Abstract
During the COVID-19 pandemic lasting now for well more than a year, nearly 247 million cases have been diagnosed and over 5 million deaths have been recorded worldwide as of November 2021. The devastating effects of the SARS-CoV-2 virus on the immune system lead to the activation of signaling pathways involved in inflammation and the production of inflammatory cytokines. SARS-CoV-2 displays a great deal of homology with other coronaviruses, especially SARS-CoV and MERS-CoV which all display similar components which may serve as targets, namely the Spike (S) protein, the main protease (MPro) which is a chymotrypsin-like protease (CLPro) and RNA-directed RNA polymerase (RdRp). Natural constituents found in traditional herbal medicines, dietary supplements and foods demonstrate activity against SARS-CoV-2 by affecting the production of cytokines, modulating cell signaling pathways related to inflammation and even by direct interaction with targets found in the virus. This has been demonstrated by the application of fluorescence resonance energy transfer (FRET) experiments, assays of cytopathic effect (CPE) and in silico molecular docking studies that estimate binding strength. Glycyrrhizin, flavonoids such as quercetin, kaempferol and baicalein, and other polyphenols are the most common constituents found in Traditional Chinese Medicines that modulate inflammation and cell signaling pathways, and bind viral targets demonstrating valuable effects against SARS-CoV-2. However, the bioavailability of these natural products and their dependence on each other in extracts make it difficult to assess their actual utility in the treatment of COVID-19. Therefore, more can be learned through rational drug design based on natural products and from well-designed clinical trials employing specific doses of standardized combinations.
Keywords: COVID-19, SARS-CoV-2, Herbal, Botanical, Natural Products
Introduction
The COVID-19 pandemic has been with us now for more than a year. With nearly 247 million cases confirmed and more than 5 million deaths worldwide [1], science continues to learn more and more about how to treat the acute effects of mild, serious and severe infection and long-term sequelae of the virus. At this point there is much concern about the rise of new variants of SARS-CoV-2 that are transmitted more easily from to person. Worries persist about these variants being able to evade the protection offered by new vaccines that have entered the marketplace since late 2020, due to mutations affecting the ability to be recognized by antibodies [2, 3].
SARS-CoV-2 is a betacoronavirus spread by airborne droplets from the respiratory tracts of infected individuals. One reason why severe infection with SARS-CoV-2 is so dangerous is the effect the virus has on the immune system where the lungs are overrun with pro-inflammatory cytokines, often called “cytokine storm.” Excessive production and release of cytokines such as Tumor Necrosis Factor Alpha (TNF-α), Interferon Gamma (IFN-γ), and Interleukins (IL-1α, IL-1β, IL-2, IL-6, IL-15 and IL-18) is believed to cause the symptoms of moderate to severe COVID-19 infection as well as multiple organ failure and cell death. Signaling pathways involved include the Nuclear Factor Kappa B (NFκB) and IFN pathways and the production of intracellular nitric oxide synthase (iNOS) and nitric oxide (NO) via the interferon regulatory factor 1/signal transducer and activator of transcription factor 1 (IRF1/STAT1) pathways [4]. Platanitis and Dekker have reviewed the roles of cytokines in these signaling pathways and their relationships to inflammation [5].
The genome of SARS-CoV-2 shares approximately 85% homology with SARS-CoV, the coronavirus that caused the SARS atypical pneumonia outbreak of 2002–2003. The proteins of SARS-CoV-2 share between 65% and 97% homology with those of SARS-CoV. The viral genome of SARS-CoV-2 also demonstrates 35–48% sequence homology with the envelope and nucleocapsid proteins and surface and membrane glycoproteins of MERS-CoV, the coronavirus that caused the Middle East Respiratory Syndrome outbreaks of 2012 and 2015 [6, 7]. MERS-CoV was originally isolated from the sputum of a man living in the Arabian Peninsula. Genetic analysis of MERS-CoV identified it as a betacoronavirus related to other betacoronaviruses transmitted by bats [8]. Civets and dromedary camels are believed to be the intermediate hosts of MERS-CoV [9].
The viral genome of SARS-CoV-2 is a single-stranded (+) RNA polymer coding for a number of proteins in open reading frames (ORFs) that play roles in viral structure, host cell penetration, replication and immunogenicity [10]. As such, SARS-CoV-2 has multiple targets for drug action that researchers are able to exploit in order to slow and diminish the effects of acute infection.
As targets, SARS-CoV-2 produces an envelope protein (E) and a nucleocapsid protein (N), important for viral structure, a main protease (MPro) which is a chymotrypsin-like protease (CLPro), a papain-like protease (PLPro), helicases and RNA-directed RNA polymerase (RdRp), all vital for viral replication. The spike protein (S), is integral for viral entry into host cells by interacting with its host cell receptor, angiotensin converting enzyme 2 (ACE2). ACE2 is found on the cells of the pulmonary epithelium as well as many other tissues throughout the body such as kidney, brain, heart and the vasculature [11]. Herbal formulae contain plant materials whose constituents are able to inhibit the proteases, thus inhibiting viral replication, and interact with either the spike protein itself, the spike protein-ACE2 receptor complex or a transmembrane serine protease (TMPRSS2) found on host cells that cleaves the spike protein into S1 and S2 segments to facilitate viral entry into host cells [12].
Such targets can be affected by a number of drugs now being repurposed, such as dexamethasone and tocilizumab, and newly marketed drugs such as remdesivir and favipiravir. Dexamethasone is a glucocorticoid used routinely for its anti-inflammatory properties and tocilizumab is a monoclonal antibody whose target is the IL-6 receptor. It is believed that blocking the IL-6 receptor could reduce the effects of massive cytokine release during severe infection [13]. Remdesivir and favipiravir are inhibitors of RdRp found to be effective in the treatment of COVID-19 [13].
Even natural constituents found in herbal medicines, dietary supplements and foods demonstrate activity against SARS-CoV-2 and related coronaviruses, SARS-CoV and MERS [14]. In fact, since the early days of the pandemic clinicians in China have used botanically based Traditional Chinese Medicines as adjunct therapy in the treatment of patients infected with SARS-CoV-2 with remarkable success in decreasing the severity of disease and the length of stay in the hospital [15–24]. However, much remains to be learned from conducting well designed clinical trials of scope and magnitude large enough to explain with certainty that natural medicines, dietary supplements and foods rich in certain phytochemicals have a place in the therapy of COVID-19 [25].
What then are the common phytochemicals and chemical classes of interest from natural sources that have activity against SARS-CoV-2, how do they work, what are their targets and how do we assess their activity? Moreover, why is it important that we understand that this activity exists and how can we exploit it? Physicians in the west may not be ready, able or willing to employ Traditional Chinese Medicine (TCM) as a modality of practice, but if we were to understand the activity of these natural constituents it may be possible to develop new drugs capable of reducing the symptoms and severity of acute infection, reduce the length of stay in hospitals and improve the quality of life in patients infected with SARS-CoV-2 [25]. The NIH Center for Complementary and Integrative Health is also looking for insight into complementary and integrative methods that may even include the use of herbs, dietary supplements and adjustments to the diet in order to mitigate the post-acute sequelae (PASC) of SARS-CoV-2 [26]. In this review, Traditional Chinese Medicines and their most common chemical constituents having activity against coronaviruses, including SARS-CoV-2, are surveyed with discussion relating to their possible mechanisms of action against SARS-CoV-2.
Traditional Chinese medicine (TCM)
During early 2020 physicians in China, where the outbreak of SARS-CoV-2 originated, began using formulae known and used in TCM to treat viral infections, or what are commonly referred to in TCM as “pestilence” [27]. TCM uses multi-component herbal formulae to achieve treatment of disease. Several formulae were employed that were found to modulate reproducibility of the virus by affecting viral protease enzymes, and modulate the concentration of cytokines in the host, thus affecting the host immune response to the virus [25].
There are at least 116 different, multicomponent TCM formulae and patent medicines composed of more than 215 different plants, plant parts and extracts used in various combinations with one another [25, 28–32]. Although vaccines are now widely used in China and around the world [33], these herbal formulae have been employed as adjuncts in the treatment of patients infected with SARS-CoV-2 for various purposes with some success [26]. Many of the plants found in these formulae are not necessarily found commonly as dietary supplements in the US, but many of their chemical constituents that have activity against the targets of SARS-CoV-2, such as kaempferol and quercetin, can be found in dietary supplements. For example, Ginkgo biloba is a popular dietary supplement that is rich in both kaempferol and quercetin [34]. The question then becomes, how can one classify these herbal formulae into a more meaningful subset of information of which to take advantage, chemically? We need to examine the constituents of plants and their activity in inhibiting SARS-CoV, MERS-CoV and SARS-CoV-2. Doing so will allow us to learn how these phytochemicals could be used as lead compounds in the development of drugs that can be used to treat and diminish the severity of COVID-19.
Assessment of activity in vitro
There are many ways to determine and analyze the interactions of various phytochemicals with the targets of SARS-CoV-2. FRET-Förster or Fluorescence resonance energy transfer (FRET) experiments use confocal microscopy to analyze the transfer of photons from a donor dye to an acceptor dye over distance through space during excitation by light in order to describe the binding strength of a labeled ligand to its labeled target [35]. Binding is often expressed as an IC50 value, the concentration necessary to inhibit 50% of target activity.
An assay of cytopathic effect (CPE) is a controlled colorimetric assay where cells are incubated in a well plate, exposed to a virus and a suspected antiviral compound in varying concentrations. A log dose-response curve is generated and the EC50 value represents the concentration of the test compound necessary to inhibit 50% of viral cell death. A lower the EC50 value indicates better ability of the test compound to inhibit viral cell death.
In Silico molecular modeling uses the established crystal structure of viral targets from SARS-CoV-2, or closely related targets of related viruses such as SARS-CoV, docked with a known subset of ligands to establish the binding strength of competitive inhibitors of the target. A variety of targets from both SARS-CoV and SARS-CoV-2 are noted in Table 1 [36–46]. Binding strength is expressed in terms of kcal/mol. It is best for the binding strength of a test compound to be as close as possible to the binding strength of a known inhibitor or control. It is also important to note that in silico docking is no indication that a compound will absolutely inhibit a target, but it offers a good start for engaging in drug design [47].
Table 1.
Targets of SARS-CoV and SARS-CoV-2 for in silico binding studies
Glycyrrhizin
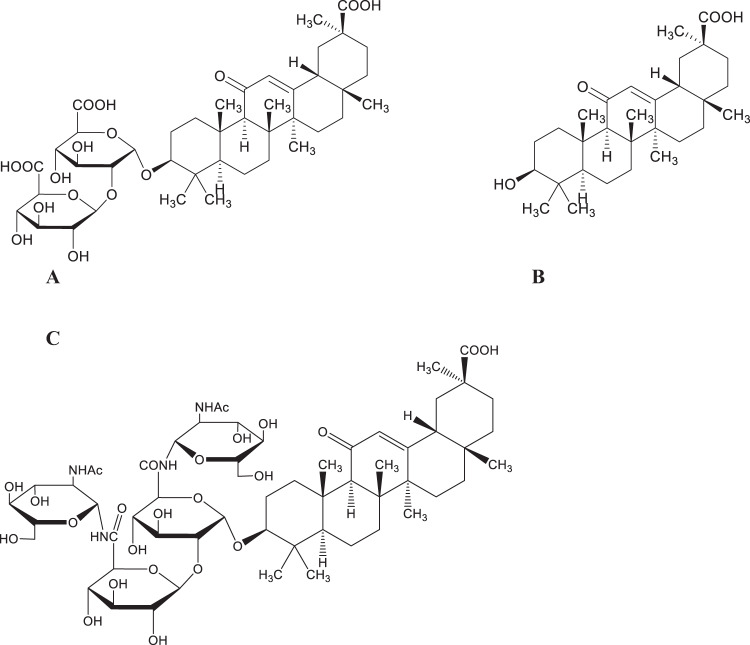
In reviewing formulae from TCM, the most commonly used herb was Glycyrrhizae radix et rhizoma, from Glycyrrhiza glabra L. and other Glycyrrhiza species, also known in the West as licorice root. This herb, with its anti-inflammatory properties also activates granulocytes, natural killer cells and eosinophils, appears to inhibit both viral entry into host cells and SARS-CoV-2 replication [29, 48–50]. Glycyrrhizin, the major triterpenoid saponin found in licorice root (Fig. 1A), inhibits the binding of SARS-CoV S proteins to ACE2 by binding four residues of ACE2 with a binding free energy similar to flavonoids, thus blocking the ability of SARS-CoV-2 to enter host cells [51]. Glycyrrhizin and its aglycone, 18β-glycyrrhetinic acid, also affect cell signaling pathways involving protein kinase C (PKC), activator protein 1 (AP1) and NFκB. These two phytochemicals mediate the production of cytokines, inflammation and apoptosis resulting from their production. By decreasing neutrophil infiltration, decreasing nitric oxide synthase and nitric oxide (NO) production, free radical sequestration the production of protective IFN-γ and apoptosis increase, while the production of pro-inflammatory cytokines IL-6, IL-10 and TNF-α decrease [28, 48, 50–53]. As used, when taken orally glycerrhizin is dosed as tablets containing approximately 300 mg of glycyrrhizin, capsules containing 150 mg diammonium glycyrrhizinate [31] or as the 1700 mg of the crude herb containing 5.65% glycyrrhizin. When administered intravenously the dose of glycyrrhizin is approximately 240 mg [51].
Fig. 1.
A Glycyrrhizin. B Glycyrrhetinic acid. C 2-acetamido-β-D-glucopyranosylglycyrrhizin
In a CPE assay glycyrrhizin’s EC50 was 365 μM and the EC50 for 18β-glycyrrhetinic acid (Fig. 1B) was >20 μM, but only glycyrrhizin was selective in maintaining cell viability with a selectivity index of >65. Interestingly, the 2-acetimido-β-D-glucopyranosylamine (N-acetylglucosamine amide-NAGA) derivative of glycyrrhizin (Fig. 1C), demonstrated an EC50 value of 40 μM with a selectivity index of >75, compared to 13 other glycyrrhizin derivatives, 6 of which were potent, but none of which were selective in their cytopathic activity. Presumably, the NAGA derivative binds well with the highly glycosylated S-proteins of SARS-CoV thus increasing the potency of this derivative [54].
Molecular docking studies were carried out in silico with glycyrrhizin and glycyrrhizic acid, and a number of SARS-CoV-2 targets. Lower docking scores indicate better binding of the compound with its putative targets. The results of molecular docking studies with glycyrrhizin and glycyrrhizic acid are shown in Table 2. These encouraging results should shed light into possible modifications relative to the rational design of anti-coronavirus agents based on the triterpenoid saponin, glycyrrhizin [55, 56].
Table 2.
Binding energies of glycyrrhizic acid and glycyrhhizin with SARS-CoV-2 target proteins
| Target/PDB Code | Glycyrrhizic Acid [55] | Glycyrrhizin [56] |
|---|---|---|
| MPro / 7K3T | −8.7 | −8.1 |
| PLPro / 6W9C | −8.2 | −7.9 |
| RdRp / 7BV2 | −9.9 | – |
| N / 6YVO | – | −7.9 |
| S / 6VXX | −9.3 | – |
| Human ACE2 / 7A98 | −9.5 | – |
MPro-Main Protease (3CLPro, Chymotripsin-like Protease); PLPro-Papain-like Protease; RdRp-RNA-dependent RNA polymerase; N-Nucleocapsid protein; S-Spike protein; Human ACE2-Angiotensin Converting Enzyme 2 (human host cell surface)
Flavonoids and polyphenolics
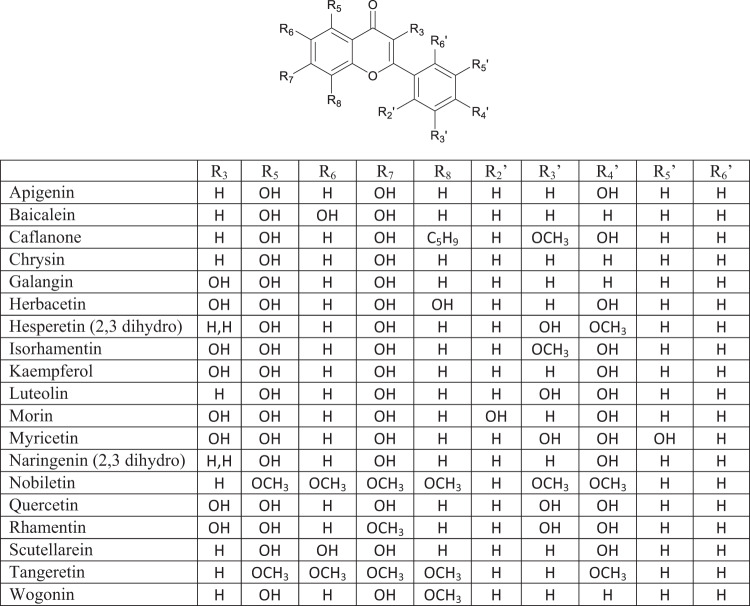
Flavonoids (Fig. 2) and other polyphenolic compounds are some of the most widely found chemical compounds in plants used both as medicinal herbs in TCM and other systems of herbal medicine, and from plants eaten in the diet [57]. Flavonoids are polyhydroxylated 2-phenylchromones. They, their glycosides and some bioisosteres demonstrate antiviral activity against SARS-CoV, MERS-CoV and other human coronaviruses as well as influenza A virus [58]. Flavonoids not only stimulate host immune responses to viral exposure; they also suppress excessive inflammatory reactions and modulate autophagy and immunoproteasome effects [57, 59, 60].
Fig. 2.
Flavonoids
The flavonoids apigenin chrysin, galangin, hesperetin, kaempferol, luteolin, naringenin and quercetin (Fig. 2), the flavonoid glycoside rutin (Fig. 3A), the isoflavone phytoestrogen daidzein (Fig. 3B), neobavaisoflavone (Fig. 3C) and the catechins from dietary sources, such as green tea (Fig. 4), are flavonoids or closely related compounds found in a variety of fruits and vegetables. These compounds diminish a number of inflammatory processes related to cyclooxygenase-2 (COX-2) and iNOS activity, NFκB, Activator Protein-1 (AP-1) and mitogen activated protein kinase (MAPK) activities [57]. Such activities reduce the synthesis of inflammatory markers such as IL-1β, IL-6, IFN-γ and TNF-α, whose elevated concentrations are noted in patients experiencing severe symptoms of SARS-CoV-2 infection [57]. Non-flavonoid anti-oxidants and anti-inflammatories found in fruits, vegetables, grains and cereals such as caffeic, ferulic, p-coumaric, syringic and vanillic acids, eriodictyol, polydatin and resveratrol (Fig. 5), also play roles in inhibiting lipid peroxidation and free radical scavenging. These activities lead to the decreased synthesis and effects of the aforementioned markers of inflammation in SARS-CoV-2. In fact, eriodictyol, one of its glycosides, and polydatin, bind ACE2 and inhibit SARS-CoV-2 main protease (MPro) [57].
Fig. 3.
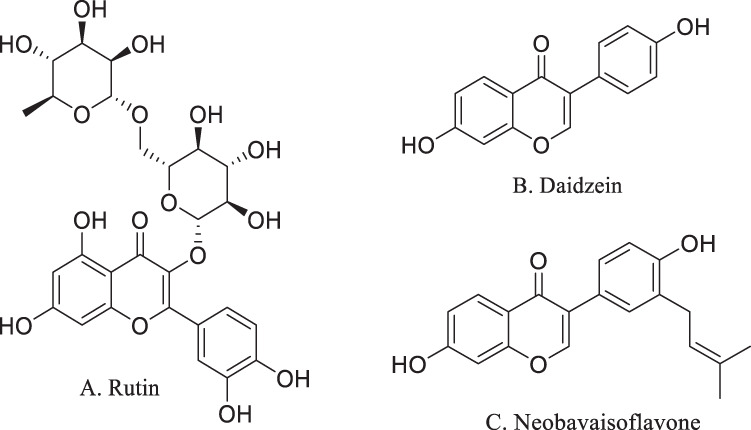
Flavonoid-related compounds. A rutin. B daidzein. C neobavaisoflavone
Fig. 4.
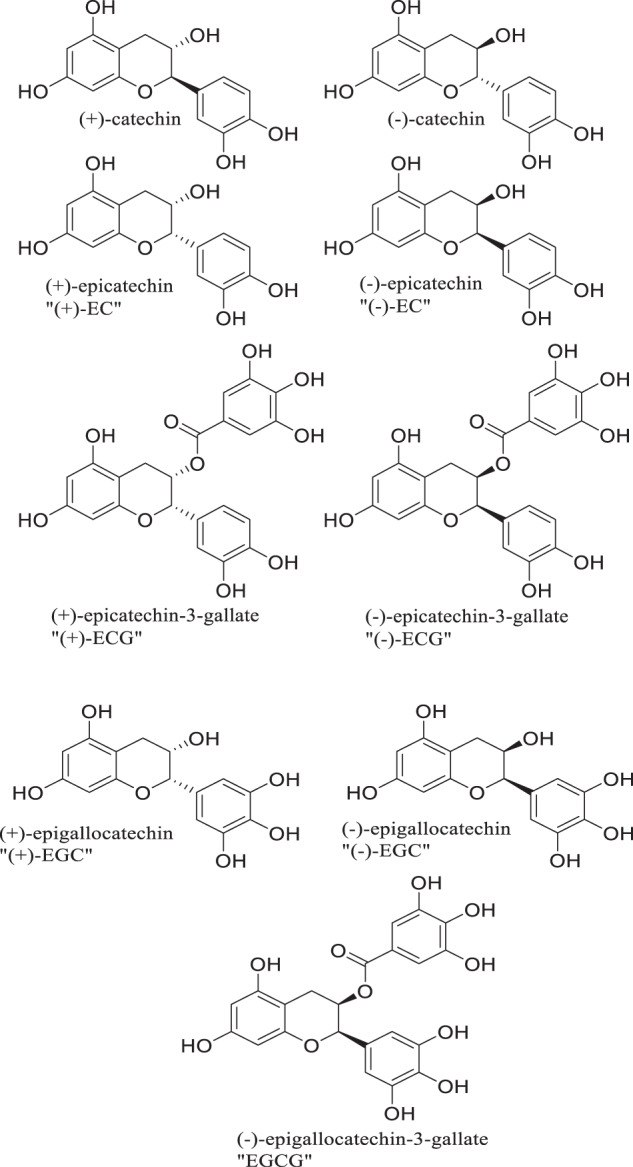
Catechins from green tea
Fig. 5.
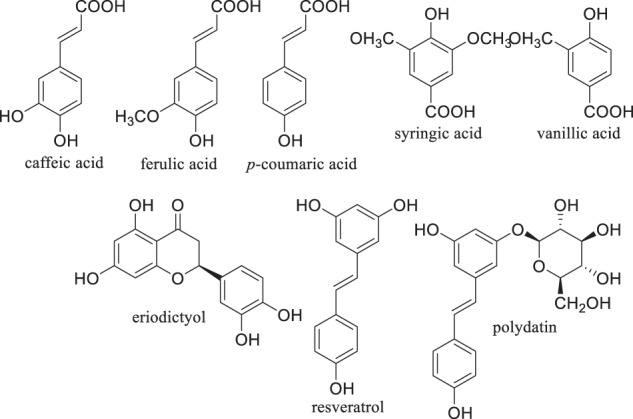
Non-flavonoid anti-oxidants and anti-inflammatories from the diet
A flavonoid enriched extract of the skullcap root, Scutellaria baicalensis, rich in the flavonoids, scutellarein, baicalein and its glycoside, baicalin, as well as other flavonoids decreased the production of inflammatory cytokines TNF-α, IL-6, monocyte chemotactic factor-1 (MCP-1) and nitric oxide in mouse lung tissues exposed to influenza A virus, while increasing the synthesis of protective cytokines IFN-γ and IL-10 [61]. In examining a TCM formula rich in flavonoids (baicalein, isorhamnetin, kaempferol, luteolin, naringenin, quercetin and wogonin), extracts rich in these compounds suppressed IL-6 production in macrophages in vitro, and downregulated TNF-α and NFκB signaling in addition to attenuating a host of other inflammatory signaling pathways [62].
Flavonoids interact with a number of viral targets including those involved with entry into host cells, genome transcription, post-translational modification and others related to viral viability. Solnier and Fladerer [59] summarized the results of flavonoids and related compounds binding to CLPro (MPro) and PLPro of SARS-CoV and MERS-CoV in FRET assays. With the lowest IC50 values being the best, flavonoids bound SARS-CoV CLPro with IC50 value ranges of 8.3–381 μM and MERS CoV CLPro with IC50 value ranges of 34.7–125.7 mM. Chalcones, dimeric structures of flavonoids, bound SARS-CoV CLPro with IC50 value ranges of 11.4–202.7 μM and MERS CoV CLPro with IC50 value ranges of 27.9–193.7 mM. Flavonoids bound SARS-CoV PLPro with IC50 value ranges of 3.7–66.2 μM and MERS-CoV PLPro with IC50 value ranges of 48.8–206.6 mM. Chalcones bound SARS-CoV PLPro with IC50 value ranges of 1.2–46.4 μM and MERS-CoV PLPro with IC50 value ranges of 42.1–171.6 mM (Table 3) [59]. Binding to CLPro is somewhat more erratic for flavonoids and chalcones, but binding to PLPro is less erratic and somewhat more potent. From this data, it may be more beneficial to base the development of PLPro inhibitors on chalcones, rather than flavonoids and to use flavonoids as the basis for the development of either CLPro or PLPro inhibitors.
Table 3.
IC50 value ranges of flavonoids and chalcones with SARS-CoV and MERS-CoV CLPro and PLPro [59]
| IC50 Value Ranges (μM) | ||||
|---|---|---|---|---|
| SARS-CoV CLPro | SARS-CoV PLPro | MERS-CoV CLPro | MERS-CoV PLPro | |
| Flavonoids | 8.3–381 | 3.7–66.2 | 34.7–125.7 | 48.8–206.6 |
| Chalcones | 11.4–202.7 | 1.2–46.4 | 27.9–193.7 | 42.1–171.6 |
Goyal and colleagues [63] completed molecular docking studies of SARS-CoV-2 RdRp with various flavonoids and triterpenes and compared them with uridine and thymidine triphosphates (UTP, TTP) and remdesivir, an effective RdRp inhibitor used in the treatment of COVID-19, as controls. Docking scores for the twenty compounds tested were all within 4 kcal/mol of the controls, and their binding free energies were on par with controls or at least within 80 kcal/mol of controls. The controls and the compounds tested appear to interact with the same amino acid residues in RdRp, namely serine, lysine, arginine, aspartate and isoleucine (Table 4). Molecular modeling demonstrates weak intermolecular interactions occurring between hydroxyl groups, ether oxygens and carbonyl oxygen atoms of the flavonoid and triterpene structures with amino acid residues of RdRp [63]. With respect to RdRp and ACE2, Goyal and colleagues found a number of binding interactions between various flavonoids/flavonoid glycosides and amino acid residues that share commonality with residues to which glycyrrhizic acid and glycyrrhizinic acid bind as noted by Vardhan and Sahoo (Table 4) [55, 63]. While molecular docking studies are not a guarantee that such compounds would interact with the target in vivo, there is some likelihood that they can, and by examining these commonalities in binding interactions these molecules such as these could serve as useful templates for antiviral drug development.
Table 4.
Binding interactions common to flavonoids and glycyrrhizic acid in RdRp, and flavonoids and glycyrrhizinic acid in ACE2 [55, 63]
| RdRp (PDB Codes 6M71 and 7BV2) [41, 89] | ACE2 (PDB Codes 6M17 and 2AJF) [90, 91] | ||
|---|---|---|---|
| Glycyrrhizic Acid [55] | Flavonoida/Flavonoid Glycosideb [63] | Glycyrrhzinic Acid [55] | |
| Arg555, Ser682 | Apigenina | Ala348 | |
| Arg555, Ile548 | Apigenin-7-O-glucosideb | Ala348, Arg393, Asn394 | |
| Arg555 | Galangina | Asn394, Asp350, Arg393 | |
| Arg555, Ser682 | Hesperetina | Ala348, Arg393, Asn394 | |
| Lys545, Ile548 | Hesperidinb | Tyr385, Ala348, Arg393 | |
| Arg555 | Kaempferola | Asn394 | |
| Ile548 | Luteolina | Ala348 | |
| Ile548 | Luteolin-7-O-glucosideb | Glu402 | |
| Ser682 | Naringenina | – | |
| Ser549, Asp623 | Naringinb | Ala348, Arg393 | |
| Ser682 | Quercetina | Ala348 | |
| Ile548, Asp623, Lys551, Arg555 | Rutina | Asn394 | |
aFlavonoid
bFlavonoid glycoside
Molecular docking studies of flavonoid and triterpene binding to ACE2 gave docking scores within 3 kcal/mol of control and binding free energy values within 30 kcal/mol of control. Compounds tested bound key alanine, aspartate and arginine residues in binding models. These residues were among those bound by the known ACE2 inhibitor, GL1001 (Ala348, Trp349, Asp350, Asp382 and Arg393). Again, models reveal weak intermolecular interactions occurring between hydroxyl groups, ether oxygens and carbonyl oxygen atoms of the flavonoid and triterpene structures with amino acid residues of ACE2 [63]. The structures of such flavonoids and triterpenes could be optimized to serve as potential leads for drug development.
Su and colleagues studied the effects of a Chinese patent medicine, whose name is Shuanghuanglian, on SARS-CoV-2 in vitro [64]. Shuanghuanglian is a multicomponent composition rich in the flavonoid, baicalein, found in Scutellaria baicalensis, and a number of other related compounds from Forsythia suspensa. As determined by molecular modeling studies, baicalein binds to a core region of the SARS-CoV-2 CLPro (MPro), by hydrogen bond interactions with leucine and glycine, glutamate and serine and glycine with the assistance of water, and by hydrophobic and/or pi stacking interactions with glutamine, arginine, methionine, cysteine and histidine residues. Baicalein inhibits SARS-CoV-2 MPro with an IC50 of ~0.94 μM, as well. Baicalein’s glycoside, baicalin, also inhibited SARS-CoV-2 MPro, but inhibition was nearly 10-fold weaker at ~6.41 μM. Interestingly, seven other compounds from Shuanghuanglian, namely the catecholic glycosides forsythoside A, B, E, H, I, isoforsythoside and scutellarein, a flavonoid closely related to baicalein, also inhibited SARS-CoV-2 MPro, albeit with potency less than that of baicalein, with IC50 values ranging from 2.88 to 10.17 μM. These compounds could also serve as initial leads for anti-SARS-CoV-2 drug development.
A number of other compounds whose structures resemble the scaffolds of natural products have been found to inhibit SARS-CoV-2 MPro in the COVID Moonshot project. The COVID Moonshot project is a crowdsourced consortium of scientists from around the world using molecular modeling and crystallographic techniques, high-throughput synthesis, solubility and screening methodology, and artificial intelligence in the effort to find lead compounds active against SARS-CoV-2 MPro [65].
Not only do flavonoids and triterpenes display potential to bind ACE2, the cell surface receptor for SARS-CoV and SARS-CoV-2, flavonoids and their glycosides, gingerols and cannabinoids all demonstrate binding to the main protease MPro and S protein of SARS-CoV-2. Recall that the S protein of the virus interacts with ACE2 on the cell surface, whereby viral entry to the cell is gained. Binding free energies of flavonoids and their glycosides, gingerols and cannabinoids are low and comparable. The best binding to S protein was accomplished by epigallocatechin gallate, a well-known constituent of green tea, the flavonoid glycosides hesperidin, rhoifolin, pectolinarin and the cannabinoids, cannabidiol and Δ9-THC [66]. Thus, all of these compounds could serve as useful scaffolds for drug design and lead development of anti-SARS-CoV-2 drugs.
Cannabis
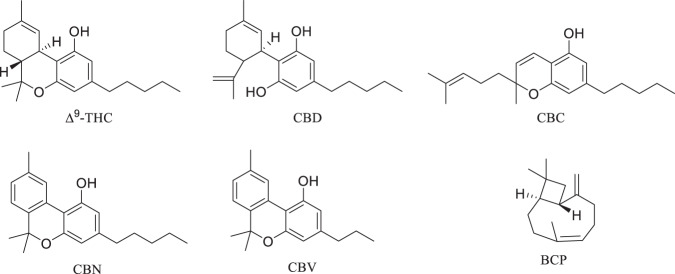
Cannabis sativa L. has been used as a phytomedicinal substance for centuries, dating back to the Han Dynasty in China [67]. Of more than 550 compounds identified from C. sativa, there are at least 113 cannabinoids, 140 terpenes and at least 23 flavonoids [68, 69]. Cannabinoids derived from Cannabis sativa L. bind CB1 and CB2 receptors in the endocannabinoid system [70]. While the psychoactive cannabinoid, Δ9-THC (Fig. 6) is known to bind CB1 receptors, promote relaxation of the vasculature and inhibit IL-1β production [71–73], cannabidiol (CBD, Fig. 6), cannabichromene (CBC, Fig. 6) and cannabinol (CBN, Fig. 6) bind CB2 receptors with resulting decreases in the proinflammatory cytokines [73, 74]. CBD has been shown to stimulate peroxisome-proliferator activated receptors alpha and gamma (PPAR-α and PPAR-γ) to attenuate the pathways leading to proinflammatory cytokine release [70, 73, 75, 76]. Through their actions at inflammasomes and the transient cation receptor potential channels (TRPs) these non-psychoactive cannabinoids depress the NFκB signaling pathway, decreasing the release of IL-6, IL-1β, IFN-γ and TNF-α in animal and in vitro models [77–79]. Thus, the interest in cannabinoids to diminish the effects of the cytokine storm as a serious manifestation of SARS-CoV-2 infection is pronounced, of late.
Fig. 6.
Selected constituents of Cannabis sativa L
In molecular docking studies of cannabinoids and their binding to ACE2, TMPSS2, IL-6 and the histone chaperone NRP1, CBD and cannabivarin (CBV, Fig. 6) demonstrated significant binding affinity with binding free energies between −8.2 and −8.9 kcal/mol [80]. Binding of the cannabinoid with the these target proteins results in downregulation of corresponding activity and an implied competition between binding of cannabinoids and the endogenous ligand [80].
Not only do cannabinoids diminish the release of proinflammatory cytokines, extracts high in CBD concentration also downregulate expression of the ACE2 receptor and TMPSS2 [81–83]. However, the effects on ACE2 and TMPSS2 expression cannot be related solely to CBD concentration [75, 82]. Extracts of cannabis contain bioactive terpenes as well. The relatively higher biological activity of cannabis extracts containing a combination of cannabinoids and terpenes may be explained by the entourage effect, where a complex mixture of compounds in a plant extract demonstrates greater biological activity than a single constituent alone [73, 82].
Terpenes are noted for the aromatic character and flavor they impart to cannabis plants [67]. Mentioned previously, terpenes found in cannabis extracts may account for an entourage effect. β-Caryophyllene (BCP, Fig. 6) is a terpene found in cannabis as well as a number of other plant species, and is a full agonist of CB2 receptors [84]. BCP also activates peroxisome-proliferator activated receptors alpha and gamma (PPAR-α and PPAR-γ) leading to attenuation of the pathways causing proinflammatory cytokine release [84]. Interestingly, BCP also binds SARS-CoV-2 MPro with a binding free energy of −7.2 kcal/mol, which is comparable to that of glycyrrhizin, −8.1 kcal/mol [69].
As stated, at least 23 flavonoids have been isolated from cannabis. Caflanone, a.k.a. isocannflavin B (Fig. 2), a flavonoid derived from cannabis, was shown to bind the metalloproteinase domain of ACE2 [85]. Other flavonoids and chloroquine also bind the metalloproteinase domain, however caflanone’s binding free energy of −7.9 kcal/mol is lower than that of chloroquine (−4.1 or −4.7 kcal/mol), implying better binding in the ACE2 metalloproteinase domain. Other flavonoids discussed above, such as myricetin and hesperitin, bind the ACE2 metalloproteinase domain with free energies of −8.9 kcal/mol and −9.1 kcal/mol respectively, comparable to that of caflanone [85].
Given the effects of cannabis constituents on systems involved in the serious complications of COVID-19 and the wide availability of cannabis in the community today, it is no wonder that there is so much interest in cannabis as a potential therapy for the severe complications of SARS-CoV-2 infection. However, the effects of smoking and/or vaping cannabis can be deleterious to the lungs, many unsubstantiated claims have been made in the media and there has not been adequate clinical research to support the use of cannabis as a treatment or preventive therapy for COVID-19 [86, 87].
Conclusion
While the flavonoids and their related compounds, and other naturally occurring anti-oxidants and anti-inflammatories discussed display these beneficial properties, it is important to realize their bioavailability from the diet is limited. When ingested these compounds are subject to hepatic first-pass metabolic transformations and conjugation reactions that decrease their absorption and promote their elimination from the body. Dabeek and Marra have reviewed the bioavailability of the flavonoids, quercetin and kaempferol. In their work they detailed a number of food sources for these flavonoids and their glycosides, and discussed the kinetics behind their absorption, metabolism and excretion [88]. Complicating matters is the notion that extracts of natural products contain constituents that often act in synergy to illicit their effects, and that when individual constituents are purified their activity diminishes or disappears. With this in mind, a diet consistently rich in fruits, vegetables, grains and cereals affords the best chances for these bioactive compounds to be present in order to have and maintain their effects on viral entry and replication, and the inflammatory processes related to the consequences of moderate to severe infection. As claims are concerned, we must be reminded that US Federal Law does not permit claims for the treatment, cure or diagnosis of diseases such as COVID-19. Regardless, at this point it would be desirable to conduct well-designed clinical trials with specific doses of standardized products in order to determine the actual utility of natural compounds in the treatment of COVID-19.
Abbreviations
- ACE2
Angiotensin Converting Enzyme Type Two
- Ala
Alanine
- AP-1
Activator Protein One
- Arg
Arginine
- Asp
Aspartic Acid
- BCP
β-Caryophyllene
- CB1
Endocannabinoid receptor type 1
- CB2
Endocannabinoid receptor type 2
- CBC
Cannabichromene
- CBD
Cannabidiol
- CBN
Cannabinol
- CBV
Cannabivarin
- CLPro
Chymotrypsin-like Protease
- COVID-19
Coronavirus Disease, 2019
- COX-2
Cyclooxygenase Two
- CPE
Cytopathic Effect
- Δ9-THC
Delta-Nine Tetrahydrocannabinol
- E
Envelope Protein
- EC50
Effective Concentration at 50% Maximal Effect
- FRET
Förster or Fluorescence Resonance Energy Transfer
- IC50
Inhibitory Concentration at 50% Inhibition
- IFN-γ
Interferon-gamma
- IL
Interleukin
- IL-1α
Interleukin 1-alpha
- IL-1β
Interleukin 1-beta
- iNOS
Intracellular Nitric Oxide Synthase
- IRF1
Interferon Regulatory Factor One
- Kcal/mol
Kilocalories per mole
- MPro
Main Protease
- MAPK
Mitogen Activated Protein Kinase
- MCP-1
Monocyte Chemotactic Factor One
- MERS-CoV
Coronavirus that causes Middle-East Acute Respiratory Syndrome, in the outbreaks of 2012 and 2015
- Mg
Milligram
- μM
Micromolar
- N
Nucleocapsid Protein
- NAGA
N-Acetylglucosamine Amide
- NFκB
Nuclear Factor Kappa B
- NIH
National Institutes of Health (U.S.A.)
- NO
Nitric Oxide
- NRP1
Nucleosome Assembly Protein-1 Related Protein-1; Protein neuropilin 1
- ORF
Open Reading Frame
- PASC
Post-acute sequalae
- PLPro
Papain-like Protease
- PPAR
Peroxisome-proliferator activated receptor
- RNA
Ribonucleic Acid
- RdRp
RNA-directed RNA Polymerase
- S
Spike Protein
- S1
Spike Protein Segment One
- S2
Spike Protein Segment Two
- SARS-CoV
Coronavirus that causes Systemic Acute Respiratory Syndrome, in the epidemic of 2002-2003
- SARS-CoV-2
Coronavirus that causes Systemic Acute Respiratory Syndrome, in the pandemic of 2020-2021
- STAT1
Signal Transducer and Activator of Transcription Factor One
- TCM
Traditional Chinese Medicine
- TMPRSS2
Transmembrane Serine Protease Type Two
- TNF-α
Tumor Necrosis Factor-alpha
- Trp
Tryptophan
- TRP
Transient cation receptor potential channel
- TTP
Thymidine Triphosphate
- UTP
Uridine Triphosphate
Compliance with ethical standards
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Organization WH. WHO Coronavirus (COVID-19) Dashboard. World Health Organization. 2021. https://covid19.who.int/?adgroupsurvey=%7badgroupsurvey%7d&gclid=EAIaIQobChMIoejY_42S7wIVHj6tBh3IggpTEAAYASABEgJ8v_D_BwE. Accessed Aug 24 2021.
- 2.Chen AT, Altschuler K, Zhan SH, Chan YA, Deverman BE. COVID-19 CG enables SARS-CoV-2 mutation and lineage tracking by locations and dates of interest. Elife. 2021;10. 10.7554/eLife.63409. [DOI] [PMC free article] [PubMed]
- 3.To KK, Hung IF, Ip JD, Chu AW, Chan WM, Tam AR, et al. COVID-19 re-infection by a phylogenetically distinct SARS-coronavirus-2 strain confirmed by whole genome sequencing. Clin Infect Dis. 2020. 10.1093/cid/ciaa1275. [DOI] [PMC free article] [PubMed]
- 4.Karki R, Sharma BR, Tuladhar S, Williams EP, Zalduondo L, Samir P, et al. Synergism of TNF-α and IFN-γ triggers inflammatory cell death, tissue damage, and mortality in SARS-CoV-2 infection and cytokine shock syndromes. Cell. 2021;184:149–68.e17. doi: 10.1016/j.cell.2020.11.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Platanitis E, Decker T. Regulatory Networks Involving STATs, IRFs, and NFκB in Inflammation. Front Immunol. 2018;9:2542. doi: 10.3389/fimmu.2018.02542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Xu J, Zhao S, Teng T, Abdalla AE, Zhu W, Xie L, et al. Systematic comparison of two animal-to-human transmitted human coronaviruses: SARS-CoV-2 and SARS-CoV. Viruses. 2020;12. 10.3390/v12020244. [DOI] [PMC free article] [PubMed]
- 7.Grifoni A, Sidney J, Zhang Y, Scheuermann RH, Peters B, Sette A. A sequence homology and bioinformatic approach can predict candidate targets for immune responses to SARS-CoV-2. Cell Host Microbe. 2020;27:671–80.e2. doi: 10.1016/j.chom.2020.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zaki AM, van Boheemen S, Bestebroer TM, Osterhaus AD, Fouchier RA. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N. Engl J Med. 2012;367:1814–20. doi: 10.1056/NEJMoa1211721. [DOI] [PubMed] [Google Scholar]
- 9.Liu Z, Xiao X, Wei X, Li J, Yang J, Tan H, et al. Composition and divergence of coronavirus spike proteins and host ACE2 receptors predict potential intermediate hosts of SARS-CoV-2. J Med Virol. 2020;92:595–601. doi: 10.1002/jmv.25726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Malik YS, Sircar S, Bhat S, Sharun K, Dhama K, Dadar M, et al. Emerging novel coronavirus (2019-nCoV)-current scenario, evolutionary perspective based on genome analysis and recent developments. Vet Q. 2020;40:68–76. doi: 10.1080/01652176.2020.1727993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Samudrala PK, Kumar P, Choudhary K, Thakur N, Wadekar GS, Dayaramani R, et al. Virology, pathogenesis, diagnosis and in-line treatment of COVID-19. Eur J Pharm. 2020;883:173375. doi: 10.1016/j.ejphar.2020.173375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Khare P, Sahu U, Pandey SC, Samant M. Current approaches for target-specific drug discovery using natural compounds against SARS-CoV-2 infection. Virus Res. 2020;290:198169. doi: 10.1016/j.virusres.2020.198169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tleyjeh IM, Kashour Z, Damlaj M, Riaz M, Tlayjeh H, Altannir M, et al. Efficacy and safety of tocilizumab in COVID-19 patients: a living systematic review and meta-analysis. Clin Microbiol Infect. 2021;27:215–27. doi: 10.1016/j.cmi.2020.10.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fuzimoto AD, Isidoro C. The antiviral and coronavirus-host protein pathways inhibiting properties of herbs and natural compounds - Additional weapons in the fight against the COVID-19 pandemic? J Tradit Complement Med. 2020;10:405–19. doi: 10.1016/j.jtcme.2020.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Du HZ, Hou XY, Miao YH, Huang BS, Liu DH. Traditional Chinese Medicine: an effective treatment for 2019 novel coronavirus pneumonia (NCP) Chin J Nat Med. 2020;18:206–10. doi: 10.1016/s1875-5364(20)30022-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ren JL, Zhang AH, Wang XJ. Traditional Chinese medicine for COVID-19 treatment. Pharm Res. 2020;155:104743. doi: 10.1016/j.phrs.2020.104743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liu CX. Pay attention to situation of SARS-CoV-2 and TCM advantages in treatment of novel coronavirus infection. Chin Herb Med. 2020;12:97–103. doi: 10.1016/j.chmed.2020.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhang L, Yu J, Zhou Y, Shen M, Sun L. Becoming a Faithful Defender: Traditional Chinese Medicine against Coronavirus Disease 2019 (COVID-19) Am J Chin Med. 2020;48:763–77. doi: 10.1142/s0192415x2050038x. [DOI] [PubMed] [Google Scholar]
- 19.Zhong LLD, Lam WC, Yang W, Chan KW, Sze SCW, Miao J, et al. Potential Targets for Treatment of Coronavirus Disease 2019 (COVID-19): A Review of Qing-Fei-Pai-Du-Tang and Its Major Herbs. Am J Chin Med. 2020;48:1051–71. doi: 10.1142/s0192415x20500512. [DOI] [PubMed] [Google Scholar]
- 20.Huang ST, Lai HC, Lin YC, Huang WT, Hung HH, Ou SC, et al. Principles and treatment strategies for the use of Chinese herbal medicine in patients at different stages of coronavirus infection. Am J Cancer Res. 2020;10:2010–31. [PMC free article] [PubMed] [Google Scholar]
- 21.Pang W, Liu Z, Li N, Li Y, Yang F, Pang B, et al. Chinese medical drugs for coronavirus disease 2019: a systematic review and meta-analysis. Integr Med Res. 2020;9:100477. doi: 10.1016/j.imr.2020.100477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fan TT, Cheng BL, Fang XM, Chen YC, Su F. Application of Chinese Medicine in the management of critical conditions: a review on Sepsis. Am J Chin Med. 2020;48:1315–30. doi: 10.1142/s0192415x20500640. [DOI] [PubMed] [Google Scholar]
- 23.Zhang XR, Li TN, Ren YY, Zeng YJ, Lv HY, Wang J, et al. The important role of volatile components from a Traditional Chinese Medicine Dayuan-Yin Against the COVID-19 Pandemic. Front Pharm. 2020;11:583651. doi: 10.3389/fphar.2020.583651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhang Y, Li Y, Wang X, Qu R, Li J, Li T, et al. Herbal plants coordinate COVID-19 in multiple dimensions - an insight analysis for clinically applied remedies. Int J Med Sci. 2020;17:3125–45. doi: 10.7150/ijms.50260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yang Y, Islam MS, Wang J, Li Y, Chen X. Traditional Chinese Medicine in the treatment of patients infected with 2019-New Coronavirus (SARS-CoV-2): a review and perspective. Int J Biol Sci. 2020;16:1708–17. doi: 10.7150/ijbs.45538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.H L. Shining a Light on COVID-19’s Long Shadow, Director’s Page. 2021. https://www.nccih.nih.gov/about/offices/od/director/past-messages/shining-a-light-on-covid-19s-long-shadow?nav=govd. Accessed February 24 2021.
- 27.Luo H, Tang QL, Shang YX, Liang SB, Yang M, Robinson N, et al. Can Chinese Medicine be used for prevention of Corona Virus Disease 2019 (COVID-19)? a review of Historical Classics, Research Evidence and Current Prevention Programs. Chin J Integr Med. 2020;26:243–50. doi: 10.1007/s11655-020-3192-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Huang F, Li Y, Leung EL, Liu X, Liu K, Wang Q, et al. A review of therapeutic agents and Chinese herbal medicines against SARS-COV-2 (COVID-19) Pharm Res. 2020;158:104929. doi: 10.1016/j.phrs.2020.104929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ang L, Lee HW, Choi JY, Zhang J, Soo Lee M. Herbal medicine and pattern identification for treating COVID-19: a rapid review of guidelines. Integr Med Res. 2020;9:100407. doi: 10.1016/j.imr.2020.100407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhang D, Zhang B, Lv JT, Sa RN, Zhang XM, Lin ZJ. The clinical benefits of Chinese patent medicines against COVID-19 based on current evidence. Pharm Res. 2020;157:104882. doi: 10.1016/j.phrs.2020.104882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.López-Alcalde J, Yan Y, Witt CM, Barth J. Current State of Research about Chinese Herbal Medicines (CHM) for the treatment of Coronavirus Disease 2019 (COVID-19): A Scoping Review. J Alter Complement Med. 2020;26:557–70. doi: 10.1089/acm.2020.0189. [DOI] [PubMed] [Google Scholar]
- 32.Zhao ZH, Zhou Y, Li WH, Huang QS, Tang ZH, Li H. Analysis of Traditional Chinese Medicine Diagnosis and Treatment Strategies for COVID-19 Based on "The Diagnosis and Treatment Program for Coronavirus Disease-2019" from Chinese Authority. Am J Chin Med. 2020;48:1035–49. doi: 10.1142/s0192415x20500500. [DOI] [PubMed] [Google Scholar]
- 33.Liu R, Zhang Y, Nicholas S, Leng A, Maitland E, Wang J. COVID-19 Vaccination Willingness among Chinese Adults under the Free Vaccination Policy. Vaccines (Basel). 2021;9. 10.3390/vaccines9030292. [DOI] [PMC free article] [PubMed]
- 34.Kang JW, Kim JH, Song K, Kim SH, Yoon JH, Kim KS. Kaempferol and quercetin, components of Ginkgo biloba extract (EGb 761), induce caspase-3-dependent apoptosis in oral cavity cancer cells. Phytother Res. 2010;24:S77–82. doi: 10.1002/ptr.2913. [DOI] [PubMed] [Google Scholar]
- 35.Okamoto K, Sako Y. Recent advances in FRET for the study of protein interactions and dynamics. Curr Opin Struct Biol. 2017;46:16–23. doi: 10.1016/j.sbi.2017.03.010. [DOI] [PubMed] [Google Scholar]
- 36.Yang H, Xie W, Xue X, Yang K, Ma J, Liang W, et al. Design of wide-spectrum inhibitors targeting coronavirus main proteases. PLoS Biol. 2005;3:e324. doi: 10.1371/journal.pbio.0030324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Andi B KD, Kreitler DF, Soares AS, Shi W, Jakoncic J, Fuchs MR, et al. Virus NSP3/NSP4A inhibitors as promising lead compounds for the design of new covalent inhibitors for SARS-CoV-2 3CLpro/Mpro Protease.
- 38.Ratia K, Saikatendu KS, Santarsiero BD, Barretto N, Baker SC, Stevens RC, et al. Severe acute respiratory syndrome coronavirus papain-like protease: structure of a viral deubiquitinating enzyme. Proc Natl Acad Sci USA. 2006;103:5717–22. doi: 10.1073/pnas.0510851103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Osipiuk J, Jedrzejczak R, Tesar C, Endres M, Stols L, Babnigg G, et al. The crystal structure of papain-like protease of SARS CoV-2. 10.2210/pdb6W9C/pdb.
- 40.Kirchdoerfer RN, Ward AB. Structure of the SARS-CoV nsp12 polymerase bound to nsp7 and nsp8 co-factors. Nat Commun. 2019;10:2342. doi: 10.1038/s41467-019-10280-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yin W, Mao C, Luan X, Shen DD, Shen Q, Su H, et al. Structural basis for inhibition of the RNA-dependent RNA polymerase from SARS-CoV-2 by remdesivir. Science. 2020;368:1499–504. doi: 10.1126/science.abc1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yu IM, Oldham ML, Zhang J, Chen J. Crystal structure of the severe acute respiratory syndrome (SARS) coronavirus nucleocapsid protein dimerization domain reveals evolutionary linkage between corona- and arteriviridae. J Biol Chem. 2006;281:17134–9. doi: 10.1074/jbc.M602107200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chang C, Michalska K, Jedrzejczak R, Maltseva N, Endres M, Godziks A, et al. Crystal structure of RNA binding domain of nucleocapsid phosphoprotein from SARS coronavirus 2. 10.2210/pdb6VYO/pdb.
- 44.Walls AC, Tortorici MA, Bosch BJ, Frenz B, Rottier PJM, DiMaio F, et al. Cryo-electron microscopy structure of a coronavirus spike glycoprotein trimer. Nature. 2016;531:114–7. doi: 10.1038/nature16988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, Veesler D. Structure, function, and antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell. 2020;181:281–92.e6. doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Clark HF, Gurney AL, Abaya E, Baker K, Baldwin D, Brush J, et al. The secreted protein discovery initiative (SPDI), a large-scale effort to identify novel human secreted and transmembrane proteins: a bioinformatics assessment. Genome Res. 2003;13:2265–70. doi: 10.1101/gr.1293003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hall DC, Jr., Ji HF. A search for medications to treat COVID-19 via in silico molecular docking models of the SARS-CoV-2 spike glycoprotein and 3CL protease. Travel Med Infect Dis. 2020;35:101646. doi: 10.1016/j.tmaid.2020.101646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Boukhatem MN, Setzer WN. Aromatic Herbs, Medicinal Plant-Derived Essential Oils, and Phytochemical Extracts as Potential Therapies for Coronaviruses: Future Perspectives. Plants (Basel). 2020;9. 10.3390/plants9060800. [DOI] [PMC free article] [PubMed]
- 49.Jalali A, Dabaghian F, Akbrialiabad H, Foroughinia F, Zarshenas MM. A pharmacology-based comprehensive review on medicinal plants and phytoactive constituents possibly effective in the management of COVID-19. Phytother Res. 2021;35:1925–38. doi: 10.1002/ptr.6936. [DOI] [PubMed] [Google Scholar]
- 50.Akalın E, Ekici M, Alan Z, Özbir Elevli E, Yaman Bucak A, Aobuliaikemu N, et al. Traditional Chinese medicine practices used in COVID-19 (Sars-cov 2/Coronavirus-19) treatment in clinic and their effects on the cardiovascular system. Turk Kardiyol Dern Ars. 2020;48:410–24. doi: 10.5543/tkda.2020.03374. [DOI] [PubMed] [Google Scholar]
- 51.Zhou J, Huang J. Current findings regarding natural components with potential Anti-2019-nCoV Activity. Front Cell Dev Biol. 2020;8:589. doi: 10.3389/fcell.2020.00589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Oladele JO, Ajayi EI, Oyeleke OM, Oladele OT, Olowookere BD, Adeniyi BM, et al. A systematic review on COVID-19 pandemic with special emphasis on curative potentials of Nigeria based medicinal plants. Heliyon. 2020;6:e04897. doi: 10.1016/j.heliyon.2020.e04897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Adhikari B, Marasini BP, Rayamajhee B, Bhattarai BR, Lamichhane G, Khadayat K, et al. Potential roles of medicinal plants for the treatment of viral diseases focusing on COVID-19: a review. Phytother Res. 2021;35:1298–312. doi: 10.1002/ptr.6893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hoever G, Baltina L, Michaelis M, Kondratenko R, Baltina L, Tolstikov GA, et al. Antiviral activity of glycyrrhizic acid derivatives against SARS-coronavirus. J Med Chem. 2005;48:1256–9. doi: 10.1021/jm0493008. [DOI] [PubMed] [Google Scholar]
- 55.Vardhan S, Sahoo SK. In silico ADMET and molecular docking study on searching potential inhibitors from limonoids and triterpenoids for COVID-19. Comput Biol Med. 2020;124:103936. doi: 10.1016/j.compbiomed.2020.103936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Muhseen ZT, Hameed AR, Al-Hasani HMH, Ahmad S, Li G. Computational Determination of Potential Multiprotein Targeting Natural Compounds for Rational Drug Design Against SARS-COV-2. Molecules. 2021;26. 10.3390/molecules26030674. [DOI] [PMC free article] [PubMed]
- 57.Khalil A, Tazeddinova D. The upshot of Polyphenolic compounds on immunity amid COVID-19 pandemic and other emerging communicable diseases: An appraisal. Nat Prod Bioprospect. 2020;10:411–29. doi: 10.1007/s13659-020-00271-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Mani JS, Johnson JB, Steel JC, Broszczak DA, Neilsen PM, Walsh KB, et al. Natural product-derived phytochemicals as potential agents against coronaviruses: A review. Virus Res. 2020;284:197989. doi: 10.1016/j.virusres.2020.197989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Solnier J, Fladerer JP. Flavonoids: a complementary approach to conventional therapy of COVID-19? Phytochem Rev. 2020:1–23. 10.1007/s11101-020-09720-6. [DOI] [PMC free article] [PubMed]
- 60.Limanaqi F, Busceti CL, Biagioni F, Lazzeri G, Forte M, Schiavon S, et al. Cell clearing systems as targets of polyphenols in viral infections: potential implications for COVID-19 Pathogenesis. Antioxidants (Basel). 2020;9. 10.3390/antiox9111105. [DOI] [PMC free article] [PubMed]
- 61.Zhi HJ, Zhu HY, Zhang YY, Lu Y, Li H, Chen DF. In vivo effect of quantified flavonoids-enriched extract of Scutellaria baicalensis root on acute lung injury induced by influenza A virus. Phytomedicine. 2019;57:105–16. doi: 10.1016/j.phymed.2018.12.009. [DOI] [PubMed] [Google Scholar]
- 62.Huang YF, Bai C, He F, Xie Y, Zhou H. Review on the potential action mechanisms of Chinese medicines in treating Coronavirus Disease 2019 (COVID-19) Pharm Res. 2020;158:104939. doi: 10.1016/j.phrs.2020.104939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Goyal RK, Majeed J, Tonk R, Dhobi M, Patel B, Sharma K, et al. Current targets and drug candidates for prevention and treatment of SARS-CoV-2 (COVID-19) infection. Rev Cardiovasc Med. 2020;21:365–84. doi: 10.31083/j.rcm.2020.03.118. [DOI] [PubMed] [Google Scholar]
- 64.Su HX, Yao S, Zhao WF, Li MJ, Liu J, Shang WJ, et al. Anti-SARS-CoV-2 activities in vitro of Shuanghuanglian preparations and bioactive ingredients. Acta Pharm Sin. 2020;41:1167–77. doi: 10.1038/s41401-020-0483-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Consortium TCM, Achdout H, Aimon A, Bar-David E, Barr H, Ben-Shmuel A, et al. COVID Moonshot: Open Science Discovery of SARS-CoV-2 Main Protease Inhibitors by Combining Crowdsourcing, High-Throughput Experiments, Computational Simulations, and Machine Learning. bioRxiv. 2020:2020.10.29.339317. 10.1101/2020.10.29.339317.
- 66.Tallei TE, Tumilaar SG, Niode NJ, Fatimawali, Kepel BJ, Idroes R, et al. Potential of plant bioactive compounds as SARS-CoV-2 Main Protease (M(pro)) and Spike (S) Glycoprotein Inhibitors: A Molecular Docking Study. Sci (Cairo) 2020;2020:6307457.. doi: 10.1155/2020/6307457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lowe H, Steele B, Bryant J, Toyang N, Ngwa W. Non-cannabinoid metabolites of cannabis sativa L. with Therapeutic Potential. Plants (Basel). 2021;10. 10.3390/plants10020400. [DOI] [PMC free article] [PubMed]
- 68.Elsohly MA, Slade D. Chemical constituents of marijuana: the complex mixture of natural cannabinoids. Life Sci. 2005;78:539–48. doi: 10.1016/j.lfs.2005.09.011. [DOI] [PubMed] [Google Scholar]
- 69.Pollastro F, Minassi A, Fresu LG. Cannabis phenolics and their bioactivities. Curr Med Chem. 2018;25:1160–85. doi: 10.2174/0929867324666170810164636. [DOI] [PubMed] [Google Scholar]
- 70.Sexton M. Cannabis in the Time of Coronavirus Disease 2019: The Yin and Yang of the Endocannabinoid System in Immunocompetence. J Alter Complement Med. 2020;26:444–8. doi: 10.1089/acm.2020.0144. [DOI] [PubMed] [Google Scholar]
- 71.Sainz-Cort A, Heeroma JH. The interaction between the endocannabinoid system and the renin angiotensin system and its potential implication for COVID-19 infection. J Cannabis Res. 2020;2:23. doi: 10.1186/s42238-020-00030-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Anil SM, Shalev N, Vinayaka AC, Nadarajan S, Namdar D, Belausov E, et al. Cannabis compounds exhibit anti-inflammatory activity in vitro in COVID-19-related inflammation in lung epithelial cells and pro-inflammatory activity in macrophages. Sci Rep. 2021;11:1462. doi: 10.1038/s41598-021-81049-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Stasiłowicz A, Tomala A, Podolak I, Cielecka-Piontek J. Cannabis sativa L. as a Natural Drug Meeting the Criteria of a Multitarget Approach to Treatment. Int J Mol Sci. 2021;22. 10.3390/ijms22020778. [DOI] [PMC free article] [PubMed]
- 74.Khodadadi H, Salles ÉL, Shin E, Jarrahi A, Costigliola V, Kumar P, et al. A potential role for cannabichromene in modulating TRP channels during acute respiratory distress syndrome. J Cannabis Res. 2021;3:45. doi: 10.1186/s42238-021-00101-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Malinowska B, Baranowska-Kuczko M, Kicman A, Schlicker E. Opportunities, Challenges and Pitfalls of Using Cannabidiol as an Adjuvant Drug in COVID-19. Int J Mol Sci. 2021;22. 10.3390/ijms22041986. [DOI] [PMC free article] [PubMed]
- 76.Khodadadi H, Salles ÉL, Jarrahi A, Chibane F, Costigliola V, Yu JC, et al. Cannabidiol modulates cytokine storm in acute respiratory distress syndrome induced by simulated viral infection using synthetic RNA. Cannabis Cannabinoid Res. 2020;5:197–201. doi: 10.1089/can.2020.0043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Paland N, Pechkovsky A, Aswad M, Hamza H, Popov T, Shahar E, et al. The Immunopathology of COVID-19 and the Cannabis Paradigm. Front Immunol. 2021;12:631233. doi: 10.3389/fimmu.2021.631233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Nguyen LC, Yang D, Nicolaescu V, Best TJ, Ohtsuki T, Chen SN, et al. Cannabidiol inhibits SARS-CoV-2 replication and promotes the host innate immune response. bioRxiv. 2021. 10.1101/2021.03.10.432967.
- 79.Suryavanshi SV, Kovalchuk I, Kovalchuk O. Cannabinoids as key regulators of inflammasome signaling: a current perspective. Front Immunol. 2020;11:613613. doi: 10.3389/fimmu.2020.613613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Sarkar I, Sen G, Bhattacharya M, Bhattacharyya S, Sen A. In silico inquest reveals the efficacy of Cannabis in the treatment of post-Covid-19 related neurodegeneration. J Biomol Struct Dyn. 2021:1–10. 10.1080/07391102.2021.1905556. [DOI] [PubMed]
- 81.Hill KP. Cannabinoids and the Coronavirus. Cannabis Cannabinoid Res. 2020;5:118–20. doi: 10.1089/can.2020.0035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Wang B, Kovalchuk A, Li D, Rodriguez-Juarez R, Ilnytskyy Y, Kovalchuk I, et al. In search of preventive strategies: novel high-CBD Cannabis sativa extracts modulate ACE2 expression in COVID-19 gateway tissues. Aging (Albany NY) 2020;12:22425–44. doi: 10.18632/aging.202225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Esposito G, Pesce M, Seguella L, Sanseverino W, Lu J, Corpetti C, et al. The potential of cannabidiol in the COVID-19 pandemic. Br J Pharm. 2020;177:4967–70. doi: 10.1111/bph.15157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Jha NK, Sharma C, Hashiesh HM, Arunachalam S, Meeran MN, Javed H, et al. β-Caryophyllene, a natural dietary CB2 receptor selective cannabinoid can be a candidate to target the trinity of infection, immunity, and inflammation in COVID-19. Front Pharm. 2021;12:590201. doi: 10.3389/fphar.2021.590201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Ngwa W, Kumar R, Thompson D, Lyerly W, Moore R, Reid TE, et al. Potential of Flavonoid-Inspired Phytomedicines against COVID-19. Molecules. 2020;25. 10.3390/molecules25112707. [DOI] [PMC free article] [PubMed]
- 86.Shover CL, Humphreys K. Debunking Cannabidiol as a Treatment for COVID-19: Time for the FDA to Adopt a Focused Deterrence Model? Cureus. 2020;12:e8671. doi: 10.7759/cureus.8671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Onaivi ES, Sharma V. Cannabis for COVID-19: can cannabinoids quell the cytokine storm? Future Sci OA. 2020;6:Fso625. doi: 10.2144/fsoa-2020-0124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Dabeek WM, Marra MV. Dietary Quercetin and Kaempferol: Bioavailability and Potential Cardiovascular-Related Bioactivity in Humans. Nutrients. 2019;11. 10.3390/nu11102288. [DOI] [PMC free article] [PubMed]
- 89.Gao Y, Yan L, Huang Y, Liu F, Zhao Y, Cao L, et al. Structure of the RNA-dependent RNA polymerase from COVID-19 virus. Science. 2020;368:779–82. doi: 10.1126/science.abb7498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Yan R, Zhang Y, Li Y, Xia L, Guo Y, Zhou Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science. 2020;367:1444–8. doi: 10.1126/science.abb2762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Li F, Li W, Farzan M, Harrison SC. Structure of SARS coronavirus spike receptor-binding domain complexed with receptor. Science. 2005;309:1864–8. doi: 10.1126/science.1116480. [DOI] [PubMed] [Google Scholar]