Abstract
Background: Patients with primary antibody deficiencies are at risk in the current COVID-19 pandemic due to their impaired response to infection and vaccination. Specifically, patients with common variable immunodeficiency (CVID) generated poor spike-specific antibody and T cell responses after immunization. Methods: Thirty-four CVID convalescent patients after SARS-CoV-2 infection, 38 CVID patients immunized with two doses of the BNT162b2 vaccine, and 20 SARS-CoV-2 CVID convalescents later and immunized with BNT162b2 were analyzed for the anti-spike IgG production and the generation of spike-specific memory B cells and T cells. Results: Spike-specific IgG was induced more frequently after infection than after vaccination (82% vs. 34%). The antibody response was boosted in convalescents by vaccination. Although immunized patients generated atypical memory B cells possibly by extra-follicular or incomplete germinal center reactions, convalescents responded to infection by generating spike-specific memory B cells that were improved by the subsequent immunization. Poor spike-specific T cell responses were measured independently from the immunological challenge. Conclusions: SARS-CoV-2 infection primed a more efficient classical memory B cell response, whereas the BNT162b2 vaccine induced non-canonical B cell responses in CVID. Natural infection responses were boosted by subsequent immunization, suggesting the possibility to further stimulate the immune response by additional vaccine doses in CVID.
Keywords: common variable immunodeficiencies, SARS-CoV-2, COVID-1, BNT162b2, vaccine, third dose, memory B cells, spike protein, antibody response
1. Introduction
Due to the severely impaired immune response to infection and immunization, patients with primary antibody defects (PADs) may be at increased risk for severe or prolonged infections [1,2]. In particular, patients with common variable immunodeficiencies (CVIDs), the most common symptomatic PAD, have an impaired response to infections and vaccination, severely reduced circulating class-switched memory B cells (MBCs), and strongly decreased plasmablast/plasma cell production, associated with impaired post-germinal center (GC) B cell maturation and differentiation in blood and secondary lymphoid tissues [3,4].
Since the start of the SARS-CoV-2 pandemic, clinical descriptions of COVID-19 in CVID patients are expanding, with a clinical presentation varying from asymptomatic or mild symptoms to death [5,6,7,8,9,10,11]. In Italy, we demonstrated that CVID patients have a cumulative incidence and an infection fatality rate similar to the SARS-CoV-2-positive general population [12]. Different from the general population, CVID patients display a lower median age at death and do not present the same risk factors predisposing to severe COVID-19 [13,14,15] with the exception of the underlying chronic lung disease (CLD) [16].
Immunization is the safest and most effective tool to achieve a protective response against SARS-CoV-2 infection and to terminate the pandemic [17,18]. In immunocompetent individuals, mRNA vaccine elicits high SARS-CoV-2-neutralizing antibodies and robust antigen-specific CD8+ and CD4+ T cell responses [19,20]. Clinical trials showed an effectiveness of almost 95% in preventing severe COVID-19 disease [17]. In Italy, COVID-19 immunization has been made available for fragile patients since March 2021 [21]. Thanks to its safety profile, SARS-CoV-2 immunization is highly recommended also in PAD patients [22]. However, due to the immune defect, their responses to vaccines are variable [23,24].
Here, we compared the adaptive responses induced by natural SARS-CoV-2 infection and immunization with an mRNA vaccine in patients with CVID. Our results showed that vaccination and infection prime different B cells responses and that the humoral immune response induced by natural infection can be significantly enhanced by subsequent immunization.
2. Methods
2.1. Study Design and Patients
Interventional study carried out in two groups of CVID patients: 34 subjects previously infected by SARS-CoV-2 (thereafter indicated as convalescent) and 38 subjects naive to SARS-CoV-2 infection, who were immunized by the BNT162b2 vaccine (reported as immunized). Participants were diagnosed as having CVID according to the ESID criteria [25]. Eligible patients were informed on the study, including its safety profile and supply procedures.
SARS-CoV-2-positive patients were identified by RT-PCR on nasopharyngeal swabs within 48 h from the symptom onset or in case of family contact. COVID-19 clinical symptoms, demographic characteristics, and comorbidities data were collected by study physicians.
In the immunized group, the BNT162b2 vaccine was administered in two doses, with 21 days apart. Blood samples were obtained for serological and cellular immunity assessment at baseline (BL) before immunization and seven days after the second dose. Samples from SARS-CoV-2-convalescent patients were obtained after a negative RT-PCR.
Blood samples were also collected in a group of 20/34 convalescent patients who underwent immunization with a single dose of BNT162b2 vaccine (indicated as convalescent/immunized). During the study, the participants were allowed to continue their therapies, including immunoglobulin substitution as a standard therapy for the underlying antibody deficiency. The study was approved by the Ethical Committee of the Sapienza University of Rome (Prot. 0521/2020, 13 July 2020) and was performed in accordance with the Good Clinical Practice guidelines, the International Conference on Harmonization guidelines, and the most recent version of the Declaration of Helsinki.
2.2. ELISA for Specific IgG Detection
A semi-quantitative in vitro determination of human IgG antibodies against the SARS-CoV-2 (S1) was performed on serum samples by using the anti-SARS-CoV-2 spike ELISA (EUROIMMUN, Lübeck, Germany), according to the manufacturer’s instructions. Values were then normalized by comparison with a calibrator. Results were obtained by calculating the ratio between the extinction of samples and the extinction of the calibrator. Results are reported as the ratio between the optical density (OD) from the sample and the OD from the calibrator. The ratio interpretation was as follows: negative, <0.8; borderline, ≥0.8 to <1.1; positive, ≥1.1.
2.3. Detection of SARS-CoV-2-Specific B Cells
The analysis of spike-specific cellular immunity was performed in 23, 17, and 3 selected immunized, convalescent, and convalescent/immunized patients, respectively. We did not analyze spike-specific B cells frequency in 15 immunized, 17 convalescents, and 17 convalescent/immunized patients, due to the lack of blood samples. Peripheral blood mononuclear cells (PBMCs) were isolated by Ficoll Paque™ Plus 206 (Amersham PharmaciaBiotech, Amersham, United Kingdom) density-gradient centrifugation and immediately frozen and stored in liquid nitrogen until use. The freezing medium contained 90% fetal bovine serum (FBS, Gibco, MA, USA) and 10% DMSO (Sigma-Aldrich, MI, USA). To detect SARS-CoV-2-specific B cells, biotinylated protein antigens were individually multimerized with fluorescently labelled streptavidin at 4 °C for 1 h, as previously described [26]. Briefly, a recombinant biotinylated SARS-CoV-2 spike (S1 + S2; aa16-1211) was purchased from R&D systems (Minnesota, USA) (BT10549).
B cell subsets were identified based on the expression of CD19, CD27, CD24, and CD38 markers by flow cytometry performed before freezing cell samples. MBCs were defined as CD19+CD24+CD27+CD38– and atypical MBCs (ATMs) were identified as CD19+CD27–CD24–CD38– [27,28]. Stained PBMC samples were acquired on FACs LSRFortessa (BD Bioscience, NJ, USA). At least 4 × 106 cells were acquired and analyzed using FlowJo10.7.1 (BD Bioscience). The phenotype analysis of antigen-specific B cells was only performed in subjects with at least 10 cells detected in the corresponding antigen-specific gate. Figure S1 shows the gating strategy.
2.4. Detection of SARS-CoV-2-Specific T Cells
T cell subsets were identified based on the expression of CD3, CD4, CD8, and CD45 markers by flow cytometry performed before freezing cell samples. The analysis of spike-specific cellular immunity was performed in 9 immunized, 15 convalescent, and 3 selected convalescent/immunized patients. We did not analyze spike-specific T cells frequency in 19 immunized, 19 convalescents, and 17 convalescent/immunized patients, due to the lack of blood samples.
We used an IFNγ ELISpot assay (Mabtech, Nacka Strand, Sweden), as previously described [29]. Briefly, T cells were plated in duplicate, with 2 × 105 cells/well, stimulated with 1 µg/mL CRUDE PepMix™ SARS-CoV-2 (Spike Glycoprotein, JPT Peptide Technologies, Berlin, Germany) and incubated at 37 °C for 24 h. In all experiments, T cells were also incubated with serum-free CellGenixTM GMP (Cell Genix, GMBH, Freiburg, Germany) as a negative control. As a positive control, PBMCs were stimulated with 5 µg/mL of phytohemoagglutinin-P (PHA, Sigma-Aldrich). The IFNγ+ spot-forming unit (SFU) was counted with EliScan (Epson) by Automated ELisa-Spot Assay Video Analysis Systems (A.EL.VIS GmbH, Hannover, Germany). Data were presented as the percentage of IFNγ SFUs obtained after pepMix stimulation, compared to that of the total SFUs obtained in the positive control condition (PHA).
2.5. Statistical Analysis
Data obtained post-immunization, post-SARS-CoV-2 infection, and post-immunization in convalescent participants were separately analyzed. Only for the spike-specific IgG evaluation, data from patients treated with SARS-CoV-2 monoclonal antibodies (MoAbs) were separately analyzed. Demographics were summarized with descriptive statistics (continuous values of the median and the interquartile range IQR). A univariate analysis assessed the impact of variables of interest. Values were compared by two-tailed Mann–Whitney U-test or by Wilcoxon matched-pairs signed-rank test (convalescents vs. convalescents/immunized). Data were represented as individual data and median data (horizontal lines). The dotted area represented the IQR range recorded in healthy donors, as previously reported [26]. The comparison between continuous parameters was assessed by simple linear regression analysis. Differences were deemed significant when p < 0.05. The Statistical Package for Social Sciences version 15 (SPSS Inc., 233 South Wacker Drive, 11th Floor, Chicago, USA) was used for the analysis.
3. Results
3.1. Patients
Blood samples were collected from 34 SARS-CoV-2 convalescent CVID patients (median age = 49.5 years; IQR = 45–59; 47% females) after a median of 86.5 days (IQR = 51.7–120) days from the first positive RT-PCR by nasopharyngeal swab. Convalescent patients were compared to 38 CVID patients naïve to SARS-CoV-2 who were immunized by two doses of the BNT162b2 vaccine (median age = 54 years; IQR = 42–60; 66% females). Demographic and immunological characteristics and comorbidities in the study groups are summarized in Table 1.
Table 1.
Demographics, immunological, and primary antibody defect (PAD)-related characteristics in common variable immunodeficiency (CVID) participants.
| Group 1 Immunized n = 38 | Group 2 Convalescents n = 34 | Group 3 Convalescent/Immunized n = 20 | Groups 1 vs. 2 p-Value | Groups 1 vs. 3 p-Value | Groups 2 vs. 3 p-Value | |
|---|---|---|---|---|---|---|
| Sex, n (%) | 25 (66) | 16 (47) | 12 (60) | 0.157 | 0.776 | 0.408 |
| Age, years, median (IQR) | 54.5 (42.5–60.0) | 49.5 (44.7–59.2) | 49.5 (44.2–62.7) | 0.840 | 0.973 | 0.943 |
| PAD-related complication | ||||||
| Autoimmunity, n (%) | 18 (47) | 9 (26) | 7 (35) | 0.094 | 0.414 | 0.550 |
| Chronic lung disease, n (%) | 8 (21) | 20 (58) | 10 (50) | 0.001 | 0.036 | 0.560 |
| Cancer, n (%) | 2 (5) | 2 (6) | 2 (10) | 1.000 | 0.602 | 0.622 |
| Immunosuppressive treatment, n (%) | 10 (26) | 3 (9) | 2 (10) | 0.069 | 0.186 | 1.000 |
| IgG, g/L, median (IQR) | 6.9 (5.8–8.1) | 7.5 (6.5–8.8) | 7.6 (7.2–8.8) | 0.069 | 0.122 | 0.881 |
| IgA, mg/dL, median (IQR) | 7.0 (0–25.0) | 6.0 (0–28.0) | 4.5 (0–12.8) | 0.541 | 0.599 | 0.401 |
| IgM, mg/dL, median (IQR) | 19.6 (4–42) | 20.0 (5–25.1) | 9.5 (4–25) | 0.576 | 0.355 | 0.648 |
| CD3+, cell/mm3, median (IQR) | 924 (748–1512) | 974 (675–1548) | 879 (639–1412) | 0.513 | 0.255 | 0.335 |
| CD4+, cell/mm3, median (IQR) | 452 (356–811) | 511 (356–811) | 479 (335–838) | 0.756 | 0.801 | 0.992 |
| CD8+, cell/mm3, median (IQR) | 421 (186–692) | 369 (269–710) | 297 (242–521) | 0.741 | 0.281 | 0.095 |
| CD19+, cell/mm3, median (IQR) | 87 (20–184) | 91 (29–172) | 55 (30–131) | 0.564 | 0.150 | 0.272 |
| CD19+CD27+ IgM–IgD–, %, median (IQR) | 3.5 (1–7.7) | 2.0 (0–5.0) | 2.5 (0–5.5) | 0.649 | 0.921 | 0.771 |
| CD19+CD27+IgM–IgD–, cell/mm3, median (IQR) | 1 (0–5.8) | 1.6 (0–4.1) | 2.3 (0.1–4.6) | 0.480 | 0.474 | 0.739 |
| Atypical MBC (ATM) CD19+CD24-CD27–CD38–CD21–, % of CD19+, median (IQR) | 3.69 (2.8–8.1) | 3.4 (2.3–5.2) | 3.1 (2.9–4.1) | 0.469 | 0.613 | 0.863 |
The clinical symptoms of COVID-19, treatments, and outcomes in the convalescents are summarized in Table 2. According to the World Health Organization criteria 2020, 14 (41%) patients were classified as asymptomatic COVID-19, 9 (26%) were classified as mild, 9 (26%) were classified as moderate, and 2 (7%) were classified as severe COVID-19 [30]. The median time of SARS-CoV-2 RT-PCR positivity was 27 days (IQR = 17–51). Nine patients developed pneumonia, and no patients were admitted to the ICU. Fourteen out of 34 patients received COVID-19 treatment. Five of them were treated with SARS-CoV-2 MoAbs (median age = 59 years; IQR = 50–64; 40% females). Infection outcome was favorable in all patients, with the exception of two patients who required long-term oxygen therapy due to the worsening in their underlying CLD.
Table 2.
SARS-CoV-2 infections, treatments, and outcomes in 34 CVID patients.
| ID | Age (Range) | Time Range of SARS-CoV-2 Infection | SARS-CoV-2 Infection Severity | SARS-CoV-2Associated Symptoms | Pneumonia | Days of SARS-CoV-2 qPCR Positivity | Additional COVID-19 Specific Therapy | Outcome | BNT162b2 Immunization |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 30–39 | March–May 2020 | moderate | fever and cough dyspnea | yes | 45 | lopinavir/ritonavir, tocilizumab, and dexamethasone | recovery | no |
| 2 | 50–59 | June–September 2020 | asymptomatic | no | 79 | no | recovery | yes, 180 days from infection | |
| 3 | 40–49 | January–April 2021 | asymptomatic | no | 30 | no | recovery | no | |
| 4 | 60–60 | October–December 2020 | asymptomatic | no | 14 | no | recovery | no | |
| 5 | 50–59 | October–December 2020 | asymptomatic | no | 81 | no | recovery | no | |
| 6 | 50–59 | June–September 2020 | asymptomatic | no | 51 | no | recovery | no | |
| 7 | 30–39 | January–April 2021 | moderate | fever, cough, and dyspnea | no | 23 | no | recovery | no |
| 8 | 40–49 | October–December 2020 | moderate | fever, cough, and dyspnea | yes | 75 | lopinavir/ritonavir, tocilizumab, and dexamethasone | dyspnoea and O2 therapy | no |
| 9 | 30–39 | January–April 2021 | mild | fever | no | 21 | dexamethasone | recovery | no |
| 10 | 40–49 | January–April 2021 | moderate | fever, cough, and dyspnea | yes | 22 | remdesivir and dexamethasone | recovery | no |
| 11 | 60–69 | January–April 2021 | mild | fever | no | 17 | bamlanivimab/ etesevimab | recovery | no |
| 12 | 50–59 | January–April 2021 | moderate | fever, cough, and dyspnea | yes | 112 | calsirimab/imdevimab, remdesivir, and dexamethasone | recovery | no |
| 13 | 40–49 | January–April 2021 | moderate | fever, cough, and dyspnea | yes | 40 | calsirimab/imdevimab, remdesivir, and dexamethasone | dyspnoea and O2 therapy | no |
| 14 | 60–69 | January–April 2021 | asymptomatic | no | 33 | bamlanivimab/ etesevimab | recovery | no | |
| 15 | 50–59 | January–April 2021 | asymptomatic | no | 23 | bamlanivimab | recovery | no | |
| 16 | 40–49 | October–December 2020 | asymptomatic | no | NA | no | recovery | yes, 150 days from infection | |
| 17 | 30–39 | October–December 2020 | asymptomatic | no | NA | no | recovery | yes, 150 days from infection | |
| 18 | 40–49 | January–April 2021 | moderate | fever and mild dyspnea | yes | 49 | dexamethasone and azithromycin | recovery | yes, 120 days from infection |
| 19 | 20–29 | October–December 2020 | asymptomatic | no | 15 | no | recovery | yes, 120 days from infection | |
| 20 | 50–59 | March–May 2020 | severe | fever, cough, and severe dyspnea | yes | 10 | lopinavir/ritonavir, azithromycin, and hydroxychloroquine | recovery | yes, 330 days from infection |
| 21 | 60–69 | October–December 2020 | moderate | fever and mild dyspnea | yes | 60 | NA | recovery | yes, 150 days from infection |
| 22 | 50–59 | January–April 2021 | mild | ageusia | no | 9 | no | recovery | yes, 90 days from infection |
| 23 | 50–59 | October–December 2020 | mild | fever | no | NA | no | recovery | yes, 180 days from infection |
| 24 | 40–49 | January–April 2021 | asymptomatic | no | 22 | no | recovery | yes, 90 days from infection | |
| 25 | 40–49 | January–April 2021 | asymptomatic | no | 9 | no | recovery | yes, 120 days from infection | |
| 26 | 50–59 | October–December 2020 | asymptomatic | no | 16 | no | recovery | yes, 120 days from infection | |
| 27 | 40–49 | October–December 2020 | mild | fever and cough | no | 23 | dexamethasone and azithromycin | recovery | yes, 150 days from infection |
| 28 | 60–69 | October–December 2020 | mild | fever and arthralgia | no | 20 | no | recovery | yes, 150 days from infection |
| 29 | ≥70 | October–December 2020 | moderate | fever, mild dyspnea, and ageusia | no | 27 | dexamethasone and azithromycin | dyspnoea | yes, 90 days from infection |
| 30 | 40–49 | October–December 2020 | asymptomatic | no | 30 | no | recovery | yes, 150 days from infection | |
| 31 | 20–29 | January–April 2021 | mild | ageusia and anosmia | no | 59 | no | recovery | yes, 120 days from infection |
| 32 | 60–69 | October–December 2020 | mild | anosmia | no | 34 | no | recovery | yes, 150 days from infection |
| 33 | 60–69 | January–April 2021 | mild | arthralgia | no | 12 | no | dyspnoea | yes, 120 days from infection |
| 34 | 40–49 | October–December 2020 | severe | fever and moderate dyspnea | yes | 53 | dexamethasone and azithromycin | recovery | yes, 120 days from infection |
Among convalescents, 20 patients were immunized by BNT162b2 COVID-19 vaccine after a median of 94.2 days (IQR = 80.2–140.5) from the first SARS-CoV-2-negative test. In these convalescent/immunized patients, blood samples were collected after a median of 21 days (IQR = 21–45) from immunization. The immunized patients, convalescent patients, and convalescent/immunized patients did not differ for demographics and PAD comorbidities. Only CLD was more frequently observed in the SARS-CoV-2-infected patients (Table 1).
3.2. SARS-CoV-2 Antibodies
Anti-spike IgG response was assessed in immunized, convalescent, and convalescents/immunization. In the three groups, the values of IgG S1 antibodies were significantly increased in comparison to the BL values. After infection, a higher number of convalescent CVID patients showed a detectable anti-spike IgG antibody response (IgG S1 > 1.1 OD ratio; 24/29, 82%) in comparison to the number of responders in the immunized group (14/38; 34%; p = 0.0002). However, the median anti-spike IgG serum level was not significantly higher in the convalescent group than in the immunized group (p = 0.553; Figure 1A).
Figure 1.
Spike-specific IgG levels in the CVID cohort (panel (A)) Data are shown at baseline (BL) after immunization (immunized, n = 38), after infection (convalescent, n = 34), and after immunization in convalescents (convalescent/immunized n = 20). Data from monoclonal antibodies (MoAbs, n = 5) recipients are shown separately. Changes in individual data points of spike-specific IgG antibodies from BL to post-immunization and from SARS-CoV-2 recovery to post-immunization are shown in panel (B). The positive cut-off values are represented by a dashed line. For each group, medians are plotted as horizontal bars. The dotted area represents the IQR range in healthy donors (HD) after immunization as previously reported [26]. **** p < 0.0001; *** p < 0.001; p-value represents the level of significance by two-tailed Mann–Whitney U-test (immunized vs convalescent/immunized and convalescent vs convalescent (MoAbs)) or by Wilcoxon matched-pairs signed-rank test (BL vs immunized and convalescents vs convalescents/immunized). Abbreviation: BL baseline, IgG S1: Spike specific IgG, OD: optical density, MoAbs: Monoclonal antibodies treatment.
The highest IgG S1 response was observed in the group of convalescents/immunized, where the post-infection antibody response was boosted after immunization (p = 0.001), and in infected patients who underwent anti-spike MoAbs treatment. To note, two out of five convalescent patients who were seronegative after SARS-CoV-2 infection seroconverted after immunization. The summary of IgG S1 in CVID was reported in Table 3, showing spike-specific IgG antibodies in 38 CVID patients before (BL) and one week after the second dose of the BNT162b2 vaccine (immunized) and in 20 CVID patients after recovery from SARS-CoV2 infection (convalescent) and after one dose of the BNT162b2 vaccine (convalescent/immunized).
Table 3.
SARS-CoV-2 anti-spike antibody immunoassay results and S1 memory B cells (MBCs) in CVID patients.
| IgG S1 (OD Ratio) | MBC S + (% of CD24 + CD27 + CD38–Inside CD19 + Cells) |
ATM S + (% of CD24–CD27–CD38–CD21–Inside CD19+ Cells) | |
|---|---|---|---|
| Baseline, median (IQR) | 0.11 (0.08–0.18) | 0 (0–0.98) | 0 (0–0) |
| Immunized, median (IQR) | 0.30 (0.09–5.39) | 0.07 (0–0.13) | 0 (0–0.27) |
| Convalescent, median (IQR) | 2.5 (1.07–4.04) | 0.16 (0.22) | 0 (0–0) |
| Convalescent/immunized median (IQR) | 9.6 (2.75–12.33) | 0.25 (1.17–0.31) | 0.50 (0.39–1.40) |
| MoAbs treatment, median (IQR) | 6.91 (6.56–9.42) | Nap | Nap |
Abbreviations: MBC S+, spike-specific memory B cells; ATM S+, spike-specific atypical memory B cells; Nap, not applicable.
The individual data points of spike-specific IgG antibodies from BL to post-immunization and from SARS-CoV-2 recovery to post-immunization after one dose of the BNT162b2 vaccine (convalescent/immunized) are presented in Figure 1, panel B. The positive cut-off values are represented by a dashed line.
In our cohort, patients who did not mount a detectable antibody response after immunization had a lower frequency of switched MBCs (p = 0.0005) and lower serum IgA and IgM levels (p = 0.0002 and p = 0.042, respectively). In detail, 10% of those with low frequencies of switched MBCs (<2%) showed a detectable humoral response. Differently, in the convalescents, we could not identify any immunological or clinical signatures associated with the different antibody response after infection. To note, 50% of patients with low frequencies of switched MBCs were able to mount a detectable humoral response. The convalescents with more severe COVID-19 courses did not develop more frequently a detectable IgG S1 response (Table S1).
3.3. Spike-Specific SARS-CoV-2 MBCs
High specificity and affinity are the most important characteristics of protective MBCs, generated by the adaptive immune system in response to infection or vaccination in the GC [31], thanks to the mechanisms of somatic mutation and affinity selection [32]. After immunization or infection, ATMs become transiently detectable in the peripheral blood, mostly generated by extrafollicular reactions where antigen selection does not occur [33].
Recently, we showed that vaccination induces spike-specific (S+) ATMs in about one-third of patients in CVID [26], different from healthy subjects who respond by generating S+ MBCs (Figure S2) [34].
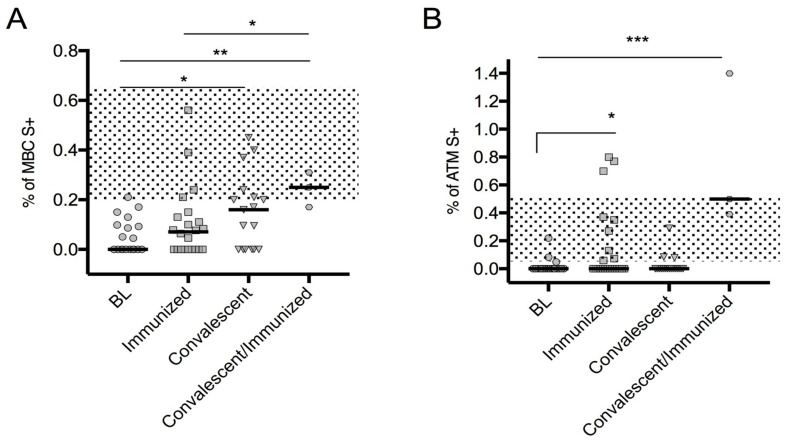
Here, we recorded that convalescent CVID generated S+ MBCs cells, but not S+ ATMs. Remarkably, after immunization, the convalescents generated also S+ ATMs and developed an even more evident classical spike-specific MBC response. In this cohort, we confirmed that immunized CVID patients generated specific S+ ATMs, but not S+ MBCs (Figure 2).
Figure 2.
Spike-specific MBC subsets in the CVID cohort. Spike-specific MBCs (A) and ATMs (B) data are shown at BL (n = 23), after immunization (immunized, n = 23), after SARS-CoV-2 infection (convalescent, n = 17), and after immunization in SARS-CoV-2 convalescents (convalescent/immunized, n = 3). Medians are plotted as horizontal bars. The dotted area represents the IQR range in HD after immunization as previously reported [26]. Levels of significance were measured by two-tailed Mann–Whitney U-test and for Wilcoxon matched-pairs signed-rank test (comparison between convalescents and convalescents/immunized only): *** p < 0.001; ** p < 0.01; * p < 0.05. Abbreviations: BL, baseline; MBC S+, spike-specific memory B cells; ATM S+, atypical memory B cells.
The spike-specific B cells subset frequencies were similar among patients with severe/moderate COVID-19 or mild/asymptomatic infection (data not shown). Moreover, the B cells subset frequencies were similar in patients when grouped accordingly to their ability to develop a detectable IgG S1 antibody response, both after immunization and after infection (Table S1). No linear correlation was observed between the frequencies of the specific B cells subset and IgG S1 levels (R = 0.003; p = 0.855). The Frequencies of MBCs++ and ATMs S++ were not shown, because CVID patients are unable to produce them.
3.4. SARS-CoV-2 T Cell-Specific ELISpot Response
T cell activity was indirectly analyzed by measuring the concentration of IFNγ secreted by activated lymphocytes after 24 h of the in vitro stimulation. On the basis of the cut-off of spot increments of IFNγ, we calculated the positive SARS-CoV-2 T cell-specific response after stimulation with the spike SARS-CoV-2. The median percentage of the positive response was 1.2% in immunized CVID (IQR = 0.15–3.15) and 1.04% (IQR = 0.0–4.8) in convalescents. These values were lower than previously reported in the healthy donors (median = 9%; IQR = 5–30) [25]. The absence of specific T cell responses was also observed in convalescent/immunized CVID patients (Figure 3).
Figure 3.
SARS-CoV-2 T cell-specific response in CVID patients. Data are shown as the percentage of increment IFNγ production after T cells stimulation with the spike SARS-CoV-2 protein at BL (n = 9), after immunization (immunized, n = 9), after COVID-19 recovery (convalescent, n = 15), and in convalescents after immunization (convalescent/immunized, n = 3). Percentage distributions and medians (horizontal lines) are shown. The dotted area represents the IQR range in HD after immunization as previously reported [26]. Values were compared by two-tailed Mann–Whitney U-test or Wilcoxon matched-pairs signed-rank test (comparison between convalescents and convalescents/immunized only). Abbreviations: BL, baseline; IFN-γ, interferon gamma spot-forming unit.
4. Discussion
In CVID, the impaired immune response after infections and immunization accounts for the high infection susceptibility [3], requiring specific prevention and treatment strategies to minimize morbidity and mortality. In the current SARS-CoV-2 pandemic, 6.3% of CVID patients attending our collaborative centers became infected. Consistent with other reports [10,12,35], about 65% of patients had a mild or asymptomatic COVID-19 course. Since vaccination became available, almost all 335 CVID patients from this cohort were immunized with an mRNA COVID-19 vaccine (BNT162b2). A single dose of mRNA vaccine was administered in 20 convalescent patients starting from 30 days from the infection recovery.
This study was planned in order to evaluate if natural SARS-CoV-2 infection and immunization induced different adaptive immune responses in CVID patients. Our results showed that vaccination and infection primed different B cells responses. Moreover, the humoral immune response induced by natural infection was significantly enhanced by subsequent immunization. The antibody response was influenced neither by the severity of COVID-19 nor by the duration of viral replication.
Natural infection generated a detectable antibody IgG response in more than 80% of convalescent patients, a surprisingly higher proportion in comparison to in fully immunized CVID patients, who seroconverted only in 34% of cases. Here, we confirmed our previous data showing low adaptive responses to immunization with the BNT162b2 vaccine in CVID subjects [26]. Our data contrasted with the high frequency of response reported in small heterogeneous cohorts of immunized PAD patients [35,36,37,38].
However, the improved antibody response by a single vaccine dose in convalescent patients is in line with observations in convalescent/vaccinated immunocompetent subjects [39,40]. In our cohort, immunization was able to induce a detectable antibody response also in two out of five CVID seronegative convalescents. An analogous increase of the seroconversion rate after immunization has been recently observed also in other SARS-CoV-2 convalescent fragile patients, including organ transplant recipients [20,40].
The differences in antibody response between immunization and natural infection mirrored the B cell subsets phenotype and the spike-specific B cell responses. The frequency of switched CD27+ MBCs affected the antibody response in vaccinated, but not in convalescent CVID, patients, pointing out that natural infection can induce an antibody response also in those having a severe B memory cell defect. More importantly, only convalescents generated spike-specific MBCs, whereas immunized generated spike-specific ATM only, revealing alternative patterns of the B cell response. Interestingly, we recorded that spike-specific ATMs were also elicited by immunization in convalescents.
Impaired post-GC B cell maturation, severely reduced circulating class-switched MBCs, and persistent circulating ATMs are the most consistent defects in CVID [4,40,41]. In comparison to classical MBCs, ATMs display hypo-responsiveness to B cell receptor (BCR) stimulation, upregulation of inhibitory receptors, and limited antibody secretion upon stimulation [42,43].
Different from the spike-specific B cell response observed in immunized CVID, the B cell response in convalescents CVID was consistent with that observed in immunized immunocompetent individuals, who generated spike-specific MBCs switching from the IgM+ to IgM– isotype, due to the process of affinity maturation and class-switching in the GC [26]. In immunocompetent subjects, it has been suggested that the infection generates more robust GC responses than the mRNA vaccine [44] and spike-specific B cell responses differ in immunized and convalescents with striking differences in the cell composition and transcriptional profiles of circulating immune cells, possibly linked to the different milieu induced by activated innate immune cells in infected patients [45]. Likewise, it is then possible to hypothesize a different pattern of response observed in immunized, convalescent, and convalescent/immunized patients, which is even more complex by the multiple innate [46,47,48] and adaptive immune abnormalities described in CVID [4,20,40,49,50,51,52]. Thus, also in CVID, the comparison of immune responses generated by the vaccine and the infection shed light on the difference between an antigen-driven response and an infection-driven response where the inflammation directs the subsequent adaptive immune response.
Different from what was observed in immunocompetent individuals after immunization and in small cohorts of PAD patients that mounted a robust antigen-specific CD8+ and CD4+ T cell responses after vaccination and natural infection [53,54], we recorded a poor T cell response after immunization, after infection, and after vaccination in convalescent patients. While in CVID influenza virus immunization generates specific T cells after multiple exposures to viral antigen, the poor spike-specific T cell response might be a consequence of a limited antigenic stimulation by a new pathogen, which has never been encountered before [55]. At present, it might be assumed that patients with poor T cell responses might require multiple doses or combinations of SARS-CoV-2 vaccines to obtain a possible protection. The main limitation of this study is the limited number of samples analyzed for specific B and T cell responses in convalescents who have been immunized. Further analysis is ongoing to assess in the future for the dynamic of the immune responses in this population of patients. Our observations underline the need of vaccination in convalescent CVID patients. Then, immunization by a single dose of mRNA vaccine should be recommended in all CVID convalescent patients from SARS-CoV-2 infection starting from 30 days from infection recovery [56]. For patients naive to infection, a third “booster” dose could be necessary to achieve a better immune response against COVID-19.
Acknowledgments
We would thank patients and their families.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/cells10112915/s1, Figure S1. Gating strategy to identify S+ and S++ MBCs and ATMs. Flow cytometry plots showing the staining patterns of SARS-CoV-2 antigen probes in MBCs (identified as CD24+CD27+CD38–) and ATMs (identified as CD24CD27–CD38–CD21–) populations in one HD and one CVID subjects. The color code identifies spike-positive MBCs (blue) and ATMs (pink). Figure S2. Spike-specific memory B cell subsets in the healthy cohort. Spike-specific MBCs (A) and ATMs (B) data are shown at BL (n = 28), after immunization (immunized, n = 28), and after immunization in SARS-CoV-2 convalescents (convalescent/immunized, n = 9). Medians are plotted as horizontal bars. Levels of significance by Wilcoxon matched-pairs signed-rank test or two-tailed Mann–Whitney U-test: **** p < 0.0001, *** p < 0.001, ** p < 0.01. Table S1. Demographic, clinical, and immunological characteristics of CVID enrolled, stratified by antibody response to a two-dose mRNA vaccine or to natural infection.
Author Contributions
F.P., A.F.S., R.C., G.S., F.L. and I.Q. designed the study. A.F.S., E.P.M., S.T., C.Q., M.G., C.A. and S.D.C. performed data experiments. F.P., A.F.S., E.P.M., S.T. and C.M. performed data analysis and helped with manuscript preparation. G.L., A.P., F.V., S.A., C.M. and L.B. helped with the collection of study samples and clinical information and with the manuscript preparation. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by: Italian Ministry of Health RF2013-02358960 grant; Italian Ministry of Health COVID-2020-12371817 grant.
Institutional Review Board Statement
The study was approved by the Ethical Committee of the Sapienza University of Rome (Prot. 0521/2020, 13 July 2020).
Informed Consent Statement
All participants signed the informed consents for vaccination and the immunological study and provided consent for the publication of data.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
Authors declare that they have no competing interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Picard C., Gaspar H.B., Al-Herz W., Bousfiha A., Casanova J.-L., Chatila T., Crow Y.J., Cunningham-Rundles C., Etzioni A., Franco J.L., et al. International Union of Immunological Societies: 2017 Primary Immunodeficiency Diseases Committee Report on Inborn Errors of Immunity. J. Clin. Immunol. 2017;38:96–128. doi: 10.1007/s10875-017-0464-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Quinti I., Soresina A., Spadaro G., Martino S., Donnanno S., Agostini C., Claudio P., Franco D., Pesce A.M., Borghese F., et al. Long-Term Follow-Up and Outcome of a Large Cohort of Patients with Common Variable Immunodeficiency. J. Clin. Immunol. 2007;27:308–316. doi: 10.1007/s10875-007-9075-1. [DOI] [PubMed] [Google Scholar]
- 3.Bonilla F.A., Barlan I., Chapel H., Costa-Carvalho B.T., Cunningham-Rundles C., De La Morena M.T., Espinosa-Rosales F.J., Hammarström L., Nonoyama S., Quinti I., et al. International Consensus Document (ICON): Common Variable Immunodeficiency Disorders. J. Allergy Clin. Immunol. Pract. 2015;4:38–59. doi: 10.1016/j.jaip.2015.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wehr C., Kivioja T., Schmitt C., Ferry B., Witte T., Eren E., Vlkova M., Hernandez-Gonzalez M., Detkova D., Bos P.R., et al. The EUROclass trial: Defining subgroups in common variable immunodeficiency. Blood. 2008;111:77–85. doi: 10.1182/blood-2007-06-091744. [DOI] [PubMed] [Google Scholar]
- 5.Guan W.-J., Liang W.-H., Zhao Y., Liang H.-R., Chen Z.-S., Li Y.-M., Liu X.-Q., Chen R.-C., Tang C.-L., Wang T., et al. Comorbidity and its impact on 1590 patients with COVID-19 in China: A nationwide analysis. Eur. Respir. J. 2020;55:2000547. doi: 10.1183/13993003.00547-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Minotti C., Tirelli F., Barbieri E., Giaquinto C., Dona D. How is immunosuppressive status affecting children and adults in SARS-CoV-2 infection? A systematic review. J. Infect. 2020;8:e61–e66. doi: 10.1016/j.jinf.2020.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Van Damme K.F.A., Tavernier S., Van Roy N., De Leeuw E., Declercq J., Bosteels C., Maes B., De Bruyne M., Bogaert D., Bosteels V., et al. Case Report: Convalescent Plasma, a Targeted Therapy for Patients with CVID and Severe COVID-19. Front. Immunol. 2020;11:596761. doi: 10.3389/fimmu.2020.596761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ribeiro L.C., Benites B.D., Ulaf R.G., Nunes T.A., Costa-Lima C., Addas-Carvalho M., Proenca-Modena J.L., Granja F., da Costa V.A., Duarte A.D.S.S., et al. Rapid clinical recovery of a SARS-CoV-2 infected common variable immunodeficiency patient following the infusion of COVID-19 convalescent plasma. Allergy Asthma Clin. Immunol. 2021;17:14. doi: 10.1186/s13223-021-00518-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gupta S., Su H., Narsai T., Agrawal S. SARS-CoV-2-Associated T-Cell Responses in the Presence of Humoral Immunodeficiency. Int. Arch. Allergy Immunol. 2021;182:195–209. doi: 10.1159/000514193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Meyts I., Bucciol G., Quinti I., Neven B., Fischer A., Seoane E., Lopez-Granados E., Gianelli C., Robles-Marhuenda A., Jeandel P.-Y., et al. Coronavirus disease 2019 in patients with inborn errors of immunity: An international study. J. Allergy Clin. Immunol. 2020;147:520–531. doi: 10.1016/j.jaci.2020.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Quinti I., Lougaris V., Milito C., Cinetto F., Pecoraro A., Mezzaroma I., Mastroianni C.M., Turriziani O., Bondioni M.P., Filippini M., et al. A possible role for B cells in COVID-19? Lesson from patients with agammaglobulinemia. J. Allergy Clin. Immunol. 2020;146:211–213. doi: 10.1016/j.jaci.2020.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Milito C., Lougaris V., Giardino G., Punziano A., Vultaggio A., Carrabba M., Cinetto F., Scarpa R., Delle Piane R.M., Baselli L., et al. Clinical outcome, incidence, and SARS-CoV-2 infection-fatality rates in Italian patients with inborn errors of immunity. J. Allergy Clin. Immunol. Pract. 2021;9:2904–2906. doi: 10.1016/j.jaip.2021.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ho H.-E., Mathew S., Peluso M.J., Cunningham-Rundles C. Clinical outcomes and features of COVID-19 in patients with primary immunodeficiencies in New York City. J. Allergy Clin. Immunol. Pract. 2020;9:490–493. doi: 10.1016/j.jaip.2020.09.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Aljaberi R., Wishah K. Positive outcome in a patient with coronavirus disease 2019 and common variable immunodeficiency after intravenous immunoglobulin. Ann. Allergy Asthma Immunol. 2020;125:349–350. doi: 10.1016/j.anai.2020.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Marcus N., Frizinsky S., Hagin D., Ovadia A., Hanna S., Farkash M., Maoz-Segal R., Agmon-Levin N., Broides A., Nahum A., et al. Minor Clinical Impact of COVID-19 Pandemic on Patients With Primary Immunodeficiency in Israel. Front. Immunol. 2021;11:614086. doi: 10.3389/fimmu.2020.614086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Milito C., Soccodato V., Auria S., Pulvirenti F., Quinti I. COVID-19 in complex common variable immunodeficiency patients affected by lung diseases. Curr. Opin. Allergy Clin. Immunol. 2021 doi: 10.1097/ACI.0000000000000789. epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 17.Polack F.P., Thomas S.J., Kitchin N., Absalon J., Gurtman A., Lockhart S., Perez J.L., Pérez Marc G., Moreira E.D., Zerbini C., et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N. Engl. J. Med. 2020;383:2603–2615. doi: 10.1056/NEJMoa2034577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Saad-Roy C.M., Morris S.E., Metcalf C.J., Mina M.J., Baker R.E., Farrar J., Holmes E.C., Pybus O.G., Graham A.L., Levin S.A., et al. Epidemiological and evolutionary considerations of SARS-CoV-2 vaccine dosing regimens. Science. 2021;372:363–370. doi: 10.1126/science.abg8663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sahin U., Muik A., Vogler I., Derhovanessian E., Kranz L.M., Vormehr M., Quandt J., Bidmon N., Ulges A., Baum A., et al. BNT162b2 vaccine induces neutralizing antibodies and poly-specific T cells in humans. Nature. 2021;595:572–577. doi: 10.1038/s41586-021-03653-6. [DOI] [PubMed] [Google Scholar]
- 20.Wei J., Stoesser N., Matthews P.C., Ayoubkhani D., Studley R., Bell I., Bell J.I., Newton J.N., Farrar J., Diamond I., et al. Antibody responses to SARS-CoV-2 vaccines in 45,965 adults from the general population of the United Kingdom. Nat. Microbiol. 2021;6:1140–1149. doi: 10.1038/s41564-021-00947-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Raccomandazioni ad Interim sui Gruppi Target della Vaccinazione Anti-SARS-CoV-2/COVID-19. [(accessed on 16 September 2021)]; Available online: https://www.trovanorme.salute.gov.it/norme/renderPdf.spring?seriegu=SG&datagu=24/03/2021&redaz=21A01802&artp=1&art=1&subart=1&subart1=10&vers=1&prog=002.
- 22.ESID Positions on SARS-CoV-2 Infections in Inborn Errors of Immunity. [(accessed on 16 September 2021)]. Available online: https://esid.org/COVID-19/ESID-COVID-19-Statement.
- 23.Kostinova A.M., Akhmatova N.K., Latysheva E.A., Dagil Y.A., Klimova S.V., Vlasenko A.E., Khromova E.A., Latysheva T.V., Kostinov M.P. Assessment of Immunogenicity of Adjuvanted Quadrivalent Inactivated Influenza Vaccine in Healthy People and Patients With Common Variable Immune Deficiency. Front. Immunol. 2020;11:1876. doi: 10.3389/fimmu.2020.01876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pulvirenti F., Milito C., Cavaliere F.M., Mezzaroma I., Cinetto F., Quinti I. IGA Antibody Induced by Immunization With Pneumococcal Polysaccharides Is a Prognostic Tool in Common Variable Immune Deficiencies. Front. Immunol. 2020;11:1283. doi: 10.3389/fimmu.2020.01283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Seidel M.G., Kindle G., Gathmann B., Quinti I., Buckland M., Van Montfrans J., Scheible R., Rusch S., Gasteiger L.M., Grimbacher B., et al. The European Society for Immunodeficiencies (ESID) Registry Working Definitions for the Clinical Diagnosis of Inborn Errors of Immunity. J. Allergy Clin. Immunol. Pract. 2019;7:1763–1770. doi: 10.1016/j.jaip.2019.02.004. [DOI] [PubMed] [Google Scholar]
- 26.Salinas A.F., Mortari E.P., Terreri S., Quintarelli C., Pulvirenti F., Di Cecca S., Guercio M., Milito C., Bonanni L., Auria S., et al. SARS-CoV-2 Vaccine Induced Atypical Immune Responses in Antibody Defects: Everybody Does their Best. J. Clin. Immunol. 2021;20:1–14. doi: 10.1007/s10875-021-01133-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ehrhardt G.R., Hsu J., Gartland L., Leu C.-M., Zhang S., Davis R.S., Cooper M.D. Expression of the immunoregulatory molecule FcRH4 defines a distinctive tissue-based population of memory B cells. J. Exp. Med. 2005;202:783–791. doi: 10.1084/jem.20050879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fecteau J.F., Côté G., Néron S. A new memory CD27-IgG+ B cell population in peripheral blood expressing VH genes with low frequency of somatic mutation. J. Immunol. 2006;177:3728–3736. doi: 10.4049/jimmunol.177.6.3728. [DOI] [PubMed] [Google Scholar]
- 29.Quintarelli C., Dotti G., Hasan S.T., De Angelis B., Hoyos V., Errichiello S., Mims M., Luciano L., Shafer J., Leen A.M., et al. High-avidity cytotoxic T lymphocytes specific for a new PRAME-derived peptide can target leukemic and leukemic-precursor cells. Blood. 2011;117:3353–3362. doi: 10.1182/blood-2010-08-300376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.World Health Organization Country & Technical Guidance—Coronavirus Disease 2019. [(accessed on 16 September 2021)]. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance.
- 31.Tangye S.G., Tarlinton D. Memory B cells: Effectors of long-lived immune responses. Eur. J. Immunol. 2009;39:2065–2075. doi: 10.1002/eji.200939531. [DOI] [PubMed] [Google Scholar]
- 32.Mesin L., Ersching J., Victora G.D. Germinal Center B Cell Dynamics. Immunity. 2016;45:471–482. doi: 10.1016/j.immuni.2016.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Braddom A.E., Batugedara G., Bol S., Bunnik E.M. Potential functions of atypical memory B cells in Plasmodium-exposed individuals. Int. J. Parasitol. 2020;50:1033–1042. doi: 10.1016/j.ijpara.2020.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Piano Mortari E., Russo C., Vinci M.R., Terreri S., Fernandez Salinas A., Piccioni L., Alteri C., Colagrossi L., Coltella L., Ranno S., et al. Highly Specific Memory B Cells Generation after the 2nd Dose of BNT162b2 Vaccine Compensate for the Decline of Serum Antibodies and Absence of Mucosal IgA. Cells. 2021;10:2541. doi: 10.3390/cells10102541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Goudouris E.S., Pinto-Mariz F., Mendonça L.O., Aranda C.S., Guimarães R.R., Kokron C., Barros M.T., Anísio F., Alonso M.L.O., Marcelino F., et al. Outcome of SARS-CoV-2 Infection in 121 Patients with Inborn Errors of Immunity: A Cross-Sectional Study. J. Clin. Immunol. 2021;23:1–11. doi: 10.1007/s10875-021-01066-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Squire J., Joshi A. Seroconversion after coronavirus disease 2019 vaccination in patients with immune deficiency. Ann. Allergy Asthma Immunol. 2021;127:383–384. doi: 10.1016/j.anai.2021.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Romano C., Esposito S., Donnarumma G., Marrone A. Detection of neutralizing anti-severe acute respiratory syndrome coronavirus 2 antibodies in patients with common variable immunodeficiency after immunization with messenger RNA vaccines. Ann. Allergy Asthma Immunol. 2021;4:499–501. doi: 10.1016/j.anai.2021.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hagin D., Freund T., Navon M., Halperin T., Adir D., Marom R., Levi I., Benor S., Alcalay Y., Freund N.T. Immunogenicity of Pfizer-BioNTech COVID-19 vaccine in patients with inborn errors of immunity. J. Allergy Clin. Immunol. 2021;148:739–749. doi: 10.1016/j.jaci.2021.05.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Steensels D., Pierlet N., Penders J., Mesotten D., Heylen L. Comparison of SARS-CoV-2 Antibody Response Following Vaccination With BNT162b2 and mRNA-1273. JAMA. 2021;15:1533–1535. doi: 10.1001/jama.2021.15125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Driessen G.J., Van Zelm M.C., Van Hagen P.M., Hartwig N.G., Trip M., Warris A., De Vries E., Barendregt B.H., Pico I., Hop W., et al. B-cell replication history and somatic hypermutation status identify distinct pathophysiologic backgrounds in common variable immunodeficiency. Blood. 2011;118:6814–6823. doi: 10.1182/blood-2011-06-361881. [DOI] [PubMed] [Google Scholar]
- 41.Kalina T., Stuchly J., Janda A., Hrušák O., Růžičková S., Šedivá A., Litzman J., Vlkova M. Profiling of polychromatic flow cytometry data on B-cells reveals patients’ clusters in common variable immunodeficiency. Cytom. Part A. 2009;75:902–909. doi: 10.1002/cyto.a.20801. [DOI] [PubMed] [Google Scholar]
- 42.Knox J.J., Buggert M., Kardava L., Seaton K.E., Eller M.A., Canaday D.H., Robb M.L., Ostrowski M.A., Deeks S.G., Slifka M.K., et al. T-bet+ B cells are induced by human viral infections and dominate the HIV gp140 response. JCI Insight. 2017;2:e92943. doi: 10.1172/jci.insight.92943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Jenks S.A., Cashman K.S., Zumaquero E., Marigorta U.M., Patel A.V., Wang X., Tomar D., Woodruff M.C., Simon Z., Bugrovsky R., et al. Distinct effector B cells induced by unregulated toll-like receptor 7 contribute to pathogenic responses in systemic lupus erythematosus. Immunity. 2018;49:725–739. doi: 10.1016/j.immuni.2018.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lederer K., Castaño D., Gómez Atria D., Oguin T.H., Wang S., Manzoni T.B., Muramatsu H., Hogan M.J., Amanat F., Cherubin P., et al. SARS-CoV-2 mRNA Vaccines Foster Potent Antigen-Specific Germinal Center Responses Associated with Neutralizing Antibody Generation. Immunity. 2020;53:1281–1295. doi: 10.1016/j.immuni.2020.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ivanova E.N., Devlin J.C., Buus T.B., Koide A., Cornelius A., Samanovic M.I., Herrera A., Zhang C., Desvignes L., Odum N., et al. Discrete immune response signature to SARS-CoV-2 mRNA vaccination versus infection. medRxiv. 2021 doi: 10.1101/2021.04.20.21255677. [DOI] [Google Scholar]
- 46.Casulli S., Coignard-Biehler H., Amazzough K., Shoai-Tehrani M., Bayry J., Mahlaoui N., Elbim C., Kaveri S.V. Defective functions of polymorphonuclear neutrophils in patients with common variable immunodeficiency. Immunol. Res. 2014;60:69–76. doi: 10.1007/s12026-014-8555-7. [DOI] [PubMed] [Google Scholar]
- 47.Trujillo C.M., Muskus C., Arango J., Patino P., Montoya C.J. Quantitative and functional evaluation of innate immune responses in patients with common variable immunodeficiency. J. Investig. Allergol. Clin. Immunol. 2011;21:207–215. [PubMed] [Google Scholar]
- 48.Park J., Munagala I., Xu H., Blankenship D., Maffucci P., Chaussabel D., Banchereau J., Pascual V., Cunningham-Rundles C. Interferon Signature in the Blood in Inflammatory Common Variable Immune Deficiency. PLoS ONE. 2013;8:e74893. doi: 10.1371/journal.pone.0074893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Azizi G., Rezaei N., Kiaee F., Tavakolinia N., Yazdani R., Mirshafiey A., Aghamohammadi A. T Cell Abnormalities in Common Variable Immunodeficiency. J. Investig. Allergol. Clin. Immunol. 2016;26:233–243. doi: 10.18176/jiaci.0069. [DOI] [PubMed] [Google Scholar]
- 50.Viallard J.-F., Ruiz C., Guillet M., Pellegrin J.-L., Moreau J.-F. Perturbations of the CD8+ T-cell repertoire in CVID patients with complications. Results Immunol. 2013;3:122–128. doi: 10.1016/j.rinim.2013.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Vlkova M., Ticha O., Nechvatalova J., Kalina T., Litzman J., Mauri C., Blair P.A. Regulatory B cells in CVID patients fail to suppress multifunctional IFN-γ+TNF-α+CD4+ T cells differentiation. Clin. Immunol. 2015;160:292–300. doi: 10.1016/j.clim.2015.06.013. [DOI] [PubMed] [Google Scholar]
- 52.Ochtrop M.L.G., Goldacker S., May A.M., Rizzi M., Draeger R., Hauschke D., Stehfest C., Warnatz K., Goebel H., Technau-Ihling K., et al. T and B lymphocyte abnormalities in bone marrow biopsies of common variable immunodeficiency. Blood. 2011;118:309–318. doi: 10.1182/blood-2010-11-321695. [DOI] [PubMed] [Google Scholar]
- 53.Ameratunga R., Longhurst H., Steele R., Lehnert K., Leung E., Brooks A.E., Woon S.-T. Common Variable Immunodeficiency Disorders, T-Cell Responses to SARS-CoV-2 Vaccines, and the Risk of Chronic COVID-19. J. Allergy Clin. Immunol. Pract. 2021;9:3575–3583. doi: 10.1016/j.jaip.2021.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kinoshita H., Durkee-Shock J., Jensen-Wachspress M., Kankate V.V., Lang H., Lazarski C.A., Keswani A., Webber K.C., Montgomery-Recht K., Walkiewicz M., et al. Robust Antibody and T Cell Responses to SARS-CoV-2 in Patients with Antibody Deficiency. J. Clin. Immunol. 2021;41:1146–1153. doi: 10.1007/s10875-021-01046-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Friedmann D., Goldacker S., Peter H.-H., Warnatz K. Preserved Cellular Immunity Upon Influenza Vaccination in Most Patients with Common Variable Immunodeficiency. J. Allergy Clin. Immunol. Pract. 2020;8:2332–2340. doi: 10.1016/j.jaip.2020.04.019. [DOI] [PubMed] [Google Scholar]
- 56.Ministry of Health Circular “Vaccination of Individuals with SARS-CoV.2 Infection”. [(accessed on 16 September 2021)]; Available online: https://www.trovanorme.salute.gov.it/norme/renderNormsanPdf?anno=2021&codLeg=79033&parte=1%20&serie=null.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data presented in this study are available on request from the corresponding author.