Abstract
Low back pain arising from disc degeneration is one of the most common causes of limited function in adults. A number of tissue engineering strategies have been used to develop composite tissue engineered total disc replacements to restore native tissue structure and function. In this study we fabricated a composite engineered disc based on the combination of a porous polycaprolactone (PCL) foam annulus fibrosus (AF) and a hyaluronic acid (HA) hydrogel nucleus pulposus (NP). To evaluate whether native tissue cells or mesenchymal stem cells (MSCs) would perform better, constructs were seeded with native AF/NP cells or with MSCs in the foam and/or gel region. Maturation of these composite engineered discs was evaluated for 9 weeks in vitro culture by biochemical content, histological analysis and mechanical properties. To evaluate the performance of these constructs in the in vivo space, engineered discs were implanted into the caudal spines of athymic rats for 5 weeks. Our findings show that engineered discs comprised of AF/NP cells and MSCs performed similarly and maintained their structure after 5 weeks in vivo. However, for both cell types, loss of proteoglycan was evident in the NP region. These data support the continued development of the more clinically relevant MSCs population for disc replacement applications.
Statement of Significance
A number of tissue engineering strategies have emerged that are focused on the creation of a composite disc replacement. We fabricated a composite engineered disc based on the combination of a porous foam AF and a HA gel NP. We used these constructs to determine whether the combination of AF/NP cells or MSCs would mature to a greater extent in vitro and which cell type would best retain their phenotype after implantation. Engineered discs comprised of AF/NP cells and MSCs performed similarly, maintaining their structure after 5 weeks in vivo. These data support the successful fabrication and in vivo function of an engineered disc composed of a PCL foam AF and a hydrogel NP using either disc cells or MSCs.
Keywords: Intervertebral disc degeneration, Disc cells, Mesenchymal stem cells, Tissue engineering, Total disc replacement
1. Introduction
Low back pain is one of the most common causes of limited function in adults, and intervertebral disc (IVD) degeneration is considered to be a major contributing factor [1]. IVD degeneration occurs as a result of a variety of factors; ageing, high mechanical loads, changes in extracellular matrix composition, and poor vascularity are all associated with degeneration [2,3]. Despite its prevalence, current treatment options for IVD degeneration are limited, favoring either conservative physical therapy and pain management or surgical methods to fuse the motion segment. Neither approach restores native tissue structure and function, and so a number of tissue engineering strategies have emerged that focus on the creation of composite tissue engineered total disc replacements [4-6]. While early versions of these implants showed promise in vitro, most were limited in their mechanical properties, and it remains unclear whether they can restore long term native function in vivo [7,8]. Therefore, further improvements in engineered disc properties, such as cell and scaffold types, will be required for the next generation of IVD replacements.
The native IVD is composed of three component structures: the nucleus pulposus (NP), the annulus fibrosus (AF) and the vertebral end-plates. The central region of the disc is called the NP, a proteoglycan-rich gelatinous structure. Surrounding the NP is the AF, composed primarily of a collagen type I-rich matrix that is organized into a lamellar architecture [9]. The AF resists radial expansion of the NP due to osmotic swelling and axial compression during loading of the spine [10]. During degeneration, damage to the AF or a decrease in proteoglycan content in the NP results in dehydration of the NP and a loss of disc height. This loss of disc height and load bearing function may impinge on adjacent spinal nerves, resulting in disability and pain [2,11].
In response to these degenerative processes and their prevalence, a number of biological therapies have been proposed for the treatment of disc degeneration. These proposed interventions are predominantly cell based, and several cell types have been proposed, including NP cells (NPCs) [12,13], chondrocytes [14,15] and stem cells [16,17]. Mesenchymal stem cells (MSCs) are of particular interest as they can be readily isolated in an autologous fashion, possess a robust self-renewal capacity, and can be differentiated into lineages that recapitulate aspects of the NP phenotype [17-20]. MSCs have been used in preclinical studies involving rat, rabbit, goat and porcine animal models [21-24]. For instance, in a recent study [22], MSCs were transplanted to the nucleus pulposus of degenerative discs in a rabbit model, and resulted in increased proteoglycan content and expression of matrix-related genes compared to control discs. David et al. also recently showed that intradiscal MSC treatment in an ovine model improved extracellular matrix production and disc height, and reduced the severity of radiological and histological degeneration metrics [25].
In addition to direct cell therapies, a variety of biomaterials and polymeric scaffolds have been developed for cell-based engineered AF and NP tissues, and their combination as a whole disc construct. For example, several studies have sought to promote organized AF-like tissue using self-organizing collagen-based gels [5], lamellar nanofibrous scaffolds [26], or mesh scaffolds composed of silk protein [27] or decellularized for the AF [28]. Others have encapsulated cells within various hydrogels, including agarose [29], fibrin/hyaluronic acid (HA) [30], carboxymethylcellulose [31], or alginate gels to generate NP-like tissue constructs [32]. Increasingly, these disparate scaffolds have been combined to reconstruct the entirety of the IVD in an engineered composite. For example, we recently fabricated disc-like angle ply structures (DAPS) for total disc arthroplasty that include a PCL electrospun nanofibrous AF and a HA hydrogel core NP [33]. Despite the promise of these engineered discs, we noted poor cell infiltration into each layer of the AF and lack of integration between the NP and AF. Alternative material formulations, such as porous foams, may encourage cell infiltration and more robust integration between the NP and AF components.
It is also still not clear which cell type is most appropriate for each disc region. While native disc cells are the most appropriate in terms of phenotype, mesenchymal or other progenitor cells are more readily available for regenerative applications. Additionally, a number of recent studies have shown that there is a ‘co-culture’ effect, where native tissue cells can impact the differentiation and function of various stem cell populations [34-36]. To examine these concepts, in this study we fabricated a composite engineered disc based on the combination of a porous PCL foam AF and a HA gel NP. These constructs were seeded with either native AF/NP cells (AFCs/NPCs) or with MSCs and were matured in vitro and then evaluated in vivo in a rat tail model of disc replacement [33]. In vitro analysis included the evaluation of biochemical composition, biomechanical properties, and extracellular matrix composition over long term culture in a chemically-defined medium formulation. In vivo analysis included assays to evaluate the integration and function of the engineered IVD in situ in terms of disc height, hydration level, histological features, and mechanical function post-implantation.
2. Materials and methods
2.1. Research design
The overall study design is shown in Fig. 1. AFCs, NPCs and MSCs were harvested from bovine tissue and expanded in monolayer. AFCs or MSCs were seeded onto PCL foam constructs to generate the AF regions, whereas NPCs or MSCs were encapsulated in HA hydrogels to form the NP regions. Four types of engineered discs were fabricated: (1) AFC AF region and NPC NP region, (2) AFC AF region and MSC NP region, (3) MSC AF region and NPC NP region, (4) MSC AF region and MSC NP region. The AF and NP regions were cultured separately in a chemically-defined medium containing TGF-β3 and combined at 2 weeks. At regular intervals over 9 weeks, glycosaminoglycan (GAG) and collagen content, compressive mechanical properties, and histological features (Al-cian blue staining of sulfated GAGs and picrosirius red staining of collagens) were evaluated.
Fig. 1.
A scheme of tissue engineering for intervertebral discs. AFCs or MSCs were seeded onto the PCL foam at a density of 2 × 106 cells/construct, whereas NPCs or MSCs were encapsulated in HA at a density of 20 × 106 cells/ml. AF and NP regions were cultured separately in chemically-defined media and combined at 2 weeks. The tissue engineered intervertebral disc constructs were harvested and evaluated at regular intervals over 9 weeks for in vitro study. Additionally, AFCs/NPCs and MSCs/MSCs seeded constructs were implanted into the rat caudal disc space after 5 weeks of priming and after 5 weeks in vivo, constructs were evaluated.
In an additional arm of the study, AFC/NPC seeded and MSC/MSC seeded constructs were implanted into the rat caudal disc space after 5 weeks of pre-culture or priming. We commonly utilize bovine cells as a starting source, based on the robust growth, differentiation capacity, and matrix formation by these cells. Given the source is bovine, we implant these constructs into the tails of athymic rats, so as to minimize the response to xenogenic cell sources. Rats were returned to normal cage activity and euthanized 5 weeks after implantation. Evaluation of functional outcomes such as disc height and hydration level, using μCT and MRI, respectively, and histological assessment and mechanical tests were performed.
2.2. Study 1: in vitro culture of engineered discs
2.2.1. Fabrication of poly(ε-caprolactone) (PCL) foams and hyaluronic acid (HA) hydrogels
Porous PCL foams were fabricated by salt-leaching to form the AF regions of the engineered discs. PCL (Sigma Aldrich; St Louis, MO, USA) was dissolved in chloroform (EMD Millipore; Billerica, MA, USA) at a 20% (w/v) concentration. NaCl (Sigma Aldrich) particles were sieved to yield a range of size of ~106 μm and loaded and mixed into PCL solution at a PCL/NaCl mass ratio of 1:4 (%W/W). The mixture was poured into a Teflon mould and solvent evaporation and solidification was allowed to proceed for 48 h. The resultant solidified PCL sheet with entrapped salt particles was 1.5 mm in height. Individual plugs were extracted using a 4 mm biopsy punch for the other diameter and a 2 mm biopsy punch for the inner diameter; this geometry approximates that of the rat caudal disc. The NaCl particles were subsequently removed from the foams in distilled water for 48 h, and the construct was lyophilized. After characterization, the porous PCL foams were observed by scanning electron microscopy (SEM). For preparation, the samples were vacuum dried at room temperature and mounted onto a sample holder with conductive adhesive tape. Images were taken after sputter-coating with gold/palladium (Au/Pd) for 40 mins, and viewed under SEM at an accelerating voltage of 10 kV. To form the NP regions of the engineered discs, methacrylated HA (MeHA) was produced by reacting 65 kDa HA (Lifecore; Chaska, MN, USA) with methacrylic anhydride (Sigma Aldrich) as previously described [30]. The degree of methacrylation was ~25%, as assessed by proton nuclear magnetic resonance [30]. Lyophilized MeHA was sterilized by exposure to a biocidal UV light for 15 min. Prior to cell encapsulation, the macromer was dissolved at 1% w/vol in sterile phosphate-buffered saline (PBS) with addition of 0.05% w/vol of the photoinitiator, Irgacure 2959 (2-methyl-1-[4-(hydroxyethoxy)phenyl]—2-methyl-1-propanone; Ciba-Geigy; Tarrytown, NY, USA).
2.2.2. Cell isolation and culture of AFCs, NPCs, and MSCs
AF and NP tissue was isolated from four adult bovine caudal discs purchased from a local slaughterhouse. AFCs and NPCs were isolated from minced tissue via digestion for 1 h with 2.5 mg/mL pronase followed by further digestion with 0.5 mg/mL collagenase at 37°C until the tissue was fully digested (NP: ~4 h, AF: ~18 h). After digestion, the cell suspension was filtered through a 70 μm strainer. Isolated cells were expanded in high-glucose Dulbecco’s modified Eagle’s medium (DMEM) containing 10% foetal bovine serum (FBS) and 1% penicillin–streptomycin–fungizone (PSF). Bovine bone marrow-derived MSCs were isolated from the tibia and femurs of 3–6 month old calves (Research 87, Boylston, MA, USA). Typically, six separate marrow isolations (minimum of three animals) were carried out. Trabecular regions were removed with a saw and agitated in a solution of high glucose Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 2% penicillin/streptomycin/Fungizone (PSF) and 300 U/mL of heparin. The resulting solution was centrifuged (5 min at 300 × g) and plated into 10 cm tissue culture plates. Cultures were maintained and expanded through passage 2 in high glucose DMEM supplemented with 1% PSF and 10% foetal bovine serum (FBS) changed twice weekly until confluence [30,33].
2.2.3. Cell seeding for 3D culture
Prior to cell seeding, PCL foam AF regions were rehydrated in decreasing concentrations of ethanol (100%, 70%, 50%, 30%), UV sterilized for 30 min, and coated with 20 μg/ml solution of fibronectin for 16 h. A 50 μL aliquot containing 2 million cells (either AFCs or MSCs) were seeded directly onto each side of the PCL foam, followed by 1 h of incubation to allow for cell attachment. Either NPCs or MSCs were seeded into 1% w/vol MeHA solutions at a density of 20 million cells/ml [30]. Constructs were formed from gel slabs using a 2 mm biopsy punch (Dermal Biopsy Punch, Miltex Inc, Bethpage, NY). AF and NP regions were cultured separately in a chemically-defined medium containing TGF-β3 [30,33] (10 ng/ml; R&D Systems, Minneapolis, MN, USA) (1 ml per construct) and combined at 2 weeks and then cultured for up to 9 weeks. Media were changed three times weekly.
2.2.4. Mechanical testing
For the in vitro study, unconfined compression tests were performed at weeks 4 and 8 using an electromechanical testing system (Instron 5542, Instron, Norwood, MA) to determine construct mechanical properties as a function of time and cell type. For the normalization of mechanical properties, sample cross-sectional area was measured from digital images with a custom MATLAB program (The MathWorks Inc., Natick, MA) and sample height was defined as the platen-to-platen distance after applying a pre-load to each construct. Testing took place in a PBS bath and the protocol consisted of cyclic compression (20 cycles, 0 to −3 N/−0.24 MPa, 0.5 Hz) followed by creep (−3 N/−0.24 MPa, 10 min hold) [33]. Strain was calculated as the displacement divided by sample height; stress was calculated as force divided by cross-sectional area. The linear region modulus was calculated from the 20th cycle of the stress-strain response using a bilinear model [33]. After testing, constructs were frozen at −20 °C for subsequent biochemical evaluation.
2.2.5. Biochemical composition
After mechanical testing, samples were divided into AF and NP regions, weighed and digested with proteinase K at 60°C for 24 h [37]. Glycosaminoglycan (GAG) content was quantified using the 1,9-dimethylmethylene blue (DMMB) dye binding assay with chondroitin-6 sulfate as a standard, and collagen content quantified using the orthohydroxyproline (OHP) assay, assuming a ratio of OHP to collagen of 1:7.14 [38,39]. Data are reported as a fraction of the wet weight.
2.2.6. Histological analysis of matrix distribution
At each time point, specimens were fixed and embedded in paraffin, and serial sections of 8 μm were cut using a microtome. Sections were stained with Alcian blue or picrosirius red to visualize proteoglycans and collagens, respectively [40]. Staining was visualized and imaged by light microcopy (Eclipse 90i, Nikon, Tokyo, Japan).
2.3. Study 2: in vivo model
2.3.1. Surgical implantation of engineered disc into the rat caudal spine
Engineered discs were implanted into the caudal spines of athymic retired male breeder rats (Foxn1rnu, Harlan Laboratories, Inc., Indianapolis, IN) in accordance with local institutional guidelines as previously described [33]. This was approved by the Corporal Michael J. Crescenz VA Medical centre IACUC (protocol #: 01,252). After rats were anesthetized, the caudal (Cd) Cd8 and Cd9 vertebral bodies were marked under fluoroscopy (Orthoscan HD, Orthoscan, Inc., Scottsdale, AZ). To implant the engineered disc, two Kirschner wires were passed laterally through both the Cd8 and Cd9 vertebrae and an Ilizarov-type external fixation device was secured to these wires to immobilize the tail segment. A skin incision was made dorsally, the native Cd8/Cd9 disc was removed with a scalpel and micro-curette, and an engineered disc was implanted into the disc space. The rats were maintained up to 5 weeks in normal cage conditions. Prophylactic treatments were performed for post-surgical management including infection and pain [41].
2.3.2. Radiographic analysis of disc height
Rat caudal spines were imaged fluoroscopically pre-operatively, immediately post-operatively, and at euthanasia to evaluate implant stability. As previously described [41], the disc height index (DHI) [41,42] which is a standard technique used to normalize disc height to vertebral body length, was determined from lateral fluoroscopic images using a custom Matlab program. The results were expressed as a percentage of pre-operative DHI (%DHI0).
2.3.3. Imaging: μCT and MRI
Three-dimensional reconstructions of the vertebrae adjacent to the implant site were obtained from μCT scans (vivaCT 75, SCANCO Medical AG, Bruttisellen, Switzerland) with an isotropic resolution of 20.5 μm in order to visualize any bony remodelling or potential intervertebral fusion. Magnetic resonance imaging (MRI) was performed at 4.7T on intact rat tail discs and AF/NP and MSC/MSC constructs 5 weeks following implantation. A multi-echo-multi-spin (MEMS) sequence was used to acquire coronal slices for quantitative T2 mapping (three 0.5 mm thick slices, 16 echoes, TE/TR = 7.84 ms/2000 ms, FOV = 15 × 15 mm2, matrix = 128 × 128, 4 averages). Population average T2 maps and the mean NP T2 value of each experimental group (adjacent control, AF/NP construct, MSC/MSC construct) were generated as previously described [33,43].
2.3.4. Mechanical, histological, and immunohistological analyses
Bone-engineered disc-bone motion segments were tested in compression using an Instron 5948 and an optical displacement tracking technique as in [33]. To do so, the skin around the motion segment was carefully removed, while adjacent muscle and tendons were left intact and the ventral bony surface adjacent to the disc space was cleared of soft tissue using a micro-curette. Ink spots drawn onto the vertebral surfaces directly above and below the disc space allowed for pattern tracking. A digital camera (acA3800-14um; Basler AG, Ahrensburg, Germany) fit with a close-focusing macro video lens (Zoom 7000; Navitar Inc., Rochester, NY) and custom software were used to record images during testing. Motion segments were potted in low melting temperature alloy and testing was conducted in a PBS bath. We applied 3 N compression, which when normalized to the average area of the rat tail disc, equates to 0.24 MPa, or half body weight. Texture tracking was performed in Matlab [43,44] and the 20th cycle of stress-strain was analysed using a bilinear fit routine. Linear region modulus was calculated as described above. In addition, the toe region modulus, the strain at the transition between the toe and linear region, and the maximum strain were calculated as these are related to the degree of integration of the engineered disc into the adjacent vertebrae [45]. Mechanical properties were normalized as described above using the pre-implantation geometry (outer diameter 4 mm, height 1.5 mm).
All segments from native IVDs, and AF/NP, and MSC/MSC constructs were fixed in buffered formalin and decalcified in formic acid. Segments were sectioned at 10 μm thickness in the dorsoventral plane on a paraffin microtome. Sections were double stained (Alcian blue/picrosirius red). For immunohistochemical detection of collagen type I (MAB3391, Milipore, Billerica, MA, USA), collagen type II (II-II6B3, Developmental Studies Hybridoma Bank (DSHB); Iowa City, IA, USA), and collagen type X (X-AC9, DSHB) sections were incubated in proteinase K at room temperature for 4 mins followed by blocking with 10% NGS for 30 mins at RT. Primary antibody was applied at a concentration of 10 μg/mL at 4°C overnight, followed by a one hour incubation with secondary antibody at RT. Sections were visualized using a DAB chromagen reagent (DAB150 IHC Select; EMD Millipore; Bellirica, MA, USA) according to manufacturer’s protocol. Sections were imaged under brightfield (Eclipse 90i).
2.4. Statistical analysis
Statistical analyses were performed by two-way analyses of variance (ANOVA) using SYSTAT 10.3 (Systat Software; Point Rich-mond, CA, USA). All results are expressed as the mean ± the standard deviation. Significance was set at p ≤ 0.05, with comparisons between groups carried out using Tukey’s post-hoc testing.
3. Results
3.1. Characterization of PCL foam AF regions
SEM micrographs (Fig. 2) illustrate the morphology of the porous PCL scaffolds used for the AF regions of the engineered disc. On a macroscopic scale, scaffolds were characterized by a multimodal pore structure with distinct pore architectures (Fig. 2A). Pores appeared to be homogeneously distributed in both the inner and outer portions of the structure (Fig. 2B and C). Higher magnification views showed good pore interconnection, with pore size varying from tens to hundreds of microns, as would be expected based on the staring size of the salt particulate (Fig. 2D).
Fig. 2.
SEM images of porous PCL foam for AF regions prepared by salt leaching of mixtures of PCL and salt particles. (A) Overview of the PCL foam (scale = 1 mm) (B) the inner surface (scale = 500 μm) (C) the outer surface (scale = 500 μm) (D) higher magnification image of the surface (scale = 100 μm).
3.2. Biochemical composition and ECM distribution in engineered discs
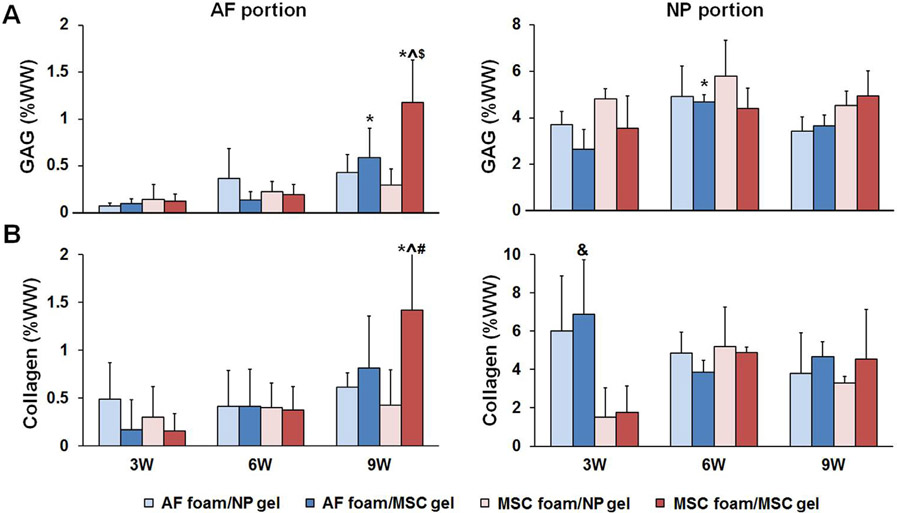
Over the course of 9 weeks, construct biochemical composition and histological features were analysed (Fig. 3). In the AF region, at 6 weeks, few differences were observed between groups (Fig. 3A). By 9 weeks, however, GAG content in AF/MSC and MSC/MSC groups significantly increased compared to week 3. GAG content in the AF region of the MSC/MSC group was significantly higher than that of the other groups at week 9. In the NP region, GAG content had reached >2% by 3 weeks, with no significant differences amongst groups. There was a modest increase in GAG content in all groups with time in culture (Fig. 3A). Collagen content in the AF region remained relatively low for all groups with culture (<1%), with the exception of the AF region of the MSC/MSC group, which showed a significant increase by week 9 (reaching ~1.5% collagen by wet weight). In the NP region, collagen content rose quickly to values ranging between 2–6% wet weight by week 3. Interestingly, at week 3, collagen content in the AF/MSC group was significantly higher than that in either the MSC/NP or MSC/MSC group in the NP region (Fig. 3B).
Fig. 3.
Biochemical composition of AFCs or MSCs seeded onto the PCL foam at a density of 2 × 106 cells/construct and NPCs or MSCs encapsulated HA constructs at a density of 20 × 106 cells/ml after 3, 6 and 9 weeks of in vitro culture. (A) GAG content per wet weight of AF region and NP region, n = 4~5. (B) Collagen content per wet weight of AF region and NP region, n = 4~5. (* indicates p < 0.05 vs. 3 W within same group; ^ indicates p < 0.05 vs. 6 W within same group; $ indicates p < 0.05 vs. AF/NP, AF/MSC and MSC/NP within 9 W; # indicates p < 0.05 vs. MSC/NP within 9 W; & indicates p < 0.05 vs. MSC/NP and MSC/MSC within 3 W).
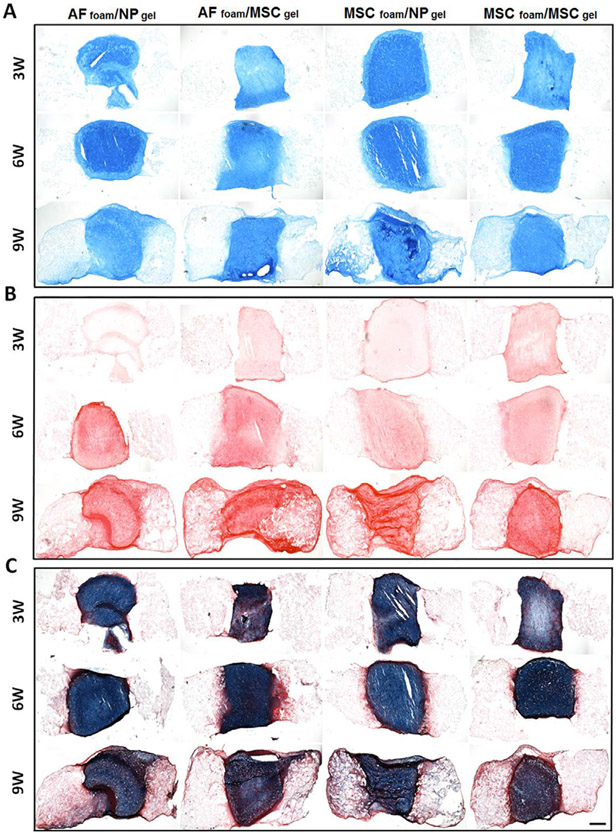
In terms of matrix distribution, by 3 weeks, the NP region of all the groups stained intensely for proteoglycans, while collagen staining in the NP region began lower but increased with culture duration. In the AF region, staining gradually increased with time, though to a lesser extent than in the NP (Fig. 4A and B). Costaining with Alcian blue and picrosirius red showed that the NP region was well-connected to the AF and that collagen was more heavily deposited in the AF region adjacent to the NP region at 6 weeks (Fig. 4C).
Fig. 4.
Histological staining of AF/NP, AF/MSC, MSC/NP and MSC/MSC engineered disc with time in-vitro culture. (A) Alcian blue, (B) picrosirius red, and (C) Alcian blue/picrosirius red staining (Scale = 500 μm).
3.3. Mechanical properties of engineered discs
Mechanical properties of intact engineered discs were assessed at 4 and 8 weeks. At 4 weeks, only the AF/MSC group was significantly greater than control levels (acellular engineered disc, p < 0.05). By 8 weeks, both the AF/NP and AF/MSC groups were significantly stiffer than acellular controls, with the AF/MSC group reaching a compressive modulus >2.8 MPa, which was significantly higher than the MSC/MSC group (p < 0.05, Fig. 5).
Fig. 5.
Compressive mechanical modulus of AF/NP, AF/MSC, MSC/NP and MSC/MSC engineered disc at weeks 4 and 8 of in vitro culture. (* indicates p < 0.05 vs. control; & indicates p < 0.05 vs. MSC/MSC within 8 W, n = 4~5 per group per time point, dashed line indicates acellular engineered disc).
3.4. Engineered disc implantation into the rat caudal spine
After 5 weeks of priming, engineered discs were implanted into the Cd8/Cd9 disc space in an athymic rat model (Fig. 6A). For this, either all disc cell constructs (AF/NP) or all stem cell constructs (MSC/MSC) were utilized. Immediately after implantation, an increase in disc height index (DHI) was noted on fluoroscopy images, as the constructs were press-fit into place (Fig. 6B). After 5 weeks of implantation, DHI [41] for remained significantly greater (as a result of external fixation) than pre-operative levels, with only small differences between groups (Fig. 6C, Supplemental Fig. S1).
Fig. 6.
Implantation of the engineered disc of MSC/MSC and AF/NP into the rat caudal spine. (A) Intraoperative images of implanted engineered disc into the Cd8/Cd9 disc space. (B) Fluoroscopy images of the vertebral bodies of pre or post-operation. (C) Three-dimensional μCT reconstructions for MSC/MSC and AF/NP engineered disc after 5 weeks implantation. Implanted discs of MSC/MSC and AF/NP did not result intervertebral fusion. Scale = 2 mm.
3.5. Histological appearance and mechanical properties of implanted discs
At five weeks post-implantation, both AF/NP and MSC/MSC engineered discs were resident in the disc space (Fig. 7A). Alcian blue/picrosirius red staining showed abundant collagen in the engineered disc, though little proteoglycan remained in the NP region in either group; however, type II collagen staining was intense and localized to the NP at this time point. There was little to no staining for either type I or X collagen in the NP region in either group after 5 weeks of implantation (Fig. 7A). After 5 weeks of implantation, there were no differences in the mechanical properties of engineered discs between MSC/MSC and AF/NP groups, in either the transition or maximum strains and the compressive modulus in toe region or linear region (p > 0.05, Fig. 7B). The compressive modulus in the linear region reached 4.4 and 2.8 MPa for MSC/MSC and AF/NP implants, respectively. These values were not significantly different from the linear region modulus of the native disc tested similarly [33] (dashed lines, Fig. 7B).
Fig. 7.
Histological appearance and mechanical properties of implanted engineered discs with native disc after 5 weeks. (A) Histology double staining (alcian blue and picrosirious red) and immunostaining (Type I, II and X collagen) after 5 weeks implantation. Scale = 500 μm. (B) Mechanical properties for vertebra-engineered disc-vertebra. Mechanical properties of MSC/MSC and AF/NP constructs were not significantly different with native discs. (dashed line indicates that properties of native disc).
3.6. T2-weighted MRI of implanted discs and native controls
Five weeks following implantation of engineered disc constructs, quantitative MRI T2 mapping was performed. The T2 relaxation constant in the disc has been previously shown to correlate with water and proteoglycan content [46]. This analysis showed that implanted discs had a similar structure to native discs (Fig. 8A and B). The NP T2 constant in the MSC/MSC group was significantly less than the native discs (p = 0.009). Similarly, the NP T2 constant of NP/AF constructs were reduced relative to native discs, however, though this difference was not significant (Fig. 8C).
Fig. 8.
5 weeks following implantation of constructs of MSC/MSC and AF/NP engineered disc in the rat caudal spine. (A) T2 MRI images of implanted discs had a similar structure to native discs. (B) T2 mapping showed reduced signal in the NP for both groups compared to native discs. (C) Quantification at 5 weeks after implantation. No significant difference between AF/NP group and native discs (*:p = 0.009 vs. adjacent control, n = 16 for adjacent control, n = 4 for MSC/MSC, n = 3 for AF/NP).
4. Discussion
In this study, we demonstrated that a tissue engineered disc composed of a PCL foam AF region and a hydrogel NP region could be fabricated, matured in vitro, and implanted and maintained in the rat caudal spine. The engineered disc showed progressive tissue formation with time, as indicated by the increasing quantity of ECM deposition. Engineered discs comprised of AFCs/NPCs and MSCs performed similarly, maintaining their structure and mechanical properties after 5 weeks in vivo, though a loss of proteoglycan was evident in the NP region for both groups. This suggests that, following 5 weeks of implantation, water and proteoglycan content are less than in the native disc, perhaps reflecting the inflammatory nature of the operative site and unwanted remodelling post-implantation. This finding is consistent with other studies involving engineered discs of other formulations implanted into the rat tail [33].
While a number of studies have introduced engineered composite discs [4-6,47], one issue with these formulations relates to the poor integration between the AF and NP regions [48,49]. In this study, gross and histological morphology indicated excellent integration between the AF and NP regions during 9 weeks of in-vitro culture and after in vivo implantation. After 5 weeks of implantation, engineered discs also showed abundant collagen in the disc, but little proteoglycan in the NP region, despite the intense staining for type II collagen in the NP at the time point. This finding is consistent with our previous studies [33] using other whole engineered disc formulations. Other studies showed that there were negative effects with this transition from the hospitable in vitro culture environment to the harsh in vivo environment. In addition, it has been reported that the pre-implantation characteristics of engineered tissues can be lost after short periods of in vivo implantation [50]. For MSC-based tissue engineering, it may be necessary to improve priming and pre-treatment procedures to promote advanced features of the NP/AF phenotype to further successful translation of stem cell technologies.
In this study, we also employed MSCs as an alternative cell source to fabricate the engineered discs and explored various combinations of MSCs taking the place of either or both the AFCs and NPCs. Experimental MSC transplantation therapies have been reported for the treatment of a variety of musculoskeletal conditions, including the articular cartilage, and the induction of articular chondrocytes from MSCs in vitro is well established [51]. In this study, we found no differences in terms of the histological appearance, mechanical properties, and the NP T2 constant of engineered disc that were fabricated with either all MSCs (MSC/MSC group) compared to those made up of AFCs/NPCs after 5 weeks of implantation.
In addition to the appropriate cellular phenotype and matrix composition, engineered implants for the IVD should have appropriate mechanical properties [33,52] in order to support the large spinal loads (i.e. tension, torsion, compression or combinations) that will be experienced after implantation. Ideally, cell-seeded constructs would gradually increase in mechanical properties with cultures as cells within the scaffold secret large amounts of ECM [53]. Findings in this study suggest, however, that the large majority of the mechanical properties of the implant were governed by the PCL foam itself. The compressive modulus of the combined scaffolds showed no differences between AF/NP seeded constructs and MSC/MSC seeded constructs during in-vitro culture over 8 weeks, despite the robust accumulation of extracellular matrix. Similarly, after implantation, the mechanical properties of the engineered disc were similar in MSC/MSC and AF/NP constructs, with the transition and maximum strains and compressive modulus in the toe region and linear region being comparable across groups (p > 0.05). Additional, longer term studies will need to be performed to determine whether, once the PCL foam begins to biodegrade in vivo, the new matrix has sufficient mechanical integrity to sustain physiologic loading.
In addition to mechanical properties, we used T2 MRI imaging to evaluate the development and maintenance of disc-like properties in our IVD implants. MRI is commonly used to examine the morphological features and composition of cartilage, disc, and other tissues in the clinic [46,54]. T2 mapping, in particular, is a promising method that can quantify the water content in tissues such as cartilage and IVD [46]. Our findings demonstrate that, at the time of implantation, discs had a similar MRI T2 characteristics as the native disc at 5 weeks. T2 mapping also showed, however reduced signal in the NP for both groups, indicative of some loss in hydration. To address this, we may need to include an endplate, which forms the interface between the disc and vertebral body and plays crucial role in nutrient supply and homoeostasis of IVD [55]. For instance, we recently showed [33] that an entire IVD unit including both a NP, an AF and an adjacent bony endplate preserved the structure and composition of the NP region to a greater extent.
This study is not without limitations. It is possible that there may be some differential response based on sex as a biological variable, both in terms of the donor cells and the host into which the implant is placed. This was not considered here, but is a point for consideration in future studies as we scale this technology up to larger animal species. Another limitation is that the fixator was left in place for the duration of this study. This likley protects the cells within the implant from mechanical stimulation and may contribute to a lack of robust ECM production. For this consideration, we have current studies under way examining the impact of fixator removal and remobilization in long term studies. Finally, it should be noted that we did not carry out a sham (no implant) surgery or carry out implantation of acellular implants in the current study. The decision to not include these groups was based on our past work which showed boney fusion in the absence of cells [44]. Specifically, when cells are not present (with no implant or with a cell-free implant), we observed fusion occurring in most cases. For this reason, we only examined cell-based implants in the current study.
5. Conclusion
Here, we fabricated and characterized the maturation of tissue-engineered discs in vitro and after implantation in the rat caudal spine. Engineered discs comprised of AFCs/NPCs and MSCs performed similarly, maintaining their structure after 5 weeks in vivo, though loss of proteoglycan was evident in the NP region regardless of cell type. These data support the continued development of a tissue engineered disc based on the more clinically relevant MSC population for future translational studies.
Supplementary Material
Acknowledgments
This work was funded by the Department of Veterans Affairs (Grant numbers I01 RX001321, IK2 RX001476, I01 RX002274, IK1 RX002445, IK6 RX003416), and the National Institutes of Health, through the Penn centre for Musculoskeletal Disorders (Grant number AR069619). We have no additional disclosures.
Footnotes
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Supplementary materials
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.actbio.2020.05.039.
References
- [1].Kauppila LI, Atherosclerosis and disc degeneration/low-back pain- a systematic review, Eur. J. Vasc. Endovasc. Surg 37 (2009) 661–670. [DOI] [PubMed] [Google Scholar]
- [2].Urban JPG, Roberts S. Degeneration of the intervertebral disc, Arthritis. Res. Ther 5 (2003) 120–130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Guiot BH, Fessler RG, Molecular biology of degenerative disc disease, Neurosurgery 47 (2000) 1034–1040. [DOI] [PubMed] [Google Scholar]
- [4].Mizuno H, Roy AK, Vacanti CA, Kojima K, Ueda M, Bonassar LJ, Tissue-engineered composites of anulus fibrosus and nucleus pulposus for inter-vertebral disc replacement, Spine 29 (2004) 1290–1298 [DOI] [PubMed] [Google Scholar]
- [5].Bowles RD, Williams RM, Zipfel WR, Bonassar LJ, Self-assembly of aligned tissue-engineered annulus fibrosus and intervertebral disc composite via collagen gel contraction, Tissue Eng. Part A 16 (2010) 1339–1348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Park SH, Gil ES, Cho H, Mandal BB, Tien LW, Min BH, et al. , Intervertebral disk tissue engineering using biphasic silk composite scaffolds, Tissue Eng. Part A 18 (2012) 447–458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Wilke HJ, Heuer F, Neidlinger-Wilke C, Claes L, Is a collagen scaffold for a tissue engineered nucleus replacement capable of restoring disc height and stability in an animal model? Eur. Spine J 15 (2006) 433–438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Korge A, Nydegger T, Polard JL, Mayer HM, Husson JL, A spiral implant as nucleus prosthesis in the lumbar spine, Eur. Spine J 11 (2002) S149–S153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Cassidy J, Hiltner A, Hierarchical structure of the intervertebral disc, Connect. Tissue Res 23 (1989) 75–88. [DOI] [PubMed] [Google Scholar]
- [10].Humzah M, Soames R, Human intervertebral disc: structure and function, Anat. Rec 220 (1988) 337–356. [DOI] [PubMed] [Google Scholar]
- [11].American Academy of Orthopaedic Surgeons, The Burden of Musculoskeletal Diseases in the United States, Academy of Orthopaedic Surgeons, Rosemont, IL, 2008. [Google Scholar]
- [12].Smith LJ, Chiaro JA, Nerurkar NL, Cortes DH, Horava SD, Hebela NM, et al. Nucleus pulposus cells synthesize a functional extracellular matrix and respond to inflammatory cytokine challenge following long term agarose culture, Eur. Cell. Mater 22 (2012) 291–301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Chou AI, Nicoll SB, Characterization of photocrosslinked alginate hydrogels for nucleus pulposus cell encapsulation, J. Biomed. Mater Res. Part A 91A (2009) 187–194. [DOI] [PubMed] [Google Scholar]
- [14].Meisel HJ, Siodla V, Ganey T, Minkus Y, Hutton WC, Alasevic OJ, Clinical experience in cell-based therapeutics: disc chondrocyte transplantation: a treatment for degenerated or damaged intervertebral disc, Biomol. Eng 24 (2007) 5–21. [DOI] [PubMed] [Google Scholar]
- [15].Ganey T, Libera J, Moos V, Alasevic O, Fritsch KG, Joerg Meisel H, Disc chondrocyte transplantation in a canine model: a treatment for degenerated or damaged intervertebral disc, Spine 28 (2003) 2609–2620. [DOI] [PubMed] [Google Scholar]
- [16].Sakai D, Mochida J, Yamamoto Y, Nomura T, Okuma M, Nishimura K, Transplantation of mesenchymal stem cells embedded in Atelocollagen® gel to the intervertebral disc: a potential therapeutic model for disc degeneration, Biomaterials 24 (2003) 3531–3541. [DOI] [PubMed] [Google Scholar]
- [17].Graf T, Differentiation plasticity of hematopoietic cells, Blood 99 (2002) 3089–3101. [DOI] [PubMed] [Google Scholar]
- [18].Watt FM, Hogan BLM, Out of eden: stem cells and their niches, Science 287 (2000) 1427–1430 [DOI] [PubMed] [Google Scholar]
- [19].Porada CD, Zanjani ED, Almeida-Porad G, Adult mesenchymal stem cells: a pluripotent population with multiple applications, Curr. Stem Cell Res. Ther 1 (2006) 365–369. [DOI] [PubMed] [Google Scholar]
- [20].da Silva Meirelles L. Chagastelles PC, Nardi NB, Mesenchymal stem cells reside in virtually all post-natal organs and tissues, J. Cell Sci 119 (2006) 2204–2213. [DOI] [PubMed] [Google Scholar]
- [21].Crevensten G, Walsh AJL, Ananthakrishnan D, Page P, Wahba GM, Lotz JC, et al. Intervertebral disc cell therapy for regeneration: mesenchymal stem cell implantation in rat intervertebral discs, Ann. Biomed. Eng 32 (2004) 430–434. [DOI] [PubMed] [Google Scholar]
- [22].Sakai D, Mochida J, Iwashina T, Watanabe T, Nakai T, Ando K, et al. Differentiation of mesenchymal stem cells transplanted to a rabbit degenerative disc model, Spine 30 (2005) 2379–2387. [DOI] [PubMed] [Google Scholar]
- [23].Gullbrand SE, Schaer TP, Agarwal P, Bendigo JR, Dodge GR, Chen W, et al. Translation of an injectable triple-interpenetrating-network hydrogel for intervertebral disc regeneration in a goat model, Acta Biomater 60 (2017) 201–209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Acosta FL Jr, Metz L, Adkisson HD IV, Liu J, Carruthers-Liebenberg E, Milliman C, et al. Allogeneic Juvenile Articular Chondrocytes or Mesenchymal Stem Cells, Tissue Eng. Part A 17 (2011) 3045–3055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Oehme D, Ghosh P, Goldschlager T, Shimon S, Wu J, Stuckey S, Radiological, morphological, histological and biochemical changes of lumbar discs in an animal model of disc degeneration suitable for evaluating the potential regenerative capacity of novel biological agents, J Tissue Sci Eng. 6 (2015), doi: 10.4172/2157-7552.1000153. [DOI] [Google Scholar]
- [26].Nerurkar NL, Elliott DM, Mauck RL, Mechanics of oriented electrospun nanofibrous scaffolds for annulus fibrosus tissue engineering, J. Orthop. Res 25 (2007) 1018–1028. [DOI] [PubMed] [Google Scholar]
- [27].Chang G, Kim H−J, Kaplan D, Vunjak-Novakovic G, Kandel RA, Porous silk scaffolds can be used for tissue engineering annulus fibrosus, Eur Spine J 16 (2007) 1848–1857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Xu H, Xu B, Yang Q, Li X, Ma X. Xia Q. Comparison of decellularization protocols for preparing a decellularized porcine annulus fibrosus scaffold, PLoS ONE 9 (2014) e86723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Smith LJ, Chiaro JA, Nerurkar NL, Cortes DH, Horava SD, Hebela NM, Nucleus pulposus cells synthesize a functional extracellular matrix and respond to inflammatory cytokine challenge following long term agarose culture, Eur Cell Mater 22 (2012) 291–301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Kim DH, Martin JT, Elliott DM, Smith LJ, Mauck RL. Phenotypic stability, matrix elaboration and functional maturation of nucleus pulposus cells encapsulated in photocrosslinkable hyaluronic acid hydrogels, Acta Biomater 12 (2015) 21–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Reza AT, Nicoll SB, Characterization of novel photocrosslinked carboxymethylcellulose hydrogels for encapsulation of nucleus pulposus cells, Acta Biomater 6 (2010) 179–186. [DOI] [PubMed] [Google Scholar]
- [32].Masuda K, Takegami K, An H, Kumano F, Chiba K, Andersson GBJ, Recombinant osteogenic protein-1 upregulates extracellular matrix metabolism by rabbit annulus fibrosus and nucleus pulposus cells cultured in alginate beads, J. Orthop. Res 21 (2003) 922–930. [DOI] [PubMed] [Google Scholar]
- [33].Martin JT, Gullbrand SE, Kim DH, Ikuta K, Pfeifer CG, Ashinsky BG, In vitro maturation and in vivo integration and function of an engineered cell-seeded disc-like angle ply structure (DAPS) for total disc arthroplasty, Sci. Rep 7 (2017) 15765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Strassburg S, Richardson SM, Freemont AJ, Hoyland JA, Co-culture induces mesenchymal stem cell differentiation and modulation of the degenerate human nucleus pulposus cell phenotype, Regen. Med 5 (2010) 701–711. [DOI] [PubMed] [Google Scholar]
- [35].Allon AA, Schneider RA, Lotz JC, Co-culture of adult mesenchymal stem cells and nucleus pulposus cells in bilaminar pellets for intervertebral disc regeneration, SAS J 3 (2009) 41–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Allon AA, Schneider RA, Butcher K, Lotz JC, Structured coculture of mesenchymal stem cells and disc cells enhances differentiation and proliferation, Cells Tissues Organs 196 (2012) 99–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Huang AH. Yeger-McKeever M, Stein A, Mauck RL, Tensile properties of engineered cartilage formed from chondrocyte- and MSC-laden hydrogels, Osteoarthr Cartilage 16 (9) (2008) 1074–1082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Stegemann H, Stalder K, Determination of hydroxyproline, Clin. Chim. Acta 18 (1967) 267–273. [DOI] [PubMed] [Google Scholar]
- [39].Neuman RE, Logan MA, The determination of hydroxypoline, J. Biol. Chem (1949) 299–306. [PubMed] [Google Scholar]
- [40].Huang AH. Farrell MJ, Mauck RL, Mechanics and mechanobiology of mesenchymal stem cell-based engineered cartilage, J. Biomech 43 (2010) 128–136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Martin JT, Milby AH, Chiaro JA, Kim DH, Hebela NM, Smith LJ. Translation of an engineered nanofibrous disc-like angle-ply structure for intervertebral disc replacement in a small animal model, Acta Biomater 10 (2014) 2473–2481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Masuda K, Aota Y, Muehleman C, Imai Y, Okuma M, Thonar EJ, A novel rabbit model of mild, reproducible disc degeneration by an Anulus needle puncture: correlation between the degree of disc injury and radiological and histological appearances of disc degeneration, Spine 30 (2005) 5–14. [DOI] [PubMed] [Google Scholar]
- [43].Gullbrand SE, Kim DH, Bonnevie E, Ashinsky BG, Smith LJ, Elliott DM, Towards the scale up of tissue engineered intervertebral discs for clinical application, Acta Biomater 70 (2018) 154–164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Martin JT, Kim DH, Milby AH, Pfeifer CG, Smith LJ, Elliott DM, In vivo performance of an acellular disc-like angle ply structure (DAPS) for total disc replacement in a small animal model, J. Orthop. Res 35 (2017) 23–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Gullbrand SE, Ashinsky BG, Bonnevie ED, Kim DH, Engiles JB, Smith LJ, Long-term mechanical function and integration of an implanted tissue-engineered intervertebral disc, Sci. Transl. Med 10 (2018) eaau0670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Gullbrand SE, Ashinsky BG, Martin JT, Pickup S, Smith LJ, Mauck RL. Correlations between quantitative T2 and T1rho MRI, mechanical properties and biochemical composition in a rabbit lumbar intervertebral disc degeneration model, J. Orthop. Res 34 (2016) 1382–1388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Ruan DK, Xin H, Zhang C, Wang C, Xu C, Li C, Experimental intervertebral disc regeneration with tissue-engineered composite in a canine model, Tissue Eng. Part A 16 (2010) 2381–2389. [DOI] [PubMed] [Google Scholar]
- [48].Lazebnik M, Singh M, Glatt P, Friis LA, Berkland CJ, Detamore MS, Biomimetic method for combining the nucleus pulposus and annulus fibrosus for intervertebral disc tissue engineering, J. Tissue. Eng. Regen. Med 5 (2011) 179–187. [DOI] [PubMed] [Google Scholar]
- [49].Dormer NH, Berkland CJ, Detamore MS, Emerging techniques in stratified designs and continuous gradients for tissue engineering of interfaces, Ann. Biomed. Eng 38 (2010) 2121–2141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Pelttari K, Winter A, Steck E, Goetzke K, Hennig T, Ochs BG, Premature induction of hypertrophy during in vitro chondrogenesis of human mesenchymal stem cells correlates with calcification and vascular invasion after ectopic transplantation in SCID mice, Arthritis Rheum 54 (2006) 3254–3266. [DOI] [PubMed] [Google Scholar]
- [51].Koga H, Engebretsen L, Brinchmann JE, Muneta T, Sekiya I, Mesenchymal stem cell-based therapy for cartilage repair: a review, Knee Surg. Sports Traumatol. Arthrosc 17 (2009) 1289–1297. [DOI] [PubMed] [Google Scholar]
- [52].Bowles RD, Gebhard HH, Härtl R, Bonassar LJ. Tissue-engineered intervertebral discs produce new matrix, maintain disc height, and restore biomechanical function to the rodent spine, Proc. Natl. Acad. Sci. U S A 9 (2011) 13106–13111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Khoshgoftar M, Wilson W, Ito K, van Donkelaar CC, Influence of tissue- and cell-scale extracellular matrix distribution on the mechanical properties of tissue-engineered cartilage, Biomech. Model. Mechanobiol 12 (2013) 901–913. [DOI] [PubMed] [Google Scholar]
- [54].Guermazi A, Alizai H, Crema MD, Trattnig S, Regatte RR, Roemer FW, Compositional MRI techniques for evaluation of cartilage degeneration in osteoarthritis, Osteoarthr. Cartilage 23 (2015) 1639–1653. [DOI] [PubMed] [Google Scholar]
- [55].Moore RJ, The vertebral endplate: disc degeneration, disc regeneration, Eur. Spine J 15 (2006) S333–S337. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.