Graphical abstract
Keywords: Coronavirus, Fatigue, Sequelae, Post-infection, Chronic fatigue syndrome, Myalgic encephalomyelitis, Post COVID-19 condition, SARS-CoV-2
Highlights
-
•
Serious side effects and post-infection sequelae have been reported in SARS-CoV-2-infected patients after acute disease phase.
-
•
Long-COVID patient characteristics and symptoms were compared to myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS).
-
•
The onset, progression, and symptom profile of long-COVID patients have considerable overlap with ME/CFS.
-
•
Longitudinal monitoring of COVID-19 patients is warranted to understand the virus long-term effects.
Abstract
Background
The severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) disease (COVID-19) triggers the development of numerous pathologies and infection-linked complications and exacerbates existing pathologies in nearly all body systems. Aside from the primarily targeted respiratory organs, adverse SARS-CoV-2 effects were observed in nervous, cardiovascular, gastrointestinal/metabolic, immune, and other systems in COVID-19 survivors. Long-term effects of this viral infection have been recently observed and represent distressing sequelae recognised by the World Health Organisation (WHO) as a distinct clinical entity defined as post-COVID-19 condition. Considering the pandemic is still ongoing, more time is required to confirm post COVID-19 condition diagnosis in the COVID-19 infected cohorts, although many reported post COVID-19 symptoms overlap with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS).
Aims of Review
In this study, COVID-19 clinical presentation and associated post-infection sequelae (post-COVID-19 condition) were reviewed and compared with ME/CFS symptomatology.
Key Scientific Concepts of Review
The onset, progression, and symptom profile of post COVID-19 condition patients have considerable overlap with ME/CFS. Considering the large scope and range of pro-inflammatory effects of this virus, it is reasonable to expect development of post COVID-19 clinical complications in a proportion of the affected population. There are reports of a later debilitating syndrome onset three months post COVID-19 infection (often described as long-COVID-19), marked by the presence of fatigue, headache, cognitive dysfunction, post-exertional malaise, orthostatic intolerance, and dyspnoea. Acute inflammation, oxidative stress, and increased levels of interleukin-6 (IL-6) and tumor necrosis factor α (TNFα), have been reported in SARS-CoV-2 infected patients. Longitudinal monitoring of post COVID-19 patients is warranted to understand the long-term effects of SARS-CoV-2 infection and the pathomechanism of post COVID-19 condition.
Introduction
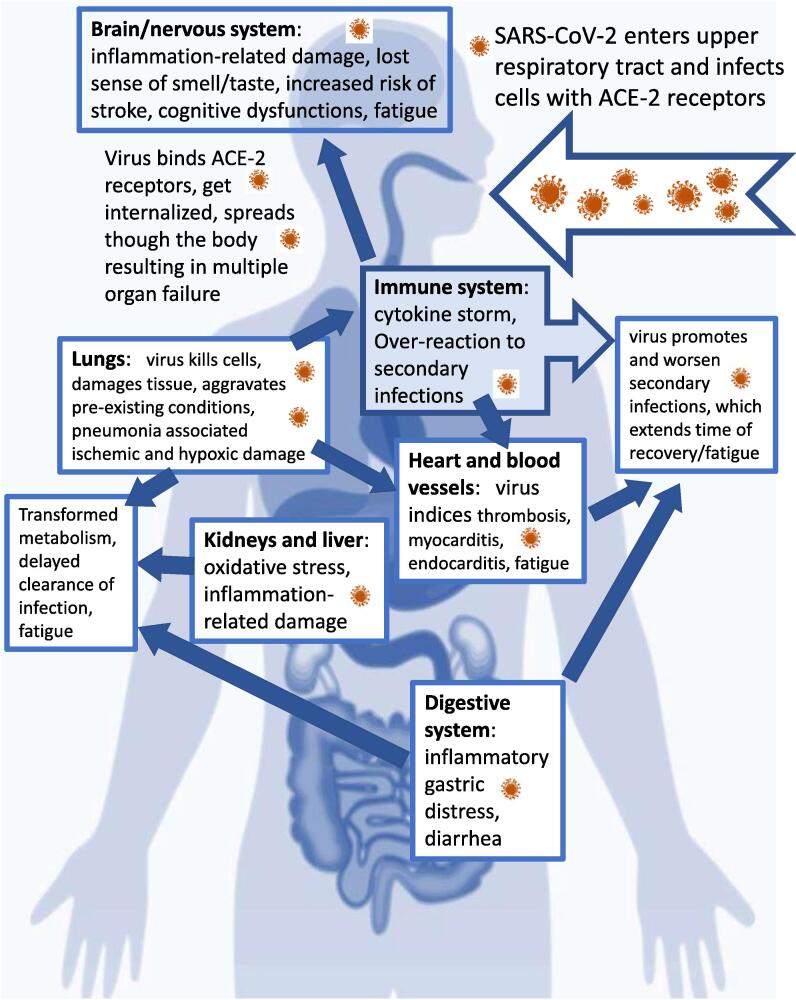
The severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), defined as coronavirus disease of 2019 (COVID-19), has caused a viral pandemic emergency of international concern. Public health services registered over 249 million SARS-CoV-2 infections and nearly five million associated deaths worldwide on 25 October 2021 [1]. The majority of those infected with COVID-19 demonstrate mild flu-like symptoms. However, a subset of COVID-19 patients developed severe pathology including bilateral interstitial pneumonitis and acute respiratory distress syndrome (ARDS), with a mortality rate of over 50% [2], [3]. Although the virus primarily affects the respiratory organs [2], SARS-CoV-2 pathogens were shown to impact metabolism and multiple organs and systems, including the cardiovascular [3], [4], [5], gastrointestinal [4], and nervous systems [6], [7] (Fig. 1). The proportion of the affected population is constantly growing as the pandemic is ongoing and is accompanied by large variety of symptoms with various duration. The number of patients with serious post-COVID complications is of great concern. For instance, persistence of dyspnoea and neuropsychological symptoms were registered in 35% of non-hospitalized and 87% of hospitalized COVID-19 patients [5], [6].
Fig. 1.
SARS-CoV-2 induces multiple organ failure. Viral particles and associated cytokines (“cytokine storm”) provoke development of long-lasting complications, including chronic fatigue.
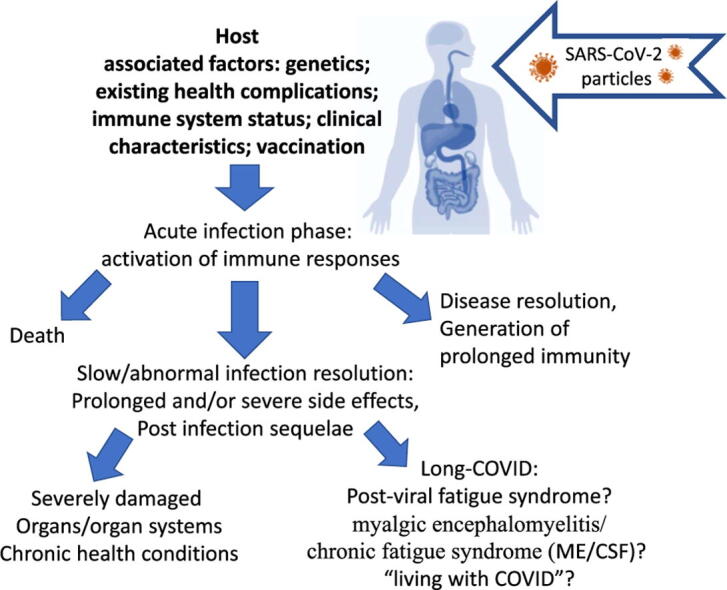
Recent reports indicated that several months after acute SARS-CoV-2 infection, some patients were still experiencing severe fatigue and other systemic symptoms described as long-COVID [7], [8]. Symptoms that persist for longer than three weeks have been classified as “post-acute COVID-19”, and those that persist beyond 12 weeks as “chronic COVID-19” [9]. The WHO guidelines now describe a post COVID-19 condition which occurs three months post-SARS-CoV-2 infection [10]. The onset is suggested to be independent of initial infection severity, pre-existing conditions, or age [11]. Some studies, however, have reported a greater amount of post-viral symptoms in more severe and critically ill patients [12]. A hospital-based study of 143 COVID-19 patients registered debilitating fatigue in 53% of survivors approximately two months after discharge from the hospital in Rome, Italy [6]. Another study in China observed that 16% of COVID-19 patients experienced weakening fatigue and 25% suffered aberrant lung function three months after discharge from the intensive care unit (ICU) [13]. A longitudinal study on non-hospitalized COVID-19 patients found that symptoms such as loss of smell, loss of taste, fatigue, or shortness of breath were most commonly reported four and seven months after SARS-CoV-2 infection [14]. Another report indicated that 1–3% of post COVID-19 condition patients were found to have symptoms that persisted longer than 12 weeks [11]. Larger controlled studies should confirm the observed symptoms and percentages mentioned above. However, the reported prolonged pathological post-viral condition may represent or closely relate to a range of diseases including myalgic encephalitis/chronic fatigue syndrome (ME/CFS) or other post-infection sequelae. Post-viral and multifactorial aetiology of ME/CFS revealed neuroimmune roots of the illness [15], which may be compared with post COVID-19 conditions.
This study will review current key signs and symptoms of post-COVID-19 condition, discuss SARS-CoV-2 pathological effects in nervous, cardiovascular, gastrointestinal/metabolic, immune, and other systems and potential overlap with ME/CFS, and suggest directions for relevant interventions in COVID-19 survivors. Considering that SARS-CoV-2 effects and host-related consequences are highly heterogenous, multiple mechanisms of post COVID-19 complications will be discussed with an accent in the most relevant ME/CFS-related targets.
Earlier and current evidence of SARS post-viral pathologies and associated symptoms: overlap with ME/CFS
Several studies of the first SARS pandemic (2003) indicated that 60% of study participants complained of unresolved health problems including fatigue after being infected with the coronavirus [16], [17], [18], [19]. When interviewed four years after the acute infection phase, 40% of the SARS (2003) infection survivors in Hong Kong complained of ongoing fatigue and other complications [17], [18]. SARS survivors reported diffuse muscle pain, various symptoms of depression, muscle weakness, and sleep-related abnormalities, defined as chronic post-SARS syndrome [19]. Post-SARS patients in Canada reported feeling unfit to work 1–3 years after infection due to various symptoms including persistent fatigue [19]. Other studies that monitored SARS survivors also reported a 40% rate of chronic fatigue development and associated social stigmatization [18], [20]. However, many facets of first SARS infection were not investigated. Only a few studies investigated the mechanism of post-infection sequelae resulting in inconclusive outcomes [13].
Regarding the current SARS-CoV-2 pandemic, the number of COVID-19 patients is growing, however, many have not completely recovered [5], [6], [7], [8], [11], [21]. Post COVID-19 condition follow-up studies were initiated primarily for those patients who were hospitalized with the most severe infection and spent on average two weeks at an intensive care unit (ICU). The follow-up data indicated serious consequences in a large number of the patients [6], [13], [22]. Persistent functional disabilities (including fatigue) long after ICU discharge were reported [23]. In a study conducted by Freitag et al, 45.2% of COVID-19 patients subsequently met symptom-specific ME/CFS criteria six months post-infection [24]. In a large study (across 56 countries) comprising 3762 COVID-19 patients with persisting symptoms, 56.8% of participants reported post-exertional malaise (PEM), one of the major ME/CFS symptoms [25]. Post COVID-19 condition has been recently described as having considerable overlap of symptoms with ME/CFS [26].
ME/CFS is a complex illness marked by persistent fatigue that is unrelieved by rest in combination with a range of other debilitating symptoms such as autonomic disturbances, endocrine dysfunction, and impaired cellular energy metabolism and ion transport [27]. Symptom presentation varies from patient to patient, ranging from mild to severe, with approximately 25% of ME/CFS patients being bedbound [28]. Many factors were shown to influence development of fatigue which was also linked to multiple intracellular mechanisms indicating a heterogeneity of this illness. Biochemical determinants of fatigue were often associated with immunopathogenesis and metabolic abnormalities [29]. Debilitating fatigue has often been marked by chronic muscle weakness and pain [30].
Viral and bacterial infections have been shown to trigger development of ME/CFS [20], [31]. Development of ME/CFS has been associated with Epstein–Barr virus (EBV) [32], [33], human herpesviruses (HHV-6, HHV-7, HHV-8) [34], [35], [36], cytomegalovirus (CMV) [37], human parvovirus B19 (B19V), lentivirus [33], and enterovirus infections [38]. The absence of targeted treatment and molecular-based diagnostic tools for this disease are attributed to the heterogeneity and unclear molecular mechanisms of ME/CFS. Diagnosis for this disease is derived using symptom-specific case criteria [27], [39], [40], including presence of post-exertional malaise (PEM) [41].
SARS-CoV-2: The mechanism of virus ACE receptor-mediated effects with links to ME/CFS
SARS-CoV-2 consists of a single linear RNA molecule which is ≈ 89 % identical to previously detected bat SARS-like-CoVZXC21 and ≈ 82 % identical to human SARS-CoV [42]. The virus belongs to the large family of coronaviruses and includes viral envelope-stabilizing components: S (spike; angiotensin converting enzyme 2 (ACE2)-interacting protein), E (envelope), M (membrane proteins), and N (nucleocapsid protein). Moreover, SARS-CoV-2 yields virulence factors (non-structural proteins) that can manipulate the host immune system and cell physiology [42], [43]. The mechanism of interactions between the host immune system and SARS-CoV-2 (pathogenesis) remains largely unclear. However, it has been documented that SARS-CoV-2 affects the host immune system resulting in a variety of clinical COVID-19 symptoms, ranging from asymptomatic to severe complications [44]. One of the confirmed COVID-19 systemic responses is a strong and disproportionate immune reaction to SARS-CoV-2 particles. The virus stimulates avalanche-like increases in pro-inflammatory cytokines, including IL-6 and TNFα (Table 1) [45], [46], [47]. Multiple recent studies reported that SARS-CoV-2 severity is associated with “cytokine storm”, the out-of-control cytokine cascades produced by the host immune system after the introduction of the viral particles [8], [48], [49].
Table 1.
Comparisons between ME/CFS and COVID-19.
| Author/year | COVID‐19 | Immunologic changes | ME/CFS | Author/year |
|---|---|---|---|---|
| [46], [77], [178], [201], [202], [43] | Lymphopenia (reduction of all lymphocyte subsets including CD4+ and CD8+ T-cells, NK, and B cells) has been associated with increased COVID-19 severity in < 20% of patients. COVID-19 non-survivors had significantly lower lymphocyte counts than survivors. Increased CD25 + T helper population and activation (severe COVID-19). Decreased dendritic cells (DCs) and increased level of dysfunctional monocytes resembling myeloid-derived suppressor cells (MDSCs) correlated with lymphopenia in severe COVID-19 patients. |
T cell count, populations, and responses | ME/CFS patients show several immunological changes suggestive of decreased cellular immunity. Mixed/complex T cell responses: larger proportions of effector memory CD8+ T-cells and decreased proportions of terminally differentiated effector CD8+ T cells was observed in ME-CSF patients. Severely affected ME/CFS patients demonstrated high proportion of mucosal associated invariant T cells (MAIT). |
[89], [203], [204] |
| [205], [206] | High CD25 + T cells with downregulated FOXP3 differentiate abnormally into hyperactivated T-cells in severe COVID-19 patients. Activated CD4+ T cell subsets, including Th1, Th2, and Th17-like cells were increased with higher clonally expanded in severe COVID-19 patients. |
CD4 + T cells | Infection was associated with chronically activated immune responses under control of CD4 + T-cell repertoire with increased density and percentages of Tregs in ME/CFS patients. | [89], [204] |
| [86], [205], [207], [208] | CD8+ T cells lymphopenia was associated with severe COVID-19 disease. CD56lowCD16+ NK cell numbers were increased. Dysregulated NK T cells responses. |
CD8+ T cells/ NK T cells | The proportions of CD3+, CD8+, CD8+/CD38 + and CD8 + HLA-DR + T cells were significantly higher in ME/CFS patients than controls. iNKT CD62L expression was significantly increased in moderate CFS patients. Naïve CD8 + T cells, CD8-/CD4- and CD56-/CD16- iNKT phenotypes, γδ2T cells and T memory subsets were significantly increased in severe ME/CFS patients at 6 months. However, reduced/dysregulated NK T cell functions were also reported in ME/CFS patients. Cytotoxic activity CD8+/CD56(bright) NK cells were significantly decreased in CFS/ME patients. Indirect evidence of diminished NK cell responsiveness to IL-12 and LT-α stimulus. |
[83], [89], [209], [210] |
| [81], [201], [211], [212] | Severe COVID-19 shows a systemic severe inflammatory response from mainly Th1 repertoire. Dysfunctional T cell differentiation with abnormal TNFα accumulation. However, in another study, Th1, Th2, and Th17 cell percentages were significantly lower in COVID-19 patients. Elevated Th17 (as well as Th1) responses or enhanced IL-17-related pathways were observed in SARS-CoV patients. |
Th1‐Th2 responses | CFS is dominated by Th1 cytokines network, consisting of cytokine nodes IL-1b, IL-4, IFN-γ and TNFα. However, the shift toward an allergic or Th2 pattern associated with autoimmunity was also observed in ME/CFS patients who also exhibited poorer sleep and high levels of basal salivary cortisol. Highly attenuated Th1 and Th17 immune responses were observed in ME/CFS. High Th2 marker expression but weak interaction patterns pointed to an established Th2 inflammatory milieu. Another study did not find any differences in the level of IL-17 between ME/CFS and control patients. |
[83], [213], [214], [215] |
| [216] | Decreased circulating eosinophil numbers in 50%‐80% of the hospitalized patients. Eosinopenia (52.9%) was observed in most severe COVID-19 patients. |
Eosinophils | Inconclusive findings: Eosinophils count was not significantly different to controls, but eosinophil count was significantly associated with indices of autonomic nervous activity, plasma cortisol, and blood monocyte in ME/CFS group. increase in CCL11 (eotaxin), a chemokine involved in eosinophil recruitment was detected in ME/CFS. |
[83], [92] |
| [76], [216] | IgM antibody appeared within a week post-disease onset, lasted for 1 month, and gradually decreased, whereas IgG antibody was produced 10 days after infection and lasted for a longer time (the exact time is unknown). However, in 44 moderate cases, 52 severe cases, and 37 critical cases with no differences in gender and age, the IgM and IgG antibodies concentrations were not found to be different. |
Specific antibody levels | Epstein-Barr virus (EBV) (a member of the herpes family) infection resulted in prolonged elevated IgM antibody level in a subset of ME/CFS patients. Higher levels of anti-human herpes virus (HHV)-6 IgM and IgG antibodies, and higher human cytomegalovirus (HCMV) IgM serum antibodies were present in ME/CFS patients. Accordingly, retrospective analysis in 300 ME/CFS patients indicated a decrease in IgG3 and 4, but an increase in IgG2 and IgM. |
[36], [217], [218] |
| [46], [96], [219], [220] | B cells were gradually decreased (loss of transitional and follicular B cells) with increased severity of illness. SARS-CoV-2 spike-specific neutralizing antibodies, memory B cells and circulating TFH cells may be lowered in COVID-19 non-survivors. Reduced numbers of in Bcl-6+germinal centres/B cells were detected post mortem in thoracic lymph nodes and spleens in acute SARS-CoV-2 cases. |
B cells | Dysregulated numbers of naïve, transitional, and memory B cells were reported in subsets of ME/CFS patients. Autoimmune B cells pathogenic responses following chronic or recurrent viral infections were seen in many patients with ME/CFS. Production of autoantibodies was linked to severe metabolic disturbances in ME/CFS. EBV was shown to infect B lymphocytes leading to their immortalisation with consequent weakening of cellular immunity. Increased percentage of CD24 + viable B cells was shown in ME/CFS blood samples in vitro. Gene set enrichment analyses suggested impairment of B cell differentiation and survival in ME/CFS group. |
[89], [92], [111], [221], [222], [223] |
| [46], [77], [211], [224], [225] | Innate and adaptive systems, monocyte-macrophages of COVID-19 patients produced massive amounts of cytokines and chemokines, including IFNg, TNF, CCL4, CCL5, IL-1α and IL-1β, neutrophil chemoattractant chemokine IL-8 (CXCL8). IL-6 concentrations were significantly higher in COVID-19 non-survivors. Accordingly, Tocilizumab (TCZ), a monoclonal antibody against interleukin-6 (IL-6) appears to be an effective treatment option in COVID-19 patients with a risk of cytokine storms. |
Cytokine storm | The increased levels of pro-inflammatory cytokines (IL-1, IL-4, IL-5, TNFα, IL-10, IFN-γ, IL-12, LTa,), CD4+/ CD25 + T cells, increased expression of FOXP3 and VPACR2, and activation of NF-κB were detected in ME/CFS patients. IL-4 median concentration was increased 3-fold in CFS. Increased IL-6, produced by EBV-infected B cells, together with depressed levels of IL-15 was suggested to interfere with LT-α and IL-12 activation of NK cells and the resulting decreased IFN-γ production. Another study also observed lower INFg production by CD4 + T cells from ME/CFS patients. A larger post-exercise increase in IL-10 and Toll-like receptor 4 (TLR4) gene transcripts in CFS was observed. The following cytokines were decreased in ME/CFS: IL-8, IL-13, and IL-15. |
[89], [94], [215], [226], [227], [228], [229] |
| [66], [230] | Significantly higher levels of D-dimer, C-reactive protein (CRP), and procalcitonin were associated with severe patients compared to non-severe patients. The levels of chloride and calcium in blood of COVID-19 patients were significantly lower than those of non-COVID-19 patients. The levels of lactate dehydrogenase (LDH) and potassium were significantly higher in COVID-19. However, another study found decreased levels of CRP in COVID-19 patients when we compared CRP levels among patients with endogenously elevated CRP. |
Acute‐phase reactants | The elevated level of high-sensitivity CRP (hsCRP) was found in ME/CFS patients. Patients with ME/CFS had significantly elevated level of TGF-β. |
[231], [232] |
| [125], [233], [234], [235] | Oxidative stress and hypoxia were observed in COVID-19 patients with severe pneumonia. Proinflammatory cytokines IL-6 and IL-1β release is triggered by activation of the Nod-like receptor family, pyrin domain-containing 3 (NLRP3) inflammasome pathway that is associated with oxidative burst in mitochondria. However, COVID-19 monocytes had a reduced ability to perform oxidative burst, although they produced TNF and IFN-γ in vitro. A significantly high number of COVID-19 monocytes had depolarized mitochondria and abnormal mitochondrial ultrastructure. Dysfunctional mitochondria/oxidative phosphorylation was associated with aging and defective immunological response to viral infections and chronic inflammation. |
Cell death/mitochondria/ energy metabolism | Low ATP production and mitochondrial dysfunction is a source of autoimmunity accompanied by necrotic cell death. Mitochondrial dysfunctions, including decreased phosphocreatine synthesis and impaired oxidative phosphorylation, are involved in the ME/CFS pathophysiology. The gut and immune cell mitochondria are proposed to be two important effectors that influence the circadian rhythm in ME/CFS pathophysiology. ME/CFS CD8 + T cells had reduced mitochondrial membrane potential. Both CD4 + and CD8 + T cells from patients with ME/CFS had reduced glycolysis at rest, whereas CD8 + T cells also had reduced glycolysis following activation. |
[15], [89], [134], [135], [141], [236] |
| [237], [238] | Exogenous or endogenous GC excess leads to the increased susceptibility to viral infections. GC excess was suggested to impact on COVID-19 infection clinical outcome. However, in hospitalized COVID-19 patients, the use of dexamethasone (DEX) resulted in lower 28-day mortality among those who were receiving either invasive mechanical ventilation or oxygen alone at randomization but not among those receiving no respiratory support. | Glucocorticoids (GCs) |
ME/CFS appears to be associated with a disturbed hypothalamus–pituitaryadrenal (HPA)-axis. GCs suppress Th1 cells and cellular immunity but may favour Th2 responses and humoral immunity. It was postulated that in ME/CFS patients a decreased Th1/Th2 balance may be the result of selective effects of GC on the IL-10/IL-12 regulatory circuit. With CD4 + T cells from ME/CFS patients (compared with cells from controls), a 10- to 20-fold lower DEX concentration was needed to achieve 50% inhibition of IL-4 production and proliferation, indicating an increased sensitivity to DEX in ME/CFS patients. |
[229], [239] |
SARS-CoV-2 has been shown to preferentially infect cells with higher expression of the virus spike protein-binding ACE2 receptor [50]. The penetration of the virus into cells starts with ACE2 binding, internalization of the ACE2-virus, and ACE2 shedding by tumour necrosis factor α (TNFα) convertase, a disintegrin and metalloprotease 17 (ADAM17) [51]. Activated ADAM17 signalling has been reported to disrupt biological barriers, cause endothelial senescence, and induce aging-associated brain pathologies [52], [53]. The hypothesis of involvement of ADAM17 in development of post-COVID-19 syndrome was recently reviewed [54]. Soluble human ACE2 was shown to inhibit SARS-CoV-2 infection in human blood vessel and kidney organoids [55], [56]. Consequently, several trials tested pathological influence of ACE2 gene single nucleotide polymorphisms (SNPs) [57], [58]. COVID-19-related morbidity and mortality were linked to D allele frequency of ACE2 [59], [60]. Multiple ACE2 variants were identified, suggesting ACE Del/Del polymorphisms influence COVID-19 susceptibility and facilitate development of a more aggressive infection profile [56], [61]. Accordingly, ACE Del/Del polymorphisms may be associated with post-COVID-19 condition onset. ACE2 polymorphisms may act as a predictive biomarker of ME/CFS. A study that previously tested association between ACE polymorphism and ME/CFS was published in 2009 and used a diathesis-allostatic load (AL) model [62] to assess pathological stress-related symptoms including ME/CFS [63]. Notably, high AL scoring was associated with ACE T allele rs4968591 and TT genotype was more common in subjects with high AL in ME/CFS. ME/CFS participants with TT genotype had the highest median C-reactive protein (CRP) levels, lowest urinary cortisol level (females), and increasing linear trend in IL-6 [63]. Considering that ACE2 polymorphism is associated with cytokine storm and COVID-19 severity [64], further investigations would be necessary to identify whether the ACE2 polymorphism correlates with onset and development of ME/CFS-like symptoms in patients with post COVID conditions.
Immune system changes in COVID-19, post-COVID-19 condition, and ME/CFS
Innate immune system abnormalities and cytokine storm associated with SARS- CoV-2 infection
Immune system is responsible for the recognition of viral particles and their elimination. Previous research has indicated that SARS-CoV-2 may predominantly infect macrophages (innate immunity cell effectors), although this assumption requires experimental confirmation and is currently based on previous findings for the SARS-CoV strain which was shown to infect alveolar epithelial cells, vascular endothelial cells, and macrophages [65]. Macrophages, dendritic cells, and neutrophils provide the initial immune defence barrier alongside airway mucosa. Encountering infection, macrophages can trigger anti-viral immune reactions and define the intensity of immune response. Elevated levels of macrophages and their over-activation were detected in severe cases of COVID-19 (Table 1) [66], [67]. Macrophages that express ACE2 (the virus-binding protein) along with incorporated SARS-CoV-2 nucleoprotein antigen were shown to infiltrate the spleen and lymph nodes in COVID-19 patients [68].
Infiltrating monocytes/macrophages and neutrophils express virus-recognizing receptors and can identify viruses and virus-infected cells. The virus is then recognized by cytosol-located pathogen recognition receptors (PRRs) after viral RNA enters its reproductive cycle, duplicates, and becomes double-stranded [69]. Coronaviruses, including SARS-CoV-2, encode specific proteins that can prevent PRR-mediated detection and activation of anti-viral mechanisms [70]. Accordingly, alveolar macrophages and monocytes were indicated as the original source of cytokines associated with abnormal systemic responses and multiple organ dysfunction in COVID-19 patients [70]. Previous studies have indicated that coronaviruses inhibit interferon (INF)-based responses and/or viral RNA identification by pathogen-associated molecular pattern (PAMP) receptors [71]. In contrast to the previous SARS strain, SARS-CoV-2-linked over-production of cytokines indicates effective recognition of the virus by the immune system [8], [48]. It is possible that Toll-like receptor 3 (TLR-3) may be able to identify SARS-CoV-2 [49], [72]. However, the mechanisms of this potential interaction have not yet been experimentally tested. Following the virus-sensing process, immune cells start to produce large amounts of pro-inflammatory cytokines and ROS, which are necessary for the propagation of defensive responses and elimination of viral particles. The key immune-pathologies related to ME/CFS and COVID-19 are presented in Table 1.
Many T-cell activating cytokines can be produced by macrophages. The level of produced cytokines in an immune response should be balanced to avoid tissue damage. Cytokine storm was registered during severe clinical manifestations of COVID-19 [8], [48]. SARS-CoV-2 infected macrophages have been shown to produce excessive amounts of interleukin-6 (IL-6) and IL-1, contributing to extreme inflammation in COVID-19 patients [68]. High IL-6 serum levels were detected in >50% of the COVID-19 patients [73]. The progression of disease severity was marked by the macrophage over-reactivity defined as macrophage activation syndrome (MAS) [48], [68]. Presence of MAS in adult COVID-19 patients with ARDS is indicated by high CRP levels in serum, which are not commonly observed during viral infections. McGonagle et al suggested a new MAS classification that can be used to uncover the role of macrophages in the progression of COVID-19-induced pneumonia towards ARDS [48].
The overproduction of pro-inflammatory cytokines in some patients with COVID-19 resulted in cardiovascular complications, multiple organ dysfunctions, and is considered lethal [74], [75]. Along with IL-6, enhanced production of T helper type-1 and type-2 (Th1 and Th2) cytokines (galectins, tumour necrosis factor alpha (TNFα), interferon-γ (INFγ), IL-2, and IL-17) was detected in COVID-19 patients [76], [77]. This increase in pro-inflammatory cytokines was also linked to subsequent T-cell exhaustion and PD-1 upregulation [78], [79]. Many recent studies have detected higher levels of IL-6 and IL-10 in patients with severe COVID-19 [4], [76], [77]. Notably, high IL-6 was determined to be a lethal risk factor in SARS-CoV-2 infected patients [80]. Therefore, IL-6 was named as a potential therapeutic target in SARS-CoV-2-infected patients. Tocilizumab (anti-IL-6 receptor monoclonal antibody) was tested for minimizing inflammation, with promising results [81]. The suggestion was supported by Ruan et al, who demonstrated a significant difference in serum IL-6 levels between COVID-19 survivors and non-survivors [77]. However, another study detected no association among various routine laboratory markers of inflammation (leukocyte, neutrophil, and lymphocyte counts, neutrophil-to-lymphocyte ratio (NLR), lactate dehydrogenase (LDH), CRP) and fatigue scores [82]. The study also found no association between fatigue scores and serum levels of IL-6 or soluble CD25, although the relatively small sample size of the study (128 participants) should be noted [82].
Abnormal immune system signalling was previously associated with debilitating fatigue [83], [84], [85] and fibromyalgia [86]. Changes in cytokine levels have been reported in ME/CFS patients [84], [86], [87]. A study published by Hornig et al. reported that early in the onset of the illness, the immune system of ME/CFS patients produces both disbalanced proportions of pro- and anti-inflammatory cytokines [83]. A recent systematic review indicated that cytokines may have ambiguous roles in ME/CFS [88], supporting the hypothesis that this illness is highly heterogeneous. The dynamic changes in ME/CFS cytokine profile require longitudinal investigations as the illness progresses [83].
The link between high levels of IL-6 (and other pro-inflammatory cytokines) and the development of post-COVID-19 symptoms, including fatigue and/or related sequelae, remains to be confirmed. However, the contribution of immune disbalances towards development of chronic fatigue is supported by several post-infection ME/CFS studies that analysed post-viral complications [20], [32], [35], [39], [41]. The wide scope of ME/CFS-associated pathologies supports the assumption that ME/CFS is a multifactorial illness with diverse molecular backgrounds. The pathomechanism of ME/CFS development is complex, where the majority of studies indicated immunological changes, including modest systemic inflammation [15], [89], significant reduction of Natural Killer (NK) cell function [90], [91], altered B cell phenotypes [92], significant changes in the level of circulating cytokines, including IL-1 (IL-1α and IL-1β), IL-6 [88], [93], and TNFα [94]. However, the cellular mechanisms of innate immune system responses, their role in potentially precipitating fatigue, and their therapeutic potential are yet to be examined in current and recovered COVID-19 patients.
Immune cell phenotypes were found to be significantly different between severe and non-severe COVID-19 patients. Higher NK cell numbers were found in peripheral blood and were associated with higher clearance rate of the virus and testing negative for COVID-19. Conversely, patients with lower NK cell numbers had presented longer duration of viral shedding and the significantly lower survival outcomes in the lymphocytopenia group [95]. Interestingly, the only consistently reported finding in ME/CFS patients is significantly reduced NK cell function [91].
Adaptive immune system abnormalities associated with SARS-CoV-2 infection
Susceptibility to SARS-CoV-2 is defined by individual immune system characteristics, as the virus has been shown to affect both innate and adaptive immune responses [48], [72], [73], [74], [75], [76], [77]. Asymptomatic COVID-19 disease resolution relies on anti-viral adaptive immune responses that can naturally eliminate SARS-CoV-2 virus particles in the majority of infected people [96], [97]. The adaptive T-cell responses are activated during acute and progressive infection stages and mediated by CD8 + T cells, which are key effectors for acute infection eradication and life-long protective immunity [96]. T-cell-activating cytokines, together with antigenic signals, are necessary for differentiation and clonal expansion of the naïve T cells [98]. However, in some circumstances (often unknown) the viral effects were associated with over-activation or reduction in immune responses in some patients. The dysfunctional reactivity of immune system results in exaggerated responses and organ failure, while reduced immune function may lead to inability to clear the infection and the development of chronic conditions [99], [100].
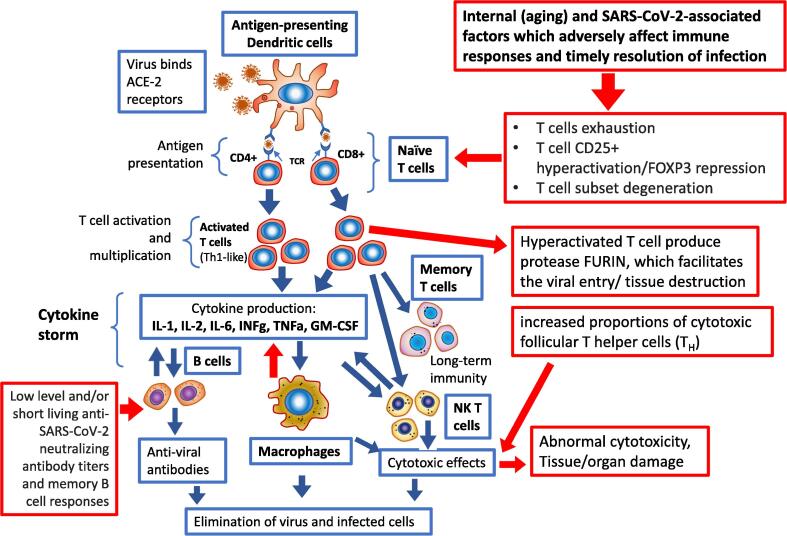
To provide effective anti-viral defence, adaptive T-cell populations proceed through tightly regulated stages. During the acute infection stage, CD8 + T-cells undergo clonal virus-specific differentiation into effector T-cells to eliminate the virus [96], [99]. Some effector CD8 + T-cells gain a memory precursor phenotype. After the infection is eliminated, the T-cell response is reduced, effector T-cells are removed by apoptosis, and the memory precursor cells are kept and develop into the anti-viral memory T-cell pool [96], [99]. However, in some individuals, including patients with severe COVID-19, the normal development of adaptive T-cell responses is compromised (Fig. 2).
Fig. 2.
Immune system responses to SARS-CoV-2 and associated complications. Red arrows indicate abnormal or dysfunctional signalling associated with severity of COVID-19. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
CD8 + T-cell differentiation and anti-viral functions are directed by a set of proinflammatory cytokines. Type I interferons (IFN) and IL-12/IL-6/IL-1 family members were shown to direct the intensity of the inflammatory milieu and T-cell responses [96], [99]. Overwhelming increases in the production of these cytokines can be devastating for viral control and facilitate development of faulty CD8 + T-cells, described as exhausted T-cells [96], [99]. Commonly, during chronic viral infections, prolonged viral antigen exposure, and/or excessive inflammation (overproduction of cytokines/cytokine storm), CD8 + T-cells develop the exhausted phenotype, marked by limited capacity to eliminate viral pathogens [99], [101]. Exhausted T-cells express ensembles of inhibitory receptors and fail to accomplish their key anti-viral functions [96], [99]. The exhausted T-cell phenotype was reported in severe COVID-19 patients [79]. Cytotoxic CD8 + T-cells in COVID-19 patients are activated by pro-inflammatory cytokines and display functional exhaustion characteristics including expression of CD94/NK group 2 member A receptor (NKG2A), programmed cell death protein 1 (PD-1), and T-cell immunoglobulin and mucin domain 3 (TIM-3) [78].
Aside from signs of T-cell exhaustion, SARS-CoV-2 was shown to downregulate the production of interferons (including INFγ, IL-2, and granzyme B) and slow down immune responses [49], [102]. General T cell degeneration was also marked by lymph node and spleen atrophy, as well as a decreased number of CD8 + T-cells in severe COVID-19 patients [103]. Inhibition of T-cell-mediated immune responses was linked to downregulation of MHC class I and II molecules and problems with antigen presentation [49]. CD8 + T-cell abnormality was associated with decreased CD107a externalization [102]. However, a link between T-cell exhaustion and development of post-COVID-19 illness requires confirmation. Immune cells from ME/CFS patients were not checked for presentation of exhausted phenotype markers either.
The relatively slow initial progression of immune responses was also identified as one of the immune-system-based defects in severe COVID-19 cases [43]. A potential problem with circulating memory T and B cells was suggested as being responsible for the initial low rate of anti-viral responses [104]. A lower level of memory CD4+ T-cells contributed to poorer T cell responses in SARS patients [105]. Currently, there is limited information about memory B-cell kinetics in COVID-19 patients. To the best of our knowledge, B-cell kinetics in association with development of ME/CFS as a post-viral complication has not yet been investigated.
Autoimmunity in the pathophysiology of COVID-19, post-COVID-19 condition, and ME/CFS
The presence of autoantibodies has been described in COVID-19 patients [106]. The autoantibodies were detected using the rapid extracellular antigen profiling method, which indicated increased auto-responses in COVID-19 patients compared to healthy controls. Autoantibodies were also systemically found throughout various tissues and implicated in the development of post COVID-19 condition. However, it remains to be determined whether these autoantibodies persist beyond the acute infection phase of COVID-19 and whether they are casually associated with post COVID-19 condition [106]. Arthur et al detected presence of ACE2-specific autoantibodies in many post COVID patients [107]. Consequently, patients with post-acute sequelae were found to have lower activity of soluble ACE2 in blood plasma and increased circulation of angiotensin II which may lead to enhanced proinflammatory state in post COVID-19 patients [107].
Another study indicated the presence of active G-protein coupled receptors (GPCR)-targeting autoantibodies, including β2- and α1-adrenoreceptors, angiotensin-II AT1-receptors, and muscarinic M2-receptors in post COVID-19 condition patients [108]. Using lumbar puncture, Franke et al assessed autoantibody levels in cerebrospinal fluids (CSF) from eleven severe COVID-19 patients (median age 67, 8 male) with unclear aetiology neurological symptoms diagnosed after admission to ICU and onset of respiratory complications [109]. The study analysis addressed antigen binding to a large panel of intracellular and surface antigen markers for CNS diseases, including antibodies against amphiphysin, Delta/Notch -like EGF repeat containing receptor, collapsin response-mediator protein-5, glutamic acid decarboxylase 65-kilodalton isoform, cardiolipin, beta2- glycoprotein/annexin/glutamate receptors, dipeptidyl-peptidase–like protein 6, γ-aminobutyric acid type A receptors, GPCRs for gamma-aminobutyric acid receptors, metabotropic glutamate receptor, leucine-rich glioma-inactivated 1 protein, contactin-associated protein-like 2, dopamine-2 receptor, aquaporin-4, myelin, and skeletal muscle proteins [109]. The collected data indicated elevated level of autoantibodies affecting specific brain targets, although the comparison was not impeccable as control CSF samples were not collected in patients without neurological illness. Despite this limitation, the authors suggested the development of neurological pathologies as contributing factor to the severity of the illness [109]. It was indicated that generation of autoantibodies may initiate and/or impact multi-organ pathologies in COVID-19. Accordingly, the development of targeted immunotherapies was indicated as a potential therapeutic approach for affected COVID-19 patients. Whether such autoreactivity can cause persisting neurological morbidity after clearance of SARS-CoV-2 and remission of COVID-19 requires further studies [109].
In a prospective study, Virhammar et al investigated 19 mild to critical COVID-19 patients with neurological symptoms [110]. Extensive analysis of CSF content, including measurement of CNS injury biomarkers (neurofilament light chain protein (NFLP), glial fibrillary acidic protein (GFAP), and total tau), was performed and correlated to neurological features and the disease severity. Elevated levels of NFLP, GFAP, and total tau in CSF were found in COVID-19 patients with neurological symptoms [110]. Conclusively, autoimmunity mechanisms may play a role in development of COVID-19-associated pathologies, however, studies have been limited by small sample sizes and lack of suitable controls. Previously, autoantibody levels were found to be elevated in ME/CFS patients [111], [112]. Aptamer BC 007 [113], [114], an autoantibody blocker against GPCRs has also been assessed for efficacy in “long COVID-19” patients in an initial treatment trial comprising two patients. BC 007 is currently being investigated in a bigger placebo-controlled clinical trial called reCOVer [113], [114]. However, the role of autoimmune antibodies in the development of post COVID condition requires further investigation.
Significantly increased levels of autoantibodies to beta-adrenergic and muscarinic cholinergic receptors were found in a cohort of ME/CFS patients in Sweden, although the pathomechanism of the autoantibody contribution to the development of in ME/CFS is unknown [115].
Metabolism changes in the pathophysiology of COVID-19, post COVID-19 condition, and ME/CFS
Energy metabolism and oxidative processes in mitochondria have been shown to influence innate and adaptive immune responses to SARS-CoV-2 [116], [117]. Mitochondria are the organelles responsible for structural and functional support of oxidative phosphorylation (OXPHOS), intracellular energy metabolism, and homeostasis. Consequently, dysbalanced mitochondrial functions have been suggested to result in immune cell failure and dangerous progression of COVID-19 [118]. Mitochondrial mutations and modified OXPHOS were associated with the development of numerous age-associated pathologies, muscle degeneration, fatigue, and metabolic transformations [47], which have been reported to contribute to the severity of COVID-19 [3], [119].
Bidirectional connections were observed between inflammatory signalling and energy metabolism. Abnormal mitochondrial functions and oxidative stress may be activated by inflammation and pro-inflammatory cytokines [120]. Viral infection strongly impacts energy supply and dysregulates metabolism in many tissues, including circulating immune cells [121], [122], [123]. Increased IL-6 was also associated with disbalanced OXPHOS and lowered levels of ATP [122]. Insufficient energy/ATP supply compromises the ability of T-cells to proliferate and function as anti-viral effectors [124], [125]. ROS and metabolic starvation contribute to T-cell exhaustion [126]. Aside from direct activation of apoptosis, oxidative stress and ROS can impact gene activation via epigenetic mechanisms and transform T-cell differentiation towards the exhausted phenotype [101], [127].
Adaptive antigen-specific responses rely on balanced mitochondrial activity, which is significantly transformed during T-cell activation. A minimal amount of nutrients is required for basal glycolytic rate and effective OXPHOS process in naïve and resting T-cells, which are described as “metabolically quiescent”. T-cell receptor (TCR) stimulation triggers a ‘glycolytic switch’ towards greater utilization of ATP generated via OXPHOS [128]. More differentiated CD8 + memory T-cells were found to have more mitochondrial mass, which provides them with a metabolic advantage and higher reactivity compared to naïve CD8 + T-cells [129]. Additionally, differentiated and/or activated CD8 + T-cells start to utilize glycolysis to support their anti-viral effector functions [130]. Stimulation of glycolysis in CD8 + T-cells is mediated by Akt and mTOR signalling, which is downregulated during chronic infection and in exhausted T-cells [131]. Suppressed mTOR activity in T-cells is marked by mitochondrial depolarization, lowered mitochondrial biogenesis, and enhanced ROS production [99]. The process of metabolic reprogramming was observed in T-cell subpopulations after acute viral infection [131]. The association between changes in mitochondrial functions, T-cell exhaustion, and development of post-COVID-19 complications including chronic fatigue warrants further detailed investigation.
Aside from T-cell transformations, altered bioenergetics was observed in peripheral blood mononuclear cells (PBMCs) from SARS-CoV-2 infected patients. The PBMCs demonstrated decreased respiration rates and diminished ability to support a reliable level of aerobic glycolysis. Monocytes from COVID-19 patients failed to support a high level of oxidative burst despite normal synthesis and release of TNFα and IFNγ in vitro [125]. Depolarized mitochondria with transformed mitochondrial ultrastructure were detected in those PBMCs. The abnormality was marked by high levels of inflammatory cytokines [125].
In both COVID-19 and ME/CFS patients, serious tissue and organ damage was associated with increased reactive oxygen species (ROS) formation, which follows the disbalanced cytokine production [132], [133]. Pro-inflammatory cytokines, dysregulated T-cell signalling, abnormal mitochondrial OXPHOS, and elevated level of ROS (oxidative stress) may facilitate development of ME/CFS [134], [135]. High serum TNFα content in patients with ME/CFS was shown to downregulate mitochondrial and cellular bioenergetics [94], [136]. TNFα-induced signalling through death receptors activates production of ROS, which were shown to be responsible for the destruction of mitochondrial DNA (mtDNA) [137]. Cytokine-induced destruction of mitochondrial signalling was suggested in ME/CFS [94], [136]. However, further investigations are warranted.
Circulating platelets (non-nucleated cells) contain mitochondria, which influence the pro-thrombotic function of these cells. Dis-regulated OXPHOS leads to activation of apoptosis in platelets, which triggers coagulation cascades and aggravates blood clotting and thrombosis [138]. SARS-CoV-2 pathogenesis was associated with increased incidence of thrombosis and lower survival in susceptible patients [139], [140]. The role of platelets and thrombosis-related factors in ME/CFS and other post-viral complications remains largely unclear, although there are indications of transformed coagulation in ME/CFS patients [87].
The role of mitochondrial pathways in ME/CFS pathogenesis has been explored in several clinical studies and critically assessed in a systematic review conducted by Holden et al [141]. Despite the described mitochondrial changes in ME/CFS patients, the role of mitochondria in development of ME/CFS pathology remains unclear [141]. The long-term changes in mitochondrial and cellular bioenergetics in post COVID-19 condition patients have not yet been reported.
Transient receptor potentiation channel dysfunction in development of post COVID-19 symptoms: A link to ME/CFS
Genetic screening has indicated that several single nucleotide polymorphisms (SNPs) of transient receptor potential (TRP) ion channel family members are commonly exhibited by ME/CFS patients compared to healthy controls [142]. TRP ion channels are membrane bound proteins that are non-selectively permeable to various essential ions including calcium, magnesium, sodium, and zinc. TRPs mediate responses to stress factors associated with environmental changes including fluctuations in temperature, pressure, and chemical and pathogen-related (bacterial and viral infections) exposures [143]. Specifically, several SNPs were detected in the TRP melastatin 3 (TRPM3) group [142], [144]. TRPM3 expression is decreased in NK cells in ME/CFS patients, resulting in impaired Ca2+ influx [142], [144].
Bousquet et al discussed the potential involvement of TRP ankyrin A1 (TRPA1) and TRP vanilloid (TRPV1) channels in COVID-19 pathology [145]. TRPV4 has been associated with lung oedema, a frequent symptom in critically ill COVID-19 patients. TRPV4-facilitated ion transport has been shown to disrupt alveolo-capillary barrier integrity. Accordingly, desensitization and/or inhibition of these channels by specific antagonists, including GSK2798745, have shown promising results in treatment of chronic cough or cardiogenic lung oedema patients in phase I clinical trial [146]. However, the role of these channels has not been assessed in post-COVID-19 condition. It remains to test whether TRP expression levels is changed in patients with post-COVID-19 complications. Genetic screening of post COVID-19 condition patients may offer insights about gene signatures and molecular pathways involved in the pathomechanism of this illness.
Importantly, Ca2+ channels and associated ion signalling pathways are involved in the regulation of numerous processes, including energy metabolism and OXPHOS, cytokine effects, and immune responses [147]. Lower levels of Ca2+ were detected in COVID-19 patient serum samples [148]. The observed data suggests that low serum Ca2+ may be an indicator of higher COVID-19 severity. In mild and moderate COVID-19 patients, low Ca2+ levels correlated to severe multi-organ injuries and changes in IL-6 levels [148]. Hypocalcaemia (low Ca2+ levels), however, has also been associated with activation of host defence mechanisms [149]. It has been suggested that the SARS-CoV-2 virus utilizes host Ca2+ channels to facilitate cell entry and efficient replication (virulence) [149]. Given Ca2+ is a critical regulator of many cellular pathways, including oxidative stress and NK cell cytotoxicity, irregular Ca2+ levels may have systemic consequences. Long-term involvement of Ca2+ transport in post COVID-19 pathology has yet to be investigated. Considerable Ca2+ dysregulation in ME/CFS and the potential overlap with pathomechanism of post COVID-19 condition remains to be confirmed.
Neurological and autonomic dysfunctions in COVID-19, post COVID-19 condition, and ME/CFS
Persistence of dyspnoea and neuropsychological symptoms were registered in 35% of non-hospitalized and 87% of hospitalized COVID-19 patients [5], [6]. In a three-month follow- up study, 55% of COVID-19 patients had neurological symptoms [150]. These characteristics included higher bilateral gray matter volumes (GMVs) in olfactory cortices, hippocampi, insulas, left Rolandic operculum, left Heschl’s gyrus, and right cingulate gyrus. Global mean diffusivity of white matter and GMV in the aforementioned regions were correlated with memory loss. GMVs in the right cingulate gyrus and left hippocampus were correlated with the loss of smell [150]. Neurological manifestations have also been described in children infected by SARS-CoV-2 [151]. The most frequent observation found was postinfectious immune-mediated encephalomyelitis-like changes of the brain, which were found in 16 out of 38 child patients. Neural enhancement and myelitis were also observed in 13 and 8 patients (out of 38), respectively [151]. The same study reported splenial legions and myositis associated with multisystem inflammatory post-COVID-19 syndrome [151].
Sollini et al. investigated post COVID-19 condition hallmarks using positron emission tomography/computed tomography ([18F]FDG-PET/CT) [152], a highly precise visual tool used frequently in oncological settings and commonly applied in inflammatory conditions of the CNS. Patients included in this study exhibited brain hypometabolism in the right parahippocampal gyrus and thalamus. Changes in metabolism were also observed in other regions of the body, which suggests the systemic nature of this illness. Hypometabolism was associated with persistent anosmia/ageusia, fatigue, and vascular complications [152]. CNS-linked pathologies may be caused by viral particle accumulation in cerebrospinal fluids. Supporting this hypothesis, autopsy investigations revealed SARS-CoV-2 RNAs and viral proteins concentrated at the brainstem [139]. SARS-CoV-2 has shown an involuntary growth-orienting response to the brainstem due to the presence of high ACE2 receptor levels in this organ compared to other brain regions. A significant part of the assessed post-COVID patient samples (67%) had SARS-CoV-2 RNA and spike proteins located in the olfactory mucosal-neuronal junction. Inflammatory responses, neurodegeneration, and viral invasion in brainstem have been described [153].
Another report described SARS-CoV-2-caused neuroinflammation [154]. Proinflammatory changes in cerebral fluid composition were shown to provoke cognitive dysfunction, impact sleep-wake cycles, and contribute symptoms of fatigue [155], [156]. Accordingly, IL-6 has been indicated as a potential mediator of long-term neuropsychiatric symptoms in COVID-19 survivors [157]. Linked to inflammation, psychiatric sequelae have also been described in COVID-19 patients [12], [158]. In a study conducted by Mazza et al., 402 recovered COVID-19 patients were assessed for the presence of psychological changes after COVID-19 and 56% of the participants were scored with the psychiatric pathologies [158]. It has been suggested that these psychopathologies may be fostered directly via viral infection of the CNS or through dysfunctional immune processes such as neuroinflammation as well as disruption of the blood–brain barrier and/or hypothalamic-pituitary adrenal axis [158]. Stigma, social isolation, and other stressors may also contribute to these symptoms [158]. A longitudinal study by Jason et al reported that some post COVID condition patients experienced progressive worsening of neurocognitive symptoms over time [159].
Among other inflammation-related symptoms, fatigue was found to be the most common symptom (46.9%) among 130 surveyed COVID-19 patients [160]. Significant age difference was detected between patients with fatigue (53.9 ± 13.5 years) compared with those without fatigue (48.5 ± 13.3 years). Patients with fatigue were also hospitalized for longer periods, although no association was observed between the disease severity and presence of fatigue [160]. However, after adjustments for the relevant criteria (defined by the Institute of Medicine as “systemic exertion intolerance disease” characteristics), only 13% patients in this cohort were diagnosed with ME/CFS, indicating that the observed clinical symptoms overlap with ME/CFS in a set of post COVID-19 condition patients [160].
Nervous system abnormalities have been reported in ME/CFS patients, although the pathomechanism remains unclear. ME/CFS is considered a neurological disorder by the WHO [161]. Infection of microglia, changes in cerebral blood flow, and gray and white matter volumes have been described in ME/CFS patients [162], [163]. Brainstem abnormalities have also been reported, including changes in brainstem functional connectivity [164]. These cognitive impairments may result in brain fog and restless sleep [165]. In conclusion, the observed post-COVID-19 nervous system pathophysiology indicates a potential predisposition to develop ME/CFS.
In contrast to the described observations, Sykes et al reported the lack of correlation between symptom burden and radiographic or biochemical abnormalities in brain [166]. Accordingly, it has been suggested that symptoms may be attributable to biopsychosocial effects of COVID-19. This study, however, included a limited number of tests [166] that may have overlooked clinical characteristics of post COVID-19 condition described above.
Cardiovascular dysfunction in COVID-19, post COVID-19 condition, and ME/CFS
Several inflammation-linked cardiovascular issues have been defined in hospitalized patients with COVID-19, including inflammatory plaque rupture, cardiac stress due to high cardiac output, and thrombosis [167], [168]. Those with pre-existing conditions were shown to have an increased risk of heart failure [169]. Cardiovascular sequelae using cardiovascular magnetic resonance imaging in patients who recovered from recent infection with COVID-19 were investigated in 100 patients [169]. Of these patients, 78% had ongoing cardiac involvement and 60% had myocardial inflammation [169]. Evidence of vasculitis, the inflammation of blood vessels, has also been described [170]. Reports of palpitations and orthostatic intolerance have also been made, but these are limited to single case studies [171]. ME/CFS patients also have reported cardiovascular problems, including abnormal blood pressure, hypertension, and orthostatic intolerance [172]. It currently remains unclear whether these symptoms of autonomic dysfunction in COVID-19 patients are caused by direct viral invasion and tissue destruction or indirect effects of ARDS, including hypoxemia, inflammation, and/or thrombosis. It is possible that a combination of both mechanisms is involved.
The permanent and often uncurable vascular damage caused by the SARS strain was marked by thromboembolism, micro-infarctions, pulmonary embolism, and other dysfunctions [173]. Cardiovascular post-COVID-19 complications have been diagnosed in hospitalized patients and included inflammatory plaque rupture, cardiac stress, and stent thrombosis [169]. Investigations into cardiovascular sequelae in 100 patients who have recovered from recent SARS-CoV-2 infection indicated that 78% of patients had ongoing cardiac complications and 60% had myocardial inflammation [169]. Another similar study also detected signs of vasculitis, the inflammation of blood vessels [170]. One other single case study reported palpitations and orthostatic intolerance [171]. The observed pathologies could be associated with virus-induced hypoxemia, inflammation, and/or thrombosis. In comparison to COVID-19, ME/CFS patients were also diagnosed with cardiovascular pathologies, abnormal blood pressure, hypertension, and orthostatic intolerance [172].
Gastrointestinal (GI) dysfunction in COVID-19, post COVID-19 condition, and ME/CFS
A variety of GI manifestations have been reported in COVID-19 patients, including nausea, vomiting and abdominal pain. The second most common COVID-19 symptom is diarrhea, which was registered in up to 70% of patients [174], [175], [176]. COVID-19 patients presented with diarrhea had 2.7 times higher odds of having longer duration of symptoms [177]. Some patients have reported GI symptoms in the absence of respiratory symptoms [74]. ACE2 receptors are highly expressed in the glandular cells of gastric, duodenal, and rectal epithelia. Direct viral invasion into GI tissue may contribute to the symptom onset [178], consequently, causing nervous system dysregulation, including disruptions in the dorsal vagal complex (DVC). Vagal neurocircuits have important roles in regulating nausea and vomiting [179], which were reported in COVID-19 patients [180].
Changes in microbiome composition, including underrepresentation of key commensal bacterial colonies such as Faecalibacterium prausnitzii, Eubacterium rectale and bifidobacterial, were associated with immunomodulation in COVID-19 patients [181]. Microbiome changes, including SARS-CoV-2 invasion, often result in variations of cytokine and inflammatory marker levels [181]. A study investigating alterations in gut microbiota suggested that dysbiosis in COVID-19 patients persisted even after infection clearance [182]. Changes in microbiome and GI-related symptoms have been reported in individual studies in ME/CFS patients [183]. It has been suggested that gut-located pathogens and/or infection-related toxins produced by invaded pathogens may translocate from the GI tract into other tissues and trigger ME/CFS symptoms [54]. A systematic review on enteric dysbiosis in ME/CFS, however, found that there was limited evidence of this condition being involved in the pathomechanism of ME/CFS [184].
GI-associated pathologies were reported in post COVID-19 condition patients [185], [186]. Interaction between SARS-CoV-2 particles and host GI proteins, including Bromodomain-Containing Proteins 2 and 4 (BRD2 and BRD4) was detected [187]. Expression of BRD4, the protein involved in the innate immune reaction to SARS-CoV-2, has been previously observed in the small intestine [187]. Notably, the crosstalk between SARS-CoV-2-located protein E and BRD4 have been shown to activate macrophage senescence [187]. Previous investigations suggested that BRD4 signalling may be associated with muscle weakness (a potential link to PEM, a major ME/CFS symptom) [188]. Importantly, there is limited information available on the pathomechanism of post COVID-19 condition in the GI system.
Intervention strategies and future directions for post COVID-19 condition -illness
There is evidence of persistent physiological and psychological sequelae in COVID-19 survivors. The multi-system post-COVID-19 sequalae that have been observed include disruption in neurological, cardiovascular, and musculoskeletal pathways [128]. However, the diversity of the observed complications hinders the development of efficient treatments. Post COVID-19 condition may represent a number of different post-viral syndromes that require appropriate classification [189]. All long-term symptoms of post COVID-19 condition should be monitored and recorded. Large data collection and registration of symptoms is required to determine whether long-term symptoms are due to a post-viral syndrome such as ME/CFS. The process of data collection and analysis should be controlled for all confounding factors including consequences of intensive care hospitalization, social isolation, and diet-related effects (diet-linked anaemia). Timely collection of data and identification of physiological mechanisms underlying the long-term clinical manifestations of COVID-19 and post COVID-19 condition are vital for the relevant design of effective therapies. While available vaccinations are expected to reduce the devastating health effects of SARS-CoV-2, it is also predicted that SARS-CoV-2 mutations may turn the pandemic into an ongoing issue [190].
COVID-19 and post COVID-19 condition follow-up studies have been initiated. Some of these studies are focused on damaged organ systems (e.g. respiratory and cardiovascular system organs), while others will monitor and record all adverse effects. For instance, the post-hospitalisation COVID-19 study (PHOSP-COVID https://www.phosp.org/) intends to follow 10,000 patients for 1 year in the United Kingdom [191]. A similar study was initiated in the USA (go.nature.com/3mfqqxc). The British Heart Foundation also declared several new COVID-19-based research programmes. One of these projects will follow COVID-19 hospitalized patients for six months and all signs of damage will be recorded. In the European Union, the CAPACITY registry initiative is looking to collect follow-up reports about SARS-CoV-2 infected patients from dozens of European hospitals [192]. The studies aim to collect data from blood tests and scans, using virus-related biomarkers.
Several antiviral and anti-inflammatory agents (including remdesivir, dexamethasone), monoclonal antibodies and immunomodulatory substances (bamlanivimab, casirivimab, baricitinib, tocilizumab) were used as emergency treatment during the management of severe acute COVID-19 in risk factors [193], [194]. However, there is currently no validated treatment for post COVID-19 condition. Considering prospective interventions, Ca2+-regulating treatment can be explored as promising, given Ca2+ signalling was shown to be modified in both post-COVID-19 and ME/CFS patients. The use of dihydropyridine Ca2+ channel blockers, including nifedipine (TRPM3 agonist [195]) and amlodipine, have been suggested as an effective therapeutic approach to reduce mortality in elderly COVID-19 patients [196]. The decreased serum Ca2+ level may serve similar anti-viral purposes [149]. Accordingly, Cabanas et al have recently found that TRPM3 function in ME/CFS patients can be restored by in the presence of the mu (μ)-opioid receptor antagonist, naltrexone, which also indicated anti-inflammatory properties [197], [198]. Recently, a pilot study was registered to test the effect of low-dose naltrexone in combination with nicotinamide adenine dinucleotide supplement for the treatment of post COVID-19 condition (Clinical trial #NCT04604704).
The use of ACE inhibitors was suggested as a potential therapeutic approach for prevention of post-COVID-19 complications, including organ injury [2], [199]. However, ACE inhibitors should be used cautiously as they were found to increase plasma angiotensin levels (pro-inflammatory effect) and cardiac ACE2 expression [200]. To minimize the potential complications, the ACE2-related pathway was also explored [199]. The ACE2-pathway-targeting agent and Mas receptor activator BIO101 (20-hydroxyecdysone) is currently being tested (the trial started on September 1, 2020) for management of acute SARS-CoV-2 infection and related complications [199]. BIO101 activates the Mas-receptor and triggers anti-inflammatory, anti-thrombotic, and anti-fibrotic effects, thus restoring internal homeostasis in COVID-19 patients [199].
Conclusions
Nearly two years into the worldwide COVID-19 pandemic, post COVID-19 condition syndrome has proven to be a serious and lingering problem for many recovering patients. This multifactorial illness is characterised by a variety of debilitating symptoms, including fatigue, brain fog, and post-exertional malaise. Many of the pathological observations of post COVID-19 condition, including changes in immune, cardiovascular, metabolic, gastrointestinal, nervous and autonomic systems, are shared with or similar to the symptoms described in ME/CFS patients. Considering the current evidence presented in this study, it is possible that large groups of post COVID-19 condition patients may eventually meet the criteria for ME/CFS diagnosis. Comprehensive longitudinal symptom monitoring is required to confirm this diagnosis, uncover the mechanisms of post-COVID-19 -associated ME/CFS development, and design or develop relevant prevention and treatment measures. Current absence of the effective treatment reflects the unclear causes of the post COVID-19 conditions which cannot be targeted properly until the mechanism is established and confirmed.
Compliance with Ethics Requirements
This article (the review article) does not contain any studies with human or animal subjects.
CRediT authorship contribution statement
Olga A. Sukocheva: Writing – review & editing, Conceptualization, Visualization. Rebekah Maksoud: Writing – review & editing. Narasimha M. Beeraka: Writing – review & editing, Formal analysis. SabbaRao V. Madhunapantula: Conceptualization. Mikhail Sinelnikov: Conceptualization. Vladimir N. Nikolenko: Conceptualization. Margarita E. Neganova: Writing – review & editing, Visualization. Sergey G. Klochkov: Conceptualization. Mohammad Amjad Kamal: Conceptualization. Sonya Marshall-Gradisnik: Conceptualization, review & editing. Donald R Staines: Conceptualization, editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgement
None.
Biographies

Dr Sukocheva completed Ph.D. in Biophysics in 1997 (Uzbekistan). She was awarded Muskie Fellowship in 1999 and worked as a Postdoctoral Scientist at New York State Department of Health, Wadsworth Laboratories (Albany, NY, USA). In 2001, Dr Sukocheva was appointed as research officer at Division of Human Immunology, Hanson Cancer Research Institute in Adelaide, South Australia. During her work at the institute, Dr Sukocheva joined Professor Barrie Marmion and Professor Stephen Graves research group which investigated post-infection sequelae and associated chronic fatigue syndrome. In 2010, Dr Sukocheva was awarded C.M.E. Kelly Cancer Research Fellowship at Flinders University where she worked as Lecturer in Health Sciences till May 2021. In May 2021, Dr Sukocheva accepted Senior Public Health Officer position at SA Health (Department of Health and Wellbeing) and works now with COVID Operations Team. Recently, Dr Sukocheva joined in the Menzies Health Institute NCN as Adjunct-Lecturer.
Scopus ID: https://www.scopus.com/authid/detail.uri?authorId=6602940090
ORCID ID: https://orcid.org/0000-0003-1041-3311
ResearchGate ID: https://www.researchgate.net/profile/Olga-Sukocheva
Publons ID: https://publons.com/researcher/1169737/olga-sukocheva/metrics/
Google Scholar ID: https://scholar.google.co.in/citations?user=f21zMfMAAAAJ&hl=en

Ms Rebekah Maksoud is an early career researcher. She has been working at the National Centre for Neuroimmunology and Emerging Diseases for the last four years. During her work at the centre, Rebekah has gained significant clinical, laboratory and research experience in the field of immunology. She is currently involved into research using a large variety of immunological methods, including natural killer cell isolation, flow cytometry, and cytotoxic assays. Her research aims are linked to determination of the expression and cytotoxic potential of natural killer T-cells and the role of T-cells in development of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). Rebekah’s current PhD project is about the assessing of disability measures in ME/CFS patients. Rebekah contributed various other projects and published reviews about neurological, mitochondrial, and sleep-rel ated changes in ME/CFS patients. Rebekah currently has got five publications in the field of ME/CFS and will presented her research data at the International Association of Chronic Fatigue Syndrome (IACFS) conference (19-21st August 2021).

Narasimha M. Beeraka, MSci by Research (UK), PhD, is an early career research fellow at Prof. M.V.S.S.T. Subba Rao Madhunapantula’s Laboratory at JSS Higher Education and Research Academy, Mysuru, Karnataka, India. He successfully published 28 research manuscripts (scopus; ORCID) and currently involved in several inflammation-related clinical and bio-medical investigations. Dr Beeraka is interested in identification and development of clinical biomarkers for chronic diseases including inflammation-linked pathologies.

Dr. SubbaRao V. Madhunapantula received Ph.D. in Biochemistry from University of Mysore, Karnataka, India. During his post-doctoral research at Penn State Cancer Institute, Hershey, Pennsylvania, USA, he investigated key signaling cascades involved in regulation of melanoma carcinogenesis. Dr. Madhunapantula joined JSS Academy of Higher Education & Research (JSS AHER), Mysore, Karnataka, India in 2011 and established a biomedical research center which is known as Center of Excellence in Molecular Biology and Regenerative Medicine (CEMR). Currently he continues his work as a professor of cellular and molecular biology in CEMR laboratory. His current research interests belong to the field of clinical pathology and inflammation-related signaling.

Dr Mikhail Y. Sinelnikov, M.D., Ph.D. is an assistant professor at the department of Human anatomy at Sechenov University. He is the Clinical Morphology lab Head at Research Institute of Human Morphology and a practicing reconstructive surgeon at Russian National Scientific Center of Surgery. He grew up in New York, USA, but chose to finish medical school in Russia. He graduated with honours in 2016, and later attained residencies in both surgery and oncology. Dr. Sinelnikov's research interests include reparative regeneration of tissues, microvascular aspects of tissue degeneration, lymphatic diseases, and stem cell therapy. Dr Sinelnikov’s current research aim is focused on investigation of degenerative disorders of lymphatic tissue associated with various pro-inflammatory conditions.

Professor Vladimir N. Nikolenko, M.D., Ph.D., D. Med. Sc., is the Head of the Department of Human Anatomy, at N. V. Sklifosovsky Institute of Clinical Medicine, FSAEI HE and I. M. Sechenov First Moscow State Medical University (Sechenov University). He is an expert in neuroscience and biomechanics of the nervous system. Professor Nikolenko authored and co-authored over 500 scientific publications, including 21 books, 8 textbooks, and 31 patents. Prof Nikolenko established a new scientific direction in CNS neuro-morphology and medical biomechanics associated with adaptation of nervous system to new environment and tsress-related conditions. He formulated background laws of spinal cord kinesiology. Prof Nikolenko made significant contributions to the field of nervous system regeneration (Scopus ID: 14016274500; ORCID: https://orcid.org/0000-0001-9532-9957). His current research interests belong to the field of CNS-related pathologies.

Margarita E. Neganova, PhD, is the Head of the Group of Enzymatic Processes of Pathological Conditions of the Institute of Physiologically Active Compounds of Russian Academy of Sciences (IPAC RAS). After graduating with honors from Ivanovo State University in 2007, she entered the postgraduate study at the IPAC RAS, specializing in bioorganic chemistry, and in 2012 received her Ph.D. She is a specialist in the field of biochemistry, neurobiology, cytology, oncology, and preclinical drug development. Margarita E. Neganova is the author of more than 70 publications, including publications in various peer-reviewed journals, patents, and monographs. https://www.scopus.com/authid/detail.uri?authorId=49864350200

Sergey G. Klochkov, Ph.D, is the acting director of Institute of Physiologically Active Compounds of Russian Academy of Sciences (IPAC RAS). He is the Head of laboratory of Natural Products Institute of Physiologically Active Compounds. After graduating from Moscow State University, he received his Ph.D. at Biological Department of Moscow State University. He is a well-known specialist in the field of medicinal and natural chemistry, biochemistry, neurobiology and oncology. His current research interests are focused on the field of neuroprotection and drug design, as well as development of new clinical testing methods. https://www.scopus.com/authid/detail.uri?authorId=6603406902

Mohammad Amjad Kamal is a Distinguish Adjunct Professor at King Fahd Medical Research Center (KFMRC), King Abdulaziz University, Jeddah, Saudi Arabia, where he was a full Professor from March 2011 to March 2013. Prof. Kamal's overall biochemical research output had culminated in more than 560 publications in internationally respected journals. His research was pivotal in supporting the development of the novel anti-Alzheimer's agents from the laboratory to the clinic via collaboration with Dr Nigel H. Greig (Chief of Laboratory of Neurosciences and Gerontology, NIA, NIH, USA). Prof. Kamal is serving Novel Global Community Educational Foundation, Australia as a Foundation President since 2016 (http://www.ngcef.net). He has collaboration/affiliations with many institutes and scholars all around the world such as the following: West China School of Nursing / Institutes for Systems Genetics, Frontiers Science Center for Disease-related Molecular Network, West China Hospital, Sichuan University, Chengdu 610041, Sichuan, China. Moreover, he is a regional/editorial member/guest editor of several reputed scientific journals - this is undertaken on an honourable basis. His publications are obvious on the following links: http://www.researcherid.com/rid/J-2918-2014 https://www.scopus.com/authid/detail.uri?authorId=56340070000https://www.ncbi.nlm.nih.gov/pubmed/?term=Kamal-mahttps://loop.frontiersin.org/people/196702/overviewhttps://sites.google.com/site/globesciencement/

Professor Staines is a registered specialist medical practitioner; Fellow of the Australasian Faculty of Occupational and Environmental Medicine (FAFOEM,1986) and Fellow of the Australasian Faculty of Public Health Medicine (FAFPHM,1986). He has served as a Senior Public Health Medical Officer for 35 years, including Gold Coast Public Health Unit, Queensland Health (2003-2014), and Quarantine Medical Officer. In 2013, he was appointed as Clinical Professor and with Professor Sonya Marshall-Gradisnik, he founded the National Centre for Neuroimmunology and Emerging Diseases, Griffith University, with the aim of translating research into clinical settings to improve diagnosis of chronic conditions. In 2019, Professor Staines was awarded the first four-year international Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) full Professorial Fellowship. Professor Staines is an outstanding independent clinical researcher in the field of ME/CFS. His research is successfully supported by competitive/philanthropic national and international funding bodies. Professor Staines has been successful in attaining over $12 million research support funding to explore the clinical presentation and pathophysiology of ME/CFS. His significantly contributed the ME/CSF-related epidemiology, demographic, socioeconomic, and diagnostic ME/CFS guidelines, and development of ME-CFS-targeting treatment.

Professor Marshall-Gradisnik, immunologist, completed her PhD in immunology, Southern Cross University (2003) and completed her Post-Doctoral Research Fellowship with Southern Cross University and the University of Copenhagen (2004-2006). In 2006, Professor Marshall-Gradisnik established her own research group at Bond University (Australia) with primarily focused on Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS)- The Public Health & Neuroimmunology Unit. In 2012, Prof Marshall-Gradisnik relocated, along with her research team, to Griffith University (GU) and with Professor Staines established the National Centre for Neuroimmunology and Emerging Disease (NCNED). In 2018, she was appointed to the National Health and Medical Research ME/CFS Advisory Committee and in the following year (2019), she was awarded the first international ME/CFS Professorial Medical Research Fellowship by the Stafford Fox Medical Research Foundation (2019-2022). Professor Marshall-Gradisnik has sustained success as an independent researcher and has been supported by competitive/philanthropic national and international funding bodies for over ten years. She has been successful in attaining over $12 million research support funding and published 110 publications in the field of immunological and neurological research leading to understanding of the ME/CFS pathophysiology.
Footnotes
Peer review under responsibility of Cairo University.
References
- 1.Organization WH. WHO Coronavirus (COVID-19) Dashboard. https://covid19.who.int/; 2021 (accessed 05/08/2021.).
- 2.Wu C., Chen X., Cai Y., Xia J., Zhou X., Xu S., et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020;180(7):934–943. doi: 10.1001/jamainternmed.2020.0994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mei Q., Wang A.Y., Bryant A., Yang Y., Li M., Wang F., et al. Survival factors and metabolic pathogenesis in elderly patients (≥65) with COVID-19: a multi-center study. Front Med (Lausanne) 2020;7 doi: 10.3389/fmed.2020.595503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhang L., Han C., Zhang S., Duan C., Shang H., Bai T., et al. Diarrhea and altered inflammatory cytokine pattern in severe coronavirus disease 2019: impact on disease course and in-hospital mortality. J Gastroenterol Hepatol. 2021;36(2):421–429. doi: 10.1111/jgh.15166. [DOI] [PubMed] [Google Scholar]
- 5.Tenforde Symptom duration and risk factors for delayed return to usual health among outpatients with COVID-19 in a multistate health care systems network — United States. Morb Mortal Wkly Rep. 2020;69(30):993–998. doi: 10.15585/mmwr.mm6930e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Carfì A, Bernabei R, Landi F, Group ftGAC-P-ACS. Persistent symptoms in patients after acute COVID-19, JAMA 2020;324(6):603–5. [DOI] [PMC free article] [PubMed]
- 7.Marshall M. The lasting misery of coronavirus long-haulers. Nature. 2020:339–341. doi: 10.1038/d41586-020-02598-6. [DOI] [PubMed] [Google Scholar]
- 8.Fu B., Xu X., Wei H. Why tocilizumab could be an effective treatment for severe COVID-19? J Transl Med. 2020;18(1):164. doi: 10.1186/s12967-020-02339-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Greenhalgh T., Knight M., A’Court C., Buxton M., Husain L. Management of post-acute covid-19 in primary care. BMJ. 2020;370 doi: 10.1136/bmj.m3026. [DOI] [PubMed] [Google Scholar]
- 10.Organization WH. A clinical case definition of post COVID-19 condition by a Delphi consensus, 6 October 2021; 2021.
- 11.Ladds E., Rushforth A., Wieringa S., Taylor S., Rayner C., Husain L., et al. Persistent symptoms after Covid-19: qualitative study of 114 “long Covid” patients and draft quality principles for services. BMC Health Serv Res. 2020;20(1):1144. doi: 10.1186/s12913-020-06001-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huang C., Huang L., Wang Y., Li X., Ren L., Gu X., et al. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. The Lancet. 2021;397(10270):220–232. doi: 10.1016/S0140-6736(20)32656-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhao Y.M., Shang Y.M., Song W.B., Li Q.Q., Xie H., Xu Q.F., et al. Follow-up study of the pulmonary function and related physiological characteristics of COVID-19 survivors three months after recovery. EClinicalMedicine. 2020;25 doi: 10.1016/j.eclinm.2020.100463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Augustin M., Schommers P., Stecher M., Dewald F., Gieselmann L., Gruell H., et al. Post-COVID syndrome in non-hospitalised patients with COVID-19: a longitudinal prospective cohort study. Lancet Reg Health – Europe. 2021;6 doi: 10.1016/j.lanepe.2021.100122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Morris G., Puri B.K., Walker A.J., Maes M., Carvalho A.F., Walder K., et al. Myalgic encephalomyelitis/chronic fatigue syndrome: from pathophysiological insights to novel therapeutic opportunities. Pharmacol Res. 2019;148 doi: 10.1016/j.phrs.2019.104450. [DOI] [PubMed] [Google Scholar]
- 16.Tansey C.M., Louie M., Loeb M., Gold W.L., Muller M.P., de Jager J., et al. One-year outcomes and health care utilization in survivors of severe acute respiratory syndrome. Arch Intern Med. 2007;167(12):1312–1320. doi: 10.1001/archinte.167.12.1312. [DOI] [PubMed] [Google Scholar]
- 17.Zhong N.S., Zheng B.J., Li Y.M., Poon, Xie Z.H., Chan K.H., et al. Epidemiology and cause of severe acute respiratory syndrome (SARS) in Guangdong, People's Republic of China, in February, 2003. Lancet. 2003;362(9393):1353–1358. doi: 10.1016/S0140-6736(03)14630-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lam M.H., Wing Y.K., Yu M.W., Leung C.M., Ma R.C., Kong A.P., et al. Mental morbidities and chronic fatigue in severe acute respiratory syndrome survivors: long-term follow-up. Arch Intern Med. 2009;169(22):2142–2147. doi: 10.1001/archinternmed.2009.384. [DOI] [PubMed] [Google Scholar]
- 19.Moldofsky H., Patcai J. Chronic widespread musculoskeletal pain, fatigue, depression and disordered sleep in chronic post-SARS syndrome; a case-controlled study. BMC Neurol. 2011;11:37. doi: 10.1186/1471-2377-11-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gerwyn M., Maes M. Mechanisms explaining muscle fatigue and muscle pain in patients with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): a review of recent findings. Curr Rheumatol Rep. 2017;19(1):1. doi: 10.1007/s11926-017-0628-x. [DOI] [PubMed] [Google Scholar]
- 21.Mao L., Jin H., Wang M., Hu Y., Chen S., He Q., et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77(6):683–690. doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Biehl M., Sese D. Post-intensive care syndrome and COVID-19 - Implications post pandemic. Cleve Clin J Med. 2020 doi: 10.3949/ccjm.87a.ccc055. [DOI] [PubMed] [Google Scholar]
- 23.Sasannejad C., Ely E.W., Lahiri S. Long-term cognitive impairment after acute respiratory distress syndrome: a review of clinical impact and pathophysiological mechanisms. Crit Care. 2019;23(1):352. doi: 10.1186/s13054-019-2626-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kedor C., Freitag H., Meyer-Arndt L., Wittke K., Zoller T., Steinbeis F., et al. Chronic COVID-19 Syndrome and Chronic Fatigue Syndrome (ME/CFS) following the first pandemic wave in Germany – a first analysis of a prospective observational study. medRxiv. 2021 2021.02.06.21249256. [Google Scholar]
- 25.Davis H.E., Assaf G.S., McCorkell L., Wei H., Low R.J., Re’em Y., et al. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. medRxiv. 2020 doi: 10.1016/j.eclinm.2021.101019. 2020.12.24.20248802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Komaroff A.L., Bateman L. Will COVID-19 lead to myalgic encephalomyelitis/chronic fatigue syndrome? Front Med. 2021;7(1132) doi: 10.3389/fmed.2020.606824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Carruthers B.M., van de Sande M.I., De Meirleir K.L., Klimas N.G., Broderick G., Mitchell T., et al. Myalgic encephalomyelitis: International Consensus Criteria. J Intern Med. 2011;270(4):327–338. doi: 10.1111/j.1365-2796.2011.02428.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Eaton-Fitch N., Johnston S.C., Zalewski P., Staines D., Marshall-Gradisnik S. Health-related quality of life in patients with myalgic encephalomyelitis/chronic fatigue syndrome: an Australian cross-sectional study. Qual Life Res. 2020;29(6):1521–1531. doi: 10.1007/s11136-019-02411-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tomas C., Newton J. Metabolic abnormalities in chronic fatigue syndrome/myalgic encephalomyelitis: a mini-review. Biochem Soc Trans. 2018;46(3):547–553. doi: 10.1042/BST20170503. [DOI] [PubMed] [Google Scholar]
- 30.Nicolson G.L. Metabolic syndrome and mitochondrial function: molecular replacement and antioxidant supplements to prevent membrane peroxidation and restore mitochondrial function. J Cell Biochem. 2007;100(6):1352–1369. doi: 10.1002/jcb.21247. [DOI] [PubMed] [Google Scholar]
- 31.Marmion B.P., Sukocheva O., Storm P.A., Lockhart M., Turra M., Kok T., et al. Q fever: persistence of antigenic non-viable cell residues of Coxiella burnetii in the host–implications for post Q fever infection fatigue syndrome and other chronic sequelae. QJM. 2009;102(10):673–684. doi: 10.1093/qjmed/hcp077. [DOI] [PubMed] [Google Scholar]
- 32.Straus S.E., Tosato G., Armstrong G., Lawley T., Preble O.T., Henle W., et al. Persisting illness and fatigue in adults with evidence of Epstein-Barr virus infection. Ann Intern Med. 1985;102(1):7–16. doi: 10.7326/0003-4819-102-1-7. [DOI] [PubMed] [Google Scholar]
- 33.Holmes G.P., Kaplan J.E., Stewart J.A., Hunt B., Pinsky P.F., Schonberger L.B. A cluster of patients with a chronic mononucleosis-like syndrome. Is Epstein-Barr virus the cause? JAMA. 1987;257(17):2297–2302. [PubMed] [Google Scholar]
- 34.Buchwald D., Cheney P.R., Peterson D.L., Henry B., Wormsley S.B., Geiger A., et al. A chronic illness characterized by fatigue, neurologic and immunologic disorders, and active human herpesvirus type 6 infection. Ann Intern Med. 1992;116(2):103–113. doi: 10.7326/0003-4819-116-2-103. [DOI] [PubMed] [Google Scholar]
- 35.Yalcin S., Kuratsune H., Yamaguchi K., Kitani T., Yamanishi K. Prevalence of human herpesvirus 6 variants A and B in patients with chronic fatigue syndrome. Microbiol Immunol. 1994;38(7):587–590. doi: 10.1111/j.1348-0421.1994.tb01827.x. [DOI] [PubMed] [Google Scholar]
- 36.Ablashi D.V., Eastman H.B., Owen C.B., Roman M.M., Friedman J., Zabriskie J.B., et al. Frequent HHV-6 reactivation in multiple sclerosis (MS) and chronic fatigue syndrome (CFS) patients. J Clin Virol. 2000;16(3):179–191. doi: 10.1016/s1386-6532(99)00079-7. [DOI] [PubMed] [Google Scholar]
- 37.Martin W.J. Detection of RNA sequences in cultures of a stealth virus isolated from the cerebrospinal fluid of a health care worker with chronic fatigue syndrome. Case report. Pathobiology. 1997;65(1):57–60. doi: 10.1159/000164104. [DOI] [PubMed] [Google Scholar]
- 38.McGarry F., Gow J., Behan P.O. Enterovirus in the chronic fatigue syndrome. Ann Intern Med. 1994;120(11):972–973. doi: 10.7326/0003-4819-120-11-199406010-00020. [DOI] [PubMed] [Google Scholar]
- 39.Carruthers B.M., Jain A.K., De Meirleir K.L., Peterson D.L., Klimas N.G., Lerner A.M., et al. Myalgic encephalomyelitis/chronic fatigue syndrome. J Chronic Fatigue Syndrome. 2003;11(1):7–115. [Google Scholar]
- 40.Fukuda K., Straus S.E., Hickie I., Sharpe M.C., Dobbins J.G., Komaroff A. The chronic fatigue syndrome: a comprehensive approach to its definition and study. International Chronic Fatigue Syndrome Study Group. Ann Intern Med. 1994;121(12):953–959. doi: 10.7326/0003-4819-121-12-199412150-00009. [DOI] [PubMed] [Google Scholar]
- 41.Ghali A., Richa P., Lacout C., Gury A., Beucher A.-B., Homedan C., et al. Epidemiological and clinical factors associated with post-exertional malaise severity in patients with myalgic encephalomyelitis/chronic fatigue syndrome. J Transl Med. 2020;18(1):246. doi: 10.1186/s12967-020-02419-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chan J.F., Kok K.H., Zhu Z., Chu H., To K.K., Yuan S., et al. Genomic characterization of the 2019 novel human-pathogenic coronavirus isolated from a patient with atypical pneumonia after visiting Wuhan. Emerg Microbes Infect. 2020;9(1):221–236. doi: 10.1080/22221751.2020.1719902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhou R., To K.K.-W., Wong Y.-C., Liu L., Zhou B., Li X., et al. Acute SARS-CoV-2 infection impairs dendritic cell and T cell responses. Immunity. 2020;53(4):864–877.e5. doi: 10.1016/j.immuni.2020.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dong X., Cao Y.Y., Lu X.X., Zhang J.J., Du H., Yan Y.Q., et al. Eleven faces of coronavirus disease 2019. Allergy. 2020;75(7):1699–1709. doi: 10.1111/all.14289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Xu B., Fan C.Y., Wang A.L., Zou Y.L., Yu Y.H., He C., et al. Suppressed T cell-mediated immunity in patients with COVID-19: a clinical retrospective study in Wuhan, China. J Infect. 2020;81(1):e51–e60. doi: 10.1016/j.jinf.2020.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kaneko N., Kuo H.H., Boucau J., Farmer J.R., Allard-Chamard H., Mahajan V.S., et al. Loss of Bcl-6-Expressing T follicular helper cells and germinal centers in COVID-19. Cell. 2020;183(1):143–157. doi: 10.1016/j.cell.2020.08.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Smith M., Honce R., Schultz-Cherry S. Metabolic syndrome and viral pathogenesis: lessons from influenza and coronaviruses. J Virol. 2020;94(18) doi: 10.1128/JVI.00665-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.McGonagle D., Sharif K., O'Regan A., Bridgewood C. The role of cytokines including interleukin-6 in COVID-19 induced pneumonia and macrophage activation syndrome-like disease. Autoimmun Rev. 2020;19(6) doi: 10.1016/j.autrev.2020.102537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Paces J., Strizova Z., Smrz D., Cerny J. COVID-19 and the immune system. Physiol Res. 2020;69(3):379–388. doi: 10.33549/physiolres.934492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fehr A.R., Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol. 2015;1282:1–23. doi: 10.1007/978-1-4939-2438-7_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lambert D.W., Yarski M., Warner F.J., Thornhill P., Parkin E.T., Smith A.I., et al. Tumor necrosis factor-alpha convertase (ADAM17) mediates regulated ectodomain shedding of the severe-acute respiratory syndrome-coronavirus (SARS-CoV) receptor, angiotensin-converting enzyme-2 (ACE2) J Biol Chem. 2005;280(34):30113–30119. doi: 10.1074/jbc.M505111200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Dou H., Feher A., Davila A.C., Romero M.J., Patel V.S., Kamath V.M., et al. Role of adipose tissue endothelial ADAM17 in age-related coronary microvascular dysfunction. Arterioscler Thromb Vasc Biol. 2017;37(6):1180–1193. doi: 10.1161/ATVBAHA.117.309430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Qian M., Shen X., Wang H. The distinct role of ADAM17 in APP proteolysis and microglial activation related to Alzheimer's disease. Cell Mol Neurobiol. 2016;36(4):471–482. doi: 10.1007/s10571-015-0232-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sfera A., Osorio C., Zapata Martín Del Campo C.M., Pereida S., Maurer S., Maldonado J.C., Kozlakidis Z. Endothelial senescence and chronic fatigue syndrome, a COVID-19 based hypothesis. Front Cell Neurosci. 2021;15 doi: 10.3389/fncel.2021.673217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Monteil V., Kwon H., Prado P., Hagelkrüys A., Wimmer R.A., Stahl M., et al. Inhibition of SARS-CoV-2 infections in engineered human tissues using clinical-grade soluble human ACE2. Cell. 2020;181(4):905–913.e7. doi: 10.1016/j.cell.2020.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bellone M., Calvisi S.L. ACE polymorphisms and COVID-19-related mortality in Europe. J Mol Med (Berl) 2020;98(11):1505–1509. doi: 10.1007/s00109-020-01981-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Burrell L.M., Harrap S.B., Velkoska E., Patel S.K. The ACE2 gene: its potential as a functional candidate for cardiovascular disease. Clin Sci (Lond) 2013;124(2):65–76. doi: 10.1042/CS20120269. [DOI] [PubMed] [Google Scholar]
- 58.Pan Y., Wang T., Li Y., Guan T., Lai Y., Shen Y., et al. Association of ACE2 polymorphisms with susceptibility to essential hypertension and dyslipidemia in Xinjiang, China. Lipids Health Dis. 2018;17(1):241. doi: 10.1186/s12944-018-0890-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Delanghe J.R., Speeckaert M.M., De Buyzere M.L. The host's angiotensin-converting enzyme polymorphism may explain epidemiological findings in COVID-19 infections. Clin Chim Acta. 2020;505:192–193. doi: 10.1016/j.cca.2020.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Imai Y., Kuba K., Ohto-Nakanishi T., Penninger J.M. Angiotensin-converting enzyme 2 (ACE2) in disease pathogenesis. Circ J. 2010;74(3):405–410. doi: 10.1253/circj.cj-10-0045. [DOI] [PubMed] [Google Scholar]
- 61.Cao Y., Cai K., Xiong L. Coronavirus disease 2019: A new severe acute respiratory syndrome from Wuhan in China. Acta Virol. 2020;64(2):245–250. doi: 10.4149/av_2020_201. [DOI] [PubMed] [Google Scholar]
- 62.Maloney E.M., Gurbaxani B.M., Jones J.F., de Souza Coelho L., Pennachin C., Goertzel B.N. Chronic fatigue syndrome and high allostatic load. Pharmacogenomics. 2006;7(3):467–473. doi: 10.2217/14622416.7.3.467. [DOI] [PubMed] [Google Scholar]
- 63.Smith A.K., Maloney E.M., Falkenberg V.R., Dimulescu I., Rajeevan M.S. An angiotensin-1 converting enzyme polymorphism is associated with allostatic load mediated by C-reactive protein, interleukin-6 and cortisol. Psychoneuroendocrinology. 2009;34(4):597–606. doi: 10.1016/j.psyneuen.2008.10.022. [DOI] [PubMed] [Google Scholar]
- 64.Shikov A.E., Barbitoff Y.A., Glotov A.S., Danilova M.M., Tonyan Z.N., Nasykhova Y.A., et al. Analysis of the spectrum of ACE2 variation suggests a possible influence of rare and common variants on susceptibility to COVID-19 and severity of outcome. Front Genet. 2020;11 doi: 10.3389/fgene.2020.551220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Gu J., Gong E., Zhang B., Zheng J., Gao Z., Zhong Y., et al. Multiple organ infection and the pathogenesis of SARS. J Exp Med. 2005;202(3):415–424. doi: 10.1084/jem.20050828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zhang J., Wu H., Yao X., Zhang D., Zhou Y., Fu B., et al. Pyroptotic macrophages stimulate the SARS-CoV-2-associated cytokine storm. Cell Mol Immunol. 2021;18(5):1305–1307. doi: 10.1038/s41423-021-00665-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Merad M., Martin J.C. Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nat Rev Immunol. 2020;20(6):355–362. doi: 10.1038/s41577-020-0331-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Park M.D. Macrophages: a Trojan horse in COVID-19? Nat Rev Immunol. 2020;20(6):351. doi: 10.1038/s41577-020-0317-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Züst R., Cervantes-Barragan L., Habjan M., Maier R., Neuman B.W., Ziebuhr J., et al. Ribose 2′-O-methylation provides a molecular signature for the distinction of self and non-self mRNA dependent on the RNA sensor Mda5. Nat Immunol. 2011;12(2):137–143. doi: 10.1038/ni.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Tan Y., Zhang W., Zhu Z., Qiao N., Ling Y., Guo M., et al. Integrating longitudinal clinical laboratory tests with targeted proteomic and transcriptomic analyses reveal the landscape of host responses in COVID-19. Cell Discov. 2021;7(1):42. doi: 10.1038/s41421-021-00274-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Frieman M., Heise M., Baric R. SARS coronavirus and innate immunity. Virus Res. 2008;133(1):101–112. doi: 10.1016/j.virusres.2007.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Totura A.L., Whitmore A., Agnihothram S., Schäfer A., Katze M.G., Heise M.T., et al. Toll-like receptor 3 signaling via TRIF contributes to a protective innate immune response to severe acute respiratory syndrome coronavirus. Infection. mBio 2015,;6(3):e00638–e715. doi: 10.1128/mBio.00638-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Prompetchara E., Ketloy C., Palaga T. Immune responses in COVID-19 and potential vaccines: lessons learned from SARS and MERS epidemic. Asian Pac J Allergy Immunol. 2020;38(1):1–9. doi: 10.12932/AP-200220-0772. [DOI] [PubMed] [Google Scholar]
- 74.Villapol S. Gastrointestinal symptoms associated with COVID-19: impact on the gut microbiome. Transl Res. 2020;226:57–69. doi: 10.1016/j.trsl.2020.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Catanzaro M., Fagiani F., Racchi M., Corsini E., Govoni S., Lanni C. Immune response in COVID-19: addressing a pharmacological challenge by targeting pathways triggered by SARS-CoV-2. Signal Transduct Target Ther. 2020;5(1):84. doi: 10.1038/s41392-020-0191-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Liu R., Liu X., Yuan L., Han H., Shereen M.A., Zhen J., et al. Analysis of adjunctive serological detection to nucleic acid test for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection diagnosis. Int Immunopharmacol. 2020;86 doi: 10.1016/j.intimp.2020.106746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ruan Q., Yang K., Wang W., Jiang L., Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46(5):846–848. doi: 10.1007/s00134-020-05991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Moon C. Fighting COVID-19 exhausts T cells. Nat Rev Immunol. 2020;20(5):277. doi: 10.1038/s41577-020-0304-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.He L., Zhang Q., Zhang Y., Fan Y., Yuan F., Li S. Single-cell analysis reveals cell communication triggered by macrophages associated with the reduction and exhaustion of CD8(+) T cells in COVID-19. Cell Commun Signal. 2021;19(1):73. doi: 10.1186/s12964-021-00754-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Shan H., Lyu Z., Xiao Y., Li C., Wang J., He L., et al. Analysis of the changes of inflammatory cytokine levels in patients with critical coronavirus disease 2019 undergoing invasive mechanical ventilation. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2020;32(9):1051–1055. doi: 10.3760/cma.j.cn121430-20200414-00519. [DOI] [PubMed] [Google Scholar]
- 81.Xu Z., Shi L., Wang Y., Zhang J., Huang L., Zhang C., et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8(4):420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Townsend L., Dyer A.H., Jones K., Dunne J., Mooney A., Gaffney F., et al. Persistent fatigue following SARS-CoV-2 infection is common and independent of severity of initial infection. PLoS ONE. 2020;15(11) doi: 10.1371/journal.pone.0240784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Hornig M., Montoya J.G., Klimas N.G., Levine S., Felsenstein D., Bateman L., et al. Distinct plasma immune signatures in ME/CFS are present early in the course of illness. Sci Adv. 2015;1(1) doi: 10.1126/sciadv.1400121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Bansal A.S., Bradley A.S., Bishop K.N., Kiani-Alikhan S., Ford B. Chronic fatigue syndrome, the immune system and viral infection. Brain Behav Immun. 2012;26(1):24–31. doi: 10.1016/j.bbi.2011.06.016. [DOI] [PubMed] [Google Scholar]
- 85.Mullington J.M., Hinze-Selch D., Pollmächer T. Mediators of inflammation and their interaction with sleep: relevance for chronic fatigue syndrome and related conditions. Ann N Y Acad Sci. 2001;933:201–210. doi: 10.1111/j.1749-6632.2001.tb05825.x. [DOI] [PubMed] [Google Scholar]
- 86.Lucas H.J., Brauch C.M., Settas L., Theoharides T.C. Fibromyalgia–new concepts of pathogenesis and treatment. Int J Immunopathol Pharmacol. 2006;19(1):5–10. [PubMed] [Google Scholar]
- 87.Moneghetti K.J., Skhiri M., Contrepois K., Kobayashi Y., Maecker H., Davis M., et al. Value of circulating cytokine profiling during submaximal exercise testing in myalgic encephalomyelitis/chronic fatigue syndrome. Sci Rep. 2018;8(1):2779. doi: 10.1038/s41598-018-20941-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Corbitt M., Eaton-Fitch N., Staines D., Cabanas H., Marshall-Gradisnik S. A systematic review of cytokines in chronic fatigue syndrome/myalgic encephalomyelitis/systemic exertion intolerance disease (CFS/ME/SEID) BMC Neurology. 2019;19(1):207. doi: 10.1186/s12883-019-1433-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Morris G., Maes M. Mitochondrial dysfunctions in myalgic encephalomyelitis/chronic fatigue syndrome explained by activated immuno-inflammatory, oxidative and nitrosative stress pathways. Metab Brain Dis. 2014;29(1):19–36. doi: 10.1007/s11011-013-9435-x. [DOI] [PubMed] [Google Scholar]
- 90.Rivas J.L., Palencia T., Fernández G., García M. Association of T and NK cell phenotype with the diagnosis of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) Front Immunol. 2018;9:1028. doi: 10.3389/fimmu.2018.01028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Eaton-Fitch N., du Preez S., Cabanas H., Staines D., Marshall-Gradisnik S. A systematic review of natural killer cells profile and cytotoxic function in myalgic encephalomyelitis/chronic fatigue syndrome. Syst Rev. 2019;8(1):279. doi: 10.1186/s13643-019-1202-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Nguyen C.B., Alsøe L., Lindvall J.M., Sulheim D., Fagermoen E., Winger A., et al. Whole blood gene expression in adolescent chronic fatigue syndrome: an exploratory cross-sectional study suggesting altered B cell differentiation and survival. J Transl Med. 2017;15(1):102. doi: 10.1186/s12967-017-1201-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Milrad S.F., Hall D.L., Jutagir D.R., Lattie E.G., Ironson G.H., Wohlgemuth W., et al. Poor sleep quality is associated with greater circulating pro-inflammatory cytokines and severity and frequency of chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME) symptoms in women. J Neuroimmunol. 2017;303:43–50. doi: 10.1016/j.jneuroim.2016.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Maes M., Twisk F.N., Kubera M., Ringel K. Evidence for inflammation and activation of cell-mediated immunity in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): increased interleukin-1, tumor necrosis factor-α, PMN-elastase, lysozyme and neopterin. J Affect Disord. 2012;136(3):933–939. doi: 10.1016/j.jad.2011.09.004. [DOI] [PubMed] [Google Scholar]
- 95.van Eeden C., Khan L., Osman M.S., Cohen Tervaert J.W. Natural killer cell dysfunction and its role in COVID-19. Int J Mol Sci. 2020;21(17) doi: 10.3390/ijms21176351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Cox M.A., Kahan S.M., Zajac A.J. Anti-viral CD8 T cells and the cytokines that they love. Virology. 2013;435(1):157–169. doi: 10.1016/j.virol.2012.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.de Candia P., Prattichizzo F., Garavelli S., Matarese G., Cells T. Warriors of SARS-CoV-2 infection. Trends Immunol. 2021;42(1):18–30. doi: 10.1016/j.it.2020.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Kaech S.M., Wherry E.J. Heterogeneity and cell-fate decisions in effector and memory CD8+ T cell differentiation during viral infection. Immunity. 2007;27(3):393–405. doi: 10.1016/j.immuni.2007.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Saeidi A., Zandi K., Cheok Y.Y., Saeidi H., Wong W.F., Lee C.Y.Q., et al. T-cell exhaustion in chronic infections: reversing the state of exhaustion and reinvigorating optimal protective immune responses. Front Immunol. 2018;9:2569. doi: 10.3389/fimmu.2018.02569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Kahan S.M., Wherry E.J., Zajac A.J. T cell exhaustion during persistent viral infections. Virology. 2015;479–480:180–193. doi: 10.1016/j.virol.2014.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Wherry E.J. T cell exhaustion. Nat Immunol. 2011;12(6):492–499. doi: 10.1038/ni.2035. [DOI] [PubMed] [Google Scholar]
- 102.Zheng H.Y., Zhang M., Yang C.X., Zhang N., Wang X.C., Yang X.P., et al. Elevated exhaustion levels and reduced functional diversity of T cells in peripheral blood may predict severe progression in COVID-19 patients. Cell Mol Immunol. 2020;17(5):541–543. doi: 10.1038/s41423-020-0401-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Wang W., Thomas R., Oh J., Su D.M. Thymic aging may be associated with COVID-19 pathophysiology in the elderly. Cells. 2021;10(3) doi: 10.3390/cells10030628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Sariol A., Perlman S. Lessons for COVID-19 immunity from other coronavirus infections. Immunity. 2020;53(2):248–263. doi: 10.1016/j.immuni.2020.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Rydyznski Moderbacher C., Ramirez S.I., Dan J.M., Grifoni A., Hastie K.M., Weiskopf D., et al. Antigen-specific adaptive immunity to SARS-CoV-2 in acute COVID-19 and associations with age and disease severity. Cell. 2020;183(4):996–1012.e19. doi: 10.1016/j.cell.2020.09.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Wang E.Y., Mao T., Klein J., Dai Y., Huck J.D., Liu F., et al. Diverse functional autoantibodies in patients with COVID-19. medRxiv. 2021 doi: 10.1038/s41586-021-03631-y. 2020.12.10.20247205. [DOI] [PubMed] [Google Scholar]
- 107.Arthur J.M., Forrest J.C., Boehme K.W., Kennedy J.L., Owens S., Herzog C., et al. Development of ACE2 autoantibodies after SARS-CoV-2 infection. PLoS ONE. 2021;16(9) doi: 10.1371/journal.pone.0257016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Wallukat G., Hohberger B., Wenzel K., Fürst J., Schulze-Rothe S., Wallukat A., et al. Functional autoantibodies against G-protein coupled receptors in patients with persistent Long-COVID-19 symptoms. J Transl Autoimmun. 2021;4 doi: 10.1016/j.jtauto.2021.100100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Franke C., Ferse C., Kreye J., Reincke S.M., Sanchez-Sendin E., Rocco A., et al. High frequency of cerebrospinal fluid autoantibodies in COVID-19 patients with neurological symptoms. Brain Behav Immun. 2021;93:415–419. doi: 10.1016/j.bbi.2020.12.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Virhammar J., Nääs A., Fällmar D., Cunningham J.L., Klang A., Ashton N.J., et al. Biomarkers for central nervous system injury in cerebrospinal fluid are elevated in COVID-19 and associated with neurological symptoms and disease severity. Eur J Neurol. 2021;28(10):3324–3331. doi: 10.1111/ene.14703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Sotzny F., Blanco J., Capelli E., Castro-Marrero J., Steiner S., Murovska M., et al. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome – Evidence for an autoimmune disease. Autoimmun Rev. 2018;17(6):601–609. doi: 10.1016/j.autrev.2018.01.009. [DOI] [PubMed] [Google Scholar]
- 112.Wirth K., Scheibenbogen C. A Unifying Hypothesis of the Pathophysiology of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): recognitions from the finding of autoantibodies against ß2-adrenergic receptors. Autoimmun Rev. 2020;19(6) doi: 10.1016/j.autrev.2020.102527. [DOI] [PubMed] [Google Scholar]
- 113.Results of first successful treatment confirmed with two other Long COVID patients. Friedrich-Alexander-Universität Erlangen-Nürnberg, Erlangen-Nürnberg, Germany; 2021.
- 114.Haberland A., Krylova O., Nikolenko H., Göttel P., Dallmann A., Müller J., et al. Aptamer BC 007's affinity to specific and less-specific anti-SARS-CoV-2 neutralizing antibodies. Viruses. 2021;13(5) doi: 10.3390/v13050932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Bynke A., Julin P., Gottfries C.G., Heidecke H., Scheibenbogen C., Bergquist J. Autoantibodies to beta-adrenergic and muscarinic cholinergic receptors in Myalgic Encephalomyelitis (ME) patients - A validation study in plasma and cerebrospinal fluid from two Swedish cohorts. Brain Behav Immun Health. 2020;7 doi: 10.1016/j.bbih.2020.100107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.de Las Heras N., Martín Giménez V.M., Ferder L., Manucha W., Lahera V. Implications of oxidative stress and potential role of mitochondrial dysfunction in COVID-19: therapeutic effects of vitamin D. Antioxidants (Basel) 2020;9(9) doi: 10.3390/antiox9090897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Singh K.K., Chaubey G., Chen J.Y., Suravajhala P. Decoding SARS-CoV-2 hijacking of host mitochondria in COVID-19 pathogenesis. Am J Physiol-Cell Physiol. 2020;319(2):C258–C267. doi: 10.1152/ajpcell.00224.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Burtscher J., Cappellano G., Omori A., Koshiba T., Millet G.P. Mitochondria: in the cross fire of SARS-CoV-2 and immunity. iScience. 2020;23(10):101631. doi: 10.1016/j.isci.2020.101631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Wu X., Li C., Chen S., Zhang X., Wang F., Shi T., et al. Association of body mass index with severity and mortality of COVID-19 pneumonia: a two-center, retrospective cohort study from Wuhan, China. Aging (Albany NY) 2021;13(6):7767–7780. doi: 10.18632/aging.202813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Fischer R., Maier O. Interrelation of oxidative stress and inflammation in neurodegenerative disease: role of TNF. Oxid Med Cell Longev. 2015;2015 doi: 10.1155/2015/610813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Saleh J., Peyssonnaux C., Singh K.K., Edeas M. Mitochondria and microbiota dysfunction in COVID-19 pathogenesis. Mitochondrion. 2020;54:1–7. doi: 10.1016/j.mito.2020.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Wolsk E., Mygind H., Grøndahl T.S., Pedersen B.K., van Hall G. IL-6 selectively stimulates fat metabolism in human skeletal muscle. Am J Physiol Endocrinol Metab. 2010;299(5):E832–E840. doi: 10.1152/ajpendo.00328.2010. [DOI] [PubMed] [Google Scholar]
- 123.Bondia-Pons I., Ryan L., Martinez J.A. Oxidative stress and inflammation interactions in human obesity. J Physiol Biochem. 2012;68(4):701–711. doi: 10.1007/s13105-012-0154-2. [DOI] [PubMed] [Google Scholar]
- 124.Geltink R.I.K., Kyle R.L., Pearce E.L. Unraveling the complex interplay between T cell metabolism and function. Annu Rev Immunol. 2018;36:461–488. doi: 10.1146/annurev-immunol-042617-053019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Gibellini L., De Biasi S., Paolini A., Borella R., Boraldi F., Mattioli M., et al. Altered bioenergetics and mitochondrial dysfunction of monocytes in patients with COVID-19 pneumonia. EMBO Mol Med. 2020;12(12) doi: 10.15252/emmm.202013001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Moro-García M.A., Mayo J.C., Sainz R.M., Alonso-Arias R. Influence of inflammation in the process of T lymphocyte differentiation: proliferative, metabolic, and oxidative changes. Front Immunol. 2018;9:339. doi: 10.3389/fimmu.2018.00339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Assmann N., Finlay D.K. Metabolic regulation of immune responses: therapeutic opportunities. J Clin Invest. 2016;126(6):2031–2039. doi: 10.1172/JCI83005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Pearce E.L., Pearce E.J. Metabolic pathways in immune cell activation and quiescence. Immunity. 2013;38(4):633–643. doi: 10.1016/j.immuni.2013.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.van der Windt G.J., O'Sullivan D., Everts B., Huang S.C., Buck M.D., Curtis J.D., et al. CD8 memory T cells have a bioenergetic advantage that underlies their rapid recall ability. Proc Natl Acad Sci U S A. 2013;110(35):14336–14341. doi: 10.1073/pnas.1221740110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.MacIver N.J., Michalek R.D., Rathmell J.C. Metabolic regulation of T lymphocytes. Annu Rev Immunol. 2013;31:259–283. doi: 10.1146/annurev-immunol-032712-095956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Gupta S.S., Wang J., Chen M. Metabolic reprogramming in CD8(+) T cells during acute viral infections. Front Immunol. 2020;11:1013. doi: 10.3389/fimmu.2020.01013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Schönrich G., Raftery M.J., Samstag Y. Devilishly radical NETwork in COVID-19: oxidative stress, neutrophil extracellular traps (NETs), and T cell suppression. Adv Biol Regul. 2020;77 doi: 10.1016/j.jbior.2020.100741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Castellheim A., Brekke O.L., Espevik T., Harboe M., Mollnes T.E. Innate immune responses to danger signals in systemic inflammatory response syndrome and sepsis. Scand J Immunol. 2009;69(6):479–491. doi: 10.1111/j.1365-3083.2009.02255.x. [DOI] [PubMed] [Google Scholar]
- 134.Sweetman E., Kleffmann T., Edgar C., de Lange M., Vallings R., Tate W. A SWATH-MS analysis of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome peripheral blood mononuclear cell proteomes reveals mitochondrial dysfunction. J Transl Med. 2020;18(1):365. doi: 10.1186/s12967-020-02533-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Anderson G., Maes M. Mitochondria and immunity in chronic fatigue syndrome. Prog Neuropsychopharmacol Biol Psychiatry. 2020;103 doi: 10.1016/j.pnpbp.2020.109976. [DOI] [PubMed] [Google Scholar]
- 136.Morris G., Anderson G., Maes M. Hypothalamic-pituitary-adrenal hypofunction in myalgic encephalomyelitis (ME)/chronic fatigue syndrome (CFS) as a consequence of activated immune-inflammatory and oxidative and nitrosative pathways. Mol Neurobiol. 2017;54(9):6806–6819. doi: 10.1007/s12035-016-0170-2. [DOI] [PubMed] [Google Scholar]
- 137.Suematsu N., Tsutsui H., Wen J., Kang D., Ikeuchi M., Ide T., et al. Oxidative stress mediates tumor necrosis factor-alpha-induced mitochondrial DNA damage and dysfunction in cardiac myocytes. Circulation. 2003;107(10):1418–1423. doi: 10.1161/01.cir.0000055318.09997.1f. [DOI] [PubMed] [Google Scholar]
- 138.Pietraforte D., Vona R., Marchesi A., de Jacobis I.T., Villani A., Del Principe D., et al. Redox control of platelet functions in physiology and pathophysiology. Antioxid Redox Signal. 2014;21(1):177–193. doi: 10.1089/ars.2013.5532. [DOI] [PubMed] [Google Scholar]
- 139.Eslamifar Z., Behzadifard M., Soleimani M., Behzadifard S. Coagulation abnormalities in SARS-CoV-2 infection: overexpression tissue factor. Thromb J. 2020;18(1):38. doi: 10.1186/s12959-020-00250-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Ortega-Paz L., Capodanno D., Montalescot G., Angiolillo D.J. Coronavirus disease 2019–associated thrombosis and coagulopathy: review of the pathophysiological characteristics and implications for antithrombotic management. J Am Heart Assoc. 2021;10(3) doi: 10.1161/JAHA.120.019650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Holden S., Maksoud R., Eaton-Fitch N., Cabanas H., Staines D., Marshall-Gradisnik S. A systematic review of mitochondrial abnormalities in myalgic encephalomyelitis/chronic fatigue syndrome/systemic exertion intolerance disease. J Transl Med. 2020;18(1):290. doi: 10.1186/s12967-020-02452-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Marshall-Gradisnik S., Huth T., Chacko A., Johnston S., Smith P., Staines D. Natural killer cells and single nucleotide polymorphisms of specific ion channels and receptor genes in myalgic encephalomyelitis/chronic fatigue syndrome, Appl. Clin Genet. 2016;9:39–47. doi: 10.2147/TACG.S99405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Dhakal S., Lee Y. Transient receptor potential channels and metabolism. Mol Cells. 2019;42(8):569–578. doi: 10.14348/molcells.2019.0007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Nguyen T., Staines D., Nilius B., Smith P., Marshall-Gradisnik S. Novel identification and characterisation of Transient receptor potential melastatin 3 ion channels on Natural Killer cells and B lymphocytes: effects on cell signalling in Chronic fatigue syndrome/Myalgic encephalomyelitis patients. Biol Res. 2016;49(1):27. doi: 10.1186/s40659-016-0087-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Bousquet J., Czarlewski W., Zuberbier T., Mullol J., Blain H., Cristol J.-P., et al. Potential interplay between Nrf2, TRPA1, and TRPV1 in nutrients for the control of COVID-19. Int Arch Allergy Immunol. 2021;182(4):324–338. doi: 10.1159/000514204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Kuebler W., Jordt S.-E., Liedtke W. COVID-19: urgent reconsideration of lung edema as a preventable outcome: inhibition of TRPV4 as a promising and feasible approach. SSRN. 2020 doi: 10.1152/ajplung.00161.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Quintana A., Hoth M. Mitochondrial dynamics and their impact on T cell function. Cell Calcium. 2012;52(1):57–63. doi: 10.1016/j.ceca.2012.02.005. [DOI] [PubMed] [Google Scholar]
- 148.Zhou X., Chen D., Wang L., Zhao Y., Wei L., Chen Z., et al. Low serum calcium: a new, important indicator of COVID-19 patients from mild/moderate to severe/critical. Biosci Rep. 2020;40(12) doi: 10.1042/BSR20202690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Crespi B., Alcock J. Conflicts over calcium and the treatment of COVID-19. Evol Med Public Health. 2021;9(1):149–156. doi: 10.1093/emph/eoaa046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Lu Y., Li X., Geng D., Mei N., Wu P.Y., Huang C.C., et al. Cerebral micro-structural changes in COVID-19 patients - an MRI-based 3-month follow-up study. EClinicalMedicine. 2020;25 doi: 10.1016/j.eclinm.2020.100484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Lindan C.E., Mankad K., Ram D., Kociolek L.K., Silvera V.M., Boddaert N., et al. Neuroimaging manifestations in children with SARS-CoV-2 infection: a multinational, multicentre collaborative study. Lancet Child Adolescent Health. 2021;5(3):167–177. doi: 10.1016/S2352-4642(20)30362-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Sollini M., Ciccarelli M., Cecconi M., Aghemo A., Morelli P., Gelardi F., et al. Vasculitis changes in COVID-19 survivors with persistent symptoms: an [(18)F]FDG-PET/CT study. Eur J Nucl Med Mol Imaging. 2021;48(5):1460–1466. doi: 10.1007/s00259-020-05084-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Meinhardt J., Radke J., Dittmayer C., Franz J., Thomas C., Mothes R., et al. Olfactory transmucosal SARS-CoV-2 invasion as a port of central nervous system entry in individuals with COVID-19. Nat Neurosci. 2021;24(2):168–175. doi: 10.1038/s41593-020-00758-5. [DOI] [PubMed] [Google Scholar]
- 154.Kempuraj D., Selvakumar G.P., Ahmed M.E., Raikwar S.P., Thangavel R., Khan A., et al. COVID-19, mast cells, cytokine storm, psychological stress, and neuroinflammation. Neuroscientist. 2020;26(5–6):402–414. doi: 10.1177/1073858420941476. [DOI] [PubMed] [Google Scholar]
- 155.Perrin R., Riste L., Hann M., Walther A., Mukherjee A., Heald A. Into the looking glass: post-viral syndrome post COVID-19. Med Hypotheses. 2020;144 doi: 10.1016/j.mehy.2020.110055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Goldstein D.S. The extended autonomic system, dyshomeostasis, and COVID-19. Clin Auton Res. 2020;30(4):299–315. doi: 10.1007/s10286-020-00714-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Kappelmann N., Dantzer R., Khandaker G.M. Interleukin-6 as potential mediator of long-term neuropsychiatric symptoms of COVID-19. Psychoneuroendocrinology. 2021;131 doi: 10.1016/j.psyneuen.2021.105295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Mazza M.G., De Lorenzo R., Conte C., Poletti S., Vai B., Bollettini I., et al. Anxiety and depression in COVID-19 survivors: role of inflammatory and clinical predictors. Brain Behav Immun. 2020;89:594–600. doi: 10.1016/j.bbi.2020.07.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Jason L.A., Islam M.F., Conroy K., Cotler J., Torres C., Johnson M., et al. COVID-19 symptoms over time: comparing long-haulers to ME/CFS. Fatigue: Biomed Health Behav. 2021:1–10. doi: 10.1080/21641846.2021.1922140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.González-Hermosillo J.A., Martínez-López J.P., Carrillo-Lampón S.A., Ruiz-Ojeda D., Herrera-Ramírez S., Amezcua-Guerra L.M., et al. Post-acute COVID-19 symptoms, a potential link with myalgic encephalomyelitis/chronic fatigue syndrome: a 6-month survey in a Mexican cohort. Brain Sci. 2021;11(6) doi: 10.3390/brainsci11060760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Cortes Rivera M., Mastronardi C., Silva-Aldana C.T., Arcos-Burgos M., Lidbury B.A. Myalgic encephalomyelitis/chronic fatigue syndrome: a comprehensive review. Diagnostics (Basel) 2019;9(3) doi: 10.3390/diagnostics9030091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Maksoud R., du Preez S., Eaton-Fitch N., Thapaliya K., Barnden L., Cabanas H., et al. A systematic review of neurological impairments in myalgic encephalomyelitis/ chronic fatigue syndrome using neuroimaging techniques. PLoS ONE. 2020;15(4) doi: 10.1371/journal.pone.0232475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Shan Z.Y., Barnden L.R., Kwiatek R.A., Bhuta S., Hermens D.F., Lagopoulos J. Neuroimaging characteristics of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): a systematic review. J Transl Med. 2020;18(1):335. doi: 10.1186/s12967-020-02506-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Barnden L.R., Shan Z.Y., Staines D.R., Marshall-Gradisnik S., Finegan K., Ireland T., et al. Intra brainstem connectivity is impaired in chronic fatigue syndrome. Neuroimage Clin. 2019;24 doi: 10.1016/j.nicl.2019.102045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Shan Z.Y., Kwiatek R., Burnet R., Del Fante P., Staines D.R., Marshall-Gradisnik S.M., et al. Medial prefrontal cortex deficits correlate with unrefreshing sleep in patients with chronic fatigue syndrome. NMR Biomed. 2017;30(10) doi: 10.1002/nbm.3757. [DOI] [PubMed] [Google Scholar]
- 166.Sykes D.L., Holdsworth L., Jawad N., Gunasekera P., Morice A.H., Crooks M.G. Post-COVID-19 symptom burden: what is long-COVID and how should we manage it? Lung. 2021;199(2):113–119. doi: 10.1007/s00408-021-00423-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Tanaka T., Narazaki M., Masuda K., Kishimoto T. Regulation of IL-6 in immunity and diseases. Adv Exp Med Biol. 2016;941:79–88. doi: 10.1007/978-94-024-0921-5_4. [DOI] [PubMed] [Google Scholar]
- 168.Guzik T.J., Mohiddin S.A., Dimarco A., Patel V., Savvatis K., Marelli-Berg F.M., et al. COVID-19 and the cardiovascular system: implications for risk assessment, diagnosis, and treatment options. Cardiovasc Res. 2020;116(10):1666–1687. doi: 10.1093/cvr/cvaa106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Puntmann V.O., Carerj M.L., Wieters I., Fahim M., Arendt C., Hoffmann J., et al. Outcomes of cardiovascular magnetic resonance imaging in patients recently recovered from coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5(11):1265–1273. doi: 10.1001/jamacardio.2020.3557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Becker R.C. COVID-19-associated vasculitis and vasculopathy. J Thromb Thrombolysis. 2020;50(3):499–511. doi: 10.1007/s11239-020-02230-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Dani M., Dirksen A., Taraborrelli P., Torocastro M., Panagopoulos D., Sutton R., et al. Autonomic dysfunction in 'long COVID': rationale, physiology and management strategies. Clin Med (Lond) 2021;21(1):e63–e67. doi: 10.7861/clinmed.2020-0896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Bozzini S., Albergati A., Capelli E., Lorusso L., Gazzaruso C., Pelissero G., et al. Cardiovascular characteristics of chronic fatigue syndrome. Biomed Rep. 2018;8(1):26–30. doi: 10.3892/br.2017.1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.O'Sullivan O. Long-term sequelae following previous coronavirus epidemics. Clin Med (Lond) 2021;21(1):e68–e70. doi: 10.7861/clinmed.2020-0204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Han H., Ma Q., Li C., Liu R., Zhao L., Wang W., et al. Profiling serum cytokines in COVID-19 patients reveals IL-6 and IL-10 are disease severity predictors. Emerg Microbes Infect. 2020;9(1):1123–1130. doi: 10.1080/22221751.2020.1770129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.D'Amico F., Baumgart D.C., Danese S., Peyrin-Biroulet L. Diarrhea during COVID-19 infection: pathogenesis, epidemiology, prevention, and management. Clin Gastroenterol Hepatol. 2020;18(8):1663–1672. doi: 10.1016/j.cgh.2020.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Wong S.H., Lui R.N., Sung J.J. Covid-19 and the digestive system. J Gastroenterol Hepatol. 2020;35(5):744–748. doi: 10.1111/jgh.15047. [DOI] [PubMed] [Google Scholar]
- 177.Hegazy M.A., Lithy R.M., Abdel-Hamid H.M., Wahba M., Ashoush O.A., Hegazy M.T., et al. COVID-19 disease outcomes: does gastrointestinal burden play a role? Clin Exp Gastroenterol. 2021;14:199–207. doi: 10.2147/CEG.S297428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Guo J.Y., Chen C.Y. How is the digestive system affected by coronavirus disease? J Chin Med Assoc. 2020;83(9):795–796. doi: 10.1097/JCMA.0000000000000371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Babic T., Browning K.N. The role of vagal neurocircuits in the regulation of nausea and vomiting. Eur J Pharmacol. 2014;722:38–47. doi: 10.1016/j.ejphar.2013.08.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Groff A., Kavanaugh M., Ramgobin D., McClafferty B., Aggarwal C.S., Golamari R., et al. Gastrointestinal manifestations of COVID-19: a review of what we know. Ochsner J. 2021;21(2):177–180. doi: 10.31486/toj.20.0086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Yeoh Y.K., Zuo T., Lui G.C.-Y., Zhang F., Liu Q., Li A.Y., et al. Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut. 2021;70(4):698–706. doi: 10.1136/gutjnl-2020-323020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Zuo T., Zhang F., Lui G.C.Y., Yeoh Y.K., Li A.Y.L., Zhan H., et al. Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology. 2020;159(3):944–955.e8. doi: 10.1053/j.gastro.2020.05.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Maes M., Leunis J.C., Geffard M., Berk M. Evidence for the existence of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) with and without abdominal discomfort (irritable bowel) syndrome. Neuro Endocrinol Lett. 2014;35(6):445–453. [PubMed] [Google Scholar]
- 184.Du Preez S., Corbitt M., Cabanas H., Eaton N., Staines D., Marshall-Gradisnik S. A systematic review of enteric dysbiosis in chronic fatigue syndrome/myalgic encephalomyelitis. Systematic Reviews. 2018;7(1):241. doi: 10.1186/s13643-018-0909-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Walsh-Messinger J., Manis H., Vrabec A., Sizemore Bs J., Bishof K., Debidda M., et al. The kids are not alright: a preliminary report of Post-COVID syndrome in university students. J Am Coll Health. 2021:1–7. doi: 10.1080/07448481.2021.1927053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Silva Andrade B., Siqueira S., de Assis Soares W.R., de Souza Rangel F., Santos N.O., Dos Santos Freitas A., et al. Long-COVID and post-COVID health complications: an up-to-date review on clinical conditions and their possible molecular mechanisms. Viruses. 2021;13(4) doi: 10.3390/v13040700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Huang Q., Wu X., Zheng X., Luo S., Xu S., Weng J. Targeting inflammation and cytokine storm in COVID-19. Pharmacol Res. 2020;159 doi: 10.1016/j.phrs.2020.105051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Segatto M., Fittipaldi R., Pin F., Sartori R., Dae Ko K., Zare H., et al. Epigenetic targeting of bromodomain protein BRD4 counteracts cancer cachexia and prolongs survival. Nat Commun. 2017;8(1) doi: 10.1038/s41467-017-01645-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.NIHR, A dynamic review of the evidence around ongoing covid-19 symptoms (often called long covid). https://evidence.nihr.ac.uk/themedreview/living-with-covid19; 2020).
- 190.Williams TC, Burgers WA. SARS-CoV-2 evolution and vaccines: cause for concern? Lancet Respir Med. [DOI] [PMC free article] [PubMed]
- 191.Arnold D.T., Hamilton F.W., Milne A., Morley A.J., Viner J., Attwood M., et al. Patient outcomes after hospitalisation with COVID-19 and implications for follow-up: results from a prospective UK cohort. Thorax. 2021;76(4):399–401. doi: 10.1136/thoraxjnl-2020-216086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Linschoten M., Asselbergs F.W. CAPACITY-COVID: a European Registry to determine the role of cardiovascular disease in the COVID-19 pandemic. Eur Heart J. 2020;41(19):1795–1796. doi: 10.1093/eurheartj/ehaa280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Kazama I. Stabilizing mast cells by commonly used drugs: a novel therapeutic target to relieve post-COVID syndrome? Drug Discov Ther. 2020;14(5):259–261. doi: 10.5582/ddt.2020.03095. [DOI] [PubMed] [Google Scholar]
- 194.Kumar S., Çalışkan D.M., Janowski J., Faist A., Conrad B.C.G., Lange J., et al. Beyond vaccines: clinical status of prospective COVID-19 therapeutics. Front Immunol. 2021;12 doi: 10.3389/fimmu.2021.752227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.de Oliveira B.A., Alves Rodrigues Santos S.A., Menezes Pereira E.W., Nogueira A.B., Vieira Neto A.E., de Melo Júnior J.M.A., et al. Orofacial Antinociceptive effect of nifedipine in rodents is mediated by TRPM3, TRPA1, and NMDA processes. J Oral Facial Pain Headache. 2020;34(2):174–186. doi: 10.11607/ofph.2491. [DOI] [PubMed] [Google Scholar]
- 196.Solaimanzadeh I. Nifedipine and amlodipine are associated with improved mortality and decreased risk for intubation and mechanical ventilation in elderly patients hospitalized for COVID-19. Cureus. 2020;12(5) doi: 10.7759/cureus.8069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Cabanas H., Muraki K., Eaton-Fitch N., Staines D.R., Marshall-Gradisnik S. Potential therapeutic benefit of low dose naltrexone in myalgic encephalomyelitis/chronic fatigue syndrome: role of transient receptor potential melastatin 3 ion channels in pathophysiology and treatment. Front Immunol. 2021;12 doi: 10.3389/fimmu.2021.687806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Cabanas H., Muraki K., Staines D., Marshall-Gradisnik S. Naltrexone restores impaired transient receptor potential melastatin 3 ion channel function in natural killer cells from myalgic encephalomyelitis/chronic fatigue syndrome patients. Front Immunol. 2019;10:2545. doi: 10.3389/fimmu.2019.02545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Dioh W., Chabane M., Tourette C., Azbekyan A., Morelot-Panzini C., Hajjar L.A., et al. Testing the efficacy and safety of BIO101, for the prevention of respiratory deterioration, in patients with COVID-19 pneumonia (COVA study): a structured summary of a study protocol for a randomised controlled trial. Trials. 2021;22(1):42. doi: 10.1186/s13063-020-04998-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Ni W., Yang X., Yang D., Bao J., Li R., Xiao Y., et al. Role of angiotensin-converting enzyme 2 (ACE2) in COVID-19. Crit Care. 2020;24(1):422. doi: 10.1186/s13054-020-03120-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.De Biasi S., Meschiari M., Gibellini L., Bellinazzi C., Borella R., Fidanza L., et al. Marked T cell activation, senescence, exhaustion and skewing towards TH17 in patients with COVID-19 pneumonia. Nat Commun. 2020;11(1):3434. doi: 10.1038/s41467-020-17292-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Yang X., Dai T., Zhou X., Qian H., Guo R., Lei L., et al. Naturally activated adaptive immunity in COVID-19 patients. J Cell Mol Med. 2020;24(21):12457–12463. doi: 10.1111/jcmm.15771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Cliff J.M., King E.C., Lee J.S., Sepúlveda N., Wolf A.S., Kingdon C., et al. Cellular immune function in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) Front Immunol. 2019;10:796. doi: 10.3389/fimmu.2019.00796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Sepúlveda N., Carneiro J., Lacerda E., Nacul L. Myalgic encephalomyelitis/chronic fatigue syndrome as a hyper-regulated immune system driven by an interplay between regulatory T cells and chronic human herpesvirus infections. Front Immunol. 2019;10:2684. doi: 10.3389/fimmu.2019.02684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Kalfaoglu B., Almeida-Santos J., Tye C.A., Satou Y., Ono M. T-cell hyperactivation and paralysis in severe COVID-19 infection revealed by single-cell analysis. Front Immunol. 2020;11 doi: 10.3389/fimmu.2020.589380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Xu T., Zhao J., Wang X., Meng Y., Zhao Z., Bao R., et al. CXCL4 promoted the production of CD4(+)CD25(+)FOXP3(+)treg cells in mouse sepsis model through regulating STAT5/FOXP3 pathway. Autoimmunity. 2020;53(5):289–296. doi: 10.1080/08916934.2020.1777283. [DOI] [PubMed] [Google Scholar]
- 207.Jeannet R., Daix T., Formento R., Feuillard J., François B. Severe COVID-19 is associated with deep and sustained multifaceted cellular immunosuppression. Intensive Care Med. 2020;46(9):1769–1771. doi: 10.1007/s00134-020-06127-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Diao B., Wang C., Tan Y., Chen X., Liu Y., Ning L., et al. Reduction and functional exhaustion of T cells in patients with coronavirus disease 2019 (COVID-19) Front Immunol. 2020;11:827. doi: 10.3389/fimmu.2020.00827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Maes M., Bosmans E., Kubera M. Increased expression of activation antigens on CD8+ T lymphocytes in Myalgic Encephalomyelitis/chronic fatigue syndrome: inverse associations with lowered CD19+ expression and CD4+/CD8+ ratio, but no associations with (auto)immune, leaky gut, oxidative and nitrosative stress biomarkers. Neuro Endocrinol Lett. 2015;36(5):439–446. [PubMed] [Google Scholar]
- 210.Hardcastle S.L., Brenu E.W., Johnston S., Nguyen T., Huth T., Ramos S., et al. Longitudinal analysis of immune abnormalities in varying severities of Chronic Fatigue Syndrome/Myalgic Encephalomyelitis patients. J Transl Med. 2015;13:299. doi: 10.1186/s12967-015-0653-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Alosaimi R.A., Albalwi R. Acute upper limb ischemia in a patient with COVID-19 pneumonia. Clin Med. 2020;7 [Google Scholar]
- 212.Fathi F., Sami R., Mozafarpoor S., Hafezi H., Motedayyen H., Arefnezhad R., et al. Immune system changes during COVID-19 recovery play key role in determining disease severity. Int J Immunopathol Pharmacol. 2020;34 doi: 10.1177/2058738420966497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Fletcher M.A., Zeng X.R., Barnes Z., Levis S., Klimas N.G. Plasma cytokines in women with chronic fatigue syndrome. J Transl Med. 2009;7:96. doi: 10.1186/1479-5876-7-96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Torres-Harding S., Sorenson M., Jason L.A., Maher K., Fletcher M.A. Evidence for T-helper 2 shift and association with illness parameters in chronic fatigue syndrome (CFS) Bull IACFS ME. 2008;16(3):19–33. [PMC free article] [PubMed] [Google Scholar]
- 215.Broderick G., Fuite J., Kreitz A., Vernon S.D., Klimas N., Fletcher M.A. A formal analysis of cytokine networks in chronic fatigue syndrome. Brain Behav Immun. 2010;24(7):1209–1217. doi: 10.1016/j.bbi.2010.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Zhang G., Nie S., Zhang Z., Zhang Z. Longitudinal change of severe acute respiratory syndrome coronavirus 2 antibodies in patients with coronavirus disease 2019. J Infect Dis. 2020;222(2):183–188. doi: 10.1093/infdis/jiaa229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Lerner A.M., Ariza M.E., Williams M., Jason L., Beqaj S., Fitzgerald J.T., et al. Antibody to Epstein-Barr virus deoxyuridine triphosphate nucleotidohydrolase and deoxyribonucleotide polymerase in a chronic fatigue syndrome subset. PLoS ONE. 2012;7(11) doi: 10.1371/journal.pone.0047891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Guenther S., Loebel M., Mooslechner A.A., Knops M., Hanitsch L.G., Grabowski P., et al. Frequent IgG subclass and mannose binding lectin deficiency in patients with chronic fatigue syndrome. Hum Immunol. 2015;76(10):729–735. doi: 10.1016/j.humimm.2015.09.028. [DOI] [PubMed] [Google Scholar]
- 219.Wang H., Fu H., Zhu R., Wu X., Ji X., Li X., et al. BRD4 contributes to LPS-induced macrophage senescence and promotes progression of atherosclerosis-associated lipid uptake. Aging (Albany NY) 2020;12(10):9240–9259. doi: 10.18632/aging.103200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Juno J.A., Tan H.X., Lee W.S., Reynaldi A., Kelly H.G., Wragg K., et al. Humoral and circulating follicular helper T cell responses in recovered patients with COVID-19. Nat Med. 2020;26(9):1428–1434. doi: 10.1038/s41591-020-0995-0. [DOI] [PubMed] [Google Scholar]
- 221.Bradley A.S., Ford B., Bansal A.S. Altered functional B cell subset populations in patients with chronic fatigue syndrome compared to healthy controls. Clin Exp Immunol. 2013;172(1):73–80. doi: 10.1111/cei.12043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Kerr J.R. Epstein-barr virus induced gene-2 upregulation identifies a particular subtype of chronic fatigue syndrome/myalgic encephalomyelitis. Front Pediatr. 2019;7:59. doi: 10.3389/fped.2019.00059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Mensah F.F.K., Armstrong C.W., Reddy V., Bansal A.S., Berkovitz S., Leandro M.J., et al. CD24 expression and B cell maturation shows a novel link with energy metabolism: potential implications for patients with myalgic encephalomyelitis/chronic fatigue syndrome. Front Immunol. 2018;9:2421. doi: 10.3389/fimmu.2018.02421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Luo X.H., Zhu Y., Mao J., Du R.C. T cell immunobiology and cytokine storm of COVID-19. Scand J Immunol. 2021;93(3) doi: 10.1111/sji.12989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Zhao Z., Xie J., Yin M., Yang Y., Ding C., Gao Y., et al. Interleukin-6 and severity of COVID-19 patients in Hefei, China. Med Mal Infect. 2020;50(7):629–631. doi: 10.1016/j.medmal.2020.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Brenu E.W., van Driel M.L., Staines D.R., Ashton K.J., Ramos S.B., Keane J., et al. Immunological abnormalities as potential biomarkers in Chronic Fatigue Syndrome/Myalgic Encephalomyelitis. J Transl Med. 2011;9:81. doi: 10.1186/1479-5876-9-81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Nijs J., Nees A., Paul L., De Kooning M., Ickmans K., Meeus M., et al. Altered immune response to exercise in patients with chronic fatigue syndrome/myalgic encephalomyelitis: a systematic literature review. Exerc Immunol Rev. 2014;20:94–116. [PubMed] [Google Scholar]
- 228.Kerr J.R., Tyrrell D.A. Cytokines in parvovirus B19 infection as an aid to understanding chronic fatigue syndrome. Curr Pain Headache Rep. 2003;7(5):333–341. doi: 10.1007/s11916-003-0031-3. [DOI] [PubMed] [Google Scholar]
- 229.Visser J., Graffelman W., Blauw B., Haspels I., Lentjes E., de Kloet E.R., et al. LPS-induced IL-10 production in whole blood cultures from chronic fatigue syndrome patients is increased but supersensitive to inhibition by dexamethasone. J Neuroimmunol. 2001;119(2):343–349. doi: 10.1016/s0165-5728(01)00400-3. [DOI] [PubMed] [Google Scholar]
- 230.Nakanishi H., Suzuki M., Maeda H., Nakamura Y., Ikegami Y., Takenaka Y., et al. Differential diagnosis of COVID-19: importance of measuring blood lymphocytes, serum electrolytes, and olfactory and taste functions. Tohoku J Exp Med. 2020;252(2):109–119. doi: 10.1620/tjem.252.109. [DOI] [PubMed] [Google Scholar]
- 231.Groven N., Fors E.A., Reitan S.K. Patients with fibromyalgia and chronic fatigue syndrome show increased hsCRP compared to healthy controls. Brain Behav Immun. 2019;81:172–177. doi: 10.1016/j.bbi.2019.06.010. [DOI] [PubMed] [Google Scholar]
- 232.Strawbridge R., Sartor M.L., Scott F., Cleare A.J. Inflammatory proteins are altered in chronic fatigue syndrome-A systematic review and meta-analysis. Neurosci Biobehav Rev. 2019;107:69–83. doi: 10.1016/j.neubiorev.2019.08.011. [DOI] [PubMed] [Google Scholar]
- 233.Freeman T.L., Swartz T.H. Targeting the NLRP3 inflammasome in severe COVID-19. Front Immunol. 2020;11:1518. doi: 10.3389/fimmu.2020.01518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Moreno Fernández-Ayala D.J., Navas P., López-Lluch G. Age-related mitochondrial dysfunction as a key factor in COVID-19 disease. Exp Gerontol. 2020;142 doi: 10.1016/j.exger.2020.111147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Wood E., Hall K.H., Tate W. Role of mitochondria, oxidative stress and the response to antioxidants in myalgic encephalomyelitis/chronic fatigue syndrome: a possible approach to SARS-CoV-2 'long-haulers'? Chronic Dis Transl Med. 2021;7(1):14–26. doi: 10.1016/j.cdtm.2020.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Mandarano A.H., Maya J., Giloteaux L., Peterson D.L., Maynard M., Gottschalk C.G., et al. Myalgic encephalomyelitis/chronic fatigue syndrome patients exhibit altered T cell metabolism and cytokine associations. J Clin Invest. 2020;130(3):1491–1505. doi: 10.1172/JCI132185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Guarnotta V., Ferrigno R., Martino M., Barbot M., Isidori A.M., Scaroni C., et al. Glucocorticoid excess and COVID-19 disease. Rev Endocr Metab Disord. 2020:1–12. doi: 10.1007/s11154-020-09598-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Tomazini B.M., Maia I.S., Cavalcanti A.B., Berwanger O., Rosa R.G., Veiga V.C., et al. Effect of dexamethasone on days alive and ventilator-free in patients with moderate or severe acute respiratory distress syndrome and COVID-19: the CoDEX randomized clinical trial. JAMA. 2020;324(13):1307–1316. doi: 10.1001/jama.2020.17021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Visser J., Blauw B., Hinloopen B., Brommer E., de Kloet E.R., Kluft C., et al. CD4 T lymphocytes from patients with chronic fatigue syndrome have decreased interferon-gamma production and increased sensitivity to dexamethasone. J Infect Dis. 1998;177(2):451–454. doi: 10.1086/517373. [DOI] [PubMed] [Google Scholar]