Abstract
Aims: Soluble suppression of tumorigenicity 2 (sST2) was validated to independently predict prognosis for heart failure (HF) and ST-segment elevation myocardial infarction (STEMI). In this study, we aimed to evaluate the relation between sST2 and coronary artery stenosis, and no-reflow phenomenon and one-year prognosis in patients with non-ST-segment elevation acute coronary syndrome (NSTE-ACS).
Methods: This prospective study consecutively enrolled 205 patients who were diagnosed with NSTE-ACS and underwent percutaneous coronary intervention (PCI). sST2 was measured for all patients during admission. Patients were divided into two groups based on the optimal cutoff value: sST2 >34.2 ng/ml and sST2 ≤ 34.2 ng/ml groups.
Results: Patients in the sST2 >34.2 ng/ml group was associated with higher Gensini scores and multivessel disease. sST2 had weak predictive value for no-reflow phenomenon (area under the curve [AUC], 0.662; 95% confidence interval [CI], 0.53–0.79; P =0.015) with 66.7% sensitivity and 65.2% specificity, and it also had independent predictive value of no-reflow phenomenon after adjusting for confounding factors (odds ratio [OR], 3.802; 95% CI, 1.03–14.11; P =0.046). sST2 >34.2 ng/ml had a commendable predictive value for the one-year prognosis (AUC, 0.84; 95% CI, 0.75–0.93; P <0.001) with 72% sensitivity and 84% specificity, and it independently predicted one-year major cardiovascular and cerebrovascular events (MACCE) (hazard ratio [HR], 10.22; 95% CI, 4.05–25.7; P <0.001).
Conclusion: The sST2 concentration on admission is correlated with the degree of coronary artery stenosis. sST2 can predict both no-reflow and MACCE in patients with NSTE-ACS after PCI and was an independent predictor of MACCE and no-reflow phenomenon.
Keywords: Soluble ST2, Non-ST-elevation acute coronary syndrome, Percutaneous coronary intervention, No-reflow phenomenon, Prognosis
Introduction
Acute coronary syndrome (ACS) is a common critical illness in cardiovascular disease and encompasses ST-segment elevation myocardial infarction (STEMI), non-ST-segment elevation myocardial infarction (NSTEMI), and unstable angina (UA) 1) . With the current technology improvement and new drugs, the prognosis of ACS remains unsatisfactory. Biomarkers (e.g., cardiac troponin I [cTnI] and N-terminal pro-B-type natriuretic peptide [NT-proBNP]) are acknowledged as useful clinical tools 2) . They have the advantage of being convenient and fast detection; therefore, finding new and more valuable markers to predict the complication and prognosis of ACS is of great significance.
The suppression of tumorigenicity 2 (ST2) is a novel biomarker in cardiovascular disease and is a member of interleukin (IL)-1 receptor family 3) . There are two main subtypes of proteins that are encoded by the ST2 gene. One is ST2L, a membrane-bound receptor. When binding to IL-33 ligand, it can promote a T helper 2 (Th2) immune response and protect the heart. The other subtype is soluble suppression of tumorigenicity 2 (sST2), which can bind to IL-33 and block the binding of IL-33 and ST2L, thereby weakening the cardiovascular protection of IL-33/ST2L signaling pathway 1 , 4) . As a mechanical stress-induced myocardial protein, sST2 is expressed from cardiac myocytes, cardiac fibroblasts, and endothelial cells in patients with acute myocardial infarction and heart failure (HF) 5 , 6) . A large amount of sST2 binds to free IL-33 in extracellular fluid, which effectively reduces the binding of ST2L to IL-33 and weakens the protective effect of IL-33 on the myocardium 6) . Previous studies suggested that sST2 is associated with the severity and prognosis of HF 7 - 9) . Studies also proved that sST2 is related to no-reflow phenomenon and prognosis in patients with STEMI 10 , 11) . Whether the relationship between sST2 with STEMI is also applicable to patients with NSTE-ACS, however, is unclear. To our knowledge, we were the first to investigate the predictive value of sST2 for coronary stenosis, no-reflow phenomenon, and prognosis in patients with NSTE-ACS after percutaneous coronary intervention (PCI).
Materials and Methods
In this prospective single-center study, we consecutively included 205 patients who were admitted to the Department of Cardiology of Shengjing Hospital between October 2018 and February 2019, diagnosed as NSTE-ACS, and treated with PCI. Overall, 57 patients were diagnosed with NSTEMI, and 148 were diagnosed with UA. This study was approved by the Ethics Committee of Shengjing Hospital (ethics number: 2018PS519K), and all patients signed informed consent. The registration number of this study is ChiCTR1800018952.
All patients were diagnosed according to the criteria of the American College of Cardiology (ACC) or American Heart Association (AHA), which include angina, ST-segment depression, T wave negativity or flattening, and troponin value. Patients with any of the following diseases were excluded: 1) STEMI, 2) asthma, 3) chronic obstructive pulmonary disease, 4) liver and/or kidney dysfunction, and 5) immune and hematological diseases.
Peripheral venous blood was immediately obtained from all patients during hospital admission. Plasma was separated by centrifuging blood samples at 4000 rpm for 15 minutes and stored at −80℃ refrigerator. sST2 was examined by performing Presage ® ST2 assay (Critical Diagnostics, San Diego, USA). ST2 assay accurately detected plasma ST2 levels in the range of 3.1–200 ng/ml. Routine laboratory parameters were all tested in the Department of Clinical Laboratory by standard assay techniques. Baseline characteristics and angiographic features were collected from the information system of Shengjing Hospital.
All patients received 300 mg aspirin and 300 mg clopidogrel or 180 mg ticagrelor after admission. According to patients’ blood pressure, heart rates, and liver and kidney functions, beta-blockers, angiotensin-converting enzyme inhibitors (ACEI) or angiotensin receptor blockers (ARB), and statin were added. Coronary angiography and stent implantation were performed with standard techniques. Thrombolysis in myocardial infarction (TIMI) flow grades were decided by cardiologists who were blinded to the study.
The no-reflow phenomenon was defined as TIMI flow grade of ≤ 2 despite successfully reopening the culprit vessel and lack of mechanical obstructions. TIMI blood flow is defined as follows: grade 0, no antegrade blood flow; grade 1, with antegrade blood flow but cannot fill the distal blood vessels; grade 2, with antegrade blood flow that can fill the distal blood vessels but is significantly slow; and grade 3, with antegrade blood flow that can fill the distal coronary bed quickly and completely.
All patients continued to take drugs following doctors’ advice. The primary end point of our study was major cardiovascular and cerebrovascular event (MACCE) during one-year follow-up. MACCE was defined as cardiovascular death, worsening HF (new-onset HF and worsening chronic HF), stroke, and ischemia-driven revascularization. The second end point was all-cause death, worsening HF, stroke, and ischemia-driven revascularization.
Statistical Analysis
The statistical software SPSS 22.0 (SPSS Inc, Chicago, IL, USA) was applied for statistics analysis. If they met the normal distribution, continuous variables were recorded as mean±standard deviation (SD), and if they did not meet the normal distribution, they were expressed as median and quartile range. To compare between the two groups, unpaired Studentʼs t -test for normal distribution or Mann–Whitney U test for non-normal distribution was adopted. Categorical data was presented as number and percentage and was analyzed using the chi-square test. The receiving operating characteristic (ROC) curve was utilized to analyze the predictive value of sST2 for MACCE and no-reflow phenomenon. Youdenʼ index determined the best cutoffs of sST2. Patients were divided into two groups based on the cutoff value from ROC: sST2 ≤ 34.2 ng/ml and sST2 >34.2 ng/ml groups. The correlation among sST2 and left ventricular ejection fraction (LVEF), NT-pro BNP, cTnI, and Gensini score was calculated by the Spearman rank correlation coefficient. We adopted multivariate logistic regression analysis to identify independent predictors of the no-reflow phenomenon. The Kaplan–Meier survival curves and cox proportional hazard models were used for survival analysis to identify independent predictors of the MACCE. The two-sided P value of <0.05 was considered statistically significant.
Results
Overall, we recruited 205 patients who were diagnosed with NSTE-ACS (143 males and 62 females); their age ranged from 33 to 83. No-reflow phenomenon occurred in a total of 21 (10.2%) patients after PCI. At first, we conducted a ROC curve to evaluate the predictive power of sST2 to predict MACCE. In Fig.1A , the area under the curve (AUC) was 0.84 (95% confidence interval [CI], 0.75–0.93; P <0.001). The best cutoff value was 34.2 ng/ml, the sensitivity was 72%, and the specificity was 84%.
Fig.1.
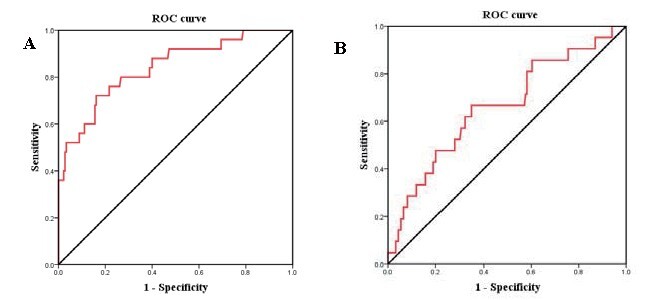
A: ROC curve analyses for the correlation of sST2 with MACCE. B: ROC curve analyses for the correlation of sST2 with no-reflow phenomenon.
Table 1 summarizes the baseline characteristics of patients in the sST2 ≤ 34.2 and sST2 >34.2 ng/ml groups. Patients in the sST2 >34.2 ng/ml group had higher total cholesterol, white blood cell (WBC), peak cTnI, and NT-proBNP than those in the sST2 ≤ 34.2 ng/ml group. The estimated glomerular filtration rate (eGFR) was lower in the sST2 >34.2 ng/ml group. Compared with the sST2 ≤ 34.2 ng/ml group, the sST2 >34.2 ng/ml group had significantly higher sST2 (42.8 [36.02–52.7] vs. 19.7 [13.68–26.4]; P <0.001). We also divided patients into single-vessel, two-vessel, and three-vessel lesion group, and it was found that sST2 was statistically different among the three groups (F: 5.163; P =0.007). Comparison between the two groups revealed that there was statistical difference between the single- and three-vessel lesion groups ( P =0.002). There was no statistical difference in age, gender, medical history, blood pressure, heart rate, creatinine, left ventricular ejection fraction, and platelet counts between the two groups. The angiographic features between the two groups were also compared, as shown in Table 1 . The multivessel disease and higher Gensini scores were more frequent in the sST2 >34.2 ng/ml group. Patients in the sST2 >34.2 ng/ml group have more grades 0–1 initial TIMI flow, and more stents were implanted. We also analyzed the correlation between sST2 and clinical parameters. sST2 is positively correlated with NT-proBNP, peak cTnI, WBC, and Gensini score and negatively correlated with LVEF and initial TIMI flow grade ( Table 2 ) .
Table 1. Baseline characteristics and angiographic features.
| Variables | sST2 ≤ 34.2 ng/ml ( n = 157) | sST2 > 34.2 ng/ml ( n = 48) | P value |
|---|---|---|---|
| Age, years, mean±SD | 61.13±9.67 | 63.71±10.02 | 0.111 |
| Male, n (%) | 106 (67.5) | 37 (77.1) | 0.207 |
| Hypertension, n (%) | 65 (41.4) | 22 (45.8) | 0.587 |
| Diabetes mellitus, n (%) | 104 (66.2) | 35 (72.9) | 0.386 |
| Smoking, n (%) | 74 (47.1) | 18 (37.5) | 0.24 |
| History of stroke/TIA, n (%) | 14 (8.9%) | 5 (10.4) | 0.778 |
| SBP, mmHg, mean±SD | 132.18±18.49 | 137.08±18.49 | 0.129 |
| DBP, mmHg, mean±SD | 80.37±11.23 | 80.35±11.65 | 0.993 |
| HR, beats/min, mean±SD | 68.81±10.49 | 71.98±12.19 | 0.08 |
| Creatinine, µmol/L, mean±SD | 70.14±17.16 | 77.06±17.16 | 0.287 |
| eGFR, ml/min/1.73 m 2 , mean±SD | 96.31±21.39 | 86.77±18.26 | 0.006 |
| TC, mmol/L, mean±SD | 3.89±1.02 | 4.31±1.05 | 0.014 |
| LDL-C, mmol/L, mean±SD | 2.36±0.89 | 2.63±0.95 | 0.07 |
| HDL-C, mmol/L, mean±SD | 0.94±0.23 | 1.03±0.35 | 0.092 |
| WBC counts, ×10 9 /L, mean±SD | 6.31±1.72 | 7.33±2.29 | 0.006 |
| Neutrophil counts, ×10 9 /L, mean±SD | 3.9±2.96 | 3.3±2.23 | 0.211 |
| Platelet counts, ×10 9 /L, mean±SD | 195.75±45.73 | 195.25±42.65 | 0.946 |
| Peak cTnI, µg/L, mean±SD | 0.093 (0.035-0.57) | 0.7 (0.115-4.08) | <0.001 |
| NT-pro BNP, pg/mL, mean±SD | 104.2 (46.4-388.7) | 400.4 (132.8-848.2) | <0.001 |
| LVEF, %, mean±SD | 62.18±5.53 | 60.5±4.93 | 0.066 |
| Medications, n (%) | |||
| Ticagrelor | 84 (53.5) | 28 (58.3) | 0.556 |
| Clopidogrel | 73 (46.5) | 20 (41.7) | 0.556 |
| Beta blocker | 148 (94.3) | 46 (95.8) | 0.665 |
| ACEI/ARB | 144 (91.7) | 45 (93.8) | 0.639 |
| Nicorandil | 69 (43.9) | 21 (43.8) | 0.981 |
| Statin | 157 (100) | 48 (100) | 1 |
| sST2, ng/mL, mean±SD | 19.7 (13.68-26.4) | 42.8 (36.02-52.7) | <0.001 |
| Multivessel disease, n (%) | 102 (65) | 42 (87.5) | 0.002 |
| Initial TIMI flow grade, n (%) | <0.001 | ||
| 0-1 | 13 (8.3) | 11 (22.9) | |
| ≥ 2 | 144 (91.7) | 37 (77.1) | |
| Multistent, n (%) | 82 (52.2%) | 35 (72.9) | 0.01 |
| Gensini Score | 34.7±25.09 | 53.67±24.44 | <0.001 |
| No-reflow phenomenon, n (%) | 11 (7) | 10 (20.8) | 0.012 |
Abbreviations: eGFR, estimated glomerular filtration rate; TIA, transient ischemic attack; SBP, systolic blood pressure; DBP, diastolic blood pressure; HR, heart rate; TC, total cholesterol; HDL-C, high-density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; cTnI, cardiac troponin I; NT-pro BNP, N-terminal pro-B-type natriuretic peptide; LVEF, left ventricular ejection fraction; ACEI: angiotensin-converting enzyme inhibitors; ARB: angiotensin receptor blockers; sST2: soluble suppression of tumorigenicity 2; WBC:white blood cell; SD, standard deviation
Table 2. Correlation between sST2 and clinical parameters.
| Variables | Spearman r | P |
|---|---|---|
| LVEF | -0.156 | 0.027 |
| NT-pro BNP | 0.339 | <0.001 |
| Peak cTnI | 0.297 | <0.001 |
| White blood cell | 0.277 | <0.001 |
| Initial TIMI flow grade | -0.27 | <0.001 |
| Gensini score | 0.352 | <0.001 |
Abbreviations: cTnI, cardiac troponin I; NT-pro BNP, N-terminal pro-B-type natriuretic peptide; LVEF, left ventricular ejection fraction; sST2: soluble suppression of tumorigenicity 2; TIMI, thrombolysis in myocardial infarction.
ROC curve analysis was applied to determine the cutoff value of sST2 and predict no-reflow phenomenon ( Fig.1B ) . AUC was 0.662 (95% CI, 0.534–0.79; P =0.015). The optimal sST2 cutoff value was 27.8 ng/ml, with 66.7% sensitivity and 65.2% specificity. sST2 >34.2 ng/ml, peak cTnI, initial TIMI flow grade, multivessel disease, and neutrophil counts had predictive values for the no-reflow in univariate logistic regression, and sST2 >34.2 ng/ml also had independent predictive value from multivariate logistic regression after adjusting peak cTnI and neutrophil counts ( Table 3 ) .
Table 3. Univariate and Multivariate Logistic Regression Analysis for Independent Predictors of no-reflow phenomenon.
| Variables | Univariate OR (95% CI) | P value | Multivariate OR (95% CI) | P value |
|---|---|---|---|---|
| sST2 > 34.2 ng/ml | 3.49 (1.38-8.83) | 0.008 | 3.802 (1.03-14.11) | 0.046 |
| eGFR | 0.994 (0.97-1.02) | 0.595 | - | - |
| Age | 1.016 (0.969-1.07) | 0.517 | - | - |
| White blood cell | 1.116 (0.901-1.38) | 0.314 | - | - |
| Peak cTnI | 1.06 (1.01-1.113) | 0.018 | 1.06 (0.99-1.12) | 0.099 |
| NT-pro BNP | 1.0 (0.999-1.001) | 0.789 | - | - |
| Gensini score | 1.012 (0.996-1.03) | 0.143 | - | - |
| Initial TIMI flow grade | 0.368 (0.251-0.54) | <0.001 | 0.434 (0.28-0.68) | <0.001 |
| Diabetes mellitus | 2.16 (0.697-6.695) | 0.182 | - | - |
| Smoking | 1.26 (0.44-3.62) | 0.667 | - | - |
| Multivessel disease | 0.27 (0.11-0.69) | 0.006 | 0.13 (0.04-0.48) | 0.002 |
| LDL-C | 0.71 (0.41-1.22) | 0.214 | - | - |
| Neutrophil counts | 0.71 (0.51-0.997) | 0.048 | 0.93 (0.69-1.25) | 0.636 |
Abbreviations: cTnI, cardiac troponin I; NT-pro BNP, N-terminal pro-B-type natriuretic peptide; sST2: soluble suppression of tumorigenicity 2; TIMI, thrombolysis in myocardial infarction; eGFR, estimated glomerular filtration rate; LDL-C, low density lipoprotein cholesterol OR: odds ratio.
All patients completed one-year follow-up. One patient died of acute cerebral infarction, 14 had worsening HF, and 10 had ischemia-driven revascularization ( Table 4 ) . According to Kaplan–Meier curves, the high sST2 concentration (sST2 >34.2 ng/ml) was strongly related with one-year prognosis, thus worsening HF and ischemia-driven revascularization ( Fig.2 ) . In the univariate cox regression analysis, sST2 >34.2 ng/ml, NT-proBNP, no-reflow phenomenon, and diabetes mellitus all are statistically significant. From multivariate cox regression analysis, sST2 >34.2 ng/ml and diabetes mellitus were independent predictors of MACCE within 1 year after adjusting for LVEF, no-reflow phenomenon, and NT-proBNP ( Table 5 ) .
Table 4. Long-term follow-up outcomes.
| Variables | sST2 ≤ 34.2 ng/ml ( n = 157) | sST2 > 34.2 ng/ml ( n = 48) | P value |
|---|---|---|---|
| All-cause mortality | 0 (0%) | 1 (2.1%) | 0.234 |
| Cardiovascular mortality | 0 | 0 | 1 |
| ischemia-driven revascularization | 3 (1.9%) | 7 (14.6%) | 0.002 |
| worsening heart failure | 4 (2.5%) | 10 (20.8%) | <0.001 |
| MACE | 7 (4.5%) | 17 (35.4%) | <0.001 |
Fig.2.
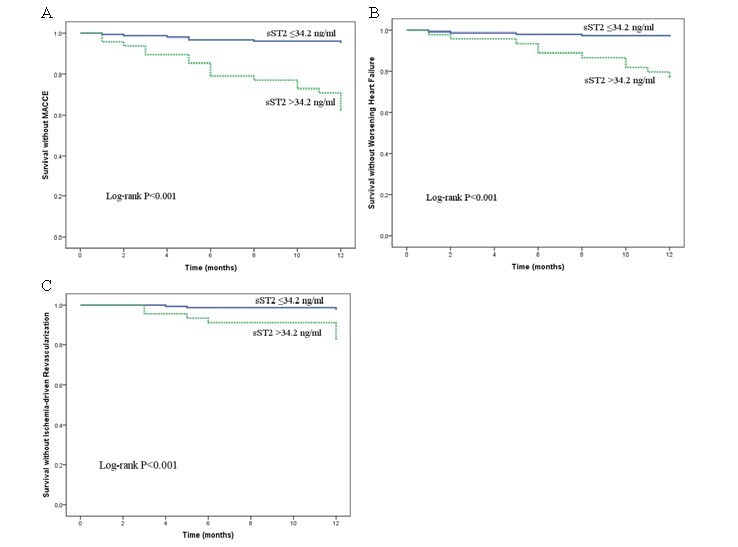
Kaplan–Meier survival curves for the relation between sST2 and MACCE (A), between sST2 and worsening HF (B), and between sST2 and ischemia-driven revascularization (C)
Table 5. Predictors of MACCE by Univariate and Multivariate Cox Regression Analysis.
| Variables | Univariate HR (95% CI) | P value | Multivariate HR (95% CI) | P value |
|---|---|---|---|---|
| sST2 > 34.2 ng/ml | 9.76 (4.1-23.4) | <0.001 | 10.22 (4.05-25.7) | <0.001 |
| eGFR | 0.997 (0.98-1.02) | 0.747 | - | - |
| Age | 1.04 (0.992-1.08) | 0.113 | - | - |
| White blood cell | 1.1 (0.92-1.32) | 0.287 | - | - |
| Peak cTnI | 1.01 (0.95-1.06) | 0.824 | - | - |
| NT-pro BNP | 1.00 (1.00-1.001) | 0.004 | 1.00 (1.00-1.001) | 0.826 |
| Gensini | 1.004 (0.99-1.02) | 0.561 | - | - |
| No-reflow phenomenon | 0.36 (0.15-0.91) | 0.031 | 0.48 (0.17-1.33) | 0.158 |
| Diabetes mellitus | 0.35 (0.16-0.76) | 0.008 | 0.25 (0.11-0.595) | 0.002 |
| Hypertension | 0.64 (0.28-1.48) | 0.294 | - | |
| Multivessel disease | 1.78 (0.67-4.73) | 0.251 | - | |
| LVEF | 0.92 (0.87-0.98) | 0.011 | 0.96 (0.89-1.04) | 0.316 |
| Neutrophil counts | 0.87 (0.67-1.12) | 0.279 |
Abbreviations: cTnI, cardiac troponin I; NT-pro BNP, N-terminal pro-B-type natriuretic peptide; sST2: soluble suppression of tumorigenicity 2; eGFR, estimated glomerular filtration rate; LVEF, left ventricular ejection fraction; HR: hazard ratio.
Discussion
In this study, our main findings were as follow: (1) Among the 205 patients with NSTE-ACS enrolled in this study, 21 (10.2%) underwent no-reflow phenomenon and 25 (12.2%) developed MACCE. (2) sST2 was weakly related to the degree of coronary artery stenosis, and patients with high level of sST2 had higher Gensini score. (3) sST2 had a great predictive value for the occurrence of adverse outcomes but had a low predictive ability for no-reflow. (4) After the adjustment of confounding variables, sST2 remained independent predictors of MACCE and no-reflow phenomenon.
ST2 was first discovered in 1989 and was considered as an orphan receptor. The specific ligand IL-33 was discovered in 2005 12) . Its gene sequence and molecular structure are similar to IL-18 and IL-1β. The effect of IL-33/ST2L pathway on atherosclerosis is mainly achieved by maintaining a balanced Th1/Th2 cell ratio 13) . The combination of ST2L and IL-33 can promote Th2-type immune response and induce the production of Th2-type cytokines such as IL-5, as well as IL-6, thereby slowing the development of atherosclerosis 14) . At the same time, it can also reduce Th1 cytokine production. For example, IFN-γcan inhibit the inflammatory response involved in Th1 15) . IL-33/ST2L can also exert anti-myocardial hypertrophy and fibrosis by blocking the effects of angiotensin II or epinephrine on the myocardium. Weir et al. proposed that sST2 levels at admission in patients with myocardial infarction can predict the recovery of long-term left ventricular function, and it has a correlation with infarcted myocardial volume and cardiac remodeling. Patients with higher level of ST2 have a severe infarct transmurality and endocardial extent in the presence of microvascular obstruction 16) . The results of another study revealed that the sST2 level in STEMI group was higher than that in NSTEMI, SA, and control groups. The sST2 level in the NSTEMI group was also significantly higher than that in the SA control group. It was observed that the sST2 level in patients with STEMI continued to increase from SA to NSTEMI, to STEMI, indicating that it was related to the development stage and severity of coronary artery disease 17) . Therefore, ST2 levels at admission can reflect the degree of inflammatory response, myocardial injury, and myocardial fibrosis in patients developing myocardial infarction.
In our study, it was obvious that the Gensini score of the sST2 >34.2 ng/ml group was significantly higher than that of the sST2 ≤ 34.2 ng/ml group and that sST2 had a weak positive correlation with Gensini score. When we divided patients into 1, 2, and 3 coronary lesions, there was statistically significant difference in sST2 concentration among the three groups. Wang et al. concluded that sST2 had a positive correlation with Gensini score (r=0.29) 18) , and this may due to the high sST2 level of patients with NSTE-ACS, which inhibits the anti-atherosclerotic effect of IL-33, thus leading to the formation of coronary atherosclerosis. However, atherosclerosis is a long-term process, and the level of sST2 before the onset of the disease cannot be known, which requires further monitoring of sST2 in patients with high risk of coronary heart disease to be determined.
No-reflow phenomenon was defined after PCI, and the myocardium is still unable to receive sufficient reperfusion, excluding the reduction of coronary blood flow that is caused by mechanical obstruction. Studies proved that no-reflow, as one of the serious complications after PCI, can aggravate myocardial ischemia, enlarge the area of myocardial infarction, and increase the incidence of HF 19) . It was also a predictor of short- and long-term mortality 20 , 21) . Although the exact mechanism of no-reflow was not completely clear at present, researches illustrated that the occurrence of no-reflow may be related to microcirculatory dysfunction, myocardial ischemia-reperfusion injury, distal vascular embolism, and inflammatory cell infiltration 22) . This was the first study that assessed the relationship between no-reflow phenomenon and sST2 in patients with NSTE-ACS. Somuncu et al. put forward that sST2 is the independent predictor of no-reflow in patients with STEMI. The cutoff value of sST2 is 11.6 ng/ml with an 82.7% sensitivity and 64.0% specificity and had an AUC of 0.699 in the ROC curve 11) . In our study, we revealed that the AUC of 0.662 is similar to 0.699, but the optimal cutoff value was 27.8 ng/ml, which was significantly different from 11.6 ng/ml. This may be related to the different diseases of patients in the two studies. sST2 has predictive value for no-reflow in univariate logistic regression analysis ( P =0.009). After adjusting for peak cTnI and neutrophil counts, multivariate regression analysis displayed that sST2 >34.2 ng/ml is also an independent predictor of no-reflow phenomenon. Such experimental results are also consistent with the mechanism of no-reflow. The increase of ST2 indicates the degree of inflammatory response. Severe inflammation likely means that no-reflow will occur.
Recently, ST2 was included in ACC/AHA guidelines as a prognostic marker for patients with HF, which also made this cardiac biomarker being widely used in the clinical setting 23) . A meta-analysis of seven studies displayed that sST2 could be applied as a predictor of death in patients with chronic heart failure (CHF), and 35 ng/ml was discovered to be a cutoff value for high incidence of cardiovascular events 24) . At the same time, the cut point of 35 ng/ml was also written into the guide 23) . A multicenter, prospective study on CHF showed that sST2 expression levels are related to HF severity. Patients with elevated sST2 expression levels have significantly increased risk of death and the need for heart transplantation 25) . In addition, Min-Seok et al. also confirmed that BNP was significantly higher in severe renal insufficiency, and sST2 levels did not significantly change in the degree of renal insufficiency compared with BNP levels, so ST2 level was not correlated with the severity of renal function 26) , suggesting that ST2 has better stability tA large number of studies currently explored the relationship between sST2 and patients with STEMI. Previous researches illustrated that sST2 levels in patients with acute myocardial infartion (AMI) have predictive value for prognosis, especially HF and death, and have complementary effects with NT-proBNP and grace scores 27 , 28) . Sabatine et al. demonstrated that, in patients with STEMI, the high level of sST2 was a significant predictor of cardiovascular death and HF independent of NT-proBNP. The combination of ST2 and NT-proBNP can improve risk stratification 10) .
However, there are relatively few studies on the relationship between sST2 and patients with NSTE-ACS. In MERLIN-TIMI36 study, by observing 4,426 patients with NSTE-ACS, patients with sST2 >35 µg/L may be older and male and have diabetes and renal dysfunction. High sST2 levels were significantly associated with 30-day and 1-year risk of cardiovascular death/HF but were only weakly related to BNP and troponin 29) . Eggers et al. observed the dynamic changes of sST2 levels in 403 patients with NSTE-ACS and concluded that sST2 peaked at 6–17 hours after the onset and then gradually decreased. However, after adjusting for NT-proBNP, sST2 was not the independent predictor of mortality in patients with NSTE-ACS within 1 year 30) . In the results of our study, high-sST2 remains an independent predictor of MACCE in patients with NSTE-ACS and had great predictive value (AUC, 0.84). The difference between the two studies may be caused by different measurement times of sST2 and different levels of sST2 used for grouping. In our study, high ST2 was associated with not only increased risk for HF at 1 year but also increased ischemia-driven revascularization risk at 1 year. The cutoff value is 34.2 ng/ml, which is similar to the recommended value of 35 ng/ml in patients with HF; the sensitivity was 72% and specificity 84%. Therefore, whether we can apply 35 ng/ml as the optimal risk stratification for patients with HF and ACS is a direction for further investigation.
Nevertheless, this study has several limitations. This was a single-center study, and the number of patients included was relatively small. In this study, we examined the plasma sST2 levels only for patients with NSTE-ACS. It would be better to collect the data in patients with STEMI, stable coronary artery, and control and further compare the levels among those groups. Most importantly, we only detected the ST2 level of patients at the time of admission but did not observe subsequent changes.
Conclusion
This is the first study to explore the relationship between sST2 and the degree of coronary stenosis and the predictive ability of no-reflow phenomenon after PCI in patients with NSTE-ACS. Moreover, we concluded that high sST2 at admission is associated with the 1-year prognosis and no-reflow phenomenon. Finally, we believe that 35 ng/ml can be adopted as a recommended value for risk stratification to HF and ACS.
Conflicts of Interest
The authors declared that there was no conflict of interest.
Funding
No funding was received for this research.
References
- 1).Kokkoz Ç, Bilge A, Irik M, Dayangaç HI, Hayran M, Akarca FK, Erdem NB, Çavuş M. Prognostic value of plasma ST2 in patients with non-ST segment elevation acute coronary syndrome. Turk J Emerg Med, 2018; 18: 62-66 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2).Bassand JP, Hamm CW, Ardissino D, Boersma E, Budaj A, Fernández-Avilés F, Fox KA, Hasdai D, Ohman EM, Wallentin L, Wijns W. Guidelines for the diagnosis and treatment of non-ST-segment elevation acute coronary syndromes. Eur Heart J, 2007; 28: 1598-1660 [DOI] [PubMed] [Google Scholar]
- 3).Weinberg EO, Shimpo M, De Keulenaer GW, MacGillivray C, Tominaga S, Solomon SD, Rouleau JL, Lee RT. Expression and regulation of ST2, an interleukin-1 receptor family member, in cardiomyocytes and myocardial infarction. Circulation, 2002; 106: 2961-2966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4).Kakkar R, Lee RT. The IL-33/ST2 pathway: therapeutic target and novel biomarker. Nat Rev Drug Discov, 2008; 7: 827-840 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5).Tymińska A, Kapłon-Cieślicka A, Ozierański K, Budnik M, Wancerz A, Sypień P, Peller M, Maksym J, Balsam P, Opolski G, Filipiak KJ. Association of galectin-3 and soluble ST2 with in-hospital and 1-year outcomes in patients with ST-segment elevation myocardial infarction treated with primary percutaneous coronary intervention. Pol Arch Intern Med, 2019; 129: 770-780 [DOI] [PubMed] [Google Scholar]
- 6).Sanada S, Hakuno D, Higgins LJ, Schreiter ER, McKenzie AN, Lee RT. IL-33 and ST2 comprise a critical biomechanically induced and cardioprotective signaling system. J Clin Invest, 2007; 117: 1538-1549 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7).Manzano-Fernández S, Mueller T, Pascual-Figal D, Truong QA, Januzzi JL. Usefulness of soluble concentrations of interleukin family member ST2 as predictor of mortality in patients with acutely decompensated heart failure relative to left ventricular ejection fraction. Am J Cardiol, 2011; 107: 259-267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8).Weinberg EO, Shimpo M, Hurwitz S, Tominaga S, Rouleau JL, Lee RT. Identification of serum soluble ST2 receptor as a novel heart failure biomarker. Circulation, 2003; 107: 721-726 [DOI] [PubMed] [Google Scholar]
- 9).Rehman SU, Mueller T, Januzzi JL. Characteristics of the novel interleukin family biomarker ST2 in patients with acute heart failure. J Am Coll Cardiol, 2008; 52: 1458-1465 [DOI] [PubMed] [Google Scholar]
- 10).Sabatine MS, Morrow DA, Higgins LJ, MacGillivray C, Guo W, Bode C, Rifai N, Cannon CP, Gerszten RE, Lee RT. Complementary roles for biomarkers of biomechanical strain ST2 and N-terminal prohormone B-type natriuretic peptide in patients with ST-elevation myocardial infarction. Circulation, 2008; 117: 1936-1944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11).Somuncu MU, Akgun T, Cakır MO, Akgul F, Serbest NG, Karakurt H, Can M, Demir AR. The Elevated Soluble ST2 Predicts No-Reflow Phenomenon in ST-Elevation Myocardial Infarction Undergoing Primary Percutaneous Coronary Intervention. J Atheroscler Thromb, 2019; 26: 970-978 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12).Dattagupta A, Immaneni S. ST2: Current status. Indian Heart J, 2018 : S96-S101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13).Miller AM, Xu D, Asquith DL, Denby L, Li Y, Sattar N, Baker AH, McInnes IB, Liew FY. IL-33 reduces the development of atherosclerosis. J Exp Med, 2008; 205: 339-346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14).Miller AM, Liew FY. The IL-33/ST2 pathway--A new therapeutic target in cardiovascular disease. Pharmacol Ther, 2011; 131: 179-186 [DOI] [PubMed] [Google Scholar]
- 15).Aimo A, Migliorini P, Vergaro G, Franzini M, Passino C, Maisel A, Emdin M. The IL-33/ST2 pathway, inflammation and atherosclerosis: Trigger and target. Int J Cardiol, 2018; 267: 188-192 [DOI] [PubMed] [Google Scholar]
- 16).Weir RA, Miller AM, Murphy GE, Clements S, Steedman T, Connell JM, McInnes IB, Dargie HJ, McMurray JJ. Serum soluble ST2: a potential novel mediator in left ventricular and infarct remodeling after acute myocardial infarction. J Am Coll Cardiol, 2010; 55: 243-250 [DOI] [PubMed] [Google Scholar]
- 17).Demyanets S, Speidl WS, Tentzeris I, Jarai R, Katsaros KM, Farhan S, Krychtiuk KA, Wonnerth A, Weiss TW, Huber K, Wojta J. Soluble ST2 and interleukin-33 levels in coronary artery disease relation to disease activity and adverse outcome. PloS one, 2014; 9: e95055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18).Wang YP, Wang JH, Wang XL, Liu JY, Jiang FY, Huang XL, Hang JY, Qin W, Ma SX, Zhang J, Yuan MJ, Li JB, Lu ZG, Wei M. Roles of ST2, IL-33 and BNP in predicting major adverse cardiovascular events in acute myocardial infarction after percutaneous coronary intervention. J Cell Mol Med, 2017; 21: 2677-2684 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19).Choo EH, Kim PJ, Chang K, Ahn Y, Jeon DS, Lee JM, Kim DB, Her SH, Park CS, Kim HY, Yoo KD, Jeong MH, Seung KB. The impact of no-reflow phenomena after primary percutaneous coronary intervention: a time-dependent analysis of mortality. Coron Artery Dis, 2014; 25: 392-398 [DOI] [PubMed] [Google Scholar]
- 20).Erkol A, Oduncu V, Pala S, Kızılırmak F, Kılıcgedik A, Yılmaz F, Güler A, Karabay CY, Kırma C. Plasma osteoprotegerin level on admission is associated with no-reflow phenomenon after primary angioplasty and subsequent left ventricular remodeling in patients with acute ST-segment elevation myocardial infarction. Atherosclerosis, 2012; 221: 254-259 [DOI] [PubMed] [Google Scholar]
- 21).Gür M, Türkoğlu C, Taşkın A, Uçar H, Börekçi A, Seker T, Gözükara MY, Kaypaklı O, Akyol S, Selek S, Sahin DY, Elbasan Z, Caylı M. Paraoxonase-1 activity and oxidative stress in patients with anterior ST elevation myocardial infarction undergoing primary percutaneous coronary intervention with and without no-reflow. Atherosclerosis, 2014; 234: 415-420 [DOI] [PubMed] [Google Scholar]
- 22).Durante A, Camici PG. Novel insights into an "old" phenomenon: the no reflow. Int J Cardiol, 2015; 187: 273-280 [DOI] [PubMed] [Google Scholar]
- 23).Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, Drazner MH, Fonarow GC, Geraci SA, Horwich T, Januzzi JL, Johnson MR, Kasper EK, Levy WC, Masoudi FA, McBride PE, McMurray JJ, Mitchell JE, Peterson PN, Riegel B, Sam F, Stevenson LW, Tang WH, Tsai EJ, Wilkoff BL. 2013 ACCF/AHA guideline for the management of heart failure: executive summary: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation, 2013; 128: 1810-1852 [DOI] [PubMed] [Google Scholar]
- 24).Aimo A, Vergaro G, Passino C, Ripoli A, Ky B, Miller WL, Bayes-Genis A, Anand I, Januzzi JL, Emdin M. Prognostic Value of Soluble Suppression of Tumorigenicity-2 in Chronic Heart Failure: A Meta-Analysis. JACC Heart Fail, 2017; 5: 280-286 [DOI] [PubMed] [Google Scholar]
- 25).Lassus J, Gayat E, Mueller C, Peacock WF, Spinar J, Harjola VP, van Kimmenade R, Pathak A, Mueller T, Disomma S, Metra M, Pascual-Figal D, Laribi S, Logeart D, Nouira S, Sato N, Potocki M, Parenica J, Collet C, Cohen-Solal A, Januzzi JL, Mebazaa A. Incremental value of biomarkers to clinical variables for mortality prediction in acutely decompensated heart failure: the Multinational Observational Cohort on Acute Heart Failure (MOCA) study. Int J Cardiol, 2013; 168: 2186-2194 [DOI] [PubMed] [Google Scholar]
- 26).Kim MS, Jeong TD, Han SB, Min WK, Kim JJ. Role of Soluble ST2 as a Prognostic Marker in Patients with Acute Heart Failure and Renal Insufficiency. J Korean Med Sci, 2015; 30: 569-575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27).Zhang K, Zhang XC, Mi YH, Liu J. Predicting value of serum soluble ST2 and interleukin-33 for risk stratification and prognosis in patients with acute myocardial infarction. Chin Med J (Engl), 2013; 126: 3628-3631 [PubMed] [Google Scholar]
- 28).Wang TJ, Wollert KC, Larson MG, Coglianese E, McCabe EL, Cheng S, Ho JE, Fradley MG, Ghorbani A, Xanthakis V, Kempf T, Benjamin EJ, Levy D, Vasan RS, Januzzi JL. Prognostic utility of novel biomarkers of cardiovascular stress: the Framingham Heart Study. Circulation, 2012; 126: 1596-1604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29).Kohli P, Bonaca MP, Kakkar R, Kudinova AY, Scirica BM, Sabatine MS, Murphy SA, Braunwald E, Lee RT, Morrow DA. Role of ST2 in non-ST-elevation acute coronary syndrome in the MERLIN-TIMI 36 trial. Clin Chem, 2012; 58: 257-266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30).Eggers KM, Armstrong PW, Califf RM, Simoons ML, Venge P, Wallentin L, James SK. ST2 and mortality in non-ST-segment elevation acute coronary syndrome. Am Heart J, 2010; 159: 788-794 [DOI] [PubMed] [Google Scholar]


