Abstract
Purpose
We conducted a systematic review and a retrospective study to investigate the relationship between spleen irradiation and lymphocyte toxicity.
Methods and Materials
Forty-six patients diagnosed with locally advanced gastric, esophageal, and pancreatic cancer who underwent radiation therapy were included in this study. The spleen was contoured for each patient. Volumes that received 5 up to 40 Gy (5 Gy increments), minimum, mean, and maximum dose were considered along with lymphocyte count to determine toxicity. Comprehensive and systematic literature searches were performed using PubMed, SCOPUS, Cochrane Central Databases, and Google Scholar.
Results
Literature review on spleen unintended irradiation and lymphocyte toxicity resulted in 408 patients from 5 studies. In our study, univariate and multivariate linear regressions found an association between V15 (chemotherapy as controlling factor) and nadir lymphocyte count (P = .04) and between DMAX and nadir lymphocyte count (P = .046). An increase of 1 Gy in mean splenic dose was associated with a 1% decrease in absolute lymphocyte count at nadir.
Conclusions
Although there is no consensus regarding lymphopenia spleen dose volume threshold, all studies found that higher splenic dose increases the risk of lymphopenia. Our study's results suggest that spleen unintentional V15 and maximum dose irradiation were associated with lymphopenia during chemoradiation therapy.
Introduction
For upper abdominal tumors (eg, gastric, pancreatic, lower esophagus), alongside surgery and chemotherapy, external beam radiation therapy plays an essential role in the multidisciplinary treatment approach of the locally advanced disease.1, 2, 3
Radiation affects tumoral cells as well as healthy surrounding tissues, and the extent of damage depends on the irradiation technique, treatment planning, and area of irradiation. When the target volume includes upper abdominal or lower thoracic structures, then liver, lung, intestinal, heart, gastric, and bone marrow toxicities might be expected. Although concurrent chemotherapy increases radiation response,4 hematologic toxicity might occur more frequently, associating leukopenia, thrombocytopenia, and neutropenia.5, 6, 7 These side effects might delay the treatment, which is associated with a decreased overall survival.8
Unlike for lower abdomen tumors, where bone marrow irradiation is linked to a negative prognosis,9, 10, 11, 12 for upper abdominal/lower chest irradiation, although not routinely contoured, the spleen suffers damage, largely to an unknown extent. The spleen plays an important part in the immune system as the site for T cell activation and B lymphocyte reactions. This further leads to antibody production and adaptive immune response initiation, combining in a particular manner the innate and adaptive immune systems.13 In another manner, tumor infiltrating lymphocytes might be an indicator of tumor prognosis,14 and more information on how lymphocytes affect treatment response is needed.
The spleen is not considered as an organ at risk (OAR) in radiation therapy and is not routinely delineated, and this is probably because there are a lack of data regarding the hematological effect regarding spleen irradiation.15
Our study's main objective was to identify if spleen radiation exposure dose can be associated with nadir lymphocyte count during radiation therapy in patients. Apart from previous studies, we selected a more diverse array of tumors to obtain more variability in spleen toxicity.
Methods and Materials
All procedures were performed in accordance with the ethical standards in the 1964 Declaration of Helsinki and its later amendments. Informed consent was obtained from all patients included in the study.
Literature review
We conducted a systematic search of PubMed, SCOPUS, Cochrane Central Databases, and Google Scholar databases to identify studies that focused on spleen unintended irradiation. Published articles were identified by using database-specific search engines on “spleen” AND “irradiation.” Titles and abstracts screened for data “unintended OR lymphopenia AND unintentional OR lymphopenia AND radiation therapy OR lymphopenia AND/OR chemoradiation” were collated.
Patient population and treatment planning
Our study population included patients diagnosed with locally advanced gastric, lower third esophageal, and pancreatic cancer that underwent radiation therapy between January 2015 and July 2020 as part of their treatment. Patient population characteristics are presented in Table 1. Considering that our study followed spleen radiation dose and lymphocyte count relationship, we excluded patients undergoing fewer than 10 radiation therapy fractions and patients with initial hematologic parameters outside normal range. Also, patients undergoing altered fractionation schedules of radiation therapy were excluded. Forty-six out of 69 consecutive patients were eligible for this study following patient population stratification based on exclusion criteria.
Table 1.
Patient characteristics
| Patient characteristics | Features | n (%) or median (range) | |
|---|---|---|---|
| Total number of patients | 46 | ||
| Sex | Male | 39 | |
| Female | 7 | ||
| Age | Years | 67 (44-83) | |
| Diagnosis | Esophagus | 17 (37) | |
| Esogastric junction | 12 (26) | ||
| Gastric | 13 (28) | ||
| Pancreatic | 4 (9) | ||
| Stage | I-III | 45 (98) | |
| IV | 1 (2) | ||
| Treatment characteristics | |||
| Chemotherapy | Sequential | Concomitant | |
| Total | 30 (65) | 16 (34) | |
| Platinum salts (CBDA/CDDP/OX) | 24 | 9 | |
| 5FU/Cape | 15 | 8 | |
| Taxanes (PTX) | 7 | 5 | |
| Other | 4 | 0 | |
| Radiation therapy | Total dose | 45 (25-56.6) Gy | |
| Dose/fraction | 1.8 Gy | ||
| Number of fractions | 25 (16-25) |
Abbreviations: 5FU = 5 fluorouracil; Cape = capecitabine; CBDA = carboplatin; CDDP = cisplatin; OX = oxaliplatin; PTX = paclitaxel.
Radiation treatment was delivered using the 3-dimensional conformal radiotherapy technique up to a total dose of 45 to 50.4 Gy, using a conventional fractionation regimen. Concurrent chemotherapy was used as summarized in Table 1. For OAR the dose restrictions used were according to Quantitative Analyses of Normal Tissue Effects in the Clinic recommendations.16
In our institution the spleen is not contoured as an OAR, therefore we delineated it after radiation therapy ending on the patients’ treatment plans. The spleen was delineated on the radiation therapy planning computed tomography scan using 3-mm slides. Subsequently, the following dosimetric parameters were extracted from the resulting spleen dose-volume histogram (DVH): minimum dose (DMIN), maximum dose (DMAX), mean dose (DMEAN), and V5 up to V40 in 5 Gy increments for spleen. For DVH extraction we used Eclipse version 9 and for data processing of each DVH we used RStudio with libraries "tidyverse," "ggplot2," and "DVH metrics."17
Laboratory data
As per institutional treatment protocol, blood tests, including complete blood count (CBC), were done before and weekly during treatment. From patients’ lab test results we extracted white blood cell count, absolute neutrophil count, hemoglobin, platelet count, and lymphocyte count (LYM). Pretreatment initial CBC results were used as the baseline. The LYM nadir values were then registered along with the week of occurrence and graded according to Common Terminology Criteria for Adverse Events v5.0. For CBC extraction we used Python 3.18
Statistical analysis
Statistical analysis LYM parameter was compared with baseline while the week of occurrence was registered. Univariate and multivariate linear regressions were used to verify if any link could be found between nadir value (toxicity) and increasing dosimetric values, depending on the use of chemotherapy. Toxicity was graded using Common Terminology Criteria for Adverse Events 5.0 system. For data analysis we used RStudio,19 and 1-way analysis of variance test was used by grouping nadir lymphocyte toxicity with or without chemotherapy with DMAX, DMEAN values.
Results
Literature review on spleen unintended irradiation and lymphocyte toxicity resulted in 408 patients from 5 studies, as summarized in Table 2.
Table 2.
Dosimetric parameters and level 2 lymphocyte toxicity
| Spleen dosimetry | DMAX | 43.74 Gy (0.5-53) | |
|---|---|---|---|
| DMEAN | 14.4 (22.5-37.14) | ||
| DMIN | 0.9 (0-19.6) | ||
| V10 Gy | 72 (0-100) | ||
| V15 Gy | 44 (0-100) | ||
| V20 Gy | 34 (0-99.9) | ||
| V25 Gy | 23.72 (0-98.4) | ||
| V30 Gy | 10 (0-92.7) | ||
| V35 Gy | 4.35 (0-51.6) | ||
| V40 Gy | 1 (0-37.1) | ||
| Lymphocyte toxicity | |||
| <2 (0.8-0.5 × 10e9/L) | 14 (30) | ||
| >=2 (<0.5 × 10e9/L) | 32 (70) |
Abbreviations: DMAX = maximum dose; DMEAN = mean dose; DMIN = minimum dose; VxGy = volume that received × Gy.
In our study, after patient population stratification according to exclusion criteria, 46 patients remained. The median age at time of radiation therapy was 66 years. Regarding tumor location, 17 patients were diagnosed with lower esophageal cancer, 12 with esogastric junction cancer, and 13 with gastric cancer. Forty-five (97%) patients enrolled in this study were patients with locally advanced stage I-III cancer. Regarding chemotherapy, 66% of the patients underwent sequential chemoradiation therapy, while 34% underwent concurrent chemoradiation therapy. Radiation therapy was administered with a conventional fractionation regimen, using 1.8 Gy/fraction, up to a median total dose of 45 Gy (25-56.5 Gy).
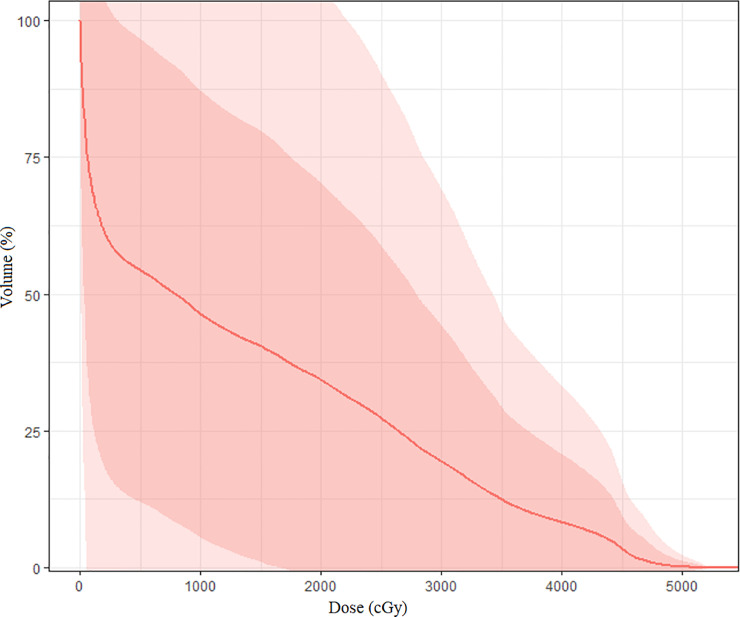
All dosimetric parameter testing results are presented in Table 2. These data highlight V40, which was increased in 26 patients, with means of 72%, 34%, and 10%. Figure 1 encompasses a cumulative distribution of DMEAN with 1 and 2 standard deviations for all patients. Grade 2 or higher lymphocyte toxicity was found in 32 patients (69%). Mean nadir lymphocyte count (NLC) was 0.4 × 109/L (range, 0.06-0.93 × 109/L).
Figure 1.
Spleen mean dose cumulative dose distribution with first 2 standard deviations
Using linear modeling with NLC value as a response to DMAX, DMEAN, DMIN, or V15 Gy and chemotherapy administration, a weak but significant statistical association was found between DMAX and NLC (P = .046 for DMAX, r-squared = 0.11). In the linear model chemotherapy administration, no statistical significance was observed (P = .3). In a similar manner, a weak association was found between NLC and V15 Gy (P = .04; r2 = 0.07), whereas the addition of chemotherapy administration did not produce a statistically significant association (P = .087). An increase in mean dose by 1 Gy resulted in a 1% decrease of NLC, although the result was not statistically significant (P = .052).
We also tested the existence of a relationship between the moment of chemotherapy use, sequential or concurrent, DMEAN, and NLC, but no such connection was found using multivariable regression (sequential chemotherapy P = .4, concomitant chemotherapy P = .38, mean dose P = .082).
Using logistic regression, V15, V20, DMEAN, and DMEAN (controlling for chemotherapy) were not found to be associated with lymphocyte toxicity (odds ratio, 1.021; P = .54). Splitting DMEAN and DMAX dosimetric parameters to values less than or greater than their corresponding means (mean DMAX = 43.4 Gy, mean DMEAN = 14.4 Gy) and all these as groups along with chemotherapy status, no significant differences were found on 1-way analysis of variance between the groups regarding NLC (P = .08 for DMAX group, P = .07 for DMEAN group).
Discussion
Our literature review found that, before 2015 most studies20, 21, 22 evaluated intentional and therapeutic irradiation of the spleen. Later, although modest regarding patient population, spleen unintended irradiation became a new and emerging field of research, as summarized in Table 3.
Table 3.
Current literature data available for spleen as an organ at risk
| Author | Pts | Tumor | Tx intent | RTdose | CHT | MSD (Gy) | CBC | Predictors |
|---|---|---|---|---|---|---|---|---|
| Trip et al. (2015)15 | 90 | Gastric | A | 45 Gy (25 fx) |
Cape CDDP |
40 | - | V44 for SV decrease |
| Chadha et al. (2016)23 | 177 | Pancr. | C | 50.4 Gy (25-28 fx) |
5FU Gem Cape |
6.8 | ↓ALC | V15>20% (lymphopenia SDV threshold) |
| Liu et al. (2017)24 | 59 | HCC | C | 50-60 Gy (25-30 fx) |
- | 2.27 | ↓ALC | V5 and MSD for ALC |
| Chin et al. (2018)26 | 21 | Esoph. | Na/C | 50 Gy (25 fx-SIB) |
CBDA- PTX 5FU/Cape |
23.4 | HT > gr.3 | V5-V20 and MSD for HT > gr.3 |
| Saito et al. (2018)25 | 61 | Esoph. | C | 60 Gy (30-33 fx) |
5FU CDDP TXT |
2.87 | ↓ALC | V5-V30 and MSD for ALC (–2.9% ALC for each Gy of MSD) |
Abbreviations: 5FU = 5 flurouracil; ALC = absolute lymphocyte count; Cape = capecitabine; CBC = complete blood count; CBDA = carboplatin; CDDP = cisplatin; CHT = chemotherapy; Esoph. = esophagus; fx = fractions; Gem = gemcitabine; HCC = hepatocellular carcinoma; HT = hematological toxicity; MSD = mean spleen dose; Pancr. = pancreatic; PTX = paclitaxel; RT = radiation therapy; SDV = splenic dose volume; SIB = simultaneous integrated boost; SV = spleen volume; Tx = treatment; TXT = docetaxel.
In 2015, the first study considering the spleen as an OAR was conducted by Trip et al15 evaluating retrospectively the effect of radiation-induced dose-dependent changes of the spleen after chemoradiation therapy. The study population included 46 patients with gastric cancer who underwent postoperative chemoradiation therapy. The mean total external beam radiation therapy dose was 45 Gy using a conventional fractionation regimen while concurrent capecitabine and cisplatin were administered. The mean radiation dose to the spleen was 40 Gy. This study was also the first to demonstrate a radiation–dose effect relationship to the spleen and the blood-cell count. This is supported by the fact that 4 years following splenic irradiation the spleen volume was reduced to one-third, while the average leukocyte count was significantly higher than baseline. In this study, spleen volume reduction was a result of splenic hilum irradiation, which further leads to vascular side effects. The authors concluded that the threshold for spleen radiation exposure was 12 Gy.
It is known that radiation therapy has both immune-stimulating and immune-suppressive effects. The radiation immune-suppressive effect has been linked to bone marrow suppression and circulating lymphocyte depletion. Based on this hypothesis, the first study evaluating a link between spleen radiation exposure and lymphocyte count was from Chadha et al.23 This is also one of the largest patient population trials, encompassing 177 patients with pancreatic adenocarcinoma, which evaluated the hematological effect of spleen radiation exposure. All patients enrolled in this study underwent induction chemotherapy, followed by concurrent chemoradiation therapy. According to the results, postchemoradiation therapy lymphopenia was independent of the chemotherapy regimen used and could be predicted by baseline and after induction chemotherapy lymphocyte values. The authors found that the risk of severe lymphopenia increases proportional to the medium spleen dose and to the fractional volume of spleen receiving low radiation doses (V10, V20). The medium spleen dose and splenic dose-volume thresholds for lymphopenia to develop were 9 Gy and V15 of 20%, respectively.
In Liu et al's24 study, a correlation between spleen irradiation and peripheral blood lymphocytes was investigated for 59 patients with hepatocellular carcinoma. Chemotherapy was used only for transarterial chemoembolization. Owing to its curative intent, radiation therapy was administered up to 60 Gy. The study identified a significant correlation between spleen irradiation dose and peripheral lymphocyte decrease during radiation therapy. The only independent parameter associated with a decrease in absolute lymphocyte count was V5; therefore, the authors concluded that in order to avoid decreasing the peripheral lymphocyte count the spleen low-dose area should be radiation spared.
In 2018, 2 studies25,26 evaluated the effect of spleen irradiation for patients with esophageal cancer undergoing radiation therapy. Saito et al. performed a retrospective study on 61 patients with esophageal cancer undergoing definitive chemoradiation therapy. Using multivariable linear regression, V5, V10, V20, V30, and medium spleen dose were analyzed separately and were linked to lymphocyte count. Although an important drawback of this study is that lab tests were not always available during chemoradiation therapy, this is the first study to report the relationship between the mean splenic dose and lymphocyte count. Although from the multivariate analysis an increase of 1 Gy in mean splenic dose predicted a 2.9% decrease in absolute lymphocyte count at nadir, the univariable logistic regression analysis found the mean splenic dose to be a significant predictor of grade 4 lymphopenia. In another study, Chin et al26 included 21 esophagus and gastroesophageal junction patients and evaluated dosimetric and volumetric parameters for spleen, thoracic spine, and posterior ribs. Although the study also included metastatic patients, no data regarding the use of chemotherapy prior to radiation therapy are available. This is the first study to report the radiation technique used and the use of simultaneous integrated boost. The authors found that hematological toxicity decreases with the increase in the mean spleen radiation dose and that initial splenic volume, before radiation therapy, was a predictor of an increase of the hematological toxicity.
Compared with the previous data, our study is the only to include a patient population with different tumor locations: esophageal, esogastric junction, pancreatic, and gastric. All our patients underwent 3-dimensional conformal radiation therapy, therefore, unlike the study of Liu et al,24 no patients with hepatocellular carcinoma were included in our study. Similar to the Trip et al study,15 we also included patients with metastatic cancer. Mean spleen dose in our study was 14.4 Gy compared with 6.8 Gy in the Chadha et al study23 and 23.1 Gy in the Chin et al study.26
Our study evaluated general hematologic parameters, all grades, but lymphocyte toxicity was the main objective of our study. Compared with the study of Chin et al,26 where grade 3 leukopenia was reported in 85% of the patients, in our study only 24% of the patients experienced this side effect. This is probably owing to the use of curative radiation doses up to 59 Gy and simultaneous integrated boost in the study of Chin et al,26 whereas in our study, most patients underwent pre/postoperative radiation therapy with total doses up to 45 Gy using standard fractionation schedules.
In Trip et al's study, probably owing to the 4-year follow-up period, the authors reported a significant effect of chemotherapy use on CBC, especially for the thrombocyte count. Alongside the studies of Chin et al26 and Chadha et al,23 in our study, chemotherapy was not a predictive factor for hematological toxicity.
As in the studies of Chin et al,26 Chadha et al,23 and Dailey et al,22 the splenic volume is an important parameter to be monitored because it can be used as an independent predictor of lymphocyte count. However, due to a lack of posttherapeutic follow-up data, splenic volume was not evaluated in our study. Also, another limitation of our study was that due to the increased number of deaths among patients enrolled in this study, no association between treatment-related lymphopenia and survival could be evaluated.
Conclusions
Spleen is not routinely considered an OAR; therefore, there are a lack of data regarding radiation and the dose-effect relationship to the spleen. A decrease in lymphocyte count has been associated with an increase in incidence of infectious events and, in the current international context of the severe acute respiratory syndrome coronavirus-2 pandemic, more attention should be given to the spleen, especially for oncological patients undergoing radiation therapy.
According to previously published data, independent of its value, Mean spleen dose is associated with a decrease in lymphocyte count. After this literature review we can observe that there is no consensus regarding dosimetric parameters proposed as lymphopenia spleen dose-volume thresholds. However, what is unanimously accepted is that an increase in spleen exposure to higher doses leads to a decrease in volume.
Our study is the first to address spleen as an OAR in a heterogeneous cancer patient population. From its findings, alongside previously published data, we can conclude that an increase of 1 Gy of the mean spleen dose might generate a decrease in NLC of 1%, although more patients would be needed to obtain statistical significance.
All these data support the recommendation of delineating the spleen as an OAR. However, more data from studies assessing lymphocyte toxicity in patients who have undergone spleen sparing during radiation therapy are needed.
Footnotes
Sources of support: This work had no specific funding.
Disclosures: none.
Research data are not available at this time.
Note–An online CME test for this article can be taken at https://academy.astro.org.
References
- 1.Lambert A, Schwarz L, Borbath I, et al. An update on treatment options for pancreatic adenocarcinoma. Ther Adv Med Oncol. 2019;11 doi: 10.1177/1758835919875568. 1758835919875568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kato H, Nakajima M. Treatments for esophageal cancer: A review. Gen Thorac Cardiovasc Surg. 2013;61:330–335. doi: 10.1007/s11748-013-0246-0. [DOI] [PubMed] [Google Scholar]
- 3.National Comprehensive Cancer Network. Esophageal and esophagogastric junction cancers (version 4.2020). Available at:https://www.nccn.org/professionals/physician_gls/pdf/esophageal.pdf. Accessed December 30, 2020.
- 4.Georgescu MT, Patrascu T, Serbanescu LG, et al. When should we expect curative results of neoadjuvant treatment in locally advanced rectal cancer patients? Chirurgia (Bucur) 2021;116:16–23. doi: 10.21614/chirurgia.116.1.16. [DOI] [PubMed] [Google Scholar]
- 5.Kroep JR, Peters GJ, van Moorsel CJ, et al. Gemcitabine-cisplatin: A schedule finding study. Ann Oncol. 1999;10:1503–1510. doi: 10.1023/a:1008339425708. [DOI] [PubMed] [Google Scholar]
- 6.Saito H, Ohta A, Abe E, et al. Definitive chemoradiation therapy with low-dose continuous 5-fluorouracil reduces hematological toxicity without compromising survival in esophageal squamous cell carcinoma patients. Clin Transl Radiat Oncol. 2018;9:12–17. doi: 10.1016/j.ctro.2017.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schwab M, Zanger UM, Marx C, et al. Role of genetic and nongenetic factors for fluorouracil treatment-related severe toxicity: A prospective clinical trial by the German 5-FU Toxicity Study Group. J Clin Oncol. 2008;26:2131–2138. doi: 10.1200/JCO.2006.10.4182. [DOI] [PubMed] [Google Scholar]
- 8.González Ferreira JA, Olasolo JJ, Azinovic I, Jeremic B. Effect of radiation therapy delay in overall treatment time on local control and survival in head and neck cancer: Review of the literature. Rep Pract Oncol Radiother. 2015;20:328–339. doi: 10.1016/j.rpor.2015.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Georgescu MT, Anghel R. Variation in uterus position prior to brachytherapy of the cervix: A case report. J Med Life. 2017;10:90–93. [PMC free article] [PubMed] [Google Scholar]
- 10.Georgescu MT, VGM Ileanu BV, Anghel R. Dosimetric influence of uterus position in cervix cancer high-dose-rate brachytherapy. Rom Journ Phys. 2015;61:1557–1566. [Google Scholar]
- 11.Sini C, Fiorino C, Perna L, et al. Dose-volume effects for pelvic bone marrow in predicting hematological toxicity in prostate cancer radiation therapy with pelvic node irradiation. Radiother Oncol. 2016;118:79–84. doi: 10.1016/j.radonc.2015.11.020. [DOI] [PubMed] [Google Scholar]
- 12.Kumar T, Schernberg A, Busato F, et al. Correlation between pelvic bone marrow radiation dose and acute hematological toxicity in cervical cancer patients treated with concurrent chemoradiation. Cancer Manag Res. 2019;11:6285–6297. doi: 10.2147/CMAR.S195989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lewis SM, Williams A, Eisenbarth SC. Structure and function of the immune system in the spleen. Sci Immunol. 2019;4 doi: 10.1126/sciimmunol.aau6085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zgura A, Gales L, Haineala B, et al. Correlations between known prognostic markers and tumor - infiltrating lymphocytes in breast cancer. Rev Chim. 2019;70:2363–2366. [Google Scholar]
- 15.Trip AK, Sikorska K, van Sandick JW, et al. Radiation-induced dose-dependent changes of the spleen following postoperative chemoradiation therapy for gastric cancer. Radiother Oncol. 2015;116:239–244. doi: 10.1016/j.radonc.2015.07.036. [DOI] [PubMed] [Google Scholar]
- 16.Marks LB, Ten Haken RK, Martel MK. Guest editor's introduction to QUANTEC: A users guide. Int J Radiat Oncol Biol Phys. 2010;76(3 Suppl):S1–S2. doi: 10.1016/j.ijrobp.2009.08.075. [DOI] [PubMed] [Google Scholar]
- 17.R Foundation for Statistical Computing; Vienna, Austria: 2020. R: A Language and Environment for Statistical Computing. [Google Scholar]
- 18.Rossum Gv. Python Tutorial, Techical Report CS-R9526. Centrum voor Wiskunde en Informatica (CWI); Amsterdam, Netherlands: 1995. [Google Scholar]
- 19.Mebius RE, Kraal G. Structure and function of the spleen. Nat Rev Immunol. 2005;5:606–616. doi: 10.1038/nri1669. [DOI] [PubMed] [Google Scholar]
- 20.Knecht H, Jost R, Gmur J, Burger J, Fehr J. Functional hyposplenia after allogeneic bone marrow transplantation is detected by epinephrine stimulation test and splenic ultrasonography. Eur J Haematol. 1988;41:382–387. doi: 10.1111/j.1600-0609.1988.tb00213.x. [DOI] [PubMed] [Google Scholar]
- 21.Weinmann M, Becker G, Einsele H, Bamberg M. Clinical indications and biological mechanisms of splenic irradiation in chronic leukaemias and myeloproliferative disorders. Radiother Oncol. 2001;58:235–246. doi: 10.1016/s0167-8140(00)00316-9. [DOI] [PubMed] [Google Scholar]
- 22.Dailey MO, Coleman CN, Kaplan HS. Radiation-induced splenic atrophy in patients with Hodgkin's disease and non-Hodgkin's lymphomas. N Engl J Med. 1980;302:215–217. doi: 10.1056/NEJM198001243020406. [DOI] [PubMed] [Google Scholar]
- 23.Chadha AS, Liu G, Chen H-C, et al. Does unintentional splenic radiation predict outcomes after pancreatic cancer radiation therapy? Int J Radiat Oncol Biol Phys. 2017;97:323–332. doi: 10.1016/j.ijrobp.2016.10.046. [DOI] [PubMed] [Google Scholar]
- 24.Liu J, Zhao Q, Deng W, et al. Radiation-related lymphopenia is associated with spleen irradiation dose during radiation therapy in patients with hepatocellular carcinoma. Radiat Oncol. 2017;12:90. doi: 10.1186/s13014-017-0824-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Saito T, Toya R, Yoshida N, et al. Spleen dose-volume parameters as a predictor of treatment-related lymphopenia during definitive chemoradiation therapy for esophageal cancer. In Vivo. 2018;32:1519–1525. doi: 10.21873/invivo.11409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chin AL, Aggarwal S, Pradhan P, et al. The role of bone marrow and spleen irradiation in the development of acute hematologic toxicity during chemoradiation for esophageal cancer. Adv Radiat Oncol. 2018;3:297–304. doi: 10.1016/j.adro.2018.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]