Abstract
Background
The main cause of death in patients with hepatocellular carcinoma (HCC) with portal hypertension is esophageal and gastric variceal bleeding caused by severe portal hypertension; therefore, the treatment of portal hypertension is particularly important to prolong the survival of patients. The therapeutic efficacy and safety of transarterial chemoembolization (TACE) combined with a transjugular intrahepatic portosystemic shunt (TIPS) for HCC with esophageal and gastric variceal bleeding has been rarely reported. The aim of this study was to analyze the clinical efficacy of TIPS combined with TACE in the treatment of HCC with esophageal and gastric variceal bleeding.
Material/Methods
A total of 80 patients with HCC with esophageal and gastric variceal bleeding from July 2015 to November 2019 were retrospectively investigated. Clinical outcomes, biochemical indexes, and complications were compared between TIPS plus TACE and endoscopy plus TACE treatments.
Results
Gastrointestinal rebleeding and adverse reactions (P<0.05) after TIPS combined with TACE were lower than that after endoscopy combined with TACE treatment. Furthermore, TIPS plus TACE had superior clinical outcomes than endoscopy plus TACE, which was associated with promising progression-free survival, overall survival, objective response rate, and disease control rate, and improved liver function.
Conclusions
TIPS combined with TACE was better than endoscopy combined with TACE in the treatment of patients with HCC and esophageal and gastric variceal bleeding. TIPS combined with TACE had a better therapeutic effect on improving liver function and prolonging patient survival time.
Keywords: Banti’s Syndrome; Chemoembolization, Therapeutic; Fibrolamellar Hepatocellular Carcinoma
Background
Liver cirrhosis is a common chronic disease of the digestive system caused by chronic injury, including hepatitis infection and alcoholism, and occurs most frequently in adult men. The occurrence of diffuse fibrosis and regenerative nodules in liver tissue is an important cause of portal hypertension, and long-term portal hypertension can cause esophageal and gastric varices bleeding (EGVB) [1]. Variceal bleeding caused by portal hypertension associated with cirrhosis is a serious complication with a high mortality rate; the 6-week mortality rate is 15% to 20% [2]. Endoscopic hemostasis, endoscopic varicose vein ligation, and injection sclerotherapy have been extensively used in the treatment of esophageal and gastric fundus bleeding, which is more effective than conservative treatment with traditional drugs, such as octreotide, proton pump inhibitors, and hemostatic drugs. It is worth noting that 10% to 15% of patients still need repeated endoscopic hemostasis or even repeated blood transfusion treatment due to procedural ineffectiveness [3], and patients are prone to major complications, including ulcer bleeding and stenosis and perforation of the esophagus, so endoscopic treatment has certain clinical application limitations [4,5]. In general, endoscopic treatment is unable to fundamentally solve the problem of portal hypertension, resulting in a high risk of rebleeding and poor prognosis, and the pain caused by endoscopic therapy is intolerable for some patients.
The transjugular intrahepatic portosystemic shunt (TIPS), which is used to build a shunt between the hepatic vein or the inferior vena cava and the branches of the portal vein, shunts the portal vein blood flow and directly reduces the pressure in the portal vein, further reducing the pressure of the collateral circulation, resulting in a reduction of the rebleeding rate of the varicose vein. With postoperative stent stenosis, blockage, displacement, hepatic encephalopathy, and other complications occurring, TIPS is often recommended as a remedial treatment after the failure of drug or endoscopic therapy [6]. It has recently become necessary to reevaluate the clinical efficacy and safety of TIPS owing to the application of the covered stent, which has reduced stent restenosis to a certain extent, significantly improved the patency rate of shunts, reduced the incidence of bleeding and ascites, and raised the quality of life of patients. Increasing evidence shows that TIPS is an effective method for the treatment of portal hypertension and its complications [7,8].
There is a correlation between performing TIPS early and reduced mortality caused by acute portal hypertension variceal bleeding [9]. The advantage of TIPS performed within 72 h after the control of acute esophagogastric variceal bleeding, compared with endoscopic therapy combined with drug therapy, is a lower rebleeding rate, mortality rate, and incidence of hepatic encephalopathy [9]. In addition, with the development of TIPS-related technologies and the continuous expansion of indications, TIPS provides an option for resolving esophageal and gastric variceal bleeding caused by portal hypertension with hepatocellular carcinoma (HCC) [10].
Indeed, nearly 70% to 90% of patients with HCC with liver cirrhosis in China also have portal hypertension to varying degrees [11]. Therefore, it is essential to consider how to improve varicose bleeding or ascites to raise the quality of life, which has been shown to be an important factor influencing the prognosis of patients with liver cancer [12]. Similarly, for patients with HCC with portal hypertension, variceal bleeding caused by the occurrence and aggravation of portal hypertension is the main cause of death [13]. It is therefore more urgent to treat the portal hypertension than the tumor itself, which will prolong the survival time of patients and gain time and the opportunity to further treat the cause of the tumor. However, in the past, it was believed that TIPS could cause tumor metastasis, tumor rupture, and liver failure, and HCC was regarded as a relative contraindication of TIPS.
Nevertheless, the accumulation of evidence has demonstrated that, for patients with HCC with portal hypertension, TIPS relieved esophageal gastric venous bleeding and other complications and had a lower mortality rate associated with TIPS surgery, indicating that TIPS may be an effective and safe therapeutic method to treat HCC with portal hypertension [14]. More importantly, it is believed that TIPS for patients with HCC with portal hypertension requires that the tumor must be controllable, with good liver function and an unblocked portal vein, to have a significant therapeutic effect. Transarterial chemoembolization (TACE) achieved the effect of shrinking the tumor by blocking the blood supply of the tumor, and was regarded as the first-line therapy for patients with stage BCLC-B HCC [15–17]. It is noteworthy that it was required to repeat TACE several times owing to tumor recurrence and metastasis, which resulted in hepatic failure [18–20]. Stent placement cannot only achieve shunting and reduce portal vein pressure, but it can also restore the blood flow to the liver to a certain extent, increasing the liver blood perfusion to restore liver function. In addition, the advantage of TACE is that it relieves the compression of the portal vein by the tumor by shrinking the tumor body, also leaving room for the establishment of the shunt of the TIPS procedure, thereby relieving the effect of portal hypertension. Therefore, the unblocked portal vein and the recovery of liver function may make patients after TIPS more likely to benefit from the combined treatment with TACE. However, the number of similar clinical studies is relatively small, and we need more clinical studies to confirm that this treatment measure is effective. The purpose of this study was to investigate the feasibility and clinical value of TIPS with sequential TACE in the treatment of HCC with EGVB.
Material and Methods
Study Sample
This was a retrospective analysis including 80 patients with EGVB combined with HCC (mean age, 47.2±8.1 years; 55 men, 25 women) that were treated in our hospital from July 2015 to December 2019. Patients diagnosed with EGVB were divided into 2 groups: (1) the TIPS combined with TACE group received TIPS and TACE surgical therapy under local or regional anesthesia (n=42) and (2) the endoscopy combined with TACE treatment group (n=38) received routine endoscopic ligation, sclerosing agent injection, and TACE surgical therapy. In the TIPS group, there were a total of 42 patients, 39 of whom underwent early TIPS treatment after hemostatic treatment with medical drugs at the first admission, and the remaining 3 patients received endoscopic treatment followed by TIPS. The 38 patients in the endoscopic treatment group (control group) were treated with endoscopic ligation for hemostasis instead of TIPS treatment for reasons including the high economic burden of TIPS, large liver tumor size, tumor in the puncture route, and tumor thrombus in the right atrium, for which the number of endoscopic interventions was 1.77±0.29. Whether patients receiving TIPS were continued on a spring coil or received a medical glue emulsion depended on the condition of gastric fundus varices evaluated by intraoperative angiography. Patients with severe heart, kidney, and lung diseases or other serious life-threatening conditions were excluded from the study.
The trial was conducted according to the Declaration of Helsinki and was approved by the Ethics Committee of the First Affiliated Hospital of Sun Yat-sen University [no. 2015-082].
Treatment Procedures
The TIPS procedure was performed in the interventional radiology operating room under local anesthesia. The inferior vena cava and hepatic vein were then accessed through the right internal jugular vein and the pressure of the inferior vena cava was measured. Indirect portal angiography was performed by accessing the right femoral artery to reach the superior mesenteric artery. To determine the morphology of the portal vein, a RUPS-100 puncture set was plugged into the portal vein by the right internal jugular vein. First, portal pressure measurements and the portal pressure gradient (PPG) baseline value were obtained. The PPG corresponds to the difference between the direct portal pressure and the inferior vena cava pressure. An 8-mm diameter Viatorr stent (VCX; W.L. Gore & Associates, Flagstaff, Arizona, USA) was positioned using a guide wire under fluoroscopy. A balloon catheter (6 or 8 mm in diameter) was then inserted into the stent for dilation, after which the portal vein pressure was measured, and PPG was calculated. Intraoperative gastric variceal vein embolization was performed with a coil or a mixed emulsion of medical glue and iodized oil. Patients in the endoscopic treatment group received endoscopic ligation or sclerosing agent injection therapy. When the symptoms of EGVB were controlled, all patients received TACE treatment. The TACE procedure was carried out rigorously according to the TACE protocol [16,21].
Follow-Up and Outcome Assessment
All patients were followed up until death or loss to follow-up. We observed rebleeding rates, complication type and incidence, blood routine examination, AFP, blood coagulation, liver and kidney function, and blood ammonia and performed color ultrasound or gastroscopy and computed tomography (CT)/magnetic resonance imaging (MRI). Tumor response was measured every 4 to 8 weeks using the modified solid tumor efficacy evaluation standard (mRECIST) criteria through contrast-enhanced dynamic CT or MRI [16]. Based on the mRECIST criteria, complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) were calculated. The co-primary endpoints were progression-free survival (PFS, time from start of treatment to progression of HCC) and overall survival (OS, time interval from ERVB to death or loss of follow-up).
Statistical Analysis
SPSS version 22.0 statistical software (IBM Corp, Armonk, NY, USA) was used for data processing and analyses. Quantitative data are expressed as mean±standard deviation and those conforming to a normal distribution according to the Kolmogorov-Smirnov test were compared using the t test. The chi-squared test was performed for categorical data, which are expressed as the number of cases (percentage). Pearson’s chi-squared test and the independent t test were used to compare and analyze categorical variables and continuous variables, respectively. The analyses of PFS and OS were carried out using the Kaplan-Meier method for analysis, and 2-sided 95% confidence intervals (CIs) were calculated for the median time to event. Statistical significance was set at P<0.05.
Results
Patient Characteristics
A total of 169 patients were included, of which, 80 met the eligibility criteria. There were 42 (52.50%) patients in TIPS plus TACE group and 38 (47.50%) patients in endoscopy plus TACE group (Figure 1). The TIPS plus TACE group received TIPS and TACE surgical therapy under local or regional anesthesia (Figure 2). The patients in the endoscopy plus TACE group received routine endoscopic ligation, sclerosing agent injection, and TACE treatment (Figure 3). Patients were diagnosed by the evaluation of medical history, clinical symptoms, blood samples, routine stool examination, abdominal enhanced CT, and gastroscopy (Figures 2, 3). Patient characteristics are summarized in Table 1. The 2 groups were similar in terms of sex, age, and Child-Pugh class (P>0.05; Table 1). The size and number of HCCs in the 2 groups were similar, and most HCC tumors in the 2 groups were large (more than 5 cm in diameter). The number of tumors was mostly less than 5 in the TIPS plus TACE group (83.33%) and in the endoscopy plus TACE group (84.21%). Few patients with HCC were diagnosed with macroscopic vascular invasion or extrahepatic spread. The number of times the TACE treatment was performed in the 2 groups is described in Table 1.
Figure 1.
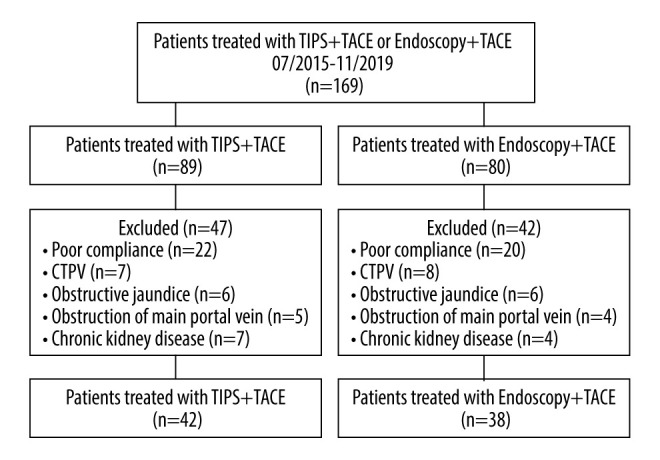
Flow diagram showed selection criteria. CTPV – cavernous transformation of portal vein.
Figure 2.
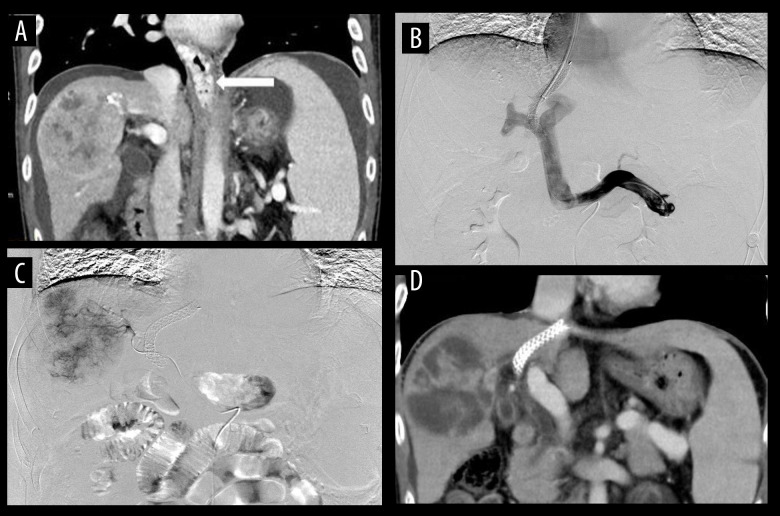
(A) The examination of esophageal and gastric varices (white arrow), peritoneal effusion, and hepatocellular carcinoma by computed tomography (CT) in patients. (B) The effective shunt function of the stent through angiography under transjugular intrahepatic portosystemic shunt (TIPS), with implantation of 8×70-mm Viatorr stent. (C) In patients undergoing transarterial chemoembolization (TACE), the staining of hepatocellular carcinoma was obvious and embolization with drug-loaded microspheres was performed. (D) At the 1-month follow-up after TACE, most of the hepatocellular carcinoma was necrotic, the stent was unobstructed, and the peritoneal effusion disappeared.
Figure 3.
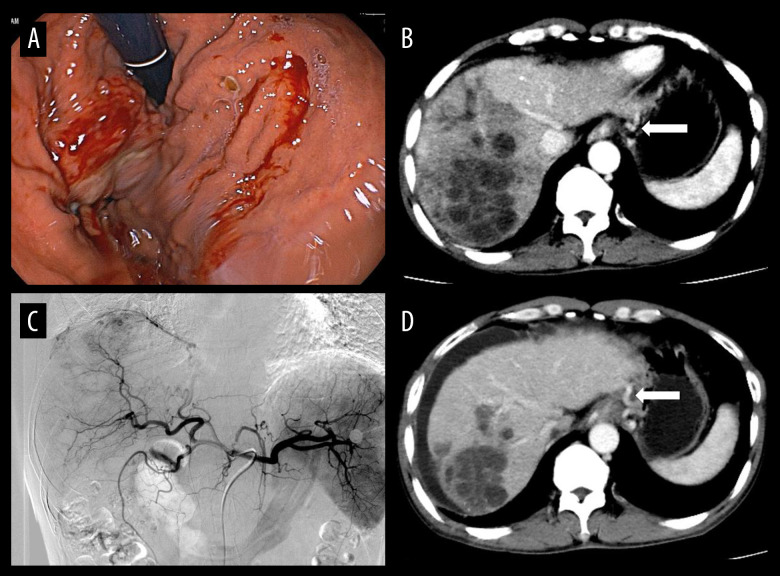
(A) Gastroscopy revealed varices in the fundus of the stomach, which showed bleeding and rupture of blood vessels. Under the gastroscope, application of titanium clips to hemostatic varicose veins and tissue glue injection were used to stop the bleeding. (B) The hepatocellular carcinoma and gastric fundus varices (white arrow) by computed tomography examination. (C) In patients undergoing transarterial chemoembolization (TACE), the staining of hepatocellular carcinoma was obvious and embolization with drug-loaded microspheres was performed. (D) At the 1-month follow-up after TACE, most of the hepatocellular carcinoma was necrotic, but there was peritoneal effusion, and the gastric fundus varices were still not relieved (white arrow).
Table 1.
Baseline demographic and clinical characteristics of patients enrolled in this study.
| Characteristic | TIPS+TACE (n=42) | Endoscopy+TACE (n=38) | χ2 | t | P | |
|---|---|---|---|---|---|---|
| Sex (n/%) | Male | 29 (69.05%) | 26 (68.42%) | 0.004 | – | 0.952 |
| Female | 13 (30.95%) | 12 (31.58%) | ||||
| Age (median±SD) | Years | 47.24±7.86 | 46.08±7.32 | – | 0.684 | 0.496 |
| Child-Pugh classification (n/%) | A | 9 (21.43%) | 9 (23.68%) | 0.067 | – | 0.967 |
| B | 22 (52.38%) | 19 (50.00%) | ||||
| C | 11 (26.19) | 10 (26.32%) | ||||
| Viral hepatitis | HBV | 38 (90.48%) | 34 (89.48%) | 0.557 | – | 0.757 |
| HCV | 1 (2.38%) | 2 (5.26%) | ||||
| None | 3 (7.14%) | 2 (5.26%) | ||||
| HCC size | >5 cm | 23 (54.76%) | 19 (50.00%) | 0.290 | – | 0.865 |
| 3–5 cm | 13 (30.95%) | 12 (31.58%) | ||||
| <3 cm | 6 (14.29%) | 7 (18.42%) | ||||
| Number of tumors | >5 | 7 (16.67%) | 6 (15.79%) | 0.297 | – | 0.862 |
| 3–5 | 14 (33.33%) | 15 (39.47%) | ||||
| <3 | 21 (50.00%) | 17 (44.74%) | ||||
| MVI | With | 8 (19.05%) | 6 (15.79%) | 0.942 | – | 0.332 |
| Without | 34 (80.95%) | 32 (84.21%) | ||||
| EHS | Lung | 2 (4.76%) | 2 (5.26%) | 0.750 | – | 0.687 |
| Lymph nodes | 3 (7.15%) | 2 (5.26%) | ||||
| Bone | 2 (4.76%) | 0 (0.00%) | ||||
| TACE times | – | 2.87±0.32 | 2.75±0.41 | – | 1.359 | 0.183 |
TIPS – transjugular intrahepatic portosystemic shunt; TACE – transarterial chemoembolization; HBV – viral hepatitis B; HCV – viral hepatitis C; HCC – hepatocellular carcinoma; EHS – extrahepatic spread; MVI – macroscopic vascular invasion.
General Curative Effect
The general curative effect was compared between the 2 groups. Endoscopy and intensive CT/MRI examinations were performed on patients at 4 to 8 weeks after surgery. Good hemostatic effect was obtained in both groups, as shown in Figures 2 and 3. However, the incidence of local mucosal necrosis was significantly increased (P<0.05) in the endoscopic treatment group (34.21%) compared with in the TIPS group (0.00%). The incidence of gastrointestinal rebleeding was significantly lower (P<0.05) in the TIPS group (7.14%) than in the endoscopy group (15.79%; Table 2).
Table 2.
Comparison of postoperative complications between groups (n [%]).
| Group(n/%) | Local mucosal necrosis | Gastrointestinal rebleeding | χ2 | P |
|---|---|---|---|---|
| TIPS+TACE (n=42) | 0 (0.00%) | 3 (7.14%) | 5.018 | 0.025 |
| Endoscopy+TACE (n=38) | 13 (34.21%) | 6 (15.79%) |
TIPS – transjugular intrahepatic portosystemic shunt; TACE – transarterial chemoembolization.
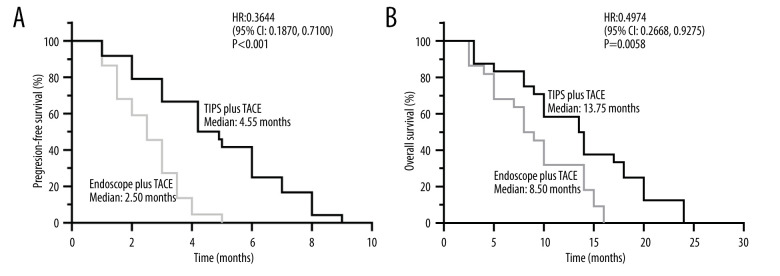
As shown in Table 3, based on the evaluation of the mRECIST criteria in the endoscopy plus TACE group undergoing TACE after endoscopic hemostasis, the disease control rate (DCR) was 55.26% and the objective response rate (ORR) was 23.68%. For the TIPS plus TACE group, DCR was seen in 78.56% of patients and ORR in 38.09% of patients, which was higher than that of the endoscopy plus TACE group, and the results were statistically significant (P=0.041, Table 3). Furthermore, the median PFS was significantly prolonged in the TIPS plus TACE group compared with in the endoscopy plus TACE group (4.55 vs 2.50 months; hazard ratio [HR], 0.3644; 95% CI, 0.1870–0.7100; P<0.0001; Figure 4A). The median OS was 13.75 months for the TIPS plus TACE group and 8.50 months for the endoscopy plus TACE group (HR, 0.4974; 95% CI, 0.2668–0.9275; P=0.0058; Figure 4B).
Table 3.
Tumor responses in patients to transjugular intrahepatic portosystemic shunt (TIPS) plus transarterial chemoembolization (TACE) in the TIPS group and endoscopy plus TACE in the endoscopic treatment group. Results reported as n (%).
| Group (n/%) | Best response | DCR | ORR | χ2 | P | |||
|---|---|---|---|---|---|---|---|---|
| CR | PR | SD | PD | |||||
| TIPS+TACE (n=42) | 4 (9.52%) | 12 (28.57%) | 17 (40.47%) | 9 (21.44%) | 33 (78.56%) | 16 (38.09%) | 7.895 | 0.041 |
| Endoscopy+TACE (n=38) | 2 (5.26%) | 7 (18.42%) | 12 (31.58%) | 17 (44.74%) | 21 (55.26%) | 9 (23.68%) | ||
CR – complete response; PR – partial response; PD – progressive disease; SD – stable disease; DCR (CR+PR+SD) – disease control rate; ORR (CR+PR), objective response rate; TIPS – transjugular intrahepatic portosystemic shunt; TACE – transarterial chemoembolization.
Figure 4.
Kaplan-Meier plots of median (A) progression-free survival and (B) overall survival in the transjugular intrahepatic portosystemic shunt (TIPS) plus transarterial chemoembolization (TACE) group and endoscopy plus TACE group. GraphPad Prim version 8.0 (GraphPad Software Inc, San Diego, CA, USA) was used for the survival analysis and figure editing.
In addition, the survival analysis based on relative reasons of death between the TIPS group and endoscopy group is shown in Table 4. One patient in the TIPS group died of gastrointestinal rebleeding, compared with 4 patients in the endoscopy group, indicating that the main cause of death in the 2 groups was liver failure caused by tumor progression, and the long-term effect of TIPS in controlling gastrointestinal bleeding was significantly better than that of endoscopy.
Table 4.
The survival analysis based on relative reasons of death between groups.
| Reasons of death | TIPS+TACE (n=42) | Endoscopy+TACE (n=38) |
|---|---|---|
| Liver failure | 19 (45.24%) | 17 (44.74%) |
| Gastrointestinal rebleeding | 1 (2.38%) | 4 (10.53%) |
| Hepatic encephalopathy | 2 (4.76%) | 3 (7.89%) |
| Multiple organ failure | 1 (2.38%) | 0 (0.00%) |
| Uncontrollable infection | 2 (4.76%) | 2 (5.26%) |
| Uncontrollable intrahepatic tumor progression | 10 (23.81%) | 8 (21.05%) |
| Uncontrolled progression of extrahepatic metastases | 7 (16.67%) | 4 (10.53%) |
TIPS – transjugular intrahepatic portosystemic shunt; TACE – transarterial chemoembolization. Results reported as N (%) unless otherwise indicated.
Biochemical Indices of Blood
Biochemical indices of blood were compared between the 2 groups at 4 to 6 weeks after surgery. After treatment, the coagulation index, platelet count, and alanine aminotransferase and cholinesterase levels of the 2 groups of patients were improved compared with those before treatment (P<0.05). After treatment, improvements in the coagulation index, platelet count, and alanine aminotransferase and cholinesterase levels were better in the TIPS group than in the endoscopic treatment group (P<0.05). Hepatic encephalopathy developed in 2 patients each in the TIPS plus TACE group (4.8%) and in the endoscopy plus TACE group (5.3%). After treatment, the blood ammonia level was 34.38±11.89 μg/L in the TIPS plus TACE group and 32.17±10.52 μg/L in the endoscopy plus TACE group, with no significant difference between the groups (P>0.05; Tables 5, 6). In addition, these 2 groups were similar in alpha-fetoprotein (AFP) testing before treatment (P>0.05); however, the level of AFP in the TIPS plus TACE group was lower than that in the endoscopy plus TACE group (203.96±63.51 μg/L vs 356.17±52.76 μg/L, P<0.05; Table 5), which showed that the TIPS plus TACE group had greater effectiveness in patients with HCC with esophageal and gastric variceal bleeding caused by portal hypertension than did the endoscopy plus TACE group.
Table 5.
Comparison of biochemical indexes between the 2 groups after treatment.
| Indicators | After treatment | t | p | |
|---|---|---|---|---|
| TIPS+TACE (n=42) | Endoscopy+TACE (n=38) | |||
| PLT (×109/L) | 110.07±18.72 | 90.65±17.72 | 4.751 | 0.000 |
| PT (s) | 11.51±1.78 | 14.59±1.70 | −7.854 | 0.000 |
| TBIL (μmol/L) | 41.52±3.80 | 47.61±4.42 | 5.020 | 0.000 |
| ALB (g/L) | 31.13±2.85 | 29.92±3.19 | −1.366 | 0.179 |
| ALT (IU/L) | 38.17±9.17 | 75.73±14.49 | −13.983 | 0.000 |
| CHE (IU/L) | 2746.02±275.60 | 2339.87±227.56 | 7.143 | 0.000 |
| AFP (μg/L) | 203.96±63.51 | 356.17±52.76 | 8.796 | 0.000 |
| NH3 (μg/L) | 34.38±11.89 | 32.17±10.52 | 0.876 | 0.383 |
PLT – platelets; PT – prothrombin time; ALT – alanine aminotransferase; TBIL – total bilirubin; ALB – albumin; AFP – alpha-fetoprotein; CHE – cholinesterase. TIPS – transjugular intrahepatic portosystemic shunt; TACE – transarterial chemoembolization. Results are presented as mean±SD.
Table 6.
Comparison of biochemical indices between groups.
| Indicator | TIPS+TACE (n=42) | t | Endoscopy+TACE (n=38) | t | ||
|---|---|---|---|---|---|---|
| Pre-treatment | After treatment | Pre-treatment | After treatment | |||
| PLT (×109/L) | 37.72±8.66 | 110.07±18.72 | −45.510* | 41.12±8.45 | 90.65±17.72 | −32.934* |
| PT (s) | 16.25±1.92 | 11.51±1.78 | 87.320* | 16.05±2.02 | 14.59±1.70 | 28.300* |
| TBIL (μmol/L) | 40.68±4.17 | 41.52±3.80 | −9.434* | 41.92±6.17 | 47.61±4.42 | −11.855* |
| ALB (g/L) | 30.48±2.19 | 31.13±2.85 | −3.189* | 29.83±2.76 | 29.92±3.19 | −0.869 |
| ALT (IU/L) | 82.74±11.99 | 38.17±9.17 | 86.722* | 80.14±13.15 | 75.73±14.49 | 17.936* |
| CHE (IU/L) | 2319.39±186.7 | 2746.02±275.60 | −31.081* | 2288.76±220.50 | 2339.87±227.56 | −40.994* |
| AFP (μg/L) | 655.34±219.90 | 203.96±63.51 | 13.948* | 634.01±189.16 | 356.17±52.76 | 9.544* |
| NH3 (μg/L) | 28.50±11.64 | 34.38±11.89 | −34.021* | 27.54±7.36 | 32.17±10.52 | −7.661* |
P<0.05.
PLT – platelets; PT – prothrombin time; ALT – alanine aminotransferase; TBIL – total bilirubin; ALB – albumin; AFP – alpha-fetoprotein; CHE – cholinesterase; TIPS – transjugular intrahepatic portosystemic shunt; TACE – transarterial chemoembolization. Results are presented as mean±SD.
Discussion
EGVB remains one of the most critical complications of cirrhosis and portal hypertension, resulting in death when an appropriate intervention is not delivered promptly and effectively, with an in-hospital mortality rate of 10% to 20% [22]. When it is combined with other diseases that occurred before surgery and complications that occurred after surgery, the mortality rate is even higher [23–25]. However, the latest clinical analyses showed that even if conservative and optimal endoscopic treatment is delivered in time, around 25% of patients with acute portal hypertension begin to bleed again within 5 days [26,27].
It is difficult for clinicians to determine which treatment method or combination therapy is capable of restoring hemostasis quickly, effectively, and safely. Although conservative medications such as octreotide and proton pump inhibitors and hemostatic and blood transfusion products can partially alleviate the symptoms of excessive bleeding, they cannot fundamentally treat esophageal varices caused by portal hypertension, meaning that the risk of rebleeding remains high. Endoscopic variceal ligation and endoscopic variceal sclerotherapy have been used most often for clinical emergency diagnosis and hemostasis. However, most patients with variceal hemorrhage resulting from portal hypertension due to cirrhosis of the liver have already entered the decompensation period, usually accompanied by liver and coagulation dysfunction. In this case, endoscopic treatment frequently fails to control bleeding, and the pain caused by endoscopic ligation is intolerable for some patients.
TIPS is able to reduce portal pressure and inhibit gastrointestinal bleeding by opening the portal and hepatic veins to increase hepatic blood flow and perfusion, with small trauma, less bleeding, and regional or local anesthesia, which is suitable for EGVB. Some studies have shown that TIPS achieved good clinical results in cases of EGVB that were difficult to control with endoscopic treatment [28–30]. A meta-analysis reported that the risk of rebleeding of the esophagogastric vein after TIPS was reduced (RR, 0.46, 95% CI, 0.36–0.58, P<0.001) and that TIPS was superior to endoscopic hemostasis for internal bleeding control [31]. As our results showed, the rebleeding rate of the esophagogastric fundus vein after TIPS was lower than that with endoscopic treatment (7.14% vs 15.79%, P<0.05). First, TIPS not only directly reduces the pressure in the main portal vein, but also further reduces the pressure of portal vein collateral circulation, and the risk of bleeding is further reduced. Second, similarly, the application of a Viatorr stent with a diameter of 8 mm during TIPS cannot only ensure the effective shunt flow and shunt opening rate, but can also reduce the rebleeding rate and the incidence of severe hepatic encephalopathy and other complications [32]. In the present study, we demonstrated that the incidence of local mucosal necrosis was significantly increased (P<0.05) in the endoscopic treatment group (34.21%) compared with in the TIPS group (0.00%). It is certain that the blood ammonia level, as a risk factor for hepatic encephalopathy, was not significantly different in the 2 groups, which may be because TIPS can significantly reduce the amount of blood in the collateral circulation, thereby reducing the incidence of hepatic encephalopathy. A controlled trial showed that early TIPS treatment before announcing the failure of drugs or endoscopic treatment can reduce the risk of rebleeding and mortality [9]. Therefore, according to the guidelines developed by the American Liver Disease Society, TIPS has priority over surgical or endoscopic therapy in cases of fatal bleeding from esophageal and gastric varices. Notably, it is certain that if not prevented for patients with varicose veins who have already experienced bleeding, the incidence of rebleeding in 1 to 2 years is more than half, especially in patients with portal vein thrombosis.
Indeed, many patients with HCC in China have EGVB mostly due to portal hypertension, which in turn causes hepatic failure and hepatic encephalopathy, eventually leading to death [11]. The strategy is that, while actively treating tumors, portal hypertension is also supposed to be effectively reduced in sufficient time for patients with HCC, especially for patients with HCC with portal vein tumor thrombus, for whom treatment is expected to relieve portal hypertension caused by a prehepatic obstruction. Because of the dual blood supply characteristics of the hepatic portal vein and artery and according to the interdependence of portal vein and hepatic artery after TIPS, the blood flow of the portal vein into the liver is decreased, while the blood flow of the hepatic artery is increased to compensate for the decrease of liver blood supply [33]. The portal vein stent placed in TIPS can reduce the portal vein pressure, restore the liver blood flow to a certain extent, and increase the liver blood flow perfusion to protect the liver function of patients. We found that patients treated with TIPS had significant improvement in levels of alanine aminotransferase and cholinesterase compared with those receiving endoscopic treatment, indicating that TIPS improved biochemical indices of liver function and was beneficial for the recovery of liver function. Therefore, the patency of the portal vein improved the blood supply of the liver and made the patients better tolerate the follow-up treatment, including TACE, ablation, and targeted drug therapy, which turned contraindications into indications [34]. In the present study, the median PFS and OS rates in the TIPS plus TACE group were higher than those of patients undergoing TACE after endoscopic hemostasis (P<0.05), suggesting that the survival time of patients with HCC with portal hypertension was significantly prolonged when TACE was successfully performed after TIPS surgery (Figure 4). The ORR, as a secondary endpoint of this trial, was relatively increased in the TIPS plus TACE group (38.09%) compared with in the endoscopy plus TACE group (23.68%), as was the DCR (78.56% vs 55.26%, P=0.041). Therefore, TACE combined with TIPS is the main therapy for patients with HCC with EGVB caused by portal hypertension. Studies have found that repeated TACE was a secure and effective treatment for patients with HCC after TIPS and that the tumor response of multiple TACE treatments was the only predictor of mortality [19,20]; however, there is a need to pay attention to liver function. As shown in the present study, on the premise of restoring liver function, patients with HCC with EGVB received multiple TACE procedures with a low incidence of hepatic encephalopathy and other complications and prolonged patient survival. Moreover, it was found that early TIPS treatment is advocated for patients with a MELD score ≥19 or liver function of Child-Pugh class C, whereas patients with MELD ≤11 or liver function of Child-Pugh class A may not need TIPS [35] owing to the increase of adverse consequences through undergoing TIPS. Most patients included in the present study had a Child-Pugh class of B or C. The benefit of early TIPS over endoscopic treatment in the group of patients with Child-Pugh class B or C (score <14) with esophageal and gastric variceal bleeding was preliminarily demonstrated in our study but confirmation in further studies is needed. Therefore, it is necessary to fully evaluate patient liver function and liver reserve, especially under the effective and safe timing of TACE and other local treatment that can be carried out repeatedly if necessary, to prolong the survival time of patients.
This study had limitations. First, owing to the small sample size, the conclusions of the study may not be generalizable. Further confirmation by clinical trials with large sample sizes is therefore required in the future. Second, this was a retrospective study, and there was therefore potential selection bias in patients recruited at a single center. The results of this study thus need to be verified in different populations.
Conclusions
TIPS combined with TACE treatment had significantly better outcomes than endoscopy combined with TACE for patients with HCC and EGVB caused by portal hypertension. TIPS combined with TACE is recommended as the main therapy for patients with HCC with EGVB caused by portal hypertension.
Footnotes
Conflict of interest: None declared
Declaration of Figures’ Authenticity
All figures submitted have been created by the authors who confirm that the images are original with no duplication and have not been previously published in whole or in part.
Financial support: This work was supported by the Major Scientific and Technological Project of Guangdong Province (no. 2017B030308006) and the Major Program for Tackling Key Problems of Guangzhou City, China (no. 201704020144)
References
- 1.GBD 2017 Cirrhosis Collaborators. The global, regional, and national burden of cirrhosis by cause in 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. 2020;5(3):245–66. doi: 10.1016/S2468-1253(19)30349-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Garcia-Tsao G, Bosch J. Management of varices and variceal hemorrhage in cirrhosis [published correction appears in N Engl J Med. 2011;364(5):490. Dosage error in article text] N Engl J Med. 2010;362(9):823–32. doi: 10.1056/NEJMra0901512. [DOI] [PubMed] [Google Scholar]
- 3.Kochhar GS, Navaneethan U, Hartman J, et al. Comparative study of endoscopy vs. transjugular intrahepatic portosystemic shunt in the management of gastric variceal bleeding. Gastroenterol Rep (Oxf) 2015;3(1):75–82. doi: 10.1093/gastro/gou095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Xu L, Ji F, Xu QW, et al. Risk factors for predicting early variceal rebleeding after endoscopic variceal ligation. World J Gastroenterol. 2011;17(28):3347–52. doi: 10.3748/wjg.v17.i28.3347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lo GH. The role of endoscopy in secondary prophylaxis of esophageal varices. Clin Liver Dis. 2010;14(2):307–23. doi: 10.1016/j.cld.2010.03.009. [DOI] [PubMed] [Google Scholar]
- 6.Tripathi D, Stanley AJ, Hayes PC, et al. U.K. guidelines on the management of variceal haemorrhage in cirrhotic patients. Gut. 2015;64(11):1680–704. doi: 10.1136/gutjnl-2015-309262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Patidar KR, Sydnor M, Sanyal AJ. Transjugular intrahepatic portosystemic shunt. Clin Liver Dis. 2014;18(4):853–76. doi: 10.1016/j.cld.2014.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tripathi D, Stanley AJ, Hayes PC, et al. Transjugular intrahepatic portosystemic stent-shunt in the management of portal hypertension. Gut. 2020;69(7):1173–92. doi: 10.1136/gutjnl-2019-320221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.García-Pagán JC, Caca K, Bureau C, et al. Early use of TIPS in patients with cirrhosis and variceal bleeding. N Engl J Med. 2010;362(25):2370–79. doi: 10.1056/NEJMoa0910102. [DOI] [PubMed] [Google Scholar]
- 10.Deltenre P, Trépo E, Rudler M, et al. Early transjugular intrahepatic portosystemic shunt in cirrhotic patients with acute variceal bleeding: A systematic review and meta-analysis of controlled trials. Eur J Gastroenterol Hepatol. 2015;27(9):e1–e9. doi: 10.1097/MEG.0000000000000403. [DOI] [PubMed] [Google Scholar]
- 11.Ye JZ, Zhang YQ, Ye HH, et al. Appropriate treatment strategies improve survival of hepatocellular carcinoma patients with portal vein tumor thrombus. World J Gastroenterol. 2014;20(45):17141–47. doi: 10.3748/wjg.v20.i45.17141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee YH, Hsu CY, Hsia CY, et al. A prognostic model for patients with hepatocellular carcinoma within the Milan criteria undergoing non-transplant therapies, based on 1106 patients. Aliment Pharmacol Ther. 2012;36(6):551–59. doi: 10.1111/j.1365-2036.2012.05226.x. [DOI] [PubMed] [Google Scholar]
- 13.Liu L, Zhao Y, Qi X, et al. Transjugular intrahepatic portosystemic shunt for symptomatic portal hypertension in hepatocellular carcinoma with portal vein tumor thrombosis. Hepatol Res. 2014;44(6):621–30. doi: 10.1111/hepr.12162. [DOI] [PubMed] [Google Scholar]
- 14.Qiu B, Li K, Dong X, Liu FQ. Transjugular intrahepatic portosystemic shunt for portal hypertension in hepatocellular carcinoma with portal vein tumor thrombus. Cardiovasc Intervent Radiol. 2017;40(9):1372–82. doi: 10.1007/s00270-017-1655-8. [DOI] [PubMed] [Google Scholar]
- 15.Loustaud-Ratti V, Debette-Gratien M, Carrier P. European Association for the Study of the Liver and French hepatitis C recent guidelines: The paradigm shift. World J Hepatol. 2018;10(10):639–44. doi: 10.4254/wjh.v10.i10.639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Heimbach JK, Kulik LM, Finn RS, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67(1):358–80. doi: 10.1002/hep.29086. [DOI] [PubMed] [Google Scholar]
- 17.Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: Chemoembolization improves survival. Hepatology. 2003;37(2):429–42. doi: 10.1053/jhep.2003.50047. [DOI] [PubMed] [Google Scholar]
- 18.Hiraoka A, Kumada T, Kudo M, et al. Hepatic Function during repeated TACE procedures and prognosis after introducing sorafenib in patients with unresectable hepatocellular carcinoma: Multicenter analysis. Dig Dis. 2017;35(6):602–10. doi: 10.1159/000480256. [DOI] [PubMed] [Google Scholar]
- 19.Kang JW, Kim JH, Ko GY, et al. Transarterial chemoembolization for hepatocellular carcinoma after transjugular intrahepatic portosystemic shunt. Acta Radiol. 2012;53(5):545–50. doi: 10.1258/ar.2012.110476. [DOI] [PubMed] [Google Scholar]
- 20.Wang Z, Zhang H, Zhao H, et al. Repeated transcatheter arterial chemoembolization is safe for hepatocellular carcinoma in cirrhotic patients with transjugular intrahepatic portosystemic shunt. Diagn Interv Radiol. 2014;20(6):487–91. doi: 10.5152/dir.2014.13493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Park JW, Park KW, Cho SH, et al. Risk of hepatitis B exacerbation is low after transcatheter arterial chemoembolization therapy for patients with HBV-related hepatocellular carcinoma: Report of a prospective study. Am J Gastroenterol. 2005;100(10):2194–200. doi: 10.1111/j.1572-0241.2005.00232.x. [DOI] [PubMed] [Google Scholar]
- 22.Thabut D, Bernard-Chabert B. Management of acute bleeding from portal hypertension. Best Pract Res Clin Gastroenterol. 2007;21(1):19–29. doi: 10.1016/j.bpg.2006.07.010. [DOI] [PubMed] [Google Scholar]
- 23.Sato M, Tateishi R, Yasunaga H, et al. Variceal hemorrhage: Analysis of 9987 cases from a Japanese nationwide database. Hepatol Res. 2015;45(3):288–93. doi: 10.1111/hepr.12357. [DOI] [PubMed] [Google Scholar]
- 24.Jairath V, Rehal S, Logan R, et al. Acute variceal haemorrhage in the United Kingdom: Patient characteristics, management and outcomes in a nationwide audit. Dig Liver Dis. 2014;46(5):419–26. doi: 10.1016/j.dld.2013.12.010. [DOI] [PubMed] [Google Scholar]
- 25.Bissonnette J, Garcia-Pagán JC, Albillos A, et al. Role of the transjugular intrahepatic portosystemic shunt in the management of severe complications of portal hypertension in idiopathic noncirrhotic portal hypertension. Hepatology. 2016;64(1):224–31. doi: 10.1002/hep.28547. [DOI] [PubMed] [Google Scholar]
- 26.Bosch J, Thabut D, Albillos A, et al. Recombinant factor VIIa for variceal bleeding in patients with advanced cirrhosis: A randomized, controlled trial. Hepatology. 2008;47(5):1604–14. doi: 10.1002/hep.22216. [DOI] [PubMed] [Google Scholar]
- 27.Villanueva C, Ortiz J, Sàbat M, et al. Somatostatin alone or combined with emergency sclerotherapy in the treatment of acute esophageal variceal bleeding: A prospective randomized trial. Hepatology. 1999;30(2):384–89. doi: 10.1002/hep.510300222. [DOI] [PubMed] [Google Scholar]
- 28.Azoulay D, Castaing D, Majno P, et al. Salvage transjugular intrahepatic portosystemic shunt for uncontrolled variceal bleeding in patients with decompensated cirrhosis. J Hepatol. 2001;35(5):590–97. doi: 10.1016/s0168-8278(01)00185-4. [DOI] [PubMed] [Google Scholar]
- 29.Vangeli M, Patch D, Burroughs AK. Salvage tips for uncontrolled variceal bleeding. J Hepatol. 2002;37(5):703–4. doi: 10.1016/s0168-8278(02)00321-5. [DOI] [PubMed] [Google Scholar]
- 30.Monescillo A, Martínez-Lagares F, Ruiz-del-Arbol L, et al. Influence of portal hypertension and its early decompression by TIPS placement on the outcome of variceal bleeding. Hepatology. 2004;40(4):793–801. doi: 10.1002/hep.20386. [DOI] [PubMed] [Google Scholar]
- 31.Zhou GP, Sun LY, Wei L, et al. Comparison between portosystemic shunts and endoscopic therapy for prevention of variceal re-bleeding: A systematic review and meta-analysis. Chin Med J (Engl) 2019;132(9):1087–99. doi: 10.1097/CM9.0000000000000212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang Q, Lv Y, Bai M, et al. Eight millimetre covered TIPS does not compromise shunt function but reduces hepatic encephalopathy in preventing variceal rebleeding. J Hepatol. 2017;67(3):508–16. doi: 10.1016/j.jhep.2017.05.006. [DOI] [PubMed] [Google Scholar]
- 33.Stankovic Z, Rössle M, Euringer W, et al. Effect of TIPS placement on portal and splanchnic arterial blood flow in 4-dimensional flow MRI. Eur Radiol. 2015;25(9):2634–40. doi: 10.1007/s00330-015-3663-x. [DOI] [PubMed] [Google Scholar]
- 34.Ishikawa T, Kubota T, Abe H, et al. Percutaneous transhepatic portal vein stent placement can improve prognosis for hepatocellular carcinoma patients with portal vein tumor thrombosis. Hepatogastroenterology. 2014;61(130):413–16. [PubMed] [Google Scholar]
- 35.Lv Y, Zuo L, Zhu X, et al. Identifying optimal candidates for early TIPS among patients with cirrhosis and acute variceal bleeding: A multicentre observational study. Gut. 2019;68(7):1297–310. doi: 10.1136/gutjnl-2018-317057. [DOI] [PubMed] [Google Scholar]