Abstract
Autism spectrum disorder (ASD) is typically characterized by common deficits in social skills and repetitive/stereotyped behaviors. It is widely accepted that genetic and environmental factors solely or in combination cause ASD. However, the underlying pathogenic mechanism is unclear due to its highly heterogeneous nature. To better understand the pathogenesis of ASD, various animal models have been generated, which can be generally divided into genetic, environment-induced, and idiopathic animal models. In this review, we summarize the common animals used for ASD study and then discuss the applications, clinical insights, as well as challenges and prospects of current ASD animal models.
Keywords: Autism spectrum disorders, Animal models, Neurodevelopment, Genetics, Environment
INTRODUCTION
Autism spectrum disorder (ASD) comprises a clinically heterogeneous group of disorders characterized by social deficits, narrow interests, stereotyped behaviors, and verbal and non-verbal communication difficulties (Sandin et al., 2017). The worldwide incidence of ASD has dramatically increased in the past several decades, increasing from 1/150 (0.67%) in 2000 to 1/54 (1.85%) in 2016 in American children, with a 4.3 times higher incidence in boys than in girls (Maenner et al., 2020). In China, the most extensive population-based study (~120 000 children) indicates that the prevalence of ASD in Chinese children aged 6 to 12 is ~0.70%. According to China’s 2016 national census data, this estimate translates to approximately 700 000 Chinese children (6 to 12 years) with ASD (Zhou et al., 2020). As early as 1943, American pediatric psychiatrists reported on clinical autistic symptoms in 11 children, which became an important basis for the diagnosis of ASD (Kanner, 1968). However, there is still a lack of practical methods for the diagnosis and treatment of ASD, mainly due to high disease heterogeneity. ASD is not only a medical problem for patients but also an urgent social problem, imposing a heavy mental and financial burden on families and society (Manoli & State, 2021). Therefore, systematic investigations on the pathogenic regulators of ASD are critical to provide theoretical and experimental support for the development of new clinical diagnosis, treatment, and intervention measures.
As indicated in twin and family studies, development of ASD is predominantly attributed to genetic factors (Sandin et al., 2017; Tick et al., 2016). Dozens of rare Mendelian disorders, including fragile X syndrome, neurofibromatosis, Rett syndrome, tuberous sclerosis complex, and structural chromosomal variations, are also considered high risk factors for the development of ASD (Bourgeron, 2015; Geschwind & State, 2015). Genome-wide studies have identified hundreds of risk loci for ASD, with a considerable number also related to other neurodevelopmental diseases such as schizophrenia and intellectual disability (ID) (Geschwind & State, 2015). Copy number variants (CNVs) are another major factor responsible for ASD etiology (Chung et al., 2014). Recent studies have also revealed the contribution of rare de novo single nucleotide mutations, which together contribute significantly to the pathogenesis of ASD (Bourgeron, 2015; Gaugler et al., 2014; Geschwind & Flint, 2015; Geschwind & State, 2015). Dysregulated immune response during early embryonic development due to environmental factors, such as chemical exposure, infection, inflammation, and emotional health of pregnant mothers, can also increase ASD risk (Ornoy et al., 2015).
Given the difficulties in obtaining samples from ASD patients, animal models that restore the clinical features of ASD are the best choice for exploring the pathogenic regulators of ASD (Wintler et al., 2020). The establishment and selection of a stable and reliable animal model of ASD is critical for elucidating the relationship between the central nervous system (CNS) and ASD pathogenesis and for investigating pathophysiological processes, behavioral changes, diagnosis, and treatment. To date, many ASD animal models have been developed, and each animal species and modeling method has its own unique advantages and disadvantages. In this review, we first summarize the standard animals used to study ASD, and then discuss the applications and clinical insights of ASD animal models based on genetic and environmental factors. Finally, we discuss the challenges and prospects of current ASD animal models. This review should provide a better understanding of ASD research and treatment.
COMMON ANIMALS USED IN ASD STUDY
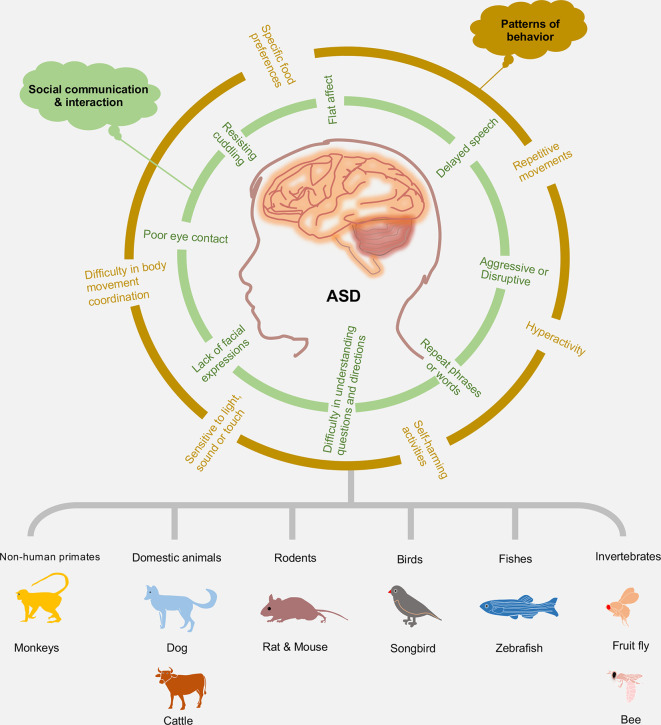
Impaired social relationships and repetitive/stereotypic behaviors distinguish ASD from other developmental disorders (Rapin & Tuchman, 2008; Yenkoyan et al., 2017). Therefore, advances in the pathophysiology of ASD symptoms are classified into two categories: social communication/interaction and patterns of behavior (Mukherjee, 2017; Peretti et al., 2019) (Figure 1). Social communication and interaction deficits include poor eye contact, lack of facial expressions, flat affect, delayed (or no) speech, difficulty in understanding questions and directions, aggressive or disruptive, repeated phrases or words, and resistance to cuddling and holding. Patterns of behavior are highlighted as repetitive movements like handshaking, spinning, or rocking; hyperactivity; difficulty in body movement coordination; sensitivity to light, sound, or touch; self-harming activities such as head-banging; and specific food preferences or food patterns.
Figure 1.
Diverse animal models of ASD
Advances in understanding of ASD symptom pathophysiology can be classified into two categories: social communication/interaction and patterns of behavior. Non-human primates, domestic animals, rodents, birds, fish, and invertebrates are the most commonly used in ASD research.
Non-human primates (NHPs), domestic animals, rodents, birds, fish, and invertebrates have been used as animal models to mimic the clinical features of ASD (Figure 1; Table 1). In this section, we summarize those animals used in ASD study and how their research advantages benefit our understanding of the pathogenesis of ASD.
Table 1. Advantages and limitations of different animals in ASD modeling.
| Category | Animal | Advantage | Limitation |
| Non-human primates | Rhesus, cynomolgus | Relatives of humans, similar brain structure, tracible eye contact | Expensive and inconvenient experiment operation |
| Domestic animals | Dogs, cattle | Similarity of human in core clinical phenotypes, temperament, brain structure, physiology, nutritional metabolism, and ethics | Large size, expensive, inconvenient experiment operation |
| Rodents | Mice, rats, and prairie voles | Relatively low cost, powerful genetic tools, suitable for drug screening and preclinical study | No sulcus and gyrus structure |
| Songbird | Zebra finch | Mimic language defect | Brain is underdeveloped |
| Fish | Zebrafish | Low cost, stereotypical behavior | Low homology, large differences in organizational structure |
| Invertebrates | Fruit fly, nematode, bee, Aplysia | Low cost, powerful genetic tools, stereotypical behavior, habituation behavior, suitable for large-scale screening for ASD risk genes and drugs | Large differences in brain structure |
NHPs
As animal models, monkeys, including rhesus (Macaca mulatta) and crab-eating macaques (M. fascicularis), best simulate human social behavior. Compared with rodents that have evolved for more than 70 million years, macaques separated from human evolution nearly 25 million years ago, and thus show greater similarity to humans in terms of genetics, neurobiology, and behavior (Kumar & Hedges, 1998; Rat Genome Sequencing Project Consortium, 2004).
Macaques mature about four times faster than humans. However, the early social development of macaque infants is similar to that of human infants in many aspects (Weinstein & Capitanio, 2012; Weinstein et al., 2014), including complex social groupings, communication through facial expressions, body language, and sound (Testard et al., 2021), and behavioral defects (Ghazanfar & Santos, 2004). In addition, several key brain regions associated with social behavior in macaques are similar to those in humans (Bauman & Schumann, 2018). For example, the amygdala has a very similar nuclear structure, neurochemical distribution, connectivity, and functional characteristics as humans and other NHPs (Rutishauser et al., 2015), whereas nuclear distribution differs significantly in rodent brains (Chareyron et al., 2011; Pitkänen & Kemppainen, 2002).
The Social Responsiveness Scale (SRS) provides a quantitative measure of behavioral variability to identify individuals who do not meet the diagnostic criteria for ASD but still exhibit atypical social behaviors compared to the general population (Constantino et al., 2006). It is adaptive to NHPs. For instance, well-trained caregivers using the monkey SRS can identify macaques that exhibit atypical social response patterns related to social avoidance, social anxiety/rigidity, lack of social self-confidence, and social embarrassment (Feczko et al., 2016). Moreover, chromosome-scale genome assemblies of NHPs not only allow us to better understand these animal models but also provide an important basis for human biomedical research (Jayakumar et al., 2021). However, although NHP models can bridge rodent models of human diseases, increased costs and ethical considerations limit their use in research.
Domestic animals
Several studies have proposed that domestic animal models are complementary to traditional rodent models. For example, the dog is an innovative and unique model for many human neuropsychiatric diseases, including ASD (Bunford et al., 2017; Sándor et al., 2021). The advantages of dog models include: (a) significant inter-individual differences in social cognitive performance in dogs; (b) greater phenotypic similarity between dogs and humans than between rodents and humans; (c) symptoms that are functionally similar to the human condition; and (d) similar causes in dogs than in rodents (Topál et al., 2019). Cattle have also been utilized to study the molecular mechanism underlying ASD. A recent study tested enrichment of polymorphisms associated with cattle temperament in genes involved in four characteristics of human psychosis and personality disorder, including schizophrenia, ASD, neuroticism, and developmental delay disorder (Costilla et al., 2020). As relatively advanced mammals, domestic animals have a similar brain structure, physiology, and nutritional metabolism to humans. However, due to their large size, their maintenance and experimental operation can be inconvenient and expensive.
Rodents
Rodents (namely mice and rats) have similar neuroanatomy, biochemistry, electrophysiology, and genetics to humans. As classic animal models, they are widely used in basic scientific studies and preclinical trials due to the advantages of low price, short pregnancy, and many offspring (Simmons et al., 2021). Other rodents, such as prairie voles, have a stable spousal relationship and parental behavior, which is beneficial for studying social cognitive defects and ASD (Donovan et al., 2020). Most current ASD animal models are generated in mice and rats and will be discussed in detail in the following sections.
Birds
Little is known about the neural and genetic basis of human language development and related neurodevelopmental disorders (including ASD), in which language deficits are considered a comorbidity. Although no animal model can fully capture the behavior and genetic complexity of ASD, as an experimental language learning animal model, songbirds can supplement the shortcomings of rodent genetic models and provide essential insights into communication deficiencies. For instance, zebra finches communicate through learned vocalizations and can be used to simulate language communication disorders (Ahmadiantehrani & London, 2017; Hacohen-Kleiman et al., 2020; Panaitof, 2012). Moreover, recent genomic research has identified potential genetic substrates for the evolution and regulation of vocal communication (Warren et al., 2010). Furthermore, expression of the ASD gene FOXP1 is reported to cause severe impairment in speech/language learning and affect the cultural transmission of bird songs between adult and juvenile zebra finches (Garcia-Oscos et al., 2021).
Fish
Zebrafish serve as a model system due to their precocious development, small transparent larval morphology, sensitive pharmacology, and genetic and physiological similarities to humans (de Abreu et al., 2020; James et al., 2021). For neurological research, zebrafish have been widely used in studies on brain development, synaptic growth, and other functions that regulate the CNS, making it a powerful tool for studying ASD (de Abreu et al., 2021; Gawel et al., 2020; Rea & Van Raay, 2020). With homologous preference and community aggregation, zebrafish can be used to study social behaviors (Tang et al., 2020). The phenotypic characteristics of zebrafish can be reliably evaluated by automatically reconstructing three-dimensional swimming trajectories (Stewart et al., 2015).
Invertebrates
The fruit fly (Drosophila melanogaster) is a unique and powerful genetic model organism with a high degree of genetic conservatism and easy genome manipulation, and thus can be used to study a wide range of biological issues (Cheng & Chen, 2018). Drosophila can help identify genes associated with ASD (Hope et al., 2019). For instance, Drosophila fed different doses of bisphenol propane show more repetitive behavior (grooming behavior) and abnormal social interactions (shorter distance from individual flies) compared with the controls (Kaur et al., 2015). Recent research on 286 gene orthologs implicated in ID with or without comorbid ASD, specifically in Drosophila neurons, with light-off jump habituation testing, identified nearly 100 ID genes that regulate habituation learning, with some “habituation-deficient” genes implicated in ASD (Cheng & Jin, 2019; Fenckova et al., 2019).
In addition to Drosophila, other invertebrates such as bees (Kocher et al., 2018), Caenorhabditis elegans (Calahorro & Ruiz-Rubio, 2011), and Aplysia (Choi et al., 2011) have been used for ASD study. Although there are significant differences in brain structure between invertebrates and humans, these animals can be utilized as models to study the underlying mechanism of ASD.
DIVERSE ANIMAL MODELS EXPLORING KEY FEATURES OF ASD
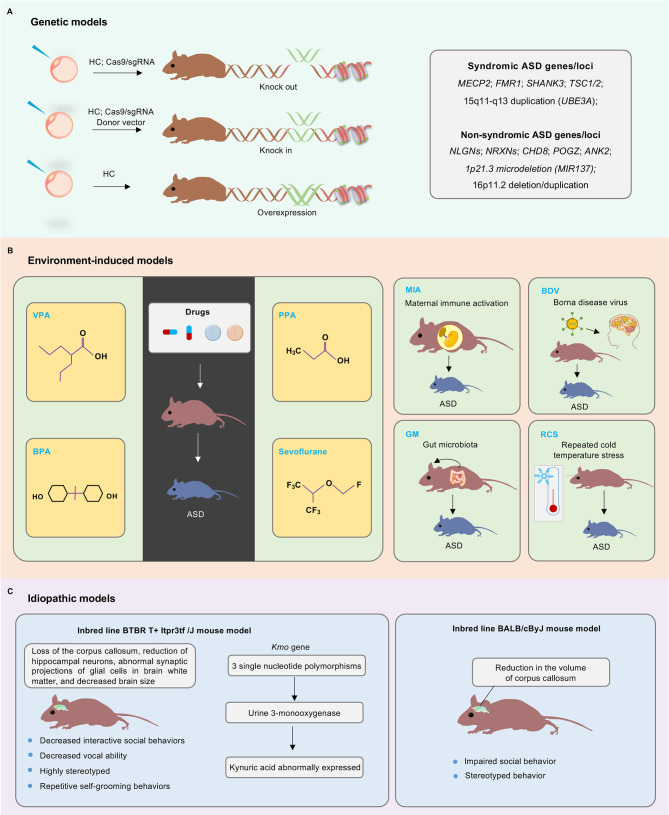
Based on different construction methods, animal models of ASD mainly include genetic models, environment-induced models, and idiopathic models (Figure 2).
Figure 2.
Schematic of major animal models of ASD
ASD animal models mainly include genetic, environment-induced, and idiopathic models. A: Developed technologies, such as homologous recombination (HC) and CRISPR/Cas9, have been used to generate ASD risk gene knock-in, knockout, and overexpressed animal models. B: Environmental factors can act as critical pathogenic regulators in ASD etiology, including valproic acid (VPA), propionic acid (PPA), bisphenol propane (BPA), sevoflurane, maternal immune activation (MIA), borna disease virus (BDV), and repeated cold temperature stress (RCS). C: Idiopathic models include BTBR T+Itpr3tfJ (BTBR) and BALB/cByJ mouse strains. BTBR mice mainly show decreased interactive social behaviors, decreased vocal ability, and highly stereotyped and repetitive self-grooming behaviors. Abnormal behaviors in BTBR mice are primarily caused by three single nucleotide polymorphisms in the Kmo gene. BALB/cByJ is another inbred mouse strain that shows significant social disorder and stereotyped behavior. Imaging studies have demonstrated that BALB/cByJ mice have a reduction corpus callosum volume.
Genetic models
The genetic risks of ASD are conferred by hundreds of genes (Satterstrom et al., 2020), broadly affecting various biological processes, such as synaptic function and neuronal activity, postsynaptic density protein metabolism, neuronal cell adhesion, WNT signaling, and chromatin remodeling in the process of neurogenesis (Abrahams & Geschwind, 2008; Miles, 2011). Advanced sequencing and gene-editing technologies, such as homologous recombination and CRISPR/Cas9, have together resulted in the rapid development of genetic animal models of ASD (Figure 2A). To date, more than 200 ASD risk genes/loci are targeted to generate corresponding animal models (https://gene.sfari.org/database/animal-models/genetic-animal-models/). In this section, we discuss the animal models of several typical syndromic and non-syndromic ASD genes/loci. Many of these genetic models have been used to examine the rescue effectiveness of pharmaceutical agents, as summarized in Table 2.
Table 2. Genetic animal models targeting classical ASD risk genes.
| Genes | Neuronal function | Clinical relevancea | Rescue agentc | References | |||||||||||||||||
| Typical symptoms | Animal performancesb | ||||||||||||||||||||
| ① | ② | ③ | ④ | ⑤ | ① | ② | ③ | ④ | ⑤ | Genetic | Pharmaceutical | ||||||||||
| a: M: Monkey; R: Rodents; F: Fish; I: Invertebrates. b: ①: Social communication disorders; ②: Repetitive/stereotyped behaviors; ③: Narrow interests; ④: Impaired linguistics; ⑤: Intellectual disability. +: Positive, –: Negative. c: Based on the public data from SFARI GENE: https://gene.sfari.org/database/animal-models/rescue-animal-models. | |||||||||||||||||||||
| Syndromic | |||||||||||||||||||||
| MECP2 | Starts or inhibits transcription; neuron maturation; regulated by development | + | + | + | + | + | M | + | + | + | – | + | One copy knocked out of Crh, or Crhr1, or Oprm; Reducing Mecp2 expression from a transgene, MECP2-dNIC; Reinstatement of Mecp2 | 7,8-Dihydroxyflavone; Acetyl-L-carnitine; D-Cycloserine; CX546; Antalarmin; Cysteamine; RU-486; Isoguvacine; Midazolam; Fenobam; SCH-23390; SCH-39166 | Achilly et al., 2021; Zhan et al., 2021 | ||||||
| R | + | + | + | + | + | ||||||||||||||||
| F | + | + | + | – | + | ||||||||||||||||
| I | + | + | + | – | + | ||||||||||||||||
| FMR1 | Involves in translation; affects neuronal proliferation and migration | + | + | + | + | + | R | + | + | + | + | + | Inactivation of one allele of App gene; inactivation of one copy of S6K1; inactivation of S6K1 gene | Cyclosporine-A; Dexpramipexole; Isoguvacine; CTEP; Bryostatin1; MPEP; BW-723C86; MDL-11,939; JQ1; Sulpiride; CX-4945, Recombinant IL-17a, Lithium; Minocycline; Lipopolysaccharide; Bumetanide; FRAX486 | Dockendorff et al., 2002; Kim et al., 2014b; Reynolds et al., 2021; Schiavi et al., 2020 | ||||||
| F | + | + | + | – | + | ||||||||||||||||
| I | + | + | + | – | + | ||||||||||||||||
| SHANK3 | Promotes formation, maturation, and stability of dendritic spines | + | + | + | + | + | M | + | + | + | – | + | Reinstatement of Shank3; beta-catenin shRNA; constitutively active Rac1 | CDPPB; MPEP; Insulin-like growth factor 1 and its peptide derivative; CX-546; Lipopolysaccharide; Cocaine; Isoguvacine; D-Cycloserine; 7,8-Dihydroxyflavone; TG003; Valproic acid; Aripiprazole; Clozapine; Romidepsin; Fluoxetine; Phospho-cofilin peptide; Oxytocin; Risperidone; Trichostatin A | Han et al., 2013; Kozol et al., 2015; Liu et al., 2018; Matas et al., 2021; Mossa et al., 2021 | ||||||
| R | + | + | + | + | + | ||||||||||||||||
| F | + | + | + | – | + | ||||||||||||||||
| I | + | + | + | – | + | ||||||||||||||||
| TSC1/2 | Regulates mTORC1 pathway, neuronal differentiation, and Purkinje cell excitability | + | + | + | + | + | R | + | + | + | + | + | – | D-Cycloserine, Fenobam, SCH-23390, SCH-39166, Rapamycin | Normand et al., 2013; Tsai et al., 2012 | ||||||
| UBE3A | Regulates neuronal homeostatic synaptic plasticity | + | + | + | + | + | R | + | + | + | – | + | Reinstatement of Ube3a in GABAergic neurons | Cbln1 expression; THIP; Pamin | Krishnan et al, 2017; Xu et al, 2018 | ||||||
| Non-syndromic | |||||||||||||||||||||
| NLGNs | Regulates the formation of hippocampal neurons and post-glutamatergic synapse proteins | + | + | + | + | + | R | + | + | + | + | + | Nlgn3,shRNAs against Nlgn1,shRNAs against Nlgn2,DIO-NL3 transgene in nucleus accumbens,Optogenetic activation of Pvalb interneurons, Nlgn3 reexpression | ETC-168, ACEA, HU-210, Quinpirole, URB597 | Camacho et al., 2014; Kolozsi et al., 2009; Sledziowska et al., 2020 | ||||||
| NRXNs | Encodes neuronal transmembrane protein; interacts glial cells | + | + | + | + | + | R | + | + | + | + | + | – | – | Etherton et al., 2009; Tromp et al., 2021 | ||||||
| CHD8 | Controls epigenetic and transcript regulation; affects brain phenotype | + | + | + | + | + | R | + | + | + | – | + | Human CHD8; stabilized Ctnnb1(S37A) | CPI-455; Oxytocin | Kunkel et al, 2018; Platt et al, 2017; Sugathan et al, 2014 | ||||||
| F | – | – | – | – | – | ||||||||||||||||
| POGZ | Regulates neuronal development | + | + | + | + | + | R | + | + | + | – | – | – | NBQX; Oxytocin; Perampanel | Matsumura et al, 2020; Suliman-Lavie et al, 2020; White et al, 2016 | ||||||
| ANK2 | Affects axonal branching; regulates postnatal development of excitatory synapses | + | + | + | – | – | R | + | + | + | – | – | – | – | Iossifov et al, 2014; Sztainberg et al, 2016 | ||||||
| MIR137 | Regulates neuronal gene expression and neurogenesis | + | + | + | + | + | R | + | + | + | – | – | Ablation of Pde10a | Papaverine | Cheng et al., 2018 | ||||||
| 16p11.2 | Controls prefrontal connectivity; endothelium-dependent structural and functional neurovascular | + | + | + | + | + | R | + | + | + | + | + | – | – | Portmann et al, 2014; Yin et al, 2021 | ||||||
| F | – | – | – | – | – | ||||||||||||||||
Syndromic ASD genes:MECP2: Rett syndrome (RTT) is a progressive neurodevelopmental disorder that causes mental retardation, with an incidence of 1/10 000–15 000 (Amir et al., 1999). It is frequently classified within ASD given their shared clinical symptoms of repetitive movements, impaired motor coordination, and social withdrawal. The primary cause of RTT is mutation of the methyl CpG binding protein 2 (MECP2) gene, a typical monogenic cause of ASD. MECP2 is located on the X chromosome, and MECP2 mutation in males is usually lethal; thus, RTT affects females almost exclusively (Amir et al., 1999). Mouse models of MECP2 mimic the symptoms of RTT, including impaired social behavior (Amir et al., 1999; Li et al., 2020a; Orefice et al., 2019; Pizzamiglio et al., 2021).
At present, classic Mecp2-mutant mice include deletion of exon 3 (Chen et al., 2001), deletion of exons 3 and 4 (Guy et al., 2001), and 308X point mutations (Shahbazian et al., 2002). Although Mecp2-knockout mice show most of the developmental and behavioral defects of patients with RTT, identifying ASD-like behaviors in Mecp2-overexpressing mouse models can be challenging (Collins et al., 2004). Mecp2-knockout mice have also been used to explore the regulatory mechanisms of other ASD risk genes, such as Mir137, whose expression is controlled by Mecp2 in a promoter binding-dependent manner (Szulwach et al., 2010). In rat models, loss of Mecp2 results in growth retardation, malocclusion, lack of exercise, weak forelimb grip, and significant social communication defects (Wu et al., 2016).
Transgenic cynomolgus monkeys (Macaca fascicularis) overexpressing human MECP2 show ASD-like behaviors and stable inheritance of transgenic germlines (Liu et al., 2016). Moreover, a MECP2 mutant cynomolgus monkey model generated using transcriptional activator-like effector nuclease (TALEN) technology to edit the MECP2 gene (Chen et al., 2017) has been applied to explore related phenotypes using unique eye-tracking tests and magnetic resonance imaging analysis, showing similar physiological, behavioral, and brain structural abnormalities to those in ASD patients (Zhang et al., 2019). Recently, the circuit abnormalities related to MECP2 and autism-like traits in monkeys have been mapped to homogenous ASD subgroups, thus providing a new strategy to deconstruct the clinical heterogeneity of ASD (Cai et al., 2020). These studies suggest that NHP ASD models are feasible and reliable and can be applied to study ASD-related neural mechanisms and potential therapeutic interventions.
FMR1: Fragile X syndrome (FXS) is mainly caused by the excessive expansion of the CGG trinucleotide in the 5'UTR of the fragile X mental retardation 1 (FMR1) gene, with a small portion caused by point mutations of the FMR1 gene (Richter & Zhao, 2021). Boys tend to exhibit more severe clinical symptoms than girls, including ID, speech delay, anxiety, attention deficit disorder, hyperactivity, seizures, and physical deformities (Richter & Zhao, 2021). Similar to MECP2, FMR1 is considered as another monogenic cause of ASD.
Loss of Fmr1 in male mice is not lethal. The dendritic spines of cerebral cortex neurons in Fmr1 knockout mice are longer, thinner, and more curved than those in wild-type mice, and the dendritic spines of the apical dendrites are denser (Comery et al., 1997). In vivo two-photon calcium imaging found that the adaptability of neurons in Fmr1-knockout mice to repeated whisker stimulation is inadequate, suggesting that adaptive impairment of the cortical sensory circuit may be a potential cause of ASD tactile defense (He et al., 2017). In addition to Fmr1-knockout mouse models, several clinically relevant mouse models have been generated. For example, the recurrent missense mutations in FMR1 encoding protein FMRP (FMRP-R138Q) are reportedly associated with FXS (Collins et al., 2010). In a knock-in mouse model expressing the FMRP-R138Q protein, neuronal density increasing in the hippocampus is related to synaptic ultrastructural defects and increased surface expression of AMPA receptors (Prieto et al., 2021). Based on biochemical analysis, high-resolution imaging, electrophysiological recording, and behavioral testing, the R138Q mutation can lead to impaired long-term hippocampal enhancement and social cognitive deficits in mice (Prieto et al., 2021). Social dominance and hierarchy changes in Fmr1-knockout rats have been evaluated through a penetrating experiment, providing new insight for understanding complex social dynamics in an Fmr1-dependent manner (Saxena et al., 2018).
SHANK3: SH3 and multiple ankyrin repeat domains protein 3 (SHANK3) is a postsynaptic density protein that interacts with a variety of ionotropic and metabotropic glutamate receptors and is associated with the actin cytoskeleton (Duffney et al., 2013). SHANK3 mutation is associated with ASD and Phelan-McDermid syndrome, with the latter characterized by global brain retardation, mental disability, speech delay or loss, and mild deformities (Tatavarty et al., 2020; Wang et al., 2020). In Shank3-knockout mice, the cortex-striatum-thalamic loop in mutant mice is overactive, leading to impaired social behaviors (Wang et al., 2016). Early recovery of Shank3 expression in Shank3-knockout mice can prevent the ASD-like behavior phenotype (Jaramillo et al., 2020). Loss of Shank3 in male rats does not lead to the enhanced social approach behavior that typically occurs after playback of pro-social ultrasonic vocalizations (Berg et al., 2018).
In addition to rodent models, the first heritable Shank3b-mutant zebrafish model also show ASD-like behavior and changes in synaptic protein homer1 and synaptophysin levels (Liu et al., 2018). Moreover, SHANK3-knockout cynomolgus monkeys have been generated (Tu et al., 2019; Zhou et al., 2019b), which show motor deficits, repetitive behaviors, social and learning disorders, and sleep disorders.
TSC1/2: Tuberous sclerosis complex (TSC) is an autosomal dominant disease characterized by tuber-like benign tumors in multiple organs (including the brain and kidney). It can develop into malignant tumors, often accompanied by ASD (Lam et al., 2018). TSC1 or TSC2 mutation can lead to a high incidence of complications in ASD, and the protein product dimerizes and negatively regulates mTOR signaling transduction in mammalian targets.
The cerebellum primarily controls motor activity, while its non-motor functions are also associated with psychiatric disorders, such as ASD (Wolf et al., 2009). For example, compared with TSC patients without ASD, TSC patients with ASD show hypermetabolism in the deep structure of the cerebellum based on functional imaging. Furthermore, compared with the control group, human induced pluripotent stem cell (hiPSC)-derived Purkinje cells (major cerebellum neurons) from patients with pathogenic TSC2 mutations exhibit excessive mTORC1 pathway activation, neuronal differentiation deficiency, RNA regulation deficiency, decreased excitability, and lower synaptic activity (Sundberg et al., 2018). However, upon loss of TSC2, the expression levels of FMRP, glutamate receptor δ2 (GRID2), and pre- and post-synaptic markers (such as synaptophysin and PSD95) decrease in hiPSC-derived Purkinje cells (Sundberg et al., 2018). In mouse models, following specific deletion of Tsc1 or Tsc2 in Purkinje cells, mice exhibit a battery of ASD behaviors, including impaired memory, repetitive behaviors, and altered vocalizations (Reith et al., 2013; Tsai et al., 2012). Since the absence of Purkinje cells is one of the most common anatomical abnormalities in ASD patients, this model provides experimental support for related pathophysiological studies.
15q11–q13 duplication (UBE3A): Among the known causes of ASD, duplication of human chromosome 15q11–q13, which contains large repeat sequences, is the most frequently associated cytogenetic abnormality. Disruption of this region causes different disorders, including autism, Angelman syndrome, and Prader-Willi syndrome (Pinto et al., 2010). Duplications of chromosome 15q11–q13 account for up to 3% of ASD, where increased levels of UBE3A, an E3 ubiquitin ligase, are usually observed (Baron et al., 2006; Smith et al., 2011). In patients with ASD, a mutation that impairs UBE3A phosphorylation (p. T485A) can cause elevated UBE3A activity (Iossifov et al., 2014; Yi et al., 2015). Increased expression of UBE3A negatively regulates the function of cerebellin 1 precursor (Cbln1) in the ventral tegmental area, resulting in impaired synaptic transmission and sociability (Krishnan et al., 2017). In mice, ASD-like behaviors can be induced when expressing excessive UBE3A, resulting in impaired retinoic acid (RA)-mediated neuronal homeostatic synaptic plasticity (Xu et al., 2018). UBE3A can regulate ALDH1A2, the rate-limiting enzyme of RA synthesis, and administration of an ALDH1A antagonist can rescue impaired social behaviors associated with ASD (Xu et al., 2018). Other genes involved in 15q11–q13 and their corresponding animal models have been reviewed extensively (see Takumi, 2011).
Non-syndromic ASD genes:NLGNs: Neuroligins (NLGNs) are cell adhesion molecules on the postsynaptic membrane and consist of excitatory and inhibitory synapses. NLGNs interact with neurexins (NRXNs) to promote the formation of presynaptic and posterior membranes (Vieira et al., 2021). The five NLGN genes expressed in humans (NLGN1, NLGN2, NLGN3, NLGN4X, and NLGN4Y) are all associated with ASD, with NLGN3 and NLGN4 most closely related to ASD symptoms (Heshmati et al., 2018). Indeed, animal models suggest that NLGN-gene knockout can cause synaptic changes and abnormalities in neurotransmitters of the brain, leading to the emergence of ASD symptoms (Südhof, 2017).
Both Nlgn1-knockout and Nlgn1-P89L-knock-in mice show spatial memory impairment, but only Nlgn1-knockout mice show vigorous combing and stereotyped behavioral phenotypes (Blundell et al., 2010; Nakanishi et al., 2017). Nlgn2-knockout mice also exhibit ASD-like behaviors, normal social behaviors, increased anxiety-like behaviors, decreased pain sensitivity, and poor motor coordination (Wöhr et al., 2013).
Mice with Nlgn3 gene deletion show impaired ultrasound vocalization and social deficits (Modi et al., 2019; Radyushkin et al., 2009). Mutation in Nlgn3 can lead to impaired oxytocin signaling in dopaminergic neurons and changes in the behavioral responses of mice to social novelty tests (Hörnberg et al., 2020). Additionally, Nlgn3-R451C-knock-in mice exhibit ASD-related behavioral phenotypes, including deficiency in social novelty preference, hyperactivity, and repetitive behavior, indicating that Nlgn3 mutations damage striatal circuits, leading to repetitive behavior (Rothwell et al., 2014). Other studies suggest that abnormal gamma oscillations in the prefrontal cortex may be a leading cause of social behavior disorders in Nlgn3-R451C-knock-in mice. Social defects can be effectively reversed by manipulating the inhibitory effect of parvalbumin intermediate neurons in the prefrontal cortex (Cao et al., 2018).
NLGN4 gene mutations are found in many patients with ASD and other neurodevelopmental disorders. For example, single amino acid substitution (R101Q) mutations in NLGN4 can cause synaptic dysfunction in autistic patients (Cast et al., 2021). In Nlgn4-knockout mice, PSD-95 in the cerebral cortex and GABAA receptor and porphyrin immunoreactive synapse density in the hippocampal CA3 region are significantly decreased (Hammer et al., 2015).
NRXNs: Neurotoxins (NRXNs) serve as the presynaptic binding ligand of NLGN and play important roles in synaptic adhesion, differentiation, and maturation (Missler et al., 2003). At present, most models come from mice in which all three genes (NRXN-1, NRXN-2, and NRXN-3) are knocked out or from NRXN-1α/2α double knockout mice. Compared with wild-type mice, triple- and double-knockout mice show fewer inhibitory synapses in the brainstem and neocortex, respectively (Missler et al., 2003).
ASD patients often show high comorbidity with attention deficit hyperactivity disorder (ADHD). In rats, loss of Nrxn-1α results in significant non-social cognitive deficits and hyperactivity, similar to ADHD (Esclassan et al., 2015). CNTNAP2, which encodes contactin-associated protein-like 2 protein, is another member of the neurexin family. The CNTNAP2 gene is indispensable in promoting dendritic axons, maintaining synaptic stability, and transporting transmitters (Gdalyahu et al., 2015; Varea et al., 2015). Mice with Cntnap2 mutations exhibit behavioral defects, hyperactivity, and seizures (Peñagarikano et al., 2011). Other studies have revealed that loss of Cntnap2 in rats can lead to changes in social interaction, stereotyped behavior, and sensory behavior (Scott et al., 2020). Interestingly, recent studies on songbirds have shown that, across taxonomic classes, Cntnap2 plays an important role in neural connectivity critical for vocal learning (Condro & White, 2014).
CHD8: Chromodomain helicase DNA-binding protein 8 (CHD8) controls epigenetic and transcriptomic regulation and acts as a master regulator of many ASD risk genes (Cotney et al., 2015). Mutation of CHD8 is a highly penetrant risk factor of ASD (De Rubeis et al., 2014; Talkowski et al., 2012; Werling et al., 2018).
As a recently identified ASD gene, the functions of CDH8 have been investigated in several animal models. For example, CHD8 ortholog knockdown in mice increases brain weight and volume and in zebrafish results in macrocephaly, consistent with the macrocephaly of ASD patients with CHD8 mutations (Bernier et al., 2014; Katayama et al., 2016; O'Roak et al., 2012; Platt et al., 2017). CHD8 haploinsufficiency in mice causes ASD-like phenotypes associated with a delay in neuronal development and small but global changes in the expression of many genes in the brain during development (Katayama et al., 2016). In germline heterozygous frameshift Chd8 mutation mice, Chd8+/del5 mice show standard core features of ASD, including social interactions and repetitive behaviors, but also exhibit cognitive impairment correlated with increased regional brain volume (Gompers et al., 2017).
There is a male bias (84%) in autistic subjects with CHD8 mutations (n=24) (Stessman et al., 2017). In mice carrying heterozygous mutation of Chd8 (Chd8+/N2373K), which mimics human CHD8 (Asn2373LysfsX2) mutation, male mutant mice display a range of abnormal ASD-like behaviors during development from pup to adult, but their female counterparts do not (Jung et al., 2018). The differential patterns in male and female Chd8+/N2373K mice are determined by sexually dimorphic changes in neuronal activity, synaptic transmission, and transcriptomic profiles, validating that human CHD8 mutation indeed results in sexually dimorphic changes in mice (Jung et al., 2018).
Moreover, recent study has also shown that cerebellar granule neuron progenitor (GNP)-specific deletion of CHD8 in mice impairs cellular proliferation and differentiation, and leads to cerebellar hypoplasia and motor coordination defects, but not to ASD-like behavioral abnormalities (Kawamura et al., 2021).
POGZ: Pogo transposable element derived with ZNF domain (POGZ) is widely expressed in human tissues, including the brain. Expression analysis has shown that POGZ is expressed in the mouse cortex and hippocampus in the early developmental stages but decreases in the nucleus of both cortical and hippocampal neurons at postnatal day 30 (P30) (Ibaraki et al., 2019). In the developing cerebellum, POGZ is mainly detected in the nucleus of Purkinje cells, while at P15 and P30, POGZ expression is observed in the granular and molecular layers (Ibaraki et al., 2019).
Recent studies provide compelling evidence that loss-of-function mutations in POGZ are associated with abnormal development and behavior (Stessman et al., 2016; White et al., 2016; Ye et al., 2015). POGZ is one of the chromatin regulators of several genes implicated in ASD (De Rubeis et al., 2014; Suliman-Lavie et al., 2020). Many individuals with POGZ mutations, known as White-Sutton syndrome, manifest ID, ASD, specific facial features, and other phenotypic spectra (Fukai et al., 2015). A recent study suggested that ASD-related de novo mutations in Pogz impair neuronal development in the developing mouse brain and induce pluripotent cell lines from an ASD patient (Matsumura et al., 2020). In addition, mice with heterozygous de novo Pogz mutation exhibit ASD-like abnormalities and reduced anxiety-related avoidance (Cunniff et al., 2020; Matsumura et al., 2020). Nervous system-specific deletion of Pogz in mice leads to microcephaly, impaired growth, and ASD-like behaviors that mimic several human symptoms, suggesting that Pogz-dependent heterochromatin dysregulation can lead to cerebellar circuit dysfunction and behavioral abnormalities in ASD (Suliman-Lavie et al., 2020).
ANK2: ANK2 is a member of the ankyrin gene family, which encodes two primary ankyrin B (ankB) polypeptides, including a 440 kDa polypeptide (giant ankB) expressed only in the nervous system (Chan et al., 1993; Kunimoto, 1995), and a 220 kDa polypeptide expressed in various tissues. Clinical history shows that defect/mutation of giant ankB alone is sufficient to cause non-syndromic ASD. ANK2 is not directly related to gene regulation or synaptic function, and most ASD patients diagnosed with ANK2 mutations are non-syndromic and show average intelligence (Iossifov et al., 2014; Sztainberg & Zoghbi, 2016).
Mice lacking the two ankB peptides die during the neonatal period, and long axon bundles are lost, including pyramidal tracts and corpus callosum (Lorenzo et al., 2014; Scotland et al., 1998). For the neuronal-specific giant ankB, giant ankB-mutant mice show increased axonal branching in cultured neurons and a transient increase in excitatory synapses during postnatal development, providing evidence that normal structural connections require a considerable amount of ankB in the CNS (Yang et al., 2019). Dysfunction of giant ankB leads to inheritable impaired communicative and social behaviors, as well as superior executive function, suggesting giant ankB-deficiency/mutation is a potential cause for the abnormal structural connectivity and penetrant behaviors in mice and humans carrying ASD-related ANK2 mutations (Yang et al., 2019).
1p21.3 microdeletion (MIR137): Among chromosome 1p21.3 microdeletion carriers, most have been shown to have ASD (11 out of 12 patients) and all have ID (Carter et al., 2011; D'Angelo et al., 2015; Tucci et al., 2016; Willemsen et al., 2011). Interestingly, although each patient exhibits different genomic deletions on chromosome 1p21.3, the minimal overlapping regions only include the MIR137 gene. Genetic studies have also identified that the MIR137 gene is highly associated with ASD, schizophrenia, and bipolar disorder (Duan et al., 2014; Pinto et al., 2014; Schizophrenia Psychiatric Genome-Wide Association Study (GWAS) Consortium, 2011). Thus, miR-137 appears to be a critical pathogenic regulator mediating the core clinical features of ASD in these patients.
To study the role of miR-137, we generated Mir137 germline-knockout (gKO) and nervous system-knockout (cKO) mice and found that complete loss of miR-137 in gKO and cKO mice leads to postnatal lethality (Cheng et al., 2018). In contrast, heterozygous gKO and cKO mice are viable, indicating that all chromosome 1p21.3 microdeletion carriers are hemizygous. The partial loss of miR-137 in heterozygous gKO and cKO mice can cause synaptic overgrowth and impaired synaptic plasticity related to ASD, as well as impaired repetitive behaviors, learning, and social behaviors (Cheng et al., 2018). Multi-omics studies have revealed that one of the miR-137 mRNA targets, phosphodiesterase 10a (Pde10a), is elevated in Mir137-knockout mice, and the application of the Pde10a inhibitor papaverine or lentivirus-induced Pde10a knockdown significantly ameliorates the deficits observed in heterozygous Mir137-knockout mice. Given the known genetic link between MIR137 and neuropsychiatric diseases, our study provides direct evidence that the dysregulation of miR-137 is involved in the molecular pathogenesis of ASD.
16p11.2 microdeletion and duplication: Microdeletion and microduplication at 16p11.2 (containing 29 genes) are strongly associated with autism, accounting for about 1% of cases (Weiss et al., 2008). To explore which of the 29 genes in this genomic region are critical for the corresponding phenotypes of 16p11.2 microdeletion and microduplication, Golzio et al. (2012a) conducted functional screening in a zebrafish model and identified KCTD13 as a key gene responsible for the mirrored neuroanatomical phenotypes in humans.
Given that 16p11.2 microduplication is also reported in schizophrenia (McCarthy et al., 2009), animal models used for ASD often focus on loss-of-function of 16p11.2. For example, 16p11.2-deficient mice exhibit diminished prefrontal connectivity, thalamo-prefrontal miswiring, and reduced long-range functional synchronization (Bertero et al., 2018). Compared to wild-type male littermates, male mice with 16p11.2 heterozygous deletion (16p11.2+/–) produce strikingly fewer vocalizations during first exposure to an unfamiliar estrous female (Yang et al., 2015) and a complete lack of habituation similar to that observed in some autistic individuals (Portmann et al., 2014). In addition, partial loss of 16p11.2 can lead to endothelium-dependent structural and functional neurovascular abnormalities in 16p11.2+/– male mice and in male mice with endothelium-specific 16p11.2 deletion, suggesting a potential role for endothelial impairment in ASD (Ouellette et al., 2020). Moreover, based on in vivo two-photon imaging analysis in awake mice, layer 2/3 excitatory neurons in the motor cortex of adult male 16p11.2+/– mice are dysregulated, showing overactivation during the initial phase of learning, with prolonged learning-induced spine reorganization (Yin et al., 2021). Local infusion or intraperitoneal injection of clozapine N-oxide (CNO) is sufficient to ameliorate these impaired cellular and behavioral phenotypes, suggesting a novel role of noradrenergic neuromodulation in improving delayed motor learning in 16p11.2+/– male mice (Yin et al., 2021).
Environment-induced models
Environmental factors can also act as important pathogenic regulators in the etiology of ASD (Cheroni et al., 2020; Hertz-Picciotto et al., 2018). Children with ASD often show oxidative stress and methylation damage, which may be related to environmental pollution, exposure to chemical or toxic substances, and viral infection (Bölte et al., 2019; Varghese et al., 2017). In addition, when the mother is affected by certain antibodies and neurotoxins, her offspring can exhibit ASD-like symptoms. Animal models of ASD induced by drugs or other substances (~45 models) have the advantages of simple, fast, and low-cost operation. However, each animal model only reflects a few aspects of the possible pathogenesis of ASD (https://gene.sfari.org/database/animal-models/induced-animal-models). Nevertheless, these environment-induced animal models can be used to study nervous system function, screen new drugs, and explore treatment protocols for ASD (Figure 2B; Table 3).
Table 3. Environmental-induced ASD animal models.
| Environmental factors | Brain impacts | Clinical relevancea | References | |||||||||||||
| Typical symptoms | Animal performanceb | |||||||||||||||
| ① | ② | ③ | ④ | ⑤ | ① | ② | ③ | ④ | ⑤ | |||||||
| a: ①: Social communication disorders; ②: Repetitive/stereotyped behaviors; ③: Narrow interests; ④: Impaired linguistics; ⑤: Intellectual disability. +: Positive, –: Negative; NT: Not tested. b: M: Monkey; R: Rodents; F: Fish; I: Invertebrates. | ||||||||||||||||
| VPA | Affects expression of BDFN mRNA in brain tissue | + | + | + | + | + | R | + | + | + | – | + | Almeida et al., 2014; Banerjee et al., 2014; Kim et al., 2014a; Reynolds et al., 2021 | |||
| F | + | + | + | – | + | |||||||||||
| PPA | Reactive astrocyte keratinization of brain tissue; microglia are activated; oxidative stress markers rise; glutathione declines | NT | NT | NT | NT | NT | R | + | + | + | – | + | El-Ansary et al., 2012; Foley et al., 2015; MacFabe et al., 2011; Shultz et al., 2009 | |||
| BPA | Changes in number of neurons and glia in the medial prefrontal lobe | + | + | + | + | + | R | + | + | + | – | + | Braniste et al., 2010; Sadowski et al., 2014; Vermeer et al., 2014; Xu et al., 2015 | |||
| Sevoflurane | Increases the number of apoptotic cells in brain; inhibits the axon development of hippocampal neurons | NT | NT | NT | NT | NT | R | + | + | + | – | + | Chen et al., 2020; Jin et al., 2020 | |||
| MIA | Abnormal increase of offspring’s brain volume | + | + | + | + | + | M | + | + | + | – | + | Bauman et al., 2013; Braunschweig et al., 2012; Breach et al., 2021 | |||
| R | + | + | + | – | + | |||||||||||
| BDV | Abnormal hippocampal and cerebellar development | + | + | + | + | + | R | + | + | + | – | + | Pletnikov et al., 2002; Taieb et al., 2001 | |||
| GM | Regulation of neuroactive metabolites | + | + | + | + | + | M | + | + | + | – | + | Sharon et al., 2019 | |||
| R | + | + | + | – | + | |||||||||||
| F | + | + | + | – | + | |||||||||||
| I | + | + | + | – | + | |||||||||||
| RCS | Changes in neurotransmitter and corticosterone levels | NT | NT | NT | NT | NT | R | + | + | + | – | + | Hata et al., 1987; Namimatsu et al., 1992 | |||
Drug-induced models: Valproic acid (VPA): VPA was first used as an anti-epileptic drug and later as a mood stabilizer in epilepsy, bipolar disorder, migraine, and schizophrenia (Evatt et al., 2009). In animal experiments, researchers have simulated typical ASD models of newborn mice by exposing female mice to VPA during pregnancy. Prenatal and postnatal VPA exposure in mice can induce autism-like behaviors, and inflammatory cytokines and oxidative/nitrosative stress markers in the prefrontal cortex and hippocampal homogenate are increased in VPA-treated offspring (Elnahas et al., 2021). The degree of neuropathological changes depends on the dose and duration of VPA exposure (Bringas et al., 2013). Combined with histopathology and immunohistochemistry, the prenatal VPA model is better than the postnatal VPA model at inducing behavioral and neuropathological changes, thus highlighting the superiority of prenatal VPA exposure as a translation model for understanding the pathophysiology of ASD and developing new targets for treatment. In rat models where mothers are exposed to VPA, their offspring show changes in serotonin levels in the prefrontal cortex, hippocampus, cerebellum, and peripheral blood, similar to the results of current human clinical studies, suggesting that VPA application may be a good candidate for generating animal models of ASD (Sacco et al., 2010).
Propionic acid (PPA): PPA is a short-chain fatty acid, the final product of intestinal bacterial metabolism, and a common food preservative. Many studies have demonstrated that PPA can cause ASD-like behaviors and neuroinflammatory responses in rats. For example, rats treated with PPA show less interest in specific objects and weaker social behavior, as well as reactive astrocyte proliferation and microglia activation in brain tissue, indicating that PPA application can lead to a congenital neuroinflammatory response (MacFabe et al., 2011). The impacts of PPA on social behavior, anxiety behavior, and ultrastructure of central amygdaloid nucleus have also been explored in rats, with acute administration of a relatively low dose of PPA (175 mg/kg) found to significantly affect social behavior (Lobzhanidze et al., 2019). Although these results provide an experimental basis for using PPA to establish rodent ASD models, the direct correlation between PPA models of ASD and human ASD requires further verification.
Bisphenol propane (BPA): BPA is an important organic chemical material and a significant derivative of phenol and acetone and is widely used in the packaging of canned food and beverages. Various studies have provided evidence on neural correlates of human exposure to BPA. For instance, prenatal maternal exposure to BPA can lead to changes in the microstructure of white matter in preschool-aged children and plays an intermediary role in the relationship between BPA exposure in early life and internalization (Grohs et al., 2019). In addition, as an endocrine disruptor, exposure to BPA during fetal and perinatal periods can lead to abnormal neurodevelopment and behavioral changes in children (Elsworth et al., 2013; Nakamura et al., 2012). Studies exploring the relationship between endocrine interferon and ASD/ADHD have revealed that BPA is a low-dose developmental neurotoxic drug (Mustieles & Fernández, 2020).
Social cognition tests on three consecutive generations of mice after BPA exposure during pregnancy show that these mice exhibit more exploratory behaviors compared with the controls (Wolstenholme et al., 2013). However, third-generation mice cannot recognize new stimuli (strange mice), indicating long-term disruptive social memory in offspring. These results suggest that BPA may cause neurodevelopmental diseases in a cross-generational way (Wolstenholme et al., 2019). A recent study of 46 autistic children and 52 normal children found that those with ASD show a direct association with BPA exposure (Stein et al., 2015). Metabolomics analysis has also shown a correlation between ASD and the metabolic pathways of essential amino acids, i.e., precursors of neurotransmitters, such as tryptophan and serotonin. Furthermore, compared with age-matched non-autistic children, there are two fundamentally different types of serotonin synthesis abnormalities in autistic children, i.e., overall brain capabilities and focal abnormalities (Sarrouilhe & Dejean, 2017).
In mouse models, during neural development, intergenerational exposure to BPA can disrupt social interactions in mice and the normal expression of the PSD gene (Wolstenholme et al., 2019). In Drosophila, the application of different doses of BPA (0, 0.1, 0.5, 1 mmol/L) can lead to differences in autonomous movement (Kaur et al., 2015). Drosophila treated with BPA (especially 0.5 mmol/L BPA) also exhibit more ASD-related behaviors than the controls, including repetitive behaviors (grooming behavior) and abnormal social interactions (shortened distance from surrounding flies) (Kaur et al., 2015).
Sevoflurane: During the rapid and sensitive period of CNS development, harmful stimuli can cause serious consequences. The effect of anesthetic exposure during pregnancy on CNS development in offspring is still unclear but has aroused widespread concern. For instance, long-term exposure to sevoflurane, a commonly used general anesthetic in pediatrics, can lead to social defects in mice (Zhou et al., 2019a). Moreover, in addition to neurons, astrocytes may be important targets of sevoflurane toxicity, suggesting that the morphological integrity of astrocytes is essential for synaptic formation and neurobehavior (Zhou et al., 2019a).
In mouse models, narcotic sevoflurane can cause neurotoxicity in the developing brain, as well as adverse neurobehavioral outcomes, including inattention, insufficient social interaction, and learning and memory disabilities (Jin et al., 2020). Newborn mice inhaling sevoflurane exhibit dysfunction in learning ability (Satomoto et al., 2009). The number of apoptotic cells in the brain tissue of neonatal mice exposed to sevoflurane increase significantly after anesthesia, leading to the continuous loss of learning behavior in adulthood (Satomoto et al., 2009). Furthermore, mice exposed to sevoflurane exhibit impaired social communication, which is not caused by loss of interest or smell as they tend to show normal exploratory behavior and olfaction (Satomoto et al., 2009). These results indicate that exposure to sevoflurane during the neonatal period can lead to abnormal learning and social behaviors, resulting in ASD-like characteristics.
In pregnant mice exposed to sevoflurane when germ cells are epigenetically reprogrammed during embryonic development, >38% of F1 mice exhibited anxiety and social interaction disorders and 44%–47% of F2 and F3 mice without direct sevoflurane contact showed the same behavioral problems ( Wang et al., 2021). In addition, in the sperm of F1 mice, more than 1 200 differentially accessible loci were identified, 69 of which were also found in the sperm of F2 mice, most of which were found in the regulatory regions of genes closely related to ASD, including Arid1b, Ntrk2, and Stmn2 (Wang et al., 2021). These results suggest a long-lasting impact of sevoflurane in the pathogenesis of ASD, transmitting through male germlines between and across generations.
In rat models, compared with other brain regions of offspring, the hippocampus, a vital component of the brain involved in learning and memory, is more susceptible to repeated sevoflurane exposure (Chen et al., 2020). In addition to affecting motor sense and emotional and social behavior in offspring mice, repeated exposure to sevoflurane can cause memory deficits, probably by inhibiting the axonal development of hippocampal neurons (Chen et al., 2020).
Maternal immune activation (MIA) models: Many studies have shown that MIA in pregnant women can cause significant damage to fetal development. During pregnancy, maternal immunoglobin G (IgG) can provide passive immunity to the fetus through the placenta; however, protective and pathogenic antibodies will be transmitted to the fetus in the same way (Zimmerman et al., 2007). In the presence of autoimmune diseases or antigens, autoantibodies are activated during or before pregnancy, and damage caused by maternal autoantibodies to the fetus can cause congenital developmental disorders (Haddad et al., 2020; Zimmerman et al., 2007).
There is a known association between maternal IgG antibodies that are reactive to proteins in the fetal brain and autistic childhood outcome. Animal experiments have shown that maternal anti-fetal brain protein antibodies are closely related to offspring ASD-like symptoms (Gładysz et al., 2018). After injecting IgG in utero, offspring mice respond more positively to new things during adolescence but show anxiety-like behaviors at the adult stage and react strongly to external stimuli (Ariza et al., 2017; Singer et al., 2009). In addition, in mice injected intravenously with brain-reactive IgG antibodies from the “mothers of autistic children” and “mothers of normal children”, offspring exposed to IgG antibodies from the “mothers of autistic children” show impaired sensory and increased anxiety, suggesting that maternal IgG in autistic children can cause long-term behavioral abnormalities (Braunschweig et al., 2012). Similarly, in a maternal autoantibody model of rhesus monkeys, offspring brain volume is abnormally increased (Bauman et al., 2013).
Maternal infections or fever during pregnancy are also associated with autism or developmental delay (Zerbo et al., 2013). For example, the pro-inflammatory cytokine interleukin-6 (IL-6) has been identified as the cause of the ASD-like phenotype associated with prenatal exposure to MIA (Kumari et al., 2020). Moreover, tumor necrosis factor α (TNF-α), IL-2, IL-6, and IL-17A are also important in the pathogenesis of ASD (Eftekharian et al., 2018). For example, recent evidence suggests that the underlying inflammatory pathway links MIA-related ASD with the activity of T helper 17 (Th17) lymphocytes and its effector IL-17A. Furthermore, antibody blockade of IL-17A signaling can prevent ASD-like behavior in offspring exposed to MIA (Wong & Hoeffer, 2018).
Borna disease virus (BDV) models: BDV is a widely distributed, non-segmental, non-cytolytic neurotrophic, single-stranded RNA virus. It can infect vertebrates and lead to multiple immune-mediated CNS diseases, depending on various host and viral factors (Taieb et al., 2001). In animal infections, BDV can persist in the CNS and lead to changes in brain cell function, neurodevelopmental abnormalities, and behavioral disorders (Taieb et al., 2001). Although BDV infection has been observed in both humans and animals, the epidemiology of BDV in humans, especially children, is unclear. Based on immunofluorescence, western blotting, and radioligand analysis, Honda et al. (2018) reported on the prevalence of BDV antibodies (7.4%) in Japanese ASD children, thus providing valuable baseline data on the epidemiology of BDV in children for future research. Therefore, as a unique teratogenic factor, BDV could be used to study the pathophysiological mechanism underlying the interaction between heredity and the environment and to help carry out preclinical drug treatment trials. Additionally, in BDV-induced ASD rat models, infected young rats exhibit various behavioral dysfunctions, including impaired sensory, motor, social, emotional, and cognitive functions (Pletnikov et al., 2002).
Gut microbiota (GM) models: Traditionally, the core symptoms of many neurological diseases are considered to involve genetic variations that affect brain development and function. However, as a new research hotspot, the gut microbiome is now recognized as another important source of variation that can also affect specific behaviors of ASD (Wu et al., 2021). For example, recent studies have shown that GM can affect mouse behavior by regulating neuroactive metabolites, indicating that the gut-brain axis may be closely related to the pathogenesis and pathophysiological processes of ASD (Hsiao et al., 2013). Transplant experiments have confirmed that the GM in ASD patients is sufficient to induce obvious ASD-like behaviors (Sharon et al., 2019). In Cntnap2-knockout mice, it has been shown that the microbiome and host genes unexpectedly regulate impaired behaviors in an interdependent manner (Buffington et al., 2021). Furthermore, the hyperactive phenotype of Cntnap2-knockout mice is caused by host inheritance, while the gut microbiome mediates the social behavior phenotype. Interestingly, specific microbial interventions can selectively save social deficits in Cntnap2-knockout mice by up-regulating metabolites in the tetrahydrobiopterin synthesis pathway. These results indicate that behavioral abnormalities may have different origins (host genetics and microbes), changing the way we think about neurological diseases and their treatment (Buffington et al., 2021).
Repeated cold temperature stress (RCS) models: Other environment-induced models, such as RCS, can lead to changes in neurotransmitter and corticosterone levels in the rat brain (Hata et al., 1987; Namimatsu et al., 1992). Rats exhibit impaired locomotor activities and anxiety after RCS (Hata et al., 1988). Moreover, offspring of rats treated with RSC (during pregnant days 9–12.5) show autism-like behavioral abnormalities (Tazumi et al., 2005). Given its operational reproducibility, the RCS model has become a commonly used autism model, but the underlying mechanism is currently unclear; thus, its reliability needs to be further demonstrated.
Idiopathic models:The etiology of ASD is complex, and various factors together lead to the pathogenesis of ASD. Genetic and environment-induced models cannot simulate all pathological features of ASD. Therefore, strains of mice and rats that better mimic the core symptoms of ASD can be generated using idiopathic models, which are helpful for identifying novel ASD risk genes. At present, eight inbred lines have been used as idiopathic models, most of which are based on BTBR T+Itpr3tf/J and BALB/cByJ strains (https://gene.sfari.org/database/animal-models/inbred-animal-models/; Figure 2C).
Inbred line BTBR T+Itpr3tf/J mouse model: The BTBR T+Itpr3tf/J (BTBR) mouse strain is a widely used animal model of ASD (Meyza & Blanchard, 2017; Queen et al., 2020). BTBR mice show behavior consistent with most core clinical features of ASD and exhibit stable progeny replication (Endo et al., 2019; Meyza & Blanchard, 2017). In behavioral experiments, BTBR mice show decreased interactive social behaviors, decreased vocal ability, and highly stereotyped and repetitive self-grooming behaviors (Dodero et al., 2013; Ellegood et al., 2013). The abnormal behaviors of BTBR mice are mainly caused by three single nucleotide polymorphisms in the Kmo gene, which encodes urine 3-monooxygenase (McFarlane et al., 2008). Urine 3-monooxygenase can regulate the synthesis of kynurenic acid, one of the metabolites of tryptophan, which is abnormally expressed in other mental diseases such as schizophrenia (McFarlane et al., 2008).
Clinically, ASD patients often show corpus callosum hypoplasia or corpus callosum volume reduction, leading to speech disorders and social communication disorder symptoms (Frazier & Hardan, 2009). Based on imaging studies, the most significant neuroanatomical features of BTBR mice include loss of the corpus callosum, extreme reduction of hippocampal neurons, abnormal synaptic projections of glial cells in brain white matter, and decreased brain size (Stark et al., 2008). At the same time, gray matter volume in BTBR mice is reduced in the ventral tegmental area, cingulate gyrus, lateral thalamus, posterior thalamus, occipital and parietal cortices, and subcortex, but increased in the olfactory bulb, medial prefrontal and insular cortices, amygdala, and dorsal hippocampus (Pagani et al., 2016). These findings are consistent with the continuous decrease of gray matter volume in the brains of clinical ASD patients over time. Thus, abnormalities in other brain structures of BTBR mice may be helpful in clinical diagnosis in future studies.
Inbred line BALB/cByJ mouse model: Compared with highly social inbred mice, such as C57BL/6J and FVB/NJ, the inbred BALB/cByJ mouse strain shows significant social disorder and stereotyped behavior. Imaging studies have demonstrated that BALB/cByJ mice have a reduced corpus callosum volume. However, BALB/cByJ mice require a specific control group and do not fully present all typical clinical symptoms of ASD (Fairless et al., 2008, 2012).
CLINICAL INSIGHTS OF ASD ANIMAL MODELS
There is currently no effective treatment for ASD, resulting in a heavy burden on individuals, families, and society. Many drug therapies only treat peripheral symptoms, such as aggression, anxiety, and depression, rather than improving the core symptoms of ASD and lack effectiveness and safety. Therefore, systematic research and evaluation of innovative treatment methods to ameliorate the core social defects of ASD are needed (Anagnostou & Hansen, 2011; Doyle & McDougle, 2012a, 2012b).
Based on ASD animal models, the effects of hundreds of genetic and pharmaceutical rescue agents have been examined, indicating the great value of these models. For example, it has been shown that mTOR signaling is significantly associated with the neurological and behavioral phenotypes of certain ASD animal models, such as Tsc1/2-mutant mice, and impaired social behaviors can be corrected using the mTOR inhibitor rapamycin (Haji et al., 2020; Sato et al., 2012). Complete loss of eukaryotic initiation factor 4E (eIF4E)-binding protein 2 gene (Eif4ebp2) can lead to an imbalance in the excitatory/inhibitory ratio and ASD-like behaviors, while application of metabotropic glutamate receptor 1 (mGluR1) antagonists (JNJ16259685) or mGluR5 antagonists (fenobam) can reverse these defects (Aguilar-Valles et al., 2015). Similarly, in VPA mouse models of autism, mGluR5-antagonist (2-methyl-6-phenylethyl-pyrididine, MPEP) can ameliorate stereotyped repetitive behaviors (Mehta et al., 2011). Moreover, by inhibiting ionotropic glutamate receptors (NMDARs), d-cycloserine (DCS) can rescue ASD-associated social defects and repetitive behaviors in multiple animal models, including NLGN-1−/−, Shank2−/−, and BTBR inbred mice (Budreck et al., 2013; Burket et al., 2013; Won et al., 2012).
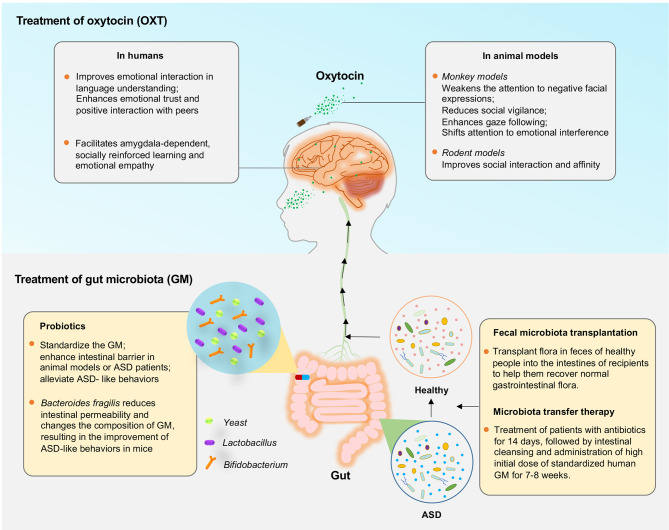
In the section below, we will briefly discuss several recent hotspots of ASD treatment that have benefited from animal model studies (Figure 3).
Figure 3.
Clinical insights of ASD animal models
Oxytocin (OXT) and gut microbiota (GM) treatment in ASD patients are recent hotspots. In humans, OXT is associated with social interaction and trust, and application of OXT may improve social function in some autistic patients. In support, monkey and rodent models have shown that OXT is closely related to social interaction and affinity and can selectively weaken attention of rhesus monkeys to negative facial expressions, reduce social vigilance, enhance gaze following response, and shift attention to emotional interference factors. Patients with ASD often exhibit changes in GM composition, as demonstrated in ASD animal models. Current GM treatments, including probiotics, fecal microbial transplantation (FMT), and microbial transfer therapy (MTT), can effectively alleviate ASD-like behaviors.
Oxytocin (OXT) treatment
As a brain neuropeptide, OXT is related to human social interactions and trust, and its application may improve social function in some autistic patients (Guastella & Hickie, 2016). Intranasal application of OXT is practical for autism, especially for enhancing language comprehension, emotional trust, and positive interactions with peers (Kosaka et al., 2012). Moreover, OXT can facilitate amygdala-dependent, socially reinforced learning and emotional empathy in men (Hurlemann et al., 2010).
In monkey models, OXT application can selectively weaken the attention of rhesus monkeys to negative facial expressions (Parr et al., 2013), reduce social vigilance (Ebitz et al., 2013), enhance gaze following response (Putnam et al., 2016), and shift attention to emotional interference factors (Landman et al., 2014). Application of OXT can also reduce fMRI response to fear and aggressive faces in macaques and selectively reduce functional coupling between the amygdala and occipital and infratemporal cortical regions (Liu et al., 2015). Moreover, in rodent models, OXT is reported to be closely related to social interaction and affinity (Silverman & Crawley, 2014). At present, there are about 10 genetic models targeting OXT and its receptor (OXTR) (https://gene.sfari.org/database/animal-models/rescue-animal-models). These animal models have confirmed the rescue effects of several pharmaceutical agents, including OXT, cocaine, and arginine vasopressin, strongly suggesting the clinical value of drug treatments targeting OXT and OXTR.
Encouraged by successful OXT treatment in animal models, a randomized placebo-controlled study of 38 male autistic patients aged 7–16 years was conducted to evaluate the effects of OXT (nasal spray); however, the study found no significant improvement in emotional cognition, social skills, or problem behavior, highlighting that intervention methods for ASD patients should be considered carefully (Yatawara et al., 2016). However, in a recent long-term follow-up treatment study, multi-dose intranasal OXT treatment effectively induced long-lasting adaptations in core social brain regions (posterior superior temporal sulcus and amygdala) from the four weeks of intranasal OXT administration until four weeks (even up to one year) post-treatment (Bernaerts et al., 2020). Therefore, OXT may be a promising molecule to promote social behavior (Gauthier et al., 2016). OXTR gene expression is low in POGZWT/Q1038R mice, and intranasal OXT administration can effectively restore impaired social behavior in these mice (Kitagawa et al., 2021). Up to 20 clinical trials are currently testing compounds for ASD treatment, including vasopressin in OXT and OXTR animal models.
Treatment of GM
Potential probiotic therapies for specific behavioral symptoms of gastrointestinal and human neurodevelopmental disorders have been identified using ASD mouse models (Hsiao et al., 2013). Indeed, changes in the GM can regulate gastrointestinal physiology, immune function, and even behavior, suggesting a certain correlation between specific bacteria in the GM and ASD-related phenotypes (Coretti et al., 2017; Wu, 2017). Thus, probiotics, fecal microbial transplantation (FMT), and microbial transfer therapy (MTT) have been applied for treating ASD.
Many studies have shown that probiotics, such as Bifidobacterium, yeast, Lactococcus, and Lactobacillus, can prevent and treat animal models and human diseases, such as obesity, depression, colorectal cancer, and Crohn’s disease (Tomova et al., 2015). Indeed, in rodent models of ASD, application of Bacteroides fragilis can reduce intestinal permeability and change intestinal microbiota composition, resulting in improved ASD-like behaviors (Hsiao et al., 2013). In a recent study of 131 autistic children and adolescents (male:female=122:19; age: 86.1±41.1 months), probiotics were shown to ameliorate several symptoms of ASD (Mensi et al., 2021), suggesting that probiotics can standardize intestinal microbiota, enhance the intestinal barrier in animal models or ASD patients, and alleviate ASD-like behaviors. However, whether probiotics play a positive role in humans remains controversial, and more clinical evidence is needed.
The transplantation (i.e., FMT) of fecal flora from healthy people into the intestines of recipients to help them recover normal gastrointestinal flora has been used to treat gastrointestinal and other diseases (Antushevich, 2020). FMT from EphB6-deficient mice results in ASD-like behavior in antibiotic-treated C57BL/6J mice, whereas FMT from wild-type mice ameliorates autism-like behavior in EphB6-deficient mice (Li et al., 2020b). Although it is workable in animal models, there are many adverse reactions caused by FMT, such as diarrhea, abdominal colic, and transient low fever. Nevertheless, FMT can normalize the intestinal microbiota of ASD patients and improve their constipation symptoms (100%) (Rossen et al., 2015).
Similarly, MTT, in which patients are treated with antibiotics for 14 days, followed by intestinal cleansing, and administration of a high dose of standardized human GM for 7–8 weeks, may be another way to treat ASD (Hamilton et al., 2012). MTT not only improves gastrointestinal symptoms (such as constipation, diarrhea, dyspepsia, abdominal pain) and ASD-related symptoms, but also normalizes the microbiota of ASD patients, with improvement for up to two years post-MTT (Kang et al., 2017, 2019).
CHALLENGES AND PROSPECTS
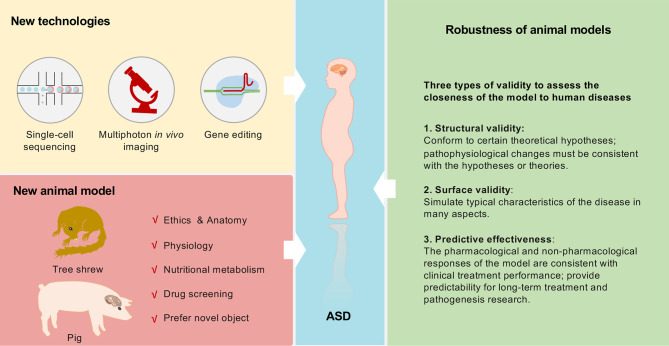
ASD is a complex neurodevelopmental disorder caused by a variety of pathogenic regulators. Although susceptible factors have been clarified through epidemiological data and clinical observations, our understanding of this disease remains relatively poor. At present, animal models remain the best choice for studies on the causes and treatment of ASD. Moreover, continuous technological improvements and breakthroughs, such as multiphoton in vivo imaging and single-cell technology, will deepen our understanding of the pathogenesis of ASD and provide necessary support for the development of new diagnostic methods and treatment (Figure 4).
Figure 4.
Development of robust and novel animal models for ASD
Animal model robustness can be evaluated based on structural validity, surface validity, and predictive effectiveness. Newly developed technologies and animal models using animals evolutionarily close to humans, such as tree shrews and pigs, are of great value for investigating ASD pathogenesis.
Assessment of current animal models
The more effective a model is, the better it can reflect human diseases. Validation is essential for evaluating the reliability of animal models and cannot be replaced when assessing the efficacy of drug treatments (Figure 4). Three types of validation are widely used to assess the closeness of a model to human disease (Chadman, 2017). Firstly, structural validity requires that the model conform to a certain theoretical hypothesis, and pathophysiological changes must be consistent with the hypothesis or theory. From this point of view, models more consistent with structural validity include ASD animal models with single-gene variation and confirmed toxic drugs. Secondly, surface validity requires that the model simulate typical characteristics of the disease in many aspects, such as behavior. From this point of view, almost all current ASD animal models conform to this feature to different degrees, given that typical clinical symptoms (such as stereotyped and impaired social behavior) are present. However, except for songbirds, no animal models are suitable for studying linguistic defects in ASD patients. Thirdly, predictive effectiveness requires that the pharmacological and non-pharmacological responses of the model are consistent with clinical treatment performance and can provide predictability for long-term treatment and pathogenesis research. From this point of view, although there is no ideal drug to treat impaired social communication and repetitive stereotyped behavior in human ASD, current ASD animal models are capable of predictive effectiveness to a certain extent. Importantly, many drugs approved for other diseases can effectively alleviate ASD symptoms without knowing the underlying mechanism, and thus basic research using ASD animal models is necessary. Of note, given its high heterogeneity, there is no current agreement on which animal model is best to investigate the pathogenesis of ASD, and finding shared causes from independent models is an excellent way to identify novel pathogenic regulators of ASD.
Developing new animal models for ASD
In addition to current ASD animal models, developing new models with novel animals that are evolutionarily close to humans, such as tree shrews and pigs, is important (Figure 4).
The tree shrew (order Scandentia) is a small animal closely related to primates and is considered an intermediary between rodents and primates (Savier et al., 2021). It has a small body size, low maintenance cost, and a relatively short reproductive cycle, making it an ideal model for studying various human diseases (Chen et al., 2020; Xu et al., 2020; Zhang et al., 2020). Extensive characterization of critical factors and signaling pathways in the immune and nervous systems shows that tree shrews have conservative and unique characteristics compared with primates (Fan et al., 2019; Yao, 2017; Ye et al., 2021). Tree shrews have a more developed nervous system and a stress system similar to humans, suggesting they may be a good choice for models of mental illness behavior. Indeed, tree shrews exhibit strong novelty preferences similar to those in rodents and primates (Khani & Rainer, 2012). A recent study on social avoidance behavior toward unfamiliar conspecifics showed notably differences between tree shrews and mice, suggesting that tree shrews may be an ideal animal model for exploring social avoidance and prosocial behaviors (Ni et al., 2020). Tree shrews have also been used to establish social frustration, learned helplessness, and chronic mild stress models (Meng et al., 2016). Based on drug antidepressant experiments, clomipramine has been shown to reverse social withdrawal behavior in depressed animals, while fluoxetine has a reversal effect on learned helplessness. In contrast, carbetocin has been shown to have a significant therapeutic impact on decreased interest, social withdrawal, and learned helplessness (Meng et al., 2016). Moreover, visuospatial cognitive task experiments have demonstrated that trees shrews have much higher cognitive abilities than rodents, including reverse learning, reward, and punishment expectations (Ohl et al., 1998). Thus, using tree shrews to explore the pathogenesis of ASD is likely to become a hotspot in ASD research.
Pigs are another potential candidate animal. Compared with rodents, pigs have advantages in anatomy, physiology, and nutritional metabolism, and are more suitable for animal models of human diseases. Various human disease model pigs have been generated by gene-editing and somatic cell-cloning technology. In recent years, pig models have been used in studies on diabetes, cardiovascular diseases, genetic diseases, tumors, and neurodegenerative diseases. Studies have shown that mini pigs explore novel objects significantly more than familiar objects, indicating that pigs exhibit measurable social novelty behavior (Moustgaard et al., 2002; Søndergaard et al., 2012). Previous studies have identified promising tasks in pig cognitive research, e.g., universal space that allows simultaneous measurement of multiple behavioral domains. The use of appropriate tasks can facilitate the collection of reliable data on pig cognition (Gieling et al., 2011). Recent study investigated the effects of intrauterine growth retardation level (IUGR, score 0−3; i.e., “normal” to “severe”) in pigs at birth, and found that some low birth-weight piglets can perform spatial tasks and object recognition tests, but performance is modulated by IUGR levels (Schmitt et al., 2019). Moreover, their relatively large littermate size makes pigs an ideal model for drug screening.
COMPETING INTERESTS
The authors declare that they have no competing interests.
AUTHORS’ CONTRIBUTIONS
Z.L. and Y.C. conceived the review. Y.X.Z., L.J.G., and Y.C. prepared the illustrations. Z.L. and Y.C. wrote the manuscript. All authors read and approved the final version of the manuscript.
ACKNOWLEDGEMENTS
We thank Dr. Da-Hua Chen for critical discussion.
Funding Statement
This work was supported by the National Natural Science Foundation of China (82071540) and Yunnan University (CZ21623201)
References
- 1.Abrahams BS, Geschwind DH Advances in autism genetics: on the threshold of a new neurobiology. Nature Reviews Genetics. 2008;9(5):341–355. doi: 10.1038/nrg2346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Achilly NP, Wang W, Zoghbi HY Presymptomatic training mitigates functional deficits in a mouse model of Rett syndrome. Nature. 2021;592(7855):596–600. doi: 10.1038/s41586-021-03369-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Aguilar-Valles A, Matta-Camacho E, Khoutorsky A, Gkogkas C, Nader K, Lacaille JC, et al Inhibition of group I metabotropic glutamate receptors reverses autistic-like phenotypes caused by deficiency of the translation repressor eiF4E binding protein 2. The Journal of Neuroscience. 2015;35(31):11125–11132. doi: 10.1523/JNEUROSCI.4615-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ahmadiantehrani S, London SE Bidirectional manipulation of mTOR signaling disrupts socially mediated vocal learning in juvenile songbirds. Proceedings of the National Academy of Sciences of the United States of America. 2017;114(35):9463–9468. doi: 10.1073/pnas.1701829114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Almeida LEF, Roby CD, Krueger BK Increased BDNF expression in fetal brain in the valproic acid model of autism. Molecular and Cellular Neuroscience. 2014;59:57–62. doi: 10.1016/j.mcn.2014.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Amir RE, Van den Veyver IB, Wan MM, Tran CQ, Francke U, Zoghbi HY Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2 . Nature Genetics. 1999;23(2):185–188. doi: 10.1038/13810. [DOI] [PubMed] [Google Scholar]
- 7.Anagnostou E, Hansen R Medical treatment overview: traditional and novel psycho-pharmacological and complementary and alternative medications. Current Opinion in Pediatrics. 2011;23(6):621–627. doi: 10.1097/MOP.0b013e32834cba3e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Antushevich H Fecal microbiota transplantation in disease therapy. Clinica Chimica Acta. 2020;503:90–98. doi: 10.1016/j.cca.2019.12.010. [DOI] [PubMed] [Google Scholar]
- 9.Ariza J, Hurtado J, Rogers H, Ikeda R, Dill M, Steward C, et al Maternal autoimmune antibodies alter the dendritic arbor and spine numbers in the infragranular layers of the cortex. PLoS One. 2017;12(8):e0183443. doi: 10.1371/journal.pone.0183443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Banerjee A, Engineer CT, Sauls BL, Morales AA, Kilgard MP, Ploski JE. 2014. Abnormal emotional learning in a rat model of autism exposed to valproic acid in utero. Frontiers in Behavioral Neuroscience, 8: 387.
- 11.Baron CA, Tepper CG, Liu SY, Davis RR, Wang NJ, Schanen NC, et al Genomic and functional profiling of duplicated chromosome 15 cell lines reveal regulatory alterations in UBE3A-associated ubiquitin-proteasome pathway processes. Human Molecular Genetics. 2006;15(6):853–869. doi: 10.1093/hmg/ddl004. [DOI] [PubMed] [Google Scholar]
- 12.Bauman MD, Iosif AM, Ashwood P, Braunschweig D, Lee A, Schumann CM, et al Maternal antibodies from mothers of children with autism alter brain growth and social behavior development in the rhesus monkey. Translational Psychiatry. 2013;3(7):e278. doi: 10.1038/tp.2013.47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bauman MD, Schumann CM Advances in nonhuman primate models of autism: Integrating neuroscience and behavior. Experimental Neurology. 2018;299:252–265. doi: 10.1016/j.expneurol.2017.07.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Berg EL, Copping NA, Rivera JK, Pride MC, Careaga M, Bauman MD, et al Developmental social communication deficits in the Shank3 rat model of phelan-mcdermid syndrome and autism spectrum disorder . Autism Research. 2018;11(4):587–601. doi: 10.1002/aur.1925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bernaerts S, Boets B, Steyaert J, Wenderoth N, Alaerts K Oxytocin treatment attenuates amygdala activity in autism: a treatment-mechanism study with long-term follow-up. Translational Psychiatry. 2020;10(1):383. doi: 10.1038/s41398-020-01069-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bernier R, Golzio C, Xiong B, Stessman HA, Coe BP, Penn O, et al Disruptive CHD8 mutations define a subtype of autism early in development . Cell. 2014;158(2):263–276. doi: 10.1016/j.cell.2014.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bertero A, Liska A, Pagani M, Parolisi R, Masferrer ME, Gritti M, et al Autism-associated 16p11.2 microdeletion impairs prefrontal functional connectivity in mouse and human. Brain. 2018;141(7):2055–2065. doi: 10.1093/brain/awy111. [DOI] [PubMed] [Google Scholar]
- 18.Blundell J, Blaiss CA, Etherton MR, Espinosa F, Tabuchi K, Walz C, et al Neuroligin-1 deletion results in impaired spatial memory and increased repetitive behavior. The Journal of Neuroscience. 2010;30(6):2115–2129. doi: 10.1523/JNEUROSCI.4517-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bölte S, Girdler S, Marschik PB The contribution of environmental exposure to the etiology of autism spectrum disorder. Cellular and Molecular Life Sciences. 2019;76(7):1275–1297. doi: 10.1007/s00018-018-2988-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bourgeron T From the genetic architecture to synaptic plasticity in autism spectrum disorder. Nature Reviews Neuroscience. 2015;16(9):551–563. doi: 10.1038/nrn3992. [DOI] [PubMed] [Google Scholar]
- 21.Braniste V, Jouault A, Gaultier E, Polizzi A, Buisson-Brenac C, Leveque M, et al Impact of oral bisphenol A at reference doses on intestinal barrier function and sex differences after perinatal exposure in rats. Proceedings of the National Academy of Sciences of the United States of America. 2010;107(1):448–453. doi: 10.1073/pnas.0907697107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Braunschweig D, Golub MS, Koenig CM, Qi LH, Pessah IN, Van de Water J, et al. 2012. Maternal autism-associated IgG antibodies delay development and produce anxiety in a mouse gestational transfer model. Journal of Neuroimmunology, 252(1–2): 56–65.
- 23.Breach MR, Dye CN, Joshi A, Platko S, Gilfarb RA, Krug AR, et al Maternal allergic inflammation in rats impacts the offspring perinatal neuroimmune milieu and the development of social play, locomotor behavior, and cognitive flexibility. Brain, Behavior, and Immunity. 2021;95:269–286. doi: 10.1016/j.bbi.2021.03.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bringas ME, Carvajal-Flores FN, López-Ramírez TA, Atzori M, Flores G Rearrangement of the dendritic morphology in limbic regions and altered exploratory behavior in a rat model of autism spectrum disorder. Neuroscience. 2013;241:170–187. doi: 10.1016/j.neuroscience.2013.03.030. [DOI] [PubMed] [Google Scholar]
- 25.Budreck EC, Kwon OB, Jung JH, Baudouin S, Thommen A, Kim HS, et al Neuroligin-1 controls synaptic abundance of NMDA-type glutamate receptors through extracellular coupling. Proceedings of the National Academy of Sciences of the United States of America. 2013;110(2):725–730. doi: 10.1073/pnas.1214718110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Buffington SA, Dooling SW, Sgritta M, Noecker C, Murillo OD, Felice DF, et al Dissecting the contribution of host genetics and the microbiome in complex behaviors. Cell. 2021;184(7):1740–1756.e16. doi: 10.1016/j.cell.2021.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bunford N, Andics A, Kis A, Miklósi Á, Gácsi M Canis familiaris as a model for non-invasive comparative neuroscience . Trends in Neurosciences. 2017;40(7):438–452. doi: 10.1016/j.tins.2017.05.003. [DOI] [PubMed] [Google Scholar]
- 28.Burket JA, Benson AD, Tang AH, Deutsch SI D-Cycloserine improves sociability in the BTBR T+ Itpr3tf/J mouse model of autism spectrum disorders with altered Ras/Raf/ERK1/2 signaling. Brain Research Bulletin. 2013;96:62–70. doi: 10.1016/j.brainresbull.2013.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cai DC, Wang ZW, Bo TT, Yan SY, Liu YL, Liu ZW, et al MECP2 duplication causes aberrant GABA pathways, circuits and behaviors in transgenic monkeys: neural mappings to patients with autism . The Journal of Neuroscience. 2020;40(19):3799–3814. doi: 10.1523/JNEUROSCI.2727-19.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Calahorro F, Ruiz-Rubio M Caenorhabditis elegans as an experimental tool for the study of complex neurological diseases: Parkinson's disease, Alzheimer's disease and autism spectrum disorder . Invertebrate Neuroscience. 2011;11(2):73–83. doi: 10.1007/s10158-011-0126-1. [DOI] [PubMed] [Google Scholar]
- 31.Camacho J, Jones K, Miller E, Ariza J, Noctor S, Van de Water J, et al Embryonic intraventricular exposure to autism-specific maternal autoantibodies produces alterations in autistic-like stereotypical behaviors in offspring mice. Behavioural Brain Research. 2014;266:46–51. doi: 10.1016/j.bbr.2014.02.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cao W, Lin S, Xia QQ, Du YL, Yang Q, Zhang MY, et al Gamma oscillation dysfunction in mPFC leads to social deficits in neuroligin 3 R451C knockin mice. Neuron. 2018;97(6):1253–1260.e7. doi: 10.1016/j.neuron.2018.02.001. [DOI] [PubMed] [Google Scholar]
- 33.Carter MT, Nikkel SM, Fernandez BA, Marshall CR, Noor A, Lionel AC, et al Hemizygous deletions on chromosome 1p21.3 involving the DPYD gene in individuals with autism spectrum disorder . Clinical Genetics. 2011;80(5):435–443. doi: 10.1111/j.1399-0004.2010.01578.x. [DOI] [PubMed] [Google Scholar]
- 34.Cast TP, Boesch DJ, Smyth K, Shaw AE, Ghebrial M, Chanda S An autism-associated mutation impairs neuroligin-4 glycosylation and enhances excitatory synaptic transmission in human neurons. The Journal of Neuroscience. 2021;41(3):392–407. doi: 10.1523/JNEUROSCI.0404-20.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chadman KK Animal models for autism in 2017 and the consequential implications to drug discovery. Expert Opinion on Drug Discovery. 2017;12(12):1187–1194. doi: 10.1080/17460441.2017.1383982. [DOI] [PubMed] [Google Scholar]
- 36.Chan W, Kordeli E, Bennett V. 1993. 440-kD ankyrinB: structure of the major developmentally regulated domain and selective localization in unmyelinated axons. The Journal of Cell Biology, 123(6 Pt 1): 1463–1473.
- 37.Chareyron LJ, Lavenex PB, Amaral DG, Lavenex P Stereological analysis of the rat and monkey amygdala. The Journal of Comparative Neurology. 2011;519(16):3218–3239. doi: 10.1002/cne.22677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen B, Liu YJ, Cai YR, Tang D, Xu SH, Gao P, et al Hippocampus is more vulnerable to neural damages induced by repeated sevoflurane exposure in the second trimester than other brain areas. Acta Biochimica et Biophysica Sinica. 2020;52(8):864–874. doi: 10.1093/abbs/gmaa060. [DOI] [PubMed] [Google Scholar]
- 39.Chen Q, Ma ZX, Xia LB, Ye ZN, Liu BL, Ma TK, et al A tree shrew model for steroid-associated osteonecrosis. Zoological Research. 2020;41(5):564–568. doi: 10.24272/j.issn.2095-8137.2020.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chen RZ, Akbarian S, Tudor M, Jaenisch R Deficiency of methyl-CpG binding protein-2 in CNS neurons results in a Rett-like phenotype in mice. Nature Genetics. 2001;27(3):327–331. doi: 10.1038/85906. [DOI] [PubMed] [Google Scholar]
- 41.Chen YC, Yu JH, Niu YY, Qin DD, Liu HL, Li G, et al Modeling rett syndrome using TALEN-edited MECP2 mutant cynomolgus monkeys . Cell. 2017;169(5):945–955.e10. doi: 10.1016/j.cell.2017.04.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cheng Y, Chen DH Fruit fly research in China. Journal of Genetics and Genomics. 2018;45(11):583–592. doi: 10.1016/j.jgg.2018.09.003. [DOI] [PubMed] [Google Scholar]
- 43.Cheng Y, Jin P Dysfunction of habituation learning: a novel pathogenic paradigm of intellectual disability and autism spectrum disorder. Biological Psychiatry. 2019;86(4):253–254. doi: 10.1016/j.biopsych.2019.06.012. [DOI] [PubMed] [Google Scholar]
- 44.Cheng Y, Wang ZM, Tan WQ, Wang XN, Li YJ, Bai B, et al Partial loss of psychiatric risk gene Mir137 in mice causes repetitive behavior and impairs sociability and learning via increased Pde10a . Nature Neuroscience. 2018;21(12):1689–1703. doi: 10.1038/s41593-018-0261-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cheroni C, Caporale N, Testa G Autism spectrum disorder at the crossroad between genes and environment: contributions, convergences, and interactions in ASD developmental pathophysiology. Molecular Autism. 2020;11(1):69. doi: 10.1186/s13229-020-00370-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Choi YB, Li HL, Kassabov SR, Jin I, Puthanveettil SV, Karl KA, et al. 2011. Neurexin-neuroligin transsynaptic interaction mediates learning-related synaptic remodeling and long-term facilitation in Aplysia. Neuron, 70(3): 468–481.
- 47.Chung BHY, Tao VQ, Tso WWY Copy number variation and autism: new insights and clinical implications. Journal of the Formosan Medical Association. 2014;113(7):400–408. doi: 10.1016/j.jfma.2013.01.005. [DOI] [PubMed] [Google Scholar]
- 48.Collins AL, Levenson JM, Vilaythong AP, Richman R, Armstrong DL, Noebels JL, et al Mild overexpression of MeCP2 causes a progressive neurological disorder in mice. Human Molecular Genetics. 2004;13(21):2679–2689. doi: 10.1093/hmg/ddh282. [DOI] [PubMed] [Google Scholar]
- 49.Collins SC, Bray SM, Suhl JA, Cutler DJ, Coffee B, Zwick ME, et al. 2010. Identification of novel FMR1 variants by massively parallel sequencing in developmentally delayed males. American Journal of Medical Genetics Part A, 1 52(10): 2512–2520.
- 50.Comery TA, Harris JB, Willems PJ, Oostra BA, Irwin SA, Weiler IJ, et al Abnormal dendritic spines in fragile X knockout mice: maturation and pruning deficits. Proceedings of the National Academy of Sciences of the United States of America. 1997;94(10):5401–5404. doi: 10.1073/pnas.94.10.5401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Condro MC, White SA Distribution of language-related Cntnap2 protein in neural circuits critical for vocal learning. The Journal of Comparative Neurology. 2014;522(1):169–185. doi: 10.1002/cne.23394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Constantino JN, Lajonchere C, Lutz M, Gray T, Abbacchi A, McKenna K, et al Autistic social impairment in the siblings of children with pervasive developmental disorders. The American Journal of Psychiatry. 2006;163(2):294–296. doi: 10.1176/appi.ajp.163.2.294. [DOI] [PubMed] [Google Scholar]
- 53.Coretti L, Cristiano C, Florio E, Scala G, Lama A, Keller S, et al Sex-related alterations of gut microbiota composition in the BTBR mouse model of autism spectrum disorder. Scientific Reports. 2017;7:45356. doi: 10.1038/srep45356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Costilla R, Kemper KE, Byrne EM, Porto-Neto LR, Carvalheiro R, Purfield DC, et al Genetic control of temperament traits across species: association of autism spectrum disorder risk genes with cattle temperament. Genetics Selection Evolution. 2020;52(1):51. doi: 10.1186/s12711-020-00569-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Cotney J, Muhle RA, Sanders SJ, Liu L, Willsey AJ, Niu W, et al The autism-associated chromatin modifier CHD8 regulates other autism risk genes during human neurodevelopment. Nature Communications. 2015;6:6404. doi: 10.1038/ncomms7404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cunniff MM, Markenscoff-Papadimitriou E, Ostrowski J, Rubenstein JL, Sohal VS. 2020. Altered hippocampal-prefrontal communication during anxiety-related avoidance in mice deficient for the autism-associated gene Pogz. eLife, 9: e54835.
- 57.D'Angelo CS, Dos Santos MFM, Alonso LG, Koiffmann CP Two new cases of 1p21.3 deletions and an unbalanced translocation t(8;12) among individuals with syndromic obesity. Molecular Syndromology. 2015;6(2):63–70. doi: 10.1159/000371600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.de Abreu MS, Genario R, Giacomini ACVV, Demin KA, Lakstygal AM, Amstislavskaya TG, et al Zebrafish as a model of neurodevelopmental disorders. Neuroscience. 2020;445:3–11. doi: 10.1016/j.neuroscience.2019.08.034. [DOI] [PubMed] [Google Scholar]
- 59.de Abreu MS, Giacomini ACVV, Genario R, dos Santos BE, Marcon L, Demin KA, et al Color as an important biological variable in zebrafish models: implications for translational neurobehavioral research. Neuroscience & Biobehavioral Reviews. 2021;124:1–15. doi: 10.1016/j.neubiorev.2020.12.014. [DOI] [PubMed] [Google Scholar]
- 60.De Rubeis S, He X, Goldberg AP, Poultney CS, Samocha K, Cicek AE, et al Synaptic, transcriptional and chromatin genes disrupted in autism. Nature. 2014;515(7526):209–215. doi: 10.1038/nature13772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Dockendorff TC, Su HS, McBride SM, Yang ZH, Choi CH, Siwicki KK, et al Drosophila lacking dfmr1 activity show defects in circadian output and fail to maintain courtship interest . Neuron. 2002;34(6):973–984. doi: 10.1016/S0896-6273(02)00724-9. [DOI] [PubMed] [Google Scholar]
- 62.Dodero L, Damiano M, Galbusera A, Bifone A, Tsaftsaris SA, Scattoni ML, et al Neuroimaging evidence of major morpho-anatomical and functional abnormalities in the BTBR T+TF/J mouse model of autism. PLoS One. 2013;8(10):e76655. doi: 10.1371/journal.pone.0076655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Donovan M, Mackey CS, Platt GN, Rounds J, Brown AN, Trickey DJ, et al Social isolation alters behavior, the gut-immune-brain axis, and neurochemical circuits in male and female prairie voles. Neurobiology of Stress. 2020;13:100278. doi: 10.1016/j.ynstr.2020.100278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Doyle CA, McDougle CJ Pharmacologic treatments for the behavioral symptoms associated with autism spectrum disorders across the lifespan. Dialogues in Clinical Neuroscience. 2012a;14(3):263–279. doi: 10.31887/DCNS.2012.14.3/cdoyle. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Doyle CA, McDougle CJ Pharmacotherapy to control behavioral symptoms in children with autism. Expert Opinion on Pharmacotherapy. 2012b;13(11):1615–1629. doi: 10.1517/14656566.2012.674110. [DOI] [PubMed] [Google Scholar]
- 66.Duan JB, Shi JX, Fiorentino A, Leites C, Chen XN, Moy W, et al A rare functional noncoding variant at the GWAS-implicated MIR137/MIR2682 locus might confer risk to schizophrenia and bipolar disorder . The American Journal of Human Genetics. 2014;95(6):744–753. doi: 10.1016/j.ajhg.2014.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Duffney LJ, Wei J, Cheng J, Liu WH, Smith KR, Kittler JT, et al Shank3 deficiency induces NMDA receptor hypofunction via an actin-dependent mechanism. The Journal of Neuroscience. 2013;33(40):15767–15778. doi: 10.1523/JNEUROSCI.1175-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ebitz RB, Watson KK, Platt ML Oxytocin blunts social vigilance in the rhesus macaque. Proceedings of the National Academy of Sciences of the United States of America. 2013;110(28):11630–11635. doi: 10.1073/pnas.1305230110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Eftekharian MM, Ghafouri-Fard S, Noroozi R, Omrani MD, Arsang-Jang S, Ganji M, et al Cytokine profile in autistic patients. Cytokine. 2018;108:120–126. doi: 10.1016/j.cyto.2018.03.034. [DOI] [PubMed] [Google Scholar]
- 70.El-Ansary AK, Ben Bacha A, Kotb M Etiology of autistic features: the persisting neurotoxic effects of propionic acid. Journal of Neuroinflammation. 2012;9:74. doi: 10.1186/1742-2094-9-74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ellegood J, Babineau BA, Henkelman RM, Lerch JP, Crawley JN Neuroanatomical analysis of the BTBR mouse model of autism using magnetic resonance imaging and diffusion tensor imaging. NeuroImage. 2013;70:288–300. doi: 10.1016/j.neuroimage.2012.12.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Elnahas EM, Abuelezz SA, Mohamad MI, Nabil MM, Abdelraouf SM, Bahaa N, et al Validation of prenatal versus postnatal valproic acid rat models of autism: a behavioral and neurobiological study. Progress in Neuro-Psychopharmacology and Biological Psychiatry. 2021;108:110185. doi: 10.1016/j.pnpbp.2020.110185. [DOI] [PubMed] [Google Scholar]
- 73.Elsworth JD, Jentsch JD, VandeVoort CA, Roth RH, Redmond DE Jr, Leranth C Prenatal exposure to bisphenol A impacts midbrain dopamine neurons and hippocampal spine synapses in non-human primates. NeuroToxicology. 2013;35:113–120. doi: 10.1016/j.neuro.2013.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Endo N, Makinodan M, Somayama N, Komori T, Kishimoto T, Nishi M Characterization of behavioral phenotypes in the BTBR T+ Itpr3tf/ J mouse model of autism spectrum disorder under social housing conditions using the multiple animal positioning system . Experimental Animals. 2019;68(3):319–330. doi: 10.1538/expanim.18-0177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Esclassan F, Francois J, Phillips KG, Loomis S, Gilmour G Phenotypic characterization of nonsocial behavioral impairment in neurexin 1α knockout rats. Behavioral Neuroscience. 2015;129(1):74–85. doi: 10.1037/bne0000024. [DOI] [PubMed] [Google Scholar]
- 76.Etherton MR, Blaiss CA, Powell CM, Sudhof TC Mouse neurexin-1α deletion causes correlated electrophysiological and behavioral changes consistent with cognitive impairments. Proceedings of the National Academy of Sciences of the United States of America. 2009;106(42):17998–18003. doi: 10.1073/pnas.0910297106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Evatt ML, DeLong MR, Grant WB, Cannell JJ, Tangpricha V Autism spectrum disorders following in utero exposure to antiepileptic drugs. Neurology. 2009;73(12):997. doi: 10.1212/WNL.0b013e3181af0b95. [DOI] [PubMed] [Google Scholar]
- 78.Fairless AH, Dow HC, Kreibich AS, Torre M, Kuruvilla M, Gordon E, et al Sociability and brain development in BALB/cJ and C57BL/6J mice. Behavioural Brain Research. 2012;228(2):299–310. doi: 10.1016/j.bbr.2011.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Fairless AH, Dow HC, Toledo MM, Malkus KA, Edelmann M, Li HZ, et al Low sociability is associated with reduced size of the corpus callosum in the BALB/cJ inbred mouse strain. Brain Research. 2008;1230:211–217. doi: 10.1016/j.brainres.2008.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Fan Y, Ye MS, Zhang JY, Xu L, Yu DD, Gu TL, et al Chromosomal level assembly and population sequencing of the Chinese tree shrew genome. Zoological Research. 2019;40(6):506–521. doi: 10.24272/j.issn.2095-8137.2019.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Feczko EJ, Bliss-Moreau E, Walum H, Pruett JR Jr, Parr LA The macaque social responsiveness scale (mSRS): a rapid screening tool for assessing variability in the social responsiveness of rhesus monkeys (Macaca mulatta) . PLoS One. 2016;11(1):e0145956. doi: 10.1371/journal.pone.0145956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Fenckova M, Blok LER, Asztalos L, Goodman DP, Cizek P, Singgih EL, et al Habituation learning is a widely affected mechanism in Drosophila models of intellectual disability and autism spectrum disorders . Biological Psychiatry. 2019;86(4):294–305. doi: 10.1016/j.biopsych.2019.04.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Foley KA, MacFabe DF, Kavaliers M, Ossenkopp KP Sexually dimorphic effects of prenatal exposure to lipopolysaccharide, and prenatal and postnatal exposure to propionic acid, on acoustic startle response and prepulse inhibition in adolescent rats: relevance to autism spectrum disorders. Behavioural Brain Research. 2015;278:244–256. doi: 10.1016/j.bbr.2014.09.032. [DOI] [PubMed] [Google Scholar]
- 84.Frazier TW, Hardan AY A meta-analysis of the corpus callosum in autism. Biological Psychiatry. 2009;66(10):935–941. doi: 10.1016/j.biopsych.2009.07.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Fukai R, Hiraki Y, Yofune H, Tsurusaki Y, Nakashima M, Saitsu H, et al. 2015. A case of autism spectrum disorder arising from a de novo missense mutation in POGZ. Journal of Human Genetics, 60(5): 277–279.
- 86.Garcia-Oscos F, Koch TMI, Pancholi H, Trusel M, Daliparthi V, Co M, et al Autism-linked gene FoxP1 selectively regulates the cultural transmission of learned vocalizations. Science Advances. 2021;7(6):eabd2827. doi: 10.1126/sciadv.abd2827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Gaugler T, Klei L, Sanders SJ, Bodea CA, Goldberg AP, Lee AB, et al Most genetic risk for autism resides with common variation. Nature Genetics. 2014;46(8):881–885. doi: 10.1038/ng.3039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Gauthier C, Doyen C, Amado I, Lôo H, Gaillard R Therapeutic effects of oxytocin in autism: current status of the research. L'Encéphale. 2016;42(1):24–31. doi: 10.1016/j.encep.2015.07.006. [DOI] [PubMed] [Google Scholar]
- 89.Gawel K, Langlois M, Martins T, van der Ent W, Tiraboschi E, Jacmin M, et al Seizing the moment: zebrafish epilepsy models. Neuroscience & Biobehavioral Reviews. 2020;116:1–20. doi: 10.1016/j.neubiorev.2020.06.010. [DOI] [PubMed] [Google Scholar]
- 90.Gdalyahu A, Lazaro M, Penagarikano O, Golshani P, Trachtenberg JT, Geschwind DH The autism related protein contactin-associated protein-like 2 (CNTNAP2) stabilizes new spines: an in vivo mouse study . PLoS One. 2015;10(5):e0125633. doi: 10.1371/journal.pone.0125633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Geschwind DH, Flint J Genetics and genomics of psychiatric disease. Science. 2015;349(6255):1489–1494. doi: 10.1126/science.aaa8954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Geschwind DH, State MW Gene hunting in autism spectrum disorder: on the path to precision medicine. The Lancet Neurology. 2015;14(11):1109–1120. doi: 10.1016/S1474-4422(15)00044-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Ghazanfar AA, Santos LR Primate brains in the wild: the sensory bases for social interactions. Nature Reviews Neuroscience. 2004;5(8):603–616. doi: 10.1038/nrn1473. [DOI] [PubMed] [Google Scholar]
- 94.Gieling ET, Nordquist RE, van der Staay FJ Assessing learning and memory in pigs. Animal Cognition. 2011;14(2):151–173. doi: 10.1007/s10071-010-0364-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Gładysz D, Krzywdzińska A, Hozyasz KK Immune abnormalities in autism spectrum disorder-could they hold promise for causative treatment? Molecular Neurobiology. 2018;55(8):6387–6435. doi: 10.1007/s12035-017-0822-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Golzio C, Willer J, Talkowski ME, Oh EC, Taniguchi Y, Jacquemont S, et al KCTD13 is a major driver of mirrored neuroanatomical phenotypes of the 16p11.2 copy number variant . Nature. 2012;485(7398):363–367. doi: 10.1038/nature11091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Gompers AL, Su-Feher L, Ellegood J, Copping NA, Riyadh MA, Stradleigh TW, et al Germline Chd8 haploinsufficiency alters brain development in mouse . Nature Neuroscience. 2017;20(8):1062–1073. doi: 10.1038/nn.4592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Grohs MN, Reynolds JE, Liu JY, Martin JW, Pollock T, Lebel C, et al Prenatal maternal and childhood bisphenol a exposure and brain structure and behavior of young children. Environmental Health. 2019;18(1):85. doi: 10.1186/s12940-019-0528-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Guastella AJ, Hickie IB Oxytocin treatment, circuitry, and autism: a critical review of the literature placing oxytocin into the autism context. Biological Psychiatry. 2016;79(3):234–242. doi: 10.1016/j.biopsych.2015.06.028. [DOI] [PubMed] [Google Scholar]
- 100.Guy J, Hendrich B, Holmes M, Martin JE, Bird A A mouse Mecp2-null mutation causes neurological symptoms that mimic Rett syndrome . Nature Genetics. 2001;27(3):322–326. doi: 10.1038/85899. [DOI] [PubMed] [Google Scholar]
- 101.Hacohen-Kleiman G, Moaraf S, Kapitansky O, Gozes I Sex-and region-dependent expression of the autism-linked ADNP correlates with social- and speech-related genes in the canary brain . Journal of Molecular Neuroscience. 2020;70(11):1671–1683. doi: 10.1007/s12031-020-01700-x. [DOI] [PubMed] [Google Scholar]
- 102.Haddad FL, Patel SV, Schmid S Maternal immune activation by Poly I: C as a preclinical model for neurodevelopmental disorders: a focus on autism and schizophrenia. Neuroscience & Biobehavioral Reviews. 2020;113:546–567. doi: 10.1016/j.neubiorev.2020.04.012. [DOI] [PubMed] [Google Scholar]
- 103.Haji N, Riebe I, Aguilar-Valles A, Artinian J, Laplante I, Lacaille JC Tsc1 haploinsufficiency in Nkx2.1 cells upregulates hippocampal interneuron mTORC1 activity, impairs pyramidal cell synaptic inhibition, and alters contextual fear discrimination and spatial working memory in mice . Molecular Autism. 2020;11(1):29. doi: 10.1186/s13229-020-00340-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Hamilton MJ, Weingarden AR, Sadowsky MJ, Khoruts A Standardized frozen preparation for transplantation of fecal microbiota for recurrent Clostridium difficile infection . American Journal of Gastroenterology. 2012;107(5):761–767. doi: 10.1038/ajg.2011.482. [DOI] [PubMed] [Google Scholar]
- 105.Hammer M, Krueger-Burg D, Tuffy LP, Cooper BH, Taschenberger H, Goswami SP, et al Perturbed hippocampal synaptic inhibition and γ-oscillations in a neuroligin-4 knockout mouse model of autism. Cell Reports. 2015;13(3):516–523. doi: 10.1016/j.celrep.2015.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Han K, Holder JL Jr, Schaaf CP, Lu H, Chen HM, Kang H, et al SHANK3 overexpression causes manic-like behaviour with unique pharmacogenetic properties . Nature. 2013;503(7474):72–77. doi: 10.1038/nature12630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Hata T, Kita T, Kamanaka Y, Honda S, Kakehi K, Kawabata A, et al Catecholamine levels in the brain of SART (repeated cold)-stressed rats. Journal of Autonomic Pharmacology. 1987;7(3):257–266. doi: 10.1111/j.1474-8673.1987.tb00154.x. [DOI] [PubMed] [Google Scholar]
- 108.Hata T, Nishimura Y, Kita T, Itoh E, Kawabata A The abnormal open-field behavior of SART-stressed rats and effects of some drugs on it. Japanese Journal of Pharmacology. 1988;48(4):479–490. doi: 10.1254/jjp.48.479. [DOI] [PubMed] [Google Scholar]
- 109.He CX, Cantu DA, Mantri SS, Zeiger WA, Goel A, Portera-Cailliau C Tactile defensiveness and impaired adaptation of neuronal activity in the Fmr1 knock-out mouse model of autism . The Journal of Neuroscience. 2017;37(27):6475–6487. doi: 10.1523/JNEUROSCI.0651-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Hertz-Picciotto I, Schmidt RJ, Krakowiak P Understanding environmental contributions to autism: causal concepts and the state of science. Autism Research. 2018;11(4):554–586. doi: 10.1002/aur.1938. [DOI] [PubMed] [Google Scholar]
- 111.Heshmati M, Aleyasin H, Menard C, Christoffel DJ, Flanigan ME, Pfau ML, et al Cell-type-specific role for nucleus accumbens neuroligin-2 in depression and stress susceptibility. Proceedings of the National Academy of Sciences of the United States of America. 2018;115(5):1111–1116. doi: 10.1073/pnas.1719014115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Honda T, Sofuku K, Matsunaga H, Tachibana M, Mohri I, Taniike M, et al Prevalence of antibodies against Borna disease virus proteins in Japanese children with autism spectrum disorder. Microbiology and Immunology. 2018;62(7):473–476. doi: 10.1111/1348-0421.12603. [DOI] [PubMed] [Google Scholar]
- 113.Hope KA, Flatten D, Cavitch P, May B, Sutcliffe JS, O'Donnell J, et al The Drosophila gene Sulfateless modulates autism-like behaviors . Frontiers in Genetics. 2019;10:574. doi: 10.3389/fgene.2019.00574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Hörnberg H, Pérez-Garci E, Schreiner D, Hatstatt-Burklé L, Magara F, Baudouin S, et al Rescue of oxytocin response and social behaviour in a mouse model of autism. Nature. 2020;584(7820):252–256. doi: 10.1038/s41586-020-2563-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Hsiao EY, McBride SW, Hsien S, Sharon G, Hyde ER, McCue T, et al Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell. 2013;155(7):1451–1463. doi: 10.1016/j.cell.2013.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Hurlemann R, Patin A, Onur OA, Cohen MX, Baumgartner T, Metzler S, et al Oxytocin enhances amygdala-dependent, socially reinforced learning and emotional empathy in humans. The Journal of Neuroscience. 2010;30(14):4999–5007. doi: 10.1523/JNEUROSCI.5538-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Ibaraki K, Hamada N, Iwamoto I, Ito H, Kawamura N, Morishita R, et al. 2019. Expression analyses of POGZ, a responsible gene for neurodevelopmental disorders, during mouse brain development. Developmental Neuroscience, 41(1–2): 139–148.
- 118.Iossifov I, O'Roak BJ, Sanders SJ, Ronemus M, Krumm N, Levy D, et al The contribution of de novo coding mutations to autism spectrum disorder . Nature. 2014;515(7526):216–221. doi: 10.1038/nature13908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.James DM, Davidson EA, Yanes J, Moshiree B, Dallman JE The gut-brain-microbiome axis and its link to autism: emerging insights and the potential of zebrafish models. Frontiers in Cell and Developmental Biology. 2021;9:662916. doi: 10.3389/fcell.2021.662916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Jaramillo TC, Xuan Z, Reimers JM, Escamilla CO, Liu SN, Powell CM Early restoration of Shank3 Expression in Shank3 knock-out mice prevents core ASD-like behavioral phenotypes . eNeuro. 2020;7(3):ENEURO.0332–19.2020. doi: 10.1523/ENEURO.0332-19.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Jayakumar V, Nishimura O, Kadota M, Hirose N, Sano H, Murakawa Y, et al Chromosomal-scale de novo genome assemblies of cynomolgus macaque and common marmoset . Scientific Data. 2021;8(1):159. doi: 10.1038/s41597-021-00935-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Jin X, Ji L, Chen QC, Sheng R, Ji FH, Yang JP Anesthesia plus surgery in neonatal period impairs preference for social novelty in mice at the juvenile age. Biochemical and Biophysical Research Communications. 2020;530(3):603–608. doi: 10.1016/j.bbrc.2020.07.108. [DOI] [PubMed] [Google Scholar]
- 123.Jung H, Park H, Choi Y, Kang H, Lee E, Kweon H, et al Sexually dimorphic behavior, neuronal activity, and gene expression in Chd8-mutant mice. Nature Neuroscience. 2018;21(9):1218–1228. doi: 10.1038/s41593-018-0208-z. [DOI] [PubMed] [Google Scholar]
- 124.Kang DW, Adams JB, Coleman DM, Pollard EL, Maldonado J, McDonough-Means S, et al Long-term benefit of Microbiota Transfer Therapy on autism symptoms and gut microbiota. Scientific Reports. 2019;9(1):5821. doi: 10.1038/s41598-019-42183-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Kang DW, Adams JB, Gregory AC, Borody T, Chittick L, Fasano A, et al Microbiota Transfer Therapy alters gut ecosystem and improves gastrointestinal and autism symptoms: an open-label study. Microbiome. 2017;5(1):10. doi: 10.1186/s40168-016-0225-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Kanner L Autistic disturbances of affective contact. Acta Paedopsychiatrica. 1968;35(4):100–136. [PubMed] [Google Scholar]
- 127.Katayama Y, Nishiyama M, Shoji H, Ohkawa Y, Kawamura A, Sato T, et al CHD8 haploinsufficiency results in autistic-like phenotypes in mice. Nature. 2016;537(7622):675–679. doi: 10.1038/nature19357. [DOI] [PubMed] [Google Scholar]
- 128.Kaur K, Simon AF, Chauhan V, Chauhan A Effect of bisphenol A on Drosophila melanogaster behavior-A new model for the studies on neurodevelopmental disorders . Behavioural Brain Research. 2015;284:77–84. doi: 10.1016/j.bbr.2015.02.001. [DOI] [PubMed] [Google Scholar]
- 129.Kawamura A, Katayama Y, Kakegawa W, Ino D, Nishiyama M, Yuzaki M, et al The autism-associated protein CHD8 is required for cerebellar development and motor function. Cell Reports. 2021;35(1):108932. doi: 10.1016/j.celrep.2021.108932. [DOI] [PubMed] [Google Scholar]
- 130.Khani A, Rainer G Recognition memory in tree shrew (Tupaia belangeri) after repeated familiarization sessions . Behavioural Processes. 2012;90(3):364–371. doi: 10.1016/j.beproc.2012.03.019. [DOI] [PubMed] [Google Scholar]
- 131.Kim JW, Seung H, Kwon KJ, Ko MJ, Lee EJ, Oh HA, et al Subchronic treatment of donepezil rescues impaired social, hyperactive, and stereotypic behavior in valproic acid-induced animal model of autism. PLoS One. 2014a;9(8):e104927. doi: 10.1371/journal.pone.0104927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Kim L, He L, Maaswinkel H, Zhu LQ, Sirotkin H, Weng W Anxiety, hyperactivity and stereotypy in a zebrafish model of fragile X syndrome and autism spectrum disorder. Progress in Neuro-Psychopharmacology and Biological Psychiatry. 2014b;55:40–49. doi: 10.1016/j.pnpbp.2014.03.007. [DOI] [PubMed] [Google Scholar]
- 133.Kitagawa K, Matsumura K, Baba M, Kondo M, Takemoto T, Nagayasu K, et al Intranasal oxytocin administration ameliorates social behavioral deficits in a POGZWT/Q1038R mouse model of autism spectrum disorder . Molecular Brain. 2021;14(1):56. doi: 10.1186/s13041-021-00769-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Kocher SD, Mallarino R, Rubin BER, Yu DW, Hoekstra HE, Pierce NE The genetic basis of a social polymorphism in halictid bees. Nature Communications. 2018;9(1):4338. doi: 10.1038/s41467-018-06824-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Kolozsi E, Mackenzie RN, Roullet FI, Decatanzaro D, Foster JA Prenatal exposure to valproic acid leads to reduced expression of synaptic adhesion molecule neuroligin 3 in mice. Neuroscience. 2009;163(4):1201–1210. doi: 10.1016/j.neuroscience.2009.07.021. [DOI] [PubMed] [Google Scholar]
- 136.Kosaka H, Munesue T, Ishitobi M, Asano M, Omori M, Sato M, et al Long-term oxytocin administration improves social behaviors in a girl with autistic disorder. BMC Psychiatry. 2012;12:110. doi: 10.1186/1471-244X-12-110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Kozol RA, Cukier HN, Zou B, Mayo V, De Rubeis S, Cai GQ, et al Two knockdown models of the autism genes SYNGAP1 and SHANK3 in zebrafish produce similar behavioral phenotypes associated with embryonic disruptions of brain morphogenesis . Human Molecular Genetics. 2015;24(14):4006–4023. doi: 10.1093/hmg/ddv138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Krishnan V, Stoppel DC, Nong Y, Johnson MA, Nadler MJS, Ozkaynak E, et al Autism gene Ube3a and seizures impair sociability by repressing VTA Cbln1 . Nature. 2017;543(7646):507–512. doi: 10.1038/nature21678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Kumar S, Hedges SB A molecular timescale for vertebrate evolution. Nature. 1998;392(6679):917–920. doi: 10.1038/31927. [DOI] [PubMed] [Google Scholar]
- 140.Kumari E, Velloso FJ, Nasuhidehnavi A, Somasundaram A, Savanur VH, Buono KD, et al Developmental IL-6 exposure favors production of PDGF-responsive multipotential progenitors at the expense of neural stem cells and other progenitors. Stem Cell Reports. 2020;14(5):861–875. doi: 10.1016/j.stemcr.2020.03.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Kunimoto M. 1995. A neuron-specific isoform of brain ankyrin, 440-kD ankyrinB, is targeted to the axons of rat cerebellar neurons. The Journal of Cell Biology, 131(6 Pt 2): 1821–1829.
- 142.Kunkel GR, Tracy JA, Jalufka FL, Lekven AC. 2018. CHD8short, a naturally-occurring truncated form of a chromatin remodeler lacking the helicase domain, is a potent transcriptional coregulator. Gene, 641: 303–309.
- 143.Lam HC, Siroky BJ, Henske EP Renal disease in tuberous sclerosis complex: pathogenesis and therapy. Nature Reviews Nephrology. 2018;14(11):704–716. doi: 10.1038/s41581-018-0059-6. [DOI] [PubMed] [Google Scholar]
- 144.Landman R, Sharma J, Sur M, Desimone R Effect of distracting faces on visual selective attention in the monkey. Proceedings of the National Academy of Sciences of the United States of America. 2014;111(50):18037–18042. doi: 10.1073/pnas.1420167111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Li CH, Coffey EL, Dall'Agnese A, Hannett NM, Tang X, Henninger JE, et al MeCP2 links heterochromatin condensates and neurodevelopmental disease. Nature. 2020a;586(7829):440–444. doi: 10.1038/s41586-020-2574-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Li Y, Luo ZY, Hu YY, Bi YW, Yang JM, Zou WJ, et al The gut microbiota regulates autism-like behavior by mediating vitamin B6 homeostasis in EphB6-deficient mice . Microbiome. 2020b;8(1):120. doi: 10.1186/s40168-020-00884-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Liu CX, Li CY, Hu CC, Wang Y, Lin J, Jiang YH, et al CRISPR/Cas9-induced shank3b mutant zebrafish display autism-like behaviors . Molecular Autism. 2018;9:23. doi: 10.1186/s13229-018-0204-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Liu N, Hadj-Bouziane F, Jones KB, Turchi JN, Averbeck BB, Ungerleider LG Oxytocin modulates fMRI responses to facial expression in macaques. Proceedings of the National Academy of Sciences of the United States of America. 2015;112(24):E3123–E3130. doi: 10.1073/pnas.1508097112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Liu Z, Li X, Zhang JT, Cai YJ, Cheng TL, Cheng C, et al Autism-like behaviours and germline transmission in transgenic monkeys overexpressing MeCP2. Nature. 2016;530(7588):98–102. doi: 10.1038/nature16533. [DOI] [PubMed] [Google Scholar]
- 150.Lobzhanidze G, Lordkipanidze T, Zhvania M, Japaridze N, MacFabe DF, Pochkidze N, et al Effect of propionic acid on the morphology of the amygdala in adolescent male rats and their behavior. Micron. 2019;125:102732. doi: 10.1016/j.micron.2019.102732. [DOI] [PubMed] [Google Scholar]
- 151.Lorenzo DN, Badea A, Davis J, Hostettler J, He J, Zhong GS, et al A PIK3C3-ankyrin-B-dynactin pathway promotes axonal growth and multiorganelle transport. Journal of Cell Biology. 2014;207(6):735–752. doi: 10.1083/jcb.201407063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.MacFabe DF, Cain NE, Boon F, Ossenkopp KP, Cain DP Effects of the enteric bacterial metabolic product propionic acid on object-directed behavior, social behavior, cognition, and neuroinflammation in adolescent rats: relevance to autism spectrum disorder. Behavioural Brain Research. 2011;217(1):47–54. doi: 10.1016/j.bbr.2010.10.005. [DOI] [PubMed] [Google Scholar]
- 153.Maenner MJ, Shaw KA, Baio J, Washington A, Patrick M, DiRienzo M, et al Prevalence of autism spectrum disorder among children aged 8 years - autism and developmental disabilities monitoring network, 11 sites, United States, 2016. Morbidity and Mortality Weekly Report. Surveillance Summaries. 2020;69(4):1–12. doi: 10.15585/mmwr.ss6904a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Manoli DS, State MW Autism spectrum disorder genetics and the search for pathological mechanisms. The American Journal of Psychiatry. 2021;178(1):30–38. doi: 10.1176/appi.ajp.2020.20111608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Matas E, Maisterrena A, Thabault M, Balado E, Francheteau M, Balbous A, et al Major motor and gait deficits with sexual dimorphism in a Shank3 mutant mouse model . Molecular Autism. 2021;12(1):2. doi: 10.1186/s13229-020-00412-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Matsumura K, Seiriki K, Okada S, Nagase M, Ayabe S, Yamada I, et al Pathogenic POGZ mutation causes impaired cortical development and reversible autism-like phenotypes. Nature Communications. 2020;11(1):859. doi: 10.1038/s41467-020-14697-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.McCarthy SE, Makarov V, Kirov G, Addington AM, McClellan J, Yoon S, et al Microduplications of 16p11.2 are associated with schizophrenia. Nature Genetics. 2009;41(11):1223–1227. doi: 10.1038/ng.474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.McFarlane HG, Kusek GK, Yang M, Phoenix JL, Bolivar VJ, Crawley JN Autism-like behavioral phenotypes in BTBR T+tf/J mice. Genes, Brain, and Behavior. 2008;7(2):152–163. doi: 10.1111/j.1601-183X.2007.00330.x. [DOI] [PubMed] [Google Scholar]
- 159.Mehta MV, Gandal MJ, Siegel SJ mGluR5-antagonist mediated reversal of elevated stereotyped, repetitive behaviors in the VPA model of autism. PLoS One. 2011;6(10):e26077. doi: 10.1371/journal.pone.0026077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Meng XL, Shen F, Li CL, Li YH, Wang XW Depression-like behaviors in tree shrews and comparison of the effects of treatment with fluoxetine and carbetocin. Pharmacology Biochemistry and Behavior. 2016;145:1–8. doi: 10.1016/j.pbb.2016.03.006. [DOI] [PubMed] [Google Scholar]
- 161.Mensi MM, Rogantini C, Marchesi M, Borgatti R, Chiappedi M Lactobacillus plantarum PS128 and other probiotics in children and adolescents with autism spectrum disorder: a real-world experience . Nutrients. 2021;13(6):2036. doi: 10.3390/nu13062036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Meyza KZ, Blanchard DC The BTBR mouse model of idiopathic autism - Current view on mechanisms. Neuroscience & Biobehavioral Reviews. 2017;76:99–110. doi: 10.1016/j.neubiorev.2016.12.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Miles JH Autism spectrum disorders-A genetics review. Genetics in Medicine. 2011;13(4):278–294. doi: 10.1097/GIM.0b013e3181ff67ba. [DOI] [PubMed] [Google Scholar]
- 164.Missler M, Zhang WQ, Rohlmann A, Kattenstroth G, Hammer RE, Gottmann K, et al α-Neurexins couple Ca2+ channels to synaptic vesicle exocytosis . Nature. 2003;423(6943):939–948. doi: 10.1038/nature01755. [DOI] [PubMed] [Google Scholar]
- 165.Modi B, Pimpinella D, Pazienti A, Zacchi P, Cherubini E, Griguoli M Possible implication of the CA2 hippocampal circuit in social cognition deficits observed in the neuroligin 3 knock-out mouse, a non-syndromic animal model of autism. Frontiers in Psychiatry. 2019;10:513. doi: 10.3389/fpsyt.2019.00513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Mossa A, Pagano J, Ponzoni L, Tozzi A, Vezzoli E, Sciaccaluga M, et al Developmental impaired Akt signaling in the Shank1 and Shank3 double knock-out mice. Molecular Psychiatry. 2021;26(6):1928–1944. doi: 10.1038/s41380-020-00979-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Moustgaard A, Lind NM, Hemmingsen R, Hansen AK Spontaneous object recognition in the Göttingen minipig. Neural Plasticity. 2002;9:845262. doi: 10.1155/NP.2002.255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Mukherjee SB Autism spectrum disorders-diagnosis and management. The Indian Journal of Pediatrics. 2017;84(4):307–314. doi: 10.1007/s12098-016-2272-2. [DOI] [PubMed] [Google Scholar]
- 169.Mustieles V, Fernández MF Bisphenol A shapes children's brain and behavior: towards an integrated neurotoxicity assessment including human data. Environmental Health. 2020;19(1):66. doi: 10.1186/s12940-020-00620-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Nakamura K, Itoh K, Dai HM, Han LZ, Wang XH, Kato S, et al Prenatal and lactational exposure to low-doses of bisphenol A alters adult mice behavior. Brain & Development. 2012;34(1):57–63. doi: 10.1016/j.braindev.2010.12.011. [DOI] [PubMed] [Google Scholar]
- 171.Nakanishi M, Nomura J, Ji X, Tamada K, Arai T, Takahashi E, et al Functional significance of rare neuroligin 1 variants found in autism. PLoS Genetics. 2017;13(8):e1006940. doi: 10.1371/journal.pgen.1006940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Namimatsu A, Go K, Ohara H, Yoneda R Changes in muscarinic acetylcholine receptors in the isolated duodenum from repeatedly cold-stressed rats and the effect of neurotropin. Life Sciences. 1992;50(25):1993–2000. doi: 10.1016/0024-3205(92)90529-X. [DOI] [PubMed] [Google Scholar]
- 173.Ni RJ, Tian Y, Dai XY, Zhao LS, Wei JX, Zhou JN, et al Social avoidance behavior in male tree shrews and prosocial behavior in male mice toward unfamiliar conspecifics in the laboratory. Zoological Research. 2020;41(3):258–272. doi: 10.24272/j.issn.2095-8137.2020.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Normand EA, Crandall SR, Thorn CA, Murphy EM, Voelcker B, Browning C, et al Temporal and mosaic Tsc1 deletion in the developing thalamus disrupts thalamocortical circuitry, neural function, and behavior . Neuron. 2013;78(5):895–909. doi: 10.1016/j.neuron.2013.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Ohl F, Oitzl MS, Fuchs E. 1998. Assessing cognitive functions in tree shrews: visuo-spatial and spatial learning in the home cage. Journal of Neuroscience Methods, 81(1–2): 35–40.
- 176.Orefice LL, Mosko JR, Morency DT, Wells MF, Tasnim A, Mozeika SM, et al Targeting peripheral somatosensory neurons to improve tactile-related phenotypes in ASD models. Cell. 2019;178(4):867–886.e24. doi: 10.1016/j.cell.2019.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Ornoy A, Weinstein-Fudim L, Ergaz Z Prenatal factors associated with autism spectrum disorder (ASD) Reproductive Toxicology. 2015;56:155–169. doi: 10.1016/j.reprotox.2015.05.007. [DOI] [PubMed] [Google Scholar]
- 178.O'Roak BJ, Vives L, Fu WQ, Egertson JD, Stanaway IB, Phelps IG, et al Multiplex targeted sequencing identifies recurrently mutated genes in autism spectrum disorders. Science. 2012;338(6114):1619–1622. doi: 10.1126/science.1227764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Ouellette J, Toussay X, Comin CH, da F. Costa L, Ho M, Lacalle-Aurioles M, et al Vascular contributions to 16p11.2 deletion autism syndrome modeled in mice. Nature Neuroscience. 2020;23(9):1090–1101. doi: 10.1038/s41593-020-0663-1. [DOI] [PubMed] [Google Scholar]
- 180.Pagani M, Damiano M, Galbusera A, Tsaftaris SA, Gozzi A Semi-automated registration-based anatomical labelling, voxel based morphometry and cortical thickness mapping of the mouse brain. Journal of Neuroscience Methods. 2016;267:62–73. doi: 10.1016/j.jneumeth.2016.04.007. [DOI] [PubMed] [Google Scholar]
- 181.Panaitof SC A songbird animal model for dissecting the genetic bases of autism spectrum disorder. Disease Markers. 2012;33:727058. doi: 10.3233/DMA-2012-0918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Parr LA, Modi M, Siebert E, Young LJ Intranasal oxytocin selectively attenuates rhesus monkeys' attention to negative facial expressions. Psychoneuroendocrinology. 2013;38(9):1748–1756. doi: 10.1016/j.psyneuen.2013.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Peñagarikano O, Abrahams BS, Herman EI, Winden KD, Gdalyahu A, Dong HM, et al Absence of CNTNAP2 leads to epilepsy, neuronal migration abnormalities, and core autism-related deficits. Cell. 2011;147(1):235–246. doi: 10.1016/j.cell.2011.08.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Peretti S, Mariano M, Mazzocchetti C, Mazza M, Pino MC, Di Pianella AV, et al Diet: the keystone of autism spectrum disorder? Nutritional Neuroscience. 2019;22(12):825–839. doi: 10.1080/1028415X.2018.1464819. [DOI] [PubMed] [Google Scholar]
- 185.Pinto D, Delaby E, Merico D, Barbosa M, Merikangas A, Klei L, et al Convergence of genes and cellular pathways dysregulated in autism spectrum disorders. American Journal of Human Genetics. 2014;94(5):677–694. doi: 10.1016/j.ajhg.2014.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Pinto D, Pagnamenta AT, Klei L, Anney R, Merico D, Regan R, et al Functional impact of global rare copy number variation in autism spectrum disorders. Nature. 2010;466(7304):368–372. doi: 10.1038/nature09146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Pitkänen A, Kemppainen S Comparison of the distribution of calcium-binding proteins and intrinsic connectivity in the lateral nucleus of the rat, monkey, and human amygdala. Pharmacology Biochemistry and Behavior. 2002;71(3):369–377. doi: 10.1016/S0091-3057(01)00690-6. [DOI] [PubMed] [Google Scholar]
- 188.Pizzamiglio L, Focchi E, Cambria C, Ponzoni L, Ferrara S, Bifari F, et al The DNA repair protein ATM as a target in autism spectrum disorder. JCI Insight. 2021;6(3):e133654. doi: 10.1172/jci.insight.133654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Platt RJ, Zhou Y, Slaymaker IM, Shetty AS, Weisbach NR, Kim JA, et al Chd8 mutation leads to autistic-like behaviors and impaired striatal circuits . Cell Reports. 2017;19(2):335–350. doi: 10.1016/j.celrep.2017.03.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Pletnikov MV, Moran TH, Carbone KM Borna disease virus infection of the neonatal rat: developmental brain injury model of autism spectrum disorders. Frontiers in Bioscience. 2002;7(4):593–607. doi: 10.2741/A797. [DOI] [PubMed] [Google Scholar]
- 191.Portmann T, Yang M, Mao R, Panagiotakos G, Ellegood J, Dolen G, et al Behavioral abnormalities and circuit defects in the basal ganglia of a mouse model of 16p11.2 deletion syndrome. Cell Reports. 2014;7(4):1077–1092. doi: 10.1016/j.celrep.2014.03.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Prieto M, Folci A, Poupon G, Schiavi S, Buzzelli V, Pronot M, et al Missense mutation of Fmr1 results in impaired AMPAR-mediated plasticity and socio-cognitive deficits in mice . Nature Communications. 2021;12(1):1557. doi: 10.1038/s41467-021-21820-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Putnam PT, Roman JM, Zimmerman PE, Gothard KM Oxytocin enhances gaze-following responses to videos of natural social behavior in adult male rhesus monkeys. Psychoneuroendocrinology. 2016;72:47–53. doi: 10.1016/j.psyneuen.2016.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Queen NJ, Boardman AA, Patel RS, Siu JJ, Mo XK, Cao L Environmental enrichment improves metabolic and behavioral health in the BTBR mouse model of autism. Psychoneuroendocrinology. 2020;111:104476. doi: 10.1016/j.psyneuen.2019.104476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Radyushkin K, Hammerschmidt K, Boretius S, Varoqueaux F, El-Kordi A, Ronnenberg A, et al Neuroligin-3-deficient mice: model of a monogenic heritable form of autism with an olfactory deficit. Genes, Brain, and Behavior. 2009;8(4):416–425. doi: 10.1111/j.1601-183X.2009.00487.x. [DOI] [PubMed] [Google Scholar]
- 196.Rapin I, Tuchman RF Autism: definition, neurobiology, screening, diagnosis. Pediatric Clinics of North America. 2008;55(5):1129–1146. doi: 10.1016/j.pcl.2008.07.005. [DOI] [PubMed] [Google Scholar]
- 197.Rat Genome Sequencing Project Consortium Genome sequence of the Brown Norway rat yields insights into mammalian evolution. Nature. 2004;428(6982):493–521. doi: 10.1038/nature02426. [DOI] [PubMed] [Google Scholar]
- 198.Rea V, Van Raay TJ Using zebrafish to model autism spectrum disorder: a comparison of ASD risk genes between zebrafish and their mammalian counterparts. Frontiers in Molecular Neuroscience. 2020;13:575575. doi: 10.3389/fnmol.2020.575575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Reith RM, McKenna J, Wu H, Hashmi SS, Cho SH, Dash PK, et al Loss of Tsc2 in Purkinje cells is associated with autistic-like behavior in a mouse model of tuberous sclerosis complex . Neurobiology of Disease. 2013;51:93–103. doi: 10.1016/j.nbd.2012.10.014. [DOI] [PubMed] [Google Scholar]
- 200.Reynolds KE, Wong CR, Scott AL Astrocyte-mediated purinergic signaling is upregulated in a mouse model of Fragile X syndrome. Glia. 2021;69(7):1816–1832. doi: 10.1002/glia.23997. [DOI] [PubMed] [Google Scholar]
- 201.Richter JD, Zhao XY The molecular biology of FMRP: new insights into fragile X syndrome. Nature Reviews Neuroscience. 2021;22(4):209–222. doi: 10.1038/s41583-021-00432-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Rossen NG, MacDonald JK, de Vries EM, D'Haens GR, de Vos WM, Zoetendal EG, et al Fecal microbiota transplantation as novel therapy in gastroenterology: a systematic review. World Journal of Gastroenterology. 2015;21(17):5359–5371. doi: 10.3748/wjg.v21.i17.5359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Rothwell PE, Fuccillo MV, Maxeiner S, Hayton SJ, Gokce O, Lim BK, et al Autism-associated neuroligin-3 mutations commonly impair striatal circuits to boost repetitive behaviors. Cell. 2014;158(1):198–212. doi: 10.1016/j.cell.2014.04.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Rutishauser U, Mamelak AN, Adolphs R The primate amygdala in social perception - insights from electrophysiological recordings and stimulation. Trends in Neurosciences. 2015;38(5):295–306. doi: 10.1016/j.tins.2015.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Sacco R, Curatolo P, Manzi B, Militerni R, Bravaccio C, Frolli A, et al Principal pathogenetic components and biological endophenotypes in autism spectrum disorders. Autism Research. 2010;3(5):237–252. doi: 10.1002/aur.151. [DOI] [PubMed] [Google Scholar]
- 206.Sadowski RN, Wise LM, Park PY, Schantz SL, Juraska JM Early exposure to bisphenol A alters neuron and glia number in the rat prefrontal cortex of adult males, but not females. Neuroscience. 2014;279:122–131. doi: 10.1016/j.neuroscience.2014.08.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Sandin S, Lichtenstein P, Kuja-Halkola R, Hultman C, Larsson H, Reichenberg A The heritability of autism spectrum disorder. The Journal of the American Medical Association. 2017;318(12):1182–1184. doi: 10.1001/jama.2017.12141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Sándor S, Czeibert K, Salamon A, Kubinyi E Man's best friend in life and death: scientific perspectives and challenges of dog brain banking. GeroScience. 2021;43(4):1653–1668. doi: 10.1007/s11357-021-00373-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Sarrouilhe D, Dejean C Autism spectrum disorders and bisphenol A: is serotonin the lacking link in the chain? L'Encéphale. 2017;43(4):402–404. doi: 10.1016/j.encep.2016.04.007. [DOI] [PubMed] [Google Scholar]
- 210.Sato A, Kasai S, Kobayashi T, Takamatsu Y, Hino O, Ikeda K, et al Rapamycin reverses impaired social interaction in mouse models of tuberous sclerosis complex. Nature Communications. 2012;3:1292. doi: 10.1038/ncomms2295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Satomoto M, Satoh Y, Terui K, Miyao H, Takishima K, Ito M, et al Neonatal exposure to sevoflurane induces abnormal social behaviors and deficits in fear conditioning in mice. Anesthesiology. 2009;110(3):628–637. doi: 10.1097/ALN.0b013e3181974fa2. [DOI] [PubMed] [Google Scholar]
- 212.Satterstrom FK, Kosmicki JA, Wang JB, Breen MS, De Rubeis S, An JY, et al Large-scale exome sequencing study implicates both developmental and functional changes in the neurobiology of autism. Cell. 2020;180(3):568–584.e23. doi: 10.1016/j.cell.2019.12.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Savier E, Sedigh-Sarvestani M, Wimmer R, Fitzpatrick D A bright future for the tree shrew in neuroscience research: summary from the inaugural Tree Shrew Users Meeting. Zoological Research. 2021;42(4):478–481. doi: 10.24272/j.issn.2095-8137.2021.178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Saxena K, Webster J, Hallas-Potts A, Mackenzie R, Spooner PA, Thomson D, et al Experiential contributions to social dominance in a rat model of fragile-X syndrome. Proceedings of the Royal Society B:Biological Sciences. 2018;285(1880):20180294. doi: 10.1098/rspb.2018.0294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Schiavi S, Carbone E, Melancia F, Buzzelli V, Manduca A, Campolongo P, et al Perinatal supplementation with omega-3 fatty acids corrects the aberrant social and cognitive traits observed in a genetic model of autism based on FMR1 deletion in rats. Nutritional Neuroscience. 2020;11:1–14. doi: 10.1080/1028415X.2020.1819107. [DOI] [PubMed] [Google Scholar]
- 216.Schmitt O, O'Driscoll K, Baxter EM Exploratory study of the effects of intra-uterine growth retardation and neonatal energy supplementation of low birth-weight piglets on their post-weaning cognitive abilities. Animal Cognition. 2019;22(3):373–385. doi: 10.1007/s10071-019-01251-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Scotland P, Zhou DX, Benveniste H, Bennett V Nervous system defects of AnkyrinB (-/-) mice suggest functional overlap between the cell adhesion molecule L1 and 440-kD AnkyrinB in premyelinated axons . Journal of Cell Biology. 1998;143(5):1305–1315. doi: 10.1083/jcb.143.5.1305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Scott KE, Kazazian K, Mann RS, Möhrle D, Schormans AL, Schmid S, et al Loss of Cntnap2 in the rat causes autism-related alterations in social interactions, stereotypic behavior, and sensory processing . Autism Research. 2020;13(10):1698–1717. doi: 10.1002/aur.2364. [DOI] [PubMed] [Google Scholar]
- 219.Shahbazian MD, Young JI, Yuva-Paylor LA, Spencer CM, Antalffy BA, Noebels JL, et al Mice with truncated MeCP2 recapitulate many Rett syndrome features and display hyperacetylation of histone H3. Neuron. 2002;35(2):243–254. doi: 10.1016/S0896-6273(02)00768-7. [DOI] [PubMed] [Google Scholar]
- 220.Sharon G, Cruz NJ, Kang DW, Gandal MJ, Wang B, Kim YM, et al Human gut microbiota from autism spectrum disorder promote behavioral symptoms in mice. Cell. 2019;177(6):1600–1618.e17. doi: 10.1016/j.cell.2019.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Shultz SR, MacFabe DF, Martin S, Jackson J, Taylor R, Boon F, et al Intracerebroventricular injections of the enteric bacterial metabolic product propionic acid impair cognition and sensorimotor ability in the Long-Evans rat: further development of a rodent model of autism. Behavioural Brain Research. 2009;200(1):33–41. doi: 10.1016/j.bbr.2008.12.023. [DOI] [PubMed] [Google Scholar]
- 222.Silverman JL, Crawley JN The promising trajectory of autism therapeutics discovery. Drug Discovery Today. 2014;19(7):838–844. doi: 10.1016/j.drudis.2013.12.007. [DOI] [PubMed] [Google Scholar]
- 223.Simmons DH, Titley HK, Hansel C, Mason P Behavioral tests for mouse models of autism: an argument for the inclusion of cerebellum-controlled motor behaviors. Neuroscience. 2021;462:303–319. doi: 10.1016/j.neuroscience.2020.05.010. [DOI] [PubMed] [Google Scholar]
- 224.Singer HS, Morris C, Gause C, Pollard M, Zimmerman AW, Pletnikov M. 2009. Prenatal exposure to antibodies from mothers of children with autism produces neurobehavioral alterations: a pregnant dam mouse model. Journal of Neuroimmunology, 211(1–2): 39–48.
- 225.Sledziowska M, Kalbassi S, Baudouin SJ. 2020. Complex interactions between genes and social environment cause phenotypes associated with autism spectrum disorders in mice. eNeuro, 7(4),doi: 10.1523/ENEURO.0124-20.2020.
- 226.Smith SEP, Zhou YD, Zhang GP, Jin Z, Stoppel DC, Anderson MP Increased gene dosage of Ube3a results in autism traits and decreased glutamate synaptic transmission in mice . Science Translational Medicine. 2011;3(103):103ra97. doi: 10.1126/scitranslmed.3002627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Søndergaard LV, Ladewig J, Dagnæs-Hansen F, Herskin MS, Holm IE Object recognition as a measure of memory in 1–2 years old transgenic minipigs carrying the APPsw mutation for Alzheimer's disease. Transgenic Research. 2012;21(6):1341–1348. doi: 10.1007/s11248-012-9620-4. [DOI] [PubMed] [Google Scholar]
- 228.Stark KL, Xu B, Bagchi A, Lai WS, Liu H, Hsu R, et al Altered brain microRNA biogenesis contributes to phenotypic deficits in a 22q11-deletion mouse model. Nature Genetics. 2008;40(6):751–760. doi: 10.1038/ng.138. [DOI] [PubMed] [Google Scholar]
- 229.Stein TP, Schluter MD, Steer RA, Guo LN, Ming X Bisphenol A exposure in children with autism spectrum disorders. Autism Research. 2015;8(3):272–283. doi: 10.1002/aur.1444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Stessman HAF, Willemsen MH, Fenckova M, Penn O, Hoischen A, Xiong B, et al Disruption of POGZ is associated with intellectual disability and autism spectrum disorders . The American Journal of Human Genetics. 2016;98(3):541–552. doi: 10.1016/j.ajhg.2016.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Stessman HAF, Xiong B, Coe BP, Wang TY, Hoekzema K, Fenckova M, et al Targeted sequencing identifies 91 neurodevelopmental-disorder risk genes with autism and developmental-disability biases. Nature Genetics. 2017;49(4):515–526. doi: 10.1038/ng.3792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Stewart AM, Grieco F, Tegelenbosch RAJ, Kyzar EJ, Nguyen M, Kaluyeva A, et al A novel 3D method of locomotor analysis in adult zebrafish: implications for automated detection of CNS drug-evoked phenotypes. Journal of Neuroscience Methods. 2015;255:66–74. doi: 10.1016/j.jneumeth.2015.07.023. [DOI] [PubMed] [Google Scholar]
- 233.Südhof TC Synaptic neurexin complexes: a molecular code for the logic of neural circuits. Cell. 2017;171(4):745–769. doi: 10.1016/j.cell.2017.10.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Sugathan A, Biagioli M, Golzio C, Erdin S, Blumenthal I, Manavalan P, et al. 2014. CHD8 regulates neurodevelopmental pathways associated with autism spectrum disorder in neural progenitors. Proceedings of the National Academy of Sciences of the United States of America, 111(42): E4468–E4477.
- 235.Suliman-Lavie R, Title B, Cohen Y, Hamada N, Tal M, Tal N, et al Pogz deficiency leads to transcription dysregulation and impaired cerebellar activity underlying autism-like behavior in mice . Nature Communications. 2020;11(1):5836. doi: 10.1038/s41467-020-19577-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Sundberg M, Tochitsky I, Buchholz DE, Winden K, Kujala V, Kapur K, et al Purkinje cells derived from TSC patients display hypoexcitability and synaptic deficits associated with reduced FMRP levels and reversed by rapamycin. Molecular Psychiatry. 2018;23(11):2167–2183. doi: 10.1038/s41380-018-0018-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Sztainberg Y, Zoghbi HY Lessons learned from studying syndromic autism spectrum disorders. Nature Neuroscience. 2016;19(11):1408–1417. doi: 10.1038/nn.4420. [DOI] [PubMed] [Google Scholar]
- 238.Szulwach KE, Li XK, Smrt RD, Li YJ, Luo YP, Lin L, et al Cross talk between microRNA and epigenetic regulation in adult neurogenesis. Journal of Cell Biology. 2010;189(1):127–141. doi: 10.1083/jcb.200908151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Taieb O, Baleyte JM, Mazet P, Fillet AM Borna disease virus and psychiatry. European Psychiatry. 2001;16(1):3–10. doi: 10.1016/S0924-9338(00)00529-0. [DOI] [PubMed] [Google Scholar]
- 240.Takumi T The neurobiology of mouse models syntenic to human chromosome 15q. Journal of Neurodevelopmental Disorders. 2011;3(3):270–281. doi: 10.1007/s11689-011-9088-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Talkowski ME, Rosenfeld JA, Blumenthal I, Pillalamarri V, Chiang C, Heilbut A, et al Sequencing chromosomal abnormalities reveals neurodevelopmental loci that confer risk across diagnostic boundaries. Cell. 2012;149(3):525–537. doi: 10.1016/j.cell.2012.03.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Tang WL, Davidson JD, Zhang GQ, Conen KE, Fang J, Serluca F, et al Genetic control of collective behavior in zebrafish. iScience. 2020;23(3):100942. doi: 10.1016/j.isci.2020.100942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Tatavarty V, Pacheco AT, Kuhnle CG, Lin H, Koundinya P, Miska NJ, et al Autism-associated Shank3 is essential for homeostatic compensation in rodent V1. Neuron. 2020;106(5):769–777.e4. doi: 10.1016/j.neuron.2020.02.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Tazumi T, Hori E, Uwano T, Umeno K, Tanebe K, Tabuchi E, et al Effects of prenatal maternal stress by repeated cold environment on behavioral and emotional development in the rat offspring. Behavioural Brain Research. 2005;162(1):153–160. doi: 10.1016/j.bbr.2005.03.006. [DOI] [PubMed] [Google Scholar]
- 245.Testard C, Tremblay S, Platt M From the field to the lab and back: neuroethology of primate social behavior. Current Opinion in Neurobiology. 2021;68:76–83. doi: 10.1016/j.conb.2021.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.The Schizophrenia Psychiatric Genome-Wide Association Study (GWAS) Consortium Genome-wide association study identifies five new schizophrenia loci. Nature Genetics. 2011;43(10):969–976. doi: 10.1038/ng.940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Tick B, Bolton P, Happé F, Rutter M, Rijsdijk F Heritability of autism spectrum disorders: a meta-analysis of twin studies. Journal of Child Psychology and Psychiatry. 2016;57(5):585–595. doi: 10.1111/jcpp.12499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Tomova A, Husarova V, Lakatosova S, Bakos J, Vlkova B, Babinska K, et al Gastrointestinal microbiota in children with autism in Slovakia. Physiology & Behavior. 2015;138:179–187. doi: 10.1016/j.physbeh.2014.10.033. [DOI] [PubMed] [Google Scholar]
- 249.Topál J, Román V, Turcsán B The dog (Canis familiaris) as a translational model of autism: it is high time we move from promise to reality . Wiley Interdisciplinary Reviews Cognitive Science. 2019;10(4):e1495. doi: 10.1002/wcs.1495. [DOI] [PubMed] [Google Scholar]
- 250.Tromp A, Mowry B, Giacomotto J Neurexins in autism and schizophrenia-a review of patient mutations, mouse models and potential future directions. Molecular Psychiatry. 2021;26(3):747–760. doi: 10.1038/s41380-020-00944-8. [DOI] [PubMed] [Google Scholar]
- 251.Tsai PT, Hull C, Chu YX, Greene-Colozzi E, Sadowski AR, Leech JM, et al Autistic-like behaviour and cerebellar dysfunction in Purkinje cell Tsc1 mutant mice . Nature. 2012;488(7413):647–651. doi: 10.1038/nature11310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Tu ZC, Zhao H, Li B, Yan S, Wang L, Tang YJ, et al CRISPR/Cas9-mediated disruption of SHANK3 in monkey leads to drug-treatable autism-like symptoms . Human Molecular Genetics. 2019;28(4):561–571. doi: 10.1093/hmg/ddy367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Tucci A, Ciaccio C, Scuvera G, Esposito S, Milani D MIR137 is the key gene mediator of the syndromic obesity phenotype of patients with 1p21.3 microdeletions . Molecular Cytogenetics. 2016;9:80. doi: 10.1186/s13039-016-0289-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Varea O, Martin-de-Saavedra MD, Kopeikina KJ, Schürmann B, Fleming HJ, Fawcett-Patel JM, et al Synaptic abnormalities and cytoplasmic glutamate receptor aggregates in contactin associated protein-like 2/Caspr2 knockout neurons . Proceedings of the National Academy of Sciences of the United States of America. 2015;112(19):6176–6181. doi: 10.1073/pnas.1423205112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Varghese M, Keshav N, Jacot-Descombes S, Warda T, Wicinski B, Dickstein DL, et al Autism spectrum disorder: neuropathology and animal models. Acta Neuropathologica. 2017;134(4):537–566. doi: 10.1007/s00401-017-1736-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Vermeer LMM, Gregory E, Winter MK, McCarson KE, Berman NEJ Exposure to bisphenol A exacerbates migraine-like behaviors in a multibehavior model of rat migraine. Toxicological Sciences. 2014;137(2):416–427. doi: 10.1093/toxsci/kft245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Vieira MM, Jeong J, Roche KW The role of NMDA receptor and neuroligin rare variants in synaptic dysfunction underlying neurodevelopmental disorders. Current Opinion in Neurobiology. 2021;69:93–104. doi: 10.1016/j.conb.2021.03.001. [DOI] [PubMed] [Google Scholar]
- 258.Wang HLV, Forestier S, Corces VG Exposure to sevoflurane results in changes of transcription factor occupancy in sperm and inheritance of autism. Biology of Reproduction. 2021;105(3):705–719. doi: 10.1093/biolre/ioab097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Wang XM, Bey AL, Katz BM, Badea A, Kim N, David LK, et al Altered mGluR5-Homer scaffolds and corticostriatal connectivity in a Shank3 complete knockout model of autism . Nature Communications. 2016;7:11459. doi: 10.1038/ncomms11459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Wang ZJ, Zhong P, Ma KJ, Seo JS, Yang FW, Hu ZH, et al Amelioration of autism-like social deficits by targeting histone methyltransferases EHMT1/2 in Shank3-deficient mice . Molecular Psychiatry. 2020;25(10):2517–2533. doi: 10.1038/s41380-019-0351-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Warren WC, Clayton DF, Ellegren H, Arnold AP, Hillier LW, Kunstner A, et al The genome of a songbird. Nature. 2010;464(7289):757–762. doi: 10.1038/nature08819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Weinstein TAR, Bales KL, Maninger N, Hostetler CM, Capitanio JP Early involvement in friendships predicts later plasma concentrations of oxytocin and vasopressin in juvenile rhesus macaques (Macaca mulatta) . Frontiers in Behavioral Neuroscience. 2014;8:295. doi: 10.3389/fnbeh.2014.00295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Weinstein TAR, Capitanio JP Longitudinal stability of friendships in rhesus monkeys (Macaca mulatta): individual- and relationship-level effects . Journal of Comparative Psychology. 2012;126(1):97–108. doi: 10.1037/a0025607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Weiss LA, Shen YP, Korn JM, Arking DE, Miller DT, Fossdal R, et al Association between microdeletion and microduplication at 16p11.2 and autism. The New England Journal of Medicine. 2008;358(7):667–675. doi: 10.1056/NEJMoa075974. [DOI] [PubMed] [Google Scholar]
- 265.Werling DM, Brand H, An JY, Stone MR, Zhu LX, Glessner JT, et al An analytical framework for whole-genome sequence association studies and its implications for autism spectrum disorder. Nature Genetics. 2018;50(5):727–736. doi: 10.1038/s41588-018-0107-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.White J, Beck CR, Harel T, Posey JE, Jhangiani SN, Tang S, et al POGZ truncating alleles cause syndromic intellectual disability . Genome Medicine. 2016;8(1):3. doi: 10.1186/s13073-015-0253-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Willemsen MH, Vallès A, Kirkels LAMH, Mastebroek M, Loohuis NO, Kos A, et al Chromosome 1p21.3 microdeletions comprising DPYD and MIR137 are associated with intellectual disability. Journal of Medical Genetics. 2011;48(12):810–818. doi: 10.1136/jmedgenet-2011-100294. [DOI] [PubMed] [Google Scholar]
- 268.Wintler T, Schoch H, Frank MG, Peixoto L Sleep, brain development, and autism spectrum disorders: Insights from animal models. Journal of Neuroscience Research. 2020;98(6):1137–1149. doi: 10.1002/jnr.24619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Wöhr M, Silverman JL, Scattoni ML, Turner SM, Harris MJ, Saxena R, et al Developmental delays and reduced pup ultrasonic vocalizations but normal sociability in mice lacking the postsynaptic cell adhesion protein neuroligin2. Behavioural Brain Research. 2013;251:50–64. doi: 10.1016/j.bbr.2012.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Wolf U, Rapoport MJ, Schweizer TA Evaluating the affective component of the cerebellar cognitive affective syndrome. The Journal of Neuropsychiatry and Clinical Neurosciences. 2009;19(2):245–253. doi: 10.1176/jnp.2009.21.3.245. [DOI] [PubMed] [Google Scholar]
- 271.Wolstenholme JT, Drobná Z, Henriksen AD, Goldsby JA, Stevenson R, Irvin JW, et al Transgenerational bisphenol A causes deficits in social recognition and alters postsynaptic density genes in mice. Endocrinology. 2019;160(8):1854–1867. doi: 10.1210/en.2019-00196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Wolstenholme JT, Goldsby JA, Rissman EF Transgenerational effects of prenatal bisphenol A on social recognition. Hormones and Behavior. 2013;64(5):833–839. doi: 10.1016/j.yhbeh.2013.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Won H, Lee HR, Gee HY, Mah W, Kim JI, Lee J, et al Autistic-like social behaviour in Shank2-mutant mice improved by restoring NMDA receptor function . Nature. 2012;486(7402):261–265. doi: 10.1038/nature11208. [DOI] [PubMed] [Google Scholar]
- 274.Wong H, Hoeffer C Maternal IL-17A in autism. Experimental Neurology. 2018;299:228–240. doi: 10.1016/j.expneurol.2017.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Wu WL Association among gut microbes, intestinal physiology, and autism. EBioMedicine. 2017;25:11–12. doi: 10.1016/j.ebiom.2017.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Wu WL, Adame MD, Liou CW, Barlow JT, Lai TT, Sharon G, et al Microbiota regulate social behaviour via stress response neurons in the brain. Nature. 2021;595(7867):409–414. doi: 10.1038/s41586-021-03669-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Wu Y, Zhong WW, Cui NR, Johnson CM, Xing H, Zhang S, et al Characterization of Rett Syndrome-like phenotypes in Mecp2-knockout rats . Journal of Neurodevelopmental Disorders. 2016;8:23. doi: 10.1186/s11689-016-9156-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Xu L, Yu DD, Ma YH, Yao YL, Luo RH, Feng XL, et al COVID-19-like symptoms observed in Chinese tree shrews infected with SARS-CoV-2. Zoological Research. 2020;41(5):517–526. doi: 10.24272/j.issn.2095-8137.2020.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Xu XH, Dong FN, Yang YL, Wang YU, Wang R, Shen XY Sex-specific effects of long-term exposure to bisphenol-A on anxiety- and depression-like behaviors in adult mice. Chemosphere. 2015;120:258–266. doi: 10.1016/j.chemosphere.2014.07.021. [DOI] [PubMed] [Google Scholar]
- 280.Xu XX, Li CY, Gao XB, Xia K, Guo H, Li YL, et al Excessive UBE3A dosage impairs retinoic acid signaling and synaptic plasticity in autism spectrum disorders. Cell Research. 2018;28(1):48–68. doi: 10.1038/cr.2017.132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Yang M, Mahrt EJ, Lewis F, Foley G, Portmann T, Dolmetsch RE, et al 16p11.2 deletion syndrome mice display sensory and ultrasonic vocalization deficits during social interactions. Autism Research. 2015;8(5):507–521. doi: 10.1002/aur.1465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Yang R, Walder-Christensen KK, Kim N, Wu DW, Lorenzo DN, Badea A, et al ANK2 autism mutation targeting giant ankyrin-B promotes axon branching and ectopic connectivity . Proceedings of the National Academy of Sciences of the United States of America. 2019;116(30):15262–15271. doi: 10.1073/pnas.1904348116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Yao YG Creating animal models, why not use the Chinese tree shrew (Tupaia belangeri chinensis)? . Zoological Research. 2017;38(3):118–126. doi: 10.24272/j.issn.2095-8137.2017.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Yatawara CJ, Einfeld SL, Hickie IB, Davenport TA, Guastella AJ The effect of oxytocin nasal spray on social interaction deficits observed in young children with autism: a randomized clinical crossover trial. Molecular Psychiatry. 2016;21(9):1225–1231. doi: 10.1038/mp.2015.162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Ye MS, Zhang JY, Yu DD, Xu M, Xu L, Lv LB, et al Comprehensive annotation of the Chinese tree shrew genome by large-scale RNA sequencing and long-read isoform sequencing. Zoological Research. 2021;42(6):692–709. doi: 10.24272/j.issn.2095-8137.2021.272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Ye YZ, Cho MT, Retterer K, Alexander N, Ben-Omran T, Al-Mureikhi M, et al De novo POGZ mutations are associated with neurodevelopmental disorders and microcephaly . Cold Spring Harbor Molecular Case Studies. 2015;1(1):a000455. doi: 10.1101/mcs.a000455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Yenkoyan K, Grigoryan A, Fereshetyan K, Yepremyan D Advances in understanding the pathophysiology of autism spectrum disorders. Behavioural Brain Research. 2017;331:92–101. doi: 10.1016/j.bbr.2017.04.038. [DOI] [PubMed] [Google Scholar]
- 288.Yi JJ, Berrios J, Newbern JM, Snider WD, Philpot BD, Hahn KM, et al An autism-linked mutation disables phosphorylation control of UBE3A. Cell. 2015;162(4):795–807. doi: 10.1016/j.cell.2015.06.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Yin XM, Jones N, Yang J, Asraoui N, Mathieu ME, Cai LW, et al Delayed motor learning in a 16p11.2 deletion mouse model of autism is rescued by locus coeruleus activation. Nature Neuroscience. 2021;24(5):646–657. doi: 10.1038/s41593-021-00815-7. [DOI] [PubMed] [Google Scholar]
- 290.Zerbo O, Iosif AM, Walker C, Ozonoff S, Hansen RL, Hertz-Picciotto I Is maternal influenza or fever during pregnancy associated with autism or developmental delays? Results from the CHARGE (CHildhood Autism Risks from Genetics and Environment) study. Journal of Autism and Developmental Disorders. 2013;43(1):25–33. doi: 10.1007/s10803-012-1540-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Zhang B, Zhou ZG, Zhou Y, Zhang T, Ma YY, Niu YY, et al Social-valence-related increased attention in rett syndrome cynomolgus monkeys: an eye-tracking study. Autism Research. 2019;12(11):1585–1597. doi: 10.1002/aur.2189. [DOI] [PubMed] [Google Scholar]
- 292.Zhang J, Luo RC, Man XY, Lv LB, Yao YG, Zheng M The anatomy of the skin of the Chinese tree shrew is very similar to that of human skin. Zoological Research. 2020;41(2):208–212. doi: 10.24272/j.issn.2095-8137.2020.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Zhou B, Chen LM, Liao P, Huang L, Chen Z, Liao DQ, et al Astroglial dysfunctions drive aberrant synaptogenesis and social behavioral deficits in mice with neonatal exposure to lengthy general anesthesia. PLoS Biology. 2019a;17(8):e3000086. doi: 10.1371/journal.pbio.3000086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Zhou H, Xu X, Yan WL, Zou XB, Wu LJ, Luo XR, et al Prevalence of autism spectrum disorder in China: a nationwide multi-center population-based study among children aged 6 to 12 years. Neuroscience Bulletin. 2020;36(9):961–971. doi: 10.1007/s12264-020-00530-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Zhou Y, Sharma J, Ke Q, Landman R, Yuan JL, Chen H, et al Atypical behaviour and connectivity in SHANK3-mutant macaques . Nature. 2019b;570(7761):326–331. doi: 10.1038/s41586-019-1278-0. [DOI] [PubMed] [Google Scholar]
- 296.Zhan YF, Wei JZ, Liang J, Xu X, He R, Robbins TW, et al Diagnostic classification for human autism and obsessive-compulsive disorder based on machine learning from a primate genetic model. The American Journal of Psychiatry. 2021;178(1):65–76. doi: 10.1176/appi.ajp.2020.19101091. [DOI] [PubMed] [Google Scholar]
- 297.Zimmerman AW, Connors SL, Matteson KJ, Lee LC, Singer HS, Castaneda JA, et al Maternal antibrain antibodies in autism. Brain, Behavior, and Immunity. 2007;21(3):351–357. doi: 10.1016/j.bbi.2006.08.005. [DOI] [PubMed] [Google Scholar]