Abstract
OBJECTIVES
Recent data suggested that off-pump coronary artery bypass (OPCAB) may carry a higher risk for mortality in the long term when compared to on-pump coronary artery bypass (ONCAB). We, therefore, compared long-term survival and morbidity in patients undergoing ONCAB versus OPCAB in a large single-centre cohort.
METHODS
A total of 8981 patients undergoing isolated elective/urgent coronary artery bypass grafting between January 2009 and December 2019 were analysed. Patients were stratified into 2 groups (OPCAB n = 6649/ONCAB n = 2332). The primary end point was all-cause mortality. Secondary endpoints included repeat revascularization, stroke and myocardial infarction. To adjust for potential selection bias, 1:1 nearest neighbour propensity score (PS) matching was performed resulting in 1857 matched pairs. Moreover, sensitivity analysis was applied in the entire study cohort using multivariable- and PS-adjusted Cox regression analysis.
RESULTS
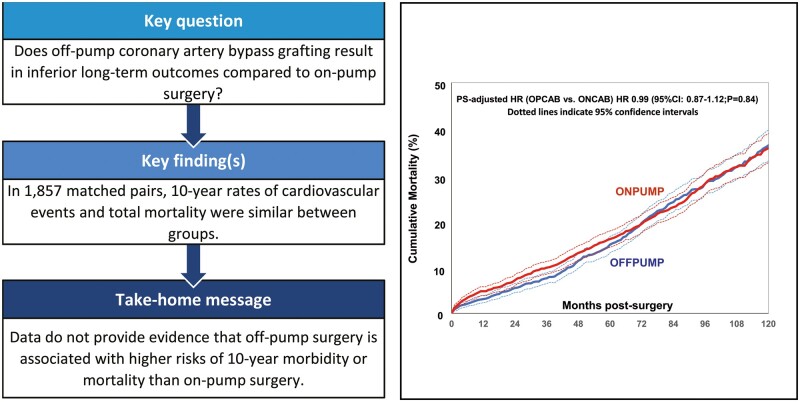
In the PS-matched cohort, 10-year mortality was similar between study groups [OPCAB 36.4% vs ONCAB 35.8%: hazard ratio (HR) 0.99, 95% confidence interval (CI) 0.87–1.12; P = 0.84]. While 10-year outcomes of secondary endpoints did not differ significantly, risk of stroke (OPCAB 1.50% vs ONCAB 2.8%: HR 0.51, 95% CI 0.32–0.83; P = 0.006) and mortality (OPCAB 3.1% vs ONCAB 4.8%: HR 0.65, 95% CI 0.47–0.91; P = 0.011) at 1 year was lower in the OPCAB group. In the multivariable- and the PS-adjusted model, mortality at 10 years was not significantly different (OPCAB 34.1% vs ONCAB 35.7%: HR 0.97, 95% CI 0.87–1.08; P = 0.59 and HR 1.01, 95% CI 0.90–1.13; P = 0.91, respectively).
CONCLUSIONS
Data do not provide evidence that elective/urgent OPCAB is associated with significantly higher risks of mortality, repeat revascularization, or myocardial infarction during late follow-up when compared to ONCAB. Patients undergoing OPCAB may benefit from reduced risks of stroke and mortality within the first year postoperatively.
Keywords: Coronary artery disease, Coronary artery bypass grafting, Myocardial revascularization, Off-pump coronary artery bypass grafting, Off-pump surgery
By avoiding potentially deleterious adverse effects associated with the use of extracorporeal circulation and aortic cross clamping, the introduction of off-pump coronary artery bypass (OPCAB) has been thought to overcome some of the limitations of conventional on-pump coronary artery bypass (ONCAB).
INTRODUCTION
By avoiding potentially deleterious adverse effects associated with the use of extracorporeal circulation and aortic cross clamping, the introduction of off-pump coronary artery bypass (OPCAB) has been thought to overcome some of the limitations of conventional on-pump coronary artery bypass (ONCAB). However, there is an ongoing controversial debate about the potential risks and benefits of OPCAB when compared to ONCAB [1, 2]. While short- and mid-term outcomes of both techniques seem to be comparable or even favour OPCAB [3–5], the results of several recent studies suggest that OPCAB may confer inferior outcomes in the long-term [2, 6–9]. However, other studies did not report significant differences [10–12]. Recently, 5-year results of 3 large randomized controlled trials (RCTs), namely the ROOBY (Randomized On/Off Bypass), the CORONARY (CABG Off- or On-Pump Revascularization Study), as well as the GOPCABE (German Off-Pump Coronary Artery Bypass Grafting in Elderly Patients) trial became available [13–15]. While the CORONARY trial did not show a significant difference, the ROOBY trial revealed poorer survival in the OPCAB group.
There is an ongoing debate as to whether OPCAB is associated with inferior long‐term outcomes. Therefore, we sought to compare long-term survival and morbidity in patients undergoing ONCAB versus OPCAB in a large single-centre propensity-matched cohort of patients.
PATIENTS AND METHODS
Ethical statement
The study was approved by the ethics committee of the Ruhr-University-Bochum, Germany, on 20 April 2020 (AZ 2020-628). The requirement for written informed consent was waived.
Patient population and study design
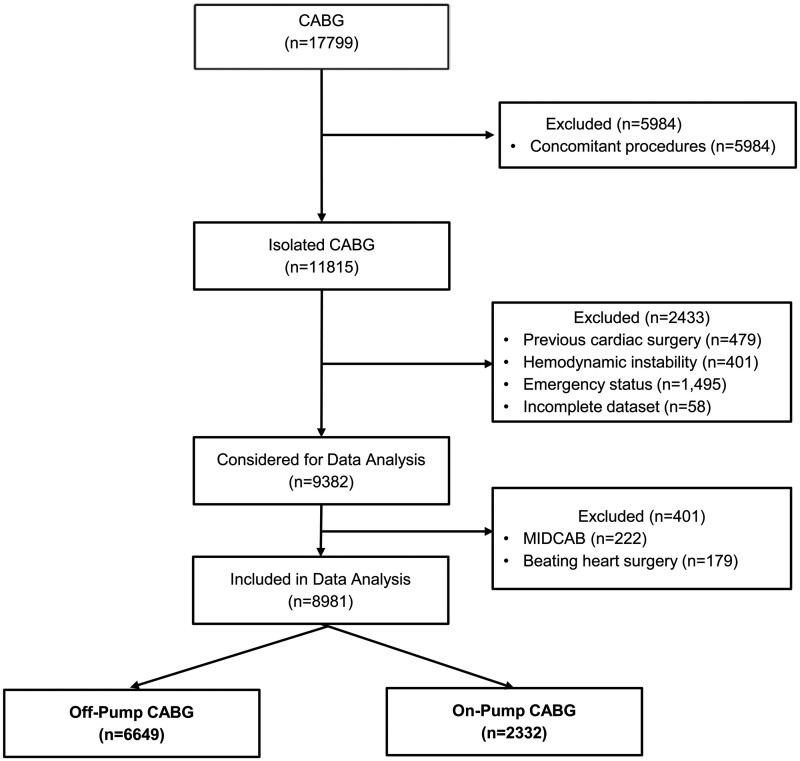
This cohort analysis included patients with coronary artery disease who underwent isolated OPCAB and ONCAB at our institution between January 2009 and December 2019. Patients with concomitant or any previous cardiac surgery, haemodynamic instability (preoperative cardiogenic shock, cardiopulmonary resuscitation), emergency status or patients undergoing minimally invasive direct coronary artery bypass grafting (MIDCAB) as well as beating heart procedures were excluded. As depicted in Fig. 1, a total of 8981 patients were finally included in our data analysis (OPCAB = 6649; ONCAB = 2332). Preoperative, intraoperative, and postoperative data were prospectively recorded in a dedicated database on a routine basis. Clinical data were obtained from a database using the cardiac surgery acquisition program THGQIMS (Münster, Germany). Biochemical parameters were obtained from Lauris (SWISSLAB, Berlin, Germany). The study was performed in accordance with the STrengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (www.strobe-statement.org).
Figure 1:
Study flow diagram. CABG: coronary artery bypass grafting; MIDCAB: minimally invasive direct coronary artery bypass grafting.
Surgical technique and general postoperative management
Surgical technique has been previously described elsewhere [16]. Both groups received 500 mg aspirin plus heparin intravenously, aiming for an activated partial thromboplastin time of 40–60 s. On the first postoperative day, all patients were placed on 100 mg aspirin orally plus weight-adjusted subcutaneous low-molecular-weight heparin. Patients with central vein graft anastomoses completed with the PAS-Port device were additionally placed on 75 mg of daily clopidogrel for 6 months.
Completeness of revascularization
Completeness of revascularization was determined by a simplified revascularization ratio, an index previously described and used by others [6, 17]. Briefly, the revascularization ratio was determined between the number of distal anastomoses and the number of diseased coronaries. Incomplete revascularization was defined as a greater number of diseased territories than the number of grafts placed (revascularization ratio < 1).
Clinical end points and follow-up
The primary endpoint was mortality of any cause up to a maximum follow-up of 10 years. Mortality was assessed by using the following sources of information: a regular review of medical records repeated contacts with the participants, contacts with family physicians, and consultation of the respective registration office in case of missing post-discharge forms. Secondary endpoints were freedom from myocardial infarction, stroke, or repeat revascularization during follow-up. Myocardial infarction was considered to have occurred in cases of new persistent ST-segment changes in combination with a rise in cardiac troponin values (perioperative: hs-troponin I > 10 000 ng/ml, troponin I > 10 mg/l; after discharge: hs-troponin I > 500 ng/ml, troponin I > 0.5 mg/l) and/or imaging evidence of new regional wall motion abnormalities. All suspected myocardial infarctions were validated by coronary angiography. A stroke of haemorrhagic or thrombotic origin was considered present when a clinically manifest motoric, sensory, or cognitive neurological deficit was recorded due to a cerebrovascular event. Transient ischaemic events were not included. Repeat revascularization was defined as postoperative percutaneous coronary intervention or redo coronary artery bypass grafting (CABG) during follow-up. Secondary clinical endpoints were assessed by the same sources used to identify the primary endpoint (exception: registration office). The completeness of follow-up was calculated according to Wu et al. [18].
Statistical analysis
Categorical variables are summarized as percentages and the number of observations. Preoperative and perioperative continuous variables are presented as means and standard deviation (SD). Patients who had undergone unplanned conversion to ONCAB (n = 133) or OPCAB (n = 8) were analysed according to the intention-to-treat principle.
Because of the non-randomized group assignment, we performed a matched propensity score (PS) analysis to assess treatment effects. The propensity matching score was estimated by multivariable logistic regression. In the regression model, the study group was the dependent variable. All parameters used in the study by Chikwe et al. [6] were also selected as independent variables for PS matching in the present study (Table 1), except liver failure and cancer. These 2 latter parameters are not routinely assessed at our institution since they are very rarely seen in our CABG patients. Instead, total arterial revascularization was also included as the independent variable for PS matching. Matching was performed using a 1:1 ratio with the logit-transformed PS. For this, an optimal-matching algorithm with a caliper width of 0.1 SD from the linear predictor was used. The balance of risk factors was judged by standardized differences. The balance was considered to be satisfactory when the standardized difference is <10%.
Table 1:
Patient characteristics before and after PS matching
| Parameter | All patients (n = 8840) |
PS-matched pairs (n = 3758) |
||||
|---|---|---|---|---|---|---|
| Off-pump (n = 6649) | On-pump (n = 2332) | Std Diff (%) | Off-pump (n = 1857) | On-pump (n = 1857) | Std Diff (%) | |
| Age (years) | 67.3 ± 9.7 | 67.6 ± 9.3 | −3.2 | 68.0 ± 9.4 | 67.8 ± 9.4 | 2.1 |
| Gender, males (%) | 5389 (81.0) | 1827 (78.3) | 9.2 | 1446 (77.9) | 1457 (78.5) | 2.0 |
| Urgent operation (%) | 287 (4.3) | 312 (13.4) | −36.4 | 131 (7.1) | 149 (8.0) | −4.4 |
| Comorbidities | ||||||
| Body mass index (kg/m2) | 28.4 ± 4.5 | 28.4 ± 4.6 | 0.0 | 28.3 ± 4.5 | 28.4 ± 4.6 | −2.2 |
| Previous myocardial infarction | ||||||
| No history | 4976 (74.8) | 1607 (68.9) | 17.9 | 1306 (70.3) | 1340 (72.2) | −6.0 |
| 0–23 h | 78 (1.2) | 24 (1.0) | 2.0 | 21 (1.1) | 21 (1.1) | 0.0 |
| 1–7 days | 189 (2.8) | 41 (1.8) | 8.5 | 46 (2.5) | 27 (1.5) | 7.7 |
| 8–21 days | 242 (3.6) | 156 (6.7) | −16.4 | 104 (5.6) | 113 (6.1) | −2.7 |
| >21 days | 1164 (17.5) | 504 (20.5) | −13.8 | 380 (20.5) | 356 (19.2) | 4.6 |
| Previous PCI | 1899 (28.6) | 648 (27.8) | 2.5 | 490 (26.4) | 488 (26.3) | 0.3 |
| NYHA functional class | ||||||
| I–II | 4397 (66.1) | 1295 (55.5) | 29.9 | 1092 (58.4) | 1101 (59.3) | −2.6 |
| III | 2111 (31.7) | 961 (41.2) | −27.0 | 706 (38.0) | 709 (38.2) | −0.6 |
| IV | 141 (2.1) | 76 (3.3) | −7.1 | 59 (3.2) | 47 (2.5) | 5.3 |
| Previous stroke | 215 (3.2) | 90 (3.9) | −4.6 | 69 (3.7) | 72 (3.9) | −1.3 |
| Renal disease | ||||||
| No renal disease | 6507 (97.9) | 2260 (96.9) | 8.1 | 1806 (97.3) | 1806 (97.3) | 0.0 |
| Dialysis-dependent | 69 (1.0) | 44 (1.9) | −7.6 | 22 (1.2) | 31 (1.7) | −4.3 |
| Creatinine > 2.3 mg/dl | 73 (1.1) | 28 (1.2) | −1.0 | 29 (1.6) | 20 (1.1) | 4.9 |
| Peripheral vascular disease | 786 (11.8) | 311 (13.3) | −6.0 | 240 (12.9) | 239 (12.9) | 0.0 |
| Diabetes mellitus (%) | ||||||
| Non-diabetic | 4322 (65.0) | 1496 (64.2) | 2.3 | 1211 (65.2) | 1187 (63.9) | 3.8 |
| Diabetes, insulin-dependent | 1348 (20.3) | 468 (20.1) | 0.7 | 376 (20.2) | 375 (20.2) | 0.0 |
| Diabetes, non-insulin-dependent | 979 (14.7) | 368 (15.8) | −4.1 | 270 (14.5) | 295 (15.9) | −5.3 |
| Smoking history | ||||||
| No history of smoking | 3867 (58.2) | 1358 (58.2) | 0.0 | 1079 (58.1) | 1086 (58.5) | −1.1 |
| Current smoker (≤1 month) | 1099 (16.5) | 374 (16.0) | 1.9 | 269 (14.5) | 297 (16.0) | −5.6 |
| Previous smoker (>1 month) | 1683 (25.3) | 600 (25.7) | −1.3 | 509 (27.1) | 474 (26.0) | 6.0 |
| COPD (%) | ||||||
| None | 6209 (93.4) | 2099 (90.0) | 15.9 | 1685 (90.7) | 1698 (91.4) | −3.5 |
| Mild | 147 (2.2) | 74 (3.2) | −7.0 | 50 (2.7) | 49 (2.6) | 0.8 |
| Moderate or severe | 293 (4.4) | 159 (6.8) | −12.6 | 122 (6.6) | 110 (5.9) | 3.9 |
| Arterial hypertension (%) | 5763 (86.7) | 1979 (84.9) | 7.1 | 1580 (85.1) | 1586 (85.4) | −1.2 |
| Atrial fibrillation (%) | 292 (4.4) | 140 (6.0) | −8.8 | 108 (5.8) | 102 (5.5) | 1.7 |
| Cardiac function | ||||||
| Ejection fraction (%) | ||||||
| <30 | 82 (1.2) | 90 (3.9) | −17.6 | 49 (2.6) | 54 (2.9) | −2.2 |
| 30–49.9 | 1024 (15.4) | 460 (19.7) | −14.9 | 341 (18.4) | 331 (17.8) | 2.2 |
| ≥50 | 5543 (83.4) | 1782 (76.4) | 23.2 | 1467 (79.0) | 1472 (79.3) | −1.0 |
| Number of diseased vessels | ||||||
| Left main stenosis >50% | 2467 (37.1) | 916 (39.3) | −6.3 | 700 (37.7) | 686 (36.9) | −2.3 |
| 1–2 | 1142 (17.2) | 384 (16.5) | 2.6 | 301 (16.2) | 298 (16.0) | 0.8 |
| 3 | 5507 (82.8) | 1948 (83.5) | −2.7 | 1556 (83.8) | 1559 (84.0) | −0.8 |
| Surgeon’s experience | ||||||
| <67 surgeries during the last year | 3326 (50.0) | 1136 (48.7) | 3.6 | 786 (42.3) | 797 (42.9) | −1.7 |
| ≥67 surgeries during the last year | 3323 (50.0) | 1196 (51.3) | −3.6 | 1071 (57.4) | 1060 (57.1) | 1.7 |
| Era | ||||||
| 2009–2014 | 3296 (49.6) | 1953 (83.7) | −127.8 | 1496 (80.6) | 1478 (79.6) | −3.5 |
| 2015–2019 | 3353 (50.4) | 379 (16.3) | 126.7 | 361 (19.4) | 379 (20.4) | 3.5 |
| Total arterial revascularization (%) | 1789 (26.9) | 180 (7.7) | 95.8 | 187 (10.1) | 177 (9.5) | 2.8 |
Values are mean ± SD or n (%).
COPD: chronic obstructive pulmonary disease; NYHA: New York Heart Association; PCI: percutaneous coronary intervention; PS: propensity score; SD: standard deviation; Std Diff: standardized difference.
We compared continuous clinical data and 30-day clinical outcomes between study groups by using the paired t-test and the McNemar test, where appropriate. We generated Kaplan–Meier estimates by study group for the primary endpoint and also for the secondary clinical endpoints. We used Cox proportional hazards models, stratified on the matched pairs, for data analysis. The proportionality of hazard assumption was tested by evaluation of time-dependent variables, which were the cross-products of the predictor variables with event-free outcomes and was satisfied for all endpoints (P > 0.05). In subgroup investigations, we tested the interaction between the study group and various baseline characteristics on the primary endpoint. Tests for interaction between study group, subgroups, and clinical outcome were based on the Wald test for the interaction term (study group × surgical group), with both off-pump/on-pump surgery and respective subgroup as categorical variables. In each subgroup, the balance of baseline characteristics between OPCAB and ONCAB patients was evaluated. When the standardized difference was >10%, the covariate was included in the Cox proportional hazards model. For long-term outcomes, we present hazard ratios (HRs, considering the total number of events and also the timing of each event) and for 30-day outcomes, we present risk ratios (total number of events by the end of follow-up). In both cases, 95% confidence intervals (CIs) are also given. We also performed a sensitivity analysis in the entire study cohort using multivariable-adjusted and PS-adjusted Cox regression analysis. We included those covariates in the multivariable analysis which were also used for PS matching. A statistical power calculation revealed that the study would have 90% power to detect a relative difference of 18% in the primary endpoint in the on-pump group compared with the off-pump group, given a total sample size of 3500 (1850 in each group), a 30% incidence of the primary endpoint, and using a two-sided alpha level of 0.05. P-values <0.05 were considered statistically significant. We used the statistical software package IBM SPSS, version 24 (IBM Corp, Armonk, NY, USA), R (version 2.15.3), and the SPE file in SPSS to perform the analyses.
RESULTS
Baseline characteristics
Out of the cohort of 8981 patients, PS matching was possible in 1857 pairs (Table 1). In the PS-matched groups, all standardized differences in preoperative covariates were <10%. The numbers of diseased vessels were similar in the OPCAB and the ONCAB group (2.82 ± 0.44 and 2.82 ± 0.42, respectively; P = 0.62). Since 2009, more and more off-pump surgeries have been performed at the expense of on-pump surgeries. OPCAB is now the preferred surgical technique at our institution (Supplementary Material, Fig. S1). In 68.0% of patients in the OPCAB group, the PAS-Port device was used for proximal graft anastomosis. In the OPCAB and ONCAB groups, the conversion rate was 3.23% (n = 60) and 0.43% (n = 8), respectively.
Primary end point
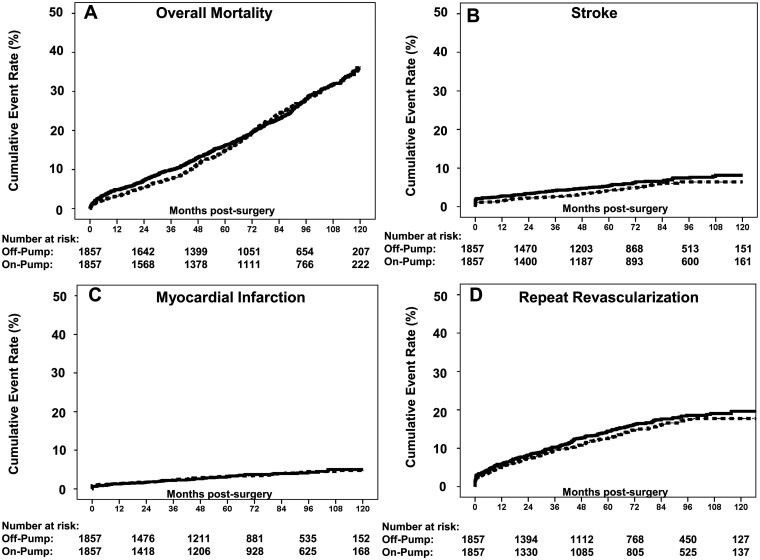
In the unmatched/unadjusted sample, OPCAB was associated with lower 10-year mortality than after ONCAB (34.1% vs 35.7%; HR 0.87, 95% CI 0.79–0.97; P = 0.008; Supplementary Material, Fig. S2). However, as mentioned above, baseline characteristics of patients in the OPCAB and the ONCAB group were considerably different. To address this issue, a 1:1 nearest neighbour PS matching analysis was performed for covariate risk adjustment (Table 1). Median follow-up in the propensity-matched cohort was 6.76 and 6.24 years in the OPCAB group and 7.02 years in the ONCAB group. The 3714 patients accrued 22 567 patient-years of observation, 11 227 in the OPCAB group and 11 339 in the ONCAB group. The completeness of follow-up was 95.0%. Thirty-day mortality did not differ significantly between study groups (OPCAB 1.0% vs ONCAB 1.2%: HR 0.82, 95% CI 0.44–1.53; P = 0.63). During follow-up, 917 patients died, 469 in the off-pump and 448 in the on-pump group (Fig. 2A). At 1 year, mortality was significantly lower after OPCAB (OPCAB 3.1% vs ONCAB 4.8%: HR 0.65, 95% CI 0.47–0.91; P = 0.011). Ten-year mortality was similar between study groups (OPCAB 36.4% vs ONCAB 35.8%: HR 0.99, 95% CI 0.87–1.12; P = 0.84; Fig. 2A).
Figure 2:
Cumulative event rate of overall mortality (A), stroke (B), myocardial infarction (C) and repeat revascularization (D). Solid line, on-pump group; dotted line, off-pump group.
Subgroup analysis
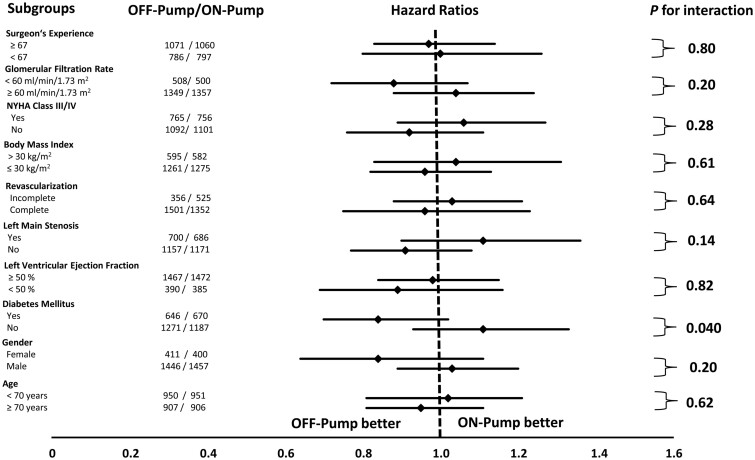
Subgroup analysis showed significant interactions of the surgical group with diabetes mellitus, but not with other patient characteristics (Fig. 3). However, the data also showed that there was no general survival benefit in either of the 2 surgical groups.
Figure 3:
Hazard ratios for mortality in off-pump surgery compared with on-pump surgery according to various subgroups. NYHA: New York Heart Association.
Secondary endpoints
At 30 days, the incidence of stroke was significantly lower in the OPCAB group. Incidences of myocardial infarction and repeat revascularization were similar between study groups (Supplementary Material, Table S1). At 1 year, risk for stroke and repeat revascularization was 1.5% vs 2.8% (HR 0.51, 95% CI 0.32–0.83; P = 0.006) and 5.1% vs 4.8% (HR 0.87, 95% CI 0.65–1.15; P = 0.33), respectively. OPCAB was not associated with significantly lower risks of stroke (OPCAB 6.4% vs ONCAB 8.1%: HR 0.76, 95% CI 0.57–1.02; P = 0.065) and repeat revascularization (OPCAB 17.7% vs ONCAB 19.6%: HR 0.90, 95% CI 0.75–1.08; P = 0.24) at 10 years, respectively. At 1 year and 10 years risk of myocardial infarction did not differ significantly between study groups (OPCAB 1.3% vs ONCAB 1.3%: HR 1.08, 95% CI 0.61–1.93; P = 0.79 at 1 year and OPCAB 4.7% vs ONCAB 5.0%: HR 1.01, 95% CI 0.70–1.44; P = 0.98 at 10 years). Figure 2B–D illustrates the results of secondary endpoint analysis by the study group.
Sensitivity analysis
In the multivariable-adjusted model applied to the entire patient population, mortality at 10 years was not significantly different between treatment groups (OPCAB 34.1% vs ONCAB 35.7%: HR 0.97, 95% CI 0.87–1.08; P = 0.59). Multivariable-adjusted 10-year risks of stroke became significant with an HR for the off-pump versus the on-pump group of 0.75 (95% CI 0.59–0.95; P = 0.015). Multivariable-adjusted HRs of myocardial infarction and repeat revascularization for the off-pump versus on-pump group at 10 years were 0.97 (95% CI 0.72–1.31; P = 0.84) and 0.88 (95% CI 0.75–1.02; P = 0.08), respectively. Supplementary Material, Fig. S3 shows the results of the multivariable-adjusted and PS-adjusted models. In the PS-adjusted model applied to the entire patient population, mortality at 10 years was also not significantly different between treatment groups HR 1.01 (95% CI 0.90–1.13; P = 0.91). Supplementary Material, Fig. S4 summarizes the results for different primary endpoint analyses.
Completeness of revascularization
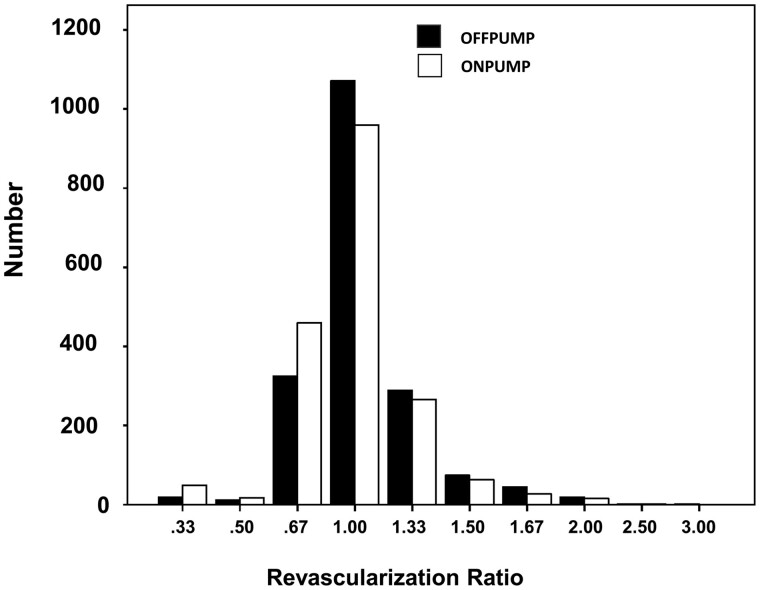
The mean number of distal anastomoses were slightly but significantly lower in the ONCAB group than in the OPCAB group (OPCAB 2.88 ± 0.80 vs ONCAB 2.74 ± 0.83, P < 0.001), and so was the mean numbers of anastomoses to the posterolateral and inferior wall (OPCAB 1.95 ± 1.06 vs ONCAB 1.65 ± 1.03, P < 0.001). The mean revascularization ratio was 1.03 ± 0.28 in the OPCAB and 0.99 ± 0.29 in the ONCAB group (P < 0.001). Figure 4 shows the distribution of individual revascularization ratios by the group. In the OPCAB group 356 patients (19.2%), and in the ONCAB group 526 patients (28.3%) had a revascularization ratio <1.0, respectively (P < 0.001).
Figure 4:
Revascularization ratio by study group.
DISCUSSION
In this risk-adjusted analysis of a large single-centre cohort of isolated elective/urgent CABG patients, 10-year mortality was similar between study groups. Moreover, 10-year outcomes of secondary endpoints did not differ significantly between study groups, whereas 1-year mortality and 1-year risk of stroke were significantly lower in the OPCAB than in the ONCAB group.
A meta‐analysis of 35 PS‐matched studies [4] indicates that OPCAB is superior to ONCAB surgery with regard to short-term outcomes including death. Similarly, a meta-analysis of RCTs reported a significantly lower risk of postoperative stroke in patients allocated to OPCAB versus ONCAB [19]. Our data also indicate beneficial OPCAB effects on short-term clinical outcomes such as the 1-year risk of stroke and 1-year mortality. Mechanisms of the surgery-related risk of stroke may include systemic inflammatory response, a dysregulated coagulation system, myocardial stunning, hypoperfusion, atrial fibrillation, and cerebral micro-and macro-embolization. The latter 2 are at least in part due to extracorporeal circulation and aortic manipulation [16]. The short-term risk of stroke may also contribute to short-term mortality risk.
There is, however, an ongoing debate as to whether OPCAB is associated with inferior long‐term outcomes. It is important to note, that for evaluation of survival at 10 years, only observational data were available. In a meta‐analysis by Filardo et al. [7] including 42 RCTs and 31 risk‐adjusted observational studies and a total of ∼1.2 million patients, OPCAB was associated with a statistically significant 10% relative increase in the probability of mortality at 5 years, which further increased to 14% at 10 years in observational studies. It was concluded that evidence from RCTs showed no differences between the techniques, whereas rigorously adjusted observational studies and the combined analysis suggested that OPCAB offers lower short-term mortality but poorer long-term survival. In another recent meta‐analysis including only RCTs with ≥4‐year outcome enrolling 8145 participants, Smart et al. reported a significantly higher odds ratio for long‐term mortality of 1.16 for OPCAB [2]. A recent analysis from the New Jersey cardiac surgery registry by Chikwe et al. [6] supports these earlier data. After PS matching, 3975 pairs of OPCAB and CABG patients were finally studied [6]. Results indicated an adjusted HR of 1.11 for mortality at 10 years. It should be noted that we applied the same covariates for PS matching that were used by Chikwe et al. [6], but did not observe a significant difference in 10-year mortality between OPCAB and ONCAB. The most obvious explanation for the differences between their study and our study points towards the aspect of incomplete revascularization. The authors found that incomplete revascularization was more common with OPCAB. Incomplete revascularization was associated with increased long-term mortality. In our series, the number of distal anastomoses, the number of grafts to the lateral and inferior wall, as well as the revascularization ratio in the OPCAB group were slightly but significantly higher in the OPCAB than in the ONCAB group. Accordingly, the incidence of incomplete revascularization, defined as a revascularization ratio of <1, was significantly lower in our OPCAB group. This may at least in part explain the similar risks of 10-year mortality and repeat revascularization in our OPCAB and ONCAB groups. Our results of similar numbers of grafts and similar long-term mortality between study groups are supported by a very recent PS-matched data analysis in octogenarians [20].
The slightly higher mean number of distal anastomoses (absolute difference 0.14) may reflect the high experience in the OPCAB techniques at our centre. A higher index of completeness of revascularization has been associated with improved long-term survival irrespective of the used CABG technique [17]. A lower number of grafts is inevitably associated with incomplete revascularization. In a recent subgroup analysis of the randomized multicentre EXCEL trial, off-pump surgery was associated with a two-fold increase in all-cause mortality at 3 years compared with on-pump surgery despite a comparable extent and complexity of coronary disease between the 2 groups. The most probable explanation for this observation was that the performance of off-pump surgery was associated with a significantly lower rate of grafting of the left circumflex and right coronary artery [21]. Moreover, in the GOPCABE trial, incomplete revascularization was associated with a lower 5-year survival rate, irrespective of the type of surgery [13]. This points to the issue of the surgeońs experience. Higher rates of incomplete revascularization have been linked to surgeońs inexperience in performing OPCAB surgery [22]. The median pre-study OPCAB experience of the surgeons who had participated in the GOPCABE trial was 322 procedures, >100 procedures in the CORONARY trial and a median of only 50 off-pump procedures in the ROOBY trial. In the ROOBY trial, OPCAB was associated with a significantly higher rate of incomplete revascularization and reduced survival. Conversely, in CORONARY and GOPCABE, the number of grafts and the incidence of incomplete revascularization between off-pump and on-pump surgery were comparable, and so were 5-year mortality rates. Conversion rates also indicate OPCAB experience and decline with increasing OPCAB surgeon and institutional experience [1]. In a large meta-analysis, the authors demonstrated that a difference in survival in favour of ONCAB is found only in the studies with a crossover rate of ≥10% [8]. In a recent post hoc analysis from the ART (Arterial Revascularization Trial), off-pump surgery performed by low-volume off-pump surgeons was associated with a significantly lower number of grafts, increased conversion rates, and increased cardiovascular death when compared with on-pump surgery performed by on-pump-only surgeons. In contrast, if the conversion rate was low, and the number of grafts was similar, 10-year mortality was comparable [23]. OPCAB-to-ONCAB conversion rate in our series was 3.2%, which can be considered low and seems to reflect the high experience with OPCAB at our centre. Since on-pump or off-pump surgery was associated with similar 10-year outcomes in our patients and our data indicate similar outcomes in various subgroups, off-pump and on-pump procedures seem to be equally effective regarding long-term clinical outcomes.
Limitations
The major limitation of the present analysis is its retrospective and non-randomized design which certainly limits the generalizability of our results. PS-based and multivariable adjustment cannot exclude that unmeasured confounding is still present. Some may also argue that only preoperative parameters should be included in the PS model. However, since arterial revascularization (i) is an important predictor of long-term clinical outcome, (ii) is largely unaffected by perioperative complications, and (iii) is also used as an inclusion criterion in randomized trials, we decided to include this parameter in our PS model. However, it is a strength of this single-centre analysis that it was performed in a high-volume centre with well-experienced surgeons proficient in performing both ONCAB and OPCAB resulting in low conversion rates. Other strengths are different methods of risk adjustment, a large number of patients and a median follow-up of >6.5 years in the PS-matched study cohort.
CONCLUSIONS
Our PS-matched, as well as PS-adjusted and multivariable-adjusted data, do not indicate that elective/urgent OPCAB is associated with higher risks of mortality, repeat revascularization or myocardial infarction during late follow-up of up to 10 years when compared to ONCAB. Patients undergoing OPCAB may benefit from reduced risks of stroke and mortality within the first 12 months post-surgery. Additionally, OPCAB when compared to ONCAB was not associated with higher rates of incomplete revascularization. Future studies evaluating long-term outcomes after CABG should prospectively analyse the completeness of revascularization and surgeons‘ experience with the respective operative technique. Patients’ clinical and anatomic characteristics should be identified to guide the choice of operative techniques and graft selection to further improve outcomes in patients treated with CABG.
SUPPLEMENTARY MATERIAL
Supplementary material is available at ICVTS online.
Supplementary Material
ACKNOWLEDGEMENTS
The authors thank all members of the ZIM (Zentrum für Informationsmanagement) for their relentless support.
Conflict of interest: none declared.
ABBREVIATIONS
- CABG
Coronary artery bypass grafting
- CI
Confidence interval
- HR
Hazard ratio
- ONCAB
On-pump coronary artery bypass
- OPCAB
Off-pump coronary artery bypass
- PS
Propensity score
- RCT
Randomized controlled trial
- SD
Standard deviation
Author contributions
Marcus-André Deutsch: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Writing—original draft. Armin Zittermann: Conceptualization; Formal analysis; Methodology; Visualization; Writing—original draft. André Renner: Writing—review & editing. René Schramm: Writing—review & editing. Julia Götte: Writing—review & editing. Jochen Börgermann: Writing—review & editing. Henrik Fox: Writing—review & editing. Sebastian V. Rojas: Writing—review & editing. Takayuki Gyoten: Writing—review & editing. Michiel Morshuis: Writing—review & editing. Andreas Koster: Writing—review & editing. Nikolai Hulde: Writing—review & editing. Dennis Hinse: Data curation; Writing—review & editing. Kavous Hakim-Meibodi: Writing—review & editing. Jan F. Gummert: Conceptualization; Investigation; Resources; Writing—review & editing.
Reviewer information
Reviewer information Interactive CardioVascular and Thoracic Surgery thanks Ari Mennander, Antonio Miceli and the other, anonymous reviewer(s) for their contribution to the peer review process of this article.
REFERENCES
- 1. Gaudino M, Angelini GD, Antoniades C, Bakaeen F, Benedetto U, Calafiore AM. et al. Off-Pump coronary artery bypass grafting: 30 years of debate. J Am Heart Assoc 2018;7:e009934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Smart NA, Dieberg G, King N.. Long-term outcomes of on- versus off-pump coronary artery bypass grafting. J Am Coll Cardiol 2018;71:983–91. [DOI] [PubMed] [Google Scholar]
- 3. Kowalewski M, Pawliszak W, Malvindi PG, Bokszanski MP, Perlinski D, Raffa GM. et al. Off-pump coronary artery bypass grafting improves short-term outcomes in high-risk patients compared with on-pump coronary artery bypass grafting: meta-analysis. J Thorac Cardiovasc Surg 2016;151:60–77.e1–58. [DOI] [PubMed] [Google Scholar]
- 4. Kuss O, von Salviati B, Borgermann J.. Off-pump versus on-pump coronary artery bypass grafting: a systematic review and meta-analysis of propensity score analyses. J Thorac Cardiovasc Surg 2010;140:829–35, 35.e1–13. [DOI] [PubMed] [Google Scholar]
- 5. Polomsky M, He X, O’Brien SM, Puskas JD.. Outcomes of off-pump versus on-pump coronary artery bypass grafting: impact of preoperative risk. J Thorac Cardiovasc Surg 2013;145:1193–8. [DOI] [PubMed] [Google Scholar]
- 6. Chikwe J, Lee T, Itagaki S, Adams DH, Egorova NN.. Long-term outcomes after off-pump versus on-pump coronary artery bypass grafting by experienced surgeons. J Am Coll Cardiol 2018;72:1478–86. [DOI] [PubMed] [Google Scholar]
- 7. Filardo G, Hamman BL, da Graca B, Sass DM, Machala NJ, Ismail S.. Efficacy and effectiveness of on- versus off-pump coronary artery bypass grafting: a meta-analysis of mortality and survival. J Thorac Cardiovasc Surg 2018;155:172–79.e5. [DOI] [PubMed] [Google Scholar]
- 8. Gaudino M, Benedetto U, Bakaeen F, Rahouma M, Tam DY, Abouarab A. et al. Off- versus on-pump coronary surgery and the effect of follow-up length and surgeons' experience: a meta-analysis. J Am Heart Assoc 2018;7:e010034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Kim JB, Yun SC, Lim JW, Hwang SK, Jung SH, Song H. et al. Long-term survival following coronary artery bypass grafting: off-pump versus on-pump strategies. J Am Coll Cardiol 2014;63:2280–8. [DOI] [PubMed] [Google Scholar]
- 10. Kirmani BH, Brazier A, Sriskandarajah S, Alshawabkeh Z, Gurung L, Azzam R. et al. Long-term survival after off-pump coronary artery bypass grafting. Ann Thorac Surg 2016;102:22–7. [DOI] [PubMed] [Google Scholar]
- 11. Kirmani BH, Holmes MV, Muir AD.. Long-term survival and freedom from reintervention after off-pump coronary artery bypass grafting: a propensity-matched study. Circulation 2016;134:1209–20. [DOI] [PubMed] [Google Scholar]
- 12. Puskas JD, Kilgo PD, Lattouf OM, Thourani VH, Cooper WA, Vassiliades TA. et al. Off-pump coronary bypass provides reduced mortality and morbidity and equivalent 10-year survival. Ann Thorac Surg 2008;86:1139–46; discussion 46. [DOI] [PubMed] [Google Scholar]
- 13. Diegeler A, Borgermann J, Kappert U, Hilker M, Doenst T, Boning A. et al. Five-year outcome after off-pump or on-pump coronary artery bypass grafting in elderly patients. Circulation 2019;139:1865–71. [DOI] [PubMed] [Google Scholar]
- 14. Lamy A, Devereaux PJ, Prabhakaran D, Taggart DP, Hu S, Straka Z. et al. Five-year outcomes after off-pump or on-pump coronary-artery bypass grafting. N Engl J Med 2016;375:2359–68. [DOI] [PubMed] [Google Scholar]
- 15. Shroyer AL, Hattler B, Wagner TH, Collins JF, Baltz JH, Quin JA. et al. Five-year outcomes after on-pump and off-pump coronary-artery bypass. N Engl J Med 2017;377:623–32. [DOI] [PubMed] [Google Scholar]
- 16. Borgermann J, Hakim K, Renner A, Parsa A, Aboud A, Becker T. et al. Clampless off-pump versus conventional coronary artery revascularization: a propensity score analysis of 788 patients. Circulation 2012;126:S176–82. [DOI] [PubMed] [Google Scholar]
- 17. Lattouf OM, Thourani VH, Kilgo PD, Halkos ME, Baio KT, Myung R. et al. Influence of on-pump versus off-pump techniques and completeness of revascularization on long-term survival after coronary artery bypass. Ann Thorac Surg 2008;86:797–805. [DOI] [PubMed] [Google Scholar]
- 18. Wu Y, Takkenberg JJ, Grunkemeier GL.. Measuring follow-up completeness. Ann Thorac Surg 2008;85:1155–7. [DOI] [PubMed] [Google Scholar]
- 19. Afilalo J, Rasti M, Ohayon SM, Shimony A, Eisenberg MJ.. Off-pump vs. on-pump coronary artery bypass surgery: an updated meta-analysis and meta-regression of randomized trials. Eur Heart J 2012;33:1257–67. [DOI] [PubMed] [Google Scholar]
- 20. Knapik P, Hirnle G, Kowalczuk-Wieteska A, Zembala MO, Pawlak S, Hrapkowicz T. et al. Off-pump versus on-pump coronary artery surgery in octogenarians (from the KROK Registry). PLoS One 2020;15:e0238880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Benedetto U, Puskas J, Kappetein AP, Brown WM 3rd, Horkay F, Boonstra PW. et al. Off-pump versus on-pump bypass surgery for left main coronary artery disease. J Am Coll Cardiol 2019;74:729–40. [DOI] [PubMed] [Google Scholar]
- 22. Magee MJ, Hebert E, Herbert MA, Prince SL, Dewey TM, Culica DV. et al. Fewer grafts performed in off-pump bypass surgery: patient selection or incomplete revascularization? Ann Thorac Surg 2009;87:1113–18; discussion 18. [DOI] [PubMed] [Google Scholar]
- 23. Taggart DP, Gaudino Mf GS, Gray A, Lees B, Sajja LR. et al. Ten-year outcomes after off-pump versus on-pump coronary artery bypass grafting: insights from the Arterial Revascularization Trial. J Thorac Cardiovasc Surg 2020 Feb 19;S0022-5223(20)30427-X. doi: 10.1016/j.jtcvs.2020.02.035. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.