Abstract
Purpose
Patients with pancreatic cancer often receive radiation therapy before undergoing surgical resection. We compared the clinical outcomes differences between stereotactic body radiation therapy (SBRT) and 3-dimensional (3D)/intensity-modulated radiation therapy (IMRT).
Methods and Materials
We retrospectively collected data from the University of Texas MD Anderson Cancer Center. Patients with borderline resectable/potentially resectable or locally advanced pancreatic cancer receiving neoadjuvant SBRT (median, 36.0 Gy/5fx), 3D conformal radiation (median, 50.4 Gy/28 fx) or IMRT (median, 50.4 Gy/28 fx) were included. Overall survival (OS) and progression-free survival were analyzed using Cox regression.
Results
In total, 104 patients were included in our study. Fifty-seven patients (54.8%) were treated with SBRT, and 47 patients (45.2%) were treated with 3D/IMRT. Patients in the SBRT group were slightly older (median age: 70.3 vs 62.7 in the 3D/IMRT group). Both groups had similar proportions of patients with locally advanced pancreatic cancer (SBRT: 30, 52.6%; 3D/IMRT: 24, 51.1%). All patients were treated with chemotherapy. Patients in the SBRT group underwent more surgical resection compared with the 3D/IMRT group (38.6% vs 23.4%, respectively). At a median follow-up of 22 months, a total of 60 patients (57.7%) died: 25 (25/57, 43.9%) in the SBRT group, and 35 (35/47, 74.5%) in the 3D/IMRT group. Median OS was slightly higher in the SBRT group (29.6 months vs 24.1 months in the 3D/IMRT group). On multivariable Cox regression, the choice of radiation therapy technique was not associated with differences in OS (adjusted hazard ratios [aHR] = 0.5; 95% confidence interval [CI], 0.2%-1.3%, P = .18). Moreover, patients that underwent surgical resection had better OS (aHR = 0.3, 95% CI, 0.1%-0.8%, P = .01). Furthermore, progression-free survival was also similar between patients treated with SBRT and those treated with 3D/IMRT (aHR = 0.9, 95% CI, 0.5%-1.8%, P = .81)
Conclusions
SBRT was associated with similar clinical outcomes compared with conventional radiation techniques, despite being delivered over a shorter period of time which would spare patients prolonged treatment burden. Future prospective data are still needed to better assess the role of SBRT in patients with pancreatic cancer.
Introduction
Pancreatic cancer is the fourth leading cause of cancer death in the developed world and is projected to become the second most common cause of cancer mortality by 2030.1,2 Despite aggressive treatment, the 5-year overall survival of patients with pancreatic cancer remains dismal at around 10%.1 Many novel treatment modalities are being suggested; however, surgical resection remains the primary curative option in those patients.3
Patients with borderline resectable/potentially resectable (BR/PR) or locally advanced (LA) pancreatic cancer have tumor involvement of critical abdominal vessels that may render an operation challenging or impossible.4 A patient's tumor may also be considered unresectable due to aggressive features like elevated CA19-9 or medical comorbidities.5 In these cases, neoadjuvant therapy4 may improve the chances of achieving a R0 resection.5 Moreover, disease that progresses through preoperative therapy may help to identify patients with biology that would have been ultimately unfavorable for resection.6
Neoadjuvant therapy often consists of systemic treatments followed by radiation therapy for consolidation.7,8 There is no consensus on the dose and fractionation or conformal technique to use,9 although if radiation is used, external beam radiotherapy delivered by 3-dimensional (3D) conformal or intensity-modulated radiation therapy (IMRT) is most common.4 Stereotactic body radiation therapy (SBRT) has gained increased popularity, due to safety and efficacy in single institution series, as well as a much shorter course of treatment, especially during the current COVID-19 pandemic.10, 11, 12 Given the differences in use of these very different radiation modalities, we sought to assess whether differences in clinical outcomes existed between patients undergoing preoperative SBRT or conventional radiation therapy at a single institution in the modern era of pancreatic cancer treatment.
Patients and Methods
Study design and population
The following study is a retrospective analysis of patients with BR/PR or LA pancreatic ductal adenocarcinoma diagnosed between 2014 and 2019, and treated at our home institution (The University of Texas MD Anderson Cancer Center). All patients included in our study received neoadjuvant chemoradiation with SBRT or conventional radiation therapy (3D/IMRT) before evaluation for potential surgical resection. The median radiation dose in the SBRT group was 36.0 Gy (range, 25-55) delivered over 5 fractions while patients in the conventional radiation group received a median of 50.4 Gy (range, 50-50.4) delivered over 25 or 28 fractions, with either 3D conformal radiation (3D CRT) or IMRT technique. Dosimetry planning and normal tissue constraints for SBRT or 3D/IMRT were based on previously published guidelines that have been adapted by the group at our institution.13,14
Patients that underwent surgery before radiation treatment were excluded from this study. Furthermore, patients presenting with resectable or metastatic pancreatic cancer were also excluded. Patient information regarding demographics and baseline tumor and treatment characteristics were collected. The University of Texas MD Anderson Cancer Center Institutional Review Board approved all protocols in this study.
Chemotherapy and radiation therapy
All patients were treated with 2 to 6 months of neoadjuvant chemotherapy using either FOLFIRINOX or a combination of gemcitabine and nab-paclitaxel. FOLFIRINOX is typically the preferred regimen, but patients with contraindications to FOLFIRINOX and patients with a poor baseline performance status are treated with gemcitabine and nab-paclitaxel instead. All chemotherapy regimens and doses were based on published guidelines and protocols, and chemotherapy dosing was modified during treatment based on patient performance status, comorbidities, and treatment toxicities.15
Patients who did not progress on initial chemotherapy were assessed every 2 months in a multidisciplinary tumor board for consideration of further definitive local treatment with radiation therapy. The decision for patients to undergo SBRT or 3D/IMRT was based on physician discretion and patient preference. Patients with extensive duodenal or surrounding tissue invasion were typically not recommended for SBRT treatment. Patients with extensive nodal involvement that would not fit in irradiated fields were also not recommended for SBRT treatment.
Planning constraints for the organs at risk are shown in Table E1. The gross tumor volume (GTV) was delineated on the planning CT. For both 3D and IMRT, the clinical target volume was derived by isotopically expanding the GTV by 1.5 cm, plus an additional 1-cm proximal expansion margin for the superior mesenteric artery and celiac trunk, if involved. The planning target volume (PTV) was derived by adding an additional 1- to 5-mm and 7- to 10-mm isotropic margin to the clinical target volume for 3D and IMRT modalities, respectively. Treatment goals included 95% of the PTV receiving at least the prescription dose and the maximum dose to not exceed 120% of the prescription dose.
For SBRT, multiple PTVs were utilized based on physician preference. Generally, a low-dose PTV (Rx = 25-33 Gy) was derived by adding an isotropic margin of 3 to 5 mm to the GTV and the tumor vessel interface. Next, a medium-dose PTV (Rx = 33-40 Gy) was created from the low-dose PTV with a gastrointestinal planning organ-at-risk volume (GI-PRV) subtracted; the GI-PRV was the union of the duodenum, bowel, and stomach with a 3- to 5-mm uniform margin. Finally, the high-dose PTV (Rx = 36-55 Gy) was prescribed to the tumor vessel interface plus a 3- to 5-mm margin, with the GI-PRV subtracted. The SBRT treatment goals typically included the PTV maximum dose to be no more than 120% of the prescription dose, as well as 95% of the high-dose PTV receiving the prescription dose; however, this was relaxed when necessary to spare the organs at risk.
Outcomes
The main endpoint of our study was overall survival (OS), defined as any cause of death from diagnosis until last follow-up. We also analyzed progression-free survival (PFS), which was defined by the occurrence of any new pancreatic cancer disease progression (local-regional recurrence/progression or distant metastasis), or the occurrence of death from diagnosis until last follow-up. Furthermore, we analyzed local-regional recurrence (LRR), defined by the occurrence of local or regional recurrence from the time of diagnosis until last follow-up. All time-to-event endpoint definitions are in line with the DATECAN classification.16 Finally, we analyzed physician-reported adverse events, which were reported and graded based on the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE).17
Statistical analysis
We used descriptive statistics to summarize the baseline patients’ demographics, disease, and treatment characteristics. Categorical variables are presented as frequencies and percentages, and continuous variables are presented as medians with interquartile ranges. Pearson χ2 and the independent t test were used to assess differences in categorical and continuous variables between the 2 groups, respectively. Multivariable Cox regression was used to calculate adjusted hazard ratios (aHR) with corresponding 95% confidence intervals (CI). All reported hazard ratios are for the comparison of SBRT to conventional radiation therapy. Survival curves for OS and PFS were generated using the Kaplan-Meier method. Statistical significance was set a priori at a 2-sided P value of .05. All statistical analyses were conducted using IBM SPSS Statistic version 26. Kaplan-Meier curves were generated using Prism version 8.
Results
Baseline patients and treatments characteristics
A total of 104 patients were included in our study. Out of those, 57 patients (54.8%) were treated with SBRT and 47 patients (45.2%) were treated with 3D/IMRT (35 patients with 3D and 12 patients with IMRT). Table 1 presents the baseline characteristics of the study groups. Patients in the SBRT group had a higher median age compared with those in the 3D/IMRT group (70.3 vs 62.7, respectively, P = .01). Most tumors (61, 58.7%) originated in the head of the pancreas regardless of treatment group. Both groups had a similar distribution of patients in regard to tumor radiological stage, with around half the patients presenting with LA and half presenting with BR/PR pancreatic cancer. However, patients in the SBRT group had more T2 (12, 21.1%), N0 (43, 75.4%) disease, and patients in the 3D/IMRT group had more T3 (19, 40.4%) and N1 (20, 42.6%) disease. Nevertheless, a similar proportion of T4 tumors was seen at baseline in both groups (SBRT: 31, 54.4%; 3D/IMRT: 26, 55.3%). Moreover, all patients in our study received modern chemotherapy with either FOLFIRINOX, gemcitabine/nab-paclitaxel, or a combination of both. Lastly, patients in the SBRT group underwent slightly more subsequent surgical resections than those treated with 3D/IMRT (22, 38.6% vs 11, 23.4%), although this difference was not statistically significant (P = .10).
Table 1.
Patients, tumors, and treatment characteristics
| Patient characteristics | Radiation therapy | P value* | |
|---|---|---|---|
| SBRT (n = 57) | 3D/IMRT (n = 47) | ||
| Age (median, IQR), y | 70.3 (59.8-70.3) | 62.7 (57.4-68.4) | .01 |
| Sex | |||
| Male | 30 (52.6) | 31 (66.0) | .17 |
| Female | 27 (47.4) | 16 (34.0) | |
| Tumor origin | |||
| Head | 31 (54.4) | 30 (63.8) | .61 |
| Body | 11 (19.3) | 10 (21.3) | |
| Neck | 4 (7.0) | 2 (4.3) | |
| Tail | 2 (3.5) | 2 (4.3) | |
| Uncinate process | 3 (5.3) | 2 (4.3) | |
| Multiple pancreatic sites | 6 (10.5) | 1 (2.1) | |
| Tumor radiological stage | |||
| BR/PR | 27 (47.4) | 23 (48.9) | .87 |
| LA | 30 (52.6) | 24 (51.1) | |
| ECOG PS | |||
| 0 | 12 (21.1) | 17 (36.2) | .17 |
| 1 | 44 (77.2) | 30 (63.8) | |
| 2 | 1 (1.8) | 0 (0) | |
| T stage | |||
| T1 | 2 (3.5) | 1 (2.1) | .01 |
| T2 | 12 (21.1) | 1 (2.1) | |
| T3 | 12 (21.1) | 19 (40.4) | |
| T4 | 31 (54.4) | 26 (55.3) | |
| N stage | |||
| N0 | 43 (75.4) | 22 (46.8) | .01 |
| N1 | 12 (21.1) | 20 (42.6) | |
| Nx | 2 (3.5) | 5 (10.6) | |
| Baseline CA19-9 (median, IQR) | 129.9 (35.3-474.6) | 219.0 (72.2-980.2) | .32 |
| Chemotherapy | |||
| FOLFIRINOX/nabPGem | 9 (15.8) | 9 (19.1) | .39 |
| FOLFIRINOX | 21 (36.8) | 22 (46.8) | |
| nabPGem | 27 (47.4) | 16 (34.0) | |
| BED >70 Gy | 27 (47.4) | 0 (0) | <.001 |
| Surgical resection | |||
| No | 35 (61.4) | 36 (76.6) | .10 |
| Yes | 22 (38.6) | 11 (23.4) | |
Abbreviations: 3D = three-dimensional radiation therapy; BED = biologically effective dose; BR = borderline resectable; ECOG PS = Eastern Cooperative Oncology Group Performance Status; Gem = Gemcitabine; IMRT = intensity-modulated radiation therapy; IQR = interquartile range; LA = locally advanced; nabP = nab-paclitaxel; PR = potentially resectable; PS = performance status; SBRT = stereotactic body radiation therapy.
The P values were calculated using Pearson χ2 method for categorical variables, and the independent t test for continuous variables.
Values are for frequency (percentage).
Clinical outcomes
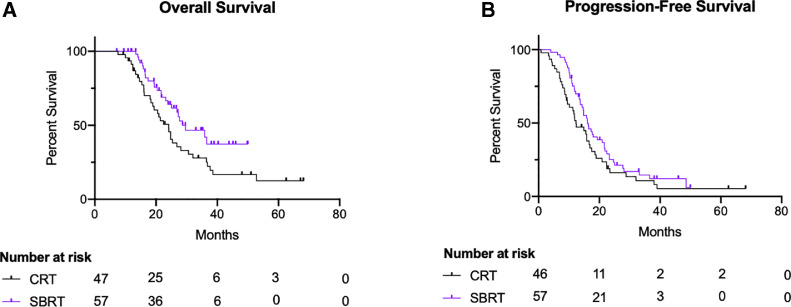
At a median follow-up of approximately 22 months (SBRT group: 23 months, 3D/IMRT group: 21 months), a total of 60 patients died (57.7%): 25 (43.9%) in the SBRT group, and 35 (74.5%) in the 3D/IMRT group. Despite follow-up periods being similar between the 2 groups, more censoring occurred in the SBRT group owing to the smaller number of patients followed-up beyond 40 months (Fig. 1 A). Median OS was slightly higher in the SBRT group compared with patients in the 3D/IMRT group (29.6 months, IQR 19.8-39.4 vs 24.1 months, IQR 20.2-28.0, respectively; Fig 1 A). On multivariable Cox regression, no difference in OS was noted between patients regardless of radiation therapy technique (SBRT vs 3D/IMRT aHR = 0.5; 95% CI, 0.2%-1.3%; P = .18; Table 2). Furthermore, patients that underwent surgical resection had a statistically significant improvement in OS (aHR: 0.3; 95% CI, 0.1%-0.8%; P = .01; Table 2).
Fig. 1.
Kaplan-Meier survival curves for overall survival (A) and progression-free survival (B) divided by stereotactic body radiation therapy (SBRT; purple) and 3-dimensional/intensity-modulated radiation therapy (black). Abbreviation: CRT = conformal radiation.
Table 2.
Multivariable COX regression for overall survival and progression-free survival
| Overall survival |
Progression-free survival |
|||
|---|---|---|---|---|
| aHR (95% CI) | P value | aHR (95% CI) | P value | |
| Radiation therapy | ||||
| 3D/IMRT | - | - | - | - |
| SBRT | 0.5 (0.2-1.3) | .18 | 0.9 (0.5-1.8) | .81 |
| Age | 1.0 (0.9-1.0) | .13 | 1.0 (0.9-1.0) | .80 |
| Sex | ||||
| Male | - | - | - | - |
| Female | 1.1 (0.6-2.1) | .83 | 0.8 (0.5-1.3) | .37 |
| Tumor origin | ||||
| Head | - | - | - | - |
| Body | 2.6 (1.1-5.8) | .03 | 2.6 (1.3-5.3) | .007 |
| Neck | 2.1 (0.4-11.0) | .37 | 1.2 (0.3-4.6) | .78 |
| Tail | 0.4 (0.1-3.6) | .44 | 3.2 (0.8-12.1) | .09 |
| Uncinate process | 2.0 (0.5-8.0) | .35 | 1.8 (0.5-6.2) | .35 |
| Multiple pancreatic sites | 0.8 (0.2-3.1) | .80 | 2.7 (1.0-6.9) | .04 |
| ECOG PS | ||||
| 0 | - | - | - | - |
| ≥1 | 0.6 (0.3-1.2) | .14 | 0.7 (0.4-1.2) | .19 |
| T stage | ||||
| T1 | - | - | - | - |
| T2 | 5.7 (0.52-61.8) | .16 | 2.8 (0.5-15.6) | .25 |
| T3 | 3.3 (0.4-27.8) | .28 | 1.8 (0.4-8.7) | .49 |
| T4 | 2.2 (0.3-17.8) | .44 | 1.5 (0.3-6.7) | .59 |
| N stage | ||||
| N0 | - | - | ||
| N1 | 0.9 (0.5-1.9) | .80 | 1.4 (0.8-2.5) | .30 |
| Baseline CA19-9 | 1.0 (1.0-1.0) | .005 | 1.0 (1.0-1.0) | .12 |
| Chemotherapy | ||||
| FOLFIRINOX/nabPGem | - | - | - | - |
| FOLFIRINOX | 4.4 (1.4-13.6) | .01 | 0.9 (0.4-2.1) | .82 |
| nabPGem | 4.0 (1.3-12.9) | .02 | 1.0 (0.4-2.2) | .94 |
| BED >70 Gy | 0.8 (0.3-2.5) | .74 | 0.7 (0.3-1.5) | .37 |
| Surgical resection | ||||
| No | - | - | - | - |
| Yes | 0.3 (0.1-0.8) | .01 | 0.4 (0.2-0.8) | .01 |
Abbreviations: 3D = 3-dimensional radiation therapy; aHR = adjusted hazard ratio; BED = biologically effective dose; CI = confidence interval; ECOG PS = Eastern Cooperative Oncology Group Performance Status; Gem = Gemcitabine; IMRT = intensity-modulated radiation therapy; nabP = nab-paclitaxel; PS = performance status; SBRT = stereotactic body radiation therapy.
When analyzing PFS, patients in the SBRT group had a slightly higher median PFS when compared to the 3D/IMRT group (16.1 months vs 12.3 months, respectively; Fig 1 B). Moreover, on multivariable Cox regression, no difference in PFS was noted between the 2 groups (aHR: 0.9, P = .81; Table 2). Additionally, surgical resection was shown to be independently associated with better PFS (aHR: 0.4; 95% CI, 0.2%-0.8%; P = .01; Table 2). Furthermore, a total of 29 patients (27.9%) developed LRR: 15 (15/57, 26.3%) in the SBRT group, and 14 (14/47, 29.8%) in the 3D/IMRT group. On multivariable Cox regression, no difference in LRR was appreciated between patients treated with SBRT and those treated with 3D/IMRT (aHR: 1.0; 95% CI, 0.3%-2.9%; P = .95). Lastly, we compared the clinical outcomes differences between patients treated with 3D and those treated with IMRT, and no difference was noted in OS (log-rank P = .93) or PFS (log-rank P = .89).
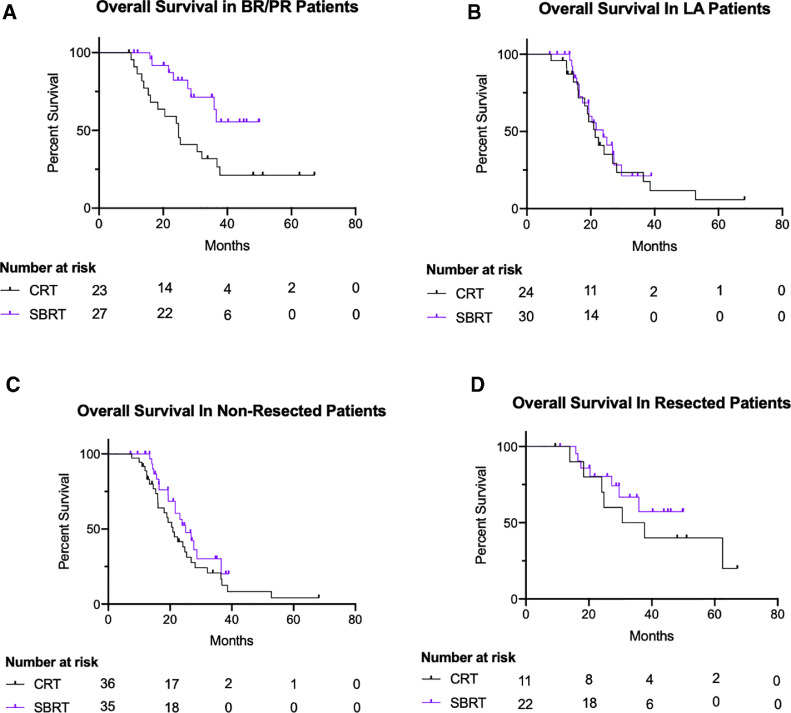
On subgroup analysis, our data show that patients with BR/PR pancreatic cancer treated with SBRT had a slightly better OS compared with those in the 3D/IMRT group (log-rank P = .01) (Fig. 2 A), whereas no difference in OS was noted among patients with LA pancreatic cancer (log-rank P = .64; Fig 2 B). Moreover, when stratifying by surgical resection, no difference in OS was noted between the 2 radiation groups, whether patients did not get a surgical resection (log-rank P = .15; Fig. 2 C) or got a surgical resection (log rank P = .41; Fig. 2 D).
Fig. 2.
Kaplan-Meier survival curves for overall survival in patients with borderline resectable/potentially resectable (BR/PR; log-rank P = .01). (A) Locally advanced (LA; log-rank P = .64), (B) who did not get surgical resection (log-rank P = .15), (C) and who did get surgical resection (log-rank P = .41), (D) divided by stereotactic body radiation therapy (SBRT; purple) and 3-dimensional/intensity-modulated radiation therapy (black). Abbreviation: CRT = conformal radiation.
Table 3 presents the sites of first local-regional or distant recurrence. Overall, the patterns of recurrence were similar in both groups. The majority of local-regional recurrences happened locally within the original tumor (29/104, 27.9%), with no difference being noted between the 2 groups (P = .69). Moreover, most distant metastases occurred in the liver (26, 25.0%), or the lungs (23, 22.1%). There were slightly more liver metastases in the SBRT group, and slightly more peritoneal metastases in the 3D/IMRT group, but all of those differences were not statistically significant (Table 3).
Table 3.
Sites of first local-regional and/or distant recurrence
| First recurrence site | SBRT (n = 57) | 3D/IMRT (n = 47) | P value* |
|---|---|---|---|
| Local-regional | |||
| Tumor | 15 (26.3) | 14 (29.8) | .69 |
| Regional lymph node | 4 (7.0) | 5 (10.6) | .51 |
| Distant | |||
| Liver | 16 (28.1) | 10 (21.3) | .43 |
| Lungs | 13 (22.8) | 10 (21.3) | .85 |
| Peritoneal | 7 (12.3) | 11 (23.4) | .14 |
Abbreviations: 3D = 3-dimensional radiation therapy; IMRT = intensity-modulated radiation therapy; SBRT = stereotactic body radiation therapy.
The P values were calculated using Pearson χ2 method. Values are for frequency (percentage).
Adverse events
Table 4 summarizes the main differences in adverse events between the 2 groups. Treatment with SBRT and 3D/IMRT was overall well tolerated with minimal grade 2 or more toxicity. In our study, only one patient from the SBRT group developed grade 3 abdominal pain (1/57, 1.8%), and no other patients developed any grade 3 adverse events. Patients in the 3D/IMRT developed more diarrhea or constipation compared with those in the SBRT group, but most of those were grade 1 toxicity, with only 1 patient in the 3D/IMRT group (1/47 2.1%) developing grade 2 diarrhea. Lastly, more patients in the 3D/IMRT group had grade 2 nausea (6/47, 12.8%) compared with those in the SBRT group (2/57, 3.5%).
Table 4.
Adverse events differences between SBRT and 3D/IMRT
| Adverse event | SBRT (n = 57) | 3D/IMRT (n = 47) | ||
|---|---|---|---|---|
| Any grade | Grade ≥2 | Any grade | Grade ≥2 | |
| Abdominal pain | 17 (29.8) | 1 (1.8)* | 18 (38.3) | 3 (6.4) |
| Fatigue | 22 (38.6) | 1 (1.8) | 22 (46.8) | 2 (4.3) |
| Nausea | 19 (33.3) | 2 (3.5) | 22 (46.8) | 6 (12.8) |
| Vomiting | 4 (7.0) | 1 (1.8) | 2 (4.3) | 1 (2.1) |
| Diarrhea | 1 (1.8) | 0 (0) | 12 (25.5) | 1 (2.1) |
| Constipation | 2 (3.5) | 0 (0) | 7 (14.9) | 0 (0) |
Abbreviations: 3D = 3-dimensional radiation therapy; IMRT = intensity-modulated radiation therapy; SBRT = stereotactic body radiation therapy.
Only one patient in the SBRT group developed grade 3 abdominal pain. All other adverse events are either grade 1 or 2.
Discussion
Patients with pancreatic cancer are often treated with neoadjuvant chemoradiation4 to potentially downstage tumors and improve surgical outcomes.6,18,19 A recent phase 2 trial by the Alliance for clinical trials in oncology (A021501) compared the use of extended chemotherapy to chemoradiation with SBRT in patients with borderline resectable pancreatic cancer.20 Data from this trial showed that patients receiving extended chemotherapy had better clinical outcomes with improved 18-months OS, and better surgical outcomes.21 Unfortunately, there are no clear guidelines regarding the type of radiation therapy to use in the neoadjuvant setting. In this article, we show that SBRT could offer similar clinical outcomes to conventional radiation modalities, with the advantage of being delivered over a shorter fractionation schedule that would be beneficial to patients.
In our analysis, patients who received SBRT had similar OS and PFS to those treated with 3D or IMRT. SBRT treatment, when delivered at similar total doses, offers a higher biologically effective dose (BED) to the pancreatic tumor when compared to 3D/IMRT. This could explain the slight improvement in clinical outcomes for patients in the SBRT group. The higher BED to the pancreatic tumor could have also contributed to improved tumor control, and hence have led to a modestly higher rate of subsequent surgical resection in the SBRT group. This is particularly important to highlight because approximately half of the patients in the SBRT group (27, 47.4%) had a BED >70 Gy, whereas no patients in the 3D/IMRT group had a BED >70 Gy.14 It should be noted that the 3D/IMRT group had slightly worse baseline tumor characteristics, with more advanced T and N stages, and this also could have contributed to the lower rate of surgical resection in this group of patients. However, even after adjusting for T stage, N stage, chemotherapy, and surgical resection, SBRT still showed similar clinical outcomes to conventional radiation modalities in our analysis. Furthermore, patients in the SBRT group did not receive elective nodal coverage. However, patients with nodal involvement were recommended for SBRT treatment only when nodal involvement was proximal to the irradiated fields. This could explain the lower rates of local and regional recurrences in our SBRT cohort compared with previous data.22
It is important to note that the patients analyzed in this study did not routinely receive radiation therapy. Instead, patients received chemotherapy treatment first, and only those who did not progress on chemotherapy and subsequently discussed in a multidisciplinary setting were then consolidated with radiation therapy. This is in contradiction to patients in the Alliance trial (A021501), who were randomly assigned to the extended chemotherapy or chemoradiation arm.20,21 Furthermore, despite the trial showing favorable outcomes for patients that received extended chemotherapy, the trial included 2 patients that showed pathologic complete response, and both of those patients were in the radiation arm. As such, growing evidence is showing that not all patients with pancreatic cancer benefit from radiation therapy since they may harbor disease with more aggressive metastatic biology that reduces the immediate need for local treatment with radiation. Our findings underscore the critical need for biomarker development to predict which patients would benefit from intensive local treatment like SBRT or surgery to guide future therapeutic trials.
SBRT treatment allows for the delivery of a full course of radiation therapy in 5 days, instead of more prolonged treatments with conventional techniques that take more than 5 weeks. There are several established benefits of this hypofractionated approach. The reduced radiation treatment time shrinks the window between systemic treatment and potential surgery, which may be beneficial.11,23
In addition, reducing the number of daily radiation treatment would reduce the time spent merely on transportation and waiting for care, which was shown to be up to 10% of patients’ survival time in the setting of metastatic pancreatic cancer.24 Moreover, the shorter fractionation schedule offered with SBRT has been shown to be more cost-effective when compared to conventional radiation therapy.25 Finally, the current COVID-19 pandemic has made patients reluctant to all forms of hospital visits.26 Together, these issues may favor the use of SBRT instead of conventionally fractionated RT for some patients. Our data suggest that for patients who complete induction chemotherapy without significant disease progression, SBRT may be a reasonable alternative approach.
Our study has a few limitations worth noting. First of all, our study included 104 patients, and as such our sample size may have not provided enough statistical power to detect significant differences in subgroup analyses. Also, our analysis was based on a cohort collected from a single institution, limiting the generalizability of our findings to other medical settings. Furthermore, our study is a retrospective analysis, and as such, radiation therapy delivery and planning were not standardized across all patients, but rather delivered at the discretion of each physician. Lastly, dosimetric information was not collected, and thus not analyzed. As such, differences in treatment parameters could have been present between the 2 groups, and our study does not account for that. Despite these limitations, our study still presents a large comparison of patients with pancreatic cancer treated with SBRT or conventional radiation therapy in the modern era of chemotherapy. These patients were treated by the same group of radiation physicists and radiation oncologists, which reduces confounding factors that might occur by comparing different institutions or treatment techniques.
Conclusions
Our data show that SBRT affords similar clinical outcomes to conventional radiation therapy in pancreatic cancer. In this disease, surgical resection remains the main predictor of OS and PFS. The use of shorter fractionation patterns with SBRT could save patients the burden, cost, and time associated with prolonged radiation treatments which has been particularly relevant during the current COVID-19 pandemic, where patients are being reluctant to all hospital visits. Future randomized trials analyzing the use of SBRT are still needed to better assess the role of radiation therapy in this patient population.
Acknowledgments
We would like to thank all the patients and patient families who allowed us to learn from their cancer journey in this analysis.
Footnotes
Sources of support: The authors report no funding for this work.
Disclosures: Dr Das reports personal fees from Adlai Nortye and personal fees from MD Anderson Cancer Center Madrid Spain. Dr Herman has received research support from OncoSil, Galera, and Augmenix, and works as a consultant at 1440 Foundation. Dr Koong acts as a consultant at Aravive, Inc. Dr Taniguchi is supported by funding from the National Institutes of Health under award R01CA227517-01A1, Cancer Prevention & Research Institute of Texas (CPRIT) grant RR140012, V Foundation (V2015-22), the Kimmel Foundation, Sabin Family Foundation Fellowship, and the McNair Foundation. No other financial disclosures or conflicts of interest to report. All authors report no conflicts of interest related to the present article.
Research data are stored in an institutional repository and will be shared on reasonable request to the corresponding authors.
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.adro.2021.100763.
Appendix. Supplementary materials
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA: A Cancer Journal for Clinicians. 2019;69:7–34. doi: 10.3322/caac.21551. [DOI] [PubMed] [Google Scholar]
- 2.Rahib L, Smith BD, Aizenberg R, et al. Projecting cancer incidence and deaths to 2030: The unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014;74:2913–2921. doi: 10.1158/0008-5472.CAN-14-0155. [DOI] [PubMed] [Google Scholar]
- 3.Chakraborty S, Singh S. Surgical resection improves survival in pancreatic cancer patients without vascular invasion: A population based study. Ann Gastroenterol. 2013;26:346–352. [PMC free article] [PubMed] [Google Scholar]
- 4.Toesca DAS, Koong AJ, Poultsides GA, et al. Management of borderline resectable pancreatic cancer. Int J Radiat Oncol Biol Phys. 2018;100:1155–1174. doi: 10.1016/j.ijrobp.2017.12.287. [DOI] [PubMed] [Google Scholar]
- 5.Katz MHG, Pisters PWT, Evans DB, et al. Borderline resectable pancreatic cancer: The importance of this emerging stage of disease. J Am Coll Surg. 2008;206:833–846. doi: 10.1016/j.jamcollsurg.2007.12.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Winter JM, Cameron JL, Campbell KA, et al. 1423 Pancreaticoduodenectomies for pancreatic cancer: a single-institution experience. J Gastrointest Surg. 2006;10:1199–1211. doi: 10.1016/j.gassur.2006.08.018. [DOI] [PubMed] [Google Scholar]
- 7.Kaufmann B, Hartmann D, D'Haese JG, et al. Neoadjuvant treatment for borderline resectable pancreatic ductal adenocarcinoma. Dig Surg. 2019;36:455–461. doi: 10.1159/000493466. [DOI] [PubMed] [Google Scholar]
- 8.Janssen QP, O'Reilly EM, van Eijck CHJ, et al. Neoadjuvant treatment in patients with resectable and borderline resectable pancreatic cancer. Front Oncol. 2020:10. doi: 10.3389/fonc.2020.00041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Palta M, Godfrey D, Goodman KA, et al. Radiation therapy for pancreatic cancer: Executive summary of an ASTRO clinical practice guideline. Pract Radiat Oncol. 2019;9:322–332. doi: 10.1016/j.prro.2019.06.016. [DOI] [PubMed] [Google Scholar]
- 10.Ling DC, Vargo JA, Beriwal S. Breast, prostate, and rectal cancer: Should 5-5-5 be a new standard of care? Int J Radiat Oncol Biol Phys. 2020;108:390–393. doi: 10.1016/j.ijrobp.2020.06.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Herman JM, Chang DT, Goodman KA, et al. Phase 2 multi-institutional trial evaluating gemcitabine and stereotactic body radiotherapy for patients with locally advanced unresectable pancreatic adenocarcinoma. Cancer. 2015;121:1128–1137. doi: 10.1002/cncr.29161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Koong AC, Le QT, Ho A, et al. Phase I study of stereotactic radiosurgery in patients with locally advanced pancreatic cancer. Int J Radiat Oncol Biol Phys. 2004;58:1017–1021. doi: 10.1016/j.ijrobp.2003.11.004. [DOI] [PubMed] [Google Scholar]
- 13.Colbert LE, Rebueno N, Moningi S, et al. Dose escalation for locally advanced pancreatic cancer: How high can we go? Adv Radiat Oncol. 2018;3:693–700. doi: 10.1016/j.adro.2018.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Krishnan S, Chadha AS, Suh Y, et al. Focal radiation therapy dose escalation improves overall survival in locally advanced pancreatic cancer patients receiving induction chemotherapy and consolidative chemoradiation. Int J Radiat Oncol Biol Phys. 2016;94:755–765. doi: 10.1016/j.ijrobp.2015.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Balaban EP, Mangu PB, Khorana AA, et al. Locally advanced, unresectable pancreatic cancer: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2016;34:2654–2668. doi: 10.1200/JCO.2016.67.5561. [DOI] [PubMed] [Google Scholar]
- 16.Bonnetain F, Bonsing B, Conroy T, et al. Guidelines for time-to-event end-point definitions in trials for pancreatic cancer. Results of the DATECAN initiative (Definition for the Assessment of Time-to-event End-points in CANcer trials) Eur J Cancer. 2014;50:2983–2993. doi: 10.1016/j.ejca.2014.07.011. [DOI] [PubMed] [Google Scholar]
- 17.National Institutes of Health National Cancer Institute . 2009. Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0. [Google Scholar]
- 18.Sohn TA, Yeo CJ, Cameron JL, et al. Resected adenocarcinoma of the pancreas—616 patients: Results, outcomes, and prognostic indicators. J Gastrointest Surg. 2000;4:567–579. doi: 10.1016/s1091-255x(00)80105-5. [DOI] [PubMed] [Google Scholar]
- 19.Evans DB, George B, Tsai S. Non-metastatic pancreatic cancer: Resectable, borderline resectable, and locally advanced-definitions of increasing importance for the optimal delivery of multimodality therapy. Ann Surg Oncol. 2015;22:3409–3413. doi: 10.1245/s10434-015-4649-2. [DOI] [PubMed] [Google Scholar]
- 20.Katz MHG, Ou F-S, Herman JM, et al. Alliance for clinical trials in oncology (ALLIANCE) trial A021501: Preoperative extended chemotherapy vs. chemotherapy plus hypofractionated radiation therapy for borderline resectable adenocarcinoma of the head of the pancreas. BMC Cancer. 2017;17:505. doi: 10.1186/s12885-017-3441-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Katz MHG, Shi Q, Meyers JP, et al. Alliance A021501: Preoperative mFOLFIRINOX or mFOLFIRINOX plus hypofractionated radiation therapy (RT) for borderline resectable (BR) adenocarcinoma of the pancreas. J Clin Oncol. 2021;39(Suppl 3) doi: 10.1016/j.ijrobp.2024.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhu X, Ju X, Cao Y, et al. Patterns of local failure after stereotactic body radiation therapy and sequential chemotherapy as initial treatment for pancreatic cancer: Implications of target volume design. Int J Radiat Oncol Biol Phys. 2019;104:101–110. doi: 10.1016/j.ijrobp.2019.01.075. [DOI] [PubMed] [Google Scholar]
- 23.Chuong MD, Springett GM, Freilich JM, et al. Stereotactic body radiation therapy for locally advanced and borderline resectable pancreatic cancer is effective and well tolerated. Int J Radiat Oncol Biol Phys. 2013;86:516–522. doi: 10.1016/j.ijrobp.2013.02.022. [DOI] [PubMed] [Google Scholar]
- 24.Bange EM, Doucette A, Gabriel PE, et al. Opportunity costs of receiving palliative chemotherapy for metastatic pancreatic ductal adenocarcinoma. JCO Oncol Pract. 2020;16:e678–e687. doi: 10.1200/JOP.19.00328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Murphy JD, Chang DT, Abelson J, et al. Cost-effectiveness of modern radiotherapy techniques in locally advanced pancreatic cancer. Cancer. 2012;118:1119–1129. doi: 10.1002/cncr.26365. [DOI] [PubMed] [Google Scholar]
- 26.Rosenbaum L. The untold toll: The pandemic's effects on patients without COVID-19. N Engl J Med. 2020;382:2368–2371. doi: 10.1056/NEJMms2009984. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.