Abstract
While it took decades to arrive to a conclusion that ferritin is more than an indicator of iron storage level, it took a short period of time through the COVID-19 pandemic to wonder what the reason behind high levels of ferritin in patients with severe COVID-19 might be. Unsurprisingly, acute phase reactant was not a satisfactory explanation. Moreover, the behavior of ferritin in patients with severe COVID-19 and the subsequent high mortality rates in patients with high ferritin levels necessitated further investigations to understand the role of ferritin in the diseases. Ferritin was initially described to accompany various acute infections, both viral and bacterial, indicating an acute response to inflammation. However, with the introduction of the hyperferritinemic syndrome connecting four severe pathological conditions such as adult-onset Still's disease, macrophage activation syndrome, catastrophic antiphospholipid syndrome, and septic shock added another aspect of ferritin where it could have a pathogenetic role rather than an extremely elevated protein only. In fact, suggesting that COVID-19 is a new member in the spectrum of hyperferritinemic syndrome besides the four mentioned conditions could hopefully direct further search on the pathogenetic role of ferritin. Doubtlessly, improving our understanding of those aspects of ferritin would enormously contribute to better coping with severe diseases in terms of treatment and prevention of complications. The origin, history, importance, and the advances of searching the role of ferritin in various pathological and clinical processes are presented hereby in our article. In addition, the implications of ferritin in COVID-19 are addressed.
Keywords: Ferritin, Iron homeostasis, Acute phase reactant, Hyperferritinemia, COVID-19
1. Introduction
Initially addressed as a surrogate marker for the status of iron storage in the human body, ferritin which is derived from the Latin word “ferratus” for iron-bearing, was considered to be merely related to iron and iron level in the human body [1]. Low ferritin levels indicated decreased body iron levels whereas high levels indicated normal or increased iron levels [2].
Later by the seventies, ferritin was shown to be increased in acute infectious diseases [3] hence aided in the explanation of increased ferritin levels in acute inflammatory conditions, the majority caused by infections. Consequently, ferritin starred in medical textbooks as a marker that differentiated between iron deficiency anemia and anemia of chronic diseases since it is higher in the latter due to inflammation and low in the first secondary to decreased iron levels [4].
Later on, elevated ferritin levels that were previously related to acute or chronic inflammatory conditions whether or not caused by an infection, were shown to play a central role in the pathogenesis of various inflammatory and autoimmune diseases [5,6]. In fact, the description of hyperferritinemia, Shoenfeld's syndrome, as “an iron sword of autoimmunity” [7] sums up many years of searching for the significance of ferritin in severe diseases. Indeed, the mere fact that high ferritin levels were addressed as a syndrome “hyperferritinemic syndrome” sheds light on the huge implication of high levels of ferritin in the pathogenesis and severity of those diseases [8]. The correlation between hyperferritinemia and the four major pathologies of adult-onset Still's disease, macrophage activation syndrome, catastrophic antiphospholipid syndrome and septic shock is a great example of the implication of ferritin, particularly as all share in common a very severe disease course with high mortality [9].
And finally, when the pandemic of COVID-19 came to the attention of the entire world by the start of the year 2020 [10], ferritin was forced back into the debate regarding its role in severe diseases such as the newly born COVID-19 [[11], [12], [13]]. For instance, and early in the pandemic, ferritin served as a marker of severity and prognostic factor of COVID-19 [[14], [15], [16]].
Therefore and based on that, we directed our work toward the origin of ferritin, its role in iron hemostasis, the appearance of the acute phase protein era, the special features of hyperferritinemia, the introduction and significance of the hyperferritinemic syndrome, and eventually the early presence of ferritin in the pandemic of COVID-19, to an extent of calling ferritin an additional piece in the puzzle of the hyperferritinemic syndrome [17].
2. Ferritin
First isolated from a horse spleen in 1937, ferritin was defined as a crystallizable protein that contained more than 20% of iron [18]. Few years later, Granick showed that ferritin consisted of 54.5% protein, 12.1% nucleic acid, and 35% ferric oxide-hydroxide [19]. Furthermore, the same author wrote later on about the importance of ferritin in iron metabolism and homeostasis [20].
Detection and quantification of ferritin were not possible until 1972 when ferritin purification was achieved and anti-ferritin antibodies were developed [21]. Following the year 1975, ferritin was widely accepted as an iron storage protein of both prokaryotes and eukaryotes where ferritin was found to store iron in most tissues as a cytosolic protein [22]. Since then, measurement of serum ferritin level served as a new and indirect method to assess the intracellular iron. Considering the fact that ferritin is the primary iron storage mechanism, ferritin plays a critical role in iron homeostasis. For instance, ferritin makes iron available for essential cellular activities including protecting DNA and proteins from the potentially toxic effects of iron [23]. Furthermore, ferritin is now known to protect cells from the toxic effects of free iron, besides being involved in different functions including immune regulation [24].
In regard to iron storage, iron is stored in two proteins, either in ferritin or in hemosiderin. Hemosiderin is predominantly present within macrophages and low to undetectable levels in the plasma and thought to be a result of phagocytosis of damaged red blood cells. When the quantity of iron exceeds ferritin storage capacity, ferritin cages become damaged [25]. Ferritin and hemosiderin have high affinity and sensitivity to iron however, ferritin stores slightly more iron than hemosiderin in low iron conditions such as iron deficiency anemia, whereas in iron overload hemosiderin stores more iron than ferritin [26].
2.1. Structure of ferritin
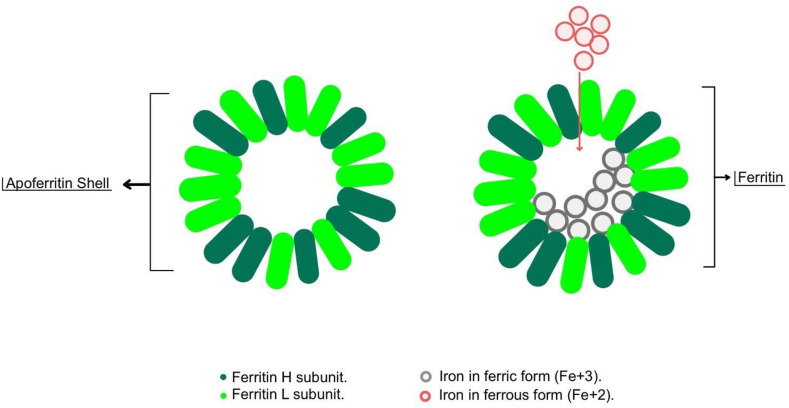
Ferritin is a large molecule of approximately 10–12 nm in diameter, formed by a spherical molecule called “apoferritin shell” which stores iron inside its hollow center and thus called ferritin [1,27] (Fig. 1 ). Each apoferritin shell can store up to approximately 4500 iron atoms [28].
Fig. 1.
Structure of ferritin. Ferritin is a large molecule formed by a spherical molecule called “apoferritin shell” which stores iron inside its hollow center and thus called ferritin.
In vertebrates, ferritin consist of 2 subunits forming the apoferritin shell, L and H subunits which are encoded by the FTL and FTH genes respectively [24]. The expression of the L and H subunits varies depending on the tissue type and physiological status of the cells. For instance, in spleen and liver, L subunit is the predominant one, while in the heart, H subunit predominates [29]. Both L and H subunits attach to each other and form the outer apoferritin shell. The N-terminal peptides, which are present in L and H subunits, extend in order to form the gates that allow ferrous to exit from ferritin molecule [30].
2.2. Synthesis of ferritin
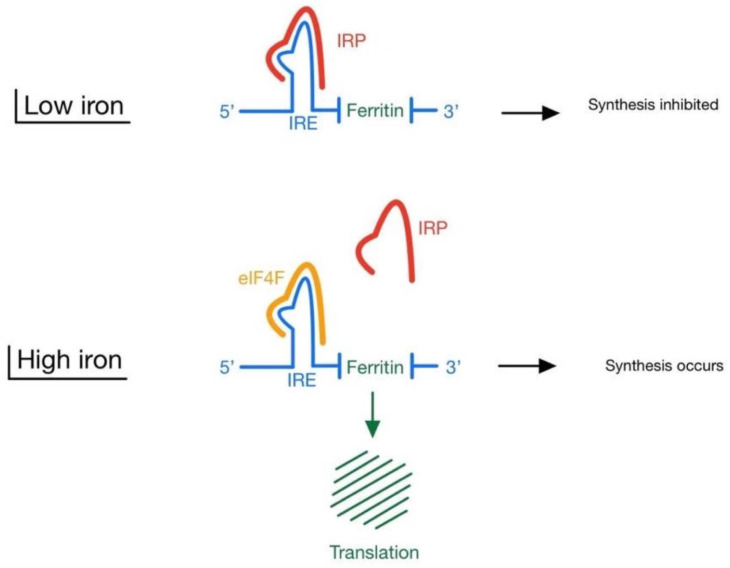
The process of ferritin synthesis has been mostly studied in animals. Synthesis of ferritin takes place at the level of the cytoplasmic 5′-untranslated mRNA [31]. Iron regulatory proteins 1 and 2 (IRP1 and IRP2) regulate ferritin synthesis at translational level by interacting with iron responsive element (IRE) found in 5′-untranslated region of ferritin mRNA [32]. In high intracellular iron conditions, IRP dissociates from IRE which in turn permits eIF4F binding to IRE. Following eIF4F-IRE binding, the IRE-RNA translation of ferritin begins thus building up new molecules of ferritin. On the other hand, when iron levels are low, IRP binds to IRE preventing eIF4F binding which blocks the process of ferritin synthesis [25] (Fig. 2 ).
Fig. 2.
Synthesis of ferritin: in low iron conditions, IRP binds to IRE preventing eIF4F binding thus blocking ferritin synthesis. In high intracellular iron conditions, IRP dissociates from IRE which in turn permits eIF4F binding to IRE. Following eIF4F-IRE binding, the IRE-RNA translation of ferritin begins and hence building up new molecules of ferritin. IRE: iron responsive element; IRP: iron regulatory protein.
The IRE-containing flanking region produces a highly stable stem-loop structure, with the six-membered loop demonstrating the high affinity interaction with IRP. It is noteworthy to mention that the interaction between the IRE and the IRP is an iron-dependent process [32].
2.3. Release of ferritin
Serum ferritin is predominantly L-ferritin which is released from the liver [33]. Several experiments conducted in liver of rats showed that secretion of ferritin is elicited by increased serum iron levels [34]. Moreover, several cytokines such as IL-6 and TNF-alpha were shown to stimulate the release of ferritin into the blood stream [35]. The latter explains high ferritin levels seen in inflammatory conditions, which is discussed below.
2.4. Functions of ferritin
Though ferritin is a major contributor for iron homeostasis [36], it is implicated in various physiological and pathological conditions (Fig. 3 ). In terms of iron homeostasis, both iron deficiency and overload might lead to deleterious consequences, hence the role of ferritin in balancing iron concentration cannot be overemphasized [37]. The pathological manifestations of iron excess correlate with the tissue site where iron accumulates. For example, pancreatic deposition of ferritin leads to diabetes [38], pituitary deposition leads to hypogonadism [39], skin iron deposits lead to hyperpigmentation [40], and liver accumulation leads to liver disease that may progress to cirrhosis [41]. Furthermore, recent studies have indicated that accumulation of iron has also been implicated in oncogenesis, aging, and progression of chronic neurodegenerative conditions, such as Alzheimer and Parkinson disease [23].
Fig. 3.
Functions and implications of ferritin.
Ferritin has multiple sites that catalyze the oxidoreduction of ferrous and O2 producing di-ferric mineral precursors. Those precursors migrate into the central cavity of ferritin and form ferric hydrated oxo mineral [42]. Though elevated ferritin levels in infections are viewed as an inflammatory marker, high levels are an important host defense mechanism due to toxicity of iron for bacteria resulting in inhibiting bacterial growth [43].
Ferritin can be found in cell organelles other than cytosol, such as the nuclei, mitochondria, and lysosome, where it can perform various different functions depending on the cellular context. As an example, mitochondrial ferritin can protect the cell from the ROS (reactive oxygen species) and thus, ferroptosis (programmed cell death depending on iron) [44]. In addition, nuclear ferritin was shown to be effective in protecting DNA from iron-induced oxidative damage [45]. In the cytosol, a small fraction of intracellular iron is maintained in the labile iron pool, while the majority is utilized in enzymes or sequestered in ferritin to prevent iron mediated oxidative damage [43].
2.5. Regulation of ferritin
While ferritin can store iron and release it when there is a decreased level, intracellular iron is the first and main regulatory factor of ferritin synthesis at both the transcriptional and the translational levels [21]. Ferritin production is tightly controlled at the translational stage by the interaction between iron regulatory protein (IRP) and the conserved iron responsive element (IRE) in the 5′-untranslated region of ferritin H and L subunit mRNAs as mentioned earlier [32]. Ferritin synthesis is also regulated by oxidative stress, hormones (thyroid and insulin), growth factors, second messengers, hypoxia-ischemia and hyperoxia [46].
In the liver, particularly in HepG2 hepatic cell line, while IL-1b increases the synthesis of both H and L subunits, IL-6 primarily induces the synthesis of the L-subunit [47]. In excess iron levels, iron generates toxic free radicals which might directly damage the hepatocytes [43].
2.6. Ferritin as an acute phase reactant
Acute phase response is a series of reactions conducted by host cells secondary to injury, trauma, infection, autoimmune diseases, or neoplasms [48]. Acute phase responses are aimed at inhibiting the processes involved in cell damage while promoting the processes involved in tissue repair [49]. Acute phase reactants are in fact acute phase proteins which are mainly produced and secreted by hepatocytes. Cytokines were found to have a major role in the regulation of acute phase protein synthesis. For instance, interleukin-1β (Il-1β), tumor necrosis factor-alpha (TNF- α) and interleukin-6 (Il-6) are pro-inflammatory cytokines thus stimulating the production of acute phase proteins [50]. In turn, interleukin-10 (Il-10) and transforming growth factor beta (TGF-β) are examples of anti-inflammatory cytokines downregulating the synthesis of acute phase proteins [51].
Acute phase reactants that are synthesized by the liver in relatively large quantities (over expressed) are denoted as “positive” acute phase proteins (APP) and include Hp, SA, fibrinogen, Cp, AGP, α-1-antitrypsin, lactoferrin, and CRP. The APP's synthesized at a lower level are termed “negative” and include proteins such as albumin, transferrin, and transthyretin [52]. Ferritin, a positive acute phase reactant presents in high concentrations in both intracellular and extracellular media. Ferritin has multiple forms depending on the ratio between its two subunits, the H and L subunits [53]. Since the functions of these two subunits differ, they determine the metabolic features of ferritin [54]. H subunit rich ferritins contribute to the intracellular iron traffic in terms of dynamism by accumulating and releasing iron faster [55]. On the other hand, L subunit rich ferritins with its ability of accumulating more iron increase preferentially when iron load occurs since L subunit rich ferritin is more stable than H subunit rich ferritin [56,57]. Up-regulated expression of both H and L subunit of ferritin is effective for protection against oxidative stress [58]. However, most stimuli directing ferritin synthesis in inflammation cause upregulation of H subunit rich ferritin synthesis more than L subunit rich ferritin indicating importance of rapid sequestration of iron and decrease in available cell iron in inflammation. Upregulation of H subunit rich ferritin also provides resistance to oxidative damage by hydroxyl radicals during inflammation by leaving little amount of iron available to produce these radicals by Fenton reaction [59].
Acting as an acute phase reactant, ferritin sequesters and stores iron intracellularly. The role of ferritin in iron homeostasis in the context of inflammation has a great importance in body protection against infection, injury, and cancer. For instance, Fenton reaction includes ferrous iron (Fe+2) reacting with H2O2 to generate hydroxyl radical, one of the most active oxygen radicals [60]. Oxygen radicals assist neutrophils and macrophages during phagocytosis by reacting with cellular components of phagocytosed materials. During inflammatory and infectious conditions, large amounts of oxygen radicals are produced. Subsequently, oxygen radicals leak into the fluids and tissues around inflammation leading to a considerable amount of cellular damage by reacting with the cellular components [61]. Therefore, a decrease in available iron by increasing ferritin levels protect against the damage free radical could produce at the inflammation site.
2.7. Autoimmune diseases and ferritin
Elevated ferritin levels have been described in various autoimmune diseases. The mechanism behind high levels of ferritin in autoimmune diseases is believed to be secondary to immune stimulation of ferritin synthesis by cytokines [62,63].
Rheumatoid arthritis is an autoimmune inflammatory disease characterized by inflammation of joints and increased level of TNFα and IL-1α [64]. Serum ferritin levels in patients with rheumatoid arthritis can be within the normal range however, synovial fluid and synovial cells have been shown to have an increased levels of ferritin [65]. In turn, patients with systemic juvenile arthritis were found to have elevated serum ferritin levels at the time of diagnosis [66]. The levels of ferritin decrease during therapy and its levels help guide the physicians in the use of glucocorticoids as described in the same study. Even though C-reactive protein demonstrates significant correlation with rheumatoid arthritis disease activity, similar and less significant correlation with serum ferritin levels was observed [67].
Systemic lupus erythematosus is a chronic autoimmune disease that has been shown to affect multiple organs and tissues due to various types of autoantibodies with diffuse inflammation [68]. Elevated acute phase proteins are not a characteristic of systemic lupus erythematosus unless an infection is accompanied however, high ferritin concentrations were detected in the urine of lupus patients with nephritis [69]. Another study demonstrated that serum ferritin levels are elevated in patients with more active disease [70].
Multiple sclerosis is another autoimmune disease which results in demyelination of central nervous system (CNS) [71]. As iron is required for myelin formation; iron dysregulation was shown to be involved in the pathogenesis of multiple sclerosis [72]. In fact, iron delivery to brain tissue is dependent on transferrin as transferrin receptors are located in the gray matter areas whereas ferritin binding sites are found in the white matter areas. Moreover, multiple sclerosis leads to differences in the levels of transferrin and ferritin binding sites within the demyelinating areas [73]. Whether a cause or result of demyelination, the causative correlation between demyelination and loss of ferritin binding remains unestablished. Additionally, hyperferritinemia was more common in patients with multiple sclerosis compared to healthy controls in a study conducted among 150 patients with multiple sclerosis [74].
Other autoimmune diseases shown to be associated with high ferritin levels are polymyositis and dermatomyositis especially in the elderly when compared to younger patients [75]. In addition, patients with thyroiditis were found to have elevated ferritin levels which decreased following treatment with anti-inflammatory drugs [76].
2.8. Infectious diseases and ferritin
High ferritin levels during acute infectious diseases have for a long period of time served as a good example of a strong correlate. A Scandinavian study from the seventies [3] which included 18 patients with acute infections, demonstrated an abrupt increase in serum ferritin immediately following infection. The authors showed that rapid elevation of serum ferritin level was similar among the enrolled patients regardless of whether the etiological agent was viral or bacterial. Furthermore, ferritin and haptoglobin displayed parallel behavior in terms of the rise and fall in serum levels. In contrast to the rapid elevation of serum ferritin at the beginning of the disease; it took as long as five weeks for ferritin levels to start dropping. An even longer duration until ferritin started to decrease in infectious diseases was reported in another study [77].
Bacterial infections were found to contribute to an increase in ferritin levels as well. Kawamata et al. [78] investigated ferritin levels in 22 Japanese children with Mycoplasma pneumonia infection. Although serum ferritin was increased in the acute phase of pneumonia, it rapidly decreased during the recovery phase. The authors suggested that ferritin levels could serve as an indicator for the severity of the disease. Moreover, ferritin was also proposed to serve as a diagnostic marker in patients with Legionella pneumonia lung infection [79].
In addition to bacteria, high ferritin concentrations have also been reported in viral infections. Elevated serum ferritin levels were described in influenza viral infections. For instance, increased ferritin levels were described in 6 out of 22 patients infected with H5N1 influenza virus [80]. Moreover, elevated ferritin levels were used as a rapid diagnostic surrogate marker in the diagnosis of Legionnaire's disease during the H1N1 epidemic, a marker that could aid in distinguishing H1N1 infection from Legionnaire's disease during the influenza season [81]. Furthermore, high ferritin levels were associated with worse prognosis in patients infected with influenza A [82]. Similarly, higher ferritin levels were correspondent with a poor immune response to the influenzae vaccine [83]. Besides influenza viruses, high ferritin levels were associated with hemorrhage and death in Ebola hemorrhagic fever [84]. An interesting case report of 2 patients with Chikungunya infection who developed hyperferritinemic syndrome manifested as adult-onset Still's diseases following the infection and reasoned to be due to the triggering of high ferritin levels by the viral infection and the subsequent development of auto-inflammatory disease [85].
In addition to the previously mentioned infectious agents, an elevation of serum ferritin has also been demonstrated in other bacterial and viral infections, such as EBV, HIV and tuberculosis [86].
Previous outbreaks of coronaviruses and ferritin:
In fact, data is scarce in regard to levels of ferritin during the severe acute respiratory syndrome (SARS) epidemic [87,88] and the Middle-East respiratory syndrome (MERS) outbreak in Saudi Arabia [89,90]. However, during the early SARS epidemic, a study from Taiwan describing the first 10 patients infected with the SARS virus showed high ferritin levels in 7 out the 10 patients enrolled [91]. The levels of ferritin ranged from 590 ng/ml to 4984 ng/ml. Furthermore, rising ferritin levels were found to be correlated with clinical deterioration in the patients studied.
2.9. Obesity and ferritin
Obesity is a worldwide health problem affecting millions of people across the globe [92]. The correlation of ferritin with obesity has been widely studied. Ferritin concentrations were significantly associated with metabolic syndrome and obesity in a study conducted among Mexican American men [93]. Interestingly, the findings were independent of age and body mass index. Another study was aimed at addressing the significance of ferritin levels in obese and overweight individuals [94]. Ferritin was found to have a positive correlation with both CRP levels and body mass index however, a negative association was reported with regards to correlations with levels of hemoglobin, iron, and transferrin. Based on the findings, the authors concluded that ferritin is a marker of inflammation in obese and overweight persons rather than a marker for iron deficiency.
2.10. Malignancy and ferritin
Ferritin levels are also overexpressed in many malignancies. In fact, the medical literature is replete with papers that describe an association between ferritin levels and a variety of malignancies. For instance, high ferritin levels were reported in solid tumors such as breast cancer in old and new studies, suggesting a correlation with the pathogenesis of the diseases alongside a marker for prognosis [95,96], non-small cell lung cancer [97] and pancreatic cancer [98]. In the latter, ferritin was demonstrated to act as an independent predictor of mortality in patients with pancreatic cancer.
In CNS malignancies, elevated cerebrospinal fluid (CSF) to serum ferritin ratio was postulated to mark active tumor synthesis in patients with glioblastoma, whereas serum ferritin levels were shown to serve for diseases activity and guide to therapy in patients with neuroblastoma [99]. Certain studies correlated the elevation of serum ferritin to higher risk of certain cancers. For instance, a Taiwanese cohort study demonstrated that the elevation of serum ferritin increased the risk for hepatocellular carcinoma [100]. The increase in serum ferritin was due to localized release inside the tumor site, the authors explained. Furthermore, surgical excision of tumors reduced serum ferritin levels by nearly 50% in individuals with increased serum ferritin, indicating a link between tumor bulk and serum ferritin elevation [101]. In addition, it has been demonstrated in vitro, that cancer cells from various malignancies release ferritin or ferritin-like compounds [[102], [103], [104]].
2.11. Hyperferritinemia and hyperferritinemic syndrome
The association of high levels of ferritin with various inflammatory as well as infectious diseases has been known for decades [8] and described in details above (Table 1 ). However, extremely high levels of ferritin were addressed as a separate pathological entity. For instance, hyperferritinemia is considered when ferritin levels increase up to more than 400 ng/ml [11]. Such high levels of ferritin were shown to be correlated with a constellation of symptoms constituting the “hyperferritinemic syndrome”, Shoenfeld's syndrome [9].
Table 1.
Autoimmune, inflammatory, and infectious diseases associated with high levels of ferritin.
| Autoimmune and Inflammatory diseases |
|---|
| Rheumatoid arthritis |
| Systemic juvenile arthritis |
| Systemic lupus erythematosus |
| Multiple sclerosis |
| Polymyositis |
| Dermatomyositis |
| Adult-onset Still's disease |
| Catastrophic antiphospholipid syndrome |
| Macrophage activation syndrome |
| Infections |
| Septic shock |
| Mycoplasma pneumonia |
| Legionella pneumonia |
| Influenza viral infections |
| H5N1 |
| H1N1 |
| Influenza A |
| Ebola hemorrhagic fever |
| Chikungunya |
| EBV |
| HIV |
| Tuberculosis |
| COVID-19 |
In 1998, Alberto Piperno addressed various diseases which share together elevated ferritin levels as a common mechanism. Piperno named the diseases as “hyperferritinemic conditions” [105]. The conditions, according to the author, were inconsistent and unlike previously known pathologies of iron overload due to congenital or acquired disorders, which were correlated mainly with iron overload.
In the year 2013, the group of Shoenfeld et al. presented hyperferritinemia as a continuation of a spectrum of disorders called “hyperferritinemic syndrome” [9]. The authors illustrated four pathological processes where hyperferritinemia was the common base and connector. The disorders were adult-onset Still's disease (AOSD), catastrophic antiphospholipid syndrome (cAPS), macrophage activation syndrome (MAS) and septic shock. The article concluded the similarities in clinical, laboratory and therapeutical aspects of the disorders eventually proposing to include those four uncommon diseases under one entity “hyperferritinemic syndrome”. Earlier during the year 2013, prior to suggesting the term “hyperferritinemic syndrome”, the same group illustrated that hyperferritinemia could serve as an early marker for secondary antiphospholipid syndrome in patients with systemic lupus erythematosus (SLE) [106].
The spectrum of hyperferritinemic syndrome consists of several inflammatory, autoimmune, and infectious diseases as follows:
3. Adult-onset Still's disease
Adult-onset Still's disease is a rare systemic auto-inflammatory disorder of unknown etiology [107]. Several infectious agents have been suspected as a trigger of the disease due to the similarities in clinical presentation [108,109] however, none has been proven to be the etiologic agent. Adult-onset Still's disease is characterized by spikes of fever accompanied by joints pain, and an evanescent salmon pink macular or maculopapular rash [110]. In addition, myalgia, lymphadenopathy, splenomegaly, and hepatomegaly are also found. Laboratory findings include among others elevated C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR), leukocytosis with neutrophilic predomination, anemia, elevated liver enzymes and hyperferritinemia [111].
Hyperferritinemia has been proposed as a tool to aid in the diagnosis of adult-onset Still's disease. For instance, several studies indicated a threshold of 1000 ng/ml for a possible diagnosis of adult-onset Still's disease [112]. Higher levels, to the upwards of 30,000 ng/ml were reported to be a common finding, reaching even as much as 250,000 ng/ml in some studies [113]. Therefore, even though the threshold was proposed to be at 1000 ng/ml; most patients presented with much higher levels.
Moreover, the value of ferritin in adult-onset Still's disease has been used as an indicator of the disease activity especially as it normalizes following the initiation of treatment and when the disease goes into remission [112]. In addition, levels of ferritin were directly correlated with disease activity [114,115]. Furthermore, in a study which included 147 patients with adult-onset Still's disease, ferritin alongside CRP were shown to serve as a predictor of both mortality and macrophage activation syndrome [116]. As macrophage activation syndrome is a life-threatening complication of adult-onset Still's disease, ferritin levels in this regard serve to guide clinicians in terms of early diagnosis, treatment, and prevention of complications.
Along with the previously mentioned implications of ferritin in adult-onset Still's disease which included disease activity, severity, and complications, it has been recently suggested that ferritin could be part of the pathogenesis of the disease, particularly following the several researches made in regard to ferritin in COVID-19 [117]. The findings were based on the fact that ferritin stimulates and amplifies the secretion of numerous cytokines.
4. Macrophage activation syndrome (MAS)
Macrophage activation syndrome (MAS) or often and interchangeably called secondary hemophagocytic lymphohistiocytosis (HLH), is a hyperimmune complication of a number of autoimmune diseases that include juvenile arthritis and systemic lupus erythematosus [118]. The disease is characterized by a rapid expansion and activation of T-lymphocytes and macrophages that express hemophagocytic activity [119]. Due to the similarities of clinical presentation with systemic inflammatory diseases, macrophage activation syndrome is often misdiagnosed or diagnosed late in the disease course which directly leads to high morbidity and mortality rates [120].
The clinical presentation of macrophage activation syndrome consists of high fever, skin manifestations such as purpura or petechiae, and edema [121]. Multiorgan involvement is also common including respiratory symptoms manifested by dyspnea, cough, or respiratory failure in severe cases; gastrointestinal as well as renal complications were also reported [122]. Neurological symptoms were also documented in patients with macrophage activation syndrome particularly during the early stages of the disease [123]. The CNS manifestations vary and include meningitis, encephalomyelitis, and cerebral hemorrhage. Laboratory findings in macrophage activation syndrome consist of pancytopenia, hyperferritinemia, abnormal liver function tests and elevated triglycerides [124].
Since macrophage activation syndrome resembles in clinical presentation several autoimmune and inflammatory diseases and while early diagnosis and treatment are lifesaving, a set of criteria for diagnosis were established. Ferritin level constituted one of the cornerstones of these criteria. For instance, a minimum of 500 ng/ml level is considered the threshold for the diagnosis of macrophage activation syndrome [120]. Nevertheless, levels of ferritin can be as high as 5000 ng/ml or even higher. One study showed that ferritin levels above 1000 ng/ml are almost specific for the diagnosis of macrophage activation syndrome [125]. In fact, hyperferritinemia is a must criterion for the diagnosis of macrophage activation syndrome. For example, and while juvenile idiopathic arthritis (JIA) is one of the most common associations with macrophage activation syndrome, Ravelli and a panel of experts [126] developed a diagnostic guideline for the diagnosis of MAS as a complication of systemic JIA. Unsurprisingly, a threshold of ferritin level higher than 684 ng/ml was included as an obligatory requirement for the diagnosis. The authors stressed the importance of the criteria in facilitating the diagnosis of macrophage activation syndrome, as well as providing a tool for better enrollment of patients in studies and hence permitting further evaluation of potential therapies.
It is noteworthy to mention, that previously proposed diagnostic criteria for macrophage activation syndrome were based on the HLH-2004 diagnostic guidelines [127]. The latter which set ferritin levels at 500 ng/ml, were used mainly for the diagnosis of primary HLH.
The presence and importance of ferritin and its value in all of the mentioned criteria and guidelines, shed light on the enormously important role of ferritin in macrophage activation syndrome.
5. Catastrophic anti-phospholipid syndrome
Antiphospholipid syndrome is a systemic autoimmune disease caused by the formation of autoantibodies against the phospholipid bound proteins of platelets leading to widespread intravascular thrombosis of arteries and veins [128]. Antiphospholipid syndrome may occur alone and called primary, or concomitantly with other systemic autoimmune disease, most commonly systemic lupus erythematosus thus named secondary [129]. The diagnosis of antiphospholipid syndrome is based on both clinical and laboratory criteria indicating thromboembolic phenomena and the presence of antiphospholipid antibodies [130].
A small percentage of patients with antiphospholipid syndrome may develop a severe form of the disease called catastrophic antiphospholipid syndrome [131]. The latter is characterized by disseminated microvascular thrombosis leading to multiorgan failure and high mortality rate [132]. Interestingly, ferritin was shown to be involved in antiphospholipid syndrome as well as in its catastrophic form. In a study which included 176 patients with antiphospholipid syndrome [133], 9% of the patients had high serum ferritin levels versus 0% of 98 healthy age and sex matched controls. Among the hyperferritinemic patients, ferritin levels correlated with venous thrombosis alongside cardiac, neurological, and hematological events. In regard to 14 patients with catastrophic antiphospholipid syndrome in the study, hyperferritinemia was reported in 71% of the patients. Ferritin levels were found significantly higher in comparison to patients with antiphospholipid syndrome without the catastrophic variant (816 ng/ml vs. 120 ng/ml). Based on their findings, the authors suggested a pathogenetic role of ferritin in the severe form of the disease especially that patients with milder form had significantly lower levels of ferritin.
6. Septic shock
As opposed to the above mentioned autoimmune and autoinflammatory members of the hyperferritinemic syndrome, septic shock represents the infectious part of the spectrum of hyperferritinemic syndrome. This connection is highly important when it comes to the following section of COVID-19 and ferritin.
Septic shock is a severe and life-threatening complication of sepsis [134]. While sepsis is a response to severe infection, septic shock is marked by a systemic hypoperfusion resulting in multiorgan failure [135]. Among 36 children with severe sepsis and septic shock admitted to the intensive care unit, Garcia et al. [136] demonstrated three subgroups according to ferritin levels as follows: 13 children had less than 200 ng/mL, 11 children had levels of 200–500 ng/mL, and 12 children had levels higher than 500 ng/mL. Accordingly, the mortality rate was found to be 23%, 9% and 58%, respectively. The authors presented ferritin levels of more than 500 ng/mL were associated with a significantly high mortality rate in comparison with lower levels. Moreover, a pathogenetic and immunomodulatory role of ferritin in sepsis were addressed in patients with sepsis-induced acute kidney injury in a recent study [137].
6.1. The pandemic of COVID-19
By the end of 2019, a new corona virus was reported to cause acute and severe respiratory illness in Wuhan, China [138]. At the beginning it was designated as a novel-corona virus however, it was changed latter on to be named SARS-CoV-2 based on the genomic similarities with the SARS virus that emerged during the winter season of 2002–2003 [87].
The constellation of the symptoms of SARS-CoV-2 infection were called COVID-19 which stands for corona virus disease of 2019. Due to the rapid and worldwide spread of the SARS-CoV-2 infection, COVID-19 was declared as a pandemic and global health emergency by the world health organization (WHO) in March 2020 [10].
As those words were being written, around 250 millions people were infected with SARS-CoV-2 worldwide and approximately 5 millions died secondary to COVID-19, according to the weekly epidemiological report issued by the WHO [139].
6.2. COVID-19 and ferritin
Following the global spread of the COVID-19 pandemic and early throughout the pandemic, elevated levels of ferritin in patients with COVID-19 brought the attention of medical staff treating huge numbers of patients [140] as well as researchers by predicting poor outcomes [141]. The possible correlation between ferritin and worse prognosis however, barely scratched the surface of the hidden elements beneath it which necessitated further investigation. Subsequently, ferritin levels progressed from a prognostic point of view, to serve as a severity risk factor. More importantly, the dramatic increase in ferritin levels, alongside lymphopenia, reduced NK cell number and activity, abnormal liver function tests, and coagulopathy, had researchers speculating, and later agreeing that COVID-19 might be the newest member in the group of hyperferritinemic syndromes [142]. In fact, the aspects of COVID-19 that resemble hyperferritinemic syndromes in terms of severe manifestations include systemic inflammatory response syndromes (SIRS) and acute respiratory disease syndrome (ARDS) [17,143]. The common ground beneath the hyperferritinemic syndrome and the mentioned complications encompasses the combination of high serum ferritin and a life-threatening hyperinflammation which eventually leads to multi-organ failure [142]. In this regard, the severe consequences of COVID-19 rely on two main pathological aspects, those produced by the SARS-CoV-2 itself, and those related to the host immune-inflammatory response [144]. Based on that interplay between the viral and immune system a clinical-therapeutic staging of SARS-CoV-2 infection was proposed: stage I (mild), stage II (moderate), and stage III (severe). In the first stage, SARS-CoV-2 invades the lungs and enters pulmonary cells by binding to the angiotensin-converting enzyme receptor 2 (ACE2), a receptor abundant on human lungs, intestinal epithelium, and vascular epithelium. During the first stage of the disease, symptoms are non-specific, and diagnosis is made mainly by PCR. Complete blood counts may only show lymphopenia and neutrophilia as abnormalities [145]. Therefore, ferritin at this stage does not seem to have a noticeable role. Patients progressing to the second stage of SARS-CoV-2 infection suffer from the effects of viral replication and local inflammation. The infection of the lung is manifested by pneumonia and characterized by cough, fever, and in some cases, hypoxia, whereas blood tests may reveal increased lymphopenia and elevated transaminase levels [146]. In the third and most severe stage, the formerly localized inflammation of the lungs becomes a systemic and extrapulmonary hyperinflammation syndrome [147]. In blood samples at this stage decreased T lymphocyte counts and an enormous increase in inflammatory cytokines and biomarkers including ferritin are seen [148].
Indeed, when it comes to the cellular level, the exact interplay of ferritin in the pathophysiology of COVID-19 has not been fully established yet. However, the current knowledge is that in response to injury, cytokines stimulate the production of defense proteins by the liver, including C-reactive protein and ferritin. The transcription and translation of ferritin is induced mainly by IL-1β, IL-6, and molecules of IFN-γ [11]. Macrophages as well as damaged cells constitute additional pathway explaining high ferritin levels. Importantly, ferritin promotes further pro-inflammatory mediator release, increasing the inflammatory burden and resulting in a vicious cycle. Ferritin accomplishes this by the activation of NF-ԟB leading to upregulation of ferritin gene transcription [117].
6.3. The implications of ferritin in COVID-19
The implication of ferritin in severe and advanced stages of COVID-19 as mentioned above, raised questions regarding the role of ferritin in COVID-19: is it solely a biomarker of inflammation indicating a stage or rather a trigger for further propagation of the inflammatory cascade?
Actually, if a line is drawn throughout the pandemic of COVID-19, the interpretation of elevated ferritin levels shows the following pattern:
-
1.
Acute phase reactant: increased ferritin levels were reported among the first series of patients with COVID-19 as part of the laboratory findings parallel to decreased lymphocyte count and high CRP levels [145,146].
-
2.
Indicator of severity of the disease: a strong correlation between elevated ferritin levels and the severity of COVID-19 was reported during the pandemic. Ferritin levels were significantly higher among patients with severe COVID-19 versus non-severe patients (2800 ng/ml versus 708 ng/ml, respectively). Similarly, ferritin levels were directly associated with increased risk of ARDS in patients with COVID-19 in an Italian study [149].
-
3.
Prognostic factor: patients with COVID-19 and increased ferritin levels were shown to have longer time period for viral clearance as well as longer hospital-stay [150]. The same implications were reported in another study which included the correlation between elevated ferritin levels and in-hospital mortality and invasive ventilator dependence [151].
-
4.
A member of the hyperferritinemic syndrome: COVID-19, as addressed earlier in our paper, was introduced during the pandemic as part of the hyperferritinemic syndrome due to the similarities in the presentation and severity [17,142].
-
5.
Other aspects and future studies: ferritin was investigated as a nanoparticle-based SARS-CoV-2 vaccine product tested in mice [152]. In addition, several studies exploring ferritin as a potential therapeutic target [153], possibly applicable to COVID-19. Furthermore, due to the harmful effects of iron excess and the resultant high ferritin levels, iron-depleting therapy was suggested as a treatment potential in patients with COVID-19 [154].
7. Conclusion
One of the interesting consequences of COVID-19 is that it has brought ferritin back again to the attention of researchers. While being more than an iron status indicator is a matter of fact nowadays, the exact role of ferritin, its implication in the pathogenesis of inflammatory and autoimmune diseases, and its proinflammatory properties are yet to be completely established. Better understanding of those aspects will inevitably improve diagnosis, treatment, and prevention of life-threatening complications of many diseases.
Credit author statement
Naim Mahroum: Supervision, Writing- Reviewing and Editing; Amal Alghory: Writing - Original Draft; Zeynep Kiyak: Writing - Original Draft; Abdulkarim Alwani: Writing - Original Draft; Ravend Seida: Writing - Original Draft; Mahmoud Alrais: Writing - Original Draft; Yehuda Shoenfeld: Supervision.
Declaration of competing interest
The authors declare no competing of interests.
References
- 1.Granick S., Michaelis L. Ferritin and apoferritin. Science. 1942;95(2469):439–440. doi: 10.1126/science.95.2469.439. [DOI] [PubMed] [Google Scholar]
- 2.Jacobs A., Miller F., Worwood M., Beamish M.R., Wardrop C.A. Ferritin in the serum of normal subjects and patients with iron deficiency and iron overload. Br. Med. J. 1972;4(5834):206–208. doi: 10.1136/bmj.4.5834.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Birgegard G., Hallgren R., Killander A., Stromberg A., Venge P., Wide L. Serum ferritin during infection. A longitudinal study. Scand. J. Haematol. 1978;21(4):333–340. doi: 10.1111/j.1600-0609.1978.tb00374.x. [DOI] [PubMed] [Google Scholar]
- 4.Weiss G., Ganz T., Goodnough L.T. Anemia of inflammation. Blood. 2019;133(1):40–50. doi: 10.1182/blood-2018-06-856500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Recalcati S., Invernizzi P., Arosio P., Cairo G. New functions for an iron storage protein: the role of ferritin in immunity and autoimmunity. J. Autoimmun. 2008;30(1–2):84–89. doi: 10.1016/j.jaut.2007.11.003. [DOI] [PubMed] [Google Scholar]
- 6.Sharif K., Vieira Borba V., Zandman-Goddard G., Shoenfeld Y. Eppur Si Muove: ferritin is essential in modulating inflammation. Clin. Exp. Immunol. 2018;191(2):149–150. doi: 10.1111/cei.13069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Giemza-Stoklosa J., Islam M.A., Kotyla P.J. Hyperferritinaemia: an iron sword of autoimmunity. Curr. Pharmaceut. Des. 2019;25(27):2909–2918. doi: 10.2174/1381612825666190709202804. [DOI] [PubMed] [Google Scholar]
- 8.Rosario C., Shoenfeld Y. The hyperferritinemic syndrome. Isr. Med. Assoc. J. 2014;16(10):664–665. [PubMed] [Google Scholar]
- 9.Rosario C., Zandman-Goddard G., Meyron-Holtz E.G., D'Cruz D.P., Shoenfeld Y. The hyperferritinemic syndrome: macrophage activation syndrome, Still's disease, septic shock and catastrophic antiphospholipid syndrome. BMC Med. 2013;11:185. doi: 10.1186/1741-7015-11-185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Organization W.H. 2020. WHO Director-General's Opening Remarks at the Media Briefing on COVID-19 - 11 March 2020.https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 [updated 11, March 2020. Available from: [Google Scholar]
- 11.Gomez-Pastora J., Weigand M., Kim J., Wu X., Strayer J., Palmer A.F., et al. Hyperferritinemia in critically ill COVID-19 patients - is ferritin the product of inflammation or a pathogenic mediator? Clin. Chim. Acta. 2020;509:249–251. doi: 10.1016/j.cca.2020.06.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kappert K., Jahic A., Tauber R. Assessment of serum ferritin as a biomarker in COVID-19: bystander or participant? Insights by comparison with other infectious and non-infectious diseases. Biomarkers. 2020;25(8):616–625. doi: 10.1080/1354750X.2020.1797880. [DOI] [PubMed] [Google Scholar]
- 13.Shoenfeld Y. Corona (COVID-19) time musings: our involvement in COVID-19 pathogenesis, diagnosis, treatment and vaccine planning. Autoimmun. Rev. 2020;19(6):102538. doi: 10.1016/j.autrev.2020.102538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cheng L., Li H., Li L., Liu C., Yan S., Chen H., et al. Ferritin in the coronavirus disease 2019 (COVID-19): a systematic review and meta-analysis. J. Clin. Lab. Anal. 2020;34(10) doi: 10.1002/jcla.23618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dahan S., Segal G., Katz I., Hellou T., Tietel M., Bryk G., et al. Ferritin as a marker of severity in COVID-19 patients: a fatal correlation. Isr. Med. Assoc. J. 2020;22(8):494–500. [PubMed] [Google Scholar]
- 16.Para O., Caruso L., Pestelli G., Tangianu F., Carrara D., Maddaluni L., et al. Ferritin as prognostic marker in COVID-19: the FerVid study. Postgrad. Med. 2021:1–6. doi: 10.1080/00325481.2021.1990091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ruscitti P., Berardicurti O., Di Benedetto P., Cipriani P., Iagnocco A., Shoenfeld Y., et al. Severe COVID-19, another piece in the puzzle of the hyperferritinemic syndrome. An immunomodulatory perspective to alleviate the storm. Front. Immunol. 2020;11:1130. doi: 10.3389/fimmu.2020.01130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cermak J., Neuwirt J. [A half century since the isolation of crystalline ferritin by Professor Laufberger] Vnitr. Lek. 1986;32(9):833–835. [PubMed] [Google Scholar]
- 19.Granick S. Structure and physiological functions of ferritin. Physiol. Rev. 1951;31(4):489–511. doi: 10.1152/physrev.1951.31.4.489. [DOI] [PubMed] [Google Scholar]
- 20.Granick S. Ferritin; its properties and significance for iron metabolism. Chem. Rev. 1946;38(3):379–403. doi: 10.1021/cr60121a001. [DOI] [PubMed] [Google Scholar]
- 21.Wang W., Knovich M.A., Coffman L.G., Torti F.M., Torti S.V. Serum ferritin: past, present and future. Biochim. Biophys. Acta. 2010;1800(8):760–769. doi: 10.1016/j.bbagen.2010.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jacobs A., Worwood M. Ferritin in serum. Clinical and biochemical implications. N. Engl. J. Med. 1975;292(18):951–956. doi: 10.1056/NEJM197505012921805. [DOI] [PubMed] [Google Scholar]
- 23.Knovich M.A., Storey J.A., Coffman L.G., Torti S.V., Torti F.M. Ferritin for the clinician. Blood Rev. 2009;23(3):95–104. doi: 10.1016/j.blre.2008.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Moreira A.C., Mesquita G., Gomes M.S. Ferritin: an inflammatory player keeping iron at the core of pathogen-host interactions. Microorganisms. 2020;8(4) doi: 10.3390/microorganisms8040589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Theil E.C. Ferritin: the protein nanocage and iron biomineral in health and in disease. Inorg. Chem. 2013;52(21):12223–12233. doi: 10.1021/ic400484n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Saito H. Metabolism of iron stores. Nagoya J. Med. Sci. 2014;76(3–4):235–254. [PMC free article] [PubMed] [Google Scholar]
- 27.Theil E.C. Ferritin protein nanocages-the story. Nanotechnol Percept. 2012;8(1):7–16. doi: 10.4024/n03th12a.ntp.08.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Torti F.M., Torti S.V. Regulation of ferritin genes and protein. Blood. 2002;99(10):3505–3516. doi: 10.1182/blood.v99.10.3505. [DOI] [PubMed] [Google Scholar]
- 29.Harrison P.M., Arosio P. The ferritins: molecular properties, iron storage function and cellular regulation. Biochim. Biophys. Acta. 1996;1275(3):161–203. doi: 10.1016/0005-2728(96)00022-9. [DOI] [PubMed] [Google Scholar]
- 30.Tosha T., Behera R.K., Ng H.L., Bhattasali O., Alber T., Theil E.C. Ferritin protein nanocage ion channels: gating by N-terminal extensions. J. Biol. Chem. 2012;287(16):13016–13025. doi: 10.1074/jbc.M111.332734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kuriyama-Matsumura K., Sato H., Yamaguchi M., Bannai S. Regulation of ferritin synthesis and iron regulatory protein 1 by oxygen in mouse peritoneal macrophages. Biochem. Biophys. Res. Commun. 1998;249(1):241–246. doi: 10.1006/bbrc.1998.9046. [DOI] [PubMed] [Google Scholar]
- 32.Klausner R.D., Harford J.B. cis-trans models for post-transcriptional gene regulation. Science. 1989;246(4932):870–872. doi: 10.1126/science.2683086. [DOI] [PubMed] [Google Scholar]
- 33.Cragg S.J., Wagstaff M., Worwood M. Detection of a glycosylated subunit in human serum ferritin. Biochem. J. 1981;199(3):565–571. doi: 10.1042/bj1990565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tran T.N., Eubanks S.K., Schaffer K.J., Zhou C.Y., Linder M.C. Secretion of ferritin by rat hepatoma cells and its regulation by inflammatory cytokines and iron. Blood. 1997;90(12):4979–4986. [PubMed] [Google Scholar]
- 35.Fahmy M., Young S.P. Modulation of iron metabolism in monocyte cell line U937 by inflammatory cytokines: changes in transferrin uptake, iron handling and ferritin mRNA. Biochem. J. 1993;296(Pt 1):175–181. doi: 10.1042/bj2960175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Arosio P., Levi S. Ferritin, iron homeostasis, and oxidative damage. Free Radic. Biol. Med. 2002;33(4):457–463. doi: 10.1016/s0891-5849(02)00842-0. [DOI] [PubMed] [Google Scholar]
- 37.Kotze M.J., van Velden D.P., van Rensburg S.J., Erasmus R. Pathogenic mechanisms underlying iron deficiency and iron overload: new insights for clinical application. EJIFCC. 2009;20(2):108–123. [PMC free article] [PubMed] [Google Scholar]
- 38.Simcox J.A., McClain D.A. Iron and diabetes risk. Cell Metabol. 2013;17(3):329–341. doi: 10.1016/j.cmet.2013.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.El Osta R., Grandpre N., Monnin N., Hubert J., Koscinski I. Hypogonadotropic hypogonadism in men with hereditary hemochromatosis. Basic Clin Androl. 2017;27:13. doi: 10.1186/s12610-017-0057-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chacon A.H., Morrison B., Hu S. Acquired hemochromatosis with pronounced pigment deposition of the upper eyelids. J Clin Aesthet Dermatol. 2013;6(10):44–46. [PMC free article] [PubMed] [Google Scholar]
- 41.Kowdley K.V. Iron overload in patients with chronic liver disease. Gastroenterol. Hepatol. 2016;12(11):695–698. [PMC free article] [PubMed] [Google Scholar]
- 42.Tosha T., Hasan M.R., Theil E.C. The ferritin Fe2 site at the diiron catalytic center controls the reaction with O2 in the rapid mineralization pathway. Proc. Natl. Acad. Sci. U. S. A. 2008;105(47):18182–18187. doi: 10.1073/pnas.0805083105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kernan K.F., Carcillo J.A. Hyperferritinemia and inflammation. Int. Immunol. 2017;29(9):401–409. doi: 10.1093/intimm/dxx031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Plays M., Muller S., Rodriguez R. Chemistry and biology of ferritin. Metall. 2021;13(5) doi: 10.1093/mtomcs/mfab021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Thompson K.J., Fried M.G., Ye Z., Boyer P., Connor J.R. Regulation, mechanisms and proposed function of ferritin translocation to cell nuclei. J. Cell Sci. 2002;115(Pt 10):2165–2177. doi: 10.1242/jcs.115.10.2165. [DOI] [PubMed] [Google Scholar]
- 46.Koorts A.M., Viljoen M. Ferritin and ferritin isoforms I: structure-function relationships, synthesis, degradation and secretion. Arch. Physiol. Biochem. 2007;113(1):30–54. doi: 10.1080/13813450701318583. [DOI] [PubMed] [Google Scholar]
- 47.Rogers J.T. Ferritin translation by interleukin-1and interleukin-6: the role of sequences upstream of the start codons of the heavy and light subunit genes. Blood. 1996;87(6):2525–2537. [PubMed] [Google Scholar]
- 48.Kushner I. The phenomenon of the acute phase response. Ann. N. Y. Acad. Sci. 1982;389:39–48. doi: 10.1111/j.1749-6632.1982.tb22124.x. [DOI] [PubMed] [Google Scholar]
- 49.Pannen B.H., Robotham J.L. The acute-phase response. New Horiz. 1995;3(2):183–197. [PubMed] [Google Scholar]
- 50.Moshage H. Cytokines and the hepatic acute phase response. J. Pathol. 1997;181(3):257–266. doi: 10.1002/(SICI)1096-9896(199703)181:3<257::AID-PATH756>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- 51.Koj A. Termination of acute-phase response: role of some cytokines and anti-inflammatory drugs. Gen. Pharmacol. 1998;31(1):9–18. doi: 10.1016/s0306-3623(97)00435-7. [DOI] [PubMed] [Google Scholar]
- 52.Gabay C., Kushner I. Acute-phase proteins and other systemic responses to inflammation. N. Engl. J. Med. 1999;340(6):448–454. doi: 10.1056/NEJM199902113400607. [DOI] [PubMed] [Google Scholar]
- 53.Worwood M. Serum ferritin. Clin. Sci. (Lond.) 1986;70(3):215–220. doi: 10.1042/cs0700215. [DOI] [PubMed] [Google Scholar]
- 54.Levi S., Santambrogio P., Cozzi A., Rovida E., Corsi B., Tamborini E., et al. The role of the L-chain in ferritin iron incorporation. Studies of homo and heteropolymers. J. Mol. Biol. 1994;238(5):649–654. doi: 10.1006/jmbi.1994.1325. [DOI] [PubMed] [Google Scholar]
- 55.Arosio P., Levi S., Santambrogio P., Cozzi A., Luzzago A., Cesareni G., et al. Structural and functional studies of human ferritin H and L chains. Curr. Stud. Hematol. Blood Transfus. 1991;(58):127–131. doi: 10.1159/000419350. [DOI] [PubMed] [Google Scholar]
- 56.Bomford A., Conlon-Hollingshead C., Munro H.N. Adaptive responses of rat tissue isoferritins to iron administration. Changes in subunit synthesis, isoferritin abundance, and capacity for iron storage. J. Biol. Chem. 1981;256(2):948–955. [PubMed] [Google Scholar]
- 57.Boyd D., Vecoli C., Belcher D.M., Jain S.K., Drysdale J.W. Structural and functional relationships of human ferritin H and L chains deduced from cDNA clones. J. Biol. Chem. 1985;260(21):11755–11761. [PubMed] [Google Scholar]
- 58.Orino K., Lehman L., Tsuji Y., Ayaki H., Torti S.V., Torti F.M. Ferritin and the response to oxidative stress. Biochem. J. 2001;357(Pt 1):241–247. doi: 10.1042/0264-6021:3570241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Cozzi A., Corsi B., Levi S., Santambrogio P., Albertini A., Arosio P. Overexpression of wild type and mutated human ferritin H-chain in HeLa cells: in vivo role of ferritin ferroxidase activity. J. Biol. Chem. 2000;275(33):25122–25129. doi: 10.1074/jbc.M003797200. [DOI] [PubMed] [Google Scholar]
- 60.Di Virgilio F. New pathways for reactive oxygen species generation in inflammation and potential novel pharmacological targets. Curr. Pharmaceut. Des. 2004;10(14):1647–1652. doi: 10.2174/1381612043384727. [DOI] [PubMed] [Google Scholar]
- 61.Closa D., Folch-Puy E. Oxygen free radicals and the systemic inflammatory response. IUBMB Life. 2004;56(4):185–191. doi: 10.1080/15216540410001701642. [DOI] [PubMed] [Google Scholar]
- 62.Zandman-Goddard G., Shoenfeld Y. Ferritin in autoimmune diseases. Autoimmun. Rev. 2007;6(7):457–463. doi: 10.1016/j.autrev.2007.01.016. [DOI] [PubMed] [Google Scholar]
- 63.Zandman-Goddard G., Shoenfeld Y. Hyperferritinemia in autoimmunity. Isr. Med. Assoc. J. 2008;10(1):83–84. [PubMed] [Google Scholar]
- 64.Sparks J.A. Rheumatoid arthritis. Ann. Intern. Med. 2019;170(1):ITC1–ITC16. doi: 10.7326/AITC201901010. [DOI] [PubMed] [Google Scholar]
- 65.Abe E., Arai M. Synovial fluid ferritin in traumatic hemarthrosis, rheumatoid arthritis and osteoarthritis. Tohoku J. Exp. Med. 1992;168(3):499–505. doi: 10.1620/tjem.168.499. [DOI] [PubMed] [Google Scholar]
- 66.Pelkonen P., Swanljung K., Siimes M.A. Ferritinemia as an indicator of systemic disease activity in children with systemic juvenile rheumatoid arthritis. Acta Paediatr. Scand. 1986;75(1):64–68. doi: 10.1111/j.1651-2227.1986.tb10158.x. [DOI] [PubMed] [Google Scholar]
- 67.Yildirim K., Karatay S., Melikoglu M.A., Gureser G., Ugur M., Senel K. Associations between acute phase reactant levels and disease activity score (DAS28) in patients with rheumatoid arthritis. Ann. Clin. Lab. Sci. 2004;34(4):423–426. [PubMed] [Google Scholar]
- 68.Xiao Z.X., Miller J.S., Zheng S.G. An updated advance of autoantibodies in autoimmune diseases. Autoimmun. Rev. 2021;20(2):102743. doi: 10.1016/j.autrev.2020.102743. [DOI] [PubMed] [Google Scholar]
- 69.Vanarsa K., Ye Y., Han J., Xie C., Mohan C., Wu T. Inflammation associated anemia and ferritin as disease markers in SLE. Arthritis Res. Ther. 2012;14(4):R182. doi: 10.1186/ar4012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Beyan E., Beyan C., Demirezer A., Ertugrul E., Uzuner A. The relationship between serum ferritin levels and disease activity in systemic lupus erythematosus. Scand. J. Rheumatol. 2003;32(4):225–228. doi: 10.1080/03009740310003712. [DOI] [PubMed] [Google Scholar]
- 71.Dobson R., Giovannoni G. Multiple sclerosis - a review. Eur. J. Neurol. 2019;26(1):27–40. doi: 10.1111/ene.13819. [DOI] [PubMed] [Google Scholar]
- 72.Stephenson E., Nathoo N., Mahjoub Y., Dunn J.F., Yong V.W. Iron in multiple sclerosis: roles in neurodegeneration and repair. Nat. Rev. Neurol. 2014;10(8):459–468. doi: 10.1038/nrneurol.2014.118. [DOI] [PubMed] [Google Scholar]
- 73.Hulet S.W., Powers S., Connor J.R. Distribution of transferrin and ferritin binding in normal and multiple sclerotic human brains. J. Neurol. Sci. 1999;165(1):48–55. doi: 10.1016/s0022-510x(99)00077-5. [DOI] [PubMed] [Google Scholar]
- 74.Da Costa R., Szyper-Kravitz M., Szekanecz Z., Csepany T., Danko K., Shapira Y., et al. Ferritin and prolactin levels in multiple sclerosis. Isr. Med. Assoc. J. 2011;13(2):91–95. [PubMed] [Google Scholar]
- 75.Marie I., Hatron P.Y., Levesque H., Hachulla E., Hellot M.F., Michon-Pasturel U., et al. Influence of age on characteristics of polymyositis and dermatomyositis in adults. Medicine (Baltim.) 1999;78(3):139–147. doi: 10.1097/00005792-199905000-00001. [DOI] [PubMed] [Google Scholar]
- 76.Sakata S., Nagai K., Maekawa H., Kimata Y., Komaki T., Nakamura S., et al. Serum ferritin concentration in subacute thyroiditis. Metabolism. 1991;40(7):683–688. doi: 10.1016/0026-0495(91)90084-a. [DOI] [PubMed] [Google Scholar]
- 77.Hulthen L., Lindstedt G., Lundberg P.A., Hallberg L. Effect of a mild infection on serum ferritin concentration--clinical and epidemiological implications. Eur. J. Clin. Nutr. 1998;52(5):376–379. doi: 10.1038/sj.ejcn.1600573. [DOI] [PubMed] [Google Scholar]
- 78.Kawamata R., Yokoyama K., Sato M., Goto M., Nozaki Y., Takagi T., et al. Utility of serum ferritin and lactate dehydrogenase as surrogate markers for steroid therapy for Mycoplasma pneumoniae pneumonia. J. Infect. Chemother. 2015;21(11):783–789. doi: 10.1016/j.jiac.2015.07.009. [DOI] [PubMed] [Google Scholar]
- 79.Cunha B.A. Highly elevated serum ferritin levels as a diagnostic marker for Legionella pneumonia. Clin. Infect. Dis. 2008;46(11):1789–1791. doi: 10.1086/588053. [DOI] [PubMed] [Google Scholar]
- 80.Soepandi P.Z., Burhan E., Mangunnegoro H., Nawas A., Aditama T.Y., Partakusuma L., et al. Clinical course of avian influenza A(H5N1) in patients at the Persahabatan Hospital, Jakarta, Indonesia. 2005-2008. Chest. 2010;138(3):665–673. doi: 10.1378/chest.09-2644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Cunha B.A., Mickail N., Syed U., Strollo S., Laguerre M. Rapid clinical diagnosis of Legionnaires' disease during the "herald wave" of the swine influenza (H1N1) pandemic: the Legionnaires' disease triad. Heart Lung. 2010;39(3):249–259. doi: 10.1016/j.hrtlng.2009.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lalueza A., Ayuso B., Arrieta E., Trujillo H., Folgueira D., Cueto C., et al. Elevation of serum ferritin levels for predicting a poor outcome in hospitalized patients with influenza infection. Clin. Microbiol. Infect. 2020;26(11) doi: 10.1016/j.cmi.2020.02.018. 1557 e9- e15. [DOI] [PubMed] [Google Scholar]
- 83.Eiselt J., Kielberger L., Sedlackova T., Racek J., Pazdiora P. High ferritin, but not hepcidin, is associated with a poor immune response to an influenza vaccine in hemodialysis patients. Nephron Clin. Pract. 2010;115(2):c147–c153. doi: 10.1159/000312878. [DOI] [PubMed] [Google Scholar]
- 84.McElroy A.K., Erickson B.R., Flietstra T.D., Rollin P.E., Nichol S.T., Towner J.S., et al. Ebola hemorrhagic Fever: novel biomarker correlates of clinical outcome. J. Infect. Dis. 2014;210(4):558–566. doi: 10.1093/infdis/jiu088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Betancur J.F., Navarro E.P., Echeverry A., Moncada P.A., Canas C.A., Tobon G.J. Hyperferritinemic syndrome: still's disease and catastrophic antiphospholipid syndrome triggered by fulminant Chikungunya infection: a case report of two patients. Clin. Rheumatol. 2015;34(11):1989–1992. doi: 10.1007/s10067-015-3040-9. [DOI] [PubMed] [Google Scholar]
- 86.Kim S.E., Kim U.J., Jang M.O., Kang S.J., Jang H.C., Jung S.I., et al. Diagnostic use of serum ferritin levels to differentiate infectious and noninfectious diseases in patients with fever of unknown origin. Dis. Markers. 2013;34(3):211–218. doi: 10.3233/DMA-130962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Peiris J.S., Yuen K.Y., Osterhaus A.D., Stohr K. The severe acute respiratory syndrome. N. Engl. J. Med. 2003;349(25):2431–2441. doi: 10.1056/NEJMra032498. [DOI] [PubMed] [Google Scholar]
- 88.Anderson R.M., Fraser C., Ghani A.C., Donnelly C.A., Riley S., Ferguson N.M., et al. Epidemiology, transmission dynamics and control of SARS: the 2002-2003 epidemic. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2004;359(1447):1091–1105. doi: 10.1098/rstb.2004.1490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Zaki A.M., van Boheemen S., Bestebroer T.M., Osterhaus A.D., Fouchier R.A. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N. Engl. J. Med. 2012;367(19):1814–1820. doi: 10.1056/NEJMoa1211721. [DOI] [PubMed] [Google Scholar]
- 90.Azhar E.I., El-Kafrawy S.A., Farraj S.A., Hassan A.M., Al-Saeed M.S., Hashem A.M., et al. Evidence for camel-to-human transmission of MERS coronavirus. N. Engl. J. Med. 2014;370(26):2499–2505. doi: 10.1056/NEJMoa1401505. [DOI] [PubMed] [Google Scholar]
- 91.Hsueh P.R., Chen P.J., Hsiao C.H., Yeh S.H., Cheng W.C., Wang J.L., et al. Patient data, early SARS epidemic, Taiwan. Emerg. Infect. Dis. 2004;10(3):489–493. doi: 10.3201/eid1003.030571. [DOI] [PubMed] [Google Scholar]
- 92.Organization W.H. 2021. Obesity and Overweight.https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight Available from: [Google Scholar]
- 93.Gillum R.F. Association of serum ferritin and indices of body fat distribution and obesity in Mexican American men--the Third National Health and Nutrition Examination Survey. Int. J. Obes. Relat. Metab. Disord. 2001;25(5):639–645. doi: 10.1038/sj.ijo.0801561. [DOI] [PubMed] [Google Scholar]
- 94.Khan A., Khan W.M., Ayub M., Humayun M., Haroon M. Ferritin is a marker of inflammation rather than iron deficiency in overweight and obese people. J Obes. 2016;2016:1937320. doi: 10.1155/2016/1937320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Alkhateeb A.A., Han B., Connor J.R. Ferritin stimulates breast cancer cells through an iron-independent mechanism and is localized within tumor-associated macrophages. Breast Cancer Res. Treat. 2013;137(3):733–744. doi: 10.1007/s10549-012-2405-x. [DOI] [PubMed] [Google Scholar]
- 96.Weinstein R.E., Bond B.H., Silberberg B.K., Vaughn C.B., Subbaiah P., Pieper D.R. Tissue ferritin concentration and prognosis in carcinoma of the breast. Breast Cancer Res. Treat. 1989;14(3):349–353. doi: 10.1007/BF01806307. [DOI] [PubMed] [Google Scholar]
- 97.Kukulj S., Jaganjac M., Boranic M., Krizanac S., Santic Z., Poljak-Blazi M. Altered iron metabolism, inflammation, transferrin receptors, and ferritin expression in non-small-cell lung cancer. Med. Oncol. 2010;27(2):268–277. doi: 10.1007/s12032-009-9203-2. [DOI] [PubMed] [Google Scholar]
- 98.Kalousova M., Krechler T., Jachymova M., Kubena A.A., Zak A., Zima T. Ferritin as an independent mortality predictor in patients with pancreas cancer. Results of a pilot study. Tumour Biol. 2012;33(5):1695–1700. doi: 10.1007/s13277-012-0426-z. [DOI] [PubMed] [Google Scholar]
- 99.Hann H.W., Levy H.M., Evans A.E. Serum ferritin as a guide to therapy in neuroblastoma. Cancer Res. 1980;40(5):1411–1413. [PubMed] [Google Scholar]
- 100.Stevens R.G., Beasley R.P., Blumberg B.S. Iron-binding proteins and risk of cancer in Taiwan. J. Natl. Cancer Inst. 1986;76(4):605–610. doi: 10.1093/jnci/76.4.605. [DOI] [PubMed] [Google Scholar]
- 101.Tappin J.A., George W.D., Bellingham A.J. Effect of surgery on serum ferritin concentration in patients with breast cancer. Br. J. Cancer. 1979;40(4):658–660. doi: 10.1038/bjc.1979.232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Gray C.P., Franco A.V., Arosio P., Hersey P. Immunosuppressive effects of melanoma-derived heavy-chain ferritin are dependent on stimulation of IL-10 production. Int. J. Cancer. 2001;92(6):843–850. doi: 10.1002/ijc.1269. [DOI] [PubMed] [Google Scholar]
- 103.Kikyo N., Hagiwara K., Fujisawa M., Kikyo N., Yazaki Y., Okabe T. Purification of a cell growth factor from a human lung cancer cell line: its relationship with ferritin. J. Cell. Physiol. 1994;161(1):106–110. doi: 10.1002/jcp.1041610113. [DOI] [PubMed] [Google Scholar]
- 104.Kikyo N., Suda M., Kikyo N., Hagiwara K., Yasukawa K., Fujisawa M., et al. Purification and characterization of a cell growth factor from a human leukemia cell line: immunological identity with ferritin. Cancer Res. 1994;54(1):268–271. [PubMed] [Google Scholar]
- 105.Piperno A. Classification and diagnosis of iron overload. Haematologica. 1998;83(5):447–455. [PubMed] [Google Scholar]
- 106.Zandman-Goddard G., Orbach H., Agmon-Levin N., Boaz M., Amital H., Szekanecz Z., et al. Hyperferritinemia is associated with serologic antiphospholipid syndrome in SLE patients. Clin. Rev. Allergy Immunol. 2013;44(1):23–30. doi: 10.1007/s12016-011-8264-0. [DOI] [PubMed] [Google Scholar]
- 107.Narvaez J. Adult onset still's disease. Med. Clin. 2018;150(9):348–353. doi: 10.1016/j.medcli.2017.10.035. [DOI] [PubMed] [Google Scholar]
- 108.Jia J., Shi H., Liu M., Liu T., Gu J., Wan L., et al. Cytomegalovirus infection may trigger adult-onset still's disease onset or relapses. Front. Immunol. 2019;10:898. doi: 10.3389/fimmu.2019.00898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Wouters J.M., van der Veen J., van de Putte L.B., de Rooij D.J. Adult onset Still's disease and viral infections. Ann. Rheum. Dis. 1988;47(9):764–767. doi: 10.1136/ard.47.9.764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Mahroum N., Mahagna H., Amital H. Diagnosis and classification of adult Still's disease. J. Autoimmun. 2014;48–49:34–37. doi: 10.1016/j.jaut.2014.01.011. [DOI] [PubMed] [Google Scholar]
- 111.Gerfaud-Valentin M., Jamilloux Y., Iwaz J., Seve P. Adult-onset Still's disease. Autoimmun. Rev. 2014;13(7):708–722. doi: 10.1016/j.autrev.2014.01.058. [DOI] [PubMed] [Google Scholar]
- 112.Mehta B., Efthimiou P. Ferritin in adult-onset still's disease: just a useful innocent bystander? Int. J. Inflamm. 2012;2012:298405. doi: 10.1155/2012/298405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Efthimiou P., Paik P.K., Bielory L. Diagnosis and management of adult onset Still's disease. Ann. Rheum. Dis. 2006;65(5):564–572. doi: 10.1136/ard.2005.042143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Fautrel B. Adult-onset still disease. Best Pract. Res. Clin. Rheumatol. 2008;22(5):773–792. doi: 10.1016/j.berh.2008.08.006. [DOI] [PubMed] [Google Scholar]
- 115.Fautrel B., Le Moel G., Saint-Marcoux B., Taupin P., Vignes S., Rozenberg S., et al. Diagnostic value of ferritin and glycosylated ferritin in adult onset Still's disease. J. Rheumatol. 2001;28(2):322–329. [PubMed] [Google Scholar]
- 116.Di Benedetto P., Cipriani P., Iacono D., Pantano I., Caso F., Emmi G., et al. Ferritin and C-reactive protein are predictive biomarkers of mortality and macrophage activation syndrome in adult onset Still's disease. Analysis of the multicentre Gruppo Italiano di Ricerca in Reumatologia Clinica e Sperimentale (GIRRCS) cohort. PLoS One. 2020;15(7) doi: 10.1371/journal.pone.0235326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Ruscitti P., Berardicurti O., Barile A., Cipriani P., Shoenfeld Y., Iagnocco A., et al. Severe COVID-19 and related hyperferritinaemia: more than an innocent bystander? Ann. Rheum. Dis. 2020;79(11):1515–1516. doi: 10.1136/annrheumdis-2020-217618. [DOI] [PubMed] [Google Scholar]
- 118.Ravelli A., Davi S., Minoia F., Martini A., Cron R.Q. Macrophage activation syndrome. Hematol. Oncol. Clin. N. Am. 2015;29(5):927–941. doi: 10.1016/j.hoc.2015.06.010. [DOI] [PubMed] [Google Scholar]
- 119.Crayne C.B., Albeituni S., Nichols K.E., Cron R.Q. The immunology of macrophage activation syndrome. Front. Immunol. 2019;10:119. doi: 10.3389/fimmu.2019.00119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Bojan A., Parvu A., Zsoldos I.A., Torok T., Farcas A.D. Macrophage activation syndrome: a diagnostic challenge (Review) Exp Ther Med. 2021;22(2):904. doi: 10.3892/etm.2021.10336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Sen E.S., Clarke S.L., Ramanan A.V. Macrophage activation syndrome. Indian J. Pediatr. 2016;83(3):248–253. doi: 10.1007/s12098-015-1877-1. [DOI] [PubMed] [Google Scholar]
- 122.Ramos-Casals M., Brito-Zeron P., Lopez-Guillermo A., Khamashta M.A., Bosch X. Adult haemophagocytic syndrome. Lancet. 2014;383(9927):1503–1516. doi: 10.1016/S0140-6736(13)61048-X. [DOI] [PubMed] [Google Scholar]
- 123.Deiva K., Mahlaoui N., Beaudonnet F., de Saint Basile G., Caridade G., Moshous D., et al. CNS involvement at the onset of primary hemophagocytic lymphohistiocytosis. Neurology. 2012;78(15):1150–1156. doi: 10.1212/WNL.0b013e31824f800a. [DOI] [PubMed] [Google Scholar]
- 124.Minoia F., Davi S., Horne A., Demirkaya E., Bovis F., Li C., et al. Clinical features, treatment, and outcome of macrophage activation syndrome complicating systemic juvenile idiopathic arthritis: a multinational, multicenter study of 362 patients. Arthritis Rheum. 2014;66(11):3160–3169. doi: 10.1002/art.38802. [DOI] [PubMed] [Google Scholar]
- 125.Jordan M.B., Allen C.E., Greenberg J., Henry M., Hermiston M.L., Kumar A., et al. Challenges in the diagnosis of hemophagocytic lymphohistiocytosis: recommendations from the north American consortium for histiocytosis (NACHO) Pediatr. Blood Cancer. 2019;66(11) doi: 10.1002/pbc.27929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Ravelli A., Minoia F., Davi S., Horne A., Bovis F., Pistorio A., et al. Classification criteria for macrophage activation syndrome complicating systemic juvenile idiopathic arthritis: a European league against rheumatism/American college of rheumatology/paediatric rheumatology international trials organisation collaborative initiative. Ann Rheum Dis. 2016. 2016;75(3):481–489. doi: 10.1136/annrheumdis-2015-208982. [DOI] [PubMed] [Google Scholar]
- 127.Henter J.I., Horne A., Arico M., Egeler R.M., Filipovich A.H., Imashuku S., et al. HLH-2004: diagnostic and therapeutic guidelines for hemophagocytic lymphohistiocytosis. Pediatr. Blood Cancer. 2007;48(2):124–131. doi: 10.1002/pbc.21039. [DOI] [PubMed] [Google Scholar]
- 128.Sammaritano L.R. Antiphospholipid syndrome. Best Pract. Res. Clin. Rheumatol. 2020;34(1):101463. doi: 10.1016/j.berh.2019.101463. [DOI] [PubMed] [Google Scholar]
- 129.Cervera R., Boffa M.C., Khamashta M.A., Hughes G.R. The Euro-Phospholipid project: epidemiology of the antiphospholipid syndrome in Europe. Lupus. 2009;18(10):889–893. doi: 10.1177/0961203309106832. [DOI] [PubMed] [Google Scholar]
- 130.Groot N., de Graeff N., Avcin T., Bader-Meunier B., Dolezalova P., Feldman B., et al. European evidence-based recommendations for diagnosis and treatment of paediatric antiphospholipid syndrome: the SHARE initiative. Ann. Rheum. Dis. 2017;76(10):1637–1641. doi: 10.1136/annrheumdis-2016-211001. [DOI] [PubMed] [Google Scholar]
- 131.Nayer A., Ortega L.M. Catastrophic antiphospholipid syndrome: a clinical review. J Nephropathol. 2014;3(1):9–17. doi: 10.12860/jnp.2014.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Bucciarelli S., Cervera R., Espinosa G., Gomez-Puerta J.A., Ramos-Casals M., Font J. Mortality in the catastrophic antiphospholipid syndrome: causes of death and prognostic factors. Autoimmun. Rev. 2006;6(2):72–75. doi: 10.1016/j.autrev.2006.06.007. [DOI] [PubMed] [Google Scholar]
- 133.Agmon-Levin N., Rosario C., Katz B.S., Zandman-Goddard G., Meroni P., Cervera R., et al. Ferritin in the antiphospholipid syndrome and its catastrophic variant (cAPS) Lupus. 2013;22(13):1327–1335. doi: 10.1177/0961203313504633. [DOI] [PubMed] [Google Scholar]
- 134.Angus D.C., van der Poll T. Severe sepsis and septic shock. N. Engl. J. Med. 2013;369(9):840–851. doi: 10.1056/NEJMra1208623. [DOI] [PubMed] [Google Scholar]
- 135.Cecconi M., Evans L., Levy M., Rhodes A. Sepsis and septic shock. Lancet. 2018;392(10141):75–87. doi: 10.1016/S0140-6736(18)30696-2. [DOI] [PubMed] [Google Scholar]
- 136.Garcia P.C., Longhi F., Branco R.G., Piva J.P., Lacks D., Tasker R.C. Ferritin levels in children with severe sepsis and septic shock. Acta Paediatr. 2007;96(12):1829–1831. doi: 10.1111/j.1651-2227.2007.00564.x. [DOI] [PubMed] [Google Scholar]
- 137.McCullough K., Bolisetty S. Iron homeostasis and ferritin in sepsis-associated kidney injury. Nephron. 2020;144(12):616–620. doi: 10.1159/000508857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Zhu H., Wei L., Niu P. The novel coronavirus outbreak in Wuhan, China. Glob Health Res Policy. 2020;5:6. doi: 10.1186/s41256-020-00135-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Organization W.H. 2021. Weekly Operational Update on COVID-19 - 3 November 2021.https://www.who.int/publications/m/item/weekly-operational-update-on-covid-19---3-november-2021 [Available from: [Google Scholar]
- 140.Wang D., Hu B., Hu C., Zhu F., Liu X., Zhang J., et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in wuhan, China. J. Am. Med. Assoc. 2020;323(11):1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Huang I., Pranata R., Lim M.A., Oehadian A., Alisjahbana B. C-reactive protein, procalcitonin, D-dimer, and ferritin in severe coronavirus disease-2019: a meta-analysis. Ther. Adv. Respir. Dis. 2020;14 doi: 10.1177/1753466620937175. 1753466620937175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Colafrancesco S., Alessandri C., Conti F., Priori R. COVID-19 gone bad: a new character in the spectrum of the hyperferritinemic syndrome? Autoimmun. Rev. 2020;19(7):102573. doi: 10.1016/j.autrev.2020.102573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Alunno A., Carubbi F., Rodriguez-Carrio J. Storm, typhoon, cyclone or hurricane in patients with COVID-19? Beware of the same storm that has a different origin. RMD Open. 2020;6(1) doi: 10.1136/rmdopen-2020-001295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Siddiqi H.K., Mehra M.R. COVID-19 illness in native and immunosuppressed states: a clinical-therapeutic staging proposal. J. Heart Lung Transplant. 2020;39(5):405–407. doi: 10.1016/j.healun.2020.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Guan W.J., Ni Z.Y., Hu Y., Liang W.H., Ou C.Q., He J.X., et al. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020;382(18):1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Goyal P., Choi J.J., Pinheiro L.C., Schenck E.J., Chen R., Jabri A., et al. Clinical characteristics of covid-19 in New York city. N. Engl. J. Med. 2020;382(24):2372–2374. doi: 10.1056/NEJMc2010419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Qin C., Zhou L., Hu Z., Zhang S., Yang S., Tao Y., et al. Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in wuhan, China. Clin. Infect. Dis. 2020;71(15):762–768. doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J., et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395(10229):1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Gandini O., Criniti A., Ballesio L., Giglio S., Galardo G., Gianni W., et al. Serum ferritin is an independent risk factor for acute respiratory distress syndrome in COVID-19. J. Infect. 2020;81(6):979–997. doi: 10.1016/j.jinf.2020.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Cao P., Wu Y., Wu S., Wu T., Zhang Q., Zhang R., et al. Elevated serum ferritin level effectively discriminates severity illness and liver injury of coronavirus disease 2019 pneumonia. Biomarkers. 2021;26(3):207–212. doi: 10.1080/1354750X.2020.1861098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Qeadan F., Tingey B., Gu L.Y., Packard A.H., Erdei E., Saeed A.I. Prognostic values of serum ferritin and D-dimer trajectory in patients with COVID-19. Viruses. 2021;13(3) doi: 10.3390/v13030419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Wang W., Huang B., Zhu Y., Tan W., Zhu M. Ferritin nanoparticle-based SARS-CoV-2 RBD vaccine induces a persistent antibody response and long-term memory in mice. Cell. Mol. Immunol. 2021;18(3):749–751. doi: 10.1038/s41423-021-00643-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Zhang B., Tang G., He J., Yan X., Fan K. Ferritin nanocage: a promising and designable multi-module platform for constructing dynamic nanoassembly-based drug nanocarrier. Adv. Drug Deliv. Rev. 2021;176:113892. doi: 10.1016/j.addr.2021.113892. [DOI] [PubMed] [Google Scholar]
- 154.Perricone C., Bartoloni E., Bursi R., Cafaro G., Guidelli G.M., Shoenfeld Y., et al. COVID-19 as part of the hyperferritinemic syndromes: the role of iron depletion therapy. Immunol. Res. 2020;68(4):213–224. doi: 10.1007/s12026-020-09145-5. [DOI] [PMC free article] [PubMed] [Google Scholar]