Visual Abstract
Keywords: thyroid, microcarcinoma, minimal extrathyroidal extension
Abstract
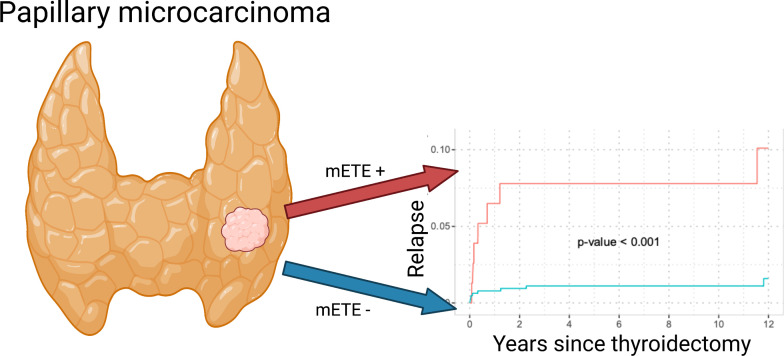
Minimal extrathyroidal extension (mETE) of papillary thyroid microcarcinoma (PTMC) is no longer considered in the new eighth edition of the staging manual of the American Joint Committee on Cancer and the International Union Against Cancer. Therefore, PTMC with mETE previously staged as pT3 will now be staged as pT1a and most likely not receive adjuvant radioiodine therapy. However, it remains unclear whether mETE is associated with higher aggressiveness in PTMC. Therefore, the aim of this study was to investigate whether mETE is associated with a higher risk of lymph node metastases (LNMs) or distant metastases. Methods: In total, 721 patients with PTMC presenting at our department for postoperative counseling from May 1983 to August 2012 were included in this retrospective analysis (median follow-up time, 9.30 y). The impact of mETE on the presence of LNMs at thyroidectomy and relapse through LNMs and distant metastases was assessed by logistic regression and Fine–Gray model analyses. Results: mETE was present in 10.7% (n = 77) of patients and was an independent risk factor for LNMs at thyroidectomy, with an adjusted odds ratio of 4.33 (95% CI, 2.02–9.60; P < 0.001) in multivariable analysis. Patients with mETE had significantly more relapses through LNMs (over 5 y: 13.1% vs. 1.25%; P < 0.001) and distant metastases (over 5 y: 7.8% vs. 1.1%; P < 0.001) than did patients without mETE. mETE was an independent risk factor for relapse through LNMs and distant metastases in multivariable analysis (hazard ratio of 7.78 and 95% CI of 2.87–21.16 for LNMs [P < 0.001]; hazard ratio of 4.09 and 95% CI of 1.25–13.36 for distant metastases [P = 0.020]). Conclusion: mETE is a statistically significant and independent risk factor for relapse through LNMs and distant metastases in PTMC. Therefore, future studies should evaluate whether patients with mETE and PTMC might benefit from intensified surveillance and therapy.
Differentiated thyroid cancer is classified according to the TNM staging system of the American Joint Committee on Cancer and the International Union Against Cancer and has an increasing incidence, especially of papillary tumors that are 10 mm or smaller (1). Up to the seventh edition of the TNM manual, minimal extrathyroidal extension (mETE) was considered in determining the T stage. However, mETE was found to have no impact on disease-related mortality in several studies, such as a study by Hay et al. with 3,524 patients and a metaanalysis by Diker-Cohen with 23,816 patients (2–6). Also, there are studies demonstrating no impact of mETE on recurrence-free survival in differentiated thyroid cancer (7–9).
Because the TNM system strives for optimal prediction of cancer-related overall survival, the new eighth edition of the cancer staging manual of the American Joint Committee on Cancer and the International Union Against Cancer no longer considers mETE (10). In consequence, tumors smaller than 4 cm with mETE, which would have been classified as T3 according to the seventh edition, are now classified as T1 when no larger than 2 cm or as T2 when larger than 2 cm but no larger than 4 cm (11–13).
However, patients with mETE in these studies had radioiodine therapy (RAI) significantly more often than did patients without mETE. This fact is not surprising since, according to the seventh edition of the TNM manual, tumors with mETE were classified as at least T3, a stage for which RAI should be considered, according to the American Thyroid Association and the European Society for Medical Oncology (14,15). Hence, tumors with mETE could represent a more aggressive subset, with the patients showing survival rates comparable to those of patients with completely intrathyroidal tumors only because of higher rates of initial RAI according to the higher T stages.
Moreover, in these studies, the impact of mETE was studied not in a group with only papillary thyroid microcarcinoma (PTMC) but in a group with tumors of various sizes. In the case of a large, 4-cm, tumor, it seems plausible that mETE might not additionally influence survival. Yet, in small tumors, with a diameter of no more than 1 cm, mETE might still be clinically relevant. The impact of removing mETE from T staging in the eighth edition of the TNM manual is especially pronounced for papillary carcinoma no larger than 10 mm with mETE, which will now be assigned the lowest possible T-stage, pT1a, instead of T3.
Therefore, the aim of the present study was to investigate whether mETE is an independent risk factor for tumor relapse in a large cohort of patients with PTMC and should be accounted for.
MATERIALS AND METHODS
Patients, Postoperative Management, and Follow-up
For this retrospective analysis, 721 consecutive patients with PTMC initially presenting in our department for postoperative counseling from May 1983 to August 2012 were enrolled. The median follow-up time was 9.30 y, with the last follow-up data recorded in December 2020.
The extent of thyroidectomy and lymph node resection is detailed in Table 1. Histologic and TNM classifications were present for all patients. To ensure consistent TNM classification, all patients were classified or reclassified according to the sixth edition of the staging manual of the American Joint Committee on Cancer and the International Union Against Cancer (1997). Patients were divided into 2 groups (patients with tumors confined to the thyroid and patients with mETE), as previously published (16,17). Tumors were designated as mETE when the pathology report of the thyroidectomy sample stated mETE (12,18). After initial presentation, 74.9% of patients received adjuvant RAI with consecutive 131I-whole-body scintigraphy. Between surgery and 131I treatment, l-thyroxine treatment was withheld or stopped for 4–6 wk (19). Those patients who received RAI underwent stimulated thyroglobulin measurement, cervical ultrasound, and diagnostic whole-body scintigraphy with 131I at 3–6 mo and 1 y after initial adjuvant RAI in accordance with national and international standards prevailing at the time (20,21). If one or more of these diagnostic tests were positive, further courses of 131I were given as needed (19). Long-term follow-up consisted of thyroglobulin measurement on levothyroxine therapy, serum thyrotropin measurement, and neck ultrasound yearly.
TABLE 1.
Characteristics of Included Cohort
| Characteristic | Total cohort | Confined | mETE | P* (univariate analysis) |
|---|---|---|---|---|
| Patients | 721 | 644 (89.3) | 77 (10.7) | |
| Sex (male) | 154 (21.4) | 145 (22.5) | 9 (11.7) | 0.041 |
| Age (y) | 47.95 ± 12.83 | 48.02 ± 12.77 | 47.38 ± 13.42 | 0.678 |
| Median follow-up (y)† | 9.30 (95% CI, 8.89–9.94) | 8.94 (95% CI, 8.40–9.43) | 12.85 (95% CI, 10.76–14.38) | 0.002 |
| Extent of thyroidectomy | <0.001 | |||
| Hemithyroidectomy | 18 (2.5) | 18 (2.8) | 0 (0.0) | |
| Subtotal thyroidectomy | 126 (17.5) | 125 (19.4) | 1 (1.3) | |
| Total thyroidectomy | 577 (80.0) | 501 (77.8) | 76 (98.7) | |
| Lymph node dissection | <0.001 | |||
| No lymph nodes resected | 505 (70.0) | 472 (73.3) | 33 (42.9) | |
| Node picking | 51 (7.1) | 46 (7.1) | 5 (6.5) | |
| Central compartment node dissection | 120 (16.6) | 93 (14.4) | 27 (35.1) | |
| Central and lateral neck node dissection | 45 (6.2) | 33 (5.1) | 12 (15.6) | |
| Number of removed lymph nodes | 0 (IQR, 0–1) | 0 (IQR, 0–0] | 2 (IQR, 0–10) | <0.001 |
| Adjuvant RAI | 540 (74.9) | 463 (71.9) | 77 (100.0) | <0.001 |
| Cumulative RAI activity (GBq) | 3.00 (IQR, 0.00–6.00) | 3.00 (IQR, 0.00–4.00) | 6.00 (IQR, 3.00–10.00) | <0.001 |
| Number of RAIs | 1.00 (IQR, 0.00–2.00) | 1.00 (IQR, 0.00–1.00) | 1.00 (IQR, 1.00–2.00) | <0.001 |
| Pre-RAI TSH (μU/mL) | 43.06 (IQR, 15.62–77.26) | 39.17 (IQR, 13.24–76.89) | 59.36 (IQR, 42.20–77.72) | 0.001 |
| Pre-RAI thyroglobulin (ng/mL) | 2.30 (IQR, 0.62–8.45) | 2.50 (IQR, 0.60–8.67) | 2.20 (IQR, 0.80–7.00) | 0.935 |
*Determined with Fisher exact test for categoric variables and Wilcoxon test for continuous variables.
†Calculated by reverse Kaplan–Meier test.
IQR = interquartile range.
Qualitative data are number followed by percentage; continuous data are mean ± SD, or median followed by 95% CI or IQR.
Table 1 describes the patients’ characteristics in detail. The study protocol was approved by the local ethics committee (2019-459-f-S) and was performed in accordance with the 1964 Declaration of Helsinki and its later amendments.
Endpoints
The primary endpoints were the cumulative incidences of lymph node metastasis (LNM) relapse (1) and of distant metastasis relapse (2). The secondary endpoint was the presence of LNMs in the thyroidectomy sample. Analyses were adjusted for extent of thyroidectomy and lymph node dissection if numerically feasible.
Diagnosis of distant metastases was based on surgery with histologic workup or on a composite score requiring positive RAI imaging findings and elevated thyroglobulin levels. Relapse through distant metastases was considered present when distant metastases were diagnosed after thyroidectomy, including recurrence in nonregional lymph nodes or visceral sites. Table 2 provides details on the detection of distant metastases. LNM relapse was considered in 2 cases. The first was when LNMs were diagnosed in patients without LNMs in the thyroidectomy sample after thyroidectomy. The second was when LNMs became apparent in patients who had LNMs in the thyroidectomy sample in the course of follow-up after initial adjuvant RAI. In both cases, diagnoses were based on a composite score consisting of RAI imaging and elevated thyroglobulin levels or surgery with histologic work-up. The presence of LNMs in the thyroidectomy sample was histologically ascertained in all patients who underwent lymph node resection.
TABLE 2.
Detailed Report on Patients with Distant Metastases
| mETE | Age at initial presentation | Type of distant metastasis | Initial LNM | Tumor diameter (mm) | Days between postoperative counseling and confirmation of metastasis |
|---|---|---|---|---|---|
| No | 65 | Mediastinal LNM | No | 4 | 119 |
| Yes | 68 | Bone | No | 5 | 4,213 |
| No | 69 | Bone | No | 1 | −25* |
| No | 30 | Pulmonary | No | 7 | 454 |
| No | 26 | Pulmonary | No | 10 | 830 |
| No | 48 | Bone | No | 4 | 7 |
| No | 25 | Bone | No | 9 | 0 |
| No | 44 | Pulmonary | No | 7 | 40 |
| Yes | 54 | Bone and pulmonary | Yes | 9 | 50 |
| Yes | 58 | Pulmonary | No | 9 | 121 |
| Yes | 55 | Pulmonary | Yes | 5 | 61 |
| No | 48 | Pulmonary | Yes | 4 | 4,309 |
| No | 15 | Pulmonary | Yes | 10 | 14 |
| Yes | 15 | Pulmonary | Yes | 8 | 28 |
| Yes | 37 | Pulmonary | Yes | 3 | 256 |
| Yes | 23 | Pulmonary | Yes | 5 | 442 |
| No | 6 | Pulmonary | Yes | 10 | 22 |
*Initial diagnosis of thyroid cancer was made after bone metastasis confirmation.
Statistical Analysis
Normally distributed data are described using mean and SD, and nonnormally distributed data are described using median and interquartile range. Normality was assessed by histograms and skewness statistics. Univariable and multivariable logistic regression analyses were performed to evaluate the effect of age, sex, tumor size, multifocality, extent of lymph node resection, and mETE on the presence of LNMs at thyroidectomy, in the subgroup of patients who had lymph node resection (i.e., known nodal status at thyroidectomy). Results are reported as odds ratios, corresponding 95% CIs, and the P values of the Wald test.
Time-to-event data were analyzed within a competing-risk framework accounting for the competing risk of death. Because distant metastases at thyroidectomy were considered a terminal event, the corresponding endpoint was analyzed in the subgroup of patients without distant metastases at thyroidectomy. Cumulative incidences of distant metastases and LNM relapse after thyroidectomy were estimated on the basis of the Fine–Gray model and compared between mETE-positive and -negative patients using the Gray test (22,23). To adjust for further factors, a multivariable Fine–Gray subdistribution hazard regression was conducted. Results are presented as subdistribution hazard ratios (HRs), corresponding 95% CIs, and P values. Follow-up times were calculated by reverse Kaplan–Meier testing. All inferential statistics were intended to be exploratory and were interpreted accordingly. The reported 2-sided P values were used only to generate new hypotheses. P values of 0.05 or less were considered statistically significant. Analyses were performed using R statistical software, version 3.6.1 (The R Foundation, https://www.r-project.org/). The graphical abstract was created with BioRender.com.
RESULTS
Patient Characteristics
Between May 1983 and August 2012, 721 patients with PTMC presented for postoperative counseling in our department. The patients were followed up annually (median follow-up time, 9.30 y). No patient died of thyroid cancer. mETE was present in 77 (10.7%) of the patients. The patient characteristics are summarized in Table 1. Comparative characteristics between patients with confined tumors and patients with mETE are presented in Table 3.
TABLE 3.
Characteristics of Patients With and Without mETE
| Characteristic | Total cohort | Confined | mETE | P* (univariable analysis) |
|---|---|---|---|---|
| Number of patients | 721 | 644 (89.3) | 77 (10.7) | |
| Number of metastasized lymph nodes | 0 (IQR–0,0) | 0 (IQR–0,0) | 0 (IQR–0,1) | <0.001 |
| Nodal stage given by histopathologic examination of surgical specimen | <0.001 | |||
| pN0 | 137 (19.0) | 120 (18.6) | 17 (22.1) | |
| pN1a | 57 (7.9) | 39 (6.1) | 18 (23.4) | |
| pN1b | 22 (3.1) | 13 (2.0) | 9 (11.7) | |
| pNx | 505 (70.0) | 472 (73.3) | 33 (42.9) | |
| Five-year cumulative incidence of LNM relapse (%) | 2.54 (95% CI, 1.57–3.91) | 1.25 (95% CI, 0.59–2.37) | 13.12 (95% CI, 6.69–21.77) | <0.001 |
| Five-year cumulative incidence distant metastasis relapse‡ (%) | 1.83 (95% CI, 1.03–3.03) | 1.11 (95% CI, 0.50–2.19) | 7.79 (95% CI, 3.16–15.19) | <0.001 |
| Tumor size (mm) | 5.00 (IQR, 3.00–8.00) | 5.00 (IQR, 3.00–8.00) | 8.00 (IQR, 6.00–10.00) | <0.001 |
| Multifocal disease | 135 (18.7) | 118 (18.3) | 17 (22.1) | 0.520 |
*Determined with Fisher exact test for categoric variables and Wilcoxon test for continuous variables.
†Assessed in subset of patients without distant metastases at thyroidectomy (n = 719).
IQR = interquartile range.
Qualitative data are number followed by percentage, except for 5-y cumulative incidence of LNM relapse; continuous data are mean ± SD, or median followed by 95% CI or IQR.
Differences Between Patients With and Without mETE
The proportion of patients with LNM in the thyroidectomy sample (subcohort of patients with lymph nodes resected at the thyroidectomy, n = 216) was higher in patients with mETE (27/44, 61.4%) than in those without mETE (49/172, 28.5%, P < 0.001). LNM relapse occurred in 11 of 77 patients (14.3%) with mETE in the course of follow-up and in 10 of 644 patients without mETE (1.6%). Relapse through distant metastases (subcohort of patients without known distant metastases at thyroidectomy, n = 719) occurred in 7 of 77 patients in the subgroup with mETE (9.1%) and in 8 of 642 patients with confined tumors (1.2%). Table 3 shows additional differences between patients with mETE and patients with confined tumors. To adjust for interfering effects and loss to follow-up, multivariate regressions and survival analysis were performed in the next section.
Risk Factors Associated with LNMs Detected at Thyroidectomy
Risk factors associated with LNMs detected at thyroidectomy were analyzed in the subset of patients who had lymph nodes removed at thyroidectomy (n = 216). In 140 of those 216 patients (64.8%), no LNMs were found; 55 (25.5%) had central LNMs, and 21 (9.7%) had LNMs in the lateral compartment.
The proportion of patients with LNMs was higher in patients with mETE (27/44, 61.4%) than in those without mETE (49/172, 30.2%, P < 0.001).
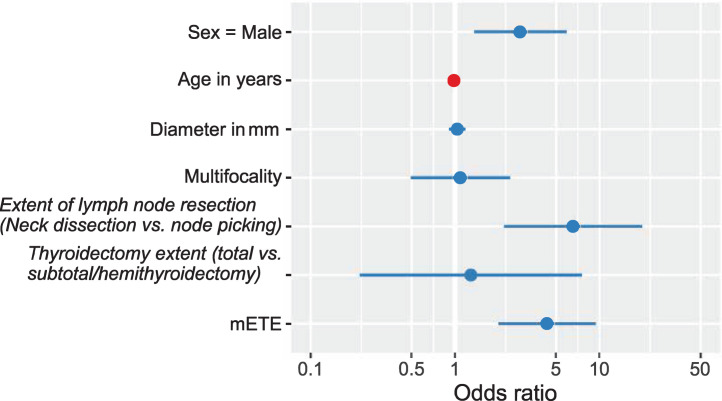
The results of the multivariable analysis using logistic regression are summarized in Table 4 and visualized in Figure 1. Sex, extent of lymph node resection (neck dissection vs. node picking), and mETE were significant risk factors for LNM, whereas tumor size was not. The adjusted odds ratio for LNMs in regard to mETE was 4.33 (95% CI, 2.02–9.60; P < 0.001).
TABLE 4.
Univariable and Multivariable Logistic Regression Analysis of Risk Factors for LNM Diagnosed in Histologic Workup of Thyroidectomy Sample Adjusted for Lymph Node and Thyroidectomy Extent
| Risk factor | LNM-negative | LNM-positive | Univariable analysis | Multivariable analysis | ||||
|---|---|---|---|---|---|---|---|---|
| Odds ratio | 95% CI | P | Odds ratio | 95% CI | P | |||
| n | 140 | 76 | ||||||
| Sex (male) | 24 (17.1) | 25 (32.9) | 2.37 | 1.24–4.56 | 0.009 | 2.83 | 1.36–5.98 | 0.006 |
| Age | 44.79 ± 12.50 | 41.54 ± 13.92 | 0.98 | 0.96–1.00 | 0.082 | 0.98 | 0.96–1.01 | 0.183 |
| Tumor size in mm | 7.00 (IQR, 4.00–9.00) | 7.00 (IQR, 5.00–9.00) | 1.05 | 0.94–1.18 | 0.378 | 1.04 | 0.91–1.18 | 0.579 |
| Multifocality | 27 (19.3) | 16 (21.1) | 1.12 | 0.55–2.21 | 0.756 | 1.09 | 0.49–2.39 | 0.834 |
| Neck dissection vs. node picking | 93 (66.4) | 72 (94.7) | 9.10 | 3.50–31.17 | <0.001 | 6.55 | 2.41–23.05 | <0.001 |
| Total thyroidectomy vs. subtotal or hemithyroidectomy | 131 (93.6) | 74 (97.4) | 2.54 | 0.63–16.96 | 0.241 | 1.28 | 0.25–9.94 | 0.782 |
| mETE | 17 (12.1) | 27 (35.5) | 3.99 | 2.02–8.09 | <0.001 | 4.33 | 2.02–9.60 | <0.001 |
IQR = interquartile range.
Only patients who had lymph nodes resected are included (n = 216). Qualitative data are number followed by percentage; continuous data are mean ± SD, or median followed by IQR.
FIGURE 1.
Adjusted odds ratios for LNMs at thyroidectomy (evaluated in patients who had lymph nodes resected at thyroidectomy, n = 216). Odds ratios are tabulated in Table 4.
Risk Factors Associated with LNM Relapse
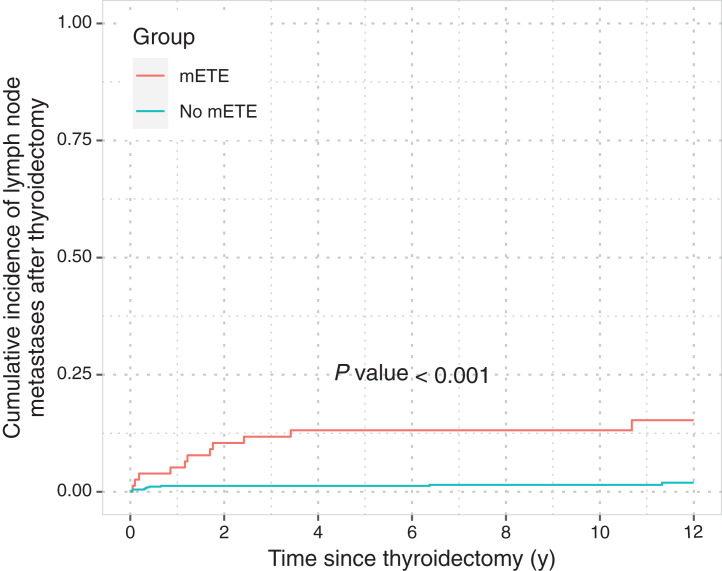
LNM relapse occurred in 21 of 721 patients during the course of follow-up (11/77 patients with mETE, 10/644 patients without mETE). The 5-y cumulative incidences were 13.12% (95% CI, 6.69–21.77) and 1.25% (95% CI, 0.59–2.37, P < 0.001) for patients with mETE and with confined tumors, respectively (Fig. 2).
FIGURE 2.
Cumulative incidence function of LNM relapse after thyroidectomy stratified by mETE. Curves were compared using Gray test (n = 721).
In the multivariable competing-risk regression (Table 5), mETE (HR, 7.80; 95% CI, 2.87–21.16; P < 0.001), male sex (HR, 4.17; 95% CI, 1.63– 10.67; P = 0.003), and tumor size (HR, 1.15; 95% CI, 1.02–1.30; P = 0.022) were found to be independent risk factors for LNM relapse, whereas age and multifocality were not.
TABLE 5.
Multivariable Competing-Risk Regression Analysis for LNM Relapse After Thyroidectomy Based on Fine–Gray Proportional Subdistribution Hazards Model
| Variable | HR | 95% CI | P |
|---|---|---|---|
| Sex (male–female) | 4.17 | 1.63–10.67 | 0.003 |
| Age | 1.00 | 0.98–1.03 | 0.690 |
| Tumor size | 1.15 | 1.02–1.30 | 0.022 |
| Multifocality (yes–no) | 2.44 | 0.97–6.17 | 0.060 |
| Lymph nodes resected at thyroidectomy (yes–no) | 3.23 | 1.33–7.86 | 0.010 |
| mETE (yes–no) | 7.80 | 2.87–21.16 | <0.001 |
Risk Factors Associated with Distant Metastasis Relapse
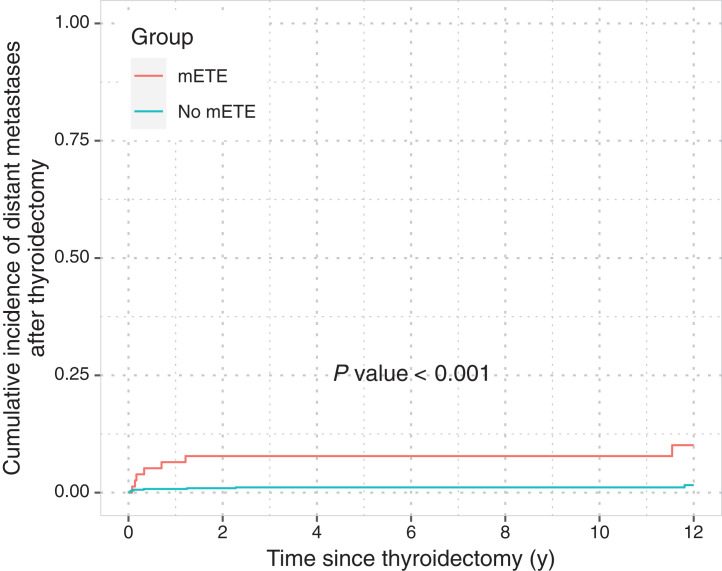
Distant metastases occurred in 15 of 719 patients without distant metastases at thyroidectomy during the course of follow-up (mETE, 7/77; confined, 8/642). The 5-y cumulative incidences were 7.79% (95% CI, 3.16–15.19) and 1.11% (95% CI, 0.50–2.19; P < 0.001) for patients with mETE and those with confined tumors, respectively (Fig. 3).
FIGURE 3.
Cumulative incidence function of distant metastasis relapse after thyroidectomy in group of patients without distant metastases at thyroidectomy (n = 719), stratified by mETE. Curves were compared using Gray test.
In multivariable competing-risk regression (Table 6), both mETE and the presence of LNMs at thyroidectomy (N1 vs. N0) were found to be independent risk factors for distant metastasis occurrence after thyroidectomy (HR of 4.09 and 95% CI of 1.25–13.36 for mETE [P = 0.020] and HR of 8.76 and 95% CI of 1.16–66.17 for LNM [P = 0.035]).
TABLE 6.
Multivariable Competing-Risk Regression Analysis for Distant Metastasis Relapse After Thyroidectomy Based on Fine–Gray Proportional Subdistribution Hazards Model
| Variable | HR | 95% CI | P |
|---|---|---|---|
| Sex (male–female) | 1.52 | 0.48–4.82 | 0.470 |
| Age | 0.96 | 0.91–1.01 | 0.140 |
| Tumor size | 1.06 | 0.85–1.32 | 0.610 |
| Multifocality (yes–no) | 2.25 | 0.73–6.90 | 0.160 |
| N status at thyroidectomy (Nx–N0) | 2.60 | 0.30–22.51 | 0.390 |
| N status at thyroidectomy (N1–N0) | 8.76 | 1.16–66.17 | 0.035 |
| mETE (yes–no) | 4.09 | 1.25–13.36 | 0.020 |
DISCUSSION
The risk of LNMs at thyroidectomy and relapse through LNMs and distant metastases in patients with PTMC was analyzed with regard to mETE. mETE was found to be an independent risk factor for the presence of LNMs at thyroidectomy and an independent risk factor for relapse through both distant metastases and LNMs.
The implications of mETE in papillary thyroid cancer are highly controversial. Multiple studies have reported that mETE is not associated with higher rates of LNMs, distant metastases, or mortality in differentiated thyroid cancer (3,4,24). Therefore, mETE was removed from the new eighth edition of the TNM staging manual. However, it seems unjustified to compare the outcome of patients with mETE with the outcome of those without, as the two groups have been treated with different intensities: patients with mETE significantly more often received RAI than did patients without mETE (2). Therefore, the lack of difference in outcome between the groups might have been caused by confounding factors.
The impact of removing mETE from T staging is especially pronounced for papillary carcinoma no larger than 10 mm with mETE, which would previously have been staged as T3. In the new eighth edition of the TNM manual, these tumors are now attributed to the lowest possible stage, pT1a. Very recent studies including differentiated thyroid cancer of all sizes indicate that not only gross extrathyroidal extension but also mETE is associated with increased mortality and recurrence (25,26). To further corroborate these findings, the present study assessed the implications of mETE in a large, homogeneous group of patients with PTMC.
The study found an increased odds that LNMs would be present at surgery in patients with mETE. This observation indicates that mETE tumors show a more aggressive phenotype, as is well in line with the results of Zhi et al. and others, who found mETE to be a risk factor for LNMs in patients with PTMC (25–31).
Castagna et al. reported that mETE is a risk factor for LNM only in patients with a tumor size greater than 1.5 cm (32). That report is in contrast to our findings that mETE-positive patients are more frequently affected by LNM at thyroidectomy than are mETE-negative patients, irrespective of tumor size (Fig. 1).
The role of mETE in relapse through distant metastasis in PTMC is likewise controversial. Six previous studies with PTMC-only cohorts could not find an impact of mETE on any cancer recurrence (5,26, 29,31,33,34). However, the cohorts in those studies were rather small, ranging from 144 to 288 patients. Three studies with larger PTMC-only cohorts, ranging from 287 to 531 patients, were able to demonstrated an impact of mETE on relapse in univariable analysis (17,27,35). However, these studies investigated not distant metastasis relapse but any cancer recurrence. To date, there has been no evidence for a higher rate of relapse for distant metastases associated with mETE. In contrast, our study, using a cohort of 721 patients, could show for the first time (to our knowledge) that mETE is a statistically significant independent risk factor both for LNMs and for distant metastasis relapse. As distant metastases are associated with a significantly worsened prognosis, this finding is of great clinical relevance (15).
The presence of LNMs at thyroidectomy was identified as a further independent risk factor for relapse through distant metastases. Therefore, patients with LNMs and mETE seem to have a particularly high risk of distant metastases after thyroidectomy, compared with patients without both characteristics. Because of the delayed effects associated with mETE (i.e., relapse through distant metastases), it seems advisable to still integrate mETE in the T stage as proposed by Schmid et al. (12).
Given the higher cumulative incidence of LNM and distant metastasis relapse in patients with PTMC and mETE than in patients without, adjuvant RAI might be advisable. A study by Rosario et al. investigated disease recurrence rates in patients with mETE who did not receive adjuvant RAI (36). Only 2% of their patients had recurrent disease, leading the investigators to conclude that RAI can be omitted in patients with mETE. However, the fact that only patients without LNMs at diagnosis and only 20 patients with PTMC were enrolled hampers the transferability of the results. Because mETE is a risk factor for LNMs and especially distant metastases, mETE-positive tumors might be biologically more aggressive and could deserve an intensified treatment. In particular, patients with both LNMs in the thyroidectomy sample and mETE might benefit from an initial RAI. Until further studies explicitly investigate the benefit of RAI in patients with PTMC and mETE, the clinician has to carefully review the pathologic report after thyroidectomy and discuss the option of adjuvant RAI when mETE is present.
The present study faced some limitations. It was conducted retrospectively and might therefore have been influenced by selection biases. To counteract this effect, all patients from an interval larger than 30 y were included. However, the accuracy of diagnostic procedures has increased and could therefore influence the detection of metastases. Despite the long recruitment period, the number of included patients was relatively small. The presence of LNMs at thyroidectomy could be evaluated only in patients who underwent lymph node resection. Because these patients might not represent a random subset of PTMC patients, our findings regarding this endpoint could be limited. The extent of thyroidectomy could not be included in the competing-risk models of LNM and distant metastasis recurrence for numeric reasons (nearly all patients had total thyroidectomy). Given the low incidence of mETE, multicentric analyses have to be performed to corroborate the present initial findings. Another limitation arose from the controversy among pathologists about what constitutes mETE, with there being no standardized histopathologic criteria currently (18,37). The pathology reports from which the presence of mETE was derived for this study were issued by numerous pathologists from different institutions, possibly applying varying criteria for the diagnosis of mETE.
CONCLUSION
mETE is an independent risk factor for cancer relapse through LNMs and distant metastases in PTMC. Therefore, future studies should evaluate whether patients with mETE and PTMC might benefit from intensified surveillance or therapy.
DISCLOSURE
No potential conflict of interest relevant to this article was reported.
KEY POINTS.
QUESTION: Is mETE an independent risk factor for cancer relapse in PTMC?
PERTINENT FINDINGS: mETE was found to be a statistically significant and independent risk factor of cancer relapse through both LNMs and distant metastases.
IMPLICATIONS FOR PATIENT CARE: PTMC patients with mETE might benefit from intensified surveillance or therapy.
REFERENCES
- 1. Davies L, Welch HG. Increasing incidence of thyroid cancer in the United States, 1973-2002. JAMA. 2006;295:2164–2167. [DOI] [PubMed] [Google Scholar]
- 2. Diker-Cohen T, Hirsch D, Shimon I, et al. Impact of minimal extra-thyroid extension in differentiated thyroid cancer: systematic review and meta-analysis. J Clin Endocrinol Metab. March 1, 2018. [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 3. Ito Y, Tomoda C, Uruno T, et al. Minimal extrathyroid extension does not affect the relapse-free survival of patients with papillary thyroid carcinoma measuring 4 cm or less over the age of 45 years. Surg Today. 2006;36:12–18. [DOI] [PubMed] [Google Scholar]
- 4. Hay ID, Johnson TR, Thompson GB, Sebo TJ, Reinalda MS. Minimal extrathyroid extension in papillary thyroid carcinoma does not result in increased rates of either cause-specific mortality or postoperative tumor recurrence. Surgery. 2016;159:11–19. [DOI] [PubMed] [Google Scholar]
- 5. Woo CG, Sung CO, Choi YM, et al. Clinicopathological significance of minimal extrathyroid extension in solitary papillary thyroid carcinomas. Ann Surg Oncol. 2015;22(suppl 3):S728–S733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Al-Qurayshi Z, Shama MA, Randolph GW, Kandil E. Minimal extrathyroidal extension does not affect survival of well-differentiated thyroid cancer. Endocr Relat Cancer. 2017;24:221–226. [DOI] [PubMed] [Google Scholar]
- 7. Nixon IJ, Ganly I, Patel S, et al. The impact of microscopic extrathyroid extension on outcome in patients with clinical T1 and T2 well-differentiated thyroid cancer. Surgery. 2011;150:1242–1249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Shin JH, Ha TK, Park HK, et al. Implication of minimal extrathyroidal extension as a prognostic factor in papillary thyroid carcinoma. Int J Surg. 2013;11:944–947. [DOI] [PubMed] [Google Scholar]
- 9. Jin BJ, Kim MK, Ji YB, Song CM, Park JH, Tae K. Characteristics and significance of minimal and maximal extrathyroidal extension in papillary thyroid carcinoma. Oral Oncol. 2015;51:759–763. [DOI] [PubMed] [Google Scholar]
- 10. Tuttle RM, Haugen B, Perrier ND. Updated American Joint Committee on Cancer/Tumor-Node-Metastasis staging system for differentiated and anaplastic thyroid cancer (eighth ed.): what changed and why? Thyroid. 2017;27:751–756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Amin MB, Greene FL, Edge SB, eds. AJCC Cancer Staging Manual. 8th ed. Springer; 2017:881–889. [Google Scholar]
- 12. Schmid KW, Synoracki S, Dralle H, Wittekind C. Proposal for an extended pTNM classification of thyroid carcinoma: commentary on deficits of the 8th edition of the TNM classification [in German]. Pathologe. 2018;39:49–56. [DOI] [PubMed] [Google Scholar]
- 13. Amin MB, Greene FL, Edge SB, et al. The eighth edition AJCC cancer staging manual: continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017;67:93–99. [DOI] [PubMed] [Google Scholar]
- 14. Filetti S, Durante C, Hartl D, et al. Thyroid cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2019;30:1856–1883. [DOI] [PubMed] [Google Scholar]
- 15. Haugen BR, Alexander EK, Bible KC, et al. 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26:1–133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Youngwirth LM, Adam MA, Scheri RP, Roman SA, Sosa JA. Extrathyroidal extension is associated with compromised survival in patients with thyroid cancer. Thyroid. 2017;27:626–631. [DOI] [PubMed] [Google Scholar]
- 17. Mercante G, Frasoldati A, Pedroni C, et al. Prognostic factors affecting neck lymph node recurrence and distant metastasis in papillary microcarcinoma of the thyroid: results of a study in 445 patients. Thyroid. 2009;19:707–716. [DOI] [PubMed] [Google Scholar]
- 18. Mete O, Rotstein L, Asa SL. Controversies in thyroid pathology: thyroid capsule invasion and extrathyroidal extension. Ann Surg Oncol. 2010;17:386–391. [DOI] [PubMed] [Google Scholar]
- 19. Vrachimis A, Riemann B, Mäder U, Reiners C, Verburg FA. Endogenous TSH levels at the time of 131I ablation do not influence ablation success, recurrence-free survival or differentiated thyroid cancer-related mortality. Eur J Nucl Med Mol Imaging. 2016;43:224–231. [DOI] [PubMed] [Google Scholar]
- 20. Luster M, Clarke SE, Dietlein M, et al. Guidelines for radioiodine therapy of differentiated thyroid cancer. Eur J Nucl Med Mol Imaging. 2008;35:1941–1959. [DOI] [PubMed] [Google Scholar]
- 21. Dietlein M, Eschner W, Grünwald F, Lassmann M, Verburg FA, Luster M. DGN recommendation for action (S1 guideline): radioiodine therapy for differentiated thyroid cancer (version 4) [in German]. https://www.nuklearmedizin.de/leistungen/leitlinien/docs/031-002l_S1_Radioiodtherapie_differenziertes_Schilddruesenkarziom_2015-10.pdf. Published October 2015. Accessed August 25, 2021.
- 22. Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509. [Google Scholar]
- 23. Gray RJ. A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat. 1988;16:1141–1154. [Google Scholar]
- 24. Ito Y, Tomoda C, Uruno T, et al. Prognostic significance of extrathyroid extension of papillary thyroid carcinoma: massive but not minimal extension affects the relapse-free survival. World J Surg. 2006;30:780–786. [DOI] [PubMed] [Google Scholar]
- 25. Liu Z, Huang Y, Chen S, et al. Minimal extrathyroidal extension affects the prognosis of differentiated thyroid cancer: is there a need for change in the AJCC classification system? PLoS One. 2019;14:e0218171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Danilovic DLS, Castroneves LA, Suemoto CK, et al. Is there a difference between minimal and gross extension into the strap muscles for the risk of recurrence in papillary thyroid carcinomas? Thyroid. 2020;30:1008–1016. [DOI] [PubMed] [Google Scholar]
- 27. Lombardi CP, Bellantone R, de Crea C, et al. Papillary thyroid microcarcinoma: extrathyroidal extension, lymph node metastases, and risk factors for recurrence in a high prevalence of goiter area. World J Surg. 2010;34:1214–1221. [DOI] [PubMed] [Google Scholar]
- 28. Zhi J, Zhao J, Gao M, et al. Impact of major different variants of papillary thyroid microcarcinoma on the clinicopathological characteristics: the study of 1041 cases. Int J Clin Oncol. 2018;23:59–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Moon HJ, Kim E-K, Chung WY, Yoon JH, Kwak JY. Minimal extrathyroidal extension in patients with papillary thyroid microcarcinoma: is it a real prognostic factor? Ann Surg Oncol. 2011;18:1916–1923. [DOI] [PubMed] [Google Scholar]
- 30. Ahmaddy F, Wenter V, Ilhan H, et al. Effects of the minimal extrathyroidal extension on early response rates after (adjuvant) initial radioactive iodine therapy in PTC patients. Cancers (Basel). 2020;12:3357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Siddiqui S, White MG, Antic T, et al. Clinical and pathologic predictors of lymph node metastasis and recurrence in papillary thyroid microcarcinoma. Thyroid. 2016;26:807–815. [DOI] [PubMed] [Google Scholar]
- 32. Castagna MG, Forleo R, Maino F, et al. Small papillary thyroid carcinoma with minimal extrathyroidal extension should be managed as ATA low-risk tumor. J Endocrinol Invest. 2018;41:1029–1035. [DOI] [PubMed] [Google Scholar]
- 33. Besic N, Pilko G, Petric R, Hocevar M, Zgajnar J. Papillary thyroid microcarcinoma: prognostic factors and treatment. J Surg Oncol. 2008;97:221–225. [DOI] [PubMed] [Google Scholar]
- 34. Chow S-M, Law SCK, Chan JKC, Au S-K, Yau S, Lau W-H. Papillary microcarcinoma of the thyroid: prognostic significance of lymph node metastasis and multifocality. Cancer. 2003;98:31–40. [DOI] [PubMed] [Google Scholar]
- 35. Tran B, Roshan D, Abraham E, et al. An analysis of the American Joint Committee on Cancer 8th edition T staging system for papillary thyroid carcinoma. J Clin Endocrinol Metab. 2018;103:2199–2206. [DOI] [PubMed] [Google Scholar]
- 36. Rosario PW, Mourão G, Calsolari MR. Risk of recurrence in patients with papillary thyroid carcinoma and minimal extrathyroidal extension not treated with radioiodine. J Endocrinol Invest. 2019;42:687–692. [DOI] [PubMed] [Google Scholar]
- 37. Su HK, Wenig BM, Haser GC, et al. Inter-observer variation in the pathologic identification of minimal extrathyroidal extension in papillary thyroid carcinoma. Thyroid. 2016;26:512–517. [DOI] [PMC free article] [PubMed] [Google Scholar]