Abstract
OBJECTIVES
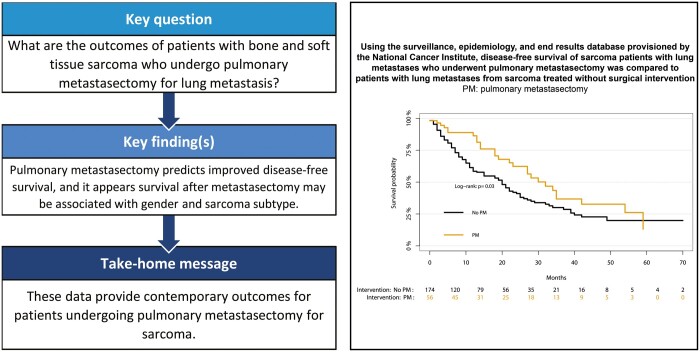
This study investigated the outcomes of sarcoma patients with lung metastases who underwent pulmonary metastasectomy (PM), compared to patients who underwent medical management alone. The secondary objective was to compare survival after PM between variables of interest.
METHODS
This was a retrospective review of 565 sarcoma patients with confirmed, isolated pulmonary metastasis identified from the Surveillance, Epidemiology and End Results database between 2010 and 2015. 1:4 propensity score matching was used to select PM and non-PM groups. The multivariable Cox proportional hazards model was used to analyse prognostic factors of disease-free survival (DFS).
RESULTS
Of the eligible 565 patients, 59 PM patients were matched to 202 non-PM patients in a final ratio of 3.4. After propensity matching, there were no significant differences in baseline characteristics between PM and non-PM patients. The median DFS after PM was 32 months (interquartile range 18–59), compared to 20 months (interquartile range 7–40) in patients without PM (P = 0.032). Using a multivariable Cox proportional hazards model, metastasectomy (hazard ratio 0.536, 95% confidence interval 0.33–0.85; P = 0.008) was associated with improved DFS. In a subset analysis of patients who underwent PM only, the median DFS was longer in males compared to females (P = 0.021), as well as in bone sarcoma compared to soft tissue sarcoma (P = 0.014).
CONCLUSIONS
For sarcoma patients with metastatic lung disease, PM appears to improve the prognosis compared to medical management. Furthermore, there may be a survival association with gender and tumour origin in patients who underwent PM. These data may be used to inform the surgical indications and eligibility criteria for metastasectomy in this setting.
Keywords: Sarcoma, Pulmonary metastasectomy, Survival, Prognosis, Outcomes
Sarcomas are a heterogeneous tumour group that accounts for 1% of adult malignancies [1].
INTRODUCTION
Sarcomas are a heterogeneous tumour group that accounts for 1% of adult malignancies [1]. These tumours have a propensity for lung metastasis, and patients with lung metastases have poor clinical outcomes. Surgery (metastasectomy) and chemotherapy are often utilized for the treatment of advanced sarcoma, and some data suggest metastasectomy alone is more cost-effective than chemotherapy in prolonging short-term survival in this population [2].
Five-year survival of patients undergoing pulmonary metastasectomy (PM) for sarcoma ranges from 15% to 50% [3–11]. However, there is no randomized evidence to support PM for metastatic sarcoma, and therefore the current indications rely on data derived mostly from single institutions and retrospective series. It has been proposed that PM affords favourable survival in patients with advanced bone and soft tissue sarcoma (STS) [11–14]. However, there is no consensus on the best way to select these patients as surgical candidates. In an attempt to address the role of PM for sarcoma, the current study reviewed the Surveillance, Epidemiology and End Results (SEER) database in order to identify patients with primary bone or STS and isolated lung metastases. After propensity matching, the primary objective was to determine whether an association exists between PM and disease-free survival (DFS). The secondary objective was to compare survival between variables of interest among patients who were treated with PM only.
MATERIALS AND METHODS
Selection
Ethical or institutional review board approval was not required for the completion of this study. The SEER database of the National Cancer Institute was queried from 2010 to 2015 [15]. Inclusion criteria were diagnostically confirmed bone or STS with lung metastases. Cases were first filtered by Histologic International Classification of Diseases for Oncology, Third Edition (ICD-O-3) codes: 8800–9059 and 9120–9269. Patients with synchronous or metachronous metastasis to the liver, bones and brain were excluded, leaving those with isolated metastasis only. For surgery, a distant site surgical procedure in the setting of isolated lung metastasis was presumed to indicate PM. Additional variables of interest were age, sex, race, primary tumour grade at diagnosis, primary tumour size, primary tumour histology and origin and T and N stage defined by the American Joint Committee on Cancer (AJCC) criteria [16]. Radiation (yes or no) and chemotherapy (yes or no/unknown) were recorded as dichotomous variables with respect to the treatment of the primary tumour and not the metastatic disease.
Statistical analyses
Propensity score matching was performed, and propensity scores were estimated by conditional backwards elimination multivariable logistic regression that incorporated potential predictors of PM group membership as independent variables. The entry value was P-value <0.020 and the remaining value was P-value <0.030, to maximize discrimination of the model between PM and non-PM patients (c-statistic, or area under receiving operating characteristics curve). These variables included diagnosis year, age, race, sex, primary tumour grade and size and T and N stage, which were each defined a priori as clinically meaningful. The propensity score represents the probability of surgical intervention in the study population. Each PM patient then was matched to up to 4 non-PM patients based on a greedy match algorithm [17]. PM patients with no matches (propensity score not within 0.1) were not included in the final grouping.
Categorical variables were described using frequencies and percentages, and continuous data (age and tumour size) were tested for normality and reported as the median with interquartile range (IQR). The standardized mean difference was used to measure the comparison of individual variables before and after matching. Disease-free survival (DFS) was estimated using Kaplan–Meier methods, and survival between variables of interest was compared using the log-rank test. DFS was defined as the survival in months from diagnosis until a cancer-specific death per the SEER database. Censoring patients were those alive without a cancer-specific death at last follow-up. The median follow-up time (months) was estimated using the reverse Kaplan–Meier method wherein survival is the event of interest and death is a censored event [18].
A multivariable Cox model was used to assess the association of individual variables with DFS. The final model was constructed using a backward stepwise elimination method of sequential variable exclusion with the highest P-value variable being excluded at each step, until only those with P-value <0.15 remained. Preselected variables were those of clinical interest and included diagnosis year, age, sex, race, sarcoma origin (bone or soft tissue), tumour size, grade and T stage, radiation and chemotherapy for the primary tumour and PM. Factors with <10 events per variable were excluded. Statistical significance was set to P-value <0.05, and all analyses were performed on SPSS version 26.0 (IBM, Armonk, NY, USA) and R-Studio version (RStudio: Integrated Development for R. RStudio, PBC, Boston, MA, USA).
RESULTS
Clinicopathological data with propensity score matching
Overall, 565 patients met the inclusion criteria. Before propensity matching, there were significant differences in median age between PM (n = 66) and non-PM (n = 499) groups [45 years (IQR 16–62) vs 52 years (IQR 18–63); P < 0.001] (Table 1). The propensity score model evaluated up to 8 co-variables related to baseline characteristics. The final model included 4 co-variables (age, size, grade and N stage) with a c-statistic of 0.71. Of the eligible 565 patients, 59 PM patients were matched to 202 non-PM patients in a final ratio of 3.4 (Table 1). Among the matched PM patients, metastasectomy was performed most commonly for advanced osteosarcoma (32.2%, n = 19), leiomyosarcoma (18.6%, n = 11) and chondrosarcoma (8.5%, n = 5) (Supplementary Material, Table S1). The majority of all matched PM and non-PM patients were treated with chemotherapy (neoadjuvant and/or adjuvant) (76.3%, n = 45 vs 74.8%, n = 151), respectively. However, after matching, a comparably higher proportion of patients in the PM group received radiation to the primary site compared to the non-PM group (39.0%, n = 23 vs 17.8%, n = 36), respectively. Trends in the yearly utilization of chemotherapy and radiation among all matched patients are shown in Supplementary Material, Fig. S1.
Table 1:
Baseline clinicopathological data of included patients
| Variables | All patients (n = 565) |
Propensity-matched (n = 261) |
||||
|---|---|---|---|---|---|---|
| PM (n = 66) | No-PM (n = 499) | SMD | PM (n = 59) | No-PM (n = 202) | SMD | |
| Age (years), median (IQR) | 45.0 (16–62) | 52.0 (18–63) | 0.571 | 45.0 (16–62) | 45 (20.8–63) | 0.114 |
| Sex, n (%) | 0.132 | 0.021 | ||||
| Male | 44 (66.7) | 301 (60.3) | 40 (67.8) | 135 (66.8) | ||
| Female | 22 (33.3) | 198 (39.7) | 19 (32.2) | 67 (33.2) | ||
| Race, n (%) | 0.112 | 0.114 | ||||
| White | 53 (80.3) | 389 (78.0) | 48 (81.4) | 155 (76.7) | ||
| Black | 9 (13.6) | 65 (13.0) | 7 (11.9) | 30 (14.9) | ||
| Other | 4 (6.1) | 45 (9.0) | 4 (6.4) | 17 (8.4) | ||
| Tissue, n (%) | 0.412 | 0.139 | ||||
| Bone | 32 (48.5) | 145 (29.1) | 27 (45.8) | 77 (38.1) | ||
| Soft tissue | 33 (50.0) | 346 (69.3) | 32 (54.2) | 121 (59.9) | ||
| Overlapping | 1 (1.5) | 8 (1.6) | 0 (0.0) | 4 (2.0) | ||
| Size (cm), median (IQR) | 10.8 (7.5–13.5) | 10.0 (7.7–15.0) | 0.233 | 11.0 (7.6–13.8) | 10.3 (7.0–150.0) | 0.011 |
| Grade, n (%) | 0.164 | 0.016 | ||||
| Low/intermediate | 11 (16.7) | 55 (11.0) | 10 (16.9) | 33 (16.3) | ||
| High | 55 (83.3) | 444 (89.0) | 49 (83.1) | 169 (83.7) | ||
| T stage,an (%) | 0.169 | 0.110 | ||||
| T1 | 13 (19.7) | 62 (12.4) | 12 (20.3) | 32 (15.8) | ||
| T2/T3 | 46 (69.7) | 339 (67.9) | 46 (78.0) | 163 (80.7) | ||
| Missing | 7 (10.6) | 98 (19.6) | 1 (1.7) | 7 (3.5) | ||
| N stage,an (%) | 0.389 | 0.080 | ||||
| N0 | 63 (95.5) | 378 (75.8) | 56 (94.9) | 195 (96.5) | ||
| N1 | 3 (4.5) | 73 (14.6) | 3 (5.1) | 7 (3.5) | ||
| Missing | 0 (0.0) | 48 (9.6) | 0 (0.0) | 0 (0.0) | ||
| Chemotherapy,bn (%) | 0.310 | 0.035 | ||||
| Yes | 50 (75.8) | 307 (61.5) | 45 (76.3) | 151 (74.8) | ||
| No/unknown | 16 (24.2) | 192 (38.5) | 14 (23.7) | 51 (25.2) | ||
| Radiation,bn (%) | 0.471 | 0.483 | ||||
| Yes | 23 (34.8) | 75 (15.0) | 23 (39.0) | 36 (17.8) | ||
| No | 43 (65.2) | 424 (85.0) | 36 (61.0) | 166 (82.2) | ||
AJCC 7th edition staging.
Adjuvant and/or neoadjuvant with respect to the primary disease.
IQR: interquartile range; PM: pulmonary metastasectomy; SMD: standardized mean difference.
Survival and follow-up
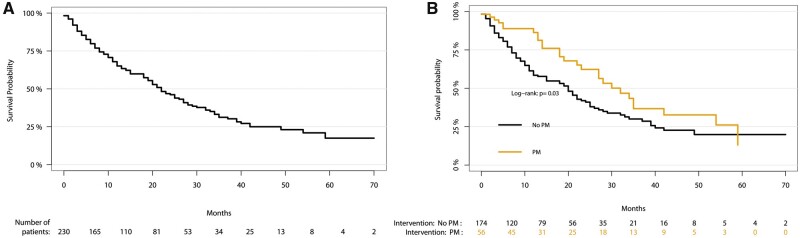
According to quantiles of potential follow-up from the reverse Kaplan–Meier method, the median estimated follow-up was 31 months (IQR 14–46). Among all matched patients the median DFS was 22 months (IQR 8–49), with 1-, 3- and 5-year estimates of 65.1% [95% confidence interval (CI) 58.5–71.7%], 31.2% (95% CI 23.6–38.9%) and 17.5% (95% CI 8.3–26.7%), respectively (Fig. 1A).
Figure 1:
(A) Kaplan–Meier survival analysis demonstrating disease-free survival of all patients with metastatic sarcoma after propensity matching. At-risk individuals are shown along the x-axis. (B) Kaplan–Meier survival analysis demonstrating disease-free survival between propensity-matched sarcoma patients who underwent PM and those treated with medical management alone (no metastasectomy). At-risk individuals are shown along the x-axis. PM: pulmonary metastasectomy.
PM was associated with improved DFS compared to medical management. The median DFS after PM was 32 months (IQR 18–59) compared to a median DFS of 20 months (IQR 7–40) without PM (P = 0.032). After PM, the DFS probability at 1 and 3 years were 86.3% (95% CI 76.8–95.9%) and 36.8% (95% CI 20.4–53.1%), respectively. With medical management, the 1- and 3-year DFS probabilities were 58.5% (95% CI 50.7–66.3%) and 30.0% (95% CI 21.5–38.5%), respectively (Fig. 1B). There were zero at-risk PM patients at 5 years, whereas for those without PM, the 5-year DFS probability was 19.9% (95% CI 10.8–29.0%).
A subset survival analysis among matched PM-only patients (n = 59) is tabulated in Supplementary Material, Table S2. Among these patients, the median DFS was longer in males compared to female patients (P = 0.021; Supplementary Material, Fig. S2), as well as in patients that underwent PM for lung metastasis from a primary bone sarcoma compared to a primary STS (P = 0.014; Supplementary Material, Fig. S3).
Multivariable Cox proportional hazards model
Among all matched patients on the basis of a multivariable Cox proportional hazards model of regression, age [hazard ratio (HR) 1.016, 95% CI 1.01–1.03; P = 0.001] and high-grade sarcoma (HR 1.893, 95% CI 1.09–3.28; P = 0.023) were found to be independent negative prognostic factors. However, chemotherapy (HR 0.569, 95% CI 0.35–0.92; P = 0.021) and metastasectomy (HR 0.536, 95% CI 0.33–0.85; P = 0.008) were found to be independent positive prognostic factors of DFS (Table 2).
Table 2:
Results of the final model of multivariable Cox proportional hazards regression among all patients (n = 261) after propensity matching
| Variables | HR (95% CI) | P-value |
|---|---|---|
| Age (years) (n = 261) | 1.016 (1.01–1.03) | 0.001a |
| Grade | ||
| Low/intermediate (n = 36) | Ref | |
| High (n = 183) | 1.893 (1.09–3.28) | 0.023a |
| Pulmonary metastasectomy | ||
| No (n = 164) | Ref | |
| Yes (n = 55) | 0.536 (0.34–0.85) | 0.008a |
| T stageb | ||
| T1 (n = 37) | Ref | |
| T2/3 (n = 182) | 1.499 (0.86–2.61) | 0.15 |
| Chemotherapyc | ||
| No/unknown (n = 48) | Ref | |
| Yes (n = 171) | 0.569 (0.35–0.92) | 0.021a |
| Radiationc | ||
| None (n = 163) | Ref | |
| Yes (n = 56) | 0.717 (0.47–1.10) | 0.13 |
Frequencies for each variable are indicated. Cases with missing survival data excluded.
Significant.
AJCC 7th edition staging for primary sarcoma.
Adjuvant and/or neoadjuvant with respect to the primary disease.
CI: confidence interval; HR: hazard ratio; Ref: reference variable.
DISCUSSION
Sarcomas are among the most common of tumours that metastasize to the lungs [19]. In the absence of lung metastases, 5-year survival ranges from ∼30% to 80% [20]. In contrast, 5-year survival with metastatic disease remains at or below 50%, with modern estimates nearing only 34% [21]. In this study, the 5-year DFS of all sarcoma patients with lung metastasis regardless of metastasectomy was 17.5%. Treatment options for these patients are relatively limited, though typically consist of chemotherapy and metastasectomy in an attempt to eradicate systemic disease. We therefore investigated the role of PM in a group of propensity-matched patients with advanced sarcoma to assess outcomes in comparison to patients treated medically. We found that PM portended an improved prognosis, though moreover, our data imply certain patients may be more eligible for PM than others based on survival differences by gender and sarcoma subtype.
The current population-based study of the SEER database found that PM improved the prognosis of patients with advanced sarcoma on multivariable analysis. In addition, chemotherapy for the primary tumour improved the prognosis on multivariable analysis, while older patients and those with high-grade disease had an association with worse DFS. After PM in particular, the median DFS was 32 months and was statistically significantly higher than the median DFS of 20 months in patients with advanced sarcoma treated medically. Although low, the 3-year DFS probability after PM was 36.8%, which aligns with the current literature [3–11, 22].
Treasure et al. [23] note the average age of patients undergoing PM for metastatic bone sarcoma was considerably lower (∼17 years old) in comparison to patients with STS undergoing PM (∼46 years old). The current study made no distinction in age between bone and sarcoma patients, though after propensity matching the median age of all patients undergoing PM was 45 years. There was a slightly higher proportion of STS patients included in this study, which may explain the higher median age as those who present with STS are typically older [1].
Interestingly, the current study found that males with advanced sarcoma who underwent PM had a statistically significant lower median DFS than females. While most existing studies fail to prognosticate gender in this setting, Buddingh et al. [24] do evidence worse outcomes for males with osteosarcoma that underwent PM in comparison to females with osteosarcoma who underwent PM. Therefore, the consensus is unclear, and the current study cohort was comprised of a variety of different diagnoses and patients of varying ages, which confounds the observed differences in survival. Although these data may imply certain patients should be considered more eligible than others with respect to indications for PM, future research is likely needed to reliably confirm an association between gender and survival after PM for sarcoma.
To address potential differences in the clinicopathological data in the SEER database, propensity matching was performed using up to a 1:4 ratio of PM to non-PM patients. After propensity matching, we found that osteosarcoma, leiomyosarcoma, and chondrosarcoma were among the most frequently metastatic primary tumour subtypes in the PM group. This profile is relatively similar to the context of a routine clinical sarcoma practice, and we believe the SEER was therefore at least clinically amenable to an analysis of PM in sarcoma. Each patient who underwent PM was further divided into a bone sarcoma and STS group, as well as into a group based on primary tumour grade (low and intermediate vs high grade).
Notably, patients with STS who underwent PM had a statistically significantly lower median DFS than those with bone sarcoma who underwent PM, which aligns with the current literature [6, 23]. However, this finding may be confounded by age, wherein younger patients with bone sarcoma may tolerate metastasectomy better, compared to older patients in whom STS is more commonly seen and in whom PM may be a higher morbidity procedure.
With respect to the outcomes of STS-only patients after PM, some data do suggest those with advanced leiomyosarcoma have improved survival compared to patients with rhabdomyosarcoma, alveolar soft part sarcoma, malignant fibrous histiocytoma, liposarcoma, and/or malignant peripheral nerve sheath tumour [3, 7, 25–28]. However, other reports have failed to find a survival difference among STS subtypes [3, 29]. Nonetheless, the findings of a worse median DFS after PM for advanced STS in the current study may mean the decision to pursue metastasectomy should be more readily considered in younger patients with, for example, advanced osteosarcoma or Ewing sarcoma, especially compared to an older patient with advanced STS.
With respect to the grade of the primary tumour, the current study found no difference in median DFS after PM for high-grade sarcoma versus PM for low or intermediate grade sarcoma. Although a lack of an association between survival after PM and tumour grade has largely been confirmed in the literature, Billingsley et al. [25] did report an improved survival of patients with low-grade compared to high-grade primary tumours [12]. However, based on the findings of this study, which failed to find an association of primary grade and survival after PM, it is more likely that no matter the primary tumour grade, metastasis represents an advanced state of disease and by the time of systemic spread, the initial grade has little prognostic significance.
Limitations
There are limitations to this study, most of which are inherent to the use of a multi-institutional database. First, this study was retrospective in nature, and although it collected data from a variety of participating hospitals, the cohort of PM patients is smaller than other single-institutional studies. This is likely because the SEER database only recorded PM data from 2010 to 2015. Second, we utilized propensity matching to reduce the influence of confounding bias, however, we did not have baseline patient data such as pulmonary reserve or comorbid diseases, nor data stating whether there was progression of initial disease on systemic medical treatment. These data would have helped to more clearly define outcomes. Third, a large number of patients were not subjected to PM. By nature of the SEER database, it was impossible to know whether these patients had, for example, multiple and bilateral metastases not amenable to complete resection. A complete resection (R0) is thought to be of critical importance in determining outcomes after PM [21, 22, 29]. Welter et al. [11] also found that certain characteristics such as pleural penetration portended a worse overall survival, in addition to the interval between resection of the primary tumour and metastasectomy. These data were not studied. Furthermore, given that the SEER database only records characteristics of the primary tumour with no such interval data, or data on the number or size of nodules, this study was unable to explore an association with the surgical approach (video-assisted thoracic surgery or open). This lack of a description of the technique precluded a meaningful comparison of each approach to one another, or to medical management alone. Therefore, the results must be interpreted accordingly.
CONCLUSIONS
Using a large, multi-institutional database, propensity-matched patients with advanced sarcoma and isolated lung metastasis who underwent PM had improved survival compared to patients treated with medical management alone. PM was identified as a positive prognostic factor of survival in this group, although it appears the benefit is limited to the short- to mid-term. Furthermore, it appears there may be an association of survival after PM by gender and tumour origin, which may have implications for selecting these patients as surgical candidates. Until randomized clinical trials for the treatment of pulmonary metastasis in sarcoma are successfully completed, PM should be considered an effective treatment strategy in select patients.
SUPPLEMENTARY MATERIAL
Supplementary material is available at ICVTS online.
Conflict of interest: none declared.
Author contributions
Charles A. Gusho: Data curation; Formal analysis; Investigation; Methodology; Software; Writing—original draft; Writing—review & editing. Christopher W. Seder: Formal analysis; Methodology; Writing—original draft; Writing—review & editing. Nicolas Lopez-Hisijos: Methodology; Writing—original draft; Writing—review & editing. Alan T. Blank: Conceptualization; Investigation; Methodology; Project administration; Supervision; Validation; Visualization; Writing—review & editing. Marta Batus: Conceptualization; Investigation; Methodology; Project administration; Supervision; Validation; Visualization; Writing—review & editing.
Reviewer information
Interactive CardioVascular and Thoracic Surgery thanks William Grossi, Mohamed Rahouma, Stefan B. Watzka and the other anonymous reviewer(s) for their contribution to the peer review process of this article.
Supplementary Material
ABBREVIATIONS
- CI
Confidence interval
- DFS
Disease-free survival
- HR
Hazard ratio
- IQR
Interquartile range
- PM
Pulmonary metastasectomy
- SEER
Surveillance, Epidemiology and End Results
- STS
Soft tissue sarcoma
REFERENCES
- 1. Spiguel A. Soft tissue sarcomas. Cancer Treat Res 2014;162:203–23. [DOI] [PubMed] [Google Scholar]
- 2. Porter GA, Cantor SB, Walsh GL, Rusch VW, Leung DH, DeJesus AY. et al. Cost-effectiveness of pulmonary resection and systemic chemotherapy in the management of metastatic soft tissue sarcoma: a combined analysis from the University of Texas M. D. Anderson and Memorial Sloan-Kettering Cancer Centers. J Thorac Cardiovasc Surg 2004;127:1366–72. [DOI] [PubMed] [Google Scholar]
- 3. Blackmon SH, Shah N, Roth JA, Correa AM, Vaporciyan AA, Rice DC. et al. Resection of pulmonary and extrapulmonary sarcomatous metastases is associated with long-term survival. Ann Thorac Surg 2009;88:877–85; discussion 884–5. [DOI] [PubMed] [Google Scholar]
- 4. Dear RF, Kelly PJ, Wright GM, Stalley P, McCaughan BC, Tattersall MHN.. Pulmonary metastasectomy for bone and soft tissue sarcoma in Australia: 114 patients from 1978 to 2008. Asia Pac J Clin Oncol 2012;8:292–302. [DOI] [PubMed] [Google Scholar]
- 5. Kim HS, Nam CM, Jang S-Y, Choi SK, Han M, Kim S. et al. Characteristics and treatment patterns of patients with advanced soft tissue sarcoma in Korea. Cancer Res Treat 2019;51:1380–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Dossett LA, Toloza EM, Fontaine J, Robinson L, Reed D, Druta M. et al. Outcomes and clinical predictors of improved survival in a patients undergoing pulmonary metastasectomy for sarcoma. J Surg Oncol 2015;112:103–6. [DOI] [PubMed] [Google Scholar]
- 7. Lin AY, Kotova S, Yanagawa J, Elbuluk O, Wang G, Kar N. et al. Risk stratification of patients undergoing pulmonary metastasectomy for soft tissue and bone sarcomas. J Thorac Cardiovasc Surg 2015;149:85–92. [DOI] [PubMed] [Google Scholar]
- 8. Okiror L, Peleki A, Moffat D, Bille A, Bishay E, Rajesh P. et al. Survival following pulmonary metastasectomy for sarcoma. Thorac Cardiovasc Surg 2016;64:146–9. [DOI] [PubMed] [Google Scholar]
- 9. Reza J, Sammann A, Jin C, Horvai A, Hudnall M, Jablons DM. et al. Aggressive and minimally invasive surgery for pulmonary metastasis of sarcoma. J Thorac Cardiovasc Surg 2014;147:1193–200; discussion 1200–1. [DOI] [PubMed] [Google Scholar]
- 10. Suzuki M, Iwata T, Ando S, Iida T, Nakajima T, Ishii T. et al. Predictors of long-term survival with pulmonary metastasectomy for osteosarcomas and soft tissue sarcomas. J Cardiovasc Surg (Torino) 2006;47:603–8. [PubMed] [Google Scholar]
- 11. Welter S, Grabellus F, Bauer S, Schmid KW, Stamatis G, Tötsch M.. Growth patterns of lung metastases from sarcomas. Virchows Arch 2011;459:213–19. [DOI] [PubMed] [Google Scholar]
- 12. Smith R, Demmy TL.. Pulmonary metastasectomy for soft tissue sarcoma. Surg Oncol Clin N Am 2012;21:269–86. [DOI] [PubMed] [Google Scholar]
- 13. Dudek W, Schreiner W, Mykoliuk I, Higaze M, Sirbu H.. Pulmonary metastasectomy for sarcoma—survival and prognostic analysis. J Thorac Dis 2019;11:3369–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Marulli G, Mammana M, Comacchio G, Rea F.. Survival and prognostic factors following pulmonary metastasectomy for sarcoma. J Thorac Dis 2017;9:S1305–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.SEERStat Databases: November 2012 Submission. SEER. https://seer.cancer.gov/data-software/documentation/seerstat/nov2012/index.html (7 July 2020, date last accessed).
- 16. Amin MB, Edge S, Greene F, Byrd D, Brookland R, Washington MK. et al. (eds). AJCC Cancer Staging Manual. 8th edn. Springer International Publishing, 2017. https://www.springer.com/gp/book/9783319406176 (23 September 2020, date last accessed). [Google Scholar]
- 17. Parsons LS. Reducing Bias in a Propensity Score Matched-Pair Sample Using Greedy Matching Techniques. The Twenty-Sixth Annual SAS Users Group International Conference. 2001. 214–26.
- 18. Schemper M, Smith TL.. A note on quantifying follow-up in studies of failure time. Control Clin Trials 1996;17:343–6. [DOI] [PubMed] [Google Scholar]
- 19. Pastorino U, Buyse M, Friedel G, Ginsberg RJ, Girard P, Goldstraw P. et al. ; International Registry of Lung Metastases. Long-term results of lung metastasectomy: prognostic analyses based on 5206 cases. J Thorac Cardiovasc Surg 1997;113:37–49. [DOI] [PubMed] [Google Scholar]
- 20. Stiller CA, Trama A, Serraino D, Rossi S, Navarro C, Chirlaque MD. et al. ; RARECARE Working Group. Descriptive epidemiology of sarcomas in Europe: report from the RARECARE project. Eur J Cancer 2013;49:684–95. [DOI] [PubMed] [Google Scholar]
- 21. Chudgar NP, Brennan MF, Munhoz RR, Bucciarelli PR, Tan KS, D'Angelo SP. et al. Pulmonary metastasectomy with therapeutic intent for soft-tissue sarcoma. J Thorac Cardiovasc Surg 2017;154:319–30.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Mizuno T, Taniguchi T, Ishikawa Y, Kawaguchi K, Fukui T, Ishiguro F. et al. Pulmonary metastasectomy for osteogenic and soft tissue sarcoma: who really benefits from surgical treatment? Eur J Cardiothorac Surg 2013;43:795–9. [DOI] [PubMed] [Google Scholar]
- 23. Treasure T, Fiorentino F, Scarci M, Møller H, Utley M.. Pulmonary metastasectomy for sarcoma: a systematic review of reported outcomes in the context of Thames Cancer Registry data. BMJ Open 2012;2:e001736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Buddingh EP, Anninga JK, Versteegh MIM, Taminiau A, Egeler R, van Rijswijk C. et al. Prognostic factors in pulmonary metastasized high-grade osteosarcoma. Pediatr Blood Cancer 2010;54:216–21. [DOI] [PubMed] [Google Scholar]
- 25. Billingsley KG, Burt ME, Jara E, Ginsberg RJ, Woodruff J, Leung D. et al. Pulmonary metastases from soft tissue sarcoma: analysis of patterns of diseases and postmetastasis survival. Ann Surg 1999;229:602–10; discussion 610–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Burt BM, Ocejo S, Mery CM, Dasilva M, Bueno R, Sugarbaker DJ. et al. Repeated and aggressive pulmonary resections for leiomyosarcoma metastases extends survival. Ann Thorac Surg 2011;92:1202–7. [DOI] [PubMed] [Google Scholar]
- 27. Gadd MA, Casper ES, Woodruff JM, McCormack PM, Brennan MF.. Development and treatment of pulmonary metastases in adult patients with extremity soft tissue sarcoma. Ann Surg 1993;218:705–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Casson AG, Putnam JB, Natarajan G, Johnston DA, Mountain C, McMurtrey M. et al. Five-year survival after pulmonary metastasectomy for adult soft tissue sarcoma. Cancer 1992;69:662–8. [DOI] [PubMed] [Google Scholar]
- 29. Smith R, Pak Y, Kraybill W, Kane JM.. Factors associated with actual long-term survival following soft tissue sarcoma pulmonary metastasectomy. Eur J Surg Oncol 2009;35:356–61. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.