almost 10 folds relative to the bacterial host cells. Phages do not lyse all bacterial cells due to some factors proposed by KtW supporters. Mainly, bacteria defend against phages by imposing genetic mutations that resist phage adherence and infection. As a consequence, the phages evolve to overcome the resistant barrier to invade the bacterial host. The coevolution process of phage and bacteria in response to each other is referred to as “arms race” (Hanifin et al., 2008) which leads to flourishing the diversity in the ecosystem (Thingstad, 2000, Thingstad and Lignell, 1997).
The recent discoveries in phage-bacteria dynamics could not be explained by the old version of KtW model. The raising studies show huge gap between the experimental findings and the theoretical calculations of KtW model. Unlike our previous knowledge of phages lytic behavior against the high density bacteria, studies revealed phages go lysogenic in cases of high density bacteria (Zhen et al., 2019, Chen et al., 2019). This suggests that phages do not kill the bacteria, but piggybacking them in order to pass the genome to other bacterial hosts. This finding shakes the ground of KtW model and establishes the new foundations for Piggyback the winner (PtW) model. While KtW speculates controlling the growth of the dominating bacteria in an ecosystem, PtW predicts increasing the bacterial biomass in the ecosystem (McDole et al., 2012).
PtW model could reconcile experimental findings which KtW failed to explain. Moreover, Bacteriophage Adherence to Mucus (BAM) model reviles many facts about bacteria-phage interactions. BAM model studies the phage dynamics and behavior in response to the microbes in mucus (e.g, in mucins of coral reefs, lung and gut mucosa) (Almeida et al., 2019). In BAM model, a phage concentration gradient is observed, where phages are more abundant in the middle mucosal layer, which is mucus rich, relative to the upper layer. Whereas, the bacterial gradient is the opposite, higher in the upper layer and lower in the middle. Recent studies showed that phages in the upper layer prefer the lysogenic mode of action, which is reconciled with PtW model due to the high bacterial abundance. Reversely, the phages in the middle layer with lower bacteria count experience induction and lytic mode of action (Silveira and Rohwer, 2016). To date, the debate between both theoretical models is ongoing. KtW supporters try to update and consider several factors that solve the weakness and gaps in their model (Xue and Goldenfeld, 2017).
3.4. Approaches to study the lytic-lysogenic switch
Three approaches are used to study the lytic/ lysogenic switch: traditional, genome sequencing and advanced experimental approaches. Traditional approaches are usually performed after isolating, propagating and structurally characterizing the phage, where lysogenic test is done to verify the phage replication mode (Bae et al., 2006). Then, induction experiment is done to quantify the phage particles lysed. Usually the universal inducer, Mitomycin-C, is used in the induction (Cochran et al., 1998). The induction process is essential to differentiate the phage forming lysogens from the phage for which bacteria are resistant. Then, phages particles and burst size are calculated. However, many issues are found in the traditional approaches. Other issues are reviewed by Howard-Varona et al. (2017).
Sequencing approaches can minimize the weakness and uncertainties of the traditional approach. They include scanning prophage sequencing; whole microbial metagenomics (Waller et al., 2014) or even single cell genomes. Then, the results are tested against virome databases (Sutton and Hill, 2019, Rampelli et al., 2016, Elbehery et al., 2018, Gregory et al., 2019). Otherwise, researches might scan for marker genes which are specific for lytic or lysogenic modes of action (Schmidt et al., 2014). Then, results are compared to the literature. However, sequencing approaches have limitations in the data availability and they also need further verifications using experimental evidences.
Advanced experimental approaches rely on more trusted methodology, unlike the traditional approaches. More efforts are devoted to develop models and systems which are capable of mimicking the phage behavior in ecosystems. In addition, increasing the resolution of the detecting tools to capture a florescent labeled phages and marker genes in single cell (Cenens et al., 2013, Dang and Sullivan, 2014).
4. History of phage cheating
One of the key roles of phages is to balance the bacterial population in every shared environment. Their abundance in biosphere is estimated to be around 1030–1032 virions (Ashelford et al., 2003, Weinbauer, 2004). They were identified as a vehicle for different genes in the environment since they act as a vector for genes substitution in a process known as transduction. Phages have four types of life cycles, however, transduction commonly happens through either the lytic cycle or the lysogenic cycle (Chiang et al., 2019, Zinder, 1992)
Genetic transduction is one of the main evolutionary methods which used by the bacteria to adapt with a lot of natural and artificial stresses. Transduction term was discovered and announced by Norton Zinder and Joshua Lederberg in 1952 (Chiang et al., 2019, Zinder and Lederberg, 1952), when they were investigating through an experiment on Salmonella typhimurium if the conjugation occurs only in E. coli or also in other types of bacteria Zinder (1992). They used two various strains: one was met− his− and the other was phe− trp− tyr−. These mutations negatively affect the nutrition of each strain (Griffiths et al., 2000). When they cultured separately, each strain on media that contained only inorganic salts, a carbon source, and water, the wild type cells have shown no growth, but the growth was observed when both strains were mixed showing that recombination has taken place in between Salmonella typhimurium strains. However, a U-shaped glass tube, that consisted from two arms were also filled separately with the two Salmonella typhimurium strains and supplied with a bacterial sterile filters of different sizes to prohibit any physical connection between the bacterial cells of the two different strains to ensure that if the conjugation is responsible for the recombination or not. It was concluded that the temperate bacteriophage p22 was responsible for horizontal gene transfer (HGT). Consequently, this process was called transduction (Zinder and Lederberg, 1952). Zinder and Lederberg have discovered the first type of transduction is called generalized transduction where the phage can take any part of the host's genome and pack it in the phage virion, with 0.1% possibility (Griffiths et al., 2000). Then after four years the second type of transduction was discovered in E. coli that took place by λ phage in 1956 and named specialized transduction (Morse et al., 1956). In specialized transduction, the phage cleaves specific portions from host genome, particularly; regions surround the prophage integration site. The last transduction phenomenon was explored in Staphylococcus aureus (S. aureus) and was called lateral transduction Davidson (2018). It occurs through a temperate phage that excises late in its atypical lytic cycle (Chiang et al., 2019). Unusually, phage replicates first, then, starts the excision process from the bacterial genome. This type is distinguished from the other types as being able to transfer large DNA stretches at high frequencies in addition to the prophage (Chen et al., 2018).
5. Evolution of pathogenic bacteria
Virtually, phages may carry genes that help their hosts to adapt and to stabilize by transferring new traits to them. Shiga toxins (Stx) are toxins expressed by genes found in the lambdoid prophages and were produced by different bacterial strains but the most popular one is Shiga toxin-producing E. coli (STEC) that cause human illness and death (Liu, 2014, Schnaar and Lopez, 2018). It was found that various Enterobacter cloacae and E. coli strains acquired new Shiga toxin genes (e.g. Stx1e and Stx1a) which are originally produced by Enterobacter cloacae M12 × 01451 strain and E. coli O157:H7 strain; RM8530. Stx1a- and Stx1e-temperate converting phages were responsible for this gene transfer process (Khalil et al., 2016). Biofilm formation is also associated with HGT. Moreover, Anthrax, which is a serious infectious disease caused by Bacillus anthracis, used to have no capability to survive completely outside the mammalian hosts (Schuch and Fischetti, 2009). However, phages increased its chance to cause illness for humans and animals through introducing a phenotypic changes that enabled them to survive as dormant spores outside their original host, and potentiated their endosymbiotic capabilities (Schuch and Fischetti, 2009). Furthermore, Cholera is an epidemic disease caused by V. cholera bacterium (Das et al., 2011). Some environmental non-disease causing strains acquired virulence genes, e.g., cholera toxin (CT) and toxin coregulated pilus (TCP), via temperate bacteriophage CTXφ (Faruque and Mekalanos, 2012).
5.1. Bacteriophages contribute to the development of antibiotic resistance
Antibiotics have been used for a while to treat bacterial infections since the ancient Egyptians. They, applied moldy bread to infected wounds. In 1909, Paul Ehrlich discovered a chemical agent called arsphenamine that was effective chemotherapy in the treatment of the bacterial infection (syphilis) (Valent et al., 2016). Then, Alexander Fleming discovered accidentally the first antimicrobial compound: penicillin Fleming (1979). By the time, the scientists proved the effectiveness of antibiotics in reducing the morbidity and mortality of humans but soon it became dangerous on human public health through generation of multi-drug resistance (MDR) bacteria phenomenon. It was not anticipated that microbes would overcome these various chemical compounds and adapted with them. However, this adaptation have been achieved through many resistance mechanisms: (i) Permeability changes in the bacterial cell wall/membrane which restrict antimicrobial access to target sites; (ii) Vigorous efflux of the antimicrobial from the cell; (iii) Mutation in the target site; (iv) Enzymatic alteration or disintegration of the antimicrobial; (v) Gaining of alternative metabolic pathways to those inhibited by the drug (McDermott et al., 2003). There is various phenotype of resistance: including intrinsic resistance (also named insensitivity) which is the innate ability of a bacterial species to resist the activity of a particular antimicrobial agent through its inherent structural or functional characteristics. It allows a tolerance of particular antimicrobial drugs that have never been susceptible to them. While the other resistance pattern, the acquired resistance, occurs when the microorganism obtains the Antibiotic Resistance (AR) genes via mutation of genes involved in normal physiological processes and cellular structures as in Mycobacterium tuberculosis which is resistant to rifamycins. This resistance is achieved by point mutations in the rifampin-binding region of rpoB (Smith et al., 2013), and also via (HGT) mechanism (bacteriophage transduction) as shown in:
5.1.1. Methicillin-resistant Staphylococcus aureus (MRSA)
S. aureus is a pathogenic bacterium that causes sepsis, toxic shock syndrome and necrotizing pneumonia in humans. They acquired resistance to methicillin by transferring of mecA gene among staphylococci via generalized transduction mediated by lysogenic phage. mecA gene is carried on Staphylococcal Cassette Chromosome mec (SCCmec) which codes for penicillin-binding proteins (PBPs). PBP 2a is not sensitive to ß-lactam inhibition. In 1970, Cohen and Sweeney reported that the ideal transfer occurs only to S. aureus recipients that had a prophage (Cohen and Sweeney, 1970). Although SCCmec can be transferred by homologous recombination due to the ccr-recombinase gene products encoded within SCCmec or by ccr-mediated recombination (Katayama et al., 2000), there is a recent study suggests that the acquisition of SCCmec is due to homologous recombination since ccr-mediated recombination requires complementary SCCmec flanking sequences. The study has also reported the importance of the chromosomal integration and expression for effective chromosomal gene transfer (Scharn et al., 2013).
5.1.2. Vancomycin-resistant enterococcus faecium
Enterococcus faecium has intrinsic resistance to β-lactams via mutations in the penicillin-binding protein-5 (PBP5) which is encoded by a horizontally transferred gene. WHO has reported the limitation of the drugs that can cure or eliminate infections with Enterococcus faecium which has acquired resistance to vancomycin through obtaining one of two linked gene clusters VanA and VanB. These gene clusters code for enzymes that modify peptidoglycan precursor, decreasing its affinity to vancomycin. These genes are acquired by HGT including transduction via bacteriophages (Delpech et al., 2019).
5.1.3. Multidrug-resistant salmonella typhimurium
The generalized transduction and specialized transduction help in acquiring and transferring the resistance genes among the Salmonella serotypes. Salmonella Typhimurium DT104 is considered as an epidemic strain due to their ability to show multi-drug resistant (Mather et al., 2013). Phages have accelerated their resistance progress. phage PDT17 that is emitted by S. Typhimurium DT104 and P22-like phage ES18 have introduced many antibiotic resistance genes: ampicillin (amp), chloramphenicol (cam), sulfonamide (sul) and streptomycin (str) (Schmieger and Schicklmaier, 1999). Carbadox-induced phages transfere tetracycline gene (tet) from S. Typhimurium DT104 NCTC13348 to another S. Typhimurium DT104 strain (Bearson et al., 2014). P24 phage has already mediated the transduction of antibiotic resistance genes: beta-lactamase (blaCMY-2) and tetracycline resistance genes (tetA and tetB) from S. Heidelberg S25 to S. Typhimurium MZ1262 (Zhang and LeJeune, 2008), while Fluoroquinolone-induced phages have transferred the plasmid that has kanamycin resistance genes from S. Typhimurium DT120 (BBS 1162) and S. Typhimurium DT104 (BBS 1170) to S. Typhimurium BBS 243 (B.L. and B.W., 2015).
5.2. In epidemic conditions
5.2.1. Cholera
As mentioned above, Cholera is caused by pathogenic V. cholera (Das et al., 2011, Faruque et al., 1998, Kaper et al., 1995). Researchers have shown that the transmission of V. cholerae is due to its presence of the infectious organism in the stools of cholera patients (Merrell et al., 2002). Phages could play a dual role in cholera epidemic: lytic phages can control the spread of cholera pathogens in water or food, and also can contributes in the evolution of pathogenic V. cholera and affect the dynamics of cholera epidemic Faruque (2013).
5.3. The impacts of bacteriophage in dairy industry
Phages are not only enemies to pathogenic bacteria but also may be enemies to human. Although phages could contribute in maintaining our health, they may be menacing our food industry which reflects badly on our health and economy. The dairy industry -which is based on the use of bacteria in making products such as cheeses, buttermilk, and sour cream- is threatened by virulent phages whose presence result in fermentation failure since they invade the lactic acid bacteria (LAB) and kill them. The sources and causes of phage contamination are various: (a) Raw ingredients e.g., the raw milk which is a well-known container of phages; (b) reusing of ingredients such as whey proteins; (c) via aerosols throughout the working area (Verreault et al., 2011); (d) invalid sanitation procedures e.g., bad handling of material, unsafe movement of employees; (e) using contaminated starter cultures (Bacteriophages of lactic acid bacteria and their impact on milk fermentations). Although the presence of prophages in the bacterial strains may help in good-quality cheese production through the expression of prophage-encoded endolysins which in turn could activate autolysis and the release of intracellular flavor-generating enzymes, prophages could contribute in generating of new lytic phages eliminating the bacterial resistance (Labrie and Moineau, 2007, Lepeuple et al., 1998).
5.4. Phages as a vaccine
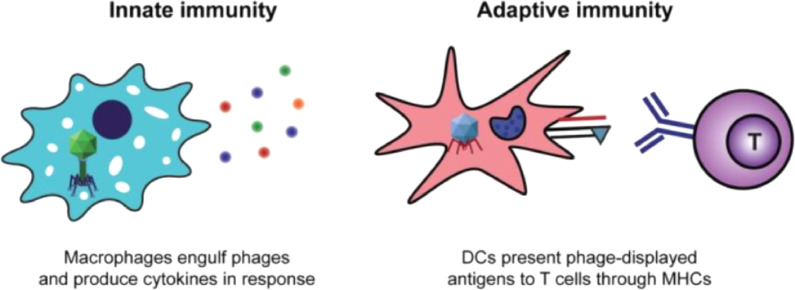
Phages can trigger the innate and adaptive immune systems of the organism against several pathogens such as bacteria and viruses. Induction of the pattern recognition receptors (PRRs), which is important in innate immune system, takes place in the host cells that recognize pathogen-associated molecular patterns (PAMPs) (Oakes et al., 2019). Toll‐like receptors (TLRs) which is a type of PRRs, can be activated by the phages (Krut and Bekeredjian-Ding, 2018). Upon activation, TLRs generate inflammatory signals such as cytokines that in turn control the evolution of the adaptive immune response (Hess and Jewell, 2020). Phages can also induce the adaptive immune response by interacting with the antigen‐presenting cells (APC) that lead to forming major histocompatibility complexes (MHCs) on the surface of (APC) cells (Fig. 3) (Tao et al., 2019). these findings made the use of phages as a delivery system tool for foreign antigens is possible and opened new gates for phages, such as filamentous phages, T4 phage, T7 phage, λ phage, MS2, and Qβ phage, to contribute in the development of vaccines for diseases by incorporating the gene of foreign peptide or protein (antigen) into their coat protein genes and expressing them on their surfaces (Smith and Petrenko, 1997).
Fig. 3.
Phages induce both innate and adaptive immune responses to antigens (Tao et al., 2019).
5.4.1. Anthrax and plague diseases
Anthrax and plague are epidemic diseases that has been used as a biological weapon (Whitby et al., 2002). As mentioned above, Anthrax is caused by Bacillus anthracus bacteria, and it was the first bacterial disease that infect humans and animals. However, plague is caused by Yersinia pestis bacteria and the disease has caused around 25–40 million death cases in Europe Brightman (2019). Since there was no approved or developed vaccine for plague but there was an authorized one for Anthrax, the scientists created one vaccine formula for Anthrax and plague together using T4 phage as a delivery system of their antigens. The two genes of the antigens were incorporated into the capsid protein Soc (9 kDa). T4 phage-based vaccine has proved its efficiency in the elimination of anthrax and plague diseases in animal models and has shown high efficiency compared to the other vaccines. In addition, supportive agent wasn't needed to provoke T-helper cells in the 1st and 2nd immune responses (Tao et al., 2018).
5.4.2. Zika disease
Zika is an epidemic disease that caused by flavivirus and transmitted by mosquito. Zika virus was found in East Africa in 1947 (Gubler et al., 2017). In 2007, it was the first year in which Zika virus was reported as an outbreak that occurred in Island of Yap (Federated States of Micronesia) (World Health Organization 2018). The virus had a group of envelope protein epitopes that contributed in the development of vaccines. The researchers elicited the immunity against the virus through the use of various phages (e.g., Qβ, MS2, and PP7) which acted similar to the nanoparticles that carries virus epitopes. The results have shown that phages mixture which contains various virus epitopes is effective in the reduction of infection more than using of just one epitope with phages in an experiment applied to mice (Basu et al., 2018).
5.4.3. Influenza
Influenza is a viral disease affects the respiratory system of the organism menacing the public health. Latest studies constructed a T7 phage vaccine against influenza A by engineering the phage to display M2e protein on its surface to evoke the production of IgG2a and IgG1 antibodies against influenza A virus. This vaccine has proved its effectiveness on health and economic levels (Hashemi et al., 2012).
5.4.4. Hepatitis B
Hepatitis B is a disease caused by the hepatitis B virus and transmits from an infected person to another through body fluids such as the blood or semen. The researchers designed a vaccine consisting of the λ phage-displayed hepatitis B antigen (λ-HBsAg) (March et al., 2004).
5.4.5. COVID-19
COVID-19 is a pandemic disease caused by SARS-CoV-2 that struck firstly in Wuhan, China in December 2019. As a consequence of its outbreak, Wuhan was isolated from other provinces. According to European Centre for Disease Prevention and Control, the confirmed cases until 2ed of September 2020 reached 25,983,958 cases involving 862,568 deaths (COVID-19 Situation Update Worldwide, as of 2ed of September 2020, n.d.). This virus has become a universal challenge and each country is racing to discover the vaccine to stem the outbreaks of the virus. Like most of single-stranded RNA viruses, SARS-CoV-2 is more susceptible to mutations (Korber et al., 2020). Its virion size is 60 nm and can extend to be 140 nm in diameter when considering its surface spike (S) protein Singhai (2020). S protein is considered the virus key to invade the human host cells through binding to angiotensin-converting enzyme 2 (ACE2) receptors. Subunits S1 are responsible for receptor binding and S2 are responsible for virion fusion (Scudellari, 2020, Shang et al., 2020). In a recent study, researchers have affirmed the essential role of phage display in determining of SARS-CoV-2 antibodies by using it to display a single-domain antibody library. After the different rounds of bio-panning, they recognized human single-domain antibodies for five distinct epitopes on SARS-CoV-2 receptor-binding domain (RBD) which is able to attack the cryptic epitope that positioned in the spike trimeric interface to suppress SARS-CoV-2 (Wu et al., 2020).
6. Conclusion
Phage lifecycle decision has a great impact on many aspects of determining their use, host pathogenesis, and ecosystem understanding. Many questions need future investigation, starting from the lytic-lysogenic switch decision, such as the study approaches used in detecting switch decision. In general, the techniques used in studying lytic-lysogenic switch will revolutionize our understanding of phages replication modes in the real and complex systems. Investment in the methodology approaches will cascade our knowledge about phage behavior and advance our capabilities of using phages in several applications. Further research into the surprising variation of temperate phages impact on their host evolution should elucidate aspects of antimicrobial resistance and bacterial adaptation to surrounding environment. While phage display is a promising method that has been already investigated for eradication of many infections, its role in immunization or vaccination could help in solving the current situation the world face against COVID-19 disease.
Funding
This research was funded by the Egyptian Sciences and Technology Development Fund (STDF), grant #41909 and Zewail City of Science and Technology internal grant No. ZC 019-2019.
Declaration of Competing Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a possible conflict of interest.
References
- Łoś M, Węgrzyn G. Pseudolysogeny. Adv. Virus Res. 2012;82:339–349. doi: 10.1016/B978-0-12-394621-8.00019-4. Academic Press Inc. [DOI] [PubMed] [Google Scholar]
- Almeida GM, Laanto E, Ashrafi R, Sundberg L-R. Bacteriophage adherence to mucus mediates preventive protection against pathogenic bacteria. BioRxiv. 2019 doi: 10.1101/592097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ashelford KE, Day MJ, Fry JC. Elevated abundance of bacteriophage infecting bacteria in soil. Appl. Environ. Microbiol. 2003;69:285–289. doi: 10.1128/AEM.69.1.285-289.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- B.L. B, B.W. B. Fluoroquinolone induction of phage-mediated gene transfer in multidrug-resistant Salmonella. Int. J. Antimicrob. Agents. 2015 doi: 10.1016/j.ijantimicag.2015.04.008. [DOI] [PubMed] [Google Scholar]
- Bae T, Baba T, Hiramatsu K, Schneewind O. Prophages of staphylococcus aureus Newman and their contribution to virulence. Mol. Microbiol. 2006 doi: 10.1111/j.1365-2958.2006.05441.x. [DOI] [PubMed] [Google Scholar]
- Basu R, Zhai L, Contreras A, Tumban E. Immunization with phage virus-like particles displaying Zika virus potential B-cell epitopes neutralizes Zika virus infection of monkey kidney cells. Vaccine. 2018 doi: 10.1016/j.vaccine.2018.01.056. [DOI] [PubMed] [Google Scholar]
- Bearson BL, Allen HK, Brunelle BW, Lee IS, Casjens SR, Stanton TB. The agricultural antibiotic carbadox induces phage-mediated gene transfer in Salmonella. Front. Microbiol. 2014 doi: 10.3389/fmicb.2014.00052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brightman C. Plague and anthrax: diagnosis and management. Trends Urol. Men's Heal. 2019;10:27–30. [Google Scholar]
- Brown-Jaque M, Calero-Cáceres W, Muniesa M. Transfer of antibiotic-resistance genes via phage-related mobile elements. Plasmid. 2015;79:1–7. doi: 10.1016/j.plasmid.2015.01.001. [DOI] [PubMed] [Google Scholar]
- Campoy S, Hervàs A, Busquets N, Erill I, Teixidó L, Barbé J. Induction of the SOS response by bacteriophage lytic development in Salmonella enterica. Virology. 2006;351:360–367. doi: 10.1016/j.virol.2006.04.001. [DOI] [PubMed] [Google Scholar]
- Casjens SR, Hendrix RW. Bacteriophage lambda: early pioneer and still relevant. Virology. 2015;479–480:310–330. doi: 10.1016/j.virol.2015.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cenens W, Makumi A, Mebrhatu MT, Lavigne R, Aertsen A. Phage–host interactions during pseudolysogeny. Bacteriophage. 2013;3:e25029. doi: 10.4161/bact.25029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang Y, Shin H, Lee JH, Park CJ, Paik SY, Ryu S. Isolation and genome characterization of the virulent Staphylococcus aureus bacteriophage SA97. Viruses. 2015 doi: 10.3390/v7102870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chanishvili N. Bacteriophages as therapeutic and prophylactic means: summary of the soviet and post Soviet experiences. Curr. Drug. Deliv. 2016 doi: 10.2174/156720181303160520193946. [DOI] [PubMed] [Google Scholar]
- Chen J, Quiles-Puchalt N, Chiang YN, Bacigalupe R, Fillol-Salom A, Chee MSJ, et al. Genome hypermobility by lateral transduction. Science (80-) 2018;362:207–212. doi: 10.1126/science.aat5867. [DOI] [PubMed] [Google Scholar]
- Chen X, Ma R, Yang Y, Jiao N, Zhang R. Viral regulation on bacterial community impacted by lysis-lysogeny switch: a microcosm experiment in eutrophic coastal waters. Front. Microbiol. 2019;10:1763. doi: 10.3389/fmicb.2019.01763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiang YN, Penadés JR, Chen J. Genetic transduction by phages and chromosomal islands: the new and noncanonical. PLoS Pathog. 2019 doi: 10.1371/journal.ppat.1007878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi J, Kotay SM, Goel R. Various physico-chemical stress factors cause prophage induction in Nitrosospira multiformis 25196- an ammonia oxidizing bacteria. Water Res. 2010 doi: 10.1016/j.watres.2010.04.040. [DOI] [PubMed] [Google Scholar]
- Cochran PK, Kellogg CA, Paul JH. Prophage induction of indigenous marine lysogenic bacteria by environmental pollutants. vol. 164. 1998.
- Cohen S, Sweeney HM. Transduction of methicillin resistance in staphylococcus aureus dependent on an unusual specificity of the recipient strain. J. Bacteriol. 1970 doi: 10.1128/jb.104.3.1158-1167.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornuault JK, Moncaut E, Loux V, Mathieu A, Sokol H, Petit MA, et al. The enemy from within: a prophage of Roseburia intestinalis systematically turns lytic in the mouse gut, driving bacterial adaptation by CRISPR spacer acquisition. ISME J. 2020;14:771–787. doi: 10.1038/s41396-019-0566-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dang VT, Sullivan MB. Emerging methods to study bacteriophage infection at the single-cell level. Front. Microbiol. 2014 doi: 10.3389/fmicb.2014.00724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das B, Bischerour J, François-Xavier B. VGJφ integration and excision mechanisms contribute to the genetic diversity of Vibrio cholerae epidemic strains. Proc. Natl. Acad. Sci. USA. 2011 doi: 10.1073/pnas.1017061108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davidson AR. A common trick for transferring bacterial DNA. Science. 2018 doi: 10.1126/science.aav1723. (80-) [DOI] [PubMed] [Google Scholar]
- Delpech G, Allende LG, Sparo M. Mobile genetic elements in vancomycin-resistant enterococcus faecium population. In Vancomycin-Resistant Enterococcus. 2019 [Google Scholar]
- Diard M, Bakkeren E, Cornuault JK, Moor K, Hausmann A, Sellin ME, et al. Inflammation boosts bacteriophage transfer between Salmonella spp. Science. 2017;355:1211–1215. doi: 10.1126/science.aaf8451. (80-) [DOI] [PubMed] [Google Scholar]
- Dolgin E. The secret social lives of viruses. Nature. 2019;570:290–292. doi: 10.1038/D41586-019-01880-6. [DOI] [PubMed] [Google Scholar]
- El-Shibiny A, Dawoud A. In: Biocommunication of Phages. 1st ed. Witzany G, editor. Springer International Publishing; 2020. Bacteriophage applications for food safety; pp. 463–484. [Google Scholar]
- Elbehery AHA, Feichtmayer J, Singh D, Griebler C, Deng L. The human virome protein cluster database (HVPC): a human viral metagenomic database for diversity and function annotation. Front. Microbiol. 2018;9:1110. doi: 10.3389/fmicb.2018.01110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faruque SM, Mekalanos JJ. Phage-bacterial interactions in the evolution of toxigenic Vibrio cholerae. Virulence. 2012 doi: 10.4161/viru.22351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faruque SM, Albert MJ, Mekalanos JJ. Epidemiology, genetics, and ecology of toxigenicvibrio cholerae. Microbiol. Mol. Biol. Rev. 1998 doi: 10.1128/mmbr.62.4.1301-1314.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faruque SM. In: Cholera Outbreaks. Nair GB, Takeda Y, editors. Springer; Berlin, Heidelberg: 2013. Role of phages in the epidemiology of cholera; pp. 165–180. [Google Scholar]
- Filipiak M, Łoś JM, Łoś M. Efficiency of induction of Shiga-toxin lambdoid prophages in Escherichia coli due to oxidative and antibiotic stress depends on the combination of prophage and the bacterial strain. J. Appl. Genet. 2020;61:131–140. doi: 10.1007/s13353-019-00525-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fleming A. On the antibacterial action of cultures of a penicillium, with special reference to their use in the isolation of B. influenzae. Br. J. Exp. Pathol. 1979 [PMC free article] [PubMed] [Google Scholar]
- Gómez-Gómez C, Blanco-Picazo P, Brown-Jaque M, Quirós P, Rodríguez-Rubio L, Cerdà-Cuellar M, et al. Infectious phage particles packaging antibiotic resistance genes found in meat products and chicken feces. Sci. Rep. 2019;9:1–11. doi: 10.1038/s41598-019-49898-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gregory A, Zablocki O, Howell A, Bolduc B, Sullivan M. The human gut virome database. BioRxiv. 2019 doi: 10.1101/655910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffiths AJ, Wessler SR, Lewontin RC, Gelbart WM, Suzuki DT, Griffiths JHMJ, et al. Transduction. An. Introd. Genet. Anal. 2000 [Google Scholar]
- Gubler DJ, Vasilakis N, Musso D. History and emergence of Zika virus. J. Infect. Dis. 2017 doi: 10.1093/infdis/jix451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanifin CT, Brodie ED, Brodie ED. Phenotypic mismatches reveal escape from arms-race coevolution. PLoS Biol. 2008;6:e60. doi: 10.1371/journal.pbio.0060060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hashemi H, Pouyanfard S, Bandehpour M, Noroozbabaei Z, Kazemi B, Saelens X, et al. Immunization with M2e-Displaying T7 bacteriophage nanoparticles protects against influenza a virus challenge. PLoS One. 2012 doi: 10.1371/journal.pone.0045765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hay ID, Lithgow T. Filamentous phages: masters of a microbial sharing economy. EMBO Rep. 2019;20 doi: 10.15252/embr.201847427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorsten Heinzel, Mathias Velleman and HS. The c1 repressor inactivator protein coi of bacteriophage P1. Cloning and expression of coi and its interference with c1 repressor function. 1992. [PubMed]
- Hernández SB, Cota I, Ducret A, Aussel L, Casadesús J. Adaptation and Preadaptation of salmonella enterica to bile. PLoS Genet. 2012;8 doi: 10.1371/journal.pgen.1002459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hess KL, Jewell CM. Phage display as a tool for vaccine and immunotherapy development. Bioeng. Transl. Med. 2020 doi: 10.1002/btm2.10142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard-Varona C, Hargreaves KR, Abedon ST, Sullivan MB. Lysogeny in nature: mechanisms, impact and ecology of temperate phages. ISME J. 2017;11:1511–1520. doi: 10.1038/ismej.2017.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaper JB, Jr JGM, Levine MM. Cholera. Clin. Microbiol. Rev. 1995:48–86. doi: 10.1128/cmr.8.1.48. 8th ed. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katayama Y, Ito T, Hiramatsu K. A new class of genetic element, staphylococcus cassette chromosome mec, encodes methicillin resistance in Staphylococcus aureus. Antimicrob. Agents Chemother. 2000 doi: 10.1128/AAC.44.6.1549-1555.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khalil RK, Skinner C, Patfield S, He X. Phage-mediated Shiga toxin (Stx) horizontal gene transfer and expression in non-Shiga toxigenic Enterobacter and Escherichia coli strains. Pathog Dis. 2016;74 doi: 10.1093/femspd/ftw037. [DOI] [PubMed] [Google Scholar]
- Korber B, Fischer W, Gnanakaran SG, Yoon H, Theiler J, Abfalterer W, et al. Spike mutation pipeline reveals the emergence of a more transmissible form of SARS-CoV-2. BioRxiv. 2020 doi: 10.1101/2020.04.29.069054. [DOI] [Google Scholar]
- Krut O, Bekeredjian-Ding I. Contribution of the immune response to phage therapy. J. Immunol. 2018 doi: 10.4049/jimmunol.1701745. [DOI] [PubMed] [Google Scholar]
- Labrie SJ, Moineau S. Abortive infection mechanisms and prophage sequences significantly influence the genetic makeup of emerging lytic lactococcal phages. J. Bacteriol. 2007 doi: 10.1128/JB.01111-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lepeuple AS, Van Gemert E, Chapot-Chartier MP. Analysis of the bacteriolytic enzymes of the autolytic Lactococcus lactis subsp. cremoris strain AM2 by renaturing polyacrylamide gel electrophoresis: identification of a prophage-encoded enzyme. Appl. Environ. Microbiol. 1998 doi: 10.1128/aem.64.11.4142-4148.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang X, Radosevich M. Commentary: a host-produced quorum-sensing autoinducer controls a phage lysis-lysogeny decision. Front. Microbiol. 2019;10:1201. doi: 10.3389/fmicb.2019.01201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu D. Diarrhoeagenic Escherichia coli. Mol. Med. Microbiol. Second Ed., 2014 doi: 10.1016/B978-0-12-397169-2.00064-0. [DOI] [Google Scholar]
- M Tulio Pardini G, Silva B L, Aguiar A LA, Elisa Soto L M. Bacteriophage genome sequencing: a new alternative to understand biochemical interactions between prokaryotic cells and phages. J. Microb. Biochem. Technol. 2017 doi: 10.4172/1948-5948.1000362. [DOI] [Google Scholar]
- March JB, Clark JR, Jepson CD. Genetic immunisation against hepatitis B using whole bacteriophage lambda particles. Vaccine. 2004;22:1666–1671. doi: 10.1016/j.vaccine.2003.10.047. [DOI] [PubMed] [Google Scholar]
- Mather AE, Reid SWJ, Maskell DJ, Parkhill J, Fookes MC, Harris SR, et al. Distinguishable epidemics of multidrug-resistant Salmonella typhimurium DT104 in different hosts. Science. 2013 doi: 10.1126/science.1240578. (80-) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maxwell KL. Phages tune in to host cell quorum sensing. Cell. 2019;176:7–8. doi: 10.1016/j.cell.2018.12.007. [DOI] [PubMed] [Google Scholar]
- McDaniel L, Paul JH. Effect of nutrient addition and environmental factors on prophage induction in natural populations of marine Synechococcus species. Appl. Environ. Microbiol. 2005;71:842–850. doi: 10.1128/AEM.71.2.842-850.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDermott PF, Walker RD, White DG. Antimicrobials: modes of action and mechanisms of resistance. Int. J. Toxicol. 2003;22:135–143. doi: 10.1080/10915810305089. [DOI] [PubMed] [Google Scholar]
- McDole T, Nulton J, Barott KL, Felts B, Hand C, Hatay M, et al. Assessing coral reefs on a pacific-wide scale using the microbialization score. PLoS One. 2012;7 doi: 10.1371/journal.pone.0043233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merrell DS, Butler SM, Qadri F, Dolganov NA, Alam A, Cohen MB, et al. Host-induced epidemic spread of the cholera bacterium. Nature. 2002;417:642–645. doi: 10.1038/nature00778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Modell JW, Kambara TK, Perchuk BS, Laub MT. A DNA damage-induced, SOS-independent checkpoint regulates cell division in caulobacter crescentus. PLoS Biol. 2014;12 doi: 10.1371/journal.pbio.1001977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morse ML, Lederberg EM, Lederberg J. Transduction in Escherichia coli K-12. Genetics. 1956;41:142. doi: 10.1093/genetics/41.1.142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oakes RS, Froimchuk E, Jewell CM. Engineering Biomaterials to Direct Innate Immunity. Adv. Ther. 2019 doi: 10.1002/adtp.201800157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Obeng N, Pratama AA, Elsas JD van. The significance of mutualistic phages for bacterial ecology and evolution. Trends Microbiol. 2016;24:440–449. doi: 10.1016/j.tim.2015.12.009. [DOI] [PubMed] [Google Scholar]
- Ormaasen I. An investigation of wild-type P1 prophage induction. n.d. 2021.
- Osterhout RE, Figueroa IA, Keasling JD, Arkin AP. Global analysis of host response to induction of a latent bacteriophage. BMC Microbiol. 2007;7:82. doi: 10.1186/1471-2180-7-82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul JH. MINI-REVIEW Prophages in marine bacteria: dangerous molecular time bombs or the key to survival in the seas? ISME J. 2008;2:579–589. doi: 10.1038/ismej.2008.35. [DOI] [PubMed] [Google Scholar]
- Penadés JR, Chen J, Quiles-Puchalt N, Carpena N, Novick RP. Bacteriophage-mediated spread of bacterial virulence genes. Curr. Opin. Microbiol. 2015;23:171–178. doi: 10.1016/j.mib.2014.11.019. [DOI] [PubMed] [Google Scholar]
- Rakonjac J, Bennett NJ, Spagnuolo J, Gagic D, Russel M. Filamentous bacteriophage: biology, phage display and nanotechnology applications. Curr. Issues Mol. Biol. 2011 doi: 10.21775/cimb.013.051. [DOI] [PubMed] [Google Scholar]
- Rakonjac J, Russel M, Khanum S, Brooke SJ, Rajič M. Filamentous phage: structure and biology. Adv. Exp. Med. Biol. 2017;1053:1–20. doi: 10.1007/978-3-319-72077-7_1. Springer New York LLC. [DOI] [PubMed] [Google Scholar]
- Rampelli S, Soverini M, Turroni S, Quercia S, Biagi E, Brigidi P, et al. ViromeScan: a new tool for metagenomic viral community profiling. BMC Genomics. 2016;17:165. doi: 10.1186/s12864-016-2446-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savageau MA. Phenotypes and design principles in system design space. Handb. Syst. Biol. 2013:287–310. doi: 10.1016/B978-0-12-385944-0.00015-0. Elsevier Inc. [DOI] [Google Scholar]
- Scanlan JG, Hall AR, Scanlan PD. Impact of bile salts on coevolutionary dynamics between the gut bacterium Escherichia coli and its lytic phage PP01. Infect. Genet. Evol. 2019;73:425–432. doi: 10.1016/j.meegid.2019.05.021. [DOI] [PubMed] [Google Scholar]
- Scharn CR, Tenover FC, Goering R V. Transduction of staphylococcal cassette chromosome mec elements between strains of Staphylococcus aureus. Antimicrob. Agents Chemother. 2013;57:5233–5238. doi: 10.1128/AAC.01058-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt HF, Sakowski EG, Williamson SJ, Polson SW, Wommack KE. Shotgun metagenomics indicates novel family A DNA polymerases predominate within marine virioplankton. ISME J. 2014;8:103–114. doi: 10.1038/ismej.2013.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmieger H, Schicklmaier P. Transduction of multiple drug resistance of Salmonella enterica serovar typhimurium DT104. FEMS Microbiol. Lett. 1999 doi: 10.1111/j.1574-6968.1999.tb13381.x. [DOI] [PubMed] [Google Scholar]
- Schnaar RL, Lopez PHH. Gangliosides in health and disease. Prog. Mol. Biol. Transl. Sci. 2018 doi: 10.1016/S1877-1173(18)30062-0. [DOI] [PubMed] [Google Scholar]
- Schuch R, Fischetti VA. The secret life of the anthrax agent Bacillus anthracis: Bacteriophage-mediated ecological adaptations. PLoS One. 2009 doi: 10.1371/journal.pone.0006532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuster H, Beyersmann D, Mikolajczyk M, Schlicht M. Prophage Induction by High Temperature in Thermosensitive dna Mutants Lysogenic for Bacteriophage Lambda. 1973. [DOI] [PMC free article] [PubMed]
- Scudellari M. The sprint to solve coronavirus protein structures—and disarm them with drugs. Nature. 2020;581:252–255. doi: 10.1038/d41586-020-01444-z. [DOI] [PubMed] [Google Scholar]
- Shang J, Wan Y, Luo C, Ye G, Geng Q, Auerbach A, et al. Cell entry mechanisms of SARS-CoV-2. Proc. Natl. Acad. Sci. USA. 2020 doi: 10.1073/pnas.2003138117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silveira CB, Rohwer FL. Piggyback-the-winner in host-associated microbial communities. NPJ Biofilms Microbiomes. 2016;2 doi: 10.1038/npjbiofilms.2016.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singhai T. A review of the coronavirus disease-2019. Indian J. Pediatr. 2020;87:281–286. doi: 10.1007/s12098-020-03263-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith GP, Petrenko VA. Phage display. Chem Rev. 1997;97:391–410. doi: 10.1021/cr960065d. [DOI] [PubMed] [Google Scholar]
- Smith TC, Gebreyes WA, Abley MJ, Harper AL, Forshey BM, Male MJ, et al. Methicillin-Resistant staphylococcus aureus in pigs and farm workers on conventional and antibiotic-free swine farms in the USA. PLoS One. 2013 doi: 10.1371/journal.pone.0063704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sutton TDS, Hill C. Gut bacteriophage: current understanding and challenges. Front. Endocrinol. (Lausanne) 2019;10:784. doi: 10.3389/fendo.2019.00784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan D, Hansen MF, de Carvalho LN, Røder HL, Burmølle M, Middelboe M, et al. High cell densities favor lysogeny: induction of an H20 prophage is repressed by quorum sensing and enhances biofilm formation in Vibrio anguillarum. ISME J. 2020;14 doi: 10.1038/s41396-020-0641-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tao P, Mahalingam M, Zhu J, Moayeri M, Sha J, Lawrence WS, et al. A bacteriophage T4 nanoparticle-based dual vaccine against anthrax and plague. MBio. 2018;9 doi: 10.1128/mBio.01926-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tao P, Zhu J, Mahalingam M, Batra H, Rao VB. Bacteriophage T4 nanoparticles for vaccine delivery against infectious diseases. Adv. Drug Deliv. Rev. 2019;145:57–72. doi: 10.1016/j.addr.2018.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thingstad TF, Lignell R. Theoretical models for the control of bacterial growth rate, abundance, diversity and carbon demand. Aquat. Microb. Ecol. 1997;13:19–27. doi: 10.3354/ame013019. [DOI] [Google Scholar]
- Thingstad TF. Elements of a theory for the mechanisms controlling abundance, diversity, and biogeochemical role of lytic bacterial viruses in aquatic systems. Limnol. Oceanogr. 2000;45:1320–1328. doi: 10.4319/lo.2000.45.6.1320. [DOI] [Google Scholar]
- Torres-Barceló C. The disparate effects of bacteriophages on antibiotic-resistant bacteria. Emerg. Microbes. Infect. 2018;7 doi: 10.1038/s41426-018-0169-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valent P, Groner B, Schumacher U, Superti-Furga G, Busslinger M, Kralovics R, et al. Paul Ehrlich (1854-1915) and his contributions to the foundation and birth of translational medicine. J. Innate. Immun. 2016;8:111–120. doi: 10.1159/000443526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verreault D, Gendron L, Rousseau GM, Veillette M, Massé D, Lindsley WG, et al. Detection of airborne lactococcal bacteriophages in cheese manufacturing plants. Appl. Environ. Microbiol. 2011;77:491–497. doi: 10.1128/AEM.01391-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waller AS, Yamada T, Kristensen DM, Kultima JR, Sunagawa S, Koonin EV., et al. Classification and quantification of bacteriophage taxa in human gut metagenomes. ISME J. 2014;8:1391–1402. doi: 10.1038/ismej.2014.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinbauer MG. Ecology of prokaryotic viruses. FEMS Microbiol. Rev. 2004 doi: 10.1016/j.femsre.2003.08.001. [DOI] [PubMed] [Google Scholar]
- Whitby M, Ruff TA, Street AC, Fenner F. Biological agents as weapons 2: anthrax and plague. Med. J. Aust. 2002;176:605–608. doi: 10.5694/j.1326-5377.2002.tb04594.x. [DOI] [PubMed] [Google Scholar]
- Williamson SJ, Houchin LA, McDaniel L, Paul JH. Seasonal variation in lysogeny as depicted by prophage induction in Tampa Bay, Florida. Appl. Environ. Microbiol. 2002;68:4307–4314. doi: 10.1128/AEM.68.9.4307-4314.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Health Organization. WHO | Zika virus. Who 2018.
- Wu Y, Li C, Xia S, Tian X, Kong Y, Wang Z, et al. Identification of human single-domain antibodies against SARS-CoV-2. Cell Host Microbe. 2020 doi: 10.1016/j.chom.2020.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue C, Goldenfeld N. Coevolution maintains diversity in the stochastic “kill the winner” model. Phys. Rev. Lett. 2017;119 doi: 10.1103/PhysRevLett.119.268101. [DOI] [PubMed] [Google Scholar]
- Young R. Phage lysis: Three steps, three choices, one outcome. J. Microbiol. 2014 doi: 10.1007/s12275-014-4087-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Z H, ST A. Diversity of phage infection types and associated terminology: the problem with “lytic or lysogenic. FEMS Microbiol. Lett. 2016;363 doi: 10.1093/FEMSLE/FNW047. [DOI] [PubMed] [Google Scholar]
- Zhang Y, LeJeune JT. Transduction of blaCMY-2, tet(A), and tet(B) from Salmonella enterica subspecies enterica serovar Heidelberg to S. Typhimurium. Vet. Microbiol. 2008 doi: 10.1016/j.vetmic.2007.11.032. [DOI] [PubMed] [Google Scholar]
- Zhang X, McDaniel AD, Wolf LE, Keusch GT, Waldor MK, Acheson DWK. Quinolone antibiotics induce Shiga toxin–encoding bacteriophages, toxin production, and death in mice. J. Infect. Dis. 2000;181:664–670. doi: 10.1086/315239. [DOI] [PubMed] [Google Scholar]
- Zhen X, Zhou H, Ding W, Zhou B, Xu X, Perčulija V, et al. Structural basis of AimP signaling molecule recognition by AimR in Spbeta group of bacteriophages. Protein Cell. 2019;10:131–136. doi: 10.1007/s13238-018-0588-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zinder ND, Lederberg J. Genetic exchange in salmonella. J. Bacteriol. 1952 doi: 10.1128/jb.64.5.679-699.1952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zinder ND. Forty years ago: the discovery of bacterial transduction. Genetics. 1992;132:291. doi: 10.1093/genetics/132.2.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sime-Ngando T, Bertrand JC, Bogusz D, Brugère JF, Franche C, Fardeau ML, et al. The evolution of living beings started with prokaryotes and in interaction with prokaryotes. Prokaryotes Evol. 2019 doi: 10.1007/978-3-319-99784-1_5. [DOI] [Google Scholar]