Abstract
Background
Household contacts of people with pulmonary tuberculosis (TB) have greater risk of developing TB. Recent guidelines conditionally recommended TB preventive treatment (TPT) for household contacts of any age living in TB high-incidence countries, expanding earlier guidance to provide TPT to household contacts under five. The all-age population of household contacts has not been estimated.
Methods
Our model-based estimation included 20 countries with >80% of incident TB globally in 2019. We developed country-specific distributions of household composition by age and sex using bootstrap resampling from health surveys and census data. We incorporated age-, sex-, year-, and location-specific estimates of pulmonary TB incidence from the Global Burden of Diseases, Injuries, and Risk Factors Study 2019 to estimate the population in each country sharing a household with someone with incident pulmonary TB, and quantified uncertainty using a Monte Carlo approach.
Findings
We estimate that 38 million [95% uncertainty interval (UI) 33- 43 million] individuals lived in a household with someone with incident pulmonary TB in 2019 in these 20 countries. Children under five made up 12% of the population with household exposure, while adults were 65%. Zimbabwe, Mozambique, Zambia, and Pakistan had the highest proportion of the population with household exposure, while India had the highest number of contacts (11·4 million, 95% UI 9·7-13·4 million).
Interpretation
Expanding TPT evaluation to household contacts of all ages in high-incidence countries could include a population more than 7-times larger than the under-5 contacts previously prioritized. This would substantially increase the impact of household contact investigation on reducing TB morbidity and mortality.
Funding
JMR is supported by the National Institute of Allergy and Infectious Diseases (K01 AI138620). This research was funded in part by a 2020 developmental grant from the University of Washington / Fred Hutch Center for AIDS Research, an NIH funded program under award number AI027757 which is supported by the following NIH Institutes and Centers: NIAID, NCI, NIMH, NIDA, NICHD, NHLBI, NIA, NIGMS, NIDDK. This work was funded in part by the National Science Foundation (DMS-1839116).
Research in Context.
Evidence before this study
We reviewed PubMed for studies reporting the population of household contacts of people with pulmonary tuberculosis (TB). Many studies reported contacts by age and sex, but most were limited to a single country, and studies used different methodologies that made it challenging to compare values between countries. We identified three studies estimating the population of childhood household contacts globally or across high-burden countries. These studies analysed adult TB prevalence estimates or TB case notifications with census data or household surveys to estimate child contacts age less than five or less than 15, but not older adolescents or adults.
Added value of this study
To our knowledge, this is the first estimate of the all-age population at elevated risk for TB due to sharing a household with someone with incident pulmonary TB across high-burden countries. This work is responsive to 2018 guidelines from the World Health Organization offering a conditional recommendation for TB preventive treatment (TPT) to a household contact of any age of someone with bacteriologically confirmed TB in high-incidence settings. We used a standardized estimation process for all locations, which allows for age-, sex-, and location-specific estimates that are comparable between countries.
Implications of all the available evidence
We estimate that there were 38 million individuals living in a household with someone with incident pulmonary TB in 2019 in 20 high-burden countries. Since we estimate 88% of this population is age 5 or older, the new guidelines correspond to a substantial increase in the number of people potentially considered for TPT. Our estimates likely represent an upper bound, as they include all people with incident pulmonary TB in these 20 countries as index cases rather than the fraction of the people with TB who reach health services for TB diagnosis and bacteriological confirmation.
Alt-text: Unlabelled box
1. Introduction
Nearly 10 million people develop tuberculosis (TB) each year despite decades of research and public health effort to prevent this bacterial infection [1,2]. Global rates of new TB illness are not declining rapidly enough to reach international morbidity and mortality targets, such as the Sustainable Development Goals Target 3·3 and the Stop TB Partnership target for TB incidence [3,4]. Effective TB prevention efforts include evaluating persons who are at elevated risk for TB due to sharing a household with someone with active TB, which is primarily acquired via inhalation of bacteria that become aerosolized from the respiratory system of a person with pulmonary TB [5], [6], [7]. Providing courses of anti-tuberculous medications as TB preventive treatment (TPT) reduces the risk of developing active TB among household contacts without signs or symptoms of active TB at the time of screening [8].
Recently, the World Health Organization (WHO) offered a conditional recommendation to broaden the age range of household contacts in countries with high TB incidence who may receive TPT [9,10]. Historically, children under the age of 5 years living in the household of a person with pulmonary TB have been prioritized due to their greater risk for developing TB disease than older children or adults [11]. The guidelines updated in 2018 and 2020 expanded the eligible population of household contacts to include children older than 5 years, adolescents, and adults in countries with high TB incidence in the context of clinical assessment of likely benefits and risks and in the context of local TB epidemiology [9,10]. The UN High-Level Meeting on TB in 2018 achieved a commitment to provide TPT to 20 million persons over age 5 by 2022, though fewer than 1 million household contacts age 5 or older received TPT in 2018 and 2019 [2,12].
While the initial commitment to reach 20 million people represents a rapid expansion of TPT, the proportional contribution is unclear, as the denominator of the number of people considered for TPT due to household TB exposure has not been systematically estimated across age groups in TB high-burden countries. Earlier efforts aligned with the prior focus on young children, including an estimate by Dodd, et al, that 15·3 million children age 0-14 shared a household with someone with infectious TB in 2010 across 22 high-burden countries; Yuen, et al estimated there were approximately 2·4 million children under five and 5.1 million children aged 5-14 living in households were adults with known TB in 2014; and Hamada, et al estimated that 1·27 million children under age five globally were eligible for TPT in 2017 due to living in a household with someone with bacteriologically-confirmed pulmonary TB and having a positive tuberculin skin test (TST) or interferon gamma release assay (IGRA) for children in low-burden countries [13], [14], [15]. Quantifying the full population of household contacts now considered for TPT across the new age groups is critical for benchmarking progress and for estimating the impact that TPT expansion will have in averting TB morbidity and death under the new guidelines.
The Global Burden of Diseases, Injuries, and Risk Factors Study 2019 (GBD 2019) estimated that 9.65 million people developed active TB disease in 2019 [1]. To fill the knowledge gap for household contacts, we applied age-, sex-, and location-specific results from GBD 2019 in a model-based analysis to estimate the number of persons by age and sex at elevated risk for TB due to living in a household with a person with incident active pulmonary TB in 20 TB high-burden countries in which 82% of the global total of new TB cases occurred in 2019.
2. Methods
2.1. Overview
This model-based estimation study analyzes age-, sex-, and location-specific estimates of TB burden from GBD 2019 together with an age-, sex-, and location-specific household composition analysis to generate comparable estimates of the population (by age and sex) exposed to someone with new pulmonary TB in their household in 20 countries in 2019. We selected the countries for modelling to include those among the top 20 countries for TB incidence counts in GBD 2017.
This analysis complies with the Guidelines for Accurate and Transparent Health Estimates Reporting (GATHER) statement [16]. We conducted estimation using Python version 3·6 and published a code repository.[17] The analysis uses publicly available de-identified data and did not require participant consent or human subjects review.
2.2. Data sources
We extracted results from GBD 2019 to inform the age-, sex-, and location- specific population and active TB incidence for each modelled country in 2019. We conducted our primary analysis using incident cases in 2019 and a supplemental analysis modelled using prevalent cases to align with different public health intervention scenarios. The methodology for population and disease burden estimation for GBD 2019 is described elsewhere [1,18]. We used age-, sex-, and location-specific estimates of pulmonary TB cases from GBD 2019 to include only cases of pulmonary TB (and exclude extrapulmonary TB) from our subsequent estimation (Fig S1). The GBD 2019 process calculated the proportions of pulmonary versus extrapulmonary TB using WHO case notification data and a spatiotemporal Gaussian process regression [19,20]. We included active pulmonary TB among people living with HIV and people without HIV. We extracted data on household composition by age and sex from the most recent Demographic and Health Survey (DHS) or Integrated Public Use Microdata Series (IPUMS) available for each country (Table S1). We used the sample of households from the most recent available data source in each location to approximate the probability of each household size and age/sex composition, as described below.
2.3. Calculating exposure probabilities and counts of exposed individuals
Our estimate of the number of people in each age-/sex-group who are living in a household with exposure to incident pulmonary TB depends on 1) the number of people in the age-/sex-group, 2) the age-/sex-specific incidence of pulmonary TB, and 3) the distribution of household composition in each country (both the household size and the age-/sex-composition). Household contacts in our analysis are people living in a household with at least one person with incident pulmonary TB, but who do not have pulmonary TB themselves. Intuitively, the more people in a household, the more likely it is that at least one of them has pulmonary TB, and therefore that others are exposed to TB in the household; this risk is higher in locations where the incidence of active TB is higher; and, since age and sex are important determinants of TB risk, the age and sex composition of households also impact our calculation. Full details of the mathematical formulation of these estimates are presented in the supplementary methods. In brief, we calculated an age- and sex-specific household contact matrix incorporating both household size and household demographics. We then applied this to the age-/sex-specific incidence of pulmonary TB to obtain estimates of the number of people living with a household exposure to pulmonary TB in each age-group and sex.
We quantified uncertainty in the number of exposed household contacts using a Monte Carlo approach [21]. The estimates from GBD 2019 of incidence of pulmonary TB for each group (e.g. by age, sex, country, and year) quantify uncertainty by producing 1,000 "draws" from a posterior distribution. We combined these draws with the household structure that results from bootstrap resampling of households from the household survey data, where we chose up to 50,000 households (or the total sample of households if less than 50,000) with replacement and used this resampled dataset to calculate the probability of exposure for each age-/sex-group.
2.4. Role of the funding source
The sponsor of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
3. Results
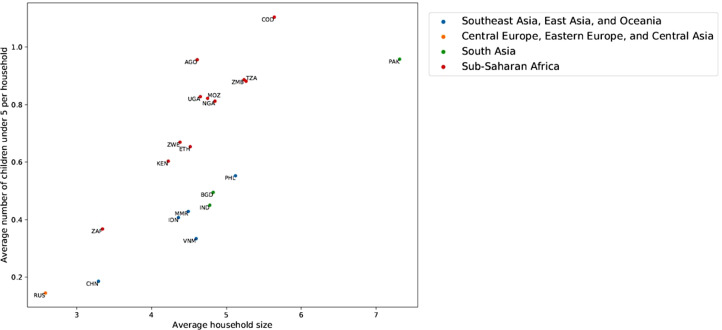
The mean household size varied from below three in Russia to above seven in Pakistan (Fig 1). All countries had a mean household size of four or larger except Russia, China, and South Africa. Age composition within households also varied by country, with the mean number of children under five ranging from 0·15 in Russia to 1·1 in the Democratic Republic of the Congo (DR Congo).
Figure 1.
Mean household size and mean number of children under five in 20 TB high-burden countries. AGO = Angola, BGD = Bangladesh, CHN = China, COD = Democratic Republic of the Congo, ETH = Ethiopia, IDN = Indonesia, IND = India, KEN = Kenya, MMR = Myanmar, MOZ = Mozambique, NGA = Nigeria, PAK = Pakistan, PHL = Philippines, RUS = Russia, TZA = Tanzania, UGA = Uganda, VNM = Vietnam, ZAF = South Africa, ZMB = Zambia, ZWE = Zimbabwe.
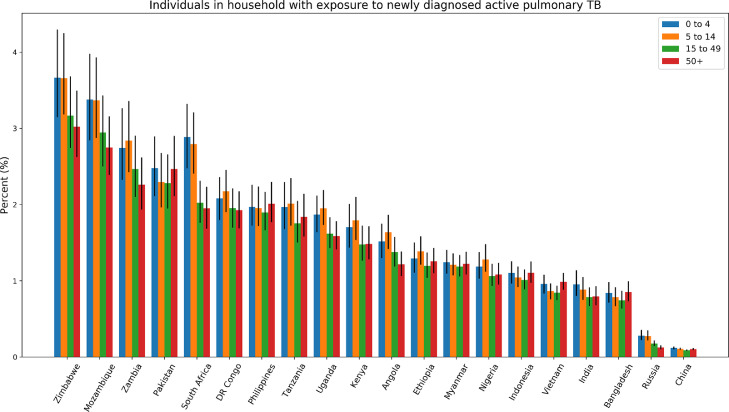
The proportion of the population living in a household with a person with incident pulmonary TB across age groups ranged from 0·10% in China to 3·3% in Zimbabwe (Fig 2). The countries with the highest proportion of the population living in a household with someone with incident pulmonary TB were Zimbabwe (3·3%), Mozambique (3·1%), Zambia (2·6%), and Pakistan (2·3%). For children under five, the proportion with household exposure to incident pulmonary TB varied from 0·12% in China to 3·7% in Mozambique.
Figure 2.
The percentage of individuals living in a household with someone with incident pulmonary TB by age group and country in 2019. Error bars represent 2.5% - 97.5% uncertainty intervals for age group estimates. Countries are ordered by the proportion across age groups.
The number of individuals with exposure in their household to someone with incident pulmonary TB across all ages and 20 countries was 38 million (95% UI 33 to 43 million)(Table 1). Children under five made up 12% of the population with household exposure to pulmonary TB, while adults ages 15 and older accounted for 65%. We estimated 33 million (95% UI 29 to 38 million) household contacts across the modeled countries were age 5 or older, corresponding to persons considered under the new TPT criteria. India was the country with the largest population with household exposure to incident pulmonary TB, with an estimated 11·4 million people (95% UI 9·7-13·4), followed by Pakistan (5·2 million), Indonesia (2·7 million), Nigeria (2·5 million), and the Philippines (2·2 million). We estimated a larger population of 62 million (95% UI 55 to 71) with household exposure to someone with prevalent pulmonary TB across all ages and the 20 countries (Table S2).
Table 1.
Estimated number of persons with household exposure to incident pulmonary TB in 2019, by age*
| Count Rank | Age 0-4 (95% UI) | Age 5-14 (95% UI) | Age 15-49 (95% UI) | Age 50+ (95% UI) | Total | |
|---|---|---|---|---|---|---|
| 20 countries | 458.2 (399.0 - 528.2) | 866.0 (758.6 - 990.0) | 1856.8 (1631.6 - 2107.9) | 583.1 (509.0 - 662.1) | 3773.5 (3312.1 - 4297.8) | |
| Angola | 19 | 7.8 (6.7 - 9.0) | 14.5 (12.5 - 16.5) | 18.6 (16.1 - 21.3) | 3.2 (2.8 - 3.6) | 44.4 (38.5 - 50.7) |
| Bangladesh | 10 | 11.5 (9.8 - 13.5) | 23.7 (20.2 - 27.7) | 64.1 (54.8 - 74.9) | 24.8 (21.2 - 28.9) | 123.9 (106.1 - 144.8) |
| China | 7 | 9.8 (8.4 - 11.3) | 15.4 (13.6 - 17.6) | 64.1 (56.7 - 72.3) | 49.7 (44.0 - 56.2) | 139.2 (123.2 - 157.0) |
| DR Congo | 6 | 28.4 (24.5 - 32.1) | 52.3 (45.7 - 59.0) | 81.5 (70.8 - 92.2) | 15.9 (13.9 - 17.9) | 178.5 (155.4 - 202.0) |
| Ethiopia | 8 | 21.6 (18.4 - 25.0) | 40.7 (35.6 - 46.5) | 62.7 (54.5 - 71.9) | 11.3 (9.9 - 12.9) | 136.6 (119.0 - 156.0) |
| India | 1 | 111.9 (93.8 - 133.1) | 228.9 (193.9 - 271.7) | 594.8 (508.9 - 693.1) | 202.6 (172.9 - 237.0) | 1142.9 (973.8 - 1340.7) |
| Indonesia | 3 | 21.7 (19.0 - 24.7) | 47.3 (41.7 - 53.9) | 144.3 (126.9 - 164.2) | 57.0 (50.1 - 64.6) | 270.0 (238.0 - 305.4) |
| Kenya | 14 | 10.9 (9.2 - 12.9) | 22.8 (19.5 - 26.8) | 38.5 (32.9 - 44.9) | 7.4 (6.4 - 8.6) | 80.1 (68.4 - 93.5) |
| Mozambique | 12 | 17.2 (14.5 - 20.3) | 29.2 (24.9 - 34.0) | 39.2 (33.3 - 45.7) | 6.7 (5.8 - 7.7) | 92.8 (79.1 - 108.1) |
| Myanmar | 16 | 6.2 (5.5 - 7.0) | 12.0 (10.7 - 13.5) | 34.3 (30.5 - 38.7) | 13.2 (11.7 - 14.9) | 65.7 (58.6 - 74.1) |
| Nigeria | 4 | 39.8 (34.4 - 46.1) | 77.1 (67.5 - 89.2) | 107.3 (94.0 - 123.3) | 21.8 (19.2 - 24.9) | 247.1 (216.8 - 284.7) |
| Pakistan | 2 | 75.6 (64.4 - 88.3) | 129.9 (111.0 - 151.6) | 254.8 (217.6 - 297.1) | 62.0 (53.0 - 72.9) | 521.8 (445.7 - 610.5) |
| Philippines | 5 | 24.9 (21.8 - 28.6) | 44.7 (39.4 - 51.2) | 110.7 (97.2 - 126.7) | 36.4 (32.0 - 41.6) | 216.6 (190.4 - 247.6) |
| Russia | 20 | 2.5 (2.0 - 3.3) | 4.9 (3.9 - 6.3) | 12.2 (9.9 - 15.0) | 6.4 (5.3 - 7.8) | 26.5 (21.5 - 32.8) |
| South Africa | 9 | 14.7 (12.6 - 16.9) | 27.8 (23.9 - 31.9) | 61.6 (53.6 - 70.4) | 19.7 (17.0 - 22.6) | 125.2 (108.3 - 142.9) |
| Tanzania | 11 | 18.3 (15.6 - 21.4) | 30.7 (26.3 - 35.8) | 46.7 (40.0 - 54.6) | 10.1 (8.7 - 11.8) | 106.2 (91.1 - 123.4) |
| Uganda | 15 | 13.2 (11.6 - 15.0) | 23.4 (20.8 - 26.2) | 30.6 (27.0 - 34.6) | 5.0 (4.5 - 5.6) | 72.6 (64.2 - 81.6) |
| Vietnam | 13 | 6.6 (5.8 - 7.5) | 12.5 (10.9 - 13.9) | 44.8 (39.6 - 49.6) | 21.8 (19.4 - 24.3) | 85.3 (75.7 - 94.6) |
| Zambia | 18 | 7.7 (6.5 - 9.2) | 13.9 (11.9 - 16.5) | 22.3 (19.0 - 26.2) | 3.3 (2.9 - 3.9) | 47.5 (40.7 - 56.1) |
| Zimbabwe | 17 | 7.7 (6.6 - 9.1) | 14.1 (12.3 - 16.4) | 23.8 (20.6 - 27.7) | 4.6 (4.0 - 5.3) | 50.5 (43.7 - 58.6) |
Population sizes are in tens of thousands
The age distribution of the proportion of the population living in a household with a person with incident pulmonary TB showed higher probabilities among children in more than half of the countries (Angola, India, Kenya, Mozambique, Nigeria, Russia, Tanzania, Uganda, South Africa, Zambia, and Zimbabwe), while the distribution of this population was relatively even across age groups in the remaining countries (Fig 2). No country had a clear increasing trend in the probability across age groups, but the proportion was modestly higher in the 50 and older age group in Bangladesh, Indonesia, Pakistan, and Vietnam. Individual probabilities did not differ substantially by sex.
4. Discussion
We found that an estimated 38 million people in 20 countries were at elevated risk for TB in 2019 due to sharing a household with someone with incident pulmonary TB. To our knowledge, this is the first estimate of household contacts across high-incidence countries to include people of all ages. The size of the household contact population composed of older children, adolescents, and adults is more than 7-fold larger than estimated population of contacts under 5, indicating a large population that may be newly considered for TPT under recent guidelines.
Despite each country in our study having a high burden of TB, the factors driving the size of the population of household contacts vary somewhat by setting. In general, the factors contributing to a high total number of contacts include large populations, high TB incidence, and large household sizes, particularly for household configurations including people in age groups with high TB incidence. We estimated more than twice as many household contacts in India than any other country due to high values across these domains. Similarly, factors contributing to Pakistan's ranking with the second-largest number of contacts were the largest mean household size, high TB incidence, and a large population. The southern African countries of Mozambique, Zimbabwe, Zambia, and South Africa had high proportions of the population living in a household with someone with incident pulmonary TB, but lower absolute counts due to relatively smaller populations.
Our population size estimates likely represent upper bounds for the number of people potentially eligible for TPT in these 20 countries for two reasons. First, our calculation incorporates the number of people estimated to have developed incident pulmonary TB in 2019 in each location rather than the smaller number of people who access health services and have TB diagnosis confirmed and notified to public health programs. Second, local TB epidemiology and individual risk factors including TST or IGRA test results may further inform clinical decision making around TPT [10]. Using incidence estimates instead of case notification data generates a more comprehensive estimate of household contacts due to the substantial gap in TB diagnosis, where an estimated 2·9 million of the 10 million people who develop TB each year are not diagnosed and linked with public health reporting systems [2]. Though this gap is narrowing over time, reaching the “missing people with TB” and their household contacts will require a massive increase in effort globally. In contrast to our approach, prior studies began their estimation process using notifications of bacteriologically-confirmed TB, which may be a closer estimate to what is achievable by TB programs in the near-term, though these estimates did not include household contacts who were adolescents or adults [14,15,22].
Expanding household contact investigation to reach this large population would require a massive increase in time and resources, and so cost-effectiveness analysis of these interventions is critical for program planning. Depending on current local practice, implementing TPT to household contacts of all ages may require contacting additional households, screening additional people for active TB within households, and providing TPT associated clinical care. Swindells et al reported 512 attempts to reach 308 households for contact investigation, requiring a median time of four hours of staff person-time per attempt across sites in eight countries [23]. Alsdurf and colleagues estimated the time required for health care workers to complete all steps of evaluation of household contacts to be 1·8 hours to 5·2 hours per TB positive patient, though this estimate included steps for tuberculin skin test placement and reading, which may not be conducted in all settings [24]. Engaging community health workers may support this effort; Zawedde-Muyanja et al found improvements in screening of household contacts and initiation and completion of TPT among children in Uganda following training of community health workers [25]. Economic analysis of a multi-country (Benin, Canada, Ghana, Indonesia, and Vietnam) trial to strengthen management of household contacts of people with confirmed TB estimated a cost of the intervention and TPT-associated clinical care of CA$1348 per contact [26]. Investigators working in Vietnam calculated an incremental cost-effectiveness ratio of $544 per disability-adjusted life year averted for a serial screening program among household contacts of adults with smear-positive TB [27]. Despite the challenges of scale-up, a 2018 review indicated that screening for TB among child contacts <5 was included in national guidelines in 25 (83%) of TB high-burden countries, indicating a program priority for household contact investigation [28].
Our study brings several strengths and new contributions to this area of work in TB household contact investigation. First, our estimation process leverages the GBD 2019 study, which applies a standardized estimation framework across locations to yield results that are comparable across countries, are age- and sex-specific, and are updated annually. An incidence-based analysis is less subject to fluctuations that may impact case notifications, such as increases from enhanced case finding efforts or the substantial declines in case notifications observed during the COVID-19 pandemic [2]. Second, we simulated the distribution of household compositions by age-, sex-, and location using a large dataset of publicly available, nationally representative household surveys including more than 8·5 million households across the 20 countries. Further, both the GBD 2019 results and our household composition model include measures of uncertainty, which we propagate through our estimation framework to yield uncertainty estimates for each output measure. Finally, in a supplementary analysis, we calculated the household contacts of people with prevalent pulmonary TB who might be identified through population-based screening efforts like a TB prevalence survey.
Our study also has limitations. First, as our household composition estimation uses national surveys of the general population, systematic differences in the composition of TB-affected households versus unaffected households may bias our results. For example, socioeconomic factors and urban versus rural residence likely correlate with household size and TB risk in many settings. However, our focus on TB high-burden countries likely reduces the bias that may occur in lower-incidence countries where there may be greater concentration of TB risk among members of key and vulnerable populations. Our estimates of the number of household contacts aligns well with a systematic review and meta-analysis that identified an average of 3.8 contacts per TB index patient in 95 household contact investigation studies conducted in low- and middle-income countries [6]. Our household size estimates were lower than reported by several studies, including by Beyanga et al who identified a mean household size of 5·9 among 93 index patients with TB at several sites in Tanzania (our estimate of 5·3), and higher than others, including by Jerene et al who registered a mean household size of 3·7 among 6,015 index patients with TB in Ethiopia (our estimate 4·5) and Fox et al who reported household sizes of 3·3 in the control population and 3·9 in the intervention groups in a study of 10,964 index patients with TB in Vietnam (our estimate 4·6) [5,29,30]. Second, while TB prevalence and household composition vary within countries, our analysis does not consider subnational variation [31,32]. Third, GBD 2019 incidence estimates also have limitations due to data availability and inconsistencies between prevalence and mortality data [1]. Fourth, our household composition analysis used the most recent dataset available for each country, which for China was from 2000, and household composition has likely changed since then. Fifth, as we selected countries for our model based on incidence counts, some countries with high TB incidence rates but relatively smaller populations (e.g. eSwatini, Lesotho, Democratic People's Republic of Korea) were not included and may not be well-represented by countries with larger populations. Additionally, our analysis did not include Brazil or Thailand, despite being high-burden countries for TB in the updated ranking in GBD 2019. Finally, we assume that there is one person per household with prevalent TB per household in our estimate of the number of affected households. However, we estimate this effect of this assumption to be small based on the systematic review and meta-analysis by Hamada et al that found a summary estimate of 1·06 people per household simultaneously detected with prevalent TB in a synthesis of 58 studies (including >70% conducted in countries with medium or high TB burden) [15]. This indicates that our estimates are likely approximately 6% higher than they would be without assuming one person with TB per affected household.
Our country-specific estimates have important implications for policy. The UN High-Level Meeting on TB set an ambitious target to reach 20 million household contacts ages five and older for TPT by 2022, though current progress is far short of this target [2,4]. These estimates provide country-specific information to help reach this goal in the 20 countries that had more than 80% of the global total of new TB cases in 2019. As estimated for child household contacts globally or in the WHO Southeast Asia region, future modeling studies could build on this work by estimating the impact of reaching household contacts of all ages in averting TB disease and death and the associated cost [22,33]. Household contact investigation studies continue to show the benefit of TPT among household contacts of all ages, including the recent study by Paradkar et al reporting that 2% of household contacts at study sites in India developed TB in the first 24 months of follow-up [34]. The feasibility of scaling up household contact investigation to reach this larger population remains a concern, particularly in light of the negative impact of the COVID-19 pandemic on essential TB services globally [35]. However, the shared importance of household contact investigation in response to TB and COVID-19 has been identified as an opportunity to address both infections in parallel [36,37].
In conclusion, we estimated that the recent recommendation that household contacts ages five and older in high-incidence countries may receive TPT could expand the eligible population by more than 7-fold. This population is even larger than the target set at the 2018 UN High-Level Meeting on TB. Successfully reaching these household contacts for clinical evaluation and TPT may avert substantial TB-related morbidity and mortality.
Contributors
JMR and ADF conceptualized and designed the study. YX, YW, JRL, and ADF conducted the analysis. JC, CH, and ADF developed estimation software. JBD and PL managed the project. JMR wrote the first draft of the manuscript. All authors critically revised the manuscript.
Data Sharing Statement
These estimates will be available in the Global Health Data Exchange upon publication. http://ghdx.healthdata.org/
Funding
JMR is supported by the National Institute of Allergy and Infectious Diseases (K01 AI138620). This research was funded in part by a 2020 developmental grant from the University of Washington / Fred Hutch Center for AIDS Research, an NIH funded program under award number AI027757 which is supported by the following NIH Institutes and Centers: NIAID, NCI, NIMH, NIDA, NICHD, NHLBI, NIA, NIGMS, NIDDK. This work was funded in part by the National Science Foundation (DMS-1839116).
Declaration of Competing Interest
JMR reports grants from the National Institutes of Health during the conduct of the study. JBD reports other financial or non-financial interests from Sanofi. AES reports grant funding from the National Institutes of Health, Bill & Melinda Gates Foundation, and Vir Biotechnology. AES also reports serving on a data safety monitoring board. ADF reports work related to data licensing and analysis with Sanofi during the conduct of the study. ADF reports personal fees from Kaiser Permanente and NORC and other relationship with Agathos, Ltd outside of the submitted work. All other authors have nothing to declare.
Acknowledgements
The authors thank Meixin Zhang for help with literature review.
Footnotes
Supplementary material associated with this article can be found in the online version at doi:10.1016/j.eclinm.2021.101206.
Appendix. Supplementary materials
References
- 1.GBD 2019 Tuberculosis Collaborators Global, regional, and national sex differences in the global burden of tuberculosis by HIV status, 1990-2019: results from the Global Burden of Disease Study 2019. Lancet Infect Dis. 2021 doi: 10.1016/S1473-3099(21)00449-7. S1473-3099(21)00449-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.World Health Organization. Global Tuberculosis Report 2020. Geneva, Switzerland: World Health Organization, 2020.
- 3.United Nations General Assembly. Transforming our world : the 2030 Agenda for Sustainable Development. 2015 https://sdgs.un.org/goals.
- 4.Fukunaga R, Glaziou P, Harris JB, Date A, Floyd K, Kasaeva T. Epidemiology of Tuberculosis and Progress Toward Meeting Global Targets - Worldwide, 2019. MMWR Morb Mortal Wkly Rep. 2021;70:427–430. doi: 10.15585/mmwr.mm7012a4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fox GJ, Nhung NV, Sy DN, et al. Household-Contact Investigation for Detection of Tuberculosis in Vietnam. N Engl J Med. 2018;378:221–229. doi: 10.1056/NEJMoa1700209. [DOI] [PubMed] [Google Scholar]
- 6.Fox GJ, Barry SE, Britton WJ, Marks GB. Contact investigation for tuberculosis: a systematic review and meta-analysis. Eur Respir J. 2013;41:140–156. doi: 10.1183/09031936.00070812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Morrison J, Pai M, Hopewell PC. Tuberculosis and latent tuberculosis infection in close contacts of people with pulmonary tuberculosis in low-income and middle-income countries: a systematic review and meta-analysis. Lancet Infect Dis. 2008;8:359–368. doi: 10.1016/S1473-3099(08)70071-9. [DOI] [PubMed] [Google Scholar]
- 8.Smieja MJ, Marchetti CA, Cook DJ, Smaill FM. Isoniazid for preventing tuberculosis in non-HIV infected persons. Cochrane Database Syst Rev. 2000 doi: 10.1002/14651858.CD001363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.World Health Organization. Latent tuberculosis infection: updated and consolidated guidelines for programmatic management. Geneva, Switzerland, 2018. [PubMed]
- 10.WHO consolidated guidelines on tuberculosis: tuberculosis preventive treatment: Module 1: prevention. Geneva: World Health Organization, 2020 http://www.ncbi.nlm.nih.gov/books/NBK554956/. (accessed Oct 8, 2021). [PubMed]
- 11.Martinez L, Cords O, Horsburgh CR, Andrews JR. Pediatric TB Contact Studies Consortium. The risk of tuberculosis in children after close exposure: a systematic review and individual-participant meta-analysis. Lancet. 2020;395:973–984. doi: 10.1016/S0140-6736(20)30166-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stop TB Partnership. UN high-level meeting on TB: key targets & commitments for 2022. 2018 http://www.stoptb.org/assets/documents/global/advocacy/unhlm/UNHLM_Targets&Commitments.pdf. (accessed Feb 11, 2020).
- 13.Dodd PJ, Gardiner E, Coghlan R, Seddon JA. Burden of childhood tuberculosis in 22 high-burden countries: a mathematical modelling study. Lancet Glob Health. 2014;2:e453–e459. doi: 10.1016/S2214-109X(14)70245-1. [DOI] [PubMed] [Google Scholar]
- 14.Yuen CM, Jenkins HE, Chang R, Mpunga J, Becerra MC. Two methods for setting child-focused tuberculosis care targets. Public Health Action. 2016;6:83–96. doi: 10.5588/pha.16.0022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hamada Y, Glaziou P, Sismanidis C, Getahun H. Prevention of tuberculosis in household members: estimates of children eligible for treatment. Bull World Health Organ. 2019;97:534–547D. doi: 10.2471/BLT.18.218651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Stevens GA, Alkema L, Black RE, et al. Guidelines for Accurate and Transparent Health Estimates Reporting: the GATHER statement. Lancet. 2016;388:e19–e23. doi: 10.1016/S0140-6736(16)30388-9. [DOI] [PubMed] [Google Scholar]
- 17.Horst C, Swedin K, Xie Y, Yaqi W, Flaxman A, Collins J. Vivarium LTBI. 2020 https://github.com/ihmeuw/vivarium_csu_ltbi/tree/main/src/vivarium_csu_ltbi/household_tb_model.
- 18.Wang H, Abbas KM, Abbasifard M, et al. Global age-sex-specific fertility, mortality, healthy life expectancy (HALE), and population estimates in 204 countries and territories, 1950–2019: a comprehensive demographic analysis for the Global Burden of Disease Study 2019. The Lancet. 2020;396:1160–1203. doi: 10.1016/S0140-6736(20)30977-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.GBD 2019 Risk Factors Collaborators Global burden of 87 risk factors in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020;396:1223–1249. doi: 10.1016/S0140-6736(20)30752-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tymeson H. Adapting spatiotemporal Gaussian process regression for multinomial data. 2019. http://hdl.handle.net/1773/43877.
- 21.Flaxman AD, Vos T, Murray CJL, editors. An integrative metaregression framework for descriptive epidemiology. University of Washington Press; Seattle: 2015. [Google Scholar]
- 22.Dodd PJ, Yuen CM, Becerra MC, Revill P, Jenkins HE, Seddon JA. Potential effect of household contact management on childhood tuberculosis: a mathematical modelling study. Lancet Glob Health. 2018;6:e1329–e1338. doi: 10.1016/S2214-109X(18)30401-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Swindells S, Gupta A, Kim S, et al. Resource utilization for multidrug-resistant tuberculosis household contact investigations (A5300/I2003) Int J Tuberc Lung Dis. 2018;22:1016–1022. doi: 10.5588/ijtld.18.0163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Alsdurf H, Oxlade O, Adjobimey M, et al. Resource implications of the latent tuberculosis cascade of care: a time and motion study in five countries. BMC Health Serv Res. 2020;20:341. doi: 10.1186/s12913-020-05220-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zawedde-Muyanja S, Nakanwagi A, Dongo JP, et al. Decentralisation of child tuberculosis services increases case finding and uptake of preventive therapy in Uganda. Int J Tuberc Lung Dis Off J Int Union Tuberc Lung Dis. 2018;22:1314–1321. doi: 10.5588/ijtld.18.0025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Oxlade O, Benedetti A, Adjobimey M, et al. Effectiveness and cost-effectiveness of a health systems intervention for latent tuberculosis infection management (ACT4): a cluster-randomised trial. Lancet Public Health. 2021 doi: 10.1016/S2468-2667(20)30261-9. published online March 19. [DOI] [PubMed] [Google Scholar]
- 27.Lung T, Marks GB, Nhung NV, et al. Household contact investigation for the detection of tuberculosis in Vietnam: economic evaluation of a cluster-randomised trial. Lancet Glob Health. 2019;7:e376–e384. doi: 10.1016/S2214-109X(18)30520-5. [DOI] [PubMed] [Google Scholar]
- 28.Jagger A, Reiter-Karam S, Hamada Y, Getahun H. National policies on the management of latent tuberculosis infection: review of 98 countries. Bull World Health Organ. 2018;96:173–184F. doi: 10.2471/BLT.17.199414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Beyanga M, Kidenya BR, Gerwing-Adima L, Ochodo E, Mshana SE, Kasang C. Investigation of household contacts of pulmonary tuberculosis patients increases case detection in Mwanza City, Tanzania. BMC Infect Dis. 2018;18:110. doi: 10.1186/s12879-018-3036-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jerene D, Melese M, Kassie Y, et al. The yield of a tuberculosis household contact investigation in two regions of Ethiopia. Int J Tuberc Lung Dis. 2015;19:898–903. doi: 10.5588/ijtld.14.0978. [DOI] [PubMed] [Google Scholar]
- 31.Mistry D, Litvinova M, Pastore Y, Piontti A, et al. Inferring high-resolution human mixing patterns for disease modeling. Nat Commun. 2021;12:323. doi: 10.1038/s41467-020-20544-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ross JM, Henry NJ, Dwyer-Lindgren LA, et al. Progress toward eliminating TB and HIV deaths in Brazil, 2001-2015: a spatial assessment. BMC Med. 2018;16:144. doi: 10.1186/s12916-018-1131-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mandal S, Bhatia V, Sharma M, Mandal PP, Arinaminpathy N. The potential impact of preventive therapy against tuberculosis in the WHO South-East Asian Region: a modelling approach. BMC Med. 2020;18:163. doi: 10.1186/s12916-020-01651-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Paradkar M, Padmapriyadarsini C, Jain D, et al. Tuberculosis preventive treatment should be considered for all household contacts of pulmonary tuberculosis patients in India. PloS One. 2020;15 doi: 10.1371/journal.pone.0236743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.World Health Organization. Impact of the COVID-19 pandemic on TB detection and mortality in 2020. 2021. https://www.who.int/publications/m/item/impact-of-the-covid-19-pandemic-on-tb-detection-and-mortality-in-2020.
- 36.Hopewell PC, Reichman LB, Castro KG. Parallels and Mutual Lessons in Tuberculosis and COVID-19 Transmission, Prevention, and Control. Emerg Infect Dis. 2021;27:681–686. doi: 10.3201/eid2703.203456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Loveday M, Cox H, Evans D, et al. Opportunities from a new disease for an old threat: Extending COVID-19 efforts to address tuberculosis in South Africa. S Afr Med J. 2020;110:1160–1167. doi: 10.7196/SAMJ.2020.v110i12.15126. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.