Abstract
Background: The study provides a novel prediction model for COVID-19 progression and outcome by the combination of the CD8+: B-cells ratio with neutrophil-to-lymphocyte ratio (NLR). Patients and Methods: Immune phenotyping was performed in 120 COVID-19 patients. Results: A decrease in CD8+:B-cell (p<0.0001) and in lymphocyte-to-CRP (LCR) ratio (p<0.0001) was observed in intubated patients versus non-intubated with an increase for CD4+:CD8+ (p<0.01), NLR (p<0.0001) and CRP: Albumin (p<0.001). Receiving operating curve (ROC) analysis predicting requirement for mechanical ventilation revealed the highest AUC for CD8+:B-cells, (AUC=0.795, p<0.001) versus NLR (AUC=0.783, p<0.001), LCR (AUC=0.779, p<0.001), Albumin:CRP (AUC=0.750, p<0.001) and CD4+:CD8+ (AUC=0.779, p<0.001). Combination of the CD8+: B-cell ratio with the NLR increased the AUC (AUC=0.845, p<0.001). The combined ratios correlated with outcome defined as duration of hospital (r=0.435, p<0.001) or ICU stay (r=0.596, p<0.001). Conclusion: Combination of the CD8+: B-cell ratio and NLR serves as a useful prognostic tool for COVID-19 patient progression.
Keywords: SARS-COV-2, COVID-19, immune phenotyping, prognostic markers, disease progression, mechanical ventilation, intensive care unit
More than a year on, the SARS-COV-2 pandemic has led to the death of thousands of people worldwide and has posed an unprecedented burden on health systems across the world. Although the majority of patients experience mild symptoms mimicking those of the common cold, in a small number of cases disease from SARS-COV-2 known as coronavirus 2019 disease (COVID-19) causes pneumonia leading to acute respiratory distress syndrome (ARDS) and possible need for mechanical ventilation and intensive care unit (ICU) treatment (1). These patients usually exhibit elevated levels of cytokines such as IL-6, known as ‘cytokine storm’ accompanied by an increase in pro-inflammatory markers such as ferritin and C-reactive protein (CRP). In this regard, more specific markers indicating disease deterioration and outcome have become an urgent need.
Immune phenotyping allowed the characterization of the immune profile of COVID-19 patients, thus providing an insight to the immune response towards COVID-19; it is now known that COVID-19 patients with severe and/or critical disease exhibit markedly reduced blood cell subpopulations (2). Therefore, immune phenotyping may also serve as a potential source for markers of COVID-19 progression. To this end, CD4+ and CD8+ counts have already been proposed as markers for COVID-19 progression (3).
The ability of previously reported ratios based on either immune phenotyping such as the CD8+:B-cells and the CD4+:CD8+ or on standard clinical parameters such as the neutrophil to lymphocyte ratio (NLR), the lymphocyte to CRP ratio (LCR) and the CRP:Albumin ratio to predict the need for intubation in COVID-19 patients was assessed in the present study. The combination of the CD8+:B-cells with the NLR was found as a powerful prediction tool for disease progression and outcome.
Patients and Methods
Patients. A written informed consent was obtained from all patients. Ethical approval was obtained from the Ethics committee of Evangelismos Hospital. A total of 120 patients from the second wave of COVID-19 were recruited. All patients had a positive polymerase chain reaction (PCR) test for SARS-COV-2 from a nasopharyngeal sample. Patients were grouped into those who did not require mechanical ventilation (non-intubated, n=71) and those who required mechanical ventilation (intubated, n=49). Blood samples were obtained on admission for all patients and analyzed on the same day. Demographic and clinical data collected included age, gender, comorbidities, lab values on admission, initial vital signs upon presentation to hospital, duration of hospital or ICU stay (days), duration of mechanical ventilation (days), blood cell counts and in-hospital mortality. Blood cell counts on admission were used to calculate the NLR, LCR and the CRP:Albumin ratio. More specifically, the absolute neutrophil count was divided by the absolute lymphocyte count, the absolute lymphocyte count was divided by CRP values and CRP values were divided by albumin values to obtain the NLR, LCR and CRP:Albumin ratios, respectively.
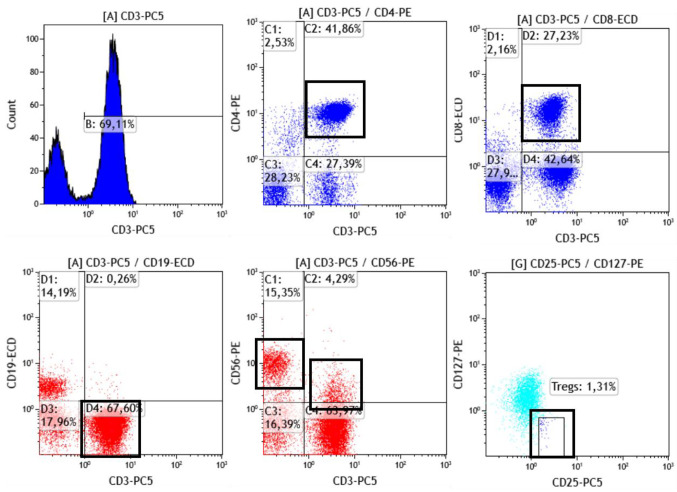
Immune phenotyping. Immune phenotyping was performed in whole blood samples of patients by flow cytometric analysis. The TQprep workstation was used for lysis, lymphocyte preparation and staining prior to flow cytometric analysis in a Navios EX flow cytometer (Beckman Coulter, Brea, CA, USA). Antibodies used for cell staining are listed in Table I. Data were analyzed using the Kaluza flow cytometric analysis software (Beckman Coulter). A representative flow cytometric analysis of lymphocyte subsets and gating strategy is shown in Figure 1.
Table I. List of antibodies utilized for immune profiling of patients by flow cytometric analysis.
Figure 1. Representative gating strategy for immune profiling of coronavirus disease 2019 (COVID-19) patients.
Statistical analysis. Results are reported as absolute numbers, medians, or means and standard deviations, as appropriate. No imputation was made for missing data. Statistical analysis was performed using the SPSS (IBM, Armonk, NY, USA) and GraphPad Prism 8.0 software (GraphPad, San Diego, CA, USA). Data were tested for normality using the Shapiro-Wilk test. Unpaired t-test or Mann-Whitney U-test were used for normally and non-normally distributed data, respectively. A paired t-test was used for comparisons between variables within the same group in the event of normally distributed data and a Wilcoxon test was used for comparison of data within the same group without normality. Spearman correlation was used for correlation of data. p<0.05 was considered statistically significant.
Results
Patient characteristics. Patient characteristics and clinical data on admission are provided in Table II. The mean age of non-intubated and intubated patients was 56.31±15.99 and 63.69±10.95 years, respectively. The most frequent symptoms were fever, cough and dyspnea. Statistically significant differences were observed for levels of various clinical parameters recorded, such as CRP, troponin, urea, aspartate aminotransferase (AST), alanine transaminase (ALT), lactate dehydrogenase (LDH) and albumin (Table II). A better outcome was observed for the non-intubated patients, estimated by the smaller duration of hospitalization (days) as well as survival.
Table II. Patient clinical and demographic data.
Data are presented as mean±SD or absolute number (percentage of group).
Prediction of the need for intubation in COVID-19 patients. A statistically significant decrease of CD8+:B-cell ratio (p<0.0001) was observed in intubated compared to non-intubated patients (Figure 2), whereas a statistically significant increase of the CD4+:CD8+ ratio (p<0.001) was shown. A significant increase was observed in intubated patients versus non-intubated for NLR (p<0.0001) and CRP:Albumin (p<0.0001), whereas a significant decrease was shown for LCR (p<0.0001) (Figure 2). The same ratios were also determined in a subsequent analysis between survivors (n=99) and non-survivors (n=19) of the same cohort of patients. CD8+:B-cell ratio significantly decreased in the non-survivors group (p<0.0001) (Figure 3). Although significant, the increase of CD4+:CD8+ (p<0.01), NLR (p<0.05), LCR (p<0.05) and CRP:Albumin (p<0.05) ratios in non-survivors versus survivors was markedly weaker compared to CD8+:B-cell ratio (Figure 3).
Figure 2. Severe decrease of the CD8+: B-cell ratio in intubated patients. A significant decrease of the CD8+: B-cell ratio was observed between non-intubated and intubated (A) as well as an increase for CD4+:CD8+ (B), neutrophil-to-lymphocyte ratio (NLR), a decrease in lymphocyte-toCRP ratio (C) and an increase in CRP:Albumin ratio (E). Statistical analysis was performed by Mann-Whitney U-test. ****p<0.0001, ***p<0.001.
Figure 3. Severe decrease of the CD8+: B-cell ratio in non-survivors versus survivors. A significant decrease of the CD8+: B-cell ratio was observed between non-intubated and intubated patients (A) as well as an increase for CD4+:CD8+ (B), neutrophil-to-lymphocyte ratio (NLR), a decrease in lymphocyte-to-CRP ratio (C) and an increase in CRP:Albumin (E). Statistical analysis was performed by Mann-Whitney U-test. *p<0.05, **p<0.01, ****p<0.0001.
Prognostic value of the CD8+:B-cell ratio in combination with the NLR. Receiving operating curve (ROC) analysis was performed in order to compare the aforementioned ratios on their ability to predict the need for intubation in COVID-19 patients. In Figure 4A, curves derived from ROC analysis on all ratios are shown. As described in Table III, the AUC of the CD8+:B-cell ratio was the highest among the ratios included in the study (AUC=0.795, p<0.0001) followed by NLR which also had a high AUC (AUC=0.783, p<0.0001) whereas the lowest AUC value was obtained for the CD4+:CD8+ ratio (AUC=0.664, p<0.002) confirming its lower potential as a predictor for COVID-19 progression.
Figure 4. Prognostic value of the CD8+: B cell ratio. Receiving operating curve analysis for predicting the requirement of mechanical ventilation in COVID-19 patients by (A) CD8+:B cell, CD4+:CD8+, neutrophil-toly mphocyte ratio (NLR), lymphocyte-to-CRP ratio (LCR) and CRP:Albumin ratio and (B) by the combination of CD8+: B cells with NLR.
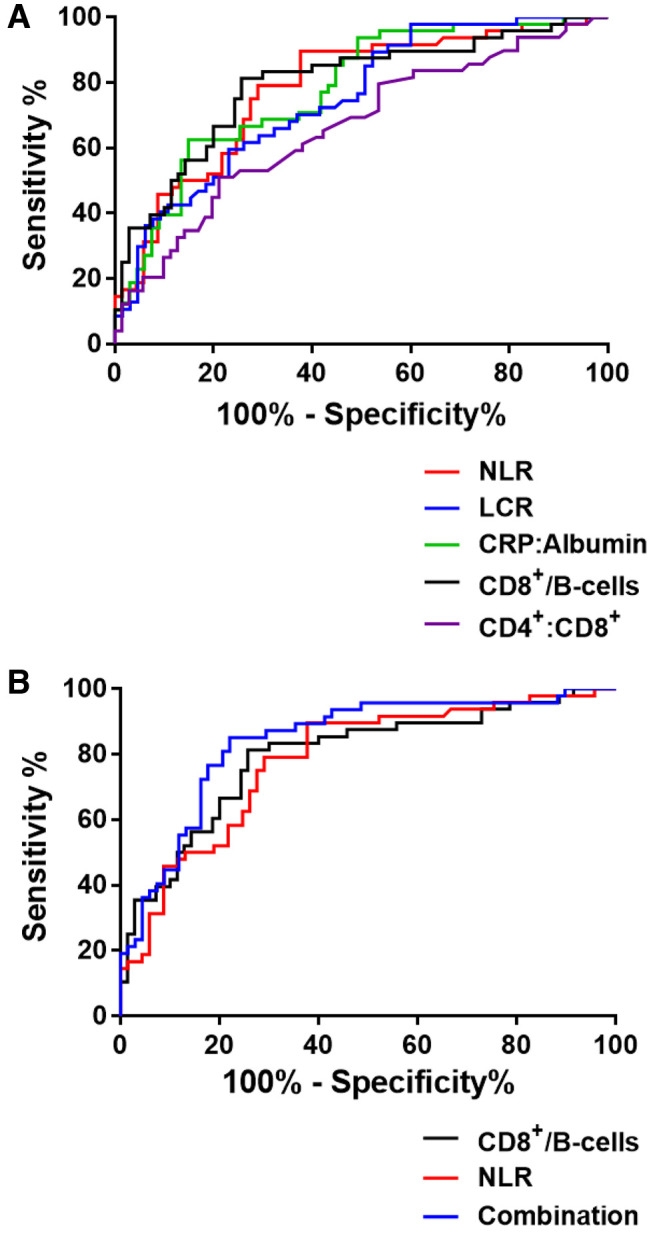
Table III. Receiving operating curve (ROC) analysis of ratios for prediction of COVID-19 patient progression.
We next combined the CD8+:B-cell ratio with the NLR in order to examine the performance of the combined ratios as a prognostic marker. The combination of the CD8+:B-cell ratio with the NLR exhibited the highest AUC (AUC=0.845, p<0.0001) (Table III). Specificity of the combined ratios was 85.11% and sensitivity was 77.94%, with a cut-off at 0.388 and odds ratio (OR) of 3.858, suggesting that a combination of the two ratios could be utilized as a highly predictive tool for COVID-19 progression (Figure 4B).
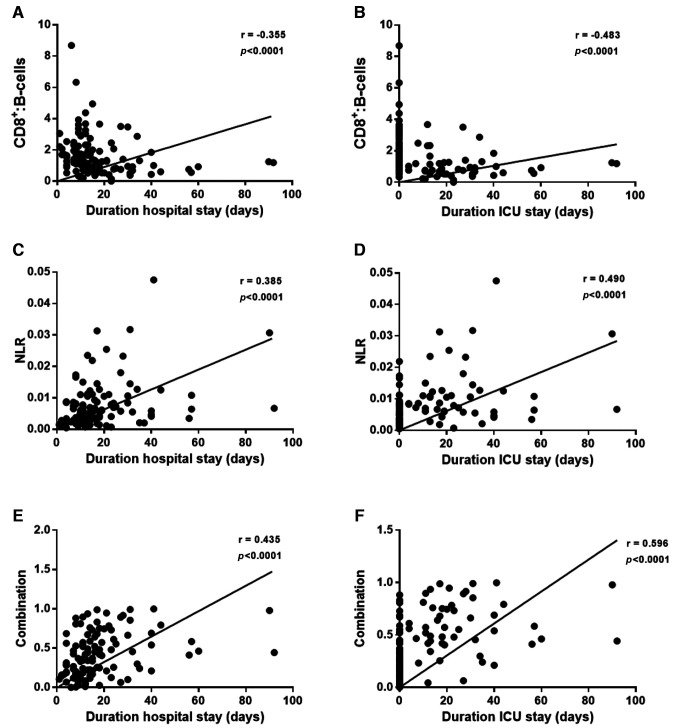
The correlation of the CD8+:B-cell, NLR and combination ratios with outcomes defined as either duration of hospitalization (days) or duration of ICU stay (days) were examined next. A statistically significant correlation with outcome was obtained for CD8+:B-cell ratio, although this was stronger for ICU stay (r=-0.483, p<0.0001) compared to duration of hospitalization (r=-0.355, p<0.0001) (Figure 5). Similarly, a statistically significant positive correlation was observed for NLR with outcome and again this was stronger for outcome in ICU (r=0.490, p<0.0001) than for duration of hospitalization (r=0.385, p<0.0001). Combination of the two ratios resulted in a statistically significant correlation with outcome for duration of hospitalization (r=0.435, p<0.0001) and ICU stay (r=0.596 p<0.0001) (Figure 5) thus indicating that combining the two ratios can be a powerful tool for predicting both progression and outcome of COVID-19 patients.
Figure 5. Combined CD8+: B cell and NLR ratios as a prognostic tool for COVID-19 outcome. Correlation analysis revealed a negative correlation of CD8+:B cells with (A) outcome defined as duration of hospitalization (days) and (B) outcome defined as duration of ICU stay. Positive correlation with outcome for NLR (C, D) and combined CD8+: B cell ratio with NLR (E, F).
Discussion
COVID-19 has been particularly challenging in terms of successful patient management, partly due to the individual disease characteristics such as rapid deterioration of patients into ARDS and the need for mechanical ventilation as well as due to the lack of successful therapeutic regiments. To guide clinical practice and assist clinicians in predicting progression of patients into severe and/or critical cases, numerous markers have been proposed as prediction tools for COVID-19 progression.
To this end we reported a ratio of CD8+:B-cells with an increased sensitivity in predicting the need of COVID-19 patients for intubation and thus ICU treatment (4). In the current study, our initial findings have been verified in a large cohort of patients of the second wave of COVID-19, confirming its predictive value. Our results showed a severe decrease of CD8+:B-cells in intubated compared to non-intubated patients (p<0.0001), as well as between non-survivors and survivors (p<0.0001) and significant correlations of CD8+:B-cells with CRP, PaO2/FiO2, LDH and NKT cells, in line with our previous findings (4).
Immune phenotyping of COVID-19 patients has assisted in the characterization of the immune profiles such as severe lymphopenia (5-6). Due to the characteristic reduction in lymphocytes, specific subtypes such as CD4+ and CD8+ cells have been proposed as markers for COVID-19 progression. A prospective study by Calvet et al., reported low CD4+ and CD8+ counts as predictors for COVID-19 patients progressing into severe and/or critical disease and suggested immune phenotype as an admission routine test for COVID-19 patients (3). CD8+ counts have also been reported as a predictor for poor patient prognosis, especially in combination with obesity (7), whereas Huang et al. reported CD4+ and CD8+ counts as independent predictors for COVID-19 patient outcomes (8). Furthermore, an increase of the CD4+:CD8+ ratio has been proposed as a risk factor for critical illness in COVID-19 (9). In our study, although an increase in CD4+:CD8+ was observed, it exhibited a low AUC for predicting the need for intubation compared to the CD8+:B-cell ratio, suggesting a poor prognostic value of this specific ratio.
However, immune phenotyping is not always performed as standard of care and various other markers have been reported for COVID-19 progression and are widely used mainly based on routine clinical parameters. The NLR is one of the most frequently reported ratios for COVID-19 progression with a high predictive value. Numerous studies have reported the NLR as a strong predictor of poor prognosis and progression to critical illness (10-13) as well as a prognosis factor of intubation (14) and an independent risk factor for mortality (14-16). Similar reports have been published for the LCR (17-16) and the CRP:Albumin ratio (18). Comparing the CD8+:B-cell ratio and NLR as predictors of patient need for intubation, the CD8+:B-cell showed a higher performance with an AUC of 0.795 compared to an AUC of 0.783 for the NLR. However, combining the two ratios resulted in an AUC of 0.842, suggesting a powerful marker for prediction of the need for mechanical ventilation. An improved performance of the correlations with outcomes, defined as either duration of hospitalization or duration of ICU stay was also observed, pointing to the combined ratio as a strong predictor for COVID-19 progression and outcome. Taken together, these observations introduce the combination of the CD8+:B-cell ratio with NLR as a means to improve performance of both markers for prediction of COVID-19 patient deterioration. Furthermore, the current study underlines the importance of immune phenotyping in monitoring of COVID-19 patient progression and its use as a source of successful prognostic markers.
Conclusion
In summary, the current study identifies the combination of the CD8+:B-cell ratio with NLR as a novel predictor of COVID-19 progression and outcome and stresses the importance of the correct use of markers for assessment of COVID-19 patients.
Conflicts of Interest
The Authors declare no competing interests.
Authors’ Contributions
MGD analyzed and interpreted the data and wrote the manuscript. EG, KP, EJ, TS, AN, and AZ contributed to the acquisition of data, ID, SEO contributed to data analysis, AT contributed to the conception and co-ordination of the study, AK contributed to conception and design of the study and supervised the analysis. All Authors read and approved the manuscript.
References
- 1.Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liu J, Li S, Liu J, Liang B, Wang X, Wang H, Li W, Tong Q, Yi J, Zhao L, Xiong L, Guo C, Tian J, Luo J, Yao J, Pang R, Shen H, Peng C, Liu T, Zhang Q, Wu J, Xu L, Lu S, Wang B, Weng Z, Han C, Zhu H, Zhou R, Zhou H, Chen X, Ye P, Zhu B, Wang L, Zhou W, He S, He Y, Jie S, Wei P, Zhang J, Lu Y, Wang W, Zhang L, Li L, Zhou F, Wang J, Dittmer U, Lu M, Hu Y, Yang D, Zheng X. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55:102763. doi: 10.1016/j.ebiom.2020.102763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Calvet J, Gratacós J, Amengual MJ, Llop M, Navarro M, Moreno A, Berenguer-Llergo A, Serrano A, Orellana C, Cervantes M. CD4 and CD8 lymphocyte counts as surrogate early markers for progression in SARS-CoV-2 pneumonia: A prospective study. Viruses. 2020;12(11):1277. doi: 10.3390/v12111277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Detsika MG, Ampelakiotou K, Grigoriou E, Psarra K, Jahaj E, Roussos C, Dimopoulou I, Orfanos SE, Tsirogianni A, Kotanidou A. A novel ratio of CD8+:B-cells as a prognostic marker of coronavirus disease 2019 patient progression and outcome. Virology. 2021;556:79–86. doi: 10.1016/j.virol.2021.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chen G, Wu D, Guo W, Cao Y, Huang D, Wang H, Wang T, Zhang X, Chen H, Yu H, Zhang X, Zhang M, Wu S, Song J, Chen T, Han M, Li S, Luo X, Zhao J, Ning Q. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. 2020;130(5):2620–2629. doi: 10.1172/JCI137244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Diao B, Wang C, Tan Y, Chen X, Liu Y, Ning L, Chen L, Li M, Liu Y, Wang G, Yuan Z, Feng Z, Zhang Y, Wu Y, Chen Y. Reduction and functional exhaustion of T cells in patients with Coronavirus disease 2019 (COVID-19) Front Immunol. 2020;11:827. doi: 10.3389/fimmu.2020.00827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Urra JM, Cabrera CM, Porras L, Ródenas I. Selective CD8 cell reduction by SARS-CoV-2 is associated with a worse prognosis and systemic inflammation in COVID-19 patients. Clin Immunol. 2020;217:108486. doi: 10.1016/j.clim.2020.108486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Huang W, Berube J, McNamara M, Saksena S, Hartman M, Arshad T, Bornheimer SJ, O’Gorman M. Lymphocyte subset counts in COVID-19 patients: A meta-analysis. Cytometry A. 2020;97(8):772–776. doi: 10.1002/cyto.a.24172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pallotto C, Suardi LR, Esperti S, Tarquini R, Grifoni E, Meini S, Valoriani A, Di Martino S, Cei F, Sisti E, Piani F, Botta A, Salomoni E, Baragli F, Blanc P. Increased CD4/CD8 ratio as a risk factor for critical illness in coronavirus disease 2019 (COVID-19): a retrospective multicentre study. Infect Dis (Lond) 2020;52(9):675–677. doi: 10.1080/23744235.2020.1778178. [DOI] [PubMed] [Google Scholar]
- 10.Lian J, Jin C, Hao S, Zhang X, Yang M, Jin X, Lu Y, Hu J, Zhang S, Zheng L, Jia H, Cai H, Zhang Y, Yu G, Wang X, Gu J, Ye C, Yu X, Gao J, Yang Y, Sheng J. High neutrophil-to-lymphocyte ratio associated with progression to critical illness in older patients with COVID-19: a multicenter retrospective study. Aging (Albany NY) 2020;12(14):13849–13859. doi: 10.18632/aging.103582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ma A, Cheng J, Yang J, Dong M, Liao X, Kang Y. Neutrophil-to-lymphocyte ratio as a predictive biomarker for moderate-severe ARDS in severe COVID-19 patients. Crit Care. 2020;24(1):288. doi: 10.1186/s13054-020-03007-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Liu L, Zheng Y, Cai L, Wu W, Tang S, Ding Y, Liu W, Kou G, Xiong Z, Wang S, Zheng S. Neutrophil-to-lymphocyte ratio, a critical predictor for assessment of disease severity in patients with COVID-19. Int J Lab Hematol. 2021;43(2):329–335. doi: 10.1111/ijlh.13374. [DOI] [PubMed] [Google Scholar]
- 13.Yang AP, Liu JP, Tao WQ, Li HM. The diagnostic and predictive role of NLR, d-NLR and PLR in COVID-19 patients. Int Immunopharmacol. 2020;84:106504. doi: 10.1016/j.intimp.2020.106504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tatum D, Taghavi S, Houghton A, Stover J, Toraih E, Duchesne J. Neutrophil-to-lymphocyte ratio and outcomes in Louisiana COVID-19 patients. Shock. 2020;54(5):652–658. doi: 10.1097/SHK.0000000000001585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vafadar Moradi E, Teimouri A, Rezaee R, Morovatdar N, Foroughian M, Layegh P, Rezvani Kakhki B, Ahmadi Koupaei SR, Ghorani V. Increased age, neutrophil-to-lymphocyte ratio (NLR) and white blood cells count are associated with higher COVID-19 mortality. Am J Emerg Med. 2021;40:11–14. doi: 10.1016/j.ajem.2020.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lagunas-Rangel FA. Neutrophil-to-lymphocyte ratio and lymphocyte-to-C-reactive protein ratio in patients with severe coronavirus disease 2019 (COVID-19): A meta-analysis. J Med Virol. 2020;92(10):1733–1734. doi: 10.1002/jmv.25819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ullah W, Basyal B, Tariq S, Almas T, Saeed R, Roomi S, Haq S, Madara J, Boigon M, Haas DC, Fischman DL. Lymphocyte-to-C-reactive protein ratio: A novel predictor of adverse outcomes in COVID-19. J Clin Med Res. 2020;12(7):415–422. doi: 10.14740/jocmr4227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Karakoyun I, Colak A, Turken M, Altin Z, Arslan FD, Iyilikci V, Yilmaz N, Kose S. Diagnostic utility of C-reactive protein to albumin ratio as an early warning sign in hospitalized severe COVID-19 patients. Int Immunopharmacol. 2021;91:107285. doi: 10.1016/j.intimp.2020.107285. [DOI] [PMC free article] [PubMed] [Google Scholar]