Abstract
Background
Antipseudomonal antibiotics are often used to treat community-acquired intra-abdominal infections (CA-IAIs) despite common causative pathogens being susceptible to more narrow-spectrum agents. The purpose of this study was to compare treatment-associated complications in adult patients treated for CA-IAI with antipseudomonal versus narrow-spectrum regimens.
Methods
This retrospective cohort study included patients >18 years admitted for CA-IAI treated with antibiotics. The primary objective of this study was to compare 90-day treatment-associated complications between patients treated empirically with antipseudomonal versus narrow-spectrum regimens. Secondary objectives were to compare infection and treatment characteristics along with patient outcomes. Subgroup analyses were planned to compare outcomes of patients with low-risk and high-risk CA-IAIs and patients requiring surgical intervention versus medically managed.
Results
A total of 350 patients were included: antipseudomonal, n=204; narrow spectrum, n=146. There were no differences in 90-day treatment-associated complications between groups (antipseudomonal 15.1% vs narrow spectrum 11.3%, P=.296). In addition, no differences were observed in hospital length of stay, 90-day readmission, Clostridiodes difficile, or mortality. In multivariate logistic regression, treatment with a narrow-spectrum regimen (odds ratio [OR], 0.75; 95% confidence interval, 0.39–1.45) was not independently associated with the primary outcome. No differences were observed in 90-day treatment-associated complications for (1) patients with low-risk (antipseudomonal 15% vs narrow spectrum 9.6%, P=.154) or high-risk CA-IAI (antipseudomonal 15.8% vs narrow spectrum 22.2%, P=.588) or (2) those who were surgically (antipseudomonal 8.5% vs narrow spectrum 9.2%, P=.877) or medically managed (antipseudomonal 23.1 vs narrow spectrum 14.5, P=.178).
Conclusions
Treatment-associated complications were similar among patients treated with antipseudomonal and narrow-spectrum antibiotics. Antipseudomonal therapy is likely unnecessary for most patients with CA-IAI.
Keywords: antimicrobial resistance, antipseudomonal, appendicitis, diverticulitis, intra-abdominal infections
Antipseudomonal regimens are often used to treat community-acquired intra-abdominal infections (CA-IAIs) despite common causative pathogens being susceptible to more narrow-spectrum agents. This retrospective cohort study demonstrated no difference in 90-day treatment-associated complications between patients treated with antipseudomonal or narrow-spectrum regimens.
Community-acquired intra-abdominal infections (CA-IAIs) are a leading cause of hospitalization in the United States annually [1]. Successful treatment depends on a variety of factors including patient characteristics, surgical source control, and appropriate antimicrobial therapy [1]. Complications after CA-IAI are also a concern, with an estimated recurrence rate of 15%–20% and 3%–6%, respectively, for diverticulitis and appendicitis [2, 3]. Although the most common causative pathogens in CA-IAI include Enterobacterales, Streptococcus species, and anaerobic bacteria, which are typically susceptible to more narrow-spectrum antibiotics, antipseudomonal regimens are often used to treat CA-IAI [4].
The overuse of broad-spectrum antibiotics is a leading cause of antimicrobial resistance, and multidrug-resistant Pseudomonas is classified by the Centers for Disease Control and Prevention as a serious threat of concern to human health affecting approximately 33000 hospitalized patients each year [5]. In addition to intrinsic mechanisms of resistance, excessive use of antibiotics significantly contributes to the development of multidrug-resistant Pseudomonas [6]. Optimizing antimicrobial use and stewardship efforts when treating CA-IAI plays an essential role in combating the emergence of local and global resistance. In 2010, CA-IAI treatment guidelines outlining empiric antibiotic recommendations were published by the Infectious Diseases Society of America (IDSA) and Surgical Infection Society (SIS) [7]. Since then, SIS published revised guidelines in 2017 with updated empiric antibiotic recommendations taking into consideration antimicrobial stewardship goals in light of rising antimicrobial resistance [8]. These most recent guidelines recommend narrow-spectrum antimicrobial regimens for the majority of patients presenting with CA-IAI. Therapy should be based on local susceptibilities and resistance patterns, stressing that the choice of empiric antibiotic therapy should take into consideration targeted pathogens and patient risk factors to minimize the overuse of antipseudomonal agents [1, 8]. In addition, the guidelines further recommend stratifying patients as either low- or high-risk based on their risk for complications and adverse outcomes, with low-risk patients being appropriate to receive narrow-spectrum therapy. Despite the updated guidance supporting more narrow-spectrum regimens, surveillance data showed the majority of adult patients presenting with low-risk CA-IAI were still receiving antipseudomonal regimens [4]. Research is lacking to indicate whether a difference exists in treatment failure rates in adult patients who are hospitalized with CA-IAI treated with antipseudomonal versus more narrow-spectrum antibiotic regimens. The purpose of this study was to compare treatment-associated complication rates between adult patients treated with antipseudomonal antibiotic regimens versus narrow-spectrum regimens for the treatment of CA-IAI.
METHODS
Patient Consent Statement
This study qualified for exemption after institutional review board review and did not require patient consent.
Study Site and Patient Selection
This retrospective cohort study was conducted at a 350-bed community teaching hospital. Patients were eligible for inclusion if they were >18 years and admitted for acute appendicitis or diverticulitis that required empiric treatment with antibiotics between January 1, 2013 and December 31, 2019. Patients were excluded if they were empirically treated with both an antipseudomonal and narrow-spectrum regimen during admission. In addition, patients were excluded (1) if they were found to have a healthcare-associated intra-abdominal infection upon chart review or (2) if they were noted to have peritonitis or bacteremia. Healthcare-associated intra-abdominal infection was defined as hospitalization for at least 48 hours during the previous 30 days, intravenous antimicrobial therapy within the previous 90 days, diverticulitis within 90 days of admission, or diagnosis of a postoperative infection.
Data Collection and Study Endpoints
A report was generated from the electronic medical record using International Classification of Diseases, Ninth Revision (ICD-9) and ICD-10 codes for diverticulitis and appendicitis. Patient eligible for screening were then put into randomized order and screened for inclusion into the analysis until the desired sample size was met. Patient, treatment, and infection characteristics were collected. The primary objective of this study was to compare treatment-associated complications within 90 days between patients treated empirically for CA-IAI empirically with antipseudomonal regimens versus narrow-spectrum regimens. Treatment-associated complication was defined as postoperative infection, including surgical site infection or development of new intra-abdominal abscess, recurrence of diverticulitis within 90 days, or mortality within 90 days. Table 1 defines the regimens included within either the antipseudomonal or narrow-spectrum groups. Secondary objectives were to compare length of antibiotic therapy, hospital length of stay (LOS), hospital readmission within 30 and 90 days, development of Clostridiodes difficile within 30 days, and in-hospital and 90-day mortality between groups. Subgroup analyses were planned a priori to compare outcomes between patients classified as low-risk CA-IAI versus high-risk CA-IAI as well as between patients who required source control versus medically managed. High-risk CA-IAI was defined as patients having 2 or more of the following characteristics: age >70, current malignancy, significant cardiovascular disease defined as patients in acute decompensated heart failure exacerbation at the time of admission, significant liver disease defined as cirrhosis, significant renal disease defined as chronic kidney disease stage 4 or worse, hypoalbuminemia, delayed source control defined as >24 hours after hospital admission, and history of extended-spectrum beta lactamase, vancomycin-resistant Enterococcus, or Klebsiella pneumoniae carbapenemase. Infectious source control was defined as surgical intervention or via interventional radiology procedure. A breakdown of the source control interventions can be found in Table 7.
Table 1.
Empiric Antibiotic Regimens
| Antipseudomonal Regimen, n (%) | n=146 |
| Piperacillin/tazobactam | 90 (61.7) |
| Fluoroquinolonea/metronidazole | 53 (36.3 |
| Cefepime/metronidazole | 1 (0.7) |
| Ceftazidime/metronidazole | 1 (0.7) |
| Aztreonam/metronidazole | 1 (0.7) |
| Narrow-Spectrum Regimen, n (%) | n=204 |
| Ceftriaxone/metronidazole | 162 (79.3) |
| Cefoxitin | 32 (15.7) |
| Ertapenem | 4 (2) |
| Cefazolin/metronidazole | 2 (1) |
| Ampicillin/sulbactam | 1 (0.5) |
| Cefazolin | 1 (0.5) |
| Ceftriaxone | 1 (0.5) |
| Cefuroxime/metronidazole | 1 (0.5) |
Ciprofloxacin or levofloxacin.
Table 7.
Subgroup Analysis: Surgical Candidate Outcomes
| Surgical Candidate Outcomes | Antipseudomonal (n=83) | Narrow Spectrum (n=120) | P Value |
|---|---|---|---|
| Infection Type | .189 | ||
| Uncomplicated diverticulitis, n (%) | 0 (0) | 0 (0) | |
| Complicated diverticulitis, n (%) | 11 (13.3) | 10 (8.3) | |
| Uncomplicated appendicitis, n (%) | 25 (30.1) | 50 (41.7) | |
| Complicated appendicitis, n (%) | 47 (56.6) | 60 (50.0) | |
| Surgery-related infection at 30 days, n (%) | 6 (7.3) | 9 (7.5) | .961 |
| Surgery-related infection 90 days, n (%) | 7 (8.4) | 11 (9.2) | .857 |
| Laparoscopic procedures, n (%) | (64) 77.1 | 95 (79.2) | .726 |
| Interventional radiology procedure, n (%) | 5 (6) | 19 (15.8) | .033 |
| Open surgical procedure, n (%) | 14 (16.9) | 6 (5) | 0.005 |
| Gross perforation/ contamination, n (%) | 55 (66.3) | 62 (51.7) | .039 |
| LOS, days, median [IQR] | 3 [2–5] | 3 [2–4] | .179 |
| In-hospital mortality, n (%) | 1 (1.2) | 0 (0) | .409 |
| Hospital Readmission 30-Day, n (%) | 8 (9.6) | 8 (6.7) | .440 |
| IAI related | 7 (8.4) | 7 (6.0) | 1.0 |
| Hospital Readmission 90-Day, n (%) | 16 (19.3) | 11 (9.2) | .037 |
| IAI related | 12 (14.5) | 8 (6.7) | 1.0 |
| Clostridiodes difficile 30 days, n (%) | 0 (0) | 0 (0) | 1.0 |
Abbreviations: IAI, intra-abdominal infection; IQR, interquartile range; LOS, length of stay.
Statistical Analysis
Considering diverticulitis recurrence rates are estimated to be 15%–20% and appendicitis treatment-associated complication rates are estimated to be between 3% and 6%, we assumed similar rates of diverticulitis and appendicitis in each group and therefore a combined baseline treatment-associated complication rate of 10% to 13% in our total study population. Based on these assumptions, we determined that 350 patients were needed to detect at least a 7%–10% absolute difference in treatment-associated complication rates for patients empirically treated with either antipseudomonal versus narrow-spectrum regimens using a 2-sided test with α=0.05 and 80% power [2, 3]. Nominal data were assessed with χ2 or Fisher’s exact test, as appropriate, and continuous data were assessed with Student’s t test or Mann-Whitney U based on the distribution of data. Multivariate logistic regression was performed to assess for independent risk factors of 90-day treatment-associated complications. Factors with a P ≤ .3 in the bivariate analysis were eligible for inclusion into the model. SPSS statistical software version 22 (IBM Corporation, Armonk, NY) was used for all statistical analyses.
RESULTS
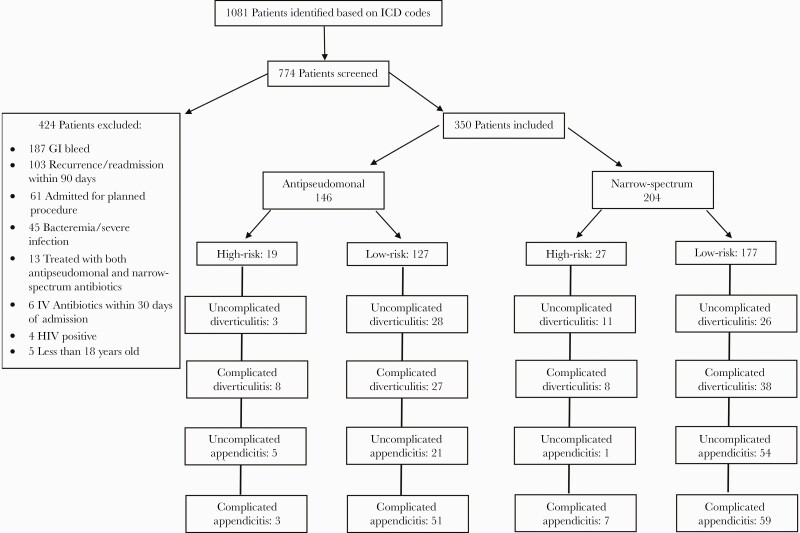
Within the 7-year study period, the initial query identified 1081 patients with CA-IAI. A randomized sample of 774 patients were screened until 350 patients met criteria for inclusion (Figure 1). A total of 204 (58.3%) patients were included in the narrow-spectrum group with 177 (86.7%) determined to be low risk and 146 (41.7%) patients in the antipseudomonal group with 127 (87%) low-risk patients (Figure 1). There were no differences observed in baseline characteristics between groups (Table 2). The majority of patients in the antipseudomonal group were empirically treated with piperacillin/tazobactam (61.7%), followed by a fluoroquinolone plus metronidazole (36.3%). Patients in the narrow-spectrum group were most commonly treated with ceftriaxone and metronidazole (79.3%), followed by cefoxitin (15.7%). A list of empirically prescribed antibiotic regimens by group is available in Table 1.
Figure 1.
Flowchart summarizing patient inclusion screening, including reasons for exclusion. GI, gastrointestinal; HIV, human immunodeficiency virus; ICD, International Classification of Diseases; IV, intravenous.
Table 2.
Patient and Infection Characteristics
| Characteristics | Antipseudomonal (n=146) | Narrow Spectrum (n=204) | P Value |
|---|---|---|---|
| Age, years (mean±SD) | 51.8±16.3 | 53.9±19.2 | .257 |
| Sex, female, n (%) | 82 (56.2) | 107 (52.5) | .492 |
| BMI, classification, n (%)a | .547 | ||
| Underweight | 0 (0) | 4 (2) | |
| Normal | 32 (21.9) | 48 (23.5) | |
| Overweight | 56 (38.4) | 68 (33.3) | |
| Obese Class 1 | 32 (21.9) | 45 (22.1) | |
| Obese Class 2 | 16 (11) | 21 (10.3) | |
| Obese Class 3 | 10 (6.8) | 18 (8.8) | |
| Antibiotic Allergy, n (%) | |||
| Beta-lactam | 23 (15.8) | 31 (15.2) | .887 |
| Fluoroquinolone | 2 (1.4) | 4 (2) | 1.0 |
| Duration of symptoms PTA, days, median [IQR] | 2 [1–4] | 2 [1–4] | .975 |
| SIRS, ≥2, n (%) | 55 (37.7) | 80 (39.2) | .77 |
| qSOFA score, median [IQR] | 0 [0–1] | 0 [0–1] | .935 |
| Charlson comorbidity index, median [IQR] | 0 [0–2] | 1 [0–3] | .277 |
| High risk, n (%)b | 19 (13) | 27 (13.2) | .952 |
| Age ≥70 | 22 (15.1) | 40 (19.6) | .273 |
| Current malignancy | 6 (4.1) | 3 (1.5) | .172 |
| Significant CV disease | 0 (0) | 0(0) | 1.0 |
| Significant liver disease | 0 (0) | 2 (1) | 0.512 |
| Significant renal disease | 6 (4.1) | 9 (4.4) | .891 |
| Hypoalbuminemia | 15 (10.3) | 17 (8.3) | .535 |
| Source control ≥24 hours | 91 (62.3) | 125 (61.4) | .888 |
| History of MDRO | 0 (0) | 3 (1.5) | .269 |
| ID consulted, n (%) | 1 (0.7) | 10 (0.5) | 1.0 |
Abbreviations: BMI, body mass index; CV, cardiovascular; ID, Infectious Diseases physician; IQR, interquartile range; MDRO, multidrug-resistant organism; PTA, prior to admission; SD, standard deviation; qSOFA, quick Sequential Organ Failure Assessment; SIRS, systemic inflammatory response syndrome.
BMI classification based on World Health Organization.
High risk defined as ≥2 risk factors.
Main Outcomes
There was no difference observed between groups in the primary outcome of treatment-associated complication within 90 days (antipseudomonal 15.1% vs narrow-spectrum 11.3%, P=.296). A breakdown of the individual components of treatment-associated complications can be found in Table 3. The majority of patients in both groups were transitioned to oral therapy (antipseudomonal 81.5% vs narrow-spectrum 73%, P=.065); however, patients treated empirically with a narrow-spectrum regimen had a shorter median length of therapy compared with patients treated with an antipseudomonal regimen (9 days [interquartile range {IQR}, 5–12 days] vs 11 days [IQR, 8–14 days]; P<.001). Patient-centered outcomes are displayed in Table 3. There were no differences observed in hospital LOS, readmission within 30 or 90 days, C difficile infections within 30 days, or mortality between groups.
Table 3.
Treatment-Associated Complication Rates and Patient Centered Outcomes
| Antipseudomonal (n=146) | Narrow Spectrum (n=204) | P Value | |
|---|---|---|---|
| Any Treatment-Associated Complication Within 90 Days, n (%) | 22 (15.1) | 23 (11.3) | .296 |
| Postoperative infection | 15 (10.3) | 12 (5.9) | .942 |
| Recurrence of diverticulitis | 6 (4.1) | 8 (4.4) | .178 |
| 90-day mortality | 1 (0.7) | 0 | .417 |
| LOS, days, median [IQR] | 3 [2–5] | 3 [2–4.75] | .441 |
| In-hospital mortality, n (%) | 1 (0.7) | 0 (0) | .417 |
| Hospital Readmission 30-Day, n (%) | 15 (10.3) | 16 (7.8) | .430 |
| IAI related | 12 (8.2) | 14 (6.9) | 1.0 |
| Hospital Readmission 90-Day, n (%) | 29 (19.9) | 21 (10.3) | .012 |
| IAI related | 23 (15.8) | 15 (7.4) | .520 |
| Clostridiodes difficile 30 days, n (%) | 0.7 | 0 (0) | .417 |
Abbreviations: IAI, intra-abdominal infection; IQR, interquartile range; LOS, length of stay.
Multivariate Logistic Regression Analysis
Factors included in the model are shown in Table 4. Both surgical intervention (odds ratio [OR], 3.03; 95% confidence interval [CI], 1.12–8.19) and having a diagnosis of diverticulitis (OR, 6.24; 95% CI, 2.27–17.17) were found to be independently associated with an increase in treatment-associated complications within 90 days. Treatment with a narrow-spectrum regimen (OR, 0.75; 95% CI, 0.39–1.45) or meeting high-risk criteria (OR, 1.41; 95% CI, 0.59–3.38) were not independently associated with the primary outcome.
Table 4.
Multivariate Logistic Regression: Risk Factors for 90-Day Postoperative Complication
| Variable | Odds Ratio (95% CI) | P Value |
|---|---|---|
| Surgery | 3.0 (1.1–8.2) | .029 |
| Diverticulitis | 6.2 (2.3–17.2) | <.001 |
| Perforation or abscess | 1.2 (0.6–2.5) | .539 |
| Obesity | 1.6 (0.8–3.2) | .157 |
| High-risk IAI classification | 1.4 (0.6–3.4) | .437 |
| Narrow spectrum | 0.8 (0.4–1.5) | .398 |
Abbreviations: CI, confidence interval; IAI, intra-abdominal infection.
Subgroup Analyses
Data for low- and high-risk patients are summarized in Tables 5 and 6. For patients who met the criteria for low-risk CA-IAI, there was no difference observed in treatment-associated complication rates in patients who received antipseudomonal versus narrow-spectrum regimens (15% vs 9.6%, P=.154). Likewise, for patients who met the criteria for high-risk CA-IAI, there was no difference observed in treatment-associated complication rates in patients who received antipseudomonal versus narrow-spectrum regimens (15.8% vs 22.2%, P=.588). In addition, there was no difference observed in patient-centered outcomes except in the low-risk CA-IAI group where 90-day readmission occurred more frequently in patients who received an antipseudomonal regimen (18.9% vs 10.2%, P=.03).
Table 5.
Subgroup Analysis: Low-Risk Patients
| Outcomes | Antipseudomonal (n=127) | Narrow Spectrum (n=177) | P Value |
|---|---|---|---|
| Any Treatment-Associated Complication Within 90 Days, n (%) | 19 (15) | 17 (9.6) | .154 |
| Postoperative infection | 6 (4.7) | 9 (5.1) | .886 |
| Recurrence of diverticulitis | 13 (10.3) | 8 (4.5) | .112 |
| 90-day mortality | 0 (0) | 0 (0) | 1.0 |
| LOS, days, median [IQR] | 3 [2–5] | 3 [2–4] | .228 |
| In-hospital mortality, n (%) | 0 (0) | 0 (0) | 1.0 |
| Hospital Readmission 30-Day, n (%) | 14 (11.0) | 12 (6.8) | .192 |
| IAI related | 11 (8.7) | 11 (6.2) | .598 |
| Hospital Readmission 90-Day, n (%) | 24 (18.9) | 18 (10.2) | .030 |
| IAI related | 19 (15.0) | 13 (7.4) | .720 |
| Clostridiodes difficile 30 days, n (%) | 1 (0.8) | 0 (0) | .418 |
Abbreviations: IAI, intra-abdominal infection; IQR, interquartile range; LOS, length of stay.
Table 6.
Subgroup Analysis: High-Risk Patients
| Outcomes | Antipseudomonal (n=19) | Narrow Spectrum (n=27) | P Value |
|---|---|---|---|
| Any Treatment-Associated Complication Within 90 Days, n (%) | 3 (15.8) | 6 (22.2) | .588 |
| Postoperative infection | 1 (5.3) | 2 (7.4) | .772 |
| Recurrence of diverticulitis | 2 (11.1) | 4 (14.8) | 1.0 |
| 90-day mortality | 1 (5.3) | 0 (0) | .413 |
| LOS, days, median [IQR] | 4 [3–6] | 4 [3–6] | .534 |
| In-hospital mortality, n (%) | 1 (5.3) | 0 (0) | .413 |
| Hospital Readmission 30-Day, n (%) | 1 (5.3) | 4 (14.8) | .305 |
| IAI related | 1 (5.3) | 2 (7.4) | 1.0 |
| Hospital Readmission 90-Day, n (%) | 5 (26.3) | 3 (11.1) | .246 |
| IAI related | 4 (21.0) | 2 (7.4) | 1.0 |
| Clostridiodes difficile 30 days, (%) | 0 (0) | 0 (0) | 1.0 |
Abbreviations: IAI, intra-abdominal infection; IQR, interquartile range; LOS, length of stay.
Of the total study population, 203 (58%) patients underwent intervention for source control (antipseudomonal, n=83 vs narrow spectrum, n=120). Most patients had laparoscopic procedures in both groups (antipseudomonal, n=64 [77.1%] vs narrow spectrum, n=95 [79.2%]; P=.726). More patients who received an antipseudomonal regimen had open procedures (antipseudomonal, 16.9% vs narrow spectrum, 5%; P=.005), whereas patients who received narrow-spectrum therapy had more interventional radiology procedures (antipseudomonal, 6% vs narrow spectrum, 15.8%; P=.033). The operative note identified gross perforation or contamination in more patients who received antipseudomonal therapy (66.3% vs 51.7%, P=.039). One patient in the antipseudomonal group and 2 patients in the narrow-spectrum group required repeat surgical management for source control (P=1.0). The median duration of therapy for the patients who underwent surgical intervention was 7 days (IQR, 2–11 days). There were no differences observed in 90-day treatment-associated complications between groups in patients who underwent surgical management of CA-IAI (antipseudomonal 8.4% vs narrow spectrum 9.2%, P=.857). In addition, there was no difference in median hospital LOS (antipseudomonal, 3 days [IQR, 2–5 days] vs narrow spectrum, 3 days [IQR, 2–4 days]; P=.179) or hospital readmission within 30 days (antipseudomonal 9.6% vs narrow spectrum 6.7%, P=.44). More patients were readmitted within 90 days of index visit who received antipseudomonal therapy (antipseudomonal 19.3% vs narrow spectrum 9.2%, P<.037); however, when considering whether the readmission was related to the index intra-abdominal infection, there was no difference between groups (antipseudomonal 75% vs narrow spectrum 72.7%, P=.895). No episodes of C difficile within 30 days were reported in either group.
Of the 81 patients with uncomplicated appendicitis, 55 (67.9%) were treated with narrow-spectrum agents vs 26 (32.1%) with antipseudomonal therapy. Of the 55 patients treated with narrow-spectrum agents, 76.4% received 24- to 48-hour perioperative antibiotics; the remaining patients were medically managed and received 3–17 days of therapy. Of the 26 patients who received antipseudomonal therapy, 73.1% received 24–48 hours of perioperative prophylaxis; the remaining patients were medically managed and received 4–17 days of therapy. When patients with uncomplicated appendicitis were removed from the primary outcome, there was no difference in 90-day postoperative complication of the remaining 269 patients (narrow spectrum, n=149; antipseudomonal, n=120) analyzed (narrow spectrum, 20 [13.4%] vs antipseudomonal, 20 [16.7%]; P=.457).
Medical management was the primary strategy for 147 (42%) of the total study population (antipseudomonal, n=63 vs narrow spectrum, n=84). A breakdown of the types of infections in each group is available in Tables 7 and 8. The median duration of therapy for the patients who underwent medical management was 11 days (IQR, 9–14 days). There were no differences observed in 90-day treatment-associated complications between groups (antipseudomonal [17.5%] vs narrow spectrum [13.1%]; P=.463). In addition, there were no differences observed in hospital LOS, readmission within 30 or 90 days, or C difficile infections within 30 days between groups (Tables 7 and 8).
Table 8.
Subgroup Analysis: Medically Managed Outcomes
| Medically Managed Outcomes | Antipseudomonal (n=63) | Narrow Spectrum (n=84) | P Value |
|---|---|---|---|
| Treatment-associated complication, n (%) | 11 (17.5) | 11 (13.1) | .463 |
| Diverticulitis recurrence, n (%) | 15 (23.8) | 15 (17.9) | .178 |
| Infection Type | .435 | ||
| Uncomplicated diverticulitis, n (%) | 31 (49.2) | 37 (44) | |
| Complicated diverticulitis, n (%) | 24 (38.1) | 36 (42.9) | |
| Uncomplicated appendicitis, n (%) | 1 (1.6) | 5 (6) | |
| Complicated appendicitis, n (%) | 7 (11.1) | 6 (7.1) | |
| LOS, days, median [IQR] | 4 [3–5] | 3.5 [3–5] | .713 |
| In-hospital mortality, n (%) | 0 (0) | 0 (0) | 1.0 |
| Hospital readmission 30-day, n (%) | 7 (11.1) | 8 (9.5) | .753 |
| IAI related | 5 (7.9) | 6 (7.1) | 1.0 |
| Hospital Readmission 90-Day, n (%) | 13 (20.6) | 10 (11.9) | .149 |
| IAI related | 11 (17.4) | 7 (8.3) | .618 |
| Clostridiodes difficile 30 days, n (%) | 0 (0) | 1 (1.6) | .429 |
Abbreviations: IAI, intra-abdominal infection; IQR, interquartile range; LOS, length of stay.
DISCUSSION
This study sought to compare outcomes of adult patients treated for CA-IAI with antipseudomonal versus narrow-spectrum antibiotic regimens. Our findings demonstrate that there was no difference in treatment-associated complication rates or patient-centered outcomes between antipseudomonal or narrow-spectrum regimens. Although current national guidelines support narrow-spectrum regimens in low-risk CA-IAI, to our knowledge, there are no studies comparing outcomes of antipseudomonal versus narrow-spectrum antibiotic regimens in adult patients. However, these findings augment the limited number of published studies within the pediatric population that support the use of narrow-spectrum antibiotic regimens for the treatment of CA-IAI. St. Peter et al [9] were the first to compare a more narrow-spectrum, 2-drug combination, ceftriaxone and metronidazole, to an antipseudomonal, triple-drug combination, ampicillin with gentamicin and clindamycin, for surgical infection prophylaxis in 100 pediatric patients with perforated appendicitis who underwent laparoscopic appendectomy. The authors found no difference in 5-day postoperative abscess development between groups (narrow spectrum 40.8% vs antipseudomonal 32.6%, P=.60) or surgical wound infection (narrow spectrum 0% vs antipseudomonal 4.1%, P=.99). In addition, cost for antibiotic therapy was significantly more in the antipseudomonal group. Although this study was limited in that it only evaluated aminoglycoside-containing regimens for antipseudomonal therapy, Kronman et al [10] also evaluated narrow-spectrum versus antipseudomonal regimens for the treatment of appendicitis in 24984 children; all antipseudomonal agents were beta-lactam antibiotics. Antipseudomonal regimens were associated with significantly higher rates of treatment failure (30-day readmission for wound infection or repeat abdominal surgery) in children with complicated appendicitis (adjusted OR, 1.43; 95% CI, 1.06–1.93). No increase in risk was observed in uncomplicated appendicitis (adjusted OR, 1.32; 95% CI, 0.88–1.98). Hamdy et al [11] specifically evaluated 353 children with perforated appendicitis who received antipseudomonal antibiotics versus ceftriaxone and metronidazole. They similarly found no difference in 6-week postoperative complications between regimens (antipseudomonal 17.8% vs ceftriaxone/metronidazole 14.7%, P=.46). Finally, Cameron et al [12] compared the antipseudomonal regimen piperacillin/tazobactam with ceftriaxone and metronidazole or cefoxitin regimens in pediatric patients with uncomplicated appendicitis. The rates of surgical site infections and hospital revisits were similar between the 2 groups.
Antimicrobial resistance continues to be a global and public health concern [13]. The misuse and overuse of unnecessary, broad-spectrum agents continues to be a driving factor for antibiotic resistance [14]. A major concern with the overuse of antipseudomonal regimens in CA-IAI is that exposure may lead to selective pressure and resistant Pseudomonas strains that can affect the institutional microbiota [15]. Even a short course of unnecessary perioperative antibiotic exposure with antipseudomonal agents may result in significantly altered microbiota and increased risk for drug-resistant organisms [16]. Although the most recent Surgical Infection Society guidelines support the use of narrow-spectrum agents for patients with low-risk CA-IAI, antipseudomonal agents continue to be favored in many institutions. Lodise et al [4] recently evaluated national patterns of prescribing for CA-IAI to assess concordance with national guideline recommendations. They conducted a multicenter, retrospective, observational study evaluating treatment patterns among 46722 hospitalized adult patients and found that more than 75% of patients empirically received an antipseudomonal regimen. The most common agent prescribed was piperacillin/tazobactam (54%) followed by an antipseudomonal fluoroquinolone-containing regimen (20%). Despite rising rates of antimicrobial resistance of Enterobacterales to fluoroquinolones, they are commonly used as empiric therapy. The results of our study were consistent with their findings, with piperacillin/tazobactam being the most prescribed antipseudomonal agent, followed by fluoroquinolones. A further concern with the overuse of piperacillin/tazobactam is the emergence of ampicillin-resistant Enterococcus strains [17]. It is also important to note that we found no difference in patient outcomes of length of stay, hospital revisit, mortality, or development of C difficile between groups, which further supports the safety of using narrow-spectrum regimens for CA-IAI [8]. The results of our study support current guideline recommendations to avoid antipseudomonal regimens for empiric treatment of low-risk CA-IAI. In addition, our findings suggest that narrow-spectrum regimens may also be effective in a subset of high-risk CA-IAI patients and that future research should focus on this patient population to further identify significant risk factors for drug-resistant infections to limit the overuse of broad-spectrum regimens.
Antimicrobial stewardship programs may benefit from targeting educational and audit-and-feedback interventions toward CA-IAIs to limit the overuse of broad-spectrum antimicrobial agents. We found that patients who received antipseudomonal antibiotics were more likely to have open surgical procedures as well as perforation or contamination documented in their operative notes. We hypothesize that these patients may have been considered by the surgical team to be sicker and therefore require a broader-spectrum antimicrobial regimen. Despite this, neither the receipt of an antipseudomonal agent or being classified as a high-risk CA-IAI were independently associated with an increased risk for treatment-associated complication. In addition to focusing on the use of more narrow-spectrum regimens, our study demonstrated that focus is also needed on prescribed durations of therapy for CA-IAI. Although current literature supports durations of antibiotic therapy between 4 and 7 days for the majority of patients, we found that patients in our study were being treated for a median of 10 days, with patients receiving antipseudomonal agents being treated longer than those receiving narrow-spectrum regimens. We hypothesize that this difference in duration of therapy between groups may be due to more patients who received antipseudomonal agents having perforation or contamination noted within their operative notes. Finally, there is additional emerging evidence from randomized, controlled trials demonstrating that certain patients with uncomplicated, medically managed CA-IAIs had no differences in clinical outcomes when managed with supportive care alone versus with short courses of antibiotics. These data are important for surgical teams and antimicrobial stewardship programs to consider because they demonstrate that some patients with appendicitis and diverticulitis may not require antibiotic therapy, which can further limit antimicrobial resistance and adverse effects of antibiotics. Future research and guidance are needed focusing on the optimal patient to withhold antibiotics in CA-IAI [18, 19].
Our study has limitations that should be considered. First, we included patients from over a 6-year timespan; therefore, surgical management practices may have evolved during the study period, which is to be expected as new best practices emerge. Another limitation of our study is the possibility of missing treatment-associated complication or hospital readmission within 90 days in patients who were treated outside of our hospital, although the rates of treatment-associated complication and recurrence of diverticulitis found in our study were similar to rates estimated previously. In addition, confounding variables may have impacted the primary outcome or readmission rates; however, we attempted to control for these with logistic regression as well as further characterizing whether readmissions were related to the initial infection. We also chose to include both low-risk and high-risk patients in our study cohort; although we found no differences in outcomes between either risk group, the results of our high-risk patient population should be interpreted carefully because the sample size is small and we were likely underpowered to find a difference. Finally, as with all single-center and retrospective studies, there is concern for external validity as well as a strong reliance on documentation within the medical record. Although documentation practices did change over time, all data were collected by a single investigator to limit discrepancies with interpretation. Despite these limitations, our study findings are important and add to the limited available literature in demonstrating the role of narrow-spectrum agents in the treatment of adult patients with CA-IAI.
CONCLUSIONS
The results of our study support national guideline recommendations for using narrow-spectrum antibiotics to empirically treat adult patients with CA-IAI. By avoiding the use of unnecessary antipseudomonal antibiotics in CA-IAI, antimicrobial stewardship teams have the opportunity to limit antimicrobial resistance.
Acknowledgments
Potential conflicts of interest. All authors: No reported conflicts of interest. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest.
References
- 1. Sartelli M, Catena F, Abu-Zidan FM, et al. Management of intra-abdominal infections: recommendations by the WSES 2016 consensus conference. World J Emerg Surg 2017; 12:22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Humes DJ, Simpson J.. Acute appendicitis. BMJ 2006; 333:530–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Peery AF. Recent advances in diverticular disease. Curr Gastroenterol Rep 2016; 18:37. [DOI] [PubMed] [Google Scholar]
- 4. Lodise TP, Izmailyan S, Olesky M, Lawrence K.. An evaluation of treatment patterns and associated outcomes among adult hospitalized patients with lower-risk community-acquired complicated intra-abdominal infections: how often are expert guidelines followed? Open Forum Infect Dis 2020; 7:ofaa237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Centers for Disease Control and Prevention. Multidrug-resistant Pseudomonas aeruginosa. Available at: https://www.cdc.gov/hai/organisms/pseudomonas.html. Accessed 5 May 2021.
- 6. Pang Z, Raudonis R, Glick BR, et al. Antibiotic resistance in Pseudomonas aeruginosa: mechanisms and alternative therapeutic strategies. Biotechnol Adv 2019; 37:177–92. [DOI] [PubMed] [Google Scholar]
- 7. Solomkin JS, Mazuski JE, Bradley JS, et al. Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Clin Infect Dis 2010; 50:133–64. [DOI] [PubMed] [Google Scholar]
- 8. Mazuski JE, Tessier JM, May AK, et al. The surgical infection society revised guidelines on the management of intra-abdominal infection. Surg Infect (Larchmt) 2017; 18:1–76. [DOI] [PubMed] [Google Scholar]
- 9. St. Peter SD, Tsao K, Spilde TL, et al. Single daily dosing ceftriaxone and metronidazole vs standard triple antibiotic regimen for perforated appendicitis in children: a prospective randomized trial. J Pediatr Surg 2008; 43:981–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Kronman MP, Oron AP, Ross RK, et al. Extended- versus narrower-spectrum antibiotics for appendicitis. Pediatrics 2016; 138:e20154547. [DOI] [PubMed] [Google Scholar]
- 11. Hamdy RF, Handy LK, Spyridakis E, et al. Comparative effectiveness of ceftriaxone plus metronidazole versus anti-pseudomonal antibiotics for perforated appendicitis in children. Surg Infect (Larchmt) 2019; 20:399–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Cameron DB, Melvin P, Graham DA, et al. Extended versus narrow-spectrum antibiotics in the management of uncomplicated appendicitis in children: a propensity-matched comparative effectiveness study. Ann Surg 2018; 268:186–92. [DOI] [PubMed] [Google Scholar]
- 13. Centers for Disease Control and Prevention. Antibiotic/antimicrobial resistance. Available at: https://www.cdc.gov/drugresistance/index.html. Accessed 5 May 2021.
- 14. Dadgostar P. Antimicrobial resistance: implications and costs. Infect Drug Resist 2019; 12:3903–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Cobos-Trigueros N, Solé M, Castro P, et al. Acquisition of Pseudomonas aeruginosa and its resistance phenotypes in critically ill medical patients: role of colonization pressure and antibiotic exposure. Crit Care 2015; 19:218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Ramirez J, Guarner F, Bustos Fernandez L, et al. Antibiotics as major disruptors of gut microbiota. Front Cell Infect Microbiol 2020; 10:572912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Miller WR, Munita JM, Arias CA.. Mechanisms of antibiotic resistance in enterococci. Expert Rev Anti Infect Ther 2014; 12:1221–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Park HC, Kim MJ, Lee BH.. Randomized clinical trial of antibiotic therapy for uncomplicated appendicitis. Br J Surg 2017; 104:1785–90. [DOI] [PubMed] [Google Scholar]
- 19. Daniels L, Ünlü Ç, de Korte N, et al. ; Dutch Diverticular Disease (3D) Collaborative Study Group. Randomized clinical trial of observational versus antibiotic treatment for a first episode of CT-proven uncomplicated acute diverticulitis. Br J Surg 2017; 104:52–61. [DOI] [PubMed] [Google Scholar]