Abstract
Bacteria-targeting nanomaterials have been widely used in the diagnosis and treatment of bacterial infectious diseases. These nanomaterials show great potential as antimicrobial agents due to their broad-spectrum antibacterial capacity and relatively low toxicity. Recently, nanomaterials have improved the accurate detection of pathogens, provided therapeutic strategies against nosocomial infections and facilitated the delivery of antigenic protein vaccines that induce humoral and cellular immunity. Biomaterial implants, which have traditionally been hindered by bacterial colonization, benefit from their ability to prevent bacteria from forming biofilms and spreading into adjacent tissues. Wound repair is improving in terms of both the function and prevention of bacterial infection, as we tailor nanomaterials to their needs, select encapsulation methods and materials, incorporate activation systems and add immune-activating adjuvants. Recent years have produced numerous advances in their antibacterial applications, but even further expansion in the diagnosis and treatment of infectious diseases is expected in the future.
Keywords: nanoparticle, silver nanoparticle, biofilms, implants, bacterial infection, polymers
1. Introduction
Antimicrobial resistance in bacteria has become a substantial threat to human health in recent years; however, few new antimicrobial agents with innovative mechanisms of action are available to effectively combat these bacteria [1]. Moreover, many bacteria form complex communities called biofilms in their environment; biofilms provide advantages to bacteria, such as enhanced resistance toward antibiotic challenge [2]. Nanomedicine is a category that means the application of nanotechnology in medical conditions and is defined as the practice of nanotechnology for the detection, prevention, and therapy of diseases, for example infectious diseases [3]. One of the most commonly used nanomaterials in nanomedicine is nanoparticles (NPs), which show highly tunable physical and optical properties and the ability to produce a wide library of compounds [3]. A variety of organic, inorganic, and hybrid NPs have been synthesized using various approaches in the past few decades. Nanomaterials comprised of different organic and/or inorganic constitutes are defined as hybrid nanomaterials. The most notable example is organic-inorganic hybrid NPs, which are commonly prepared by absorbing or grafting polymers onto inorganic NPs. For example, several studies have demonstrated that hybrid NPs based on silver (Ag) NPs are promising in the therapy of antimicrobial-resistant bacterial biofilm-related infectious diseases [4,5].
Nanomaterial-based therapies represent promising tools to fight against bacterial infections that are difficult to treat, for example, those characterized by biofilm formation or antimicrobial resistance [6]. In this review, we introduce the different types of nanomaterials and highlight their current applications as treatments for bacterial infections characterized by possible antibiotic resistance and biofilms. In this review article, we emphasize the importance of nanomaterials in combating bacterial infections in different body systems.
2. The Development of Organic, Inorganic and Hybrid NPs for Infectious Diseases
Bacteria-targeting nanomaterials have been widely used to treat bacterial, fungal, viral and parasitic infectious diseases in recent years [7,8,9,10,11,12,13,14], and their applications include direct bactericidal effects [15,16,17,18], encapsulation of probiotics [19], and encapsulation of bactericidal agents [20].
Inorganic nanomaterials with direct bactericidal effects include calcium silicate (CaSi) [15,21], silver (Ag) [22,23], copper (Cu) [24,25], and gold (Au) NPs [17,18]. Among the inorganic nanomaterials, Ag NPs have been one of the most widely used antimicrobial materials due to their broad-spectrum antibacterial capacity and low propensity to engender drug resistance [18]. Ag NPs had a good antimicrobial effect in both gram-positive and gram-negative bacteria, for example methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus (VRE), Pseudomonas aeruginosa, and Klebsiella pneumoniae [18,22,23]. Au NPs have been shown to decrease the effect of lipopolysaccharide (LPS), an important toxin in gram-negative bacteria that causes fulminant immune responses through its effect on dendritic cells and immune reactions [26]. Transition metal oxides and chalcogenide (TMO&C) nanomaterials have been characterized as having excellent physiochemical, electronic, and optical properties. They possess a functional architectural assembly, proving their benefits in combating bacterial infections [27]. Similarly, magnetic iron oxide NPs (MIONPs) exhibit improved adherence to Staphylococcus and extracellular polymeric substances, thus increasing bacterial biofilm eradication through the encapsulation of antimicrobial agents [28]. In summary, inorganic nanomaterials have been widely used to treat various bacterial infectious diseases, according to the characteristics of inorganic materials. Notably, the intrinsic toxicity of some inorganic materials (i.e., heavy metals) and their ability to accumulate and persist in the human body are of great concern when used in clinical patients [29]. Successful clinical application of these NPs primarily depends on their stability, circulation time, access and bioavailability at disease sites, and safety profile [29].
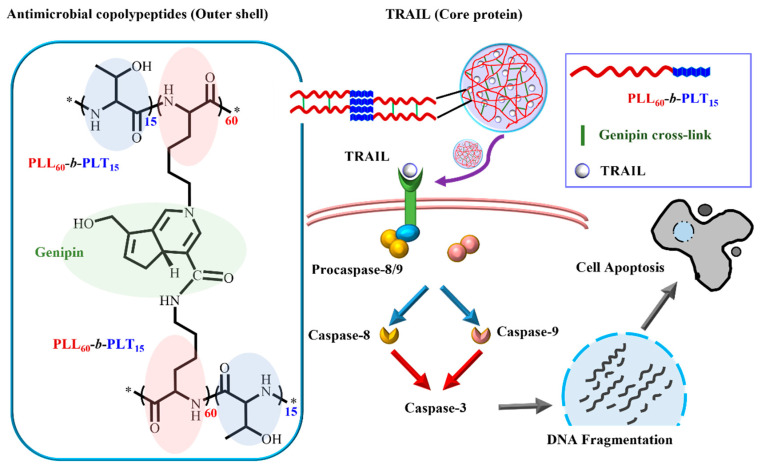
Organic nanomaterials have also been developed for the treatment of bacterial infectious diseases in the past few decades. Among them, polymeric NPs have become an important class of nanomaterials and have recently attracted intense interest. Polymeric NPs such as micelles and vesicles are formed by the self-assembly of amphiphilic copolymers in aqueous solutions through various physical interactions, such as hydrogen bonding and electrostatic or hydrophobic interactions [30,31,32]. Chemical cross-linking of polymers or in situ polymerization of monomers in nanoconfined environments also promotes the formation of NPs, which are typically named nanogels [33,34,35]. For example, antimicrobial agents have been encapsulated in cross-linked nanogels, achieving better antibacterial activity with less cytotoxicity [36]. Tumor necrosis factor (TNF)-related apoptosis-inducing ligand (TRAIL), which triggers an extrinsic apoptotic pathway via death receptors, was encapsulated in a bactericidal polypeptide-crosslinked nanogel that suppressed K. pneumoniae and overactive macrophages. Interestingly, nanogel and TRAIL nanogel treatments were more toxic toward LPS-activated cells than toward naïve cells (Figure 1) [36]. The highly selective character of antimicrobial agent-encapsulated cross-linked nanogels is beneficial for increasing the local concentration of the antimicrobial agent without damaging normal tissue.
Figure 1.
Schematic illustration of TRAIL encapsulated in bactericidal polypeptide nanogels, adapted from [36], published by Elsevier, 2019.
An important category is hybrid nanomaterials, and many of them have been applied for treatments of infectious disease [4,5,37,38,39,40,41,42,43,44]. There are some examples of hybrid nanomaterials:
-
(a)
Ag-based hybrid NPs: Intracellular protein with gentamycin attached was analyzed to create the biocompatible Ag NPs [5]. The created nanohybrid had been shown to be effective during gram-positive, gram-negative and drug-resistant bacteria infection [5]. A hybrid antimicrobial particle with improved antimicrobial efficacy and good aqueous dissolubility relying on Ag NPs and curcumin had been made with oxidized amylose as an environmentally-friendly NP [4]. The hybrid antimicrobial agent revealed good dissolubility in aqueous solution, improved antimicrobial efficacy, excellent antioxidant effect and better cell compatibility [4]. A hybrid hydrogel of organic collagen capped on an inorganic Ag NP and melatonin had been used to promote tissue regeneration of wounds with skin defects during multi-drug resistant bacterial infection [37]. Assemblies of organic thiolated hyaluronic acid with Ag NP hydrogels were effective against S. aureus infection in chronic wounds [38]. The hybrid NP with coating Ag on organic fluorhydroxyapatite had been shown to enhance its antimicrobial ability when applied in skeletal system implants [39].
-
(b)
Anti-bacterial agent-based hybrid: An anti-bacterial peptide attached with biomimetic phage-platelet hybrid NP was created for extended circulation in blood and enhanced anti-microbial efficacy [40]. Poly lactic-co-glycolic acid (PLGA)-based NP distribution systems were analyzed to treat the bacterial biofilm-related infections. Azithromycin (a macrolide antibiotic)-loaded carbon quantum dots (CQDs)–PLGA hybrid NPs showed chemo-photothermally synergistic anti-biofilm effects against P. aeruginosa biofilms [41]. Au NP combined with β-lactam antibiotic had been shown to be effective against MRSA infection [42].
-
(c)
Theranostics hybrid nanomaterial: The hybrid of inorganic material (Mn3O4 NPs) with organic citrate (C-Mn3O4) and folic acid (FA-Mn3O4) ligands were bactericidal against S. hominis by causing membrane rupture [43]. The multielement physical characters of these organic–inorganic hybrid materials revealed their further application both in diagnosis and therapy (theranostics) [43].
-
(d)
Bifunctional hybrid nano-flowers: A bifunctional hybrid nano-flower composed of an organic part (the rabbit polyclonal antibody of H. pylori) and inorganic part (the Cu NP), prepared with an enzyme-linked immunosorbent assay, was invented for the detection of H. pylori infection [44].
Overall the hybrid nanomaterials showed enhanced antibacterial activity with less adverse drug reactions compared to conventional treatment.
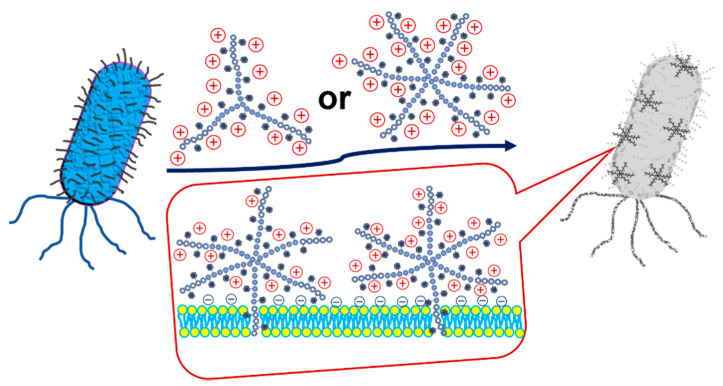
In recent years, nanostructured polymers have also been broadly applied as treatments for infectious disease [45,46,47,48,49]. The preparation of polymeric nanomaterials with nanoscale architectures, such as star-shaped polymers, can be achieved by exploiting polymer topology. Among these materials, star-shaped polymers provide distinct advantages over their linear counterparts. Star-shaped polymers are typically synthesized using dendrimers as initiators. The star-shaped topology effectively decreases the tendency of the cationic polymers to interact with other cells, reducing cytotoxicity when the arms are “locked” in a nanoparticle form [45,46,47,48,49]. The so-called ‘structurally nanoengineered antimicrobial polymers’ (SNAPs) are different from existing self-assembled antimicrobial macromolecules, which dissociate into monomers below their critical micelle concentration [39]. Since 2016, SNAPs have shown great potential as low-cost, effective antimicrobial agents that may represent a tool for combating multiple-drug resistant (MDR) pathogens, such as MDR Acinetobacter baumannii [47]. Additionally, the star-shaped structure has been further modified and optimized for the subsequent development of antimicrobial agents used in clinical applications [45].
A new SNAP (14–26 nm in diameter) that contains mixtures of poly(L-lysine) (PLL) and glycopolymer arms was reported to have antimicrobial activity toward gram-positive bacteria, including MRSA and VRE, while being nonhemolytic and showing excellent mammalian cell biocompatibility [45]. Star-shaped polymers based on PLL modified with an indole group (PLL-g-indo) were revealed to have the best antibacterial activity against enterohaemorrhagic E. coli (EHEC), improving cytotoxic selectivity toward pathogens over mammalian cells without hemolytic activities (Figure 2) [48]. A hybrid SNAP synthesized by atom transfer radical polymerization (ATRP) and composed of poly[2-(dimethylamino)ethyl methacrylate] (PDMA) as the arms and polyhedral oligomeric silsesquioxane (POSS) as the core (POSS-g-PDMA) exerted antibacterial effects against E. coli infection in zebrafish embryos while maintaining low cytotoxicity and was used in aquaculture applications [46]. A polymer synthesized by ring-opening polymerization and composed of copolymer poly(L-lactide)-block-poly(phenylalanine-stat-lysine) [PLLA31-b-poly(Phe24-stat-Lys36)] chains self-assembles to form micelles in aqueous solution and has antibacterial properties against both gram-positive and gram-negative bacteria without evident antibiotic resistance while being biodegradable [49]. SNAPs are predicted to be more widely used in bacterial infections in the future to increase the efficacy and decrease adverse events compared to traditional NPs.
Figure 2.
Schematic illustration of the antibacterial mechanism of star-shaped graft copolypeptides, adapted from [36], published by Elsevier, 2019.
3. The Wide Application of Nanotechnology in Bacterial Infectious Diseases
3.1. Diagnosis/Increased Accuracy and Efficacy of Diagnostic Tests
In addition to serving as treatments, nanotechnology has improved the accuracy of diagnosing bacterial infectious diseases [44,45,46,47]. The rapid development of nanoscience and nanotechnology has been used to design nanomaterial-assisted biosensors with better detection performance for bacterial infections, as shown in the examples listed below [50,51].
3.1.1. Graphene-Based Biosensors
The unique 2D structure and single-atom thickness of graphene sheets enable graphene-based biosensors to be highly flexible to any tiny changes in the surrounding environmental conditions [51]. Graphene-based biosensors have been widely used in infectious disease, especially in coronavirus (COVID-19) infection [52,53]. For example, an novel aptameric dual channel graphene-TWEEN 80 field effect transistor (DGTFET) biosensing machine with on-site analyzing of interferon (IFN)-γ, TNF-α, and interleukin (IL)-6 works as quickly as 7 min with limits of detection (LODs) and as low as 476–611 × 10−15 m in bio-fluids [52].
For bacterial infection, surfaces of graphene products, graphite, graphene, graphene oxide and reduced graphene oxide have been revealed to be a good platform for antibacterial effects and improved biocompatibility in tissue engineering, biosensors or antimicrobial agent-carriage [54]. A graphene (SLG)/Au NP-based biosensor has been used in the diagnosis of S. aureus gene sequences, with detection limits as low as 12.4 pg/mL [55]. A biosensor using reduced graphene oxide-carbon nanocomposites prepared using the hydrothermal method has been used for precise and fast label-free electrochemical detection of pathogenic bacteria, such as S. enterica, in a wide linear dynamic range from 101 to 108 cfu mL−1 and with a 101 cfu mL−1 limit of detection [56]. A fluorogenic DNAzyme probe was utilized in diagnosis of E. coli infection and could detect with a detection limit of 1000 CFU [57]. A label-free electrical biosensor coupled with graphene exhibits accurate diagnosis of pathogenic E.coli O157:H7 with the sensors transduced through electric charges [58]. Thermally decreased graphene oxide-based field-effect transistor (rGO FET) coupled with an ultrathin layer of Al2O3 shows on-time diagnosis of E. coli bacteria [59]. The sensor has the ability to detect a single E. coli cell within 50 s in a 1 µL sample volume [59]. A quick detection device for Vibrio parahaemolyticus was developed by merging loop-mediated isothermal amplification (LAMP) and one-use electrochemical sensors with screen-printed graphene electrodes (SPGEs) [60]. The LAMP responses using primers targeting V. parahaemolyticus toxR gene could diagnose V. parahaemolyticus within 45 min with the detection limit of 0.3 CFU per 25 g of uncooked seafood [60]. A novel self-propelled micromotor-based immunoassay (MIm) has been used for C-reactive protein (CRP) detection of blood of preterm infants suspected to have sepsis [61]. Graphene sensors could be merged with various signal transduction technologies to improve their applications in the future.
3.1.2. NP-Based Lateral Flow Biosensor
An assay that combines LAMP with an NP-based lateral flow biosensor (LFB), named LAMP-LFB, enables the simple, reliable, and objective detection of Mycoplasma pneumoniae [62]. The development of NP-enhanced antibodies and DNA sensors for detecting Salmonella using a microfluidic-based electrochemical system provides a rapid detection method with high sensitivity and specificity [63]. Multiple cross displacement amplification (MCDA) combined with Au NP-based lateral flow biosensors (LFBs) provides a simple, fast and sensitive method for the accurate identification of P. aeruginosa, among other pathogens [64]. Au/Ag hybrid NPs composed of gold nanorods (AuNRs) coated with Ag serve as excellent theranostic agents in photoacoustic (PA) imaging, as well as the treatment of bacterial infections [65]. The multiple cross displacement amplification (MCDA) and NP-based LFB (MCDA-LFB) were combined to detect H. influenzae, which has been proven to be reliable, rapid, and not complicated [66]. An electrochemiluminescent lateral flow immunosensor (ECL-LFI), Ru(bpy)32+-labeled AuNPs, has been used to detect CRP [67]. Another multiplex loop-mediated isothermal amplification coupled with a NP-based lateral flow biosensor (m-LAMP-LFB) has been used in detection of all S. aureus species [68]. The LAMP-LFB also provides a reliable and rapid assay for E. faecalis, Neisseria meningitidis and M. pneumoniae detection, which is significant for diagnosis and follow-up treatment [62,69,70].
3.1.3. Sensitive Label-Free Immunosensing
An H. pylori immunosensor based on a platinum NPs/poly(3,4-ethylenedioxythiophene)/reduced graphene oxide (Ptnano/PEDOT/red-GOx)-modified gold electrode (Au-ET) was fabricated in a stepwise manner for the detection of cytotoxin-associated gene A antibody [71]. This novel immunosensor exhibited a good accuracy, precision and reliability, and the sensor has an excellent linear range of 0.1 ng/mL to 30 ng/mL by limiting the detection range to 0.1 ng/mL [71]. A label-free electrochemical immunosensor was developed for quick detection of H pylori after covalent conjugation of the antibody (CagA) on the nanomaterials altered the Au electrode [72]. An Au disc electrode coupled with anti-P. syringae pv. lachrymans antibodies and conjugated with bovine plasma albumin was developed and sensor development was characterized by cyclic voltammetry (CV) and antigen detection by electrochemical impedance spectroscopy (EIS) measurements [73]. A new paper-based sensing platform has been applied in a label-free potentiometric immunosensor for Salmonella typhimurium detection based on the blocking surface principle [74]. A photonic interferometer biosensor based on a bimodal waveguide (BiMW) was developed for the rapid and label-free detection of bacteria, such as E.coli directly in ascitic fluid [75].
3.1.4. Nanosieving Microfluidic System
An electropolymerized self-assembled layer of Au NPs was fabricated in a portable nanosieving microfluidic system (NS-MFS). Redox-active gold NPs (raGNPs) enhanced the electrical conductivity and provided the detection limit of the device, reaching 10 CFU/mL for P. aeruginosa and S. aureus spiked in plasma [76]. A microwave-microfluidic biosensor has been developed for quick, contactless and non-invasive detection of the concentration and growth of E. coli [77].
3.1.5. Au NPs
Au NPs have been widely used as biosensors for bacteria detection. Thiolation of the chimeric phages directed on various bacterial pathogens caused aggregation of AuNPs, resulting in a visible colorimetric reaction in front of at least about 100 cells of the target bacteria [78]. The photoelectronic Au nanostructure biosensor could detect citrullinated histone H3, an important biomarker during neutrophil cell death in bacterial infection [79]. The Au NP-based biosensor has been used to detect the colistin (an important antimicrobial agent for gram negative bacteria) resistance gene mcr-1 [80].
3.1.6. Carbon Nanotube Biosensor
This is an electrochemical biosensor that is constructed from single walled carbon nanotubes that could detect the change in intracellular hydrogen peroxide during contact with lipopolysaccharides, an important component of gram-negative bacteria [81].
These materials are some examples of using nanotechnology to diagnose bacterial infections. With the increasing application of nanotechnology in the detection of bacteria, the accuracy and efficacy of diagnostic tests in bacterial infections will increase substantially.
3.2. Vaccination/Vectors or Adjuvants to Deliver Antigenic Proteins
NPs can be used as vaccine vectors to deliver antigenic proteins that induce humoral and cellular immune responses [82,83]. NPs carrying outer membrane vesicles (OMVs) from carbapenem-resistant K. pneumoniae (CRKP) are believed to be potential vaccines for intractable CRKP infection [82]. NPs containing polysorbitol transporter (PST) and pneumococcal surface protein A (PspA) induce protective immunity against Streptococcus pneumoniae, with long-term reaction mediated by T helper (Th)2 or T follicular helper (Tfh) cells [83]. T and B cell immune responses are mediated by antigen-presenting cells through the activation of a peroxisome proliferator-activated receptor gamma (PPAR-c) pathway [83]. These immune reactions suggest that NPs might serve as a mucosal adjuvant in a subunit vaccine [83]. Similarly, NPs coated with membranes of extracellular vesicles secreted by S. aureus (i.e., NP@EV) may serve as an active-targeting antibiotic carrier [84]. NPs coated with a PEGylated lipid bilayer (i.e., NP@Lipo; PEG = poly(ethylene glycol)) are internalized by S. aureus-infected macrophages with higher efficiency and accumulate within major organs (such as kidneys, lungs, spleen, and heart), suggesting that they are beneficial for treating metastatic S. aureus infections [84]. NPs have been shown to be excellent vaccine vectors or adjuvants to deliver antigenic proteins that induce host immunity against bacterial infections.
4. Current Applications of Nanotechnology as Treatments for Bacterial Infections in Regard to Possible Antibiotic Resistance and Biofilms
4.1. Medical Device/Efficacy against Biofilms
Biofilm formation is a key step in the bacterial infection of implants, catheters and naive tissues [85]. Many bacteria, such as S. aureus, E. coli or P. aeruginosa, have the capacity to self-secrete a matrix of extracellular polymeric substances (EPSs) that encapsulate bacterial societies. This matrix then progresses to form an advanced 3D structure called a biofilm that functions as a protective barrier against host immunity, environmental pressure, and the inner diffusion of antimicrobial agents [85,86,87,88,89].
4.1.1. Application of Nanotechnology in Biofilms in Biomaterial Implants
A common application of nanotechnology is biomaterial implants. These devices generally present a 5% failure rate, with most cases attributed to the bacterial colonization of biomaterial surfaces, formation of biofilms and spread into adjacent tissues [85,86,90,91,92,93]. S. aureus, for example, recognizes host extracellular matrix (ECM) molecules and initiates colonization of the epithelium through microbial surface recognizing adhesive matrix molecules (MSCRAMMs), such as fibronectin binding protein A (FnBPA) [92]. In contrast, NPs interfere with the function and binding property of FnBPA, enabling their use in a wide variety of biomedical applications [92]. Similarly, 316L stainless steel coated with a polyelectrolyte multilayer and a mesoporous bioactive glass exhibited controlled antibiotic (tetracycline) release and biocompatibility, showing great potential in endowing orthopedic implants with antibacterial and osteoconductive properties [93]. Silver/copper (Ag/Cu)-coated catheters were investigated as tools to prevent MRSA infection and biofilm formation [89].
4.1.2. Improve Biofilm Penetration with Nanotechnology
Bacteria-sensitive NPs prepared from poly(lactic-coglycolic acid) and poly(Ɛ-caprolactone), which were decorated with chitosan and combined with dissolved microneedles of doxycycline, could serve as antimicrobial agents with improved biofilm penetration specifically in the delivery of doxycycline to an infection site [94]. Zeolite-based imidazole framework (ZIF-8)-covered mesoporous polydopamine (MPDA) NPs were combined with Pifithrin-μ (PES), a natural inhibitor of heat-shock protein (HSP) that is important in bacterial related destruction and inflammation [95]. The ZIF-8 shell of the MPDA@ZIF-8/PES nanoplatform could quickly degrade under the acidic microbial environment, which activated the release of PES and Zn ions [95]. In general, this penetration of biofilms to target enclosed bacteria has been an important therapeutic aim when developing NPs for bacterial infectious diseases in different organs or systems [16,85,86,87,88,89,94].
4.1.3. Nanotechnology-Based Therapy in Destroying Biofilm Formation
NPs have been used as one of the therapeutic strategies for nosocomial bacterial infections [96]. A nanoscale surface coating containing silicon NPs forms a hydrophobic surface that prevented the growth of bacteria with a significant reduction in the frequency of isolation of Acinetobacter spp. on coated surfaces [96]. A synthetic iron NP (FeOOH-NP) was shown to be an active dose-dependent agent that inhibited the formation of P. aeruginosa biofilms, modulating bacterial motility and other important virulence factors related to biofilm formation [86]. Another sulfur-functionalized fullerene NP (SFF NP) prepared by chemical vapor deposition effectively inhibits biofilm formation, eradicating preformed biofilms of P. aeruginosa and disrupting the expression of the exotoxin A (toxA) gene [87]. Similarly, zinc oxide NPs (ZnO-NPs) have been shown to inhibit biofilm formation and flu gene expression in uropathogenic E. coli (UPEC) strains [88].
4.1.4. Biofilm Monitor with Nanotechnology
Biofilm formation is critical in antimicrobial agent-resistance and treatment failure; however, so far there is no detection method to monitor the formation of biofilms. Anti-S. aureus antibodies were coupled with Au NPs to develop an immunochromatographic strip (ICS) [97]. The invented ICS could detect 102 CFU/mL S. aureus in 15 min among patients with neonatal sepsis [97]. NP-based biosensors have been used to detect and prevent the formation of biofilms by disturbing the adhesion of bacteria to the surface of food [98]. The developed prototype ICS could detect as low as 5 µg purified polypeptide and 102 CFU/mL S. aureus within 15 min. An optoelectronic machine by the dual array of interdigitated micro- and nanoelectrodes in parallel could detect the bacterial biofilm development by the optical and electrical monitor methods [99].
4.1.5. Support of Tissue Regeneration in Biofilms
Tissue regeneration is often retarded when biofilms are formed during bacterial infections. Injectable calcium phosphate bone cement-chitosan paste containing graphene oxide has been used for endodontic therapy, with antimicrobial effect against E. faecalis biofilms, together with the support for the dental pulp stem cells [100]. The fast coverage of the nano-textured titanium by human mesenchymal stem cells together with stimulation of bony differentiation could decrease the chance of biofilm formation during implant integration with the human bone tissue [101]. Ag NPs coupled with polyetheretherketone have been developed for osteogenesis and angiogenesis in addition to the antibacterial ability [102]. Copper-incorporating mesoporous bioactive glass NPs have been invented for bone regeneration as well as antimicrobial ability [103].
Biofilm formation is a troublesome problem when treating bacterial infections, and NPs have been shown to be effective at inhibiting biofilm formation and eradicating preformed biofilms.
4.2. Application of Nanotechnology in Detecting Possible Antibiotic Resistance in Bacterial Infectious Diseases
As mentioned above the m-LAMP-LFB was applied not only to detect all S. aureus strains, but also to detect methicillin-resistant genes from all S. aureus species [68]. Multiplex MCDA (m-MCDA), which aims to detect the nuc gene (S. aureus-specific gene) and mecA gene (encoding penicillin-binding protein-2′), has been shown to detect S. aureus strains and MRSA within 85 min [68]. Fast dissemination of the colistin resistance (mcr) gene mcr-1 in Enterobacteriaceae is very important in combating bacterial infections [80]. MCDA merged with the sensing of agents by Au NP-based LFB assay was identified to detect the mcr-1 gene with simplicity, rapidity, specificity, and sensitivity [80]. Coupled with the sensitivity of magnetoresistive (MR) sensors, the possibility of a lab-on-chip device, and the specificity of phage receptor binding proteins (RBPs) was constructed as probes for the quick diagnosis of Enterococcus and Staphylococcus with possible antimicrobial agent resistance [104]. The multicomponent nucleic acid enzyme-Au NP (MNAzyme-GNP) device was used in patients with central line related bloodstream infectious disease and revealed a sensitivity and specificity as high as 86% and 100%, respectively [105]. MRSA was detected in patient swabs with 90% clinical sensitivity and 95% clinical specificity [105]. Detecting mecA resistance genes in uncultured nasal, groin, axilla, and wound swabs from patients was done with 90% clinical sensitivity and 95% clinical specificity [105]. Rapid detection of bacteria with multiple drug resistance had been achieved by combining electrochemical immunoassays (EC-IA) for pathogen identification with confirmatory quantitative mass spectral immunoassays (MS-IA) based on signal ion emission reactive release amplification (SIERRA) nanoparticles with unique mass labels [88]. The nanotechnology-based detection method has been shown to provide a more efficient and more accurate way of detecting multi-drug resistant bacteria.
4.3. Advances in Nanotechnology in Treating Bacterial Infectious Diseases with Possible Antibiotic Resistance
Nanotechnology-based therapeutic methods have revealed an effective therapeutic result against multi-drug resistant bacterial infection. There are some examples of advances in nanotechnology in treating bacterial infectious diseases with possible antibiotic resistance.
4.3.1. Silver (Ag) NPs
Ag NPs have shown synergic action with colistin, and imipenem against pandrug-resistant A. baumannii isolated from patients [106]. The efficacy of combined neomycin, an antibiotic with Ag-NPs in bacteria-infected mice with burn wounds revealed good healing ability with excellent wound contraction when the neomycin-Ag-NPs were used in the spray method [107]. Ag NPs@organic frameworks/graphene oxide in sericin/chitosan/polyvinyl alcohol hydrogel were constructed for the redox ability of tyrosine residues in silk sericin without extra chemicals [108]. The composite material was noted to be an ideal dressing for accelerating hemostasis, preventing multi-drug resistant bacterial infection and promoting wound healing [108]. Based on the good antibacterial ability of Ag NPs combating numerous bacterial strains compared with other antimicrobial agents, the IPM@AgNPs-PEG-NOTA nanocomposite (Ag NPs covered with SH-PEG-NOTA and loaded by imipenem) was developed [109]. The IPM@AgNPs-PEG-NOTA is a pH-sensitive nanodrug device with good efficacy in reducing resistance and is synergistically active against carbapenem-resistant A. baumannii [109].
4.3.2. Nanotechnology Based Phage Therapy
Bacteriophages, or “phages” can attack bacteria, takeover their intracellular mechanism to let themselves survive, and thus kill the bacteria. Phage therapy has been proposed as good weapon in combating bacteria with multi-antimicrobial resistance [110]. However, how to prepare and deliver this “genetic material” into a human body is a major problem so far [110]. With the nanotechnology-based method, the phage might be delivered after remodeling with lipid based nano-carriers, microfluidic-based methods, nano-emulsification, or phage loaded nanofibers [111]. The advances could improve the stability of phage therapy, enhancing host retention times, and facilitate the biofilm penetration [111].
4.3.3. Development of Nano-Cargos to Deliver Antimicrobial Agents
Surface engineering of nano-cargos has the advantages of aiming and controlling the antimicrobial-resistance mechanisms, and thus possibly overwhelms the bacterial resistance [112]. The incorporation of ivacaftor-colistin nanosuspensions (Iva-Col-NPs) was effective against the multi-drug resistance of P. aeruginosa, an overwhelming cause of terminal and persistent lung infections in cystic fibrosis patients [113]. Chitosan propolis nanocomposite coupled with apramycin was used for therapy for multiantimicrobial agent-resistant S. Typhimurium [114]. Nano-encapsulated daptomycin has been used in therapy for an experimental MRSA bone and joint infection model [115].
4.3.4. Nanotechnology-Based New Antimicrobial Agent Delivery
Some new antimicrobial agents might be effective in combating antimicrobial agent resistant bacteria, but the clinical use might be limited because of the hydrophobicity or low oral bioavailability. A nanotechnology-based delivery method might provide a practicable way for therapy with these new antimicrobial agents. Platensimycin (PTM), a natural agent, could target bacterial fatty acid synthases and treat infections by MRSA [116]. To enable the usage of PTM in local MRSA infections, polyacrylamide hydrogels containing polyamidoamine was constructed for the organized release of PTM [116]. Luteolin-loaded methoxy poly-poly micelles could overcome the hydrophobicity and low oral bioavailability characters of luteolin. The luteolin-loaded NP has been shown to be effective in treating pulmonary infection with multi-antimicrobial agent resistant K. pneumoniae [117].
4.3.5. Chitosan-Based Nanomaterial
Chitosan, itself an antimicrobial polysaccharide, is produced to be readily soluble in neutral aqueous solution during systemic therapy [118]. After modification, the acid-transforming chitosan (ATC) become an insoluble material in a slight acidic intracellular environment [118]. ATC, coupled with fragment DNA could form nano-sized spherical polyplexes and combat the S. typhimurium in macrophages [118]. Chitosan-Au loaded with embelin has shown synergistic activity with ciprofloxacin, an antimicrobial agent against multidrug-resistant P. aeruginosa and E. coli, by inhibiting the efflux pumps [119].
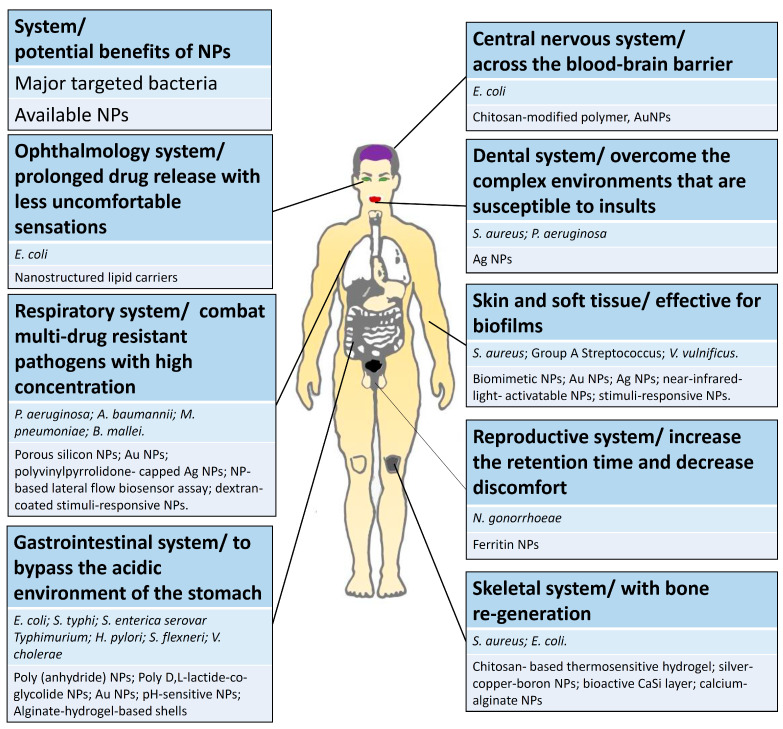
5. Clinical Applications of NPs for Bacterial Infections in Various Systems
Recent advances in the development of NPs to treat bacterial infectious diseases are summarized in Table 1. NPs have been used for the treatment of bacterial infections in clinical situations, with applications across different systems, as described below (Figure 3).
Table 1.
Advances in the development of NPs to treat bacterial infectious diseases.
| System/Disease | Targeted Pathogens | Material | Authors | Publish Year | Major Findings | References |
|---|---|---|---|---|---|---|
| Central nervous system | ||||||
| Neonatal meningitis |
Escherichia coli K1 |
Chitosan-modified poly (lactic-coglycolic acid) (PLGA) | Zhang J, et al. | 2021 | PLGA NPs as vector for the recombinant protein OmpAVac (Vo), which induced protective immunity against E. coli K1 infections. | [120] |
| Respiratory system | ||||||
| Glanders | Burkholderia mallei | Gold NPs (AuNPs) | Gregory AE, et al. | 2015 | AuNPs functionalized with a glycoconjugate vaccines bind to LPS and protect against lethal inhalation of B. mallei. | [121] |
| Pneumonia | Acinetobacter baumannii | Polyvinylpyrrolidone (PVP)-capped Ag NPs | Tiwari V, et al. | 2017 | PVP-capped Ag NPs has shown antibacterial activity against a carbapenem-resistant strain of A. baumannii. | [122] |
| Pneumonia | Pseudomonas aeruginosa | Porous silicon NPs (pSiNPs) | Kwon EJ, et al. | 2017 | Selected antibacterial peptides loaded into pSiNPs exerted therapeutic effects on P. aeruginosa infections in the lung. | [123] |
| Pneumonia | Mycoplasma pneumoniae | Nanoparticle-based lateral flow biosensor (LFB) assay | Wang Y, et al. | 2019 | Loop-mediated isothermal amplification (LAMP) combined with LFBs for the rapid and accurate detection of Mycoplasma pneumoniae | [62] |
| Pneumonia | Drug-resistant P. aeruginosa | Dextran-coated stimuli-responsive nanoparticle | Ye M, et al. | 2021 | Encapsulation the hydrophobic antibiotic rifampicin, and strong affinity for P. aeruginosa, activated by either low pH or high reactive oxygen species levels. | [16] |
| Gastrointestinal system | ||||||
| Intestinal infection |
Shigella flexneri (Shigellosis) | Poly (anhydride) NPs | Camacho AI, et al. | 2013 | Outer membrane vesicles encapsulated poly NPs for mucosal protection against S. flexneri. | [124] |
| Intestinal infection |
Salmonella enterica serovar Typhimurium | Poly D,L-lactide-coglycolide (PLGA) nanoparticle | Lee JA, et al. | 2014 | PLGA nanoparticle-encapsulated honeybee (Apis mellifera) venom promoted the clearance of S. enterica serovar Typhimurium infection. | [125] |
| Intestinal infection |
S. typhi | Gold nanoparticle | Chowdhury R, et al. | 2017 | A designed antimicrobial peptide, VG16KRKP, delivered via gold nanoparticles exhibited strong bacteriolytic activity against intracellular S. typhi. | [126] |
| Stomach | Helicobacter pylori | Targeting the UreI channel protein, pH-sensitive NPs | Luo M, et al. | 2018 | Amoxicillin-loaded NPs protected antimicrobial drugs from an acidic environment, delivering them safely to eradicate H. pylori at the infected site. | [127] |
| Intestinal infection |
Vibrio cholerae | Host receptor for cholera toxin was coated onto polymeric NPs | Das S, et al. | 2018 | The key host receptor for V. cholerae toxin, monosialotetrahexosylganglioside (GM1), was coated onto the surface of polymeric NPs and functioned as toxin decoys, neutralizing its actions. | [128] |
| Intestinal infection |
E. coli | Alginate-hydrogel-based shells | Yuan L, et al. | 2021 | Bifidobacterium breve, when encapsulated within alginate-hydrogel-based shells yielded protection against gastric acid and antimicrobial agents, subsequently killing E. coli adhering to the intestinal epithelium. | [19] |
| Skeletal system | ||||||
| Osteomyelitis | Staphylococcus aureus | Chitosan (CS)-based thermosensitive hydrogel | Tao J, et al. | 2020 | NPs containing vancomycin, an antimicrobial agent, combined with CS gel were effective against osteomyelitis caused by S. aureus. | [129] |
| Bone infection | S. aureus | Silver-copper-boron (ACB) | Abdulrehman T, et al. | 2020 | ACB NPs for bone infections that targeted intracellular S. aureus in bone cells. | [130] |
| Bone infection | E. coli and S. aureus | Bioactive CaSi layer on titanium substrate | Buga C, et al. | 2021 | A nanostructured CaSi layer exerts an antibacterial effect on E. coli and S. aureus species with osteogenic properties when coated on bone tissue | [15] |
| Osteomyelitis | Methicillin- resistant S. aureus (MRSA) | Calcium-alginate nanoparticle (Ca-Alg) |
Gowri M, et al. | 2021 | Ca-Alg crosslinked phosphorylated polyallylamine (PPAA) encapsulating clindamycin, an antimicrobial agent, was effective against MRSA osteomyelitis. | [131] |
| Skin and soft tissue | ||||||
| Skin infection | MRSA | Biomimetic nanoparticle |
Wang F, et al. | 2016 | Nanoparticle-based detained staphylococcal α-hemolysin vaccine protected against MRSA skin infection and further decreased dissemination. | [132] |
| Necrotizing fasciitis | Group A Streptococcus (GAS) | Red blood cell derived biomimetic NPs (“nanosponges”) | Escajadillo T, et al. | 2017 | Nanosponges sequester pore-forming streptolysin O (SLO) and block the injury to host cells by GAS, preserving innate immune function and increasing bacterial clearance. | [133] |
| Wound infection |
Vibrio vulnificus | Gold nanoparticle-DNA aptamer (AuNP-Apt) | Lee B, et al. | 2017 | An antimicrobial peptide (AMP), HPA3Phis, loaded onto AuNP-Apt reduced the intracellular V. vulnificus load by 90% and increased the viability of the infected cells. | [134] |
| Wound infection |
MRSA | Biocompatible enzyme-responsive Ag nanoparticle assemblies (ANAs) | Zuo YM, et al. | 2020 | High-efficiency ANAs were an antimicrobial treatment for MRSA when applied as a wound dressing, accelerating healing. | [17] |
| Wound infection |
S. aureus | Near-infrared-light (NIR)-activatable–carbon monoxide (CO) | Yuan Z, et al. | 2021 | Delivery of bactericidal CO gas that penetrated the impaired biofilms to achieve effective S. aureus biofilm elimination. | [20] |
| Soft tissue infection |
S. aureus | The dextran-coated stimuli-responsive nanoparticle | Ye M, et al. | 2021 | Encapsulation of the hydrophobic antibiotic rifampicin, and then accumulating in infected soft tissues to eradicate drug-resistant S. aureus infection. | [16] |
| Ophthalmology | ||||||
| Ocular wound | E. coli | Nanostructured lipid carriers mixed with a collagen/gelatin/alginate (CGA) biodegradable material | Chang MC, et al. | 2020 | Moxifloxacin (a type of antibiotic) and dexamethasone (a type of steroid)-loaded nanostructured lipid carriers for anti-inflammatory ocular disease treatment. | [135] |
| Dental tissues | S. aureus and P. aeruginosa | Silver-nanoparticle-coated collagen membrane | Chen P, et al. | 2018 | Antibacterial effects against S. aureus and P. aeruginosa, effective anti-inflammatory properties, and induction of the osteogenic differentiation of mesenchymal stem cells. | [136] |
| Reproductive system |
Neisseria
gonorrhoeae |
Ferritin nanoparticle |
Wang L, et al. | 2017 | Ferritin nanoparticles were utilized as a vector for MtrE, the outer membrane channel of gonococcal MtrCDE active efflux pump, in gonorrhea vaccines. |
[137] |
Figure 3.
Advances in the use of NPs to treat bacterial infectious diseases in different body systems.
5.1. Central Nervous System: Transport of Antimicrobial Agents across the Blood–Brain Barrier (BBB)
When treating patients with central nervous system (CNS) infection, the most important challenge for drug delivery is the ability of antimicrobial agents to penetrate the BBB [138]. The BBB is highly selective and serves as the interface between the central nervous system and circulation, which is critical for maintaining brain homeostasis [138]. Since most of the pathogens infect the CNS through phagocytosis or receptor (e.g., EphA2)-mediated transcytosis, the majority of the NPs cross the BBB via receptor-mediated transcytosis (e.g., antibody, peptide, or protein) [139]. In neonatal meningitis caused by E. coli K1 infection, a chitosan-modified poly(lactic-coglycolic acid) (PLGA) nanoparticle was used as a vector for the recombinant protein OmpAVac (Vo), inducing protective immunity against E. coli K1 infection [120]. Transcranial picosecond laser stimulation of Au NPs after an intravenous injection has been noted to increase BBB permeability, which might be specifically graded by laser intensity and entirely reversible, without inducing significant structural damage [138]. For the treatment of CNS infections, drug-loaded NPs are safe and could be up to ten-fold more efficient than drugs alone [140]; therefore, nanotechnology is anticipated to be useful as vectors transporting antimicrobial agents across the BBB and subsequently provide new avenues for drug screening and therapeutic interventions in the central nervous system. However, the main bacterial pathogens infecting the CNS are S. pneumoniae, H. influenzae, and N. meningitides [141]. The efficacy of nanomedicine toward meningitis caused by these common pathogens remains unclear. Moreover, the most troublesome problem in the management of meningitis is the difficulty in detecting pathogens in the CNS. Methods to apply nanotechnology in the diagnosis of CNS infections should be analyzed in the future.
5.2. Respiratory System: Combatting Multidrug Resistant Pathogens at High Concentrations
Respiratory tract infections, especially pneumonia, are often caused by some multidrug-resistant bacterial strains. MDR may be overcome through the nanoparticle-mediated delivery of high-concentration antibacterial agents at the site of infection [16,122,123]. A dextran-coated stimuli-responsive nanoparticle that encapsulated the hydrophobic antibiotic rifampicin had a strong affinity for a variety of pathogens and effectively accumulated in bacteria-infected tissues. NPs were activated by either low pH or high concentrations of reactive oxygen species in the infectious microenvironment and released both cationic polymers and rifampicin that exerted synergistic effects on pneumonia in a mouse model of drug-resistant P. aeruginosa [16]. Antibacterial peptides loaded into porous silicon NPs (pSiNPs) exert a therapeutic effect on lung infection caused by P. aeruginosa, resulting in an obvious reduction in the bacterial load and improved survival in a mouse model [123].
Chemically engineered polyvinylpyrrolidone (PVP)-capped Ag NPs showed antibacterial activity against carbapenem-resistant strains of A. baumannii, a critical pathogen causing nosocomial respiratory tract infections. Ag NPs decreased the number of intracellular bacteria and bacterial adherence without any cytotoxic effects on human pulmonary cells [122].
Multidrug resistant, high mortality Burkholderia mallei is associated with weaponization by inducing aerosol inhalation, which results in glanders [98]. Au NPs functionalized with a glycoconjugate vaccine covalently conjugated to one of three different protein carriers (TetHc, Hcp1 and FliC) followed by conjugation to lipopolysaccharide (LPS) purified from a nonvirulent clonal relative, B. thailandensis, protect against lethal inhalations of B. mallei [121].
NPs have also been utilized for the accurate diagnosis of respiratory infectious disease [44]. LAMP-LFB provides simple, reliable, and objective detection of M. pneumoniae, one of the most common pathogens causing respiratory tract infection. This method has been particularly effective in the diagnosis of community-acquired pneumonia in school-age children [62].
Nanomaterials have been widely used to detect or treat bacterial infections in the respiratory system. In an era of respiratory tract infection with multidrug resistant pathogens, the high concentration of the drug delivered by NPs is expected to have more potent antibacterial activity. However, the main nanotechnologies used in the diagnosis or treatment of the respiratory system target only one or a few specific pathogens. In the real world, some of the pathogens in the respiratory system are multidrug resistant and polymicrobial, indicating that infection is caused by many bacteria. The treatment of multidrug-resistant and polymicrobial pathogens in the respiratory system will be the aim of future investigations.
5.3. Gastrointestinal System: Bypassing the Acidic Environment of the Stomach
The acidic environment of the stomach is an important defense mechanism of the human body, which eradicates ingested pathogens, but this acidity is also a threat to the therapeutic efficacy of antibiotics or probiotics [19,127]. The construction of delivery vectors that allow therapeutic agents to bypass the acidic environment of the stomach has always been a central aim in the development of NPs for treating bacterial infections in the gastrointestinal system [19,127]. Helicobacter pylori is the most well-known pathogen associated with gastric ulcers and gastric cancer, but the acidity of the stomach limits the therapeutic efficacy of antibiotics that eradicate H. pylori at the infection site [127]. Urea-modified UCCs-2 were used as a targeting moiety for UreI channel proteins that are specifically expressed on H. pylori. These pH-sensitive amoxicillin-loaded AMX-PLGA/UCCs-2 NPs protected antimicrobial drugs from the acidic environment of the stomach, safely delivering them to the H. pylori infection site [127].
Probiotics, such as Bifidobacterium, are vulnerable to gastric juices or gastrointestinal enzymes, and thus encapsulation is crucial to bypassing erosion and reaching target environments in the intestine or colon [19]. After encapsulation within alginate hydrogel-based shells, B. breve protected against simulated gastric acid and antimicrobial agents (tetracycline), thus successfully eradicating E. coli adhering to intestinal epithelial layers and maintaining surface coverage of Transwell membranes and their barrier integrity [19].
One of the most widespread toxin-mediated gastrointestinal pathogens is V. cholerae, which induces diarrhea by secreting cholera toxins [128]. The key host receptor for cholera toxin, monosialotetrahexosylganglioside (GM1), was coated onto the surface of polymeric NPs to function as toxin decoys. These NPs selectively and stably bound cholera toxins, neutralizing their actions on epithelial cells [128]. Another poly D,L-lactide-coglycolide (PLGA) nanoparticle encapsulated honeybee (Apis mellifera) venom and was capable of promoting the clearance of S. enterica serovar Typhimurium infection through the upregulation of T helper type 1-specific immune responses [125].
An antimicrobial peptide, VG16KRKP (VARGWKRKCPLFGKGG), delivered by gold nanoparticles (Au-VG16KRKP) was designed to tag against S. typhi, a common intestinal pathogen. These NPs were characterized as noncytotoxic to eukaryotic cells but exhibited strong bacteriolytic activity against intracellular S. typhi [126]. Another leading cause of diarrhea worldwide, Shigella flexneri (Shigellosis) was fortunately prevented by some outer membrane vesicles (OMVs) [124]. OMVs encapsulated in poly(anhydride) NPs were successfully developed for mucosal protection against S. flexneri when delivered through the nasal route [124].
NPs have been widely used as delivery vectors for therapeutic agents to bypass the acidic environment of the stomach as treatments for bacterial infections in the gastrointestinal system. The gastrointestinal system is a very complex environment composed of the microbiome and metabolome. The effects of NPs on the microbiota and its metabolites in the gut requires further evaluation.
5.4. Skeletal System: Effective in Bone Regeneration
Many NPs with bactericidal effects have been used to treat bone infection (such as osteomyelitis) or as part of the components of implants in the skeletal system [15,129,130,131]. Bioactive nanostructured CaSi layers on titanium substrates constructed by electrospray deposition were shown to exert antibacterial effects against gram-negative E. coli and gram-positive S. aureus species, with additional osteogenic properties when coated on bone tissue [15]. Calcium-alginate (Ca-Alg) NPs crosslinked with phosphorylated polyallylamine (PPAA) and prepared by salting-out achieved 82.55% encapsulation of clindamycin, an antimicrobial agent for bone S. aureus infections [131]. This Ca-Alg/PPAA/clindamycin nanoparticle was effective against MRSA osteomyelitis, with benefits in bone regeneration that prompted the recovery of infected fragments [131]. Another chitosan (CS)-based thermosensitive hydrogel was engineered to support vancomycin, an antimicrobial agent, in an osteomyelitis-NP/gel local drug delivery system. Vancomycin-NPs produced with quaternary ammonium chitosan and carboxylated chitosan NPs using positive and negative charge adsorption promoted osteoblast proliferation and were effective against osteomyelitis caused by S. aureus infection [129]. Silver-copper-boron (ACB) NPs were also used to treat infected osteoblasts by targeting intracellular S. aureus in bone cells, decreasing systemic toxicity and forming the basis of targeting specific markers expressed in bone infections [130]. In addition to antimicrobial effects, NPs are effective at promoting bone regeneration during the treatment of bone infection or as part of the components of implants in the skeletal system ex vivo and in vivo. The beneficial effect of its application in clinical patients remains unclear.
5.5. Skin and Soft Tissue: Targeting Bacteria and Their Biofilms
S. aureus is the most noticeable threat contributing to skin and soft tissue infection worldwide, with the capacity to form biofilms during infection [16,17,85]. Near-infrared-light (NIR)-activatable deoxyribonuclease (DNase)-carbon monoxide (CO) MPDA NPs successfully delivered bactericidal CO gas, penetrating impaired S. aureus biofilms and effectively eliminating them through the collaborative therapeutic effect of DNase I and CO gas-potentiated photothermal treatment [20]. A dextran-coated stimuli-responsive NP that encapsulated the hydrophobic antibiotic rifampicin accumulated in bacteria-infected tissues and was activated by either low pH or high levels of reactive oxygen species. When activated, the NPs released both cationic polymers and rifampicin, showing synergistic effects on drug-resistant S. aureus soft tissue (thigh) infection in a mouse model [16].
Biocompatible enzyme-responsive Ag NP assemblies (ANAs) are high-efficiency targeted antimicrobial treatments for MRSA and have been utilized as effective wound dressings to accelerate the healing of wounds with MRSA infections [17]. A biomimetic NP-based antivirulence (detained staphylococcal α-hemolysin (Hla)) vaccine that efficiently induced high anti-Hla titers exhibits protective immunity against MRSA skin infections and subsequently decreases the invasiveness of MRSA, preventing dissemination into other organs [132].
Group A Streptococcus (GAS), along with its infamous pore-forming streptolysin O (SLO) toxin, is a major pathogen causing severely invasive skin infections, including necrotizing fasciitis [133]. The pharmacological appearance of red blood cell (RBC)-derived biomimetic NPs (“nanosponges”) sequestered SLO and blocked the capacity of GAS to injure host cells, thereby preserving innate immune function while increasing bacterial clearance in a mouse model of GAS necrotizing skin infection [133].
V. vulnificus could cause quick and severe tissue damage, with the mortality rate as high as 50% [134,142]. The antimicrobial peptide (AMP) HPA3PHis loaded onto a Au NP-DNA aptamer (AuNP-Apt) coupled (AuNP-Apt-HPA3PHis) was effective against V. vulnificus infection by damaging membrane integrity, and decreasing intracellular V. vulnificus numbers up to 90% [134].
NPs have the advantages of targeting bacteria and their biofilms during the treatment of skin and soft tissue infections, especially tissues infected with S. aureus in vivo. However, evidence of the effect of NPs on eradicating bacteria hidden in biofilms or implants in clinical patients is still limited. More clinical studies are needed to confirm the effect of NPs on eradicating bacteria in biofilms or implants.
5.6. Ophthalmology System/Prolonged Drug Release with Less Uncomfortable Sensations
Several ocular drug delivery systems, such as hydrogels, microparticles, nanoemulsions, microemulsions, and liposomes, have been analyzed for drug release in the eye and sustain therapeutic levels for a prolonged period [143]. Nanomaterial-containing eye drops with the advantage of less uncomfortable sensations, fewer blinking reflexes, and less blurred visions are needed to better treat infectious ophthalmological diseases [135]. Moxifloxacin (MFX, an antibiotic) and dexamethasone (DEX, a type of steroid)-loaded nanostructured lipid carrier (Lipo-MFX/DEX) mixed with collagen/gelatin/alginate (CGA) biodegradable materials (CGA-Lipo-MFX/DEX) have been used as anti-inflammatory agents for treating ocular diseases, with properties that sustain drug release for at least 12 h, inhibit pathogen microorganism growth and improve corneal wound healing [135]. Zinc oxide (ZnO)/cyclized polyacrylonitrile (CPAN) composite-loaded contact lenses showed broad-spectrum antibacterial properties with the capacity to selectively block UVB and UVA, as well as blue light, under the premise of ensuring hydrophilicity and a certain transparency [144]. Artificial eyes containing Ag NPs significantly inhibited the growth of S. pneumoniae, S. aureus, P. aeruginosa, and E. coli [145]. In addition to bactericidal effects, various emerging nanomaterials, such as NPs, nanowires, hybrid nanostructures, and nanoscaffolds, have been tested in mouse models for ocular tissue engineering and regeneration [146]. Although NP-containing eye drops or ocular equipment provide prolonged drug release with less uncomfortable sensations, their long-term effects on the cornea and other eye structures require further evaluation.
5.7. Dental System/Overcoming the Complex Environments That Are Susceptible to Insults
The oral cavity and oropharynx are complex locations that face physical, chemical, and microbiological damage and have pathological and cancerous variations [147]. Alveolar bone loss is a common problem that affects dental implant placement and is influenced by fibrotic tissue ingrowth and bacterial infection [136]. Micro/nanoencapsulants with unique structures and properties are more favorable drug-release platforms for treating dental diseases than conventional treatment approaches [147]. Ag coating methods provide bioactive collagen barriers that help guide bone regeneration, and they are currently being coupled with antibacterial and anti-inflammatory properties [136]. An Ag-NP-coated collagen membrane showed outstanding antibacterial effects on S. aureus and P. aeruginosa, as well as anti-inflammatory effects achieved by reducing the expression and release of inflammatory cytokines, including IL-6 and TNF-α, and inducing the osteogenic differentiation of mesenchymal stem cells that guide bone regeneration [136]. Micro/nanomaterials of bioactive glasses (BGs) with biocompatibility and osteogenesis-promoting agents were analyzed using Zn-doped bioactive glass micro/nanospheres for dental pulp capping to combat bacterial infection and stimulate tissue renewal [148]. NPs can be used in the oral cavity and oropharynx to overcome complex environments that are susceptible to physical, chemical, and microbiological insults. However, the long-term efficacy and safety of these NPs in the complex environment of the oral cavity of clinical patients warrant further evaluation.
5.8. Reproductive System (Sexually Transmitted Disease)/Increasing the Retention Time and Decreasing Discomfort
The incidence of reproductive system infections (sexually transmitted disease), especially vaginal infections, has recently increased, especially among women of reproductive age, and requires an effective concentration of topical antimicrobial treatment for its direct therapeutic action, reduced drug doses and side effects, and self-insertion [149]. However, alterations in the physiological conditions of the vagina significantly alter the effectiveness of vaginally delivered drugs [149]. Consequently, nanomedicine has been applied with topical antimicrobial agents to increase the retention time in the vagina and decrease discomfort [149].
Neisseria gonorrhoeae, the causative agent of gonorrhea, is an important target for the treatment of sexually transmitted disease. MtrE, the outer membrane channel of the highly conserved gonococcal MtrCDE active efflux pump, is a surrogate for vaccine targets because of its significance in defending N. gonorrhoeae from host innate effectors [137]. Thus, ferritin NPs (ferritin nanocages) have been utilized as vectors for MtrE in gonorrhea vaccines [137].
Although NPs have not been used as often as treatments for the reproductive system thus far, the use of nanomedicine to increase the retention time and decrease discomfort is expected in the future. However, the effects of these NPs on the fertility of clinical patients should be evaluated.
5.9. Reticuloendothelial System
The reticuloendothelial system (RES) is also called the macrophage system because of the common property of phagocytosis, where the cells engulf and destroy bacteria, viruses, and other foreign substances, as well as ingest worn-out or abnormal cells in the body. Since intravenously administered NPs are recognized and quickly cleared from systemic circulation by the RES [150], the use of activated phagocytes for NPs delivering antimicrobial drugs to treat infectious diseases is an ideal strategy. This approach has limited use, however, when fighting local infections [151]. Gupta et al. (2004) revealed that the surface coating of superparamagnetic iron oxide NPs with hydrophilic-hydrophobic polymeric compounds (PEG) can be used to deliver antimicrobial agents [152]. The result was attained by reducing plasma protein adsorption to the NPs and eliminating RES uptake [152,153]. Consistently, NPs modified with PEGylated polymers could reduce the adherence of serum proteins, coupled with opsonins, and cleared by the RES [154]. However, some of these antimicrobials agents, although effective against some bacteria, might be poisonous to mammalian cells [155,156]. Polymers have nonetheless been used in NP preparations to increase the appropriate host response to cationic antimicrobial agents in a balanced manner [157]. Although NPs have been used in the RES to facilitate eradication of systemic bacterial infection ex vivo and in vivo, the efficacy and adverse side effects of NPs on the RES system of clinical patients are still uncertain.
6. Advances in Developing Nanotechnology for Bacterial Infectious Diseases
Many methods have been shown to increase the function of NPs in preventing bacterial infection or improving wound repair, including encapsulation methods and material design [19], the incorporation of activation systems in NPs [16,20], and the addition of immune-activating adjuvants [158]. The protection strength of encapsulating shells, however, varies among encapsulation methods and materials [19]. Alginate gelation (intermediate interaction due to hydrogen bonding) provided better protection of probiotic B. breve against simulated gastric fluid and antimicrobial agents (tetracycline) than protamine-assisted SiO2 NPs, yolk−shell packing (weak interactions across a void), and ZIF-8 mineralization (strong interaction due to coordinated covalent bonding) [19]. The selection of methods to increase the function of nanotechnology depends on the purpose of the diagnosis or treatment of the bacterial infectious disease.
The hydrophilicity and roughness of NPs modulate their ability to prevent biomaterial-associated infections [90]. Au NPs that are hydrophilic/rough (water contact angle of 30 degrees/surface roughness of 118 nm) provide better cellular interactions, facilitating macrophage-mediated repair of damage [90]. Within the same nanomaterial, the incorporation of photothermal or photodynamic properties [20] or an activation system triggered by low pH or high concentrations of reactive oxygen species both improve antibacterial effects [16]. NPs containing both DNase I and CO gas-potentiated photothermal therapy are activated after permeating the impaired biofilms with the help of NIR to combat S. aureus hiding inside biofilms [20]. A dextran-coated stimuli-responsive NP that encapsulated a hydrophobic antibiotic has strong affinity for a variety of pathogens and effectively accumulates in bacterial infected tissues. NPs are then activated by either low pH or high concentrations of reactive oxygen species in the infectious microenvironment to release the payload of antimicrobial agents [16]. The modification of the hydrophilicity and roughness of the NPs influences their effect on the infectious microenvironment.
Combinations of biomaterials encapsulated in NPs might provide additive therapeutic effects on bacterial infectious diseases [159]. Combining the nitric oxide donor S-nitroso-N-acetyl-penicillamine with strong antioxidant cerium oxide NPs (CNPs) induced a synergistic therapeutic interaction that inhibited the growth of S. aureus, E. coli and Candida albicans [159]. Liposomal NPs have also been investigated for drug delivery, with advantages of increasing the cellular uptake of drugs, as well as stabilizing the physicochemical properties and bioavailability of both water-soluble drugs and drugs with poor water solubility [135]. Antibiotics incorporated in liposomal NPs prolong the drug retention time in the local area and have been used to treat ophthalmological diseases [135]. An enzyme-responsive NP has been shown to be highly effective at targeting MRSA. NPs were prepared from enzyme-responsive branched copolymers (BPs) synthesized by living radical polymerizations and underwent stable/collapsed transitions upon exposure to serine protease-like B enzyme proteins (SplB) secreted by MRSA [17]. The strategy of combining biomaterials is anticipated to provide additive therapeutic effects.
The addition of immune-activating adjuvants represents another method for enhancing the effect of NPs [158]. Invariant NK T (iNKT) cells are innate glycolipid-specific T cells restricted to the nonpolymorphic Ag-presenting molecule CD1d, and an iNKT glycolipid agonist, α-galactosylceramide (αGC), was used as an adjuvant for a human protective epitope [158]. An NP that carries αGC plus antigenic polysaccharides from S. pneumoniae αGC-embedded NPs was used to trigger murine iNKT cells and B cells while avoiding iNKT anergy [158]. NPs containing αGC plus S. pneumoniae polysaccharides provoked vigorous IgM and IgG responses and defended mice against lethal S. pneumoniae infections [158]. Magnetic NP/alternating magnetic field (MNP/AMF) hyperthermia combined with conventional antibiotics was more effective against S. aureus biofilms, inducing increased uptake of antibiotics to the bacterial cells and promoting the bactericidal activity of macrophages against intracellular bacteria via the generation of reactive oxygen species (ROS) [160]. The incorporation of immune-activating adjuvants might further potentiate the effect of NPs.
Modification of the hydrophilicity and roughness of the NPs, combinations of biomaterials, or incorporation of immune-activating adjuvants are predicted to increase the therapeutic effect of NPs without increasing the adverse effects. However, the selection of the appropriate methods to increase the function of nanotechnology depends on the aims of the diagnosis or treatment of the bacterial infectious disease.
7. Conclusions
In an era of bacterial infections characterized by increasing antimicrobial resistance, medicine is running out of weapons to combat these pathogens. Although some new antimicrobial agents have been developed in recent years, the emergence of bacterial resistance against these new drugs was reported a few years later. The development of NPs against these troublesome bacteria might be an alternative therapeutic choice with more bactericidal effects and less cytotoxicity. In recent years, numerous advances in the application of organic, inorganic and hybrid NPs to combat bacterial infectious diseases have been reported. SNAPs have excellent antibacterial activity and low toxicity; expanded use of star-shaped polymers for both the diagnosis and treatment of infectious diseases is expected in the future. To date, most developed NPs have been applied superficially on the body, such as in wound infections, skin and soft tissue infections, dental infections, or ophthalmological infections. Thus, the use of NPs inside the body to combat internal organ infections still has some challenges to overcome, including methods to guide the particle to the target organ or area, confirm the biosafety of the particle when administered inside the body, protect the particle against the damage of gastric juices or intestinal enzymes (for orally administered NPs) and confirm that encapsulated agents had higher efficacy than unencapsulated agents. Fortunately, many methods to increase the function of NPs in preventing bacterial infection or reducing cytotoxicity have been developed, including changing encapsulation methods and materials, incorporating activation systems, or adding immune-activating adjuvants. The construction of star-shaped polymers, for example, has shown the potential for increased antibacterial effects and decreased toxicity to mammalian cells. The development of appropriate NPs specific for the target organ using an effective delivery method will be a challenge to overcome when developing NPs as treatments for bacterial infections in the future.
Abbreviations
| A. baumannii | Acinetobacter baumannii |
| Ag | silver |
| Au | gold |
| B. mallei | Burkholderia mallei |
| E. coli | Escherichia coli |
| H. pylori | Helicobacter pylori |
| M. pneumoniae | Mycoplasma pneumoniae |
| NP | nanoparticle |
| N. gonorrhoeae | Neisseria gonorrhoeae |
| P. aeruginosa | Pseudomonas aeruginosa |
| S. typhi | Salmonella typhi |
| S. enterica serovar Typhimurium | Salmonella enterica serovar Typhimurium |
| S. flexneri | Shigella flexneri |
| S. aureus | Staphylococcus aureus |
| V. cholerae | Vibrio cholerae |
| V. vulnificus | Vibrio vulnificus |
Author Contributions
Y.-P.H., Y.-F.C.: writing—original draft preparation; P.-J.T., I.-H.H.: review and editing; J.-S.J., W.-C.K.: read and approved the final version of the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
The present study is supported by research grants from the Ministry of Science and Technology, Taiwan (MOST 110-2314-B-675-001, 109-2314-B-006-089-MY3, 108-2321-B-006-004, 108-2320-B-006-043-MY3, 108-2221-E-006-034-MY3, and 107-2923-M-006-002-MY3).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors have no conflict of interest to declare.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Duval R.E., Grare M., Demore B. Fight against Antimicrobial Resistance: We Always Need New Antibacterials but for Right Bacteria. Molecules. 2019;24:3152. doi: 10.3390/molecules24173152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Penesyan A., Gillings M., Paulsen I.T. Antibiotic discovery: Combatting bacterial resistance in cells and in biofilm communities. Molecules. 2015;20:5286–5298. doi: 10.3390/molecules20045286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Aguilera-Correa J.J., Esteban J., Vallet-Regi M. Inorganic and Polymeric Nanoparticles for Human Viral and Bacterial Infections Prevention and Treatment. Nanomaterials. 2021;11:137. doi: 10.3390/nano11010137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lyu Y., Yu M., Liu Q., Zhang Q., Liu Z., Tian Y., Li D., Changdao M. Synthesis of silver nanoparticles using oxidized amylose and combination with curcumin for enhanced antibacterial activity. Carbohydr. Polym. 2020;230:115573. doi: 10.1016/j.carbpol.2019.115573. [DOI] [PubMed] [Google Scholar]
- 5.Nag S., Biswas A., Chattopadhyay D., Bhattacharyya M. Protein-stabilized silver nanoparticles encapsulating gentamycin for the therapy of bacterial biofilm infections. Nanomedicine. 2021;16:801–818. doi: 10.2217/nnm-2020-0451. [DOI] [PubMed] [Google Scholar]
- 6.Makabenta J.M.V., Nabawy A., Li C.H., Schmidt-Malan S., Patel R., Rotello V.M. Nanomaterial-based therapeutics for antibiotic-resistant bacterial infections. Nat. Rev. Microbiol. 2021;19:23–36. doi: 10.1038/s41579-020-0420-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brunetti G., Padovani F., De Pastina A., Rotella C., Monahan A., Hoffman S.L., Jongo S.A., Abdulla S., Corradin G., Pluschke G., et al. Nanotechnological immunoassay for rapid label-free analysis of candidate malaria vaccines. Nanoscale. 2021;13:2338–2349. doi: 10.1039/D0NR08083G. [DOI] [PubMed] [Google Scholar]
- 8.Knoblauch R., Harvey A., Ra E., Greenberg K.M., Lau J., Hawkins E., Geddes C.D. Antimicrobial carbon nanodots: Photodynamic inactivation and dark antimicrobial effects on bacteria by brominated carbon nanodots. Nanoscale. 2021;13:85–99. doi: 10.1039/D0NR06842J. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bekmukhametova A., Ruprai H., Hook J.M., Mawad D., Houang J., Lauto A. Photodynamic therapy with nanoparticles to combat microbial infection and resistance. Nanoscale. 2020;12:21034–21059. doi: 10.1039/D0NR04540C. [DOI] [PubMed] [Google Scholar]
- 10.Guo R., Li K., Qin J., Niu S., Hong W. Development of polycationic micelles as an efficient delivery system of antibiotics for overcoming the biological barriers to reverse multidrug resistance in Escherichia coli. Nanoscale. 2020;12:11251–11266. doi: 10.1039/D0NR01366H. [DOI] [PubMed] [Google Scholar]
- 11.Liu W., Zhang Y., You W., Su J., Yu S., Dai T., Huang Y., Chen X., Song X., Chen Z. Near-infrared-excited upconversion photodynamic therapy of extensively drug-resistant Acinetobacter baumannii based on lanthanide nanoparticles. Nanoscale. 2020;12:13948–13957. doi: 10.1039/D0NR01073A. [DOI] [PubMed] [Google Scholar]
- 12.Guo X., Cao B., Wang C., Lu S., Hu X. In vivo photothermal inhibition of methicillin-resistant Staphylococcus aureus infection by in situ templated formulation of pathogen-targeting phototheranostics. Nanoscale. 2020;12:7651–7659. doi: 10.1039/D0NR00181C. [DOI] [PubMed] [Google Scholar]
- 13.Yin W., Li W., Li Q., Liu Y., Liu J., Ren M., Ma Y., Zhang Z., Zhang X., Wu Y., et al. Real-time imaging of individual virion-triggered cortical actin dynamics for human immunodeficiency virus entry into resting CD4 T cells. Nanoscale. 2020;12:115–129. doi: 10.1039/C9NR07359K. [DOI] [PubMed] [Google Scholar]
- 14.Park S.M., Kim D.A., Jo J.K., Jun S.K., Jang T.S., Kim H.W., Lee J.H., Lee H.H. Ceria-Incorporated Biopolymer for Preventing Fungal Adhesion. ACS Biomater. Sci. Eng. 2021;7:1808–1816. doi: 10.1021/acsbiomaterials.0c01039. [DOI] [PubMed] [Google Scholar]
- 15.Buga C., Chen C.C., Hunyadi M., Csik A., Hegedus C., Ding S.J. Electrosprayed calcium silicate nanoparticle-coated titanium implant with improved antibacterial activity and osteogenesis. Colloids Surf. B Biointerfaces. 2021;202:111699. doi: 10.1016/j.colsurfb.2021.111699. [DOI] [PubMed] [Google Scholar]
- 16.Ye M., Zhao Y., Wang Y., Zhao M., Yodsanit N., Xie R., Andes D., Gong S. A Dual-Responsive Antibiotic-Loaded Nanoparticle Specifically Binds Pathogens and Overcomes Antimicrobial-Resistant Infections. Adv. Mater. 2021;33:2006772. doi: 10.1002/adma.202006772. [DOI] [PubMed] [Google Scholar]
- 17.Zuo Y.M., Yan X., Xue J., Guo L.Y., Fang W.W., Sun T.C., Li M., Zha Z., Yu Q., Wang Y., et al. Enzyme-Responsive Ag Nanoparticle Assemblies in Targeting Antibacterial against Methicillin-Resistant Staphylococcus Aureus. ACS Appl. Mater. Interfaces. 2020;12:4333–4342. doi: 10.1021/acsami.9b22001. [DOI] [PubMed] [Google Scholar]
- 18.Zhen J.B., Kang P.W., Zhao M.H., Yang K.W. Silver Nanoparticle Conjugated Star PCL-b-AMPs Copolymer as Nanocomposite Exhibits Efficient Antibacterial Properties. Bioconjug. Chem. 2020;31:51–63. doi: 10.1021/acs.bioconjchem.9b00739. [DOI] [PubMed] [Google Scholar]
- 19.Yuan L., Wei H., Yang X.Y., Geng W., Peterson B.W., van der Mei H.C., Busscher H.J. Escherichia coli Colonization of Intestinal Epithelial Layers In Vitro in the Presence of Encapsulated Bifidobacterium breve for Its Protection against Gastrointestinal Fluids and Antibiotics. ACS Appl Mater. Interfaces. 2021;13:15973–15982. doi: 10.1021/acsami.0c21790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yuan Z., Lin C., Dai L., He Y., Hu J., Xu K., Tao B., Liu P., Cai K. Near-Infrared Light-Activatable Dual-Action Nanoparticle Combats the Established Biofilms of Methicillin-Resistant Staphylococcus aureus and Its Accompanying Inflammation. Small. 2021;17:2007522. doi: 10.1002/smll.202007522. [DOI] [PubMed] [Google Scholar]
- 21.Ding S.J., Chu Y.H., Chen P.T. Mechanical Biocompatibility, Osteogenic Activity, and Antibacterial Efficacy of Calcium Silicate-Zirconia Biocomposites. ACS Omega. 2021;6:7106–7118. doi: 10.1021/acsomega.1c00097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lin M., Long H., Liang M., Chu B., Ren Z., Zhou P., Wu C., Liu Z., Wang Y. Antifracture, Antibacterial, and Anti-inflammatory Hydrogels Consisting of Silver-Embedded Curdlan Nanofibrils. ACS Appl. Mater. Interfaces. 2021;13:36747–36756. doi: 10.1021/acsami.1c06603. [DOI] [PubMed] [Google Scholar]
- 23.Chen A., Hernandez-Vargas J., Han R., Cortazar-Martinez O., Gonzalez N., Patel S., Keitz B.K., Luna-Barcenas G., Contreras L.M. Small RNAs as a New Platform for Tuning the Biosynthesis of Silver Nanoparticles for Enhanced Material and Functional Properties. ACS Appl. Mater. Interfaces. 2021;13:36769–36783. doi: 10.1021/acsami.1c07400. [DOI] [PubMed] [Google Scholar]
- 24.Zhao B., Zheng K., Liu C. Bio-dissolution process and mechanism of copper phosphate hybrid nanoflowers by Pseudomonas aeruginosa and its bacteria-toxicity in life cycle. J. Hazard. Mater. 2021;419:126494. doi: 10.1016/j.jhazmat.2021.126494. [DOI] [PubMed] [Google Scholar]
- 25.Lin Z., Liu L., Wang W., Jia L., Shen Y., Zhang X., Ge D., Shi W., Sun Y. The role and mechanism of polydopamine and cuttlefish ink melanin carrying copper ion nanoparticles in antibacterial properties and promoting wound healing. Biomater. Sci. 2021;9:5951–5964. doi: 10.1039/D1BM00622C. [DOI] [PubMed] [Google Scholar]
- 26.Michelini S., Barbero F., Prinelli A., Steiner P., Weiss R., Verwanger T., Andosch A., Lutz-Meindl U., Puntes V.F., Drobne D., et al. Gold nanoparticles (AuNPs) impair LPS-driven immune responses by promoting a tolerogenic-like dendritic cell phenotype with altered endosomal structures. Nanoscale. 2021;13:7648–7666. doi: 10.1039/D0NR09153G. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tang Y., Qin Z., Yin S., Sun H. Transition metal oxide and chalcogenide-based nanomaterials for antibacterial activities: An overview. Nanoscale. 2021;13:6373–6388. doi: 10.1039/D1NR00664A. [DOI] [PubMed] [Google Scholar]
- 28.Quan K., Jiang G., Liu J., Zhang Z., Ren Y., Busscher H.J., van der Mei H.C., Peterson B.W. Influence of interaction between surface-modified magnetic nanoparticles with infectious biofilm components in artificial channel digging and biofilm eradication by antibiotics in vitro and in vivo. Nanoscale. 2021;13:4644–4653. doi: 10.1039/D0NR08537E. [DOI] [PubMed] [Google Scholar]
- 29.Damasco J.A., Ravi S., Perez J.D., Hagaman D.E., Melancon M.P. Understanding Nanoparticle Toxicity to Direct a Safe-by-Design Approach in Cancer Nanomedicine. Nanomaterials. 2020;10:2186. doi: 10.3390/nano10112186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.He C., Zhuang X., Tang Z., Tian H., Chen X. Stimuli-sensitive synthetic polypeptide-based materials for drug and gene delivery. Adv. Healthc. Mater. 2012;1:48–78. doi: 10.1002/adhm.201100008. [DOI] [PubMed] [Google Scholar]
- 31.Huang Y.F., Lu S.C., Huang Y.C., Jan J.S. Cross-linked, self-fluorescent gold nanoparticle/polypeptide nanocapsules comprising dityrosine for protein encapsulation and label-free imaging. Small. 2014;10:1939–1944. doi: 10.1002/smll.201303462. [DOI] [PubMed] [Google Scholar]
- 32.Carlsen A., Lecommandoux S. Self-assembly of polypeptide-based block copolymer amphiphiles. Curr. Opin. Colloid Interface Sci. 2009;14:329–339. doi: 10.1016/j.cocis.2009.04.007. [DOI] [Google Scholar]
- 33.Siegwart D.J., Oh J.K., Matyjaszewski K. ATRP in the design of functional materials for biomedical applications. Prog. Polym. Sci. 2012;37:18–37. doi: 10.1016/j.progpolymsci.2011.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Xia L.W., Xie R., Ju X.J., Wang W., Chen Q., Chu L.Y. Nano-structured smart hydrogels with rapid response and high elasticity. Nat. Commun. 2013;4:2226. doi: 10.1038/ncomms3226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hsiao L.-W., Lai Y.-D., Lai J.-T., Hsu C.-C., Wang N.-Y., Steven S.-S.W., Jan J.-S. Cross-linked polypeptide-based gel particles by emulsion for efficient protein encapsulation. Polymer. 2017;115:261–272. doi: 10.1016/j.polymer.2017.03.055. [DOI] [Google Scholar]
- 36.Chen Y.F., Chen G.Y., Chang C.H., Su Y.C., Chen Y.C., Jiang Y.S., Jan J.S. TRAIL encapsulated to polypeptide-crosslinked nanogel exhibits increased anti-inflammatory activities in Klebsiella pneumoniae-induced sepsis treatment. Mater. Sci. Eng. C Mater. Biol. Appl. 2019;102:85–95. doi: 10.1016/j.msec.2019.04.023. [DOI] [PubMed] [Google Scholar]
- 37.Ragothaman M., Kannan Villalan A., Dhanasekaran A., Palanisamy T. Bio-hybrid hydrogel comprising collagen-capped silver nanoparticles and melatonin for accelerated tissue regeneration in skin defects. Mater. Sci. Eng. C Mater. Biol. Appl. 2021;128:112328. doi: 10.1016/j.msec.2021.112328. [DOI] [PubMed] [Google Scholar]
- 38.Perez-Rafael S., Ivanova K., Stefanov I., Puiggali J., Del Valle L.J., Todorova K., Dimitrov P., Hinojosa-Caballero D., Tzanov T. Nanoparticle-driven self-assembling injectable hydrogels provide a multi-factorial approach for chronic wound treatment. Acta Biomater. 2021;134:131–143. doi: 10.1016/j.actbio.2021.07.020. [DOI] [PubMed] [Google Scholar]
- 39.Huang Y., Song G., Chang X., Wang Z., Zhang X., Han S., Su Z., Yang H., Yang D., Zhang X. Nanostructured Ag(+)-substituted fluorhydroxyapatite-TiO2 coatings for enhanced bactericidal effects and osteoinductivity of Ti for biomedical applications. Int. J. Nanomed. 2018;13:2665–2684. doi: 10.2147/IJN.S162558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jin P., Wang L., Sha R., Liu L., Qian J., Ishimwe N., Zhang W., Qian J., Zhang Y., Wen L. A blood circulation-prolonging peptide anchored biomimetic phage-platelet hybrid nanoparticle system for prolonged blood circulation and optimized anti-bacterial performance. Theranostics. 2021;11:2278–2296. doi: 10.7150/thno.49781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Huang Z., Zhou T., Yuan Y., Natalie Klodzinska S., Zheng T., Sternberg C., Morck Nielsen H., Sun Y., Wan F. Synthesis of carbon quantum dot-poly lactic-co-glycolic acid hybrid nanoparticles for chemo-photothermal therapy against bacterial biofilms. J. Colloid Interface Sci. 2020;577:66–74. doi: 10.1016/j.jcis.2020.05.067. [DOI] [PubMed] [Google Scholar]
- 42.Kalita S., Kandimalla R., Bhowal A.C., Kotoky J., Kundu S. Functionalization of beta-lactam antibiotic on lysozyme capped gold nanoclusters retrogress MRSA and its persisters following awakening. Sci. Rep. 2018;8:5778. doi: 10.1038/s41598-018-22736-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mondal S., Ghosh R., Adhikari A., Pal U., Mukherjee D., Biswas P., Darbar S., Singh S., Bose S., Saha-Dasgupta T., et al. In vitro and Microbiological Assay of Functionalized Hybrid Nanomaterials to Validate Their Efficacy in Nanotheranostics: A Combined Spectroscopic and Computational Study. ChemMedChem. 2021 doi: 10.1002/cmdc.202100494. in press. [DOI] [PubMed] [Google Scholar]
- 44.Wang T., Li X., Chen L., Zhang Y., Zheng Y., Yu L., Ye Z., Wang H., Cui X., Zhao S. The preparation of bifunctional hybrid nano-flowers and their application in the enzyme-linked immunosorbent assay for Helicobacter pylori detection. Analyst. 2021;146:338–347. doi: 10.1039/D0AN01533D. [DOI] [PubMed] [Google Scholar]
- 45.Wong E.H., Khin M.M., Ravikumar V., Si Z., Rice S.A., Chan-Park M.B. Modulating Antimicrobial Activity and Mammalian Cell Biocompatibility with Glucosamine-Functionalized Star Polymers. Biomacromolecules. 2016;17:1170–1178. doi: 10.1021/acs.biomac.5b01766. [DOI] [PubMed] [Google Scholar]
- 46.Pu Y., Hou Z., Khin M.M., Zamudio-Vazquez R., Poon K.L., Duan H., Chan-Park M.B. Synthesis and Antibacterial Study of Sulfobetaine/Quaternary Ammonium-Modified Star-Shaped Poly[2 -(dimethylamino)ethyl methacrylate]-Based Copolymers with an Inorganic Core. Biomacromolecules. 2017;18:44–55. doi: 10.1021/acs.biomac.6b01279. [DOI] [PubMed] [Google Scholar]
- 47.Lam S.J., O’Brien-Simpson N.M., Pantarat N., Sulistio A., Wong E.H., Chen Y.Y., Lenzo J.C., Holden J.A., Blencowe A., Reynolds E.C., et al. Combating multidrug-resistant Gram-negative bacteria with structurally nanoengineered antimicrobial peptide polymers. Nat. Microbiol. 2016;1:16162. doi: 10.1038/nmicrobiol.2016.162. [DOI] [PubMed] [Google Scholar]
- 48.Chen Y.F., Lai Y.D., Chang C.H., Tsai Y.C., Tang C.C., Jan J.S. Star-shaped polypeptides exhibit potent antibacterial activities. Nanoscale. 2019;11:11696–11708. doi: 10.1039/C9NR02012H. [DOI] [PubMed] [Google Scholar]
- 49.Xi Y., Song T., Tang S., Wang N., Du J. Preparation and Antibacterial Mechanism Insight of Polypeptide-Based Micelles with Excellent Antibacterial Activities. Biomacromolecules. 2016;17:3922–3930. doi: 10.1021/acs.biomac.6b01285. [DOI] [PubMed] [Google Scholar]
- 50.Pirzada M., Altintas Z. Nanomaterials for Healthcare Biosensing Applications. Sensors. 2019;19:5311. doi: 10.3390/s19235311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Jiang Z., Feng B., Xu J., Qing T., Zhang P., Qing Z. Graphene biosensors for bacterial and viral pathogens. Biosens. Bioelectron. 2020;166:112471. doi: 10.1016/j.bios.2020.112471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hao Z., Luo Y., Huang C., Wang Z., Song G., Pan Y., Zhao X., Liu S. An Intelligent Graphene-Based Biosensing Device for Cytokine Storm Syndrome Biomarkers Detection in Human Biofluids. Small. 2021;17:2101508. doi: 10.1002/smll.202101508. [DOI] [PubMed] [Google Scholar]
- 53.Schultz A., Knoll T., Urban A., Schuck H., von Briesen H., Germann A., Velten T. Novel Cost-Efficient Graphene-Based Impedance Biosensor for the Analysis of Viral Cytopathogenicity and the Effect of Antiviral Drugs. Front. Bioeng. Biotechnol. 2021;9:718889. doi: 10.3389/fbioe.2021.718889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Pandit S., Gaska K., Kadar R., Mijakovic I. Graphene-Based Antimicrobial Biomedical Surfaces. ChemPhysChem. 2021;22:250–263. doi: 10.1002/cphc.202000769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ji J., Pang Y., Li D., Wang X., Xu Y., Mu X. Single-Layered Graphene/Au-Nanoparticles-Based Love Wave Biosensor for Highly Sensitive and Specific Detection of Staphylococcus aureus Gene Sequences. ACS Appl. Mater. Interfaces. 2020;12:12417–12425. doi: 10.1021/acsami.9b20639. [DOI] [PubMed] [Google Scholar]
- 56.Appaturi J.N., Pulingam T., Thong K.L., Muniandy S., Ahmad N., Leo B.F. Rapid and sensitive detection of Salmonella with reduced graphene oxide-carbon nanotube based electrochemical aptasensor. Anal. Biochem. 2020;589:113489. doi: 10.1016/j.ab.2019.113489. [DOI] [PubMed] [Google Scholar]
- 57.Aguirre S.D., Ali M.M., Salena B.J., Li Y. A sensitive DNA enzyme-based fluorescent assay for bacterial detection. Biomolecules. 2013;3:563–577. doi: 10.3390/biom3030563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Pandey A., Gurbuz Y., Ozguz V., Niazi J.H., Qureshi A. Graphene-interfaced electrical biosensor for label-free and sensitive detection of foodborne pathogenic E. coli O157:H7. Biosens. Bioelectron. 2017;91:225–231. doi: 10.1016/j.bios.2016.12.041. [DOI] [PubMed] [Google Scholar]
- 59.Thakur B., Zhou G., Chang J., Pu H., Jin B., Sui X., Yuan X., Yang C.H., Magruder M., Chen J. Rapid detection of single E. coli bacteria using a graphene-based field-effect transistor device. Biosens. Bioelectron. 2018;110:16–22. doi: 10.1016/j.bios.2018.03.014. [DOI] [PubMed] [Google Scholar]
- 60.Kampeera J., Pasakon P., Karuwan C., Arunrut N., Sappat A., Sirithammajak S., Dechokiattawan N., Sumranwanich T., Chaivisuthangkura P., Ounjai P., et al. Point-of-care rapid detection of Vibrio parahaemolyticus in seafood using loop-mediated isothermal amplification and graphene-based screen-printed electrochemical sensor. Biosens. Bioelectron. 2019;132:271–278. doi: 10.1016/j.bios.2019.02.060. [DOI] [PubMed] [Google Scholar]
- 61.Molinero-Fernandez A., Arruza L., Lopez M.A., Escarpa A. On-the-fly rapid immunoassay for neonatal sepsis diagnosis: C-reactive protein accurate determination using magnetic graphene-based micromotors. Biosens. Bioelectron. 2020;158:112156. doi: 10.1016/j.bios.2020.112156. [DOI] [PubMed] [Google Scholar]
- 62.Wang Y., Wang Y., Jiao W., Li J., Quan S., Sun L., Wang Y., Qi X., Wang X., Shen A. Development of loop-mediated isothermal amplification coupled with nanoparticle-based lateral flow biosensor assay for Mycoplasma pneumoniae detection. AMB Express. 2019;9:196. doi: 10.1186/s13568-019-0921-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Savas S., Ersoy A., Gulmez Y., Kilic S., Levent B., Altintas Z. Nanoparticle Enhanced Antibody and DNA Biosensors for Sensitive Detection of Salmonella. Materials. 2018;11:1541. doi: 10.3390/ma11091541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Zhao F., Niu L., Nong J., Wang C., Wang J., Liu Y., Gao N., Zhu X., Wu L., Hu S. Rapid and sensitive detection of Pseudomonas aeruginosa using multiple cross displacement amplification and gold nanoparticle-based lateral flow biosensor visualization. FEMS Microbiol. Lett. 2018;365:fny147. doi: 10.1093/femsle/fny147. [DOI] [PubMed] [Google Scholar]
- 65.Kim T., Zhang Q., Li J., Zhang L., Jokerst J.V. A Gold/Silver Hybrid Nanoparticle for Treatment and Photoacoustic Imaging of Bacterial Infection. ACS Nano. 2018;12:5615–5625. doi: 10.1021/acsnano.8b01362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Cao Q., Liang S., Wang L., Cao J., Liu M., Li S., Cao X., Guo Y. A Rapid Detection of Haemophilus influenzae Using Multiple Cross Displacement Amplification Linked with Nanoparticle-Based Lateral Flow Biosensor. Front. Cell Infect. Microbiol. 2021;11:721547. doi: 10.3389/fcimb.2021.721547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Hong D., Kim K., Jo E.J., Kim M.G. Electrochemiluminescence-Incorporated Lateral Flow Immunosensors Using Ru(bpy)3(2+)-Labeled Gold Nanoparticles for the Full-Range Detection of Physiological C-Reactive Protein Levels. Anal. Chem. 2021;93:7925–7932. doi: 10.1021/acs.analchem.1c00623. [DOI] [PubMed] [Google Scholar]
- 68.Chen X., Ma K., Yi X., Xiong L., Wang Y., Li S. The rapid and visual detection of methicillin-susceptible and methicillin-resistant Staphylococcus aureus using multiplex loop-mediated isothermal amplification linked to a nanoparticle-based lateral flow biosensor. Antimicrob. Resist. Infect. Control. 2020;9:111. doi: 10.1186/s13756-020-00774-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Chen X., Ma K., Yi X., Xiao Z., Xiong L., Wang Y., Li S. A Novel Detection of Enterococcus faecalis Using Multiple Cross Displacement Amplification Linked with Gold Nanoparticle Lateral Flow Biosensor. Infect. Drug Resist. 2019;12:3771–3781. doi: 10.2147/IDR.S235325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Li S., Liu C., Liu Y., Ma Q., Wang Y., Wang Y. Development of a multiple cross displacement amplification combined with nanoparticles-based biosensor assay to detect Neisseria meningitidis. Infect. Drug Resist. 2019;12:2077–2087. doi: 10.2147/IDR.S210735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Gupta S., Tiwari A., Jain U., Chauhan N. Synergistic effect of 2D material coated Pt nanoparticles with PEDOT polymer on electrode surface interface for a sensitive label free Helicobacter pylori CagA((Ag-Ab)) immunosensing. Mater. Sci. Eng. C Mater. Biol. Appl. 2019;103:109733. doi: 10.1016/j.msec.2019.05.018. [DOI] [PubMed] [Google Scholar]
- 72.Jain U., Gupta S., Soni S., Khurana M.P., Chauhan N. Triple-nanostructuring-based noninvasive electro-immune sensing of CagA toxin for Helicobacter pylori detection. Helicobacter. 2020;25:12706. doi: 10.1111/hel.12706. [DOI] [PubMed] [Google Scholar]
- 73.Cebula Z., Zoledowska S., Dziabowska K., Skwarecka M., Malinowska N., Bialobrzeska W., Czaczyk E., Siuzdak K., Sawczak M., Bogdanowicz R., et al. Detection of the Plant Pathogen Pseudomonas Syringae pv. Lachrymans on Antibody-Modified Gold Electrodes by Electrochemical Impedance Spectroscopy. Sensors. 2019;19:5411. doi: 10.3390/s19245411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Silva N.F.D., Almeida C.M.R., Magalhaes J., Goncalves M.P., Freire C., Delerue-Matos C. Development of a disposable paper-based potentiometric immunosensor for real-time detection of a foodborne pathogen. Biosens. Bioelectron. 2019;141:111317. doi: 10.1016/j.bios.2019.111317. [DOI] [PubMed] [Google Scholar]
- 75.Maldonado J., Gonzalez-Guerrero A.B., Dominguez C., Lechuga L.M. Label-free bimodal waveguide immunosensor for rapid diagnosis of bacterial infections in cirrhotic patients. Biosens. Bioelectron. 2016;85:310–316. doi: 10.1016/j.bios.2016.04.095. [DOI] [PubMed] [Google Scholar]
- 76.Lee C.W., Chang H.Y., Wu J.K., Tseng F.G. Ultra-sensitive electrochemical detection of bacteremia enabled by redox-active gold nanoparticles (raGNPs) in a nano-sieving microfluidic system (NS-MFS) Biosens. Bioelectron. 2019;133:215–222. doi: 10.1016/j.bios.2019.03.040. [DOI] [PubMed] [Google Scholar]
- 77.Narang R., Mohammadi S., Ashani M.M., Sadabadi H., Hejazi H., Zarifi M.H., Sanati-Nezhad A. Sensitive, Real-time and Non-Intrusive Detection of Concentration and Growth of Pathogenic Bacteria using Microfluidic-Microwave Ring Resonator Biosensor. Sci. Rep. 2018;8:15807. doi: 10.1038/s41598-018-34001-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Peng H., Borg R.E., Nguyen A.B.N., Chen I.A. Chimeric Phage Nanoparticles for Rapid Characterization of Bacterial Pathogens: Detection in Complex Biological Samples and Determination of Antibiotic Sensitivity. ACS Sens. 2020;5:1491–1499. doi: 10.1021/acssensors.0c00654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Park Y., Ryu B., Deng Q., Pan B., Song Y., Tian Y., Alam H.B., Li Y., Liang X., Kurabayashi K. An Integrated Plasmo-Photoelectronic Nanostructure Biosensor Detects an Infection Biomarker Accompanying Cell Death in Neutrophils. Small. 2020;16:1905611. doi: 10.1002/smll.201905611. [DOI] [PubMed] [Google Scholar]
- 80.Gong L., Liu E., Che J., Li J., Liu X., Xu H., Liang J. Multiple Cross Displacement Amplification Coupled with Gold Nanoparticles-Based Lateral Flow Biosensor for Detection of the Mobilized Colistin Resistance Gene mcr-1. Front. Cell Infect. Microbiol. 2019;9:226. doi: 10.3389/fcimb.2019.00226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Hicks J.M., Halkerston R., Silman N., Jackson S.K., Aylott J.W., Rawson F.J. Real-time bacterial detection with an intracellular ROS sensing platform. Biosens. Bioelectron. 2019;141:111430. doi: 10.1016/j.bios.2019.111430. [DOI] [PubMed] [Google Scholar]
- 82.Wu G., Ji H., Guo X., Li Y., Ren T., Dong H., Liu J., Liu Y., Shi X., He B. Nanoparticle reinforced bacterial outer-membrane vesicles effectively prevent fatal infection of carbapenem-resistant Klebsiella pneumoniae. Nanomedicine. 2020;24:102148. doi: 10.1016/j.nano.2019.102148. [DOI] [PubMed] [Google Scholar]
- 83.Kye Y.C., Park S.M., Shim B.S., Firdous J., Kim G., Kim H.W., Ju Y.J., Kim C.G., Cho C.S., Kim D.W., et al. Intranasal immunization with pneumococcal surface protein A in the presence of nanoparticle forming polysorbitol transporter adjuvant induces protective immunity against the Streptococcus pneumoniae infection. Acta Biomater. 2019;90:362–372. doi: 10.1016/j.actbio.2019.03.049. [DOI] [PubMed] [Google Scholar]
- 84.Gao F., Xu L., Yang B., Fan F., Yang L. Kill the Real with the Fake: Eliminate Intracellular Staphylococcus aureus Using Nanoparticle Coated with Its Extracellular Vesicle Membrane as Active-Targeting Drug Carrier. ACS Infect. Dis. 2019;5:218–227. doi: 10.1021/acsinfecdis.8b00212. [DOI] [PubMed] [Google Scholar]
- 85.Flemming H.C., Wingender J., Szewzyk U., Steinberg P., Rice S.A., Kjelleberg S. Biofilms: An emergent form of bacterial life. Nat. Rev. Microbiol. 2016;14:563–575. doi: 10.1038/nrmicro.2016.94. [DOI] [PubMed] [Google Scholar]
- 86.Pham D.T.N., Khan F., Phan T.T.V., Park S.K., Manivasagan P., Oh J., Kim Y.M. Biofilm inhibition, modulation of virulence and motility properties by FeOOH nanoparticle in Pseudomonas aeruginosa. Braz. J. Microbiol. 2019;50:791–805. doi: 10.1007/s42770-019-00108-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Darabpour E., Doroodmand M.M., Halabian R., Imani Fooladi A.A. Sulfur-Functionalized Fullerene Nanoparticle as an Inhibitor and Eliminator Agent on Pseudomonas aeruginosa Biofilm and Expression of toxA Gene. Microb. Drug Resist. 2019;25:594–602. doi: 10.1089/mdr.2018.0008. [DOI] [PubMed] [Google Scholar]
- 88.Shakerimoghaddam A., Ghaemi E.A., Jamalli A. Zinc oxide nanoparticle reduced biofilm formation and antigen 43 expressions in uropathogenic Escherichia coli. Iran. J. Basic Med. Sci. 2017;20:451–456. doi: 10.22038/IJBMS.2017.8589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ballo M.K., Rtimi S., Pulgarin C., Hopf N., Berthet A., Kiwi J., Moreillon P., Entenza J.M., Bizzini A. In Vitro and In Vivo Effectiveness of an Innovative Silver-Copper Nanoparticle Coating of Catheters to Prevent Methicillin-Resistant Staphylococcus aureus Infection. Antimicrob. Agents Chemother. 2016;60:5349–5356. doi: 10.1128/AAC.00959-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Luan Y., van der Mei H.C., Dijk M., Geertsema-Doornbusch G.I., Atema-Smit J., Ren Y., Chen H., Busscher H.J. Polarization of Macrophages, Cellular Adhesion, and Spreading on Bacterially Contaminated Gold Nanoparticle—Coatings in Vitro. ACS Biomater. Sci. Eng. 2020;6:933–945. doi: 10.1021/acsbiomaterials.9b01518. [DOI] [PubMed] [Google Scholar]
- 91.Zaat S., Broekhuizen C., Riool M. Host tissue as a niche for biomaterial-associated infection. Future Microbiol. 2010;5:1149–1151. doi: 10.2217/fmb.10.89. [DOI] [PubMed] [Google Scholar]
- 92.Ponnuvel S., Sankar S., Ponnuraj K. Analyzing the adhesion mechanism of FnBPA, a surface adhesin from Staphylococcus aureus on its interaction with nanoparticle. Microb. Pathog. 2020;146:104239. doi: 10.1016/j.micpath.2020.104239. [DOI] [PubMed] [Google Scholar]
- 93.Liu X., Chen H.H., Lin Y.C., Nabilla S.C., Liu W.C., Wang W.C., Shih S.J., Li Y., Lin C.P., Zhao G., et al. Composite Polyelectrolyte Multilayer and Mesoporous Bioactive Glass Nanoparticle Coating on 316L Stainless Steel for Controlled Antibiotic Release and Biocompatibility. J. Biomed. Nanotechnol. 2018;14:725–735. doi: 10.1166/jbn.2018.2531. [DOI] [PubMed] [Google Scholar]
- 94.Permana A.D., Mir M., Utomo E., Donnelly R.F. Bacterially sensitive nanoparticle-based dissolving microneedles of doxycycline for enhanced treatment of bacterial biofilm skin infection: A proof of concept study. Int. J. Pharm. X. 2020;2:100047. doi: 10.1016/j.ijpx.2020.100047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Peng D., Liu G., He Y., Gao P., Gou S., Wu J., Yu J., Liu P., Cai K. Fabrication of a pH-responsive core-shell nanosystem with a low-temperature photothermal therapy effect for treating bacterial biofilm infection. Biomater. Sci. 2021 doi: 10.1039/D1BM01329G. in press. [DOI] [PubMed] [Google Scholar]
- 96.Karunanayake L.I., Waniganayake Y.C., Nirmala Gunawardena K.D., Danuka Padmaraja S.A., Peter D., Jayasekera R., Karunanayake P. Use of silicon nanoparticle surface coating in infection control: Experience in a tropical healthcare setting. Infect. Dis. Health. 2019;24:201–207. doi: 10.1016/j.idh.2019.06.006. [DOI] [PubMed] [Google Scholar]
- 97.Mohamed S.A., Samir T.M., Helmy O.M., Elhosseiny N.M., Ali A.A., El-Kholy A.A., Attia A.S. A Novel Surface-Exposed Polypeptide Is Successfully Employed as a Target for Developing a Prototype One-Step Immunochromatographic Strip for Specific and Sensitive Direct Detection of Staphylococcus aureus Causing Neonatal Sepsis. Biomolecules. 2020;10:1580. doi: 10.3390/biom10111580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Rahmati F., Hosseini S.S., Mahuti Safai S., Asgari Lajayer B., Hatami M. New insights into the role of nanotechnology in microbial food safety. 3 Biotech. 2020;10:425. doi: 10.1007/s13205-020-02409-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Brunetti G., Conteduca D., Armenise M.N., Ciminelli C. Novel Micro-Nano Optoelectronic Biosensor for Label-Free Real-Time Biofilm Monitoring. Biosensors. 2021;11:361. doi: 10.3390/bios11100361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Wu S., Weir M.D., Lei L., Liu J., Xu H.H.K. Novel nanographene oxide-calcium phosphate cement inhibits Enterococcus faecalis biofilm and supports dental pulp stem cells. J. Orthop. Surg. Res. 2021;16:580. doi: 10.1186/s13018-021-02736-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Long E.G., Buluk M., Gallagher M.B., Schneider J.M., Brown J.L. Human mesenchymal stem cell morphology, migration, and differentiation on micro and nano-textured titanium. Bioact. Mater. 2019;4:249–255. doi: 10.1016/j.bioactmat.2019.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Yan J., Xia D., Zhou W., Li Y., Xiong P., Li Q., Wang P., Li M., Zheng Y., Cheng Y. pH-responsive silk fibroin-based CuO/Ag micro/nano coating endows polyetheretherketone with synergistic antibacterial ability, osteogenesis, and angiogenesis. Acta Biomater. 2020;115:220–234. doi: 10.1016/j.actbio.2020.07.062. [DOI] [PubMed] [Google Scholar]
- 103.Bari A., Bloise N., Fiorilli S., Novajra G., Vallet-Regi M., Bruni G., Torres-Pardo A., Gonzalez-Calbet J.M., Visai L., Vitale-Brovarone C. Copper-containing mesoporous bioactive glass nanoparticles as multifunctional agent for bone regeneration. Acta Biomater. 2017;55:493–504. doi: 10.1016/j.actbio.2017.04.012. [DOI] [PubMed] [Google Scholar]
- 104.Cunha A.P., Henriques R., Cardoso S., Freitas P.P., Carvalho C.M. Rapid and multiplex detection of nosocomial pathogens on a phage-based magnetoresistive lab-on-chip platform. Biotechnol. Bioeng. 2021;118:3164–3174. doi: 10.1002/bit.27841. [DOI] [PubMed] [Google Scholar]
- 105.Abdou Mohamed M.A., Kozlowski H.N., Kim J., Zagorovsky K., Kantor M., Feld J.J., Mubareka S., Mazzulli T., Chan W.C.W. Diagnosing Antibiotic Resistance Using Nucleic Acid Enzymes and Gold Nanoparticles. ACS Nano. 2021;15:9379–9390. doi: 10.1021/acsnano.0c09902. [DOI] [PubMed] [Google Scholar]
- 106.Khaled J.M., Alharbi N.S., Siddiqi M.Z., Alobaidi A.S., Nauman K., Alahmedi S., Almazyed A.O., Almosallam M.A., Al Jurayyan A.N. A synergic action of colistin, imipenem, and silver nanoparticles against pandrug-resistant Acinetobacter baumannii isolated from patients. J. Infect. Public Health. 2021;14:1679–1685. doi: 10.1016/j.jiph.2021.09.015. [DOI] [PubMed] [Google Scholar]
- 107.Khalil M.A., El Maghraby G.M., Sonbol F.I., Allam N.G., Ateya P.S., Ali S.S. Enhanced Efficacy of Some Antibiotics in Presence of Silver Nanoparticles Against Multidrug Resistant Pseudomonas aeruginosa Recovered from Burn Wound Infections. Front. Microbiol. 2021;12:648560. doi: 10.3389/fmicb.2021.648560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Zhang M., Wang D., Ji N., Lee S., Wang G., Zheng Y., Zhang X., Yang L., Qin Z., Yang Y. Bioinspired Design of Sericin/Chitosan/Ag@MOF/GO Hydrogels for Efficiently Combating Resistant Bacteria, Rapid Hemostasis, and Wound Healing. Polymers. 2021;13:2812. doi: 10.3390/polym13162812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Li X., Gui R., Li J., Huang R., Shang Y., Zhao Q., Liu H., Jiang H., Shang X., Wu X., et al. Novel Multifunctional Silver Nanocomposite Serves as a Resistance-Reversal Agent to Synergistically Combat Carbapenem-Resistant Acinetobacter baumannii. ACS Appl. Mater. Interfaces. 2021;13:30434–30457. doi: 10.1021/acsami.1c10309. [DOI] [PubMed] [Google Scholar]
- 110.Principi N., Silvestri E., Esposito S. Advantages and Limitations of Bacteriophages for the Treatment of Bacterial Infections. Front. Pharmacol. 2019;10:513. doi: 10.3389/fphar.2019.00513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Kaur S., Kumari A., Kumari Negi A., Galav V., Thakur S., Agrawal M., Sharma V. Nanotechnology Based Approaches in Phage Therapy: Overcoming the Pharmacological Barriers. Front. Pharmacol. 2021;12:699054. doi: 10.3389/fphar.2021.699054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Yang X., Ye W., Qi Y., Ying Y., Xia Z. Overcoming Multidrug Resistance in Bacteria Through Antibiotics Delivery in Surface-Engineered Nano-Cargos: Recent Developments for Future Nano-Antibiotics. Front. Bioeng. Biotechnol. 2021;9:696514. doi: 10.3389/fbioe.2021.696514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Yu S., Pu X., Uddin Ahmed M., Yu H.H., Tejasvi Mutukuri T., Li J., Tony Zhou Q. Spray-freeze-dried inhalable composite microparticles containing nanoparticles of combinational drugs for potential treatment of lung infections caused by Pseudomonas aeruginosa. Int. J. Pharm. 2021:121160. doi: 10.1016/j.ijpharm.2021.121160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.El-Sheikh S.M.A., Abd El-Alim A.E.F., Ibrahim H.A., Mobarez E.A., El-Sayed W.A., Galal A.A.A., Awad N.F.S. Chitosan propolis nanocomposite alone or in combination with apramycin: An alternative therapy for multidrug-resistant Salmonella Typhimurium in rabbits: In vitro and in vivo study. J. Med. Microbiol. 2021;70:1412. doi: 10.1099/jmm.0.001412. [DOI] [PubMed] [Google Scholar]
- 115.Jacqueline C., Caillon J., Meyer O., Dailly E., Simonsson C., Leanerts V., Asehnoune K., Reghal A., Potel G. Efficacy of Nano-Encapsulated Daptomycin in an Experimental Methicillin-Resistant Staphylococcus aureus (MRSA) Bone and Joint Infection Model. Antimicrob. Agents Chemother. 2021 doi: 10.1128/AAC.00768-21. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Wang Z., Liu X., Duan Y., Huang Y. Nanoparticle-Hydrogel Systems Containing Platensimycin for Local Treatment of Methicillin—Resistant Staphylococcus aureus Infection. Mol. Pharm. 2021;18:4099–4110. doi: 10.1021/acs.molpharmaceut.1c00523. [DOI] [PubMed] [Google Scholar]
- 117.Miao J., Lin F., Huang N., Teng Y. Improving Anti-Inflammatory Effect of Luteolin with Nano-Micelles in the Bacteria-Induced Lung Infection. J. Biomed. Nanotechnol. 2021;17:1229–1241. doi: 10.1166/jbn.2021.3101. [DOI] [PubMed] [Google Scholar]
- 118.Edson J.A., Chu W., Porwollik S., Tran K., Iribe N., McClelland M., Kwon Y.J. Eradication of Intracellular Salmonella typhimurium by Polyplexes of Acid-Transforming Chitosan and Fragment DNA. Macromol. Biosci. 2021;21:2000408. doi: 10.1002/mabi.202000408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Khare T., Mahalunkar S., Shriram V., Gosavi S., Kumar V. Embelin-loaded chitosan gold nanoparticles interact synergistically with ciprofloxacin by inhibiting efflux pumps in multidrug-resistant Pseudomonas aeruginosa and Escherichia coli. Environ. Res. 2021;199:111321. doi: 10.1016/j.envres.2021.111321. [DOI] [PubMed] [Google Scholar]
- 120.Zhang J., Sun H., Gao C., Wang Y., Cheng X., Yang Y., Gou Q., Lei L., Chen Y., Wang X., et al. Development of a chitosan-modified PLGA nanoparticle vaccine for protection against Escherichia coli K1 caused meningitis in mice. J. Nanobiotechnol. 2021;19:69. doi: 10.1186/s12951-021-00812-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Gregory A.E., Judy B.M., Qazi O., Blumentritt C.A., Brown K.A., Shaw A.M., Torres A.G., Titball R.W. A gold nanoparticle-linked glycoconjugate vaccine against Burkholderia mallei. Nanomedicine. 2015;11:447–456. doi: 10.1016/j.nano.2014.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Tiwari V., Tiwari M., Solanki V. Polyvinylpyrrolidone-Capped Silver Nanoparticle Inhibits Infection of Carbapenem-Resistant Strain of Acinetobacter baumannii in the Human Pulmonary Epithelial Cell. Front. Immunol. 2017;8:973. doi: 10.3389/fimmu.2017.00973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Kwon E.J., Skalak M., Bertucci A., Braun G., Ricci F., Ruoslahti E., Sailor M.J., Bhatia S.N. Porous Silicon Nanoparticle Delivery of Tandem Peptide Anti-Infectives for the Treatment of Pseudomonas aeruginosa Lung Infections. Adv. Mater. 2017;29 doi: 10.1002/adma.201701527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Camacho A.I., Irache J.M., de Souza J., Sanchez-Gomez S., Gamazo C. Nanoparticle-based vaccine for mucosal protection against Shigella flexneri in mice. Vaccine. 2013;31:3288–3294. doi: 10.1016/j.vaccine.2013.05.020. [DOI] [PubMed] [Google Scholar]
- 125.Lee J.A., Jung B.G., Kim T.H., Kim Y.M., Park M.H., Hyun P.M., Jeon J.W., Park J.K., Cho C.W., Suh G.H., et al. Poly d,l-lactide-co-glycolide (PLGA) nanoparticle-encapsulated honeybee (Apis melifera) venom promotes clearance of Salmonella enterica serovar Typhimurium infection in experimentally challenged pigs through the up-regulation of T helper type 1 specific immune responses. Vet. Immunol. Immunopathol. 2014;161:193–204. doi: 10.1016/j.vetimm.2014.08.010. [DOI] [PubMed] [Google Scholar]
- 126.Chowdhury R., Ilyas H., Ghosh A., Ali H., Ghorai A., Midya A., Jana N.R., Das S., Bhunia A. Multivalent gold nanoparticle-peptide conjugates for targeting intracellular bacterial infections. Nanoscale. 2017;9:14074–14093. doi: 10.1039/C7NR04062H. [DOI] [PubMed] [Google Scholar]
- 127.Luo M., Jia Y.Y., Jing Z.W., Li C., Zhou S.Y., Mei Q.B., Zhang B.L. Construction and optimization of pH-sensitive nanoparticle delivery system containing PLGA and UCCs-2 for targeted treatment of Helicobacter pylori. Colloids Surf. B Biointerfaces. 2018;164:11–19. doi: 10.1016/j.colsurfb.2018.01.008. [DOI] [PubMed] [Google Scholar]
- 128.Das S., Angsantikul P., Le C., Bao D., Miyamoto Y., Gao W., Zhang L., Eckmann L. Neutralization of cholera toxin with nanoparticle decoys for treatment of cholera. PLoS Negl. Trop. Dis. 2018;12:0006266. doi: 10.1371/journal.pntd.0006266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Tao J., Zhang Y., Shen A., Yang Y., Diao L., Wang L., Cai D., Hu Y. Injectable Chitosan-Based Thermosensitive Hydrogel/Nanoparticle-Loaded System for Local Delivery of Vancomycin in the Treatment of Osteomyelitis. Int. J. Nanomed. 2020;15:5855–5871. doi: 10.2147/IJN.S247088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Abdulrehman T., Qadri S., Skariah S., Sultan A., Mansour S., Azzi J., Haik Y. Boron doped silver-copper alloy nanoparticle targeting intracellular S. aureus in bone cells. PLoS ONE. 2020;15:0231276. doi: 10.1371/journal.pone.0231276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Gowri M., Latha N., Suganya K., Murugan M., Rajan M. Calcium alginate nanoparticle crosslinked phosphorylated polyallylamine to the controlled release of clindamycin for osteomyelitis treatment. Drug Dev. Ind. Pharm. 2021;47:280–291. doi: 10.1080/03639045.2021.1879835. [DOI] [PubMed] [Google Scholar]
- 132.Wang F., Fang R.H., Luk B.T., Hu C.J., Thamphiwatana S., Dehaini D., Angsantikul P., Kroll A.V., Pang Z., Gao W., et al. Nanoparticle-Based Antivirulence Vaccine for the Management of Methicillin-Resistant Staphylococcus aureus Skin Infection. Adv. Funct. Mater. 2016;26:1628–1635. doi: 10.1002/adfm.201505231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Escajadillo T., Olson J., Luk B.T., Zhang L., Nizet V. A Red Blood Cell Membrane-Camouflaged Nanoparticle Counteracts Streptolysin O-Mediated Virulence Phenotypes of Invasive Group A Streptococcus. Front. Pharmacol. 2017;8:477. doi: 10.3389/fphar.2017.00477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Lee B., Park J., Ryu M., Kim S., Joo M., Yeom J.H., Kim S., Park Y., Lee K., Bae J. Antimicrobial peptide-loaded gold nanoparticle-DNA aptamer conjugates as highly effective antibacterial therapeutics against Vibrio vulnificus. Sci. Rep. 2017;7:13572. doi: 10.1038/s41598-017-14127-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Chang M.C., Kuo Y.J., Hung K.H., Peng C.L., Chen K.Y., Yeh L.K. Liposomal dexamethasone-moxifloxacin nanoparticle combinations with collagen/gelatin/alginate hydrogel for corneal infection treatment and wound healing. Biomed. Mater. 2020;15:055022. doi: 10.1088/1748-605X/ab9510. [DOI] [PubMed] [Google Scholar]
- 136.Chen P., Wu Z., Leung A., Chen X., Landao-Bassonga E., Gao J., Chen L., Zheng M., Yao F., Yang H., et al. Fabrication of a silver nanoparticle-coated collagen membrane with anti-bacterial and anti-inflammatory activities for guided bone regeneration. Biomed. Mater. 2018;13:065014. doi: 10.1088/1748-605X/aae15b. [DOI] [PubMed] [Google Scholar]
- 137.Wang L., Xing D., Le Van A., Jerse A.E., Wang S. Structure-based design of ferritin nanoparticle immunogens displaying antigenic loops of Neisseria gonorrhoeae. FEBS Open Bio. 2017;7:1196–1207. doi: 10.1002/2211-5463.12267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Li X., Vemireddy V., Cai Q., Xiong H., Kang P., Li X., Giannotta M., Hayenga H.N., Pan E., Sirsi S.R., et al. Reversibly Modulating the Blood-Brain Barrier by Laser Stimulation of Molecular-Targeted Nanoparticles. Nano Lett. 2021 doi: 10.1021/acs.nanolett.1c02996. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Tamil Selvan S., Padmanabhan P., Zoltan Gulyas B. Nanotechnology-Based Diagnostics and Therapy for Pathogen-Related Infections in the CNS. ACS Chem. Neurosci. 2020;11:2371–2377. doi: 10.1021/acschemneuro.9b00470. [DOI] [PubMed] [Google Scholar]
- 140.Lashkari A., Ranjbar R. Nanoparticles and nanoformulated drugs as promising delivery system in treatment of microbial-induced CNS infection: A systematic review of literature. J. Neurovirol. 2021;27:542–549. doi: 10.1007/s13365-021-00994-w. [DOI] [PubMed] [Google Scholar]
- 141.Nakamura T., Cohen A.L., Schwartz S., Mwenda J.M., Weldegebriel G., Biey J.N.M., Katsande R., Ghoniem A., Fahmy K., Rahman H.A., et al. The Global Landscape of Pediatric Bacterial Meningitis Data Reported to the World Health Organization-Coordinated Invasive Bacterial Vaccine-Preventable Disease Surveillance Network, 2014–2019. J. Infect. Dis. 2021;224:S161–S173. doi: 10.1093/infdis/jiab217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Horseman M.A., Surani S. A comprehensive review of Vibrio vulnificus: An important cause of severe sepsis and skin and soft-tissue infection. Int. J. Infect. Dis. 2011;15:e157–e166. doi: 10.1016/j.ijid.2010.11.003. [DOI] [PubMed] [Google Scholar]
- 143.Momin M.M., Afreen S.D. Nanoformulations and Highlights of Clinical Studies for Ocular Drug Delivery Systems: An Overview. Crit. Rev. Ther. Drug Carr. Syst. 2021;38:79–107. doi: 10.1615/CritRevTherDrugCarrierSyst.2021035767. [DOI] [PubMed] [Google Scholar]
- 144.Zhu Z., Jin L., Yu F., Wang F., Weng Z., Liu J., Han Z., Wang X. ZnO/CPAN Modified Contact Lens with Antibacterial and Harmful Light Reduction Capabilities. Adv. Healthc. Mater. 2021;10:2100259. doi: 10.1002/adhm.202100259. [DOI] [PubMed] [Google Scholar]
- 145.Yang J.W., Choi J.W., Lee S.G., Kim D.S. Antibacterial properties of artificial eyes containing nano-sized particle silver. Orbit. 2011;30:77–81. doi: 10.3109/01676830.2010.538123. [DOI] [PubMed] [Google Scholar]
- 146.Sharma R., Sharma D., Hazlett L.D., Singh N.K. Nano-Biomaterials for Retinal Regeneration. Nanomaterials. 2021;11:1880. doi: 10.3390/nano11081880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Makvandi P., Josic U., Delfi M., Pinelli F., Jahed V., Kaya E., Ashrafizadeh M., Zarepour A., Rossi F., Zarrabi A., et al. Drug Delivery (Nano)Platforms for Oral and Dental Applications: Tissue Regeneration, Infection Control, and Cancer Management. Adv. Sci. 2021;8:2004014. doi: 10.1002/advs.202004014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Li Z., Xie K., Yang S., Yu T., Xiao Y., Zhou Y. Multifunctional Ca-Zn-Si-based micro-nano spheres with anti-infective, anti-inflammatory, and dentin regenerative properties for pulp capping application. J. Mater. Chem. B. 2021;9:8289–8299. doi: 10.1039/D1TB01517F. [DOI] [PubMed] [Google Scholar]
- 149.Pandey M., Choudhury H., Abdul-Aziz A., Bhattamisra S.K., Gorain B., Carine T., Wee Toong T., Yi N.J., Win Yi L. Promising Drug Delivery Approaches to Treat Microbial Infections in the Vagina: A Recent Update. Polymers. 2020;13:26. doi: 10.3390/polym13010026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Blanco E., Shen H., Ferrari M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 2015;33:941–951. doi: 10.1038/nbt.3330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Immordino M.L., Dosio F., Cattel L. Stealth liposomes: Review of the basic science, rationale, and clinical applications, existing and potential. Int. J. Nanomed. 2006;1:297–315. [PMC free article] [PubMed] [Google Scholar]
- 152.Gupta A.K., Wells S. Surface-modified superparamagnetic nanoparticles for drug delivery: Preparation, characterization, and cytotoxicity studies. IEEE Trans. Nanobiosci. 2004;3:66–73. doi: 10.1109/TNB.2003.820277. [DOI] [PubMed] [Google Scholar]
- 153.Hennig R., Pollinger K., Veser A., Breunig M., Goepferich A. Nanoparticle multivalency counterbalances the ligand affinity loss upon PEGylation. J. Control. Release. 2014;194:20–27. doi: 10.1016/j.jconrel.2014.07.062. [DOI] [PubMed] [Google Scholar]
- 154.Adiseshaiah P.P., Hall J.B., McNeil S.E. Nanomaterial standards for efficacy and toxicity assessment. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2010;2:99–112. doi: 10.1002/wnan.66. [DOI] [PubMed] [Google Scholar]
- 155.Judzewitsch P.R., Nguyen T.K., Shanmugam S., Wong E.H.H., Boyer C. Towards Sequence-Controlled Antimicrobial Polymers: Effect of Polymer Block Order on Antimicrobial Activity. Angew. Chem. Int. Ed. 2018;57:4559–4564. doi: 10.1002/anie.201713036. [DOI] [PubMed] [Google Scholar]
- 156.Bing W., Sun H., Wang F., Song Y., Ren J. Hydrogen-producing hyperthermophilic bacteria synthesized size-controllable fine gold nanoparticles with excellence for eradicating biofilm and antibacterial applications. J. Mater. Chem. B. 2018;6:4602–4609. doi: 10.1039/C8TB00549D. [DOI] [PubMed] [Google Scholar]
- 157.Zhang L., Su W., Huang Y., Li H., Fu L., Song K., Huang X., Yu J., Lin C.T. In Situ High-Pressure X-ray Diffraction and Raman Spectroscopy Study of Ti3C2Tx MXene. Nanoscale Res. Lett. 2018;13:343. doi: 10.1186/s11671-018-2746-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Shute T., Amiel E., Alam N., Yates J.L., Mohrs K., Dudley E., Salas B., Mesa C., Serrata A., Angel D., et al. Glycolipid-Containing Nanoparticle Vaccine Engages Invariant NKT Cells to Enhance Humoral Protection against Systemic Bacterial Infection but Abrogates T-Independent Vaccine Responses. J. Immunol. 2021;206:1806–1816. doi: 10.4049/jimmunol.2001283. [DOI] [PubMed] [Google Scholar]
- 159.Estes L.M., Singha P., Singh S., Sakthivel T.S., Garren M., Devine R., Brisbois E.J., Seal S., Handa H. Characterization of a nitric oxide (NO) donor molecule and cerium oxide nanoparticle (CNP) interactions and their synergistic antimicrobial potential for biomedical applications. J. Colloid Interface Sci. 2021;586:163–177. doi: 10.1016/j.jcis.2020.10.081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Alumutairi L., Yu B., Filka M., Nayfach J., Kim M.H. Mild magnetic nanoparticle hyperthermia enhances the susceptibility of Staphylococcus aureus biofilm to antibiotics. Int. J. Hyperth. 2020;37:66–75. doi: 10.1080/02656736.2019.1707886. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.