Highlights
-
•
Cervical dystonia has significant impact on quality of life of those affected.
-
•
Dilution options allow for treatment flexibility and individualized patient care.
-
•
AboBoNT-A is efficacious with similar safety and tolerability at either dilution.
Keywords: AbobotulinumtoxinA, Cervical dystonia, Dosing flexibility, Dilution, Treatment
Abstract
Introduction
Cervical dystonia (CD) is a neurologic movement disorder with potentially disabling effects and significant impact on quality of life of those affected. AbobotulinumtoxinA (aboBoNT-A) was initially approved for a dilution of 500 U/1 mL and subsequently for a dilution of 500 U/2 mL, providing flexibility for clinicians to treat CD. Here, we explore the safety and efficacy of the 500 U/2 mL dilution versus 500 U/1 mL dilution of aboBoNT-A in a retrospective analysis based on published clinical trial data.
Methods
The safety and efficacy of aboBoNT-A in patients with CD was evaluated in three multicenter, double-blind, randomized, placebo-controlled trials and open-label extensions. Trials 1 (NCT00257660) and 2 (NCT00288509) evaluated the 500 U/1 mL dilution in 80 and 116 patients, respectively; Trial 3 (NCT01753310) evaluated the 500 U/2 mL dilution in 125 patients.
Results
Comparison of the adjusted mean difference in TWSTRS total scores at Week 4 from baseline for aboBoNT-A in Trial 1 (−6.0; 95% CI, −10.8, −1.3), Trial 2 (−8.8; 95% CI, −12.9, −4.7), and Trial 3 (−8.7; 95% CI, −13.2, −4.2) showed similar, significant improvements. Dysphagia and muscle weakness patterns were comparable across the three trials, indicating that an increased dilution of aboBoNT-A does not result in an increased risk of diffusion-related adverse events.
Conclusion
The results of these trials show that aboBoNT-A is similarly efficacious using either dilution, with similar safety and tolerability across trials. Having the 500 U/1 mL and 500 U/2 mL dilution volumes available provides further flexibility in administration, benefiting patient care.
1. Introduction
Cervical dystonia (CD) is a neurologic movement disorder characterized by sustained or repetitive involuntary contractions of the neck muscles, leading to abnormal postures [1]. CD is relatively rare, with prevalence estimates ranging from 20 to 4100 cases per million in the population worldwide [2]. Patients with CD experience significantly more pain when compared with the general population and other neurological disorders, as reflected by their lower (worse) bodily pain scores [3]. The condition also has significant impact on quality of life, with considerably lower (worse) scores on all domains of the SF-36 Health Survey of those affected [3].
The gold standard for treating CD is botulinum neurotoxin (BoNT), which is established as a safe and effective therapy based on clinical trials with all four FDA-approved toxins [4]. AbobotulinumtoxinA (aboBoNT-A) is one of three commercially available BoNT-A preparations for CD (the other two being onabotulinumtoxinA and incobotulinumtoxinA) and is recommended as a Level A treatment option for CD by the American Academy of Neurology [4]. Based on clinical trial data [5], [6], [7], aboBoNT-A was approved in the United States initially at a dilution of 500 U/1 mL and subsequently at a dilution of 500 U/2 mL and 300 U/0.6 mL [8]. Evaluation of the 500 U/2 mL dilution was prompted by feedback obtained from scientific and community experts based on clinical practice.
The 1 mL and 2 mL dilutions of aboBoNT-A contain the same total amount of neurotoxin (500 U). Administered doses are individualized per patient and are based on muscle type, size, and activity [1]. Availability of the larger, 2 mL dilution volume can provide flexibility with respect to the number of muscles and injection sites achievable per session and facilitates increased accuracy of dosing at each injection site. With any BoNT preparation, there is a risk of localized diffusion and distant spread to non-target muscles, which may induce unwanted effects, including an increase in diffusion-related adverse events (AEs) such as dysphagia [9]. Concerns have also been raised whether the risk of local diffusion increases with greater injected volume [9], [10]. In this manuscript, we explore the safety and efficacy of the 500 U/2 mL dilution versus the 500 U/1 mL dilution of aboBoNT-A, based on published clinical trial data.
2. Methods
2.1. Study designs and treatments
This is a post hoc analysis of the efficacy and safety data of aboBoNT-A in patients with CD evaluated in three multicenter, double-blind, randomized, placebo-controlled trials and their open-label extensions, the results of which have been reported previously [5], [6], [7], [11], [12]. Two trials assessed aboBoNT-A at the 500 U/1 mL dilution, and one trial evaluated a dilution of 500 U/2 mL.
In Trial 1 (NCT00257660), conducted by Truong et al. (2005), patients with CD from study centers in the United States were randomized to aboBoNT-A 500 U/1 mL or placebo [5]. Participants were monitored for 4 to 20 weeks, until they needed further treatment, and those who completed the trial were eligible to enroll in an open-label extension study and receive up to three additional aboBoNT-A treatments. In Trial 2 (NCT00257660 and NCT00288509), by Truong et al. (2010), patients with CD from study sites in the United States and Russia (16 and 4 centers, respectively) were randomized to aboBoNT-A 500 U/1 mL or placebo. Those who completed the 12-week double-blind phase were eligible to enter the open-label extension phase and receive up to four additional aboBoNT-A treatments [6]. For both 500 U/1 mL trials, injection doses during the open-label extension studies could be titrated by the investigator in 250 U increments to a minimum of 250 U or a maximum of 1000 U, based on efficacy and safety [5], [6], [11].
In Trial 3 (NCT01753310), by Lew et al. (2018), patients with CD from study centers in the United States were randomized with a 2:1 ratio to aboBoNT-A 500 U/2 mL or placebo, respectively, with an open-label extension option for those who completed the 12-week study [7]. Participants received 500 U of aboBoNT-A if they were toxin-naïve at baseline, or 250 to 500 U of aboBoNT–A based on previous onabotulinumtoxinA dose, if non-naïve. In the extension study, patients received up to three additional treatment cycles, occurring at intervals of 12 to 16 weeks, with dose adjustments in 250 U increments limited to a total dose between 250 U and 1000 U [12].
2.2. Patients
Eligible patients were aged 18 years or older and had primary idiopathic CD [5], [6], [7]. In Trials 1 and 2 (500 U/1 mL), patients had to have a baseline Toronto Western Spasmodic Torticollis Rating Scale (TWSTRS) total score ≥ 30 and a TWSTRS severity subscale score ≥ 15; in Trial 3 (500 U/2 mL), patients had to have a TWSTRS total score ≥ 20 and a TWSTRS severity subscale score > 10 [5], [6], [7].
2.3. Primary efficacy endpoint
In all three trials, the primary efficacy endpoint during the double-blind treatment phase was the change from baseline in TWSTRS total score at Week 4 compared with placebo [5], [6], [7]. TWSTRS is a composite measure to evaluate torticollis severity, disability, and pain in patients with CD. TWSTRS total score (maximum score of either 85 [5], [6] or 87 [7]) is the sum of the three subscale scores. A decrease in total or subscale score indicates an improvement in patients’ CD.
2.4. Safety
Safety assessments, including collection of treatment-emergent adverse event (AE) details, were conducted in all three studies. In Trial 1, both spontaneously reported AEs and solicited AEs of interest (e.g. dysphagia) were included, whereas in Trial 2 and Trial 3 only spontaneously reported AEs were collected.
2.5. Statistical analyses
Efficacy analyses included all patients randomized to treatment. Statistical testing for the primary efficacy endpoint was two-sided and performed at a 5% significance level, using analysis of covariance (ANCOVA) [5], [6], [7]. In Trial 1, the analysis was adjusted for center, treatment history (i.e. previous BoNT treatment vs. no previous BoNT treatment), and baseline score [5]. The analysis in Trial 2 was controlled for center, treatment history, baseline score, and treatment group [6]. Trial 3 used an ANCOVA with baseline TWSTRS total score as covariate and stratified by pre-treatment status (onabotulinumtoxinA naïve vs. non-naïve) [7]. Results from the open-label extension studies are presented descriptively [11], [12]. Safety variables are summarized here by descriptive statistics.
3. Results
3.1. Patients
In total, 330 patients participated in the three trials; 181 participants were treated with aboBoNT–A and 149 received placebo (Table 1). The mean age was 53 years in Trial 1, 52 years in Trial 2, and 57 years in Trial 3. The proportion of female patients across the three trials was 63%, 65%, and 65%, respectively (Table 1) [5], [6], [7]. In Trial 1, the mean TWSTRS baseline scores were 43.83 (7.97) in the aboBoNT-A group and 45.81 (8.78) in the placebo group. In Trial 2, the open-label extension of Trial 1, the baseline TWSTRS total score was 42.48 (10.23) for the aboBoNT-A group. The mean baseline TWSTRS scores in Trial 3 were 42.5 (10.40) in the aboBoNT-A group and 42.4 (10.63) in the placebo group.
Table 1.
Baseline demographics.
| Trial 1 (500 U/1 mL) N = 80 Truong et al. 2005 |
Trial 2 (500 U/1 mL) N = 116 Truong et al. 2010 |
Trial 3 (500 U/2 mL) N = 134 Lew et al. 2018 |
||||
|---|---|---|---|---|---|---|
| AboBoNT-A | Placebo | AboBoNT-A | Placebo | AboBoNT-A | Placebo | |
| n | 37 | 43 | 55 | 61 | 89 | 45 |
| Age, mean (SD), years | 53.4 (11.6) | 53.6 (12.1) | 51.9 (13.4) | 53.9 (12.5) | 57.3 (11.1) | 56.5 (11.7) |
| Female sex, n (%) | 23 (62) | 27 (63) | 37 (67) | 38 (62) | 59 (66) | 28 (62) |
aboBoNT-A, abobotulinumtoxinA; SD, standard deviation; U, units.
3.2. Efficacy
3.2.1. TWSTRS total
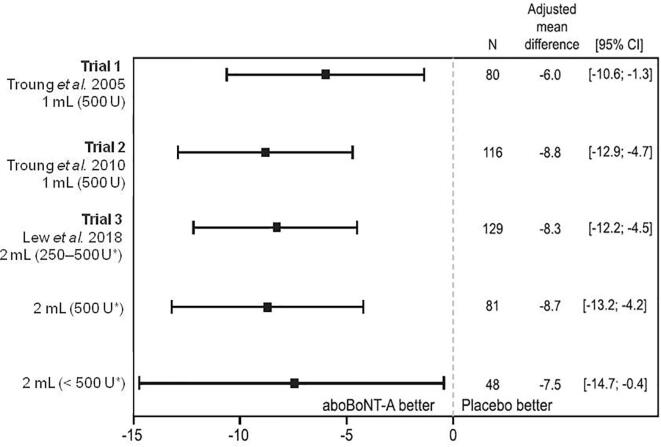
All three trials met their primary endpoint, with patients receiving aboBoNT-A experiencing significantly greater improvement from baseline in TWSTRS total score at Week 4 compared with placebo. TWSTRS total score changes in the aboBoNT-A groups were similar in the two trials using the 500 U/1 mL dilution and in the trial using the 500 U/2 mL dilution (Fig. 1).
Fig. 1.
Adjusted mean difference in TWSTRS total score at Week 4. Reprinted from International Journal of Neuroscience, Vol. 128, Lew MF, Brashear A, Dashtipour K, Isaacson S, Hauser RA, Maisonobe P, Snyder D, Ondo W. Pages 619–626, 2018, with permission from Taylor & Francis Ltd (https://www.tandfonline.com/). *Trial 3 results are presented in summary (250–500 U) and by subgroup (500 U vs. < 500 U). aboBoNT-A, abobotulinumtoxinA; CI, confidence interval; TWSTRS, Toronto Western Spasmodic Torticollis Rating Scale; U, units.
In Trial 1 (500 U/1 mL), the mean change in TWSTRS total score at Week 4 was −9.9 in the aboBoNT-A group compared with −3.8 in the placebo group (adjusted mean difference: −6.0; P ≤ 0.013); and in Trial 2 (500 U/1 mL) it was −15.6 in the aboBoNT-A group compared with −6.7 in the placebo group (adjusted mean difference: −8.8; P < 0.001) [5], [6]. In the open-label extension study of these two trials, all treatment cycles resulted in improvements in TWSTRS total scores compared with cycle baselines. The mean change in TWSTRS total scores from baseline in the open-label extension study of Trial 1 was −12.2 ± 10.3 to treatment cycle 3, and −11.3 ± 11.5 in the open-label extension of Trial 2 to treatment cycle 4 [11].
In Trial 3 (500 U/2 mL), the mean change in TWSTRS total score at Week 4 was −10.8 in the aboBoNT-A group compared with −2.5 in the placebo group, with an adjusted mean difference of −8.3 (P < 0.001) [7]. The mean baseline TWSTRS scores in the subgroup receiving a total of 500 U (n = 81) and <500 U (n = 48) of aboBoNT-A were 43.2 (SD 9.9) and 41.5 (SD 11.2), respectively. The adjusted mean difference in TWSTRS total score for aboBoNT-A versus placebo was similar in the subgroup receiving a total of 500 U of aboBoNT-A and in the subgroup receiving less than 500 U, with overlapping 95% confidence intervals (Fig. 1). During the open-label extension, mean TWSTRS total score change from baseline to Cycle 3, Week 12 was −11.7 [12]. For each subsequent cycle, TWSTRS total score significantly improved from Day 1 to Week 4 and did not return to baseline scores at the end of the injection cycle [12].
3.2.2. TWSTRS subscales
Consistent with the TWSTRS total scores, changes in TWSTRS subscale scores were also similar in the two trials using aboBoNT-A 500 U/1 mL compared with the trial using the 500 U/2 mL dilution. In Trial 1 (500 U/1 mL), the adjusted mean differences from baseline at Week 4 for the severity, disability, and pain subscales were −2.5, −1.9, and −1.6, respectively; in Trial 2 these values were −3.8, −2.4, and −2.3, respectively [5], [6]. In Trial 3, the adjusted mean differences from baseline at Week 4 were −2.0, −0.3, and −0.2 for severity, disability, and pain, respectively [13].
TWSTRS subscale score results were not reported for the open-label extension of Trial 1 [11]. During the open-label extension of Trial 3, improvement patterns in subscale scores were consistent with those for TWSTRS total score [12].
3.3. Safety
Table 2 lists the AEs occurring with aboBoNT-A during 12-week, double-blind treatment in the three trials [5], [6], [7]. The proportion of patients with any AEs in Trial 1 was considerably higher than in Trials 2 or 3, both in the aboBoNT-A group and in the placebo group. This was most likely because both spontaneously reported events and solicited events of interest (e.g. dysphagia) were included in Trial 1, whereas the other two trials included spontaneously reported AEs only [11].
Table 2.
AEs occurring with aboBoNT-A during 12-week double-blind treatment.
| Patients, n (%) | Trial 1 (500 U/1 mL)1 N = 80 Truong et al. 2005 |
Trial 2 (500 U/1 mL)2 N = 116 Truong et al. 2010 |
Trial 3 (500 U/2 mL)3 N = 134 Lew et al. 2018 |
|||
|---|---|---|---|---|---|---|
| AboBoNT-A (n = 37) | Placebo (n = 43) | AboBoNT-A (n = 55) | Placebo (n = 61) | AboBoNT-A (n = 884) | Placebo (n = 45) | |
| All TEAEs | 34 (92%) | 34 (79%) | 26 (47%) | 27 (44%) | 36 (41%) | 10 (22%) |
| Dysphagia | 6 (16%) | 4 (9%) | 5 (9%) | 0 (0%) | 8 (9%) | 0 (0%) |
| Muscular weakness | 4 (11%) | 0 (0%) | — | — | 8 (9%) | 0 (0%) |
| Neck/shoulder pain5 | 14 (38%) | 13 (30%) | 3 (5%) | 3 (5%) | 7 (8%) | 0 (0%) |
| Headache | 9 (24%) | 10 (23%) | 2 (4%) | 2 (3%) | 5 (6%) | 0 (0%) |
| Sinusitis | 3 (8%) | 1 (2%) | — | — | 3 (3%) | 0 (0%) |
| Bronchitis | 3 (8%) | 1 (2%) | — | — | 2 (2%) | 0 (0%) |
| Fatigue/tiredness | 13 (35%) | 13 (30%) | — | — | 2 (2%) | 0 (0%) |
| Blurred vision | 5 (14%) | 0 (0%) | — | — | 2 (2%) | 0 (0%) |
| Burning sensation | — | — | — | — | 2 (2%) | 0 (0%) |
| Diarrhea | — | — | — | — | 2 (2%) | 0 (0%) |
| Depression | — | — | — | — | 2 (2%) | 1 (2%) |
| Injection site pain | 14 (38%) | 10 (23%) | 3 (5%) | 2 (3%) | — | — |
| Dry mouth | 8 (22%) | 8 (19%) | — | — | — | — |
| Neck muscle weakness | 6 (16%) | 5 (12%) | — | — | — | — |
| Neck rigidity | 5 (14%) | 4 (9%) | — | — | — | — |
| Voice alteration | 4 (11%) | 4 (9%) | — | — | — | — |
| Dyspnea | 4 (11%) | 1 (2%) | — | — | — | — |
| Insomnia | 4 (11%) | 1 (2%) | — | — | — | — |
| Viral infection | 4 (11%) | 2 (5%) | — | — | — | — |
| Back pain | 3 (8%) | 3 (7%) | — | — | — | — |
| Dizziness | 3 (8%) | 2 (5%) | — | — | — | — |
| Rhinitis | 3 (8%) | 1 (2%) | — | — | — | |
| URTI | — | 2 (4%) | 2 (3%) | — | — | |
aboBoNT-A, abobotulinumtoxinA; AE, adverse event; TEAE, treatment-emergent adverse event; U, units; URTI, upper respiratory tract infection; —, not captured or reported at a rate < 5%.
AEs occurring in > 5% of aboBoNT-A–treated patients.
AEs occurring in > 5% of patients in at least 1 treatment group.
AEs occurring in > 2% of aboBoNT-A–treated patients.
One patient in the aboBoNT-A group did not receive treatment and was excluded from the safety analysis.
‘Neck/shoulder pain’ in Trial 1, ‘neck pain’ in Trials 2 and 3.
Despite the difference in AE reporting amongst the trials, patterns for dysphagia or muscle weakness (which are considered diffusion-related AEs) were similar across the three trials when comparing rates between the aboBoNT-A and placebo groups. The proportion of patients with headache was similar in the aboBoNT-A and placebo groups in Trials 1 and 2, but higher with aboBoNT-A than placebo in Trial 3. For neck pain, proportions were similar in the aboBoNT-A and placebo groups in Trial 2, but higher with aboBoNT-A than placebo in Trial 3 (neck pain incidence was not reported separately in Trial 1).
The proportions of patients with dysphagia were reported in the open-label extension studies and were 19%, 26%, and 20% in Cycles 1, 2, and 3, respectively, in the Trial 1 extension study, and 13%, 15%, 6%, and 10% in Cycles 1, 2, 3, and 4, respectively, in the Trial 2 extension study [11]. In the open-label extension of Trial 3, dysphagia occurred in 10.7% of patients [12]. In total, there were 19 patients of 180 (10.6%) in the treatment arms across all 3 trials with incidences of dysphagia.
4. Discussion
AboBoNT-A for the treatment of CD is approved in the United States at 500 U/1 mL and 500 U/2 mL dilutions [8]. The efficacy and safety of aboBoNT-A treatment for CD has been established in multicenter, randomized, controlled trials and open-label extension studies. Taken together, the results of these trials and extension studies demonstrate that aboBoNT-A is an effective long-term treatment for CD, irrespective of whether it is administered at the 500 U/1 mL or the 500 U/2 mL dilution. The primary efficacy data, summarized in the current report, show similar, significant improvements from baseline in TWSTRS total score at Week 4 in the aboBoNT-A groups in the trials using the 500 U/1 mL dilution and in the trial using the 500 U/2 mL dilution. Results for TWSTRS subscale scores mirrored those for the total score, and efficacy results in the extension studies were consistent with those in the double-blind phases of the three trials.
The efficacy and well-tolerated safety profile of BoNT is established across many therapeutic and aesthetic indications [9]. However, any BoNT preparation carries with it a risk of local diffusion and/or distant spread to non-target muscles, with a potential increase in diffusion-related AEs. When used for the treatment of CD, one of the most frequently reported AEs for any BoNT preparation is dysphagia, indicating local diffusion of unbound toxin away from the target muscle [9]. While dose is a key factor influencing diffusion, concerns have been raised about the role of dilution volume [9], [10], [14].
In the current study, the most frequent AEs, occurring in ≥ 5% of patients in Trials 1 and 2, were muscular weakness, dysphagia, dry mouth, injection site discomfort, fatigue, headache, musculoskeletal pain, dysphonia, injection site pain, and eye disorders (e.g. blurred vision) [8]. Some of these AEs suggest possible diffusion of aboBoNT-A to nearby, non-target muscles. For dysphagia and muscle weakness, however, AE patterns were similar across the three aboBoNT-A trials, indicating that an increased dilution of aboBoNT-A from 500 U/1 mL (used in Trials 1 and 2) to 500 U/2 mL (only used in Trial 3) did not result in an increased risk of these diffusion-related adverse events. Comparisons of headache and neck pain rates across the three trials were less conclusive, most likely because of differences in data collection (neck pain incidence was not reported separately in Trial 1) and differences in rates in the trials’ placebo arms.
The treatment of CD should be tailored to each patient’s specific needs [1]. Practice guidance for clinicians who inject toxins includes dose range suggestions for the different muscles that are commonly affected [1], [15]. Doses are individualized based on the muscle type and muscle size/activity level [1]. Although differences in BoNT preparations have been suggested to affect how far the toxins diffuse [12], [16], it has since been disproven to be the case [17]. It is likely, however, that these differences are primarily due to the dilution, volume, and dose used, as it has been found that a low concentration and higher volume results in greater localized diffusion and a wider area affected [12], [16], [18]. One of the most effective methods for limiting toxin diffusion is accurate localization and injection of the target muscle, with the use of electromyography or ultrasound to detect active muscles [16], [19].
The optimal concentration, number of units, and number of injections for each muscle need to be considered on a patient-by-patient basis [1]. When tailoring the dose to individual patients’ needs, a decrease or increase in dose can be achieved by adjusting the volume injected [8], [18]. Having the 500 U/1 mL and 500 U/2 mL dilution volumes available provides further flexibility and benefits patient care. The larger, 500 U/2 mL dilution volume affords versatility regarding the number of muscles and injection sites achievable per treatment session and supports enhanced accuracy of dosing for each injection site.
In conclusion, the results summarized here show that aboBoNT-A is similarly efficacious using either the 500 U/1 mL or the 500 U/2 mL dilution, with similar safety and tolerability across randomized placebo-controlled trials. Having dilution options offers clinicians treatment flexibility and an opportunity to individualize patient care.
5. Data sharing
Where patient data can be anonymized, Ipsen will share all individual participant data that underlie the results reported in this article with qualified researchers who provide a valid research question. Study documents, such as the study protocol and clinical study report, are not always available. Proposals should be submitted to DataSharing@Ipsen.com and will be assessed by a scientific review board. Data are available beginning 6 months and ending 5 years after publication; after this time, only raw data may be available.
Disclosures
M.F. Lew: Advisor/consultant: Acadia, Acorda, Adamas, Kyowa/Kirin, Lundbeck, Merz, Neurocrine, Revance, Sunovion, US World Meds; Speaker: Acorda, Adamas, Kyowa Kirin, Neurocrine; Researcher: Enterin Inc., Cala Labs, Michael J. Fox Foundation, Neuraly, NIH, Pharm2B, Sun Pharma. R.A. Hauser: Consulting: AbbVie, Academy for Continued Healthcare Learning, ACADIA, Acorda, Adamas, AstraZeneca, Back Bay Life Science, Biotie, Bracket, Cerecor, ClearView Healthcare Partners, ClinicalMind Medical and Therapeutic Communications, Cowen and Company, Cynapsus, Decision Resources Group, Eli Lilly, eResearch Technology, Expert Connect, Extera Partners, Gerson Lehrman Group, Guidepoint Global, GE Healthcare, Globe Life Sciences, Health Advances, HealthLogix, Health and Wellness Partners, Huron Consulting Group, Impax, Intec Pharma, Jazz, Kyowa, LCN Consulting, LifeMax, Lundbeck, The Lockwood Group, MEDACorp, Medscape, Medtronic, Michael J. Fox Foundation, Movement Disorder Society, National Institutes of Health (NIH), Neurocrine, Neuroderm, Neuropore Therapies, Outcomes Insights, Parkinson Foundation, Peerview Press, Pennside Partners, Pfizer, Pharma2B, Phase Five Communications, Piper Jaffray, Prexton, Projects in Knowledge, Putnam Associates, Quintiles, RMEI Medical Education for Better Outcomes, Sarepta, Schlesinger Associates, Scion Neurostim, Seagrove Partners, Slingshot Insights, Sun Pharma, Sunovion, Teva, US WorldMeds, Vista Research, WebMD, Windrose Consulting Group; Research support: AbbVie, Acorda, AstraZeneca, Axovant, Biogen, Cavion, Dart NeuroScience, Enterin, F. Hoffman-La Roche, Impax, Intec Pharma, Jazz, Lundbeck, Michael J. Fox Foundation, NeuroDerm, Parkinson’s Foundation, Prexton, Revance, Sunovion. S.H. Isaacson: Honoraria for CME/consultant/promotional speaker: AbbVie, ACADIA, Acorda, Adamas, Addex, Allergan, Amarantus, Amneal, Axovant, Biogen, Britannia, Eli Lilly, Enterin, GE Healthcare, Global Kinetics, Intec Pharma, Ipsen, Kyowa, Lundbeck, Merz, Michael J. Fox Foundation, Neurocrine, Neuroderm, Parkinson Study Group, Pharma2B, Revance, Roche, Sanofi, Sunovion, Supernus, Teva, UCB, US World Meds, Zambon; Research grants: AbbVie, ACADIA, Acorda, Adamas, Addex, Allergan, Amarantus, Amneal, Axovant, Biogen, Britannia, Eli Lilly, Enterin, GE Healthcare, Global Kinetics, Intec Pharma, Ipsen, Kyowa, Lundbeck, Michael J. Fox Foundation, Neurocrine, Neuroderm, Parkinson Study Group, Pharma2B, Revance, Roche, Sage, Sanofi, Sunovion, Teva, UCB, US WorldMeds. D. Truong: Research grants: AbbVie, Acadia, Acorda, Auspex, Cynapsus, Daiichi Sankyo, Intec, Ipsen, Kyowa, Merz, National Institute of Neurological Disorders and Stroke, Neurocrine, Neuroderm, Prexton Therapeutics, Sunovion; Fees: Adamas, Alexza, Teva, US WorldMeds. A.T. Patel: Consultant: Allergan, Ipsen, Revance; Speaker: Allergan, Ipsen, Merz; Research support: Allergan, Ipsen, Revance. A. Brashear: Consulting: Ipsen, Revance; Research support: paid to Wake Forest institution. W. Ondo: Advisor/consultant/speaker: Acorda, ACADIA, Amneal, Adamas, Neurocrine, Teva, US WorldMeds; Research support: Abbvie, Biogen, Lundbeck, Revance, Sunovian. P. Maisonobe: Employment: Ipsen. K. Dashtipour: Advisor/consultant/speaker: AbbVie, ACADIA, Acorda, Adamas, Allergan, Amneal, Ipsen, Impax, Lundbeck, Merz, Revance, Neurocrine, Sunovion, Teva, US WorldMeds. L. Bahroo: Speaker’s bureaus: Allergan, Ipsen, Us WorldMeds; Consultant: US WorldMeds. S. Wietek: Employment: former employee of Ipsen.
Funding
The study was funded by Ipsen, Cambridge, MA, USA.
CRediT authorship contribution statement
Mark F. Lew: Conceptualization, Investigation, Methodology, Writing – original draft, Writing – review & editing. Robert A. Hauser: Conceptualization, Investigation, Methodology, Writing – original draft, Writing – review & editing. Stuart H. Isaacson: Conceptualization, Investigation, Methodology, Writing – original draft, Writing – review & editing. Daniel Truong: Conceptualization, Investigation, Methodology, Writing – original draft, Writing – review & editing. Atul T. Patel: Conceptualization, Investigation, Methodology, Writing – original draft, Writing – review & editing. Allison Brashear: Conceptualization, Investigation, Methodology, Writing – original draft, Writing – review & editing. William Ondo: Conceptualization, Investigation, Methodology, Writing – original draft, Writing – review & editing. Pascal Maisonobe: Formal analysis. Khashayar Dashtipour: Conceptualization, Investigation, Methodology, Writing – original draft, Writing – review & editing. Laxman Bahroo: Conceptualization, Investigation, Methodology, Writing – original draft, Writing – review & editing. Stefan Wietek: Conceptualization, Investigation, Methodology, Writing – original draft, Writing – review & editing.
Declaration of Competing Interest
SHI received honoraria, consultancy, and promotional speaker fees from Ipsen. ATP received consultant and speaker fees from Ipsen. AB received consultancy fees from Ipsen. KD received advisor/consultant/speaker fees from Ipsen. LB received honoraria from Ipsen. SHI, DT, and ATP received research grants/support from Ipsen. PM is employed by Ipsen, and SW is a former employee of Ipsen.
Acknowledgements
The authors thank all patients involved in the study, as well as investigators and research staff in participating institutions, and James Otto, for his substantial contributions to the study. The authors also thank Rosemary Reinke, PhD, and Kate Katsaval, BSc, of The Medicine Group LLC (New Hope, PA, USA) for providing medical writing support, which was sponsored by Ipsen (Cambridge, MA, USA) in accordance with current Good Publication Practice guidelines (GPP3) and International Committee of Medical Journal Editors (ICMJE).
References
- 1.Albanese A., Abbruzzese G., Dressler D., Duzynski W., Khatkova S., Marti M.J., Mir P., Montecucco C., Moro E., Pinter M., Relja M., Roze E., Skogseid I.M., Timerbaeva S., Tzoulis C. Practical guidance for CD management involving treatment of botulinum toxin: a consensus statement. J. Neurol. 2015;262(10):2201–2213. doi: 10.1007/s00415-015-7703-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.G. Defazio, J. Jankovic, J.L. Giel, S. Papapetropoulos, Descriptive epidemiology of cervical dystonia, Tremor. Other Hyperkinet. Mov. (N Y) 3 (2013) tre-03-193-4374-4372. [DOI] [PMC free article] [PubMed]
- 3.Mordin M., Masaquel C., Abbott C., Copley-Merriman C. Factors affecting the health-related quality of life of patients with cervical dystonia and impact of treatment with abobotulinumtoxinA (Dysport): results from a randomised, double-blind, placebo-controlled study. BMJ Open. 2014;4(10):e005150. doi: 10.1136/bmjopen-2014-005150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Simpson D.M., Hallett M., Ashman E.J., Comella C.L., Green M.W., Gronseth G.S., Armstrong M.J., Gloss D., Potrebic S., Jankovic J., Karp B.P., Naumann M., So Y.T., Yablon S.A. Practice guideline update summary: botulinum neurotoxin for the treatment of blepharospasm, cervical dystonia, adult spasticity, and headache: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2016;86(19):1818–1826. doi: 10.1212/WNL.0000000000002560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Truong D., Duane D.D., Jankovic J., Singer C., Seeberger L.C., Comella C.L., Lew M.F., Rodnitzky R.L., Danisi F.O., Sutton J.P., Charles P.D., Hauser R.A., Sheean G.L. Efficacy and safety of botulinum type A toxin (Dysport) in cervical dystonia: results of the first US randomized, double-blind, placebo-controlled study. Mov. Disord. 2005;20(7):783–791. doi: 10.1002/mds.20403. [DOI] [PubMed] [Google Scholar]
- 6.Truong D., Brodsky M., Lew M., Brashear A., Jankovic J., Molho E., Orlova O., Timerbaeva S. Long-term efficacy and safety of botulinum toxin type A (Dysport) in cervical dystonia. Parkinsonism Relat. Disord. 2010;16(5):316–323. doi: 10.1016/j.parkreldis.2010.03.002. [DOI] [PubMed] [Google Scholar]
- 7.Lew M.F., Brashear A., Dashtipour K., Isaacson S., Hauser R.A., Maisonobe P., Snyder D., Ondo W. A 500 U/2 mL dilution of abobotulinumtoxinA vs. placebo: randomized study in cervical dystonia. Int. J. Neurosci. 2018;128(7):619–626. doi: 10.1080/00207454.2017.1406935. [DOI] [PubMed] [Google Scholar]
- 8.Dysport [package insert], Ipsen Biopharmaceuticals, Inc., Cambridge, MA, 2020.
- 9.Fonfria E., Maignel J., Lezmi S., Martin V., Splevins A., Shubber S., Kalinichev M., Foster K., Picaut P., Krupp J. The expanding therapeutic utility of botulinum neurotoxins. Toxins (Basel) 2018;10(5):208. doi: 10.3390/toxins10050208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Alter K.E., Wilson N.A. Springer Publishing Company; 2014. Botulinum Neurotoxin Injection Manual. [Google Scholar]
- 11.Hauser R.A., Truong D., Hubble J., Coleman C., Beffy J.L., Chang S., Picaut P. AbobotulinumtoxinA (Dysport) dosing in cervical dystonia: an exploratory analysis of two large open-label extension studies. J. Neural Transm. (Vienna) 2013;120(2):299–307. doi: 10.1007/s00702-012-0872-1. [DOI] [PubMed] [Google Scholar]
- 12.K. Dashtipour, J. Otto, P. Maisonobe, L. Bahroo, D. Truong, R. Trosch, AbobotulinumtoxinA using 2 mL dilution maintains durable functional improvements across multiple treatment cycles [abstract], Mov Disord 34 (suppl 2) (2019) https://www.mdsabstracts.org/abstract/abobotulinumtoxina-using-2-mldilution-maintains-durable-functional-improvements-across-multiple-treatment-cycles/ Accessed January 6, 2020. [DOI] [PMC free article] [PubMed]
- 13.Patel A., Lew M., Brashear A., Dashtipour K., Isaacson S., Hauser R., Ondo W., Maisonobe P., Otto J. Sustained functional benefits after a single injection with abobotulinumtoxinA using a 2 mL injection volume in adults with cervical dystonia. Mov. Disord. 2019;34(S2):S544. [Google Scholar]
- 14.Hexsel D., Hexsel C., Siega C., Schilling-Souza J., Rotta F.T., Rodrigues T.C. Fields of effects of 2 commercial preparations of botulinum toxin type A at equal labeled unit doses: a double-blind randomized trial. JAMA Dermatol. 2013;149(12):1386–1391. doi: 10.1001/jamadermatol.2013.6440. [DOI] [PubMed] [Google Scholar]
- 15.Pickett A., Rosales R.L. New trends in the science of botulinum toxin-A as applied in dystonia. Int. J. Neurosci. 2011;121(sup1):22–34. doi: 10.3109/00207454.2010.539306. [DOI] [PubMed] [Google Scholar]
- 16.M.A. Brodsky, D.M. Swope, D. Grimes, Diffusion of botulinum toxins, Tremor Other Hyperkinet Mov (N Y) 2 (2012) tre-02-85-417-411. [DOI] [PMC free article] [PubMed]
- 17.Han Y., Stevens A.L., Dashtipour K., Hauser R.A., Mari Z. A mixed treatment comparison to compare the efficacy and safety of botulinum toxin treatments for cervical dystonia. J. Neurol. 2016;263(4):772–780. doi: 10.1007/s00415-016-8050-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ramirez-Castaneda J., Jankovic J., Comella C., Dashtipour K., Fernandez H.H., Mari Z. Diffusion, spread, and migration of botulinum toxin. Mov. Disord. 2013;28(13):1775–1783. doi: 10.1002/mds.25582. [DOI] [PubMed] [Google Scholar]
- 19.Alter K.E., Karp B.I. Ultrasound guidance for botulinum neurotoxin chemodenervation procedures. Toxins (Basel) 2017;10(1):18. doi: 10.3390/toxins10010018. [DOI] [PMC free article] [PubMed] [Google Scholar]