Abstract
CDC55 encodes a Saccharomyces cerevisiae protein phosphatase 2A (PP2A) regulatory subunit. cdc55-null cells growing at low temperature exhibit a failure of cytokinesis and produce abnormally elongated buds, but cdc55-null cells producing the cyclin-dependent kinase Cdc28-Y19F, which is unable to be inhibited by Y19 phosphorylation, show a loss of the abnormal morphology. Furthermore, cdc55-null cells exhibit a hyperphosphorylation of Y19. For these reasons, we have examined in wild-type and cdc55-null cells the levels and activities of the kinase (Swe1p) and phosphatase (Mih1p) that normally regulate the extent of Cdc28 Y19 phosphorylation. We find that Mih1p levels are comparable in the two strains, and an estimate of the in vivo and in vitro phosphatase activity of this enzyme in the two cell types indicates no marked differences. By contrast, while Swe1p levels are similar in unsynchronized and S-phase-arrested wild-type and cdc55-null cells, Swe1 kinase is found at elevated levels in mitosis-arrested cdc55-null cells. This excess Swe1p in cdc55-null cells is the result of ectopic stabilization of this protein during G2 and M, thereby accounting for the accumulation of Swe1p in mitosis-arrested cells. We also present evidence indicating that, in cdc55-null cells, misregulated PP2A phosphatase activity is the cause of both the ectopic stabilization of Swe1p and the production of the morphologically abnormal phenotype.
Eukaryotic cell cycle progression requires the sequential differential regulation of cyclin-dependent protein kinases (CDKs). In general, CDK activity can be regulated at four posttranslational levels (reviewed in reference 37): the binding of a positive regulator (a cyclin), the binding of a negative regulator (a CDK inhibitor), activating phosphorylation at threonine 169 (Saccharomyces cerevisiae) or at homologous sites in other organisms (e.g., T161 in Schizosaccharomyces pombe), or inhibiting phosphorylation at tyrosine 19 (S. cerevisiae) or at homologous sites (e.g., Y15 in S. pombe).
By regulating the state of phosphorylation of tyrosine 15 of the CDK Cdc2p, organisms such as S. pombe, Xenopus laevis, and humans can effectively coordinate the timing of the entry into mitosis with the correct completion of DNA synthesis (5, 20, 39, 53) or with an assessment of the integrity of the genome (5, 20, 38). The phosphorylation of Y15 in normally growing cells also has the effect of maintaining Cdc2 kinase activity at low levels so as to prevent the ectopic initiation of mitotic events (9). Wee1 kinase and its homologs phosphorylate Y15; Cdc25 phosphatase and its homologs remove this phosphate. In S. pombe, mutations in the genes encoding Wee1p and Cdc25p can elicit severe mitotic defects (44, 45). When DNA replication is blocked or DNA is damaged, a checkpoint signal causes an enhancement of Wee1 function and an inhibition of Cdc25 activity, thereby causing cells to arrest at G2/M and preventing premature entry into mitosis. In S. cerevisiae, the phosphorylation (and dephosphorylation) of tyrosine 19 on its major CDK, Cdc28p, is not required for mitotic timing, nor is it required for DNA replication or DNA damage checkpoint-mediated cell cycle arrest (1, 55). Rather, regulating Y19 phosphorylation of Cdc28 in S. cerevisiae appears to be related to checkpoint mechanisms associated with monitoring the morphological integrity of the cell, be it the actin cytoskeleton (28, 47) or the septin ring of the developing bud neck (3). When the actin cytoskeleton is disrupted either by drug treatment or the effect of conditional mutations, Swe1p, the S. cerevisiae Wee1p homolog, is stabilized, thereby conferring a mitotic delay. When septin function is compromised, Hsl1, Gin4, and Kcc4 kinases redundantly mediate a Swe1-dependent delay of entry into mitosis. It is not known whether these two pathways share components or not.
Protein phosphatase 2A (PP2A) is a major serine/threonine phosphatase whose function has been implicated in a variety of cellular functions including DNA replication, cell cycle progression, RNA transcription, RNA splicing, and translation (60). With regard to its cell cycle function, PP2A has been identified as a negative regulator of entry into mitosis. For example, a factor purified from X. laevis egg extracts that prevents the activation of Cdc2 kinase activity was shown to be a form of PP2A (25). It was also reported that PP2A activity, as inferred from treatment with okadaic acid, a relatively specific PP2A inhibitor, was required to maintain high levels of Xe-Wee1p (the Xenopus Wee1p homolog) activity during DNA replication in X. laevis egg extracts (53). More recently, in this same system, it was shown that okadaic acid increased the rate of degradation of Xe-Wee1p and prematurely induced mitosis in the presence of a DNA replication checkpoint; inactivating the Skp1-Cdc53/Cul1-F-box protein (SCF)-ubiquitinating complex prevented this ectopic mitotic entry, most likely through the stabilization of Xe-Wee1p (32). In S. pombe, loss of one of the two PP2A catalytic subunits causes a wee1-like phenotype and is synthetically lethal with certain mutant alleles of wee1+. This same deletion partially suppressed the growth defect of cdc25 mutants, supporting the notion that PP2A acts as a positive regulator of Wee1 and/or as a negative regulator of Cdc25 (23). By contrast, in S. cerevisiae, PP2A appears to be a positive regulator of entry into mitosis. In a situation in which PP2A catalytic activity was depleted from the cell, Clb2/Cdc28 kinase activity became severely reduced and cells arrested as large budded 2N cells at G2/M (28). This raises two questions: what role, if any, does PP2A play in S. cerevisiae with regard to the regulation of phosphorylation of Y19 of Cdc28p, and, if required for this regulation, through what mechanisms does it achieve that control?
PP2A is thought to function primarily as a heterotrimeric complex composed of a catalytic subunit (C) and two others (A and B) serving regulatory functions (reviewed in references 34, 56, 58, and 60). The A subunit in S. cerevisiae is encoded by TPD3 (57). It serves as a structural platform to which one of the two B-regulatory subunits, Rts1p (46) or Cdc55p (16), and one of the catalytic subunits, Pph21p or Pph22p (40, 54), bind. The deletion of either PPH21 or PPH22 elicits no mutant phenotype, but the loss of Pph21p, Pph22p, and Pph3p (a protein sequence-related to Pph21p and Pph22p) causes abnormal bud shape, abnormal actin cytoskeleton, a G2/M delay, and a weakened cell wall (14, 28). Deletion of either RTS1 or CDC55 produces cells with completely different mutant phenotypes. rts1-null cells are temperature sensitive for growth at 37°C and are defective in their ability to respond to a number of physiological stresses (13, 46, 47). By contrast, cdc55-null cells grow slowly at low temperature and exhibit an abnormal budding morphology (16). They are also defective in establishing a spindle damage checkpoint, losing viability rapidly when exposed to normally nontoxic doses of nocodazole or benomyl (35, 59). rts1-null and cdc55-null cells share no apparent overlap of phenotypic deficiencies. The double-null rts1Δ cdc55Δ mutant exhibits a combination of the two single-null phenotypes (46).
At low temperatures, cdc55-null cells exhibit an elongation of buds and a failure of cytokinesis and cell separation (16). The abnormal morphology is abolished in these cells if they also express a CDC28 gene in which Y19 is changed to F19, thereby rendering the inhibitory phosphorylation site on Cdc28p nonphosphorylatable (59). When measured directly, the phosphorylation level on tyrosine 19 of Cdc28 in cdc55-null cells is higher in cycling and nocodazole-arrested cells than in similarly treated wild-type cells (35). These results suggested a probable role for PP2A in regulating Cdc28 tyrosine phosphorylation.
We have addressed the question of what causes the elevated phosphorylation levels of Cdc28 Y19 in cdc55-null cells. More specifically, we wanted to know if PP2A activity is required for the regulation of either Swe1 kinase activity or Mih1 (the S. cerevisiae Cdc25 homolog) phosphatase activity and, if so, how losing Cdc55p disrupts this regulation. What we have found is that the loss of Cdc55 affects the normal regulation of Swe1p turnover, causing cells to accumulate abnormally high levels of this protein at mitosis. Furthermore, this ectopic accumulation and resultant excess Swe1 kinase activity are likely caused by a gain of a formerly repressed PP2A catalytic function. Our data also show that, as in other eukaryotes, PP2A function is required in S. cerevisiae for regulating the inhibitory phosphorylation of its CDK.
MATERIALS AND METHODS
Plasmids and strains.
All strains used were derivatives of W303. The wild-type and cdc55-null (cdc55Δ::TRP1) strains have been previously described (46). A cdc55-null swe1-null strain was generated by transformation of the cdc55-null strain by one-step gene disruption (42) using a BamHI-HindIII-digested swe1Δ::LEU2 knockout construct (6). W303 strains null for PPH21 and PPH22 (DEY132-1C [pph21Δ::HIS3] and DEY10-2B [pph22Δ::URA3]) were obtained from D. Evans. Strains null for CDC55 and one or both of the genes encoding the PP2A catalytic subunits were generated by crosses, tetrad dissection, and phenotypic screening to identify the appropriate genotypic segregants. W303 strains carrying chromosomally integrated copies of CDC28:HA (ADR508) or Y19F-CDC28:HA (ADR640) were obtained from A. Murray. These genes were introduced into the appropriate deletion strains by standard crosses. A W303 strain (PS701) containing a MIH1 knockout (mih1Δ::LEU2) was obtained from P. Sorger. This strain and the cdc55-null strain were individually crossed to ADR508 to obtain mih1ΔCDC28:HA and cdc55ΔCDC28:HA segregants, respectively.
W303-derived strain 1522 expressing an integrated PDS1:HA gene was obtained from D. Koshland, who also supplied us with a plasmid (pOC42) carrying a GAL1-driven PDS1:HA gene. The chromosomal PDS1:HA was introduced into our laboratory CDC55 and cdc55Δ strains using standard crosses. We obtained strain PY1212 carrying a cdc23-1 allele from D. Pellman. This gene was also introduced into the appropriate strains by standard genetic crosses.
To create strains expressing an HA-tagged form of CLN2, we obtained the CLN2-HA-LEU2-tagging plasmid MT104 from M. Tyers. This plasmid was digested with PvuII, thereby liberating an insert that will integrate at the CLN2 locus. Using this digest, transformations to obtain single-step gene replacements were carried out using wild-type and cdc55-null strains; Leu2+ transformants were isolated, and Western blot analyses permitted us to identify strains producing HA-tagged forms of Cln2p.
To create strains expressing a MYC-tagged MIH1 gene, we obtained a plasmid from D. Lew carrying the final 683 bases of the MIH1 ORF followed by 12 MYC sequences, a stop codon, and 471 bases of the 3′ N-terminal repeat. Partial NdeI digests of this pRS306-based plasmid were used to transform either wild-type or cdc55-null cells, selecting for the URA3 marker of the plasmid. Proper integration produces cells with a full-length MIH1:MYC12 gene and an MIH1 gene truncated at its 5′ end. The correct expression of the MYC-tagged MIH1 gene was confirmed by comparing, by Western analysis, the transformed strains with a positive control (strain JMY1319: matα MIH1:MYC12 [c-term]:URA3:mih1-5′Δ) supplied by D. Lew.
The SWE1-myc-tagging plasmid construct DLB940 (31) was also obtained from D. Lew. A derivative plasmid of DLB940 (based on pRS306 [51]) was created by removing an XbaI/ClaI fragment, filling in bases to generate blunt ends, and then self-ligating the resultant plasmid. This derivative, from which the GAL1 promoter and N-terminal two-thirds of SWE1 were removed, was cut at a KpnI site near the C terminus of SWE1 and used to transform wild-type and cdc55Δ strains, selecting for uracil prototrophy. This process produced cells containing a single copy of SWE1:MYC12 and an adjacent truncated, promoterless SWE1. Transformants were grown on rich medium for 3 days and then plated on 5′-fluoroorotic acid to select for cells that had lost the URA3 gene. Western analyses were carried out to confirm that the cells produced a myc-tagged Swe1p. To generate strains that were cdc55Δ pph21Δ pph22Δ SWE1-myc, cdc55Δ::TRP1 SWE1-myc cells were mated with pph21Δ::HIS3 pph22Δ::URA3 cells, tetrads were dissected, and the triple knockout expressing an epitope-tagged SWE1 was identified by the presence of three selective markers and, as shown by a Western analysis, the presence of a myc-tagged Swe1p.
Wild-type and cdc55Δ strains with GAL1:SWE1:MYC12 as the only functional chromosomal copy of SWE1 were generated as follows. Wild-type and cdc55-null cells were transformed with the BamHI-HindIII-digested swe1Δ::LEU2 construct as described above. After confirming that the SWE1 gene was disrupted, cells were transformed with a StuI-digested DLB940 (the pRS306-GAL1-SWE1-myc-tagging construct [see above]) which would direct integration of the plasmid into the URA3 locus. This then generated a strain with GAL1:SWE1:MYC12 as the only gene expressing a functional copy of SWE1. This strain was finally crossed to the cdc55-null strain, diploids were sporulated, asci were dissected, and the segregants were screened in order to identify cells that were cdc55Δ::TRP1 swe1Δ::LEU2 and had GAL1:SWE1:MYC12 at the URA3 locus.
To create cells in which chromatid separation could be monitored, we obtained W303 strains from J. Bachant (JBY583 and JBY584), each of which contained integrated copies of TetR-GFP-LEU2 and TetO-URA3 (33) but which differed only in mating type. Crosses were made with our standard CDC55 and cdc55Δ strains, and the appropriate segregants were identified for subsequent chromatid analysis.
Cell growth conditions.
Standard yeast protocols and media were used (41). To arrest cell growth, hydroxyurea was added to culture media to a final concentration of 0.2 M; nocodazole was added to a final concentration of 20 μg/ml. In all cases, cells were microscopically examined (either by differential interference-contrast [DIC] for cell morphology or 4′,6′-diamidino-2-phenylindole [DAPI] staining/fluorescence for nuclear morphology) throughout all experiments to ensure that cell cycle arrest had occurred and that such arrest was maintained. The exact conditions used for specific experiments are given in the appropriate figure legends.
DAPI staining and microscopy.
Staining cells with DAPI to determine the state of arrest of cells was carried out by the method of Honigberg and Esposito (17). Cells growing in liquid cultures were ethanol fixed directly in growth medium. Cells growing on solid medium were scraped off, washed once in water, and then fixed. For quantitative measurements, 200 to 300 cells were counted for each time point. Visualization of cells by DIC microscopy was carried out on a Nikon Eclipse TE300 microscope. Visualization of cells using DAPI or green fluorescent protein (GFP) fluorescence was done on an Olympus BX60 microscope. For quantitation purposes, images were captured using an Optronics cooled charge-coupled device camera.
Northern analyses.
Cells to be analyzed were centrifuged and stored as frozen pellets at −80°C. Total RNAs were prepared using a hot-phenol extraction method (24). RNAs were denatured, separated on 1% agarose gels, transferred to filters, and hybridized with a radioactive probe as previously described (47). An Swe1 mRNA probe was generated by labeling a 0.6-kb HindIII-XbaI SWE1 ORF fragment with [α-32P]ATP, using a random labeling kit from Life Technologies.
Protein isolation, electrophoresis, and Western analysis.
The direct extraction of total proteins by solubilizing cells in 1.8 M NaOH–5% beta-mercaptoethanol (β-ME), the separation of proteins by sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis (PAGE), and procedures used for Western analysis have been previously described (46, 47). The antibody against phospho-Tyr15-CDC2 (New England Biolabs) was used following the instructions of the supplier. The mouse monoclonal antibodies 12CA5 and 9E10 were used to detect HA epitope-tagged proteins and MYC epitope-tagged proteins, respectively. Cdc28p and Pho85p were detected using the mouse PSTAIR monoclonal antibody (Sigma). The final visualization of proteins was done using alkaline phosphatase-conjugated second antibodies as previously described (47) or by using horseradish peroxidase (HRP)-conjugated second antibodies and enhanced chemiluminescence (ECL; Amersham).
To phosphatase treat myc-tagged Mih1p, cells were dissolved using NaOH–β-ME (46), and the solubilized proteins were trichloroacetic acid precipitated. After resolubilization of the precipitated proteins (46), 9E10 antibody and agarose-protein A beads were added to immunoprecipitate the Mih1-myc proteins. The protein A beads containing the adsorbed proteins were washed into an appropriate buffer and treated with 200 U of calf intestinal alkaline phosphatase (Promega). Following the phosphatase treatment, proteins were released from the protein A beads for SDS-PAGE separation and Western analysis.
Mih1p in vitro phosphatase activity assay.
Growing CDC55 and cdc55Δ strains expressing an MIH1-MYC gene were arrested in nocodazole for 3 h. As negative controls, CDC55 and cdc55Δ cells expressing a normal MIH1 gene were treated in the same way. Such cells were harvested and frozen at −80°C. Subsequently thawed cells were broken by glass beads at 4°C in an immunoprecipitation (IP) buffer (50 mM Tris-HCl [pH 7.5], 150 mM NaCl, 5 mM EGTA, 0.1% NP-40, 2.7 mM KCl) supplemented with a protease inhibitor cocktail (Boehringer Mannheim) and 1 mM phenylmethylsulfonate fluoride (PMSF). Following cell breakage, the lysates of the four samples were centrifuged at high speed at 4°C for 5 min (all subsequent steps were carried out at 4°C unless specified otherwise). The lysates were transferred to clean centrifuge tubes, and to each was added 20 μl of a 1:1 slurry of protein A beads equilibrated with IP buffer. The tubes were gently agitated for 20 min, after which they were centrifuged at high speed for 5 min. Each of the cleared lysates (ca. 0.5 ml) was removed and transferred to a clean centrifuge tube, and 4 μl of 9E10 anti-myc mouse monoclonal antibody (1 mg/ml; Zymed) was added. These tubes were then gently agitated for 40 min, after which 100 μl of a 1:1 protein A bead-IP buffer slurry was added and agitation was continued for an additional 45 min. The protein A beads were collected by centrifugation at 4,000 × g for 5 s. The supernate was then removed, and the beads were washed three times with 1 ml of IP solution and then twice with 1 ml of phosphatase assay buffer (50 mM Tris-Cl [pH 8.0], 50 mM NaCl, 1 mM EDTA). Following the second wash, one-tenth of the beads were collected by centrifugation and boiled in 20 μl of 2× SDS-PAGE loading buffer. These samples were used to detect the amount and modification of Mih1p in each immunoprecipitate before the phosphatase assay. The myc-tagged Mih1p from the CDC55- and cdc55-null cells adsorbed to the protein A beads served as the source of phosphatase to be assayed in vitro.
To prepare a substrate for Mih1 phosphatase activity, growing mih1-null cells expressing CDC28:HA were treated with nocodazole for 3 h and collected. Such a treatment maximizes the cellular level of phospho-Y19-Cdc28p, the substrate used to monitor the Mih1 phosphatase activity. These cells were then broken with glass beads in IP buffer supplemented as above with a protease inhibitor cocktail and PMSF. The resulting lysate was centrifuged twice for 5 min at 20,000 × g. The cleared lysate was then gently agitated at 23°C for 10 min to bring it up to assay temperature, after which it was divided into four equal parts (about 250 μl each), each then being added to one of the above four protein A-agarose bead immunoprecipitates. The mixtures were vigorously agitated at 23°C, and at 0, 15, 30, 45, and 60 min of incubation, the beads were briefly pelleted, and 14 μl of the lysates was taken from each reaction and boiled with 16 μl of 2× SDS-PAGE sample loading buffer. Also, at the end of the incubation, an aliquot containing one-sixth of the initial amount of washed protein A beads was also assayed to check Mih1p abundance and modification. The visualization of either phospho-Y19-Cdc28p, Cdc28-HA, or Mih1-myc was carried out as described above.
To quantify signals on Western blots, films or stained gels were scanned into a computer using Adobe Photoshop 4.0 LE. The images were then analyzed using either NIH Image 1.62 software (Fig. 4a, 6, and 7b) or Kodak 3.0.2 software (Fig. 3c and 3d).
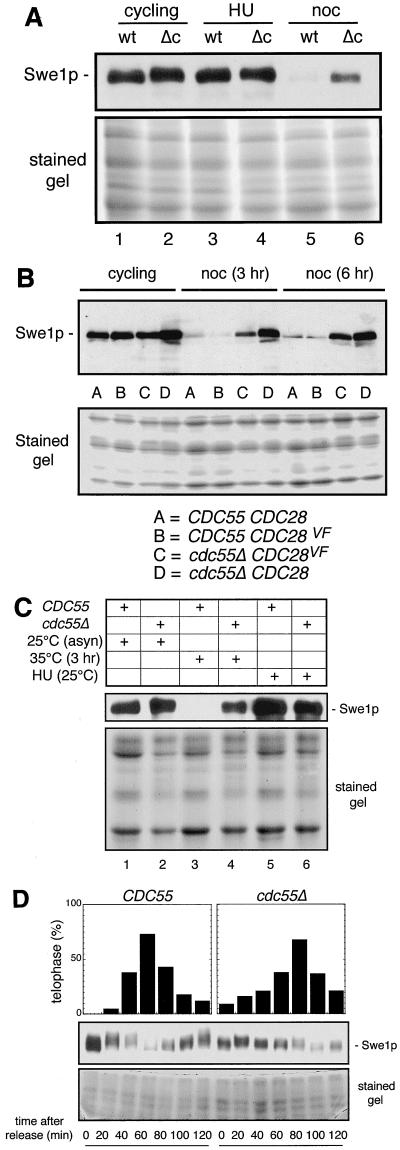
FIG. 4.
Metaphase-arrested cdc55-null cells contain more Swe1p than metaphase-arrested wild-type cells. Wild-type and cdc55-null strains expressing an SWE1 gene tagged with 12 copies of the MYC epitope were constructed (see Materials and Methods). Such cells growing in YPD medium at 30°C were collected as unsynchronized cycling cells (lanes 1 and 2), cells arrested in S phase with 0.2 M HU for 3 h (lanes 3 and 4), or cells arrested in mitosis with 20 μg of nocodazole (noc) per ml for 3 h (lanes 5 and 6). Total cell proteins were extracted and analyzed (A) as in Fig. 1, except that we used the monoclonal antibody 9E10 to detect the MYC-tagged protein. The lower half of the gel not used for the protein transfer was stained with Coomassie blue to show the relative protein loading. To see whether the phenomenon of premature sister chromatid separation exhibited by nocodazole-treated cdc55Δ cells was related to increased Swe1p levels, we determined (B) the relative levels of Swe1p in wild-type (CDC55CDC28), CDC55CDC28VF, cdc55ΔCDC28, and cdc55ΔCDC28VF strains, each expressing a MYC-tagged SWE1 gene (see Materials and Methods for details). Such cells, growing in YPD medium at 30°C, were collected from asynchronous cultures (cycling) or from cultures that had been treated with nocodazole (noc) for either 3 or 6 h; Western analyses were carried out as above. To be able to metaphase arrest wild-type (CDC55) and cdc55-null cells not using drug treatment, we created CDC55 and cdc55Δ strains expressing SWE1:MYC12 and also carrying the gene cdc23-1. The presence of the latter gene allows inactivation of the APC by raising the temperature of the culture to 35°C. Cultures of the two strains were grown at 25°C in YPD and asynchronous early-log-phase cells were collected (lanes 1 and 2), cultures were treated with HU for 3 h before cell collection (lanes 5 and 6), or cultures were shifted to 35°C for 3 h before cell collection (lanes 3 and 4). Total proteins were collected, and Western blot analyses were carried out to determine Swe1p levels (C). As another way to determine if the excess Swe1p in the metaphase-arrested cdc55-null cells was caused by the lethal effects of the nocodazole or elevated temperature treatments (D), the same wild-type and cdc55-null cells expressing SWE1:MYC12 used in panel A were arrested for 3 h with HU and then washed into fresh YPD medium containing no drug (defined as t = 0). Cells were collected at intervals following the S-phase release for Western blot analysis and for microscopically monitoring cell cycle progression as in Fig. 3. In order to magnify the electrophoretic heterogeneity of the MYC12-tagged Swe1p, SDS-PAGE was carried out using 6% gels rather than the standard 10%. Upper panels, percentages of cells in telophase; middle panels, Swe1p levels; lower panels, loading controls.
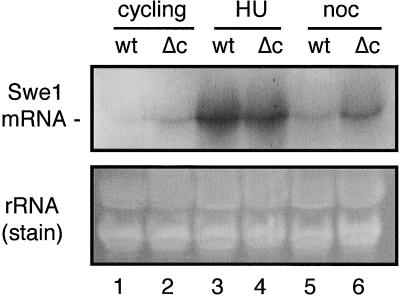
FIG. 6.
Swe1 mRNA levels in S- and M-phase-arrested wild-type and cdc55-null cells. Total RNA was extracted from cells that were treated in the same manner as those shown in Fig. 4A. The RNAs were separated on 1% agarose gels and transferred to filters for Northern blot analysis (upper panel). The probe for Swe1 mRNA was generated by random primer labeling a 0.6-kb HindIII/XbaI fragment from the SWE1 ORF. As an indication of relative lane loading, the lower panel shows the ethidium bromide-stained gel prior to transfer. Lanes 1, 3, and 5: wild-type cells as cycling, HU-arrested, and nocodazole-arrested samples, respectively; lanes 2, 4, and 6: cdc55Δ cells as cycling, HU-arrested, and nocodazole-arrested samples, respectively.
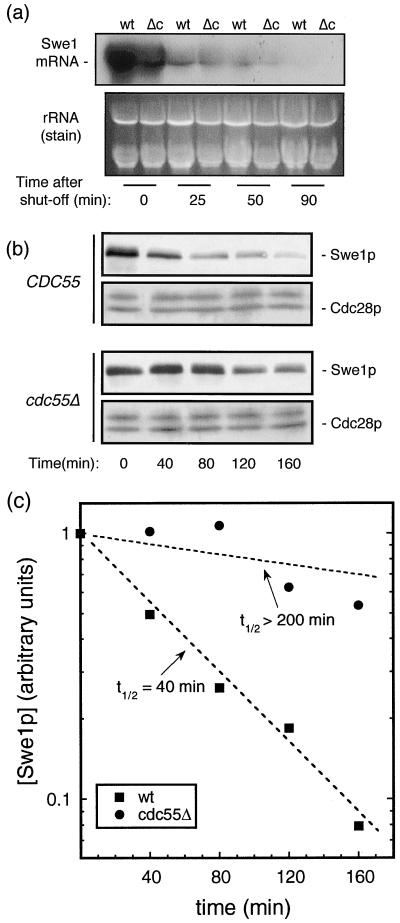
FIG. 7.
Normal Swe1p degradation is suppressed in mitosis-arrested cdc55-null cells. To determine the relative rates of degradation of Swe1 mRNA in wild-type and cdc55-null cells, strains expressing chromosomally integrated SWE1-MYC12 genes driven by a GAL1 promoter (see Materials and Methods) were developed. Such cells were grown in YP raffinose to early log phase and then arrested in nocodazole (20 μg/ml) for 3 h. The arrest phenotype was confirmed by microscopic analysis. Galactose was added to each culture to 2% for 30 min to transiently induce SWE1 transcription. Cells were then collected by centrifugation, washed, and resuspended in YPD medium containing nocodazole in order to suppress any further Swe1 mRNA production and to maintain mitotic arrest. Cells were harvested at 0, 25, 50, and 90 min after repression of transcription. Total RNA was extracted, and a Northern analysis (as in Fig. 5) was carried out (a, upper panel). The ethidium bromide-stained agarose gel prior to transfer served as a loading control (a, lower panel). Based on the above results, we measured Swe1p degradation in the following way. Swe1 mRNA was transiently induced as above. Beginning at 50 min after the SWE1 expression had been suppressed by glucose (defined as t = 0), cells were collected at intervals and the levels of Swe1p were determined (b) as in Fig. 3. Western blots were scanned and quantitated, the levels of Swe1p were plotted (c), and the degradation rates of Swe1p in wild-type and cdc55Δ cells were calculated.
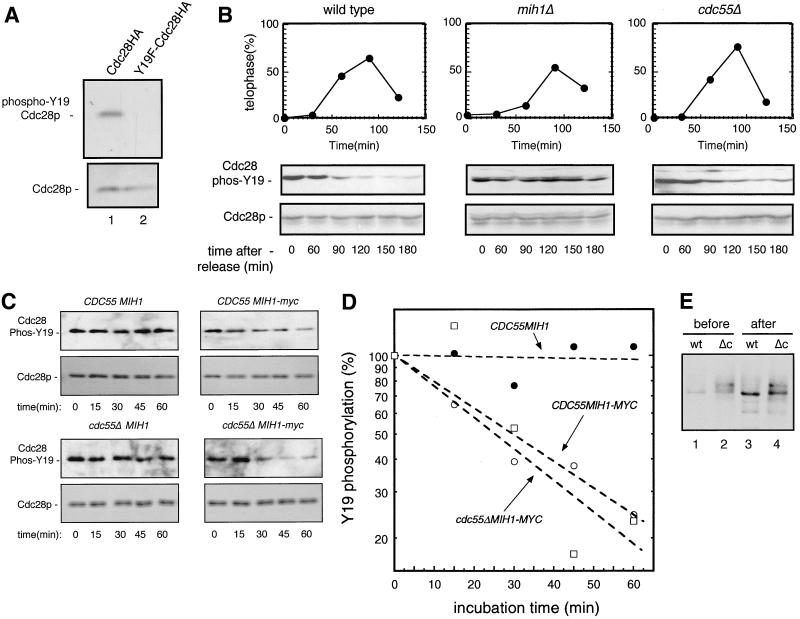
FIG. 3.
Measuring Mih1 phosphatase activity in wild-type and cdc55-null cells. To test the ability of the anti-phospho-Tyr15-Cdc2 antibody to detect S. cerevisiae phospho-Tyr19-Cdc28, total proteins were isolated from growing, unsynchronized cells expressing either an integrated HA-tagged Cdc28 or an integrated Cdc28-HA in which tyrosine 19 was converted to a phenylalanine (Y19F-Cdc28HA). Western blot analyses (A) were carried out as in Fig. 2, first using anti-phospho-Tyr15-Cdc2 antibody (upper panel) and, after washing the filter, using anti-HA antibody 12CA5 (lower panel). To compare the in vivo phosphatase activities of Mih1p in wild-type, cdc55Δ, and mih1Δ strains (all carrying integrated CDC28:HA genes), cells growing in YPD at 30°C were arrested with HU for 3 h. Cells were then collected by centrifugation, washed, and treated in one of two ways. One-fifth of the cells were resuspended in fresh YPD without HU. At various times thereafter, aliquots of cells were fixed and stained with DAPI. By microscopic examination, the percentages of large-budded cells with two separated nuclei (telophase cells) in each population were determined (B, upper panels). These samples were used to monitor the recovery of cells from S-phase arrest and their subsequent entry into and passage through telophase. The other four-fifths of the cells were resuspended in YPD plus nocodazole so that the cells released from S phase arrest would arrest again at the next metaphase. Aliquots of cells were withdrawn at the time of S-phase release (t = 0) and at various times thereafter (60, 90, 120, 150, and 180 min). At the end of the experiment, cells were stained with DAPI to make certain they were still arrested at metaphase as large-budded cells with a single nucleus. Total cell protein extracts were prepared, and a Western blot analysis was performed using the anti-phospho-Tyr15-Cdc2 antibody (B, middle panels). Subsequently, the filter was washed and reprobed with anti-PSTAIR monoclonal antibody to reveal the amount of Cdc28-HA protein in each lane (B, lower panels). To compare the in vitro Mih1 phosphatase activity of wild-type and cdc55Δ cells arrested in mitosis, Mih1p was immunoadsorbed to protein A beads from lysates of cells (either cdc55Δ or CDC55) expressing a MYC-tagged form of Mih1p (see Materials and Methods). As negative controls, similar immunoadsorptions were carried out on CDC55 and cdc55Δ strains expressing a normal (i.e., nontagged) MIH1 gene. The protein A beads were added to a soluble protein extract prepared from an mih1ΔCDC28:HA strain that had been treated with nocodazole for 3 h (see Materials and Methods for details). The phospho-Y19-Cdc28p in such extracts served as the substrate for the protein A-associated Mih1 phosphatase activity. Following protein A bead addition, the extracts were incubated with agitation at 23°C, and samples were withdrawn at 15-min intervals and, as above, analyzed for phospho-Y19-Cdc28 levels and Cdc28p levels (C). The resulting Western blots were quantitated, and the data for phospho-Y19-Cdc28 were plotted (D). To test the effects of the enzyme assay incubation on the quantity and quality of Mih1p recovered, aliquots of the protein A beads containing Mih1-myc from CDC55 (wt) and cdc55Δ (Δc) strains were collected before (one-tenth of the total) and after (one-sixth of the original total) the enzyme assay incubation. The released Mih1p was then subjected to a Western blot analysis (E).
RESULTS
cdc55-null cells remain arrested in mitosis when treated with nocodazole.
It was shown (35) that cdc55-null cells have an increased level of phosphorylation of Cdc28p tyrosine 19 (Y19) when compared with wild-type controls. The extent of phosphorylation at Y19 is controlled by two counteracting enzymes: Swe1p (a kinase) and Mih1p (a phosphatase). Thus, assuming that no other previously unrecognized enzymes are involved, either higher Swe1 kinase activity or lower Mih1 phosphatase activity in cdc55-null cells accounts for the increased Cdc28p Y19-phosphorylation level. Our goal was to establish which alternative applied for cdc55-null cells.
The extent of excess Y19 phosphorylation in cdc55-null cells relative to controls is far more pronounced when cells are treated with nocodazole (35; H. Yang, unpublished results), and we therefore wished to make our measurements in such drug-treated cells. Nocodazole causes metaphase arrest by depolymerizing microtubules (10), thereby triggering the spindle damage checkpoint (27). It was reported that cdc55-null cells have a defective spindle damage checkpoint pathway, with cells quickly losing viability in the presence of nocodazole (35, 59). Such cells initiate sister chromatid separation in the presence of spindle damage and thus, by definition, fail to arrest at metaphase. But unlike other spindle damage checkpoint mutants, nocodazole-arrested cdc55-null cells do not degrade mitotic cyclins Clb2p and Clb3p (35). It was therefore proposed that cdc55-null cells have a normal initial response to spindle damage but are unable to maintain the mitotic arrest (43). If true, we would be unable to reliably compare nocodazole-treated cdc55Δ and wild-type cells, as they would not remain arrested at the same cell cycle stage. Therefore, we asked two questions: (i) do nocodazole-treated cdc55-null cells have a normal initial spindle damage checkpoint response, and (ii) do cdc55-null cells enter the next cell cycle during prolonged nocodazole treatment?
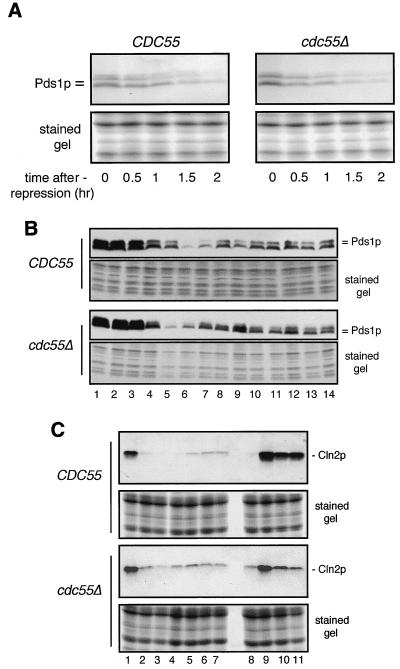
To address the first question, we compared the protein stability of Pds1p in wild-type and cdc55Δ cells. Pds1p is an anaphase inhibitor whose degradation is required for sister chromatid separation (8), and it is one of the major targets of the spindle damage checkpoint pathway (62). When spindle damage is sensed, the Mad1p-Mad2p-Mad3p complex prevents the rapid Cdc20 anaphase-promoting complex (APC)-mediated degradation or Pds1p. The relatively stabilized Pds1p then prevents sister chromatid separation (18). If the spindle damage checkpoint were not triggered by nocodazole in cdc55-null cells, Pds1p would be expected to be much more unstable in these cells than in wild-type controls and would presumably be degraded in the presence of nocodazole. To test these ideas, we transformed wild-type and cdc55-null cells with a plasmid carrying an HA-tagged PDS1 driven by the GAL1 promoter. Both strains were grown in raffinose medium and treated with nocodazole for 3 h. Galactose was added to transiently induce the production of Pds1p, and the cells were then washed into medium containing dextrose (to repress the GAL1 promoter) and nocodazole (to maintain the arrest). Samples were withdrawn at different time points after transcription repression, total cell proteins were extracted, and a Western blot analysis was performed to detect Pds1-HA. We found (Fig. 1A) that Pds1p was as stable in nocodazole-treated cdc55-null cells as in wild-type cells.
FIG. 1.
Nocodazole-treated cdc55Δ cells remain arrested in mitosis. To determine whether nocodazole-treated cdc55-null cells maintain a mitotic arrest, we examined the metabolism of Pds1p and Cln2p in these cells and compared it to that seen in wild-type controls. To examine Pds1p initially, isogenic wild-type (CDC55) and cdc55Δ strains, each transformed with a plasmid carrying an HA-tagged PDS1 gene behind a GAL1 promoter (see Materials and Methods), were grown in raffinose medium to early log phase. Nocodazole was then added to the culture medium to 20 μg/ml to arrest cell growth, and 3 h later, galactose was added to induce the accumulation of HA-tagged Pds1p. After 2 h, cells were collected by centrifugation, washed once, and then, to repress further PDS1 transcription, resuspended in dextrose medium containing nocodazole to maintain mitotic arrest. Cell samples were collected immediately and at 0.5-h intervals for 2 h after PDS1 turnoff. Total cell proteins (see Materials and Methods) were separated by SDS-PAGE, transferred to nitrocellulose filters, and immunoblotted using 12CA5 anti-HA antibody to detect Pds1p (A). The lower half of the gel not used for the protein transfer was stained with Coomassie blue to show the relative protein loading in each lane. Endogenous Pds1p levels were determined in CDC55 and cdc55-null strains at various times during an extended exposure to nocodazole or after a release from a 3-h nocodazole arrest (B). Wild-type and cdc55-null cells, each expressing an HA-tagged PDS1 gene (see Materials and Methods), were grown to early log phase in YPD medium. Nocodazole was added to both cultures, and aliquots of cells were collected at 0, 1, 2, and 3 h. After 3 h in nocodazole, the remaining wild-type and cdc55Δ cultures were split in half. One-half of the cells remained in YPD containing nocodazole. The other cells were collected, washed, and then resuspended in fresh YPD containing no nocodazole to allow cells to enter the cell cycle. Aliquots of cells were collected 20, 40, 60, 80, and 100 min later. Western analyses were carried out to measure Pds1p levels (upper panels), and those portions of the gels not used for protein transfer were stained to show the relative loading in each lane. Lanes 1 through 4, cells in nocodazole at 0, 1, 2, and 3 h; lanes 5 through 9, cells at 20, 40, 60, 80, or 100 min after nocodazole release; lanes 10 through 14, cells in nocodazole at 20, 40, 60, 80, or 100 min after the initial 3-h drug incubation. Cln2p levels were determined in CDC55 and cdc55-null strains at various times during an extended exposure to nocodazole or after release from a 3-h nocodazole arrest (C). Wild-type and cdc55-null cells, each expressing an HA-tagged CLN2 gene (see Materials and Methods), were grown to early log phase in YPD medium. Nocodazole was added to both cultures, and aliquots of cells were collected at 0, 1, 2, and 3 h. After 3 h in nocodazole, the remaining wild-type and cdc55Δ cultures were split in half. One-half of the cells remained in YPD containing nocodazole. The other cells were collected, washed, and then resuspended in fresh YPD containing no nocodazole to allow cells to reenter the cell cycle. Aliquots of cells were collected 1, 2, and 3 h later. Western blot analyses were carried out to measure Cln2p levels (upper panels), and that portion of the gel not used for protein transfer was stained to show the relative loading in each lane. Lanes 1 through 7, cells in nocodazole at 0, 1, 2, 3, 4, 5, and 6 h; lanes 8 through 11, cells at 0, 1, 2, and 3 h after nocodazole release at 3 h. Note: lanes 4 and 8 contain the same protein samples.
This similarity in Pds1p stability in the two strains predicted that the endogenous level of this protein should fluctuate in a similar fashion in strains both treated with nocodazole and subsequently released from cell cycle arrest. This proved to be the case when we measured Pds1p levels in cells arrested in nocodazole for 3 h and then washed into fresh drug-free medium (Fig. 1B). In both wild-type and cdc55-null cells, Pds1p levels remained high during nocodazole treatment (Fig. 1B, lanes 1 through 4); they decreased and subsequently increased following removal of the drug, which led to an exit from mitosis (lanes 5 through 9); and they remained relatively high in cells which were not removed from nocodazole (lanes 10 through 14). Thus, the metabolism of Pds1p does not appear to be abnormal in cdc55Δ cells, indicating that these cells have a normal mitotic checkpoint response and are able to maintain this response during prolonged nocodazole treatment.
It was still possible that, while nocodazole-treated cdc55-null cells respond correctly to spindle damage, they were unable to maintain an arrest and therefore incorrectly entered G1 of the next cell cycle without degrading Pds1p. A clear marker for such an event would be the appearance of Cln2p, one of the G1 cyclins that, while absent in mitotic cells, rapidly accumulates in G1 following completion of mitosis (61). Accordingly, wild-type and cdc55Δ strains carrying chromosomally integrated CLN2:HA genes were grown in yeast extract-peptone-dextrose (YPD) to early log phase and then treated with nocodazole. Samples were withdrawn for Western analyses at 1-h intervals for 6 h following drug addition. In addition, after 3 h in nocodazole, half of the cells in each culture were washed in fresh YPD carrying no drug (in order to monitor their ability to recover) while the other half remained in YPD-nocodazole. Cells removed from the drug were examined microscopically at 1-h intervals to assess the morphological status of these cells as well as being analyzed for Cln2p levels. As seen in Fig. 1C, Cln2p levels quickly declined in both wild-type and cdc55-null cells following nocodazole treatment (Fig. 1C, lanes 1 through 7), reaching a minimum at about 2 h, followed by a slight recovery after 3 to 4 h. While the extent of decline in Cln2p was not quite as severe in cdc55-null cells, in neither case, even after 6 h in nocodazole, did the recovery show Cln2p to approach pre-drug treatment levels (compare Fig. 1C, lanes 1 and 7). As most (>80%) of both cell types remained large budded with a single nucleus (data not shown), what this slight recovery in Cln2p levels represents is not known. The slightly higher Cln2p levels in nocodazole-treated cdc55Δ cells are likely a result of a reported low mitotic Cdc28 kinase activity (35), since this activity is required to repress CLN2 transcription (2).
For both wild-type and cdc55-null strains, cells released from a 3-h nocodazole arrest showed a dramatic increase in Cln2p levels within 1 h (Fig. 1C, lane 9), in both cases exceeding the levels detected in the original asynchronous culture. At 1 h, essentially all wild-type cells had small buds while about 30 to 40% of the cdc55-null cells showed a similar morphology (data not shown). This is consistent with the fact that wild-type cells show a much more robust increase in Cln2p levels. Presumably, the reduced response on the part of cdc55-null cells reflects the ultimately lethal effects of nocodazole treatment on these cells.
To make sure that cdc55Δ cells behaved in our experiments as has been previously reported (35), we monitored sister chromatid separation in nocodazole-treated cells. To do that, a gene expressing a tetracycline repressor (TetR)-GFP fusion construct and genes containing 112 tandem tetracycline operator (TetO2) sequences (33) were introduced into wild-type and cdc55Δ cells to help visualize sister chromatids (see Materials and Methods). Both strains were synchronized by hydroxyurea (HU) treatment and then washed into fresh medium containing nocodazole. The percentage of cells with separated sister chromatids was scored following the start of nocodazole treatment. Our findings were the same as those earlier reported (35), with cdc55Δ cells showing significant premature sister chromatid separation and with wild-type controls showing little or none (data not shown). Thus, our cdc55Δ cells treated with nocodazole separated their sister chromatids but did not exit mitosis.
In summary, mitotic cyclins are not degraded in nocodazole-arrested cdc55-null cells (35), and our results showed that Pds1p and Cln2p metabolism was similar in cdc55-null and wild-type controls. In addition, microscopic analysis of nocodazole-treated cdc55-null cells showed that even after 6 h in drug, the vast majority of cells remained large budded with a single nucleus at the mother-bud neck (data not shown). Furthermore, these cells never rebudded. As premature sister chromatid separation in cdc55-null cells occurs relatively slowly (35; H. Yang, W. Jiang, M. Gentry, and R. L. Hallberg, unpublished data), we concluded, in disagreement with Minshull et al. (35), that cdc55Δ cells treated with nocodazole did arrest at a mitotic stage similar to that of nocodazole-treated wild-type cells, and thus, we could legitimately compare Swe1p and Mih1p activities in these cells.
Mih1 phosphatase activity is similar in cdc55-null and wild-type cells.
As the hyperphosphorylation of tyrosine 19 of Cdc28p was positively correlated with (and appeared to be directly responsible for) the morphological abnormalities manifested in cdc55-null cells (35, 59), we investigated the possible causes of this state of excess phosphorylation. Swe1 kinase and Mih1 phosphatase are the only reported S. cerevisiae enzymes that either add or remove phosphates from the Y19 residue of Cdc28p. Thus, either higher Swe1 kinase activity or lower Mih1 phosphatase activity in cdc55-null cells must account for the increased phosphorylation state of Cdc28p.
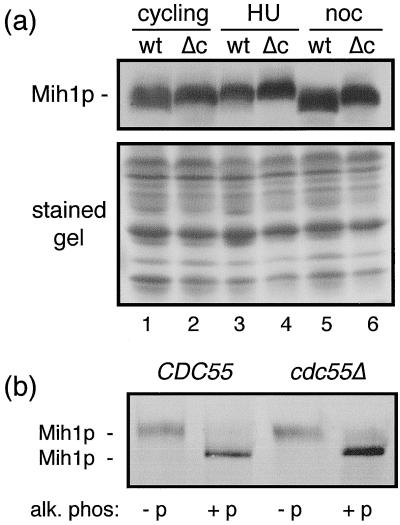
As it has been reported (31) that protein levels of Mih1 phosphatase normally remain unchanged during the cell cycle, the simplest way that Mih1 phosphatase activity could be decreased in cdc55-null cells would be if its cellular amount were to decrease at some time during the cell cycle. To test this, we tagged the MIH1 gene with a MYC epitope and measured the levels of MYC-tagged Mih1p in wild-type and cdc55-null cells either growing exponentially or arrested at different points in the cell cycle (Fig. 2a). In no cases were Mih1p levels obviously decreased in cdc55-null cells relative to controls. Unexpectedly, we did find that the electrophoretic mobility of Mih1p was different at different points in the cell cycle and, more importantly, these variations were different in cdc55-null cells. Alkaline phosphatase treatment of immunoprecipitated Mih1p showed (Fig. 2b) that the differences in electrophoretic mobility were predominantly the effects of differential phosphorylation of this protein.
FIG. 2.
Mih1p levels in wild-type and cdc55-null cells are similar. To determine Mih1p levels, wild-type (i.e., CDC55) and cdc55-null strains expressing an MIH1 gene tagged with 12 copies of the MYC epitope were constructed (see Materials and Methods). Such cells growing in YPD medium at 30°C were collected as unsynchronized cycling cells (lanes 1 and 2), cells arrested in S phase with 0.2 M HU for 3 h (lanes 3 and 4), or cells arrested in mitosis with 20 μg of nocodazole (noc) per ml for 3 h (lanes 5 and 6). Total cell proteins were extracted as described above, separated by SDS-PAGE, transferred to filters, and immunodecorated using the 9E10 anti-MYC antibody to detect Mih1p (a). The lower half of the gel not used for the protein transfer was stained with Coomassie blue to show the relative protein loading. To determine whether the heterogeneity of Mih1p gel mobility seen in (a) was due to differential phosphorylation of Mih1p, proteins were extracted from HU-arrested wild-type and cdc55Δ cells, immunoadsorbed to protein A beads, and treated with alkaline phosphatase (see Materials and Methods). Proteins released from the protein A beads were subjected to a Western blot analysis (b) as above. (−p), untreated controls; (+p), alkaline phosphatase treated.
Given the previous results, should there be a decreased Mih1 phosphatase activity in cdc55-null cells, it would have to occur by virtue of a decrease in enzyme-specific activity. As protein phosphorylation can have either positive or negative effects on the activities of both kinases and phosphatases, the obvious differences seen in the states of Mih1p phosphorylation in cdc55-null relative to control cells make this an attractive candidate for producing abnormal Mih1 phosphatase activity in these cells. To test this, we measured the in vivo Mih1 phosphatase activity in wild-type and cdc55-null cells. This was possible for the following reasons. When wild-type cells progress from S phase through G2 to M phase, Mih1p levels remain constant (31) but Swe1p levels greatly decline (31), and the extent of tyrosine 19 phosphorylation on Cdc28p decreases as a result of the activity of Mih1 phosphatase. Earlier studies (35) in which CDC28 Y19 phosphorylation was determined relied on immunoprecipitation of Cdc28p followed by subsequent assay of the immunoprecipitated protein for the presence of tyrosine phosphorylation using an antiphosphotyrosine antibody. We found that the anti-phospho-Tyr15-CDC2 antibody (New England Biolabs) cross-reacted with phospho-Tyr19-Cdc28 of S. cerevisiae in a total cell protein extract (prepared by dissolving cells directly in 1.8 M NaOH) with essentially no background (cells expressing only the gene CDC28Y19F show no immunoreactive proteins in the molecular-weight range of Cdc28p) (Fig. 3A). Thus, by using strains expressing a chromosomally integrated, HA-tagged CDC28 gene, we could directly measure in any particular cell type the levels of Cdc28p and the extent of Y19 phosphorylation of Cdc28p by a simple Western blot analysis of total cell proteins. Accordingly, wild-type, mih1-null, and cdc55-null cells in early-log-phase growth were arrested in S phase by treatment with HU for 3 h. Such cells should contain highly phosphorylated Cdc28p (35) (Fig. 3B). The arrested cells were then washed and split into two portions. One was resuspended in YPD medium without drug. The cell cycle progression of these cells was monitored by determining the percentages of telophase cells at various times after resuspension. This showed (Fig. 3B, upper panels) that each strain could recover from HU arrest and progress to telophase and into the next G1 within 2 h. The other portion of cells was resuspended in YPD medium containing nocodazole. These cells would have recovered from the S-phase arrest but would now be secondarily arrested at the subsequent mitosis, a time at which Swe1p should have significantly decreased (31), thereby substantially reducing any further Cdc28p phosphorylation. We measured the state of Cdc28 Y19 phosphorylation beginning at the S-phase arrest and at various times following the S-phase release. We found (Fig. 3B, middle panel) that the rates of initial loss of Cdc28 Y19 phosphorylation in wild-type and cdc55-null cells were similar. However, the net extent of phosphate removal was more complete in wild-type than in cdc55-null cells. (We show later that this less complete dephosphorylation is most likely due to excess Swe1 kinase activity in mitosis-arrested cdc55-null cells.) By contrast, and as a control, mih1Δ cells treated in a similar fashion showed no decrease in Y19 phosphorylation during the course of the experiment, confirming that Mih1 is the major, if not only, phosphatase that dephosphorylates Y19 on Cdc28p (Fig. 3B). From these results we concluded that Mih1 phosphatase activity is not qualitatively different in wild-type and cdc55-null cells.
To confirm the above results, we measured the in vitro Mih1p phosphatase activity of the two cell types in the following way. Mih1p in extracts made from nocodazole-arrested wild-type and cdc55-null cells, each expressing MIH1:MYC, was immunoadsorbed onto protein A-agarose beads using anti-MYC antibody. (As negative controls, anti-MYC and protein A beads were added to extracts of both cell types not expressing MYC-tagged MIH1 genes.) Then cell lysates were made from an mih1Δ strain that had also been nocodazole treated. As Mih1p phosphatase activity is absent from such a strain, the phosphorylation of Cdc28p Y19 remains maximal, and we found that the phosphorylated form of Cdc28p was stable in cell extracts for up to 3 h. Using such an extract as a source of substrate for the protein A-bound Mih1 phosphatases from wild-type and cdc55-null cells, we added the protein-bound beads to the cell extract and determined the kinetics of Y19 phosphate removal from Cdc28p. As seen in Fig. 3C, no apparent loss in level of Y19 phosphorylation was observed in the controls (i.e., when the source of Mih1p came from cells not expressing a MYC-tagged MIH1 gene), while in both cases in which Mih1-MYC was adsorbed to the beads, the extent of Y19 phosphorylation decreased. When this decline was quantitated (Fig. 3D), the rates of dephosphorylation were essentially the same whether the source of Mih1p was wild-type or cdc55-null cells. This was consistent with our in vivo findings.
As we had found that the state of phosphorylation of Mih1p was not the same in nocodazole-arrested wild-type and cdc55-null cells and to be sure that comparable amounts of Mih1p were assayed, we examined the relative amounts and the states of phosphorylation of the bead-bound Mih1p at the beginning and at the end of the incubation. As seen in Fig. 3E, the electrophoretic differences in Mih1p, reflecting the relative extents of phosphorylation, from the two cell types were maintained throughout the course of the assay. Also, the levels of the two proteins remained comparable as well (the fractions of the immunoprecipitates analyzed before and after were not the same; see figure legend). Thus, in neither our in vivo nor our in vitro assays of Mih1p phosphatase activity does the state of Mih1p phosphorylation detectably alter this activity. It remains to be determined what the physiological and biochemical significance of the altered Mih1p phosphorylation state in cdc55-null cells might be.
The level of SWE1 kinase is higher in cdc55-null cells.
Having determined that decreased levels of Mih1 phosphatase activity are not the likely cause of Y19 hyperphosphorylation in cdc55-null cells, we then measured Swe1p levels in wild-type and cdc55Δ cells. Swe1p levels normally fluctuate during the cell cycle (31). Therefore, we compared Swe1p levels in wild-type and cdc55-null cells as either asynchronous cultures, HU-arrested (S-phase) cells, or nocodazole-arrested (mitosis) cells (Fig. 4A). In asynchronous cultures, Swe1p levels in cdc55-null cells were slightly higher than that seen in wild-type cells. In HU-arrested cells, both strains had similar amounts of Swe1p. However, in nocodazole-arrested cells, while the wild-type strain had barely detectable amounts of Swe1p, cdc55-null cells had a considerably higher content of Swe1p. The relative levels of Swe1p found in these two strains, whether as asynchronous cultures or as nocodazole-arrested cells, correlated surprisingly well with the relative extent of phosphorylated Cdc28 Y19 found in these two cell types (35) (data not shown), suggesting that it is the increased amount of Swe1p in cdc55-null cells that is the direct cause of Y19 hyperphosphorylation and, consequently, the abnormal budding morphology.
Although we had shown that nocodazole-treated cdc55Δ cells remain arrested in mitosis, this arrest differed from that seen in wild-type cells, as premature sister chromatid separation occurs in cdc55-null cells (which, as indicated earlier, we independently confirmed in our strains [data not shown]). This raised the question of whether the differences in Swe1p levels we saw were a consequence of the premature sister chromatid separation. We could directly address this for the following reason. It was shown (35) that if in cdc55-null cells the wild-type CDC28 gene was replaced by CDC28VF (where threonine 18 is converted to valine and tyrosine 19 to phenylalanine), no premature sister chromatid separation occurs in such cells when they are nocodazole treated. We therefore measured Swe1p levels in cells that were either CDC55 CDC28, CDC55 cdc28VF, cdc55Δ CDC28, or cdc55Δ CDC28VF, each expressing a chromosomally integrated SWE1:MYC12 gene. We examined asynchronously dividing cells and cells that had been treated with nocodazole for 3 or 6 h. As seen in Fig. 4B, nocodazole-treated cdc55-null cells accumulated much higher levels of Swe1p than controls even when the CDC28VF gene was present. While the levels of Swe1p were not as high as in cdc55-null cells carrying a normal CDC28 gene, this decreased amount of Swe1p was probably caused by an abolition of the positive feedback of Swe1p on its own stability, an effect associated with the expression of the CDC28VF mutant allele (49). In any event, these results showed that increased Swe1p levels are not caused by premature sister chromatid separation.
Another way of inducing a metaphase arrest, thereby not relying on nocodazole treatment and spindle depolymerization, is to inactivate the APC, thereby inhibiting the proteolysis of proteins normally degraded during mitosis (19). CDC23 encodes a subunit of the APC (63). An allele of this gene, cdc23-1, encodes a protein which, at high temperature, causes the loss of the ubiquitinating activity of the APC and the subsequent metaphase arrest of cells carrying this gene (19). We therefore created wild-type and cdc55-null strains expressing SWE1:MYC12 and also carrying the cdc23-1 gene. Such cells growing asynchronously at 25°C showed comparable levels of Swe1p in both strains (Fig. 4C, lanes 1 and 2). Similarly, when these two strains were treated with HU for 3 h, both showed comparable increases in Swe1p levels (Fig. 4C, lanes 5 and 6). However, when CDC55-expressing cells were shifted to 35°C for 3 h, Swe1p levels became undetectable, while in cdc55Δ cells a substantial amount of Swe1p remained (Fig. 4C, lanes 3 and 4). In both cases, after 3 h at 35°C, most of the cells remained large budded with a single nucleus (wild type, 89%; cdc55Δ, 90%). These results showed that independent of any spindle-assembly-related defect, Swe1p levels remained high in mitotically arrested cdc55Δ cells.
One potentially trivial reason for the excess Swe1p found in cdc55-null cells was that the nocodazole-treated cells were dying (35, 59), thereby inactivating in some unknown way their protein degradation machinery. Similarly, while cdc55Δ cells grow fine at 37°C, the double mutant cdc55Δ cdc23-1 is a sick strain, and it is possible that these cells also die at 35°C. To rule out these potentially trivial reasons for seeing elevated Swe1p levels in mitotically arrested cdc55-null cells, we measured the change in levels of Swe1p in cells that were arrested in S phase with HU and then released from that arrest. While wild-type cells showed (Fig. 4D) a transient decrease in Swe1p during G2 and M, cdc55-null cells maintained a more constant level of Swe1p through G2 and M and into the next G1 phase. Furthermore, the pronounced electrophoretic-mobility changes of Swe1p seen in control cells, presumably reflecting the hyperphosphorylating activity of Hsl1 kinase (29, 48), were not obvious in cdc55-null cells. While an initial shift was observed, it was not maintained. As this Hsl1p-dependent hyperphosphorylation is a normal precursor to Swe1p destruction in wild-type cells, its apparent absence in cdc55-null cells is consistent with the maintenance of Swe1p levels. We concluded that the elevated level of Swe1p in mitosis-arrested cdc55-null cells was due to the absence of the decline of this protein that normally occurs during G2 and M phases in wild-type cells and that it was not a biological artifact induced by the nocodazole or high-temperature treatments.
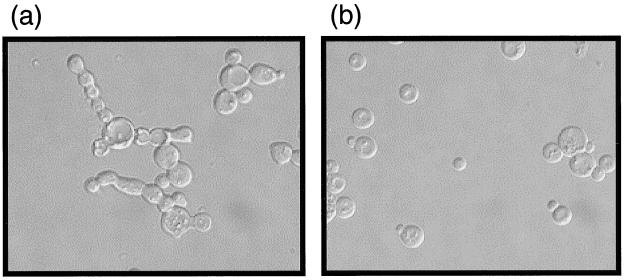
As a way of independently confirming the link between elevated Swe1p levels in mitosis-arrested cdc55-null cells, hyperphosphorylation of Cdc28 Y19, and abnormal morphology, we created the double-knockout strain cdc55Δ swe1Δ. As expected, no Cdc28 Y19 phosphorylation was detected in these cells (data not shown). More importantly, however, this strain no longer exhibited an abnormal budding morphology at low temperatures (Fig. 5). This observation strengthens the conclusion that, in cdc55-null cells, it is the elevated level of Swe1p that is responsible for ectopically inhibiting the Cdc28 kinase activity necessary for proper bud morphogenesis (59).
FIG. 5.
SWE1 expression is essential for the morphological defect of cdc55-null cells. cdc55Δ and swe1Δ cdc55Δ strains were grown on YPD plates at 30°C for 1 day and then shifted to 16°C for 2 more days. Cells were harvested, washed, fixed, and then microscopically examined using Nomarski optics. (a) cdc55Δ cells at 16°C; (b) swe1Δ cdc55Δ cells at 16°C.
An increased Swe1p level in mitosis-arrested cdc55-null cells is caused mainly by ectopic protein stabilization.
As the level of Swe1p can be regulated during the cell cycle at the transcriptional and posttranscriptional phases (36, 50), as well as the posttranslational level (protein degradation [49]), the increased levels of Swe1p found in cdc55-null cells could be due to alterations in one or more of these processes. If the rate of Swe1 transcription were to be abnormally high and/or the rate of Swe1 mRNA degradation were ectopically decreased at a particular time during the cell cycle, this would have caused an excess of Swe1p to be accumulated. In either case, the manifestation of either of these situations would be an increased steady-state level of Swe1 mRNA at that time when excess Swe1p was present in the cell. To see whether this was the case, we performed a Northern blot analysis on RNA extracted from wild-type and cdc55-null cells that had either been growing asynchronously or been arrested with HU or nocodazole (a portion of samples which were also used for Fig. 4A). These data show (Fig. 6) that as previously demonstrated (50), Swe1 mRNA levels are elevated in S-phase-arrested wild-type cells and considerably depressed in mitosis-arrested cells. They also show that while Swe1 mRNA levels in S-phase-arrested wild-type and cdc55-null cells are indistinguishable, the mRNA level in mitosis-arrested cdc55-null cells is noticeably elevated relative to that in wild-type controls. Assuming no differences in the translation efficiencies of Swe1 mRNA in the two cell types, if the hyperaccumulated Swe1p were only accounted for by the translation of excess mRNA, the relative excesses of mRNA and protein levels in cdc55-null cells versus wild-type cells should be comparable. They are not. In the case of the mRNAs, the measured ratio (i.e., cdc55-null Swe1 mRNA:wild-type Swe1 mRNA) is about 2.5:1; for Swe1p, the measured ratio (from Fig. 4A) is almost 11:1. While elevated Swe1 mRNA can account for some of the excess Swe1p present in cdc55-null cells, it clearly can account for only a small part of it. From these data we cannot tell whether the excess Swe1 mRNA in mitosis-arrested cdc55-null cells is the result of increased Swe1 transcription, a decrease in rate of Swe1 mRNA degradation, or both.
While Swe1p is relatively stable during S phase and early G2, its degradation rate increases some four- to fivefold sometime early in G2 (49). This increased degradation rate continues on into mitosis. Thus, one way that Swe1p levels could be elevated in nocodazole-arrested cdc55-null cells would be if this normal increase in rate of Swe1p degradation were somehow prevented. We examined this possibility in the following way. Wild-type and cdc55-null strains were constructed in which the SWE1 gene was replaced by a MYC-tagged SWE1 gene driven by a GAL1 promoter. Such cells, while in log-phase growth in raffinose-containing medium, were nocodazole arrested. Swe1 transcription was transiently induced in the arrested cells by adding galactose to 2% for 0.5 h and then transferring them into glucose medium. Throughout this time, nocodazole was maintained in the culture medium to keep cells in mitotic arrest. We initially monitored Swe1 mRNA levels in these cells and found (Fig. 7a) that following the final shift into glucose medium, in both wild-type and cdc55-null cells the levels of Swe1 mRNA rapidly decreased, as expected, being barely detectable at 50 min and undetectable at 90 min. Thus, 50 min following the shutoff of the SWE1 gene, the cellular level of Swe1p in each of these cells would essentially be determined solely by the degradation rate of Swe1p. Accordingly, we repeated the above experimental protocol, only this time we monitored the levels of Swe1p in the two cell types beginning 50 min after Swe1 transcription was terminated. As seen in Fig. 7b, Swe1p levels remained high for several hours in cdc55-null cells while they significantly declined in wild-type cells. Quantitation of these data (Fig. 7c) indicated that while Swe1p turned over with a half-life of approximately 40 min, the Swe1p half-life in cdc55-null cells was >200 min. Thus, it is most likely that this difference in Swe1p degradation rate is the major cause of the excess Swe1p found in nocodazole-arrested cdc55-null cells.
Unregulated PP2A activity is the cause of excess Swe1p accumulation.
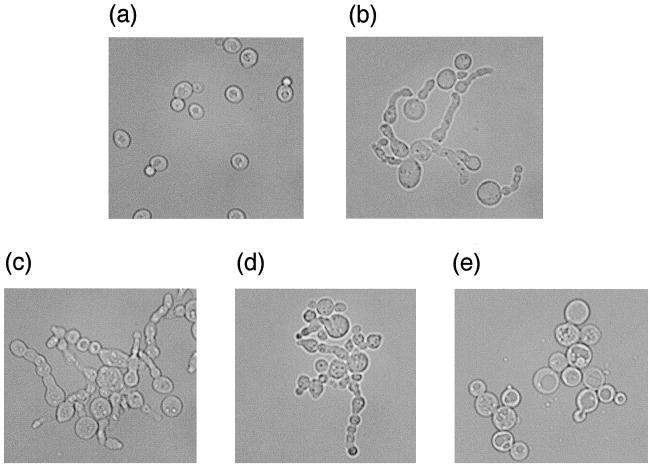
The absence of Cdc55p, the B-regulatory subunit of the trimeric PP2A, could alter PP2A activity in one of three ways. It could cause the loss of a particular phosphatase activity, it could result in the excessive increase of an already present activity, or it could elicit a novel, i.e., not normally present, phosphatase activity. If the abnormal morphology of cdc55-null cells at low temperature is caused by a loss of a particular PP2A phosphatase activity, one might have expected that the loss of one or both of the PP2A catalytic subunit genes (PPH21 and PPH22) would induce budding defects similar to those exhibited by cdc55-null cells. However, this is not the case (40). On the other hand, if loss of Cdc55p elicits a gain of PP2A function, either a novel one or one already present in the cell, removing one or both of the PP2A catalytic subunit genes in a cdc55-null strain might suppress the morphological defect. Accordingly, we constructed cdc55Δ pph21Δ, cdc55Δ pph22Δ, and cdc55Δ pph21Δ pph22Δ strains and observed their growth properties and morphologies at 16°C. While the two double-disruption strains had the same growth properties and morphologies as cdc55-null cells, the triple-knockout strain, while still showing a few cells with abnormal buds at 16°C, was essentially cured of the cdc55Δ-induced morphological phenotype (Fig. 8). These cells did grow more slowly at all temperatures when compared with cdc55-null cells, but no more slowly than pph21Δ pph22Δ cells, and their morphologies were also indistinguishable from pph21Δ pph22Δ cells (data not shown).
FIG. 8.
Disrupting both PPH21 and PPH22 in cdc55-null cells suppresses the morphological defect. Wild-type, cdc55Δ, pph21Δ cdc55Δ, pph22Δ cdc55Δ, and pph21Δ pph22Δ cdc55Δ strains were grown on YPD plates at 30°C for 1 day and then were shifted to 16°C for 2 days. Cells were harvested, washed, fixed, and then microscopically examined using Nomarski optics. (a) Wild-type cells; (b) cdc55Δ cells; (c) pph21Δ cdc55Δ cells; (d) pph22Δ cdc55Δ cells; (e) pph21Δ pph22Δ cdc55Δ cells.
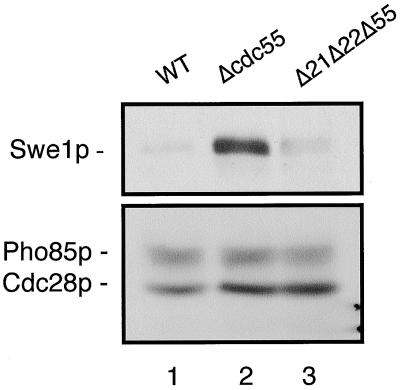
While the above data were consistent with a gain-of-phosphatase-function phenotype for cdc55-null cells, because the loss of both PP2A catalytic subunit genes disrupts the normal actin cytoskeleton (28) and produces somewhat swollen cells as a result of its abnormal cell wall (14), it was possible that the suppression of the abnormal budding phenotype in the triple-disruption strain was indirectly masked by the phenotype induced by the absence of Pph21p and Pph22p. To address the question of whether the removal of Pph21p and Pph22p from cdc55-null cells had a more direct effect on suppressing the abnormal budding morphology, we asked whether Pph21p and Pph22p activity could be shown to be responsible for elevated levels of Swe1p. To do this, we constructed a strain in which the three genes, CDC55, PPH21, and PPH22, were all disrupted and which contained a chromosomally integrated, MYC-tagged SWE1 gene. Such cells, as well as the wild-type and cdc55Δ strains expressing the MYC-tagged SWE1 gene, were grown at 30°C and then arrested in mitosis with nocodazole. Proteins from these cells were isolated, and the levels of Swe1p were determined in each by Western blot analysis (Fig. 9). As seen earlier (Fig. 4A), nocodazole-arrested cdc55-null cells contain considerably more Swe1p than wild-type cells. However, the Swe1p content of the triple-disruption strain was reduced to almost the level of that seen in wild-type cells. Taken together, these results show that it is the abnormally regulated catalytic activity of Pph21p and Pph22p in cdc55-null cells that is responsible for the elevated levels of Swe1p and, ultimately, the abnormal budding morphology of these cells. What the target(s) of this activity is remains to be determined.
FIG. 9.
Disrupting PPH21 and PPH22 in a cdc55-null strain lowers Swe1p levels in metaphase-arrested cells. Wild-type, cdc55Δ and pph21Δ pph22Δ cdc55Δ cells (all expressing MYC-tagged SWE1 genes) were grown at 30°C in YPD medium to early log phase. Wild-type and cdc55-null cells were arrested with nocodazole (20 μg/ml) for 3 h. pph21Δ pph22Δ cdc55Δ cells (Δ21Δ22Δ55) were treated with nocodazole for 5 h because of the slow growth rate of these cells. The arrest phenotype of all cells was microscopically confirmed prior to their harvest. Western analyses of the proteins isolated from these cells were carried out using anti-MYC antibody to measure Swe1p levels (upper panel) and anti-PSTAIR antibody to detect Cdc28p and Pho85p in order to determine relative protein loading in each lane (lower panel). Lane 1, wild-type cells; lane 2, cdc55Δ cells; lane 3, pph21Δ pph22Δ cdc55Δ cells.
DISCUSSION
Given that the budding defect in cdc55-null cells is elicited by the hyperphosphorylation of Cdc28p Y19 and the resulting increased inhibition of this kinase, our results show that this excess phosphorylation is due in large part, if not exclusively, to the ectopic accumulation of Swe1p in these cells. We have shown that elevated Swe1p levels occur because the increased rate of turnover of Swe1p that is normally induced during G2 in wild-type cells does not occur in cdc55-null cells. These elevated levels of Swe1p in cdc55-null cells apparently maintain continued inhibition of Cdc28 kinase activity at a time when, in wild-type cells, Cdc28 kinase is normally required to regulate the shift from axial to isotropic bud growth (26). Without that switch, abnormal bud development and, hence, the mutant phenotype, can occur.
We believe that while the increased level of Cdc28 Y19 phosphorylation seen in cdc55-null cells is important in inhibiting Cdc28 kinase activity, the increased Swe1p levels themselves can contribute to this inhibition. Swe1p has been shown to be able to inhibit Cdc28p activity through a mechanism independent of Swe1 kinase activity (30). Consistent with this, we found that when Swe1p was overproduced to a level at least 10 times higher than its normal endogenous level, thereby causing a G2/M arrest, the phosphorylation level on Y19 of Cdc28 in such cells was no higher than that of wild-type controls (data not shown). Furthermore, under these conditions, we found that Cdc28p could be coimmunoprecipitated with Swe1p (data not shown). Thus, Swe1p may bind to Cdc28p and inhibit its activity without increasing the extent of Y19 phosphorylation. In addition, mih1-null cells have a much higher Y19 phosphorylation level than do cdc55-null cells during mitosis (Fig. 2), yet mih1-null cells show only a mildly elongated morphology without a failure of cytokinesis, thus indicating that a high level of Y19 phosphorylation is not sufficient to elicit a cdc55-null-like phenotype. If in cdc55-null cells the abnormal morphology were caused solely by the increased Y19 phosphorylation, then when this phosphorylation level was further increased in a cdc55Δ mih1Δ strain, such cells should show an even more abnormal morphology, but that is not what we found (data not shown). The double-null cells appear just like cdc55-null cells, with relatively normal morphology at 30°C and a similar cdc55-null morphology at 16°C (data not shown). We conclude that increasing the Swe1p abundance in cdc55-null cells down-regulates Cdc28 kinase activity, by both Swe1 kinase-dependent and -independent mechanisms.
While we were able to show, by expressing CDC28VF in cdc55-null cells, that inhibiting premature sister chromatid separation did not in itself prevent Swe1p accumulation in nocodazole-arrested cells (Fig. 4B), we did find the abnormally high levels of Swe1p to be less than that found in cdc55-null cells expressing a normal CDC28 gene. It has been shown that Swe1p positively regulates its own stability (49). A high level of Swe1p inhibits Cdc28 kinase activity, an activity which in itself is required for efficient Swe1p degradation (49). By inhibiting Cdc28 kinase, Swe1p promotes its own accumulation. A CDC28VF mutant is resistant to Swe1p inhibition, and, consequently, Swe1p is less stable in a strain expressing this gene (49). In cdc55Δ CDC28VF cells, this positive feedback loop would be expected to be disrupted, and therefore such cells should have lower levels of Swe1p than are found in cdc55-null cells, which is precisely what we found. However, it is still possible that nocodazole-arrested cdc55-null cells accumulate at a point in mitosis where Swe1p levels normally increase, but no evidence for such an event has been reported (49). Furthermore, if premature sister chromatid separation in cdc55-null cells contributed to Swe1p accumulation significantly, a positive correlation between these two events would be expected. That is not what we observed. It has been shown that sister chromatid separation starts upon entry into mitosis and accumulates during prolonged incubation in nocodazole (35). When the cdc55-null cells were treated with nocodazole for 3 h and 6 h, the percentage of cells with separated sister chromatids should have increased significantly over this period of time (35). However, Swe1p levels did not show a corresponding increase. Instead, they remained the same at these two time points (Fig. 4B). Thus, it seems that the phenomenon of premature sister chromatid separation contributes little to the increased levels of Swe1p found in nocodazole-treated cdc55-null cells.
The step(s) in the regulatory pathway of Swe1p turnover that is adversely affected in cdc55-null cells has not been determined, but our data show that it is the unregulated activity of PP2A phosphatase activity that is responsible, either directly or indirectly, for the alteration in Swe1p degradation rate. If normally there is competition for PP2A regulatory (B or B′) subunit binding to AC dimers present in the cell, then one possible source of this unregulated activity in cells missing Cdc55p could be an excess activity of Rts1p-containing PP2A trimers. Consistent with this idea is the fact that overexpression of RTS1 in cdc55-null cells exacerbates the abnormal budding-morphology phenotype (46). Also, rts1Δ cdc55Δ strains exhibit no morphological-budding defects at any temperatures (46; H. Yang, unpublished results). While these observations are more consistent with Rts1-directed PP2A activity contributing to the mutant phenotype, other observations support a non-Rts1p-dependent PP2A phosphatase activity being responsible. For example, the strain deleted of the gene encoding Tpd3p, the A (structural) subunit, to which both catalytic and regulatory subunits bind, shows a mutant budding defect not unlike that shown by cdc55-null cells (57). As the coimmunoprecipitation of Rts1p with either of the catalytic subunits, Pph21p or Pph22p, does not occur in the absence of Tpd3p (46; Yang, unpublished results), this suggests that an Rts1p-independent phosphatase activity may elicit the mutant phenotype. Furthermore, as it has been shown that Pph21p and Pph22p can assemble into complexes with proteins other than the classically defined PP2A regulatory and structural subunits (11), it may be the elevated phosphatase activities of such complexes in cdc55-null cells that are responsible for the abnormalities of these cells. Whatever the relative contributions that Rts1-dependent and independent Pph21 and Pph22 phosphatase activities make in eliciting budding defects, what the substrates are that these phosphatases act upon in the cdc55-null strain to produce the mutant phenotype is completely unknown at this time.
Swe1p degradation is regulated at different rates at different times throughout the cell cycle, being highest during G2 and M (i.e., Swe1p is unstable) and lowest during G1 and S (48). These changes along with alterations in the Swe1 transcription rate (50) ensure that Swe1p accumulates during G1, peaking at S/G2, and then precipitously declines during G2 and M (31), thereby abolishing any further inhibition of Cdc28 kinase activity. While naturally oscillating, the turnover rate of Swe1p can also be temporarily adjusted in response to cellular checkpoint controls. For example, a temporary disruption of the actin cytoskeleton with latrunculin A during G1 or S, can bring about a temporary decrease in the Swe1p degradation rate, thereby maintaining inhibition of Cdc28 kinase activity and delaying progression of the cell cycle. The response to actin skeleton disruption does not, however, occur in cells at G2/M (31). As the abnormal change in Swe1p level in cdc55-null cells is found in mitosis-arrested cells but not S-phase-arrested cells, it would appear that deletion of CDC55 does not simply induce a constitutive actin disruption response.
It has recently been shown that the proteins Hsl1p, a kinase, and Hsl7p, a negative regulator of Swe1p with which it physically interacts, act in concert to target Swe1p for degradation, presumably by phosphorylating it (29, 48). Disruption of either or both of the genes encoding these proteins leads to a stabilization of Swe1p throughout an unperturbed cell cycle (29). Thus, accumulation of Swe1p in cdc55-null cells could conceivably be due to the loss of Hsl1p and/or Hsl7p function. The observation that the electrophoretic mobility shift of Swe1p was not as pronounced in cdc55-null cells upon mitotic entry (Fig. 4D), presumably due to a reduced level of phosphorylation, is reminiscent of what is observed in hsl1Δ cells (48). This suggests the possibility that Hsl1p kinase activity might be absent or reduced in cdc55Δ cells. However, we consider this unlikely for the following reasons. Deletion of the gene ELM1, which leads to a loss of Hsl1 kinase function (4, 12), has a synergistic effect with cdc55-null cells. If HSL1 function were already lost in a cdc55-null strain, it is not clear why eliminating ELM1 would necessarily make matters worse. In addition, hsl1Δ and hsl7Δ are each synthetically lethal with mih1Δ (29), while a strain which is cdc55Δ mih1Δ is completely viable (H. Yang, unpublished data). While these observations are inconsistent with a total loss of Hsl1 function in cdc55-null cells, further studies on strains mutant for both CDC55 and HSL1 (and possibly HSL7) would be required to establish any causative relationship between Hsl1 kinase function and the Swe1p stabilization seen in cdc55Δ cells.
Another pathway that could possibly be affected in cdc55-null cells is the one directly responsible for Swe1p degradation, namely Met30-SCF (SKP1-CDC53-F-box protein). Met30-SCF is a protein complex which ubiquitinates and therefore targets Swe1p for proteasome-mediated degradation (20). A genetic interaction between CDC55 and SCF function already exists: a deletion of GRR1, another Met30-like F-box protein known to activate the SCF complex (52), is synthetically lethal with cdc55Δ (22). It is possibly in this pathway that the uncontrolled PP2A phosphatase activity in cdc55-null cells is having its effect. This certainly is also testable.
We concluded that the contribution of Mih1 phosphatase activity to the higher level of Cdc28 Y19 phosphorylation seen in cdc55-null cells was likely to be minor. The in vivo phosphatase assay showed that, in cdc55-null cells, the MIH1 gene was functioning. Although in mitosis the Mih1p in cdc55Δ cells is hyperphosphorylated compared to the hypophosphorylated Mih1p in wild-type control cells, their specific phosphatase activities were not much different from each other based on our in vitro measurement. Coupled with the finding that cdc55Δ cells have amounts of Mih1p similar to those of wild-type cells, Mih1p phosphatase activity in cdc55Δ cells must be very close to that of wild-type cells.
While we assume that the abnormal budding morphology of cdc55-null cells is due primarily to ectopic Swe1p-induced inhibition of the Cdc28 kinase activity normally required for the apical/isotropic growth switch, is this the only factor? It is likely that elevated Swe1p is necessary, but not sufficient, for the following reasons. First of all, overproducing Swe1p arrests cells at G2/M with buds emerging from daughter cells (6), a phenotype unlike that of cdc55-null cells. Second, and most important, although Swe1p is essential for the cdc55-null abnormal morphology, Swe1p protein levels don't correlate with the severity of that phenotype (data not shown). The abnormal budding morphology of cdc55-null cells is temperature dependent; at 30°C in glucose medium, the majority of cells look normal, and the abnormal buds that are present are fairly short (46). As the temperature decreases, the frequency of cells with abnormal buds increases, as does the length of the buds. At 16°C essentially all cells have extremely long and abnormally shaped buds. If excess Swe1p were the only factor required for the manifestation of abnormal budding, then one might expect the Swe1p level in those cells at 16°C to be substantially greater than in cells growing at 30°C. This is not the case (data not shown). Thus, it has to be assumed that along with excess Swe1p levels in cdc55-null cells, some other temperature-dependent process must be affected in order to elicit the abnormal budding morphology.
While it is clear that levels of Swe1 mRNA are elevated somewhat in mitosis-arrested cdc55-null cells relative to wild-type controls (Fig. 6), that difference is insufficient to account for the differences seen in Swe1p levels. It is much more likely that an alteration in Swe1p turnover is the major cause of the hyperaccumulation of this protein in cdc55-null cells. As Swe1p has been shown to have, in some unknown way, a positive feedback effect on its own mRNA level (50), the stabilization of Swe1p may well be indirectly the cause of increased Swe1 mRNA levels in mitosis-arrested cdc55-null cells.
While earlier studies, based mainly on the inhibitory effects of okadaic acid, implicated PP2A activity as important in the regulation of mitotic events, we now report that a defined PP2A mutation can alter the turnover properties of a Wee1-like kinase activity during mitosis. This is noteworthy, as PP2A has been implicated as a negative regulator of mitotic entry in fission yeasts and higher eukaryotes and is suspected of doing so through the maintenance of high levels of Cdc2 Y15 phosphorylation. Recently it was reported that in the X. laevis egg, the PP2A-specific inhibitor okadaic acid can induce degradation of Xe-Wee1 at a time when it would normally be stabilized by the DNA replication checkpoint. Furthermore, the ability of okadaic acid to induce mitosis in S phase in the arrested cells was abolished by turning off a Cdc34-dependent ubiquitination function (32). This is certainly consistent with the notion that PP2A activity is required to maintain the SCF ubiquitinating complex activity directed against Xe-Wee1 at a low level until the completion of DNA synthesis. It will be interesting to see whether PP2A activity is also required for the regulation of turnover of Wee1-like kinases in other organisms.
ACKNOWLEDGMENTS
We thank those who supplied us with yeast strains, plasmids, and antibodies: Daniel Lew, Allen Myers, Mike Tyers, Adam Rudner, Jeff Bachant, Doug Kellogg, David Evans, David Pellman, and Dan Koshland. We are also indebted to Scott Erdman for allowing us to use his microscope facilities and assisting us in their usage. The critical review of this manuscript by the members of our lab and the continued helpful criticism from the members of the SUNY-HSC at Syracuse/Syracuse University yeast journal club were also greatly appreciated. We especially thank an anonymous reviewer whose thoughtful constructive criticism of an earlier manuscript helped make this a much stronger paper.
This work was supported by NSF grant MCB-9603733 (R.L.H.).
REFERENCES
- 1.Amon A, Surana U, Muroff I, Nasmyth K. Regulation of p34CDC28 tyrosine phosphorylation is not required for entry into mitosis in S. cerevisiae. Nature. 1992;23:368–371. doi: 10.1038/355368a0. [DOI] [PubMed] [Google Scholar]
- 2.Amon A, Tyers M, Futcher B, Nasmyth K. Mechanisms that help the yeast cell cycle clock tick: G2 cyclins transcriptionally activate G2 cyclins and repress G1 cyclins. Cell. 1993;74:993–1007. doi: 10.1016/0092-8674(93)90722-3. [DOI] [PubMed] [Google Scholar]
- 3.Barral Y, Parra M, Bidlingmaier S, Snyder M. Nim1-related kinases coordinate cell cycle progression with the organization of the peripheral cytoskeleton in yeast. Genes Dev. 1999;13:176–187. doi: 10.1101/gad.13.2.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Blacketer M J, Koehler C M, Coats S G, Myers A M, Madaule P. Regulation of dimorphism in Saccharomyces cerevisiae: involvement of the novel protein kinase homolog Elm1p and protein phosphatase 2A. Mol Cell Biol. 1993;13:5567–5581. doi: 10.1128/mcb.13.9.5567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Blasina A, Paegle E S, McGowan C H. The role of inhibitory phosphorylation of CDC2 following DNA replication block and radiation-induced damage in human cells. Mol Biol Cell. 1997;8:1013–1023. doi: 10.1091/mbc.8.6.1013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Booher R N, Deshaies R J, Kirschner M W. Properties of Saccharomyces cerevisiae wee1 and its differential regulation of p34CDC28 in response to G1 and G2 cyclins. EMBO J. 1993;12:3417–3426. doi: 10.1002/j.1460-2075.1993.tb06016.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Carroll C W, Altman R, Schieltz D, Yates J R, Kellogg D. The septins are required for the mitosis-specific activation of the Gin4 kinase. J Cell Biol. 1998;143:709–717. doi: 10.1083/jcb.143.3.709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cohen-Fix O, Peters J M, Kirschner M W, Koshland D. Anaphase initiation in Saccharomyces cerevisiae is controlled by the APC-dependent degradation of the anaphase inhibitor Pds1p. Genes Dev. 1996;10:3081–3093. doi: 10.1101/gad.10.24.3081. [DOI] [PubMed] [Google Scholar]
- 9.Coleman T R, Dunphy W G. Cdc2 regulatory factors. Curr Opin Cell Biol. 1994;6:877–882. doi: 10.1016/0955-0674(94)90060-4. [DOI] [PubMed] [Google Scholar]
- 10.De Brabander M, De May J, Joniau M, Geuens G. Ultrastructural immunocytochemical distribution of tubulin in cultured cells treated with microtubule inhibitors. Cell Biol Int Rep. 1977;1:177–183. doi: 10.1016/0309-1651(77)90038-8. [DOI] [PubMed] [Google Scholar]
- 11.Di Como C J, Arndt K T. Nutrients, via the Tor proteins, stimulate the association of Tap42 with type 2A phosphatases. Genes Dev. 1996;10:1904–1916. doi: 10.1101/gad.10.15.1904. [DOI] [PubMed] [Google Scholar]
- 12.Edgington N P, Blacketer M J, Bierwagen T A, Myers A M. Control of Saccharomyces cerevisiae filamentous growth by cyclin-dependent kinase Cdc28. Mol Cell Biol. 1999;19:1369–1380. doi: 10.1128/mcb.19.2.1369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Evangelista C C, Jr, Rodriguez Torres A M, Limbach M P, Zitomer R S. Rox3 and Rts1 function in the global stress response pathway in baker's yeast. Genetics. 1996;142:1083–1093. doi: 10.1093/genetics/142.4.1083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Evans D R, Stark M J. Mutations in the Saccharomyces cerevisiae type 2A protein phosphatase catalytic subunit reveal roles in cell wall integrity, actin cytoskeleton organization and mitosis. Genetics. 1997;145:227–241. doi: 10.1093/genetics/145.2.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hallberg E M, Shu Y, Hallberg R L. Loss of mitochondrial hsp60 function: nonequivalent effects on matrix-targeted and intermembrane-targeted proteins. Mol Cell Biol. 1993;13:3050–3057. doi: 10.1128/mcb.13.5.3050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Healy A M, Zolnierowicz S, Stapleton A E, Goebl M, Depaoliroach A A, Pringle J R. CDC55, a Saccharomyces cerevisiae gene involved in cellular morphogenesis: identification, characterization, and homology to the B subunit of mammalian type 2A protein phosphatase. Mol Cell Biol. 1991;11:5767–5780. doi: 10.1128/mcb.11.11.5767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Honigberg S M, Esposito R E. Reversal of cell determination in yeast meiosis: postcommitment arrest allows return to mitotic growth. Proc Natl Acad Sci USA. 1994;91:6559–6563. doi: 10.1073/pnas.91.14.6559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hwang L H, Lau L F, Smith D L, Mistrot C A, Hardwick K G, Hwang E S, Amon A, Murray A W. Budding yeast Cdc20: a target of the spindle checkpoint. Science. 1998;279:1041–1044. doi: 10.1126/science.279.5353.1041. [DOI] [PubMed] [Google Scholar]
- 19.Irniger S, Piatti S, Michaelis C, Nasmyth K. Genes involved in sister chromatid separation are needed for B-type cyclin proteolysis in budding yeast. Cell. 1995;21:269–278. doi: 10.1016/0092-8674(95)90337-2. [DOI] [PubMed] [Google Scholar]
- 20.Jin P, Gu Y, Morgan D O. Role of inhibitory CDC2 phosphorylation in radiation-induced G2 arrest in human cells. J Cell Biol. 1996;134:963–970. doi: 10.1083/jcb.134.4.963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kaiser P, Sia R A, Bardes E G, Lew D J, Reed R I. Cdc34 and the F-box protein Met30 are required for degradation of the Cdk-inhibitory kinase Swe1. Genes Dev. 1998;12:2587–2597. doi: 10.1101/gad.12.16.2587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kim Y J, Francisco L, Chen G C, Marcotte E, Chan C S. Control of cellular morphogenesis by the Ip12/Bem2 GTPase-activating protein: possible role of protein phosphorylation. J Cell Biol. 1994;127:1381–1394. doi: 10.1083/jcb.127.5.1381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kinoshita N, Yamano H, Niwa H, Yoshida T, Yanagida M. Negative regulation of mitosis by the fission yeast protein phosphatase ppa2. Genes Dev. 1993;7:1059–1071. doi: 10.1101/gad.7.6.1059. [DOI] [PubMed] [Google Scholar]
- 24.Köhrer K, Domdey H. Preparation of high molecular weight RNA. Methods Enzymol. 1991;194:398–401. doi: 10.1016/0076-6879(91)94030-g. [DOI] [PubMed] [Google Scholar]
- 25.Lee T H, Solomon M J, Mumby M C, Kirschner M W. INH, a negative regulator of MPF, is a form of protein phosphatase 2A. Cell. 1991;64:415–423. doi: 10.1016/0092-8674(91)90649-j. [DOI] [PubMed] [Google Scholar]
- 26.Lew D J, Reed S I. Morphogenesis in the yeast cell cycle: regulation by Cdc28 and cyclins. J Cell Biol. 1993;120:1305–1320. doi: 10.1083/jcb.120.6.1305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li R, Murray A W. Feedback control of mitosis in budding yeast. Cell. 1991;66:519–531. doi: 10.1016/0092-8674(81)90015-5. [DOI] [PubMed] [Google Scholar]
- 28.Lin F C, Arndt K T. The role of Saccharomyces cerevisiae type 2A phosphatase in the actin cytoskeleton and in entry into mitosis. EMBO J. 1995;14:2745–2759. doi: 10.1002/j.1460-2075.1995.tb07275.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.McMillan J N, Longtine M S, Sia R A, Theesfeld C L, Bardes E S, Pringle J R, Lew D J. The morphogenesis checkpoint in Saccharomyces cerevisiae: cell cycle control of Swe1p degradation by Hsl1p and Hsl7p. Mol Cell Biol. 1999;19:6929–6939. doi: 10.1128/mcb.19.10.6929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.McMillan J N, Sia R A, Bardes E S, Lew D J. Phosphorylation-independent inhibition of Cdc28p by the tyrosine kinase Swe1p in the morphogenesis checkpoint. Mol Cell Biol. 1999;19:5981–5990. doi: 10.1128/mcb.19.9.5981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.McMillan J N, Sia R A, Lew D J. A morphogenesis checkpoint monitors the actin cytoskeleton in yeast. J Cell Biol. 1998;21:1487–1499. doi: 10.1083/jcb.142.6.1487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Michael W M, Newport J. Coupling of mitosis to the completion of S phase through Cdc34-mediated degradation of Wee1. Science. 1998;282:1886–1889. doi: 10.1126/science.282.5395.1886. [DOI] [PubMed] [Google Scholar]
- 33.Michaelis C, Ciosk R, Nasmyth K. Cohesins: chromosomal proteins that prevent premature separation of sister chromatids. Cell. 1997;91:35–45. doi: 10.1016/s0092-8674(01)80007-6. [DOI] [PubMed] [Google Scholar]
- 34.Millward T A, Zolnierowicz S, Hemmings B A. Regulation of protein kinase cascades by protein phosphatase 2A. Trends Biochem Sci. 1999;24:186–191. doi: 10.1016/s0968-0004(99)01375-4. [DOI] [PubMed] [Google Scholar]
- 35.Minshull J, Straight A, Rudner A D, Dernburg A F, Belmont A, Murray A W. Protein phosphatase 2A regulates MPF activity and sister chromatid cohesion in budding yeast. Curr Biol. 1996;6:1609–1620. doi: 10.1016/s0960-9822(02)70784-7. [DOI] [PubMed] [Google Scholar]
- 36.Mizunuma M, Hirata D, Miyahara K, Tsuchiya E, Miyakawa T. Role of calcineurin and Mpk1 in regulating the onset of mitosis in budding yeast. Nature. 1998;392:303–306. doi: 10.1038/32695. [DOI] [PubMed] [Google Scholar]
- 37.Morgan D O. Principles of CDK regulation. Nature. 1995;374:131–134. doi: 10.1038/374131a0. [DOI] [PubMed] [Google Scholar]
- 38.Rhind N, Furnari B, Russell P. Cdc2 tyrosine phosphorylation is required for the DNA damage checkpoint in fission yeast. Genes Dev. 1997;11:504–511. doi: 10.1101/gad.11.4.504. [DOI] [PubMed] [Google Scholar]
- 39.Rhind N, Russell P. Tyrosine phosphorylation of cdc2 is required for the replication checkpoint in Schizosaccharomyces pombe. Mol Cell Biol. 1998;18:3782–3787. doi: 10.1128/mcb.18.7.3782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ronne H, Carlberg M, Hu G Z, Nehlin J O. Protein phosphatase 2A in Saccharomyces cerevisiae: effects on cell growth and bud morphogenesis. Mol Cell Biol. 1991;11:4876–4884. doi: 10.1128/mcb.11.10.4876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rose M D, Winston F, Hieter P. Laboratory course manual for methods in yeast genetics. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory Press; 1990. [Google Scholar]
- 42.Rothstein R J. One-step gene disruption in yeast. Methods Enzymol. 1983;101:202–211. doi: 10.1016/0076-6879(83)01015-0. [DOI] [PubMed] [Google Scholar]
- 43.Rudner A D, Murray A W. The spindle assembly checkpoint. Curr Opin Cell Biol. 1996;8:773–780. doi: 10.1016/s0955-0674(96)80077-9. [DOI] [PubMed] [Google Scholar]
- 44.Russell P, Nurse P. cdc25+ functions as an inducer in the mitotic control of fission yeast. Cell. 1986;11:145–153. doi: 10.1016/0092-8674(86)90546-5. [DOI] [PubMed] [Google Scholar]
- 45.Russell P, Nurse P. Negative regulation of mitosis by wee1+, a gene encoding a protein kinase homolog. Cell. 1987;22:559–567. doi: 10.1016/0092-8674(87)90458-2. [DOI] [PubMed] [Google Scholar]
- 46.Shu Y, Yang H, Hallberg E, Hallberg R L. Molecular genetic analysis of Rts1p, a B′ regulatory subunit of Saccharomyces cerevisiae protein phosphatase 2A. Mol Cell Biol. 1997;17:3242–3253. doi: 10.1128/mcb.17.6.3242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Shu Y, Hallberg R L. SCS1, a multicopy suppressor of hsp60-ts mutant alleles, does not encode a mitochondrially targeted protein. Mol Cell Biol. 1995;15:5618–5626. doi: 10.1128/mcb.15.10.5618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Shulewitz M J, Inouye C J, Thorner J. Hsl7 localizes to a septin ring and serves as an adapter in a regulatory pathway that relieves tyrosine phosphorylation of Cdc28 protein kinase in Saccharomyces cerevisiae. Mol Cell Biol. 1999;19:7123–7137. doi: 10.1128/mcb.19.10.7123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sia R A, Bardes E S, Lew D J. Control of Swe1p degradation by the morphogenesis checkpoint. EMBO J. 1998;17:6678–6688. doi: 10.1093/emboj/17.22.6678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sia R A, Herald H A, Lew D J. Cdc28 tyrosine phosphorylation and the morphogenesis checkpoint in budding yeast. Mol Biol Cell. 1996;7:1657–1666. doi: 10.1091/mbc.7.11.1657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sikorski R S, Hieter P. A system of shuttle vectors and yeast host strains designed for efficient manipulation of DNA in Saccharomyces cerevisiae. Genetics. 1989;122:19–27. doi: 10.1093/genetics/122.1.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Skowyra D, Craig K L, Tyers M, Elledge S J, Harper J W. F-box proteins are receptors that recruit phosphorylated substrates to the SCF ubiquitin-ligase complex. Cell. 1997;91:209–219. doi: 10.1016/s0092-8674(00)80403-1. [DOI] [PubMed] [Google Scholar]
- 53.Smythe C, Newport J W. Coupling of mitosis to the completion of S phase in Xenopus occurs via modulation of the tyrosine kinase that phosphorylates p34cdc2. Cell. 1992;68:787–797. doi: 10.1016/0092-8674(92)90153-4. [DOI] [PubMed] [Google Scholar]
- 54.Sneddon A A, Cohen P T, Stark M J. Saccharomyces cerevisiae protein phosphatase 2A performs an essential cellular function and is encoded by two genes. EMBO J. 1990;9:4339–4346. doi: 10.1002/j.1460-2075.1990.tb07883.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sorger P K, Murray A W. S-phase feedback control in budding yeast independent of tyrosine phosphorylation of p34cdc28. Nature. 1992;23:365–368. doi: 10.1038/355365a0. [DOI] [PubMed] [Google Scholar]
- 56.Stark M J. Yeast protein serine/threonine phosphatases: multiple roles and diverse regulation. Yeast. 1996;12:1647–1675. doi: 10.1002/(SICI)1097-0061(199612)12:16%3C1647::AID-YEA71%3E3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 57.van Zyl W, Huang W, Sneddon A A, Stark M, Camier S, Werner M, Marck C, Sentenac A, Broach J R. Inactivation of the protein phosphatase 2A regulatory subunit A results in morphological and transcriptional defects in Saccharomyces cerevisiae. Mol Cell Biol. 1992;12:4946–4959. doi: 10.1128/mcb.12.11.4946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Walter G, Mumby M. Protein serine/threonine phosphatases and cell transformation. Biochim Biophys Acta. 1993;1155:207–226. doi: 10.1016/0304-419x(93)90005-w. [DOI] [PubMed] [Google Scholar]
- 59.Wang Y, Burke D J. Cdc55p, the B-type regulatory subunit of protein phosphatase 2A, has multiple functions in mitosis and is required for the kinetochore/spindle checkpoint in Saccharomyces cerevisiae. Mol Cell Biol. 1997;17:620–626. doi: 10.1128/mcb.17.2.620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wera S, Hemmings B A. Serine/threonine protein phosphatases. Biochem J. 1995;311:17–29. doi: 10.1042/bj3110017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wittenberg C, Sugimoto K, Reed S I. G1-specific cyclins of S. cerevisiae: cell cycle periodicity, regulation by mating pheromone, and association with the p34CDC28 protein kinase. Cell. 1990;62:225–237. doi: 10.1016/0092-8674(90)90361-h. [DOI] [PubMed] [Google Scholar]
- 62.Yamamoto A, Guacci V, Koshland D. Pds1p, an inhibitor of anaphase in budding yeast, plays a critical role in the APC and checkpoint pathway(s) J Cell Biol. 1996;133:99–110. doi: 10.1083/jcb.133.1.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zachariae W, Shin T H, Galova M, Obermaier B, Nasmyth K. Identification of subunits of the anaphase-promoting complex of Saccharomyces cerevisiae. Science. 1996;274:1201–1204. doi: 10.1126/science.274.5290.1201. [DOI] [PubMed] [Google Scholar]