Abstract
Pooled evidence conveys the association between polyunsaturated fatty acids and infectious disease. SARS-CoV-2, an enveloped mRNA virus, was also reported to interact with polyunsaturated fatty acids. The present review explores the possible mode of action, immunology, and consequences of these polyunsaturated fatty acids during the viral infection. Polyunsaturated fatty acids control protein complex formation in lipid rafts associated with the function of two SARS-CoV-2 entry gateways: angiotensin-converting enzyme-2 and cellular protease transmembrane protease serine-2. Therefore, the viral entry can be mitigated by modulating polyunsaturated fatty acids contents in the body. α-Linolenic acid is the precursor of two clinically important eicosanoids eicosapentaenoic acid and docosahexaenoic acid, the members of ω-3 fats. Resolvins, protectins, and maresins derived from docosahexaenoic acid suppress inflammation and augment phagocytosis that lessens microbial loads. Prostaglandins of 3 series, leukotrienes of 5 series, and thromboxane A3 from eicosapentaenoic acid exhibit anti-inflammatory, vasodilatory, and platelet anti-aggregatory effects that may also contribute to the control of pre-existing pulmonary and cardiac diseases. In contrast, ω-6 linoleic acid-derived arachidonic acid increases the prostaglandin G2, lipoxins A4 and B4, and thromboxane A2. These cytokines are pro-inflammatory and enhance the immune response but aggravate the COVID-19 severity. Therefore, the rational intake of ω-3-enriched foods or supplements might lessen the complications in COVID-19 and might be a preventive measure.
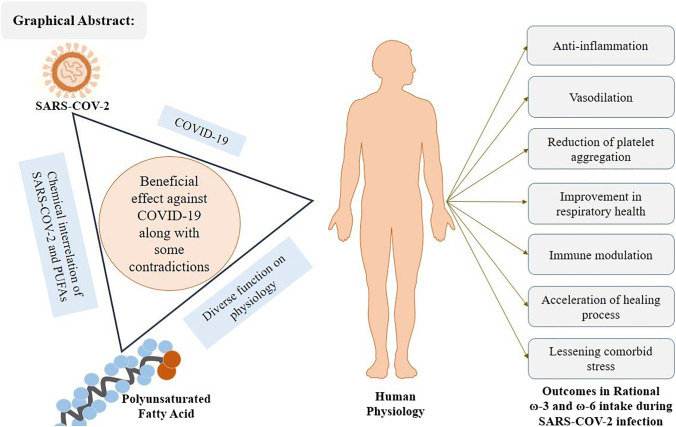
Graphic Abstract
Keywords: Anti-inflammatory, Efferocytosis, Immune system, Pro-resolving lipid mediators, Platelet aggregation, Viral replication
Introduction
Coronavirus disease-19 (COVID-19) costs above 195 million infections and 4.18 million deaths globally from December 2019 to July 2021 (WHO 2020). Countries are approaching to combat the outrageous form of the pandemic. However, they have to face different issues such as low efficacy of the vaccine, shortage of vaccine production facilities, the origin of more virulent SARS-CoV-2 variant, difficulty in the world vaccine management system, and inability to develop effective antiviral drugs (Johnson et al. 2021). So, the day-by-day, COVID-19 is becoming ferocious with multiform infections. That is why, from the outbreak to now, health experts suggest minimizing the risk by practicing standard hygiene and taking nutritious foods (de Finger et al. 2021). A nutrient-rich diet helps to reduce the risk of comorbidity and boost the body’s immune response. Polyunsaturated fatty acids (PUFAs) are fatty acids that contain more than one double bond in their backbone. This class includes many important compounds, such as essential fatty acids that give drying oils their characteristic property. Their pivotal role in improving health has been well-documented (Saini and Keum 2018; Sokoła-Wysoczańska et al. 2018).
α-Linolenic acid (ALA) and linoleic acid (LA) are two essential PUFAs which give rise to other clinically important PUFAs like eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), and arachidonic acid (AA). They impart a vital role to body homeostasis (Saini and Keum 2018). Dietary supplementation of omega-3 (ω-3) PUFAs improves the clinical condition in obesity, inflammation, hypertension, dyslipidemia, atherosclerosis, diabetes mellitus, metabolic syndrome, neurological/neuropsychiatric disorders, and eye diseases and helps fetal neurological developments (Yashodhara et al. 2009). On the other hand, eicosanoids derived from omega-6 (ω-6) PUFAs enhance the inflammatory response, concurring that the increased ratio of ω-6 and ω-3 in food may increase in chronic inflammatory diseases (Patterson et al. 2012).
Nevertheless, recent pooled evidence has consistently reported that chronic diseases like chronic obstructive pulmonary disease (COPD), cardiovascular disease (CVD), cancer, cerebrovascular accident (CVA), diabetes, hypertension, and chronic kidney disease have an association with the severity among COVID-19 patients (Baral et al. 2021). So, polyunsaturated fatty acids may have an essential role in lessening the risk of these comorbidities and improving the health of COVID-19 positive cases (Asher et al. 2021). Furthermore, the anti-inflammatory metabolites of AA, EPA, and DHA, collectively named as specialized pro-resolving lipid mediators (SPMs) such as lipoxins, resolvins, protectins, and maresins, suppress inflammation. The augmented phagocytosis of macrophages and other immunocytes decreases the microbial load and enhances the healing process (Das 2020). The present review aims to demonstrate the effect and rational selection of PUFAs to lessen COVID-19 severity with probable mechanisms.
Search Strategy
The study focused primarily on the buildup of all possible outcomes of polyunsaturated fatty acids in COVID-19 patients. Then, we had a recourse on multiple works about fatty acids’ role in viral infection published before the COVID-19 outbreak to analyze and support the outcomes. Both prospective and retrospective trials and supportive articles were picked up from PubMed, Google Scholar, and Web of Science databases from 15th May 2021 to 30th June 2021 using the English language. The more frequent searching keywords were “polyunsaturated fatty acids” or “omega-3” or “omega-6” or “PUFAs” with “COVID-19” or “coronavirus” or “SARS-CoV-2” or “severe acute respiratory syndrome-2.” We collected data from articles published by peer review journals from the 1980s to 30th June 2021.
Discussion
Classification
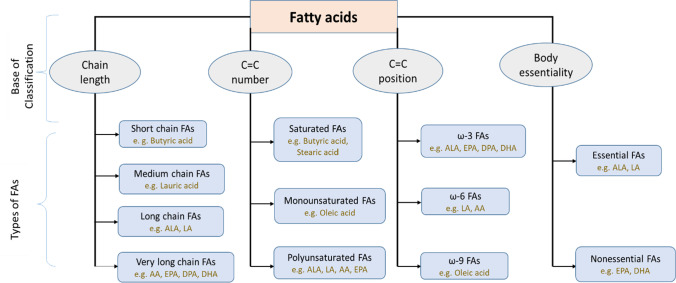
The hydrocarbon containing a one-end carboxyl group and the other end methyl group is chemically known as fatty acids. Few substantive features like the chain length in numbers of carbon, the number of carbon–carbon double bonds (unsaturations), and the position of the double bonds determine the fatty acids’ biological reactivity (Patterson et al. 2012). Human physiology can synthesize fatty acids where few acids cannot be synthesized. According to these features, the acids can be classified in four ways (Fig. 1). Although the general notion “polyunsaturated fat” indicates only multiple levels of unsaturation, its character and functions depend on all the features mentioned above.
Fig. 1.
Classification of fatty acids
According to a chain length of fatty acids, they are classified in three types: short-chain FAs (SCFAs) (C2–6), medium-chain FAs (MCFAs) (C6–12), long-chain FAs (LCFAs) (C12–18) and very-long-chain FAs (> 18) (VLCFAs). LCFAs and VLCFAs are more abundant in plants and animals, but SCFAs and MCFAs also present in comparatively small amounts (Schönfeld and Wojtczak 2016). SCFAs and MCFAs are mainly present in triglycerides in some plant oils and milk. Furthermore, SCFAs are also produced in the gut of human and other mammals by bacterial fermentation of non-starch polysaccharides and amylase-resistant starch (Wong et al. 2006). LCFAs are usually referred to as free fatty acids or unsaturated fatty acids with an even carbon count. It is synthesized in animals or plants from acetyl-CoA and is then degraded via β-oxidation reactions (Kaneko et al. 2008). VLCFAs are derived from parental 18-carbon molecules by several biochemical reactions (Agostoni and Bruzzese 1992).
The second way of classification is the degree of unsaturations. When FAs have no unsaturated bonds, it is termed as saturated fatty acids (SFAs). Very few FAs like oleic acid have only a single double bond, called monounsaturated fatty acids (MUFAs), e.g., omega-7 and omega-9 fats. Most naturally occurring FAs have more than one unsaturated double bonds, commonly known as polyunsaturated fatty acids (Agostoni and Bruzzese 1992; Saini and Keum 2018). The first double bond starting from the methyl-end (the opposite side of the molecule) is counted to categorize FAs and denoted by omega, ω, or n − FAs. All unsaturated FAs are characterized, by using this classification, in three prominent families: ω-3, ω-6, and ω-9. Alpha-linolenic acid (ALA, 18:3, n-3; C18H30O2) and linoleic acid (LA, 18:2, n-6; C18H32O2) are two FAs that cannot be synthesized within the human body, referred to as essential fatty acids (EFAs), whereas other FAs are known as nonessential FAs (Yashodhara et al. 2009).
Clinically Significant PUFAs
The essential PUFAs ALA and AA cannot be generated in humans and are therefore regarded as parent PUFAs, whereas eicosapentaenoic acid (EPA, 20:5, n-3), docosahexaenoic acid (DHA, 22:6, n-3), and γ-linolenic acid (GLA, 18:3, n-6) are endogenous nonessential PUFAs belonging to the ω-3 and ω-6 families, respectively. Dietary ALA and LA have crucial roles in maintaining ω-3 and ω-6 LC-PUFAs levels in tissue (Barceló-Coblijn and Murphy 2009). However, when there is a lack of EFAs, pathophysiological ω-9 PUFAs are synthesized in increased amounts (Burdge 2011). PUFAs exert a wide range of beneficial effects on lipoprotein concentration, regulation of blood pressure, the function of membrane enzymes and receptors, membrane fluidity, modulation of eicosanoid production, and metabolism of minerals (Simopoulos et al. 1999). Among different PUFAs, ω-3 and ω-6 fatty acids are synthesized and accumulated in specific tissues and clinically play significant roles (Saini and Keum 2018).
Omega-3 PUFAs
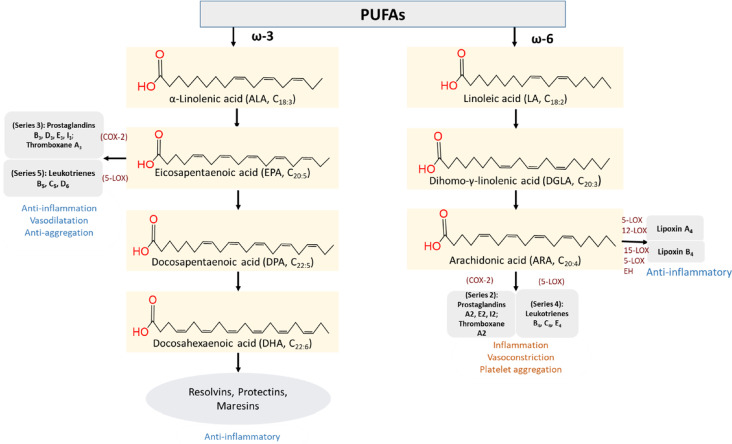
The first member of ω-3 PUFAs series is all-cis-9,12,15-octadecatrienoic (α-linolenic acid, ALA 18:3). The more important few ω-3 PUFAs derived from ALA are all-cis-5,8,11,14,17-eicosapentaenoic (EPA 20:5), all-cis-7,10,13,16,19-docosapentaenoic (DPA 22:5), and all-cis-4,7,10,13,16,19-docosahexaenoic (DHA 22:6) (Fig. 2). Bioactive signaling lipids, commonly known as eicosanoids, are derived from oxidation of EPA and DHA by several enzymes like cyclooxygenase (COX-1 and COX-2), lipoxygenase (5-LOX and 15-LOX), and epoxygenases (cytochrome P450 or CYP450).
Fig. 2.
Clinically significant polyunsaturated fatty acid families: ω-3 and ω-6 and their members associated with the pathophysiology of humans; COX, cyclooxygenase; LOX, lipoxygenase; EH, epoxide hydrolase
Eicosanoids are not produced in a steady ratio. However, after biosynthesis, esterified EPA and DHA are stored in neutral triglycerides or phospholipids, and when required, they can be mobilized to form eicosanoids and other autacoids. COX-2 mobilizes EPA to series 3 eicosanoids (PG B3, D3, E3, I3, and TXA3), and 5-LOX mobilizes to series 5 eicosanoids (LT B5, C5, and D6) (Calder 2003; Sabater et al. 2011). These are anti-inflammatory mediators which cause vasodilatation and anti-aggregation of platelets. Additionally, DHA metabolizes SPMs of the lipoxin, resolvin, protectin, and maresin families during inflammation (Scheme 1).
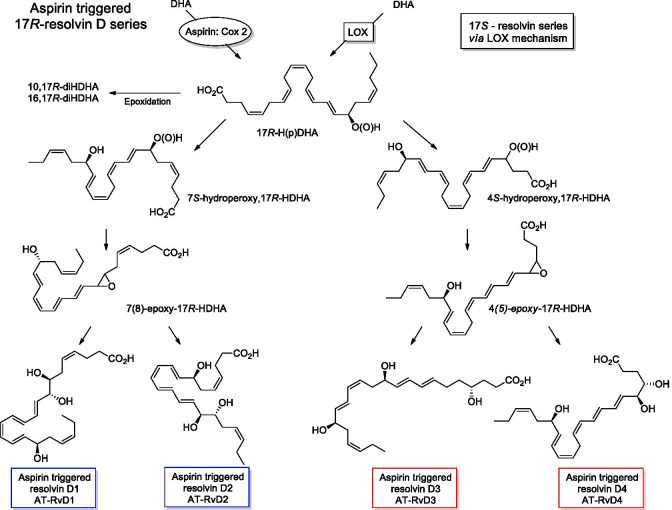
Scheme 1.
Aspirin activated biosynthesis of specialized pro-resolving mediators from docosahexaenoic acid (DHA). Aspirin not only blocks the biosynthesis of prostaglandins, but also stimulates the endogenous production of anti-inflammatory and pro-resolving lipid mediators termed aspirin-triggered specialized pro-resolving mediators (AT-SPMs), such as aspirin-triggered resolvins (AT-RvDs) and lipoxins (AT-LXs)
The SPMs initiate the process of resolution which include restriction or cessation of neutrophil infiltration, counter-regulation of chemokines and cytokines, induction of the neutrophils apoptosis and subsequent efferocytosis (the process by which apoptotic cells are removed by phagocytic cells) by macrophages, the conversion of macrophages from classically activated (M1) to alternatively activated cells (M2), return of non-apoptotic cells to the vascular system or lymphatic vessels, and the start of the healing process. These events facilitate proper return to the homeostasis balance (Park et al. 2020; Lee 2021). For example, resolvin D1 (1) and D2 (2) impart in resolving inflammation and protecting organ damage in injury (Duffield et al. 2006; Spite et al. 2009). In addition, resolvin D1 promotes the targeting and clearance of necroptotic cells (Gerlach et al. 2020). Another anti-inflammatory mediator resolvin D6 (3) induces nerve reaeration and post-infection healing (Pham et al. 2020). Neuroprotectin D1 (4) is also an anti-inflammatory mediator that reduces oxidative stress in several pathological events like stroke, retinal degenerations, and Alzheimer’s disease (Bazan 2005; 2009). Furthermore, maresins MaR1 (5) and MaR2 (6) act as anti-inflammatory and pro-resolving factors in wound healing (Wang et al. 2015; Tang et al. 2018). Ma R1 promoted de novo generation of regulatory T cells and interacted with type 2 innate lymphoid cells to inhibit TGF-β-dependent cytokine production, which effectively reduced lung inflammation and attenuate pulmonary fibrosis (Li et al. 2020). Lipoxin A4 (7) and B4 (8) attenuates or prevents an inflammatory response via the immunosuppressive activity of stem cells of the apical papilla through the activation of its receptor, ALX/FPR2 (Gaudin et al. 2018).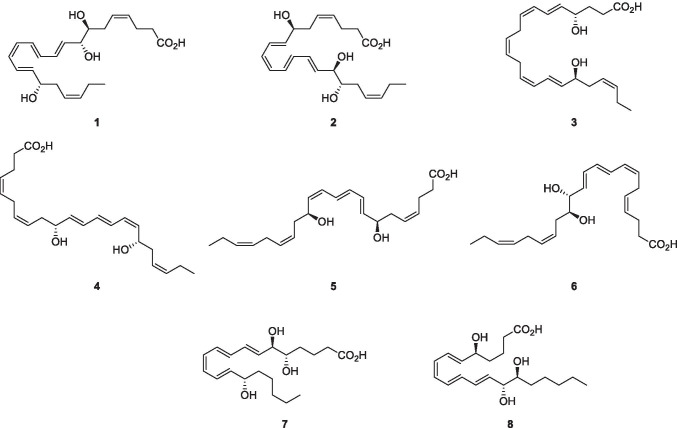
Omega-6 PUFAs
Parental all-cis-9,12-octadecadienoic (linoleic acid, LA, 18:2) of ω-6 family produces two important PUFAs: all-cis-8,11,14-eicosatrienoic (dihomo-γ-linolenic acid, DGLA, 20:3) and all-cis-5,8,11,14-eicosatetraenoic (arachidonic acid, AA, 20:4). Series 1 prostaglandin PGs (PGE1, PGF1, thromboxane A1) and series 3 leukotrienes (LTs), such as LTA3, LTC3, and LTD3, are produced by the enzyme COX and LOX, respectively (Yashodhara et al. 2009). Like EPA and DHA, AA is stored in esterified form and mobilized by phospholipase A2 (PLA2) to form eicosanoids. COX-2 converts AA to series 2 eicosanoids (PGA2, PGE2, PGI2, and TXA2). Similarly, series 4 eicosanoids (LTB4, LTC4, and LTE4) are synthesized by lipoxygenases (5-LOX) from AA (Fig. 2). Additionally, various hydroperoxy- and hydroxyl-eicosatetraenoic acid (HPETE and HETE) derivatives and lipoxin A4 are also produced from AA (Schmitz and Ecker 2008).
Most cell signaling molecules from DGLA and AA are pro-inflammatory, vasoconstrictor, platelet aggregator, and disease-propagating (Robinson and Stone 2006). However, in the case of inflammatory modulation, PGE2 exerts both pro- and anti-inflammatory roles. PGE2 induces COX-2 and 15-LOX, producing pro-inflammatory IL-6 and anti-inflammatory lipoxins, respectively (Serhan et al. 2003). In contrast, 5-lipoxygenase inhibition of PGE2 decreases the pro-inflammatory series 4 LT level (Levy et al. 2001).
PUFAs in COVID-19 Infection
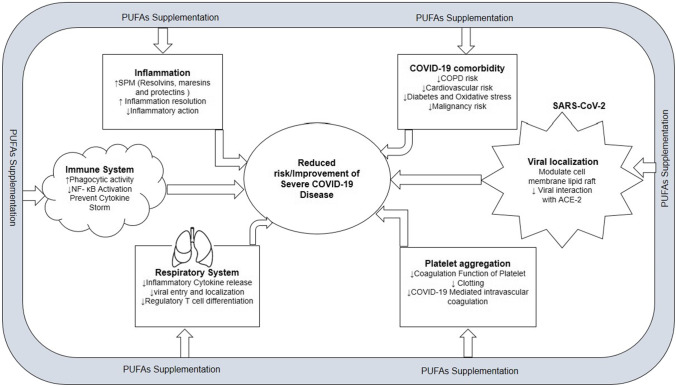
PUFAs play essential roles in the cell activity of each organ and tissue in the human body. PUFAs with structural and physiological significance have a beneficial effect on pathological conditions such as infection or inflammation. Given that COVID-19 is a viral illness associated with significant inflammation of the respiratory tract, there is a possibility of benefiting from PUFA supplementation (Lee 2021). The potential beneficial effects of PUFA supplementation based on research evidence are depicted in Fig. 3.
Fig. 3.
Potential beneficial effect of PUFA supplementation in severe COVID-19 patients. PUFAs, polyunsaturated fatty acids; SPM, specialized pro-resolving mediators; NF-κB, nuclear factor kappa-B; COPD, chronic obstructive pulmonary disease; ACE-2, angiotensin-converting enzyme-2
Role on Respiratory System
The essential PUFAs ω-3 and ω-6 modulate the inflammatory response that potentially influences respiratory health involving chronic inflammatory and infectious processes (Lemoine et al. 2020). However, PUFAs (especially ω-3) affect blood rheology, host–microbial interactions, and surfactant production in the lung (Knapp, 1995). So, commonly PUFAs are thought to be essential nutrients to prevent lung diseases like acute respiratory distress syndrome (ARDS), chronic obstructive pulmonary disease (COPD), asthma, and allergies. Nevertheless, the exact modes of these effects on the respiratory system remain primarily unknown. SPMs, such as protectins, resolvins, and maresins, from ω-3 PUFAs counter-regulate airway eosinophilic inflammation against the pro-inflammatory mediators of ω-6 PUFAs and promote the resolution of inflammation in vivo (Miyata and Arita 2015).
Two studies demonstrated that dietary fatty acid supplements in infants or children at the age range of 6 months to 5 years decreased wheeze prevalence and lessened bronchodilator use at 1.5 years of age (Mihrshahi et al. 2004). Moreover, such clinical intervention reduced the prevalence of cough allergic sensitization at 3 years of age; however, the effects on the prevalence of asthma were not observed (Damsgaard et al. 2007). Other clinical studies with ω-3 PUFAs supplementation diminished the production of LTs from polymorphonuclear cells in athlete and asthmatic adults and decreased bronchoconstriction after exercise (Mickleborough et al. 2006). Another work on ω-3-rich supplementation revealed the beneficial and suppressive effects on exhaled NO levels before and after the intervention (Schubert et al. 2009).
Masclans et al. (1999) determined plasma levels of TXB2, PGF1α, and LTB4 in peripheral arteries, pulmonary arteries, and venous system within 48 h of 21 ARDS cases. Eicosanoids were considerably more significant in ARDS patients than in the healthy control group. Furthermore, survivors presented higher eicosanoid levels in systemic pulmonary arteries than nonsurvivors. However, Auner et al. showed that LTB4 concentrations were considerably more significant in patients with respiratory problems, and it explained LTB4 levels as biochemical markers of respiratory disorders such as pneumonia, ARDS, respiratory failure, and pulmonary embolism (Auner et al., 2012). Another study also observed pro-inflammatory eicosanoid LTB4 as a correlating mediator between lung injury and severe ARDS patients (Masclans et al., 2007). Therefore, these studies draw an apparent connection between respiratory health and PUFAs, and probably the effect of ω-6 FAs family is opposed by ω-3 FAs.
Role Against Viral Replication
Long-chain and very-long-chain PUFAs EPA, DHA, AA, and ALA have a substantial role in the immunological defense against viral entry, localization, and replication to new copies (Calder et al. 2020). PUFAs can control membrane features such as membrane fluidity and protein complex formation in lipid rafts since they are the components of membrane phospholipids. Angiotensin-converting enzyme-2 (ACE2) and cellular protease transmembrane protease serine-2 (TMPRSS-2), which are the entry gateway for SARS-CoV, are most commonly found in lipid rafts (Ballout et al. 2020). ACE2 and TMPRSS2 expression, stability, and enzymatic activity may be affected by the quantity and size of lipid raft and non-raft domains. A study on Vero E6 cells showed the involvement of lipid raft on SARS-CoV entry (Lu et al. 2008), and another study by Glende et al. suggested that ACE-2 mediated viral entry can be attenuated by modulation in lipid raft (Glende et al. 2008). Comparing to other PUFAs, DHA is the most potent fatty acid to participate in lipid raft formation (Wassall et al. 2018).
Metabolic reprogramming of cells in pro-inflammatory stimulants is a recurring event. Transcription factors sterol regulatory element-binding proteins (SREBPs) and their isomers are reprogramed with metabolism in the cell if they are virally infected. Modulation of biomolecules including enzymes connected with numerous signals affecting normal and pathological cell activity is favorable for the long-chain ω-3 fatty acid. One of the strategies of the virus is to reprogram host lipid metabolism to supply sufficient lipid molecules necessary for virion replication membrane formation. One manner in which MERS-CoV might influence lipid metabolism of host cells and remodel de novo SREBP-dependent lipogenesis pathway is by maintaining an abundance of circulating fatty acids (Yuan et al. 2019). It has also been demonstrated that SREBPs are active during flavivirus infection (Pombo and Sanyal 2018). Following this, a potential therapeutic intervention against viral infection is designed targeting SREBP ½. As ω-3 PUFAs like EPA and DHA block the conversion of SREBP1 from its inactive to active mature form and accelerate the breakdown of mRNA for SREBP-1c, it may prevent the viral activating mechanism and shorten the duration of its replication. The outcome of this suppression is that cell concentrations of the matured SREBP are reduced overall, and lipogenic gene codes, especially acetyl-CoA carboxylases, fatty acid synthases, and stearoyl-CoA desaturases, are reduced (Deckelbaum et al. 2006). Viruses can cause large-scale changes in the host cell’s metabolism, including the activation of fatty acid production in human hepatocytes by the hepatitis C virus (HCV) and the consumption of cellular lipid reserves in hepatocytes by the dengue virus (Matheson et al. 2015).
Role Against Inflammation
PUFAs are crucial for membrane phospholipid structural integrity and flexibility. Immune cells such as lymph nodes or splenic lymphocytes collected from laboratory mice often contain 15–20% of PUFAs as AA and include relatively few EPA and DHA (Calder et al. 1994). Modulation of the composition of fatty acid of these animals’ diets results in changes to the fatty acid content of their immune cells, and thus, the AA-, EPA-, or DHA-enriched diet improves the accumulation of these PUFAs in the immune cell (Peterson et al. 1998). PUFAs also affect gene expression and offer a component for lipid mediator production, like cytokines (Calder, 2001). The role of the AA cascade on inflammation through viral stimulation is well-documented, while EPA- and DHA-derived eicosanoids have the anti-inflammatory potential (Ripon et al. 2021). The PLA2 enzyme activity triggered by physiological or pathological events results in the mobilization of AA for the development of inflammatory cytokines in the promotional phase of inflammation (Innes and Calder, 2020). Three individual enzymes named COX, LOX, and cytochrome P450 (CYP450) play a role in the enzymatic oxidization of AA to inflammatory and anti-inflammatory cytokines, as represented in Fig. 2. It can be said that ω-6 PUFAs have been shown to activate the inflammation promotion phase. Secondly, the affected cell or tissue tries to restore as a homeostasis process is the resolution phase of inflammation, and ω-3 PUFAs can stimulate this resolving phase at the inflammation site.
The principal precursors to inflammation resolution are EPA and DHA. Calder demonstrated that ω-3 PUFAs like EPA and DHA are incorporated across the body into the phospholipid layer of neutrophils’ cell membranes and produce various mediators, including PGs, LTs, and maresins. As a result, in case of injury, the by-products of these cell membranes can produce less inflammatory eliciting mediators than ω-6 PUFAs like AA and LA, more frequently found in the western diet (Calder 2011). EPA- and DHA-mediated SPMs known as resolvins, maresins, and protectins impede the synthesis of pro-inflammatory cytokines by diminishing NF/kB pathway (Basil and Levy 2016). E-series and D-series resolvins synthesized from EPA and DHA have potential anti-inflammatory activity by reducing the infiltration of leukocytes at the damaged tissue, whereas the DHA-derived maresins can resolve inflammation through inducing macrophage phagocytosis of neutrophils (Rius et al. 2012). Western diets are predisposed to a high ω-6/ω-3 ratio that can enhance many diseases’ pathogenesis; again US adults and children consume the insufficient amount of DHA and EPA (Sheppard and Cheatham 2018). Furthermore, a laboratory study suggested that the anti-inflammatory action of ω-3 PUFAs is inhibited by ω-6 counterparts (Innes and Calder 2018). Morin et al. (2014) suggested another minor ω-3 PUFA, docosapentaenoic acid (DPA), as significant to the inflammatory resolution of the lungs.
Enormous in vivo and in vitro studies have been launched, highlighting the anti-inflammatory role of PUFAs. One study with ω-3 PUFA emulsion to sterile peritonitis and murine polymicrobial sepsis condition showed potential anti-inflammatory activity by diminishing infiltration of neutrophils and pro-inflammatory mediator synthesis (Körner et al. 2018). Another study conducted by Saedisomeolia et al. (2009) to identify the anti-inflammatory potential of AA, DHA, and EPA in airway epithelial cell inflammation induced by a virus named rhinovirus demonstrated that DHA significantly diminished the release of IP-10 and IL-6 release from airway epithelial cells. The mechanisms underlying the anti-inflammatory activity are inhibiting the synthesis of eicosanoids from AA and decreased cellular immune function. A clinical study launched for 3 months on patients with hemodialysis showed that an additional diet with DHA and EPA lowered the serum level of C-reactive protein (CRP), which is a biomarker of inflammation (Saifullah et al. 2007). Supplementation with EPA and DHA has been shown to enhance both fatty acids in blood lipids, blood cells, and other tissue regions. Another clinical trial launched by Gerling et al. (2019) shows a 12-week supplementation of fish oil equivalent to 3 g EPA + 2 g DHA/day to healthy young men increases the ω-3 PUFA content in the mitochondrial membrane and whole muscle at the same time reduces the ω-6/ω-3 ratio. Overall, the study’s outcome represents that the incorporation of DHA and EPA in diet can reduce the AA level for eicosanoids synthesis, thus inducing anti-inflammatory potentials.
A clinical study for 8 weeks by De Souza et al. (2020) demonstrated that fish oil enriched with 1.44 g EPA and 0.96 g DHA when supplemented to obese type 2 diabetes patients diminished the inflammatory biomarker (IL-1β and IL-6 and TNF-α) level. The inhibition of leukocyte chemotaxis, reduction in cell adhesion molecule expression and leukocyte–endothelial adhesive interactions, interrupting lipid rafts, inability to activate nuclear factor kappa-B (NF-κB), activation of peroxisome proliferator-activated receptor gamma (PPARγ), and binding to G protein-coupled receptor (GPCR) are the underlying measures of anti-inflammatory action of EPA and DHA. Again, the synthesis of SPMs such as resolvins, maresins, and protectins from enzymatic oxidation of EPA and DHA can resolve inflammation by interrupting the trans-endothelial neutrophil migration and chemokine and cytokine production (Calder 2013). Inflammatory biomarkers such as tumor necrosis factor-α (TNF-α), IL-1, IL-2, IL-6, IL-7, IL-10, IL-12, IL-17, macrophage colony-stimulating factor (M-CSF), granulocyte colony-stimulating factor (G-CSF), chemokine CXCL-10 (IP-10), monocyte chemoattractant protein-1 (MCP-1), and macrophage inflammatory protein-1α ( MIP-1α) were shown to be raised in the plasma of COVID-19 patients admitted to the hospital and requiring intensive care in the clinical research (Costela-Ruiz et al. 2020; Huang et al. 2020). Moreover, pro-inflammatory TNF-α and IL-6 block desaturases, which are required to synthesize AA, EPA, and DHA from their substrates LA and ALA, accordingly. In addition, during COVID-19 infection, there is a higher level of inflammation and overexpression of inflammatory TNF-α and IL-6 that result in the shortage of AA, EPA, and DHA at the cellular level. As a result, anti-inflammatory mediators such as lipoxins, resolvins, protectins, and maresins are synthesized at a lower rate. In this case, a diet or supplement containing PUFAs can compensate for the shortage and aid in resolving inflammation while also preventing cytokine storms in severe COVID-19 (Das 2019).
Role in the Immune System
PUFAs play a critical part in the immune system’s function. Various saturated and unsaturated fatty acids provide fuel for energy production in the immune cells. ω-6 PUFAs, like AA, are one of the most critical components of the cell membrane phospholipids supporting physical and biochemical functions. In another way, PUFAs have potential in the genetic signaling process, thus regulating cellular functions. Various PUFAs like AA, DHA, and EPA serve as a precursor for synthesizing various functional eicosanoids like PGs, LTs, lipoxins, resolvins, and proteins. As a result, changes in the PUFA composition in the membrane phospholipid structure through supplementation or any other stimuli can change the immune system function (Calder 2001; Innes and Calder 2018).
Immune cell phospholipids such as lymph nodes or splenic lymphocytes contain the highest (15–20%) concentration of AA as fatty acid; for this reason, this ω-6 PUFA is considered as the primary precursor for eicosanoids which synthesis accelerated due to external stimuli like microbial infection or cellular injury (Calder et al. 1990, 1994). Among various AA-mediated inflammatory eicosanoids, PGE2 and LTB4 are predominant. The synthesis of AA-mediated inflammatory eicosanoid type depends on inflammatory cells (PGE2 synthesis is predominant in monocyte cells where PGD2 is predominant in mast cells) and the nature of stimuli. The incorporation of increased levels of EPA and DHA in cell membranes has been well-documented to reduce the production of AA-derived eicosanoid mediators such as PGE2. Peterson et al. (1998) suggested that EPA- and DHA-enriched diet to rat modified the level of AA content in the immune cell membrane phospholipids along with its predominant precursor PGE2 production. It is partially due to the reduced availability of AA as a substrate material and the inhibition of metabolic processes of AA. In contrast, oral administration of AA at a dose of 1.5 g/day to healthy humans raised the synthesis of PGE2 and LTB4 by peripheral blood mononuclear cells (Kelley et al. 1998). As EPA acts as a substrate for COX and LOX enzymes, if EPA can be incorporated into the membrane phospholipids, EPA-mediated eicosanoids (5 series LTs and PGE3) synthesis will be stimulated in immune cells. Research evidence supports this hypothesis, as Grimm et al. (2006) and Wachtler et al. (1997) suggested that the ω-3-enriched fish oil supplementation increases the production of 5 series LTs from neutrophils.
The fundamental step of the immune response against a pathogen or foreign substance is phagocytosis. This process involves the binding, in the end, a phagocytic vacuole of the pathogen or supplementary or antibody-coated pathogens on the surface receptor and subsequent invagination of the cell membrane around the pathogen. Phagocytosis also initiates acquired immunity in response to microorganisms. In addition, the level of PUFA content in the phagocytic cell membrane has a substantial role in the immune cells’ phagocytic capacity. Several in vitro and in vivo research approaches support this statement. It is suggested that the uptake of the foreign pathogen by macrophage is highly influenced by the uptake and incorporation of saturated and unsaturated fatty acid (Calder et al. 1990). The higher the uptake of PUFAs, the higher the increased in the phagocytic activity by phagocytic cells. A study conducted with human peripheral blood mononuclear cells from healthy volunteers suggested that about 80% of total neutrophils and 25% of monocytes are active. The phagocytic activity of those cells was highly correlated with the PUFA composition in membrane phospholipid. Both neutrophils and monocytes’ phagocytic activity were positively correlated with the total PUFA content, total ω-6 PUFAs, and total ω-3 PUFAs but negatively correlated with a palmitic acid content and the saturated to PUFAs ratio (Samantha Kew et al. 2003a, b). As there is a negative correlation between the phagocytic activity of the immune cell and the ω-6 to ω-3 fatty acid ratio, it can be considered that the ω-3 PUFAs have the potential to improve phagocytic activity. The outcome of clinical research on healthy volunteers also supports this, claiming that administering a mixture of ω-3 PUFAs (DHA + EPA) at a dose of 1.5 g/day improves about 40% of neutrophil and 200% of monocyte phagocytic activity (Kew et al. 2003a, b).
T cell functioning and signaling are also highly influenced by PUFAs. The underlying mechanisms behind this are (i) changes in the level of inflammatory cytokine synthesis, which influences T cell functioning; (ii) alteration in the plasma membranes’ physical structure; (iii) alteration of genetic signaling; and (iv) influence on lipid raft (Miles et al. 2003; Zeyda and Stulnig 2006). In vitro study suggested that supplementation with fish oil alters the phosphorylation of phospholipase C-λ1, a primary signaling event of T cell (Sanderson and Calder 1998). Another study on mice also demonstrated the reduced proliferation of T cell and IL-6 production, but the extent to which the human T cell lipid raft is modulated by PUFA supplementation is not studied yet (Fan et al. 2004).
An antigen-presenting cell (APC) is a cell that expresses antigen on its membrane surface via major histocompatibility complex (MHC) proteins, and this complex is recognized by T lymphocyte cells. MHC-1 and MHC-2 are involved in the presentation of pathogen-mediated antigens to T cells, resulting in the death of APCs. An in vitro study by Calder et al. (2007) suggested that antigen presentation via MHC II and expression are reduced when APCs are exposed to EPA or DHA. Sanderson et al. (1997) found a decreased expression of dendritic cells MHC-2 in fish oil-treated rats, which correlates with decreased antigen presentation by T cells. Another in vitro study examined the effect of PUFAs (AA, DHA) on MHC-1 expression and concluded that exposure to PUFAs decreases MHC-1 expression and that the effect is concentration sensitive (Shaikh and Edidin 2007). These findings of the action of AA and DHA in inhibiting MHC-1 expression provide insight into the mechanism by which PUFAs reduce antigen presentation; however, EPA has not been studied.
Role in Platelet Aggregation
PUFAs have a substantial influence on platelet structure and function. Supplementation with ω-3 and ω-6 PUFAs may alter the plasma membrane’s phospholipid composition. As a result, the platelet clotting or aggregation function may be diminished, which benefits patients with coagulopathy or cardiovascular complications. A review of in vivo and in vitro studies on the role of different PUFAs on platelet functions by Adili et al. (2018) agreed with the above statement. This is due to the action of PUFAs on the metabolizing enzyme COX-1, 12-lipoxygenase (12-LOX), and CYPs in platelets that convert the fatty acids into oxylipin products. These oxylipin products regulate platelet aggregation and thrombosis function. Barre and Holub (1992) demonstrated that AA is the primary fatty acid component (13.5%) in platelets and synthesizes various PGs that promote the coagulation process. However, supplementation with ω-3 fatty acids like EPA or DHA can modulate the AA composition in the membrane phospholipids of platelets and decrease the AA-mediated coagulation function. Another 28-day study on healthy volunteers supplemented by EPA stated that platelet function was significantly reduced, but DPA has no such function (Park and Harris 2002).
Intravascular coagulation is commonly noticed in severe cases of COVID-19. A study with 183 COVID-19 patients revealed that a significant proportion did not recover from the infection and had significantly higher levels of D-dimer, prothrombin time, and fibrin degradation products and lower levels of anti-thrombin and fibrinogen than recovered patients (Tang et al. 2020). Again, in patients with COVID-19, acute cardiac injury is particularly prominent and is correlated with severe clinical outcomes (Huang et al. 2020). However, all the heart failure cases (23%) noticed in hospitalized COVID-19 patients were not pre-existing cardiomyopathy (Zhou et al. 2020). Intra-arterial coagulation or thrombosis may promote the development of acute myocarditis. Considering these issues, it has been recommended that anticoagulants (e.g., heparin) can be used actively in patients with severe COVID-19 patients (Tang et al. 2020).
In COVID-19 patients, a moderate dose of ω-3 fatty acid can also perform heparin-like functions. A study conducted by Wander and Patton (1991) supports the correlation between ω-3 fatty acid consumption and improvement of COVID-19 coagulopathy. They demonstrated that a moderate consumption of EPA-containing fish salmon + sablefish + dover sole = 200 g/day for 18 days by healthy volunteers had increased bleeding time, increased platelet EPA content, and decreased platelet aggregation. Although substantial clinical research has not yet been conducted regarding the effect of PUFA supplementation against thrombosis in severe COVID-19 cases, a positive effect is highly expected.
Role in COVID-19 Comorbidities
Comorbidities of infectious illness increase the chances and severity of infection. In COVID-19, chronic respiratory disease, cardiovascular diseases, hypertension, diabetes, malignancy, and kidney diseases are suggested as the most influential morbidities. A tremendous report on PUFAs demonstrated a positive impact on these morbidities, although some conveyed double standards. PUFAs have both anti-inflammatory and anti-oxidative effects. Therefore, it has been mentioned to improve chronic respiratory complications like COPD. Two cohort studies of 120,175 participants revealed that fish consumption was negatively linked to the risk of COPD. Nonetheless, the effect of fatty acids and ω-3 PUFAs was non-significant (Varraso et al. 2015). Another study demonstrated the impact of ω-3 fatty acids cigarette smokers against COPD and showed an inverse relation with the risk in a quantity-dependent fashion (Shahar et al. 1994). However, interestingly, as like as ω-3, epidemiological evidence of ω-6 PUFAs inconsistently reported COPD prevention by several studies (Hirayama et al. 2010; Varraso et al. 2015).
In the 1970s, the first Greenland Eskimos have reported a lower prevalence of cardiovascular diseases (CVD) than populations taking typical Western diets. The study drew a relation between fish consumption and cardio-protection that the presence of ω-3 PUFAs series in marine food prevents ischemic processes by decreased platelet aggregability (Dyerberg and Bang 1979). A recent umbrella review found a significant reduction of CVD risks by ω-3 PUFAs, with RRs (95% CIs) range from 0.89 (0.82–0.98) to 0.90 (0.85–0.96) (Chareonrungrueangchai et al. 2020). Another pooled estimate by a meta-analysis on ω-3 effects reduced coronary heart disease and myocardial infarction risks compared to the control group (Hoang and Kim 2020). A randomized control trial in the USA on 28,100 women testified that dietary ω-3 and ω-6 PUFS was not associated with the risk of hypertension (Wang et al. 2010). In contrast, a very recent study described the vasodilatory effects of DHA and EPA, which ultimately lessen blood pressure (Bercea et al. 2021).
Moreover, several studies reported that PUFAs could prevent diabetes and attenuate the oxidant stress in chemically induced diabetic animals (Suresh and Das 2003). The Asian population intake more fish and marine ω-3 PUFAs compared to the Western population. Therefore, Asian people are at less risk of diabetes than the western population, and the same finding is described by Rice Bradley (Bradley 2018). Despite a paucity of information from various communities, few studies failed to show the association of PUFAs with end-stage renal disease (Malhotra et al. 2016). In terms of malignancy, Donat-Vargas et al. (2017) reported that EPA-DHA intake reduces the 80% risk of malignant melanoma. In comparison, a meta-analysis on 57 cases reported non-significant and weak relations between ω-3 PUFAs and different types of malignancy (Lee et al. 2020).
Safety Considerations
The ω-3 and ω-6 PUFAs are essential nutrients that should be kept in the daily food calendar to prevent nutritional deficiencies. Though the optimal daily requirements of PUFAs remain unknown, very high intakes may carry a risk of adverse effects. Clinical interventions support the notion against ω-6 PUFAs that a large intake of the FAs may enhance in vivo lipid peroxidation. Nevertheless, the influence of a high ω-3 PUFA intake on lipid peroxidation has been uncertain (Eritsland 2000). The peroxidative breakdown results in groups of products such as cyclic peroxides, aldehydes, and ketones. These molecules are involved in the free radical generation, leading to chronic and lethal pathogenesis such as inflammation, atherosclerosis, and cancer (Spiteller 2005).
High intake of ω-3 PUFAs replaces arachidonic acid with eicosapentaenoic acid in platelet membranes and results in more thromboxanes and prostacyclins. It enhances the vasodilatory and anti-aggregatory profile, leading to prolonged cutaneous bleeding (Dyerberg et al. 1978). Hypothetically, excessive n-3 PUFA intake could weaken defensive mechanisms against infections, but so far, there is no such clinical data (Eritsland 2000). However, excessive intake of PUFAs may be associated with immunosuppression, disturbance in glucose homeostasis, and hyperlipidemia. Although well-documented in vitro and in vivo assays ascertain the potential benefits of ω-3 fatty acids to lessen COVID-19-related complications, the risk of high dose supplementation during SARS-CoV-2 infection must be well-investigated.
Sources, Supplements, and Dose of PUFAs
COVID-19 patients have been reported significant improvement after ω-3 PUFA administration in few studies. So, these FAs should be suggested as palliative medicine. Effective outcomes from the treatment arise few vital questions: what are the sources of ω-3 fatty acids, do they have any supplements, which route is efficient to effective delivery, and what dose should be taken?
Nature is the best repository of essential PUFAs. Each one can take adequate ω-3 and ω-6 PUFAs from foods derived from animals and plants. The professional bodies recommend fish consumption to meet the daily needs of ω-3 PUFAs for health benefits. However, vegetable oils also contain significant amounts of ω-3 FA. The following seed oils are presented in descending order of ω-3 PUFAs: linseed oil, flaxseed oil, pumpkin seed oil, canola oil, soybean oil, safflower oil, and sunflower oil (Yashodhara et al. 2009). Box 1 elucidates ω-3 and ω-6 PUFAs enriched foods along with a recommendation, should it eat or avoid. Olivieri’s recommendation mainly focused on avoiding cardiovascular disease but may apply to COVID-19 due to similar positive/negative impacts of the two types PUFAs (Olivieri 2019).
Box 1. PUFA-enriched common foods and suggestions base on relative components.
| Fatty acid types | ω-3 PUFAs (ALA, EPA, DHA) | ω-6 PUFAs (LA, AA) | ||
|---|---|---|---|---|
| Recommended | Avoid | Recommended | Avoid | |
| Long-chain: ALA, LA | Relatively more ALA: flax, walnuts, green leafy vegetables | Relatively low ALA: canola oil, soybean oil | - | Relatively more LA: sunflower oil, safflower oil, corn oil, cottonseed oil, soybean oil |
| Very-long-chain: EPA, DHA, AA | Relatively more EPA and DHA: mackerel, salmon, anchovies, sardines, tuna, herring, squid, shellfish, wild animal meats, grass-fed animal meats | - | Relatively low AA: eggs, poultry, beef, pork, liver, human breast milk | Relatively more AA: farm-raised fish, rain feed animal milk |
Besides these dietary sources, different supplements of ω-3 FA are now available in the market. Oral capsule and intravenous lipid injection emulsion are recommended for intensive care patients. Many patients with digestive intolerance can take lipid emulsions enriched with fish oil (Sabater et al. 2011). A study on 19 septic shock patients was designed with ten patients randomly assigned to receive a ω-3 FA emulsion (350 ml/day, equivalent to 14 g DHA + EPA) intravenously for 3 days. The emulsion achieved a ratio of 2.5:1 for ω-3 and ω-6 PUFAs and significantly decreased pro-inflammatory mediators TNF-α, IL-6, and IL-8 (Mayer et al. 2003). Different dosage forms with a convenient route of administration are involved in PUFA supplement to adjust the required amounts.
The ω-3 FA content of the diet must achieve nutritional adequacy to prevent deficiency symptoms. However, exact dose requirement is difficult to measure for few reasons: interindividual variability in ω-3 FA metabolism based on genetic determinants, age, and gender; dietary composition, high ω-6 PUFA, and saturated FAs interfere with the functions of ω-3; availability of certain foodstuffs, such as wild fish; and food contaminants (Molendi-Coste et al. 2011). An ideal balanced diet is deduced from ancestral nutrition, where fat should represent below 20–30% of the total energy uptake. Moreover, the fat portion should meet 5–6 g/day of EPA- + DHA-enriched ω-3 PUFA, and the ω-6-to-ω-3 ratio should average 1 (Sanders 2000). Consumption of cold water fish or specific enteral feeding formulas can provide insufficient EPA and DHA (nearly 1 g/day). Nevertheless, Bistrian (2020) suggested 4–6 g/day of EPA and DHA to manage a hyper-inflammatory state in critically COVID-19-infected patients. PUFAs, especially ω-3 FAs, are a potential medicine to provide palliative care in coronavirus infection. Therefore, proper investigation on dosage form, dose, and administration route of its supplements is required to ensure their safety and efficacy.
Perspectives and Future Directions
PUFAs are well-known for their constructive and beneficial impact on health. A sort of study on viral infection revealed the fats and viral interaction pathway to modulate immunochemistry. Therefore, PUFAs are promising molecules considered as curative and risk reductive medicine in COVID-19 treatment worldwide. Nevertheless, negligible studies on the fats against SARS-COV-2 have been conducted till now. Most of the investigations are hypothetical based on previous works on infectious diseases.
Moreover, the viral characteristics are very complex with the implicit mode of actions. That obviously asks for comprehensive studies about the sites of interaction with biomolecules and the extent of interference using PUFAs. COVID-19 is an obdurate contagion of this decade and has recently considered its parallel coexistence with time. And so, the potential subject should be concisely explored to reduce the risks of the infection. Future studies should be highly focused on immunological effects, hematological parameters, respiratory healing, and comorbid stress reductions that will make a thorough direction for disease management.
Conclusions
SARS-CoV-2 is a highly contagious virus which spread through respiratory droplets. Besides the respiratory system, multisystem and multi-organ involvement of the virus triggers a cytokine storm that is an indicator of disease severity. Polyunsaturated fatty acids, a cluster of clinically significant fats, are the considerable option to lessen the severity. It has a substantial role in the immunological defense against viral entry, localization, and replication to new copies. However, it is also associated with respiratory health by controlling blood rheology and surfactant production in the lung. To add more, PUFAs might significantly benefit COVID-19 comorbidities such as cardiovascular disease, COPD, and diabetes. However, all PUFAs are not functionally the same. The members of ω-3 PUFAs (ALA, EPA, and DHA) are associated with anti-inflammatory action, whereas ω-6 PUFAs (LA, AA) are pro-inflammatory. Exceptionally, few SPMs like lipoxin derived from the AA show anti-inflammatory action that might lessen COVID-19 severity. Therefore, the ω-3 PUFA-enriched food or supplements is a good option in the emergence cytokine race and might decrease COVID-19 complications. Further clinical and retrospective study for the use of PUFAs in COVID-19 management is warranted.
Author Contribution
PKB and MTA equally contributed to the design and draft of the research; MMR was associated with the conception and correction of the text; MSH contributed to the conception and design of the research. All authors critically revised the manuscript, agree to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript.
Footnotes
Prodip Kumar Baral and Mohammad Tohidul Amin contributed equally to this work.
References
- Adili R, Hawley M, Holinstat M. Regulation of platelet function and thrombosis by omega-3 and omega-6 polyunsaturated fatty acids. Prostag Oth Lipid M. 2018;139:10–18. doi: 10.1016/j.prostaglandins.2018.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agostoni C, Bruzzese MG. Fatty acids: their biochemical and functional classification. Pediatr Med Chir. 1992;14:473–479. [PubMed] [Google Scholar]
- Asher A, Tintle NL, Myers M, Lockshon L, Bacareza H, Harris WS. Blood omega-3 fatty acids and death from COVID-19: a pilot study. Prostag Leukot Ess. 2021;166:102250. doi: 10.1016/j.plefa.2021.102250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Auner B, Geiger EV, Henrich D, Lehnert M, Marzi I, Relja B. Circulating leukotriene B4 identifies respiratory complications after trauma. Mediators Inflamm. 2012;2012:536156. doi: 10.1155/2012/536156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ballout RA, Sviridov D, Bukrinsky MI, Remaley AT. The lysosome: a potential juncture between SARS-CoV-2 infectivity and Niemann-Pick disease type C, with therapeutic implications. FASEB J. 2020;34:7253–7264. doi: 10.1096/fj.202000654R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baral PK, Nuruzzaman M, Uddin MS, Ferdous M, Chowdhury IH, Smrity SZ. Severe acute respiratory coronavirus-2 invasion of the central nervous system: a host-virus deadlock. Acta Virol. 2021;65:115–126. doi: 10.4149/av_2021_201. [DOI] [PubMed] [Google Scholar]
- Barceló-Coblijn G, Murphy EJ. Alpha-linolenic acid and its conversion to longer chain n-3 fatty acids: benefits for human health and a role in maintaining tissue n-3 fatty acid levels. Prog Lipid Res. 2009;48:355–374. doi: 10.1016/j.plipres.2009.07.002. [DOI] [PubMed] [Google Scholar]
- Barre DE, Holub BJ. The effect of borage oil consumption on the composition of individual phospholipids in human platelets. Lipids. 1992;27:315–320. doi: 10.1007/BF02536143. [DOI] [PubMed] [Google Scholar]
- Basil MC, Levy BD. Specialized pro-resolving mediators: endogenous regulators of infection and inflammation. Nat Rev Immunol. 2016;16:51–67. doi: 10.1038/nri.2015.4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bazan NG. Neuroprotectin D1 (NPD1): a DHA-derived mediator that protects brain and retina against cell injury-induced oxidative stress. Brain Pathol. 2005;15:159–166. doi: 10.1111/j.1750-3639.2005.tb00513.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bazan NG. Neuroprotectin D1-mediated anti-inflammatory and survival signaling in stroke, retinal degenerations, and Alzheimer’s disease. J Lipid Res. 2009;50:S400–S405. doi: 10.1194/jlr.R800068-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bercea C, Cottrell GS, Tamagnini F, McNeish AJ. Omega-3 polyunsaturated fatty acids and hypertension: a review of vasodilatory mechanisms of docosahexaenoic acid and eicosapentaenoic acid. Br J Pharmacol. 2021;178:860–877. doi: 10.1111/bph.15336. [DOI] [PubMed] [Google Scholar]
- Bistrian BR. Parenteral fish-oil emulsions in critically Ill COVID-19 emulsions. JPEN-Parenter Enter. 2020;44:1168–1168. doi: 10.1002/jpen.1871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley BHR. Dietary fat and risk for type 2 diabetes: a review of recent research. Curr Nutr Rep. 2018;7:214–226. doi: 10.1007/s13668-018-0244-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burdge GC. Polyunsaturated fatty acid intakes and α-linolenic acid metabolism. Am J Clin Nutr. 2011;93:665–666. doi: 10.3945/ajcn.110.008169. [DOI] [PubMed] [Google Scholar]
- Calder PC. Polyunsaturated fatty acids, inflammation, and immunity. Lipids. 2001;36:1007–1024. doi: 10.1007/s11745-001-0812-7. [DOI] [PubMed] [Google Scholar]
- Calder PC. Long-chain n-3 fatty acids and inflammation: potential application in surgical and trauma patients. Braz J Med Biol Res. 2003;36:433–446. doi: 10.1590/s0100-879x2003000400004. [DOI] [PubMed] [Google Scholar]
- Calder PC. Polyunsaturated fatty acids alter the rules of engagement. Future Lipidol. 2007;2:27–30. doi: 10.2217/17460875.2.1.27. [DOI] [Google Scholar]
- Calder PC. Fatty acids and inflammation: the cutting edge between food and pharma. Eur J Pharmacol. 2011;668:S50–S58. doi: 10.1016/j.ejphar.2011.05.085. [DOI] [PubMed] [Google Scholar]
- Calder PC. Long chain fatty acids and gene expression in inflammation and immunity. Curr Opin Clin Nutr Metab Care. 2013;16:425–433. doi: 10.1097/MCO.0b013e3283620616. [DOI] [PubMed] [Google Scholar]
- Calder PC, Bond JA, Harvey DJ, Gordon S, Newsholme EA. Uptake and incorporation of saturated and unsaturated fatty acids into macrophage lipids and their effect upon macrophage adhesion and phagocytosis. Biochem J. 1990;269:807–814. doi: 10.1042/bj2690807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calder PC, Yaqoob P, Harvey DJ, Watts A, Newsholme EA. Incorporation of fatty acids by concanavalin A-stimulated lymphocytes and the effect on fatty acid composition and membrane fluidity. Biochem J. 1994;300:509–518. doi: 10.1042/bj3000509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calder PC, Carr AC, Gombart AF, Eggersdorfer M. Optimal nutritional status for a well-functioning immune system is an important factor to protect against viral infections. Nutrients. 2020;12:1181. doi: 10.3390/nu12041181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chareonrungrueangchai K, Wongkawinwoot K, Anothaisintawee T, Reutrakul S. Dietary factors and risks of cardiovascular diseases: an umbrella review. Nutrients. 2020;12:1088. doi: 10.3390/nu12041088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costela-Ruiz VJ, Illescas-Montes R, Puerta-Puerta JM, Ruiz C, Melguizo-Rodríguez L. SARS-CoV-2 infection: the role of cytokines in COVID-19 disease. Cytokine Growth F R. 2020;54:62–75. doi: 10.1016/j.cytogfr.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Damsgaard CT, Lauritzen L, Kjaer TMR, Holm PMI, Fruekilde M-B, Michaelsen KF, Frøkiaer H. Fish oil supplementation modulates immune function in healthy infants. J Nutr. 2007;137:1031–1036. doi: 10.1093/jn/137.4.1031. [DOI] [PubMed] [Google Scholar]
- Das UN. Can bioactive lipids augment anti-cancer action of immunotherapy and prevent cytokine storm? Arch Med Res. 2019;50:342–349. doi: 10.1016/j.arcmed.2019.10.004. [DOI] [PubMed] [Google Scholar]
- Das UN. Can bioactive lipids inactivate coronavirus (COVID-19)? Arch Med Res. 2020;51:282–286. doi: 10.1016/j.arcmed.2020.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Finger JAFF, Lima EMF, Coelho KS, Behrens JH, Landgraf M, Franco BDGM, Pinto UM. Adherence to food hygiene and personal protection recommendations for prevention of COVID-19. Trends Food Sci Technol. 2021;112:847–852. doi: 10.1016/j.tifs.2021.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Souza DR, Pieri BLdaS, Comim VH, Marques SdeO, Luciano TF, Rodrigues MS, De Souza CT. Fish oil reduces subclinical inflammation, insulin resistance, and atherogenic factors in overweight/obese type 2 diabetes mellitus patients: a pre-post pilot study. J Diabetes Complicat. 2020;34:107553. doi: 10.1016/j.jdiacomp.2020.107553. [DOI] [PubMed] [Google Scholar]
- Deckelbaum RJ, Worgall TS, Seo T. n-3 Fatty acids and gene expression. Am J Clin Nutr. 2006;83:1520S–1525S. doi: 10.1093/ajcn/83.6.1520S. [DOI] [PubMed] [Google Scholar]
- Donat-Vargas C, Berglund M, Glynn A, Wolk A, Åkesson A. Dietary polychlorinated biphenyls, long-chain n-3 polyunsaturated fatty acids and incidence of malignant melanoma. Eur J Cancer. 2017;72:137–143. doi: 10.1016/j.ejca.2016.11.016. [DOI] [PubMed] [Google Scholar]
- Duffield JS, Hong S, Vaidya VS, Lu Y, Fredman G, Serhan CN, Bonventre JV. Resolvin D series and protectin D1 mitigate acute kidney injury. J Immunol. 2006;177:5902–5911. doi: 10.4049/jimmunol.177.9.5902. [DOI] [PubMed] [Google Scholar]
- Dyerberg J, Bang HO. Haemostatic function and platelet polyunsaturated fatty acids in Eskimos. Lancet. 1979;2:433–435. doi: 10.1016/s0140-6736(79)91490-9. [DOI] [PubMed] [Google Scholar]
- Dyerberg J, Bang HO, Stoffersen E, Moncada S, Vane JR. Eicosapentaenoic acid and prevention of thrombosis and atherosclerosis? Lancet. 1978;2:117–119. doi: 10.1016/s0140-6736(78)91505-2. [DOI] [PubMed] [Google Scholar]
- Eritsland J. Safety considerations of polyunsaturated fatty acids. Am J Clin Nutr. 2000;71:197S–201S. doi: 10.1093/ajcn/71.1.197S. [DOI] [PubMed] [Google Scholar]
- Fan Y-Y, Ly LH, Barhoumi R, McMurray DN, Chapkin RS. Dietary docosahexaenoic acid suppresses T cell protein kinase C theta lipid raft recruitment and IL-2 production. J Immunol. 2004;173:6151–6160. doi: 10.4049/jimmunol.173.10.6151. [DOI] [PubMed] [Google Scholar]
- Gaudin A, Tolar M, Peters OA. Lipoxin A4 attenuates the inflammatory response in stem cells of the apical papilla via ALX/FPR2. Sci Rep. 2018;8:8921. doi: 10.1038/s41598-018-27194-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerlach BD, Marinello M, Heinz J, Rymut N, Sansbury BE, Riley CO, Sadhu S, Hosseini Z, Kojima Y, Tang DD, Leeper NJ. Resolvin D1 promotes the targeting and clearance of necroptotic cells. Cell Death Differ. 2020;27:525–539. doi: 10.1038/s41418-019-0370-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerling CJ, Mukai K, Chabowski A, Heigenhauser GJF, Holloway GP, Spriet LL, Jannas-Vela S. Incorporation of omega-3 fatty acids into human skeletal muscle sarcolemmal and mitochondrial membranes following 12 weeks of fish oil supplementation. Front Physiol. 2019;10:348. doi: 10.3389/fphys.2019.00348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glende J, Schwegmann-Wessels C, Al-Falah M, Pfefferle S, Qu X, Deng H, Drosten C, Naim HY, Herrler G. Importance of cholesterol-rich membrane microdomains in the interaction of the S protein of SARS-coronavirus with the cellular receptor angiotensin-converting enzyme 2. Virology. 2008;381:215–221. doi: 10.1016/j.virol.2008.08.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimm H, Mertes N, Goeters C, Schlotzer E, Mayer K, Grimminger F, Fürst P. Improved fatty acid and leukotriene pattern with a novel lipid emulsion in surgical patients. Eur J Nutr. 2006;45:55–60. doi: 10.1007/s00394-005-0573-8. [DOI] [PubMed] [Google Scholar]
- Hirayama F, Lee AH, Binns CW, Hiramatsu N, Mori M, Nishimura K. Dietary intake of isoflavones and polyunsaturated fatty acids associated with lung function, breathlessness and the prevalence of chronic obstructive pulmonary disease: possible protective effect of traditional Japanese diet. Mol Nutr Food Res. 2010;54:909–917. doi: 10.1002/mnfr.200900316. [DOI] [PubMed] [Google Scholar]
- Hoang T, Kim J. Comparative effect of statins and omega-3 supplementation on cardiovascular events: meta-analysis and network meta-analysis of 63 randomized controlled trials including 264,516 participants. Nutrients. 2020;12:2218. doi: 10.3390/nu12082218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Innes JK, Calder PC. Omega-6 fatty acids and inflammation. Prostaglandins Leukot Ess. 2018;132:41–48. doi: 10.1016/j.plefa.2018.03.004. [DOI] [PubMed] [Google Scholar]
- Innes JK, Calder PC. Marine omega-3 (N-3) fatty acids for cardiovascular health: an update for 2020. Int J Mol Sci. 2020;21:1362. doi: 10.3390/ijms21041362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson JH, Bonds JM, Parnell AM, Bright CM. Coronavirus vaccine distribution: moving to a race conscious approach for a racially disparate problem. J Racial Ethn Heal Disparities. 2021;8:799–802. doi: 10.1007/s40615-021-01051-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaneko JJ, Harvey JW, Bruss ML (2008) Clinical biochemistry of domestic animals, 6th edn, Chapter 4. Academic press, San Diego, pp 81–116
- Kelley DS, Taylor PC, Nelson GJ, Mackey BE. Arachidonic acid supplementation enhances synthesis of eicosanoids without suppressing immune functions in young healthy men. Lipids. 1998;33:125–130. doi: 10.1007/s11745-998-0187-9. [DOI] [PubMed] [Google Scholar]
- Kew S, Banerjee T, Minihane AM, Finnegan YE, Muggli R, Albers R, Williams CM, Calder PC. Lack of effect of foods enriched with plant- or marine-derived n-3 fatty acids on human immune function. Am J Clin Nutr. 2003;77:1287–1295. doi: 10.1093/ajcn/77.5.1287. [DOI] [PubMed] [Google Scholar]
- Kew S, Banerjee T, Minihane AM, Finnegan YE, Williams CM, Calder PC. Relation between the fatty acid composition of peripheral blood mononuclear cells and measures of immune cell function in healthy, free-living subjects aged 25–72 y. Am J Clin Nutr. 2003;77:1278–1286. doi: 10.1093/ajcn/77.5.1278. [DOI] [PubMed] [Google Scholar]
- Knapp HR. Omega-3 fatty acids in respiratory diseases: a review. J Am Coll Nutr. 1995;14:18–23. doi: 10.1080/07315724.1995.10718468. [DOI] [PubMed] [Google Scholar]
- Körner A, Schlegel M, Theurer J, Frohnmeyer H, Adolph M, Heijink M, Giera M, Rosenberger P, Mirakaj V. Resolution of inflammation and sepsis survival are improved by dietary Ω-3 fatty acids. Cell Death Differ. 2018;25:421–431. doi: 10.1038/cdd.2017.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee CH. Role of specialized pro-resolving lipid mediators and their receptors in virus infection: a promising therapeutic strategy for SARS-CoV-2 cytokine storm. Arch Pharm Res. 2021;44:84–98. doi: 10.1007/s12272-020-01299-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee KH, Seong HJ, Kim G, Jeong GH, Kim JY, Park H, Jung E, Kronbichler A, Eisenhut M, Stubbs B. Consumption of fish and ω-3 fatty acids and cancer risk: an umbrella review of meta-analyses of observational studies. Adv Nutr. 2020;11:1134–1149. doi: 10.1093/advances/nmaa055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemoine C, Brigham E, Woo H, Koch A, Hanson C, Romero K, Putcha N, McCormack M, Hansel N. Relationship between omega-3 and omega-6 fatty acid intake and chronic obstructive pulmonary disease morbidity. AnnalsATS. 2020;17:378–381. doi: 10.1513/AnnalsATS.201910-740RL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy BD, Clish CB, Schmidt B, Gronert K, Serhan CN. Lipid mediator class switching during acute inflammation: signals in resolution. Nat Immunol. 2001;2:612–619. doi: 10.1038/89759. [DOI] [PubMed] [Google Scholar]
- Li QF, Hao H, Tu WS, Guo N, Zhou XY. Maresins: anti-inflammatory pro-resolving mediators with therapeutic potential. Eur Rev Med Pharmacol Sci. 2020;24:7442–7453. doi: 10.26355/eurrev_202007_21913. [DOI] [PubMed] [Google Scholar]
- Lu Y, Liu DX, Tam JP. Lipid rafts are involved in SARS-CoV entry into Vero E6 cells. Biochem Biophys Res Commun. 2008;369:344–349. doi: 10.1016/j.bbrc.2008.02.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malhotra R, Cavanaugh KL, Blot WJ, Ikizler TA, Lipworth L, Kabagambe EK. Dietary polyunsaturated fatty acids and incidence of end-stage renal disease in the Southern Community Cohort Study. BMC Nephrol. 2016;17:152. doi: 10.1186/s12882-016-0371-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masclans JR, Bermejo B, Picó M, de Latorre FJ, Rodríguez-Roisin R, Planas M. The prognostic value of eicosanoids in the acute respiratory distress syndrome. Med Clin. 1999;112:81–84. [PubMed] [Google Scholar]
- Masclans JR, Sabater J, Sacanell J, Chacon P, Sabin P, Roca O, Planas M. Possible prognostic value of leukotriene B(4) in acute respiratory distress syndrome. Respir Care. 2007;52:1695–1700. [PubMed] [Google Scholar]
- Matheson NJ, Sumner J, Wals K, Rapiteanu R, Weekes MP, Vigan R, Weinelt J, Schindler M, Antrobus R, Costa ASH, Frezza C, Clish CB, Neil SJD, Lehner PJ. Cell surface proteomic map of HIV infection reveals antagonism of amino acid metabolism by Vpu and Nef. Cell Host Microbe. 2015;18:409–423. doi: 10.1016/j.chom.2015.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayer K, Gokorsch S, Fegbeutel C, Hattar K, Rosseau S, Walmrath D, Seeger W, Grimminger F. Parenteral nutrition with fish oil modulates cytokine response in patients with sepsis. Am J Resp Crit Care. 2003;167:1321–1328. doi: 10.1164/rccm.200207-674OC. [DOI] [PubMed] [Google Scholar]
- Mickleborough TD, Lindley MR, Ionescu AA, Fly AD. Protective effect of fish oil supplementation on exercise-induced bronchoconstriction in asthma. Chest. 2006;129:39–49. doi: 10.1378/chest.129.1.39. [DOI] [PubMed] [Google Scholar]
- Mihrshahi S, Peat JK, Webb K, Oddy W, Marks GB, Mellis CM. Effect of omega-3 fatty acid concentrations in plasma on symptoms of asthma at 18 months of age. Pediatr Allergy Immu. 2004;15:517–522. doi: 10.1111/j.1399-3038.2004.00187.x. [DOI] [PubMed] [Google Scholar]
- Miles EA, Aston L, Calder PC. In vitro effects of eicosanoids derived from different 20-carbon fatty acids on T helper type 1 and T helper type 2 cytokine production in human whole-blood cultures. Clin Exp Allergy. 2003;33:624–632. doi: 10.1046/j.1365-2222.2003.01637.x. [DOI] [PubMed] [Google Scholar]
- Miyata J, Arita M. Role of omega-3 fatty acids and their metabolites in asthma and allergic diseases. Allergol Int. 2015;64:27–34. doi: 10.1016/j.alit.2014.08.003. [DOI] [PubMed] [Google Scholar]
- Molendi-Coste O, Legry V, Leclercq IA. Why and how meet n-3 PUFA dietary recommendations? Gastroenterol Res Pract. 2011;2011:364040. doi: 10.1155/2011/364040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morin C, Hiram R, Rousseau E, Blier PU, Fortin S. Docosapentaenoic acid monoacylglyceride reduces inflammation and vascular remodeling in experimental pulmonary hypertension. Am J Physiol Heart C. 2014;307:H574–H586. doi: 10.1152/ajpheart.00814.2013. [DOI] [PubMed] [Google Scholar]
- Olivieri C. The current state of heart disease: statins, cholesterol, fat and sugar. Int J Evid Based Healthc. 2019;17:179–186. doi: 10.1097/XEB.0000000000000197. [DOI] [PubMed] [Google Scholar]
- Park Y, Harris W. EPA, but not DHA, decreases mean platelet volume in normal subjects. Lipids. 2002;37:941–946. doi: 10.1007/s11745-006-0984-1. [DOI] [PubMed] [Google Scholar]
- Park J, Langmead CJ, Riddy DM. New advances in targeting the resolution of inflammation: implications for specialized pro-resolving mediator GPCR drug discovery. ACS Pharmacol Transl Sci. 2020;3:88–106. doi: 10.1021/acsptsci.9b00075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patterson E, Wall R, Fitzgerald GF, Ross RP, Stanton C. Health implications of high dietary omega-6 polyunsaturated fatty acids. J Nutr Metab. 2012;2012:539426. doi: 10.1155/2012/539426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson LD, Jeffery NM, Thies F, Sanderson P, Newsholme EA, Calder PC. Eicosapentaenoic and docosahexaenoic acids alter rat spleen leukocyte fatty acid composition and prostaglandin E2 production but have different effects on lymphocyte functions and cell-mediated immunity. Lipids. 1998;33:171–180. doi: 10.1007/s11745-998-0193-y. [DOI] [PubMed] [Google Scholar]
- Pham TL, Kakazu AH, He J, Jun B, Bazan NG, Bazan HEP. Novel RvD6 stereoisomer induces corneal nerve regeneration and wound healing post-injury by modulating trigeminal transcriptomic signature. Sci Rep. 2020;10:4582. doi: 10.1038/s41598-020-61390-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pombo JP, Sanyal S. Perturbation of intracellular cholesterol and fatty acid homeostasis during flavivirus infections. Front Immunol. 2018;9:1276. doi: 10.3389/fimmu.2018.01276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ripon MAR, Bhowmik DR, Amin MT, Hossain MS. Role of arachidonic cascade in COVID-19 infection: a review. Prostaglandins Oth Lipid m. 2021;154:106539. doi: 10.1016/j.prostaglandins.2021.106539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rius B, López-Vicario C, González-Périz A, Morán-Salvador E, García-Alonso V, Clária J, Titos E. Resolution of inflammation in obesity-induced liver disease. Front Immunol. 2012;3:257. doi: 10.3389/fimmu.2012.00257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson JG, Stone NJ. Antiatherosclerotic and antithrombotic effects of omega-3 fatty acids. Am J Cardiol. 2006;98:39i–49i. doi: 10.1016/j.amjcard.2005.12.026. [DOI] [PubMed] [Google Scholar]
- Sabater J, Masclans JR, Sacanell J, Chacon P, Sabin P, Planas M. Effects of an omega-3 fatty acid-enriched lipid emulsion on eicosanoid synthesis in acute respiratory distress syndrome (ARDS): a prospective, randomized, double-blind, parallel group study. Nutr Metab. 2011;8:22. doi: 10.1186/1743-7075-8-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saedisomeolia A, Wood LG, Garg ML, Gibson PG, Wark PAB. Anti-inflammatory effects of long-chain n-3 PUFA in rhinovirus-infected cultured airway epithelial cells. Br J Nutr. 2009;101:533–540. doi: 10.1017/S0007114508025798. [DOI] [PubMed] [Google Scholar]
- Saifullah A, Watkins BA, Saha C, Li Y, Moe SM, Friedman AN. Oral fish oil supplementation raises blood omega-3 levels and lowers C-reactive protein in haemodialysis patients–a pilot study. Nephrol Dial Transpl. 2007;22:3561–3567. doi: 10.1093/ndt/gfm422. [DOI] [PubMed] [Google Scholar]
- Saini RK, Keum Y-S. Omega-3 and omega-6 polyunsaturated fatty acids: dietary sources, metabolism, and significance - a review. Life Sci. 2018;203:255–267. doi: 10.1016/j.lfs.2018.04.049. [DOI] [PubMed] [Google Scholar]
- Sanders TA. Polyunsaturated fatty acids in the food chain in Europe. Am J Clin Nutr. 2000;71:176S–178S. doi: 10.1093/ajcn/71.1.176s. [DOI] [PubMed] [Google Scholar]
- Sanderson P, Calder PC. Dietary fish oil appears to prevent the activation of phospholipase C-gamma in lymphocytes. Biochim Biophys Acta. 1998;1392:300–308. doi: 10.1016/s0005-2760(98)00044-7. [DOI] [PubMed] [Google Scholar]
- Sanderson P, MacPherson GG, Jenkins C, Calder PC. Dietary fish oil diminishes the antigen presentation activity of rat dendritic cells. J Leukoc Biol. 1997;62:771–777. doi: 10.1002/jlb.62.6.771. [DOI] [PubMed] [Google Scholar]
- Schmitz G, Ecker J. The opposing effects of n-3 and n-6 fatty acids. Prog Lipid Res. 2008;47:147–155. doi: 10.1016/j.plipres.2007.12.004. [DOI] [PubMed] [Google Scholar]
- Schönfeld P, Wojtczak L. Short-and medium-chain fatty acids in energy metabolism: the cellular perspective. J Lipid Res. 2016;57:943–954. doi: 10.1194/jlr.R067629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schubert R, Kitz R, Beermann C, Rose MA, Lieb A, Sommerer PC, Moskovits J, Alberternst H, Böhles HJ, Schulze J, Zielen S. Effect of n-3 polyunsaturated fatty acids in asthma after low-dose allergen challenge. Int Arch Allergy Immunol. 2009;148:321–329. doi: 10.1159/000170386. [DOI] [PubMed] [Google Scholar]
- Serhan CN, Jain A, Marleau S, Clish C, Kantarci A, Behbehani B, Colgan SP, Stahl GL, Merched A, Petasis NA, Chan L, Van Dyke TE. Reduced inflammation and tissue damage in transgenic rabbits overexpressing 15-lipoxygenase and endogenous anti-inflammatory lipid mediators. J Immunol. 2003;171:6856–6865. doi: 10.4049/jimmunol.171.12.6856. [DOI] [PubMed] [Google Scholar]
- Shahar E, Folsom AR, Melnick SL, Tockman MS, Comstock GW, Gennaro V, Higgins MW, Sorlie PD, Ko W-J, Szklo M. Dietary n-3 polyunsaturated fatty acids and smoking-related chronic obstructive pulmonary disease. N Engl J Med. 1994;331:228–233. doi: 10.1056/NEJM199407283310403. [DOI] [PubMed] [Google Scholar]
- Shaikh SR, Edidin M. Immunosuppressive effects of polyunsaturated fatty acids on antigen presentation by human leukocyte antigen class I molecules. J Lipid Res. 2007;48:127–138. doi: 10.1194/jlr.M600365-JLR200. [DOI] [PubMed] [Google Scholar]
- Sheppard KW, Cheatham CL. Omega-6/omega-3 fatty acid intake of children and older adults in the U.S.: dietary intake in comparison to current dietary recommendations and the Healthy Eating Index. Lipids Health Dis. 2018;17:43. doi: 10.1186/s12944-018-0693-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simopoulos AP, Leaf A, Salem NJ. Essentiality of and recommended dietary intakes for omega-6 and omega-3 fatty acids. Ann Nutr Metab. 1999;43:127–130. doi: 10.1159/000012777. [DOI] [PubMed] [Google Scholar]
- Sokoła-Wysoczańska E, Wysoczański T, Wagner J, Czyż K, Bodkowski R, Lochyński S, Patkowska-Sokoła B. Polyunsaturated fatty acids and their potential therapeutic role in cardiovascular system disorders—a review. Nutrients. 2018;10:1561. doi: 10.3390/nu10101561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spite M, Norling LV, Summers L, Yang R, Cooper D, Petasis NA, Flower RJ, Perretti M, Serhan CN. Resolvin D2 is a potent regulator of leukocytes and controls microbial sepsis. Nature. 2009;461:1287–1291. doi: 10.1038/nature08541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiteller G. The relation of lipid peroxidation processes with atherogenesis: a new theory on atherogenesis. Mol Nutr Food Res. 2005;49:999–1013. doi: 10.1002/mnfr.200500055. [DOI] [PubMed] [Google Scholar]
- Suresh Y, Das UN. Long-chain polyunsaturated fatty acids and chemically induced diabetes mellitus: effect of ω-3 fatty acids. Nutrition. 2003;19:213–228. doi: 10.1016/S0899-9007(02)00855-9. [DOI] [PubMed] [Google Scholar]
- Tang S, Wan M, Huang W, Stanton RC, Xu Y. Maresins: a specialized proresolving lipid mediators and their potential role in inflammatory-related diseases. Mediators Inflamm. 2018;2018:2380319. doi: 10.1155/2018/2380319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang N, Bai H, Chen X, Gong J, Li D, Sun Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 2020;18:1094–1099. doi: 10.1111/jth.14817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varraso R, Barr RG, Willett WC, Speizer FE, Camargo CA., Jr Fish intake and risk of chronic obstructive pulmonary disease in 2 large US cohorts. Am J Clin Nutr. 2015;101:354–361. doi: 10.3945/ajcn.114.094516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wachtler P, König W, Senkal M, Kemen M, Köller M. Influence of a total parenteral nutrition enriched with omega-3 fatty acids on leukotriene synthesis of peripheral leukocytes and systemic cytokine levels in patients with major surgery. J Trauma. 1997;42:191–198. doi: 10.1097/00005373-199702000-00004. [DOI] [PubMed] [Google Scholar]
- Wander RC, Patton BD. Comparison of three species of fish consumed as part of a Western diet: effects on platelet fatty acids and function, hemostasis, and production of thromboxane. Am J Clin Nutr. 1991;54:326–333. doi: 10.1093/ajcn/54.2.326. [DOI] [PubMed] [Google Scholar]
- Wang L, Manson JE, Forman JP, Gaziano JM, Buring JE, Sesso HD. Dietary fatty acids and the risk of hypertension in middle-aged and older women. Hypertension. 2010;56:598–604. doi: 10.1161/HYPERTENSIONAHA.110.154187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C-W, Colas RA, Dalli J, Arnardottir HH, Nguyen D, Hasturk H, Chiang N, Van Dyke TE, Serhan CN. Maresin 1 biosynthesis and proresolving anti-infective functions with human-localized aggressive periodontitis leukocytes. Infect Immun. 2015;84:658–665. doi: 10.1128/IAI.01131-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wassall SR, Leng X, Canner SW, Pennington ER, Kinnun JJ, Cavazos AT, Dadoo S, Johnson D, Heberle FA, Katsaras J, Shaikh SR. Docosahexaenoic acid regulates the formation of lipid rafts: a unified view from experiment and simulation. Biochim Biophys Acta Biomembr. 2018;1860:1985–1993. doi: 10.1016/j.bbamem.2018.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WHO (2020) Novel coronavirus (2019-nCoV): situation report, 11. World Health Organization. https://apps.who.int/iris/handle/10665/330776. Accessed May 22, 2021
- Wong JMW, De Souza R, Kendall CWC, Emam A, Jenkins DJA. Colonic health: fermentation and short chain fatty acids. J Clin Gastroenterol. 2006;40:235–243. doi: 10.1097/00004836-200603000-00015. [DOI] [PubMed] [Google Scholar]
- Yashodhara BM, Umakanth S, Pappachan JM, Bhat SK, Kamath R, Choo BH. Omega-3 fatty acids: a comprehensive review of their role in health and disease. Postgrad Med J. 2009;85:84–90. doi: 10.1136/pgmj.2008.073338. [DOI] [PubMed] [Google Scholar]
- Yuan S, Chu H, Chan JF-W, Ye Z-W, Wen L, Yan B, Lai P-M, Tee K-M, Huang J, Chen D, Li C, Zhao X, Yang D, Chiu MC, Yip C, Poon VK-M, Chan CC-S, Sze K-H, Zhou J, Chan IH-Y, Kok K-H, To KK-W, Kao RY-T, Lau JY-N, Jin D-Y, Perlman S, Yuen K-Y. SREBP-dependent lipidomic reprogramming as a broad-spectrum antiviral target. Nat Commun. 2019;10:120. doi: 10.1038/s41467-018-08015-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeyda M, Stulnig TM. Lipid Rafts & Co.: an integrated model of membrane organization in T cell activation. Prog Lipid Res. 2006;45:187–202. doi: 10.1016/j.plipres.2006.01.002. [DOI] [PubMed] [Google Scholar]
- Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]