Abstract
Carcinoma cuniculatum (CC) is a rare clinicopathologic variant of squamous cell carcinoma. Histologically, it is characterized by invasive growth of bland, acanthotic, and keratinizing squamous epithelium that forms multiple rabbit burrow-like, keratin-filled crypts and sinuses. We present a 51-year-old male smoker with CC of the left vocal cord. The tumor was staged T1a and the patient was disease-free 12 months after surgery. To our knowledge, this is the fourth case of CC of the larynx reported in the English literature and the first, due to its early diagnosis, where radical surgery was not performed. We highlight the necessity for awareness of this entity and coordination between otolaryngologists, radiologists, and pathologists for early diagnosis and organ-sparing surgical treatment.
Keywords: Carcinoma cuniculatum, Verrucous carcinoma, Well-differentiated squamous cell carcinoma, Upper aero-digestive tract, Larynx, Vocal cords, Histological diagnosis
Introduction
Carcinoma cuniculatum (CC), also referred to as epithelioma cuniculatum, is a rare variant of well-differentiated squamous cell carcinoma (SCC) [1, 2]. The term cuniculatum refers to the complex pattern of deep invasion by keratinizing squamous epithelium-lined branched rabbit burrow-like crypts and sinuses. As CC shows limited low-grade cytological atypia, its histologic recognition may be exceedingly difficult, especially with small biopsies [2–4].
First described by Aird in 1954, CC may affect any cutaneous area of the body [1]. It usually occurs on the skin of the plantar surface of the foot, followed by the toes and heel [2]. Rarely, CC has been described in the mucosa of the upper aero-digestive tract [3–16].
Even though the aetiology of this tumour is not definitely established, infection, trauma, chronic irritation, and radiation are variably reported as possible predisposing factors. Specifically in the aerodigestive tract, human papillomavirus (HPV) infection, alcohol, and tobacco have been considered as potential etiologies although definitive evidence is lacking [3–16].
Clinically, CC typically presents as a slow-growing, painful, non-healing exophytic mass. Histologically, it shows both exophytic and endophytic growth patterns [1–3, 9, 16]. Due to these clinicopathologic features, CC was previously thought to be an alternative name of verrucous carcinoma (VC) and even described as “inverted VC” [11, 13]. In 2005, however, the World Health Organization (WHO) recognized CC as a specific entity of the oral cavity [17].
Since CC is locally invasive and metastases to regional lymph nodes are rarely described, local resection, usually without regional lymph node dissection, is the definitive treatment [4, 14].
To the best of our knowledge, CC has been described in the larynx in only three cases [4, 8, 12]. The aim of this paper is to present a rare case of CC of the left vocal cord with a review of the literature.
Case Report
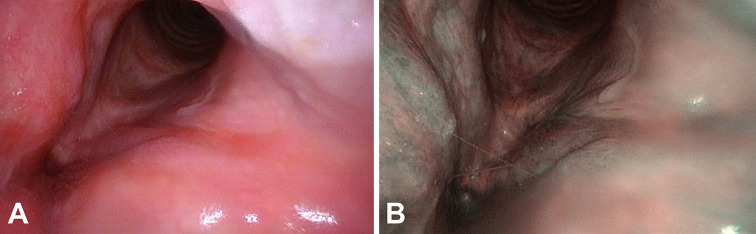
A 51-year-old Caucasian male smoker presented to our Otolaryngology clinic with a 10-month history of dysphonia. At admission, a flexible laryngoscopy with narrow-band imaging (NBI) revealed a whitish mass of the left vocal cord which extended from the anterior commissure to the posterior region of the cord. Laryngeal motility was preserved, and no cervical lymphadenopathies were clinically evident.
A computed tomography (CT) scan with contrast of the head and neck was performed and showed minimal local involvement of the left vocal cord with no extension to adjacent structures and no cervical lymphadenopathy (Fig. 1). The patient underwent removal of the lesion through microlaryngoscopy.
Fig. 1.
Computed tomography images in the axial (a) and coronal (b) planes showing minimal local involvement of the left vocal cord with no extension to the close structures and no cervical lymphadenopathy
Histologic examination (Fig. 2) revealed an endophytic network of bland, keratinizing squamous epithelium that formed multiple, complex, branching keratin-filled crypts/sinuses. The crypts “burrowed” within the superficial lamina propria. Focally, keratin within the crypts/sinuses was associated with inflammatory cells. Dyskeratosis, intra-epithelial neutrophils, and micro-abscesses were focally observed. The lesion extended 3.5 mm horizontally and showed a maximum depth of about 2.0 mm. The front of invasion was bulbous and the squamous epithelium was characterized by prominent acanthosis and keratosis. A florid inflammatory cell infiltrate was present in the lamina propria. These findings were consistent with CC. Immunostaining for HPV (clone K1H8, Dako, Glostrup, Denmark) was negative.
Fig. 2.
Low-power magnification of the lesion removed through microlaryngoscopy (a and b). The images were obtained from two different histological sections. The lesion consists of a branching, endophytic proliferation of keratinizing squamous epithelium forming crypts/sinuses that “burrow” within the superficial lamina propria. A florid inflammatory cell infiltrate is present in the lamina propria. Crypts/sinuses are lined by well-differentiated squamous epithelium and filled with keratin (c), that is focally intermixed with inflammatory cells (d). Dyskeratosis, intra-epithelial neutrophils, and micro-abscesses are illustrated in e. Staining: haematoxylin and eosin. Bars: 500 μm in a and b, 100 μm in c and 50 μm in d and e
Based on the biopsy and clinico-radiographic findings, a unilateral cordectomy of the left vocal cord was performed. Histopathologic examination of the sample revealed mucosa lined by squamous epithelium that formed a minimally branching, superficial crypt/sinus. The crypt/sinus was filled with keratin, and a florid inflammatory infiltrate within the lamina propria was considered a remnant of the lesion (Fig. 3). The tumor was staged T1a and patient was discharged with strict follow-up. Flexible white-light and NBI laryngoscopy performed at 6-month and 12-month intervals after surgery failed to detect any residual lesion (Fig. 4). Further follow-up is warranted.
Fig. 3.

A minimally branching, superficial crypt/sinus was observed in the sample obtained at cordectomy. The crypt/sinus is filled with keratin and associated with a florid inflammatory infiltrate within the lamina propria. Staining: haematoxylin and eosin. Bar: 150 μm
Fig. 4.
Flexible white-light (a) and NBI (b) laryngoscopy performed 6 months after surgery
Discussion
In the larynx, CC has previously been described in three cases (Table 1) [4, 8, 12]. Similarly to two of the previously reported cases, our patient was also a smoker [4, 12]. Unlike our case, all three tumors were identified at advanced stages that required radical surgery (partial or total laryngectomy with or without neck lymph node dissection) [4, 8, 12]. This is not surprising because the time between the onset of the tumor and definitive diagnosis may be long and require multiple biopsies. In one of these cases, where the tumor involved the glottic and supra-glottic regions, the definitive diagnosis was made on the surgical specimen after repeated biopsies [12].
Table 1.
Reported cases of carcinoma cuniculatum of the larynx
| References | Age/gender | Site | Smoker | Treatment | Follow-up |
|---|---|---|---|---|---|
| [4] | 49/M | Glottis and sub-glottis with involvement of a saccular cyst | + | Total laryngectomy with bilateral neck dissection | Disease free 6 months after surgery |
| [8] | 63/M | Right hemi-larynx with sub-glottic extension | Not available | Total laryngectomy with bilateral neck dissection | Disease free 42 months after surgery |
| [12] | 72/M | Glottis and supra-glottis | + | Supra-cricoid laryngectomy with crico-hyoidopexy extended to the pre-laryngeal muscles | Disease free 70 months after surgery |
| Present case | 51/M | Left vocal cord | + | Microlaryngoscopy with biopsy and cordectomy | Disease free 10 months after surgery |
The definitive diagnosis of CC may be difficult and require clinical examination with generous biopsies for histologic examination. The necessity for deep biopsies is related to the endophytic growth pattern of CC and deep, broad proliferations of keratinizing squamous epithelium devoid of obvious features of malignancy. The lack of cytologic atypia observed in CC is a shared characteristic of VC. Even though CC and VC may appear clinically similar and are both characterized by well-differentiated keratinizing squamous cells and minimal atypia, the pattern of invasion in VC is different and consists of broad, bulbous rete ridges with pushing margins [3, 11, 14–17]. Differentiation between CC and VC is mandatory because the clinical course of the former is more aggressive locally and, even if rarely, be associated with lymph node metastasis [4, 11, 14–17]. Since the crypts/sinuses of CC may be inflamed/infected, as evidenced by intra-luminal keratin and inflammatory cells, reactive squamous cytological atypia, brisk mitotic activity, and pseudo-epitheliomatous hyperplasia may be observed. Consequently, conventional well-differentiated SCC or a hybrid carcinoma (well-differentiated SCC in CC) may be suspected. In these cases, only the demonstration of unequivocal stromal infiltration establishes the correct diagnosis [12, 16, 17]. Finally, sampling limited to the superficial exophytic component of the lesion may also suggest a papillary SCC. However, this variant of SCC may be differentiated from CC and VC because it consists of thinner, arborizing papillae invariably lined by atypical cells [14–17].
Regardless of the location, the prognosis of CC is favorable. CC may be locally destructive, but lymph node metastases are rare [4, 14]. For these reasons, surgical excision/resection is the mainstay of treatment in combination with close and prolonged monitoring [4, 10, 12, 14–17]. Lymph node dissection should be considered with accompanying lymphadenopathy and when the diagnosis of CC is doubtful. Due to the rarity of CC in the upper aerodigestive tract, especially in the larynx, awareness of this entity and cooperation between otolaryngologists, radiologists, and pathologists is necessary for early diagnosis and organ-sparing surgical treatment.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Compliance with Ethical Standards
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
All the clinico-pathologic investigations detailed in the manuscript have been conducted in accordance with the Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed Consent
Written informed consent for publication of data and images was obtained from the patient.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Massimo Ralli and Alessandro Corsi have contributed equally
Contributor Information
Massimo Ralli, Email: massimo.ralli@uniroma1.it.
Alessandro Corsi, Email: alessandro.corsi@uniroma1.it.
References
- 1.Aird I, Johnson HD, Lennox B, Stansfeld AG. Epithelioma cuniculatum: a variety of squamous carcinoma peculiar to the foot. Br J Surg. 1954;42:245–250. doi: 10.1002/bjs.18004217304. [DOI] [PubMed] [Google Scholar]
- 2.Kao GF, Graham JH, Helwig EB. Carcinoma cuniculatum (verrucous carcinoma of the skin): a clinicopathologic study of 46 cases with ultrastructural observations. Cancer. 1982;49:2395–2403. doi: 10.1002/1097-0142(19820601)49:11<2395::aid-cncr2820491129>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- 3.Datar UV, Kale A, Mane D. Oral carcinoma cuniculatum: a new entity in the clinicopathological spectrum of oral squamous cell carcinoma. J Clin Diagn Res. 2017;11:37–39. doi: 10.7860/JCDR/2017/23437.9226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Griffith CC, Goodstein LA, Tuluc M, Prestipino A, Cognetti D, Spiegel J, et al. A rare case of carcinoma cuniculatum involving the larynx in association with a saccular cyst. Head Neck Pathol. 2015;9:421–425. doi: 10.1007/s12105-014-0603-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Flieger S, Owinski T. Epithelioma cuniculatum an unusual form of mouth and jaw neoplasm. Czas Stomatol. 1977;30:395–401. [PubMed] [Google Scholar]
- 6.Eisenberg E, Rosenberg B, Krutchkoff DJ. Verrucous carcinoma: a possible viral pathogenesis. Oral Surg Oral Med Oral Pathol. 1985;59:52–57. doi: 10.1016/0030-4220(85)90115-x. [DOI] [PubMed] [Google Scholar]
- 7.Kahn JL, Blez P, Gasser B, Weill-Bousson M, Vetter JM, Champy M. Carcinoma cuniculatum. Apropos of 4 cases with orofacial involvement. Rev Stomatol Chir Maxillofac. 1991;92:27–33. [PubMed] [Google Scholar]
- 8.Delahaye JF, Janser JC, Rodier JF, Auge B. Cuniculatum carcinoma. 6 cases and review of the literature. J Chir. 1994;131:73–78. [PubMed] [Google Scholar]
- 9.Petersen CS, Sjolin KE, Rosman N, Lindeberg H. Lack of human papillomavirus DNA in carcinoma cuniculatum. Acta Derm Venereol. 1994;74:231–232. doi: 10.2340/0001555574231232. [DOI] [PubMed] [Google Scholar]
- 10.Huault M, Laroche C, Levy J, Laxenaire A, Roucayrol AM, Scheffer P. Epithelioma cuniculatum. Apropos of a case in the anterior gingiva with involvement of the mandibular symphyseal bone and reconstruction using a fibular osteocutaneous flap and integrated implants. Rev Stomatol Chir Maxillofac. 1998;99:143–148. [PubMed] [Google Scholar]
- 11.Allon D, Kaplan I, Manor R, Calderon S. Carcinoma cuniculatum of the jaw: a rare variant of oral carcinoma. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2002;94:601–608. doi: 10.1067/moe.2002.126913. [DOI] [PubMed] [Google Scholar]
- 12.Puxeddu R, Cocco D, Parodo G, Mallarini G, Medda M, Brennan PA. Carcinoma cuniculatum of the larynx: a rare clinicopathological entity. J Laryngol Otol. 2008;122:1118–1123. doi: 10.1017/S0022215107000163. [DOI] [PubMed] [Google Scholar]
- 13.Thavaraj S, Cobb A, Kalavrezos N, Beale T, Walker DM, Jay A. Carcinoma cuniculatum arising in the tongue. Head Neck Pathol. 2012;6:130–134. doi: 10.1007/s12105-011-0270-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sun Y, Kuyama K, Burkhardt A, Yamamot H. Clinicopathological evaluation of carcinoma cuniculatum: a variant of oral squamous cell carcinoma. J Oral Pathol Med. 2012;41:303–308. doi: 10.1111/j.1600-0714.2011.01116.x. [DOI] [PubMed] [Google Scholar]
- 15.Padilla RJ, Murrach VA. Carcinoma cuniculatum of the oral mucosa: a potentially underdiagnosed entity in the absence of clinical correlation. Oral Surg Oral Med Oral Pathol Oral Radiol. 2014;118:684–693. doi: 10.1016/j.oooo.2014.08.011. [DOI] [PubMed] [Google Scholar]
- 16.Sivapathasundhara B, Kavitha B, Padmapriya VM. Carcinoma cuniculatum of the alveolar mucosa: a rare variant of squamous cell carcinoma. Head Neck Pathol. 2019;13:652–655. doi: 10.1007/s12105-018-0938-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Johnson N, Franceschi S, Ferlay J, et al. Squamous cell carcinoma. In: Barnes EL, Eveson JW, Reichart P, Sidransky D, et al., editors. World Health Organization classification of tumours. Pathology and genetics of head and neck tumours. Geneva: WHO/IARC Press; 2005. pp. 165–173. [Google Scholar]