Abstract
Recent studies have suggested that members of the Abl interactor (Abi) protein family negatively regulate cell growth and transformation. To date, however, no specific role in these cellular processes has been identified for the Abi family. Here we describe the inhibition by overexpressed Abi-1 of a mitogenic pathway activated by both growth factors and v-Abl. We have identified the guanine nucleotide exchange factors Sos1 and Sos2 as novel binding partners of Abi-1. A domain that is required for interaction with Sos in vivo has been mapped to the amino terminus of Abi-1. Overexpression of Abi-1 inhibits epidermal growth factor (EGF)-induced activation of extracellular signal-regulated kinases (Erks) but does not affect EGF-induced activation of c-Jun N-terminal kinase or Akt. In addition, overexpression of Abi-1 blocks Erk activation induced by v-Abl. In both cases, the maximal inhibitory effect requires an intact amino-terminal Sos-binding domain in Abi-1. Finally, we demonstrate that tyrosine phosphorylation of endogenous Abi-1 in fibroblasts is induced by both v-Abl and serum stimulation, further suggesting a role for Abi-1 in signal transduction initiated by v-Abl and growth factors. Taken together, these findings suggest that overexpressed Abi proteins negatively regulate cell growth and transformation by specifically targeting the Erk pathway.
Abl interactor (Abi) proteins were originally identified as binding partners of the Abl nonreceptor tyrosine kinase (5, 28). The two members of the Abi family, Abi-1 and Abi-2, are highly homologous. Both contain an amino-terminal homeobox-like domain, proline-rich regions, PEST sequences, and a carboxy-terminal SH3 domain. Mapping of interaction sites on both Abl and Abi indicates that reciprocal binding occurs: the SH3 domain of each protein binds to a proline-rich motif on the partner molecule (5). Overexpression of Abi-1 lacking amino acids 1 to 85 (AbiΔ1–85), originally reported as the full-length protein, was shown to inhibit transformation of fibroblasts by full-length p160v-abl but not by v-Src (28). This deletion mutant of Abi-1 also failed to inhibit the transforming activity of truncated p90v-abl, correlating with the inability of Abi-1 to physically associate with p90v-abl. Recently, we have found in similar assays that full-length Abi-1 can inhibit the transforming activity of p160v-abl (A. Ikeguchi and S. P. Goff, unpublished data). Overexpression of a truncated form of Abi-2 that retained only one Abl-binding site was found to potentiate c-Abl-transforming activity (5). More recently identification of Eps8 and spectrin as additional interaction partners of Abi-1 provided potential links to growth factor receptor-mediated signaling and the cytoskeleton (2, 36). In addition, oncogenic forms of Abl and Src—namely, BCR-ABL and v-Src—have been found to target Abi proteins in a Ras-independent manner for ubiquitin-dependent proteolysis (6). Expression of Abi proteins is also lost in the bone marrow cells of patients with aggressive BCR-ABL-positive leukemias, suggesting that the loss of Abi expression contributes to progression of disease. Taken together, these findings suggest that Abi proteins may act as negative regulators of cell growth and transformation.
The mechanism by which Abi-1 overexpression suppresses v-Abl-transforming activity has not been elucidated. p160v-abl, encoded by the Abelson murine leukemia virus, is a chimeric protein consisting of the amino-terminal region of Moloney murine leukemia virus Gag and the SH2, SH1, and carboxy-terminal domains of c-Abl. Similar to BCR-ABL and TEL-ABL, v-Abl displays constitutive tyrosine kinase activity that is required for transformation (19). However, Abi proteins do not appear to directly inhibit Abl kinase activity (28, 33), and it has been proposed that the binding of Abi-1 to v-Abl might block activation of critical signal transduction pathways. Signaling molecules activated downstream of v-Abl include Ras (26), phosphatidylinositol 3-kinase (PI3K) (30), Rac (25), c-Jun N-terminal kinase (JNK) (21, 25), extracellular signal-regulated kinases (Erks) (21, 25), protein kinase C (18), Janus kinases (7, 8), and signal transducers and activators of transcription (7, 8). In particular, Ras activity has been demonstrated to be necessary for v-Abl-mediated transformation (26). In addition, activation of several of the aforementioned signaling proteins requires functional Ras.
It is still unclear which signaling proteins are responsible for coupling v-Abl kinase activity to Ras activation. Ras activation normally occurs upon recruitment of the Ras-specific guanine nucleotide exchange factor (GEF) Son of sevenless (Sos) to the plasma membrane in close proximity to Ras. For example, autophosphorylation of activated receptor tyrosine kinases can create membrane-localized docking sites for Grb2-Sos or Shc-Grb2-Sos complexes. Subsequent activation of Ras can trigger multiple signaling events, including the well-characterized Raf-Mek-Erk kinase cascade. Targeting Sos to the plasma membrane by addition of myristoylation or farnesylation signals to Sos is sufficient to activate Ras-dependent signaling (1). Since the Gag moiety of v-Abl can direct localization to the plasma membrane, recruitment of Sos to v-Abl might be sufficient for Ras activation. Multiple Abl-binding proteins potentially link v-Abl to Sos. The SH2 domain of v-Abl interacts with Shc in a non-phosphotyrosine-dependent manner and can recruit Shc-Grb2-Sos complexes (20). In addition, the carboxy-terminal domain of v-Abl contains three proline-rich motifs that are docking sites for the Sos-binding Crk, Nck, and Grb2 adapter proteins (23). Dominant negative mutants of Grb2 and CrkI inhibit Erk activation induced by oncogenic Abl, suggesting that these adapter proteins participate in a positive manner in v-Abl-induced Ras signaling (29). In addition, v-Abl binds and phosphorylates p62dok, promoting the association of p62dok with RasGAP. It is not yet known whether this interaction inhibits the ability of RasGAP to negatively regulate Ras. However, overexpression of DOKL, a Dok-like protein, can inhibit v-Abl-induced Erk activation and transformation, suggesting a role for Dok proteins in v-Abl-induced Ras signaling (4). Therefore, it is possible to inhibit both v-Abl- and growth factor-mediated Ras-dependent signaling by interfering with the localization or function of regulatory, adapter, or effector components of the Ras pathway.
To investigate the potential role of Abi-1 in cell growth and transformation, we have sought additional Abi-1 interaction partners that might be modulators or effectors of Abi-1 functions. Here we describe the identification of the mammalian Sos proteins as novel binding partners for Abi-1. We demonstrate that the amino terminus of Abi-1 is necessary for this interaction in vivo. Overexpression of Abi-1 inhibits a specific epidermal growth factor (EGF)-induced signaling event downstream of Sos and Ras, namely, the activation of Erks. In addition, we show that overexpression of Abi-1 can block v-Abl-induced Erk activation. Thus, our work identifies a potential negative regulatory role for Abi proteins in a specific mitogenic pathway activated by both growth factors and v-Abl. Our findings also suggest possible mechanisms for the previously observed inhibition of v-Abl-mediated transformation by overexpression of Abi-1. A role for Abi-1 in v-Abl- and growth factor-mediated signaling in fibroblasts is further suggested by the induction of tyrosine phosphorylation of endogenous Abi-1 by both v-Abl and serum stimulation.
MATERIALS AND METHODS
Yeast two-hybrid screening.
A cDNA sequence encoding amino acids 394 to 475 of murine Abi-1 was amplified by PCR using pCGN-Abi-1 as the template. The oligonucleotides for PCR were 5′-GAAGATGGGATCCCTGCAGTAGTTCAGT-3′ and 5′-CTCTCCGTCGACCTAATCAGTATAGTGCAT-3′ (where BamHI and SalI sites are underlined). To create the bait plasmid pSH2-AbiSH3, the amplified fragment was digested with BamHI and SalI and cloned into the Saccharomyces cerevisiae expression vector pSH2-1 in frame with the LexA DNA-binding domain (LexADBD). To construct the plasmid AGP5 encoding the LexADBD-Abl hybrid, type 1 c-abl cDNA was partially digested with BglII. A BglII-BglII fragment encoding amino acids 4 to 1087 of type 1 c-abl was inserted into pSH2-1. This insert was released by BglII digestion and cloned into the yeast expression vector pGADNOT to generate the Gal4 activation domain (Gal4AD)-Abl construct. Yeast two-hybrid screening of the WEHI-3 cDNA library cloned into pGADNOT was performed as described previously (28). DNA sequencing and sequence analysis were also performed as described previously (28).
Mammalian expression constructs.
Myc-tagged constructs were created by cloning various cDNA fragments of murine abi-1, murine type IV c-abl, and human granulocyte-macrophage colony-stimulating factor (GM-CSF) receptor α chain into the EcoRI and SrfI sites of the mammalian expression vector MT21myc in frame with the myc epitope at the 3′ end of the cloning site. To generate hemagglutinin (HA)-tagged constructs, various fragments of murine abi-1 and human SOS1 cDNAs were cloned into the mammalian expression vector pCGN in frame with the HA epitope at the 5′ end of the cloning site. The HA-tagged DOKL construct has been described previously (4). To create the HA-Sos2(874–1297) construct, a DNA fragment encoding amino acids 874 to 1297 of murine Sos2 was released from the Gal4AD-Sos2 hybrid plasmid by digestion with SpeI and BglII and cloned into pCGN. SRα, SRα-HA-Erk2, and SRα-HA-JNK were gifts from Audrey Minden (Columbia University, New York, N.Y.). The plasmids pCMV-6 and pCMV-6-HA-Akt were donated by Thomas Franke (Columbia University). The v-Abl expression vector pcDNA-p160v-abl was shared with us by Paul Rothman (Columbia University). The kinase-dead v-Abl mutant expression vector pcDNA-p160v-abl (KD) was constructed by swapping a BstEII-BspEI fragment from pGD-v-Abl (KD) (courtesy of David Baltimore) into pcDNA-p160v-abl. A plasmid encoding the v-Src oncogene under the control of the simian virus 40 early promoter was a gift from A. Levinson (Genentech, Inc., South San Francisco, Calif.).
Cell culture and transfections.
COS cells and 293T cells were maintained in Dulbecco modified Eagle medium (DMEM) supplemented with 10% fetal bovine serum (FBS), 2 mM l-glutamine, and antibiotics. D5 (courtesy of Jean Wang, University of California, San Diego) and BALB/c3T3 cells were maintained in DMEM supplemented with 10% calf serum (CS), 2 mM l-glutamine, and antibiotics. D5 cells were maintained at 39°C. Parental BAF/3 cells and BAF/3 cells stably transfected with p160v-abl (7) were maintained in RPMI 1640 medium supplemented with 10% FBS, 5% WEHI-3 supernatant, 5 μM β-mercaptoethanol, 1 mM sodium pyruvate, and 1 mM l-glutamine. COS cells were transfected with 10 μg of DNA per 2 × 106 cells by the chloroquine DEAE-dextran method (22). 293T cells were transfected with 8 μg of DNA per 2 × 106 cells by the standard calcium phosphate precipitation method. The total amount of DNA per transfection was normalized using empty vector controls.
Antibodies.
Polyclonal antibodies against Sos1-Sos2, Sos1, and Sos2, and monoclonal antibody against c-Myc (9E10) were purchased from Santa Cruz Biotechnology. Anti-Grb2 (Transduction Laboratories) and anti-EGF receptor (EGFR) (Upstate Biotechnology) monoclonal antibodies were used for immunoprecipitation. Polyclonal antibodies against Grb2 and EGFR (Santa Cruz Biotechnology) were used for Western blot analysis. Anti-HA monoclonal antibody was obtained from Boehringer Mannheim and BAbCO. Anti-ACTIVE mitogen-activated protein kinase and anti-Abl antibodies were purchased from Promega and PharMingen, respectively. PhosphoPlus JNK and Akt antibody kits were obtained from New England Biolabs. Monoclonal antibodies against Eps8 and phosphotyrosine were purchased from Transduction Laboratories. Polyclonal antibodies against Abi-1 were described previously (28).
Fusion proteins and in vitro binding assays.
A cDNA fragment encoding amino acids 385 to 475 of Abi-1 was inserted into the bacterial expression vector pMAL-c2 (New England Biolabs) in frame with sequences coding for maltose-binding protein (MBP). Bacterial lysates containing the fusion proteins were prepared as previously described in TNENI (50 mM Tris-HCl [pH 7.5], 50 mM NaCl, 10 mM EDTA, 0.5% NP-40, 1 mM phenylmethylsulfonyl fluoride, 10 μg of aprotinin per ml, 10 μg of leupeptin per ml, 1 μg of pepstatin per ml) and incubated with amylose resin (New England Biolabs) for 1 h at 4°C. The resin was washed four times in lysis buffer, incubated with mammalian cell lysate for 1 h at 4°C, and again washed four times in lysis buffer. Bound proteins were boiled in 2× Laemmli buffer and resolved by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) for either staining with Coomassie brilliant blue or transfer to an Immobilon-P membrane (Millipore). Membrane-bound proteins were subjected to Western blot analysis as described below.
Growth factor deprivation and stimulation.
For serum starvation, COS and 293T cells were washed 16 to 24 h after transfection and incubated in 0.2% FBS for 24 to 30 h. Stimulation with recombinant human EGF (Upstate Biotechnology) was at a concentration of 100 ng/ml for the specified times. For tyrosine phosphorylation studies, BALB/c3T3 and D5 cells were serum starved in 0.5% CS for 24 and 48 h, respectively. Serum-starved BALB/c3T3 cells were stimulated with 20% FBS in DMEM for the indicated times. Parental BAF/3 cells and BAF/3 cells stably transfected with p160v-abl were starved in 0.5% FBS for 8 h prior to stimulation at a concentration of 108 cells/ml with 50 ng of recombinant murine interleukin-3 (IL-3) (Sigma) per ml.
Immunoprecipitation and Western blot analysis.
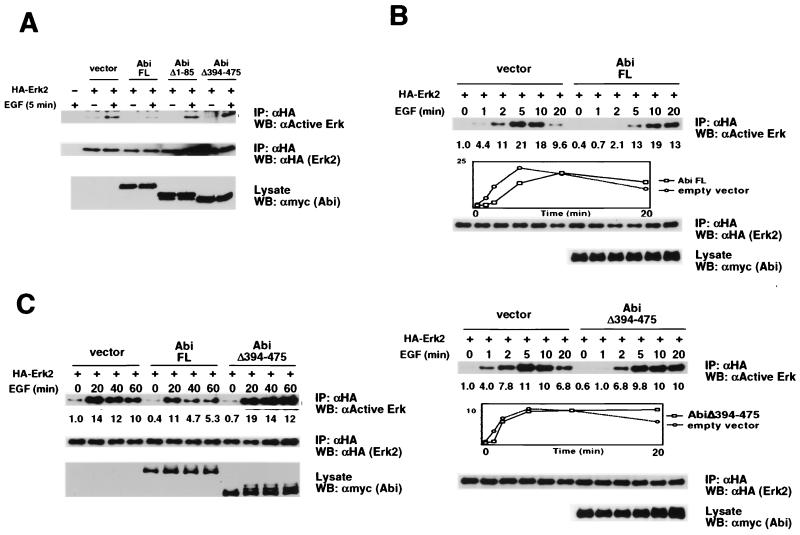
Cells for coimmunoprecipitation experiments were washed in phosphate-buffered saline (PBS)–PI (PBS plus 0.4 mM Na3VO4 and 0.4 mM EDTA) and lysed in TNENI plus 10 mM NaF and 1 mM Na3VO4. Cells for Erk2, JNK, Akt, and tyrosine phosphorylation assays were washed in PBS-PI and solubilized with lysis buffer (20 mM Tris-HCl [pH 8], 138 mM NaCl, 10% glycerol, 1% NP-40, 2 mM EDTA, 10 mM NaF, 1 mM Na3VO4, 1 mM phenylmethylsulfonyl fluoride, 10 μg of aprotinin per ml, 10 μg of leupeptin per ml, and 1 μg of pepstatin per ml). Lysates were incubated at 4°C for 20 min and clarified for 20 min at 14,000 × g and 4°C. Lysates were rocked with antibodies for 1 h at 4°C and for an additional hour with either protein A-agarose or protein G PLUS-agarose (Santa Cruz Biotechnology). The agarose beads were washed four times with lysis buffer and boiled in 2× Laemmli buffer. Eluted proteins were resolved by SDS-PAGE, transferred to an Immobilon-P membrane, and subjected to Western blot analysis as described previously using ECL and ECL Plus detection systems (Amersham) (9). Bands in Figure 4 were quantitated using a densitometer. The amount of active HA-Erk2 at each time point was normalized to the total amount of HA-Erk2. The normalized amount of active HA-Erk2 in unstimulated cells transfected with the empty vector control was assigned a relative value of 1.0.
FIG. 4.
Inhibition by Abi-1 of EGF-induced Erk2 activation but not EGF-induced JNK or Akt activation. (A) COS cells were transiently cotransfected with the indicated HA-Erk2 and myc–Abi-1 plasmids. Serum-starved cells were left untreated or were stimulated with EGF (100 ng/ml) for 5 min. HA-Erk2 was immunoprecipitated from cell lysates with anti-HA antibody (αHA) and immunoblotted with antibodies against active Erks (top). The membrane was reprobed with anti-HA antibody to confirm equal levels of protein loading (middle). Total cell lysates were probed with anti-myc antibody to confirm expression of myc-tagged Abi-1 proteins (bottom). FL, full length; IP, immunoprecipitate; WB, Western blot. (B) The experiment whose results are shown here was performed with the indicated plasmids as described for panel A except that EGF treatment was for 1, 2, 5, 10, or 20 min. The relative normalized amount of active Erk is shown beneath each lane. Graphic plots of relative amounts of active Erk versus time are included for both the upper and lower blots. (C) The experiment whose results are shown here was performed with the indicated plasmids as described for panel A except that EGF treatment was for 20, 40, or 60 min. The relative normalized amount of active Erk is shown beneath each lane. (D) The experiment whose results are shown here was performed as described for panel A except that HA-JNK and antibodies against active JNK replaced HA-Erk2 and antibodies against active Erks, respectively. (E) The experiment whose results are shown here was performed as described for panel A except that HA-Akt and antibodies against active Akt replaced HA-Erk2 and antibodies against active Erks, respectively.
RESULTS
Interaction of Abi-1 and Sos2 in yeast.
To identify proteins that interact with Abi-1, we used the yeast two-hybrid system to screen for proteins that bind to the Abi-1 SH3 domain. The bait plasmid pSH2-AbiSH3 encoding a fusion of the LexADBD and the Abi-1 SH3 domain, was used to screen a library of cDNAs derived from the mouse myelomonocytic leukemia WEHI-3 cell line and expressed as fusions to the Gal4AD. Thirty positive clones were isolated from a screen of approximately 600,000 yeast colonies cotransformed with bait and library plasmids. Four of these clones contained sequences encoding carboxy-terminal domains of the Ras-specific guanine nucleotide exchange factor Sos2. Two Gal4AD-Sos2 hybrids including different carboxy-terminal fragments of Sos2 were each isolated twice. Both hybrids contain all the proline-rich motifs that bind the SH3 domains of Grb2 (34). Neither hybrid includes the Dbl and pleckstrin homology domains or an intact Ras-specific GEF catalytic region of Sos2. Results from the yeast two-hybrid experiments are summarized in Table 1.
TABLE 1.
Yeast two-hybrid tests of the binding of the Abi-1 SH3 domain to the carboxy-terminal domain of Sos2
| Protein moiety fused to LexADBDa | Protein moiety fused to Gal4ADa | β-Galactosidase activityb |
|---|---|---|
| Abi-1(394–475) | None | − |
| None | Abl(4–1087) | − |
| Abi-1(394–475) | Abl(4–1087) | + |
| Abi-1(394–475) | Sos2(874–1297) | + |
| Abi-1(394–475) | Sos2(974–1297) | + |
| None | Sos2(874–1297) | − |
| None | Sos2(974–1297) | − |
The numbers in parentheses indicate the amino acids included in the hybrid protein.
β-Galactosidase activity as an indicator of interaction was scored by the intensity of blue color and the time for color development. −, no blue color in 24 h; +, dark blue color in <1 h.
Association of Abi-1 and Sos in vitro.
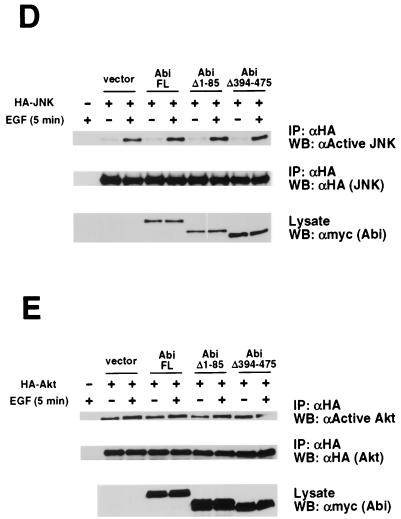
We performed in vitro binding assays to confirm the interaction observed in the yeast two-hybrid system. A fusion protein comprised of MBP and the Abi-1 SH3 domain was tested for binding to Sos proteins from mammalian cell lysates. Bacterially expressed MBP and MBP-Abi SH3 were purified on amylose resin and incubated with various cell extracts. Bound proteins were eluted, resolved by SDS-PAGE, and immunoblotted with antibodies that recognize Sos1, Sos2, or both Sos1 and Sos2. As shown in Fig. 1A, MBP-Abi SH3, but not MBP, was able to precipitate both Sos1 and Sos2 from BALB/c3T3 cell lysates. Similar results were obtained with extracts from WEHI-3 and NIH 3T3 cells (data not shown). To address the specificity of this interaction with MBP-Abi SH3, we compared the binding to Sos2 with the binding to two other proteins containing multiple PXXP motifs, the p62dok-like protein DOKL and Abi-1 itself. Cell extracts were prepared from COS cells expressing each of the truncated or full-length proteins tagged with an HA epitope. Resin-bound MBP-Abi SH3, but not resin-bound MBP, was repeatedly able to precipitate a protein consisting of amino acids 874 to 1297 of Sos2 from total cell lysates, even those containing barely detectable levels of this Sos2 fragment (Fig. 1B). No binding to MBP-Abi SH3 was detected with amino acids 86 to 475 of Abi-1 or with full-length DOKL. These data indicate that the association in vitro between the Abi-1 SH3 domain and Sos is specific.
FIG. 1.
In vitro binding of the Abi-1 SH3 domain to Sos. (A) MBP and MBP-Abi SH3 immobilized on amylose resin were incubated with BALB/c3T3 cell lysate. Bound proteins were immunoblotted with antibodies that recognize both Sos1 and Sos2 (αSos1/2) (top). The membrane was stripped and reprobed with anti-Sos1 (middle) and anti-Sos2 (bottom) antibodies. Total cell lysate and anti-Sos1 and -Sos2 immunoprecipitate (IP) from BALB/c3T3 cells were included for reference. WB, Western blot. (B) COS cells were transiently transfected with plasmids encoding HA-tagged DOKL, Abi(Δ1–85), or Sos2(874–1297) [Sos2(C)]. Cell lysates were incubated with MBP and MBP-Abi SH3 as described for panel A. Total cell lysates and bound proteins were probed with anti-HA antibody (upper). HA-Sos2(874–1297) expression in the total cell lysate was detected upon longer exposure. A fraction of the resin eluate was stained with Coomassie brilliant blue to confirm roughly equivalent amounts of MBP and MBP-Abi SH3 (lower).
Interaction of Abi-1 and Sos in vivo.
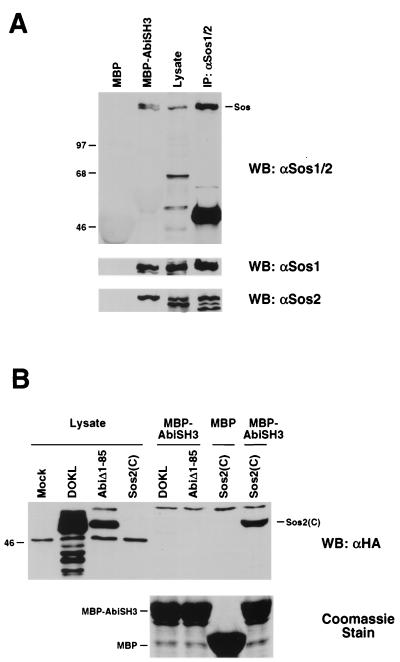
To determine whether Abi-1 and Sos can interact in vivo, we tested the ability of myc epitope-tagged Abi-1 to coimmunoprecipitate with endogenous Sos in COS cells. Plasmids encoding various myc-tagged proteins were transfected into COS cells. Cell lysates were prepared, and proteins were immunoprecipitated with antibodies that recognize both Sos1 and Sos2. Immunoprecipitates were then subjected to SDS-PAGE and Western blot analysis with anti-myc antibody. As illustrated in Fig. 2A, both Abi-1 and AbiΔ328–356, a shorter form generated by alternative splicing, coimmunoprecipitated with endogenous Sos proteins. In contrast, neither c-Abl nor the GM-CSF receptor α chain bound to Sos. These results demonstrate that Sos can associate specifically with Abi-1 in vivo.
FIG. 2.
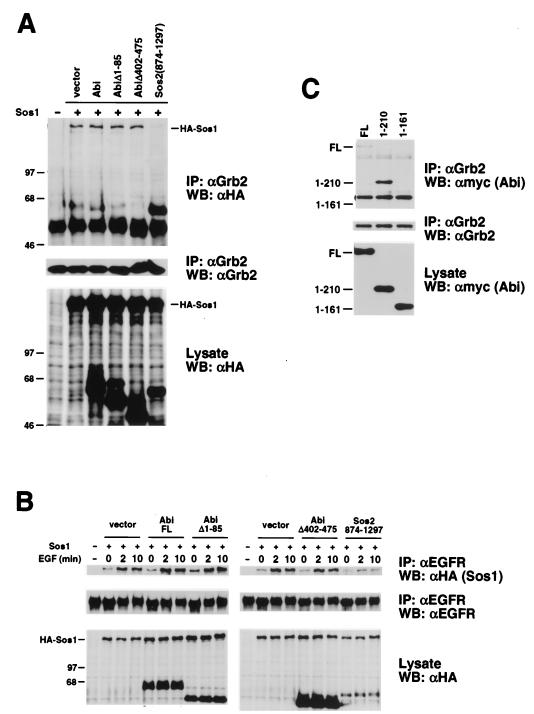
Interaction between Abi-1 and Sos in COS cells. (A) COS cells were transiently transfected with plasmids encoding myc-tagged GM-CSF receptor α chain, c-Abl, Abi-1, or AbiΔ328–356. Sos1 and Sos2 were immunoprecipitated from cell lysates. Precipitated proteins (top) were immunoblotted with anti-myc antibody (αmyc). The membrane from the top blot was reprobed with anti-Sos1 and -Sos2 antibodies to confirm equal levels of protein loading (middle). Total cell lysates were probed with anti-myc antibody to confirm expression of myc-tagged proteins (bottom). IP, immunoprecipitate; WB, Western blot. (B) Schematic representation of full-length (FL) Abi-1 and deletion mutant proteins used in panel C. The homeobox-like domain, proline-rich motifs, polyproline region, and SH3 domain are indicated. (C) COS cells were transiently transfected with plasmids encoding myc-tagged full-length Abi-1 or the indicated deletion mutant proteins. Proteins were immunoprecipitated from cell lysates with anti-Sos1 and -Sos2 antibodies and were analyzed as described for panel A. The asterisk indicates the position of the immunoglobulin heavy chain.
We next mapped the interaction domain on Abi-1 by testing various myc-tagged deletion mutant proteins of Abi-1 for binding to endogenous Sos. It should be noted that the cDNA clone previously reported as full-length Abi-1 (28) was truncated at the 5′ end; this clone is now defined as encoding a deletion mutant lacking amino acids 1 to 85. Full-length Abi-1 sequences were isolated by rapid amplification of cDNA ends and cloned into expression vectors (A. Ikeguchi and S. P. Goff, unpublished data). Full-length Abi-1 and the deletion mutant proteins examined are depicted schematically in Fig. 2B. Surprisingly, the association of Abi-1 with Sos was not eliminated but instead was slightly enhanced by deletion of the SH3 domain (AbiΔ394–475). Additional carboxy-terminal truncation of Abi-1 to amino acid 210 also failed to disrupt the interaction. Further deletion of amino acids 162 to 210, however, dramatically reduced the binding. In addition, Abi-1 mutant proteins lacking amino acids 1 to 85 failed to interact with Sos. Thus, in contrast to the results of initial in vitro binding studies by our laboratory and others (27) which detected a strong association between the Abi-1 SH3 domain and Sos, this analysis indicates that amino-terminal regions of Abi-1 are more important than the SH3 domain for binding to Sos in mammalian cells. These results, however, do not rule out the possibility that the SH3 domain of Abi-1 also interacts with Sos in vivo. It is unclear whether the two amino-terminal regions of Abi-1 required for binding, amino acids 1 to 85 and 162 to 210, are each sufficient independently to mediate this interaction.
We also investigated whether the binding of Abi-1 to Eps8 is necessary for the interaction between Abi-1 and Sos. As shown in Fig. 2C, the interaction was not disrupted by deletion of a proline-rich region (AbiΔ304–401) that includes the Eps8-binding site. This result indicates that binding to Eps8 is not required for the interaction between Abi-1 and Sos.
Association of Abi-1 and Grb2 in vivo.
One possible consequence of the association between Abi-1 and Sos might be the exclusion of other Sos interaction partners such as Grb2. We tested whether overexpression of HA–Abi-1 can disrupt the ability of cotransfected HA-Sos1 to coimmunoprecipitate with endogenous Grb2. Plasmids encoding HA-tagged Abi-1 and HA-tagged Sos1 were cotransfected into COS cells. Cell extracts were incubated with anti-Grb2 antibody, and the immunoprecipitates were subjected to Western blot analysis with anti-HA antibody. Overexpression of HA–Abi-1 did not alter the amount of HA-Sos1 that coimmunoprecipitated with endogenous Grb2 compared to that coimmunoprecipitated with the empty vector control (Fig. 3A). Overexpression of HA-AbiΔ1–85 or HA-AbiΔ402–475 also failed to disrupt the interaction. In contrast, overexpression of HA-Sos2(874–1297) abolished binding of HA-Sos1 to Grb2. In addition, cotransfection of myc-tagged Abi-1 with HA-Sos1 did not perturb the association of HA-Sos1 with endogenous Grb2 (data not shown). These data suggest that binding of Abi-1 to Sos does not disrupt Grb2-Sos complexes.
FIG. 3.
Association of Abi-1 and Grb2 in COS cells. (A) COS cells were transiently cotransfected with the indicated HA-Sos1 and HA–Abi-1 plasmids. Grb2 was immunoprecipitated from cell lysates. Precipitated proteins (top) and total cell lysates (bottom) were immunoblotted with anti-HA antibody (αHA). The membrane from the top blot was reprobed with anti-Grb2 antibody to confirm equal levels of protein loading (middle). IP, immunoprecipitate; WB, Western blot. (B) COS cells were transiently cotransfected with the indicated HA-Sos1 and HA–Abi-1 plasmids. Serum-starved cells were left untreated or were stimulated with EGF (100 ng/ml) for 2 or 10 min. Cell lysates were prepared and incubated with anti-EGFR antibody. Precipitated proteins (top) and total cell lysates (bottom) were immunoblotted with anti-HA antibody. The membrane from the top blot was reprobed with anti-EGFR antibody to confirm equal levels of protein loading. FL, full-length. (C) COS cells were transiently transfected with the indicated myc–Abi-1 plasmids. Grb2 was immunoprecipitated from cell lysates. Precipitated proteins (top) and total cell lysates (bottom) were immunoblotted with anti-myc antibody. The membrane from the top blot was reprobed with anti-Grb2 antibody to confirm equal levels of protein loading (middle).
We also tested whether overexpression of Abi-1 can block recruitment of Sos to activated EGFRs. Plasmids encoding HA-tagged Abi-1 and HA-tagged Sos1 were cotransfected into COS cells. Serum-starved cells were left untreated or were stimulated with EGF (100 ng/ml) for 2 or 10 min. Cell extracts were then incubated with anti-EGFR antibody, and the immunoprecipitates were subjected to Western blot analysis with anti-HA antibody. Stimulation with EGF for 2 or 10 min increased the amount of HA-Sos1 that coimmunoprecipitated with endogenous EGFR (Fig. 3B). Compared to the empty vector control, none of the HA–Abi-1 constructs disrupted binding of HA-Sos1 to EGFR in untreated or stimulated cells. In contrast, coimmunoprecipitation of HA-Sos1 with EGFR was significantly reduced by overexpression of HA-Sos2(874–1297). It should be noted that this effect was observed even though the level of expression of HA-Sos2(874–1297) relative to that of HA–Abi-1 was severalfold lower.
The above results indicate that overexpression of Abi-1 does not disrupt Grb2-Sos complexes or their recruitment to activated EGF receptors. These findings also suggest the possibility that Abi-1, Sos, and Grb2 can coexist in a single complex. Consistent with this idea, we found that myc-tagged Abi-1 can coimmunoprecipitate with endogenous Grb2 (Fig. 3C). The N-terminal 210 amino acids of Abi-1 that were sufficient for binding to Sos were also capable of mediating the interaction with Grb2. Thus, the amino terminus of Abi-1 can either directly or indirectly mediate binding to both Sos and Grb2. Our results to date, however, do not distinguish between simultaneous or mutually exclusive binding of Sos and Grb2 to Abi-1. It is also unclear at this time whether either Sos or Grb2 is required for bridging of Abi-1 to the other protein in vivo.
Inhibition of EGF-induced Erk2 activation by overexpression of Abi-1.
To determine the functional significance of the interaction between Abi-1 and Sos, we investigated the role of Abi-1 in Sos-dependent signal transduction. We assayed the effect of overexpression of Abi-1 on two EGF-induced signaling events downstream of Sos and Ras, namely, the activation of Erk2 and JNK (14). In addition, we assayed the effect of Abi-1 overexpression on EGF-induced activation of Akt. Activation of Akt by EGF is PI3K dependent, but the importance of Ras in this pathway is unclear (3, 10). In the Erk2 assay, plasmids encoding myc-tagged Abi-1 and HA-tagged Erk2 were cotransfected into COS cells. Serum-starved cells were left untreated or were stimulated with EGF for the times indicated in Fig. 4. To determine the effect of Abi-1 overexpression on EGF-induced Erk2 activation, HA-Erk2 was immunoprecipitated from cell lysates and then subjected to Western blot analysis with phospho-specific antibodies that recognize only the active forms of Erk1 and Erk2. As expected, treatment of serum-starved cells with EGF for 5 min strongly upregulated the amount of active HA-Erk2 (Fig. 4A). Significantly, both basal and EGF-induced HA-Erk2 activities were inhibited in cells expressing full-length Abi-1 compared to the levels in cells transfected with the empty vector control. This inhibition was not observed with either AbiΔ1–85 or AbiΔ394–475. Interestingly, a time course analysis between 0 and 20 min of EGF stimulation revealed a delay in HA-Erk2 activation in cells transfected with full-length Abi-1 as opposed to that in cells transfected with the empty vector control (Fig. 4B, upper blots). In addition, overexpression of full-length Abi-1 appeared to reduce Erk activity measured after 40 and 60 min of EGF stimulation (Fig. 4C). While the effect of Abi-1 overexpression on Erk activation at intermediate time points (10 or 20 min) was slightly variable, we consistently observed a delay at early time points (1 and 2 min) and inhibition at late time points (40 and 60 min). In contrast, cells transfected with either AbiΔ394–475 or AbiΔ1–85 exhibited less delay in Erk activation and no reduction of Erk activity after 40 and 60 min of EGF stimulation (Fig. 4B, lower blots, C and data not shown). These results suggest that full delay and inhibition of EGF-induced Erk2 activation by Abi-1 requires both the interaction between Abi-1 and Sos and the presence of an intact Abi-1 SH3 domain.
In similar experiments, we tested the effect of Abi-1 overexpression on EGF-induced HA-JNK and HA-Akt1 activation. As expected, treatment of serum-starved cells with EGF for 5 min increased the amounts of active HA-JNK and HA-Akt (Fig. 4D and E). Relative to levels induced with the empty vector control, none of the Abi-1 constructs altered either basal or EGF-induced levels of HA-JNK and HA-Akt activation. No increases or decreases of EGF-induced JNK or Akt activation by overexpression of Abi-1 were revealed by time course analyses (data not shown). These results indicate that the inhibition of EGF-induced Erk2 activation by Abi-1 is specific to the Erk pathway.
Inhibition of v-Abl-induced Erk2 activation by overexpression of Abi-1.
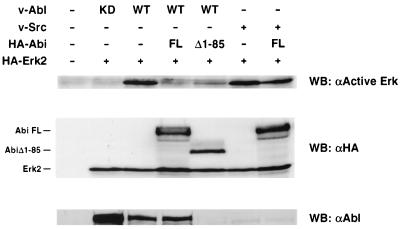
Transformation by v-Abl has been shown to require activated Ras (26). We have previously found that overexpression of either full-length Abi-1 or AbiΔ1–85 can inhibit transformation by v-Abl (28; A. Ikeguchi and S. P. Goff, unpublished data). Taken together, these findings raise the possibility that overexpression of Abi-1 inhibits v-Abl-mediated transformation by blocking Ras-dependent signaling events. Therefore, we investigated whether overexpression of Abi-1 can block v-Abl-induced Erk2 activation. 293T cells instead of COS cells were used for this experiment because the levels of v-Abl expression and v-Abl-induced Erk activation were severalfold lower in COS cells than those in 293T cells. 293T cells were cotransfected with plasmids encoding HA-Abi-1, HA-Erk2, and either v-Abl or v-Src. Cell lysates were prepared and probed with antibodies against active Erks. As shown in Fig. 5, wild-type v-Abl strongly induced Erk2 activation compared to the level induced by a mutant v-Abl lacking kinase activity. Overexpression of full-length Abi-1 completely abolished v-Abl-induced Erk2 activation. This inhibition of Erk2 activation occurred with no significant change in the levels of v-Abl protein; thus, full-length Abi-1 mediated a strong reduction in the specific activity of v-Abl for Erk signaling. Overexpression of AbiΔ1–85, which binds to Abl but not to Sos, also inhibited v-Abl-induced Erk activation but to a lesser extent than full-length Abi-1. It should be noted that in comparison to cells coexpressing full-length Abi-1 and v-Abl, cells coexpressing AbiΔ1–85 and v-Abl show markedly decreased expression of v-Abl. This phenomenon has been observed repeatedly and most likely contributes to reduced Erk activation. Thus, although AbiΔ1–85 did show significant inhibition of Erk2 activation, it did so primarily by a mechanism distinct from that used by the full-length protein. The residual Erk2 activation, when normalized to the low levels of v-Abl remaining, indicates a minimal effect of AbiΔ1–85 on the specific activity of v-Abl for Erk signaling. The effect of overexpression of the non-v-Abl-binding mutant AbiΔ394–475 on v-Abl-induced Erk2 activation could not be evaluated due to nearly undetectable levels of this mutant Abi protein when coexpressed with v-Abl (data not shown). However, Erk2 activation induced by v-Src, which does not bind Abi-1, was largely unaffected by overexpression of Abi-1 (Fig. 5). Thus, the binding of Abi-1 to Sos is not sufficient to block Erk activation by all mitogenic stimuli. Western blot analysis showed that the levels of HA–Abi-1 expression, HA-Erk2 expression, and HA–Erk2 activation in cells expressing these tyrosine kinases were similar. In summary, overexpression of Abi-1 can inhibit Erk2 activation induced by both EGF and v-Abl. In both cases, the maximal inhibitory effect requires amino acids 1 to 85 of Abi-1, a region also required for the interaction with Sos. Our results also suggest the possibility of distinct mechanisms of inhibition of v-Abl-transforming activity for full-length Abi-1 and AbiΔ1–85.
FIG. 5.
Inhibition of v-Abl-induced Erk2 activation by Abi-1. 293T cells were transiently cotransfected with plasmids encoding HA-Erk2, wild-type (WT) p160v-abl, kinase-dead (KD) p160v-abl, or v-Src, and full-length HA–Abi-1 (FL) or HA–AbiΔ1–85 (Δ1–85). Transfected cells were serum starved in 0.2% FBS for 24 h before lysis. Cell lysates were probed with antibodies against active Erks (αActive Erk) (top), HA (middle), and Abl (bottom). Expression of v-Src was not detected by our methods. WB, Western blot.
Tyrosine phosphorylation of endogenous Abi-1 induced by v-Abl.
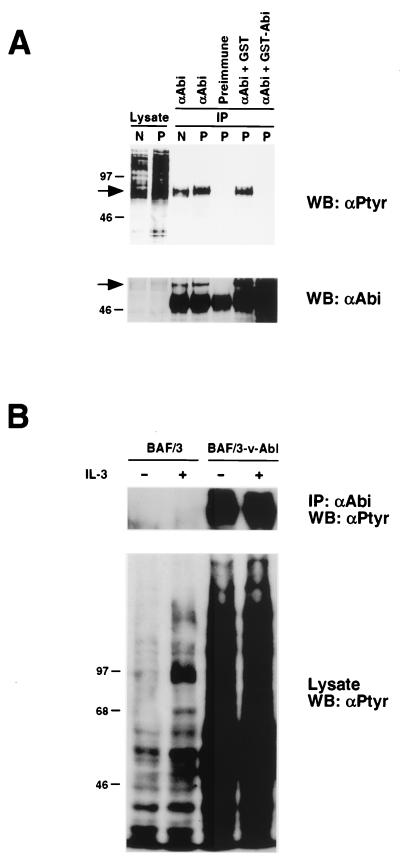
Abi-1 and v-Abl were previously shown to interact in vivo (28). In addition, earlier results demonstrated that v-Abl can phosphorylate Abi-1 in vitro. To determine whether v-Abl kinase activity can lead to tyrosine phosphorylation of endogenous Abi-1, we took advantage of the D5 cell line, an NIH 3T3 subclone transformed with a temperature-sensitive mutant v-Abl (24). The v-Abl kinase in D5 cells is inactive at 39°C, but kinase activity is restored upon shifting cells to 32°C. We investigated whether shifting D5 cells from the restrictive to the permissive temperature could increase tyrosine phosphorylation of endogenous Abi-1. Abi-1 was immunoprecipitated from serum-starved D5 cells that were maintained at 39°C or were shifted from 39 to 32°C for 1 h. Precipitated proteins were immunoblotted with antiphosphotyrosine antibody. As shown in Fig. 6A, the phosphotyrosine content of Abi-1 was increased after shifting D5 cells to the permissive temperature. In addition, the mobility of Abi-1 in gel electrophoresis was reduced after the temperature shift, consistent with postranslational modifications such as phosphorylation. Since the ability of the anti-Abi-1 antibodies to immunoprecipitate endogenous Abi-1 had not been previously characterized, we included controls to confirm this result. Preimmune serum was unable to immunoprecipitate putative Abi-1. In addition, preadsorption of anti-Abi-1 antibodies with bacterial lysate containing glutathione S-transferase (GST)–Abi-1 abolished recognition of this protein. Preadsorption of anti-Abi-1 antibodies with bacterial lysate containing GST alone did not block immunoprecipitation of putative Abi-1. These controls indicate that the anti-Abi-1 antibodies can specifically recognize endogenous Abi-1. It should be noted, however, that we have not ruled out the possibility of cross-reactivity against Abi-2. In addition, the apparent increase in Abi-1 detected after preadsorption with bacterial lysates is due to cross-reactivity against comigrating bacterial proteins (Fig. 6A and data not shown).
FIG. 6.
Tyrosine phosphorylation of endogenous Abi-1 induced by v-Abl. (A) Serum-starved D5 cells were left at 39°C (nonpermissive temperature) or shifted to 32°C (permissive temperature) for 1 h. Abi-1 was immunoprecipitated from cell lysates and immunoblotted with antiphosphotyrosine antibody (αPtyr) (top). The membranes were reprobed with anti-Abi-1 antibodies to confirm equal levels of protein loading (bottom). The arrows indicate the position of Abi-1. N and P denote nonpermissive and permissive temperatures, respectively. WB, Western blot. (B) Growth factor-deprived parental BAF/3 cells and BAF/3 cells stably transfected with p160v-abl were left untreated or were stimulated with IL-3 (50 ng/ml). Abi-1 was immunoprecipitated and immunoblotted as described for panel A. IP, immunoprecipitate.
We also examined the phosphotyrosine content of endogenous Abi-1 in cells that stably express wild-type v-Abl. The phosphotyrosine content of endogenous Abi-1 is dramatically increased in BAF/3 cells stably transfected with p160v-abl compared to that in parental BAF/3 cells (Fig. 6B). Stimulation of parental BAF/3 cells with IL-3, however, did not result in increased tyrosine phosphorylation of Abi-1. Taken together, our data demonstrate that v-Abl kinase activity can lead to tyrosine phosphorylation of endogenous Abi-1.
Tyrosine phosphorylation of endogenous Abi-1 induced by serum stimulation.
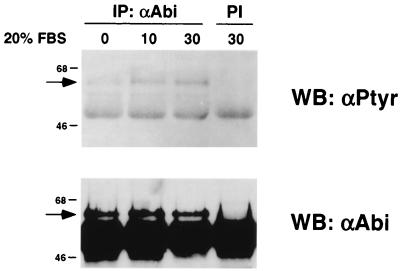
We next examined whether serum stimulation of fibroblasts could also induce tyrosine phosphorylation of endogenous Abi-1. Serum-starved BALB/c3T3 cells were left untreated or were stimulated with 20% FBS for 10 or 30 min. Abi-1 was immunoprecipitated from cell lysates and immunoblotted with antiphosphotyrosine antibody. As shown in Fig. 7, serum stimulation induced tyrosine phosphorylation of Abi-1 within 10 min. The levels of tyrosine phosphorylation were similar after 10 or 30 min of serum stimulation. These results show that serum stimulation as well as v-Abl kinase activity can result in tyrosine phosphorylation of endogenous Abi-1.
FIG. 7.
Tyrosine phosphorylation of endogenous Abi-1 induced by serum stimulation. Serum-starved BALB/c3T3 cells were stimulated with 20% FBS for the indicated times (in minutes). Abi-1 was immunoprecipitated from cell lysates with anti-Abi-1 antibodies (αAbi) and immunoblotted with antiphosphotyrosine antibody (top). The membrane was reprobed with anti-Abi-1 antibodies to confirm equal levels of protein loading (bottom). The arrows indicate the position of Abi-1. PI, preimmune serum control; IP, immunoprecipitate; WB, Western blot.
DISCUSSION
Our search for potential regulators or targets of Abi-1 functions has identified Sos as a novel interaction partner of Abi-1. The association between Abi-1 and Sos has been demonstrated both in vitro and in vivo. Our results indicate that overexpression of Abi-1 can inhibit the Erk pathway. We have mapped an indispensable Sos interaction domain in vivo to the amino terminus of Abi-1 and have shown that this region is required for the maximal inhibitory effect of Abi-1 overexpression of EGF- and v-Abl-induced Erk2 activation. In contrast to in vitro binding experiments performed by our laboratory and others (27), the SH3 domain of Abi-1 is not necessary in vivo for interaction with Sos. The SH3 domain is, however, required for full inhibition of EGF-induced Erk2 activation.
During the preparation of this paper, Scita et al. reported genetic evidence that Eps8 participates in signal transduction between Ras and Rac (27). Those authors also provided biochemical data supporting a role for a ternary complex of Eps8, Abi-1, and Sos in generating Rac-specific GEF activity. Previous work had identified a Rac-specific GEF activity of Sos and suggested that Ras-mediated PI3K signaling through Sos might couple Ras and Rac activation (16). A study using overexpression and immunodepletion of Abi-1 and Eps8 suggests a positive role for Abi-1 in linking Ras to Rac (27). Interestingly, others have shown that v-Abl-mediated induction of Erk activity can occur by Raf-independent (32) and Rac-dependent (25) pathways. However, it is unlikely that inhibition of EGF-induced Erk activation by overexpression of Abi-1 is due to inhibition of Rac activity. Although our results confirm the physical interaction between Abi-1 and Sos, we did not observe any effect of Abi-1 overexpression on EGF-induced activation of JNK and Akt downstream of Rac and PI3K, respectively. In our experiments, however, we overexpressed Abi-1 alone and not in conjunction with Eps8. In addition, antibodies used to immunodeplete Abi-1 might cross-react with other Abi family members that exhibit different cellular activities. It is possible that the overall effect on signaling components downstream of Ras is highly sensitive to the relative stoichiometries of Abi family members and their interaction partners, such as Eps8.
Regulation of the interactions of Abi-1 with its various binding partners is likely to be complex. First, phosphorylation of Abi-1 appears to be regulated during the cell cycle and can alter the association of Abi-1 with Eps8 (2). Hyperphosphorylated forms of Abi-1 fail to coimmunoprecipitate with Eps8. Recently, the SH3 domain of Eps8 has been shown to bind a novel PXXDY consensus sequence found in Abi-1 and other Eps8 interaction partners (15). Therefore, it is possible that phosphorylation of the tyrosine residue might regulate binding of the Eps8 SH3 domain to this motif. Notably, we have observed tyrosine phosphorylation of endogenous Abi-1 in response to both v-Abl kinase activation and serum stimulation. Second, the Abi-1 mRNA is subject to alternative splicing, yielding protein isoforms that can retain different sets of proline-rich motifs (36). The differential expression of proline-rich motifs may modulate the binding preferences and biological activities of Abi-1. For example, alternative splicing of the neuronal signaling adapter protein NUMB generates two types of isoforms with either a short or long proline-rich region. These two types of NUMB isoforms have distinct developmental functions (31). Third, we have recently found that Abi-1 can oligomerize (data not shown). Both the oligomerization and Sos interaction domains map to the amino terminus of Abi-1, suggesting possible interplay between homomeric and heteromeric interactions of Abi-1. There are examples of this interplay among SH3-containing proteins. The SH3 domain of Eps8 exists in monomeric and dimeric forms but binds to Abi-1 only as a monomer (15). In addition, a recent report indicates that the binding of SH3-containing amphiphysins to dynamin, a GTPase involved in endocytic membrane trafficking, can prevent dynamin self-assembly into oligomeric ring-shaped structures (17). Finally, binding of an interaction partner to Abi-1 might promote or disrupt other heteromeric interactions. Our results suggest that the amino terminus of Abi-1 interacts with both Sos and Grb2. Therefore, it is possible that the amino-terminal and SH3 domains of Abi-1 can bind simultaneously to Sos or Grb2 and Abl, respectively. However, it is not yet known whether the interaction of Abi-1 with Sos or Grb2 affects the binding of Abi-1 to Abl proteins.
Our observation that overexpression of Abi-1 results in inhibition and delay of EGF-induced Erk activation suggests a role for Abi-1 in temporal regulation of Erk activity. The timing of Erk activation within the cell can be critical in determining the biological response to growth factor stimulation. For example, in PC12 cells, sustained activation of Erks is required for neuronal differentiation in response to nerve growth factor (NGF) (13). In contrast, transient activation of Erks in response to EGF treatment of the same cells elicits a proliferative response. It has been proposed that the Ras-related GTPase Rap1 mediates the persistent activation of Erks induced by NGF (35). It is possible that Abi-1 interacts with Rap1-specific GEFs to regulate Rap1 and the duration of Erk activation. However, this role for Rap1 in Erk activation has recently been challenged (37). It is also possible that Abi-1 might interact with other GEFs belonging to the Dbl family. The Dbl family member Lfc localizes to microtubules, and overexpression of Lfc in NIH 3T3 cells induces formation of actin stress fibers and membrane ruffles (11). Staining of Abi-1 overexpressed in NIH 3T3 cells reveals a distinct filamentous pattern within the cytoplasm, consistent with a role for Abi-1 in cytoskeletal reorganization (data not shown). Therefore, Abi family members might interact with GEFs associated with the cytoskeleton during cellular responses to growth factor stimulation.
In addition to finding a novel protein-protein interaction of Abi-1, our work identifies a specific mitogenic pathway potentially regulated by Abi proteins. The inhibition of EGF-induced Erk2 activation by overexpression of Abi-1 is consistent with the hypothesized role of Abi proteins as negative regulators of cell growth. Overexpression of Abi-1 also blocked v-Abl-induced Erk2 activation, providing a possible explanation for the previously observed antagonistic role of overexpressed full-length Abi-1 in v-Abl-mediated transformation (A. Ikeguchi and S. P. Goff, unpublished data). Since Abi proteins do not appear to directly inhibit Abl kinase activity (28, 33), it has been proposed that their binding to v-Abl blocks access to critical regulators or effectors of v-Abl function. For example, interaction with Abi proteins might preclude association of v-Abl with Shc, Grb2, Crk, or Nck adapter proteins that can potentially couple Abl to Sos, Ras, and the Erk pathway. Consistent with this idea, in vitro binding experiments suggest that Grb2, Nck, and Abi-2 can bind to the same proline-rich motif in Abl (5, 23). Furthermore, the binding of Abi-1 to Sos alone is apparently not sufficient to block all signaling to Erks; for example, overexpression of Abi-1 fails to abolish v-Src-induced Erk activation. Alternatively, Abi proteins in other contexts may utilize different mechanisms to modulate signal transduction. For example, overexpression of AbiΔ1–85, which still binds to v-Abl but not to Sos, causes a strong downregulation of v-Abl protein levels in our transient-cotransfection experiments. The requirement for Abi proteins to disrupt recruitment of adapter proteins or alter expression of v-Abl in the inhibition of oncogenic transformation by Abelson murine leukemia virus remains to be investigated.
We cannot rule out the possibility that, at physiological levels, Abi-1 may participate in a positive way in transducing signals initiated by v-Abl or EGF. Experiments with Drosophila suggest that both fly and human Abi can enhance the ability of Abl to phosphorylate one of its substrates, Ena, consistent with a positive role for Abi in this pathway (12). In addition to recruitment of proteins through its SH3 domain and proline-rich motifs, tyrosine phosphorylation of Abi-1 might create docking sites for SH2-containing signaling proteins. Thus, overexpression of Abi-1 in our system may simply uncouple key signaling components. Nevertheless, it is intriguing to consider that oncogenic forms of Abl and Src trigger the ubiquitin-dependent proteolysis of Abi proteins (6). Therefore, it is possible that oncogenic forms of Abl and Src circumvent the potential inhibitory activities of Abi proteins by targeting these molecules for degradation.
ACKNOWLEDGMENTS
We thank K. Calame, T. Franke, A. Minden, P. Rothman, and J. Wang for various plasmids and cell lines. We thank F. Cong, H. Yang, and A. Ikeguchi for construction of several plasmids. We thank M. Dorsch and P. Rothman for critical reading of the manuscript and helpful discussion.
This work was supported by Public Health Service grant P01 CA 75399 from the National Cancer Institute. Support was also provided by the National Institutes of Health grant MSTP 5T35HL07616. S.P.G. is an investigator of the Howard Hughes Medical Institute.
REFERENCES
- 1.Aronheim A, Engelberg D, Li N, al-Alawi N, Schlessinger J, Karin M. Membrane targeting of the nucleotide exchange factor Sos is sufficient for activating the Ras signaling pathway. Cell. 1994;78:949–961. doi: 10.1016/0092-8674(94)90271-2. [DOI] [PubMed] [Google Scholar]
- 2.Biesova Z, Piccoli C, Wong W T. Isolation and characterization of e3B1, an eps8 binding protein that regulates cell growth. Oncogene. 1997;14:233–241. doi: 10.1038/sj.onc.1200822. [DOI] [PubMed] [Google Scholar]
- 3.Burgering B M, Coffer P J. Protein kinase B (c-Akt) in phosphatidylinositol-3-OH kinase signal transduction. Nature. 1995;376:599–602. doi: 10.1038/376599a0. [DOI] [PubMed] [Google Scholar]
- 4.Cong F, Yuan B, Goff S P. Characterization of a novel member of the DOK family that binds and modulates Abl signaling. Mol Cell Biol. 1999;19:8314–8325. doi: 10.1128/mcb.19.12.8314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dai Z, Pendergast A M. Abi-2, a novel SH3-containing protein interacts with the c-Abl tyrosine kinase and modulates c-Abl transforming activity. Genes Dev. 1995;9:2569–2582. doi: 10.1101/gad.9.21.2569. [DOI] [PubMed] [Google Scholar]
- 6.Dai Z, Quackenbush R C, Courtney K D, Grove M, Cortez D, Reuther G W, Pendergast A M. Oncogenic Abl and Src tyrosine kinases elicit the ubiquitin-dependent degradation of target proteins through a Ras-independent pathway. Genes Dev. 1998;12:1415–1424. doi: 10.1101/gad.12.10.1415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Danial N N, Losman J A, Lu T, Yip N, Krishnan K, Krolewski J, Goff S P, Wang J Y, Rothman P B. Direct interaction of Jak1 and v-Abl is required for v-Abl-induced activation of STATs and proliferation. Mol Cell Biol. 1998;18:6795–6804. doi: 10.1128/mcb.18.11.6795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Danial N N, Pernis A, Rothman P B. Jak-STAT signaling induced by the v-abl oncogene. Science. 1995;269:1875–1877. doi: 10.1126/science.7569929. [DOI] [PubMed] [Google Scholar]
- 9.Dorsch M, Fan P D, Bogenberger J, Goff S P. TPO and IL-3 induce overlapping but distinct protein tyrosine phosphorylation in a myeloid precursor cell line. Biochem Biophys Res Commun. 1995;214:424–431. doi: 10.1006/bbrc.1995.2304. [DOI] [PubMed] [Google Scholar]
- 10.Franke T F, Yang S I, Chan T O, Datta K, Kazlauskas A, Morrison D K, Kaplan D R, Tsichlis P N. The protein kinase encoded by the Akt proto-oncogene is a target of the PDGF-activated phosphatidylinositol 3-kinase. Cell. 1995;81:727–736. doi: 10.1016/0092-8674(95)90534-0. [DOI] [PubMed] [Google Scholar]
- 11.Glaven J A, Whitehead I, Bagrodia S, Kay R, Cerione R A. The Dbl-related protein, Lfc, localizes to microtubules and mediates the activation of Rac signaling pathways in cells. J Biol Chem. 1999;274:2279–2285. doi: 10.1074/jbc.274.4.2279. [DOI] [PubMed] [Google Scholar]
- 12.Juang J L, Hoffmann F M. Drosophila abelson interacting protein (dAbi) is a positive regulator of abelson tyrosine kinase activity. Oncogene. 1999;18:5138–5147. doi: 10.1038/sj.onc.1202911. [DOI] [PubMed] [Google Scholar]
- 13.Marshall C J. Specificity of receptor tyrosine kinase signaling: transient versus sustained extracellular signal-regulated kinase activation. Cell. 1995;80:179–185. doi: 10.1016/0092-8674(95)90401-8. [DOI] [PubMed] [Google Scholar]
- 14.Minden A, Lin A, McMahon M, Lange-Carter C, Derijard B, Davis R J, Johnson G L, Karin M. Differential activation of ERK and JNK mitogen-activated protein kinases by Raf-1 and MEKK. Science. 1994;266:1719–1723. doi: 10.1126/science.7992057. [DOI] [PubMed] [Google Scholar]
- 15.Mongiovi A M, Romano P R, Panni S, Mendoza M, Wong W T, Musacchio A, Cesareni G, Paolo Di Fiore P. A novel peptide-SH3 interaction. EMBO J. 1999;18:5300–5309. doi: 10.1093/emboj/18.19.5300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nimnual A S, Yatsula B A, Bar-Sagi D. Coupling of Ras and Rac guanosine triphosphatases through the Ras exchanger Sos. Science. 1998;279:560–563. doi: 10.1126/science.279.5350.560. [DOI] [PubMed] [Google Scholar]
- 17.Owen D J, Wigge P, Vallis Y, Moore J D, Evans P R, McMahon H T. Crystal structure of the amphiphysin-2 SH3 domain and its role in the prevention of dynamin ring formation. EMBO J. 1998;17:5273–5285. doi: 10.1093/emboj/17.18.5273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Owen P J, Musk P, Evans C A, Whetton A D. Cellular signaling events elicited by v-abl associated with growth factor independence in an interleukin-3-dependent cell line. J Biol Chem. 1993;268:15696–15703. [PubMed] [Google Scholar]
- 19.Prywes R, Foulkes J G, Baltimore D. The minimum transforming region of v-abl is the segment encoding protein-tyrosine kinase. J Virol. 1985;54:114–122. doi: 10.1128/jvi.54.1.114-122.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Raffel G D, Parmar K, Rosenberg N. In vivo association of v-Abl with Shc mediated by a non-phosphotyrosine-dependent SH2 interaction. J Biol Chem. 1996;271:4640–4645. doi: 10.1074/jbc.271.9.4640. [DOI] [PubMed] [Google Scholar]
- 21.Raitano A B, Halpern J R, Hambuch T M, Sawyers C L. The Bcr-Abl leukemia oncogene activates Jun kinase and requires Jun for transformation. Proc Natl Acad Sci USA. 1995;92:11746–11750. doi: 10.1073/pnas.92.25.11746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Reicin A S, Ohagen A, Yin L, Hoglund S, Goff S P. The role of Gag in human immunodeficiency virus type 1 virion morphogenesis and early steps of the viral life cycle. J Virol. 1996;170:8645–8652. doi: 10.1128/jvi.70.12.8645-8652.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ren R, Ye Z S, Baltimore D. Abl protein-tyrosine kinase selects the Crk adapter as a substrate using SH3-binding sites. Genes Dev. 1994;8:783–795. doi: 10.1101/gad.8.7.783. [DOI] [PubMed] [Google Scholar]
- 24.Renshaw M W, Kipreos E T, Albrecht M R, Wang J Y. Oncogenic v-Abl tyrosine kinase can inhibit or stimulate growth, depending on the cell context. EMBO J. 1992;11:3941–3951. doi: 10.1002/j.1460-2075.1992.tb05488.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Renshaw M W, Lea-Chou E, Wang J Y. Rac is required for v-Abl tyrosine kinase to activate mitogenesis. Curr Biol. 1996;6:76–83. doi: 10.1016/s0960-9822(02)00424-4. [DOI] [PubMed] [Google Scholar]
- 26.Sawyers C L, McLaughlin J, Witte O N. Genetic requirement for Ras in the transformation of fibroblasts and hematopoietic cells by the Bcr-Abl oncogene. J Exp Med. 1995;181:307–313. doi: 10.1084/jem.181.1.307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Scita G, Nordstrom J, Carbone R, Tenca P, Giardina G, Gutkind S, Bjarnegard M, Betsholtz C, Di Fiore P P. EPS8 and E3B1 transduce signals from Ras to Rac. Nature. 1999;401:290–293. doi: 10.1038/45822. [DOI] [PubMed] [Google Scholar]
- 28.Shi Y, Alin K, Goff S P. Abl-interactor-1, a novel SH3 protein binding to the carboxy-terminal portion of the Abl protein, suppresses v-abl transforming activity. Genes Dev. 1995;9:2583–2597. doi: 10.1101/gad.9.21.2583. [DOI] [PubMed] [Google Scholar]
- 29.Tanaka M, Gupta R, Mayer B J. Differential inhibition of signaling pathways by dominant-negative SH2/SH3 adapter proteins. Mol Cell Biol. 1995;15:6829–6837. doi: 10.1128/mcb.15.12.6829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Varticovski L, Daley G Q, Jackson P, Baltimore D, Cantley L C. Activation of phosphatidylinositol 3-kinase in cells expressing abl oncogene variants. Mol Cell Biol. 1991;11:1107–1113. doi: 10.1128/mcb.11.2.1107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Verdi J M, Bashirullah A, Goldhawk D E, Kubu C J, Jamali M, Meakin S O, Lipshitz H D. Distinct human NUMB isoforms regulate differentiation vs. proliferation in the neuronal lineage. Proc Natl Acad Sci USA. 1999;96:10472–10476. doi: 10.1073/pnas.96.18.10472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Weissinger E M, Eissner G, Grammer C, Fackler S, Haefner B, Yoon L S, Lu K S, Bazarov A, Sedivy J M, Mischak H, Kolch W. Inhibition of the Raf-1 kinase by cyclic AMP agonists causes apoptosis of v-abl-transformed cells. Mol Cell Biol. 1997;17:3229–3241. doi: 10.1128/mcb.17.6.3229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wen S T, Van Etten R A. The PAG gene product, a stress-induced protein with antioxidant properties, is an Abl SH3-binding protein and a physiological inhibitor of c-Abl tyrosine kinase activity. Genes Dev. 1997;11:2456–2467. doi: 10.1101/gad.11.19.2456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yang S S, Van Aelst L, Bar-Sagi D. Differential interactions of human Sos1 and Sos2 with Grb2. J Biol Chem. 1995;270:18212–18215. doi: 10.1074/jbc.270.31.18212. [DOI] [PubMed] [Google Scholar]
- 35.York R D, Yao H, Dillon T, Ellig C L, Eckert S P, McCleskey E W, Stork P J. Rap1 mediates sustained MAP kinase activation induced by nerve growth factor. Nature. 1998;392:622–626. doi: 10.1038/33451. [DOI] [PubMed] [Google Scholar]
- 36.Ziemnicka-Kotula D, Xu J, Gu H, Potempska A, Kim K S, Jenkins E C, Trenkner E, Kotula L. Identification of a candidate human spectrin Src homology 3 domain-binding protein suggests a general mechanism of association of tyrosine kinases with the spectrin-based membrane skeleton. J Biol Chem. 1998;273:13681–13692. doi: 10.1074/jbc.273.22.13681. [DOI] [PubMed] [Google Scholar]
- 37.Zwartkruis F J, Wolthuis R M, Nabben N M, Franke B, Bos J L. Extracellular signal-regulated activation of Rap1 fails to interfere in Ras effector signalling. EMBO J. 1998;17:5905–5912. doi: 10.1093/emboj/17.20.5905. [DOI] [PMC free article] [PubMed] [Google Scholar]