Abstract
Our previous research has demonstrated a promising capacity of streptomycetes isolated from red soils to produce novel secondary metabolites, most of which, however, remain to be explored. Co-culturing with mycolic acid-containing bacteria (MACB) has been used successfully in activating the secondary metabolism in Streptomyces. Here, we co-cultured 44 strains of red soil-derived streptomycetes with four MACB of different species in a pairwise manner and analyzed the secondary metabolites. The results revealed that each of the MACB strains induced changes in the metabolite profiles of 35–40 streptomycetes tested, of which 12–14 streptomycetes produced “new” metabolites that were not detected in the pure cultures. Moreover, some of the co-cultures showed additional or enhanced antimicrobial activity compared to the pure cultures, indicating that co-culture may activate the production of bioactive compounds. From the co-culture-induced metabolites, we identified 49 putative new compounds. Taking the co-culture of Streptomyces sp. FXJ1.264 and Mycobacterium sp. HX09-1 as a case, we further explored the underlying mechanism of co-culture activation and found that it most likely relied on direct physical contact between the two living bacteria. Overall, our results verify co-culture with MACB as an effective approach to discover novel natural products from red soil-derived streptomycetes.
Keywords: Streptomyces, co-culture, mycolic acid-containing bacteria (MACB), secondary metabolites (SMs), activation of natural products
1. Introduction
Natural products (NPs), or their semi-synthetic derivatives, are important sources of lead compounds in drug discovery [1,2]. More than 50% of clinically used antibiotics were derived from filamentous Gram-positive bacteria of the genus Streptomyces [3,4]. Analysis of the genome sequences of Streptomyces and related genera revealed that they may contain a great variety of secondary metabolite (SM) biosynthetic gene clusters (BGCs) encoding novel NPs [5,6]. However, most of these BGCs remain silent under laboratory conditions, thus triggering research in the sense of developing activating strategies for the genome mining of microbial NPs [7,8,9].
Several previous studies reported that microbial cryptic BGCs could be activated by co-culturing the host strains with other species [10,11,12,13], partially due to mimicking the in situ microbial interactions in the original environment where microorganisms coexist [14]. However, traditional co-culture methods require large-scale screening to find ideal microbial combinations, and thus, is laborious and difficult to apply at scale. In recent years, it has been shown that co-cultures with mycolic acid-containing bacteria (MACB) widely activate cryptic SM-BGCs in streptomycetes [15]. Hitherto, around 40 novel NPs have been discovered from Streptomyces, and rare actinobacteria by co-culture with MACB strains. These compounds comprise a variety of chemical scaffolds and bioactivities, validating this SM-BGC activation method of co-culture [16,17,18].
Red soils are widely distributed in tropic and subtropical areas of southern China. These soils are acidic, oligotrophic, and rich in iron and aluminum oxides, and thus provide ideal habitats for acidophilic actinobacteria [19]. Our recent studies have shown that red soil-derived streptomycetes are prolific NP producers [19], and have identified several NPs with novel scaffolds or modifications from these strains, as exemplified by azolemycins [20], NC-1 [21], and mycemycins [22]. Meanwhile, the in silico genome-mining of red soil-derived streptomycetes also reveals that these strains contain numerous unidentified SM-BGCs, the products of which remain to be unraveled.
To activate the silent SM-BGCs of red soil-derived streptomycetes for NPs discovery, we selected 44 bioactive Streptomyces isolates and co-cultured them with four MACB of different species in a pairwise manner. Metabolites of the co-cultures were subjected to multi-spectroscopic analyses and bioactivity assay, which showed that the MACB strains effectively activated secondary metabolism in most of the streptomycetes. We also tried to explore the underlying mechanism of co-culture activation. Results of the study gain a deep insight into the NP biosynthetic potential of red soil-derived streptomycetes.
2. Materials and Methods
2.1. Strains and Media
The strains used in this work are listed in Table S1. Eight MACB were isolated from soils collected in Haixi Mongolian and Tibetan Autonomous Prefecture, China [23], and two MACB and all Streptomyces strains were from red soils collected in Jiangxi Province, China [19,24]. All these strains were preliminarily identified by 16S rRNA gene sequencing in our previous studies [19,23,24]. Three indicator strains from different phyla were used for antimicrobial activity assay: Micrococcus luteus CGMCC 1.2567 (Gram-positive bacterium) and Trichoderma viride CGMCC 3.1913 (fungus) were obtained from the China General Microbiological Culture Collection Center (CGMCC), and extended-spectrum β-lactamase (ESBL)-producing Escherichia coli 4-1 (Gram-negative bacterium) was obtained from the Weifang Medical University, Shandong Province, China.
A YGGS medium (glucose 5.0 g, soluble starch 20.0 g, glycerin 20.0 g, yeast extract 3.0 g in 1 L dd-H2O, pH 7.2) [25] was used for the co-culture and pure culture of strains. A TSB medium (pancreatic digest of casein 17.0 g, papaic digest of soybean 3.0 g, dextrose 2.5 g, NaCl 5.0 g, K2HPO4 2.5 g in 1 L dd-H2O, pH 7.1–7.5) was used for seed culture. A GYM agar (yeast extract 4.0 g, malt extract 10.0 g, glucose 4.0 g, CaCO3 2.0 g, agar 15.0 g in 1 L dd-H2O) was used for recovering strains from glycerol stocks. An LB agar (tryptone 10.0 g, yeast extract 5.0 g, NaCl 10.0 g, agar 15.0 g in 1 L dd-H2O) and PDA (glucose 20.0 g, potato powder 6.0 g, agar 15.0 g in 1 L dd-H2O) were used to culture the indicator bacteria and fungus, respectively.
2.2. Co-Culture and Pure Culture of Strains
After the incubation of the strains on a GYM plate for 2–3 days, an agar block of about 1 cm2 with bacterial lawn was cut out and transferred into a 250 mL shake flask containing 50 mL of TSB medium for seed culture. The seeds of Streptomyces and MACB were cultured at 28 °C on a rotary shaker at 160 rpm for 3 and 2 days, respectively. Then, 3 mL of Streptomyces and 1 mL of MACB seed cultures were co-transferred into a 250 mL flask containing 100 mL of YGGS medium and fermented at 28 °C, 220 rpm for 7 days. Pure culture controls were performed similarly but with single strains.
Each experiment was repeated in triplicate in this study.
2.3. SM Extraction, Isolation, and Analysis
The resulting cultures were collected and extracted three times with an equal volume of ethyl acetate. The extracts were combined and concentrated in vacuo to evaporate the solvent, and the residue was re-dissolved in 1 mL of methanol. An HPLC analysis was carried out with a Shimadzu SPD-M20A HPLC system, using a Waters Xbridge ODS column (4.6 × 150 mm, 5 μm) with a linear gradient of MeOH/H2O (see Table S2). The injection volume of the sample was 20 μL. The Dionex 3000 RS system was used to set the temperature at 30 °C and the flow rate at 1.0 mL/min; the elution curves of metabolites were monitored at 220, 254, and 300 nm, respectively. Differences in the secondary metabolism between the co-cultures and pure cultures were determined by comparing their HPLC profiles based on the retention time and UV absorption spectra of peaks. Metabolites corresponding to the differential HPLC peaks were then collected and subjected to UHPLC-HRMS (Waters Xevo G2 quadrupole time of flight-ultra performance liquid chromatography, and mass spectra scanning from 100 to 2000 atomic mass units) to obtain their accurate molecular weights (MWs). The resultant mass spectrum data were analyzed by the Mass Lynx v 4.1 software system.
Compounds were identified by the comparison of MWs, UV spectra, and retention times with published chemical data from standard databases (Dictionary of Natural Products [DNP] on DVD, version 22.2 and on web, version 30.1 [http://dnp.chemnetbase.com/, accessed on 1 September 2021]; ChemSpider [http://www.chemspider.com/, accessed on 2 September 2021]) and references. The activated metabolites with characteristic information unmatched with that in the databases were inferred as putative new products. Some of these compounds were subjected to nuclear magnetic resonance (NMR) spectroscopic analysis (Bruker AVIII 500 MHz NMR spectrometer, Bruker, Karlsruhe, Germany) for further structure elucidation.
2.4. Bioactivity Assay
Bioactivities of the fermentation extracts were tested against M. luteus, ESBL-producing E. coli, and T. viride using agar-well diffusion assay. Twenty μL of each extract were added into a punched hole (7 mm in diameter) in LB/PDA plates containing indicator strains. The plates were then cultured at 37 °C for 12 h for bacterial indicators or at 28 °C for 48 h for the fungus. Antimicrobial activity was estimated by measuring the diameter of the inhibition zones: positive (7 mm < diameter ≤ 9 mm) and strongly positive (diameter > 9 mm).
2.5. Non-Contact Co-Culture of Streptomyces sp. FXJ1.264 and Mycobacterium sp. HX09-1
The co-culture was carried out in a device of two connected culture compartments separated by a 0.22-μm polyether sulfone (PES) membrane (Figure S1). Each of the compartments contained 50 mL YGSS medium and were inoculated with 3 mL seed culture of S. sp. FXJ1.264 or 1 mL seed culture of M. sp. HX09-1. The device only allowed substance exchange between the compartments, but the cells of the two strains could not contact each other. For the control groups, only one compartment in the device was inoculated, with either a single strain or two strains mixed. The device was fixed on a shaker and the strains were fermented at 28 °C, 220 rpm for 7 days. The fermentation metabolites from each compartment were analyzed using the method described in Section 2.3.
2.6. Co-Culture of S. sp. FXJ1.264 and Heat-killed M. sp. HX09-1
M. sp. HX09-1 was cultured in 250 mL flasks each containing 50 mL of the YGGS medium for 2 or 7 days. The culture broths were then heated at 121 °C for 20 min to kill the cells. After cooling down to room temperature, the flask was added with 50 mL fresh YGGS medium and 3 mL seed culture of S. sp. FXJ1.264, and the resulting culture was incubated at 28 °C, 220 rpm for 7 days. The fermentation metabolites were analyzed as above.
3. Results
3.1. Preliminary Evaluation of the Activation Ability of MACB and Selection of Red Soil-Derived Streptomycetes
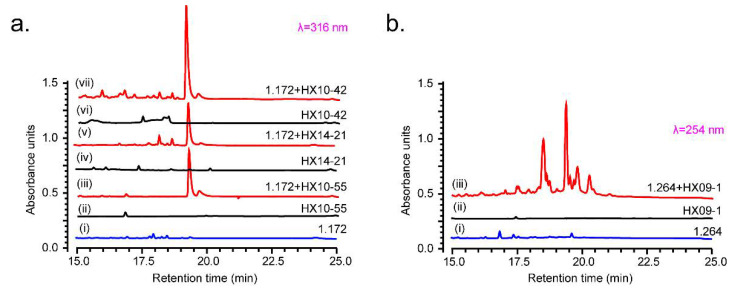
Ten MACB candidates (listed in Table S1) and two known NPs producer strains that were isolated from red soil, Streptomyces spp. FXJ1.172 and FXJ1.264 [19], were used for preliminary co-culture evaluation. The HPLC profiles of metabolites showed that three of the MACB (Mycobacterium sp. HX10-42, Nocardia sp. HX14-21, and Rhodococcus sp. HX10-55) obviously activated S. sp. FXJ1.172 to produce new peaks compared to their individual pure cultures (Figure 1a). Meanwhile, a series of unique peaks were detected in the combined culture of S. sp. FXJ1.264 and Mycobacterium sp. HX09-1 (Figure 1b). The other six MACB did not exhibit activation ability when co-cultured with the two Streptomyces strains. Therefore, the above four strains of MACB were chosen for subsequent co-culture. In addition, based on the antimicrobial activity and 16S rRNA gene similarity of the red soil-derived Streptomyces strains [19,24], 44 bioactive streptomycetes with abundant diversity were preferentially selected for this study (Table S1). The selected MACB and streptomycetes thus formed 176 (4 × 44) co-culture pairs.
Figure 1.
Preliminary evaluation of the activation ability of mycolic acid-containing bacteria (MACB). (a) HPLC analysis of the fermentation extracts of S. sp. FXJ1.172 co-cultured with different MACB and the extracts of their pure cultures; (b) HPLC analysis of the fermentation extracts of S. sp. FXJ1.264, M. sp. HX09-1, and their combined culture.
3.2. Co-Culture with MACB Changed the SM Profiles of Streptomycetes
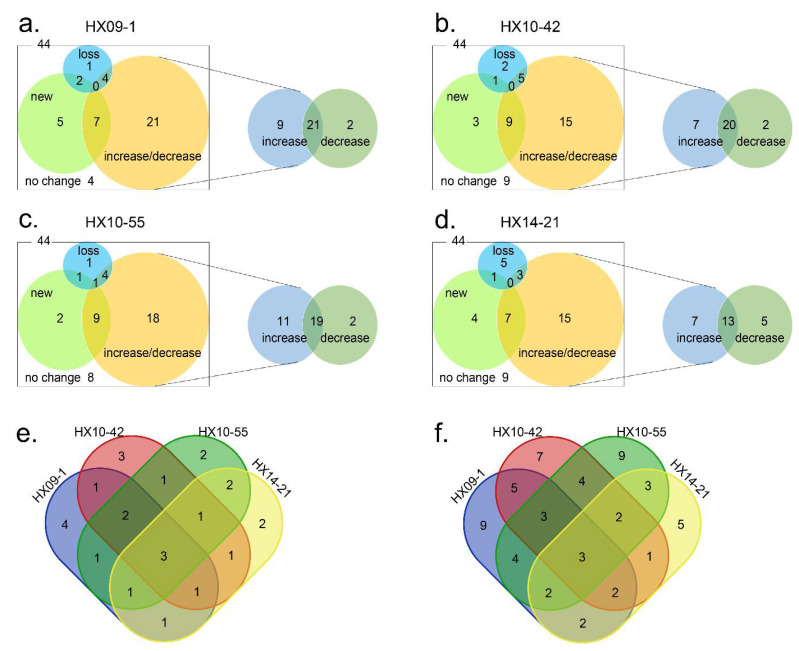
HPLC analysis showed changes in SM profiles of most (82.9%, 146/176) of the co-cultures compared to the pure culture counterparts (Figure 2 and Table 1). The differences of metabolites were characterized by four patterns of HPLC peaks: the increase/decrease in metabolite production (the integral area of peaks changed by more than 30%), appearance of new metabolite peaks, loss of original peaks, and no change. The comparison results were mostly a combination of the above patterns due to the complex secondary metabolism in Streptomyces (Figure 2). For example, compared to the pure cultures, co-culture with M. sp. HX09-1 changed the secondary metabolism of 40 Streptomyces strains. Among them, 30 and 23 strains enhanced and decreased the production of some original metabolites, respectively, 14 strains produced new metabolites that were not found in the pure cultures, and seven strains lost some metabolites (Figure 2a). M. sp. HX10-42, R. sp. HX10-55, and N. sp. HX14-21 exhibited similar activation ability (Figure 2b–d) (Table 1). In summary, 29.5% (52/176) of the co-cultures activated the production of “new” SMs and 60.8% (107/176) of the co-cultures increased the production of original metabolites (Table 1). For all four MACB strains, the proportion of positive impact (increase in production and/or appearance of “new” metabolites) on the secondary metabolism of Streptomyces is significantly higher (p < 0.01) than that of negative impact (decrease in production and/or loss of metabolites), as shown in Table 1. Only four of the co-cultured streptomycetes were not activated by any of the MACB. These results indicated that the four MACB strains effectively induced NPs biosynthesis or enhanced their yields in the co-cultured Streptomyces strains.
Figure 2.
Venn diagrams showing effects of co-culture with four different MACB on the SM profiles of 44 red soil-derived streptomycetes. Pure cultures were used as controls. The numbers represent the number of streptomycetes or co-culture pairs. (a–d) Summarization of the effects of co-culture for each MACB. Each HPLC profile was classified according to the aforementioned categories: increase/decrease, the yields of original metabolites produced in pure cultures increased/decreased in the co-cultures (the integral area of HPLC peaks changed by more than 30%); new, “new” metabolites with unique retention time and/or UV absorption that exclusively appeared in the co-cultures; loss, original pure culture metabolites lost in the co-cultures; and no change, no difference in SM profiles was detected between the co-cultures and pure cultures. The increase/decrease part is subclassified into increase, decrease, and both increase and decrease in the right panel. (e) Overlaps of the effects between MACB on inducing “new” metabolites. (f) Overlaps of the effects between MACB on increasing the yields of original metabolites.
Table 1.
Proportions of different change patterns in SM profiles of the co-cultures compared to the pure culture counterparts. The numbers represent the number of co-culture pairs.
| Pattern(s) | M. sp. HX09-1 | M. sp. HX10-42 | R. sp. HX10-55 | N. sp. HX14-21 | Total |
|---|---|---|---|---|---|
| Increase | 30/44 | 27/44 | 30/44 | 20/44 | 107/176 |
| New | 14/44 | 13/44 | 13/44 | 12/44 | 52/176 |
| Increase/New | 39/44 | 31/44 | 33/44 | 25/44 | 128/176 |
| Decrease | 23/44 | 22/44 | 21/44 | 18/44 | 84/176 |
| Loss | 7/44 | 8/44 | 7/44 | 9/44 | 31/176 |
| Decrease/Loss | 26/44 | 26/44 | 25/44 | 22/44 | 99/176 |
| Change | 40/44 | 35/44 | 36/44 | 35/44 | 146/176 |
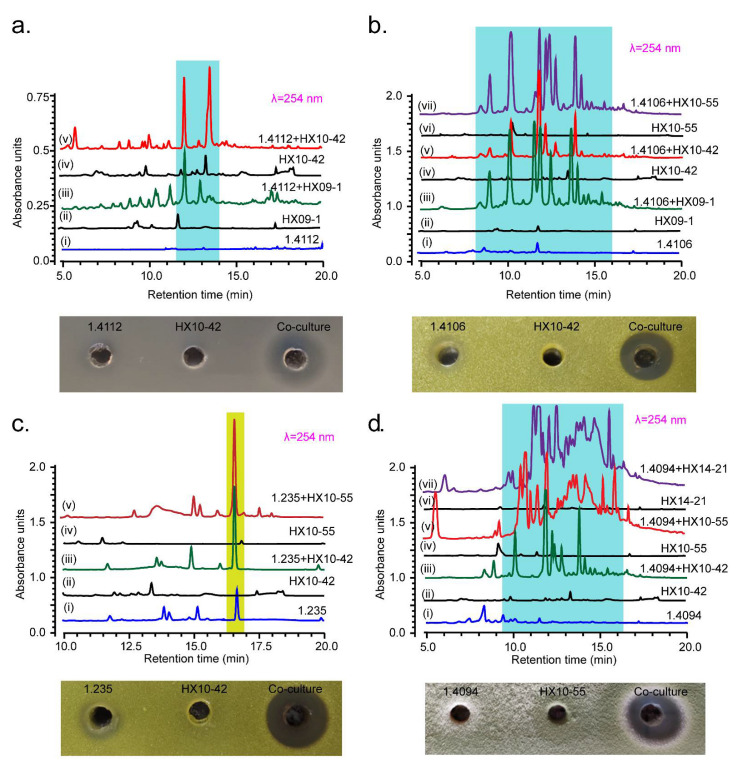
We noticed some overlaps in the activation effects (Figure 2e,f). That is, the same Streptomyces strain could be activated by different MACB to produce similar new HPLC peaks, or to increase the yields of similar original metabolites, as exemplified by Streptomyces spp. FXJ1.4112, FXJ1.4106, FXJ1.235, and FXJ1.4094 (Figure 3a–d). This result suggests that Streptomyces might use some general weapons when facing different MACB, and that the shared mycolic acids present in different MACB might be one of the key substances to change the secondary metabolism of Streptomyces.
Figure 3.
HPLC analysis of changes in SM spectra and agar-well diffusion test of emerging antimicrobial activity in the co-cultures compared to the pure cultures. (a) S. sp. FXJ1.4112 co-cultured with MACB produced “new” metabolites (blue shadowed) and strong antibacterial activity against E. coli; (b) S. sp. FXJ1.4106 co-cultured with MACB produced a series of “new” metabolites (blue shadowed) and strong antibacterial activity against M. luteus; (c) S. sp. FXJ1.235 co-cultured with MACB produced a higher yield of the original metabolite (yellow shadowed; the integral area of HPLC peaks was over twice that of control) and strong antibacterial activity against M. luteus; (d) FXJ1.4094 co-cultured with MACB produced a series of “new” metabolites (blue shadowed) and strong antifungal activity against T. viride. Antimicrobial activity was estimated by measuring the diameter of the inhibition zones: positive (7 mm [hole diameter] < diameter ≤ 9 mm) and strongly positive (diameter > 9 mm).
3.3. Changes of Antimicrobial Activity in the Co-Cultures
Besides analyzing the SM profiles (HPLC detection), we also used agar-well diffusion assay to monitor the difference of antimicrobial activity between the co-culture and pure culture extracts. Among the 176 co-culture pairs, 21 pairs exhibited distinguishing antimicrobial activities compared to the pure culture controls (Table 2), with 15 pairs displaying new activities and the other 6 pairs showing improved activities. Intriguingly, no decrease or loss of pre-existing antimicrobial activities was observed in the co-cultures.
Table 2.
Differences of antimicrobial activity between the pure cultures and co-cultures.
| Streptomycete | Mycolic Acid-Containing Bacteria | Antimicrobial Activity | |||||
|---|---|---|---|---|---|---|---|
| M. sp. HX09-1 | M. sp. HX10-42 | R. sp. HX10-55 | N. sp. HX14-21 | EC | ML | TV | |
| FXJ1.235 | |||||||
| + | |||||||
| + | |||||||
| FXJ1.4012 | |||||||
| + | |||||||
| FXJ1.4014 | |||||||
| + | |||||||
| FXJ1.4034 | |||||||
| + | |||||||
| + | |||||||
| FXJ1.4037 | |||||||
| + | |||||||
| FXJ1.4094 | |||||||
| + | |||||||
| + | |||||||
| + | |||||||
| FXJ1.4104 | |||||||
| + | |||||||
| FXJ1.4106 | |||||||
| + | |||||||
| + | |||||||
| + | |||||||
| FXJ1.4112 | |||||||
| + | |||||||
| + | |||||||
| FXJ1.535 | |||||||
| + | |||||||
| + | |||||||
| FXJ1.907 | |||||||
| + | |||||||
| + | |||||||
| + | |||||||
Note: “+”, co-culture with mycolic acid-containing bacteria;  , negative;
, negative;  , positive;
, positive;  , strongly positive; EC, multi-drug resistant Escherichia coli 4-1; ML, Micrococcus luteus CGMCC 1.2561; TV, Trichoderma viride CGMCC 3.1913. Antimicrobial activity was estimated by measuring the diameter of the inhibition zones: positive (7 mm [hole diameter] < diameter ≤ 9 mm) and strongly positive (diameter > 9 mm).
, strongly positive; EC, multi-drug resistant Escherichia coli 4-1; ML, Micrococcus luteus CGMCC 1.2561; TV, Trichoderma viride CGMCC 3.1913. Antimicrobial activity was estimated by measuring the diameter of the inhibition zones: positive (7 mm [hole diameter] < diameter ≤ 9 mm) and strongly positive (diameter > 9 mm).
The 21 co-culture pairs mentioned above all showed the “Increase/New” patterns of SM changes based on their HPLC profiling (Table 1). Thus, we speculated that the additional bioactivities might be attributed to the induced or increased production of metabolites in the combined cultures. Indeed, changes of antimicrobial activity in some co-cultures were correlated to the detected alterations of SM spectra. For instance, the co-culture of S. sp. FXJ1.4112 and M. sp. HX10-42 displayed antibacterial activity against E. coli, which was not observed in the pure cultures (Figure 3a and Table 2); correspondingly, two novel product peaks were detected in the HPLC profile of co-culture extracts (Figure 3a). After isolation and purification, the metabolites corresponding to the two peaks (further deduced as collinomycin analogs) showed antibacterial activity against E. coli. Similar results were also observed in the co-cultures of S. sp. FXJ1.4106 and MACB (Figure 3b and Table 2). In addition, co-culture also improved antibacterial activity of some streptomycetes. For example, the pure culture of S. sp. FXJ1.235 had only weak antibacterial activity against M. luteus, while the co-cultures with M. sp. HX10-42 and R. sp. HX10-55 had obvious antibacterial activity against M. luteus (Figure 3c and Table 2). The striking increase (2.08 and 2.70 times) in the yield (integral area of HPLC peaks) of the original antibacterial product in the co-cultures might contribute to the resultant enhanced activity (Figure 3c). These results confirm that co-culture can stimulate Streptomyces to produce bioactive NPs. However, not all the changes of activity in the co-cultures could be correlated to the changes of HPLC profiles. The co-culture of S. sp. FXJ1.4094 and N. sp. HX10-55 exhibited obvious antifungal activity against T. viride, which was contrary to the result of their pure cultures (Figure 3d and Table 2). But the product components corresponding to the new peaks in the HPLC profile of co-culture extracts (Figure 3d) showed no antifungal activity. Similar results were also observed in co-culturing Streptomyces spp. FXJ1.4014, FXJ1.4034, and FXJ1.4037 with MACB. These examples highlight the importance of testing antimicrobial activity as a complementary approach to secondary metabolism comparison, as metabolite differences may be overlooked if the differential compounds display low or no UV absorption in HPLC profiling.
3.4. Co-Culture of Streptomyces and MACB Is a Reliable Source of New Compounds
After UHPLC-HRMS analysis of the “new” metabolites appeared in the co-cultures, a total of 49 compounds were identified as putative novel NPs based on their unique MWs and/or UV spectra compared with those in databases and references (Table 3). Among them, 33 (67.3%) compounds were considered as NPs with new skeletons (structural class unknown) because neither their MWs nor UV spectra matched any NPs recorded in the databases or references.
Table 3.
Putative novel products from co-cultures of the streptomycetes and MACB.
| Co-Culture | Compound(s) Induced | Structural Class | Chemical Data | |
|---|---|---|---|---|
| UV (MeOH) | MS (m/z) | |||
| FXJ1.172 + HX14-21 | 2 putative novel compounds | Unknown | 221 | [M + H] + 603.3187 |
| Unknown | 221 | [M + H] + 605.2964 | ||
| FXJ1.235 + HX10-42 | 2 putative novel compounds | Unknown | 244, 256, 265, 294, 307, 340 | [M + H] + 304.0403 |
| FXJ1.235 + HX10-55 | Unknown | 244, 256, 265, 294, 307, 340 | [M + H] + 445.0760 | |
| FXJ1.264 + HX09-1 | 6 putative novel compounds | Polyketide | 194, 251, 285, 376 | [M − H] − 371.1292 |
| Polyketide | 194, 251, 285, 376 | MS no ion current | ||
| Polyketide | 194, 251, 285, 376 | [M − H] − 385.1433 | ||
| Polyketide | 194, 251, 285, 376 | [M − H] − 743.2671 (dimer of 371.1292) | ||
| Polyketide | 194, 251, 285, 376 | [M − H] − 743.2645 (dimer of 371.1292) | ||
| Polyketide | 194, 251, 285, 376 | [M − H] − 743.2634 (dimer of 371.1292) | ||
| FXJ1.4038 + HX09-1 | 1 putative novel compound | Unknown | 201, 249, 336 | [M + H] + 347.0921 |
| FXJ1.4059 + HX10-42 | 1 putative novel compound | Unknown | 199, 219, 276 | [M + H] + 319.0830 |
| FXJ1.4064 + HX10-55 | 3 putative novel compounds | Unknown | 205, 289 | [M + H] + 477.3209 |
| Unknown | 205, 294 | [M + H] + 589.3013 | ||
| Unknown | 218, 275 | [M + H] + 617.3292 | ||
| FXJ1.4075 + HX10-55 | 2 putative novel compounds | Unknown | 248, 343 | [M + H] + 253.1188 |
| Unknown | 221, 311, 351 | [M + H] + 569.0939 | ||
| FXJ1.4087 + HX10-55 | 1 putative novel compound | Unknown | 197, 224, 272, 336 | [M + H] + 288.1239 |
| FXJ1.4094 + HX09-1 | 2 putative novel compounds | Unknown | 276, 373 | [M + H] + 509.2658 |
| Unknown | None | [M − H] − 525.2604 | ||
| FXJ1.4097 + HX09-1 | 2 putative novel compounds | Unknown | 223, 265 | [M + H] + 639.3022 |
| Unknown | 225, 267 | [M + H] + 671.3289 | ||
| FXJ1.4097 + HX10-42 | 3 putative novel compounds | Unknown | 197, 245 | [M + H] + 299.1128 |
| Unknown | 197, 245 | [M + H] + 597.2175 (dimer of 299.1128) | ||
| Unknown | 224, 266 | [M + H] + 954.4636 | ||
| FXJ1.4099 + HX10-42 | 3 putative novel compounds related to fogacin | Polyketide | 221, 274, 340 | [M + H] + 863.2872 |
| Polyketide | 221, 277 | [M + H] + 893.2611 | ||
| Polyketide | 221, 271, 335 | [M + H] + 909.2637 | ||
| FXJ1.4099 + HX10-55 | 4 putative novel compounds | Unknown | 234, 276, 340 | [M + H] + 317.2076 |
| Unknown | 228, 275, 340 | [M + H] + 453.1749 | ||
| Unknown | 220, 275, 310 | [M + H] + 457.1614 | ||
| Unknown | 229, 275, 330, 350 | [M + H] + 607.1823 | ||
| FXJ1.4102 + HX10-55 | 1 putative novel compound | Unknown | 199, 286 | [M + H] + 365.1027 |
| FXJ1.4102 + HX14-21 | 2 putative novel compounds | Unknown | 237, 318, 332 | [M + H] + 622.3491 |
| Siderophore | 195, 225, 274, 404 | [M + H] + 654.2662 | ||
| FXJ1.4106 + HX09-1 | 1 putative novel compound related to tetrodecamycin | Polyketide | 206, 254 | [M + H] + 335.1494 |
| FXJ1.4106 + HX10-42 | 2 putative novel compounds | Unknown | 194, 240 | [M + H]- 229.0674 |
| Unknown | None | [M + H] + 782.5692 | ||
| FXJ1.4110 + HX14-21 | 1 putative novel compound | Unknown | 227, 272, 338 | [M + H] + 261.1125 |
| FXJ1.4111 + HX10-55 | 3 putative novel compounds related to actinoperylone | Polyketide | 231, 325 | [M + H] + 326.1597 |
| Polyketide | 226, 275, 325 | [M + H] + 543.1457 | ||
| Polyketide | 223, 325 | [M + H] + 617.1647 | ||
| FXJ1.4112 + HX09-1 | 2 putative novel compounds | Unknown | 200, 225, 278, 317, 331 | [M + H] + 654.3387 |
| Unknown | 196, 224 | [M − H] − 717.4581 | ||
| FXJ1.4112 + HX10-42 | 2 putative novel compounds related to collinomycin | Polyketide | 220, 285, 413 | [M − H] − 507.0928 |
| Polyketide | 220, 285, 415 | [M − H] − 521.1081 | ||
| FXJ1.4112 + HX10-55 | 2 putative novel compounds | Unknown | 227 | [M + H] + 747.4677 |
| Unknown | 197 | [M + Na] + 691.5305 | ||
| FXJ1.4122 + HX14-21 | 1 putative novel compound | Unknown | 234, 278 | [M + H] + 587.1566 |
Six putative new-skeleton NPs were detected in the co-culture of S. sp. FXJ1.264 and M. sp. HX09-1 (Table 3). They were analogues featuring similar UV absorption characteristics (UV: 195, 251, 285, 376 nm), color, and solubility, and were named 1.264HX-1–6 (Figure S2a,b). HRMS results revealed that the MWs of 1.264HX-1, 1.264HX-3, 1.264HX-4, 1.264HX-5, and 1.264HX-6 were 372, 386, 744, 744, and 744, respectively (Table S3 and Figure S2c,d). The MW of 1.264HX-3 was 14 Da larger than that of 1.264HX-1, indicating that it might be a methylated derivative of 1.264 HX-1 (Figure S2c,d). The MW of 1.264HX-4 was twice that of 1.264HX-1, inferring that it might be a dimer of 1.264HX-1, and so do 1.264HX-5 and 1.264HX-6 (Figure S2c,d). The MW of compound 1.264HX-2 was not determined owing to a lack of MS ion current in both ESI- and APCI- ion sources.
We isolated and purified compound 1.264HX-3 from the extracts, which unfortunately, was unstable. HRMS analysis of 1.264HX-3 supported the molecular formula of C25H22O4, indicating fifteen degrees of unsaturation. 1H-NMR analysis of 1.264HX-3 showed that no matter which peak was set as the standard, there were still many peaks whose integral areas were non-integral. For example, the integral areas of peaks at δ 5.79 and 7.90 ppm were 0.52 and 0.57, respectively, which were far smaller than 1 (presumed to be CH); and peaks at δ 1.18 and 3.80 ppm displayed integral areas of 4.98 and 4.72, respectively, which were much larger than 3 (presumed to be CH3). Accordingly, in the 13C-NMR spectrum, two or three similar signal bars were seen at each 13C chemical shift, which greatly increased the difficulty of spectral analysis (Table S4). Therefore, the possible carbon atoms and their directly connected hydrogen atoms of 1.264HX-3 were tentatively numbered by analyzing the 1H, 13C, and HSQC spectra, as shown in Table S4. Partial structural fragments of this compound were elucidated based on the current NMR data (Figures S3–S10), which revealed that it belongs to polyketides.
Meanwhile, some putative new analogues of known compounds were identified from the co-cultures as well. The co-culture of S. sp. FXJ1.4099 and M. sp. HX10-42 activated the production of fogacin and three putative new analogues with similar HPLC retention times and UV absorptions [26,27]. The co-culture of S. sp. FXJ1.4106 and M. sp. HX09-1 led to the production of a new compound bearing highly similar UV absorption, but different MW to tetracycline [28]. Two compounds with unpublished MWs produced by the co-culture of S. sp. FXJ1.4112 and M. sp. HX10-42 were deduced as collinomycin analogs due to their dark red color and characteristic UV absorption [29,30]. We speculated that the co-culture of S. sp. FXJ1.4111 and R. sp. HX10-55 produced actinoperylone and three putative new analogues, which had a similar UV absorption [31]. In addition, a putative new iron siderophore was discovered from the co-cultural broth of S. sp. FXJ1.4102 and N. sp. HX14-21, on account of its characteristic UV absorption at 404 nm and the rapid emergence of red color in solution after the addition of ferric ion [19]. Together, these data confirm that the co-culture of Streptomyces and MACB is a reliable source of novel NPs.
3.5. Direct Physical Contact between Streptomycetes and MACB Is Essential to Induce SMs in the Co-Culture
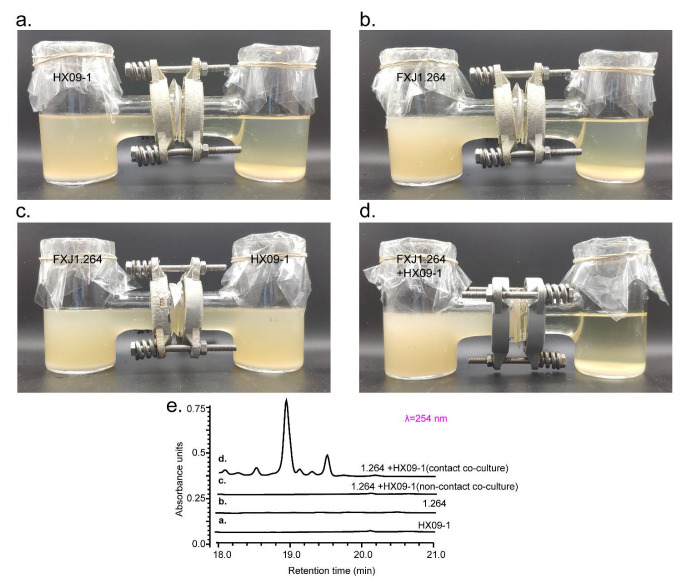
We took the co-culture of S. sp. FXJ1.264 and M. sp. HX09-1 as a case to further explore the underlying mechanism of induction. A non-contact device described in method 2.5 was used to investigate whether cell-to-cell contact was required for the activation of SMs. After fermentation for 7 days, samples from the compartments of the device (Figure 4a–d) were plated on fresh YGSS agar and incubated for 5 days. No growth of bacteria was observed for samples from the un-inoculated compartments, validating that the cells of the two strains could not pass through the 0.22 μm filter membrane. HPLC analysis of the fermentation extracts showed that, compared to the pure culture controls (Figure 4a, b), the non-contact co-culture (Figure 4c) produced no new metabolites while the contact co-culture (Figure 4d) still produced a series of new peaks (Figure 4e). Moreover, co-culturing S. sp. FXJ1.264 with heat-killed 2- or 7-day-old M. sp. HX09-1 cultures did not activate any new metabolites either (Figure S11). These results indicate that the induced production of new metabolites by co-culture of S. sp. FXJ1.264 and M. sp. HX09-1 probably relies on direct physical contact between the living cells of the two strains, instead of on the molecular elicitors secreted by MACB.
Figure 4.
Contact and non-contact co-cultures of S. sp. FXJ1.264 and M. sp. HX09-1 and related HPLC profiling. (a) S. sp. FXJ1.264 was only inoculated in the left compartment; (b) M. sp. HX09-1 was only inoculated in the left compartment; (c) S. sp. FXJ1.264 and M. sp. HX09-1 were individually inoculated in the left and right compartment, respectively; (d) S. sp. FXJ1.264 and M. sp. HX09-1 were inoculated in the left compartment; (e) HPLC analysis of fermentation extracts from (a–d).
4. Discussion
Though MACB have been proved to widely activate cryptic SM-BGCs in actinobacteria [16], it seems that the selection of actinobacteria also plays a non-negligible role in the efficiency of induction. Overall, the effect of MACB activation on Streptomyces is significantly better than that on marine Micromonosporaceae reported [32] (Figure S12), possibly because Streptomyces have a stronger ability to synthesize NPs than Micromonosporaceae. In addition, the co-cultures of MACB and red soil-derived Streptomyces display a significantly higher ratio of an increase in NP yields than the co-cultures of MACB and Streptomyces from other habitats [15] (Figure S12). This difference can be partially attributed to the outstanding biosynthetic capacity and phylogenetic diversity of the streptomycetes we used. These comparisons highlight the importance of strain selection for rational optimization of co-culture in future work.
The novel NPs obtained by the co-culture of Streptomyces and MACB prompted us to trace the original producers of these compounds. In silico genome mining of several Streptomyces strains and the type strains most closely related to the MACB used in this study reveals that the predicted diversity and number of SM-BGCs in Streptomyces (over 40 BGCs per genome) are overwhelmingly higher than those in MACB (less than 25 BGCs per genome) [20,21]. Therefore, it seems plausible that the unique metabolites emerging in the co-cultures may be derived from Streptomyces. This speculation is supported by following experimental evidences. (i) The co-culture of the same Streptomyces strain with different MACB leads to rediscovery of the same induced metabolites (Figure 1a and Figure 3a–d). (ii) Some of the activated products in co-cultures were known compounds isolated from Streptomyces. For example, the co-culture of S. sp. FXJ1.4012 and N. sp. HX14-21 produced multiple new HPLC peaks, three of which were identified as enterocin, deoxyenterocin, and oligomycin D (Figure S13). Enterocin and deoxyenterocin have been isolated from Streptomyces, but not from MACB [33,34], while oligomycin D is a macrolide antibiotic originally discovered from Streptomyces [35]. (iii) The induced NPs in the co-culture of S. sp. FXJ1.264 and M. sp. HX09-1 was also observed in the pure culture of a genetically manipulated mutant of S. sp. FXJ1.264 (data not shown). Based on the above bioinformatical and experimental evidences, we conclude that the new metabolites produced by co-cultures were mainly from Streptomyces.
Although several studies have reported that co-culture with MACB can change the SM profiles of Streptomyces [36,37,38], the specific mechanism of this phenomenon is still unclear. At present, the possible mechanisms mainly include three types: (i) direct physical contact of co-cultured strains [15,25], (ii) the induction role of molecular compounds secreted by MACB [39], and (iii) horizontal gene transfer between strains [40]. In our study, the result of the non-contact co-culture of S. sp. FXJ1.264 and M. sp. HX09-1 suggests that the production of new metabolites of S. sp. FXJ1.264 may not be mediated by molecule exchange between the two strains, but by direct contact between the cells. In order to further test the possibility of horizontal gene transfer during direct contact, we re-isolated the two strains from the co-cultured broth and cultured them separately. We did not find any new compounds from the pure cultures of the two isolated strains, and thus, essentially ruled out the possibility of activating new products by horizontal gene transfer. Moreover, a recent study reported that some SMs of Streptomyces could be activated by both living producer strains and heat-killed strains. [41,42]. However, the heat inactivated M. sp. HX09-1 strain failed to activate the production of new metabolites in S. sp. FXJ1.264 (Figure S11), indicating that the activation effect was not induced by dead cells. To summarize, direct contact between living M. sp. HX09-1 and S. sp. FXJ1.264 cells is needed to activate the new metabolites from S. sp. FXJ1.264. Nevertheless, it is limited in our work to study the activation mechanism using only one pair of co-culture, and other mechanisms may also exist in other co-culture combinations that activate new metabolites. Extensive comparative “-omics” work needs to be done to predict the activating factors in future investigation.
5. Conclusions
To explore the cryptic novel NPs encoded by red soil-derived Streptomyces, we co-cultured them with MACB and compared the SM spectra and antimicrobial activity of the co-cultures and pure cultures. We found that 82.9% (146/176) and 60.8% (107/176) of the co-culture pairs changed the SM spectra and enhanced the yield of original SMs of the pure cultures, respectively. Moreover, 29.5% (52/176) of the co-culture pairs produced unique SMs not detected in the pure cultures, of which 49 compounds were putative novel NPs. In addition, the bioactivity assay revealed that co-culture could induce and/or enhance the production of bioactive SMs. These results confirm that MACB can effectively alter the secondary metabolism of Streptomyces. Furthermore, the activation of new SMs in the co-culture of S. sp. FXJ1.264 and M. sp. HX09-1 depends on the direct contact of their living cells. In conclusion, the co-culture of MACB and red soil-derived streptomycetes is an efficient and applicable approach for novel NP discovery.
Acknowledgments
We are grateful to Luyan Z. Ma at the Institute of Microbiology, Chinese Academy of Sciences, for kindly providing us with the non-contact co-culture device.
Supplementary Materials
Supplementary data include figures, tables and associated description are available online at https://www.mdpi.com/article/10.3390/microorganisms9112187/s1, Table S1: Mycolic acid-containing bacteria (MACB) and streptomycetes used in this study. Table S2: Linear elution ratio of methanol and water in HPLC-PDA analysis. Table S3: HPLC-PDA/HRMS data of 1.264HX-1–6. Table S4: NMR data of 1.264HX-3 in CDCl3 (500 MHz). Figure S1: The device for non-contact co-culture. Figure S2: HPLC and HRMS chromatograms of compounds 1.264HX-1–6. Figure S3: Three putative structures of 1.264HX-3. Figure S4: 1H spectrum of 1.264HX-3. Figure S5: 13C spectrum of 1.264HX-3. Figure S6: H-H COSY spectrum of 1.264HX-3. Figure S7: DEPT135 spectrum of 1.264HX-3. Figure S8: HMBC spectrum of 1.264HX-3. Figure S9: HSQC spectrum of 1.264HX-3. Figure S10: NOESY spectrum of 1.264HX-3. Figure S11: HPLC analysis of fermentation extracts of co-cultures of S. sp. FXJ1.264 and heat-killed M. sp. HX09-1. Figure S12: Comparison of activation effects of MACB on different actinobacteria. Figure S13: HPLC and HRMS analysis of fermentation extracts of S. sp. FXJ1.4012, N. sp. HX14-21, and their co-culture.
Author Contributions
Conceptualization, M.L. and K.W.; methodology, K.W., N.L. and F.S.; validation, M.L., N.L. and F.S.; formal analysis, K.W., N.L. and M.L.; investigation, K.W., N.L. and F.S.; resources, B.Y. and J.H.; data curation, K.W. and M.L.; writing—original draft preparation, K.W.; writing—review and editing, M.L., N.L. and Y.H.; visualization, K.W.; supervision, Y.H.; project administration, M.L. and Y.H.; funding acquisition, M.L., N.L. and Y.H. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (grant nos. 31700008, 81773615, and 31670502).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available in the article and supplementary material.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Bade R., Chan H.F., Reynisson J. Characteristics of known drug space. Natural products, their derivatives and synthetic drugs. Eur. J. Med. Chem. 2010;45:5646–5652. doi: 10.1016/j.ejmech.2010.09.018. [DOI] [PubMed] [Google Scholar]
- 2.Newman D.J., Cragg G.M. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J. Nat. Prod. 2020;83:770–803. doi: 10.1021/acs.jnatprod.9b01285. [DOI] [PubMed] [Google Scholar]
- 3.Procópio R.E., Silva I.R., Martins M.K., Azevedo J.L., Araújo J.M. Antibiotics produced by Streptomyces. Braz. J. Infect. Dis. 2012;16:466–471. doi: 10.1016/j.bjid.2012.08.014. [DOI] [PubMed] [Google Scholar]
- 4.Devine R., Hutchings M.I., Holmes N.A. Future directions for the discovery of antibiotics from actinomycete bacteria. Emerg. Top. Life Sci. 2017;1:1–12. doi: 10.1042/ETLS20160014. [DOI] [PubMed] [Google Scholar]
- 5.Baltz R.H. Gifted microbes for genome mining and natural product discovery. J. Ind. Microbiol. Biotechnol. 2017;44:573–588. doi: 10.1007/s10295-016-1815-x. [DOI] [PubMed] [Google Scholar]
- 6.Li Y., Wang M., Sun Z.Z., Xie B.B. Comparative genomic insights into the taxonomic classification, diversity, and secondary metabolic potentials of Kitasatospora, a genus closely related to Streptomyces. Front. Microbiol. 2021;12:683814. doi: 10.3389/fmicb.2021.683814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Baltz R.H. Natural product drug discovery in the genomic era: Realities, conjectures, misconceptions, and opportunities. J. Ind. Microbiol. Biotechnol. 2019;46:281–299. doi: 10.1007/s10295-018-2115-4. [DOI] [PubMed] [Google Scholar]
- 8.Liu N., Guan H., Niu G., Jiang L., Li Y., Zhang J., Li J., Tan H. Molecular mechanism of mureidomycin biosynthesis activated by introduction of an exogenous regulatory gene ssaA into Streptomyces roseosporus. Sci. China Life Sci. 2021;9:1–15. doi: 10.1007/s11427-020-1892-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Liu Z., Zhao Y., Huang C., Luo Y. Recent advances in silent gene cluster activation in Streptomyces. Front. Bioeng. Biotechnol. 2021;9:6322–6330. doi: 10.3389/fbioe.2021.632230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cho J.Y., Kim M.S. Induction of antifouling diterpene production by Streptomyces cinnabarinus PK209 in co-culture with marine-derived Alteromonas sp. KNS-16. Biosci. Biotechnol. Biochem. 2012;76:1849–1854. doi: 10.1271/bbb.120221. [DOI] [PubMed] [Google Scholar]
- 11.Shin D., Byun W.S., Moon K., Kwon Y., Bae M., Um S., Lee S.K., Oh D.C. Co-culture of marine Streptomyces sp. with Bacillus sp. produces a new piperazic acid-bearing cyclic peptide. Front. Chem. 2018;6:498. doi: 10.3389/fchem.2018.00498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Maglangit F., Fang Q., Kyeremeh K., Sternberg J.M., Ebel R., Deng H. A co-culturing approach enables discovery and biosynthesis of a bioactive indole alkaloid metabolite. Molecules. 2020;25:256. doi: 10.3390/molecules25020256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lee N., Kim W., Chung J., Lee Y., Cho S., Jang K.S., Kim S.C., Palsson B., Cho B.K. Iron competition triggers antibiotic biosynthesis in Streptomyces coelicolor during co-culture with Myxococcus xanthus. ISME J. 2020;14:1111–1124. doi: 10.1038/s41396-020-0594-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang C., Straight P.D. Antibiotic discovery through microbial interactions. Curr. Opin. Microbiol. 2019;51:64–71. doi: 10.1016/j.mib.2019.06.006. [DOI] [PubMed] [Google Scholar]
- 15.Onaka H., Mori Y., Igarashi Y., Furumai T. Mycolic acid-containing bacteria induce natural-product biosynthesis in Streptomyces species. Appl. Environ. Microbiol. 2011;77:400–406. doi: 10.1128/AEM.01337-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hoshino S., Onaka H., Abe I. Activation of silent biosynthetic pathways and discovery of novel secondary metabolites in actinomycetes by co-culture with mycolic acid-containing bacteria. J. Ind. Microbiol. Biotechnol. 2019;46:363–374. doi: 10.1007/s10295-018-2100-y. [DOI] [PubMed] [Google Scholar]
- 17.Pan C., Kuranaga T., Cao X., Suzuki T., Dohmae N., Shinzato N., Onaka H., Kakeya H. Amycolapeptins A and B, cyclic nonadepsipeptides produced by combined-culture of Amycolatopsis sp. and Tsukamurella pulmonis. J. Org. Chem. 2021;86:1843–1849. doi: 10.1021/acs.joc.0c02660. [DOI] [PubMed] [Google Scholar]
- 18.Jiang Y., Matsumoto T., Kuranaga T., Lu S., Wang W., Onaka H., Kakeya H. Longicatenamides A-D, two diastereomeric pairs of cyclic hexapeptides produced by combined-culture of Streptomyces sp. KUSC_F05 and Tsukamurella pulmonis TP-B0596. J. Antibiot. 2021;74:307–316. doi: 10.1038/s41429-020-00400-3. [DOI] [PubMed] [Google Scholar]
- 19.Guo X., Liu N., Li X., Ding Y., Shang F., Gao Y., Ruan J., Huang Y. Red soils harbor diverse culturable actinomycetes that are promising sources of novel secondary metabolites. Appl. Environ. Microbiol. 2015;81:3086–3103. doi: 10.1128/AEM.03859-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu N., Song L., Liu M., Shang F., Anderson Z., Fox D.J., Challis G.L., Huang Y. Unique post-translational oxime formation in the biosynthesis of the azolemycin complex of novel ribosomal peptides from Streptomyces sp. FXJ1.264. Chem. Sci. 2016;7:482–488. doi: 10.1039/C5SC03021H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Liu M., Liu N., Shang F., Huang Y. Activation and identification of NC-1: A cryptic cyclodepsipeptide from red soil-derived Streptomyces sp. FXJ1.172. Eur. J. Org. Chem. 2016;23:3943–3948. doi: 10.1002/ejoc.201600297. [DOI] [Google Scholar]
- 22.Liu N., Song F., Shang F., Huang Y. Mycemycins A-E, new dibenzoxazepinones isolated from two different Streptomycetes. Mar. Drugs. 2015;13:6247–6258. doi: 10.3390/md13106247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Huang J., Yan B., Huang Y. Diversity of culturable actinobacteria from soils collected in Ali, Naqu and Haixi Districts on the Qinghai-Tibet Plateau. Acta Microbiol. Sin. 2017;57:1342–1351. [Google Scholar]
- 24.Yan B., Liu N., Liu M., Du X., Shang F., Huang Y. Soil actinobacteria tend to have neutral interactions with other co-occurring microorganisms, especially under oligotrophic conditions. Environ. Microbiol. 2021;23:4126–4140. doi: 10.1111/1462-2920.15483. [DOI] [PubMed] [Google Scholar]
- 25.Asamizu S., Ozaki T., Teramoto K., Satoh K., Onaka H. Killing of mycolic acid-containing bacteria aborted induction of antibiotic production by Streptomyces in combined-culture. PLoS ONE. 2015;10:e0142372. doi: 10.1371/journal.pone.0142372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Radzom M., Zeeck A., Antal N., Fiedler H.P. Fogacin, a novel cyclic octaketide produced by Streptomyces strain Tü 6319. J. Antibiot. 2006;59:315–317. doi: 10.1038/ja.2006.45. [DOI] [PubMed] [Google Scholar]
- 27.Sato K., Katsuyama Y., Yokota K., Awakawa T., Tezuka T., Ohnishi Y. Involvement of β-alkylation machinery and two sets of ketosynthase-chain-length factors in the biosynthesis of fogacin polyketides in Actinoplanes missouriensis. ChemBioChem. 2019;20:1039–1050. doi: 10.1002/cbic.201800640. [DOI] [PubMed] [Google Scholar]
- 28.Tsuchida T., Iinuma H., Nishida C., Kinoshita N., Sawa T., Hamada M., Takeuchi T. Tetrodecamycin and dihydrotetrodecamycin, new antimicrobial antibiotics against Pasteurella piscicida produced by Streptomyces nashvillensis MJ885-mF8. I. Taxonomy, fermentation, isolation, characterization and biological activities. J. Antibiot. 1995;48:1104–1109. doi: 10.7164/antibiotics.48.1104. [DOI] [PubMed] [Google Scholar]
- 29.Brockmann H., Renneberg K.H. Collinomycin, ein gelbes antibiotikum aus Actinomyceten. Sci. Nat.-Heidelberg. 1953;40:166–167. doi: 10.1007/BF00639946. [DOI] [Google Scholar]
- 30.Martin R., Sterner O., Alvarez M.A., de Clercq E., Bailey J.E., Minas W. Collinone, a new recombinant angular polyketide antibiotic made by an engineered Streptomyces strain. J. Antibiot. 2001;54:239–249. doi: 10.7164/antibiotics.54.239. [DOI] [PubMed] [Google Scholar]
- 31.Okamoto S., Taguchi T., Ochi K., Ichinose K. Biosynthesis of actinorhodin and related antibiotics: Discovery of alternative routes for quinone formation encoded in the act gene cluster. Chem. Biol. 2009;16:226–236. doi: 10.1016/j.chembiol.2009.01.015. [DOI] [PubMed] [Google Scholar]
- 32.Adnani N., Vazquez-Rivera E., Adibhatla S.N., Ellis G.A., Braun D.R., Bugni T.S. Investigation of interspecies interactions within marine Micromonosporaceae using an improved co-culture approach. Mar. Drugs. 2015;13:6082–6098. doi: 10.3390/md13106082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Miyairi N., Sakai H., Konomi T., Imanaka H. Enterocin, a new antibiotic taxonomy, isolation and characterization. J. Antibiot. 1976;29:227–235. doi: 10.7164/antibiotics.29.227. [DOI] [PubMed] [Google Scholar]
- 34.Piel J., Hertweck C., Shipley P.R., Hunt D.M., Newman M.S., Moore B.S. Cloning, sequencing and analysis of the enterocin biosynthesis gene cluster from the marine isolate ‘Streptomyces maritimus’: Evidence for the derailment of an aromatic polyketide synthase. Chem. Biol. 2000;7:943–955. doi: 10.1016/S1074-5521(00)00044-2. [DOI] [PubMed] [Google Scholar]
- 35.Khebizi N., Boudjella H., Bijani C., Bouras N., Klenk H.P., Pont F., Mathieu F., Sabaou N. Oligomycins A and E, major bioactive secondary metabolites produced by Streptomyces sp. strain HG29 isolated from a Saharan soil. J. Mycol. Med. 2018;28:150–160. doi: 10.1016/j.mycmed.2017.10.007. [DOI] [PubMed] [Google Scholar]
- 36.Park H.B., Kwon H.C., Lee C.H., Yang H.O. Glionitrin A, an antibiotic-antitumor metabolite derived from competitive interaction between abandoned mine microbes. J. Nat. Prod. 2009;72:248–252. doi: 10.1021/np800606e. [DOI] [PubMed] [Google Scholar]
- 37.Hoshino S., Okada M., Wakimoto T., Zhang H., Hayashi F., Onaka H., Abe I. Niizalactams A-C, multicyclic macrolactams isolated from combined culture of Streptomyces with mycolic acid-containing bacterium. J. Nat. Prod. 2015;78:3011–3017. doi: 10.1021/acs.jnatprod.5b00804. [DOI] [PubMed] [Google Scholar]
- 38.Hoshino S., Zhang L., Awakawa T., Wakimoto T., Onaka H., Abe I. Arcyriaflavin E, a new cytotoxic indolocarbazole alkaloid isolated by combined-culture of mycolic acid-containing bacteria and Streptomyces cinnamoneus NBRC 13823. J. Antibiot. 2015;68:342–344. doi: 10.1038/ja.2014.147. [DOI] [PubMed] [Google Scholar]
- 39.Adnani N., Chevrette M.G., Adibhatla S.N., Zhang F., Yu Q., Braun D.R., Nelson J., Simpkins S.W., McDonald B.R., Myers C.L., et al. Co-culture of marine invertebrate-associated bacteria and interdisciplinary technologies enable biosynthesis and discovery of a new antibiotic, Keyicin. ACS Chem. Biol. 2017;12:3093–3102. doi: 10.1021/acschembio.7b00688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kurosawa K., Ghiviriga I., Sambandan T.G., Lessard P.A., Barbara J.E., Rha C., Sinskey A.J. Rhodostreptomycins, antibiotics biosynthesized following horizontal gene transfer from Streptomyces padanus to Rhodococcus fascians. J. Am. Chem. Soc. 2008;130:1126–1127. doi: 10.1021/ja077821p. [DOI] [PubMed] [Google Scholar]
- 41.Liang L., Sproule A., Haltli B., Marchbank D.H., Berrué F., Overy D.P., McQuillan K., Lanteigne M., Duncan N., Correa H., et al. Discovery of a new natural product and a deactivation of a quorum sensing system by culturing a “Producer” bacterium with a heat-killed “Inducer” culture. Front. Microbiol. 2018;9:3351. doi: 10.3389/fmicb.2018.03351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Liang L., Wang G., Haltli B., Marchbank D.H., Stryhn H., Correa H., Kerr R.G. Metabolomic comparison and assessment of co-cultivation and a heat-killed inducer strategy in activation of cryptic biosynthetic pathways. J. Nat. Prod. 2020;83:2696–2705. doi: 10.1021/acs.jnatprod.0c00621. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data presented in this study are available in the article and supplementary material.