Abstract
Published studies agree that transient intraocular pressure (IOP) spikes are common after intravitreal injections of anti–vascular endothelial growth factor agents. Currently, there is no standard of care guiding if and when to prevent these IOP spikes. Furthermore, there are challenges in determining the impact of postinjection IOP elevation on the health of the retinal ganglion cells, particularly given the often-existing comorbidities of retinal and glaucoma pathology. This review highlights the current literature regarding both acute and chronic postinjection IOP elevations and discusses management of postinjection IOP elevation, especially in patients at high risk for glaucomatous damage.
Key Words: intravitreal injection, VEGF, intraocular pressure, glaucoma, retinal nerve fiber layer
CLINICAL SCENARIO
A patient with a diagnosis of primary open-angle glaucoma undergoes an anti–vascular endothelial growth factor (VEGF) intravitreal injection for concomitant wet age-related macular degeneration (AMD). After injection, the patient experiences the acute onset of eye pain with a decline in vision. The intraocular pressure (IOP) is noted to be 45 mm Hg. The symptoms rapidly resolve without intervention. How should this patient be managed during future injections?
Anti-VEGF agents have revolutionized the treatment of wet AMD, diabetic retinopathy, retinal vein occlusions, and other retinal pathology.1 A common intravitreal injection volume is 0.05 mL, and serial injections are often needed over months or years.2 In addition, anti-VEGF therapy is widely used to manage neovascular glaucoma.3 Bevacizumab has also been used as an adjunct to rescue failing vascularized blebs.4
Despite its utility in many diseases, a recognized adverse effect of intravitreal anti-VEGF injections is IOP elevation.1 In 2009, Kahook et al5 published a case series of 6 eyes injected with bevacizumab that developed sustained elevations in IOP, while fellow eyes remained at baseline IOP. The IOP elevations reported by the case series appeared temporally related to injections. In the past decade, many more studies have examined the relationship between intravitreal injections and IOP elevation.
This review discusses the current literature regarding acute and chronic sequelae of postinjection IOP elevations. The definition of IOP elevation and the timing of IOP measurement in published studies varies; these variables are sometimes treated as categorical variables and sometimes continuous. We try to include relevant definitions when describing individual studies in this review. Techniques used to measure IOP also vary between studies. Patients with preexisting glaucoma have often been excluded from these studies, despite their particular susceptibility to changes in IOP. We provide data from this subgroup when available and acknowledge gaps in current literature.
MAGNITUDE OF POSTINJECTION IOP ELEVATIONS
Published studies agree that transient postinjection IOP spikes are common after intravitreal injections of anti-VEGF agents. The average IOP within 1 minute of injection has been reported to be >40 mm Hg. Felfeli et al6 reported an IOP rise from mean baseline preinjection of 15.3 mm Hg to postinjection of 41.6 mm Hg, ranging broadly from 17 to 81 mm Hg. El Chehab et al7 reported a mean IOP spike of 46.4 mm Hg 1 minute after injection. In a meta-analysis of 46 articles (2872 eyes), de Vries et al8 reported that the mean difference in IOP after anti-VEGF injection (rise above preinjection) was 23.41 mm Hg immediately after injection, 2.51 mm Hg at 30 minutes, −0.63 one day after injection, and back to baseline by 1 week. For some patients, the acute IOP elevation does persist for at least several hours, and so it might make sense to define 3 categories of postinjection IOP rise, in particular to capture these sensitive eyes: early (or acute) is a rise in IOP within minutes, intermediate is a rise lasting hours, and late is a chronic elevation over months.
In contrast with acute IOP elevations, there is a lack of consensus on the sequelae of repeated intravitreal injections relating to eye pressure over a long period of time. In a post hoc analysis of 2 phase 3 clinical trials of ranibizumab for AMD, Bakri et al9 found that more (but not most) injected eyes versus control eyes had an elevation of IOP (defined as 6 or 8 mm Hg increase from baseline with concurrent IOP of at least 21 or 25 mm Hg) over 24 months. Foss et al10 demonstrated a very small but statistically significant mean IOP elevation of < 0.05 mm Hg that was sustained in 610 patients receiving ranibizumab or bevacizumab for AMD in the Alternative Treatments to Inhibit Anti-VEGF in Age-related Choroidal Neovascularization (IVAN) trial. In a meta-analysis of 5 randomized controlled trials (mostly cases of diabetic macular edema rather than AMD), Zhou et al11 demonstrated an increased risk of sustained IOP elevation (defined as IOP>21 or 5 mm Hg above baseline over 2 visits or IOP>25 mm Hg requiring treatment) in injected versus control eyes. They found that the prevalence of sustained IOP elevation was 4.7%, pooled from 13 nonrandomized controlled trials with significant heterogeneity between studies.
SEQUELAE OF POSTINJECTION IOP ELEVATION: GLAUCOMA AND RETINAL NERVE FIBER LAYER (RNFL) ARCHITECTURE
Although there is a lack of consensus on the chronic effects of anti-VEGF injections on open-angle glaucoma, several large studies support a dose-related association. In a case-control study of over 800 eyes, Eadie et al12 demonstrated higher incidence of glaucoma surgery in eyes receiving 7 or more injections per year compared with eyes receiving 3 or fewer injections per year. Cui et al13 identified 17,113 nonglaucomatous eyes receiving intravitreal injection from the Clinformatics Data Mart Database (OptumInsight, Eden Prairie, MN). Their group demonstrated that eyes receiving more injections (14 or more injections at 2 y follow-up and 20 or more injections at 3 y follow-up) had higher odds of initiating IOP-lowering therapy or having a new diagnosis of glaucoma (defined by inclusion of a glaucoma diagnosis in medical claims data). Cui and colleagues reported that 1.6% of all included eyes initiated IOP-lowering therapy within 2 years follow-up, and there was no difference in age, sex, or history of diabetes or hypertension between the group requiring IOP-lowering therapy and the group that did not.
Several case reports describe acute angle-closure glaucoma temporally related to intravitreal injection. Jeong et al14 described a case in a hyperopic eye and proposed that eyes with short axial lengths are at higher risk due to less space for the injected volume of fluid. They performed a prophylactic peripheral iridotomy in the contralateral eye to prevent future postinjection attacks. They also suggested that prevention of vitreous reflux after injection might contribute to high IOP. Kim and Baek15 described a case in an eye that was not hyperopic; they suggested that zonulopathy and mydriasis might have contributed. Alkin et al16 suggested that anterior chamber depth might shallow after intravitreal injection, in correlation with the acute rise in IOP due to increased intraocular fluid volume. It might be prudent to assess eyes for risk of angle closure, including gonioscopy, before initiating intravitreal injections.
Studies conflict on the topic of repeated injections on RNFL loss. Studies examining the effect of injections on RNFL architecture are summarized in Table 1.8,17–24 In their 2020 meta-analysis of intravitreal injections and RNFL stability, de Vries et al8 included 4 studies on this topic and concluded that RNFL was significantly reduced by −3.34 µm at 1 year, but only 1 of the 4 individual studies (Martinez-de-la-Casa and colleagues) inferred that injections were associated with thinning. Martinez-de-la-Casa et al20 completed a prospective study of 49 eyes and 17 control eyes that demonstrated RNFL thinning in injected eyes over the span of 1 year. Jo et al19 found that RNFL was not reduced in eyes treated with ranibizumab and suggested that areas of RNFL thinning might more likely be related to the macular lesion rather than the injections. Entezari et al17 also found that RNFL was at baseline at 12 months in eyes injected with bevacizumab and similarly concluded that RNFL thinning might be related to changes in macular edema. Parlak et al21 found thinning in both injected eyes and control eyes without a difference between the groups. Outside of de Vries and colleagues’ meta-analysis, Soheilian et al22 suggested that prevention of acute spikes via prophylactic anterior chamber paracentesis prevents RNFL loss in injected eyes measured 3 months after injection. In contrast, Valverde-Megias et al24 followed 20 injected eyes and their contralateral control eyes for 8 years and demonstrated RNFL loss in both groups but no difference in RNFL thickness or loss between injected and control eyes. Horsley et al18 reported no RNFL loss in a retrospective study of injected eyes that underwent RNFL measurement over the span of at least 1 year. Swaminathan et al23 reported no difference in RNFL thinning per year in a retrospective study of 53 injected eyes versus fellow eye controls. If there is an association between injections and RNFL loss, then it is likely that some but not all eyes are susceptible, and small studies might bias the data and miss susceptible eyes. Unlike IOP monitoring, monitoring of RNFL health is not currently standard of care in patients receiving anti-VEGF injections and might be impacted by injections as well as the underlying retinal pathology.
TABLE 1.
Summary of Studies Evaluating Postinjection Changes to RNFL
| References | Study Design | Anti-VEGF Drug | Retinal Pathology | N | Length of Follow-up | Eyes With Glaucoma | Summary of Results |
|---|---|---|---|---|---|---|---|
| de Vries et al8 | Meta-analysis | Anti-VEGF, dexamethasone | AMD | 5 studies included in RNFL meta-analysis; 4 studies included in anti-VEGF RNFL meta-analysis | NA | NA | RNFL thinning was significant only at 12 mo |
| Entezari et al17 | Prospective | Bevacizumab | AMD | 18 injected eyes, no controls | Measurements at 12 wk and 24 wk postinjection | Excluded | RNFL thinning was present in the temporal quadrant at 12 wk but back at baseline at 24 wk; RNFL thinning might be related to decreased macular edema |
| Horsley et al18 | Retrospective | Pegaptanib, bevacizumab, ranibizumab | AMD | 41 eyes | Mean: 27 mo | Excluded | There was no RNFL thinning in any group |
| Jo et al19 | Prospective | Ranibizumab | AMD | 20 injected eyes compared with fellow eyes | Measurements at 6 and 12 mo postinjection | Excluded | RNFL thinning was present in the clock hours of the macular lesion; RNFL might more likely be related to the macular lesion (eg, decreased macular edema) rather than injections |
| Martinez-de-la-Casa et al20 | Prospective | Ranibizumab | AMD | 49 injected eyes, 17 control eyes | 12 mo | Excluded | There was significant RNFL thinning in the injection group but not the control group at 12 mo |
| Parlak et al21 | Prospective | Ranibizumab | AMD | 22 injected eyes compared with fellow eyes | 3 mo | Excluded | There was RNFL thinning in injected eyes and control eyes, but there was no difference between these groups |
| Soheilian et al22 | Prospective, randomized | Bevacizumab | AMD, DME | 45 eyes with prophylactic paracentesis, 45 control eyes | 3 mo after injection | Excluded | Mean RNFL decreased more in eyes without paracentesis (−2 µm) than in eyes with paracentesis (0 µm) |
| Swaminathan et al23 | Retrospective | Not specified | AMD | 53 eyes compared with fellow eyes | Mean: 3.7 y | Included | Injected eyes with glaucoma did not have more RNFL thinning than control eyes with glaucoma |
| Valverde-Megias et al24 | Prospective | Ranibizumab | AMD | 20 eyes | 96 mo | Excluded | There was no difference in RNFL thickness or loss between injected eyes and contralateral eyes |
AMD indicates age-related macular degeneration; DME, diabetic macular edema; NA, not available; RNFL, retinal nerve fiber layer; VEGF, vascular endothelial growth factor.
PATHOPHYSIOLOGY
It is reasonable to suspect that injection of a volume of fluid in a relatively closed space contributes to the acute elevation in pressure. In 1 case report, an acute IOP spike following intravitreal injection in a posttrabeculectomy eye resulted in bleb rupture.25 The authors noted that the acute volume effect of an intravitreal injection could be enough to elevate IOP by 90 mm Hg. While the mechanism of acute IOP rise postintravitreal injection is agreed upon—a volume effect that is short-lasting—the mechanism of chronic IOP rise is more controversial.
Wen et al26 used electronic Schiøtz tonography to compare aqueous outflow in control eyes versus eyes that received up to 10 injections and eyes that received at least 20. They demonstrated reduced aqueous outflow in eyes receiving a greater number of injections. Chronic elevation in IOP might relate to repeated and ongoing injury to the trabecular meshwork from the repeated injection of high volume, alterations in levels of trabecular meshwork vasodilating modulators such as nitric oxide, toxic effects of drugs or drug delivery, or inflammatory damage.
In one proposed mechanism, anti-VEGF agents might decrease nitric oxide levels via inhibition of nitric oxide synthase, resulting in downstream movement of potassium and calcium in trabecular meshwork cells, alteration of trabecular meshwork cell contractility, and ultimately decreased aqueous outflow through intercellular spaces27–29 (Fig. 1). Connections have been drawn between the systemic effect and intraocular effect of nitric oxide on smooth muscle and cell contractility. For example, systemic hypertension secondary to anti-VEGF agents has been described, possibly due to decreased activity of nitric oxide synthase resulting in blood vessel constriction, and it has been proposed that anti-VEGF agents similarly increase outflow resistance in the eye.30
FIGURE 1.
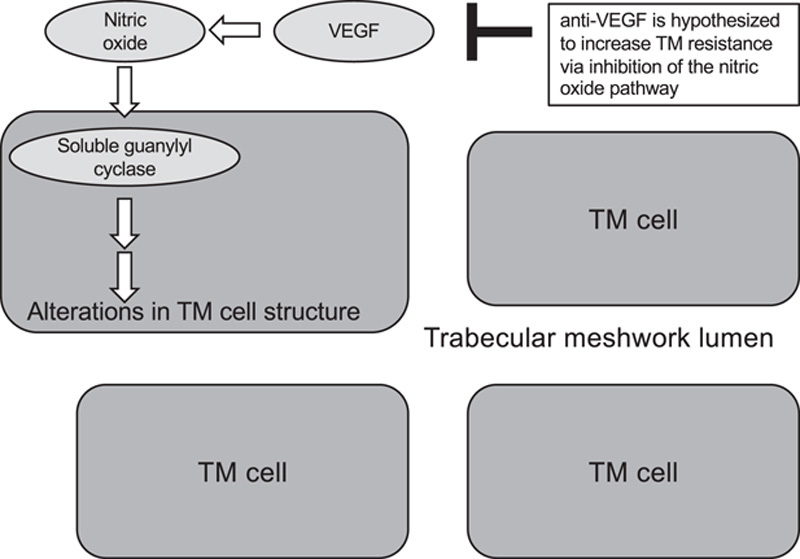
It has been proposed that vascular endothelial growth factor (VEGF) increases nitric oxide production in trabecular meshwork (TM), with downstream alteration of TM cell structure. These changes in cell structure open the TM lumen through which aqueous flows. Anti-VEGF agents would inhibit this mechanism and decrease aqueous outflow.
Animal studies demonstrate retention of injected materials in ocular structures that could affect aqueous outflow either by mechanical or other mechanisms. Gal-Or et al31 injected bevacizumab into the vitreous of rats with choroidal neovascularization to demonstrate that bevacizumab interacts with trabecular meshwork. At time points from immediately after injection to 6 hours later, bevacizumab was identified by immunofluorescence in the trabecular meshwork and Schlemm canal. At 24 hours, immunoreactivity to bevacizumab was still detectable in outflow tracts, clearing by 48 hours. Huet et al32 identified retained intraocular foreign material consistent with silicone oil droplets in eyes of monkeys given intravitreal injections. The silicone oil is theorized to originate in coatings on syringes used for injection. Silicone oil and protein particles have been found in anti-VEGF samples.33,34 Presumed silicone oil from syringes has also been described in multiple studies of injected human eyes.35 To prevent silicone retention in injected eyes, Lode et al36 described a method of compounding anti-VEGF agents in silicone-free syringes.
RISK FACTORS
Multiple factors are theorized to modulate the magnitude and duration of an acute IOP spike. Studies reporting risk factors are summarized in Table 2.7,10,37–47 For many of these factors, published studies can be found both to support and to contradict their effects on postinjection pressure. Factors related to the effect of increased volume in a closed space include axial length and lens status. Karakurt et al38 suggested a negative correlation between axial length and the immediate postinjection IOP spike. El Chehab et al7 reported no association of acute spike with axial length or lens status. Another group found Nd:YAG capsulotomy to be a risk factor for sustained postinjection IOP elevation but no difference between phakic and pseudophakic eyes, proposing that capsulotomy alters the interaction between the injected drug and the trabecular meshwork.40 In a study of ocular rigidity in eyes receiving anti-VEGF injections, Sayah et al39 suggested that increased ocular rigidity was associated with increased postinjection IOP.
TABLE 2.
Summary of Studies Evaluating Risk Factors for Postinjection IOP Elevation
| References | Study Design | Anti-VEGF Drug | Retinal Pathology | Total (N) | Summary of Results |
|---|---|---|---|---|---|
| Anterior segment anatomy | |||||
| Cui et al13 | Retrospective | Not specified | AMD, retinal vein occlusion, other | 17,113 patients | Patients that initiated IOP-lowering therapies after receiving injections were less likely to be pseudophakic than patients that did not initiate IOP-lowering therapy |
| El Chehab et al7 | Prospective | Ranibizumab | AMD | 250 injections | Phakic status and axial length were not associated with acute IOP elevation |
| Foss et al10 | Randomized controlled clinical trial | Bevacizumab, ranibizumab | AMD | 610 patients | Postinjection IOP spike was reduced in pseudophakic/aphakic eyes (compared with phakic eyes) |
| Hoang et al37 | Retrospective | Bevacizumab, ranibizumab | AMD | 207 eyes | Phakic status, status of YAG capsulotomy, and history of peripheral iridotomy were not statistically associated with sustained IOP elevation |
| Karakurt et al38 | Retrospective | Bevacizumab, ranibizumab, dexamethasone | AMD, retinal vein occlusion, diabetic retinopathy | 188 patients | Acute IOP elevation at 1 min postinjection was negatively correlated with axial length |
| Sayah et al39 | Prospective | Anti-VEGF, not otherwise specified | AMD, retinal vein occlusion, diabetic retinopathy, other | 18 eyes | Greater ocular rigidity is associated with greater postinjection IOP spike |
| Sternfeld et al40 | Retrospective | Bevacizumab, ranibizumab | AMD, diabetic macular edema, other | 119 eyes | Postcapsulotomy eyes had higher rates of IOP elevation than either phakic or pseudophakic eyes without capsulotomy. There was no difference in IOP elevation between phakic vs. pseudophakic eyes |
| Anti-VEGF drug and dosing | |||||
| Choi et al41 | Retrospective | Bevacizumab, ranibizumab, pegaptanib | AMD | 155 eyes | There was no relationship between IOP measurements and frequency of injections, total number of injections, or agents used |
| Cui et al13 | Retrospective | Not specified | AMD, retinal vein occlusion, other | 17,113 patients | Eyes with more injections had a higher odds of initiating IOP-lowering therapy |
| Foss et al10 | Randomized controlled clinical trial | Bevacizumab, ranibizumab | AMD | 610 patients | Bevacizmub-treated eyes had a postinjection IOP that was lower than in ranibizumab-treated eyes but this reduction was not statistically significant |
| Good et al42 | Retrospective | Bevacizumab, ranibizumab | AMD | 215 eyes | There was not enough statistical significance to determine a difference in elevation of IOP between bevacizumab and ranibizumab groups |
| Hoang et al37 | Retrospective | Bevacizumab, ranibizumab | AMD | 207 eyes | Greater number of injections was weakly associated with development of sustained IOP elevation |
| Mathalone et al43 | Retrospective | Bevacizumab | AMD | 201 eyes | Shorter interval between injections (<8 wk) was associated with sustained IOP elevation |
| Tseng et al44 | Series of eyes with postinjection IOP elevation | Bevacizumab, ranibizumab | AMD | 25 eyes | Eyes received mean: 20 (median: 17) injections before sustained IOP elevation developed |
| Vo Kim et al45 | Retrospective | Ranibizumab, aflibercept | Diabetic macular edema | 140 eyes | Eyes with sustained postinjection IOP elevation had received more injections than eyes without elevation; there was no difference in interval between injections or injection drug |
| Preexisting glaucoma | |||||
| Foss et al10 | Randomized controlled clinical trial | Bevacizumab, ranibizumab | AMD | 610 patients | Eyes with preexisting glaucoma had a higher postinjection IOP spike |
| Good et al42 | Retrospective | Bevacizumab, ranibizumab | AMD | 215 eyes | Eyes with preexisting glaucoma had higher prevalence of sustained IOP elevation compared with eyes without glaucoma |
| Hoang et al37 | Retrospective | Bevacizumab, ranibizumab | AMD | 207 eyes | Personal history of glaucoma was not associated with sustained IOP elevation |
| Kim et al47 | Retrospective | Bevacizumab, ranibizumab, pegaptanib, triamcinolone | AMD, other | 120 eyes | Eyes with preexisting glaucoma had a higher postinjection IOP spike at 5 min and took longer to return to baseline |
| Lam et al46 | Prospective | Bevacizumab, ranibizumab, aflibercept | Diabetic retinopathy, other | 10 postsurgical eyes, 9 control eyes | Eyes with history of incisional glaucoma surgery had a smaller postinjection rise in IOP than control eyes |
| Mathalone et al43 | Retrospective | Bevacizumab | AMD | 201 eyes | Preexisting glaucoma was not associated with sustained IOP elevation |
AMD indicates age-related macular degeneration; IOP, intraocular pressure; VEGF, vascular endothelial growth factor.
Several studies associate an increased number of injections with an increased risk of sustained IOP elevation. In a study of 140 eyes without a history of glaucoma, 7% were reported to develop sustained IOP elevation of >6 mm Hg above their preinjection IOP, and IOP elevation was reported to be associated with a number of injections.45 In a study of over 200 patients, eyes with elevated IOP sustained over 2 visits had received more injections than eyes without sustained IOP elevation.37 As discussed earlier in this review, Cui and colleagues reported an association between a greater number of injections and the risk of initiating IOP-lowering therapy based on claims data from 17,113 nonglaucomatous eyes.
The possibility that a higher number of repeated injections confers more risk raises the question of whether long-acting anti-VEGF injections confer less risk than repeated injections over the same period of time. At the time of this review, brolucizumab is the only Food and Drug Administration (FDA)-approved long-acting anti-VEGF injection for retinal disease. Brolucizumab dosed at 12-week intervals was compared with aflibercept for AMD in the phase 3 HAWK and HARRIER trials; IOP was included as an ocular adverse event.48 Eyes with potentially confounding intraocular conditions or uncontrolled glaucoma were excluded. IOP measurements were done before injection, immediately after injection, and 30 minutes after injection. Incidence of increased IOP was reported to be similar in the brolucizumab and aflibercept groups (2.5% to 3.2% and 2.2% to 2.4%, respectively). Trends in IOP over the 48-week follow-up were not specifically reported in the clinical trial. Trends in IOP over longer follow-up and RNFL measurements would be interesting to evaluate in ongoing studies of long-acting anti-VEGF injections.
Several studies suggest eyes with a history of glaucoma are at a higher risk for postinjection IOP elevation. Foss et al10 found that eyes with preexisting glaucoma have a high postinjection IOP spike. Kim et al47 found that eyes with preexisting glaucoma take longer to recover from the acute spike; fewer eyes with glaucoma had reached an IOP < 30 mm Hg at 5, 10, and 15 minutes after injection compared with nonglaucomatous eyes. Good et al42 reported higher prevalence of sustained postinjection IOP in eyes with glaucoma. Elevation of IOP in more susceptible patients receiving these injections has motivated some ophthalmologists to recommend more intensive pressure monitoring before and after injections in this specific subgroup. Surgical alterations to aqueous outflow might also affect postinjection IOP. Lam et al46 found that eyes with prior incisional glaucoma surgery have a smaller spike and recover more quickly. Mean postinjection spike was at least 15 mm Hg lower in postsurgical glaucomatous eyes and returned to baseline at least 5 minutes faster. It would be interesting to know whether minimally invasive glaucoma surgery (MIGS) mitigates the effect of intravitreal injections on IOP. Rezkallah et al49 reported a case series of MIGS for treatment of elevated IOP related to intravitreal steroid injections. We are not aware of any studies of MIGS to prevent IOP elevation associated with intravitreal anti-VEGF injections in our review of the literature.
In addition to the ocular volume and glaucoma history, other potential variables include the type of retinal pathology, volume of injected medication, and intraocular fluid dynamics.
PREVENTION
Most studies agree that pretreatment with topical IOP-lowering drops decreases acute postinjection IOP spikes. In a randomized cross-over trial, eyes pretreated with brimonidine (1 drop 20 min before injection) had a lower mean IOP immediately after injection and 20 minutes after injection.6 Fewer pretreated eyes versus control eyes reached an IOP >50 mm Hg and fewer required therapeutic anterior chamber paracentesis after injection. In a randomized double-masked placebo-controlled study, mean IOP in eyes pretreated with combination brimonidine/timolol (dosed twice daily on the day prior and the day of injection) was 28 mm Hg 5 minutes postinjection versus 34 mm Hg in eyes pretreated with artificial tears.50 At 15 minutes postinjection, all pretreated eyes but only a third of control eyes had resolved to an IOP < 20 mm Hg. Another option to control acute elevation in IOP in this setting is to perform an anterior chamber paracentesis. In a study of eyes randomized to anterior chamber paracentesis (immediately after injection), brimonidine (90 min before injection), acetazolamide (90 min before injection), or no pretreatment, only anterior chamber paracentesis prevented postinjection IOP elevation entirely.51 All pretreated groups returned to baseline more quickly than the control group. While the data strongly suggest that pretreatment is beneficial, there is no consensus around preinjection protocols to prevent acute IOP elevation. In addition, glaucoma patients who are already on IOP-lowering drops might not receive the same magnitude of mitigation as a patient who is naïve to drops. This has not been specifically studied.
In contrast, the data on how to mitigate chronic IOP elevation or impact on RNFL after injections are sparse. Methods might include minimizing injections via “treat and extend” protocols, using sustained-release anti-VEGF injections to avoid repeated injections, avoiding syringes at risk of leaving particles, or even decreasing injection volume, but in our review of the current literature, these have yet to be rigorously studied. It is difficult to study methods of mitigating chronic effects when the chronic effects are not well understood.
CHALLENGES IN RESEARCH
The interplay between glaucoma and retinal diseases as well as the complexity of each disease makes this a difficult topic to study. Some retinal nerve fiber damage is expected in retinal pathologies—regardless of injections—such as ischemic etiologies involved with retinal vein occlusions, diabetes, or retinal disease that results in neovascular glaucoma. There are likely subgroups of eyes, such as eyes with severe RNFL loss versus nonglaucomatous eyes, that are affected differently by chronic injections. The many factors theorized to modulate the magnitude and duration of the IOP spike might confound studies of this topic, thus making a homogeneous patient population hard to define.
Most published studies are indeed limited by small size and the heterogeneity of the study populations. Studies that include eyes with retinal vein occlusions might be confounding the results due to the eyes having RNFL damage secondary to the vein occlusions rather than injections. These eyes might also develop neovascular glaucoma, also confounding the results. Several studies suggest that treatment with ranibizumab increases the risk of elevated IOP when compared with bevacizumab or aflibercept.52 This difference in risk limits the interpretation of studies that mix eyes receiving a variety of different agents in a single study population. Many studies treat IOP as a categorical variable. There might be value in examining IOP as a continuous variable instead. Furthermore, the larger more homogeneous clinical studies intended for drug development and regulatory approval excluded glaucomatous eyes. In studies that do include glaucomatous eyes, it is difficult to confidently associate chronic IOP or RNFL changes with injections in eyes that are already at risk of glaucomatous progression.
Large prospective trials would be useful to determine whether prevention of acute postinjection IOP spikes protects eyes from glaucomatous damage. But even without large prospective studies, it seems intuitive that monitoring of RNFL health should be considered in eyes at risk for glaucoma while receiving chronic anti-VEGF treatment. Optic nerves in glaucomatous eyes might be particularly sensitive to variations in IOP. In future studies and in clinical practice, eyes with preexisting glaucoma should be considered a unique group with regard to postinjection IOP spikes.
CONCLUSIONS
In summary, acute IOP elevations are common after intravitreal injections; chronic IOP elevations related to injections likely occur in some eyes, and data seem to suggest that prevention of postinjection IOP spikes should be considered as part of a standardized management approach. This approach would likely vary based on individual risk factors, with vigilance in at-risk eyes more vulnerable to glaucoma damage. Vigilant monitoring of eyes receiving injections might likely include IOP and RNFL monitoring. For example, in the clinical scenario provided in this paper, management might reasonably include monitoring of RNFL imaging and prevention of future IOP spikes with topical therapies. The acute IOP elevation is likely due to an acute increase in volume in a relatively closed space; chronic IOP elevations are less understood but are theorized to result from the reduced aqueous outflow. It has been suggested that incisional glaucoma surgery mitigates the effect of intravitreal injections on IOP. Effects from MIGS on postinjection IOP is not yet well studied. In addition, the effects of long-acting anti-VEGF agents on IOP are not yet well studied.
Pretreatment with IOP-lowering drops is not a unique approach to the prevention of acute IOP spikes and is widely used before laser procedures like YAG capsulotomy and selective laser trabeculoplasty. Pretreatment should be routinely considered before intravitreal injections, particularly in eyes with glaucoma or known history of sustained IOP elevation, in whom the chronic effects are not yet known. As more attention is paid to the patient experience in healthcare valuations, the physical discomfort of postinjection IOP spikes such as acute loss of vision and eye pain are also reason to consider prevention of postinjection IOP spikes.
There are many challenges to rigorously studying the relationship between repeated intravitreal anti-VEGF injections and long-term IOP and RNFL trends. Retinal ischemia resulting in RNFL thinning or neovascular glaucoma potentially confounds our understanding of this relationship.
Indications for intravitreal injections are expanding, with studies such as the PANORAMA trial showing benefit in eyes with nonproliferative diabetic retinopathy that previously did not receive injections.53 Thus, more and more patients are likely to be impacted by the acute and chronic effects of postinjection IOP elevation. Collateral damage to the RNFL should be avoided and the impact of elevated IOP associated with intravitreal injections should be addressed. The development of intravitreal injection protocols that address both retinal and optic nerve health are perhaps long overdue.
Footnotes
Supported in part by an Unrestricted Grant from Research to Prevent Blindness, New York, NY, to the Department of Ophthalmology & Visual Sciences, University of Utah, and the Utah chapter of the Achievement Rewards for College Scientists Foundation, Salt Lake City, UT.
Disclosure: The authors declare no conflict of interest.
Contributor Information
Ariana M. Levin, Email: ariana.levin@hsc.utah.edu.
Craig J. Chaya, Email: craig.chaya@hsc.utah.edu.
Malik Y. Kahook, Email: malik.kahook@cuanschutz.edu.
Barbara M. Wirostko, Email: barbara.wirostko@hsc.utah.edu.
REFERENCES
- 1.Ghasemi Falavarjani K, Nguyen QD. Adverse events and complications associated with intravitreal injection of anti-VEGF agents: a review of literature. Eye. 2013;27:787–794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.American Academy of Ophthalmology. How to give intravitreal injections. 2013. Available at: www.aao.org/eyenet/article/how-to-give-intravitreal-injections. Accessed April 14, 2021.
- 3.Kahook MY, Schuman JS, Noecker RJ. Intravitreal bevacizumab in a patient with neovascular glaucoma. Ophthalmic Surg Lasers Imaging. 2006;37:144–146. [PubMed] [Google Scholar]
- 4.Kahook MY, Schuman JS, Noecker RJ. Needle bleb revision of encapsulated filtering bleb with bevacizumab. Ophthalmic Surg Lasers Imaging. 2006;37:148–150. [PubMed] [Google Scholar]
- 5.Kahook MY, Kimura AE, Wong LJ, et al. Sustained elevation in intraocular pressure associated with intravitreal bevacizumab injections. Ophthalmic Surg Lasers Imaging. 2009;40:293–295. [DOI] [PubMed] [Google Scholar]
- 6.Felfeli T, Hostovsky A, Trussart R, et al. Hypotensive efficacy of topical brimonidine for intraocular pressure spikes following intravitreal injections of antivascular endothelial growth factor agents: a randomised crossover trial. Br J Ophthalmol. 2019;103:1388–1394. [DOI] [PubMed] [Google Scholar]
- 7.El Chehab H, Le Corre A, Agard E, et al. Effect of topical pressure-lowering medication on prevention of intraocular pressure spikes after intravitreal injection. Eur J Ophthalmol. 2013;23:277–283. [DOI] [PubMed] [Google Scholar]
- 8.de Vries VA, Bassil FL, Ramdas WD. The effects of intravitreal injections on intraocular pressure and retinal nerve fiber layer: a systematic review and meta-analysis. Sci Rep. 2020;10:13248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bakri SJ, Moshfeghi DM, Francom S, et al. Intraocular pressure in eyes receiving monthly ranibizumab in 2 pivotal age-related macular degeneration clinical trials. Ophthalmology. 2014;121:1102–1108. [DOI] [PubMed] [Google Scholar]
- 10.Foss AJE, Scott LJ, Rogers CA, et al. Changes in intraocular pressure in study and fellow eyes in the IVAN trial. Br J Ophthalmol. 2016;100:1662–1667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhou Y, Zhou M, Xia S, et al. Sustained elevation of intraocular pressure associated with intravitreal administration of anti-vascular endothelial growth factor: a systematic review and meta-analysis. Sci Rep. 2016;6:39301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Eadie BD, Etminan M, Carleton BC, et al. Association of repeated intravitreous bevacizumab injections with risk for glaucoma surgery. JAMA Ophthalmol. 2017;135:363–368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cui QN, Gray IN, Yu Y, et al. Repeated intravitreal injections of anti-vascular endothelial growth factors and risk of intraocular pressure medication use. Graefes Arch Clin Exp Ophthalmol. 2019;257:1931–1939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jeong S, Sagong M, Chang W. Acute angle closure attack after an intravitreal bevacizumab injection for branch retinal vein occlusion: a case report. BMC Ophthalmol. 2017;17:25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kim IK, Baek J. A case of refractory acute angle closure glaucoma after an intravitreal bevacizumab injection. Korean J Ophthalmol. 2020;34:493–494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Alkin Z, Perente I, Altan C, et al. Changes in anterior segment morphology after intravitreal injection of bevacizumab and bevacizumab–triamcinolone acetate combination. Eur J Ophthalmol. 2013;23:504–509. [DOI] [PubMed] [Google Scholar]
- 17.Entezari M, Ramezani A, Yaseri M. Changes in retinal nerve fiber layer thickness after two intravitreal bevacizumab injections for wet type age-related macular degeneration. J Ophthalmic Vis Res. 2014;9:449–452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Horsley MB, Mandava N, Maycotte MA, et al. Retinal nerve fiber layer thickness in patients receiving chronic anti-vascular endothelial growth factor therapy. Am J Ophthalmol. 2010;150:558.e1–561.e1. [DOI] [PubMed] [Google Scholar]
- 19.Jo Y-J, Kim W-J, Shin I-H, et al. Longitudinal changes in retinal nerve fiber layer thickness after intravitreal anti-vascular endothelial growth factor therapy. Korean J Ophthalmol. 2016;30:114–120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Martinez-de-la-Casa JM, Ruiz-Calvo A, Saenz-Frances F, et al. Retinal nerve fiber layer thickness changes in patients with age-related macular degeneration treated with intravitreal ranibizumab. Invest Ophthalmol Vis Sci. 2012;53:6214–6218. [DOI] [PubMed] [Google Scholar]
- 21.Parlak M, Oner FH, Saatci AO. The long-term effect of intravitreal ranibizumab on retinal nerve fiber layer thickness in exudative age-related macular degeneration. Int Ophthalmol. 2015;35:473–480. [DOI] [PubMed] [Google Scholar]
- 22.Soheilian M, Karimi S, Montahae T, et al. Effects of intravitreal injection of bevacizumab with or without anterior chamber paracentesis on intraocular pressure and peripapillary retinal nerve fiber layer thickness: a prospective study. Graefes Arch Clin Exp Ophthalmol. 2017;255:1705–1712. [DOI] [PubMed] [Google Scholar]
- 23.Swaminathan SS, Kunkler AL, Quan AV, et al. Rates of RNFL thinning in patients with suspected or confirmed glaucoma receiving unilateral intravitreal injections for exudative AMD. Am J Ophthalmol. 2021;226:206–216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Valverde-Megías A, Ruiz-Calvo A, Murciano-Cespedosa A, et al. Long-term effect of intravitreal ranibizumab therapy on retinal nerve fiber layer in eyes with exudative age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol. 2019;257:1459–1466. [DOI] [PubMed] [Google Scholar]
- 25.Kahook MY, Noecker RJ, Abdelghani WM, et al. Filtering bleb rupture after intravitreal triamcinolone acetonide injection. Ophthalmic Surg Lasers Imaging. 2008;39:232–233. [DOI] [PubMed] [Google Scholar]
- 26.Wen JC, Reina-Torres E, Sherwood JM, et al. Intravitreal anti-VEGF injections reduce aqueous outflow facility in patients with neovascular age-related macular degeneration. Invest Ophthalmol Vis Sci. 2017;58:1893–1898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cavet ME, Vittitow JL, Impagnatiello F, et al. Nitric oxide (NO): an emerging target for the treatment of glaucoma. Invest Ophthalmol Vis Sci. 2014;55:5005–5015. [DOI] [PubMed] [Google Scholar]
- 28.Morshedi RG, Ricca AM, Wirostko BM. Ocular hypertension following intravitreal antivascular endothelial growth factor therapy: review of the literature and possible role of nitric oxide. J Glaucoma. 2016;25:291–300. [DOI] [PubMed] [Google Scholar]
- 29.Ricca AM, Morshedi RG, Wirostko BM. High intraocular pressure following anti-vascular endothelial growth factor therapy: proposed pathophysiology due to altered nitric oxide metabolism. J Ocul Pharmacol Ther. 2014;31:2–10. [DOI] [PubMed] [Google Scholar]
- 30.Izzedine H, Rixe O, Billemont B, et al. Angiogenesis inhibitor therapies: focus on kidney toxicity and hypertension. Am J Kidney Dis. 2007;50:203–218. [DOI] [PubMed] [Google Scholar]
- 31.Gal-Or O, Dotan A, Dachbash M, et al. Bevacizumab clearance through the iridocorneal angle following intravitreal injection in a rat model. Exp Eye Res. 2016;145:412–416. [DOI] [PubMed] [Google Scholar]
- 32.Huet F, Miller J, Miller PE, et al. Observation of silicone oil within the vitreous and sclera following intravitreal administration of biotherapeutics using insulin syringes in cynomolgus monkeys. Toxicol Pathol. 2021;49:590–597. [DOI] [PubMed] [Google Scholar]
- 33.Liu L, Ammar DA, Ross LA, et al. Silicone oil microdroplets and protein aggregates in repackaged bevacizumab and ranibizumab: effects of long-term storage and product mishandling. Invest Ophthalmol Vis Sci. 2011;52:1023–1034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kahook MY, Liu L, Ruzycki P, et al. High-molecular-weight aggregates in repackaged bevacizumab. Retina. 2010;30:887–892. [DOI] [PubMed] [Google Scholar]
- 35.Khurana RN, Chang LK, Porco TC. Incidence of presumed silicone oil droplets in the vitreous cavity after intravitreal bevacizumab injection with insulin syringes. JAMA Ophthalmol. 2017;135:800–803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lode HE, Gjølberg TT, Foss S, et al. A new method for pharmaceutical compounding and storage of anti-VEGF biologics for intravitreal use in silicone oil-free prefilled plastic syringes. Sci Rep. 2019;9:18021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hoang QV, Mendonca LS, Della Torre KE, et al. Effect on intraocular pressure in patients receiving unilateral intravitreal anti-vascular endothelial growth factor injections. Ophthalmology. 2012;119:321–326. [DOI] [PubMed] [Google Scholar]
- 38.Karakurt Y, Ucak T, Tasli G, et al. The effects of intravitreal ranibizumab, aflibercept or dexamethasone implant injections on intraocular pressure changes. Med Sci Monit. 2018;24:9019–9025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sayah DN, Szigiato A-A, Mazzaferri J, et al. Correlation of ocular rigidity with intraocular pressure spike after intravitreal injection of bevacizumab in exudative retinal disease. Br J Ophthalmol. 2021;105:392–396. [DOI] [PubMed] [Google Scholar]
- 40.Sternfeld A, Ehrlich R, Weinberger D, et al. Effect of different lens status on intraocular pressure elevation in patients treated with anti-vascular endothelial growth factor injections. Int J Ophthalmol. 2020;13:79–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Choi DY, Ortube MC, McCannel CA, et al. Sustained elevated intraocular pressures after intravitreal injection of bevacizumab, ranibizumab, and pegaptanib. Retina. 2011;31:1028–1035. [DOI] [PubMed] [Google Scholar]
- 42.Good TJ, Kimura AE, Mandava N, et al. Sustained elevation of intraocular pressure after intravitreal injections of anti-VEGF agents. Br J Ophthalmol. 2011;95:1111–1114. [DOI] [PubMed] [Google Scholar]
- 43.Mathalone N, Arodi-Golan A, Sar S, et al. Sustained elevation of intraocular pressure after intravitreal injections of bevacizumab in eyes with neovascular age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol. 2012;250:1435–1440. [DOI] [PubMed] [Google Scholar]
- 44.Tseng JJ, Vance SK, Della Torre KE, et al. Sustained increased intraocular pressure related to intravitreal antivascular endothelial growth factor therapy for neovascular age-related macular degeneration. J Glaucoma. 2012;21:241–247. [DOI] [PubMed] [Google Scholar]
- 45.Vo Kim S, Fajnkuchen F, Sarda V, et al. Sustained intraocular pressure elevation in eyes treated with intravitreal injections of anti-vascular endothelial growth factor for diabetic macular edema in a real-life setting. Graefes Arch Clin Exp Ophthalmol. 2017;255:2165–2171. [DOI] [PubMed] [Google Scholar]
- 46.Lam J, Luttrell I, Ding L, et al. Effect of prior glaucoma surgery on intraocular pressure immediately after anti-vascular endothelial growth factor injection. Graefes Arch Clin Exp Ophthalmol. 2019;257:2489–2494. [DOI] [PubMed] [Google Scholar]
- 47.Kim JE, Mantravadi AV, Hur EY, et al. Short-term intraocular pressure changes immediately after intravitreal injections of anti-vascular endothelial growth factor agents. Am J Ophthalmol. 2008;146:930.e1–934.e1. [DOI] [PubMed] [Google Scholar]
- 48.Dugel PU, Koh A, Ogura Y, et al. HAWK and HARRIER: phase 3, multicenter, randomized, double-masked trials of brolucizumab for neovascular age-related macular degeneration. Ophthalmology. 2020;127:72–84. [DOI] [PubMed] [Google Scholar]
- 49.Rezkallah A, Mathis T, Denis P, et al. XEN gel stent to treat intraocular hypertension after dexamethasone-implant intravitreal injections: 5 cases. J Glaucoma. 2019;28:e5–e9. [DOI] [PubMed] [Google Scholar]
- 50.Theoulakis PE, Lepidas J, Petropoulos IK, et al. Effect of brimonidine/timolol fixed combination on preventing the short-term intraocular pressure increase after intravitreal injection of ranibizumab. Klin Monatsbl Augenheilkd. 2010;227:280–284. [DOI] [PubMed] [Google Scholar]
- 51.Katayama BYNY, Bonini-Filho MA, Messias AMV, et al. Comparison of acetazolamide, brimonidine, and anterior chamber paracentesis for ocular hypertension control after initial intravitreal bevacizumab injection: a randomized clinical trial. J Glaucoma. 2014;23:461–463. [DOI] [PubMed] [Google Scholar]
- 52.Rusu IM, Deobhakta A, Yoon D, et al. Intraocular pressure in patients with neovascular age-related macular degeneration switched to aflibercept injection after previous anti-vascular endothelial growth factor treatments. Retina. 2014;34:2161–2166. [DOI] [PubMed] [Google Scholar]
- 53.Regeneron Pharmaceuticals. A phase 3, double-masked, randomized study of the efficacy and safety of intravitreal aflibercept injection in patients with moderately severe to severe nonproliferative diabetic retinopathy. clinicaltrials.gov; 2020. Available at: https://clinicaltrials.gov/ct2/show/NCT02718326. Accessed April 13, 2021.


